1. Introduction
Diabetes mellitus (DM) is a chronic disease
characterized by a persistent increase in blood glucose (1), which is caused by dysfunctional
insulin secretion, inefficient use of insulin by cells in
peripheral tissues due to insulin resistance, or both. The
prevalence of DM has increased by >2-fold from 1980 to 2021
(from 4 to 10.5%), and is predicted to rise to 11.3% by 2030 and to
12.2% by 2045 (2). DM is one of
the 10 top causes of mortality worldwide, thereby constituting a
serious health problem (2).
Vascular complications of the disease, such as blindness, kidney
failure, heart attack, stroke and lower limb amputation, are the
most frequent causes of mortality in patients with diabetes in the
short and medium term (3).
DM is recognized as a complex disease caused by a
combination of lifestyle and genetic factors (4). Although numerous genetic and
non-genetic risk factors interact to trigger DM and its vascular
complications, the predictive ability of genetic models remains
modest (5). Genetic models are
based on combining several gene variants or risk alleles associated
with the disease through a genetic risk score (GRS), for instance,
variants of genes such as TFCL7, PPARG,
KCNJ11, SLC30A8, HHEX, CDKAL1,
IGF2BP2 and CDKN2A/B for type 2 diabetes (T2D)
(6). Although at least 40 risk
alleles have been identified for T2D, the predictive power of the
GRS is low due to the small effect size of a number of the genetic
loci and thus the small added value of genetic risk compared with
clinical risk factors (7,8).
These clinical risk factors are phenotype-based and have higher
predictive ability, for example, body mass index, dietary habits
and glycated hemoglobin level. On the other hand, there is a lack
of appropriate models for studies of gene-gene and gene-environment
interactions in the risk prediction of DM (5). In addition, the susceptibility to
genetic factors related to the onset of macrovascular and
microvascular complications in patients with DM does not explain
all of the phenotypic variation observed during the disease course
(9). Therefore, research on the
elements involved in genetic expression control has garnered
attention in an to attempt to explain the missing heritability of
DM.
The role of non-coding RNA in the regulation of gene
expression has been investigated in the last 20 years (10-18). Covalently closed circular RNAs
(circRNAs) are an important class of non-coding RNA that have a
widespread and specific expression in cells and tissues, which are
also stable and highly conserved between species (19-22). These molecules can act as sponges
of microRNAs (miRNAs/miRs) or proteins to regulate the
transcription of their parental genes or the translation of their
targets, and consequently serve an important role in different
physiological and pathological processes (23).
Several reports have linked circRNAs to the
development and progression of different diseases, including DM and
its vascular complications (24-26). CircRNAs can modulate the
expression of extracellular matrix (ECM) components, such as
fibronectin (FN) and type IV collagen (ColIV), which are implicated
in the vascular complications of DM (27).
Although dysregulated expression of circRNAs has
been reported in diabetic nephropathy (DN) (28-30), the role of these molecules in the
modulation of the advanced glycation end products (AGE)-receptor
for AGE (RAGE) pathway by direct or indirect interaction with the
proteins that constitute this signaling pathway remains unknown.
Identifying these aspects may help to characterize the role of
circRNAs in the pathophysiology of DN, and their potential as
therapeutic targets or biomarkers of the disease.
The present study aimed to review the current
knowledge on circRNAs implicated in DN-related cell processes.
Moreover, novel potential interactions that could take place
between circRNAs expressed in renal cells under high-glucose
concentrations and the transcription factors c-Jun and c-Fos are
reported.
2. CircRNA structure, biogenesis and
degradation
CircRNAs are covalently closed circular
single-stranded RNA molecules, which can be in the nucleus,
cytoplasm or in exosomes (Fig.
1A). CircRNAs are derived from mRNA backsplicing in which the
upstream 5′ splice site of mRNA is linked to a downstream 3′ splice
site. The formation of this structure can be driven by direct
backsplicing with Arthrobacter luteus elements and inverted
repeat complementation, lariat circularization, or it can be
mediated by RNA-binding proteins (RBPs) (31,32). Consequently, circRNAs can be
generated only from the exon regions of their parental gene [exonic
circRNAs (EcircRNAs)], from lariat introns [circular intronic RNAs
(ciRNAs)] or from exons with retained introns [exon-intron circRNAs
(EIciRNAs)] (Fig. 1B).
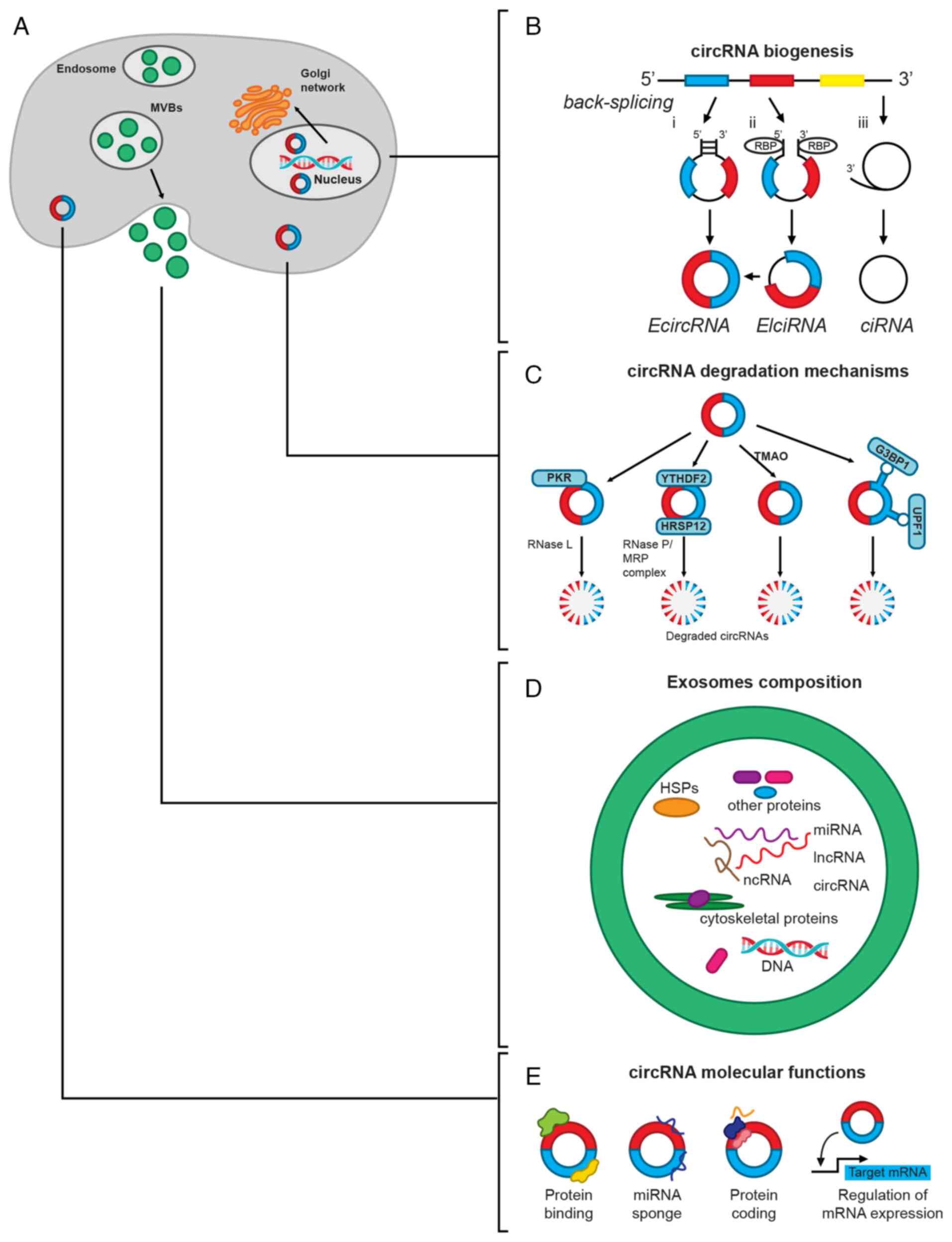 | Figure 1CircRNA biogenesis, structure,
degradation mechanisms and molecular functions. (A) CircRNAs are
located in the nucleus, cytoplasm or exported as exosomes. (B)
CircRNAs are formed by a backsplicing process driven by i) the
hybridization of introns with Arthrobacter luteus sequences
or inverted repeats; or ii) RBPs that bring the exons closer, both
of which generate EcircRNAs or EIciRNAs; or iii) the splicing of an
intron lariat, which generates the circRNAs. The colored boxes
illustrate exons, while the thin black lines indicate introns. (C)
Degradation of circRNAs by RNase L and RNase P/MRP is mediated by
PKR or RBPs (YTHDF2/HSRP12), or it is driven by the binding of
protein-stabilizers (TMAO), RNA helicases, and ATPases (UPF1) or
GTPases (G3BP1) to the circRNA. (D) CircRNAs can also be secreted
in exosomes together with other non-coding RNAs (miRNAs and
lncRNAs) and proteins, which are originated by the fusion of
multivesicular bodies with the plasma membrane and are degraded in
this way. (E) Regulation of gene expression mediated by circRNAs
mainly occurs by its binding to proteins or miRNAs, albeit circRNAs
can also be translated into proteins or directly interfere with the
translation of other proteins or the expression of their parental
genes. CircRNA, circular RNA; ciRNAs, circular intronic RNAs;
EcircRNAs, exonic circRNAs; EIciRNAs, exon-intron circRNAs; RBPs,
RNA-binding proteins; PKR, protein kinase R; RNase, ribonuclease;
YTHDF2, YTH N6-methyladenosine RBP F2; HSRP12, human heat response
protein 12; TMAO, trimethylamine-n-oxide; UPF1, up-frameshift
protein 1; G3BP1, Ras-GapSH3 domain-binding protein 1; miRNAs,
microRNAs; lncRNAs, long non-coding RNAs; MVB, multivesicular
bodies. |
According to their origins, different types of
circRNAs are located in different cell compartments. EcircRNAs are
mainly found in the cytoplasm and are usually the most abundant,
constituting ~80% of all the known circRNAs, whereas ciRNAs and
EIciRNAs are mainly located in the nuclei. The circRNA parental
gene can produce diverse isoforms, but usually only one circRNA
isoform is expressed at significant levels (15). CircRNAs are produced in the
nucleus and are transported into the cytoplasm via different
methods, depending on the length of mature circRNAs and their
N6-methyladenosine (m6A) modification status. In human cells,
circRNAs are transported into the cytoplasm by URH49 and UAP56,
which are two related DEAD box RNA helicases that compose the mRNA
processing/nuclear-exporting machinery and regulate gene expression
(33). The nuclear export of
short circRNAs (<400 nt) is regulated by URH49, whereas the
export of long circRNAs (>1,200 nt) is controlled by UAP56. The
export mechanism for circRNAs with intermediate lengths has not
been completely elucidated (34,35).
Mechanisms for circRNA degradation are mediated by
ribonucleases, by the formation of circRNA-protein complexes, by
the m6A modification of circRNA or by its packing into exosomes
(35) (Fig. 1C and D). Certain circRNAs form
DNA:RNA hybrids, displacing a single-stranded DNA (R-loops), which
are susceptible to degradation by ribonuclease (RNase) H1. On the
other hand, after cell inflammation or viral infection, circRNAs
can be degraded by activated RNase L (35) (Fig. 1C).
M6A-modified circRNAs undergo ribonuclease-mediated
cleavage via the YTH N6-methyladenosine RBP F2 (YTHDF2)-human heat
response protein 12 (HSRP12)-RNase P/MRP axis (36). YTHDF2 is a YTY-domain-containing
protein and is classified as a reading protein that recognizes
m6A-modified circRNAs, and, using HSRP12 as an adapter, binds to
RNase P/MRP to induce degradation of YTHDF2-bound circRNAs
(Fig. 1C). Degradation of
circRNAs is also mediated by trimethylamine-n-oxide, a byproduct of
high sugar and fat diet metabolism of gut microbiota (35) (Fig. 1C).
Argonaute 2 protein, a member of the Argonaute
family, also mediates circRNA degradation by recognizing, cleaving
and degrading the circRNA-miRNA complex. Other proteins, such as
glycine-trytophan protein of 182 kDa, which has an Ago-binding
domain and an RNA-recognition motif among other domains, regulate
the degradation of certain circRNAs in an Ago-independent manner
(35). Another two RBPs, namely
up-frameshift protein 1 (UPF1) and Ras-GapSH3 domain-binding
protein 1 (G3BP1), which exhibit helicase and GTPase activity,
respectively, regulate the degradation of circRNAs depending on the
highly folded tridimensional structure present in the majority of
these molecules (35) (Fig. 1C).
CircRNAs can also be released from cells packed in
exosomes or microvesicles (Fig.
1D), which facilitates their detection and isolation. This is
the most important cell mechanism for circRNA removal, and is also
a way of regulating intercellular communication through these
molecules (35-37). Notably, circRNAs are more
abundant than linear mRNAs in exosomes, compared with their
abundance in respective parental cells, and they are also more
stable, with a half-life of >48 h (38,39). Thus, the presence of large
quantities of intact and stable circRNAs in human serum and urine
exosomes, alongside their easier detection by liquid biopsies, has
suggested the use of circRNAs as a novel diagnostic biomarker and
therapeutic target for different diseases, such as cardiovascular
diseases, neurological disorders, tumors and renal diseases
(40,41).
3. Functions of circRNAs
CircRNAs can regulate gene expression by acting as
sponges for miRNAs or proteins (38,39). Other functions include
participating as scaffold and cellular translocators (38), as well as regulating the
expression of their parental gene (42), the translation of other proteins
(43) or their translation to
proteins (44) (Fig. 1E). CircRNAs are considered
competing/competitive endogenous RNAs because they contain multiple
miRNA response elements that competitively bind miRNAs, thus
modulating the regulatory function of these molecules (45).
An example of a cytoplasmic circRNA that functions
as a miRNA sponge is the cerebellar degeneration related protein 1
antisense transcript/ciRS-7, which has ~70 conserved binding sites
for miR-7 and forms a complex with Ago proteins, thus suppressing
the degradation of miR-7 target mRNAs (12,39).
CircRNAs also contain binding sites for several RBPs
(46-49). The splicing, nuclear export,
stability and subcellular localization of mRNAs are all modulated
by RBPs (50), such as are Ago
proteins, RNA polymerase II and fused in sarcoma (FUS) protein
(51). RBPs, besides mediating
the backsplicing that drives RNA circularization (Fig. 1B), interact with circRNAs to
regulate different processes, such as cell proliferation,
apoptosis, cancer cell metastasis, angiogenesis, mRNA translation,
energy metabolism and cell differentiation (50).
CircRNAs derived from a specific locus may have
binding sites for the protein codified by that locus or another
RBP, thus preventing the binding of such proteins to other targets
or the mRNA transcribed from the parental gene of the circRNA
(52).
CircRNAs may also interact with proteins to modulate
their translocation into the nucleus, consequently regulating gene
transcription. For example, circRNA_Amotl1 increases the nuclear
translocation of STAT3 to regulate the expression of its target
genes (53). By contrast,
circRNAs can maintain the nuclear retention of the c-Myc protein,
increasing its stability and binding affinity to different
promoters (52). In addition,
circRNAs can act as scaffolds for assembling protein complexes,
thus regulating several cellular functions (19). Besides, these RNA molecules can
circulate in exosomes in body fluids (40). It has been reported that circRNAs
packed in exosomes are regulated by modifying the levels of their
target miRNAs in the cells (40).
Previous studies have reported the potential coding
properties of circRNAs, albeit at low translational efficiency.
This fact is supported by the presence in some circRNAs of an
internal ribosomal entry site able to interact with the 40S subunit
of the eukaryotic ribosome and an open reading frame ready to be
translated into a polypeptide chain. For example, the zinc finger
protein 609 circRNA (circRNA_ZNF609) can be translated into a novel
ZNF609 protein isoform, which has a potential function during
myogenesis (44). CircRNAs may
also drive the translation of mRNA by binding to the mature
transcript and prevent the start of translation by blocking the
interaction between eukaryotic translation initiation factor 4G
(EIF4G) and poly-A binding protein (43). It has also been reported that
circRNAs may commonly exhibit m6A modifications in response to
environmental factors, which promote their protein translation in
human cells (27,45).
4. Regulation of the expression of circRNA
parental genes
CircRNAs regulate the transcription of their
parental genes in several ways (42). One of the methods includes
invading the RNA-binding sites in the parental gene, thus blocking
the binding of its linear isoform to the corresponding DNA sequence
(54). Another mechanism is the
conformation of DNA-RNA triple helix (R-loops), which hinders DNA
replication (55). The
transcription of the parental genes of circRNAs can also be
regulated in a cis-acting way (56). For example, circRNAs may interact
with U1 small nuclear ribonucleoproteins and RNA polymerase II at
the parental gene promoters, thus activating their initiation of
transcription. Once transcription is initiated, the expression of
circRNAs is increased generating a positive feedback loop. The
transcription of the parental gene could be suppressed if the
circRNAs interact with transcription factors that promote the
expression of such parental genes (52).
CircRNAs may activate intronic enhancers, or induce
hypomethylation at the promoter of their parental genes and
activate their transcription (57). For example, circRNA_ FECR1
activates the transcription of follicular lymphoma 1 thus
regulating the metastatic process of breast cancer (58). Another mechanism of circRNA
parental gene regulation is to sequestrate a miRNA that targets a
transcription factor, as occurs in the
circRNA_STAT3/miR-29a/b/c-3p/glioma-associated oncogene family zinc
finger 2 axis, which promotes the progression of hepatoblastoma
(59).
The ratio between mRNA-circRNA counterparts may be
≤10:1 due to the competition between backsplicing and linear
splicing (60). Consequently,
the biogenesis of circRNAs usually provokes a reduction in
protein-coding mRNA levels and inhibition of parental gene
expression (52).
5. CircRNAs in the pathogenic process of
DN
DN is a chronic microvascular complication of DM,
which is distinguished by the presence of capillary glomerular
circulation damage, which provokes alterations in renal structure
and function (61). This disease
appears in 30-40% of patients with DM, usually after the first 10
years of disease progression (62). DN is clinically characterized by
proteinuria, reduced glomerular filtration rate and high blood
pressure, and is the most frequent cause of end-stage renal disease
(62). This clinical syndrome is
mainly determined by an imbalance between the synthesis and
degradation of the ECM components, which provokes their
accumulation, as well as the generation of reactive oxygen species
(ROS), inflammatory cells recruitment and cytokines release
(63). Due to these processes,
the glomerular basal membranes and renal tubules thicken, followed
by an increase in the volume of the mesangial matrix and glomeruli
(64). These events finally lead
to inflammation, tubule interstitial renal fibrosis, glomerular
sclerosis and tubular atrophy.
Hyperglycemia and oxidative stress are essential
mediators in the progression of DN through the formation and
intracellular deposition of AGEs (63,65), which are the products of
nonenzymatic glycation and oxidation of proteins and lipids
(65). AGEs bind to a receptor
at the cell surface (i.e. RAGE), which is abundant in kidney
podocytes and endothelial cells; thus, the kidneys are considered a
major site for AGE clearance (66). The AGE-RAGE interaction triggers
multiple signaling cascades that provoke several pathophysiological
effects, including cell cycle arrest, apoptosis, increased cell
invasion, proliferation and cell migration, and generation of
pro-inflammatory cytokines (67,68). For that reason, the increase in
AGE formation, the interaction of AGE and RAGE, and the further
activation of the associated intracellular signaling pathways have
been implicated in the pathogenesis of DN (67,68).
CircRNAs are physiological regulators of multiple
intracellular signaling pathways (52); this function is performed
indirectly through their interactions with miRNAs or directly with
proteins. Several studies have revealed a differential expression
profile of circRNAs in kidney cell lines incubated in high glucose,
animal models of DN and patients with DN compared with healthy
individuals (26-29,69-111). A total of 35 dysregulated
circRNAs targeting miRNAs have been identified in these previous
studies (Table SI). Of them,
~2/3 are upregulated, one circRNA (circRNA_HIPK3) is upregulated or
downregulated depending on the cell type, and the remaining
circRNAs are downregulated (Table
SI). The target of the identified circRNA-miRNA pairs, as well
as the dysregulated axes, have been detected in the majority of
cases (28/35; Fig. 2 and
Table SI). In most cases, the
miRNA target of each circRNA and the mRNA target of the miRNA are
suggested through expression studies in the cells or tissues of
interest or bioinformatics analysis. Once a negative correlation
between the expression of circRNA/miRNA and miRNA/mRNA pairs is
verified, the deregulated axes are identified by either silencing
or overexpressing the circRNA or miRNA. Then, the expression of the
mRNA target, the abundance of its protein, as well as of the
effector molecules involved in the molecular processes related to
DN are measured (69-104). Fig. 2 shows the best characterized
targets and the regulated molecular processes related to DN. The
majority of the studied molecules mediate ECM accumulation and
fibrosis, whereas circRNAs regulating oxidative stress are less
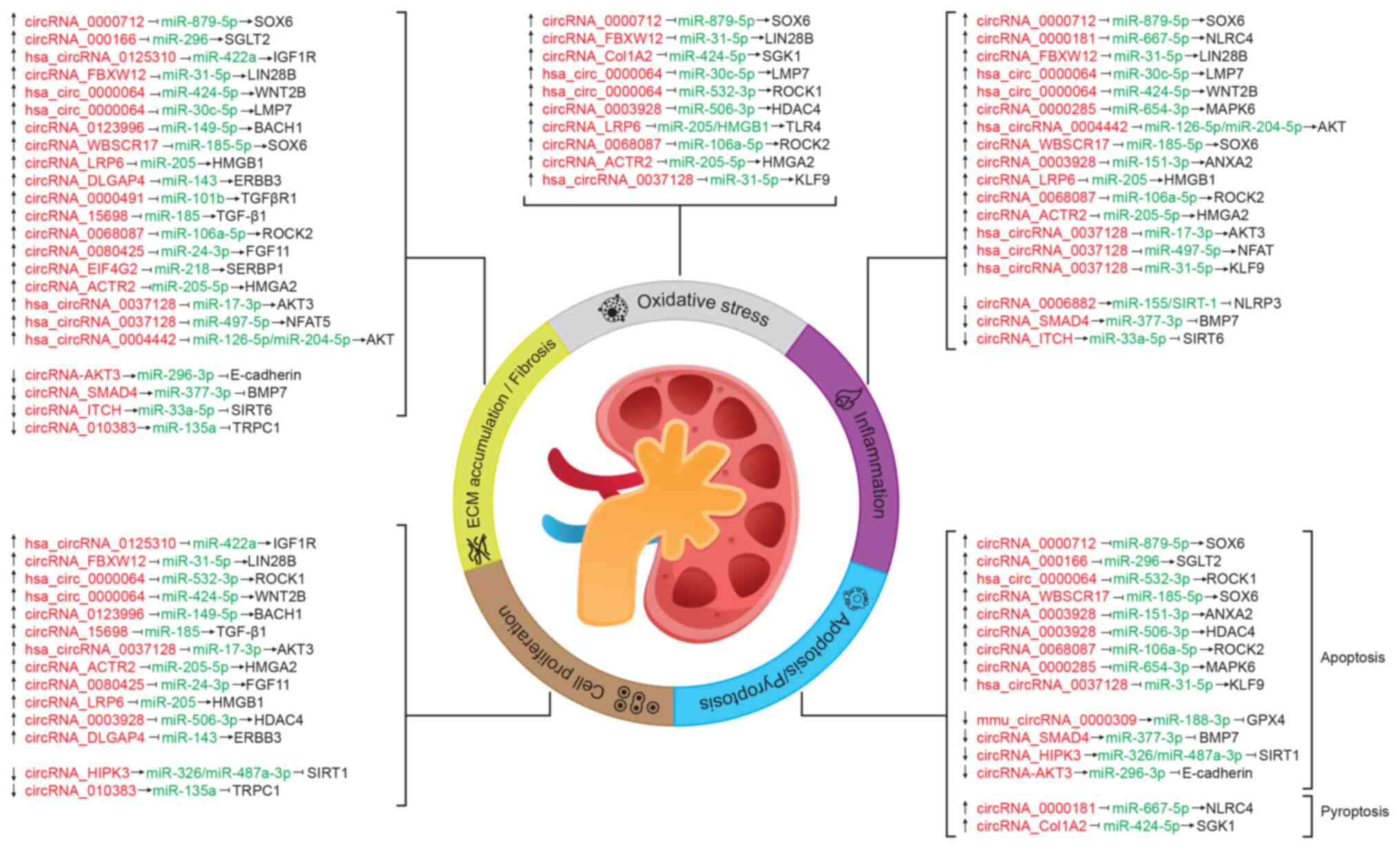
represented (Fig. 2).
Certain circRNAs (namely circRNA_ACTR2,
circRNA_LRP6, circRNA_0068087, circRNA_0003928 and circRNA_0000712)
simultaneously regulate more than one process, the most common
being: Inflammation, oxidative stress and ECM accumulation/fibrosis
(Fig. 2 and Table SI). The axes regulated by such
circRNAs include as targets transcription factors
(circRNA_0000712/miR-879-5p/SOX6), transcriptional regulators
[circRNA_ACTR2/miR-205-5p/high mobility group protein HMGI-C
(HMGA2), circRNA_LRP6/miR-205/high mobility group protein B1,
circRNA_0003928/miR-506-3p/histone deacetylase 4] or protein
kinases (circRNA_0068087/miR-106a-5p/Rho-associated protein kinase
2), which modulate different signaling pathways. Notably,
hsa_circRNA_0037128 and hsa_circRNA_0000064 mediate oxidative
stress, inflammation, apoptosis, cell proliferation and ECM
accumulation/fibrosis processes associated with DN, probably
because they have more than one target, including transcription
factors and protein kinases that are involved in several cell
processes (Fig. 2 and Table SI).
Some targets [transcription factor SOX6, RAC-γ
serine/threonine-protein kinase (AKT3) and NAD-dependent protein
deacetylase sirtuin-1] are regulated by ≥1 circRNA/miRNA pair
(Fig. 2 and Table SI), and function in the cellular
response to inflammatory, metabolic and oxidative stressors, such
as glucose, which suggests that these molecules could be explored
as potential therapeutical targets.
Some of the circRNAs that modulate the cell
processes associated with DN (Fig.
2 and Table SI) regulate
the AGE-RAGE axis and thus may be involved in the
pathophysiological events of DN. For example, circRNA_ACTR2 is
upregulated in patients with DN, and in both the proximal tubular
cell line HK-2 and mesangial cells exposed to high-glucose
concentrations (69) (Fig. 2 and Table SI). CircRNA_ACTR2 acts as a
sponge for miR-205-5p, which targets HMGA2, a molecule related to
the AGE-RAGE pathway (69).
HMGA2 is upregulated by AGEs, whereas its knockdown reverses the
AGEs-induced epithelial-to-mesenchymal transition of tubular cells
associated with DN, and inhibits the high AGEs-induced generation
of ROS and the activation of p38 MAPK (112). Consequently, silencing of
circRNA_ACTR2 inhibits cell proliferation, inflammatory mediators,
ECM deposition and oxidative stress in mesangial cells exposed to a
high-glucose concentration (69).
CircRNA_0037128 targets miR-17-3p and is upregulated
in kidney tissue of patients with DN, in the mouse mesangial cell
line SV40-MES13 when exposed to high glucose levels and in a DN
mouse model (70) (Fig. 2 and Table SI). In this previous study, it
was demonstrated that the circRNA_0037128/miR17-3p interaction
modulated the expression of AKT3, a molecule of the AGE-RAGE axis
that promotes cell proliferation and fibrosis (70). The upregulation of
circRNA_0037128 can also increase the levels of the
pro-inflammatory cytokines TNF-α, IL-1β and IL-6, as well as those
of the proteins FN, type I collagen and TGF-β1 in HK-2 tubular
cells, while knocking down this circRNA suppresses such effects
(71,72). It has been proposed that
circRNA_0037128 may regulate these cytokines, and in turn modulate
inflammation and fibrosis, through the miR-497-5p/nuclear factor of
activated T cells 5 and the miR-31-5p/Krueppel-like factor 9 axes
(71,72) (Fig. 2 and Table SI). CircRNA_0037128 is an
example of the multiple interactions and regulatory nodes in
different cell types that circRNAs can establish. By contrast,
circRNA_AKT3 has been shown to be downregulated (Fig. 2 and Table SI), and its overexpression
inhibits mouse mesangial cell apoptosis and suppresses ECM
accumulation, thus having a protective role in DN via the
circRNA_AKT3/miR-296-3p/E-cadherin pathway (73). E-cadherin is a cell adhesion
molecule that promotes cell-cell interactions, and allows cohesion
between cells and tissue integrity (113). Previous evidence has shown that
the AGE-RAGE interaction signal induces tubular
epithelial-myofibroblast transdifferentiation, as determined by the
loss of the epithelial marker E-cadherin, directly through the dual
specificity mitogen-activated protein kinase kinase
1-Ras-extracellular signal-regulated kinase1/2-MAPK pathway
(114). Notably, downregulation
of circRNA_AKT3 by high-glucose concentrations and AGEs formation
may explain the epithelial-to-mesenchymal transition of tubular
cells observed in DN.
The actions of circRNA_15698, circRNA_0000491,
circRNA_DLGAP4, circRNA_EIF4G2, cirRNA_0000285, hsa_circRNA_0004442
and circRNA_LRP6 are also mediated by molecules of the AGE-RAGE
pathway (74-80) (Fig. 2 and Table SI). Most of the remaining
circRNAs listed in Table SI
regulate molecules involved in inflammation, apoptosis, oxidative
stress and fibrosis via other signaling pathway (29,81-104). CircRNA_ANKRD36,
hsa_circRNA_0001831 and hsa_ circRNA_0000867 have been shown to be
upregulated in blood samples of patients with DN; however, their
targets and mechanisms of action remain unknown (105,106) (Table SI). Although miRNAs targeted by
hsa_circRNA_0000146 and hsa_circRNA_0000072 have been identified in
patients with DN, the evidence only suggests that such circRNAs may
be diagnostic markers of the disease (107) (Table SI). Even though several circRNAs
have been found to regulate the cellular processes linked to DN,
the available data does not support their role in the progression
of DN.
It has been recognized that circRNAs may serve a
critical role in regulating cellular events by interacting with
RBPs (50,115), thus participating in the
progression of various diseases (116); however, the role of this type
of interaction in DN has not been well explored. A notable example
of this type of interaction is the role of circRNA_Amotl1 as an
enhancer of cardiomyocyte survival in neonatal human cardiac
tissue. Zeng et al (117) reported that circRNA_Amotl1 in
primary cardiomyocytes, epithelial and endothelial cells functions
as a scaffold of pyruvate dehydrogenase kinase isoform 1 and AKT1,
facilitating AKT1 phosphorylation and its nuclear translocation,
which reduces apoptosis and enhances cardiac repair. Another
example was reported by Stoll et al (118); this previous study demonstrated
that intronic circRNA_ci-Ins2/ci-INS binds to TAR DNA-binding
protein 43 kDa at the transcriptional level for optimal insulin
secretion. Notably, circRNA_ci-Ins2/ci-INS expression is
downregulated in pancreatic β-cells of rodent models of diabetes
and in patients with T2D (118).
Although it has been demonstrated by global
interaction assays that ~8.1% of proteins binding to nucleic acids
have a dual function (119)
(that is, binding to DNA and RNA), the interaction of
circRNA_ci-Ins2/ci-INS with TAR DNA-binding protein 43 kDa in T2D
reported by Stoll et al (118) is one of the few reports between
circRNAs and proteins with a dual function found in the literature.
On the other hand, in type 1 DM, circRNA_PPM1F modulates M1
macrophage activation and inflammation of pancreatic β-cells
through the circRNA_ PPM1F/ELAV-like protein 1/protein phosphatase
1F/nuclear factor NF-κB (NF-κB) axis (120).
To the best of our knowledge, only a single report
on the interaction of circRNA/protein in DN has been published to
date. CircRNA_HIPK3 targets several miRNAs in renal tubular and
mesangial cells in rodent models of DN (29,82) (Table SI), although it can also bind
proteins. CircRNA_HIPK3 binds FUS and facilitates the enrichment of
this protein on the ectodysplasin A2 receptor (EDA2R) promoter;
this leads to the upregulation of EDA2R expression and activation
of apoptotic signaling in podocytes, which contributes to DN
progression (23).
With the aim of exploring the role of direct
interactions between circRNAs/proteins in the pathophysiology of
DN, our previous study performed an in silico analysis of
the ability of proteins involved in the AGE-RAGE pathway to bind
circRNAs expressed in renal cells (https://www.uacm.edu.mx/Portals/0/adam/Content/sshLYDUxokSSYJT-rhWTqg/Text/Geceta_22.pdf,
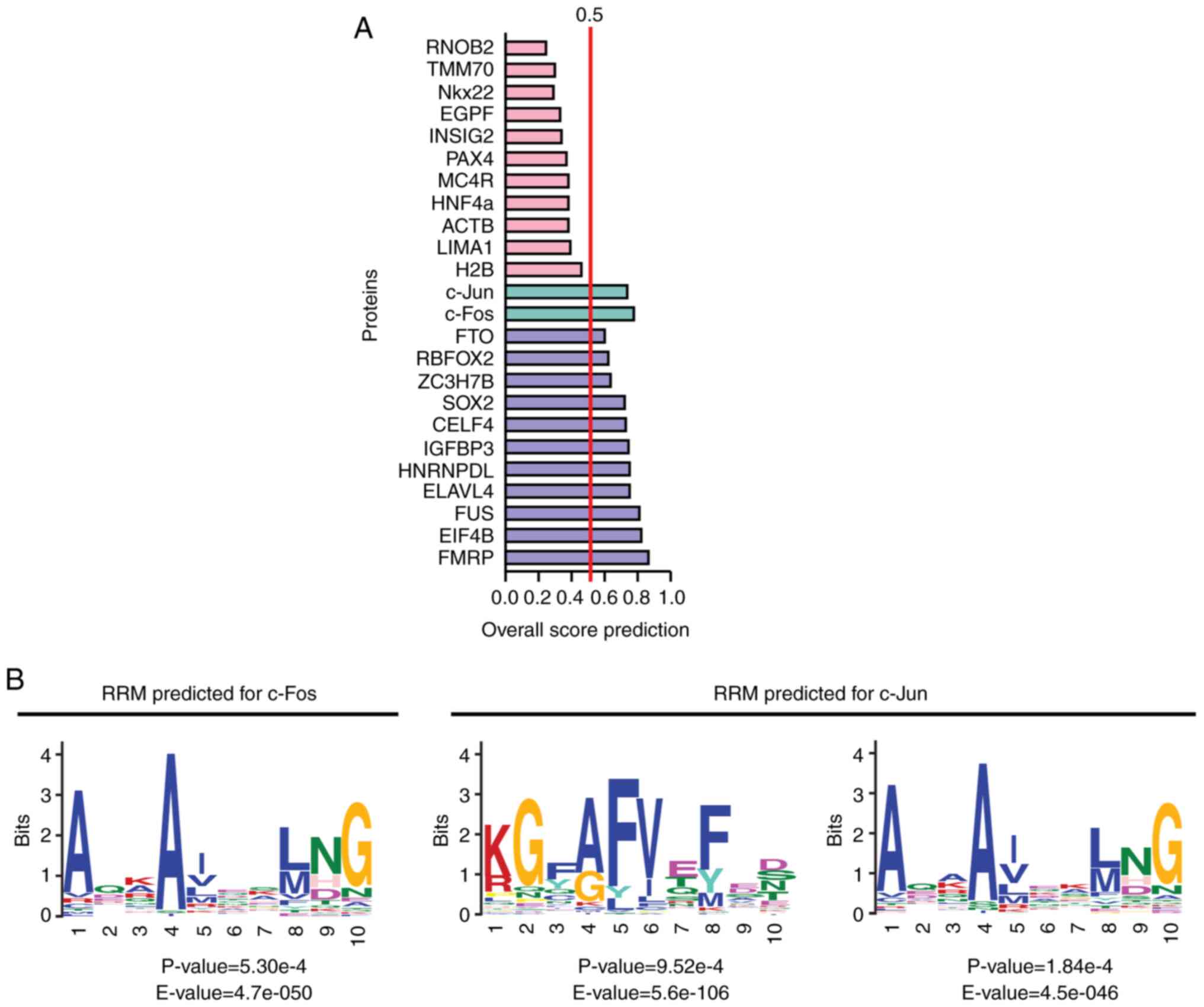
unpublished data). The results revealed that the transcription
factors c-Jun and c-Fos were potentially able to bind RNA according
to bioinformatics analysis conducted using the server catRAPID
signature (2020, RNA System Biology Italian Institute of
Technology; http://s.tartaglialab.com/page/catrapid_group), which
calculates the overall RNA-binding propensity of a protein and
predicts its RNA-binding regions (Fig. 3). The global interaction score
was 0.72 and 0.77 for c-Jun and c-Fos respectively, which was
>0.5 (the threshold recommended by the algorithm) (121) and similar to those scores of
known RBPs (Fig. 3A). This
finding suggested the presence of RNA-binding regions in the
transcription factors c-Jun and c-Fos. According to those
predictions, c-Fos would belong to the classical RNA-binding class,
while c-Jun would belong to the putative one (https://www.uacm.edu.mx/Portals/0/adam/Content/sshLYDUxokSSYJT-rhWTqg/Text/Geceta_22.pdf,
unpublished data).
Notably, c-Jun has two regions with RNA-binding
propensity: Amino acids 65-115 and 144-215. In these regions, two
potential RNA recognition motifs (RRMs) were identified by MEME
Suite (https://meme-suite.org/meme/tools/meme) and Tomtom
tool (https://meme-suite.org/meme/tools/tomtom) (122,123). For this analysis, 66 amino acid
sequences with the binding domains of 35 RBPs were used (Table SII). These RRMs were located
between amino acids 193-202 and 147-154, and were similar to the
domains SH3 LIG_G3BP_FGDF_1 and LIG_NBox_RRM1, respectively
(Fig. 3B) (https://www.uacm.edu.mx/Portals/0/adam/Content/sshLYDUxokSSYJT-rhWTqg/Text/Geceta_22.pdf,
unpublished data). By contrast, c-Fos harbors three regions with
RNA-binding propensity (amino acids 90-145, 200-257 and 264-353).
Only one RRM was identified in c-Fos at amino acids 217-226, which
was similar to the domain LIG_NBox_RRM1, and the one found in
c-Jun. This RRM was also present in ≥50% of the amino acid
sequences with the binding domains of RBPs used to analyze the
potential RNA-binding regions of c-Fos and c-Jun (Fig. 3B and Table SII) (https://www.uacm.edu.mx/Portals/0/adam/Content/sshLYDUxokSSYJT-rhWTqg/Text/Geceta_22.pdf,
unpublished data).
Our previous study also predicted (https://www.uacm.edu.mx/Portals/0/adam/Content/sshLYDUxokSSYJT-rhWTqg/Text/Geceta_22.pdf,
unpublished data) which circRNAs reported by Memczak et al
(12) that were expressed in
response to 25 mM glucose in 293 cells were able to interact with
c-Jun and/or c-Fos using the server RPI-seq (http://pridb.gdcb.iastate.edu/RPISeq/) (124). A total of 17 circRNAs that
potentially interacted with c-Jun and/or c-Fos were predicted, of
which 5 potentially interacted with c-Jun and c-Fos, 3 with c-Fos
and 9 with c-Jun, according to the results of classifiers support
vector machine and random forest, and simultaneous prediction score
with two classifiers ≥0.8 (Table
I). These circRNAs had potential binding motifs to RBP, some of
which are known motifs whereas others are unknown (https://www.uacm.edu.mx/Portals/0/adam/Content/sshLYDUxokSSYJT-rhWTqg/Text/Geceta_22.pdf,
unpublished data) according to the predictions of RPB suite
(http://www.csbio.sjtu.edu.cn/bioinf/RBPsuite/)
(125).
 | Table ICircRNAs expressed in the 25 mM
glucose-treated 293 cells that potentially interact with c-Jun
and/or c-Fos. |
Table I
CircRNAs expressed in the 25 mM
glucose-treated 293 cells that potentially interact with c-Jun
and/or c-Fos.
| Potential
interactions with proteins | CircBase ID |
|---|
| c-Fos and
c-Jun |
hsa_circ_0001181a |
|
hsa_circ_0001511 |
|
hsa_circ_0001666a |
|
hsa_circ_0001821a |
|
hsa_circ_0001461a |
| c-Fos |
hsa_circ_0001896 |
|
hsa_circ_0001294a |
|
hsa_circ_0000544a |
| c-Jun |
hsa_circ_0000211a |
|
hsa_circ_0000811 |
|
hsa_circ_0001632a |
|
hsa_circ_0001491a |
|
hsa_circ_0001784a |
|
hsa_circ_0000234a |
|
hsa_circ_0000527 |
|
hsa_circ_0000704a |
|
hsa_circ_0000745a |
These results require experimental demonstration
since i) 293 cells derived from human embryonic kidney cells, and
circRNA expression could be dependent on developmental stage; ii)
the increase in glucose concentration could lead to different
variations in circRNA expression in embryonic and adult cells
(19,22,26); and iii) the expression profile of
the predicted circRNAs in DN is not known. It should be noted that
the original kidney cell culture that served as source of 293 cells
was heterogeneous and such cells are not considered an in
vitro model of typical kidney cells (126). However, the majority of
predicted circRNAs that interact with c-Jun and c-Fos were
expressed in normal human kidney tissues (https://www.uacm.edu.mx/Portals/0/adam/Content/sshLYDUxokSSYJT-rhWTqg/Text/Geceta_22.pdf,
unpublished data) according to the results of CircAtlas 3.0
(https://ngdc.cncb.ac.cn/circatlas)
(Table I). Nevertheless, it is a
preliminary approach to identify potential interactions between
circRNAs and c-Jun or c-Fos.
It has been proposed that circRNAs can recruit
transcription factors to the promoters of target genes, and may
activate or inhibit their transcription (127). Thus, the interaction of c-Jun
and c-Fos with circRNAs could regulate the expression of genes
under the control of these transcription factors, as well as the
expression of circRNAs, their parental genes or their miRNA targets
through transcription factor sequestration via a negative feedback
mechanism. It has also been described that, during hyperglycemia,
the expression of c-Jun and c-Fos is increased, which was shown to
be mediated by the AGE-RAGE interaction, leading to the activation
of the JAK2 axis, and the production of collagen and other proteins
of the ECM (128).
Mesangial cells exposed to high-glucose
concentrations have also been reported to activate protein kinase
C, which in turn can modulate the transcription factor AP-1 (formed
by the association of c-Fos and c-Jun), thus increasing both the
mRNA and protein expression levels of c-Fos and c-Jun in the
nucleus. This increase in AP-1 is also correlated with an increased
production of ECM proteins (including FN, laminin and ColIV)
(128). Thus, it may be
hypothesized that circRNAs interacting with c-Jun and c-Fos could
exert a similar effect on the mRNA expression levels of ECM
components, whereby the potential interactions between circRNAs and
these transcription factors may explain some of the events
associated with the onset and progression of DN.
In addition, the aforementioned predictions revealed
in our previous studies (https://www.uacm.edu.mx/Portals/0/adam/Content/sshLYDUxokSSYJT-rhWTqg/Text/Geceta_22.pdf,
unpublished data) suggested that c-Jun and c-Fos may be DNA-binding
proteins and RBPs which must be validated in vitro or/and
in vivo, as well as the mechanisms by which these potential
interactions could be involved in cell processes associated with
DN.
6. CircRNAs as biomarkers of DN
The intrinsic characteristics of circRNAs, namely
stability and abundance, make them promising biomarkers for
diagnosing and evaluating DN progression or treatment efficacy.
Although circRNA expression in renal cells may provide insights
into the modulation of the pathophysiological process of DN, the
release of these circRNAs into the blood or urine would facilitate
their use as clinical biomarkers.
Upregulation of hsa_circRNA_0003928 and
downregulation of its target miR-151-3p have been observed in the
serum of patients with DN (86),
but its possible application as a diagnostic biomarker has not been
sufficiently explored. In addition, exosomal circRNA_DLGAP4
isolated from high glucose-treated mesangial cells, patients with
DN and DN rat models promotes diabetic kidney disease progression
by sponging miR-143 and targeting the receptor tyrosine-protein
kinase erbB-3/NF-κB/72 kDa type IV collagenase axis (76) (Fig. 2 and Table SI). A large number of the
circRNAs expressed in renal cells are carried by exosomes, which
are frequently involved in the pathophysiological processes
associated with DN (129). It
has been reported that high-glucose concentrations cause circRNA
differential expression in exosomes derived from human renal
tubular epithelial cells compared with controls (129,130).
Feng et al (130) reported that 7-10% of urinary
exosome transcripts correspond to circRNAs. Urinary exosomes mainly
arise from every epithelial cell of the nephron, while blood
exosomes are not able to pass through the glomerular membrane into
the urine (131). For that
reason, urinary exosomes may also be implicated in the
pathophysiological process of DN and could be a robust biomarker of
the disease. miRNAs enriched in urinary exosomes have been
associated with the progression of DN or with the early stages of
this disease (132,133). However, the diagnostic
potential of urinary exosomal circRNAs in DN has not yet been
determined. A recent study revealed that expression of
hsa_circRNA_0036649 in urinary exosomes is associated with renal
function and fibrosis degree in patients with chronic kidney
disease, which is a hallmark of DN (134). Other studies that have
supported the feasibility of the identification of circRNAs
enriched in urinary exosomes as biomarkers for kidney diseases have
focused on the dysregulation of these molecules in patients with
idiopathic membranous nephropathy or with immunoglobulin A
nephropathy compared with healthy controls (135,136). In summary, future studies
should be conducted to explore the use of serum and urine circRNAs
as biomarkers for the diagnosis and prognosis of DN.
7. Conclusions
Research related to the role of non-coding RNAs as
regulators of gene expression has increased in recent years.
Although some authors consider that circRNAs are modulators for the
initiation and development of DN, the role of these molecules in
the pathophysiological process of this disease and its progression
is still not fully understood. The complete profile of circRNAs
expressed in the kidney, blood, or urine and their interactions,
are unknown in DN. However, accumulated evidence has suggested that
circRNAs may participate in the regulation of DN-related cellular
processes.
CircRNAs are highly stable and abundant molecules,
which makes them important potential clinical biomarkers,
therapeutic targets, or novel diagnostic agents. However, numerous
questions regarding these RNA molecules and their roles in DN
remain to be addressed. Therefore, further studies are required to
reveal the function of circRNAs in the pathophysiological processes
of DN, which may also serve as the basis for developing new
diagnostic and therapeutic approaches for this disease.
Supplementary Data
Availability of data and materials
Not applicable.
Authors' contributions
LLC and MBMB conceptualized the article. EAL, YPN,
MBMB and LLC performed the literature search and analysis. YPN,
EAL, MBMB and LLC drafted the tables and figures. LLC wrote the
first draft of the manuscript. MBMB, EAL, ATC and JVF critically
revised and edited the paper. Data authentication is not
applicable. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Authors' information
Maximo B. Martinez Benitez, maximo.martinez@uacm.edu. mx;
ORCID 0000-0003-4351-0556. Yussel Perez Navarro, yussel.perez@estudiante.uacm.edu.mx.
Elisa Azuara-Liceaga, elisa.azuara@uacm.edu.mx;
ORCID 0000-0002-6392-170X. Angeles Tecalco Cruz, angeles.tecalco@uacm.edu.mx;
ORCID 0000-0001-9199-3834. Jesus Valdes Flores, jvaldes@cinvestav.mx; ORCID
0000-0003-1787-9229. Lilia Lopez-Canovas, lilia.lopez.canovas@uacm.edu.mx;
ORCID 0000-0003-0711-2569.
Acknowledgments
The authors would like to thank to Mr. Alfredo
Padilla Barberi (Postgraduate Program in Genomic Sciences, Science
and Technology School, Autonomous University of Mexico City for his
help in the composition of the figures.
Funding
This work was supported by the Science and Technology School,
Autonomous University of Mexico City [grant no. CCyT-2022-02].
References
|
1
|
American Diabetes Association: 2.
Classification and diagnosis of diabetes: Standards of medical care
in diabetes-2021. Diabetes Care. 44(Suppl 1): S15–S33. 2021.
View Article : Google Scholar
|
|
2
|
Sun H, Saeedi P, Karuranga S, Pinkepank M,
Ogurtsova K, Duncan BB, Stein C, Basit A, Chan JCN, Mbanya JC, et
al: IDF diabetes atlas: Global, regional and country-level diabetes
prevalence estimates for 2021 and projections for 2045. Diabetes
Res Clin Pract. 183:1091192022. View Article : Google Scholar
|
|
3
|
Harding JL, Pavkov ME, Magliano DJ, Shaw
JE and Gregg EW: Global trends in diabetes complications: A review
of current evidence. Diabetologia. 62:3–16. 2019. View Article : Google Scholar
|
|
4
|
Prasad RB and Groop L: Genetics of type 2
diabetes-pitfalls and possibilities. Genes (Basel). 6:87–123. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lyssenko V and Laakso M: Genetic screening
for the risk of type 2 diabetes: Worthless or valuable? Diabetes
Care. 36(Suppl 2): S120–S126. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Vassy JL and Meigs JB: Is Genetic testing
useful to predict type 2 diabetes? Best Pract Res Clin Endocrinol
Metab. 26:189–201. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Miranda-Lora AL, Vilchis-Gil J,
Juárez-Comboni DB, Cruz M and Klünder-Klünder M: A genetic risk
score improves the prediction of type 2 diabetes mellitus in
mexican youths but has lower predictive utility compared with
non-genetic factors. Front Endocrinol (Lausanne). 12:6478642021.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Willems SM, Mihaescu R, Sijbrands EJG, Van
Duijn CM and Janssens AC: A methodological perspective on genetic
risk prediction studies in type 2 diabetes: Recommendations for
future research. Curr Diab Rep. 11:511–518. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Fava S and Hattersley AT: The role of
genetic susceptibility in diabetic nephropathy: evidence from
family studies. Nephrol Dial Transplant. 17:1543–1546. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Szymañski M, Barciszewska MZ, Zywicki M
and Barciszewski J: Noncoding RNA transcripts. J Appl Genet.
44:1–19. 2003.PubMed/NCBI
|
|
11
|
Jeck WR, Sorrentino JA, Wang K, Slevin MK,
Burd CE, Liu J, Marzluff WF and Sharpless NE: Circular RNAs are
abundant, conserved, and associated with ALU repeats. RNA.
19:141–157. 2013. View Article : Google Scholar :
|
|
12
|
Memczak S, Jens M, Elefsinioti A, Torti F,
Krueger J, Rybak A, Maier L, Mackowiak SD, Gregersen LH, Munschauer
M, et al: Circular RNAs are a large class of animal RNAs with
regulatory potency. Nature. 495:333–338. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rybak-Wolf A, Stottmeister C, Glažar P,
Jens M, Pino N, Giusti S, Hanan M, Behm M, Bartok O, Ashwal-Fluss,
et al: Circular RNAs in the mammalian brain are highly abundant,
conserved, and dynamically expressed. Mol Cell. 58:870–885. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Guo JU, Agarwal V, Guo H and Bartel DP:
Expanded identification and characterization of mammalian circular
RNAs. Genome Biol. 15:4092014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Salzman J, Chen RE, Olsen MN, Wang PL and
Brown PO: Cell-Type specific features of circular RNA expression.
PLoS Genet. 9:e10037772013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang Y, Zhang XO, Chen T, Xiang JF, Yin
QF, Xing YH, Zhu S, Yang L and Chen LL: Circular intronic long
noncoding RNAs. Mol Cell. 51:792–806. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Song P, Yang F, Jin H and Wang X: The
regulation of protein translation and its implications for cancer.
Signal Transduct Target Ther. 6:682021. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liao W, Du J, Wang Z, Feng Q, Liao M, Liu
H, Yuan K and Zeng Y: The role and mechanism of noncoding RNAs in
regulation of metabolic reprogramming in hepatocellular carcinoma.
Int J Cancer. 151:337–347. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ebbesen KK, Hansen TB and Kjems J:
Insights into circular RNA biology. RNA Biol. 14:1035–1045. 2017.
View Article : Google Scholar :
|
|
20
|
Chen LL: The biogenesis and emerging roles
of circular RNAs. Nat Rev Mol Cell Biol. 17:205–211. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Qu S, Zhong Y, Shang R, Zhang X, Song W,
Kjems J and Li H: The emerging landscape of circular RNA in life
processes. RNA Biol. 14:992–999. 2017. View Article : Google Scholar :
|
|
22
|
Barrett SP and Salzman J: Circular RNAs:
Analysis, expression and potential functions. Development.
143:1838–1847. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Liu CX and Chen LL: Circular RNAs:
Characterization, cellular roles, and applications. Cell.
185:2016–2034. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chi T, Lin J, Wang M, Zhao Y, Liao Z and
Wei P: Non-Coding RNA as biomarkers for type 2 diabetes development
and clinical management. Front Endocrinol (Lausanne).
12:6300322021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang F and Zhang M: Circ_001209 aggravates
diabetic retinal vascular dysfunction through regulating
miR-15b-5p/COL12A1. J Transl Med. 19:2942021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Fan W, Pang H, Xie Z, Huang G and Zhou Z:
Circular RNAs in diabetes mellitus and its complications. Front
Endocrinol (Lausanne). 13:8856502022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Patil NS, Feng B, Su Z, Castellani CA and
Chakrabarti S: Circular RNA mediated gene regulation in chronic
diabetic complications. Sci Rep. 11:237662021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tu C, Wang L, Wei L and Jiang Z: The role
of circular RNA in diabetic nephropathy. Int J Med Sci. 19:916–923.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu R, Zhang M and Ge Y: Circular RNA
HIPK3 exacerbates diabetic nephropathy and promotes proliferation
by sponging miR-185. Gene. 765:1450652021. View Article : Google Scholar
|
|
30
|
van Zonneveld AJ, Kölling M, Bijkerk R and
Lorenzen JM: Circular RNAs in kidney disease and cancer. Nat Rev
Nephrol. 17:814–826. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lasda E and Parker R: Circular RNAs:
Diversity of form and function. RNA. 20:1829–1842. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Petkovic S and Müller S: RNA
circularization strategies in vivo and in vitro. Nucleic Acids Res.
43:2454–2465. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yamazaki T, Fujiwara N, Yukinaga H,
Ebisuya M, Shiki T, Kurihara T, Kioka N, Kambe T, Nagao M, Nishida
E and Masuda S: The Closely Related RNA helicases, UAP56 and URH49,
Preferentially Form Distinct mRNA Export Machineries and
Coordinately Regulate Mitotic Progression. Mol Biol Cell.
21:2953–2965. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Huang C, Liang D, Tatomer DC and Wilusz
JE: A length-dependent evolutionarily conserved pathway controls
nuclear export of circular RNAs. Genes Dev. 32:639–644. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ren L, Jiang Q, Mo L, Tan L, Dong Q, Meng
L, Yang N and Li G: Mechanisms of circular RNA degradation. Commun
Biol. 5:13552022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang C, Huang S, Zhuang H, Ruan S, Zhou
Z, Huang K, Ji F, Ma Z, Hou B and He X: YTHDF2 promotes the liver
cancer stem cell phenotype and cancer metastasis by regulating OCT4
expression via m6A RNA methylation. Oncogene. 39:4507–4518. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Chang W and Wang J: Exosomes and their
noncoding RNA cargo are emerging as new modulators for diabetes
mellitus. Cells. 8:8532019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hentze MW and Preiss T: Circular RNAs:
Splicing's enigma variations. EMBO J. 32:923–925. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hansen TB, Jensen TI, Clausen BH, Bramsen
JB, Finsen B, Damgaard CK and Kjems J: Natural RNA circles function
as efficient microRNA sponges. Nature. 495:384–388. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang Y, Liu J, Ma J, Sun T, Zhou Q, Wang
W, Wang G, Wu P, Wang H, Jiang L, et al: Exosomal circRNAs:
Biogenesis, effect and application in human diseases. Mol Cancer.
18:1162019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Chen XT, Li ZW, Zhao X, Li ML, Hou PF, Chu
SF, Zheng JN and Bai J: Role of Circular RNA in kidney-related
diseases. Front Pharmacol. 12:6158822021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wang H, Gao X, Yu S, Wang W, Liu G, Jiang
X and Sun D: Circular RNAs regulate parental gene expression: A new
direction for molecular oncology research. Front Oncol.
12:9477752022. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wu N, Yuan Z, Du KY, Fang L, Lyu J, Zhang
C, He A, Eshaghi E, Zeng K, Ma J, et al: Translation of
yes-associated protein (YAP) was antagonized by its circular RNA
via suppressing the assembly of the translation initiation
machinery. Cell Death Differ. 26:2758–2773. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Legnini I, Di Timoteo G, Rossi F, Morlando
M, Briganti F, Sthandier O, Fatica A, Santini T, Andronache A, Wade
M, et al: Circ-ZNF609 Is a Circular RNA that Can Be translated and
functions in myogenesis. Mol Cell. 66:22–37.e9. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhang Z, Yang T and Xiao J: Circular RNAs:
Promising biomarkers for human diseases. EBioMedicine. 34:267–274.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ashwal-Fluss R, Meyer M, Pamudurti NR,
Ivanov A, Bartok O, Hanan M, Evantal N, Memczak S, Rajewsky N and
Kadener S: CircRNA Biogenesis competes with Pre-mRNA splicing. Mol
Cell. 56:55–66. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Du WW, Yang W, Liu E, Yang Z, Dhaliwal P
and Yang BB: Foxo3 circular RNA retards cell cycle progression via
forming ternary complexes with p21 and CDK2. Nucleic Acids Res.
44:2846–2858. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Schneider T, Hung LH, Schreiner S, Starke
S, Eckhof H, Rossbach O, Reich S, Medenbach J and Bindereif A:
CircRNA-protein complexes: IMP3 protein component defines subfamily
of circRNPs. Sci Rep. 6:313132016. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Holdt LM, Stahringer A, Sass K, Pichler G,
Kulak NA, Wilfert W, Kohlmaier A, Herbst A, Northoff BH, Nicolaou
A, et al: Circular non-coding RNA ANRIL modulates ribosomal RNA
maturation and atherosclerosis in humans. Nat Commun. 7:124292016.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Das A, Sinha T, Shyamal S and Panda AC:
Emerging role of circular RNA-protein interactions. Noncoding RNA.
7:482021.PubMed/NCBI
|
|
51
|
Castello A, Fischer B, Eichelbaum K, Horos
R, Beckmann BM, Strein C, Davey NE, Humphreys DT, Preiss T,
Steinmetz LM, et al: Insights into RNA Biology from an Atlas of
Mammalian mRNA-Binding Proteins. Cell. 149:1393–1406. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Yar Saglam SA, Alp E and Ilke Onen H:
Circular RNAs and its biological functions in health and disease.
Gene Expression and Phenotypic Traits. Chen YC and Chen SJ:
IntechOpen; pp. 1–37. 2020
|
|
53
|
Yang Q, Li F, He AT and Yang BB: Circular
RNAs: Expression, localization, and therapeutic potentials. Mol
Ther. 29:1683–1702. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Wang Y, Lu T, Wang Q, Liu J and Jiao W:
Circular RNAs: Crucial regulators in the human body (Review). Oncol
Rep. 40:3119–3135. 2018.PubMed/NCBI
|
|
55
|
Wang M, Yu F, Wu W, Zhang Y, Chang W,
Ponnusamy M, Wang K and Li P: Circular RNAs: A novel type of
non-coding RNA and their potential implications in antiviral
immunity. Int J Biol Sci. 13:1497–1506. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Yang L, Fu J and Zhou Y: Circular RNAs and
their emerging roles in immune regulation. Front Immunol.
9:29772018. View Article : Google Scholar
|
|
57
|
Shao T, Pan YH and Xiong XD: Circular RNA:
an important player with multiple facets to regulate its parental
gene expression. Mol Ther Nucleic Acids. 23:369–376. 2020.
View Article : Google Scholar
|
|
58
|
Chen N, Zhao G, Yan X, Lv Z, Yin H, Zhang
S, Song W, Li X, Li L, Du Z, et al: A novel FLI1 exonic circular
RNA promotes metastasis in breast cancer by coordinately regulating
TET1 and DNMT1. Genome Biol. 19:2182018. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Liu Y, Song J, Liu Y, Zhou Z and Wang X:
Transcription activation of circ-STAT3 induced by Gli2 promotes the
progression of hepatoblastoma via acting as a sponge for
miR-29a/b/c-3p to upregulate STAT3/Gli2. J Exp Clin Cancer Res.
39:1012020. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Okholm TLH, Nielsen MM, Hamilton MP,
Christensen LL, Vang S, Hedegaard J, Hansen TB, Kjems J, Dyrskjøt L
and Pedersen JS: Circular RNA expression is abundant and correlated
to aggressiveness in early-stage bladder cancer. NPJ Genom Med.
2:362017. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Selby NM and Taal MW: An updated overview
of diabetic nephropathy: Diagnosis, prognosis, treatment goals and
latest guidelines. Diabetes Obes Metab. 22(Suppl 1): S3–S15. 2020.
View Article : Google Scholar
|
|
62
|
Gheith O, Farouk N, Nampoory N, Halim MA
and Al-Otaibi T: Diabetic kidney disease: Worldwide difference of
prevalence and risk factors. J Nephropharmacol. 5:49–56.
2015.eCollection 2016.
|
|
63
|
Brosius FC, Khoury CC, Buller CL and Chen
S: Abnormalities in signaling pathways in diabetic nephropathy.
Expert Rev Endocrinol Metab. 5:51–64. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Cooper ME: Interaction of metabolic and
haemodynamic factors in mediating experimental diabetic
nephropathy. Diabetologia. 44:1957–1972. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Makita Z, Radoff S, Rayfield EJ, Yang Z,
Skolnik E, Delaney V, Friedman EA, Cerami A and Vlassara H:
Advanced glycosylation end products in patients with diabetic
nephropathy. N Engl J Med. 325:836–842. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Busch M, Franke S, Rüster C and Wolf G:
Advanced glycation end-products and the kidney. Eur J Clin Invest.
40:742–755. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ramasamy R, Yan SF and Schmidt AM:
Receptor for AGE (RAGE): Signaling mechanisms in the pathogenesis
of diabetes and its complications. Ann N Y Acad Sci. 1243:88–102.
2011. View Article : Google Scholar
|
|
68
|
Kay AM, Simpson CL and Stewart JA Jr: The
role of AGE/RAGE signaling in diabetes-mediated vascular
calcification. J Diabetes Res. 2016:68097032016. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Yun J, Ren J, Liu Y, Dai L, Song L, Ma X,
Luo S and Song Y: Circ-ACTR2 aggravates the high glucose-induced
cell dysfunction of human renal mesangial cells through mediating
the miR-205-5p/HMGA2 axis in diabetic nephropathy. Diabetol Metab
Syndr. 13:722021. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Wang Q, Cang Z, Shen L, Peng W, Xi L,
Jiang X, Ge X, Xu B and Huang S: circ_0037128/miR-17-3p/AKT3 axis
promotes the development of diabetic nephropathy. Gene.
765:1450762021. View Article : Google Scholar
|
|
71
|
Feng T, Li W, Li T, Jiao W and Chen S:
Circular RNA_0037128 aggravates high glucose-induced damage in HK-2
cells via regulation of microRNA-497-5p/nuclear factor of activated
T cells 5 axis. Bioengineered. 12:10959–10970. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Fang R, Cao X, Zhu Y and Chen Q:
Hsa_circ_0037128 aggravates high glucose-induced podocytes injury
in diabetic nephropathy through mediating miR-31-5p/KLF9.
Autoimmunity. 55:254–263. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Tang B, Li W, Ji TT, Li XY, Qu X, Feng L
and Bai S: Circ-AKT3 inhibits the accumulation of extracellular
matrix of mesangial cells in diabetic nephropathy via modulating
miR-296-3p/E-cadherin signals. J Cell Mol Med. 24:8779–8788. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Hu W, Han Q, Zhao L and Wang L: Circular
RNA circRNA_15698 aggravates the extracellular matrix of diabetic
nephropathy mesangial cells via miR-185/TGF-β1. J Cell Physiol.
234:1469–1476. 2019. View Article : Google Scholar
|
|
75
|
Mou X, Chen JW, Zhou DY, Liu K, Chen LJ,
Zhou D and Hu YB: A novel identified circular RNA, circ-0000491,
aggravates the extracellular matrix of diabetic nephropathy
glomerular mesangial cells through suppressing miR-101b by
targeting TGFβRI. Mol Med Rep. 22:3785–3794. 2020.PubMed/NCBI
|
|
76
|
Bai S, Xiong X, Tang B, Ji T, Li X, Qu X
and Li W: Exosomal circ_DLGAP4 promotes diabetic kidney disease
progression by sponging miR-143 and targeting ERBB3/NF-κB/MMP-2
axis. Cell Death Dis. 11:10082020. View Article : Google Scholar
|
|
77
|
Xu B, Wang Q, Li W, Xia L, Ge X, Shen L,
Cang Z, Peng W, Shao K and Huang S: Circular RNA circEIF4G2
aggravates renal fibrosis in diabetic nephropathy by sponging
miR-218. J Cell Mol Med. 26:1799–1805. 2022. View Article : Google Scholar
|
|
78
|
Yao T, Zha D, Hu C and Wu X: Circ_0000285
promotes podocyte injury through sponging miR-654-3p and activating
MAPK6 in diabetic nephropathy. Gene. 747:1446612020. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Qiu B, Qi X and Wang J: CircTLK1
downregulation attenuates high glucose-induced human mesangial cell
injury by blocking the AKT/NF-κB pathway through sponging
miR-126-5p/miR-204-5p. Biochem Genet. 60:1471–1487. 2022.
View Article : Google Scholar
|
|
80
|
Chen B, Li Y, Liu Y and Xu Z: circLRP6
regulates high glucose-induced proliferation, oxidative stress, ECM
accumulation, and inflammation in mesangial cells. J Cell Physiol.
234:21249–21259. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Feng F, Yang J, Wang G, Huang P, Li Y and
Zhou B: Circ_0068087 promotes high glucose-induced human renal
tubular cell injury through regulating miR-106a-5p/ROCK2 pathway.
Nephron. 147:212–222. 2023. View Article : Google Scholar
|
|
82
|
Zhuang L, Wang Z, Hu X, Yang Q, Pei X and
Jin G: CircHIPK3 alleviates high glucose toxicity to human renal
tubular epithelial HK-2 cells through regulation of
miR-326/miR-487a-3p/SIRT1. Diabetes Metab Syndr Obes. 14:729–740.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Liu H, Wang X, Wang ZY and Li L:
Circ_0080425 inhibits cell proliferation and fibrosis in diabetic
nephropathy via sponging miR-24-3p and targeting fibroblast growth
factor 11. J Cell Physiol. 235:4520–4529. 2020. View Article : Google Scholar
|
|
84
|
Wang W, Feng J, Zhou H and Li Q:
Circ_0123996 promotes cell proliferation and fibrosis in mouse
mesangial cells through sponging miR-149-5p and inducing Bach1
expression. Gene. 761:1449712020. View Article : Google Scholar
|
|
85
|
Li G, Qin Y, Qin S, Zhou X, Zhao W and
Zhang D: Circ_WBSCR17 aggravates inflammatory responses and
fibrosis by targeting miR-185-5p/SOX6 regulatory axis in high
glucose-induced human kidney tubular cells. Life Sci.
259:1182692020. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
An L, Ji D, Hu W, Wang J, Jin X, Qu Y and
Zhang N: Interference of hsa_circ_0003928 alleviates high
glucose-induced cell apoptosis and inflammation in HK-2 cells via
mir-151-3p/anxa2. Diabetes Metab Syndr Obes. 13:3157–3168. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Liu Q, Cui Y, Ding N and Zhou C: Knockdown
of circ_0003928 ameliorates high glucose-induced dysfunction of
human tubular epithelial cells through the miR-506-3p/HDAC4 pathway
in diabetic nephropathy. Eur J Med Res. 27:552022. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Ge X, Xi L, Wang Q, Li H, Xia L, Cang Z,
Peng W and Huang S: Circular RNA Circ_0000064 promotes the
proliferation and fibrosis of mesangial cells via miR-143 in
diabetic nephropathy. Gene. 758:1449522020. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Sun L, Han Y, Shen C, Luo H and Wang Z:
Emodin alleviates high glucose-induced oxidative stress,
inflammation and extracellular matrix accumulation of mesangial
cells by the circ_0000064/miR-30c-5p/Lmp7 axis. J Recept Signal
Transduct Res. 42:302–312. 2022. View Article : Google Scholar
|
|
90
|
Wang H, Huang S, Hu T, Fei S and Zhang H:
Circ_0000064 promotes high glucose-induced renal tubular epithelial
cells injury to facilitate diabetic nephropathy progression through
miR-532-3p/ROCK1 axis. BMC Endocr Disord. 22:672022. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Li J, Min Y and Zhao Q: Circ_0000064
knockdown attenuates high glucose-induced proliferation,
inflammation and extracellular matrix deposition of mesangial cells
through miR-424-5p-mediated WNT2B inhibition in cell models of
diabetic nephropathy. Clin Exp Nephrol. 26:943–954. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Peng F, Gong W, Li S, Yin B, Zhao C, Liu
W, Chen X, Luo C, Huang Q, Chen T, et al: circRNA_010383 Acts as a
Sponge for miR-135a, and its downregulated expression contributes
to renal fibrosis in diabetic nephropathy. Diabetes. 70:603–615.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Wang Y, Qi Y, Ji T, Tang B, Li X, Zheng P
and Bai S: Circ_LARP4 regulates high glucose-induced cell
proliferation, apoptosis, and fibrosis in mouse mesangial cells.
Gene. 765:1451142021. View Article : Google Scholar
|
|
94
|
Sun A, Sun N, Liang X and Hou Z:
Circ-FBXW12 aggravates the development of diabetic nephropathy by
binding to miR-31-5p to induce LIN28B. Diabetol Metab Syndr.
13:1412021. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Wu R, Niu Z, Ren G, Ruan L and Sun L:
CircSMAD4 alleviates high glucose-induced inflammation,
extracellular matrix deposition and apoptosis in mouse glomerulus
mesangial cells by relieving miR-377-3p-mediated BMP7 inhibition.
Diabetol Metab Syndr. 13:1372021. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Liu J, Duan P, Xu C, Xu D, Liu Y and Jiang
J: CircRNA circ-ITCH improves renal inflammation and fibrosis in
streptozotocin-induced diabetic mice by regulating the
miR-33a-5p/SIRT6 axis. Inflamm Res. 70:835–846. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Zhao L, Chen H, Zeng Y, Yang K, Zhang R,
Li Z, Yang T and Ruan H: Circular RNA circ_0000712 regulates high
glucose-induced apoptosis, inflammation, oxidative stress, and
fibrosis in (DN) by targeting the miR-879-5p/SOX6 axis. Endocr J.
68:1155–1164. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Zhu Y, Zha F, Tang B, Ji TT, Li XY, Feng L
and Bai SJ: Exosomal hsa_circ_0125310 promotes cell proliferation
and fibrosis in diabetic nephropathy via sponging miR-422a and
targeting the IGF1R/p38 axis. J Cell Mol Med. 26:151–162. 2022.
View Article : Google Scholar
|
|
99
|
Jin J, Wang Y, Zheng D, Liang M and He Q:
A Novel Identified Circular RNA, mmu_mmu_circRNA_0000309, Involves
in Germacrone-Mediated Improvement of Diabetic Nephropathy Through
Regulating Ferroptosis by Targeting miR-188-3p/GPX4 Signaling Axis.
Antioxid Redox Signal. 36:740–759. 2022. View Article : Google Scholar
|
|
100
|
Chen S: Circ_000166/miR-296 aggravates the
process of diabetic renal fibrosis by regulating the SGLT2
signaling pathway in renal tubular epithelial cells. Dis Markers.
2022:61030862022.PubMed/NCBI
|
|
101
|
Wang D, Zhang Z, Si Z and Wang L: Circ
0006282/miR-155 reduced inflammation in diabetic nephropathy via
expression of SIRT1/NLRP3 signaling pathway. Food Sci Technol
(Campinas). 42:e395202022. View Article : Google Scholar
|
|
102
|
Li Y, Yu W, Xiong H and Yuan F:
Circ_0000181 regulates miR-667-5p/NLRC4 axis to promote pyroptosis
progression in diabetic nephropathy. Sci Rep. 12:119942022.
View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Zhuang L, Jin G, Qiong W, Ge X and Pei X:
Circular RNA COL1A2 mediates high glucose-induced oxidative stress
and pyroptosis by regulating MiR-424-5p/SGK1 in diabetic
nephropathy. Appl Biochem Biotechnol. 195:7652–7667. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Liu X and Wu Y: Circ_0000953 deficiency
exacerbates podocyte injury and autophage through targeting
mir-655/atg4b in diabetic nephropathy. Kidney Int Rep. 8:S198–S199.
2023. View Article : Google Scholar
|
|
105
|
Rashad NM, Sherif MH, El-Shal AS and
Abdelsamad MAE: The expression profile of circANKRD36 and ANKRD36
as diagnostic biomarkers of chronic kidney disease in patients with
type 2 diabetes mellitus. Egypt J Med Hum Genet. 22:432021.
View Article : Google Scholar
|
|
106
|
Zhang K, Wan X, Khan MA, Sun X, Yi X, Wang
Z, Chen K and Peng L: Peripheral Blood circRNA microarray profiling
identities hsa_circ_0001831 and hsa_circ_0000867 as two novel
circrna biomarkers for early type 2 diabetic nephropathy. Diabetes
Metab Syndr Obes. 15:2789–2801. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Badr AM, Elkholy O, Said M, Fahim SA,
El-Khatib M, Sabry D and Gaber RM: Diagnostic Significance of
hsa_circ_0000146 and hsa_circ_0000072 biomarkers for diabetic
kidney disease in patients with type 2 diabetes mellitus. J Med
Biochem. 42:239–248. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Ling L, Tan Z, Zhang C, Gui S, Cui Y, Hu Y
and Chen L: CircRNAs in exosomes from high glucose-treated
glomerular endothelial cells activate mesangial cells. Am J Transl
Res. 11:4667–4682. 2019.PubMed/NCBI
|
|
109
|
Liu M and Zhao J: Circular RNAs in
diabetic nephropathy: Updates and perspectives. Aging Dis.
13:1365–1380. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Loganathan TS, Sulaiman SA, Abdul Murad
NA, Shah SA, Abdul Gafor AH, Jamal R and Abdullah N: Interactions
Among Non-Coding RNAs in Diabetic Nephropathy. Front Pharmacol.
11:1912020. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Xiong X, Liu C, Shen M, Yang Q, Zhao Q, Li
X, Zhong X and Wang Z: Circular RNA expression profile in
transgenic diabetic mouse kidneys. Cell Mol Biol Lett. 26:252021.
View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Bai YH, Wang JP, Yang M, Zeng Y and Jiang
HY: SiRNA-HMGA2 weakened AGEs-induced epithelial-to-mesenchymal
transition in tubular epithelial cells. Biochem Biophys Res Commun.
457:730–735. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Birchmeier W and Behrens J: Cadherin
expression in carcinomas: Role in the formation of cell junctions
and the prevention of invasiveness. Biochim Biophys Acta.
1198:11–26. 1994.PubMed/NCBI
|
|
114
|
Li JH, Wang W, Huang XR, Oldfield M,
Schmidt AM, Cooper ME and Lan HY: Advanced glycation end products
induce tubular epithelial-myofibroblast transition through the
RAGE-ERK1/2 MAP kinase signaling pathway. Am J Pathol.
164:1389–1397. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Guria A, Sharma P, Natesan S and Pandi G:
Circular RNAs-The road less traveled. Front Mol Biosci. 6:1462020.
View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Ikeda Y, Morikawa S, Nakashima M,
Yoshikawa S, Taniguchi K, Sawamura H, Suga N, Tsuji A and Matsuda
S: CircRNAs and RNA-Binding proteins involved in the pathogenesis
of cancers or central nervous system disorders. Noncoding RNA.
9:232023.PubMed/NCBI
|
|
117
|
Zeng Y, Du WW, Wu Y, Yang Z, Awan FM, Li
X, Yang W, Zhang C, Yang Q, Yee A, et al: A circular RNA binds to
and activates AKT phosphorylation and nuclear localization reducing
apoptosis and enhancing cardiac repair. Theranostics. 7:3842–3855.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Stoll L, Rodríguez-Trejo A, Guay C, Brozzi
F, Bayazit MB, Gattesco S, Menoud V, Sobel J, Marques AC, Venø MT,
et al: A circular RNA generated from an intron of the insulin gene
controls insulin secretion. Nat Commun. 11:56112020. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Hou L, Wei Y, Lin Y, Wang X, Lai Y, Yin M,
Chen Y, Guo X, Wu S, Zhu Y, et al: Concurrent binding to DNA and
RNA facilitates the pluripotency reprogramming activity of Sox2.
Nucleic Acids Res. 48:3869–3887. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Zhang C, Han X, Yang L, Fu J, Sun C, Huang
S, Xiao W, Gao Y, Liang Q, Wang X, et al: Circular RNA circPPM1F
modulates M1 macrophage activation and pancreatic islet
inflammation in type 1 diabetes mellitus. Theranostics.
10:10908–10924. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Livi CM, Klus P, Delli Ponti R and
Tartaglia GG: CatRAPID signature: Identification of
ribonucleoproteins and RNA-binding regions. Bioinformatics.
32:773–775. 2016. View Article : Google Scholar :
|
|
122
|
Bailey TL, Johnson J, Grant CE and Noble
WS: The MEME Suite. Nucleic Acids Res. 43(W1): W39–W49. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Gupta S, Stamatoyannopoulos JA, Bailey TL
and Noble WS: Quantifying similarity between motifs. Genome Biol.
8:R242007. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Muppirala UK, Honavar VG and Dobbs D:
Predicting RNA-Protein interactions using only sequence
information. BMC Bioinformatics. 12:4892011. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Pan X, Fang Y, Li X, Yang Y and Shen HB:
RBPsuite: RNA-protein binding sites prediction suite based on deep
learning. BMC Genomics. 21:8842020. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Lin YC, Boone M, Meuris L, Lemmens I, Van
Roy N, Soete A, Reumers J, Moisse M, Plaisance S, Drmanac R, et al:
Genome dynamics of the human embryonic kidney 293 lineage in
response to cell biology manipulations. Nat Commun. 5:47672014.
View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Zhou WY, Cai ZR, Liu J, Wang DS, Ju HQ and
Xu RH: Circular RNA: Metabolism, functions and interactions with
proteins. Mol Cancer. 19:1722020. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Kreisberg JI, Radnik RA, Ayo SH, Garoni J
and Saikumar P: High glucose elevates c-fos and c-jun transcripts
and proteins in mesangial cell cultures. Kidney Int. 46:105–112.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Xu YX, Pu SD, Li X, Yu ZW, Zhang YT, Tong
XW, Shan YY and Gao XY: Exosomal ncRNAs: Novel therapeutic target
and biomarker for diabetic complications. Pharmacol Res.
178:1061352022. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Feng S, LV L, Liu B, Zhu X and Jing J:
MO619: Landscape RNA Profiling of Urinary Extracellular Vesicles in
Patients with Diabetic Nephropathy. Nephrology Dialysis
Transplantation. 37:2022. View Article : Google Scholar
|
|
131
|
Sinha N, Kumar V, Puri V, Nada R, Rastogi
A, Jha V and Puri S: Urinary exosomes: Potential biomarkers for
diabetic nephropathy. Nephrology (Carlton). 25:881–887. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Xie Y, Jia Y, Cuihua X, Hu F, Xue M and
Xue Y: Urinary exosomal MicroRNA profiling in incipient type 2
diabetic kidney disease. J Diabetes Res. 2017:69789842017.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Zhao Y, Shen A, Guo F, Song Y, Jing N,
Ding X, Pan M, Zhang H, Wang J, Wu L, et al: Urinary Exosomal
MiRNA-4534 as a novel diagnostic biomarker for diabetic kidney
disease. Front Endocrinol (Lausanne). 11:5902020. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Cao Y, Shi Y, Yang Y, Wu Z, Peng N, Xiao
J, Dou F, Xu J, Pei W, Fu C, et al: Urinary exosomes derived
circRNAs as biomarkers for chronic renal fibrosis. Ann Med.
54:1966–1976. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Ma H, Xu Y, Zhang R, Guo B, Zhang S and
Zhang X: Differential expression study of circular RNAs in exosomes
from serum and urine in patients with idiopathic membranous
nephropathy. Arch Med Sci. 15:738–753. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Luan R, Tian G, Ci X, Zheng Q, Wu L and Lu
X: Differential expression analysis of urinary exosomal circular
RNAs in patients with IgA nephropathy. Nephrology (Carlton).
26:432–441. 2021. View Article : Google Scholar : PubMed/NCBI
|