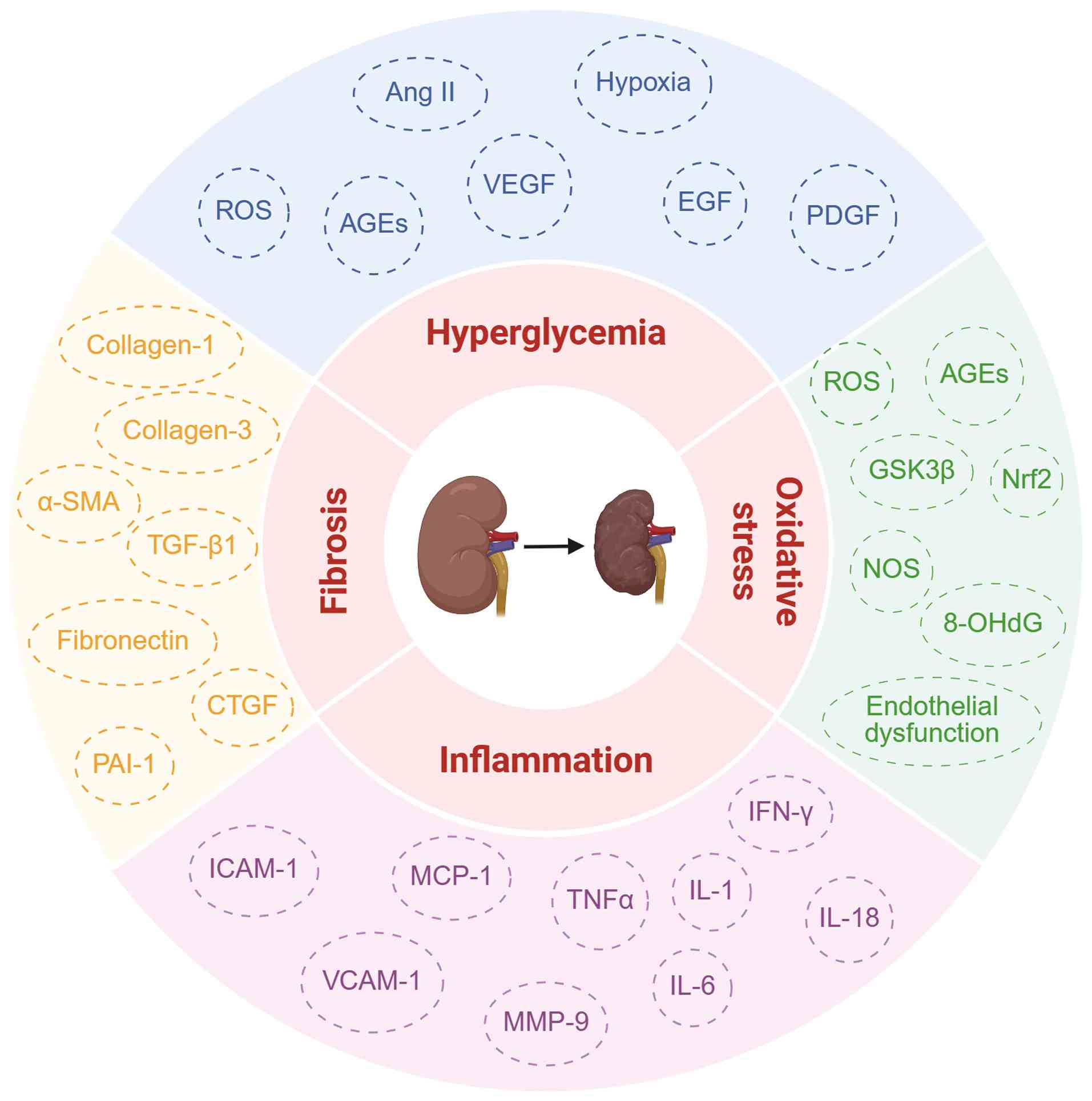
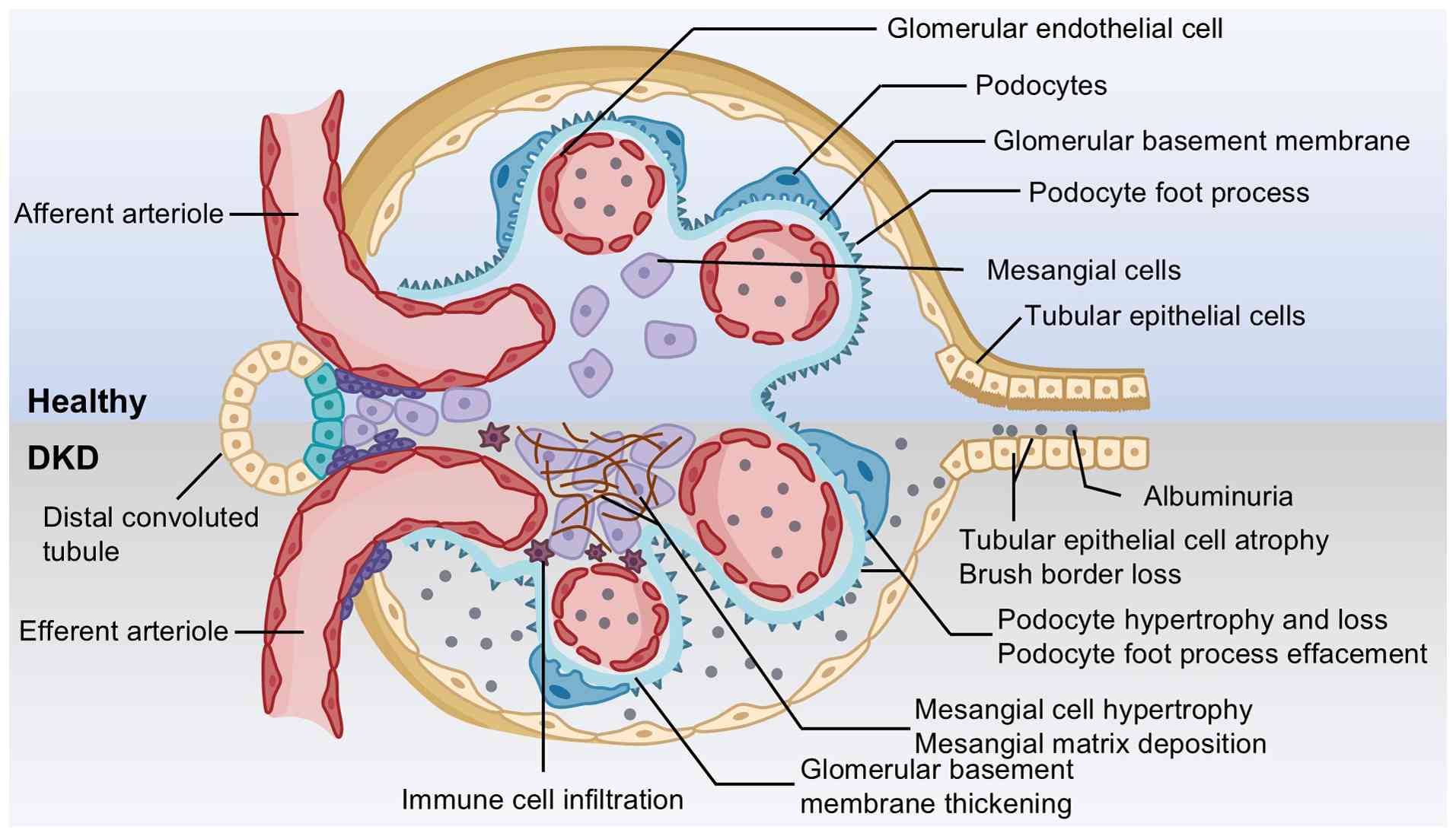
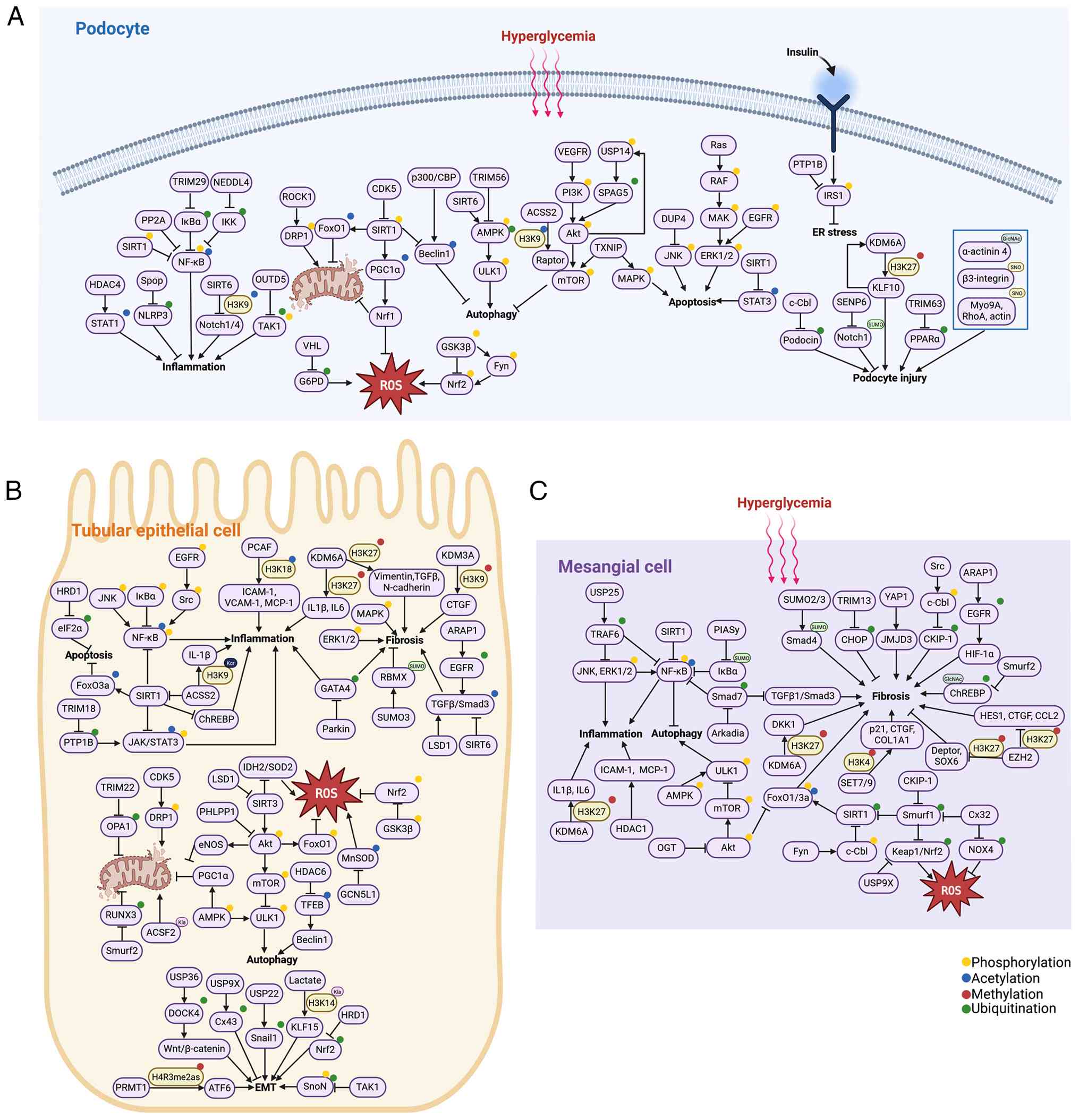
|
1
|
Martinez Leon V, Hilburg R and Susztak K:
Mechanisms of diabetic kidney disease and established and emerging
treatments. Nat Rev Endocrinol. 22:21–35. 2026. View Article : Google Scholar
|
|
2
|
Cheng HT, Xu X, Lim PS and Hung KY:
Worldwide epidemiology of diabetes-related end-stage renal disease,
2000-2015. Diabetes Care. 44:89–97. 2021. View Article : Google Scholar
|
|
3
|
Tonneijck L, Muskiet MH, Smits MM, van
Bommel EJ, Heerspink HJ, van Raalte DH and Joles JA: Glomerular
hyper-filtration in diabetes: Mechanisms, clinical significance,
and treatment. J Am Soc Nephrol. 28:1023–1039. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ye K, Zhao Y, Huang W and Zhu Y: Sodium
butyrate improves renal injury in diabetic nephropathy through
AMPK/SIRT1/PGC-1α signaling pathway. Sci Rep. 14:178672024.
View Article : Google Scholar
|
|
5
|
Lee JM, Hammarén HM, Savitski MM and Baek
SH: Control of protein stability by post-translational
modifications. Nat Commun. 14:2012023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wu X, Xu M, Geng M, Chen S, Little PJ, Xu
S and Weng J: Targeting protein modifications in metabolic
diseases: Molecular mechanisms and targeted therapies. Signal
Transduct Target Ther. 8:2202023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Burnett G and Kennedy EP: The enzymatic
phosphorylation of proteins. J Biol Chem. 211:969–980. 1954.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen T, Xie S, Cheng J, Zhao Q, Wu H,
Jiang P and Du W: AKT1 phosphorylation of cytoplasmic ME2 induces a
metabolic switch to glycolysis for tumorigenesis. Nat Commun.
15:6862024. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pearah A, Ramatchandirin B, Liu T, Wolf
RM, Ikeda A, Radovick S, Sesaki H, Wondisford FE, O'Rourke B and He
L: Blocking AMPKαS496 phosphorylation improves mitochondrial
dynamics and hyperglycemia in aging and obesity. Cell Chem Biol.
30:15852023. View Article : Google Scholar
|
|
10
|
Zhao A, Guo C, Wang L, Chen S, Xu Q, Cheng
J, Zhang J, Jiang J, Di J, Zhang H, et al: Xiebai San alleviates
acute lung injury by inhibiting the phosphorylation of the
ERK/Stat3 pathway and regulating multiple metabolisms.
Phytomedicine. 128:1553972024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu Z, Yang J, Du M and Xin W: Functioning
and mechanisms of PTMs in renal diseases. Front Pharmacol.
14:12387062023. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lu HC, Dai WN and He LY: Epigenetic
histone modifications in the pathogenesis of diabetic kidney
disease. Diabetes Metab Syndr Obes. 14:329–344. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Du C, Zhu Y, Duan J, Yang Y, Ren Y, Mu L,
Yan Z, Li G, Wang H, Shi Y and Yao F: A-485 alleviates fibrosis and
apoptosis in kidney by disrupting tandem activation of acetylation
and phosphorylation on STAT3. Biomed Pharmacother. 188:1182172025.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Natarajan R: Epigenetic mechanisms in
diabetic vascular complications and metabolic memory: The 2020
edwin bierman award lecture. Diabetes. 70:328–337. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kouzarides T: Chromatin modifications and
their function. Cell. 128:693–705. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cheng Y, Chen Y, Wang G, Liu P, Xie G,
Jing H, Chen H, Fan Y, Wang M and Zhou J: Protein methylation in
diabetic kidney disease. Front Med (Lausanne). 9:7360062022.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mevissen TET and Komander D: Mechanisms of
deubiquitinase specificity and regulation. Annu Rev Biochem.
86:159–192. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Goru SK, Kadakol A and Gaikwad AB: Hidden
targets of ubiquitin proteasome system: To prevent diabetic
nephropathy. Pharmacol Res. 120:170–179. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu B, Miao X, Shen J, Lou L, Chen K, Mei
F, Chen M, Su X, Du X, Zhu Z, et al: USP25 ameliorates diabetic
nephropathy by inhibiting TRAF6-mediated inflammatory responses.
Int Immunopharmacol. 124:1108772023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lu D, Zhang Y, Zhu P, Wu J, Yuan C and Ni
L: The roles of the ubiquitin-proteasome system in renal disease.
Int J Med Sci. 22:1791–1810. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xu X, Qin Z, Zhang C, Mi X, Zhang C, Zhou
F, Wang J, Zhang L and Hua F: TRIM29 promotes podocyte pyroptosis
in diabetic nephropathy through the NF-kB/NLRP3 inflammasome
pathway. Cell Biol Int. 47:1126–1135. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhu S, Hou S, Lu Y, Sheng W, Cui Z, Dong
T, Feng H and Wan Q: USP36-mediated deubiquitination of DOCK4
contributes to the diabetic renal tubular epithelial cell injury
via Wnt/β-Catenin signaling pathway. Front Cell Dev Biol.
9:6384772021. View Article : Google Scholar
|
|
23
|
Yang Z, Zhang Y and Sun S: Deciphering the
SUMO code in the kidney. J Cell Mol Med. 23:711–719. 2019.
View Article : Google Scholar :
|
|
24
|
Chen ZH, Li D, Zhang JY, Wei BY, Zhao HL,
Li P and Chen DQ: SUMOylation and NEDDylation in kidney diseases.
Exp Mol Pathol. 144:1050102025. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chatham JC and Patel RP: Protein
glycosylation in cardiovascular health and disease. Nat Rev
Cardiol. 21:525–544. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu Z, Qin Z, Bai W, Wang S, Huang C, Li
N, Yan L, Gu Y and Shao F: Integrating bioinformatics and machine
learning to elucidate the role of protein glycosylation-related
genes in the pathogenesis of diabetic kidney disease. PLoS One.
20:e03296402025. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ren W, Bian Q and Cai Y: Mass
spectrometry-based N-glycosylation analysis in kidney disease.
Front Mol Biosci. 9:9762982022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Magalhães A, Duarte HO and Reis CA: The
role of O-glycosylation in human disease. Mol Aspects Med.
79:1009642021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu C, Dong W, Li J, Kong Y and Ren X:
O-GlcNAc modification and its role in diabetic retinopathy.
Metabolites 2022. 12:7252022.
|
|
30
|
Ye L, Ding W, Xiao D, Jia Y, Zhao Z, Ao X
and Wang J: O-GlcNAcylation: cellular physiology and therapeutic
target for human diseases. MedComm (2020). 4:e4562023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Pasupulati AK, Nagati V, Paturi ASV and
Reddy GB: Non-enzymatic glycation and diabetic kidney disease.
Vitam Horm. 125:251–285. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ma Y, Wang X, Lin S, King L and Liu L: The
potential role of advanced glycation end products in the
development of kidney disease. Nutrients. 17:7582025. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Parwani K and Mandal P: Role of advanced
glycation end products and insulin resistance in diabetic
nephropathy. Arch Physiol Biochem. 129:95–107. 2023. View Article : Google Scholar
|
|
34
|
Chahla C, Kovacic H, Ferhat L and Leloup
L: pathological impact of redox post-translational modifications.
Antioxid Redox Signal. 41:152–180. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Peleli M, Zampas P and Papapetropoulos A:
Hydrogen sulfide and the kidney: Physiological roles, contribution
to pathophysiology, and therapeutic potential. Antioxid Redox
Signal. 36:220–243. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Vrettou S and Wirth B: S-Glutathionylation
and S-Nitrosylation in mitochondria: Focus on homeostasis and
neurodegenerative diseases. Int J Mol Sci. 23:158492022. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Nakamura T, Oh CK, Zhang X and Lipton SA:
Protein S-nitrosylation and oxidation contribute to protein
misfolding in neurodegeneration. Free Radic Biol Med. 172:562–577.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhang D, Tang Z, Huang H, Zhou G, Cui C,
Weng Y, Liu W, Kim S, Lee S, Perez-Neut M, et al: Metabolic
regulation of gene expression by histone lactylation. Nature.
574:575–580. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang Y, Li H, Jiang S, Fu D, Lu X, Lu M,
Li Y, Luo D, Wu K, Xu Y, et al: The glycolytic enzyme PFKFB3 drives
kidney fibrosis through promoting histone lactylation-mediated
NF-κB family activation. Kidney Int. 106:226–240. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ye Z, Sun Y, Yang S, Li L, Li B, Xia Y,
Yuan T, Yu W, Chen L, Zhou X and Cheng F: Lgals3 promotes calcium
oxalate crystal formation and kidney injury through histone
lactylation-mediated FGFR4 activation. Adv Sci (Weinh).
12:e24139372025. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Qiao J, Tan Y, Liu H, Yang B, Zhang Q, Liu
Q, Sun W, Li Z, Wang Q, Feng W, et al: Histone H3K18 and ezrin
lactylation promote renal dysfunction in sepsis-associated acute
kidney injury. Adv Sci (Weinh). 11:e23072162024. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Peng X and Du J: Histone and non-histone
lactylation: Molecular mechanisms, biological functions, diseases,
and therapeutic targets. Mol Biomed. 6:382025. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wei X, Long M, Yu J and Du Y: The
lactate-lactylation axis in renal fibrosis: Potential mechanisms in
diabetic kidney disease. Ann Med. 57:25873262025. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Shen R, Ruan H, Lin S, Liu B, Song H, Li L
and Ma T: Lysine succinylation, the metabolic bridge between cancer
and immunity. Genes Dis. 10:2470–2478. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Tan M, Luo H, Lee S, Jin F, Yang JS,
Montellier E, Buchou T, Cheng Z, Rousseaux S, Rajagopal N, et al:
Identification of 67 histone marks and histone lysine crotonylation
as a new type of histone modification. Cell. 146:1016–1028. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wan J, Liu H, Chu J and Zhang H: Functions
and mechanisms of lysine crotonylation. J Cell Mol Med.
23:7163–7169. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Yang P, Qin Y, Zeng L, He Y, Xie Y, Cheng
X, Huang W and Cao L: Crotonylation and disease: Current progress
and future perspectives. Biomed Pharmacother. 165:1151082023.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Li L, Xiang T, Guo J, Guo F, Wu Y, Feng H,
Liu J, Tao S, Fu P and Ma L: Inhibition of ACSS2-mediated histone
crotonylation alleviates kidney fibrosis via IL-1β-dependent
macrophage activation and tubular cell senescence. Nat Commun.
15:32002024. View Article : Google Scholar
|
|
49
|
Zhou T, Cheng X, He Y, Xie Y, Xu F, Xu Y
and Huang W: Function and mechanism of histone
beta-hydroxybutyrylation in health and disease. Front Immunol.
13:9812852022. View Article : Google Scholar
|
|
50
|
Alicic RZ, Rooney MT and Tuttle KR:
Diabetic kidney disease: Challenges, progress, and possibilities.
Clin J Am Soc Nephrol. 12:2032–2045. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Darenskaya M, Kolesnikov S, Semenova N and
Kolesnikova L: Diabetic nephropathy: Significance of determining
oxidative stress and opportunities for antioxidant therapies. Int J
Mol Sci. 24:123782023. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Mohandes S, Doke T, Hu H, Mukhi D, Dhillon
P and Susztak K: Molecular pathways that drive diabetic kidney
disease. J Clin Invest. 133:e1656542023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Jin Q, Liu T, Qiao Y, Liu D, Yang L, Mao
H, Ma F, Wang Y, Peng L and Zhan Y: Oxidative stress and
inflammation in diabetic nephropathy: Role of polyphenols. Front
Immunol. 14:11853172023. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Yao L, Liang X, Qiao Y, Chen B, Wang P and
Liu Z: Mitochondrial dysfunction in diabetic tubulopathy.
Metabolism. 131:1551952022. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Hou Y, Wang Q, Han B, Chen Y, Qiao X and
Wang L: CD36 promotes NLRP3 inflammasome activation via the mtROS
pathway in renal tubular epithelial cells of diabetic kidneys. Cell
Death Dis. 12:5232021. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Kim J, Kundu M, Viollet B and Guan KL:
AMPK and mTOR regulate autophagy through direct phosphorylation of
Ulk1. Nat Cell Biol. 13:132–141. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Chen C, Ma J, Miao CS, Zhang H, Zhang M,
Cao X and Shi Y: Trigonelline induces autophagy to protect
mesangial cells in response to high glucose via activating the
miR-5189-5p-AMPK pathway. Phytomedicine. 92:1536142021. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Jin D, Zhao Y, Sun Y, Xue J, Li X and Wang
X: Jiedu Tongluo Baoshen formula enhances renal tubular epithelial
cell autophagy to prevent renal fibrosis by activating
SIRT1/LKB1/AMPK pathway. Biomed Pharmacother. 160:1143402023.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Li A, Yi B, Han H, Yang S, Hu Z, Zheng L,
Wang J, Liao Q and Zhang H: Vitamin D-VDR (vitamin D receptor)
regulates defective autophagy in renal tubular epithelial cell in
streptozotocin-induced diabetic mice via the AMPK pathway.
Autophagy. 18:877–890. 2022. View Article : Google Scholar :
|
|
60
|
Chen Y, Zheng YF, Lin XH, Zhang JP, Lin F
and Shi H: Dendrobium mixture attenuates renal damage in rats with
diabetic nephropathy by inhibiting the PI3K/Akt/mTOR pathway. Mol
Med Rep. 24:5902021. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Wang X, Jiang L, Liu XQ, Huang YB, Wang
AL, Zeng HX, Gao L, Zhu QJ, Xia LL and Wu YG: Paeoniflorin binds to
VEGFR2 to restore autophagy and inhibit apoptosis for podocyte
protection in diabetic kidney disease through PI3K-AKT signaling
pathway. Phytomedicine. 106:1544002022. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Wang N and Zhang C: Oxidative stress: A
culprit in the progression of diabetic kidney disease. Antioxidants
(Basel). 13:4552024. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Song S, Qiu D, Wang Y, Wei J, Wu H, Wu M,
Wang S, Zhou X, Shi Y and Duan H: TXNIP deficiency mitigates
podocyte apoptosis via restraining the activation of mTOR or p38
MAPK signaling in diabetic nephropathy. Exp Cell Res.
388:1118622020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Li H, Zhao K and Li Y: Gasdermin D
protects mouse podocytes against high-glucose-induced inflammation
and apoptosis via the C-Jun N-terminal kinase (JNK) pathway. Med
Sci Monit. 27:e9284112021.PubMed/NCBI
|
|
65
|
Liu Y, Li Y, Xu L, Shi J, Yu X, Wang X, Li
X, Jiang H, Yang T, Yin X, et al: Quercetin attenuates podocyte
apoptosis of diabetic nephropathy through targeting EGFR signaling.
Front Pharmacol. 12:7927772022. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Zhai Y, Li D, Wang Z, Shao L, Yin N and Li
W: Cortex mori radicis attenuates streptozotocin-induced diabetic
renal injury in mice via regulation of transient receptor potential
canonical channel 6. Endocr Metab Immune Disord Drug Targets.
22:862–873. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Chen M, Fang Y, Ge Y, Qiu S, Dworkin L and
Gong R: The redox-sensitive GSK3β is a key regulator of glomerular
podocyte injury in type 2 diabetic kidney disease. Redox Biol.
72:1031272024. View Article : Google Scholar
|
|
68
|
Feng Q, Yu X, Xie J, Liu F, Zhang X, Li S,
Wang Y, Pan S, Liu D and Liu Z: Phillygenin improves diabetic
nephropathy by inhibiting inflammation and apoptosis via regulating
TLR4/MyD88/NF-κB and PI3K/AKT/GSK3β signaling pathways.
Phytomedicine. 136:1563142025. View Article : Google Scholar
|
|
69
|
Liang LL, He MF, Zhou PP, Pan SK, Liu DW
and Liu ZS: GSK3β: A ray of hope for the treatment of diabetic
kidney disease. FASEB J. 38:e234582024. View Article : Google Scholar
|
|
70
|
Wang H, Yu X, Liu D, Qiao Y, Huo J, Pan S,
Zhou L, Wang R, Feng Q and Liu Z: VDR activation attenuates renal
tubular epithelial cell ferroptosis by regulating Nrf2/HO-1
signaling pathway in diabetic nephropathy. Adv Sci (Weinh).
11:e23055632024. View Article : Google Scholar :
|
|
71
|
Dong D, Zhang Y, He H, Zhu Y and Ou H:
Alpinetin inhibits macrophage infiltration and atherosclerosis by
improving the thiol redox state: Requirement of GSk3β/Fyn-dependent
Nrf2 activation. FASEB J. 36:e222612022. View Article : Google Scholar
|
|
72
|
Yu X, Jiang N, Li J, Li X and He S:
Upregulation of BRD7 protects podocytes against high
glucose-induced apoptosis by enhancing Nrf2 in a GSK-3β-dependent
manner. Tissue Cell. 76:1018132022. View Article : Google Scholar
|
|
73
|
Paeng J, Chang JH, Lee SH, Nam BY, Kang
HY, Kim S, Oh HJ, Park JT, Han SH, Yoo TH and Kang SW: Enhanced
glycogen synthase kinase-3β activity mediates podocyte apoptosis
under diabetic conditions. Apoptosis. 19:1678–1690. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Xie J, Yuan Y, Yao G, Yu W and Zhu Q: Role
and mechanism of NUP160-regulated autophagy in pathogenesis of
diabetic nephropathy. Iran J Kidney Dis. 17:327–334.
2023.PubMed/NCBI
|
|
75
|
Fang X, Huang W, Sun Q, Zhao Y, Sun R, Liu
F, Huang D, Zhang Y, Gao F and Wang B: Melatonin attenuates
cellular senescence and apoptosis in diabetic nephropathy by
regulating STAT3 phosphorylation. Life Sci. 332:1221082023.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Jia J, Tan R, Xu L, Wang H, Li J, Su H,
Zhong X, Liu P and Wang L: Hederagenin improves renal fibrosis in
diabetic nephropathy by regulating Smad3/NOX4/SLC7A11
signaling-mediated tubular cell ferroptosis. Int Immunopharmacol.
135:1123032024. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Sun HJ, Xiong SP, Cao X, Cao L, Zhu MY, Wu
ZY and Bian JS: Polysulfide-mediated sulfhydration of SIRT1
prevents diabetic nephropathy by suppressing phosphorylation and
acetylation of p65 NF-κB and STAT3. Redox Biol. 38:1018132021.
View Article : Google Scholar
|
|
78
|
Lu J, Chen PP, Zhang JX, Li XQ, Wang GH,
Yuan BY, Huang SJ, Liu XQ, Jiang TT, Wang MY, et al: GPR43
deficiency protects against podocyte insulin resistance in diabetic
nephropathy through the restoration of AMPKα activity.
Theranostics. 11:4728–4742. 2021. View Article : Google Scholar
|
|
79
|
Jiang W, Xiao T, Han W, Xiong J, He T, Liu
Y, Huang Y, Yang K, Bi X, Xu X, et al: Klotho inhibits
PKCα/p66SHC-mediated podocyte injury in diabetic nephropathy. Mol
Cell Endocrinol. 494:1104902019. View Article : Google Scholar
|
|
80
|
Wang X, Zhang Y, Chi K, Ji Y, Zhang K, Li
P, Fu Z, Wang X, Cui S, Shen W, et al: IGFBP2 induces podocyte
apoptosis promoted by mitochondrial damage via integrin α5/FAK in
diabetic kidney disease. Apoptosis. 29:1109–1125. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Cao Y, Chen Z, Hu J, Feng J, Zhu Z, Fan Y,
Lin Q and Ding G: Mfn2 regulates high glucose-induced MAMs
dysfunction and apoptosis in podocytes via PERK pathway. Front Cell
Dev Biol. 9:7692132021. View Article : Google Scholar
|
|
82
|
Tian N, Gao Y, Wang X, Wu X, Zou D, Zhu Z,
Han Z, Wang T and Shi Y: Emodin mitigates podocytes apoptosis
induced by endoplasmic reticulum stress through the inhibition of
the PERK pathway in diabetic nephropathy. Drug Des Devel Ther.
12:2195–2211. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Uehara R, Yamada E, Okada S, Bastie CC,
Maeshima A, Ikeuchi H, Horiguchi K and Yamada M: Fyn phosphorylates
transglutaminase 2 (Tgm2) and modulates autophagy and p53
Expression in the development of diabetic kidney disease. Cells.
12:11972023. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Khan MF, Mathur A, Pandey VK and Kakkar P:
Endoplasmic reticulum stress-dependent activation of TRB3-FoxO1
signaling pathway exacerbates hyperglycemic nephrotoxicity:
Protection accorded by Naringenin. Eur J Pharmacol. 917:1747452022.
View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Wu H, Shi Y, Deng X, Su Y, Du C, Wei J,
Ren Y, Wu M, Hou Y and Duan H: Inhibition of c-Src/p38 MAPK pathway
ameliorates renal tubular epithelial cells apoptosis in db/db mice.
Mol Cell Endocrinol. 417:27–35. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Dusabimana T, Kim SR, Park EJ, Je J, Jeong
K, Yun SP, Kim HJ, Kim H and Park SW: P2Y2R contributes to the
development of diabetic nephropathy by inhibiting autophagy
response. Mol Metab. 42:1010892020. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Dong R, Zhang X, Liu Y, Zhao T, Sun Z, Liu
P, Xiang Q, Xiong J, Du X, Yang X, et al: Rutin alleviates EndMT by
restoring autophagy through inhibiting HDAC1 via PI3K/AKT/mTOR
pathway in diabetic kidney disease. Phytomedicine. 112:1547002023.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Lazar AG, Vlad ML, Manea A, Simionescu M
and Manea SA: Activated histone acetyltransferase p300/CBP-Related
signalling pathways mediate up-regulation of NADPH oxidase,
inflammation, and fibrosis in diabetic kidney. Antioxidants
(Basel). 10:13562021. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Li Y, Li X, He K, Li B, Liu K, Qi J, Wang
H, Wang Y and Luo W: C-peptide prevents NF-κB from recruiting p300
and binding to the inos promoter in diabetic nephropathy. FASEB J.
32:2269–2279. 2018. View Article : Google Scholar
|
|
90
|
Wang M, Huang Z, Li X, He P, Sun H, Peng Y
and Fan Q: Apabetalone, a BET protein inhibitor, inhibits kidney
damage in diabetes by preventing pyroptosis via modulating the
P300/H3K27ac/PLK1 axis. Pharmacol Res. 207:1073062024. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Lu J, Li XQ, Chen PP, Zhang JX, Liu L,
Wang GH, Liu XQ, Jiang TT, Wang MY, Liu WT, et al: Activation of
acetyl-CoA synthetase 2 mediates kidney injury in diabetic
nephropathy. JCI Insight. 8:e1658172023. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Xu J, Deng Y, Ke Y, Zhu Y, Wang P, Yu Q,
Li C and Shi B: Mutation of Beclin1 acetylation site at K414
alleviates high glucose-induced podocyte impairment in the early
stage of diabetic nephropathy by inhibiting hyperactivated
autophagy. Mol Biol Rep. 49:3919–3926. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Li X, Zhang Y, Chen H, Wu Y, Chen Y, Gong
S, Liu Y and Liu H: Inhibition of TFEB deacetylation in proximal
tubular epithelial cells (TECs) promotes TFEB activation and
alleviates TEC damage in diabetic kidney disease. FASEB J.
38:e238842024. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Brijmohan AS, Batchu SN, Majumder S,
Alghamdi TA, Thieme K, McGaugh S, Liu Y, Advani SL, Bowskill BB,
Kabir MG, et al: HDAC6 inhibition promotes transcription factor EB
activation and is protective in experimental kidney disease. Front
Pharmacol. 9:342018. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Liang T, Qi C, Lai Y, Xie J, Wang H, Zhang
L, Lin T, Jv M, Li J, Wang Y, et al: HDAC6-mediated α-tubulin
deacetylation suppresses autophagy and enhances motility of
podocytes in diabetic nephropathy. J Cell Mol Med. 24:11558–11572.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Wang X, Liu J, Zhen J, Zhang C, Wan Q, Liu
G, Wei X, Zhang Y, Wang Z, Han H, et al: Histone deacetylase 4
selectively contributes to podocyte injury in diabetic nephropathy.
Kidney Int. 86:712–725. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Wang W, Sun W, Cheng Y, Xu Z and Cai L:
Role of sirtuin-1 in diabetic nephropathy. J Mol Med (Berl).
97:291–309. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Lo CS, Shi Y, Chenier I, Ghosh A, Wu CH,
Cailhier JF, Ethier J, Lattouf JB, Filep JG, Ingelfinger JR, et al:
Heterogeneous nuclear ribonucleoprotein F stimulates sirtuin-1 gene
expression and attenuates nephropathy progression in diabetic mice.
Diabetes. 66:1964–1978. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Liu Y, Liu W, Zhang Z, Hu Y, Zhang X, Sun
Y, Lei Q, Sun D, Liu T, Fan Y, et al: Yishen capsule promotes
podocyte autophagy through regulating SIRT1/NF-κB signaling pathway
to improve diabetic nephropathy. Ren Fail. 43:128–140. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Jiao X, Li Y, Zhang T, Liu M and Chi Y:
Role of Sirtuin3 in high glucose-induced apoptosis in renal tubular
epithelial cells. Biochem Biophys Res Commun. 480:387–393. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Liu M, Liang K, Zhen J, Zhou M, Wang X,
Wang Z, Wei X, Zhang Y, Sun Y, Zhou Z, et al: Sirt6 deficiency
exacerbates podocyte injury and proteinuria through targeting Notch
signaling. Nat Commun. 8:4132017. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Liu DW, Zhang JH, Liu FX, Wang XT, Pan SK,
Jiang DK, Zhao ZH and Liu ZS: Silencing of long noncoding RNA PVT1
inhibits podocyte damage and apoptosis in diabetic nephropathy by
upregulating FOXA1. Exp Mol Med. 51:1–15. 2019. View Article : Google Scholar
|
|
103
|
Zhao Y, Li D, Zhou P, Zhao Y and Kuang J:
microRNA-29b-3p attenuates diabetic nephropathy in mice by
modifying EZH2. Hormones (Athens). 22:223–233. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Tang Y, Wan F, Tang X, Lin Y, Zhang H, Cao
J and Yang R: Celastrol attenuates diabetic nephropathy by
upregulating SIRT1-mediated inhibition of EZH2related wnt/β-catenin
signaling. Int Immunopharmacol. 122:1105842023. View Article : Google Scholar
|
|
105
|
Wang H, Wang J, Ran Q, Leng Y, Liu T,
Xiong Z, Zou D and Yang W: Identification and functional analysis
of the hub Ferroptosis-Related gene EZH2 in diabetic kidney
disease. Int Immunopharmacol. 133:1121382024. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Kim D, Ban KY, Lee GH and Jun HS:
Lysophosphatidic acid induces podocyte pyroptosis in diabetic
nephropathy by an increase of Egr1 expression via downregulation of
EzH2. Int J Mol Sci. 24:99682023. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Liebisch M and Wolf G: AGE-Induced
suppression of EZH2 mediates injury of podocytes by reducing
H3K27me3. Am J Nephrol. 51:676–692. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Lin CL, Hsu YC, Huang YT, Shih YH, Wang
CJ, Chiang WC and Chang PJ: A KDM6A-KLF10 reinforcing feedback
mechanism aggravates diabetic podocyte dysfunction. EMBO Mol Med.
11:e98282019. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Chen H, Huang Y, Zhu X, Liu C, Yuan Y, Su
H, Zhang C, Liu C, Xiong M, Qu Y, et al: Histone demethylase UTX is
a therapeutic target for diabetic kidney disease. J Physiol.
597:1643–1660. 2019. View Article : Google Scholar
|
|
110
|
Chen Q, Xie C, Tang K, Luo M, Zhang Z, Jin
Y, Liu Y, Zhou L and Kong Y: The E3 ligase Trim63 promotes podocyte
injury and proteinuria by targeting PPARα to inhibit fatty acid
oxidation. Free Radic Biol Med. 209(Pt 1): 40–54. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Liang L, He M, Zhou P, Pan S, Chen J, Lv
L, Hu M, Zhou S, Liu D and Liu Z: c-Cbl induced podocin
ubiquitination contributes to the podocytes injury in diabetic
nephropathy. FASEB J. 38:e236622024. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Xu J, Deng Y, Wang Y, Sun X, Chen S and Fu
G: SPAG5-AS1 inhibited autophagy and aggravated apoptosis of
podocytes via SPAG5/AKT/mTOR pathway. Cell Prolif. 53:e127382020.
View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Xu Y, Gao H, Hu Y, Fang Y, Qi C, Huang J,
Cai X, Wu H, Ding X and Zhang Z: High glucose-induced apoptosis and
necroptosis in podocytes is regulated by UCHL1 via RIPK1/RIPK3
pathway. Exp Cell Res. 382:1114632019. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Pontrelli P, Oranger A, Barozzino M,
Divella C, Conserva F, Fiore MG, Rossi R, Papale M, Castellano G,
Simone S, et al: Deregulation of autophagy under hyperglycemic
conditions is dependent on increased lysine 63 ubiquitination: a
candidate mechanism in the progression of diabetic nephropathy. J
Mol Med (Berl). 96:645–659. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Huang Y, Sun Y, Cao Y, Sun H, Li M, You H,
Su D, Li Y and Liang X: HRD1 prevents apoptosis in renal tubular
epithelial cells by mediating eIF2α ubiquitylation and degradation.
Cell Death Dis. 8:32022017. View Article : Google Scholar
|
|
116
|
Li X, Ma TK, Wen S, Li LL, Xu L, Zhu XW,
Zhang CX, Liu N, Wang X and Fan QL: LncRNA ARAP1-AS2 promotes high
glucose-induced human proximal tubular cell injury via persistent
transactivation of the EGFR by interacting with ARAP1. J Cell Mol
Med. 24:12994–13009. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Li X, Ma TK, Wang M, Zhang XD, Liu TY, Liu
Y, Huang ZH, Zhu YH, Zhang S, Yin L, et al: YY1-induced
upregulation of LncRNA-ARAP1-AS2 and ARAP1 promotes diabetic kidney
fibrosis via aberrant glycolysis associated with EGFR/PKM2/HIF-1α
pathway. Front Pharmacol. 14:10693482023. View Article : Google Scholar
|
|
118
|
Cai J, Wei J, Zeng Y, Yi M, Zhou Y, Ai K,
Chen W, Liu Y, Chen G, Chen A, et al: UBC9-associated SUMOylation
contributes to β-catenin activation and kidney fibrosis. Kidney
Int. 108:642–657. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Cai J, Pan J, Zeng Y, Wei J, Yi M, Xiang
Y, Liu Z, Duan S, Wang C and Dong Z: SUMOylation protects against
sepsis-associated acute kidney injury by stabilizing IκBα. Mol
Ther. Oct 27–2025.Epub ahead of print. View Article : Google Scholar
|
|
120
|
Lizotte F, Rousseau M, Denhez B, Lévesque
D, Guay A, Liu H, Moreau J, Higgins S, Sabbagh R, Susztak K, et al:
Deletion of protein tyrosine phosphatase SHP-1 restores SUMOylation
of podocin and reverses the progression of diabetic kidney disease.
Kidney Int. 104:787–802. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Wu L, Li O, Zhu F, Wang X, Chen P, Cai G,
Chen X and Hong Q: Krϋppel-like factor 15 suppresses renal
glomerular mesangial cell proliferation via enhancing P53 SUMO1
conjugation. J Cell Mol Med. 25:5691–5706. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Degrell P, Cseh J, Mohás M, Molnár GA,
Pajor L, Chatham JC, Fülöp N and Wittmann I: Evidence of O-linked
N-acetylglucosamine in diabetic nephropathy. Life Sci. 84:389–393.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Silva-Aguiar RP, Teixeira DE, Peres RAS,
Alves SAS, Novaes-Fernandes C, Dias WB, Pinheiro AAS, Peruchetti DB
and Caruso-Neves C: O-linked GlcNAcylation mediates the inhibition
of proximal tubule (Na++K+)ATPase activity in the early stage of
diabetes mellitus. Biochim Biophys Acta Gen Subj. 1867:1304662023.
View Article : Google Scholar
|
|
124
|
Gellai R, Hodrea J, Lenart L, Hosszu A,
Koszegi S, Balogh D, Ver A, Banki NF, Fulop N, Molnar A, et al:
Role of O-linked N-acetylglucosamine modification in diabetic
nephropathy. Am J Physiol Renal Physiol. 311:F1172–F1181. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Akimoto Y, Miura Y, Toda T, Wolfert MA,
Wells L, Boons GJ, Hart GW, Endo T and Kawakami H: Morphological
changes in diabetic kidney are associated with increased
O-GlcNAcylation of cytoskeletal proteins including α-actinin 4.
Clin Proteomics. 8:152011. View Article : Google Scholar
|
|
126
|
Song S, Hu T, Shi X, Jin Y, Liu S, Li X,
Zou W and Wang C: ER stress-perturbed intracellular protein
O-GlcNAcylation aggravates podocyte injury in diabetes nephropathy.
Int J Mol Sci. 24:176032023. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Sugahara S, Kume S, Chin-Kanasaki M,
Tomita I, Yasuda-Yamahara M, Yamahara K, Takeda N, Osawa N,
Yanagita M, Araki SI and Maegawa H: Protein O-GlcNAcylation is
essential for the maintenance of renal energy homeostasis and
function via lipolysis during fasting and diabetes. J Am Soc
Nephrol. 30:962–978. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Chen CH, Lin KD, Ke LY, Liang CJ, Kuo WC,
Lee MY, Lee YL, Hsiao PJ, Hsu CC and Shin SJ: O-GlcNAcylation
disrupts STRA6-retinol signals in kidneys of diabetes. Biochim
Biophys Acta Gen Subj. 1863:1059–1069. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Qin J, Chen Z, Ye M, Liang L and Ding X:
High glucose promotes O-GlcNAcylation of ACSL4 to induce
ferroptosis of renal tubular epithelial cell. Autoimmunity.
58:25768812025. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Fang M, Kang L, Wang X, Guo X, Wang W, Qin
B, Du X, Tang Q and Lin H: Inhibition of core fucosylation limits
progression of diabetic kidney disease. Biochem Biophys Res Commun.
520:612–618. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Ferreira MJ, Rodrigues TA, Pedrosa AG,
Silva AR, Vilarinho BG, Francisco T and Azevedo JE: Glutathione and
peroxisome redox homeostasis. Redox Biol. 67:1029172023. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Rashdan NA, Shrestha B and Pattillo CB:
S-glutathionylation, friend or foe in cardiovascular health and
disease. Redox Biol. 37:1016932020. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Guo Y, Liu Y, Zhao S, Xu W, Li Y, Zhao P,
Wang D, Cheng H, Ke Y and Zhang X: Oxidative stress-induced FABP5
S-glutathionylation protects against acute lung injury by
suppressing inflammation in macrophages. Nat Commun. 12:70942021.
View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Lash LH: Renal glutathione: Dual roles as
antioxidant protector and bioactivation promoter. Biochem
Pharmacol. 228:1161812024. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Li SS, Cui N, Yang Y, Trower TC, Wei YM,
Wu Y, Zhang S, Jin X and Jiang C: Impairment of the vascular KATP
channel imposes fatal susceptibility to experimental diabetes due
to multi-organ injuries. J Cell Physiol. 230:2915–2926. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Sampathkumar R, Balasubramanyam M,
Sudarslal S, Rema M, Mohan V and Balaram P: Increased
glutathionylated hemoglobin (HbSSG) in type 2 diabetes subjects
with microangiopathy. Clin Biochem. 38:892–899. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Li Q, Veron D and Tufro A: S-Nitrosylation
of RhoGAP Myosin9A is altered in advanced diabetic kidney disease.
Front Med (Lausanne). 8:6795182021. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Michelis R, Kristal B, Zeitun T, Shapiro
G, Fridman Y, Geron R and Sela S: Albumin oxidation leads to
neutrophil activation in vitro and inaccurate measurement of serum
albumin in patients with diabetic nephropathy. Free Radic Biol Med.
60:49–55. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Wu M and Ye X:
Quercetin-4'-O-β-D-glucopyranoside inhibits podocyte injury by
SIRT5-mediated desuccinylation of NEK7. Clin Exp Pharmacol Physiol.
51:e139092024. View Article : Google Scholar
|
|
140
|
Suk Kang J, Son SS, Lee JH, Lee SW, Jeong
AR, Lee ES, Cha SK, Chung CH and Lee EY: Protective effects of
klotho on palmitate-induced podocyte injury in diabetic
nephropathy. PLoS One. 16:e02506662021. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Liu H, Chen W, Lu P, Ma Y, Liang X and Liu
Y: Ginsenoside Rg1 attenuates the inflammation and oxidative stress
induced by diabetic nephropathy through regulating the
PI3K/AKT/FOXO3 pathway. Ann Transl Med. 9:17892021. View Article : Google Scholar
|
|
142
|
Park HS, Lim JH, Kim MY, Kim Y, Hong YA,
Choi SR, Chung S, Kim HW, Choi BS, Kim YS, et al: Resveratrol
increases AdipoR1 and AdipoR2 expression in type 2 diabetic
nephropathy. J Transl Med. 14:1762016. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Huang Q, Ouyang DS and Liu Q: Isoeucommin
A attenuates kidney injury in diabetic nephropathy through the
Nrf2/HO-1 pathway. FEBS Open Bio. 11:2350–2363. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Garner KL, Betin VMS, Pinto V, Graham M,
Abgueguen E, Barnes M, Bedford DC, McArdle CA and Coward RJM:
Enhanced insulin receptor, but not PI3K, signalling protects
podocytes from ER stress. Sci Rep. 8:39022018. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Li L, Zou J, Zhou M, Li H, Zhou T, Liu X,
Huang Q, Yang S, Xiang Q and Yu R: Phenylsulfate-induced oxidative
stress and mitochondrial dysfunction in podocytes are ameliorated
by Astragaloside IV activation of the SIRT1/PGC1α/Nrf1 signaling
pathway. Biomed Pharmacother. 177:1170082024. View Article : Google Scholar
|
|
146
|
Wang X, Gao Y, Tian N, Wang T, Shi Y, Xu J
and Wu B: Astragaloside IV inhibits glucose-induced
epithelial-mesenchymal transition of podocytes through autophagy
enhancement via the SIRT-NF-κB p65 axis. Sci Rep. 9:3232019.
View Article : Google Scholar
|
|
147
|
Lu J, Li XQ, Chen PP, Zhang JX, Li L, Wang
GH, Liu XQ, Jiang CM and Ma KL: Acetyl-CoA synthetase 2 promotes
diabetic renal tubular injury in mice by rewiring fatty acid
metabolism through SIRT1/ChREBP pathway. Acta Pharmacol Sin.
45:366–377. 2024. View Article : Google Scholar
|
|
148
|
Ogura Y, Kitada M, Monno I, Kanasaki K,
Watanabe A and Koya D: Renal mitochondrial oxidative stress is
enhanced by the reduction of Sirt3 activity, in Zucker diabetic
fatty rats. Redox Rep. 23:153–159. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Lv T, Lu Y, Liu Y, Feng H, Li C, Sheng W,
Cui Z, Zhu S, Gu X, Yang Z and Wan Q: General control of amino acid
synthesis 5-like 1-mediated acetylation of manganese superoxide
dismutase regulates oxidative stress in diabetic kidney disease.
Oxid Med Cell Longev. 2021:66912262021. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Chen J, Guo Y, Zeng W, Huang L, Pang Q,
Nie L, Mu J, Yuan F and Feng B: ER stress triggers MCP-1 expression
through SET7/9-induced histone methylation in the kidneys of db/db
mice. Am J Physiol Renal Physiol. 306:F916–F925. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Zhao M, Wang S, Zuo A, Zhang J, Wen W,
Jiang W, Chen H, Liang D, Sun J and Wang M: HIF-1α/JMJD1A signaling
regulates inflammation and oxidative stress following hyperglycemia
and hypoxia-induced vascular cell injury. Cell Mol Biol Lett.
26:402021. View Article : Google Scholar
|
|
152
|
Siddiqi FS, Majumder S, Thai K, Abdalla M,
Hu P, Advani SL, White KE, Bowskill BB, Guarna G, Dos Santos CC, et
al: The histone methyltransferase enzyme enhancer of zeste homolog
2 protects against podocyte oxidative stress and renal injury in
diabetes. J Am Soc Nephrol. 27:2021–2034. 2016. View Article : Google Scholar
|
|
153
|
Dai X, Liao R, Liu C, Liu S, Huang H, Liu
J, Jin T, Guo H, Zheng Z, Xia M, et al: Epigenetic regulation of
TXNIP-mediated oxidative stress and NLRP3 inflammasome activation
contributes to SAHH inhibition-aggravated diabetic nephropathy.
Redox Biol. 45:1020332021. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Li T, Yu C and Zhuang S: Histone
methyltransferase EZH2: A potential therapeutic target for kidney
diseases. Front Physiol. 12:6407002021. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Wang M, Hu J, Yan L, Yang Y, He M, Wu M,
Li Q, Gong W, Yang Y, Wang Y, et al: High glucose-induced
ubiquitination of G6PD leads to the injury of podocytes. FASEB J.
33:6296–6310. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Mathur A, Pandey VK, Khan MF and Kakkar P:
PHLPP1/Nrf2-Mdm2 axis induces renal apoptosis via influencing
nucleo-cytoplasmic shuttling of FoxO1 during diabetic nephropathy.
Mol Cell Biochem. 476:3681–3699. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Ye G, Hu ML and Xiao L: Forkhead box
A2-mediated lncRNA SOX2OT up-regulation alleviates oxidative stress
and apoptosis of renal tubular epithelial cells by promoting SIRT1
expression in diabetic nephropathy. Nephrology (Carlton).
28:196–207. 2023. View Article : Google Scholar
|
|
158
|
Li S, Lin Z, Xiao H, Xu Z, Li C, Zeng J,
Xie X, Deng L and Huang H: Fyn deficiency inhibits oxidative stress
by decreasing c-Cbl-mediated ubiquitination of Sirt1 to attenuate
diabetic renal fibrosis. Metabolism. 139:1553782023. View Article : Google Scholar
|
|
159
|
Chen ZQ, Sun XH, Li XJ, Xu ZC, Yang Y, Lin
ZY, Xiao HM, Zhang M, Quan SJ and Huang HQ: Polydatin attenuates
renal fibrosis in diabetic mice through regulating the Cx32-Nox4
signaling pathway. Acta Pharmacol Sin. 41:1587–1596. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Du L, Wang L, Wang B, Wang J, Hao M, Chen
YB, Li XZ, Li Y, Jiang YF, Li CC, et al: A novel compound AB38b
attenuates oxidative stress and ECM protein accumulation in kidneys
of diabetic mice through modulation of Keap1/Nrf2 signaling. Acta
Pharmacol Sin. 41:358–372. 2020. View Article : Google Scholar :
|
|
161
|
Gong W, Chen Z, Zou Y, Zhang L, Huang J,
Liu P and Huang H: CKIP-1 affects the polyubiquitination of Nrf2
and Keap1 via mediating Smurf1 to resist HG-induced renal fibrosis
in GMCs and diabetic mice kidneys. Free Radic Biol Med.
115:338–350. 2018. View Article : Google Scholar
|
|
162
|
Chen J, Ou Z, Gao T, Yang Y, Shu A, Xu H,
Chen Y and Lv Z: Ginkgolide B alleviates oxidative stress and
ferroptosis by inhibiting GPX4 ubiquitination to improve diabetic
nephropathy. Biomed Pharmacother. 156:1139532022. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Zhao QX, Yan SB, Wang F, Li XX, Shang GK,
Zheng ZJ, Xiao J, Lin ZW, Li CB and Ji XP: STING deficiency
alleviates ferroptosis through FPN1 stabilization in diabetic
kidney disease. Biochem Pharmacol. 222:1161022024. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Kim M, Bae JY, Yoo S, Kim HW, Lee SA, Kim
ET and Koh G: 2-Deoxy-d-ribose induces ferroptosis in renal tubular
epithelial cells via ubiquitin-proteasome system-mediated xCT
protein degradation. Free Radic Biol Med. 208:384–393. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Xu L, Zhou Y, Wang G, Bo L, Jin B, Dai L,
Lu Q, Cai X, Hu L, Liu L, et al: The UDPase ENTPD5 regulates ER
stress-associated renal injury by mediating protein
N-glycosylation. Cell Death Dis. 14:1662023. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Jo R, Shibata H, Kurihara I, Yokota K,
Kobayashi S, Murai-Takeda A, Mitsuishi Y, Hayashi T, Nakamura T and
Itoh H: Mechanisms of mineralocorticoid receptor-associated
hypertension in diabetes mellitus: the role of O-GlcNAc
modification. Hypertens Res. 46:19–31. 2023. View Article : Google Scholar
|
|
167
|
Sampathkumar R, Balasubramanyam M, Tara C,
Rema M and Mohan V: Association of hypoglutathionemia with reduced
Na+/K+ ATPase activity in type 2 diabetes and microangiopathy. Mol
Cell Biochem. 282:169–176. 2006. View Article : Google Scholar
|
|
168
|
Dursun E, Timur M, Dursun B, Süleymanlar G
and Ozben T: Protein oxidation in type 2 diabetic patients on
hemodialysis. J Diabetes Complications. 19:142–146. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Wang M, Li Q, Wang S, Zuo L, Hai Y, Yuan
S, Li X, Huang X, Yang C, Yao L, et al: Astragaloside IV protects
renal tubular epithelial cells against oxidative stress-induced
injury by upregulating CPT1A-mediated HSD17B10 lysine succinylation
in diabetic kidney disease. Phytother Res. 38:4519–4540. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Lin Q, Ma Y, Chen Z, Hu J, Chen C, Fan Y,
Liang W and Ding G: Sestrin-2 regulates podocyte mitochondrial
dysfunction and apoptosis under high-glucose conditions via AMPK.
Int J Mol Med. 45:1361–1372. 2020.PubMed/NCBI
|
|
171
|
Sun MY, Ye HJ, Zheng C, Jin ZJ, Yuan Y and
Weng HB: Astragalin ameliorates renal injury in diabetic mice by
modulating mitochondrial quality control via AMPK-dependent PGC1α
pathway. Acta Pharmacol Sin. 44:1676–1686. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Yao L, Liang X, Liu Y, Li B, Hong M, Wang
X, Chen B, Liu Z and Wang P: Non-steroidal mineralocorticoid
receptor antagonist finerenone ameliorates mitochondrial
dysfunction via PI3K/Akt/eNOS signaling pathway in diabetic
tubulopathy. Redox Biol. 68:1029462023. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Kong Z, Xiao M, Wang B, Zhang W, Che K, Lv
W, Wang Y, Huang Y, Zhao H, Zhao Y, et al: Renoprotective effect of
isoorientin in diabetic nephropathy via activating autophagy and
inhibiting the PI3K-AKT-TSC2-mTOR pathway. Am J Chin Med.
51:1269–1291. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Rousseau M, Denhez B, Spino C, Lizotte F,
Guay A, Côté A-M, Burger D and Geraldes P: Reduction of DUSP4
contributes to podocytes oxidative stress, insulin resistance and
diabetic nephropathy. Biochem Biophys Res Commun. 624:127–133.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Wang S, Yang Y, He X, Yang L, Wang J, Xia
S, Liu D, Liu S, Yang L, Liu W and Duan H: Cdk5-mediated
phosphorylation of Sirt1 contributes to podocyte mitochondrial
dysfunction in diabetic nephropathy. Antioxid Redox Signal.
34:171–190. 2021. View Article : Google Scholar
|
|
176
|
Zeng Y, Guo M, Wu Q, Tan X, Jiang C, Teng
F, Chen J, Zhang F, Ma X, Li X, et al: Gut microbiota-derived
indole-3-propionic acid alleviates diabetic kidney disease through
its mitochondrial protective effect via reducing ubiquitination
mediated-degradation of SIRT1. J Adv Res. 73:607–630. 2025.
View Article : Google Scholar :
|
|
177
|
Wang F, Xu W, Liu X and Zhang J:
Dexmedetomidine ameliorates high glucose-induced
epithelial-mesenchymal transformation in HK-2 cells through the
Cdk5/Drp1/ROS pathway. Acta Biochim Biophys Sin (Shanghai).
56:71–81. 2024. View Article : Google Scholar :
|
|
178
|
Chen Z, Ma Y, Yang Q, Hu J, Feng J, Liang
W and Ding G: AKAP1 mediates high glucose-induced mitochondrial
fission through the phosphorylation of Drp1 in podocytes. J Cell
Physiol. 235:7433–7448. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Liu S, Li X, Wen R, Chen L, Yang Q, Song
S, Xiao G, Su Z and Wang C: Increased thromboxane/prostaglandin
receptors contribute to high glucose-induced podocyte injury and
mitochondrial fission through ROCK1-Drp1 signaling. Int J Biochem
Cell Biol. 151:1062812022. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Wang W, Wang Y, Long J, Wang J, Haudek SB,
Overbeek P, Chang BH, Schumacker PT and Danesh FR: Mitochondrial
fission triggered by hyperglycemia is mediated by ROCK1 activation
in podocytes and endothelial cells. Cell Metab. 15:186–200. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Zhu X, Deng Z, Cao Y, Zhou Z, Sun W, Liu
C, Fan S and Yin XX: Resveratrol prevents Drp1-mediated
mitochondrial fission in the diabetic kidney through the PDE4D/PKA
pathway. Phytother Res. 37:5916–5931. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Picca A, Faitg J, Auwerx J, Ferrucci L and
D'Amico D: Mitophagy in human health, ageing and disease. Nat
Metab. 5:2047–2061. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Li K, Xia X and Tong Y: Multiple roles of
mitochondrial autophagy receptor FUNDC1 in mitochondrial events and
kidney disease. Front Cell Dev Biol. 12:14533652024. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Liu L, Feng D, Chen G, Chen M, Zheng Q,
Song P, Ma Q, Zhu C, Wang R, Qi W, et al: Mitochondrial
outer-membrane protein FUNDC1 mediates hypoxia-induced mitophagy in
mammalian cells. Nat Cell Biol. 14:177–185. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Zheng T, Wang HY, Chen Y, Chen X, Wu ZL,
Hu QY and Sun H: Src activation aggravates podocyte injury in
diabetic nephropathy via suppression of FUNDC1-mediated mitophagy.
Front Pharmacol. 13:8970462022. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Feng J, Chen Z, Ma Y, Yang X, Zhu Z, Zhang
Z, Hu J, Liang W and Ding G: AKAP1 contributes to impaired mtDNA
replication and mitochondrial dysfunction in podocytes of diabetic
kidney disease. Int J Biol Sci. 18:4026–4042. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Li X, Yang Q, Liu S, Song S and Wang C:
Mitochondria-associated endoplasmic reticulum membranes promote
mitochondrial fission through AKAP1-Drp1 pathway in podocytes under
high glucose conditions. Exp Cell Res. 424:1135122023. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Small DM, Morais C, Coombes JS, Bennett
NC, Johnson DW and Gobe GC: Oxidative stress-induced alterations in
PPAR-γ and associated mitochondrial destabilization contribute to
kidney cell apoptosis. Am J Physiol Renal Physiol. 307:F814–F822.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Lee J, Tsogbadrakh B, Yang S, Ryu H, Kang
E, Kang M, Kang HG, Ahn C and Oh KH: Klotho ameliorates diabetic
nephropathy via LKB1-AMPK-PGC1α-mediated renal mitochondrial
protection. Biochem Biophys Res Commun. 534:1040–1046. 2021.
View Article : Google Scholar
|
|
190
|
Pham TK, Nguyen THT, Yun HR, Vasileva EA,
Mishchenko NP, Fedoreyev SA, Stonik VA, Vu TT, Nguyen HQ, Cho SW,
et al: Echinochrome a prevents diabetic nephropathy by inhibiting
the PKC-Iota pathway and enhancing renal mitochondrial function in
db/db Mice. Mar Drugs. 21:2222023. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Zhou D, Zhou M, Wang Z, Fu Y, Jia M, Wang
X, Liu M, Zhang Y, Sun Y, Lu Y, et al: PGRN acts as a novel
regulator of mitochondrial homeostasis by facilitating mitophagy
and mitochondrial biogenesis to prevent podocyte injury in diabetic
nephropathy. Cell Death Dis. 10:5242019. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Saxena S, Anand SK, Sharma A and Kakkar P:
Involvement of Sirt1-FoxO3a-Bnip3 axis and autophagy mediated
mitochondrial turnover in according protection to hyperglycemic
NRK-52E cells by Berberine. Toxicol In Vitro. 100:1059162024.
View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Li Q, Liao J, Chen W, Zhang K, Li H, Ma F,
Zhang H, Han Q, Guo J, Li Y, et al: NAC alleviative ferroptosis in
diabetic nephropathy via maintaining mitochondrial redox
homeostasis through activating SIRT3-SOD2/Gpx4 pathway. Free Radic
Biol Med. 187:158–170. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Fan Y, Yang Q, Yang Y, Gao Z, Ma Y, Zhang
L, Liang W and Ding G: Sirt6 suppresses high glucose-induced
mitochondrial dysfunction and apoptosis in podocytes through AMPK
activation. Int J Biol Sci. 15:701–713. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Zhang Z, Zhou F, Lu M, Zhang D, Zhang X,
Xu S and He Y: WTAP-mediated m6A modification of TRIM22 promotes
diabetic nephropathy by inducing mitochondrial dysfunction via
ubiquitination of OPA1. Redox Rep. 29:24047942024. View Article : Google Scholar
|
|
196
|
Lv Z, Wang Z, Hu J, Su H, Liu B, Lang Y,
Yu Q, Liu Y, Fan X, Yang M, et al: LncRNA PVT1 induces
mitochondrial dysfunction of podocytes via TRIM56 in diabetic
kidney disease. Cell Death Dis. 15:6972024. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Xiao L and Ye G: RUNX3 alleviates
mitochondrial dysfunction and tubular damage by inhibiting
TLR4/NF-κB signalling pathway in diabetic kidney disease.
Nephrology (Carlton). 29:470–481. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Ji X, Yang X, Gu X, Chu L, Sun S, Sun J,
Song P, Mu Q, Wang Y, Sun X, et al: CUL3 induces mitochondrial
dysfunction via MRPL12 ubiquitination in renal tubular epithelial
cells. FEBS J. 290:5340–5352. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Yang Y, Ren S, Xue J, Dong W, He W, Luo J,
Li X, Xu H, Zheng Z, Wang X, et al: DeSUMOylation of RBMX regulates
exosomal sorting of cargo to promote renal tubulointerstitial
fibrosis in diabetic kidney disease. J Adv Res. 74:175–189. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Pérez-Gallardo RV, Noriega-Cisneros R,
Esquivel-Gutiérrez E, Calderón-Cortés E, Cortés-Rojo C,
Manzo-Avalos S, Campos-García J, Salgado-Garciglia R, Montoya-Pérez
R, Boldogh I and Saavedra-Molina A: Effects of diabetes on
oxidative and nitrosative stress in kidney mitochondria from aged
rats. J Bioenerg Biomembr. 46:511–518. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Rosca MG, Mustata TG, Kinter MT, Ozdemir
AM, Kern TS, Szweda LI, Brownlee M, Monnier VM and Weiss MF:
Glycation of mitochondrial proteins from diabetic rat kidney is
associated with excess superoxide formation. Am J Physiol Renal
Physiol. 289:F420–F430. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Rosca MG, Monnier VM, Szweda LI and Weiss
MF: Alterations in renal mitochondrial respiration in response to
the reactive oxoaldehyde methylglyoxal. Am J Physiol Renal Physiol.
283:F52–F59. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Chen J, Feng Q, Qiao Y, Pan S, Liang L,
Liu Y, Zhang X, Liu D and Liu Z and Liu Z: ACSF2 and lysine
lactylation contribute to renal tubule injury in diabetes.
Diabetologia. 67:1429–1443. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Fan Z, Zhang Y, Yuan L, Gao Y, Tian X,
Tian J, Wan J, Li B, Wang X, Wang S, et al: LARS1 lactylation
inhibits autophagy by activating mTORC1 to promote podocytes injury
in diabetic kidney disease. Cell Signal. 134:1119552025. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Qian J, Yin S, Ye L, Wang Z, Shu S, Mou Z,
Xu M, Chattipakorn N, Liu Z and Liang G: An Indole-2-Carboxamide
Derivative, LG4, alleviates diabetic kidney disease through
inhibiting MAPK-Mediated inflammatory responses. J Inflamm Res.
14:1633–1645. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Li X, Wang J, Yan J, He JC, Li Y and Zhong
Y: Additive renal protective effects between arctigenin and
puerarin in diabetic kidney disease. Biomed Pharmacother.
171:1161072024. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Xu X, Zhang L, Hua F, Zhang C, Zhang C, Mi
X, Qin N, Wang J, Zhu A, Qin Z and Zhou F: FOXM1-activated SIRT4
inhibits NF-κB signaling and NLRP3 inflammasome to alleviate kidney
injury and podocyte pyroptosis in diabetic nephropathy. Exp Cell
Res. 408:1128632021. View Article : Google Scholar
|
|
208
|
Ma L, Wu F, Shao Q, Chen G, Xu L and Lu F:
Baicalin alleviates oxidative stress and inflammation in diabetic
nephropathy via Nrf2 and MAPK signaling pathway. Drug Des Devel
Ther. 15:3207–3221. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Li X, Wen J, Dong Y, Zhang Q, Guan J, Liu
F, Zhou T, Li Z, Fan Y and Wang N: Wnt5a promotes renal tubular
inflammation in diabetic nephropathy by binding to CD146 through
noncanonical Wnt signaling. Cell Death Dis. 12:922021. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Pei D, Tian S, Bao Y, Zhang J, Xu D and
Piao M: Protective effect of salidroside on streptozotocin-induced
diabetic nephropathy by inhibiting oxidative stress and
inflammation in rats via the Akt/GSK-3β signalling pathway. Pharm
Biol. 60:1732–1738. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Han X, Wei J, Zheng R, Tu Y, Wang M, Chen
L, Xu Z, Zheng L, Zheng C, Shi Q, et al: Macrophage SHP2 deficiency
alleviates diabetic nephropathy via suppression of
MAPK/NF-κB-dependent inflammation. Diabetes. 73:780–796. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Li J, Zhang J, Zhao X and Tian L: MSU
crystallization promotes fibroblast proliferation and renal
fibrosis in diabetic nephropathy via the ROS/SHP2/TGFβ pathway. Sci
Rep. 14:202512024. View Article : Google Scholar
|
|
213
|
Wei TT, Yang LT, Guo F, Tao SB, Cheng L,
Huang RS, Ma L and Fu P: Activation of GPR120 in podocytes
ameliorates kidney fibrosis and inflammation in diabetic
nephropathy. Acta Pharmacol Sin. 42:252–263. 2021. View Article : Google Scholar
|
|
214
|
Yang WX, Liu Y, Zhang SM, Wang HF, Liu YF,
Liu JL, Li XH, Zeng MR, Han YZ, Liu FY, et al: Epac activation
ameliorates tubulointerstitial inflammation in diabetic
nephropathy. Acta Pharmacol Sin. 43:659–671. 2022. View Article : Google Scholar
|
|
215
|
Reddy MA, Sumanth P, Lanting L, Yuan H,
Wang M, Mar D, Alpers CE, Bomsztyk K and Natarajan R: Losartan
reverses permissive epigenetic changes in renal glomeruli of
diabetic db/db mice. Kidney Int. 85:362–373. 2014. View Article : Google Scholar :
|
|
216
|
Huang J, Wan D, Li J, Chen H, Huang K and
Zheng L: Histone acetyltransferase PCAF regulates inflammatory
molecules in the development of renal injury. Epigenetics.
10:62–72. 2015. View Article : Google Scholar
|
|
217
|
Pandey A and Gaikwad AB: Compound 21 and
telmisartan combination mitigates type 2 diabetic nephropathy
through amelioration of caspase mediated apoptosis. Biochem Biophys
Res Commun. 487:827–833. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Nicholas SB, Aguiniga E, Ren Y, Kim J,
Wong J, Govindarajan N, Noda M, Wang W, Kawano Y, Collins A and
Hsueh WA: Plasminogen activator inhibitor-1 deficiency retards
diabetic nephropathy. Kidney Int. 67:1297–1307. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Chen H, Li J, Jiao L, Petersen RB, Li J,
Peng A, Zheng L and Huang K: Apelin inhibits the development of
diabetic nephropathy by regulating histone acetylation in Akita
mouse. J Physiol. 592:505–521. 2014. View Article : Google Scholar
|
|
220
|
Du YG, Wang LP, Qian JW, Zhang KN and Chai
KF: Panax notoginseng saponins protect kidney from diabetes by
up-regulating silent information regulator 1 and activating
antioxidant proteins in rats. Chin J Integr Med. 22:910–917. 2016.
View Article : Google Scholar
|
|
221
|
Kitada M, Takeda A, Nagai T, Ito H,
Kanasaki K and Koya D: Dietary restriction ameliorates diabetic
nephropathy through anti-inflammatory effects and regulation of the
autophagy via restoration of Sirt1 in diabetic Wistar fatty (fa/fa)
rats: A model of type 2 diabetes. Exp Diabetes Res.
2011:9081852011. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Du YG, Zhang KN, Gao ZL, Dai F, Wu XX and
Chai KF: Tangshen formula improves inflammation in renal tissue of
diabetic nephropathy through SIRT1/NF-κB pathway. Exp Ther Med.
15:2156–2164. 2018.PubMed/NCBI
|
|
223
|
Liu HW, Kao HH and Wu CH: Exercise
training upregulates SIRT1 to attenuate inflammation and metabolic
dysfunction in kidney and liver of diabetic db/db mice. Nutr Metab
(Lond). 16:222019. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Li X, Liu J, Lu L, Huang T, Hou W, Wang F,
Yu L, Wu F, Qi J, Chen X, et al: Sirt7 associates with ELK1 to
participate in hyperglycemia memory and diabetic nephropathy via
modulation of DAPK3 expression and endothelial inflammation. Transl
Res. 247:99–116. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Sankrityayan H, Kale A, Shelke V and
Gaikwad AB: Cyproheptadine, a SET7/9 inhibitor, reduces
hyperglycaemia-induced ER stress alleviating inflammation and
fibrosis in renal tubular epithelial cells. Arch Physiol Biochem.
130:411–419. 2024. View Article : Google Scholar
|
|
226
|
Brasacchio D, Okabe J, Tikellis C,
Balcerczyk A, George P, Baker EK, Calkin AC, Brownlee M, Cooper ME
and El-Osta A: Hyperglycemia induces a dynamic cooperativity of
histone methylase and demethylase enzymes associated with
gene-activating epigenetic marks that coexist on the lysine tail.
Diabetes. 58:1229–1236. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Wang F, Hou W, Li X, Lu L, Huang T, Zhu M
and Miao C: SETD8 cooperates with MZF1 to participate in
hyperglycemia-induced endothelial inflammation via elevation of
WNT5A levels in diabetic nephropathy. Cell Mol Biol Lett.
27:302022. View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Hou W, Lu L, Li X, Sun M, Zhu M and Miao
C: c-Myc participates in high glucose-mediated endothelial
inflammation via upregulation of IRAK1 expression in diabetic
nephropathy. Cell Signal. 92:1102632022. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Huang T, Li X, Wang F, Lu L, Hou W, Zhu M
and Miao C: The CREB/KMT5A complex regulates PTP1B to modulate high
glucose-induced endothelial inflammatory factor levels in diabetic
nephropathy. Cell Death Dis. 12:3332021. View Article : Google Scholar : PubMed/NCBI
|
|
230
|
Wang Y, Xu J and Cheng Z: YAP1 promotes
high glucose-induced inflammation and extracellular matrix
deposition in glomerular mesangial cells by modulating NF-κB/JMJD3
pathway. Exp Ther Med. 22:13492021. View Article : Google Scholar
|
|
231
|
Zheng W, Guo J and Liu ZS: Effects of
metabolic memory on inflammation and fibrosis associated with
diabetic kidney disease: An epigenetic perspective. Clin
Epigenetics. 13:872021. View Article : Google Scholar : PubMed/NCBI
|
|
232
|
Zhao Y, Fan S, Zhu H, Zhao Q, Fang Z, Xu
D, Lin W, Lin L, Hu X, Wu G, et al: Podocyte OTUD5 alleviates
diabetic kidney disease through deubiquitinating TAK1 and reducing
podocyte inflammation and injury. Nat Commun. 15:54412024.
View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Wang B, Dai Z, Gao Q, Liu Y, Gu G and
Zheng H: Spop ameliorates diabetic nephropathy through restraining
NLRP3 inflammasome. Biochem Biophys Res Commun. 594:131–138. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
234
|
Wu W, Huang XR, You Y, Xue L, Wang XJ,
Meng X, Lin X, Shen J, Yu X, Lan HY and Chen H: Latent TGF-β1
protects against diabetic kidney disease via Arkadia/Smad7
signaling. Int J Biol Sci. 17:3583–3594. 2021. View Article : Google Scholar
|
|
235
|
Zhang X, Wang S, Chong N, Chen D, Shu J,
Sun J, Sun Z, Wang R, Wang Q and Xu Y: GDF-15 alleviates diabetic
nephropathy via inhibiting NEDD4L-mediated IKK/NF-κB signalling
pathways. Int Immunopharmacol. 128:1114272024. View Article : Google Scholar
|
|
236
|
Chen K, Chen J, Wang L, Yang J, Xiao F,
Wang X, Yuan J, Wang L and He Y: Parkin ubiquitinates GATA4 and
attenuates the GATA4/GAS1 signaling and detrimental effects on
diabetic nephropathy. FASEB J. 34:8858–8875. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Guo F, Song Y, Wu L, Zhao Y, Ma X, Wang J,
Shao M, Ji H, Huang F, Fan X, et al: SUMO specific peptidase 6
regulates the crosstalk between podocytes and glomerular
endothelial cells in diabetic kidney disease. Biochim Biophys Acta
Mol Basis Dis. 1869:1666852023. View Article : Google Scholar : PubMed/NCBI
|
|
238
|
Huang W, Xu L, Zhou X, Gao C, Yang M, Chen
G, Zhu J, Jiang L, Gan H, Gou F, et al: High glucose induces
activation of NF-κB inflammatory signaling through IκBα sumoylation
in rat mesangial cells. Biochem Biophys Res Commun. 438:568–574.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
239
|
Huang W, Liang Y, Dong J, Zhou L, Gao C,
Jiang C, Chen M, Long Y and Xu Y: SUMO E3 Ligase PIASy mediates
high glucose-induced activation of NF-κB inflammatory signaling in
rat mesangial cells. Mediators Inflamm. 2017:16851942017.
View Article : Google Scholar
|
|
240
|
Yao D, Taguchi T, Matsumura T, Pestell R,
Edelstein D, Giardino I, Suske G, Rabbani N, Thornalley PJ, Sarthy
VP, et al: High glucose increases angiopoietin-2 transcription in
microvascular endothelial cells through methylglyoxal modification
of mSin3A. J Biol Chem. 282:31038–31045. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
241
|
He Y, Xie Y, Zhou T, Li D, Cheng X, Yang
P, Luo C, Liu Y, Guo M, Wan Q, et al: Sodium crotonate alleviates
diabetic kidney disease partially via the histone crotonylation
pathway. Inflammation. 48:254–275. 2025. View Article : Google Scholar
|
|
242
|
Zhang XX, Liu Y, Xu SS, Yang R, Jiang CH,
Zhu LP, Xu YY, Pan K, Zhang J and Yin ZQ: Asiatic acid from
Cyclocarya paliurus regulates the autophagy-lysosome system via
directly inhibiting TGF-β type I receptor and ameliorates diabetic
nephropathy fibrosis. Food Funct. 13:5536–5546. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
243
|
Zhu QQ, Yang XY, Zhang XJ, Yu CJ, Pang QQ,
Huang YW, Wang XJ and Sheng J: EGCG targeting Notch to attenuate
renal fibrosis via inhibition of TGFβ/Smad3 signaling pathway
activation in streptozotocin-induced diabetic mice. Food Funct.
11:9686–9695. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
244
|
Das F, Ghosh-Choudhury N, Maity S,
Kasinath BS and Choudhury GG: Oncoprotein DJ-1 interacts with mTOR
complexes to effect transcription factor Hif1α-dependent expression
of collagen I (α2) during renal fibrosis. J Biol Chem.
298:1022462022. View Article : Google Scholar
|
|
245
|
Wu L, Liu C, Chang DY, Zhan R, Zhao M, Man
Lam S, Shui G, Zhao MH, Zheng L and Chen M: The attenuation of
diabetic nephropathy by annexin A1 via regulation of lipid
metabolism through the AMPK/PPARα/CPT1b pathway. Diabetes.
70:2192–2203. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
246
|
Cho W, Oh H, Choi SW, Abd El-Aty AM,
Birdal O, Jeong JH, Song JH and Jung TW: CTRP4 attenuates apoptosis
and epithelial-mesenchymal transition markers in podocytes through
an AMPK/autophagy-dependent pathway. Biochem Biophys Res Commun.
682:104–110. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
247
|
Han Y, Xiong S, Zhao H, Yang S, Yang M,
Zhu X, Jiang N, Xiong X, Gao P, Wei L, et al: Lipophagy deficiency
exacerbates ectopic lipid accumulation and tubular cells injury in
diabetic nephropathy. Cell Death Dis. 12:10312021. View Article : Google Scholar : PubMed/NCBI
|
|
248
|
Chen J, Chen KH, Wang LM, Luo J, Zheng QY
and He YN: Decoy receptor 2 mediates the apoptosis-resistant
phenotype of senescent renal tubular cells and accelerates renal
fibrosis in diabetic nephropathy. Cell Death Dis. 13:5222022.
View Article : Google Scholar : PubMed/NCBI
|
|
249
|
Li J, Jin S, Barati MT, Rane S, Lin Q, Tan
Y, Cai L and Rane MJ: ERK and p38 MAPK inhibition controls NF-E2
degradation and profibrotic signaling in renal proximal tubule
cells. Life Sci. 287:1200922021. View Article : Google Scholar : PubMed/NCBI
|
|
250
|
Zhang L, Zang CS, Chen B, Wang Y, Xue S
and Wu MY: Renalase regulates renal tubular injury in diabetic
nephropathy via the p38MAPK signaling pathway. FASEB J.
37:e231882023. View Article : Google Scholar : PubMed/NCBI
|
|
251
|
Wen Y, Liu Y, Huang Q, Liu R, Liu J, Zhang
F, Liu S and Jiang Y: Moringa oleifera Lam. seed extract protects
kidney function in rats with diabetic nephropathy by increasing
GSK-3β activity and activating the Nrf2/HO-1 pathway.
Phytomedicine. 95:1538562022. View Article : Google Scholar
|
|
252
|
Shin JH, Kim KM, Jeong JU, Shin JM, Kang
JH, Bang K and Kim JH: Nrf2-heme oxygenase-1 attenuates
high-glucose-induced epithelial-to-mesenchymal transition of renal
tubule cells by inhibiting ROS-Mediated PI3K/Akt/GSK-3β signaling.
J Diabetes Res. 2019:25101052019. View Article : Google Scholar
|
|
253
|
Zeng J, Liang L, Chen R, Li C, Pan L, Wen
M, Lv D, Liu M, Xu Z and Huang H: Fraxin represses NF-κB pathway
via inhibiting the activation of epidermal growth factor receptor
to ameliorate diabetic renal tubulointerstitial fibrosis. Eur J
Pharmacol. 955:1759152023. View Article : Google Scholar
|
|
254
|
Ram C, Gairola S, Verma S, Mugale MN,
Bonam SR, Murty US and Sahu BD: Biochanin A ameliorates nephropathy
in high-fat diet/streptozotocin-induced diabetic rats: Effects on
NF-kB/NLRP3 axis, pyroptosis, and fibrosis. Antioxidants (Basel).
12:10522023. View Article : Google Scholar : PubMed/NCBI
|
|
255
|
Qu X, Zhai B, Liu Y, Chen Y, Xie Z, Wang
Q, Wu Y, Liu Z, Chen J, Mei S, et al: Pyrroloquinoline quinone
ameliorates renal fibrosis in diabetic nephropathy by inhibiting
the pyroptosis pathway in C57BL/6 mice and human kidney 2 cells.
Biomed Pharmacother. 150:1129982022. View Article : Google Scholar : PubMed/NCBI
|
|
256
|
Gu LY, Yun-Sun, Tang HT and Xu ZX:
Huangkui capsule in combination with metformin ameliorates diabetic
nephropathy via the Klotho/TGF-β1/p38MAPK signaling pathway. J
Ethnopharmacol. 281:1135482021. View Article : Google Scholar
|
|
257
|
Duan YR, Chen BP, Chen F, Yang SX, Zhu CY,
Ma YL, Li Y and Shi J: LncRNA lnc-ISG20 promotes renal fibrosis in
diabetic nephropathy by inducing AKT phosphorylation through
miR-486-5p/NFAT5. J Cell Mol Med. 25:4922–4937. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
258
|
Subathra M, Korrapati M, Howell LA, Arthur
JM, Shayman JA, Schnellmann RG and Siskind LJ: Kidney
glycosphingolipids are elevated early in diabetic nephropathy and
mediate hypertrophy of mesangial cells. Am J Physiol Renal Physiol.
309:F204–F215. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
259
|
Hsu YC, Ho C, Shih YH, Ni WC, Li YC, Chang
HC and Lin CL: Knockout of KLF10 ameliorated diabetic renal
fibrosis via downregulation of DKK-1. Molecules. 27:26442022.
View Article : Google Scholar : PubMed/NCBI
|
|
260
|
Chen X, Xiao J, Tao D, Liang Y, Chen S,
Shen L, Li S, Zheng Z, Zeng Y, Luo C, et al: Metadherin
orchestrates PKA and PKM2 to activate β-catenin signaling in
podocytes during proteinuric chronic kidney disease. Transl Res.
266:68–83. 2024. View Article : Google Scholar
|
|
261
|
Chen G, Wang T, Uttarwar L, vanKrieken R,
Li R, Chen X, Gao B, Ghayur A, Margetts P and Krepinsky JC: SREBP-1
is a novel mediator of TGFβ1 signaling in mesangial cells. J Mol
Cell Biol. 6:516–530. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
262
|
Noh H, Oh EY, Seo JY, Yu MR, Kim YO, Ha H
and Lee HB: Histone deacetylase-2 is a key regulator of diabetes-
and transforming growth factor-beta1-induced renal injury. Am J
Physiol Renal Physiol. 297:F729–F739. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
263
|
Zhang L, Chen L, Gao C, Chen E, Lightle
AR, Foulke L, Zhao B, Higgins PJ and Zhang W: Loss of histone H3
K79 methyltransferase Dot1l facilitates kidney fibrosis by
upregulating endothelin 1 through histone deacetylase 2. J Am Soc
Nephrol. 31:337–349. 2020. View Article : Google Scholar
|
|
264
|
Zheng Z, Zhang S, Chen J, Zou M, Yang Y,
Lu W, Ren S, Wang X, Dong W, Zhang Z, et al: The HDAC2/SP1/miR-205
feedback loop contributes to tubular epithelial cell extracellular
matrix production in diabetic kidney disease. Clin Sci (Lond).
136:223–238. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
265
|
Feng J, Bao L, Wang X, Li H, Chen Y, Xiao
W, Li Z, Xie L, Lu W, Jiang H, et al: Low expression of HIV genes
in podocytes accelerates the progression of diabetic kidney disease
in mice. Kidney Int. 99:914–925. 2021. View Article : Google Scholar
|
|
266
|
Wang Y, Zuo B, Wang N, Li S, Liu C and Sun
D: Calcium dobesilate mediates renal interstitial fibrosis and
delay renal peritubular capillary loss through Sirt1/p53 signaling
pathway. Biomed Pharmacother. 132:1107982020. View Article : Google Scholar : PubMed/NCBI
|
|
267
|
Zhong X and Zhang J: RUNX3-activated
apelin signaling inhibits cell proliferation and fibrosis in
diabetic nephropathy by regulation of the SIRT1/FOXO pathway.
Diabetol Metab Syndr. 16:1672024. View Article : Google Scholar : PubMed/NCBI
|
|
268
|
Srivastava SP, Li J, Takagaki Y, Kitada M,
Goodwin JE, Kanasaki K and Koya D: Endothelial SIRT3 regulates
myofibroblast metabolic shifts in diabetic kidneys. iScience.
24:1023902021. View Article : Google Scholar : PubMed/NCBI
|
|
269
|
Wang X, Ji T, Li X, Qu X and Bai S: FOXO3a
protects against kidney injury in type II diabetic nephropathy by
promoting Sirt6 expression and inhibiting Smad3 Acetylation. Oxid
Med Cell Longev. 2021:55657612021. View Article : Google Scholar : PubMed/NCBI
|
|
270
|
Yang G, Jin L, Zheng D, Tang X, Yang J,
Fan L and Xie X: Fucoxanthin alleviates oxidative stress through
Akt/Sirt1/FoxO3α signaling to inhibit HG-induced renal fibrosis in
GMCs. Mar Drugs. 17:7022019. View Article : Google Scholar
|
|
271
|
Jia Y, Reddy MA, Das S, Oh HJ, Abdollahi
M, Yuan H, Zhang E, Lanting L, Wang M and Natarajan R:
Dysregulation of histone H3 lysine 27 trimethylation in
transforming growth factor-β1-induced gene expression in mesangial
cells and diabetic kidney. J Biol Chem. 294:12695–12707. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
272
|
Zhang L, Zhao S and Zhu Y: Long noncoding
RNA growth arrest-specific transcript 5 alleviates renal fibrosis
in diabetic nephropathy by downregulating matrix metalloproteinase
9 through recruitment of enhancer of zeste homolog 2. FASEB J.
34:2703–2714. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
273
|
Fang N and Li P: O-linked
N-acetylglucosaminyltransferase OGT inhibits diabetic nephropathy
by stabilizing histone methyltransferases EZH2 via the HES1/PTEN
axis. Life Sci. 274:1192262021. View Article : Google Scholar : PubMed/NCBI
|
|
274
|
Das F, Bera A, Ghosh-Choudhury N,
Sataranatarajan K, Kamat A, Kasinath BS and Choudhury GG: High
glucose-stimulated enhancer of zeste homolog-2 (EZH2) forces
suppression of deptor to cause glomerular mesangial cell pathology.
Cell Signal. 86:1100722021. View Article : Google Scholar : PubMed/NCBI
|
|
275
|
Chen YX, Zhu SY, Huang C, Xu CY, Fang XD
and Tu WP: LncRNA Dlx6os1 accelerates diabetic nephropathy
progression by epigenetically repressing SOX6 via recruiting EZH2.
Kidney Blood Press Res. 47:177–184. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
276
|
Li X, Li C, Li X, Cui P, Li Q, Guo Q, Han
H, Liu S and Sun G: Involvement of histone lysine methylation in
p21 gene expression in rat kidney in vivo and rat mesangial cells
in vitro under diabetic conditions. J Diabetes Res.
2016:38532422016. View Article : Google Scholar : PubMed/NCBI
|
|
277
|
Goru SK and Gaikwad AB: Novel
reno-protective mechanism of Aspirin involves H2AK119
monoubiquitination and Set7 in preventing type 1 diabetic
nephropathy. Pharmacol Rep. 70:497–502. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
278
|
Yuan H, Reddy MA, Deshpande S, Jia Y, Park
JT, Lanting LL, Jin W, Kato M, Xu ZG, Das S and Natarajan R:
Epigenetic histone modifications involved in profibrotic gene
regulation by 12/15-lipoxygenase and its oxidized lipid products in
diabetic nephropathy. Antioxid Redox Signal. 24:361–375. 2016.
View Article : Google Scholar
|
|
279
|
Maxwell S, Okabe J, Kaipananickal H,
Rodriguez H, Khurana I, Al-Hasani K, Chow BSM, Pitsillou E,
Karagiannis TC, Jandeleit-Dahm K, et al: Set7 methyltransferase and
phenotypic switch in diabetic glomerular endothelial cells. J Am
Soc Nephrol. 35:733–748. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
280
|
Li X, Lu L, Hou W, Wang F, Huang T, Meng Z
and Zhu M: The SETD8/ELK1/bach1 complex regulates
hyperglycaemia-mediated EndMT in diabetic nephropathy. J Transl
Med. 20:1472022. View Article : Google Scholar : PubMed/NCBI
|
|
281
|
Lu L, Zhong Z, Gu J, Nan K, Zhu M and Miao
C: ets1 associates with KMT5A to participate in high
glucose-mediated EndMT via upregulation of PFN2 expression in
diabetic nephropathy. Mol Med. 27:742021. View Article : Google Scholar : PubMed/NCBI
|
|
282
|
Lu L, Li X, Zhong Z, Zhou W, Zhou D, Zhu M
and Miao C: KMT5A downregulation participated in High
Glucose-mediated EndMT via upregulation of ENO1 expression in
diabetic nephropathy. Int J Biol Sci. 17:4093–4107. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
283
|
Chen YY, Peng XF, Liu GY, Liu JS, Sun L,
Liu H, Xiao L and He LY: Protein arginine methyltranferase-1
induces ER stress and epithelial-mesenchymal transition in renal
tubular epithelial cells and contributes to diabetic nephropathy.
Biochim Biophys Acta Mol Basis Dis. 1865:2563–2575. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
284
|
Bai S, Xiong X, Tang B, Ji T, Li X, Qu X
and Li W: hsa-miR-199b-3p prevents the epithelial-mesenchymal
transition and dysfunction of the renal tubule by regulating
E-cadherin through targeting KDM6A in diabetic nephropathy. Oxid
Med Cell Longev. 2021:88141632021. View Article : Google Scholar : PubMed/NCBI
|
|
285
|
Hung PH, Hsu YC, Chen TH, Ho C and Lin CL:
The histone demethylase inhibitor GSK-J4 Is a therapeutic target
for the kidney fibrosis of diabetic kidney disease via DKK1
Modulation. Int J Mol Sci. 23:94072022. View Article : Google Scholar : PubMed/NCBI
|
|
286
|
Shao J, Xu H, Wu X and Xu Y: Epigenetic
activation of CTGF transcription by high glucose in renal tubular
epithelial cells is mediated by myocardin-related transcription
factor A. Cell Tissue Res. 379:549–559. 2020. View Article : Google Scholar
|
|
287
|
Dong L, Yu L and Zhong J: Histone
lysine-specific demethylase 1 induced renal fibrosis via decreasing
sirtuin 3 expression and activating TGF-β1/Smad3 pathway in
diabetic nephropathy. Diabetol Metab Syndr. 14:22022. View Article : Google Scholar
|
|
288
|
Morschhauser F, Tilly H, Chaidos A, McKay
P, Phillips T, Assouline S, Batlevi CL, Campbell P, Ribrag V, Damaj
GL, et al: Tazemetostat for patients with relapsed or refractory
follicular lymphoma: An open-label, single-arm, multicentre, phase
2 trial. Lancet Oncol. 21:1433–1442. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
289
|
Huang S, Wang Z, Zhou J, Huang J, Zhou L,
Luo J, Wan YY, Long H and Zhu B: EZH2 inhibitor GSK126 suppresses
antitumor immunity by driving production of myeloid-derived
suppressor cells. Cancer Res. 79:2009–2020. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
290
|
Chen Y, Dai R, Cheng M, Wang W, Liu C, Cao
Z, Ge Y, Wang Y and Zhang L: Status and role of the
ubiquitin-proteasome system in renal fibrosis. Biomed Pharmacother.
178:1172102024. View Article : Google Scholar : PubMed/NCBI
|
|
291
|
Lv K, Li Q, Jiang N and Chen Q: Role of
TRIM29 in disease: What is and is not known. Int Immunopharmacol.
147:1139832025. View Article : Google Scholar : PubMed/NCBI
|
|
292
|
Chen Q, Gao C, Wang M, Fei X and Zhao N:
TRIM18-regulated STAT3 signaling pathway via PTP1B promotes renal
epithelial-mesenchymal transition, inflammation, and fibrosis in
diabetic kidney disease. Front Physiol. 12:7095062021. View Article : Google Scholar : PubMed/NCBI
|
|
293
|
Li Y, Ren D, Shen Y, Zheng X and Xu G:
Altered DNA methylation of TRIM13 in diabetic nephropathy
suppresses mesangial collagen synthesis by promoting ubiquitination
of CHOP. EBioMedicine. 51:1025822020. View Article : Google Scholar : PubMed/NCBI
|
|
294
|
Chen Z, Sun X, Chen Q, Lan T, Huang K,
Xiao H, Lin Z, Yang Y, Liu P and Huang H: Connexin32 ameliorates
renal fibrosis in diabetic mice by promoting K48-linked NADPH
oxidase 4 polyubiquitination and degradation. Br J Pharmacol.
177:145–160. 2020. View Article : Google Scholar
|
|
295
|
Lin Z, Li S, Xiao H, Xu Z, Li C, Zeng J,
Wang S, Liu Z and Huang H: The degradation of TGR5 mediated by
Smurf1 contributes to diabetic nephropathy. Cell Rep.
42:1128512023. View Article : Google Scholar : PubMed/NCBI
|
|
296
|
Kim D, Nam GY, Seo E and Jun HS:
Inhibition of ChREBP ubiquitination via the ROS/Akt-dependent
downregulation of Smurf2 contributes to lysophosphatidic
acid-induced fibrosis in renal mesangial cells. J Biomed Sci.
29:312022. View Article : Google Scholar : PubMed/NCBI
|
|
297
|
Yang Y, Xiao H, Lin Z, Chen R, Li S, Li C,
Sun X, Hei Z, Gong W and Huang H: The ubiquitination of CKIP-1
mediated by Src aggravates diabetic renal fibrosis (original
article). Biochem Pharmacol. 206:1153392022. View Article : Google Scholar : PubMed/NCBI
|
|
298
|
Zhao X, He X, Wei W and Huang K: USP22
aggravated diabetic renal tubulointerstitial fibrosis progression
through deubiquitinating and stabilizing Snail1. Eur J Pharmacol.
947:1756712023. View Article : Google Scholar : PubMed/NCBI
|
|
299
|
Huang K and Zhao X: USP9X prevents
AGEs-induced upregulation of FN and TGF-β1 through activating
Nrf2-ARE pathway in rat glomerular mesangial cells. Exp Cell Res.
393:1121002020. View Article : Google Scholar
|
|
300
|
Liu Z, Nan P, Gong Y, Tian L, Zheng Y and
Wu Z: Endoplasmic reticulum stress-triggered ferroptosis via the
XBP1-Hrd1-Nrf2 pathway induces EMT progression in diabetic
nephropathy. Biomed Pharmacother. 164:1148972023. View Article : Google Scholar : PubMed/NCBI
|
|
301
|
Sun XH, Xiao HM, Zhang M, Lin ZY, Yang Y,
Chen R, Liu PQ, Huang KP and Huang HQ: USP9X deubiquitinates
connexin43 to prevent high glucose-induced
epithelial-to-mesenchymal transition in NRK-52E cells. Biochem
Pharmacol. 188:1145622021. View Article : Google Scholar : PubMed/NCBI
|
|
302
|
Chang L, Wang Q, Ju J, Li Y, Cai Q, Hao L
and Zhou Y: Magnoflorine ameliorates inflammation and fibrosis in
rats with diabetic nephropathy by mediating the stability of
lysine-specific demethylase 3A. Front Physiol. 11:5804062020.
View Article : Google Scholar
|
|
303
|
Wang Y, Mao Y, Zhang X, Liu H, Peng W,
Liang L, Shi M, Xiao Y, Zhang Y, Zhang F, et al: TAK1 may promote
the development of diabetic nephropathy by reducing the stability
of SnoN protein. Life Sci. 228:1–10. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
304
|
Peng W, Zhou X, Xu T, Mao Y, Zhang X, Liu
H, Liang L, Liu L, Liu L, Xiao Y, et al: BMP-7 ameliorates partial
epithelial-mesenchymal transition by restoring SnoN protein level
via Smad1/5 pathway in diabetic kidney disease. Cell Death Dis.
13:2542022. View Article : Google Scholar : PubMed/NCBI
|
|
305
|
Gu C, Gao F, Zhang S, Kang L, Zhang W,
Feng X, Liu J, Tian Y, Wei Q, Du Y, et al: Role of SUMOylation of
STAT1 in tubular epithelial-mesenchymal transition induced by high
glucose. Mol Med Rep. 27:422023. View Article : Google Scholar
|
|
306
|
Zhou X, Gao C, Huang W, Yang M, Chen G,
Jiang L, Gou F, Feng H, Ai N and Xu Y: High glucose induces
sumoylation of Smad4 via SUMO2/3 in mesangial cells. Biomed Res
Int. 2014:7826252014. View Article : Google Scholar : PubMed/NCBI
|
|
307
|
Zhou XQ, Huang W, Xu Y and Yang MJ: High
glucose induces sumoylation of Smad4 via SUMO2/3 in glomerular
mesangial cells. Sichuan Da Xue Xue Bao Yi Xue Ban. 45:380–385.
2014.In Chinese. PubMed/NCBI
|
|
308
|
Park MJ, Kim DI, Lim SK, Choi JH, Han HJ,
Yoon KC and Park SH: High glucose-induced O-GlcNAcylated
carbohydrate response element-binding protein (ChREBP) mediates
mesangial cell lipogenesis and fibrosis: The possible role in the
development of diabetic nephropathy. J Biol Chem. 289:13519–13530.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
309
|
Goldberg H, Whiteside C and Fantus IG:
O-linked β-N-acetylglucosamine supports p38 MAPK activation by high
glucose in glomerular mesangial cells. Am J Physiol Endocrinol
Metab. 301:E713–E726. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
310
|
Aluksanasuwan S, Plumworasawat S, Malaitad
T, Chaiyarit S and Thongboonkerd V: High glucose induces
phosphorylation and oxidation of mitochondrial proteins in renal
tubular cells: A proteomics approach. Sci Rep. 10:58432020.
View Article : Google Scholar : PubMed/NCBI
|
|
311
|
Veron D, Aggarwal PK, Li Q, Moeckel G,
Kashgarian M and Tufro A: Podocyte VEGF-A knockdown induces diffuse
glomerulosclerosis in diabetic and in eNOS knockout mice. Front
Pharmacol. 12:7888862022. View Article : Google Scholar : PubMed/NCBI
|
|
312
|
Veron D, Aggarwal PK, Velazquez H,
Kashgarian M, Moeckel G and Tufro A: Podocyte-specific VEGF-a gain
of function induces nodular glomerulosclerosis in eNOS null mice. J
Am Soc Nephrol. 25:1814–1824. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
313
|
Zhang X, Chen J, Lin R, Huang Y, Wang Z,
Xu S, Wang L, Chen F, Zhang J, Pan K and Yin Z: Lactate drives
epithelial-mesenchymal transition in diabetic kidney disease via
the H3K14la/KLF5 pathway. Redox Biol. 75:1032462024. View Article : Google Scholar : PubMed/NCBI
|
|
314
|
Hong J, Xu H, Yu L, Yu Z, Chen X, Meng Z,
Zhu J, Li J and Zhu M: AARS1-mediated lactylation of H3K18 and
STAT1 promotes ferroptosis in diabetic nephropathy. Cell Death
Differ. Sep 23–2025.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
315
|
Luo W, Yu Y, Wang H, Liu K, Wang Y, Huang
M, Xuan C, Li Y and Qi J: Up-regulation of MMP-2 by histone H3K9
β-hydroxybutyrylation to antagonize glomerulosclerosis in diabetic
rat. Acta Diabetol. 57:1501–1509. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
316
|
Lyu LX, Hu MY, Lei YQ, Pan SK, Liu DW,
Zhou SJ and Liu ZS: The mechanism of β-hydroxybutyrylation in G6PDX
regulated by β-hydroxybutyric acid on alleviating podocyte injury
induced by high glucose. Zhonghua Yi Xue Za Zhi. 105:900–906.
2025.In Chinese. PubMed/NCBI
|
|
317
|
Akimoto Y, Yan K, Miura Y, Tsumoto H, Toda
T, Fukutomi T, Sugahara D, Kudo A, Arai T, Chiba Y, et al:
O-GlcNAcylation and phosphorylation of β-actin Ser199 in diabetic
nephropathy. Am J Physiol Renal Physiol. 317:F1359–F1374. 2019.
View Article : Google Scholar
|
|
318
|
Goru SK, Kadakol A, Pandey A, Malek V,
Sharma N and Gaikwad AB: Histone H2AK119 and H2BK120
mono-ubiquitination modulate SET7/9 and SUV39H1 in type 1
diabetes-induced renal fibrosis. Biochem J. 473:3937–3949. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
319
|
Li XQ, Jin B, Liu SX, Zhu Y, Li N, Zhang
QY, Wan C, Feng Y, Xing YX, Ma KL, et al: Neddylation of RhoA
impairs its protein degradation and promotes renal interstitial
fibrosis progression in diabetic nephropathy. Acta Pharmacol Sin.
46:1692–1705. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
320
|
Qu P, Li L, Jin Q, Liu D, Qiao Y, Zhang Y,
Sun Q, Ran S, Li Z, Liu T and Peng L: Histone methylation
modification and diabetic kidney disease: Potential molecular
mechanisms and therapeutic approaches (Review). Int J Mol Med.
54:1042024. View Article : Google Scholar : PubMed/NCBI
|
|
321
|
Hu H, Ji R, Hao Y, Liu Z, Yang J, Cao Y
and Yang Q: XIAP-ULK1-mediated mitophagy modulates carnitine
metabolism to mitigate diabetic kidney disease. Autophagy.
22:207–228. 2026. View Article : Google Scholar
|
|
322
|
Memarian E, t Hart LM, Slieker RC, Lemmers
RFL, van der Heijden AA, Rutters F, Nijpels G, Schoep E, Lieverse
AG, Sijbrands EJG, et al: Plasma protein N-glycosylation is
associated with cardiovascular disease, nephropathy, and
retinopathy in type 2 diabetes. BMJ Open Diabetes Res Care.
9:e0023452021. View Article : Google Scholar : PubMed/NCBI
|
|
323
|
Azushima K, Kovalik JP, Yamaji T, Ching J,
Chng TW, Guo J, Liu JJ, Nguyen M, Sakban RB, George SE, et al:
Abnormal lactate metabolism is linked to albuminuria and kidney
injury in diabetic nephropathy. Kidney Int. 104:1135–1149. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
324
|
Chen H, Du P, Jiang T, Li Y, Li Y, Liu Y,
Yang B, Kang J, Duan J, Ma Y, et al: Identification of potential
biomarkers for diabetic nephropathy via UPLC-MS/MS-based
metabolomics. Front Endocrinol (Lausanne). 16:15816912025.
View Article : Google Scholar : PubMed/NCBI
|
|
325
|
Zhang JY, Wu J, Chen ZH, Liu SY, Li P and
Chen DQ: Acetylation in renal physiology and pathophysiology. Front
Pharmacol. 16:16601092025. View Article : Google Scholar : PubMed/NCBI
|
|
326
|
Siddhi J, Sherkhane B, Kalavala AK, Arruri
V, Velayutham R and Kumar A: Melatonin prevents diabetes-induced
nephropathy by modulating the AMPK/SIRT1 axis: Focus on autophagy
and mitochondrial dysfunction. Cell Biol Int. 46:2142–2157. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
327
|
Gu W, Wang X, Zhao H, Geng J, Li X, Zheng
K, Guan Y, Hou X, Wang C and Song G: Resveratrol ameliorates
diabetic kidney injury by reducing lipotoxicity and modulates
expression of components of the junctional adhesion
molecule-like/sirtuin 1 lipid metabolism pathway. Eur J Pharmacol.
918:1747762022. View Article : Google Scholar : PubMed/NCBI
|
|
328
|
Ren H, Shao Y, Wu C, Ma X, Lv C and Wang
Q: Metformin alleviates oxidative stress and enhances autophagy in
diabetic kidney disease via AMPK/SIRT1-FoxO1 pathway. Mol Cell
Endocrinol. 500:1106282020. View Article : Google Scholar
|
|
329
|
Qi B, Chen Y, Chai S, Lu X and Kang L:
O-linked β-N-acetylglucosamine (O-GlcNAc) modification: Emerging
pathogenesis and a therapeutic target of diabetic nephropathy.
Diabet Med. 42:e154362025. View Article : Google Scholar
|
|
330
|
Fontecha-Barriuso M, Martin-Sanchez D,
Ruiz-Andres O, Poveda J, Sanchez-Niño MD, Valiño-Rivas L,
Ruiz-Ortega M, Ortiz A and Sanz AB: Targeting epigenetic DNA and
histone modifications to treat kidney disease. Nephrol Dial
Transplant. 33:1875–1886. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
331
|
Matsui S, Yamamoto T, Takabatake Y,
Takahashi A, Namba-Hamano T, Matsuda J, Minami S, Sakai S, Yonishi
H, Nakamura J, et al: Defective autophagy and AMPK inactivation
drive ferroptosis in diabetic kidney disease. Diabetologia. Nov
28–2025.Epub ahead of print. PubMed/NCBI
|
|
332
|
Zhou X, Zang X, Ponnusamy M, Masucci MV,
Tolbert E, Gong R, Zhao TC, Liu N, Bayliss G, Dworkin LD and Zhuang
S: Enhancer of zeste homolog 2 inhibition attenuates renal fibrosis
by maintaining Smad7 and phosphatase and tensin homolog expression.
J Am Soc Nephrol. 27:2092–2108. 2016. View Article : Google Scholar
|
|
333
|
Cao J, Chen G, Qiu L, Zhang L, Jiang M,
Cheng Y, Zhang Q, Liu L, Li P, Shuang Y, et al: Efficacy and safety
of tazemetostat, an EZH2 inhibitor, in Chinese patients with
relapsed/refractory follicular lymphoma: A multicentre, single-arm,
phase 2 study. EClinicalMedicine. 87:1033992025. View Article : Google Scholar : PubMed/NCBI
|
|
334
|
Zinzani PL, Izutsu K, Mehta-Shah N, Barta
SK, Ishitsuka K, Córdoba R, Kusumoto S, Bachy E, Cwynarski K,
Gritti G, et al: Valemetostat for patients with relapsed or
refractory peripheral T-cell lymphoma (VALENTINE-PTCL01): A
multicentre, open-label, single-arm, phase 2 study. Lancet Oncol.
25:1602–1613. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
335
|
Maruyama D, Jacobsen E, Porcu P, Allen P,
Ishitsuka K, Kusumoto S, Narita T, Tobinai K, Foss F, Tsukasaki K,
et al: Valemetostat monotherapy in patients with relapsed or
refractory non-Hodgkin lymphoma: A first-in-human, multicentre,
open-label, single-arm, phase 1 study. Lancet Oncol. 25:1589–1601.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
336
|
Rong QX, Chen MT, Yang W, Huang RQ, Shu
DT, Zhang Y, Xue C, Cai YC, An X, Li HF and Shi YX: Chidamide (a
Histone Deacetylase Inhibitor) Plus abiraterone in metastatic
castration-resistant prostate cancer (mCRPC): A phase Ib trial.
MedComm (2020). 6:e704702025. View Article : Google Scholar : PubMed/NCBI
|
|
337
|
Zhang Y, Chen X, Yuan L, Zhang Y, Wu J,
Guo N, Chen X and Liu J: Down-regulation of IRAK1 attenuates
podocyte apoptosis in diabetic nephropathy through PI3K/Akt
signaling pathway. Biochem Biophys Res Commun. 506:529–535. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
338
|
Chen J, Wang X, He Q, Yang HC, Fogo AB and
Harris RC: Inhibition of transcriptional coactivator YAP Impairs
the expression and function of transcription factor WT1 in diabetic
podocyte injury. Kidney Int. 105:1200–1211. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
339
|
Zhan M, Usman I, Yu J, Ruan L, Bian X,
Yang J, Yang S, Sun L and Kanwar YS: Perturbations in mitochondrial
dynamics by p66Shc lead to renal tubular oxidative injury in human
diabetic nephropathy. Clin Sci (Lond). 132:1297–1314. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
340
|
Ma Y, Yan R, Wan Q, Lv B, Yang Y, Lv T and
Xin W: Inhibitor of growth 2 regulates the high glucose-induced
cell cycle arrest and epithelial-to-mesenchymal transition in renal
proximal tubular cells. J Physiol Biochem. 76:373–382. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
341
|
Du L, Qian X, Li Y, Li XZ, He LL, Xu L,
Liu YQ, Li CC, Ma P, Shu FL, et al: Sirt1 inhibits renal tubular
cell epithelial-mesenchymal transition through YY1 deacetylation in
diabetic nephropathy. Acta Pharmacol Sin. 42:242–251. 2021.
View Article : Google Scholar
|
|
342
|
Xu H, Wu X, Qin H, Tian W, Chen J, Sun L,
Fang M and Xu Y: Myocardin-related transcription factor a
epigenetically regulates renal fibrosis in diabetic nephropathy. J
Am Soc Nephrol. 26:1648–1660. 2015. View Article : Google Scholar
|
|
343
|
Li Y, Hu Q, Li C, Liang K, Xiang Y, Hsiao
H, Nguyen TK, Park PK, Egranov SD, Ambati CR, et al: PTEN-induced
partial epithelial-mesenchymal transition drives diabetic kidney
disease. J Clin Invest. 129:1129–1151. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
344
|
Pontrelli P, Conserva F, Menghini R,
Rossini M, Stasi A, Divella C, Casagrande V, Cinefra C, Barozzino
M, Simone S, et al: Inhibition of lysine 63 ubiquitination prevents
the progression of renal fibrosis in diabetic DBA/2J mice. Int J
Mol Sci. 22:51942021. View Article : Google Scholar : PubMed/NCBI
|