Diabetic kidney disease (DKD), a prevalent form of
secondary nephropathy affecting 30-40% of the global diabetic
population, is clinically characterized by persistent
microalbuminuria accompanied by progressive deterioration of
glomerular filtration function (1,2).
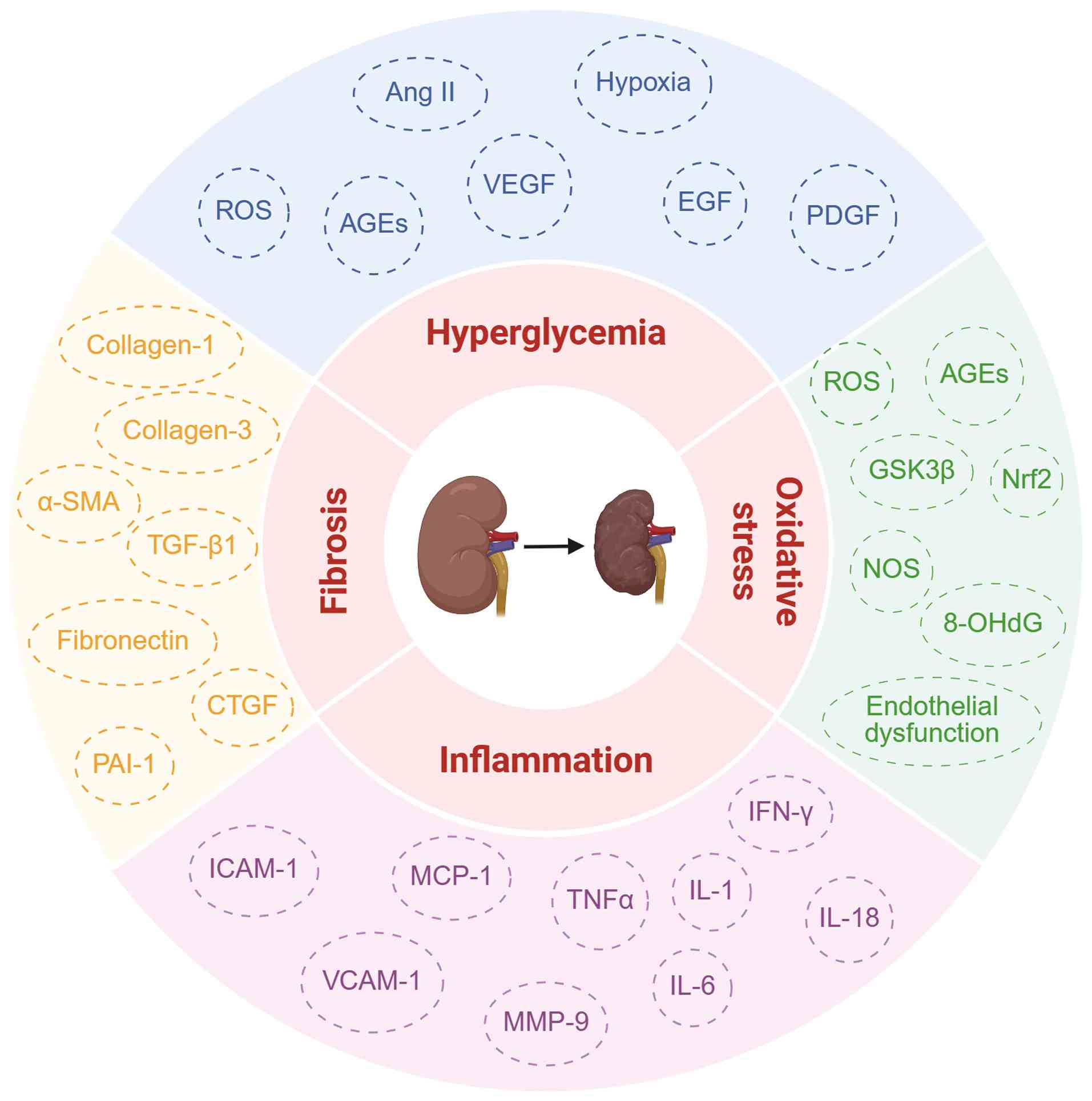
The pathophysiological alterations of DKD primarily manifest as
excessive extracellular matrix (ECM) accumulation, sustained
activation of the inflammatory microenvironment, redox
imbalance-mediated oxidative stress and mitochondrial dysfunction
(3) (Fig. 1). Hyperglycemia-induced metabolic
derangements trigger aberrant regulation of key signaling cascades,
such as the AMP-activated protein kinase (AMPK), PI3K/Akt and MAPK
pathways, which collectively orchestrate the molecular mechanisms
driving disease onset and progression (4).
Post-translational modifications (PTMs) are key
epigenetic mechanisms that govern cellular biological processes,
including phosphorylation, acetylation, methylation,
ubiquitination, small ubiquitin-like modifier (SUMO)ylation,
glycosylation, redox modification. Novel PTMs (such as lactylation
and neddylation) have also been identified with the application of
mass spectrometry-proteomics (5). PTMs refer to covalent alterations
occurring in proteins or peptides. These modifications enhance the
functional diversity of the proteome, which is achieved via the
covalent attachment or detachment of regulatory subunits, or
through the degradation of target proteins in an enzyme-dependent
or -independent manner (6).
These processes increase the intricacy of protein modulation by
affecting protein status, subcellular localization, transportation
and communication with other molecules. Consequently, understanding
of the PTM-mediated mechanisms in DKD is key for the discovery of
innovative targets for therapy development. The present review
aimed to summarize the effect of PTMs on cell death, oxidative
stress, mitochondrial dysfunction, inflammation and fibrosis in the
development and progression of DKD.
Phosphorylation is an enzymatic reaction of protein
kinases that mediates the linkage between specific amino acid
residues of target proteins and phosphate groups in ATP (7). Protein kinases and phosphatases
catalyze the transfer or removal, respectively, of phosphate groups
to their substrates, dynamically modulating the function of
proteins through allosteric regulation to activate enzyme activity
(such as Ser/Thr and Tyr residues) or via the engagement of
interaction domains to trigger signal transduction (such as Tyr
residues) (6). Protein
phosphorylation is implicated in numerous processes, including gene
transcription, cell cycle progression, activation of cell signaling
and cell apoptosis. Previous studies have shown that disruption of
phosphorylation contributes to a range of diseases, including
cancer, cardiovascular disease, respiratory illness, immune
imbalance, metabolic disorders and nervous system disease (8-10).
Acetylation is a type of histone modification that
is linked to the pathogenesis of diabetes. Histone acetylation is a
process whereby acetyl groups are transferred to lysine residues,
which alters the charge of histone proteins, facilitating the
binding of transcription factors to gene promoters and promoting
gene expression (11). The
dynamic balance of histone acetylation/deacetylation is regulated
by the coordinated action of histone acetyltransferase (HAT) and
histone deacetylase (HDAC). HATs include GCN5, p300/CBP-associated
factor (PCAF) and the MYST family. At present, four classes of
HDACs have been identified: i) Class I (HDAC1-3 and 8); ii) class
II (HDAC4-7, 9 and 10); iii) class III, sirtuin family (SIRT1-7)
and iv) class IV (HDAC11) (12).
Moreover, acetylation of non-histone proteins has been reported to
serve a key role in multiple cellular processes, including
regulation of gene expression, DNA damage repair, cell cycle
modulation, protein folding, interactions between proteins,
autophagy, signal transduction and cell metabolism (13). Consequently, abnormal acetylation
is involved in the pathogenesis of various diseases, including
DKD.
Protein methylation encompasses two primary
categories: i) Histone methylation and ii) non-histone methylation.
Histone methylation and demethylation modifications typically occur
on the amino terminal lysine or arginine residues of histones, and
are written by histone methyltransferases [such as enhancer of
zeste homologue (EZH)2, G9a and SET7/9) and erased by histone
demethylases [HDMs; such as lysine-specific demethylase (KDM)6A]
(14). Histone methylation
exerts different regulatory effects on genes due to the different
locations of the methylated histone residues. The methylation of
H3K4, H3K36 and H3K79 is typically associated with gene
transcriptional activation, whereas methylation of H3K9, H3K27 and
H4K20 is associated with gene transcriptional suppression (14,15). Apart from methylating histones,
methylation also occurs on non-histone proteins, which is
associated with the pathogenesis of DKD (16). However, the role of non-histone
methylation in DKD requires further exploration.
PTMs exert regulatory effects on all aspects of
protein function, including alteration of the proteolytic stability
of proteins. Ubiquitination, recognized as the primary PTM involved
in governing protein stability, is a reversible process (17). It can either activate or
inactivate proteins and modulate protein-protein interactions via
the ubiquitin-proteasomal system (UPS) (18). Ubiquitination relies on the
conjugation of ubiquitin, mediated by ubiquitin-activating E1
enzyme, ubiquitin-conjugating E2 proteins and ubiquitin-protein E3
ligase, whereas deubiquitinases [such as ubiquitin-specific
protease (USP)14/22 and OTU domain-containing protein 5 (OTUD5)]
remove ubiquitin and counter this process (19,20). As well as proteasomal
degradation, ubiquitination also directs substrate proteins to
participate in cell signaling pathways, such as NF-κB, TGF-β and
Wnt/β-catenin pathways. Ubiquitination is associated with numerous
processes, such as cell proliferation, DNA repair, gene
transcription, protein degradation, apoptosis and signal
transduction (21,22).
SUMOylation is a highly dynamic enzymatic cascade
similar to ubiquitination involved in multiple cellular processes,
including nuclear-cytosolic transport, transcriptional modulation,
apoptosis, regulation of protein stability, cell stress response
and cell cycle regulation (23).
It is a reversible modification associated with the covalent
attachment of SUMO1-5 to substrate proteins (24). The precursor SUMO is processed by
sentrin/SUMO-specific proteases (SENPs) to generate mature SUMOs,
which are conjugated to the target proteins through an enzymatic
cascade catalyzed by SUMO-activating E1 enzyme, SUMO-conjugating E2
enzyme and SUMO E3 ligase (6,23). SUMOylation is reversed by SENPs,
which recognize and remove the SUMO conjugate from the conjugated
proteins (6,23,24).
Hyperglycemia contributes to the pathogenesis of DKD
by disrupting the equilibrium of enzyme-driven glycosylation and
non-enzymatic glycation (NEG). Glycosylation, in which
carbohydrates are attached to specific amino acids, comprises two
primary types: i) N-linked and ii) O-linked protein glycosylation,
including O-linked N-acetylglucosamine glycosylation
(O-GlcNAcylation) (25).
N-linked glycosylation predominantly occurs in the endoplasmic
reticulum (ER) and Golgi apparatus. Through linkage to asparagine
residues of proteins via N-acetylglucosamine, glycans exerts key
roles in protein folding, stability and transportation (26). Adequate N-glycosylation is
crucial for the correct membrane localization of various key
proteins, including nephrin and podocin, enabling the interactions
of these proteins with other molecules, and further maintains the
normal function of the glomerular filtration barrier (27). O-GlcNAcylation is the reversable
addition of the O-GlcNAc moiety of uridine-diphosphate GlcNAc
(UDP-GlcNAc) to serine or threonine residues of proteins
covalently, which is catalyzed by O-GlcNAc transferase (OGT) and
hydrolyzed by O-GlcNAcase (OGA) (28,29). Disruption of this dynamic
equilibrium impacts multiple cell and metabolic processes, such as
transcriptional regulation, ferroptosis and autophagy (30). NEG is an irreversible conjugation
process that reduces sugars onto a free amino group of proteins,
resulting in the formation of initial Schiff's base, an Amadori
product and advanced glycation end products (AGEs) (31). As the role of NEG in DKD has been
summarized by Ma et al (32) and Parwani and Mandal et al
(33), the present review
focused on the role of classical enzyme-driven glycosylation in
DKD.
Redox homeostasis is key for the normal regulation
of cellular processes. Excessive generation of reactive oxygen
species (ROS) and reactive nitrogen species leads to numerous
pathologies, including diabetes complications, cancer, and
cardiovascular and neurodegenerative disease (34). Redox modifications can be divided
into reversible (S-nitrosylation, S-sulfination, S-glutathione,
S-thiothiolate, intermolecular and intramolecular disulfide bonds
and S-acylation) and irreversible (S-sulfoxide and S-sulfination)
modifications, predominantly targeting the thiol groups of cysteine
and methionine residues (6,34,35). Redox modifications occur in
numerous processes, including transcriptional regulation, protein
folding and stability, cell metabolism, antioxidant homeostasis and
signal transduction (36,37).
Succinylation refers to the covalent conjugation of
a succinyl group to the lysine residue of a substrate protein
mediated by a succinyl group donor (11). Lysine succinylation regulates
mitochondrial function, gene transcription, DNA repair and tumor
formation (44).
Histone lysine crotonylation (Kcr) was first
identified as a PTM in 2011, and is primarily associated with
active transcription (45). Kcr
is enzymatically regulated by the dynamic balance between
crotonyltransferases (such as histone crotonyltransferase, p300/CBP
and PCAF) and decrotonylases (such as HDAC1/2/3/8 and SIRT1-3),
similar to writers and erasers in histone acetylation (11,46). Abundant evidence has indicated
that crotonylation is involved in multiple cell processes including
chromatin remodeling, cell cycle progression and cell metabolism
(47,48).
Kbhb is a type of histone lysine acylation first
identified in 2016, which uses β-hydroxybutyrate (BHB) as the
substrate and has a broad impact on cell functions, including the
modulation of gene expression and cell response to starvation
(6). BHB mediates the Kbhb of
histone lysine within the promoters of certain
starvation-associated genes (such as PPAR and insulin signaling
pathways), facilitating rapid adjustment and adaption in response
to metabolic fluctuations (49).
It is facilitated by p300/CBP, and removed by SIRT13 and HDAC1-3
(11).
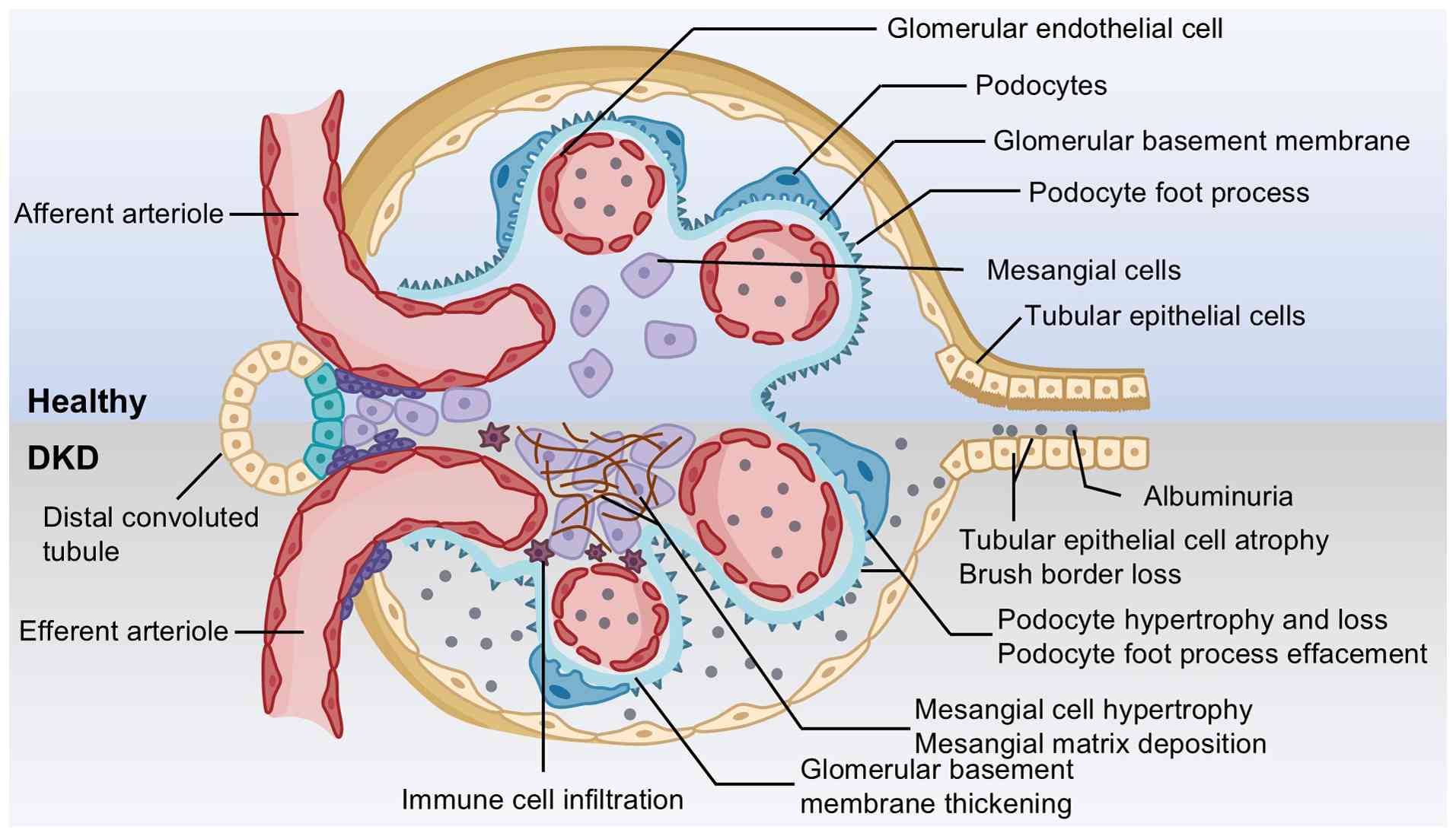
The pathophysiology of DKD is characterized by
perturbations in renal hemodynamics, excessive oxidative stress and
persistent inflammation (50). A
number of cellular processes contribute to the initiation and
development of DKD, among which oxidative stress is widely
acknowledged as a key driver of cellular injury induced by
hyperglycemia (51,52). Oxidative stress triggers renal
cell apoptosis and the release of proinflammatory factors, and
activates the signaling pathways implicated in renal fibrosis,
culminating in renal fibrosis and the deterioration of glomerular
filtration function (53).
Additionally, studies have indicated that excessive ROS production
induces mitochondrial dysfunction and contributes to the activation
of proinflammatory factors and the initiation of
epithelial-mesenchymal transition (EMT) in DKD (54,55). Collectively, these pathological
factors elicit histological changes to glomeruli and renal tubules,
thereby promoting the formation of DKD (Fig. 2).
Phosphorylation of proteins is a key PTM for
balancing glucose homeostasis, and relies on signaling cascades
mediated by protein kinases and phosphatases. Unc-51-like kinase 1
(ULK1), a key serine/threonine protein kinase in autophagy, is
modulated by AMPK and mTOR (56). The activated AMPK pathway and
activated autophagy protect numerous types of renal cells in
response to high glucose by directly activating ULK1
phosphorylation (57-59). Augmented phosphorylation levels
of PI3K/Akt/mTOR induce renal apoptosis, glomerular injury and
interstitial fibrosis by inhibiting ULK1 phosphorylation (60,61). The MAPK family is associated with
the progression of DKD, and consists of p38 MAPK, JNK and ERK,
which are involved in apoptosis of renal intrinsic cells in DKD
(62-66).
Glycogen synthase kinase 3β (GSK3β) is a highly
conserved, redox-sensitive serine/threonine protein kinase.
Exposure to the diabetic condition leads to elevated expression of
phosphorylated (p)-GSK3β at tyrosine 216 (p-GSK3βY216),
but a decrease in the expression of inhibitory p-GSK3β at serine 9
(67,68). PI3K/Akt signaling inactivates
GSK3β by phosphorylating GSK3β at serine 9 (69). Nrf2 is a key regulator in redox
balance that can translocate into the nucleus and initiate the
transcription of antioxidant genes, such as heme oxygenase-1 (HO-1)
(70). Activated GSK3β directly
phosphorylates Nrf2 and leads to its nuclear exclusion (71). Additionally, GSK3β phosphorylates
Nrf2 at the Tyr568 site and promotes the translocation of Nrf2 out
of the nucleus by increasing Fyn phosphorylation and its nuclear
retention (71). Overactivated
GSK3β phosphorylates Nrf2, thereby facilitating podocyte apoptosis
and senescence in DKD (67,72). In addition, enhanced
p-GSK3βY216 promotes phosphorylation and degradation of
β-catenin, mediating podocyte apoptosis under diabetic conditions
(73).
In addition, alteration of phosphorylation states of
specific proteins participates in the pathogenesis of DKD.
Activation of the JAK/STAT signaling pathway can affect cell
senescence, autophagy, apoptosis and ferroptosis in DKD (74-76). The phosphorylation state of STAT3
is also modulated by diverse regulators. In renal tubular
epithelial cells (RTECs), sulfhydrated SIRT1 induces
dephosphorylation and deacetylation of STAT3, thereby decreasing
high glucose-induced cell apoptosis, oxidative stress, inflammation
response and EMT progression (77). Furthermore, increasing numbers of
phosphorylated proteins have been shown to participate in the
damage of renal cells in DKD (Table
I) (78-87). This research contributes to a
more comprehensive understanding of the pathogenesis of DKD and
provides a theoretical basis for considering PTMs as a therapeutic
target for DKD.
HAT p300/CBP is a key co-transcriptional activator
that regulates the expression of numerous prooxidant,
proinflammatory and profibrotic genes by mediating H3K27
acetylation, and is associated with mediation of
diabetes-accelerated renal damage (88). Numerous targets of p300/CBP
participate in the regulation of DKD development, including
inducible nitric oxide synthase (NOS) and polo-like kinase 1 (PLK1)
(89,90). ACSS2 epigenetically activates
Raptor expression by histone H3K9 acetylation, promoting activation
of the mTOR complex (mTORC)1 pathway and inhibiting podocyte
autophagy (91). Upregulated
acetylation levels of Beclin1 are associated with exacerbation of
podocyte injury in diabetic conditions and a mutation at K414R
suppresses hyperactivated autophagy, thus ameliorating podocyte
impairment, highlighting the key role of PTMs in the pathological
process of DKD (92).
Multiple HDACs regulate the activities of numerous
proteins involved in cell death during the progression of DKD.
Specifically, inhibition of transcription factor EB (TFEB)
deacetylation induced by HDAC6 promotes TFEB activation and
alleviates RTEC damage (93,94). Moreover, HDAC6-mediated
deacetylation of α-tubulin suppresses autophagy and enhances
motility of podocytes in DKD (95). HDAC4 suppresses podocyte
autophagy and promotes podocyte apoptosis by acetylating STAT1
under diabetic conditions (96).
SIRT1 activation can deacetylate forkhead box (Fox)O3a or regulate
the NF-κB signaling pathway, while subsequently suppressing renal
cell apoptosis (97-99). SIRT3 upregulation also
antagonizes hyperglycemia-mediated tubular apoptosis by regulating
the accumulation of ROS and modulating the ROS-sensitive Akt/FoxO
signaling pathway (100). In
parallel, SIRT6 inhibits the transcription of Notch1/4 by
deacetylating histone H3K9, and protects podocytes against
apoptosis and inflammation by enhancing autophagic flux (101). These results indicate that
HDACs serve as potential therapeutic targets in DKD.
Recruitment of EZH2, a methyltransferase that
produces histone H3 lysine 27 tri-methylation (H3K27me3), at the
FoxA1 promoter region promotes podocyte damage and apoptosis in the
early stage of DKD (102,103). Inhibition of EZH2 by GSK126
attenuates podocyte injury and hyperglycemia-induced ferroptosis
(104,105). By contrast, EZH2/H3K27me3
mitigates podocyte pyroptosis in DKD by increasing early growth
response protein 1 (EGR1) in type 1 diabetic nephropathy (T1DN)
(106). Moreover, AGEs induce
downregulation of EZH2 in podocytes and decrease H3K27me3, which in
turn leads to the upregulated expression of pathological factors
(such as TGF-β1 and SNAI1) that contribute to podocyte injury in
late DKD (107). KDM6A, a HDM
that removes the di- and tri-methyl groups from histone H3K27, is
activated in patients with DKD and mice with T1DN (108). Suppression of KDM6A by
administration of GSK-J4 ameliorates the early lesions of db/db
mice, including renal dysfunction, mesangial matrix accumulation,
inflammation and apoptosis (109). The aforementioned studies
indicate that levels of H3K27 are affected by multiple factors,
such as cell types and environment, and the stage of DKD, thereby
changing the transcription of its target genes.
Numerous altered ubiquitination statuses of proteins
are associated with podocyte dysfunction. Specifically, TRIM29
promotes podocyte pyroptosis by interacting with inhibitor of
NF-κBα (IκBα) to mediate its ubiquitination-dependent degradation,
which triggers NF-κB activation (21). In addition, TRIM63 regulates
PPARα ubiquitination and degradation, contributing to podocyte
injury (110). The E3 ligase
c-Cbl binds to podocin and increases the ubiquitination of podocin,
leading to podocyte injury in DKD (111). Moreover, the deubiquitination
of sperm-associated antigen 5 by USP14 activates Akt/mTOR
signaling, exacerbating high glucose-induced autophagy and podocyte
injury (112). The
ubiquitination state of the receptor-interacting protein kinase
(RIPK)1/RIPK3 pathway downregulated by ubiquitin C-terminal
hydrolase L1, a crucial member of the deubiquitination family,
serves a key role in podocyte necroptosis and apoptosis (113).
Lysine 63 ubiquitination (Lys63-Ub) is increased in
the RTECs of DKD, and associated with autophagy deregulation and
apoptosis activation (114).
The apoptosis of RTECs is prevented by eukaryotic translation
initiation factor 2α ubiquitination and degradation mediated by
HMG-CoA reductase degradation 1 (115). Additionally, ARAP1 maintains
persistent epidermal growth factor receptor (EGFR) activation by
decreasing EGFR ubiquitination and subsequently activates
TGF-β/Smad3 and hypoxia inducible factor 1α (HIF-1α) signaling,
causing injury of RTECs and mesangial cells (MCs) (116,117). The aforementioned studies
demonstrate that targets of ubiquitination, such as Lys63-Ub, may
be a promising direction for DKD therapy.
SUMOylation and deSUMOylation serve vital roles in
the pathogenesis of numerous nephropathic diseases (118-120). In DKD, Kruppel-like factor
(KLF)15 upregulates the expression of SUMO1 and enhances the
SUMOylation of P53, subsequently inhibiting the proliferation of
MCs (121). However, the role
of SUMOylation in other aspects of DKD has not yet been
clarified.
Numerous studies have confirmed an increased number
of O-GlcNAc-positive cells in the glomeruli and enhanced staining
in the tubules in DKD (122,123). Hyperglycemia-driven elevation
of O-GlcNAc modification contributes to DKD progression via the
inhibition of Akt phosphorylation, thus abnormally activating
endothelial NOS (eNOS) (124).
A mutually reinforcing cycle is formed between activation of
O-GlcNAcylation and the intrarenal renin-angiotensin-aldosterone
system (RAAS), exacerbating glucose toxicity, while this effect is
inhibited by RAAS blockers via increasing OGA levels (124). Akimoto et al (125) found that aberrant
O-GlcNAcylation of α-actinin 4 and actin impairs cytoskeletal
structure and adhesive function, leading to morphological changes
in podocyte foot processes. This may trigger the early damage to
the glomerular filtration barrier in DKD. Pharmacological
inhibition of O-GlcNAcylation by OSMI1, an inhibitor targeting OGT,
decreases podocyte apoptosis under diabetic conditions (126). Notably, O-GlcNAc modification
exhibits a bidirectional paradox in tubular epithelial cells.
During prolonged fasting conditions in RTECs, it is essential for
renal lipolysis and exerts a protective effect against lipotoxicity
(127). However,
O-GlcNAcylation disrupts the homeostasis of retinol signals in HK2
cells (an immortalized proximal tubule epithelial cell line from
normal adult human kidney) by extensively modifying the key
molecules in the retinol signaling pathway, such as signaling
receptor and transporter of retinol 6 and retinaldehyde
dehydrogenases 1 (128).
Moreover, diabetic conditions promote OGT-mediated O-GlcNAcylation
of acyl-CoA synthetase long chain family member 4 (ACSL4), thereby
stabilizing ACSL4 and facilitating tubular ferroptosis in DKD
(129). This functional
discrepancy may be attributed to modification targets, cell type
and microenvironmental metabolic status. Additionally, as a subtype
of glycosylation, inhibition of core fucosylation mitigates renal
pathological changes, renal fibrosis and podocyte injury by
downregulating the phosphorylation of Smad2/3 and ERK (130). Targeting protein
O-GlcNAcylation may be a promising therapeutic approach for
rescuing DKD progression.
Lysine succinylation is a naturally occurring PTM
that alters the stability and function of substrate proteins. This
modification is modulated by enzymes such as SIRT5 and serves a
pivotal role in the pathogenesis of DKD. For example, upregulation
of SIRT5 inhibits the succinylation of NIMA-related kinase 7
(NEK7), disrupts the interaction between NEK7 and NOD-like receptor
protein 3 (NLRP3), and suppresses podocyte pyroptosis and oxidative
stress-induced injury under hyperglycemia (139). However, whether protein
succinylation is involved in the cell cycle of other renal cells
remains unclear.
The PI3K/Akt pathway modulates FoxO3a activity by
phosphorylating its three residues (Thr32, Ser253 and Ser315) and
excludes FoxO3a from the nucleus (140). Klotho prevents podocyte injury
against palmitate-induced oxidative stress by decreasing the
phosphorylation of FoxO3a, promoting its nuclear translocation and
upregulating the expression of antioxidants, including manganese
superoxide dismutase (MnSOD) (140). However, in streptozotocin
(STZ)-induced rats, activation of the PI3K/Akt/FoxO3 pathway
alleviates inflammation and oxidative stress in MCs (141). Furthermore, activation of the
AMPK/SIRT1/PPARγ coactivator 1α (PGC1α) pathway mitigates oxidative
stress in db/db mice (142).
Additionally, GSK3β knockdown enhances the antioxidant response
driven by Nrf2 and suppresses oxidative stress, leading to the
alleviation of oxidative stress and podocyte injury (67). Specifically, the elevated
phosphorylation of GSK3β at serine 9 reduces the protein stability
of GSK3β and diminishes its inhibitory effect on Nrf2, protecting
podocytes and RTECs from oxidative stress (67,143). The insulin receptor (IR) is key
for insulin action. Protein tyrosine-phosphatase 1B (PTP1B)
attenuates insulin signaling by dephosphorylating IR and IR
substrate 1, and knockdown of PTP1B protects podocytes from ER
stress by improving insulin sensitivity (144). Targeting protein kinases and
phosphatases may provide more options for DKD treatment.
Members of the SIRT family serve a pivotal role in
DKD pathogenesis. Activation of the SIRT1/PGC1α/Nrf1 and
SIRT1/NF-κB pathways effectively attenuates DKD, induces autophagic
flux, mitigates oxidative stress and alleviates mitochondrial
dysfunction in podocytes (77,145,146). Moreover, SIRT1 can interact
with ACSS2. ACSS2 promotes mitochondrial oxidative stress and
triggers renal tubular inflammation in DKD by modulating the
SIRT1/carbohydrate responsive element binding protein (ChREBP)
pathway (147). Polysulfides
can attenuate diabetic renal pathological lesions via inactivation
of p65 NF-κB and STAT3 phosphorylation/acetylation by sulfhydrating
SIRT1, thereby reducing high glucose-induced oxidative stress, cell
apoptosis, inflammation and EMT (77). Furthermore, the enhancement of
mitochondrial oxidative stress in DKD is mediated by the reduction
of SIRT3 activity and a subsequent increase in acetylated
isocitrate dehydrogenase 2/SOD2 (148). General control of amino acid
synthesis 5-like 1-mediated acetylation of MnSOD also exacerbates
oxidative stress-induced renal injury in DKD (149). The aforementioned studies
demonstrate the key role of the SIRT family in response to
oxidative stress in DKD.
Numerous alterations in the levels of proteins
involved in oxidative stress are regulated by the alteration of
histone methylation status. Expression of monocyte chemoattractant
protein-1 (MCP-1) is elevated due to the recruitment of SET7/9 and
H3K4me1 to its promoters in db/db mice (150). Decreased levels of HIF-1α
suppress oxidative stress and inflammation via HDM KDM3A in human
umbilical vein endothelial cells exposed to hypoxia and diabetic
conditions (151). Siddiqi
et al (152)
demonstrated that inhibition of EZH2 with DZNep can increase
podocyte injury, oxidative stress and proteinuria in diabetic rats.
Moreover, S-adenosylhomocysteine hydrolase inhibition induces
podocyte injury and oxidative stress via the
EZH2/EGR1/thioredoxin-interacting protein/NLRP3 signaling cascade
in STZ-induced T1DN mice (153). At present, the fundamental
mechanism by which EZH2 serves different roles in various DKD
models and cells remains unclear. Due to the involvement of
multiple cell types in the pathogenesis of DKD and the cell
context-specific gene regulation mediated by EZH2, EZH2 may serve a
dual role in different renal cells in DKD (154).
Hyperglycemia prompts Von Hippel-Lindau tumor
suppressor E3 ubiquitin ligase to ubiquitinate glucose-6-phosphate
dehydrogenase, leading to ROS production and podocyte injury
(155). Moreover, under
diabetic conditions, sustained activation of PH domain and
leucine-rich repeat protein phosphatase 1 promotes the nuclear
retention of FoxO1 via prevention of its ubiquitination, inducing
aberrations in renal gluconeogenesis and the activation of the
apoptotic cascade, and exacerbating oxidative stress in diabetic
rats (156). Oxidative stress
and apoptosis are decreased in RTECs by promoting SIRT1 expression
via suppressing its ubiquitination (157). Moreover, the phosphorylation of
c-Cbl at Tyr731 facilitates the combination of c-Cbl and SIRT1,
which triggers polyubiquitination of SIRT1 by c-Cbl and promotes
SIRT1 degradation, decreasing the antioxidant effects of FoxO3a in
DKD (158). Connexin32 (Cx32)
upregulates SIRT1 expression by inhibiting the ubiquitination of
Lys335 of SIRT1 by suppressing Smad specific E3 ubiquitin protein
ligase 1 (Smurf1), thus alleviating oxidative stress in DKD
(157). In addition, Cx32
decreases renal oxidative stress levels and ameliorates the
pathological progression of diabetic renal fibrosis by promoting
NADPH oxidase 4 polyubiquitination and degradation (159). Nrf2 is an important antioxidant
in response to oxidative stress. Suppression of the ubiquitination
of Nrf2 ameliorates experimental DKD through antioxidation and
regulation of the Keap1/Nrf2 signaling pathway (160,161). Additionally, ubiquitination
participates in ferroptosis induced by oxidative stress. Ginkgolide
B alleviates oxidative stress and ferroptosis by inhibiting
glutathione peroxidase (GPX)4 ubiquitination to improve changes in
renal structure in mice with DKD (162). Stimulator of interferon genes
protein inhibition alleviates ferroptosis and oxidative stress in
DKD via stabilization of ferroportin 1 (FPN1) protein levels by
decreasing FPN1 ubiquitination for proteasomal degradation
(163). In addition,
2-deoxy-d-ribose induces ferroptosis in RTECs by degrading the
cystine/glutamate antiporter SLC7A11 protein via the UPS, resulting
in decreased intracellular cystine uptake (164). These findings underscore the
key role of ubiquitination and deubiquitination in DKD development,
and highlight the potential therapeutic targets.
In early stage DKD, ectonucleoside triphosphate
diphosphohydrolase 5 (ENTPD5), a nucleotide hydrolase located in
the ER, modulates the N-glycosylation of proteins and facilitates
renal cell proliferation (165). Separately, in late stage DKD,
sustained hyperglycemia activates the hexosamine biosynthesis
pathway to increase the levels of UDP-GlcNAc, which triggers a
feedback mechanism that suppresses transcription factor SP1
activity and downregulates ENTPD5 expression, aggravating ER stress
(165). Additionally,
O-GlcNAcylation of the mineralocorticoid receptor directly enhances
both the protein abundance levels and transcriptional activity of
the receptor under diabetic conditions (166). These findings provide novel
directions for the diagnosis and targets of DKD.
Hypoglutathionemia and elevated oxidative stress
levels contribute to the early biochemical abnormalities in
diabetes (167). Additionally,
reactive carbonyl derivate levels increase in patients with
diabetes, and this elevation is more pronounced in diabetic
patients undergoing hemodialysis, suggesting that both diabetic
state and hemodialysis contribute to the enhancement of protein
oxidation (168).
A previous study showed that K99 succinylation of
hydroxysteroid 17β dehydrogenase 10 (HSD17B10) maintains
mitochondrial RNA ribonuclease P (RNase P) stability (169). Astragaloside IV alleviates
hyperglycemia-induced oxidative stress and mitochondrial
dysfunction in HK2 cells by upregulating carnitine
palmitoyltransferase 1A-mediated K99 succinylation of HSD17B10 to
maintain RNase P activity (169).
Extensive alterations of cell signaling pathways
serve a broad role in maintaining mitochondrial homeostasis and
optimizing oxidative phosphorylation. Inactivated AMPK and PI3K/Akt
signaling and abnormal activation of the JNK pathway participate in
mitochondrial dysfunction under diabetic conditions, modifying
mitochondrial dynamic homeostasis and energy metabolism disorder in
DKD (170-174). Finerenone treatment can reduce
mitochondrial fragmentation and restore mitophagy via PI3K/Akt/eNOS
signaling in HK2 cells exposed to diabetic conditions and tubular
cells of mice with DKD (172).
In addition, elevated SIRT1 phosphorylation at Ser47 is associated
with mitochondrial dysfunction in podocytes (175). Inhibiting SIRT1
phosphorylation-mediated ubiquitin-proteasome degradation restores
the capacity of SIRT1 to promote PGC1α deacetylation and nuclear
translocation, and thereby upregulates genes associated with
mitochondrial biosynthesis and antioxidant defense in DKD (176).
Phosphorylation of multiple proteins causes
disruption of mitochondrial dynamics and leads to loss of
mitochondrial voltage potential under high-glucose conditions.
Dynamin-related protein 1 (Drp1) is a key regulator of
mitochondrial fission. Cyclin-dependent kinase 5 phosphorylates
Drp1 at Ser616 and thus produces excessive ROS, leading to EMT
progression in HK2 cells (177). Enhanced phosphorylation of Drp1
at Ser637 (p-Drp1Ser637) by Rho-associated coiled
coil-containing protein kinase 1 promotes the transposition of Drp1
to the mitochondrial surface and accounts for excessive
mitochondrial fission in mouse podocytes (178-180). By contrast, resveratrol
decreases Drp1 levels while increasing p-Drp1Ser637
levels, blocking mitochondrial fission in MCs by inhibition of
phosphodiesterase-4D/protein kinase A signaling (181). The functional consequences of
serine phosphorylation of Drp1 may be dependent on cell type and
stimulation. Mitophagy is a specialized form of autophagy that
mediates the selective elimination of damaged or dysfunctional
mitochondria (182). The
phosphorylated form of FUN14 domain-containing 1 (FUNDC1) inhibits
the induction of mitophagy by blocking the interaction between the
FUNDC1 LC3-interacting region and LC3 (183,184). In addition, activated Src
kinase serves as a negative modulator of mitophagy in DKD by
inducing the phosphorylation of FUNDC1 at Tyr18, which impairs the
ability of podocytes to clear damaged mitochondria (185). Studies have demonstrated the
key role of aberrant protein phosphorylation in the progression of
DKD (Table II) (81,186-190). However, further validation at
different stages of human DKD are needed to fully understand the
impact of PTMs.
Protein acetylation is a key component of diverse
metabolic reactions. SIRT1 sustains mitochondrial homeostasis by
mediating mitochondrial biogenesis and mitophagy in DKD (191,192). It has been hypothesized that
the SIRT3/SOD2/GPX4 signaling pathway participates in the
regulation of ferroptosis in DKD via maintenance of mitochondrial
redox homeostasis (193). SIRT6
upregulation alleviates mitochondrial dysfunction and podocyte
apoptosis via AMPK activation mediated by its deacetylation of H3K9
and H3K56 (194). In addition,
activation of SIRT1 exerts a renoprotective role in restoring
mitochondrial homeostasis, providing a preclinical research basis
for small molecule drugs targeting the SIRT family.
TRIM22, a E3 ubiquitin ligase, is highly expressed
in patients with DKD, interacts with optic atrophy 1 and induces
its ubiquitination, thus altering mitochondrial fusion-associated
proteins involved in respiration/ATP synthesis, influencing ROS
production and mitochondrial function in DKD (195). Long non-coding RNA PVT1 is
involved in mitochondrial dysfunction by interacting with TRIM56 at
the post-transcriptional level to induce AMPK ubiquitination,
leading to aberrant mitochondrial biology and increasing
mitochondrial DNA leakage in podocytes in DKD (196). Additionally, interference with
Smurf2 inhibits both RUNX family transcription factor 3
ubiquitination and the TLR4/NF-κB signaling pathway, which
alleviates mitochondrial dysfunction and tubular injury (197). Another E3 ubiquitin ligase,
Cullin3, directly interacts with mitochondrial ribosomal protein
L12 to induce its ubiquitination, resulting in mitochondrial
biosynthesis dysfunction in RTECs (198). However, the effect of
ubiquitination on the mitochondrial homeostasis of MCs and
endothelial cells needs further investigation.
DeSUMOylation of RNA binding motif protein X-linked
serves a key role in determining the microRNA (miRNA/miR)
composition of renal cell exosomes, which prevents the protective
miRNAs from inhibiting mitochondrial damage in DKD (199).
In chronic diabetes, mitochondrial proteins are
susceptible to PTMs triggered by glycation and oxidation. Oxidative
and nitrosative stresses promote mitochondrial oxidative
dysfunction in STZ-induced diabetic rats (200). Carbonyl-mediated modifications
selectively target key protein components of major mitochondrial
cycles, including oxidative phosphorylation and fatty acid
β-oxidation (201).
Methylglyoxal, a dicarbonyl compound that accumulates to high
levels in the hyperglycemic environment, exerts an inhibitory
impact on both the tricarboxylic acid cycle and the electron
respiratory chain (202).
Notably, such modifications are specific to certain mitochondrial
proteins and trigger disturbances in mitochondria involved in renal
cellular toxicity and the progression of DKD (202).
The expression of lysine lactylation is notably
elevated in renal tissues from patients with diabetes as well as
db/db mice (43,203,204). Lactylation of acyl-CoA
synthetase family member 2 at lysine 182 contributes to tubular
mitochondrial dysfunction, which accelerates the progression of DKD
(203). However, research on
the roles of lactylation in other renal cell types is lacking.
NF-κB, interacting with IκB and IκB kinase (IKK),
is a key intracellular molecule regulating inflammation and is
abnormally activated in DKD (205,206). FoxM1 transcriptionally
activates SIRT4, and suppresses phosphorylation of NF-κB (Ser536)
and the levels of NLRP3 inflammasome to ameliorate renal damage and
podocyte pyroptosis in DKD (207). Inflammation in DKD is decreased
by activation of the Nrf2-mediated antioxidant pathway and
inhibition of the MAPK-mediated inflammatory pathway, such as
ERK1/2, JNK and MAPK (205,208,209). Treatment with salidroside
triggers the phosphorylation of Akt and GSK3β; suppressed
expression of p-Akt (Ser473) and p-GSK3β (Ser9) inhibits oxidative
stress and inflammation in DKD rats (210). Upregulated phosphorylation of
SH2 domain-containing protein-tyrosine phosphatase-2 (SHP2) is
detected in macrophages in both diabetic patients and mouse models
(211,212). Macrophage SHP2 deficiency
alleviates DKD via the suppression of MAPK/NF-κB-dependent
inflammation, subsequently attenuating renal dysfunction, collagen
deposition, fibrosis and inflammatory response in STZ-treated mice
(211). Dephosphorylation of
STAT3 (Tyr705) ameliorates tubulointerstitial inflammation and
glomerulosclerosis in DKD (213,214). Monitoring the dynamic changes
of the phosphorylated status of proteins during DKD progression may
improve understanding of the pathogenesis of DKD.
High-glucose stimulation increases H3K9/14Ac at the
receptor for AGEs, plasminogen activator inhibitor-1 and MCP-1
promoters, performing key roles in rat MCs in DKD, whereas losartan
reverses the H3K9/14Ac marks at targeted genes (215). PCAF serves an essential role in
regulating inflammatory molecules through H3K18ac, providing a
potential therapeutic target for inflammation-associated renal
diseases (216-218). HDACs also participate in the
inflammatory response in DKD. Apelin-13 inhibits diabetes-induced
elevation of inflammatory factors and histone hyperacetylation by
upregulation of HDAC1 (219).
Gene silencing of HDAC4 decreases the inflammatory response and
apoptosis induced by hyperglycemia in podocytes (96). Upregulation of SIRT1 inhibits
inflammation through decreasing the induction of inflammatory
cytokines and reducing acetylated-NF-κB (220-223). SIRT6 protects podocytes from
inflammation by inhibiting the Notch pathway (101). Moreover, upregulation of SIRT7
decreases inflammation and improves renal function in glomerular
endothelial cells by regulating the H3K18ac level of
death-associated protein kinase-3 (224). These findings support the
impact of acetylation on DKD and the potential mechanisms of
existing therapeutic drugs, such as losartan.
Different histone modifications are involved in the
inflammatory response in DKD. SET7/9 and H3K4me1 expression are
markedly increased, whereas H3K9me2 and H3K9me3 are decreased by
inflammation induced by hyperglycemia (225,226). Moreover, the diabetic
environment attenuates SET domain-containing protein 8 (SETD8)
levels, as well as their downstream target H4K20me1. Upregulation
of H4K20me1 inhibits endothelial inflammation in DKD by occupying
the promoter regions of diverse target genes, including IL-1
receptor-associated kinase 1, Wnt family member 5A and PTP1B
(227-229). Additionally, yes-associated
protein 1 promotes hyperglycemia-induced inflammation and ECM
deposition by triggering the activation of NF-κB/jumonji
domain-containing protein-3 signaling in MCs (230). In addition, KDM6A regulates the
transcription of inflammatory genes in a manner dependent on its
demethylase activity (109).
These results support that epigenetic alternations are associated
with sustained pro-inflammatory pathways and partly explain the
phenomenon of 'hyperglycemic memory' in DKD. This refers to the
persistent susceptibility of diabetic patients to develop
complications stemming from early hyperglycemic exposure, even
following effective implementation of blood glucose control
(231).
Ubiquitin-modifying enzymes and deubiquitinases act
in conjunction to regulate the transmission of intracellular
ubiquitin signaling to maintain normal cell activities. OTUD5, a
deubiquitinating enzyme, deubiquitinates K63-linked TGF-β-activated
kinase 1 (TAK1) at the K158 site through its active site C224,
which prevents TAK1 phosphorylation and decreases downstream
inflammatory responses in podocytes during DKD (232). E3 ubiquitin ligase speckle-type
BTB-POZ protein promotes NLRP3 degradation by elevating K48-linked
polyubiquitination of NLRP3 (233). TGF-β1 is profibrogenic in renal
fibrosis (234). Latent TGF-β1,
unlike the active form of TGF-β1, protects against inflammation and
fibrosis by blocking the E3-ligase Arkadia-mediated Smad7 ubiquitin
proteasomal degradation pathway in STZ-induced T1DN (234). Obstruction of ubiquitin
degradation of IKK induced by decreased ubiquitin ligase NEDD4L and
inhibition of TNF receptor-associated factor 6 K63
polyubiquitination mediated by USP25 can decrease the activation of
NF-κB and relieve inflammation (19,235). Parkin inhibits pathological
progression of DKD by promoting the ubiquitination of GATA-binding
protein 4 (GATA4) and downregulating GATA4/growth arrest-specific
protein 1 signaling to inhibit premature senescence, renal
inflammation and fibrosis (236). The aforementioned studies
demonstrate that protein ubiquitination performs an important role
in inflammation in DKD.
In diabetic mice, the expression of SENP6 is
decreased in glomerular tissue; this downregulation exacerbates
injury to the glomerular filtration barrier. Mechanistically, SENP6
enhances the ubiquitination of the Notch1 intracellular domain
(N1ICD) by deSUMOylating Notch1, subsequently reducing N1ICD and
inhibiting Notch1 signaling activation in podocytes (237). Moreover, SENP6 deSUMOylates
KDM6A and decreases the binding affinity of KDM6A to endothelin-1
(Edn1) via upregulation of H3K27me2/3 at its promoter (237). Numerous studies have indicated
that hyperglycemia-induced activation of NF-κB inflammatory
signaling is mediated by the SUMO E3 ligase protein inhibitor of
activated STAT y and IκBα SUMOylation (238,239). These findings indicate that the
combined effect of various PTMs regulate the pathogenesis of
DKD.
Protein glycosylation serves an important role in
protein stability, binding, folding and activity, and is a key PTM
of proteins. In mouse kidney endothelial cells, hyperglycemia
causes increased methylglyoxal modification of the corepressor
mSin3A, which results in increased recruitment of OGT and enhanced
O-GlcNAcylation of Sp3 (240).
This modification of Sp3 causes an increase in angiopoietin 2
expression, sensitizing microvascular endothelial cells to the
proinflammatory effects of TNFα (240).
TGF-β1 induces profibrotic and inflammatory genes,
which serve key roles in glomerular dysfunction and the mesangial
matrix expansion associated with DKD (242,243). Multiple protein kinases and
phosphatases have an essential role in renal fibrosis. TGF-β1
stimulation results in the phosphorylation/activation of PKCβII, a
direct substrate of mTORC2, thus modulating renal fibrosis in DKD
(244). Activation of AMPK and
Akt signaling alleviates renal injury and fibrosis in DKD (245-247). Activation of MAPK signaling
aggravates fibrosis under diabetic conditions (248-250). Activation of the GSK3β and
Nrf2/HO-1 pathways also causes inhibitory regulation of EMT and
exerts anti-renal fibrosis activity, delaying the progression of
DKD (251,252). The suppression of
phosphorylation of EGFR and NF-κB are involved in amelioration of
renal tubulointerstitial fibrosis (253-255). Targeting shared profibrotic
pathways via modulation of protein phosphorylation may serve as a
novel therapeutic strategy for DKD (Table III) (130,190,256-260).
Different acetylated states of proteins perform
different functions. For example, p300-dependent H3K27 acetylation
on the PLK1 gene promoter ameliorates renal fibrosis of DKD
(90). Moreover, sterol
regulatory element-binding transcription factor 1a K333 acetylation
mediated by the acetyltransferase CBP is key for Smad3 association
and they cooperatively mediate TGF-β transcriptional responses
(261). In addition, numerous
deacetylases contribute to renal fibrosis in DKD. HDAC2 serves a
key role in the development of ECM accumulation, EMT and renal
interstitial fibrosis in diabetic kidneys by regulating the
acetylation of substrates, including Edn1 and miR-205 (262-264). SIRT1 activation markedly
suppresses endothelial-mesenchymal transition (EndMT) progression,
and attenuates albuminuria and glomerulopathy via regulation of the
acetylation of NF-κB, FoxO1 and FoxO3a (265-267). SIRT3 deficiency in endothelial
cells stimulates TGF-β/Smad3-dependent mesenchymal transformation
in RTECs (268). SIRT6 has been
demonstrated to directly interact with Smad3, a key downstream
mediator of TGF-β, and inhibits its nuclear accumulation and
transcriptional activity by deacetylating it in HK2 cells (269). Furthermore, FoxO3a exerts a
renoprotective effect against diabetic kidney injury via the
SIRT6/Smad3 pathway (Table
III) (269,270). These studies reveal the vital
role of imbalanced acetylation in renal fibrosis in DKD.
EZH2/H3K27me3 recruitment at the promoters of
profibrotic genes is downregulated in rat MCs in T1DN and
reciprocally upregulates expression of these profibrotic genes,
such as connective tissue growth factor and Serpine1 (271). Moreover, EZH2 alleviates the
progression of renal interstitial fibrosis in T1DN by regulating
its downstream genes, such as MMP9 (272). In type 2 diabetic nephropathy,
OGT stabilizes EZH2 by promoting its glycosylation and then
inhibiting MC hyperproliferation and fibrosis by enhancing the
enrichment of EZH2/H3K27me3 in the hairy and enhancer of split 1
promoter (273). However, the
role of EZH2 in DKD remains controversial. In early DKD, enhanced
expression of EZH2 is associated with decreased DEPTOR levels and
increased mTOR activity, inducing MC hypertrophy and matrix
expansion (274). Under
diabetic conditions, recruitment of EZH2 inhibits SOX6, induces
cell proliferation, fibrosis and inflammatory cytokine release in
MCs (275). The method used to
establish DKD models and context-dependent factors affects the
function of EZH2 in renal fibrosis. To explain the role of EZH2 in
renal fibrosis, the association between EZH2 expression and
histological characteristics of patients with DKD should be
assessed.
The enhanced recruitment of SET7/9 and elevated
H3K4me at the p21 promoter, concurrent with the decreased H3K9me
level are observed in the glomeruli of diabetic rats, resulting in
increased mesangial hypertrophy (276). Histone H2AK119
mono-ubiquitination (H2AK119-Ub) downregulates SET7/9 (277). Genetic suppression of SET7/9
decreases profibrotic gene expression and prevents EndMT by
regulating insulin-like growth factor-binding protein 5 (278,279). Additionally, SETD8/H4K20me1
regulates EndMT in DKD by modulating methylation of its downstream
targets, such as profilin 2 and enolase 1 (280-282). However, additional studies are
required to examine the effects of SET7/9 and SETD8 in other cell
types in the progression of DKD.
Beyond methyltransferases that act on the lysine
sites of proteins, elevated protein arginine methyltransferase 1
expression activates activating transcription factor 6 by
recruiting H4R3me2as to the promoter, promoting ER stress and EMT
activation in HK2 cells (283).
In addition, demethylases are associated with renal
fibrosis. Expression of fibrotic proteins and dickkopf-1 is
negatively regulated by the KDM6A inhibitor GSK-J4, attenuating
glomerulosclerosis and renal fibrosis in mice with DKD (284,285). The HDM KDM3A is recruited to
the CTGF promoter to activate transcription, augmenting
hyperglycemia-induced CTGF induction in RTECs (286). Additionally, histone
lysine-specific demethylase 1 aggravates renal fibrosis by
decreasing SIRT3 expression and activating the TGF-β1/Smad3 pathway
(Table III) (287).
Numerous inhibitors (such as tazemetostat, GSK126
and GSK-J4) of methyltransferases and demethylases are undergoing
clinical trials for cancer treatment (288,289). Such preclinical research
utilizing pharmacological drugs that target methylation may advance
DKD treatment.
During the progression of renal fibrosis, numerous
proteins undergo ubiquitination and deubiquitination (290). E3 ubiquitin ligase of the TRIM
subfamily of RING-containing proteins is notably associated with
renal fibrosis in DKD (195,291). TRIM18 promotes EMT,
inflammation and fibrosis in HK2 cells via ubiquitination of PTP1B
and activates STAT3 signaling (292). Upregulation of TRIM13
suppresses mesangial collagen synthesis in DKD by promoting
ubiquitination of C/EBP homologous protein, providing insight into
the application of histone ubiquitination in the management of DKD
(293). Smurf1/2, HECT-type E3
ubiquitin ligases, participate in renal fibrosis by ubiquitinating
TGR5 and ChREBP (294-296). Additionally, serum creatinine
enhances the interaction between c-Cbl and CKIP-1 by promoting the
phosphorylation of c-Cbl, thereby increasing c-Cbl-mediated
ubiquitination of CKIP-1 to downregulate its expression, which
exacerbates renal inflammatory fibrosis in diabetic mice (297). USP modulates EMT and the
production of ECM components by deubiquitinating and stabilizing
their respective substrates (22,298). For example, USP9X decreases
Nrf2 ubiquitination and deubiquitinates Cx43 to regulate the EMT
process (299-301). Ubiquitination and degradation
of KDM3A increases TGF-β-induced factor 1 transcriptional activity,
inactivating TGF-β1/Smad2/3 signaling and suppressing the
progression of DKD (302). TAK1
mediates the phosphorylation of Ski-related novel protein N (SnoN),
leading to SnoN ubiquitination and degradation, which enhances EMT
and ECM deposition to promote renal fibrosis during DKD (Table III) (303,304). Modulation of ubiquitination may
play a promising role in the treatment of DKD.
High glucose enhances the SUMOylation of STAT1,
which prevents STAT1 from exerting an effective protective function
in inhibiting EMT (305).
Furthermore, diabetic conditions activate TGF-β/Smad signaling via
SUMO2/3 mediated SUMOylation of Smad4 in MCs (306,307).
O-GlcNAc augments the protein stability,
transcriptional activity and nuclear translocation of ChREBP.
Diabetic conditions increase the levels of O-GlcNAcylated ChREBP,
which further lead to lipid accumulation and upregulation of
fibrotic proteins in MCs (308). In addition, O-GlcNAc is, in
part, coupled to the profibrotic MAPK signaling pathway via
inhibition of Akt phosphorylation and potentially through ROS
(309).
The hyperglycemia-induced increases in
phosphorylation and oxidation of mitochondrial proteins contributes
to tubular dysfunction during DKD (310). S-nitrosylation serves a role in
the precise regulation of glomerular homeostasis by modulating
multiple important signaling pathways in DKD models. Specifically,
S-nitrosylation of laminin prevents the development of glomerular
nodules, while denitrosylation of S-nitrosoglutathione reductase
and increased S-nitrosylation of β3-integrin collectively result in
diffuse glomerulosclerosis in podocytes (311,312).
An elevation in histone lactylation is observed in
mice with DKD. H3K14la promotes the transcription of KLF5 in RTECs
of DKD (313,314). Notably, disruption of the
lactate/H3K14la/KLF5 pathway mitigates renal dysfunction and DKD
pathology (313). In addition,
6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3 (PFKFB3), a
key glycolytic enzyme, is associated with renal fibrosis and
dysfunction. Lactate generated from PFKFB3-mediated tubular
glycolytic reprogramming significantly enhances histone
lactylation, particularly H4K12la, which is enriched at the
promoters of NF-κB signaling genes (such as Ikbkb, Rela and Relb),
activating their transcription, and facilitating the inflammatory
response and renal fibrosis (39).
NaCr exerts an antidiabetic effect, decreases blood
glucose and serum lipid levels, and alleviates renal function and
DKD-associated inflammatory and fibrotic damage. NaCr induces
histone Kcr and H3K18 crotonylation. However, NaCr and
Cr-CoA-induced histone Kcr and renoprotective effects are abrogated
by inhibiting the activity of ACSS2 or histone acyltransferase p300
in vitro (241).
Moreover, ACSS2 notably increases H3K9cr levels in renal tissues
and tubular epithelial cells (48,91). Genetic and pharmacological
suppression of ACSS2 inhibits H3K9cr-mediated IL-1β expression,
which alleviates IL-1β-dependent macrophage activation and tubular
senescence to delay renal fibrosis (48). Targeting ACSS2 may serve as a
potential therapeutic intervention for the management of DKD, but
warrants further preclinical and clinical investigations.
Beyond its role in energy supply, BHB serves as a
bioactive molecule that exerts numerous protective effects,
including in DKD. BHB antagonizes glomerulosclerosis in diabetic
rats via upregulation of MMP2 production through elevation of
H3K9bhb at the MMP2 promoter (315). Moreover, BHB ameliorates
hyperglycemia-induced podocyte injury in vitro (316).
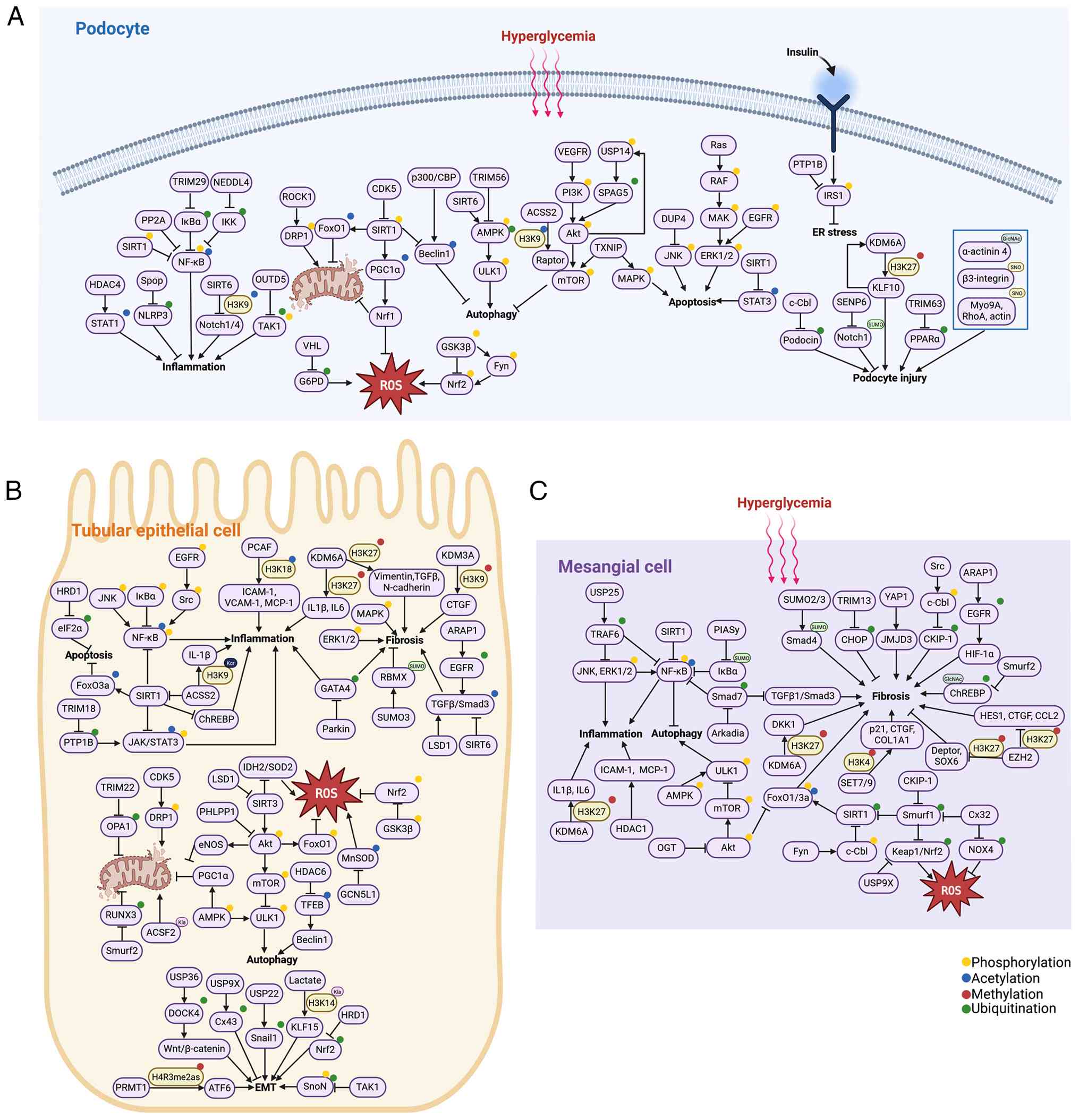
The interaction between PTMs of a protein to
modulate protein function through positive/negative regulatory
effects is termed PTM crosstalk. Positive crosstalk refers to
multiple PTMs that occur in the same local protein sequence area
(typically within a span of five amino acids) but do not happen in
the same residues, occurring concurrently or with a causal or
chronological connection (6).
Negative crosstalk is characterized by the direct competition of
two PTMs for the same residue in a causal or temporal manner.
Crosstalk between multiple PTMs is more frequently observed in key
protein domains such as histones and protein kinases in DKD. For
example, dysregulation of O-GlcNAcylation of β-actin Ser199 and
phosphorylation of β-actin Ser199 contributes to morphological
changes in DKD (317). Elevated
H2AK119-Ub and H2BK120 mono-ubiquitination (H2BK120-Ub) are
observed in diabetic rats (318). Histone H2AK119-Ub and
H2BK120-Ub promote diabetic renal fibrosis by upregulating the
expression of methyltransferases SET7/9 and SUV39H1, thereby
enhancing active H3K4Me2 and suppressive H3K9Me2 marks,
respectively (318). Zhang
et al (263) reported
that Dot1l and HDAC2 mutually inhibit their binding to the Edn1
promoter to regulate the production of Edn1, which is involved in
renal fibrosis in DKD. A novel ubiquitin-like modification,
neddylation, stabilizes RhoA by reducing its ubiquitination,
thereby activating the ERK1/2 pathway and driving interstitial
fibrosis (319). Moreover,
OTUD5 deubiquitinates K63-linked TAK1 at the K158 site, which
prevents the phosphorylation of TAK1 and decreases downstream
inflammatory responses in podocytes (230). Furthermore, different PTMs
jointly regulate DKD progression through combined or antagonistic
pathways, yet their cross-interaction networks need systematic
elucidation (Fig. 3).
PTMs modulate gene expression, and protein
stability and activity, serving a pivotal role in DKD progression
as a key event in the pathological timeline. Despite increasing
attention, the majority of PTMs in DKD are still in the preliminary
research stage, and their regulatory networks and cell
type-specific roles need further clarification (320,321). Notably, studies have identified
PTMs as potential biomarkers for DKD occurrence and development
(322-324). For example, plasma
2,6-sialylation of triantennary glycan A3E is associated with DKD
risk (322). Moreover, patients
with DKD exhibit abnormal lactate metabolism, and there is an
association between urinary lactate levels and renal tubular injury
(323,324). Overall, these findings
highlight the translational importance of PTMs in early diagnosis
and progression assessment of DKD. Accumulating evidence further
demonstrates the therapeutic prospects of targeting PTMs for DKD
treatment (320,325). Numerous drugs exert biological
effects partly by targeting PTM regulators. For example, metformin,
melatonin and resveratrol target SIRT1 to regulate autophagy,
oxidative stress, lipid deposition and renal fibrosis in DKD
(326-328). Additionally, sodium-glucose
cotransporter 2 and RAAS inhibitors exert renoprotective effects on
DKD by impacting O-GlcNAcylation (329).
At present, there are numerous small-molecule
inhibitors targeting PTMs used in the therapeutic research of DKD
models. Small-molecule inhibitors of HDACs (such as trichostatin A,
vorinostat and valproic acid targeting class I and II HDACs),
methyltransferases (such as GSK-J4 targeting KDM6A) and
phosphorylation-related enzymes (such as rapamycin targeting mTOR)
have exhibited favorable efficacy in preclinical models, as
evidenced by the mitigation of renal fibrosis and inflammation, and
improvement of renal function (320,330-332). Despite the encouraging results
of PTM activators or inhibitors in experimental models, their
clinical therapeutic efficacy is subject to limitations. The wide
presence of PTMs in vivo and the crosstalk among distinct
PTMs poses challenges to selective targeting, as targeting a single
modification may interfere with interrelated pathways and lead to
unexpected consequences. Moreover, individual enzymes typically act
on multiple substrates and signaling pathways, causing global
changes instead of gene- or organ-specific effects. At present,
clinical trials of inhibitors of methyltransferases and HDACs, such
as tazemetostat, chidamide and entinostat, are concentrated on
therapy for cancer, including various types of relapsed/refractory
lymphoma, prostate cancer and renal cell carcinoma (333-336). No specific PTM modulators have
been granted approval for clinical trials in DKD to date. The
safety and efficacy of PTM interventions in humans await rigorous
validation.
In summary, PTMs are key regulators that precisely
modulate protein function, stability, interactions and subcellular
localization. Their pervasive involvement in biological processes
provides insights into the pathogenesis of DKD. The evolving
research on PTM-regulatory agents, including novel compounds and
ongoing clinical trials, underscores their therapeutic promise.
However, current evidence for specific PTMs in DKD relies on
cross-sectional studies from preclinical models, with a notable
absence of systematic longitudinal research tracing the dynamic PTM
alterations throughout the course of DKD onset and progression.
Moreover, the intricate crosstalk among different PTM pathways
remains poorly elucidated.
To bridge these gaps and advance clinical
applications, future research should prioritize several key
directions. First, implementing single-cell and spatial multi-omics
technologies is essential to delineate PTM landscapes at cellular
and compartment-specific resolution in the kidney. Second, the
development of highly selective modulators, leveraging advanced
structural biology and proteolysis-targeting chimera technology,
will help to minimize off-target effects. Third, given the
interconnected signaling networks in DKD, investigating rational
combination strategies targeting multiple PTMs or integrating PTM
modulation with conventional therapy may enhance efficacy and
overcome resistance. Finally, translational efforts should
prioritize validating specific PTM signatures as non-invasive
biomarkers for early diagnosis and precise staging of DKD,
ultimately facilitating personalized therapeutic strategies.
Overall, a deeper understanding of PTM-driven
mechanisms, combined with innovative technology and translational
validation, is pivotal in transforming the landscape of DKD
diagnosis and treatment.
Not applicable.
YW and LY conceived the study. MH, ZM, YZ and RY
performed the literature review and constructed the figures and
tables. MH, ZW, LZ, LW and YW revised the manuscript. Data
authentication is not applicable. All authors have read and
approved the final manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
The present study was supported by the National Science Funding
in China (grant nos. 82370823 and 82400952), the Tongzhou District
Science and Technology Project (grant nos. KJ2023SS011 and
JCQN2023001), the Non-communicable Chronic Diseases-National
Science and Technology Major Project (grant no. 2023ZD0508100).
|
1
|
Martinez Leon V, Hilburg R and Susztak K:
Mechanisms of diabetic kidney disease and established and emerging
treatments. Nat Rev Endocrinol. 22:21–35. 2026. View Article : Google Scholar
|
|
2
|
Cheng HT, Xu X, Lim PS and Hung KY:
Worldwide epidemiology of diabetes-related end-stage renal disease,
2000-2015. Diabetes Care. 44:89–97. 2021. View Article : Google Scholar
|
|
3
|
Tonneijck L, Muskiet MH, Smits MM, van
Bommel EJ, Heerspink HJ, van Raalte DH and Joles JA: Glomerular
hyper-filtration in diabetes: Mechanisms, clinical significance,
and treatment. J Am Soc Nephrol. 28:1023–1039. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ye K, Zhao Y, Huang W and Zhu Y: Sodium
butyrate improves renal injury in diabetic nephropathy through
AMPK/SIRT1/PGC-1α signaling pathway. Sci Rep. 14:178672024.
View Article : Google Scholar
|
|
5
|
Lee JM, Hammarén HM, Savitski MM and Baek
SH: Control of protein stability by post-translational
modifications. Nat Commun. 14:2012023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wu X, Xu M, Geng M, Chen S, Little PJ, Xu
S and Weng J: Targeting protein modifications in metabolic
diseases: Molecular mechanisms and targeted therapies. Signal
Transduct Target Ther. 8:2202023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Burnett G and Kennedy EP: The enzymatic
phosphorylation of proteins. J Biol Chem. 211:969–980. 1954.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen T, Xie S, Cheng J, Zhao Q, Wu H,
Jiang P and Du W: AKT1 phosphorylation of cytoplasmic ME2 induces a
metabolic switch to glycolysis for tumorigenesis. Nat Commun.
15:6862024. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pearah A, Ramatchandirin B, Liu T, Wolf
RM, Ikeda A, Radovick S, Sesaki H, Wondisford FE, O'Rourke B and He
L: Blocking AMPKαS496 phosphorylation improves mitochondrial
dynamics and hyperglycemia in aging and obesity. Cell Chem Biol.
30:15852023. View Article : Google Scholar
|
|
10
|
Zhao A, Guo C, Wang L, Chen S, Xu Q, Cheng
J, Zhang J, Jiang J, Di J, Zhang H, et al: Xiebai San alleviates
acute lung injury by inhibiting the phosphorylation of the
ERK/Stat3 pathway and regulating multiple metabolisms.
Phytomedicine. 128:1553972024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu Z, Yang J, Du M and Xin W: Functioning
and mechanisms of PTMs in renal diseases. Front Pharmacol.
14:12387062023. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lu HC, Dai WN and He LY: Epigenetic
histone modifications in the pathogenesis of diabetic kidney
disease. Diabetes Metab Syndr Obes. 14:329–344. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Du C, Zhu Y, Duan J, Yang Y, Ren Y, Mu L,
Yan Z, Li G, Wang H, Shi Y and Yao F: A-485 alleviates fibrosis and
apoptosis in kidney by disrupting tandem activation of acetylation
and phosphorylation on STAT3. Biomed Pharmacother. 188:1182172025.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Natarajan R: Epigenetic mechanisms in
diabetic vascular complications and metabolic memory: The 2020
edwin bierman award lecture. Diabetes. 70:328–337. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kouzarides T: Chromatin modifications and
their function. Cell. 128:693–705. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cheng Y, Chen Y, Wang G, Liu P, Xie G,
Jing H, Chen H, Fan Y, Wang M and Zhou J: Protein methylation in
diabetic kidney disease. Front Med (Lausanne). 9:7360062022.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mevissen TET and Komander D: Mechanisms of
deubiquitinase specificity and regulation. Annu Rev Biochem.
86:159–192. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Goru SK, Kadakol A and Gaikwad AB: Hidden
targets of ubiquitin proteasome system: To prevent diabetic
nephropathy. Pharmacol Res. 120:170–179. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu B, Miao X, Shen J, Lou L, Chen K, Mei
F, Chen M, Su X, Du X, Zhu Z, et al: USP25 ameliorates diabetic
nephropathy by inhibiting TRAF6-mediated inflammatory responses.
Int Immunopharmacol. 124:1108772023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lu D, Zhang Y, Zhu P, Wu J, Yuan C and Ni
L: The roles of the ubiquitin-proteasome system in renal disease.
Int J Med Sci. 22:1791–1810. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xu X, Qin Z, Zhang C, Mi X, Zhang C, Zhou
F, Wang J, Zhang L and Hua F: TRIM29 promotes podocyte pyroptosis
in diabetic nephropathy through the NF-kB/NLRP3 inflammasome
pathway. Cell Biol Int. 47:1126–1135. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhu S, Hou S, Lu Y, Sheng W, Cui Z, Dong
T, Feng H and Wan Q: USP36-mediated deubiquitination of DOCK4
contributes to the diabetic renal tubular epithelial cell injury
via Wnt/β-Catenin signaling pathway. Front Cell Dev Biol.
9:6384772021. View Article : Google Scholar
|
|
23
|
Yang Z, Zhang Y and Sun S: Deciphering the
SUMO code in the kidney. J Cell Mol Med. 23:711–719. 2019.
View Article : Google Scholar :
|
|
24
|
Chen ZH, Li D, Zhang JY, Wei BY, Zhao HL,
Li P and Chen DQ: SUMOylation and NEDDylation in kidney diseases.
Exp Mol Pathol. 144:1050102025. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chatham JC and Patel RP: Protein
glycosylation in cardiovascular health and disease. Nat Rev
Cardiol. 21:525–544. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu Z, Qin Z, Bai W, Wang S, Huang C, Li
N, Yan L, Gu Y and Shao F: Integrating bioinformatics and machine
learning to elucidate the role of protein glycosylation-related
genes in the pathogenesis of diabetic kidney disease. PLoS One.
20:e03296402025. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ren W, Bian Q and Cai Y: Mass
spectrometry-based N-glycosylation analysis in kidney disease.
Front Mol Biosci. 9:9762982022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Magalhães A, Duarte HO and Reis CA: The
role of O-glycosylation in human disease. Mol Aspects Med.
79:1009642021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu C, Dong W, Li J, Kong Y and Ren X:
O-GlcNAc modification and its role in diabetic retinopathy.
Metabolites 2022. 12:7252022.
|
|
30
|
Ye L, Ding W, Xiao D, Jia Y, Zhao Z, Ao X
and Wang J: O-GlcNAcylation: cellular physiology and therapeutic
target for human diseases. MedComm (2020). 4:e4562023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Pasupulati AK, Nagati V, Paturi ASV and
Reddy GB: Non-enzymatic glycation and diabetic kidney disease.
Vitam Horm. 125:251–285. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ma Y, Wang X, Lin S, King L and Liu L: The
potential role of advanced glycation end products in the
development of kidney disease. Nutrients. 17:7582025. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Parwani K and Mandal P: Role of advanced
glycation end products and insulin resistance in diabetic
nephropathy. Arch Physiol Biochem. 129:95–107. 2023. View Article : Google Scholar
|
|
34
|
Chahla C, Kovacic H, Ferhat L and Leloup
L: pathological impact of redox post-translational modifications.
Antioxid Redox Signal. 41:152–180. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Peleli M, Zampas P and Papapetropoulos A:
Hydrogen sulfide and the kidney: Physiological roles, contribution
to pathophysiology, and therapeutic potential. Antioxid Redox
Signal. 36:220–243. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Vrettou S and Wirth B: S-Glutathionylation
and S-Nitrosylation in mitochondria: Focus on homeostasis and
neurodegenerative diseases. Int J Mol Sci. 23:158492022. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Nakamura T, Oh CK, Zhang X and Lipton SA:
Protein S-nitrosylation and oxidation contribute to protein
misfolding in neurodegeneration. Free Radic Biol Med. 172:562–577.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhang D, Tang Z, Huang H, Zhou G, Cui C,
Weng Y, Liu W, Kim S, Lee S, Perez-Neut M, et al: Metabolic
regulation of gene expression by histone lactylation. Nature.
574:575–580. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang Y, Li H, Jiang S, Fu D, Lu X, Lu M,
Li Y, Luo D, Wu K, Xu Y, et al: The glycolytic enzyme PFKFB3 drives
kidney fibrosis through promoting histone lactylation-mediated
NF-κB family activation. Kidney Int. 106:226–240. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ye Z, Sun Y, Yang S, Li L, Li B, Xia Y,
Yuan T, Yu W, Chen L, Zhou X and Cheng F: Lgals3 promotes calcium
oxalate crystal formation and kidney injury through histone
lactylation-mediated FGFR4 activation. Adv Sci (Weinh).
12:e24139372025. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Qiao J, Tan Y, Liu H, Yang B, Zhang Q, Liu
Q, Sun W, Li Z, Wang Q, Feng W, et al: Histone H3K18 and ezrin
lactylation promote renal dysfunction in sepsis-associated acute
kidney injury. Adv Sci (Weinh). 11:e23072162024. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Peng X and Du J: Histone and non-histone
lactylation: Molecular mechanisms, biological functions, diseases,
and therapeutic targets. Mol Biomed. 6:382025. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wei X, Long M, Yu J and Du Y: The
lactate-lactylation axis in renal fibrosis: Potential mechanisms in
diabetic kidney disease. Ann Med. 57:25873262025. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Shen R, Ruan H, Lin S, Liu B, Song H, Li L
and Ma T: Lysine succinylation, the metabolic bridge between cancer
and immunity. Genes Dis. 10:2470–2478. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Tan M, Luo H, Lee S, Jin F, Yang JS,
Montellier E, Buchou T, Cheng Z, Rousseaux S, Rajagopal N, et al:
Identification of 67 histone marks and histone lysine crotonylation
as a new type of histone modification. Cell. 146:1016–1028. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wan J, Liu H, Chu J and Zhang H: Functions
and mechanisms of lysine crotonylation. J Cell Mol Med.
23:7163–7169. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Yang P, Qin Y, Zeng L, He Y, Xie Y, Cheng
X, Huang W and Cao L: Crotonylation and disease: Current progress
and future perspectives. Biomed Pharmacother. 165:1151082023.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Li L, Xiang T, Guo J, Guo F, Wu Y, Feng H,
Liu J, Tao S, Fu P and Ma L: Inhibition of ACSS2-mediated histone
crotonylation alleviates kidney fibrosis via IL-1β-dependent
macrophage activation and tubular cell senescence. Nat Commun.
15:32002024. View Article : Google Scholar
|
|
49
|
Zhou T, Cheng X, He Y, Xie Y, Xu F, Xu Y
and Huang W: Function and mechanism of histone
beta-hydroxybutyrylation in health and disease. Front Immunol.
13:9812852022. View Article : Google Scholar
|
|
50
|
Alicic RZ, Rooney MT and Tuttle KR:
Diabetic kidney disease: Challenges, progress, and possibilities.
Clin J Am Soc Nephrol. 12:2032–2045. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Darenskaya M, Kolesnikov S, Semenova N and
Kolesnikova L: Diabetic nephropathy: Significance of determining
oxidative stress and opportunities for antioxidant therapies. Int J
Mol Sci. 24:123782023. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Mohandes S, Doke T, Hu H, Mukhi D, Dhillon
P and Susztak K: Molecular pathways that drive diabetic kidney
disease. J Clin Invest. 133:e1656542023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Jin Q, Liu T, Qiao Y, Liu D, Yang L, Mao
H, Ma F, Wang Y, Peng L and Zhan Y: Oxidative stress and
inflammation in diabetic nephropathy: Role of polyphenols. Front
Immunol. 14:11853172023. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Yao L, Liang X, Qiao Y, Chen B, Wang P and
Liu Z: Mitochondrial dysfunction in diabetic tubulopathy.
Metabolism. 131:1551952022. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Hou Y, Wang Q, Han B, Chen Y, Qiao X and
Wang L: CD36 promotes NLRP3 inflammasome activation via the mtROS
pathway in renal tubular epithelial cells of diabetic kidneys. Cell
Death Dis. 12:5232021. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Kim J, Kundu M, Viollet B and Guan KL:
AMPK and mTOR regulate autophagy through direct phosphorylation of
Ulk1. Nat Cell Biol. 13:132–141. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Chen C, Ma J, Miao CS, Zhang H, Zhang M,
Cao X and Shi Y: Trigonelline induces autophagy to protect
mesangial cells in response to high glucose via activating the
miR-5189-5p-AMPK pathway. Phytomedicine. 92:1536142021. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Jin D, Zhao Y, Sun Y, Xue J, Li X and Wang
X: Jiedu Tongluo Baoshen formula enhances renal tubular epithelial
cell autophagy to prevent renal fibrosis by activating
SIRT1/LKB1/AMPK pathway. Biomed Pharmacother. 160:1143402023.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Li A, Yi B, Han H, Yang S, Hu Z, Zheng L,
Wang J, Liao Q and Zhang H: Vitamin D-VDR (vitamin D receptor)
regulates defective autophagy in renal tubular epithelial cell in
streptozotocin-induced diabetic mice via the AMPK pathway.
Autophagy. 18:877–890. 2022. View Article : Google Scholar :
|
|
60
|
Chen Y, Zheng YF, Lin XH, Zhang JP, Lin F
and Shi H: Dendrobium mixture attenuates renal damage in rats with
diabetic nephropathy by inhibiting the PI3K/Akt/mTOR pathway. Mol
Med Rep. 24:5902021. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Wang X, Jiang L, Liu XQ, Huang YB, Wang
AL, Zeng HX, Gao L, Zhu QJ, Xia LL and Wu YG: Paeoniflorin binds to
VEGFR2 to restore autophagy and inhibit apoptosis for podocyte
protection in diabetic kidney disease through PI3K-AKT signaling
pathway. Phytomedicine. 106:1544002022. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Wang N and Zhang C: Oxidative stress: A
culprit in the progression of diabetic kidney disease. Antioxidants
(Basel). 13:4552024. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Song S, Qiu D, Wang Y, Wei J, Wu H, Wu M,
Wang S, Zhou X, Shi Y and Duan H: TXNIP deficiency mitigates
podocyte apoptosis via restraining the activation of mTOR or p38
MAPK signaling in diabetic nephropathy. Exp Cell Res.
388:1118622020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Li H, Zhao K and Li Y: Gasdermin D
protects mouse podocytes against high-glucose-induced inflammation
and apoptosis via the C-Jun N-terminal kinase (JNK) pathway. Med
Sci Monit. 27:e9284112021.PubMed/NCBI
|
|
65
|
Liu Y, Li Y, Xu L, Shi J, Yu X, Wang X, Li
X, Jiang H, Yang T, Yin X, et al: Quercetin attenuates podocyte
apoptosis of diabetic nephropathy through targeting EGFR signaling.
Front Pharmacol. 12:7927772022. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Zhai Y, Li D, Wang Z, Shao L, Yin N and Li
W: Cortex mori radicis attenuates streptozotocin-induced diabetic
renal injury in mice via regulation of transient receptor potential
canonical channel 6. Endocr Metab Immune Disord Drug Targets.
22:862–873. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Chen M, Fang Y, Ge Y, Qiu S, Dworkin L and
Gong R: The redox-sensitive GSK3β is a key regulator of glomerular
podocyte injury in type 2 diabetic kidney disease. Redox Biol.
72:1031272024. View Article : Google Scholar
|
|
68
|
Feng Q, Yu X, Xie J, Liu F, Zhang X, Li S,
Wang Y, Pan S, Liu D and Liu Z: Phillygenin improves diabetic
nephropathy by inhibiting inflammation and apoptosis via regulating
TLR4/MyD88/NF-κB and PI3K/AKT/GSK3β signaling pathways.
Phytomedicine. 136:1563142025. View Article : Google Scholar
|
|
69
|
Liang LL, He MF, Zhou PP, Pan SK, Liu DW
and Liu ZS: GSK3β: A ray of hope for the treatment of diabetic
kidney disease. FASEB J. 38:e234582024. View Article : Google Scholar
|
|
70
|
Wang H, Yu X, Liu D, Qiao Y, Huo J, Pan S,
Zhou L, Wang R, Feng Q and Liu Z: VDR activation attenuates renal
tubular epithelial cell ferroptosis by regulating Nrf2/HO-1
signaling pathway in diabetic nephropathy. Adv Sci (Weinh).
11:e23055632024. View Article : Google Scholar :
|
|
71
|
Dong D, Zhang Y, He H, Zhu Y and Ou H:
Alpinetin inhibits macrophage infiltration and atherosclerosis by
improving the thiol redox state: Requirement of GSk3β/Fyn-dependent
Nrf2 activation. FASEB J. 36:e222612022. View Article : Google Scholar
|
|
72
|
Yu X, Jiang N, Li J, Li X and He S:
Upregulation of BRD7 protects podocytes against high
glucose-induced apoptosis by enhancing Nrf2 in a GSK-3β-dependent
manner. Tissue Cell. 76:1018132022. View Article : Google Scholar
|
|
73
|
Paeng J, Chang JH, Lee SH, Nam BY, Kang
HY, Kim S, Oh HJ, Park JT, Han SH, Yoo TH and Kang SW: Enhanced
glycogen synthase kinase-3β activity mediates podocyte apoptosis
under diabetic conditions. Apoptosis. 19:1678–1690. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Xie J, Yuan Y, Yao G, Yu W and Zhu Q: Role
and mechanism of NUP160-regulated autophagy in pathogenesis of
diabetic nephropathy. Iran J Kidney Dis. 17:327–334.
2023.PubMed/NCBI
|
|
75
|
Fang X, Huang W, Sun Q, Zhao Y, Sun R, Liu
F, Huang D, Zhang Y, Gao F and Wang B: Melatonin attenuates
cellular senescence and apoptosis in diabetic nephropathy by
regulating STAT3 phosphorylation. Life Sci. 332:1221082023.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Jia J, Tan R, Xu L, Wang H, Li J, Su H,
Zhong X, Liu P and Wang L: Hederagenin improves renal fibrosis in
diabetic nephropathy by regulating Smad3/NOX4/SLC7A11
signaling-mediated tubular cell ferroptosis. Int Immunopharmacol.
135:1123032024. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Sun HJ, Xiong SP, Cao X, Cao L, Zhu MY, Wu
ZY and Bian JS: Polysulfide-mediated sulfhydration of SIRT1
prevents diabetic nephropathy by suppressing phosphorylation and
acetylation of p65 NF-κB and STAT3. Redox Biol. 38:1018132021.
View Article : Google Scholar
|
|
78
|
Lu J, Chen PP, Zhang JX, Li XQ, Wang GH,
Yuan BY, Huang SJ, Liu XQ, Jiang TT, Wang MY, et al: GPR43
deficiency protects against podocyte insulin resistance in diabetic
nephropathy through the restoration of AMPKα activity.
Theranostics. 11:4728–4742. 2021. View Article : Google Scholar
|
|
79
|
Jiang W, Xiao T, Han W, Xiong J, He T, Liu
Y, Huang Y, Yang K, Bi X, Xu X, et al: Klotho inhibits
PKCα/p66SHC-mediated podocyte injury in diabetic nephropathy. Mol
Cell Endocrinol. 494:1104902019. View Article : Google Scholar
|
|
80
|
Wang X, Zhang Y, Chi K, Ji Y, Zhang K, Li
P, Fu Z, Wang X, Cui S, Shen W, et al: IGFBP2 induces podocyte
apoptosis promoted by mitochondrial damage via integrin α5/FAK in
diabetic kidney disease. Apoptosis. 29:1109–1125. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Cao Y, Chen Z, Hu J, Feng J, Zhu Z, Fan Y,
Lin Q and Ding G: Mfn2 regulates high glucose-induced MAMs
dysfunction and apoptosis in podocytes via PERK pathway. Front Cell
Dev Biol. 9:7692132021. View Article : Google Scholar
|
|
82
|
Tian N, Gao Y, Wang X, Wu X, Zou D, Zhu Z,
Han Z, Wang T and Shi Y: Emodin mitigates podocytes apoptosis
induced by endoplasmic reticulum stress through the inhibition of
the PERK pathway in diabetic nephropathy. Drug Des Devel Ther.
12:2195–2211. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Uehara R, Yamada E, Okada S, Bastie CC,
Maeshima A, Ikeuchi H, Horiguchi K and Yamada M: Fyn phosphorylates
transglutaminase 2 (Tgm2) and modulates autophagy and p53
Expression in the development of diabetic kidney disease. Cells.
12:11972023. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Khan MF, Mathur A, Pandey VK and Kakkar P:
Endoplasmic reticulum stress-dependent activation of TRB3-FoxO1
signaling pathway exacerbates hyperglycemic nephrotoxicity:
Protection accorded by Naringenin. Eur J Pharmacol. 917:1747452022.
View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Wu H, Shi Y, Deng X, Su Y, Du C, Wei J,
Ren Y, Wu M, Hou Y and Duan H: Inhibition of c-Src/p38 MAPK pathway
ameliorates renal tubular epithelial cells apoptosis in db/db mice.
Mol Cell Endocrinol. 417:27–35. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Dusabimana T, Kim SR, Park EJ, Je J, Jeong
K, Yun SP, Kim HJ, Kim H and Park SW: P2Y2R contributes to the
development of diabetic nephropathy by inhibiting autophagy
response. Mol Metab. 42:1010892020. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Dong R, Zhang X, Liu Y, Zhao T, Sun Z, Liu
P, Xiang Q, Xiong J, Du X, Yang X, et al: Rutin alleviates EndMT by
restoring autophagy through inhibiting HDAC1 via PI3K/AKT/mTOR
pathway in diabetic kidney disease. Phytomedicine. 112:1547002023.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Lazar AG, Vlad ML, Manea A, Simionescu M
and Manea SA: Activated histone acetyltransferase p300/CBP-Related
signalling pathways mediate up-regulation of NADPH oxidase,
inflammation, and fibrosis in diabetic kidney. Antioxidants
(Basel). 10:13562021. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Li Y, Li X, He K, Li B, Liu K, Qi J, Wang
H, Wang Y and Luo W: C-peptide prevents NF-κB from recruiting p300
and binding to the inos promoter in diabetic nephropathy. FASEB J.
32:2269–2279. 2018. View Article : Google Scholar
|
|
90
|
Wang M, Huang Z, Li X, He P, Sun H, Peng Y
and Fan Q: Apabetalone, a BET protein inhibitor, inhibits kidney
damage in diabetes by preventing pyroptosis via modulating the
P300/H3K27ac/PLK1 axis. Pharmacol Res. 207:1073062024. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Lu J, Li XQ, Chen PP, Zhang JX, Liu L,
Wang GH, Liu XQ, Jiang TT, Wang MY, Liu WT, et al: Activation of
acetyl-CoA synthetase 2 mediates kidney injury in diabetic
nephropathy. JCI Insight. 8:e1658172023. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Xu J, Deng Y, Ke Y, Zhu Y, Wang P, Yu Q,
Li C and Shi B: Mutation of Beclin1 acetylation site at K414
alleviates high glucose-induced podocyte impairment in the early
stage of diabetic nephropathy by inhibiting hyperactivated
autophagy. Mol Biol Rep. 49:3919–3926. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Li X, Zhang Y, Chen H, Wu Y, Chen Y, Gong
S, Liu Y and Liu H: Inhibition of TFEB deacetylation in proximal
tubular epithelial cells (TECs) promotes TFEB activation and
alleviates TEC damage in diabetic kidney disease. FASEB J.
38:e238842024. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Brijmohan AS, Batchu SN, Majumder S,
Alghamdi TA, Thieme K, McGaugh S, Liu Y, Advani SL, Bowskill BB,
Kabir MG, et al: HDAC6 inhibition promotes transcription factor EB
activation and is protective in experimental kidney disease. Front
Pharmacol. 9:342018. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Liang T, Qi C, Lai Y, Xie J, Wang H, Zhang
L, Lin T, Jv M, Li J, Wang Y, et al: HDAC6-mediated α-tubulin
deacetylation suppresses autophagy and enhances motility of
podocytes in diabetic nephropathy. J Cell Mol Med. 24:11558–11572.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Wang X, Liu J, Zhen J, Zhang C, Wan Q, Liu
G, Wei X, Zhang Y, Wang Z, Han H, et al: Histone deacetylase 4
selectively contributes to podocyte injury in diabetic nephropathy.
Kidney Int. 86:712–725. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Wang W, Sun W, Cheng Y, Xu Z and Cai L:
Role of sirtuin-1 in diabetic nephropathy. J Mol Med (Berl).
97:291–309. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Lo CS, Shi Y, Chenier I, Ghosh A, Wu CH,
Cailhier JF, Ethier J, Lattouf JB, Filep JG, Ingelfinger JR, et al:
Heterogeneous nuclear ribonucleoprotein F stimulates sirtuin-1 gene
expression and attenuates nephropathy progression in diabetic mice.
Diabetes. 66:1964–1978. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Liu Y, Liu W, Zhang Z, Hu Y, Zhang X, Sun
Y, Lei Q, Sun D, Liu T, Fan Y, et al: Yishen capsule promotes
podocyte autophagy through regulating SIRT1/NF-κB signaling pathway
to improve diabetic nephropathy. Ren Fail. 43:128–140. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Jiao X, Li Y, Zhang T, Liu M and Chi Y:
Role of Sirtuin3 in high glucose-induced apoptosis in renal tubular
epithelial cells. Biochem Biophys Res Commun. 480:387–393. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Liu M, Liang K, Zhen J, Zhou M, Wang X,
Wang Z, Wei X, Zhang Y, Sun Y, Zhou Z, et al: Sirt6 deficiency
exacerbates podocyte injury and proteinuria through targeting Notch
signaling. Nat Commun. 8:4132017. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Liu DW, Zhang JH, Liu FX, Wang XT, Pan SK,
Jiang DK, Zhao ZH and Liu ZS: Silencing of long noncoding RNA PVT1
inhibits podocyte damage and apoptosis in diabetic nephropathy by
upregulating FOXA1. Exp Mol Med. 51:1–15. 2019. View Article : Google Scholar
|
|
103
|
Zhao Y, Li D, Zhou P, Zhao Y and Kuang J:
microRNA-29b-3p attenuates diabetic nephropathy in mice by
modifying EZH2. Hormones (Athens). 22:223–233. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Tang Y, Wan F, Tang X, Lin Y, Zhang H, Cao
J and Yang R: Celastrol attenuates diabetic nephropathy by
upregulating SIRT1-mediated inhibition of EZH2related wnt/β-catenin
signaling. Int Immunopharmacol. 122:1105842023. View Article : Google Scholar
|
|
105
|
Wang H, Wang J, Ran Q, Leng Y, Liu T,
Xiong Z, Zou D and Yang W: Identification and functional analysis
of the hub Ferroptosis-Related gene EZH2 in diabetic kidney
disease. Int Immunopharmacol. 133:1121382024. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Kim D, Ban KY, Lee GH and Jun HS:
Lysophosphatidic acid induces podocyte pyroptosis in diabetic
nephropathy by an increase of Egr1 expression via downregulation of
EzH2. Int J Mol Sci. 24:99682023. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Liebisch M and Wolf G: AGE-Induced
suppression of EZH2 mediates injury of podocytes by reducing
H3K27me3. Am J Nephrol. 51:676–692. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Lin CL, Hsu YC, Huang YT, Shih YH, Wang
CJ, Chiang WC and Chang PJ: A KDM6A-KLF10 reinforcing feedback
mechanism aggravates diabetic podocyte dysfunction. EMBO Mol Med.
11:e98282019. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Chen H, Huang Y, Zhu X, Liu C, Yuan Y, Su
H, Zhang C, Liu C, Xiong M, Qu Y, et al: Histone demethylase UTX is
a therapeutic target for diabetic kidney disease. J Physiol.
597:1643–1660. 2019. View Article : Google Scholar
|
|
110
|
Chen Q, Xie C, Tang K, Luo M, Zhang Z, Jin
Y, Liu Y, Zhou L and Kong Y: The E3 ligase Trim63 promotes podocyte
injury and proteinuria by targeting PPARα to inhibit fatty acid
oxidation. Free Radic Biol Med. 209(Pt 1): 40–54. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Liang L, He M, Zhou P, Pan S, Chen J, Lv
L, Hu M, Zhou S, Liu D and Liu Z: c-Cbl induced podocin
ubiquitination contributes to the podocytes injury in diabetic
nephropathy. FASEB J. 38:e236622024. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Xu J, Deng Y, Wang Y, Sun X, Chen S and Fu
G: SPAG5-AS1 inhibited autophagy and aggravated apoptosis of
podocytes via SPAG5/AKT/mTOR pathway. Cell Prolif. 53:e127382020.
View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Xu Y, Gao H, Hu Y, Fang Y, Qi C, Huang J,
Cai X, Wu H, Ding X and Zhang Z: High glucose-induced apoptosis and
necroptosis in podocytes is regulated by UCHL1 via RIPK1/RIPK3
pathway. Exp Cell Res. 382:1114632019. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Pontrelli P, Oranger A, Barozzino M,
Divella C, Conserva F, Fiore MG, Rossi R, Papale M, Castellano G,
Simone S, et al: Deregulation of autophagy under hyperglycemic
conditions is dependent on increased lysine 63 ubiquitination: a
candidate mechanism in the progression of diabetic nephropathy. J
Mol Med (Berl). 96:645–659. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Huang Y, Sun Y, Cao Y, Sun H, Li M, You H,
Su D, Li Y and Liang X: HRD1 prevents apoptosis in renal tubular
epithelial cells by mediating eIF2α ubiquitylation and degradation.
Cell Death Dis. 8:32022017. View Article : Google Scholar
|
|
116
|
Li X, Ma TK, Wen S, Li LL, Xu L, Zhu XW,
Zhang CX, Liu N, Wang X and Fan QL: LncRNA ARAP1-AS2 promotes high
glucose-induced human proximal tubular cell injury via persistent
transactivation of the EGFR by interacting with ARAP1. J Cell Mol
Med. 24:12994–13009. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Li X, Ma TK, Wang M, Zhang XD, Liu TY, Liu
Y, Huang ZH, Zhu YH, Zhang S, Yin L, et al: YY1-induced
upregulation of LncRNA-ARAP1-AS2 and ARAP1 promotes diabetic kidney
fibrosis via aberrant glycolysis associated with EGFR/PKM2/HIF-1α
pathway. Front Pharmacol. 14:10693482023. View Article : Google Scholar
|
|
118
|
Cai J, Wei J, Zeng Y, Yi M, Zhou Y, Ai K,
Chen W, Liu Y, Chen G, Chen A, et al: UBC9-associated SUMOylation
contributes to β-catenin activation and kidney fibrosis. Kidney
Int. 108:642–657. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Cai J, Pan J, Zeng Y, Wei J, Yi M, Xiang
Y, Liu Z, Duan S, Wang C and Dong Z: SUMOylation protects against
sepsis-associated acute kidney injury by stabilizing IκBα. Mol
Ther. Oct 27–2025.Epub ahead of print. View Article : Google Scholar
|
|
120
|
Lizotte F, Rousseau M, Denhez B, Lévesque
D, Guay A, Liu H, Moreau J, Higgins S, Sabbagh R, Susztak K, et al:
Deletion of protein tyrosine phosphatase SHP-1 restores SUMOylation
of podocin and reverses the progression of diabetic kidney disease.
Kidney Int. 104:787–802. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Wu L, Li O, Zhu F, Wang X, Chen P, Cai G,
Chen X and Hong Q: Krϋppel-like factor 15 suppresses renal
glomerular mesangial cell proliferation via enhancing P53 SUMO1
conjugation. J Cell Mol Med. 25:5691–5706. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Degrell P, Cseh J, Mohás M, Molnár GA,
Pajor L, Chatham JC, Fülöp N and Wittmann I: Evidence of O-linked
N-acetylglucosamine in diabetic nephropathy. Life Sci. 84:389–393.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Silva-Aguiar RP, Teixeira DE, Peres RAS,
Alves SAS, Novaes-Fernandes C, Dias WB, Pinheiro AAS, Peruchetti DB
and Caruso-Neves C: O-linked GlcNAcylation mediates the inhibition
of proximal tubule (Na++K+)ATPase activity in the early stage of
diabetes mellitus. Biochim Biophys Acta Gen Subj. 1867:1304662023.
View Article : Google Scholar
|
|
124
|
Gellai R, Hodrea J, Lenart L, Hosszu A,
Koszegi S, Balogh D, Ver A, Banki NF, Fulop N, Molnar A, et al:
Role of O-linked N-acetylglucosamine modification in diabetic
nephropathy. Am J Physiol Renal Physiol. 311:F1172–F1181. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Akimoto Y, Miura Y, Toda T, Wolfert MA,
Wells L, Boons GJ, Hart GW, Endo T and Kawakami H: Morphological
changes in diabetic kidney are associated with increased
O-GlcNAcylation of cytoskeletal proteins including α-actinin 4.
Clin Proteomics. 8:152011. View Article : Google Scholar
|
|
126
|
Song S, Hu T, Shi X, Jin Y, Liu S, Li X,
Zou W and Wang C: ER stress-perturbed intracellular protein
O-GlcNAcylation aggravates podocyte injury in diabetes nephropathy.
Int J Mol Sci. 24:176032023. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Sugahara S, Kume S, Chin-Kanasaki M,
Tomita I, Yasuda-Yamahara M, Yamahara K, Takeda N, Osawa N,
Yanagita M, Araki SI and Maegawa H: Protein O-GlcNAcylation is
essential for the maintenance of renal energy homeostasis and
function via lipolysis during fasting and diabetes. J Am Soc
Nephrol. 30:962–978. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Chen CH, Lin KD, Ke LY, Liang CJ, Kuo WC,
Lee MY, Lee YL, Hsiao PJ, Hsu CC and Shin SJ: O-GlcNAcylation
disrupts STRA6-retinol signals in kidneys of diabetes. Biochim
Biophys Acta Gen Subj. 1863:1059–1069. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Qin J, Chen Z, Ye M, Liang L and Ding X:
High glucose promotes O-GlcNAcylation of ACSL4 to induce
ferroptosis of renal tubular epithelial cell. Autoimmunity.
58:25768812025. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Fang M, Kang L, Wang X, Guo X, Wang W, Qin
B, Du X, Tang Q and Lin H: Inhibition of core fucosylation limits
progression of diabetic kidney disease. Biochem Biophys Res Commun.
520:612–618. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Ferreira MJ, Rodrigues TA, Pedrosa AG,
Silva AR, Vilarinho BG, Francisco T and Azevedo JE: Glutathione and
peroxisome redox homeostasis. Redox Biol. 67:1029172023. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Rashdan NA, Shrestha B and Pattillo CB:
S-glutathionylation, friend or foe in cardiovascular health and
disease. Redox Biol. 37:1016932020. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Guo Y, Liu Y, Zhao S, Xu W, Li Y, Zhao P,
Wang D, Cheng H, Ke Y and Zhang X: Oxidative stress-induced FABP5
S-glutathionylation protects against acute lung injury by
suppressing inflammation in macrophages. Nat Commun. 12:70942021.
View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Lash LH: Renal glutathione: Dual roles as
antioxidant protector and bioactivation promoter. Biochem
Pharmacol. 228:1161812024. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Li SS, Cui N, Yang Y, Trower TC, Wei YM,
Wu Y, Zhang S, Jin X and Jiang C: Impairment of the vascular KATP
channel imposes fatal susceptibility to experimental diabetes due
to multi-organ injuries. J Cell Physiol. 230:2915–2926. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Sampathkumar R, Balasubramanyam M,
Sudarslal S, Rema M, Mohan V and Balaram P: Increased
glutathionylated hemoglobin (HbSSG) in type 2 diabetes subjects
with microangiopathy. Clin Biochem. 38:892–899. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Li Q, Veron D and Tufro A: S-Nitrosylation
of RhoGAP Myosin9A is altered in advanced diabetic kidney disease.
Front Med (Lausanne). 8:6795182021. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Michelis R, Kristal B, Zeitun T, Shapiro
G, Fridman Y, Geron R and Sela S: Albumin oxidation leads to
neutrophil activation in vitro and inaccurate measurement of serum
albumin in patients with diabetic nephropathy. Free Radic Biol Med.
60:49–55. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Wu M and Ye X:
Quercetin-4'-O-β-D-glucopyranoside inhibits podocyte injury by
SIRT5-mediated desuccinylation of NEK7. Clin Exp Pharmacol Physiol.
51:e139092024. View Article : Google Scholar
|
|
140
|
Suk Kang J, Son SS, Lee JH, Lee SW, Jeong
AR, Lee ES, Cha SK, Chung CH and Lee EY: Protective effects of
klotho on palmitate-induced podocyte injury in diabetic
nephropathy. PLoS One. 16:e02506662021. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Liu H, Chen W, Lu P, Ma Y, Liang X and Liu
Y: Ginsenoside Rg1 attenuates the inflammation and oxidative stress
induced by diabetic nephropathy through regulating the
PI3K/AKT/FOXO3 pathway. Ann Transl Med. 9:17892021. View Article : Google Scholar
|
|
142
|
Park HS, Lim JH, Kim MY, Kim Y, Hong YA,
Choi SR, Chung S, Kim HW, Choi BS, Kim YS, et al: Resveratrol
increases AdipoR1 and AdipoR2 expression in type 2 diabetic
nephropathy. J Transl Med. 14:1762016. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Huang Q, Ouyang DS and Liu Q: Isoeucommin
A attenuates kidney injury in diabetic nephropathy through the
Nrf2/HO-1 pathway. FEBS Open Bio. 11:2350–2363. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Garner KL, Betin VMS, Pinto V, Graham M,
Abgueguen E, Barnes M, Bedford DC, McArdle CA and Coward RJM:
Enhanced insulin receptor, but not PI3K, signalling protects
podocytes from ER stress. Sci Rep. 8:39022018. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Li L, Zou J, Zhou M, Li H, Zhou T, Liu X,
Huang Q, Yang S, Xiang Q and Yu R: Phenylsulfate-induced oxidative
stress and mitochondrial dysfunction in podocytes are ameliorated
by Astragaloside IV activation of the SIRT1/PGC1α/Nrf1 signaling
pathway. Biomed Pharmacother. 177:1170082024. View Article : Google Scholar
|
|
146
|
Wang X, Gao Y, Tian N, Wang T, Shi Y, Xu J
and Wu B: Astragaloside IV inhibits glucose-induced
epithelial-mesenchymal transition of podocytes through autophagy
enhancement via the SIRT-NF-κB p65 axis. Sci Rep. 9:3232019.
View Article : Google Scholar
|
|
147
|
Lu J, Li XQ, Chen PP, Zhang JX, Li L, Wang
GH, Liu XQ, Jiang CM and Ma KL: Acetyl-CoA synthetase 2 promotes
diabetic renal tubular injury in mice by rewiring fatty acid
metabolism through SIRT1/ChREBP pathway. Acta Pharmacol Sin.
45:366–377. 2024. View Article : Google Scholar
|
|
148
|
Ogura Y, Kitada M, Monno I, Kanasaki K,
Watanabe A and Koya D: Renal mitochondrial oxidative stress is
enhanced by the reduction of Sirt3 activity, in Zucker diabetic
fatty rats. Redox Rep. 23:153–159. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Lv T, Lu Y, Liu Y, Feng H, Li C, Sheng W,
Cui Z, Zhu S, Gu X, Yang Z and Wan Q: General control of amino acid
synthesis 5-like 1-mediated acetylation of manganese superoxide
dismutase regulates oxidative stress in diabetic kidney disease.
Oxid Med Cell Longev. 2021:66912262021. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Chen J, Guo Y, Zeng W, Huang L, Pang Q,
Nie L, Mu J, Yuan F and Feng B: ER stress triggers MCP-1 expression
through SET7/9-induced histone methylation in the kidneys of db/db
mice. Am J Physiol Renal Physiol. 306:F916–F925. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Zhao M, Wang S, Zuo A, Zhang J, Wen W,
Jiang W, Chen H, Liang D, Sun J and Wang M: HIF-1α/JMJD1A signaling
regulates inflammation and oxidative stress following hyperglycemia
and hypoxia-induced vascular cell injury. Cell Mol Biol Lett.
26:402021. View Article : Google Scholar
|
|
152
|
Siddiqi FS, Majumder S, Thai K, Abdalla M,
Hu P, Advani SL, White KE, Bowskill BB, Guarna G, Dos Santos CC, et
al: The histone methyltransferase enzyme enhancer of zeste homolog
2 protects against podocyte oxidative stress and renal injury in
diabetes. J Am Soc Nephrol. 27:2021–2034. 2016. View Article : Google Scholar
|
|
153
|
Dai X, Liao R, Liu C, Liu S, Huang H, Liu
J, Jin T, Guo H, Zheng Z, Xia M, et al: Epigenetic regulation of
TXNIP-mediated oxidative stress and NLRP3 inflammasome activation
contributes to SAHH inhibition-aggravated diabetic nephropathy.
Redox Biol. 45:1020332021. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Li T, Yu C and Zhuang S: Histone
methyltransferase EZH2: A potential therapeutic target for kidney
diseases. Front Physiol. 12:6407002021. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Wang M, Hu J, Yan L, Yang Y, He M, Wu M,
Li Q, Gong W, Yang Y, Wang Y, et al: High glucose-induced
ubiquitination of G6PD leads to the injury of podocytes. FASEB J.
33:6296–6310. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Mathur A, Pandey VK, Khan MF and Kakkar P:
PHLPP1/Nrf2-Mdm2 axis induces renal apoptosis via influencing
nucleo-cytoplasmic shuttling of FoxO1 during diabetic nephropathy.
Mol Cell Biochem. 476:3681–3699. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Ye G, Hu ML and Xiao L: Forkhead box
A2-mediated lncRNA SOX2OT up-regulation alleviates oxidative stress
and apoptosis of renal tubular epithelial cells by promoting SIRT1
expression in diabetic nephropathy. Nephrology (Carlton).
28:196–207. 2023. View Article : Google Scholar
|
|
158
|
Li S, Lin Z, Xiao H, Xu Z, Li C, Zeng J,
Xie X, Deng L and Huang H: Fyn deficiency inhibits oxidative stress
by decreasing c-Cbl-mediated ubiquitination of Sirt1 to attenuate
diabetic renal fibrosis. Metabolism. 139:1553782023. View Article : Google Scholar
|
|
159
|
Chen ZQ, Sun XH, Li XJ, Xu ZC, Yang Y, Lin
ZY, Xiao HM, Zhang M, Quan SJ and Huang HQ: Polydatin attenuates
renal fibrosis in diabetic mice through regulating the Cx32-Nox4
signaling pathway. Acta Pharmacol Sin. 41:1587–1596. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Du L, Wang L, Wang B, Wang J, Hao M, Chen
YB, Li XZ, Li Y, Jiang YF, Li CC, et al: A novel compound AB38b
attenuates oxidative stress and ECM protein accumulation in kidneys
of diabetic mice through modulation of Keap1/Nrf2 signaling. Acta
Pharmacol Sin. 41:358–372. 2020. View Article : Google Scholar :
|
|
161
|
Gong W, Chen Z, Zou Y, Zhang L, Huang J,
Liu P and Huang H: CKIP-1 affects the polyubiquitination of Nrf2
and Keap1 via mediating Smurf1 to resist HG-induced renal fibrosis
in GMCs and diabetic mice kidneys. Free Radic Biol Med.
115:338–350. 2018. View Article : Google Scholar
|
|
162
|
Chen J, Ou Z, Gao T, Yang Y, Shu A, Xu H,
Chen Y and Lv Z: Ginkgolide B alleviates oxidative stress and
ferroptosis by inhibiting GPX4 ubiquitination to improve diabetic
nephropathy. Biomed Pharmacother. 156:1139532022. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Zhao QX, Yan SB, Wang F, Li XX, Shang GK,
Zheng ZJ, Xiao J, Lin ZW, Li CB and Ji XP: STING deficiency
alleviates ferroptosis through FPN1 stabilization in diabetic
kidney disease. Biochem Pharmacol. 222:1161022024. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Kim M, Bae JY, Yoo S, Kim HW, Lee SA, Kim
ET and Koh G: 2-Deoxy-d-ribose induces ferroptosis in renal tubular
epithelial cells via ubiquitin-proteasome system-mediated xCT
protein degradation. Free Radic Biol Med. 208:384–393. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Xu L, Zhou Y, Wang G, Bo L, Jin B, Dai L,
Lu Q, Cai X, Hu L, Liu L, et al: The UDPase ENTPD5 regulates ER
stress-associated renal injury by mediating protein
N-glycosylation. Cell Death Dis. 14:1662023. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Jo R, Shibata H, Kurihara I, Yokota K,
Kobayashi S, Murai-Takeda A, Mitsuishi Y, Hayashi T, Nakamura T and
Itoh H: Mechanisms of mineralocorticoid receptor-associated
hypertension in diabetes mellitus: the role of O-GlcNAc
modification. Hypertens Res. 46:19–31. 2023. View Article : Google Scholar
|
|
167
|
Sampathkumar R, Balasubramanyam M, Tara C,
Rema M and Mohan V: Association of hypoglutathionemia with reduced
Na+/K+ ATPase activity in type 2 diabetes and microangiopathy. Mol
Cell Biochem. 282:169–176. 2006. View Article : Google Scholar
|
|
168
|
Dursun E, Timur M, Dursun B, Süleymanlar G
and Ozben T: Protein oxidation in type 2 diabetic patients on
hemodialysis. J Diabetes Complications. 19:142–146. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Wang M, Li Q, Wang S, Zuo L, Hai Y, Yuan
S, Li X, Huang X, Yang C, Yao L, et al: Astragaloside IV protects
renal tubular epithelial cells against oxidative stress-induced
injury by upregulating CPT1A-mediated HSD17B10 lysine succinylation
in diabetic kidney disease. Phytother Res. 38:4519–4540. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Lin Q, Ma Y, Chen Z, Hu J, Chen C, Fan Y,
Liang W and Ding G: Sestrin-2 regulates podocyte mitochondrial
dysfunction and apoptosis under high-glucose conditions via AMPK.
Int J Mol Med. 45:1361–1372. 2020.PubMed/NCBI
|
|
171
|
Sun MY, Ye HJ, Zheng C, Jin ZJ, Yuan Y and
Weng HB: Astragalin ameliorates renal injury in diabetic mice by
modulating mitochondrial quality control via AMPK-dependent PGC1α
pathway. Acta Pharmacol Sin. 44:1676–1686. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Yao L, Liang X, Liu Y, Li B, Hong M, Wang
X, Chen B, Liu Z and Wang P: Non-steroidal mineralocorticoid
receptor antagonist finerenone ameliorates mitochondrial
dysfunction via PI3K/Akt/eNOS signaling pathway in diabetic
tubulopathy. Redox Biol. 68:1029462023. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Kong Z, Xiao M, Wang B, Zhang W, Che K, Lv
W, Wang Y, Huang Y, Zhao H, Zhao Y, et al: Renoprotective effect of
isoorientin in diabetic nephropathy via activating autophagy and
inhibiting the PI3K-AKT-TSC2-mTOR pathway. Am J Chin Med.
51:1269–1291. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Rousseau M, Denhez B, Spino C, Lizotte F,
Guay A, Côté A-M, Burger D and Geraldes P: Reduction of DUSP4
contributes to podocytes oxidative stress, insulin resistance and
diabetic nephropathy. Biochem Biophys Res Commun. 624:127–133.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Wang S, Yang Y, He X, Yang L, Wang J, Xia
S, Liu D, Liu S, Yang L, Liu W and Duan H: Cdk5-mediated
phosphorylation of Sirt1 contributes to podocyte mitochondrial
dysfunction in diabetic nephropathy. Antioxid Redox Signal.
34:171–190. 2021. View Article : Google Scholar
|
|
176
|
Zeng Y, Guo M, Wu Q, Tan X, Jiang C, Teng
F, Chen J, Zhang F, Ma X, Li X, et al: Gut microbiota-derived
indole-3-propionic acid alleviates diabetic kidney disease through
its mitochondrial protective effect via reducing ubiquitination
mediated-degradation of SIRT1. J Adv Res. 73:607–630. 2025.
View Article : Google Scholar :
|
|
177
|
Wang F, Xu W, Liu X and Zhang J:
Dexmedetomidine ameliorates high glucose-induced
epithelial-mesenchymal transformation in HK-2 cells through the
Cdk5/Drp1/ROS pathway. Acta Biochim Biophys Sin (Shanghai).
56:71–81. 2024. View Article : Google Scholar :
|
|
178
|
Chen Z, Ma Y, Yang Q, Hu J, Feng J, Liang
W and Ding G: AKAP1 mediates high glucose-induced mitochondrial
fission through the phosphorylation of Drp1 in podocytes. J Cell
Physiol. 235:7433–7448. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Liu S, Li X, Wen R, Chen L, Yang Q, Song
S, Xiao G, Su Z and Wang C: Increased thromboxane/prostaglandin
receptors contribute to high glucose-induced podocyte injury and
mitochondrial fission through ROCK1-Drp1 signaling. Int J Biochem
Cell Biol. 151:1062812022. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Wang W, Wang Y, Long J, Wang J, Haudek SB,
Overbeek P, Chang BH, Schumacker PT and Danesh FR: Mitochondrial
fission triggered by hyperglycemia is mediated by ROCK1 activation
in podocytes and endothelial cells. Cell Metab. 15:186–200. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Zhu X, Deng Z, Cao Y, Zhou Z, Sun W, Liu
C, Fan S and Yin XX: Resveratrol prevents Drp1-mediated
mitochondrial fission in the diabetic kidney through the PDE4D/PKA
pathway. Phytother Res. 37:5916–5931. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Picca A, Faitg J, Auwerx J, Ferrucci L and
D'Amico D: Mitophagy in human health, ageing and disease. Nat
Metab. 5:2047–2061. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Li K, Xia X and Tong Y: Multiple roles of
mitochondrial autophagy receptor FUNDC1 in mitochondrial events and
kidney disease. Front Cell Dev Biol. 12:14533652024. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Liu L, Feng D, Chen G, Chen M, Zheng Q,
Song P, Ma Q, Zhu C, Wang R, Qi W, et al: Mitochondrial
outer-membrane protein FUNDC1 mediates hypoxia-induced mitophagy in
mammalian cells. Nat Cell Biol. 14:177–185. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Zheng T, Wang HY, Chen Y, Chen X, Wu ZL,
Hu QY and Sun H: Src activation aggravates podocyte injury in
diabetic nephropathy via suppression of FUNDC1-mediated mitophagy.
Front Pharmacol. 13:8970462022. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Feng J, Chen Z, Ma Y, Yang X, Zhu Z, Zhang
Z, Hu J, Liang W and Ding G: AKAP1 contributes to impaired mtDNA
replication and mitochondrial dysfunction in podocytes of diabetic
kidney disease. Int J Biol Sci. 18:4026–4042. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Li X, Yang Q, Liu S, Song S and Wang C:
Mitochondria-associated endoplasmic reticulum membranes promote
mitochondrial fission through AKAP1-Drp1 pathway in podocytes under
high glucose conditions. Exp Cell Res. 424:1135122023. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Small DM, Morais C, Coombes JS, Bennett
NC, Johnson DW and Gobe GC: Oxidative stress-induced alterations in
PPAR-γ and associated mitochondrial destabilization contribute to
kidney cell apoptosis. Am J Physiol Renal Physiol. 307:F814–F822.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Lee J, Tsogbadrakh B, Yang S, Ryu H, Kang
E, Kang M, Kang HG, Ahn C and Oh KH: Klotho ameliorates diabetic
nephropathy via LKB1-AMPK-PGC1α-mediated renal mitochondrial
protection. Biochem Biophys Res Commun. 534:1040–1046. 2021.
View Article : Google Scholar
|
|
190
|
Pham TK, Nguyen THT, Yun HR, Vasileva EA,
Mishchenko NP, Fedoreyev SA, Stonik VA, Vu TT, Nguyen HQ, Cho SW,
et al: Echinochrome a prevents diabetic nephropathy by inhibiting
the PKC-Iota pathway and enhancing renal mitochondrial function in
db/db Mice. Mar Drugs. 21:2222023. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Zhou D, Zhou M, Wang Z, Fu Y, Jia M, Wang
X, Liu M, Zhang Y, Sun Y, Lu Y, et al: PGRN acts as a novel
regulator of mitochondrial homeostasis by facilitating mitophagy
and mitochondrial biogenesis to prevent podocyte injury in diabetic
nephropathy. Cell Death Dis. 10:5242019. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Saxena S, Anand SK, Sharma A and Kakkar P:
Involvement of Sirt1-FoxO3a-Bnip3 axis and autophagy mediated
mitochondrial turnover in according protection to hyperglycemic
NRK-52E cells by Berberine. Toxicol In Vitro. 100:1059162024.
View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Li Q, Liao J, Chen W, Zhang K, Li H, Ma F,
Zhang H, Han Q, Guo J, Li Y, et al: NAC alleviative ferroptosis in
diabetic nephropathy via maintaining mitochondrial redox
homeostasis through activating SIRT3-SOD2/Gpx4 pathway. Free Radic
Biol Med. 187:158–170. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Fan Y, Yang Q, Yang Y, Gao Z, Ma Y, Zhang
L, Liang W and Ding G: Sirt6 suppresses high glucose-induced
mitochondrial dysfunction and apoptosis in podocytes through AMPK
activation. Int J Biol Sci. 15:701–713. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Zhang Z, Zhou F, Lu M, Zhang D, Zhang X,
Xu S and He Y: WTAP-mediated m6A modification of TRIM22 promotes
diabetic nephropathy by inducing mitochondrial dysfunction via
ubiquitination of OPA1. Redox Rep. 29:24047942024. View Article : Google Scholar
|
|
196
|
Lv Z, Wang Z, Hu J, Su H, Liu B, Lang Y,
Yu Q, Liu Y, Fan X, Yang M, et al: LncRNA PVT1 induces
mitochondrial dysfunction of podocytes via TRIM56 in diabetic
kidney disease. Cell Death Dis. 15:6972024. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Xiao L and Ye G: RUNX3 alleviates
mitochondrial dysfunction and tubular damage by inhibiting
TLR4/NF-κB signalling pathway in diabetic kidney disease.
Nephrology (Carlton). 29:470–481. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Ji X, Yang X, Gu X, Chu L, Sun S, Sun J,
Song P, Mu Q, Wang Y, Sun X, et al: CUL3 induces mitochondrial
dysfunction via MRPL12 ubiquitination in renal tubular epithelial
cells. FEBS J. 290:5340–5352. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Yang Y, Ren S, Xue J, Dong W, He W, Luo J,
Li X, Xu H, Zheng Z, Wang X, et al: DeSUMOylation of RBMX regulates
exosomal sorting of cargo to promote renal tubulointerstitial
fibrosis in diabetic kidney disease. J Adv Res. 74:175–189. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Pérez-Gallardo RV, Noriega-Cisneros R,
Esquivel-Gutiérrez E, Calderón-Cortés E, Cortés-Rojo C,
Manzo-Avalos S, Campos-García J, Salgado-Garciglia R, Montoya-Pérez
R, Boldogh I and Saavedra-Molina A: Effects of diabetes on
oxidative and nitrosative stress in kidney mitochondria from aged
rats. J Bioenerg Biomembr. 46:511–518. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Rosca MG, Mustata TG, Kinter MT, Ozdemir
AM, Kern TS, Szweda LI, Brownlee M, Monnier VM and Weiss MF:
Glycation of mitochondrial proteins from diabetic rat kidney is
associated with excess superoxide formation. Am J Physiol Renal
Physiol. 289:F420–F430. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Rosca MG, Monnier VM, Szweda LI and Weiss
MF: Alterations in renal mitochondrial respiration in response to
the reactive oxoaldehyde methylglyoxal. Am J Physiol Renal Physiol.
283:F52–F59. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Chen J, Feng Q, Qiao Y, Pan S, Liang L,
Liu Y, Zhang X, Liu D and Liu Z and Liu Z: ACSF2 and lysine
lactylation contribute to renal tubule injury in diabetes.
Diabetologia. 67:1429–1443. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Fan Z, Zhang Y, Yuan L, Gao Y, Tian X,
Tian J, Wan J, Li B, Wang X, Wang S, et al: LARS1 lactylation
inhibits autophagy by activating mTORC1 to promote podocytes injury
in diabetic kidney disease. Cell Signal. 134:1119552025. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Qian J, Yin S, Ye L, Wang Z, Shu S, Mou Z,
Xu M, Chattipakorn N, Liu Z and Liang G: An Indole-2-Carboxamide
Derivative, LG4, alleviates diabetic kidney disease through
inhibiting MAPK-Mediated inflammatory responses. J Inflamm Res.
14:1633–1645. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Li X, Wang J, Yan J, He JC, Li Y and Zhong
Y: Additive renal protective effects between arctigenin and
puerarin in diabetic kidney disease. Biomed Pharmacother.
171:1161072024. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Xu X, Zhang L, Hua F, Zhang C, Zhang C, Mi
X, Qin N, Wang J, Zhu A, Qin Z and Zhou F: FOXM1-activated SIRT4
inhibits NF-κB signaling and NLRP3 inflammasome to alleviate kidney
injury and podocyte pyroptosis in diabetic nephropathy. Exp Cell
Res. 408:1128632021. View Article : Google Scholar
|
|
208
|
Ma L, Wu F, Shao Q, Chen G, Xu L and Lu F:
Baicalin alleviates oxidative stress and inflammation in diabetic
nephropathy via Nrf2 and MAPK signaling pathway. Drug Des Devel
Ther. 15:3207–3221. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Li X, Wen J, Dong Y, Zhang Q, Guan J, Liu
F, Zhou T, Li Z, Fan Y and Wang N: Wnt5a promotes renal tubular
inflammation in diabetic nephropathy by binding to CD146 through
noncanonical Wnt signaling. Cell Death Dis. 12:922021. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Pei D, Tian S, Bao Y, Zhang J, Xu D and
Piao M: Protective effect of salidroside on streptozotocin-induced
diabetic nephropathy by inhibiting oxidative stress and
inflammation in rats via the Akt/GSK-3β signalling pathway. Pharm
Biol. 60:1732–1738. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Han X, Wei J, Zheng R, Tu Y, Wang M, Chen
L, Xu Z, Zheng L, Zheng C, Shi Q, et al: Macrophage SHP2 deficiency
alleviates diabetic nephropathy via suppression of
MAPK/NF-κB-dependent inflammation. Diabetes. 73:780–796. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Li J, Zhang J, Zhao X and Tian L: MSU
crystallization promotes fibroblast proliferation and renal
fibrosis in diabetic nephropathy via the ROS/SHP2/TGFβ pathway. Sci
Rep. 14:202512024. View Article : Google Scholar
|
|
213
|
Wei TT, Yang LT, Guo F, Tao SB, Cheng L,
Huang RS, Ma L and Fu P: Activation of GPR120 in podocytes
ameliorates kidney fibrosis and inflammation in diabetic
nephropathy. Acta Pharmacol Sin. 42:252–263. 2021. View Article : Google Scholar
|
|
214
|
Yang WX, Liu Y, Zhang SM, Wang HF, Liu YF,
Liu JL, Li XH, Zeng MR, Han YZ, Liu FY, et al: Epac activation
ameliorates tubulointerstitial inflammation in diabetic
nephropathy. Acta Pharmacol Sin. 43:659–671. 2022. View Article : Google Scholar
|
|
215
|
Reddy MA, Sumanth P, Lanting L, Yuan H,
Wang M, Mar D, Alpers CE, Bomsztyk K and Natarajan R: Losartan
reverses permissive epigenetic changes in renal glomeruli of
diabetic db/db mice. Kidney Int. 85:362–373. 2014. View Article : Google Scholar :
|
|
216
|
Huang J, Wan D, Li J, Chen H, Huang K and
Zheng L: Histone acetyltransferase PCAF regulates inflammatory
molecules in the development of renal injury. Epigenetics.
10:62–72. 2015. View Article : Google Scholar
|
|
217
|
Pandey A and Gaikwad AB: Compound 21 and
telmisartan combination mitigates type 2 diabetic nephropathy
through amelioration of caspase mediated apoptosis. Biochem Biophys
Res Commun. 487:827–833. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Nicholas SB, Aguiniga E, Ren Y, Kim J,
Wong J, Govindarajan N, Noda M, Wang W, Kawano Y, Collins A and
Hsueh WA: Plasminogen activator inhibitor-1 deficiency retards
diabetic nephropathy. Kidney Int. 67:1297–1307. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Chen H, Li J, Jiao L, Petersen RB, Li J,
Peng A, Zheng L and Huang K: Apelin inhibits the development of
diabetic nephropathy by regulating histone acetylation in Akita
mouse. J Physiol. 592:505–521. 2014. View Article : Google Scholar
|
|
220
|
Du YG, Wang LP, Qian JW, Zhang KN and Chai
KF: Panax notoginseng saponins protect kidney from diabetes by
up-regulating silent information regulator 1 and activating
antioxidant proteins in rats. Chin J Integr Med. 22:910–917. 2016.
View Article : Google Scholar
|
|
221
|
Kitada M, Takeda A, Nagai T, Ito H,
Kanasaki K and Koya D: Dietary restriction ameliorates diabetic
nephropathy through anti-inflammatory effects and regulation of the
autophagy via restoration of Sirt1 in diabetic Wistar fatty (fa/fa)
rats: A model of type 2 diabetes. Exp Diabetes Res.
2011:9081852011. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Du YG, Zhang KN, Gao ZL, Dai F, Wu XX and
Chai KF: Tangshen formula improves inflammation in renal tissue of
diabetic nephropathy through SIRT1/NF-κB pathway. Exp Ther Med.
15:2156–2164. 2018.PubMed/NCBI
|
|
223
|
Liu HW, Kao HH and Wu CH: Exercise
training upregulates SIRT1 to attenuate inflammation and metabolic
dysfunction in kidney and liver of diabetic db/db mice. Nutr Metab
(Lond). 16:222019. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Li X, Liu J, Lu L, Huang T, Hou W, Wang F,
Yu L, Wu F, Qi J, Chen X, et al: Sirt7 associates with ELK1 to
participate in hyperglycemia memory and diabetic nephropathy via
modulation of DAPK3 expression and endothelial inflammation. Transl
Res. 247:99–116. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Sankrityayan H, Kale A, Shelke V and
Gaikwad AB: Cyproheptadine, a SET7/9 inhibitor, reduces
hyperglycaemia-induced ER stress alleviating inflammation and
fibrosis in renal tubular epithelial cells. Arch Physiol Biochem.
130:411–419. 2024. View Article : Google Scholar
|
|
226
|
Brasacchio D, Okabe J, Tikellis C,
Balcerczyk A, George P, Baker EK, Calkin AC, Brownlee M, Cooper ME
and El-Osta A: Hyperglycemia induces a dynamic cooperativity of
histone methylase and demethylase enzymes associated with
gene-activating epigenetic marks that coexist on the lysine tail.
Diabetes. 58:1229–1236. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Wang F, Hou W, Li X, Lu L, Huang T, Zhu M
and Miao C: SETD8 cooperates with MZF1 to participate in
hyperglycemia-induced endothelial inflammation via elevation of
WNT5A levels in diabetic nephropathy. Cell Mol Biol Lett.
27:302022. View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Hou W, Lu L, Li X, Sun M, Zhu M and Miao
C: c-Myc participates in high glucose-mediated endothelial
inflammation via upregulation of IRAK1 expression in diabetic
nephropathy. Cell Signal. 92:1102632022. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Huang T, Li X, Wang F, Lu L, Hou W, Zhu M
and Miao C: The CREB/KMT5A complex regulates PTP1B to modulate high
glucose-induced endothelial inflammatory factor levels in diabetic
nephropathy. Cell Death Dis. 12:3332021. View Article : Google Scholar : PubMed/NCBI
|
|
230
|
Wang Y, Xu J and Cheng Z: YAP1 promotes
high glucose-induced inflammation and extracellular matrix
deposition in glomerular mesangial cells by modulating NF-κB/JMJD3
pathway. Exp Ther Med. 22:13492021. View Article : Google Scholar
|
|
231
|
Zheng W, Guo J and Liu ZS: Effects of
metabolic memory on inflammation and fibrosis associated with
diabetic kidney disease: An epigenetic perspective. Clin
Epigenetics. 13:872021. View Article : Google Scholar : PubMed/NCBI
|
|
232
|
Zhao Y, Fan S, Zhu H, Zhao Q, Fang Z, Xu
D, Lin W, Lin L, Hu X, Wu G, et al: Podocyte OTUD5 alleviates
diabetic kidney disease through deubiquitinating TAK1 and reducing
podocyte inflammation and injury. Nat Commun. 15:54412024.
View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Wang B, Dai Z, Gao Q, Liu Y, Gu G and
Zheng H: Spop ameliorates diabetic nephropathy through restraining
NLRP3 inflammasome. Biochem Biophys Res Commun. 594:131–138. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
234
|
Wu W, Huang XR, You Y, Xue L, Wang XJ,
Meng X, Lin X, Shen J, Yu X, Lan HY and Chen H: Latent TGF-β1
protects against diabetic kidney disease via Arkadia/Smad7
signaling. Int J Biol Sci. 17:3583–3594. 2021. View Article : Google Scholar
|
|
235
|
Zhang X, Wang S, Chong N, Chen D, Shu J,
Sun J, Sun Z, Wang R, Wang Q and Xu Y: GDF-15 alleviates diabetic
nephropathy via inhibiting NEDD4L-mediated IKK/NF-κB signalling
pathways. Int Immunopharmacol. 128:1114272024. View Article : Google Scholar
|
|
236
|
Chen K, Chen J, Wang L, Yang J, Xiao F,
Wang X, Yuan J, Wang L and He Y: Parkin ubiquitinates GATA4 and
attenuates the GATA4/GAS1 signaling and detrimental effects on
diabetic nephropathy. FASEB J. 34:8858–8875. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Guo F, Song Y, Wu L, Zhao Y, Ma X, Wang J,
Shao M, Ji H, Huang F, Fan X, et al: SUMO specific peptidase 6
regulates the crosstalk between podocytes and glomerular
endothelial cells in diabetic kidney disease. Biochim Biophys Acta
Mol Basis Dis. 1869:1666852023. View Article : Google Scholar : PubMed/NCBI
|
|
238
|
Huang W, Xu L, Zhou X, Gao C, Yang M, Chen
G, Zhu J, Jiang L, Gan H, Gou F, et al: High glucose induces
activation of NF-κB inflammatory signaling through IκBα sumoylation
in rat mesangial cells. Biochem Biophys Res Commun. 438:568–574.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
239
|
Huang W, Liang Y, Dong J, Zhou L, Gao C,
Jiang C, Chen M, Long Y and Xu Y: SUMO E3 Ligase PIASy mediates
high glucose-induced activation of NF-κB inflammatory signaling in
rat mesangial cells. Mediators Inflamm. 2017:16851942017.
View Article : Google Scholar
|
|
240
|
Yao D, Taguchi T, Matsumura T, Pestell R,
Edelstein D, Giardino I, Suske G, Rabbani N, Thornalley PJ, Sarthy
VP, et al: High glucose increases angiopoietin-2 transcription in
microvascular endothelial cells through methylglyoxal modification
of mSin3A. J Biol Chem. 282:31038–31045. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
241
|
He Y, Xie Y, Zhou T, Li D, Cheng X, Yang
P, Luo C, Liu Y, Guo M, Wan Q, et al: Sodium crotonate alleviates
diabetic kidney disease partially via the histone crotonylation
pathway. Inflammation. 48:254–275. 2025. View Article : Google Scholar
|
|
242
|
Zhang XX, Liu Y, Xu SS, Yang R, Jiang CH,
Zhu LP, Xu YY, Pan K, Zhang J and Yin ZQ: Asiatic acid from
Cyclocarya paliurus regulates the autophagy-lysosome system via
directly inhibiting TGF-β type I receptor and ameliorates diabetic
nephropathy fibrosis. Food Funct. 13:5536–5546. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
243
|
Zhu QQ, Yang XY, Zhang XJ, Yu CJ, Pang QQ,
Huang YW, Wang XJ and Sheng J: EGCG targeting Notch to attenuate
renal fibrosis via inhibition of TGFβ/Smad3 signaling pathway
activation in streptozotocin-induced diabetic mice. Food Funct.
11:9686–9695. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
244
|
Das F, Ghosh-Choudhury N, Maity S,
Kasinath BS and Choudhury GG: Oncoprotein DJ-1 interacts with mTOR
complexes to effect transcription factor Hif1α-dependent expression
of collagen I (α2) during renal fibrosis. J Biol Chem.
298:1022462022. View Article : Google Scholar
|
|
245
|
Wu L, Liu C, Chang DY, Zhan R, Zhao M, Man
Lam S, Shui G, Zhao MH, Zheng L and Chen M: The attenuation of
diabetic nephropathy by annexin A1 via regulation of lipid
metabolism through the AMPK/PPARα/CPT1b pathway. Diabetes.
70:2192–2203. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
246
|
Cho W, Oh H, Choi SW, Abd El-Aty AM,
Birdal O, Jeong JH, Song JH and Jung TW: CTRP4 attenuates apoptosis
and epithelial-mesenchymal transition markers in podocytes through
an AMPK/autophagy-dependent pathway. Biochem Biophys Res Commun.
682:104–110. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
247
|
Han Y, Xiong S, Zhao H, Yang S, Yang M,
Zhu X, Jiang N, Xiong X, Gao P, Wei L, et al: Lipophagy deficiency
exacerbates ectopic lipid accumulation and tubular cells injury in
diabetic nephropathy. Cell Death Dis. 12:10312021. View Article : Google Scholar : PubMed/NCBI
|
|
248
|
Chen J, Chen KH, Wang LM, Luo J, Zheng QY
and He YN: Decoy receptor 2 mediates the apoptosis-resistant
phenotype of senescent renal tubular cells and accelerates renal
fibrosis in diabetic nephropathy. Cell Death Dis. 13:5222022.
View Article : Google Scholar : PubMed/NCBI
|
|
249
|
Li J, Jin S, Barati MT, Rane S, Lin Q, Tan
Y, Cai L and Rane MJ: ERK and p38 MAPK inhibition controls NF-E2
degradation and profibrotic signaling in renal proximal tubule
cells. Life Sci. 287:1200922021. View Article : Google Scholar : PubMed/NCBI
|
|
250
|
Zhang L, Zang CS, Chen B, Wang Y, Xue S
and Wu MY: Renalase regulates renal tubular injury in diabetic
nephropathy via the p38MAPK signaling pathway. FASEB J.
37:e231882023. View Article : Google Scholar : PubMed/NCBI
|
|
251
|
Wen Y, Liu Y, Huang Q, Liu R, Liu J, Zhang
F, Liu S and Jiang Y: Moringa oleifera Lam. seed extract protects
kidney function in rats with diabetic nephropathy by increasing
GSK-3β activity and activating the Nrf2/HO-1 pathway.
Phytomedicine. 95:1538562022. View Article : Google Scholar
|
|
252
|
Shin JH, Kim KM, Jeong JU, Shin JM, Kang
JH, Bang K and Kim JH: Nrf2-heme oxygenase-1 attenuates
high-glucose-induced epithelial-to-mesenchymal transition of renal
tubule cells by inhibiting ROS-Mediated PI3K/Akt/GSK-3β signaling.
J Diabetes Res. 2019:25101052019. View Article : Google Scholar
|
|
253
|
Zeng J, Liang L, Chen R, Li C, Pan L, Wen
M, Lv D, Liu M, Xu Z and Huang H: Fraxin represses NF-κB pathway
via inhibiting the activation of epidermal growth factor receptor
to ameliorate diabetic renal tubulointerstitial fibrosis. Eur J
Pharmacol. 955:1759152023. View Article : Google Scholar
|
|
254
|
Ram C, Gairola S, Verma S, Mugale MN,
Bonam SR, Murty US and Sahu BD: Biochanin A ameliorates nephropathy
in high-fat diet/streptozotocin-induced diabetic rats: Effects on
NF-kB/NLRP3 axis, pyroptosis, and fibrosis. Antioxidants (Basel).
12:10522023. View Article : Google Scholar : PubMed/NCBI
|
|
255
|
Qu X, Zhai B, Liu Y, Chen Y, Xie Z, Wang
Q, Wu Y, Liu Z, Chen J, Mei S, et al: Pyrroloquinoline quinone
ameliorates renal fibrosis in diabetic nephropathy by inhibiting
the pyroptosis pathway in C57BL/6 mice and human kidney 2 cells.
Biomed Pharmacother. 150:1129982022. View Article : Google Scholar : PubMed/NCBI
|
|
256
|
Gu LY, Yun-Sun, Tang HT and Xu ZX:
Huangkui capsule in combination with metformin ameliorates diabetic
nephropathy via the Klotho/TGF-β1/p38MAPK signaling pathway. J
Ethnopharmacol. 281:1135482021. View Article : Google Scholar
|
|
257
|
Duan YR, Chen BP, Chen F, Yang SX, Zhu CY,
Ma YL, Li Y and Shi J: LncRNA lnc-ISG20 promotes renal fibrosis in
diabetic nephropathy by inducing AKT phosphorylation through
miR-486-5p/NFAT5. J Cell Mol Med. 25:4922–4937. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
258
|
Subathra M, Korrapati M, Howell LA, Arthur
JM, Shayman JA, Schnellmann RG and Siskind LJ: Kidney
glycosphingolipids are elevated early in diabetic nephropathy and
mediate hypertrophy of mesangial cells. Am J Physiol Renal Physiol.
309:F204–F215. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
259
|
Hsu YC, Ho C, Shih YH, Ni WC, Li YC, Chang
HC and Lin CL: Knockout of KLF10 ameliorated diabetic renal
fibrosis via downregulation of DKK-1. Molecules. 27:26442022.
View Article : Google Scholar : PubMed/NCBI
|
|
260
|
Chen X, Xiao J, Tao D, Liang Y, Chen S,
Shen L, Li S, Zheng Z, Zeng Y, Luo C, et al: Metadherin
orchestrates PKA and PKM2 to activate β-catenin signaling in
podocytes during proteinuric chronic kidney disease. Transl Res.
266:68–83. 2024. View Article : Google Scholar
|
|
261
|
Chen G, Wang T, Uttarwar L, vanKrieken R,
Li R, Chen X, Gao B, Ghayur A, Margetts P and Krepinsky JC: SREBP-1
is a novel mediator of TGFβ1 signaling in mesangial cells. J Mol
Cell Biol. 6:516–530. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
262
|
Noh H, Oh EY, Seo JY, Yu MR, Kim YO, Ha H
and Lee HB: Histone deacetylase-2 is a key regulator of diabetes-
and transforming growth factor-beta1-induced renal injury. Am J
Physiol Renal Physiol. 297:F729–F739. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
263
|
Zhang L, Chen L, Gao C, Chen E, Lightle
AR, Foulke L, Zhao B, Higgins PJ and Zhang W: Loss of histone H3
K79 methyltransferase Dot1l facilitates kidney fibrosis by
upregulating endothelin 1 through histone deacetylase 2. J Am Soc
Nephrol. 31:337–349. 2020. View Article : Google Scholar
|
|
264
|
Zheng Z, Zhang S, Chen J, Zou M, Yang Y,
Lu W, Ren S, Wang X, Dong W, Zhang Z, et al: The HDAC2/SP1/miR-205
feedback loop contributes to tubular epithelial cell extracellular
matrix production in diabetic kidney disease. Clin Sci (Lond).
136:223–238. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
265
|
Feng J, Bao L, Wang X, Li H, Chen Y, Xiao
W, Li Z, Xie L, Lu W, Jiang H, et al: Low expression of HIV genes
in podocytes accelerates the progression of diabetic kidney disease
in mice. Kidney Int. 99:914–925. 2021. View Article : Google Scholar
|
|
266
|
Wang Y, Zuo B, Wang N, Li S, Liu C and Sun
D: Calcium dobesilate mediates renal interstitial fibrosis and
delay renal peritubular capillary loss through Sirt1/p53 signaling
pathway. Biomed Pharmacother. 132:1107982020. View Article : Google Scholar : PubMed/NCBI
|
|
267
|
Zhong X and Zhang J: RUNX3-activated
apelin signaling inhibits cell proliferation and fibrosis in
diabetic nephropathy by regulation of the SIRT1/FOXO pathway.
Diabetol Metab Syndr. 16:1672024. View Article : Google Scholar : PubMed/NCBI
|
|
268
|
Srivastava SP, Li J, Takagaki Y, Kitada M,
Goodwin JE, Kanasaki K and Koya D: Endothelial SIRT3 regulates
myofibroblast metabolic shifts in diabetic kidneys. iScience.
24:1023902021. View Article : Google Scholar : PubMed/NCBI
|
|
269
|
Wang X, Ji T, Li X, Qu X and Bai S: FOXO3a
protects against kidney injury in type II diabetic nephropathy by
promoting Sirt6 expression and inhibiting Smad3 Acetylation. Oxid
Med Cell Longev. 2021:55657612021. View Article : Google Scholar : PubMed/NCBI
|
|
270
|
Yang G, Jin L, Zheng D, Tang X, Yang J,
Fan L and Xie X: Fucoxanthin alleviates oxidative stress through
Akt/Sirt1/FoxO3α signaling to inhibit HG-induced renal fibrosis in
GMCs. Mar Drugs. 17:7022019. View Article : Google Scholar
|
|
271
|
Jia Y, Reddy MA, Das S, Oh HJ, Abdollahi
M, Yuan H, Zhang E, Lanting L, Wang M and Natarajan R:
Dysregulation of histone H3 lysine 27 trimethylation in
transforming growth factor-β1-induced gene expression in mesangial
cells and diabetic kidney. J Biol Chem. 294:12695–12707. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
272
|
Zhang L, Zhao S and Zhu Y: Long noncoding
RNA growth arrest-specific transcript 5 alleviates renal fibrosis
in diabetic nephropathy by downregulating matrix metalloproteinase
9 through recruitment of enhancer of zeste homolog 2. FASEB J.
34:2703–2714. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
273
|
Fang N and Li P: O-linked
N-acetylglucosaminyltransferase OGT inhibits diabetic nephropathy
by stabilizing histone methyltransferases EZH2 via the HES1/PTEN
axis. Life Sci. 274:1192262021. View Article : Google Scholar : PubMed/NCBI
|
|
274
|
Das F, Bera A, Ghosh-Choudhury N,
Sataranatarajan K, Kamat A, Kasinath BS and Choudhury GG: High
glucose-stimulated enhancer of zeste homolog-2 (EZH2) forces
suppression of deptor to cause glomerular mesangial cell pathology.
Cell Signal. 86:1100722021. View Article : Google Scholar : PubMed/NCBI
|
|
275
|
Chen YX, Zhu SY, Huang C, Xu CY, Fang XD
and Tu WP: LncRNA Dlx6os1 accelerates diabetic nephropathy
progression by epigenetically repressing SOX6 via recruiting EZH2.
Kidney Blood Press Res. 47:177–184. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
276
|
Li X, Li C, Li X, Cui P, Li Q, Guo Q, Han
H, Liu S and Sun G: Involvement of histone lysine methylation in
p21 gene expression in rat kidney in vivo and rat mesangial cells
in vitro under diabetic conditions. J Diabetes Res.
2016:38532422016. View Article : Google Scholar : PubMed/NCBI
|
|
277
|
Goru SK and Gaikwad AB: Novel
reno-protective mechanism of Aspirin involves H2AK119
monoubiquitination and Set7 in preventing type 1 diabetic
nephropathy. Pharmacol Rep. 70:497–502. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
278
|
Yuan H, Reddy MA, Deshpande S, Jia Y, Park
JT, Lanting LL, Jin W, Kato M, Xu ZG, Das S and Natarajan R:
Epigenetic histone modifications involved in profibrotic gene
regulation by 12/15-lipoxygenase and its oxidized lipid products in
diabetic nephropathy. Antioxid Redox Signal. 24:361–375. 2016.
View Article : Google Scholar
|
|
279
|
Maxwell S, Okabe J, Kaipananickal H,
Rodriguez H, Khurana I, Al-Hasani K, Chow BSM, Pitsillou E,
Karagiannis TC, Jandeleit-Dahm K, et al: Set7 methyltransferase and
phenotypic switch in diabetic glomerular endothelial cells. J Am
Soc Nephrol. 35:733–748. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
280
|
Li X, Lu L, Hou W, Wang F, Huang T, Meng Z
and Zhu M: The SETD8/ELK1/bach1 complex regulates
hyperglycaemia-mediated EndMT in diabetic nephropathy. J Transl
Med. 20:1472022. View Article : Google Scholar : PubMed/NCBI
|
|
281
|
Lu L, Zhong Z, Gu J, Nan K, Zhu M and Miao
C: ets1 associates with KMT5A to participate in high
glucose-mediated EndMT via upregulation of PFN2 expression in
diabetic nephropathy. Mol Med. 27:742021. View Article : Google Scholar : PubMed/NCBI
|
|
282
|
Lu L, Li X, Zhong Z, Zhou W, Zhou D, Zhu M
and Miao C: KMT5A downregulation participated in High
Glucose-mediated EndMT via upregulation of ENO1 expression in
diabetic nephropathy. Int J Biol Sci. 17:4093–4107. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
283
|
Chen YY, Peng XF, Liu GY, Liu JS, Sun L,
Liu H, Xiao L and He LY: Protein arginine methyltranferase-1
induces ER stress and epithelial-mesenchymal transition in renal
tubular epithelial cells and contributes to diabetic nephropathy.
Biochim Biophys Acta Mol Basis Dis. 1865:2563–2575. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
284
|
Bai S, Xiong X, Tang B, Ji T, Li X, Qu X
and Li W: hsa-miR-199b-3p prevents the epithelial-mesenchymal
transition and dysfunction of the renal tubule by regulating
E-cadherin through targeting KDM6A in diabetic nephropathy. Oxid
Med Cell Longev. 2021:88141632021. View Article : Google Scholar : PubMed/NCBI
|
|
285
|
Hung PH, Hsu YC, Chen TH, Ho C and Lin CL:
The histone demethylase inhibitor GSK-J4 Is a therapeutic target
for the kidney fibrosis of diabetic kidney disease via DKK1
Modulation. Int J Mol Sci. 23:94072022. View Article : Google Scholar : PubMed/NCBI
|
|
286
|
Shao J, Xu H, Wu X and Xu Y: Epigenetic
activation of CTGF transcription by high glucose in renal tubular
epithelial cells is mediated by myocardin-related transcription
factor A. Cell Tissue Res. 379:549–559. 2020. View Article : Google Scholar
|
|
287
|
Dong L, Yu L and Zhong J: Histone
lysine-specific demethylase 1 induced renal fibrosis via decreasing
sirtuin 3 expression and activating TGF-β1/Smad3 pathway in
diabetic nephropathy. Diabetol Metab Syndr. 14:22022. View Article : Google Scholar
|
|
288
|
Morschhauser F, Tilly H, Chaidos A, McKay
P, Phillips T, Assouline S, Batlevi CL, Campbell P, Ribrag V, Damaj
GL, et al: Tazemetostat for patients with relapsed or refractory
follicular lymphoma: An open-label, single-arm, multicentre, phase
2 trial. Lancet Oncol. 21:1433–1442. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
289
|
Huang S, Wang Z, Zhou J, Huang J, Zhou L,
Luo J, Wan YY, Long H and Zhu B: EZH2 inhibitor GSK126 suppresses
antitumor immunity by driving production of myeloid-derived
suppressor cells. Cancer Res. 79:2009–2020. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
290
|
Chen Y, Dai R, Cheng M, Wang W, Liu C, Cao
Z, Ge Y, Wang Y and Zhang L: Status and role of the
ubiquitin-proteasome system in renal fibrosis. Biomed Pharmacother.
178:1172102024. View Article : Google Scholar : PubMed/NCBI
|
|
291
|
Lv K, Li Q, Jiang N and Chen Q: Role of
TRIM29 in disease: What is and is not known. Int Immunopharmacol.
147:1139832025. View Article : Google Scholar : PubMed/NCBI
|
|
292
|
Chen Q, Gao C, Wang M, Fei X and Zhao N:
TRIM18-regulated STAT3 signaling pathway via PTP1B promotes renal
epithelial-mesenchymal transition, inflammation, and fibrosis in
diabetic kidney disease. Front Physiol. 12:7095062021. View Article : Google Scholar : PubMed/NCBI
|
|
293
|
Li Y, Ren D, Shen Y, Zheng X and Xu G:
Altered DNA methylation of TRIM13 in diabetic nephropathy
suppresses mesangial collagen synthesis by promoting ubiquitination
of CHOP. EBioMedicine. 51:1025822020. View Article : Google Scholar : PubMed/NCBI
|
|
294
|
Chen Z, Sun X, Chen Q, Lan T, Huang K,
Xiao H, Lin Z, Yang Y, Liu P and Huang H: Connexin32 ameliorates
renal fibrosis in diabetic mice by promoting K48-linked NADPH
oxidase 4 polyubiquitination and degradation. Br J Pharmacol.
177:145–160. 2020. View Article : Google Scholar
|
|
295
|
Lin Z, Li S, Xiao H, Xu Z, Li C, Zeng J,
Wang S, Liu Z and Huang H: The degradation of TGR5 mediated by
Smurf1 contributes to diabetic nephropathy. Cell Rep.
42:1128512023. View Article : Google Scholar : PubMed/NCBI
|
|
296
|
Kim D, Nam GY, Seo E and Jun HS:
Inhibition of ChREBP ubiquitination via the ROS/Akt-dependent
downregulation of Smurf2 contributes to lysophosphatidic
acid-induced fibrosis in renal mesangial cells. J Biomed Sci.
29:312022. View Article : Google Scholar : PubMed/NCBI
|
|
297
|
Yang Y, Xiao H, Lin Z, Chen R, Li S, Li C,
Sun X, Hei Z, Gong W and Huang H: The ubiquitination of CKIP-1
mediated by Src aggravates diabetic renal fibrosis (original
article). Biochem Pharmacol. 206:1153392022. View Article : Google Scholar : PubMed/NCBI
|
|
298
|
Zhao X, He X, Wei W and Huang K: USP22
aggravated diabetic renal tubulointerstitial fibrosis progression
through deubiquitinating and stabilizing Snail1. Eur J Pharmacol.
947:1756712023. View Article : Google Scholar : PubMed/NCBI
|
|
299
|
Huang K and Zhao X: USP9X prevents
AGEs-induced upregulation of FN and TGF-β1 through activating
Nrf2-ARE pathway in rat glomerular mesangial cells. Exp Cell Res.
393:1121002020. View Article : Google Scholar
|
|
300
|
Liu Z, Nan P, Gong Y, Tian L, Zheng Y and
Wu Z: Endoplasmic reticulum stress-triggered ferroptosis via the
XBP1-Hrd1-Nrf2 pathway induces EMT progression in diabetic
nephropathy. Biomed Pharmacother. 164:1148972023. View Article : Google Scholar : PubMed/NCBI
|
|
301
|
Sun XH, Xiao HM, Zhang M, Lin ZY, Yang Y,
Chen R, Liu PQ, Huang KP and Huang HQ: USP9X deubiquitinates
connexin43 to prevent high glucose-induced
epithelial-to-mesenchymal transition in NRK-52E cells. Biochem
Pharmacol. 188:1145622021. View Article : Google Scholar : PubMed/NCBI
|
|
302
|
Chang L, Wang Q, Ju J, Li Y, Cai Q, Hao L
and Zhou Y: Magnoflorine ameliorates inflammation and fibrosis in
rats with diabetic nephropathy by mediating the stability of
lysine-specific demethylase 3A. Front Physiol. 11:5804062020.
View Article : Google Scholar
|
|
303
|
Wang Y, Mao Y, Zhang X, Liu H, Peng W,
Liang L, Shi M, Xiao Y, Zhang Y, Zhang F, et al: TAK1 may promote
the development of diabetic nephropathy by reducing the stability
of SnoN protein. Life Sci. 228:1–10. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
304
|
Peng W, Zhou X, Xu T, Mao Y, Zhang X, Liu
H, Liang L, Liu L, Liu L, Xiao Y, et al: BMP-7 ameliorates partial
epithelial-mesenchymal transition by restoring SnoN protein level
via Smad1/5 pathway in diabetic kidney disease. Cell Death Dis.
13:2542022. View Article : Google Scholar : PubMed/NCBI
|
|
305
|
Gu C, Gao F, Zhang S, Kang L, Zhang W,
Feng X, Liu J, Tian Y, Wei Q, Du Y, et al: Role of SUMOylation of
STAT1 in tubular epithelial-mesenchymal transition induced by high
glucose. Mol Med Rep. 27:422023. View Article : Google Scholar
|
|
306
|
Zhou X, Gao C, Huang W, Yang M, Chen G,
Jiang L, Gou F, Feng H, Ai N and Xu Y: High glucose induces
sumoylation of Smad4 via SUMO2/3 in mesangial cells. Biomed Res
Int. 2014:7826252014. View Article : Google Scholar : PubMed/NCBI
|
|
307
|
Zhou XQ, Huang W, Xu Y and Yang MJ: High
glucose induces sumoylation of Smad4 via SUMO2/3 in glomerular
mesangial cells. Sichuan Da Xue Xue Bao Yi Xue Ban. 45:380–385.
2014.In Chinese. PubMed/NCBI
|
|
308
|
Park MJ, Kim DI, Lim SK, Choi JH, Han HJ,
Yoon KC and Park SH: High glucose-induced O-GlcNAcylated
carbohydrate response element-binding protein (ChREBP) mediates
mesangial cell lipogenesis and fibrosis: The possible role in the
development of diabetic nephropathy. J Biol Chem. 289:13519–13530.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
309
|
Goldberg H, Whiteside C and Fantus IG:
O-linked β-N-acetylglucosamine supports p38 MAPK activation by high
glucose in glomerular mesangial cells. Am J Physiol Endocrinol
Metab. 301:E713–E726. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
310
|
Aluksanasuwan S, Plumworasawat S, Malaitad
T, Chaiyarit S and Thongboonkerd V: High glucose induces
phosphorylation and oxidation of mitochondrial proteins in renal
tubular cells: A proteomics approach. Sci Rep. 10:58432020.
View Article : Google Scholar : PubMed/NCBI
|
|
311
|
Veron D, Aggarwal PK, Li Q, Moeckel G,
Kashgarian M and Tufro A: Podocyte VEGF-A knockdown induces diffuse
glomerulosclerosis in diabetic and in eNOS knockout mice. Front
Pharmacol. 12:7888862022. View Article : Google Scholar : PubMed/NCBI
|
|
312
|
Veron D, Aggarwal PK, Velazquez H,
Kashgarian M, Moeckel G and Tufro A: Podocyte-specific VEGF-a gain
of function induces nodular glomerulosclerosis in eNOS null mice. J
Am Soc Nephrol. 25:1814–1824. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
313
|
Zhang X, Chen J, Lin R, Huang Y, Wang Z,
Xu S, Wang L, Chen F, Zhang J, Pan K and Yin Z: Lactate drives
epithelial-mesenchymal transition in diabetic kidney disease via
the H3K14la/KLF5 pathway. Redox Biol. 75:1032462024. View Article : Google Scholar : PubMed/NCBI
|
|
314
|
Hong J, Xu H, Yu L, Yu Z, Chen X, Meng Z,
Zhu J, Li J and Zhu M: AARS1-mediated lactylation of H3K18 and
STAT1 promotes ferroptosis in diabetic nephropathy. Cell Death
Differ. Sep 23–2025.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
315
|
Luo W, Yu Y, Wang H, Liu K, Wang Y, Huang
M, Xuan C, Li Y and Qi J: Up-regulation of MMP-2 by histone H3K9
β-hydroxybutyrylation to antagonize glomerulosclerosis in diabetic
rat. Acta Diabetol. 57:1501–1509. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
316
|
Lyu LX, Hu MY, Lei YQ, Pan SK, Liu DW,
Zhou SJ and Liu ZS: The mechanism of β-hydroxybutyrylation in G6PDX
regulated by β-hydroxybutyric acid on alleviating podocyte injury
induced by high glucose. Zhonghua Yi Xue Za Zhi. 105:900–906.
2025.In Chinese. PubMed/NCBI
|
|
317
|
Akimoto Y, Yan K, Miura Y, Tsumoto H, Toda
T, Fukutomi T, Sugahara D, Kudo A, Arai T, Chiba Y, et al:
O-GlcNAcylation and phosphorylation of β-actin Ser199 in diabetic
nephropathy. Am J Physiol Renal Physiol. 317:F1359–F1374. 2019.
View Article : Google Scholar
|
|
318
|
Goru SK, Kadakol A, Pandey A, Malek V,
Sharma N and Gaikwad AB: Histone H2AK119 and H2BK120
mono-ubiquitination modulate SET7/9 and SUV39H1 in type 1
diabetes-induced renal fibrosis. Biochem J. 473:3937–3949. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
319
|
Li XQ, Jin B, Liu SX, Zhu Y, Li N, Zhang
QY, Wan C, Feng Y, Xing YX, Ma KL, et al: Neddylation of RhoA
impairs its protein degradation and promotes renal interstitial
fibrosis progression in diabetic nephropathy. Acta Pharmacol Sin.
46:1692–1705. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
320
|
Qu P, Li L, Jin Q, Liu D, Qiao Y, Zhang Y,
Sun Q, Ran S, Li Z, Liu T and Peng L: Histone methylation
modification and diabetic kidney disease: Potential molecular
mechanisms and therapeutic approaches (Review). Int J Mol Med.
54:1042024. View Article : Google Scholar : PubMed/NCBI
|
|
321
|
Hu H, Ji R, Hao Y, Liu Z, Yang J, Cao Y
and Yang Q: XIAP-ULK1-mediated mitophagy modulates carnitine
metabolism to mitigate diabetic kidney disease. Autophagy.
22:207–228. 2026. View Article : Google Scholar
|
|
322
|
Memarian E, t Hart LM, Slieker RC, Lemmers
RFL, van der Heijden AA, Rutters F, Nijpels G, Schoep E, Lieverse
AG, Sijbrands EJG, et al: Plasma protein N-glycosylation is
associated with cardiovascular disease, nephropathy, and
retinopathy in type 2 diabetes. BMJ Open Diabetes Res Care.
9:e0023452021. View Article : Google Scholar : PubMed/NCBI
|
|
323
|
Azushima K, Kovalik JP, Yamaji T, Ching J,
Chng TW, Guo J, Liu JJ, Nguyen M, Sakban RB, George SE, et al:
Abnormal lactate metabolism is linked to albuminuria and kidney
injury in diabetic nephropathy. Kidney Int. 104:1135–1149. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
324
|
Chen H, Du P, Jiang T, Li Y, Li Y, Liu Y,
Yang B, Kang J, Duan J, Ma Y, et al: Identification of potential
biomarkers for diabetic nephropathy via UPLC-MS/MS-based
metabolomics. Front Endocrinol (Lausanne). 16:15816912025.
View Article : Google Scholar : PubMed/NCBI
|
|
325
|
Zhang JY, Wu J, Chen ZH, Liu SY, Li P and
Chen DQ: Acetylation in renal physiology and pathophysiology. Front
Pharmacol. 16:16601092025. View Article : Google Scholar : PubMed/NCBI
|
|
326
|
Siddhi J, Sherkhane B, Kalavala AK, Arruri
V, Velayutham R and Kumar A: Melatonin prevents diabetes-induced
nephropathy by modulating the AMPK/SIRT1 axis: Focus on autophagy
and mitochondrial dysfunction. Cell Biol Int. 46:2142–2157. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
327
|
Gu W, Wang X, Zhao H, Geng J, Li X, Zheng
K, Guan Y, Hou X, Wang C and Song G: Resveratrol ameliorates
diabetic kidney injury by reducing lipotoxicity and modulates
expression of components of the junctional adhesion
molecule-like/sirtuin 1 lipid metabolism pathway. Eur J Pharmacol.
918:1747762022. View Article : Google Scholar : PubMed/NCBI
|
|
328
|
Ren H, Shao Y, Wu C, Ma X, Lv C and Wang
Q: Metformin alleviates oxidative stress and enhances autophagy in
diabetic kidney disease via AMPK/SIRT1-FoxO1 pathway. Mol Cell
Endocrinol. 500:1106282020. View Article : Google Scholar
|
|
329
|
Qi B, Chen Y, Chai S, Lu X and Kang L:
O-linked β-N-acetylglucosamine (O-GlcNAc) modification: Emerging
pathogenesis and a therapeutic target of diabetic nephropathy.
Diabet Med. 42:e154362025. View Article : Google Scholar
|
|
330
|
Fontecha-Barriuso M, Martin-Sanchez D,
Ruiz-Andres O, Poveda J, Sanchez-Niño MD, Valiño-Rivas L,
Ruiz-Ortega M, Ortiz A and Sanz AB: Targeting epigenetic DNA and
histone modifications to treat kidney disease. Nephrol Dial
Transplant. 33:1875–1886. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
331
|
Matsui S, Yamamoto T, Takabatake Y,
Takahashi A, Namba-Hamano T, Matsuda J, Minami S, Sakai S, Yonishi
H, Nakamura J, et al: Defective autophagy and AMPK inactivation
drive ferroptosis in diabetic kidney disease. Diabetologia. Nov
28–2025.Epub ahead of print. PubMed/NCBI
|
|
332
|
Zhou X, Zang X, Ponnusamy M, Masucci MV,
Tolbert E, Gong R, Zhao TC, Liu N, Bayliss G, Dworkin LD and Zhuang
S: Enhancer of zeste homolog 2 inhibition attenuates renal fibrosis
by maintaining Smad7 and phosphatase and tensin homolog expression.
J Am Soc Nephrol. 27:2092–2108. 2016. View Article : Google Scholar
|
|
333
|
Cao J, Chen G, Qiu L, Zhang L, Jiang M,
Cheng Y, Zhang Q, Liu L, Li P, Shuang Y, et al: Efficacy and safety
of tazemetostat, an EZH2 inhibitor, in Chinese patients with
relapsed/refractory follicular lymphoma: A multicentre, single-arm,
phase 2 study. EClinicalMedicine. 87:1033992025. View Article : Google Scholar : PubMed/NCBI
|
|
334
|
Zinzani PL, Izutsu K, Mehta-Shah N, Barta
SK, Ishitsuka K, Córdoba R, Kusumoto S, Bachy E, Cwynarski K,
Gritti G, et al: Valemetostat for patients with relapsed or
refractory peripheral T-cell lymphoma (VALENTINE-PTCL01): A
multicentre, open-label, single-arm, phase 2 study. Lancet Oncol.
25:1602–1613. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
335
|
Maruyama D, Jacobsen E, Porcu P, Allen P,
Ishitsuka K, Kusumoto S, Narita T, Tobinai K, Foss F, Tsukasaki K,
et al: Valemetostat monotherapy in patients with relapsed or
refractory non-Hodgkin lymphoma: A first-in-human, multicentre,
open-label, single-arm, phase 1 study. Lancet Oncol. 25:1589–1601.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
336
|
Rong QX, Chen MT, Yang W, Huang RQ, Shu
DT, Zhang Y, Xue C, Cai YC, An X, Li HF and Shi YX: Chidamide (a
Histone Deacetylase Inhibitor) Plus abiraterone in metastatic
castration-resistant prostate cancer (mCRPC): A phase Ib trial.
MedComm (2020). 6:e704702025. View Article : Google Scholar : PubMed/NCBI
|
|
337
|
Zhang Y, Chen X, Yuan L, Zhang Y, Wu J,
Guo N, Chen X and Liu J: Down-regulation of IRAK1 attenuates
podocyte apoptosis in diabetic nephropathy through PI3K/Akt
signaling pathway. Biochem Biophys Res Commun. 506:529–535. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
338
|
Chen J, Wang X, He Q, Yang HC, Fogo AB and
Harris RC: Inhibition of transcriptional coactivator YAP Impairs
the expression and function of transcription factor WT1 in diabetic
podocyte injury. Kidney Int. 105:1200–1211. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
339
|
Zhan M, Usman I, Yu J, Ruan L, Bian X,
Yang J, Yang S, Sun L and Kanwar YS: Perturbations in mitochondrial
dynamics by p66Shc lead to renal tubular oxidative injury in human
diabetic nephropathy. Clin Sci (Lond). 132:1297–1314. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
340
|
Ma Y, Yan R, Wan Q, Lv B, Yang Y, Lv T and
Xin W: Inhibitor of growth 2 regulates the high glucose-induced
cell cycle arrest and epithelial-to-mesenchymal transition in renal
proximal tubular cells. J Physiol Biochem. 76:373–382. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
341
|
Du L, Qian X, Li Y, Li XZ, He LL, Xu L,
Liu YQ, Li CC, Ma P, Shu FL, et al: Sirt1 inhibits renal tubular
cell epithelial-mesenchymal transition through YY1 deacetylation in
diabetic nephropathy. Acta Pharmacol Sin. 42:242–251. 2021.
View Article : Google Scholar
|
|
342
|
Xu H, Wu X, Qin H, Tian W, Chen J, Sun L,
Fang M and Xu Y: Myocardin-related transcription factor a
epigenetically regulates renal fibrosis in diabetic nephropathy. J
Am Soc Nephrol. 26:1648–1660. 2015. View Article : Google Scholar
|
|
343
|
Li Y, Hu Q, Li C, Liang K, Xiang Y, Hsiao
H, Nguyen TK, Park PK, Egranov SD, Ambati CR, et al: PTEN-induced
partial epithelial-mesenchymal transition drives diabetic kidney
disease. J Clin Invest. 129:1129–1151. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
344
|
Pontrelli P, Conserva F, Menghini R,
Rossini M, Stasi A, Divella C, Casagrande V, Cinefra C, Barozzino
M, Simone S, et al: Inhibition of lysine 63 ubiquitination prevents
the progression of renal fibrosis in diabetic DBA/2J mice. Int J
Mol Sci. 22:51942021. View Article : Google Scholar : PubMed/NCBI
|