Introduction
Chimeric antigen receptor (CAR) T-cell therapy is
considered to be a promising immunotherapeutic treatment strategy
for the treatment of patients with relapsed or refractory B-cell
malignancies. Clinical trials have shown promising remission rates
for patients with B-lymphoblastic acute leukemia (B-ALL) and B-cell
non-Hodgkin lymphoma who were treated with CD19-specific CAR
T-cells (1-5). Based on these results, Kymriah™
(Tisagenlecleucel, Novartis) and Yescarta™ (Axicabtagene
Ciloleucel, Kite Pharma/Gilead) were the first two CD19 CAR T-cell
products to be approved by the Food and Drug Administration (FDA)
in the United States in 2017 for the treatment of relapsed or
refractory B-ALL (r/r B-ALL) (Kymriah™) and relapsed or refractory
diffuse large B-cell lymphoma (r/r DLBCL) (Yescarta™) (6,7).
Indications were extended in the following years, and the CD19 CAR
T-cell products Tecartus™ (Brexucabtagene Autoleucel, Kite
Pharma/Gilead) and Breyanzi™ (Lisocabtagene Maraleucel, Juno
Therapeutics/BMS) have recently obtained market access (8). Additionally, academic institutions
began their own good manufacturing production (GMP) of CD19 CAR
T-cells (9). Whereas the
commercially available products contain a second-generation CAR, a
third-generation CD19 CAR is currently being evaluated for safety
and efficacy within the HD-CAR-1 trial (9). In contrast to second-generation
CARs, third-generation CARs contain two intracellular costimulatory
domains (e.g., CD28 and 4-1BB) instead of only one (10,11).
For the GMP-compliant manufacturing of CAR T-cell
products, different types of media are currently being used. Media
containing fetal bovine serum (FBS) is still most commonly used in
GMP core facilities worldwide (12). FBS is nutrient-rich containing
growth factors, hormones, vitamins, buffering proteins and ions,
but does not mirror well the condition within the human body
following the application of the CAR T-cells, as the composition of
nutrients between fetal bovine and human serum (HS) differs
significantly (13). In addition,
ethical and ecological aspects have to be taken into consideration
whenever FBS is used. Consequently, an increasing number of GMP
laboratories have begun to optimize HS containing media
formulations for CAR T-cell production (14-16). It is considered that HS supplies
nutrients and growth factors to the T-cell culture that more
closely imitates the human microenvironment. However, as the
composition of HS and FBS depend on the donor,
product-to-product-inconsistency is a major concern (17). Furthermore, the risk of
contamination of HS and FBS is also a critical aspect. FBS could
theoretically transmit bovine spongiform encephalopathy (BSE) and
viral pathogens. Moreover, HS also carries the risk of viral
contamination (17,18). Therefore, serum must be tested in
depth prior to its use in GMP manufacturing, thus leading to
increased costs.
As a result, there is a trend towards the use of
defined medium formulations with or without chemically defined
animal (xeno)-free components for the production of CAR T-cells.
Serum-free medium does not contain serum but can include purified
or synthetic ingredients. Smith et al (19) revealed similar expansion rates for
T-cells cultured in xeno-free medium compared to media supplemented
with HS or FBS. Furthermore, the T-cells expanded in xeno-free
medium exhibited a higher percentage of central memory T-cells and
the growth of lentivirus-mediated gene transduced T-cells was
comparable (19). Coeshott et
al (20) used xeno-free
medium to expand T-cells in a functionally closed, automated
bioreactor system in a large scale. The phenotyping of expanded
T-cells displayed high rates of central memory T-cells compared to
before seeding.
The aim of the present study was to compare the
effects of four different xeno-free T-cell culture media (TCM) to
the standard media (RCF) containing 10% FBS for the production of a
third-generation CD19 CAR T-cell product with respect to CAR T-cell
transduction efficiency, expansion, viability, cytotoxicity and
phenotype (Table I). CTS™
OpTmizer™ Pro SFM, Thermo Fisher Gibco™ (TF) is a serum-free medium
(SFM), whereas Prime-XV™ T Cell CDM, Fujifilm™ (FF) is a chemically
defined, animal component-free medium. LymphoONE™ T-Cell Expansion
Xeno-Free Medium, Takara Bio™ (TB) contains pharmaceutical-grade HS
albumin and recombinant human insulin. Similarly, TCM
GMP-Prototype, CellGenix™ (CG) includes HS albumin and recombinant
human insulin and transferrin.
 | Table IMedia composition. |
Table I
Media composition.
| Basal medium | CTS™ OpTmizer™ Pro
SFM, Thermo Fisher Gibco™ (TF) | Prime-XV™ T Cell
CDM, Fujifilm™ (FF) | LymphoONE™ T-Cell
Expansion Xeno-Free Medium, (TB) | TCM GMP-Prototype,
CellGenix™ (CG) | 45% Click's Medium
(EHAA), Fujifilm™ (RCF) |
|---|
| Other medium | - | - | - | - | 45% RPMI |
| Supplement | CTS™ OpTmizer™
T-Cell Expansion Supplement, Thermo Fisher Gibco™ | - | - | - | - |
| Serum | - | - | - | - | 10% FBS |
| L-glutamine | 2 mM | N/Aa | Yes | N/Aa | 2 mM |
|
Pharmaceutical-grade human proteins | Yes, e.g.,
albumin | No | Albumin | Albumin | - |
| Recombinant human
proteins | No | Yes, e.g.,
albumin | Insulin | Insulin and
transferrin | - |
| Antibiotics | - | - |
+/-Streptomycin | - | - |
Materials and methods
Ficoll separation
The isolation of peripheral blood mononuclear cells
(PBMCs) was performed for 5 healthy donor buffy coats provided by
the blood bank of the University Hospital of Mannheim
(DRK-Blutspendedienst Baden-Württemberg-Hessen) by separation with
Ficoll [Ficolite-H (Human), Linaris Biologische Produkte GmbH].
PBMCs were cryopreserved with 90% FBS (Thermo Fisher Scientific,
Inc.) and 10% dimethyl sulfoxide (DMSO; SERVA Electrophoresis GmbH)
until the CAR T-cell manufacturing process. Ethical approval and
written consent to participate was obtained. Ethical approval was
obtained from the Medical Faculty of the University of Heidelberg
(reference no. S-254/2016).
Production of retroviral vector
To produce the third-generation retroviral vector
SFG.CAR.CD19/CD28/4-1BB/CD3ζ, plasmids were kindly provided by
Professor Malcolm Brenner (Baylor College of Medicine, Houston, TX,
USA). 293T cells (DSMZ, cat. no. ACC 635) were co-transfected with
the specific retroviral vector plasmid carrying the gene of
interest (3.75 µg), the gag-pol plasmid PegPam3 (3.75
µg) and the envelope plasmid RDF (2,5 µg). 293T cells
were cultured in Iscove's modified Dulbecco's medium (IMDM)
GlutaMax-I (Thermo Fisher Scientific, Inc.) supplemented with 10%
FBS and 0,1 mM sodium pyruvate (Thermo Fisher Scientific, Inc.).
For transfection, 470 µl IMDM GlutaMax-I and 30 µl
GeneJuice Transfection Reagent (Merck KGaA) were added to the
plasmids and incubated for 15 min at room temperature. The
suspension was then added in a dropwise manner to the 100-mm dish.
Following 48 h of incubation at 37°C and 5% CO2, the
retroviral supernatant was collected, and new medium was added to
the dishes. Following 72 h of incubation in total, the collection
process was repeated. The retroviral supernatant was stored at
-80°C.
T-cell activation
The thawing and activation of PBMCs was performed on
day 0 of the transduction process. A total of 1×106
PBMCs per well were activated in a non-tissue culture 24-well plate
(Corning, Inc.) coated with anti-CD3 (clone: OKT3; cat. no. 317301;
BioLegend; dilution, 1 µg/ml;) and anti-CD28 (clone: CD28.2;
cat. no. 302901; BioLegend; dilution, 1 µg/ml) monoclonal
antibodies. The PBMCs were cultured in CTS™ OpTmizer™ Pro SFM (TF;
Thermo Fisher Scientific, Inc.), Prime-XV T Cell CDM (FF; FUJIFILM
Wako Pure Chemical Corporation), LymphoONE™ T-Cell Expansion
Xeno-Free Medium (TB; Takara Bio, Inc.), TCM GMP-Prototype (CG;
CellGenix Inc.) or in CAR medium (RCF) containing 45% Roswell Park
Memorial Institute (RPMI)-1640 medium (Thermo Fisher Scientific,
Inc.), 45% Eagle's Ham's amino acids (EHAA) Clicks medium (FUJIFILM
Wako Pure Chemical Corporation), 10% FBS and 2 mmol/l Gluta-MAX™
Supplement (Thermo Fisher Scientific, Inc.). A total of 10 ng/ml of
IL-7 (R&D Systems, Inc.) and 5 ng/ml of IL-15 (R&D Systems,
Inc.) were added to all media.
Transduction
The cells were transduced on day 2 using the vector
SFG.CAR.CD19/CD28/4-1BB/CD3ζ. The wells for the CAR T-cells were
coated with RetroNectin (Takara Bio, Inc.) at a concentration of 7
µg/ml and stored overnight at 4°C. On the day of
transduction, the wells were washed, and the retroviral supernatant
was added to the wells. The plates were then centrifuged at 2,000 ×
g for 90 min at room temperature. Activated T-cells were harvested
and 1×105 cells per well were seeded in the
corresponding medium in the 24-well plate. The plates were
centrifuged at 500 × g for 4min at room temperature and cultured at
37°C 5% CO2 until day 5.
Cell expansion and viability
Following transduction, the CAR T-cells and
non-transduced T-cells were cultured in the respective media with
the addition of cytokines. Feeding was performed every 2-3 days
adding fresh medium with cytokines. On day 12, the expansion and
viability of the CAR T-cells and non-transduced cells were
determined by counting the cells with Trypan blue (Merck KGaA) in a
counting chamber (NanoEnTek, Inc.). All cell lines were tested by
polymerase chain reaction (PCR) to be mycoplasma-free.
Cell lines
The Burkitt lymphoma cell line, Daudi (cat. no. ACC
78), obtained from the German Collection of Microorganisms and Cell
Culture (DSMZ) was used as CD19-positive target cell line. As a
CD19-negative target cell line, the chronic myeloid leukemia cell
line, K-562 (cat. no. ACC 10), also obtained from DSMZ, was used.
The target cells were cultured in RPMI (Thermo Fisher Scientific,
Inc.) supplemented with 10% FBS (Thermo Fisher Scientific, Inc.)
and 2 mM L-glutamine at 37°C and 5% CO2 and the cell
concentration were hold at 0.1-1×106/ml. The cells were
thawed 1 week prior to the experiments. The cells were washed and
cultured in the corresponding media when added to the
experiments.
Immunophenotyping
Flow cytometry was performed on a BD™ LSRII Flow
Cytometry Cell Analyzer (BD Biosciences). For immunophenotyping,
the following monoclonal antibodies were used: CD3 (UCHT1; no.
300424; 2/100 µl), CD4 (OKT4; cat. no. 317408; 2/100
µl), CD8 (SK1; cat. no. 344732; 2/100 µl), CD14
(63D3; cat. no. 367112; 2/100 µl), CD27 (M-T271; cat. no.
356418; 2/100 µl), CD45RO (UCHL1; cat. no. 304228; 2/100
µl), CCR7/CD197 (G043H7; no. 353214; 2/100 µl),
T-cell immunoglobulin and mucin-domain containing-3 (TIM-3)/CD366
(F38-2E2; cat. no. 345008; 2/100 µl), programmed cell death
protein 1 (PD-1; A17188B; cat. no. 621616; 2/100 µl) and
lymphocyte-activation gene 3 (LAG-3)/CD223 (7H2C65; cat. no.
369212; 2/100 µl), all from BioLegend, and CD45RA (MEM-56;
cat. no. MHCD45RA17; 2/100 µl) from Thermo Fisher
Scientific, Inc. In addition, 7-aminoactinomycin (7-AAD; BD
Biosciences; no. 555816; 5/100 µl) was used to detect dead
cells. To stain the CD19. CAR, a goat anti-human F(ab)2
IgG (H+L) antibody (Jackson ImmunoResearch Europe, Ltd.; cat. no.
109-116-088; 0.5/100 µl) conjugated with PE was used.
Compensation was based on PBMCs and controls including a
non-transduced control and fluorescence minus one control were
realized.
Intracellular staining
Cytokine release was measured on day 13 of CAR
T-cell manufacturing. A total of 2×105 CAR T-cells were
co-cultured with 4×105 Daudi or K562 cells for 6 h at
37°C and 5% CO2 following the addition of 1X Brefeldin A
(BioLegend) and CD107a (H4A3; BD Biosciences). After the incubation
time the plate was stored at 4°C overnight as previously described
(21). NEAR-IR (Thermo Fisher
Scientific, Inc.) staining was performed at 4°C for 30 min to
distinguish viable and dead cells. For surface marker staining, CD3
(same as above), CD4 (same as above), CD8 (same as above), CD20
(2H7; cat. no. 302310; BioLegend; 2/100 µl) and CAR antibody
(same as above) were used. For fixation and permeabilization, the
FoxP3 Staining Buffer Set (Miltenyi Biotec GmbH) was used.
Intracellular cytokine staining was performed using TNF-α (Mab11;
cat. no. 562783; BD Biosciences; 1/100 µl) and IFNγ (4S.B3;
cat. no. 502546; BioLegend; 2/100 µl) antibodies.
Multi-parametric cytometry was performed using a LSRII flow
cytometer (BD Biosciences). Unstimulated control and
unspecific-stimulated control served as negative controls.
Co-culture
Day 12 of the CAR T-cell manufacturing process was
regarded as day 0 of co-culture. A total of 0.15×105 CAR
T-cells or non-transduced T-cells were co-cultured in an
effector-to-target cell ratio of 1:2 in the corresponding medium.
Flow cytometry was performed on days 5, 10, 15, 20, 25 and 30. On
days 5, 10, 15, 20 and 25, 3×104 Daudi cells were added
to the remaining wells. The antibodies CD3 (same as above), CD4
(same as above), CAR (same as above), CD45RA (same as above), CCR7
(G043H7, cat. no. 353226; BioLegend; 2/100 µl), CD20 (same
as above), PD-1 (29F.1A12; cat. no. 135224; BioLegend; 2/100
µl), TIM-3 (same as above) and LAG-3 (11C3C65; cat. no.
369318; BioLegend; 2/100 µl) were used for staining.
Chromium release assay
The tumor cell lines, Daudi and K-562, were
incubated with 51Cr (Hartmann Analytic) for 2 h at 37°C
and 5% CO2. The ratio of effector to target cells was
30:1, 10:1, 3:1 and 1:1. A total of 5×103 target cells
were co-cultured with the effector cells in each well for 4 h at
37°C and 5% CO2; 1% Triton X-100 (Merck KGaA) served as
a maximum release control. For spontaneous release control, cell
culture medium was used. Ultima Gold (PerkinElmer, Inc.) was added
to the co-culture supernatant, and the vials were measured using a
1414 WinSpectral liquid scintillation counter (PerkinElmer, Inc.).
All experiments were performed in triplicate. To calculate the
specific lysis caused of the CAR T-cells, the following formula was
used: [(experimental release-spontaneous release)/(maximum
release-spontaneous release)] ×100.
Extraction of genomic DNA
Genomic DNA was obtained from cryopreserved CD19.CAR
T-cells. They were cryopreserved at day 14 of CAR T-cell culture in
freezing medium composed of 10% DMSO (SERVA Electrophoresis GmbH)
and 90% FBS (Thermo Fisher Scientific, Inc.). Genomic DNA was
extracted using the QIAamp® DNA Blood Mini kit (Qiagen
GmbH). DNA concentration was measured using UV spectroscopy
(NAnoDrop OneC, Thermo Fisher Scientific, Inc.) and adjusted to 20
ng/µl.
Quantitative PCR (qPCR)
For the assessment of the vector copy number of
CD19.CAR T-cells, a single copy gene (SCG)-based duplex (DP)-qPCR
assay (SCG-DP-PCR) was performed as described by Kunz et al
(22). For quantification the
2−ΔΔCt method (23)
was used. The sequence of the forward primer was 5′-AGC TGC CGA TTT
CCA GAA GA-3′ and that of the reverse primer was 5′-GCG CTC CTG CTG
AAC TTC A-3′. RNaseP served as a reference gene and the Copy Number
Reference Assay, RNaseP (cat. no. 4403326; Applied Biosystems)
which contains RNaseP gene-specific forward primer, reverse primer,
and probe (VIC/TAMRA) was used. A total of 100 ng genomic DNA were
used for the two simultaneous amplifications of the CAR transgene
and the SCG RNaseP supported by the StepOnePlus real-time PCR
system (Thermo Fisher Scientific, Inc.). Following conditions were
set up for thermocycling: 2 min for 50°C, 10 min for 95°C, followed
by 40 cycles of 15 sec 95°C and 1 min 60°C. H2O and
non-transduced cells served as negative controls.
Statistical analysis
Flow cytometric analysis was performed using FlowJo
10.8 software (BD Biosciences). GraphPad Prism 9 (GraphPad
Software, Inc.) was used for statistical analysis. To compare
multiple groups with a single independent variable, a one-way ANOVA
with a subsequent Dunnett's multiple comparisons test was used. For
the analysis of multiple groups with two independent variables, a
two-way ANOVA test with Dunnett's multiple comparisons test was
used. P<0.05 was considered to indicate a statistically
significant difference.
Results
Comparison of expansion, viability and
transduction rates
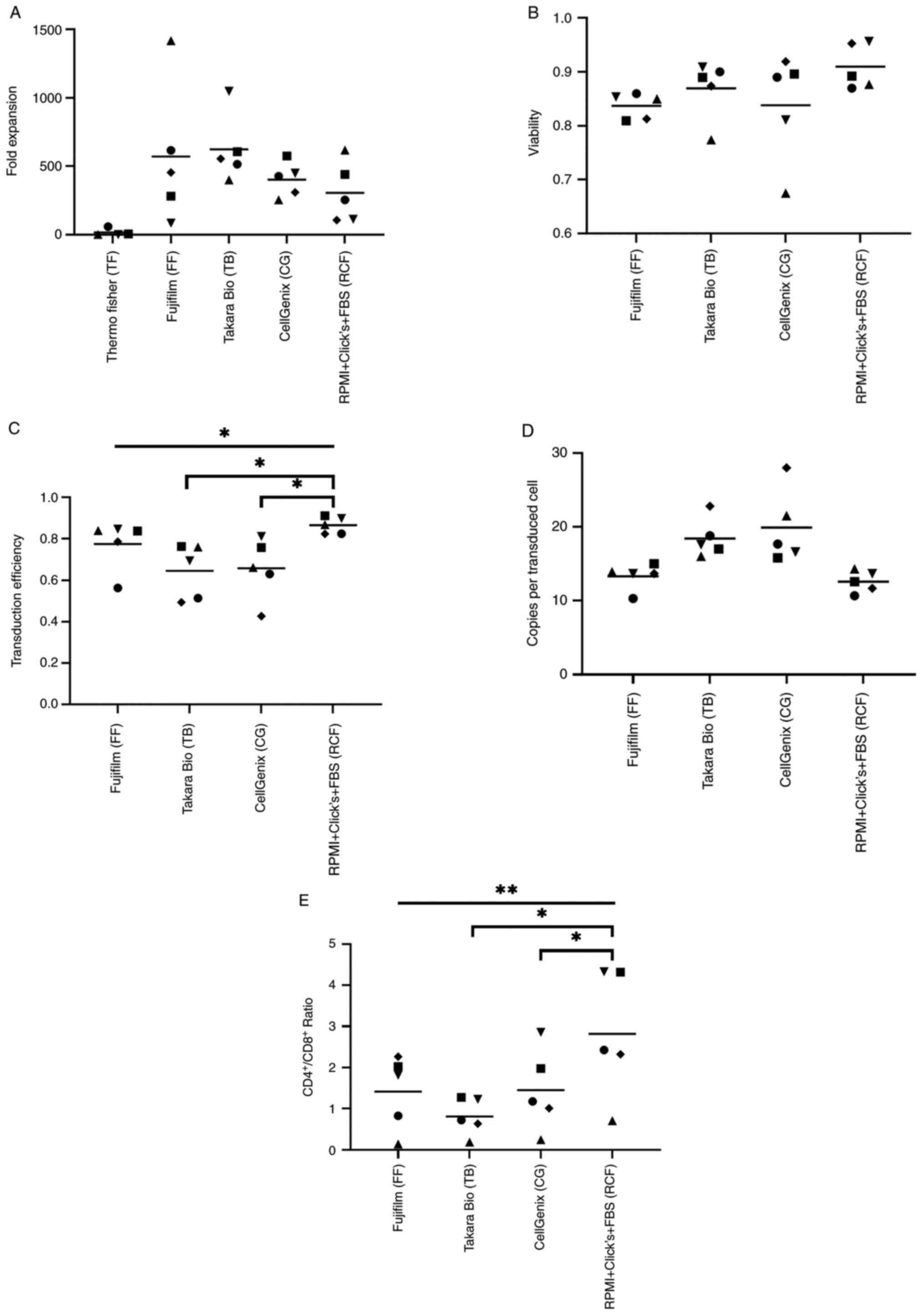
The expansion of the different CAR T-cell products
was assessed on day 12 following initial stimulation (Fig. 1A). No significant difference
(mixed-effects analysis, P=0.1248) was observed. T-cells cultured
in TF exhibited a low expansion and were excluded for further
analysis due to the small cell number which was often not
sufficient to reach similar cell concentrations in the experiments.
The viability was comparable for all four T-cell products (Fig. 1B). The transduction efficiency
(Fig. 1C) differed significantly
depending on the medium (one-way ANOVA, P=0.0094) and was highest
for the T-cells cultured in RPMI + Click's + FBS (RCF). Compared to
the cells cultured in RCF, the transduction efficiency for the
T-cells cultured in CG (Dunnett's test, P=0.0358) and TB (Dunnett's
test, P=0.0167) was lower. By contrast, a trend towards a lower
amount of integrated vector copies was noted for the CAR T-cells
manufactured in RCF and FF media, even though the difference did
not reach statistical significance (Fig. 1D). As regards the
CD4+/CD8+ ratio, CAR T-cell products cultured
in TB and CG contained a significantly lower population of
CD4+ cells than the cells cultured in RCF media
(Dunnett's test, P=0.0319 and P=0.0232, respectively; Fig. 1E).
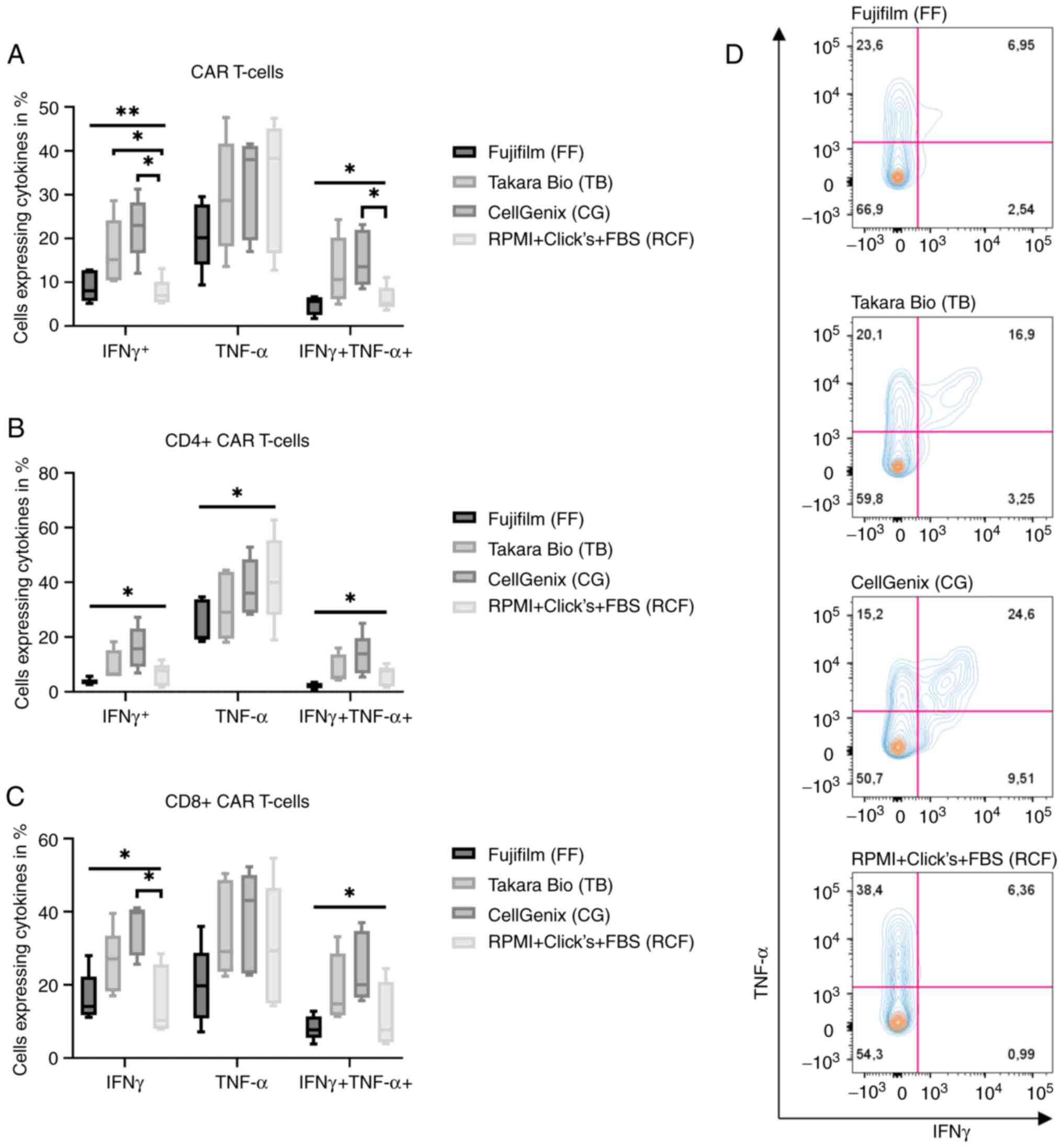
Increased IFNγ release of
third-generation CAR T-cells generated in serum-free media
In order to investigate whether the type of media
used for CAR T-cell production affects the secretion of stimulatory
cytokines upon antigen stimulation, the different CD19 CAR T-cell
products were co-cultured with CD19+ Daudi cells and the
intracellular expression of IFNγ and TNF-α was determined using
flow cytometry at 6 h following stimulation. IFNγ secretion
depended significantly on the media which was used for CAR T-cell
production (Fig. 2A; one-way
ANOVA, P=0.0098). CAR T-cells cultured in CG released the highest
levels of IFNγ in comparison to CAR T-cells cultured in RCF
(Dunnett' test, P=0.0194). The CAR T-cells produced in TB exhibited
the second highest release of IFNγ (Dunnett's test, P=0.0336). The
TNF-α release of the four different groups exhibited no significant
difference, whereas the percentage of CAR T-cells expressing IFNγ
and TNF-α was significant (one-way ANOVA, P=0.0231). CG yielded the
highest percentage of cells expressing IFNγ and TNF-α (Dunnett's
test, P=0.0399). The separate analysis of CD4+ and
CD8+ CAR T-cells revealed a similar result. The IFNγ
release of CD4+ CAR T-cells differed, depending on which
medium was used for production (one-way ANOVA, P=0.0167; Fig. 2B). The highest percentage of
IFNγ-expressing cells were the cells cultured in CG. The findings
for IFNγ- and TNF-α-positive cells in CD4+ CAR T-cells
were similar. The distribution of these cells depending on the
medium was significant (one-way ANOVA, P=0.0173), and the
CD4+ CAR T cells cultured in CG expressed the highest
levels of IFNγ and TNF-α. Additionally, the expression of TNF-α in
CD4+ CAR T-cells exhibited significant differences
(one-way ANOVA, P=0.0248). The cells produced in RCF had the
highest expression, whereas CD4+ CAR T-cells produced in
FF had the lowest level. In addition, the distribution of
IFNγ-releasing cells of CD8+ CAR T-cells was significant
(one-way ANOVA, P=0.0264; Fig.
2C) and again, CAR T-cells produced in CG had the highest
percentage of IFNγ-positive CD8+ CAR T-cells (Dunnett's
test, P=0.0450). The difference in IFNγ- and TNF-α-positive cells
was likewise significant (one-way ANOVA, P=0.0393). The
representative flow cytometry patterns in each medium plotted for
IFNγ and TNF-α and gated for CAR T-cells are presented in Fig. 2D.
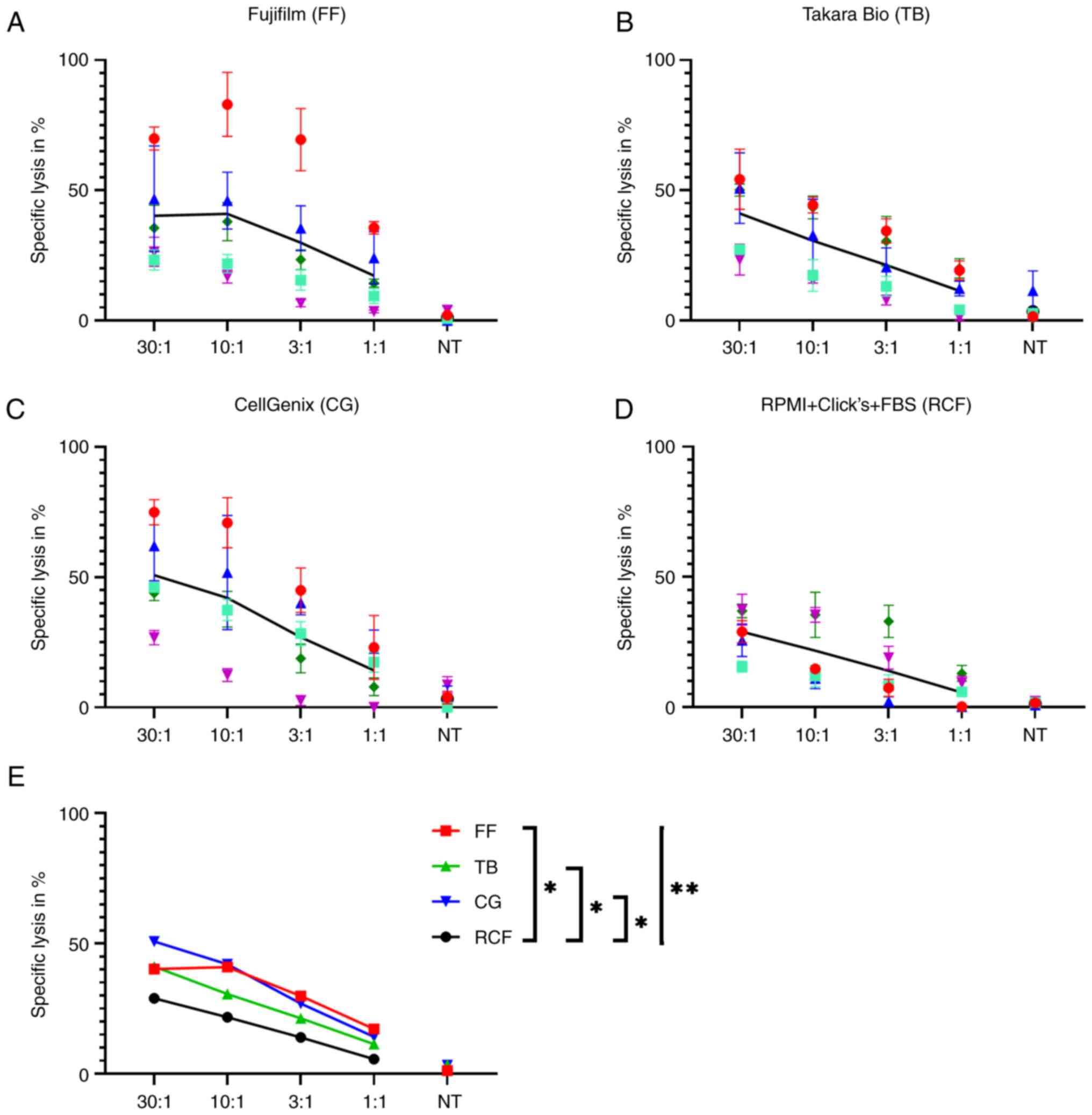
Serum-free media increase the short-term
cytotoxicity of third-generation CAR T-cells
The chromium release assay was performed to
investigate the short-term cytotoxicity of the different CAR T-cell
products, which can be reached within 4 h. The difference of the
percentage of killed tumor cells in the chromium release assay was
significant on average for all four effector-to-target-cell ratios
(Fig. 3). The P-value in a
one-way ANOVA test was P=0.0071. The CAR T-cells produced and
cultured in CG exhibited the highest cytotoxicity (CG vs. RCF:
Dunnett's test, P=0.0182). In addition, the CAR T-cells cultured in
FF exhibited a high cytotoxicity (FF vs. RCF: Dunnett's test,
P=0.0482) followed by the CAR T-cells cultured in TB (TB vs. RCF:
Dunnett's test, P=0.0428). The CAR T-cells cultured in RCF
exhibited the lowest cytotoxicity. Consequently, the lowest rate of
killed tumor cells was observed with CAR T cells generated with
FBS.
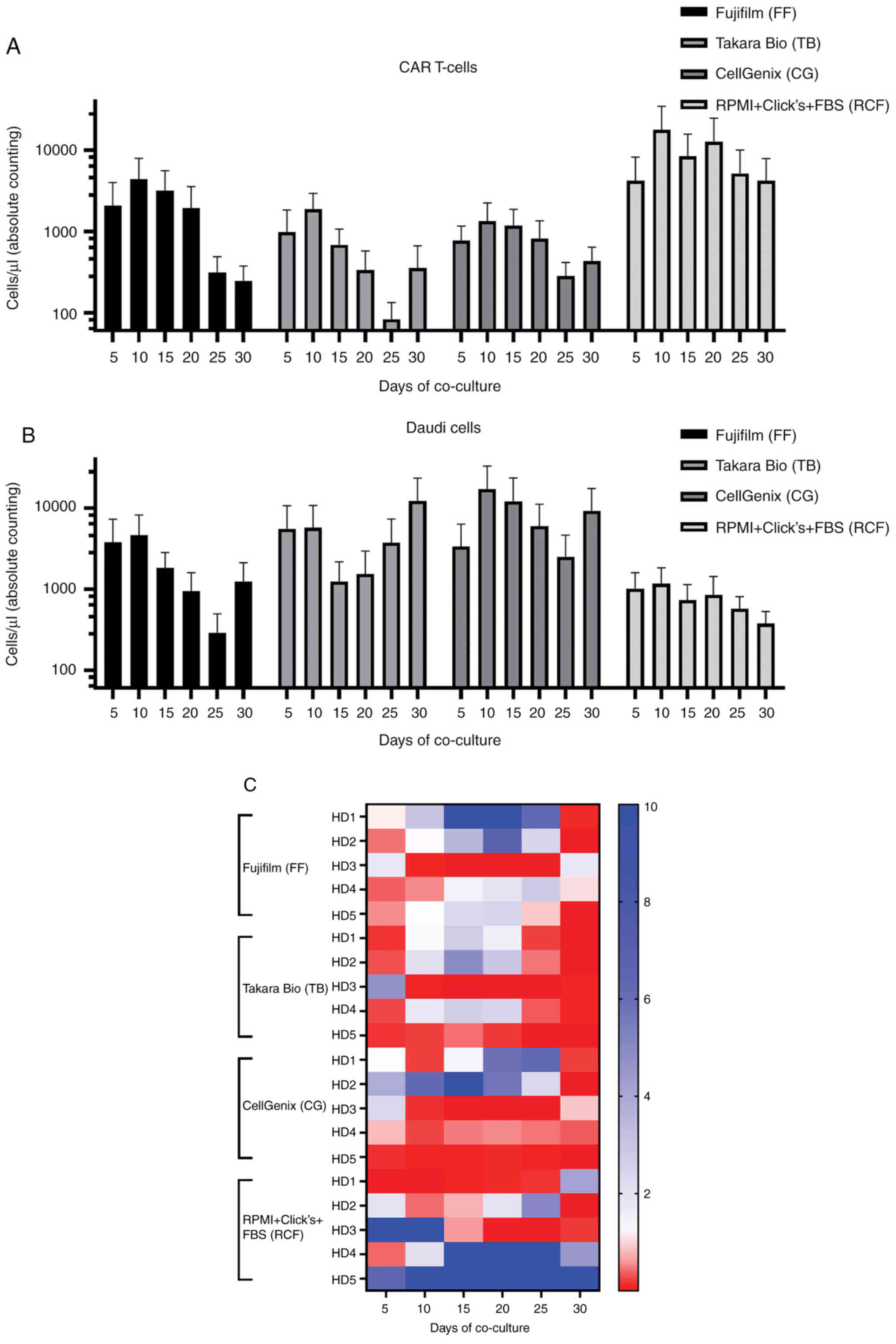
Serum-containing media improve the
long-term functionality of CAR T-cells
Long-term functionality was tested by the co-culture
of tumor cells and CAR T-cells. CAR T-cells were cultured in the
corresponding medium which was also used for expansion and on days
0, 5, 10, 15, 20 and 25 of co-culture, the tumor cells were added.
Assessment using flow cytometry was effective on days 5, 10, 15,
20, 25 and 30 prior to the addition of new tumor cells. The cells
cultured in RCF exhibited the highest expansion of CAR T-cells
(Fig. 4A) and these CAR T-cells
were most efficient in eliminating tumor cells (Fig. 4B) compared to all other CAR T-cell
products. The cells cultured in FF exhibited the second-best
result. The absolute cell counts of cells cultured in TB and CG
were comparable.
Another interesting aspect was the ratio of CAR
T-cells to Daudi cells for every single co-culture. These data are
presented in Fig. 4C. The
quotient was highest for cells cultured in RCF followed by cells
cultured in FF. Additionally, the donor-to-donor variability can be
observed in Fig. 4C. Therefore,
the different groups of co-culture were not compared statistically
and a descriptive analysis was performed. Consequently, RCF
improves the long-term functionality of CAR T-cells.
CAR T-cells cultured in serum-free media
exhibit a unique phenotype
The first phenotyping of CAR T-cells was completed
using flow cytometry on day 12 of CAR T-cell production (Fig. 5A and B). On day 12, the CAR
T-cells produced and cultured in FF exhibited a high percentage of
central memory CAR T-cells in comparison to the CAR T-cells
cultured in RCF (one-way ANOVA, P=0.0087; Dunnett's test, FF vs.
RCF, P=0.0470). The CAR T-cells cultured in TB and CG exhibited a
lower percentage of central memory than CAR T-cells cultured in RCF
(one-way ANOVA, P=0.0087; Dunnett's test, TB vs. RCF: P=0.0210 and
CG vs. RCF: P=0.0092, respectively). The distribution of naïve CAR
T-cells did not differ significantly between the CAR T-cells
cultured in TB and RCF; however, the CAR T-cells cultured in CG and
FF exhibited a significantly lower percentage of naïve CAR T-cells
(one-way ANOVA, P=0.0014; Dunnett's test, CG vs. RCF: P=0.0468 and
FF vs. RCF: P=0.0029, respectively). The percentage of effector
memory CAR T-cells cultured in FF, TB and CG did not differ
significantly in comparison to that of the CAR T-cells cultured in
RCF (one-way ANOVA, P=0.0172; Dunnett's test, not significant).
Only the CAR T-cells cultured in FF exhibited a significantly lower
percentage of terminally differentiated effector memory cells
(EMRA) in comparison to the CAR T-cells cultured in RCF (one-way
ANOVA, P=0.0098; Dunnett's test, FF vs. RCF, P=0.0010). Of note is
also the balanced distribution of all four CAR T-cell subtypes when
cultured in RCF.
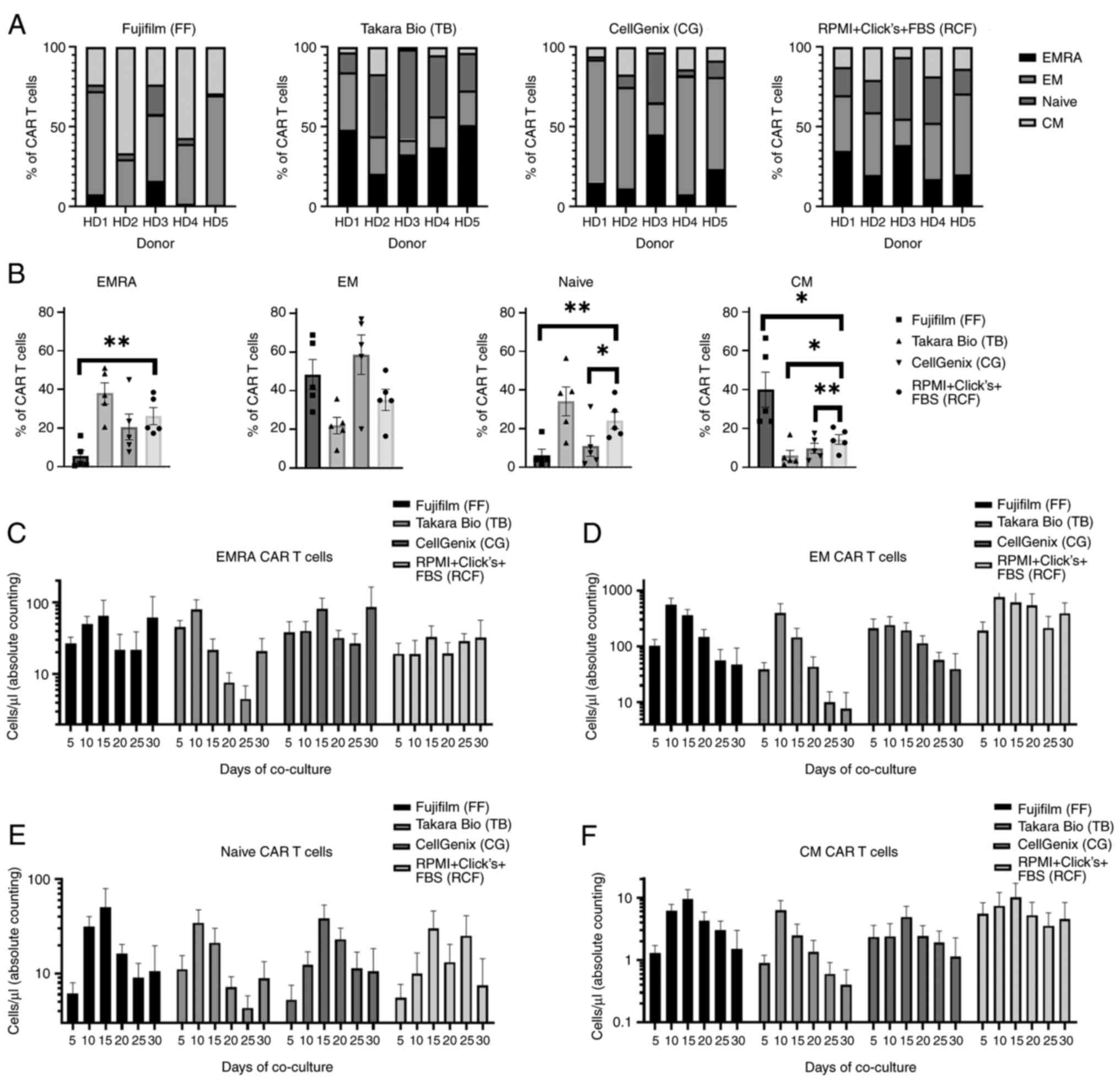 | Figure 5Phenotyping of CAR T-cells. (A)
Phenotyping of CAR T-cells on day 12 of production was assessed
using flow cytometry. Each donor is visualized separately. (B)
Phenotyping of CAR T-cells on day 12 visualized separately for
EMRA, EM, naïve and CM CAR T-cells. Data were analyzed using
one-way ANOVA of all five donors followed by Dunnett's multiple
comparisons test (represented as brackets). *P<0.05
and **P<0.01. Boxes show the mean and error bars
indicate SEM. (C-F) Phenotyping of CAR T-cells co-cultured with
tumor cells. Absolute cell count of CAR T-cells divided to the four
different CAR T-cell types, EMRA, EM, naïve and CM. Boxes show the
mean and error bars indicate SEM. CAR, chimeric antigen receptor;
EMRA, effector memory cells re-expressing RA; EM, effector memory
cells; naïve, naïve cells; CM, central memory cells. |
The phenotypic characteristics of the CAR T-cell
products were also determined upon repeated antigen stimulation
during the co-culture assay. The assessment of phenotypes was
repetitively performed for the entire co-culture (Fig. 5C-F). The CAR T-cells cultured in
FF and RCF exhibited the highest percentage of undifferentiated
cells; i.e., naïve and central memory cells. The CAR T-cells
cultured in RCF had the highest percentage of effector memory
cells, whereas the CAR T-cells cultured in CG and FF had the
highest cell count of EMRA CAR T-cells.
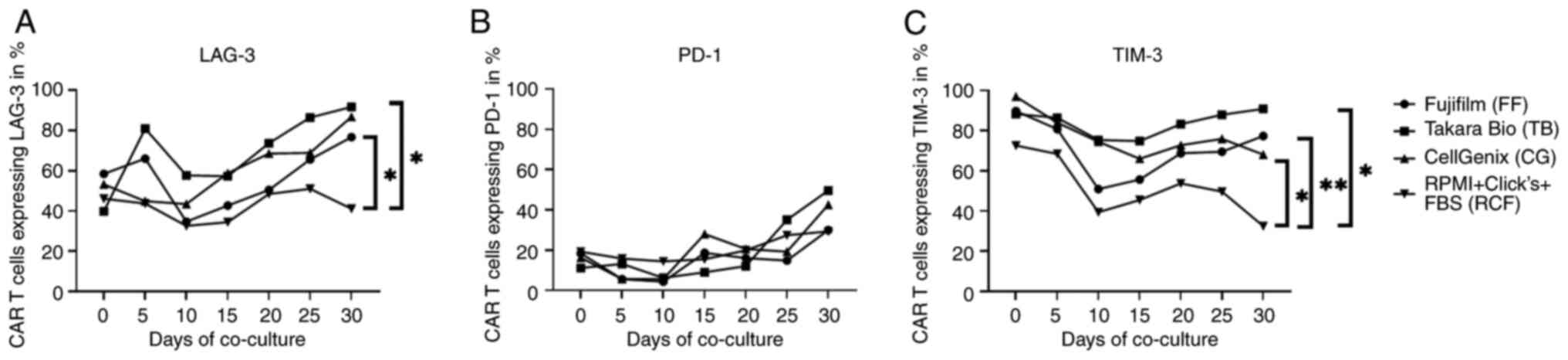
Serum-containing medium reduces the
exhaustion of third-generation CAR T-cells during long-term
co-culture
The expression of surface markers associated with
T-cell exhaustion, such as lymphocyte-activation gene 3 (LAG-3),
programmed cell death protein 1 (PD-1) and T-cell immunoglobulin
and mucin-domain containing-3 (TIM-3) were evaluated using flow
cytometry on day 12 of CAR T-cell production, which corresponds to
day 0 of co-culture and every 5 days after each antigen stimulation
thereafter (Fig. 6). The baseline
values prior to the start of the co-culture were similar for all
different CAR T-cell products. In general, a trend towards an
increase in the percentage of LAG-3-, PD-1- and TIM-3-expressing
T-cells was observed for all CAR T-cell products with repetitive
antigen stimulation. However, the CAR T-cells generated in RCF
exhibited a significantly lower expression of LAG-3 and TIM-3 at
day 30 than the CAR T-cells cultured in FF, TB and CG (LAG-3:
Dunnett's test, P=0.0205, P=0.0306 and not significant,
respectively; TIM-3: Dunnett's test, P=0.0034, P=0.0159 and
P=0.0294, respectively). By contrast, we didn't find any difference
in the level of PD-1 expression between the different groups.
Discussion
It was recently demonstrated that CAR T-cell therapy
is a promising approach which can be used to treat patients with
refractory and relapsed hematological malignancies (1-5).
Therefore, an increasing number of pharmaceutical companies and
academic institutions entered the GMP manufacturing process of CAR
T-cells. FBS and HS are widely used, leading to the intensive
testing of the different batches to reduce lot-to-lot
inconsistency. To overcome this time- and cost-consuming test
procedure, serum-free media may represent a potential solution.
Serum-free media often include pharmaceutical-grade
and/or recombinant human proteins. Pharmaceutical-grade human
proteins are obtained from pooled human plasma (24). The more donors are included, the
higher is the lot-to-lot consistency. However, differences between
the different batches cannot completely be excluded. Furthermore,
the purification process and several manufacturing steps lead to
the structural alternations of human proteins. Olsen et al
(25) reported an impaired
binding capacity of pharmaceutical-grade HS albumin for drugs and
named the stabilizers caprylic acid and N-acetyl-DL-tryptophan as
probable reason. Park et al (26) examined the crystal structure of
pharmaceutical-grade HS albumin, which had bound fatty acids and
tryptophan; they concluded a reduced biorelevance of HS
albumin.
By contrast, recombinant proteins are produced under
well-defined conditions, although the purification process also
alters the protein structure. The structure of recombinant HS
albumin was found to be of the same pharmaceutical grade as human
serum albumin (27-29). Consequently, Prime-XV™ T Cell CDM,
Fujifilm™ (FF) stands out due to the exclusive use of recombinant
human serum proteins. FF will have the highest lot-to-lot
consistency with the same structural quality. CTS™ OpTmizer™ Pro
SFM, Thermofisher Gibco™ (TF), LymphoONE™ T-Cell Expansion
Xeno-Free Medium, Takara Bio™ (TB) and TCM GMP-Prototype,
CellGenix™ (CG) all include pharmaceutical-grade HS albumin, which
is not completely chemically definable.
The present study observed a notably low expansion
rate in TF. According to the manufacturer, this medium is better
suited for workflows >15 days. Thus, this was excluded as the
experimental workflow used herein was too short for the use of
TF.
The transduction procedure and expansion of CAR
T-cells needs to be further improved with respect to media
formulations to achieve enhanced antitumor activity (18). Herein, no significant difference
was observed between the CAR T-cell cultures with four different
culture media regarding expansion, viability and vector copy
numbers. However, the transduction rate was lower in CAR T-cell
cultures with TB and CG, thus demonstrating that FF is better
suited for the transduction process than TB and CG.
In vitro potency assays reflect the
biological activity of CAR T-cell products and have to be performed
for the quality control of GMP production (30,31). They are currently used to evaluate
the functionality of CAR T-cell products, even though it is not
clear whether this is predictive of the in vivo antitumor
activity. In the present study, CAR T-cells cultured in CG
exhibited the highest short-time cytotoxicity in the chromium
release assay followed by FF and TB. These results suggest a
superior biological activity, whereas CAR T-cells cultured in RCF
had the worst in vitro potency. Moreover, CAR T-cells
cultured with CG exhibited the highest cytokine release followed by
CAR T-cells generated in TB. CAR T-cells cultured in FF and RCF had
lower cytokine levels, suggesting lower activation. However, CAR
T-cells with lower levels of cytokine release were found in
previous studies with lower levels of clinical toxicity and with
consistently good anti-lymphoma activity (32). Therefore, CAR T-cells generated in
FF appear to be advantageous, when compared to other serum-free
media due to the high potency observed in the chromium release
assay and low potential side-effects through lower cytokine levels.
However, it should be mentioned that CAR T-cells cultured in RCF
exhibited a better long-term functionality in the co-culture assay.
This assay was performed with the corresponding medium which was
also used for the production and expansion of CAR T-cells. This
fact probably influences the outcome of the single co-cultures and
highlights the importance of media providing nutrients.
Nevertheless, in vivo, all CAR T-cell products would face
the same serum-containing conditions. Therefore, the quality of the
starting product is considered of particular importance.
Furthermore, previous studies have suggested an
association between the frequency of CAR T-cells with a less
differentiated phenotype and long-term remission rates due to
enhanced persistence (33,34).
Subsequently, CAR T-cell effector function and therapeutic
potential is increased (35).
Blaeschke et al (36)
produced CAR T-cells with central memory and stem cell memory
subtype in an automated system, and reported high expansion rates,
specific cytotoxicity and cytokine expression. Apparently, CAR
T-cell cultures with mainly undifferentiated CAR T-cell subtypes
are not restricted as regards expansion and potency (34). In the present study, CAR T-cells
cultured in TB and particularly, FF exhibited a high frequency of
central memory and naïve CAR T-cells on day 12 of expansion. It can
be concluded that CAR T-cells cultured in these media have a
favorable phenotype, leading to long-term antitumor activity.
However, CAR T-cells cultured with FBS exhibited a superior
long-term cytotoxicity when co-cultured with CD19+
target cells. Additionally, CAR T-cells cultured in RCF exhibited
the lowest increase of exhaustion markers. The expression of the
surface markers, LAG3, PD-1 and TIM-3, is associated with the
exhaustion and senescence of T-cells (37). It can thus be concluded that the
use of serum-containing media during co-culture reduces the level
of exhaustion of CAR T-cells. However, in vivo, all CAR
T-cell products would experience the same conditions during
repetitive antigen stimulation. Nevertheless, a comparable quality
of RCF and the serum-free medium FF can be observed when
considering all aspects together (Table II).
 | Table IICharacteristics and rating of
different media. |
Table II
Characteristics and rating of
different media.
| Characteristic | Fujifilm (FF) | Takara (TB) | CellGenix (CG) | RPMI + Click's +
FBS (RCF) |
|---|
| Expansion | 3 | 4 | 2 | 1 |
| Viability | 1 | 3 | 2 | 4 |
| Transduction
efficiency | 3 | 1 | 2 | 4 |
|
CD4+/CD8+ ratio | 3 | 4 | 2 | 1 |
| Vector copy
number | 3 | 2 | 1 | 4 |
| Intracellular
staining | 1 | 3 | 4 | 2 |
| Chromium release
assay | 3 | 2 | 4 | 1 |
| Co-culture | 3 | 2 | 1 | 4 |
| Phenotype | 4 | 3 | 1 | 2 |
| Exhaustion | 3 | 1 | 2 | 4 |
Another critical aspect which should be considered,
is the wide range of costs of different media. The authors
calculated a price of ~440€/l for all components of the RCF medium
used herein. In comparison, TF, CG and TB are considerably more
affordable (~153€/l, ~240€/l and ~255€/l). FF is the only
serum-free medium which is more costly than the FBS-containing
medium, with ~636€/l. Consequently, the high costs of FF and RCF
are an economic disadvantage.
The use of serum, particularly FBS, is ethically and
ecologically controversial. For FBS manufacturing, >1,000,000
bovine fetuses are harvested annually (38). Hence, carbon dioxide emissions
should not be underestimated. Moreover, the harvest process is
ethically questionable. The bovine fetal blood is collected by a
cardiac puncture of the unanesthetized fetus (38).
Due to practical reasons for GMP standardization and
ethical and ecological concerns, it would be desirable to replace
current CAR T-cell culture media with serum-free media. However, it
should be mentioned that all serum-free media provide different
culture conditions with specific characteristics. Alnabhan et
al (17) observed a reduced
viability and expansion, a lower CD4/CD8 ratio and a higher
frequency of differentiated cells in CAR T-cell culture. By
contrast, Xu et al (39)
reported an improved proliferation environment in serum-free media
for T-cells. Thus, each serum-free medium needs to be regarded
individually (Table II). The
varying media formulations (Table
I) lead to this diversity.
Prime-XV™ T Cell CDM, Fujifilm™ (FF) exhibited some
advantages compared to TB and CG, as for example, the freedom of
pharmaceutical-grade human proteins, a high cytotoxicity, a lower
cytokine release and an undifferentiated phenotype. Based on these
observations, it would be reasonable to implement FF in GMP CAR
T-cell production.
In conclusion, the use of serum-free media can be
recommended for the GMP manufacturing of CAR T-cells. Serum-free
media do not lead to a loss of functionality or a slower expansion
of CAR T-cells. Serum-free media are produced under very robust
conditions. Given the unique characteristics of each serum-free
medium, it needs to be selected for each individual purpose.
Prime-XV™ T Cell CDM, Fujifilm™ (FF) proved advantageous in the
manufacturing process used herein compared to the two other
serum-free media.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
FE, AHK, MS, AS, CMT and AKe were involved in the
conceptualization of the study. FE, GJ, BN, TS and AKu were
involved in the study methodology. FE and GJ performed the analysis
of data. FE was involved in the formal analysis and in the
investigative aspects of the study, as well as in the writing and
preparation of the original draft. CMT, AS and MS were involved in
the provision of financial resources and laboratory facilities. FE,
AR, KB and AKu were involved in data curation. TS, AKe, AHK, BN,
KB, AS and MS were involved in the writing, reviewing and editing
of the manuscript. FE and GJ were involved in data visualization
and creating graphs. AK and MS supervised the study. FE and AHK
were involved in project administration. AKe and FE confirm the
authenticity of all the raw data. All authors have read and agreed
to the published version of the manuscript.
Ethics approval and consent to
participate
Ethical approval was obtained from the Medical
Faculty of the University of Heidelberg (reference no. S-254/2016).
Consent to participate was obtained in a written manner.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
Funding
No funding was received.
References
|
1
|
Maude SL, Laetsch TW, Buechner J, Rives S,
Boyer M, Bittencourt H, Bader P, Verneris MR, Stefanski HE, Myers
GD, et al: Tisagenlecleucel in Children and young adults with
B-cell lymphoblastic leukemia. N Engl J Med. 378:439–448. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Schuster SJ, Svoboda J, Chong EA, Nasta
SD, Mato AR, Anak Ö, Brogdon JL, Pruteanu-Malinici I, Bhoj V,
Landsburg D, et al: Chimeric antigen receptor T cells in refractory
B-cell lymphomas. N Engl J Med. 377:2545–2554. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Neelapu SS, Locke FL, Bartlett NL, Lekakis
LJ, Miklos DB, Jacobson CA, Braunschweig I, Oluwole OO, Siddiqi T,
Lin Y, et al: Axicabtagene ciloleucel CAR T-cell therapy in
refractory large B-cell lymphoma. N Engl J Med. 377:2531–2544.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Locke FL, Ghobadi A, Jacobson CA, Miklos
DB, Lekakis LJ, Oluwole OO, Lin Y, Braunschweig I, Hill BT,
Timmerman JM, et al: Long-term safety and activity of axicabtagene
ciloleucel in refractory large B-cell lymphoma (ZUMA-1): A
single-arm, multicentre, phase 1-2 trial. Lancet Oncol. 20:31–42.
2019. View Article : Google Scholar :
|
|
5
|
Kochenderfer JN, Dudley ME, Kassim SH,
Somerville RP, Carpenter RO, Stetler-Stevenson M, Yang JC, Phan GQ,
Hughes MS, Sherry RM, et al: Chemotherapy-refractory diffuse large
B-cell lymphoma and indolent B-cell malignancies can be effectively
treated with autologous T cells expressing an anti-CD19 chimeric
antigen receptor. J Clin Oncol. 33:540–549. 2015. View Article : Google Scholar :
|
|
6
|
Beyar-Katz O and Gill S: Advances in
chimeric antigen receptor T cells. Curr Opin Hematol. 27:368–377.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Braendstrup P, Levine BL and Ruella M: The
long road to the first FDA-approved gene therapy: Chimeric antigen
receptor T cells targeting CD19. Cytotherapy. 22:57–69. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Crees ZD and Ghobadi A: Cellular therapy
updates in B-cell lymphoma: The state of the CAR-T. Cancers
(Basel). 13. pp. 51812021, View Article : Google Scholar
|
|
9
|
Schubert ML, Schmitt A, Sellner L, Neuber
B, Kunz J, Wuchter P, Kunz A, Gern U, Michels B, Hofmann S, et al:
Treatment of patients with relapsed or refractory CD19+ lymphoid
disease with T lymphocytes transduced by RV-SFG.CD19.CD28.4-1BBzeta
retroviral vector: A unicentre phase I/II clinical trial protocol.
BMJ Open. 9:e0266442019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Abramson JS: Anti-CD19 CAR T-cell therapy
for B-cell non-Hodgkin lymphoma. Transfus Med Rev. 34:29–33. 2020.
View Article : Google Scholar
|
|
11
|
Enblad G, Karlsson H, Gammelgård G, Wenthe
J, Lövgren T, Amini RM, Wikstrom KI, Essand M, Savoldo B, Hallböök
H, et al: A phase I/IIa trial using CD19-targeted third-generation
CAR T cells for lymphoma and leukemia. Clin Cancer Res.
24:6185–6194. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang L, Gong W, Wang S, Neuber B, Sellner
L, Schubert ML, Hückelhoven-Krauss A, Kunz A, Gern U, Michels B, et
al: Improvement of in vitro potency assays by a resting step for
clinical-grade chimeric antigen receptor engineered T cells.
Cytotherapy. 21:566–578. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Heger JI, Froehlich K, Pastuschek J and
Schmidt A, Baer C, Mrowka R, Backsch C, Schleußner E, Markert UR
and Schmidt A: Human serum alters cell culture behavior and
improves spheroid formation in comparison to fetal bovine serum.
Exp Cell Res. 365:57–65. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Brentjens RJ, Rivière I, Park JH, Davila
ML, Wang X, Stefanski J, Taylor C, Yeh R, Bartido S, Borquez-Ojeda
O, et al: Safety and persistence of adoptively transferred
autologous CD19-targeted T cells in patients with relapsed or
chemotherapy refractory B-cell leukemias. Blood. 118:4817–4828.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hollyman D, Stefanski J, Przybylowski M,
Bartido S, Borquez-Ojeda O, Taylor C, Yeh R, Capacio V, Olszewska
M, Hosey J, et al: Manufacturing validation of biologically
functional T cells targeted to CD19 antigen for autologous adoptive
cell therapy. J Immunother. 32:169–180. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kalos M, Levine BL, Porter DL, Katz S,
Grupp SA, Bagg A and June CH: T cells with chimeric antigen
receptors have potent antitumor effects and can establish memory in
patients with advanced leukemia. Sci Transl Med. 3:95ra732011.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Alnabhan R, Gaballa A, Mörk LM, Mattsson
J, Uhlin M and Magalhaes I: Media evaluation for production and
expansion of anti-CD19 chimeric antigen receptor T cells.
Cytotherapy. 20:941–951. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ghassemi S, Martinez-Becerra FJ, Master
AM, Richman SA, Heo D, Leferovich J, Tu Y, García-Cañaveras JC,
Ayari A, Lu Y, et al: Enhancing chimeric antigen receptor T cell
anti-tumor function through advanced media design. Mol Ther Methods
Clin Dev. 18:595–606. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Smith C, Økern G, Rehan S, Beagley L, Lee
SK, Aarvak T, Schjetne KW and Khanna R: Ex vivo expansion of human
T cells for adoptive immunotherapy using the novel xeno-free CTS
immune cell serum replacement. Clin Transl Immunology. 4:e312015.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Coeshott C, Vang B, Jones M and Nankervis
B: Large-scale expansion and characterization of CD3+
T-cells in the quantum® cell expansion system. J Transl
Med. 17:2582019. View Article : Google Scholar
|
|
21
|
Lamoreaux L, Roederer M and Koup R:
Intracellular cytokine optimization and standard operating
procedure. Nat Protoc. 1:1507–1516. 2006. View Article : Google Scholar
|
|
22
|
Kunz A, Gern U, Schmitt A, Neuber B, Wang
L, Hückelhoven-Krauss A, Michels B, Hofmann S, Müller-Tidow C,
Dreger P, et al: Optimized assessment of qPCR-based vector copy
numbers as a safety parameter for GMP-grade CAR T cells and
monitoring of frequency in patients. Mol Ther Methods Clin Dev.
17:448–454. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Livark KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
24
|
Díez JM, Bauman E, Gajardo R and Jorquera
JI: Culture of human mesenchymal stem cells using a candidate
pharmaceutical grade xeno-free cell culture supplement derived from
industrial human plasma pools. Stem Cell Res Ther. 6:282015.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Olsen H, Andersen A, Nordbø A, Kongsgaard
UE and Børmer OP: Pharmaceutical-grade albumin: Impaired
drug-binding capacity in vitro. BMC Clin Pharmacol. 4:42004.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Park J, Kim MS, Park T, Kim YH and Shin
DH: Crystal structure of pharmaceutical-grade human serum albumin.
Int J Biol Macromol. 166:221–228. 2021. View Article : Google Scholar
|
|
27
|
Sumi A, Okuyama K, Kobayashi K, Ohtani W,
Ohmura T and Yokoyama K: Purification of recombinant human serum
albumin efficient purification using STREAMLINE. Bioseparation.
8:195–200. 1999. View Article : Google Scholar
|
|
28
|
Ohtani W, Masaki A, Ikeda Y, Hirose M,
Chuganji M, Takeshima K, Kondo M, Sumi A and Ohmura T: Structure of
recombinant human serum albumin from Pichia pastoris. Yakugaku
Zasshi. 117:220–232. 1997.In Japanese. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Watanabe H, Yamasaki K, Kragh-Hansen U,
Tanase S, Harada K, Suenaga A and Otagiri M: In vitro and in vivo
properties of recombinant human serum albumin from Pichia pastoris
purified by a method of short processing time. Pharm Res.
18:1775–1781. 2001. View Article : Google Scholar
|
|
30
|
European Medicines Agency: Guideline on
potency testing of cell based immunotherapy medicinal products for
the treatment of cancer. 2016.
|
|
31
|
Food and Drug Administration: Guidance for
industry: Potency tests for cellular and gene therapy products.
2011.
|
|
32
|
Brudno JN, Lam N, Vanasse D, Shen YW, Rose
JJ, Rossi J, Xue A, Bot A, Scholler N, Mikkilineni L, et al: Safety
and feasibility of anti-CD19 CAR T cells with fully human binding
domains in patients with B-cell lymphoma. Nat Med. 26:270–280.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Oliveira G, Ruggiero E, Stanghellini MT,
Cieri N, D'Agostino M, Fronza R, Lulay C, Dionisio F, Mastaglio S,
Greco R, et al: Tracking genetically engineered lymphocytes
long-term reveals the dynamics of T cell immunological memory. Sci
Transl Med. 7:317ra1982015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hoffmann JM, Schubert ML, Wang L,
Hückelhoven A, Sellner L, Stock S, Schmitt A, Kleist C, Gern U,
Loskog A, et al: Differences in expansion potential of naive
chimeric antigen receptor T cells from healthy donors and untreated
chronic lymphocytic leukemia patients. Front Immunol. 8:19562018.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ghassemi S, Nunez-Cruz S, O'Connor RS,
Fraietta JA, Patel PR, Scholler J, Barrett DM, Lundh SM, Davis MM,
Bedoya F, et al: Reducing ex vivo culture improves the antileukemic
activity of chimeric antigen receptor (CAR) T cells. Cancer Immunol
Res. 6:1100–1109. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Blaeschke F, Stenger D, Kaeuferle T,
Willier S, Lotfi R, Kaiser AD, Assenmacher M, Döring M, Feucht J
and Feuchtinger T: Induction of a central memory and stem cell
memory phenotype in functionally active CD4+ and
CD8+ CAR T cells produced in an automated good
manufacturing practice system for the treatment of CD19+
acute lymphoblastic leukemia. Cancer Immunol Immunother.
67:1053–1066. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wherry EJ and Kurachi M: Molecular and
cellular insights into T cell exhaustion. Nat Rev Immunol.
15:486–499. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Jochems CE, van der Valk JB, Stafleu FR
and Baumans V: The use of fetal bovine serum: Ethical or scientific
problem? Altern Lab Anim. 30:219–227. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Xu H, Wang N, Cao W, Huang L, Zhou J and
Sheng L: Influence of various medium environment to in vitro human
T cell culture. In Vitro Cell Dev Biol Anim. 54:559–566. 2018.
View Article : Google Scholar : PubMed/NCBI
|