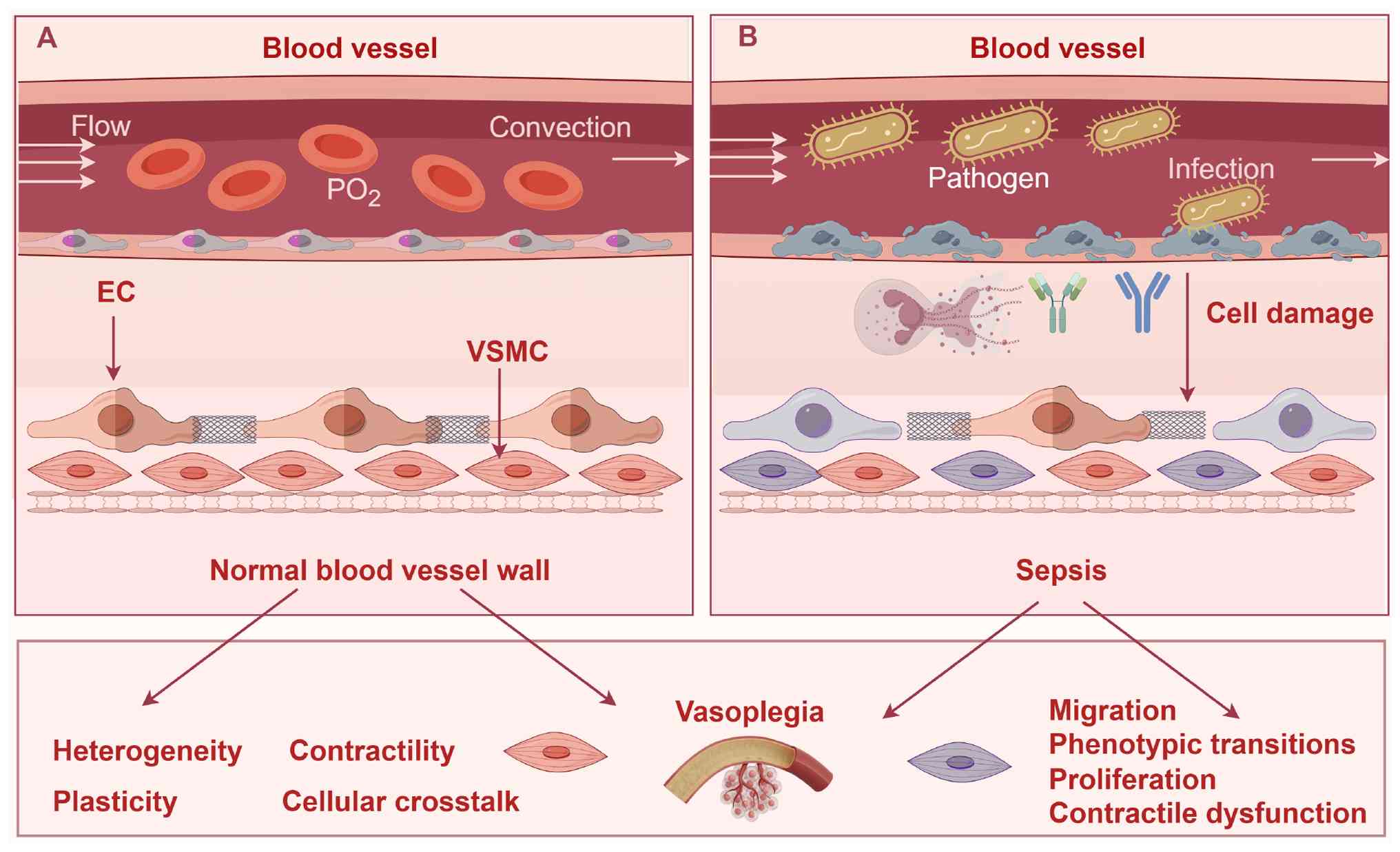
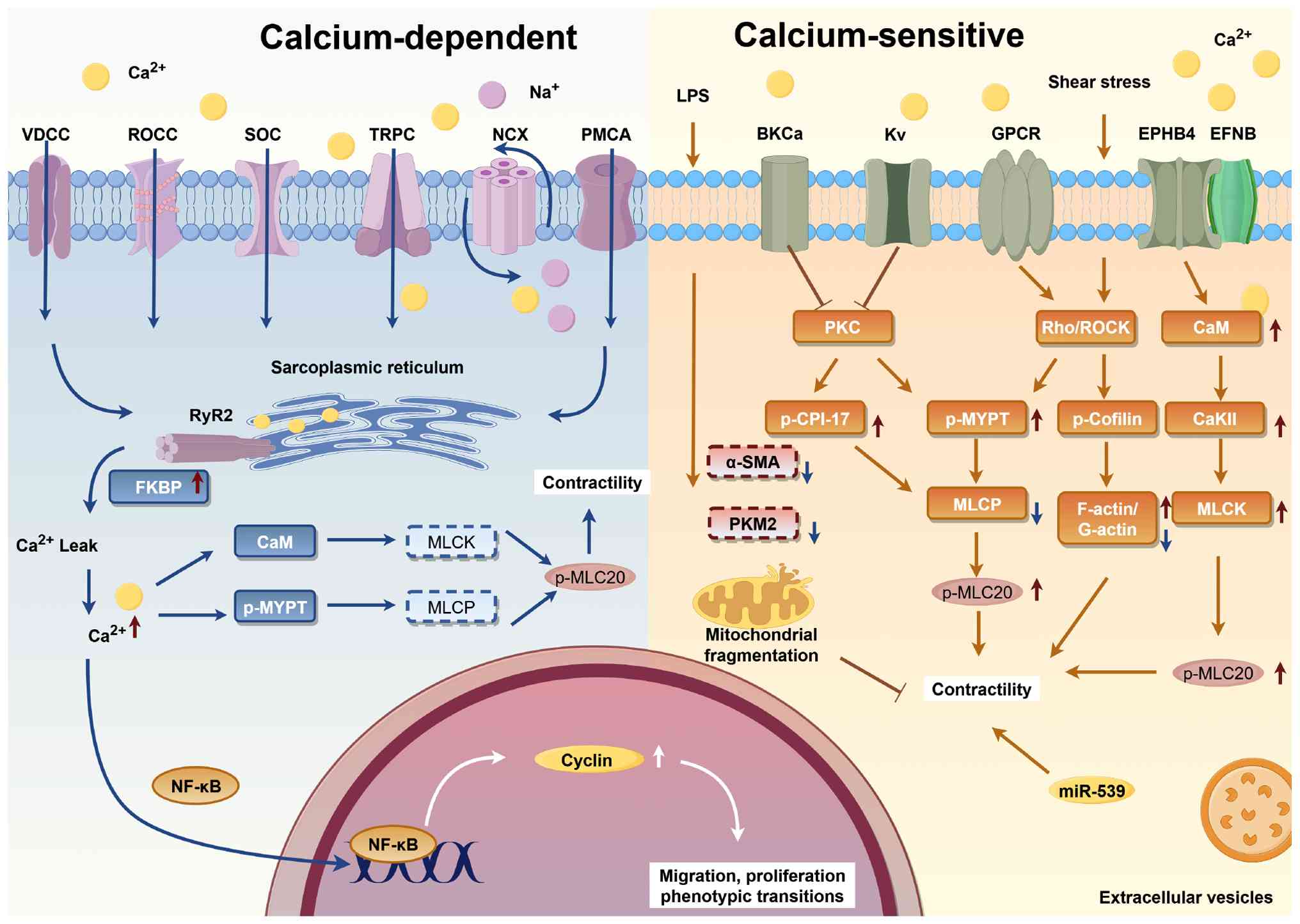
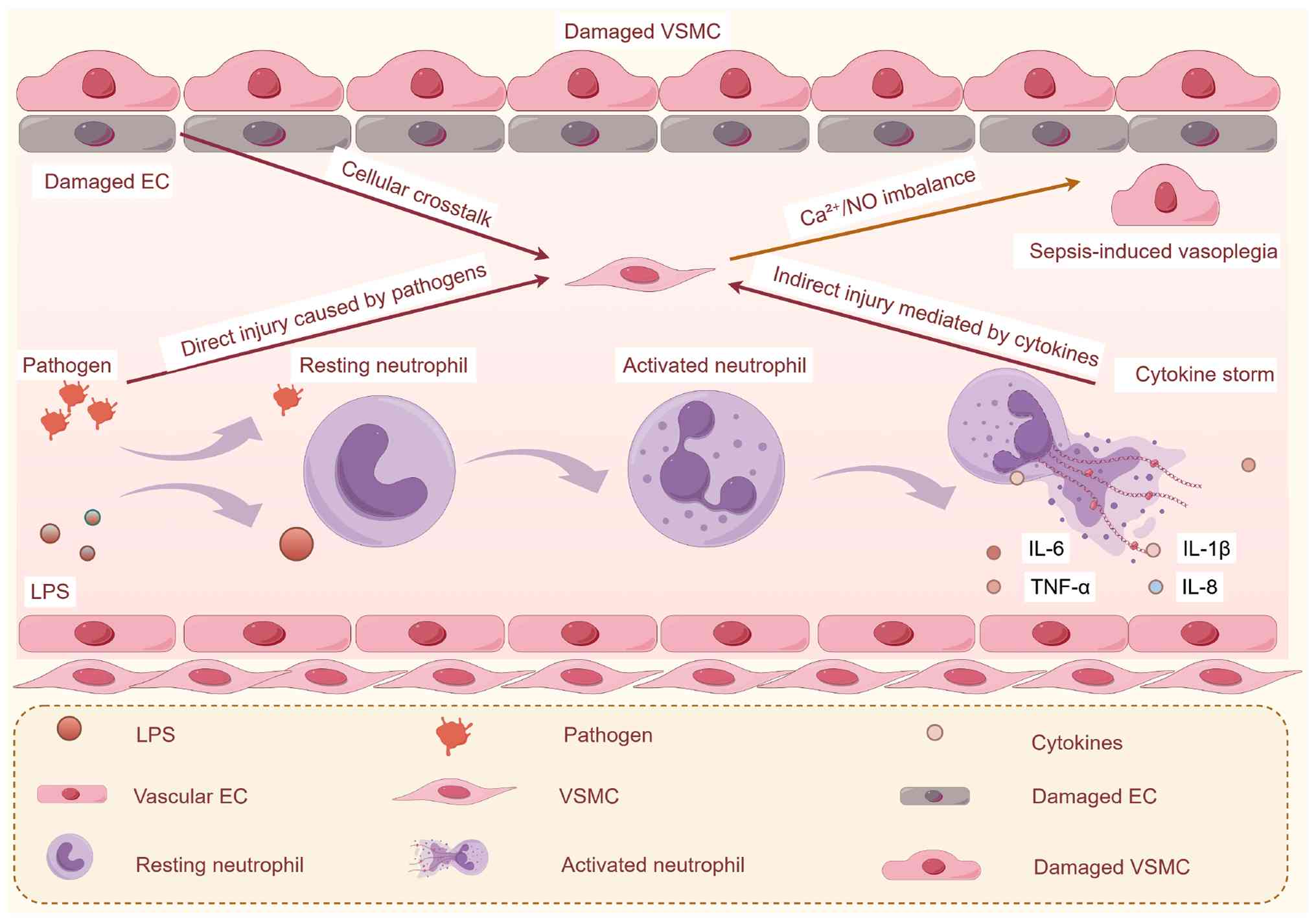
|
1
|
Lambden S, Creagh-Brown BC, Hunt J,
Summers C and Forni LG: Definitions and pathophysiology of
vasoplegic shock. Critical Care. 22:1742018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Liu Y, Bao D, She H, Zhang Z, Shao S, Wu
Z, Wu Y, Li Q, Wang L, Li T and Liu L: Role of Hippo/ACSL4 axis in
ferroptosis-induced pericyte loss and vascular dysfunction in
sepsis. Redox Biol. 78:1033532024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
GBD 2021 Global Sepsis Collaborators:
Global, regional, and national sepsis incidence and mortality,
1990-2021: A systematic analysis. Lancet Global Health.
13:e2013–e2026. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ran X, Zhang Q and Li S, Yu Z, Wan L, Wu
B, Wu R and Li S: Tissue Kallikrein exacerbating sepsis-induced
endothelial hyperpermeability is highly predictive of severity and
mortality in sepsis. J Inflamm Res. 14:3321–3333. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ruan H, Li YZ, Zhang Q, Wang BR, Wu R, Li
SS and Ran X: Identification and clinical validation of
hypoxia-inducible factor 1α protein as the potential biomarker in
patients with sepsis. Shock. 59:855–863. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ruan H, Zhang Q, Zhang YP, Li SS and Ran
X: Unraveling the role of HIF-1α in sepsis: From pathophysiology to
potential therapeutics-a narrative review. Crit Care. 28:1002024.
View Article : Google Scholar
|
|
7
|
Wurster SH, Wang P, Dean RE and Chaudry
IH: Vascular smooth muscle contractile function is impaired during
early and late stages of sepsis. J Surg Res. 56:556–561. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Terpe P, Ruhs S, Dubourg V, Bucher M and
Gekle M: The synergism of cytosolic acidosis and reduced NAD+/NADH
ratio is responsible for lactic acidosis-induced vascular smooth
muscle cell impairment in sepsis. J Biomed Sci. 31:32024.
View Article : Google Scholar
|
|
9
|
Liu R, Jia L, Yu L, Lai D, Li Q, Zhang B,
Guo E, Xu K and Luo Q: Interaction between post-tumor inflammation
and vascular smooth muscle cell dysfunction in sepsis-induced
cardiomyopathy. Front Immunol. 16:15607172025. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Donadon M and Santoro MM: The origin and
mechanisms of smooth muscle cell development in vertebrates.
Development. 148:dev1973842021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tinajero MG and Gotlieb AI: Recent
developments in vascular adventitial pathobiology: The dynamic
adventitia as a complex regulator of vascular disease. Am J Pathol.
190:520–534. 2020. View Article : Google Scholar
|
|
12
|
Méndez-Barbero N, Gutiérrez-Muñoz C and
Blanco-Colio LM: Cellular crosstalk between endothelial and smooth
muscle cells in vascular wall remodeling. Int J Mol Sci.
22:72842021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tang HY, Chen AQ, Zhang H, Gao XF, Kong XQ
and Zhang JJ: Vascular smooth muscle cells phenotypic switching in
cardiovascular diseases. Cells. 11:40602022. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Arroyo-Ataz G, Yagüe AC, Breda JC,
Mazzilli SA and Jones D: Single-cell transcriptomics and lineage
tracing unveil parallels in lymphatic muscle and venous smooth
muscle development, identity, and function. Arterioscler Thromb
Vasc Biol. 45:1207–1225. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sawada H, Rateri DL, Moorleghen JJ,
Majesky MW and Daugherty A: Smooth muscle cells derived from second
heart field and cardiac neural crest reside in spatially distinct
domains in the media of the ascending aorta-brief report.
Arterioscler Thromb Vasc Biol. 37:1722–1726. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mikawa T and Gourdie RG: Pericardial
mesoderm generates a population of coronary smooth muscle cells
migrating into the heart along with ingrowth of the epicardial
organ. Dev Biol. 174:221–232. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Khachigian LM, Black BL, Ferdinandy P, De
Caterina R, Madonna R and Geng YJ: Transcriptional regulation of
vascular smooth muscle cell proliferation, differentiation and
senescence: Novel targets for therapy. Vascular Pharmacol.
146:1070912022. View Article : Google Scholar
|
|
18
|
Martinsen A, Dessy C and Morel N:
Regulation of calcium channels in smooth muscle: New insights into
the role of myosin light chain kinase. Channels (Austin).
8:402–413. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ito M, Okamoto R, Ito H, Zhe Y and Dohi K:
Regulation of myosin light-chain phosphorylation and its roles in
cardiovascular physiology and pathophysiology. Hypertens Res.
45:40–52. 2022. View Article : Google Scholar
|
|
20
|
Phelps C, Chess-Williams R and Moro C: The
role of intracellular calcium and Rho kinase pathways in G
protein-coupled receptor-mediated contractions of urinary bladder
urothelium and lamina propria. Am J Physiol Cell Physiol.
324:C787–C797. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ran Q, Li A, Tan Y, Zhang Y, Zhang Y and
Chen H: Action and therapeutic targets of myosin light chain
kinase, an important cardiovascular signaling mechanism. Pharmacol
Res. 206:1072762024. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Rezaei M, Mehta JL, Zadeh GM, Khedri A and
Rezaei HB: Myosin light chain phosphatase is a downstream target of
Rho-kinase in endothelin-1-induced transactivation of the TGF-β
receptor. Cell Biochem Biophys. 82:1109–1120. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Weng W, Gu X, Yang Y, Zhang Q, Deng Q,
Zhou J, Cheng J, Zhu MX, Feng J, Huang O and Li Y: N-terminal
α-amino SUMOylation of cofilin-1 is critical for its regulation of
actin depolymerization. Nat Commun. 14:56882023. View Article : Google Scholar
|
|
24
|
Cao G, Xuan X, Hu J, Zhang R, Jin H and
Dong H: How vascular smooth muscle cell phenotype switching
contributes to vascular disease. Cell Commun Signal. 20:1802022.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sorokin V, Vickneson K, Kofidis T, Woo CC,
Lin XY, Foo R and Shanahan CM: Role of vascular smooth muscle cell
plasticity and interactions in vessel wall inflammation. Front
Immunol. 11:5994152020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhu Y, Zhang JL, Yan XJ, Ji Y and Wang FF:
Exploring a new mechanism between lactate and VSMC calcification:
PARP1/POLG/UCP2 signaling pathway and imbalance of mitochondrial
homeostasis. Cell Death Dis. 14:5982023. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tian Z, Ning H, Wang X, Wang Y, Han T and
Sun C: Endothelial autophagy promotes atheroprotective
communication between endothelial and smooth muscle cells via
Exosome-Mediated delivery of miR-204-5p. Arterioscler Thromb Vasc
Biol. 44:1813–1832. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Heiss EH, Schachner D, Donati M, Grojer CS
and Dirsch VM: Increased aerobic glycolysis is important for the
motility of activated VSMC and inhibited by indirubin-3'-monoxime.
Vascul Pharmacol. 83:47–56. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zein JG, Lee GL, Tawk M, Dabaja M and
Kinasewitz GT: Prognostic significance of elevated serum lactate
dehydrogenase (LDH) in patients with severe sepsis. Chest.
126(Suppl 1): 873S2004. View Article : Google Scholar
|
|
30
|
Yu H, Yang Q, Qian Y, Luo S, Kong T, Yang
G, Chen W, Xie F, Chen J, Xiong X, et al: A positive correlation
between serum lactate dehydrogenase level and in-hospital mortality
in ICU sepsis patients: Evidence from two large databases. Eur J
Med Res. 29:5252024. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chen ZH, Cao SH, Ren ZY, Zhang T, Jiang
HM, Hu ZK and Dong LH: Lactate dehydrogenase A crotonylation and
Mono-Ubiquitination maintains vascular smooth muscle cell growth
and migration and promotes neointima hyperplasia. J Am Heart Assoc.
14:e0363772025. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Meng F, Cheng H, Qian J, Dai X, Huang Y
and Fan Y: In vitro fluidic systems: Applying shear stress on
endothelial cells. Med Novel Technol Devices. 15:1001432022.
View Article : Google Scholar
|
|
33
|
Di Giantomasso D, May CN and Bellomo R:
Vital organ blood flow during hyperdynamic sepsis. Chest.
124:1053–1059. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
McMullan RR, McAuley DF, O'Kane CM and
Silversides JA: Vascular leak in sepsis: Physiological basis and
potential therapeutic advances. Crit Care. 28:972024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bateman RM, Sharpe MD, Singer M and Ellis
CG: The effect of sepsis on the erythrocyte. Int J Mol Sci.
18:19322017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang Y, Ouyang Y, Liu B, Ma X and Ding R:
Platelet activation and antiplatelet therapy in sepsis: A narrative
review. Thromb Res. 166:28–36. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Haga JH, Li YSJ and Chien S: Molecular
basis of the effects of mechanical stretch on vascular smooth
muscle cells. J Biomech. 40:947–960. 2007. View Article : Google Scholar
|
|
38
|
Sun W, Wang S, Chen Z, Zhang J, Li T,
Arias K, Griffith BP and Wu ZJ: Impact of high mechanical shear
stress and oxygenator membrane surface on blood damage relevant to
thrombosis and bleeding in a pediatric ECMO circuit. Artif Organs.
44:717–726. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lin C, Zheng X, Lin S, Zhang Y, Wu J and
Li Y: Mechanotransduction regulates the interplays between alveolar
epithelial and vascular endothelial cells in lung. Front Physiol.
13:8183942022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Shirazian S, Rios-Rojas L, Drakakis J,
Dikkala S, Dutka P, Duey M, Cho DJ and Fishbane S: The effect of
hemodialysis ultrafiltration on changes in whole blood viscosity.
Hemodial Int. 16:342–350. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Xie M, Li X, Chen L, Zhang Y, Chen L, Hua
H and Qi J: The crosstalks between vascular endothelial cells,
vascular smooth muscle cells, and adventitial fibroblasts in
vascular remodeling. Life Sci. 361:1233192025. View Article : Google Scholar
|
|
42
|
Hiroshima Y, Oyama Y, Sawasaki K, Nakamura
M, Kimura N, Kawahito K, Fujie H and Sakamoto N: A compressed
collagen construct for studying Endothelial-smooth muscle cell
interaction under high shear stress. Ann Biomed Eng. 50:951–963.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Sawasaki K, Nakamura M, Kimura N, Kawahito
K, Yamazaki M, Fujie H and Sakamoto N: Endothelial-derived nitric
oxide impacts vascular smooth muscle cell phenotypes under high
wall shear stress condition. Biochem Biophys Res Commun.
740:1510052024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Yu J, Zhang Y, Zhang X, Rudic RD, Bauer
PM, Altieri DC and Sessa WC: Endothelium derived nitric oxide
synthase negatively regulates the PDGF-survivin pathway during
flow-dependent vascular remodeling. PLoS One. 7:e314952012.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Sack KD, Teran M and Nugent MA:
Extracellular matrix stiffness controls VEGF signaling and
processing in endothelial cells. J Cell Physiol. 231:2026–2039.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ploppa A, Schmidt V, Hientz A, Reutershan
J, Haeberle HA and Nohé B: Mechanisms of leukocyte distribution
during sepsis: An experimental study on the interdependence of cell
activation, shear stress and endothelial injury. Crit Care.
14:R2012010. View
Article : Google Scholar : PubMed/NCBI
|
|
47
|
Liu Z, Ji J, Zheng D, Su L, Peng T and
Tang J: Protective role of endothelial calpain knockout in
lipopolysaccharide-induced acute kidney injury via attenuation of
the p38-iNOS pathway and NO/ROS production. Exp Mol Med.
52:702–712. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Fan Y, Moser J, van Meurs M, Kiers D, Sand
JMB, Leeming DJ, Pickkers P, Burgess JK, Kox M and Pillay J:
Neo-epitope detection identifies extracellular matrix turnover in
systemic inflammation and sepsis: An exploratory study. Crit Care.
28:1202024. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Fontaine M, Herkenne S, Ek O, Paquot A,
Boeckx A, Paques C, Nivelles O, Thiry M and Struman I:
Extracellular vesicles mediate communication between endothelial
and vascular smooth muscle cells. Int J Mol Sci. 23:3312021.
View Article : Google Scholar
|
|
50
|
Nonato AO, Olivon VC, Dela Justina V,
Zanotto CZ, Webb RC, Tostes RC, Lima VV and Giachini FR: Impaired
Ca(2+) homeostasis and decreased orai1 expression modulates
arterial hyporeactivity to vasoconstrictors during endotoxemia.
Inflammation. 39:1188–1197. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Pereira OR, Ramos VM, Cabral-Costa JV and
Kowaltowski AJ: Changes in mitochondrial morphology modulate
LPS-induced loss of calcium homeostasis in BV-2 microglial cells. J
Bioenerg Biomembr. 53:109–118. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Ni L, Lin B, Shen M, Li C, Hu L, Fu F,
Chen L, Yang J and Shi D: PKM2 deficiency exacerbates gram-negative
sepsis-induced cardiomyopathy via disrupting cardiac calcium
homeostasis. Cell Death Discov. 8:4962022. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Takeuchi K, del Nido PJ, Ibrahim AE,
Poutias DN, Glynn P, Cao-Danh H, Cowan DB and McGowan FX: Increased
myocardial calcium cycling and reduced myofilament calcium
sensitivity in early endotoxemia. Surgery. 126:231–238. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Sandbo N, Taurin S, Yau DM, Kregel S,
Mitchell R and Dulin NO: Downregulation of smooth muscle
alpha-actin expression by bacterial lipopolysaccharide. Cardiovasc
Res. 74:262–269. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Dyck B, Unterberg M, Adamzik M and Koos B:
The impact of pathogens on sepsis prevalence and outcome.
Pathogens. 13:892024. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Wang C, Mansard A, Giummelly P and
Atkinson J: Decreased aortic smooth muscle contraction in a rat
model of multibacterial sepsis. Fundam Clin Pharmacol. 18:679–683.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Zhao X, Miao G and Zhang L, Zhang Y, Zhao
H, Xu Z, Wang B and Zhang L: Chlamydia pneumoniae infection induces
vascular smooth muscle cell migration and atherosclerosis through
mitochondrial reactive oxygen Species-mediated JunB-Fra-1
activation. Front Cell Dev Biol. 10:8790232022. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
He H, Tan Y, Tang Z, Wang L, Liu S and Wu
G: ADAM9: A regulator between HCMV infection and function of smooth
muscle cells. J Med Virol. 95:e283522023. View Article : Google Scholar
|
|
59
|
Richards A, Khalil AS, Friesen M,
Whitfield TW, Gao X, Lungjangwa T, Kamm RD, Wan Z, Gehrke L, Mooney
D and Jaenisch R: SARS-CoV-2 infection of human pluripotent stem
cell-derived vascular cells reveals smooth muscle cells as key
mediators of vascular pathology during infection. Nat Commun.
15:107542024. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Hu X, He X, Zhang W, Jin C, Deng C, Ma Y,
Chen P, Ma S, Zhao R and Shi B: LPS induces RGS-1 to promote
infectious intracranial aneurysm formation and rupture by
accelerating smooth muscle cell phenotypic switching. Int
Immunopharmacol. 142:1132032024. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Hattori Y, Kasai K, Nakanishi N, Gross SS
and Thiemermann C: Induction of nitric oxide and
tetrahydrobiopterin synthesis by lipoteichoic acid from
Staphylococcus aureus in vascular smooth muscle cells. J Vasc Res.
35:104–108. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Sibelius U, Grandel U, Buerke M, Mueller
D, Kiss L, Kraemer HJ, Braun-Dullaeus R, Haberbosch W, Seeger W and
Grimminger F: Staphylococcal alpha-toxin provokes coronary
vasoconstriction and loss in myocardial contractility in perfused
rat hearts: Role of thromboxane generation. Circulation. 101:78–85.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Seki M, Mukohda M, Tajima H, Morikita N,
Imai R, Itaya K, Mizuno R and Ozaki H: Long-term treatment with the
streptococcal exotoxin streptolysin O inhibits vascular smooth
muscle contraction by inducing iNOS expression in endothelial
cells. J Pharmacol Exp Ther. 392:1000112025. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Kook H, Rhee JH, Lee SE, Kang SY, Chung
SS, Cho KW and Baik YH: Activation of particulate guanylyl cyclase
by Vibrio vulnificus hemolysin. Eur J Pharmacol. 365:267–272. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
DelVechio M, Alves JV, Saiyid AZ, Singh S,
Galley J, Awata WMC, Costa RM, Bruder-Nascimento A and
Bruder-Nascimento T: Progression of vascular function and blood
pressure in a mouse model of Kawasaki disease. Shock. 59:74–81.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Zhang B, Elmabsout AA, Khalaf H, Basic VT,
Jayaprakash K, Kruse R, Bengtsson T and Sirsjö A: The periodontal
pathogen Porphyromonas gingivalis changes the gene expression in
vascular smooth muscle cells involving the TGFbeta/Notch signalling
pathway and increased cell proliferation. BMC Genomics. 14:7702013.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Oseghale O, Liong S, Coward-Smith M, To
EE, Erlich JR, Luong R, Liong F, Miles M, Norouzi S, Martin C, et
al: Influenza A virus elicits peri-vascular adipose tissue
inflammation and vascular dysfunction of the aorta in pregnant
mice. PLoS Pathog. 18:e10107032022. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Liu MW, Duan SX, Zhao XY, Wang QF, Yang
SL, Ma N and Li X: Research status and advances in dexmedetomidine
for sepsis-induced multiple organ dysfunction syndrome (review).
Int J Mol Med. 55:942025. View Article : Google Scholar
|
|
69
|
Jarczak D and Nierhaus A: Cytokine
Storm-definition, causes, and implications. Int J Mol Sci.
23:117402022. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Reddy H, Javvaji CK, Malali S, Kumar S,
Acharya S and Toshniwal S: Navigating the cytokine storm: A
comprehensive review of chemokines and cytokines in sepsis. Cureus.
16:e542752024.PubMed/NCBI
|
|
71
|
Beasley D and Cooper AL: Constitutive
expression of interleukin-1alpha precursor promotes human vascular
smooth muscle cell proliferation. Am J Physiol. 276:H901–H912.
1999.PubMed/NCBI
|
|
72
|
Xu A, Pei J, Yang Y, Hua B and Wang J:
IL-1β promotes A7r5 and HASMC migration and invasion via the
p38-MAPK/Angpt-2 pathway. Eur J Med Res. 27:1532022. View Article : Google Scholar
|
|
73
|
Sun XH, Jiang HJ, Liu Q, Xiao C, Xu JY, Wu
Y, Mei JY, Wu ST and Lin ZY: Low concentrations of TNF-α in vitro
transform the phenotype of vascular smooth muscle cells and enhance
their survival in a three-dimensional culture system. Artif Organs.
48:839–848. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Liu W, Liu X, Luo M, Liu X, Luo Q, Tao H,
Wu D, Lu S, Jin J, Zhao Y and Zou L: dNK derived IFN-γ mediates
VSMC migration and apoptosis via the induction of LncRNA MEG3: A
role in uterovascular transformation. Placenta. 50:32–39. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Arumugam P, Carroll KL, Berceli SA,
Barnhill S and Wrenshall LE: Expression of a Functional IL-2
receptor in vascular smooth muscle cells. J Immunol. 202:694–703.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Brizzi MF, Formato L, Dentelli P, Rosso A,
Pavan M, Garbarino G, Pegoraro M, Camussi G and Pegoraro L:
Interleukin-3 stimulates migration and proliferation of vascular
smooth muscle cells: A potential role in atherogenesis.
Circulation. 103:549–554. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Hofbauer LC, Schrader J, Niebergall U,
Viereck V, Burchert A, Hörsch D, Preissner KT and Schoppet M:
Interleukin-4 differentially regulates osteoprotegerin expression
and induces calcification in vascular smooth muscle cells. Thromb
Haemost. 95:708–714. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Cimmino I, Prisco F, Orso S, Agognon AL,
Liguoro P, De Biase D, Doti N, Ruvo M, Paciello O, Beguinot F, et
al: Interleukin 6 reduces vascular smooth muscle cell apoptosis via
Prep1 and is associated with aging. FASEB J. 35:e219892021.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Yue TL, Wang X, Sung CP, Olson B, McKenna
PJ, Gu JL and Feuerstein GZ: Interleukin-8. A mitogen and
chemoattractant for vascular smooth muscle cells. Circ Res. 75:1–7.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Mazighi M, Pellé A, Gonzalez W, Mtairag el
M, Philippe M, Hénin D, Michel JB and Feldman LJ: IL-10 inhibits
vascular smooth muscle cell activation in vitro and in vivo. Am J
Physiol Heart Circ Physiol. 287:H866–H871. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Zimmerman MA, Selzman CH, Reznikov LL,
Raeburn CD, Barsness K, McIntyre RC Jr, Hamiel CR and Harken AH:
Interleukin-11 attenuates human vascular smooth muscle cell
proliferation. Am J Physiol Heart Circ Physiol. 283:H175–H180.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Cho WK, Lee CM, Kang MJ, Huang Y, Giordano
FJ, Lee PJ, Trow TK, Homer RJ, Sessa WC, Elias JA and Lee CG: IL-13
receptor α2-arginase 2 pathway mediates IL-13-induced pulmonary
hypertension. Am J Physiol Lung Cell Mol Physiol. 304:L112–L124.
2013. View Article : Google Scholar
|
|
83
|
Iwasaki S, Minamisawa S, Yokoyama U,
Akaike T, Quan H, Nagashima Y, Nishimaki S, Ishikawa Y and Yokota
S: Interleukin-15 inhibits smooth muscle cell proliferation and
hyaluronan production in rat ductus arteriosus. Pediatr Res.
62:392–398. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Park SL, Hwang B, Lee SY, Kim WT, Choi YH,
Chang YC, Kim WJ and Moon SK: p21WAF1 is required for
Interleukin-16-Induced migration and invasion of vascular smooth
muscle cells via the p38MAPK/Sp-1/MMP-9 pathway. PLoS One.
10:e01421532015. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Duncan JW, Granger JP, Ryan MJ and
Drummond HA: Interleukin-17 reduces βENaC via MAPK signaling in
vascular smooth muscle cells. Int J Mol Sci. 21:29532020.
View Article : Google Scholar
|
|
86
|
Zhang K, Zhang Y, Feng W, Chen R, Chen J,
Touyz RM, Wang J and Huang H: Interleukin-18 enhances vascular
calcification and osteogenic differentiation of vascular smooth
muscle cells through TRPM7 activation. Arterioscler Thromb Vasc
Biol. 37:1933–1943. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Cuneo AA, Herrick D and Autieri MV: Il-19
reduces VSMC activation by regulation of mRNA regulatory factor HuR
and reduction of mRNA stability. J Mol Cell Cardiol. 49:647–654.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Dale BL, Pandey AK, Chen Y, Smart CD,
Laroumanie F, Ao M, Xiao L, Dikalova AE, Dikalov SI, Elijovich F,
et al: Critical role of Interleukin 21 and T follicular helper
cells in hypertension and vascular dysfunction. JCI Insight.
5:e1292782019. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Rattik S, Hultman K, Rauch U, Söderberg I,
Sundius L, Ljungcrantz I, Hultgårdh-Nilsson A, Wigren M, Björkbacka
H, Fredrikson GN and Nilsson J: IL-22 affects smooth muscle cell
phenotype and plaque formation in apolipoprotein E knockout mice.
Atherosclerosis. 242:506–514. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Conway R, O'Neill L, McCarthy GM, Murphy
CC, Fabre A, Kennedy S, Veale DJ, Wade SM, Fearon U and Molloy ES:
Interleukin 12 and interleukin 23 play key pathogenic roles in
inflammatory and proliferative pathways in giant cell arteritis.
Ann Rheum Dis. 77:1815–1824. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Lee KM, Kang HA, Park M, Lee HY, Choi HR,
Yun CH, Oh JW and Kang HS: Interleukin-24 attenuates
β-glycerophosphate-induced calcification of vascular smooth muscle
cells by inhibiting apoptosis, the expression of calcification and
osteoblastic markers, and the Wnt/β-catenin pathway. Biochem
Biophys Res Commun. 428:50–55. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Hao N, Zhou Z, Zhang F, Li Y, Hu R, Zou J,
Zheng R, Wang L, Xu L, Tan W, et al: Interleukin-29 accelerates
vascular calcification via JAK2/STAT3/BMP2 signaling. J Am Heart
Assoc. 12:e0272222023. View Article : Google Scholar :
|
|
93
|
Son DJ, Jung YY, Seo YS, Park H, Lee DH,
Kim S, Roh YS, Han SB, Yoon DY and Hong JT: Interleukin-32α
inhibits endothelial inflammation, vascular smooth muscle cell
activation, and atherosclerosis by upregulating Timp3 and reck
through suppressing microRNA-205 biogenesis. Theranostics.
7:2186–2203. 2017. View Article : Google Scholar
|
|
94
|
DeVallance E, Bowdridge E, Garner K,
Griffith J, Seman M, Batchelor T, Velayutham M, Goldsmith WT,
Hussain S, Kelley EE and Nurkiewicz TR: The alarmin,
interleukin-33, increases vascular tone via extracellular signal
regulated kinase-mediated Ca2+ sensitization and endothelial nitric
oxide synthase inhibition. J Physiol. 602:6087–6107. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Skowron W, Zemanek K, Wojdan K, Gorzelak
P, Borowiec M, Broncel M and Chalubinski M: The effect of
interleukin-35 on the integrity, ICAM-1 expression and apoptosis of
human aortic smooth muscle cells. Pharmacol Rep. 67:376–381. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Ding Y, Wang Y, Cai Y, Pan C, Yang C, Wang
M, Qi X, Ye J, Ji Q, Yu J, et al: IL-37 expression in patients with
abdominal aortic aneurysm and its role in the necroptosis of
vascular smooth muscle cells. Oxid Med Cell Longev.
2022:18065132022. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Niessner A, Shin MS, Pryshchep O, Goronzy
JJ, Chaikof EL and Weyand CM: Synergistic proinflammatory effects
of the antiviral cytokine interferon-alpha and Toll-like receptor 4
ligands in the atherosclerotic plaque. Circulation. 116:2043–2052.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Sano E, Tashiro S, Tsumoto K and Ueda T:
Differential effects of IFN-β on the survival and growth of human
vascular smooth muscle and endothelial cells. Biores Open Access.
4:1–15. 2015. View Article : Google Scholar
|
|
99
|
Vasudevan SS, Lopes NH, Seshiah PN, Wang
T, Marsh CB, Kereiakes DJ, Dong C and Goldschmidt-Clermont PJ:
Mac-1 and Fas activities are concurrently required for execution of
smooth muscle cell death by M-CSF-stimulated macrophages.
Cardiovasc Res. 59:723–733. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Plenz G, Reichenberg S, Koenig C,
Rauterberg J, Deng MC, Baba HA and Robenek H:
Granulocyte-macrophage colony-stimulating factor (GM-CSF) modulates
the expression of type VIII collagen mRNA in vascular smooth muscle
cells and both are codistributed during atherogenesis. Arterioscler
Thromb Vasc Biol. 19:1658–1668. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Rinaldi B, Finicelli M, Donniacuo M, Di
Bernardo G, Gritti G, Gaudio SD, Forte A, Peluso G, Cipollaro M,
Rossi F and Galderisi U: G-CSF contributes at the healing of tunica
media of arteriotomy-injured rat carotids by promoting
differentiation of vascular smooth muscle cells. J Cell Physiol.
231:215–223. 2016. View Article : Google Scholar
|
|
102
|
Li Y, Ren S and Zhou S: Advances in sepsis
research: Insights into signaling pathways, organ failure, and
emerging intervention strategies. Exp Mol Pathol. 142:1049632025.
View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Chen C, Du Y, Nie R, Wang S, Wang H and Li
P: Notch signaling in cancers: Mechanism and potential therapy.
Front Cell Dev Biol. 13:15429672025. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Singh V, Akash R, Chaudhary G, Singh R,
Choudhury S, Shukla A, Prabhu SN, Gangwar N and Garg SK: Sepsis
downregulates aortic Notch signaling to produce vascular
hyporeactivity in mice. Sci Rep. 12:29412022. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Zeng H, He X, Tuo QH, Liao DF, Zhang GQ
and Chen JX: LPS causes pericyte loss and microvascular dysfunction
via disruption of Sirt3/angiopoietins/Tie-2 and HIF-2α/Notch3
pathways. Sci Rep. 6:209312016. View Article : Google Scholar
|
|
106
|
Guan G, Chen Y and Dong Y: Unraveling the
AMPK-SIRT1-FOXO pathway: The In-depth analysis and breakthrough
prospects of oxidative Stress-induced diseases. Antioxidants
(Basel). 14:702025. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Yang H, Luo YY, Zhang LT, He KR and Lin
XJ: Extracellular histones induce inflammation and senescence of
vascular smooth muscle cells by activating the AMPK/FOXO4 signaling
pathway. Inflamm Res. 71:1055–1066. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Millar MW, Fazal F and Rahman A:
Therapeutic targeting of NF-κB in acute lung injury: A Double-edged
sword. Cells. 11:33172022. View Article : Google Scholar
|
|
109
|
Canovas B and Nebreda AR: Diversity and
versatility of p38 kinase signalling in health and disease. Nat Rev
Mol Cell Biol. 22:346–366. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Liu Z, Ting Y, Li M, Li Y, Tan Y and Long
Y: From immune dysregulation to organ dysfunction: Understanding
the enigma of Sepsis. Front Microbiol. 15:14152742024. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Miao S, Zhang R, Guo G, Wang X, Zhang B,
Li L, Wang J, Weng Y, Shi X, Ma S and Lu C: p38 protein as a
therapeutic target for sepsis-induced organ dysfunction. Eur J
Pharmacol. 1002:1778332025. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Chan CH, Jo U, Kohrman A, Rezaeian AH,
Chou PC, Logothetis C and Lin HK: Posttranslational regulation of
Akt in human cancer. Cell Biosci. 4:592014. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Liu Y, Meng J, Liu J, Zhang F, An C and
Yang J: NLRP3 inflammasome in acute lung injury: Cumulative
evidence for traditional Chinese medicine and natural products.
Chin Herbal Med. 17:703–719. 2025. View Article : Google Scholar
|
|
114
|
Zhan X, Li Q, Xu G, Xiao X and Bai Z: The
mechanism of NLRP3 inflammasome activation and its pharmacological
inhibitors. Front Immunol. 13:11099382022. View Article : Google Scholar
|
|
115
|
Hao QY, Yan J, Wei JT, Zeng YH, Feng LY,
Que DD, Li SC, Guo JB, Fan Y, Ding YF, et al: Prevotella copri
promotes vascular calcification via lipopolysaccharide through
activation of NF-κB signaling pathway. Gut Microbes.
16:23515322024. View Article : Google Scholar
|
|
116
|
Li Z, Wu H, Yao F, Li Y, Li Y, Xie SA, Yu
F, Fu Y, Wang L, Zhou J and Kong W: Runx2-NLRP3 axis orchestrates
matrix stiffness-evoked vascular smooth muscle cell inflammation.
Am J Physiol Cell Physiol. 328:C467–C482. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Kim EJ, Park SY, Baek SE, Jang MA, Lee WS,
Bae SS, Kim K and Kim CD: HMGB1 Increases IL-1β production in
vascular smooth muscle cells via NLRP3 inflammasome. Front Physiol.
9:3132018. View Article : Google Scholar
|
|
118
|
James AM and Murphy MP: How mitochondrial
damage affects cell function. J Biomed Sci. 9:475–487. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Tsuji N, Tsuji T, Yamashita T, Hayase N,
Hu X, Yuen PS and Star RA: BAM15 treats mouse sepsis and kidney
injury, linking mortality, mitochondrial DNA, tubule damage, and
neutrophils. J Clin Invest. 133:e1524012023. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Hu D, Sheeja Prabhakaran H, Zhang YY, Luo
G, He W and Liou YC: Mitochondrial dysfunction in sepsis:
Mechanisms and therapeutic perspectives. Critical Care. 28:2922024.
View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Bauernfeind F, Bartok E, Rieger A, Franchi
L, Núñez G and Hornung V: Cutting edge: Reactive oxygen species
inhibitors block priming, but not activation, of the NLRP3
inflammasome. J Immunol. 187:613–617. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Li PF, Dietz R and von Harsdorf R:
Differential effect of hydrogen peroxide and superoxide anion on
apoptosis and proliferation of vascular smooth muscle cells.
Circulation. 96:3602–3609. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Brown MR, Miller FJ Jr, Li WG, Ellingson
AN, Mozena JD, Chatterjee P, Engelhardt JF, Zwacka RM, Oberley LW,
Fang X, et al: Overexpression of human catalase inhibits
proliferation and promotes apoptosis in vascular smooth muscle
cells. Circ Res. 85:524–533. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Bieling F, Uhle F, Weissmüller K, Weigand
MA and Henrich M: Sepsis varies arterial two-pore-domain potassium
channel messenger RNA in mice. J Surg Res. 193:816–824. 2015.
View Article : Google Scholar
|
|
125
|
Yan Z, Niu L, Wang S, Gao C and Pan S:
Intestinal Piezo1 aggravates intestinal barrier dysfunction during
sepsis by mediating Ca2+ influx. J Transl Med. 22:3322024.
View Article : Google Scholar
|
|
126
|
Orliac ML, Peroni RN, Abramoff T, Neuman
I, Podesta EJ and Adler-Graschinsky E: Increases in vanilloid TRPV1
receptor protein and CGRP content during endotoxemia in rats. Eur J
Pharmacol. 566:145–152. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Zhang FR, Tang J, Lai Y, Mo SQ, Lin ZM,
Lei QQ, Han CC, Zhou AD, Lv XF, Wang C, et al: Smooth muscle cell
Piezo1 is essential for phenotypic switch and neointimal
hyperplasia. Br J Pharmacol. 182:2031–2048. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Zhao C, Shen J, Lu Y, Ni H, Xiang M and
Xie Y: Dedifferentiation of vascular smooth muscle cells upon
vessel injury. Int Immunopharmacol. 144:1136912025. View Article : Google Scholar
|
|
129
|
Han M, Wen JK, Zheng B, Cheng Y and Zhang
C: Serum deprivation results in redifferentiation of human
umbilical vascular smooth muscle cells. Am J Physiol Cell Physiol.
291:C50–C58. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Dang YY, Chen C, Wei QF, Gao LF, Zhang SC,
Li YX and Dai XY: Dehydrocorydaline maintains the vascular smooth
muscle cell contractile phenotype by upregulating Spta1. Acta
Pharmacol Sin. 46:1303–1316. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Zhang HS, Hong H, Zeng DY, Xie LN, Cheng
Q, Pang XF and Guan QG: Atorvastatin suppresses vascular
hypersensitivity and remodeling induced by transient adventitial
administration of lipopolysaccharide in rats. Ann Transl Med.
7:3862019. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Kazune S, Piebalga A, Strike E and Vanags
I: Impaired vascular reactivity in sepsis-a systematic review with
meta-analysis. Arch Med Sci Atheroscler Dis. 4:e151–e161. 2019.
View Article : Google Scholar
|
|
133
|
Glavaš M, Gitlin-Domagalska A, Dębowski D,
Ptaszyńska N, Łęgowska A and Rolka K: Vasopressin and its
analogues: From natural hormones to multitasking peptides. Int J
Mol Sci. 23:30682022. View Article : Google Scholar
|
|
134
|
Yoshimura M, Conway-Campbell B and Ueta Y:
Arginine vasopressin: Direct and indirect action on metabolism.
Peptides. 142:1705552021. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Moreau R, Barrière E, Tazi KA, Lardeux B,
Dargère D, Urbanowicz W, Poirel O, Chauvelot-Moachon L, Guimont MC,
Bernuau D and Lebrec D: Terlipressin inhibits in vivo aortic iNOS
expression induced by lipopolysaccharide in rats with biliary
cirrhosis. Hepatology. 36:1070–1078. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Russell JA, Walley KR, Singer J, Gordon
AC, Hébert PC, Cooper DJ, Holmes CL, Mehta S, Granton JT, Storms
MM, et al: Vasopressin versus norepinephrine infusion in patients
with septic shock. N Engl J Med. 358:877–887. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Gordon AC, Russell JA, Walley KR, Singer
J, Ayers D, Storms MM, Holmes CL, Hébert PC, Cooper DJ, Mehta S, et
al: The effects of vasopressin on acute kidney injury in septic
shock. Intensive Care Med. 36:83–91. 2010. View Article : Google Scholar
|
|
138
|
Morelli A, Ertmer C, Rehberg S, Lange M,
Orecchioni A, Cecchini V, Bachetoni A, D'Alessandro M, Van Aken H,
Pietropaoli P and Westphal M: Continuous terlipressin versus
vasopressin infusion in septic shock (TERLIVAP): A randomized,
controlled pilot study. Crit Care. 13:R1302009. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Laterre PF, Berry SM, Blemings A, Carlsen
JE, François B, Graves T, Jacobsen K, Lewis RJ, Opal SM, Perner A,
et al: Effect of selepressin vs placebo on Ventilator- and
Vasopressor-free days in patients with septic shock: The SEPSIS-ACT
randomized clinical trial. JAMA. 322:1476–1485. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Chaudhary A, Bansal S, Saha S, Arora S,
Akram W and Kumar S: Phosphodiesterase inhibitors for diabetes:
From mechanistic insights to therapeutic innovations. N Emirates
Med J. 5:2024.
|
|
141
|
Zhang Z, Liang M and Wan X: Roflumilast, a
type of phosphodiesterase 4 inhibitor, can reduce intestinal injury
caused by sepsis. Exp Ther Med. 22:13982021. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Ferreira Alves G, Oliveira JG, Nakashima
MA, Delfrate G, Sordi R, Assreuy J, da Silva-Santos JE, Collino M
and Fernandes D: Cardiovascular effects of Roflumilast during
sepsis: Risks or benefits? Eur J Pharmacol. 983:1770152024.
View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Nakashima MA, Delfrate G, Albino LB, Alves
GF, Oliveira JG and Fernandes D: Impact of tadalafil on
cardiovascular and organ dysfunction induced by experimental
sepsis. Acute Crit Care. 40:46–58. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Schmidt W, Tinelli M, Secchi A, Gebhard
MM, Martin E and Schmidt H: Milrinone improves intestinal villus
blood flow during endotoxemia. Can J Anaesth. 47:673–679. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
145
|
de Miranda ML, Pereira SJ, Santos AO,
Villela NR, Kraemer-Aguiar LG and Bouskela E: Milrinone attenuates
arteriolar vasoconstriction and capillary perfusion deficits on
endotoxemic hamsters. PLoS One. 10:e01170042015. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Shati AA, Zaki MSA, Alqahtani YA,
Al-Qahtani SM, Haidara MA, Dawood AF, AlMohanna AM, El-Bidawy MH,
Alaa Eldeen M and Eid RA: Antioxidant activity of Vitamin C against
LPS-Induced septic cardiomyopathy by Down-regulation of oxidative
stress and inflammation. Curr Issues Mol Biol. 44:2387–2400. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Pehlivan VF, Pehlivan B, Duran E, Taskın
A, Koyuncu I and Çakmak Y: Effects of Vitamin E on Redox balance in
regulating thiol/disulfide homeostasis in sepsis: An antioxidant
therapy perspective. PLoS One. 20:e03363342025. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Mao H, Zhang Y, Xiong Y, Zhu Z, Wang L and
Liu X: Mitochondria-targeted antioxidant mitoquinone maintains
mitochondrial homeostasis through the Sirt3-Dependent pathway to
mitigate oxidative damage caused by renal Ischemia/Reperfusion.
Oxid Med Cell Longev. 2022:22135032022. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Ning R, Li Y, Du Z, Li T, Sun Q, Lin L, Xu
Q, Duan J and Sun Z: The mitochondria-targeted antioxidant MitoQ
attenuated PM2.5-induced vascular fibrosis via regulating
mitophagy. Redox Biol. 46:1021132021. View Article : Google Scholar
|
|
150
|
Liang B, Su J, Shao H, Chen H and Xie B:
The outcome of IV vitamin C therapy in patients with sepsis or
septic shock: A meta-analysis of randomized controlled trials. Crit
Care. 27:1092023. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Lamontagne F, Masse MH, Menard J, Sprague
S, Pinto R, Heyland DK, Cook DJ, Battista MC, Day AG, Guyatt GH, et
al: Intravenous vitamin C in adults with sepsis in the intensive
care unit. N Engl J Med. 386:2387–2398. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
He Y, Li Y, Lao Q, Qin T, Xie X and Jiang
W: Association between vitamin E and 28-day mortality in adult
sepsis patients: A retrospective cohort analysis. Am J Med Sci.
370:552–558. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Yang CH, Qiu HQ, Wang C, Tang YT, Zhang
CR, Fan YY and Jiao XY: Levosimendan relaxes thoracic aortic smooth
muscle in mice by inhibiting PKC and activating inwardly rectifying
potassium channels. J Cardiovasc Pharmacol. 83:474–481. 2024.
View Article : Google Scholar
|
|
154
|
Lei Y, Peng X, Hu Y, Xue M, Li T, Liu L
and Yang G: The calcilytic drug calhex-231 ameliorates vascular
hyporesponsiveness in traumatic hemorrhagic shock by inhibiting
oxidative stress and miR-208a-Mediated mitochondrial fission. Oxid
Med Cell Longev. 2020:41327852020. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Rodríguez EE, Jaramillo GAD, Cuellar LMR,
Herran SEP, Lima DRR and Herpain A: Levosimendan vs. Dobutamine in
patients with septic shock: A systematic review and Meta-analysis
with trial sequential analysis. J Clin Med. 14:54962025. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Morelli A, Donati A, Ertmer C, Rehberg S,
Lange M, Orecchioni A, Cecchini V, Landoni G, Pelaia P, Pietropaoli
P, et al: Levosimendan for resuscitating the microcirculation in
patients with septic shock: A randomized controlled study. Crit
Care. 14:R2322010. View
Article : Google Scholar : PubMed/NCBI
|
|
157
|
Nielsen J, Langgård M, Tengberg JF and
Kehler J: The molecular mechanism of PDE1 Regulation. Cells.
14:17222025. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Flemming S, Schlegel N, Wunder C, Meir M,
Baar W, Wollborn J, Roewer N, Germer CT and Schick MA:
Phosphodiesterase 4 inhibition dose dependently stabilizes
microvascular barrier functions and microcirculation in a rodent
model of polymicrobial sepsis. Shock. 41:537–545. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Tai-Passmann S, Slegers CAD, Hemelaar P,
Waalders N, Koopmans M, van den Bogaard B, van Lookeren Campagne M,
Goedegebuur J, Kuindersma M, Schroten N, et al: Phosphodiesterase 3
inhibitors do not influence lactate kinetics and clinical outcomes
in patients with septic shock: A multicentre cohort study. J Crit
Care. 83:1548272024. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Yamamoto M, Hosoya Y, Hanada H, Osawa T,
Arai M, Sakamoto K, Okazaki Y, Katasako-Yabumoto A, Ishizu T, Kondo
T, et al: Clinical importance of Phosphodiesterase 3 inhibitors on
outcomes in patients with cardiogenic shock-A systematic review.
Circ Rep. 7:1021–1028. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Sahoo DK, Wong D, Patani A, Paital B,
Yadav VK, Patel A and Jergens AE: Exploring the role of
antioxidants in sepsis-associated oxidative stress: A comprehensive
review. Front Cell Infect Microbiol. 14:13487132024. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Kakade S and Mani G: A comparative study
of the effects of vitamin C, sirolimus, and paclitaxel on the
growth of endothelial and smooth muscle cells for cardiovascular
medical device applications. Drug Des Devel Ther. 7:529–544.
2013.PubMed/NCBI
|
|
163
|
Ulrich-Merzenich G, Zeitler H, Panek D,
Bokemeyer D and Vetter H: Vitamin C promotes human endothelial cell
growth via the ERK-signaling pathway. Eur J Nutr. 46:87–94. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Dang H, Li J, Liu C and Xu F: The
association between Vitamin E deficiency and critically Ill
children with sepsis and septic shock. Front Nutr. 8:6484422021.
View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Cui L, Zhou Q, Zheng X, Sun B and Zhao S:
Mitoquinone attenuates vascular calcification by suppressing
oxidative stress and reducing apoptosis of vascular smooth muscle
cells via the Keap1/Nrf2 pathway. Free Radic Biol Med. 161:23–31.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Supinski GS, Murphy MP and Callahan LA:
MitoQ administration prevents endotoxin-induced cardiac
dysfunction. Am J Physiol Regul Integr Comp Physiol.
297:R1095–R1102. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Schmid D, Svoboda M, Sorgner A, Moravcevic
I, Thalhammer T, Chiba P and Möslinger T: Glibenclamide reduces
proinflammatory cytokines in an ex vivo model of human
endotoxinaemia under hypoxaemic conditions. Life Sci. 89:725–734.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Zhou R, Chen F, Li Q, Hu DY and Liu LM:
Stimulation of the adenosine A3 receptor reverses vascular
hyporeactivity after hemorrhagic shock in rats. Acta Pharmacol Sin.
31:413–420. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Li T, Liu L, Liu J, Ming J, Xu J, Yang G
and Zhang Y: Mechanisms of Rho kinase regulation of vascular
reactivity following hemorrhagic shock in rats. Shock. 29:65–70.
2008. View Article : Google Scholar
|
|
170
|
Gibraeil HD, Dittrich P, Saleh S and Mayer
B: Inhibition of endotoxin-induced vascular hyporeactivity by
4-amino-tetrahydrobiopterin. Br J Pharmacol. 131:1757–1765. 2000.
View Article : Google Scholar
|
|
171
|
Squadrito F, Altavilla D, Squadrito G,
Ferlito M, Deodato B, Arlotta M, Minutoli L, Campo GM, Bova A,
Quartarone C, et al: Protective effects of Cyclosporin-A in
splanchnic artery occlusion shock. Br J Pharmacol. 130:339–344.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Luo MH, Luo SX, Cheng Z, Yang X, Lv D, Li
X, Guo Y, Li C and Yan J: Tubeimoside I improves survival of mice
in sepsis by inhibiting inducible nitric oxide synthase expression.
Biomed Pharmacother. 126:1100832020. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Hai-Mei L, Song-Yin H, Run-Mei L,
Xiao-Huang X, Le-Quan Z, Xiao-Ping L and Jin-Wen X: Andrographolide
protects against Lipopolysaccharide-induced vascular hyporeactivity
by suppressing the expression of inducible nitric oxide in
periaortic adipose. J Cardiovasc Pharmacol. 62:154–159. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Altavilla D, Squadrito F, Campo GM,
Squadrito G, Arlotta M, Urna G, Sardella A, Quartarone C, Saitta A
and Caputi AP: The lazaroid, U-74389G, inhibits inducible nitric
oxide synthase activity, reverses vascular failure and protects
against endotoxin shock. Eur J Pharmacol. 369:49–55. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Crankshaw DJ, Pistilli MJ, O'Brien YM,
Sweeney EM, Dockery P, Holloway AC and Morrison JJ: The effects of
extracellular calcium-sensing receptor ligands on the contractility
of pregnant human myometrium in vitro. Reprod Sci. 20:882–890.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Winter A, Nepper P, Hermann M, Bayer F,
Riess S, Salem R, Hlavicka J, Prinzing A, Hecker F, Holubec T, et
al: Glibenclamide serves as a potent vasopressor to treat
vasoplegia after cardiopulmonary bypass and reperfusion in a
porcine model. Int J Mol Sci. 26:40402025. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Warrillow S, Egi M and Bellomo R:
Randomized, double-blind, placebo-controlled crossover pilot study
of a potassium channel blocker in patients with septic shock. Crit
Care Med. 34:980–985. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Corriden R, Self T, Akong-Moore K, Nizet
V, Kellam B, Briddon SJ and Hill SJ: Adenosine-A3 receptors in
neutrophil microdomains promote the formation of bacteria-tethering
cytonemes. EMBO Rep. 14:726–732. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Yang G, Xu J, Li T, Ming J, Chen W and Liu
L: Role of V1a receptor in AVP-induced restoration of vascular
hyporeactivity and its relationship to MLCP-MLC20 phosphorylation
pathway. J Surg Res. 161:312–320. 2010. View Article : Google Scholar
|
|
180
|
Wan L, Langenberg C, Bellomo R and May CN:
Angiotensin II in experimental hyperdynamic sepsis. Crit Care.
13:R1902009. View
Article : Google Scholar : PubMed/NCBI
|
|
181
|
Tsuruoka S, Schwartz GJ, Wakaumi M,
Nishiki K, Yamamoto H, Purkerson JM and Fujimura A: Nitric oxide
production modulates cyclosporin A-Induced distal renal tubular
acidosis in the rat. J Pharmacol Exp Ther. 305:840–845. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Gui Q, Jiang Z and Zhang L: Insights into
the modulatory role of cyclosporine A and its research advances in
acute inflammation. Int Immunopharmacol. 93:1074202021. View Article : Google Scholar : PubMed/NCBI
|