Osteoporosis is a systemic skeletal disease
characterized by decreased bone mass, degradation of bone
microstructural integrity, increased bone fragility and a
heightened risk of fracture (1).
Its onset and progression are influenced by a range of factors,
including genetic predisposition, sex, aging, physical inactivity
and dietary deficiency, especially an inadequate intake of calcium
and vitamin D (2,3). As global life expectancy is
increasing, age-associated osteoporosis has become a growing public
health concern (4,5).
Current treatment strategies primarily rely on
pharmacological agents, such as bisphosphonates, calcitonin and
selective estrogen receptor modulators (6,7).
However, these therapies are often associated with high cost and
marked side effects, leading to reduced patient adherence and
limited long-term efficacy (8-10). Consequently, there is need to
explore safer, more effective and economically viable alternative
therapies, including non-pharmacological interventions such as
exercise.
Gut microbiota are a key player in various metabolic
disorders, including osteoporosis (11,12). Increasing evidence suggests that
gut microbiota and its metabolites contribute to bone health
through diverse mechanisms, including immune modulation, endocrine
regulation and nutrient absorption (13,14). Notably, individuals with
osteoporosis have distinct gut microbiota compositions compared
with healthy controls (15) and
microbial dysbiosis may disrupt host metabolism and immune
homeostasis, which has a negative impact on bone turnover (16,17).
Interventions targeting the gut microbiota,
including probiotic supplementation, microbial colonization and
exercise, have promise in modulating bone health (18-22). In particular, exercise has been
recognized for its ability to mitigate bone loss through by
mechanical loading on the skeleton, thereby enhancing calcium
absorption and regulating bone-associated signaling pathways
(5,23-25). Attention has turned to the
gut-bone axis, with studies suggesting that exercise-induced
changes in gut microbiota may mediate part of these bone-protective
effects (3,10,13,17,20,26).
Despite these advances, however, the underlying
mechanisms that link gut microbiota, exercise and bone metabolism
remain incompletely understood. Moreover, clinical and
translational studies in this area are limited. The present review
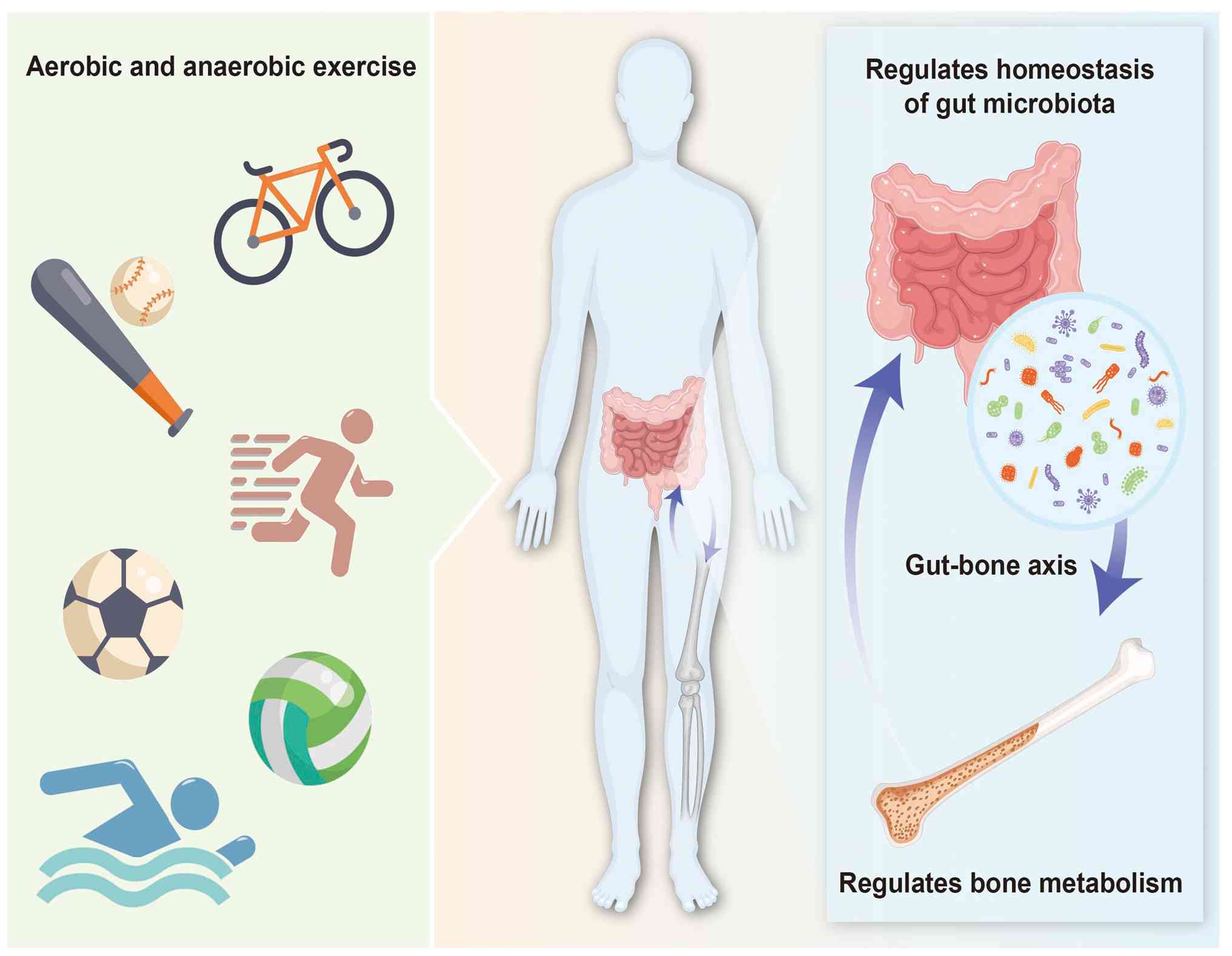
summarizes how exercise influences the gut microbiota, and how
these changes affect bone metabolism, with the goals of informing
future research and developing potential therapeutic strategies for
osteoporosis. A conceptual overview of how different exercise
modalities influence bone metabolism via the gut microbiota is
shown in Fig. 1.
Gut microbiota constitute a complex and dynamic
microbial ecosystem, comprising primarily bacteria alongside
archaea, fungi, viruses and other microorganisms (27). Throughout human evolution, the
gut microbiota has formed a mutually beneficial relationship with
the host, serving essential roles in physiological processes, such
as maintaining intestinal barrier integrity, regulating immune
responses and contributing to metabolic functions (28,29).
The composition and function of the gut microbiota
are influenced by internal and external factors, including diet,
genetics, age, physical activity and exposure to environmental
agents (30). Alterations in
microbial composition, commonly referred to as 'dysbiosis', have
been implicated in the pathogenesis of numerous types of disease,
including neurodegenerative and metabolic disorders, as well as
osteoporosis (31).
Exercise is a non-pharmacological intervention
capable of modifying gut microbiota composition and diversity. Both
aerobic and resistance training increase the abundance of
health-promoting bacterial genera, including Lactobacillus,
Bifidobacterium and Akkermansia, which reduces
systemic inflammation (32,33). These findings suggested that
exercise-induced modulation of the gut microbiota may serve as a
key mechanism in the prevention and management of bone metabolic
disorders.
Gut microbiota is predominantly composed of five
major bacterial phyla: Firmicutes, Proteobacteria, Bacteroidetes,
Actinobacteria and Verrucomicrobia (34,35).
Other gut residents include archaea, which mediate
methane metabolism and modulate intestinal gas production, as well
as fungi and viruses that maintain microecological balance and
regulate bacterial dynamics via bacteriophage interactions
(41-43).
Gut microbiota exerts physiological functions
largely through the production of metabolites and interactions with
host cells. SCFAs, including acetate, propionate and butyrate,
which are derived from the fermentation of dietary fibers, serve
key roles, modulating intestinal pH, suppressing pathogenic
bacteria and maintaining epithelial integrity and immune balance
(44). Certain gut microbiota
species contribute to the biosynthesis of vitamins (for example, B
vitamins and vitamin K) and regulate bile acid metabolism, thereby
influencing lipid absorption and energy homeostasis (45). Furthermore, probiotics such as
Lactobacillus and Bifidobacterium reinforce the
mucosal immune barrier and suppress inflammatory cytokine
production (46,47).
Exercise has been shown to exert a positive
influence on gut microbiota composition and function: Voluntary
wheel running in mice increases the numbers of SCFA-producing
bacteria, whereas treadmill exercise in moderate treadmill exercise
in Sprague-Dawley rats enhances epithelial barrier function and
microbial diversity (48,49).
Collectively, these findings support the role of physical activity
as a key modulator of host-microbiota interactions.
Accumulating evidence has suggested the gut
microbiota serves a critical role in the regulation of bone
metabolism, mediated via immune, metabolic and endocrine pathways
(50-52). One mechanism involves the
integrity of the intestinal barrier (53). Dysbiosis has been shown to
increase intestinal permeability, facilitating the translocation of
bacterial components, such as lipopolysaccharide (LPS), into
systemic circulation (54). This
activates immune responses, elevating the levels of circulating
pro-inflammatory cytokines, including tumor necrosis factor
(TNF)-α, IL-6 and IL-1β, which leads to the promotion of osteoclast
differentiation and bone resorption (55,56). SCFAs, a major group of microbial
metabolites, serve an important role in bone homeostasis. By acting
through G protein-coupled receptors, SCFAs decrease inflammation,
support intestinal integrity and promote osteoblast
differentiation, while inhibiting osteoclastogenesis (57-60). The gut microbiota also regulates
bone metabolism through endocrine and metabolic pathways. For
example, bile acids, which serve metabolic roles, also act through
receptors such as the farnesoid X receptor (FXR) and the G
protein-coupled receptor TGR5, which are involved in endocrine
signaling, which enhances osteoblast differentiation and
mineralization (61,62). Furthermore, gut microbiota
influence circulating levels of insulin-like growth factor-1
(IGF-1), a key hormone for bone formation and remodeling. Dysbiosis
is associated with decreased expression of IGF-1, which impairs
osteogenesis and contributes to bone loss (63).
In addition, the gut microbiota modulates immune
cell subsets involved in bone remodeling, especially regulatory T
cells (Tregs) and T helper 17 (Th17) cells. Tregs secrete
anti-inflammatory cytokines that suppress osteoclast activity,
whereas Th17 cells release pro-inflammatory mediators that promote
osteoclastogenesis. An imbalance in the Treg/Th17 ratio, typically
associated with gut microbiota dysbiosis, may therefore disrupt
bone homeostasis (67-69). Exercise may help to restore this
balance, both by enhancing gut microbiota diversity and SCFA
production, thereby improving barrier function, and by modulating
the immune system. Mechanistically, exercise-induced mechanical
loading has been shown to activate key osteogenic signaling
pathways, including the Wnt/β-catenin and morphogenetic protein
(BMP)/Smad signaling pathways, which promote osteoblast
differentiation and bone formation (24). Additionally, exercise upregulates
the expression of IGF-1 and activates the PI3K/Akt and MAPK/ERK
pathways, enhancing osteoblast proliferation and activity (63,70). Through gut microbiota-mediated
mechanisms, exercise has also been shown to increase levels of
circulating SCFAs, which activates signaling pathways mediated via
the G protein-coupled receptors GPR41 and GPR43 (60), whereas secondary bile acids
produced by gut microbes activate TGR5- and FXR-mediated signaling
pathways; all of these effects contribute to enhanced osteoblast
function and bone mineralization (61,62,71-73). Collectively, these findings
demonstrate that regular physical activity supports bone health
through integrated direct mechanical and gut microbiota-mediated
endocrine mechanisms.
The gut microbiota contributes to bone metabolism by
regulating immune responses, producing key metabolites and
influencing endocrine factors. Specifically, microbiota-derived
SCFAs modulate the Treg/Th17 cell balance to suppress
osteoclastogenesis via the osteoprotegerin/receptor activator for
nuclear factor κB ligand (RANKL) signaling pathway (67,74,75), while also activating GPR41/GPR43
signaling to inhibit bone resorption (59,60). Secondary bile acids promote
osteoblast differentiation through the TGR5 and FXR (61,62,71,72), and microbial regulation of IGF-1
enhances bone formation via the PI3K/Akt and MAPK/ERK signaling
pathways (63,70). These mechanisms highlight the key
role of gut microbiota in maintaining bone homeostasis.
Mounting evidence has demonstrated that the gut
microbiota shapes bone metabolism through intertwined immune,
endocrine and metabolic pathways (13,14). Alterations in microbial
composition and metabolite production influence the differentiation
and activity of both osteoclasts and osteoblasts, thereby
modulating skeletal homeostasis. Within the framework of the
gut-bone axis, several molecular routes have been identified via
which gut-derived signals affect bone remodeling. Among these, the
LPS/NF-κB osteoclast and SCFA GPR41/43 osteoblast signaling
pathways are the most extensively characterized, representing key
links between microbial metabolism and bone physiology (76-84).
Microbial dysbiosis compromises intestinal barrier
integrity, thereby permitting LPS from Gram-negative bacteria to
enter circulation. LPS engages toll-like receptor 4 (TLR4) on
immune and bone marrow-derived precursor cells, thereby activating
NF-κB signaling and inducing pro-inflammatory cytokines such as
TNF-α, IL-1β and IL-6 (55,56,76). These cytokines upregulate RANKL
expression, which drives osteoclast differentiation, resulting in
excessive bone resorption and microarchitectural deterioration
(76-81). Sustained NF-κB activation
amplifies inflammatory osteolysis. On the other hand, probiotics,
natural compounds (such as quercetin and chondroitin sulfate) and
moderate exercise restore barrier integrity, suppressing
LPS-induced osteoclastogenesis (82-84). This inflammatory cascade
constitutes a principal mechanism by which gut microbial imbalance
contributes to osteoporosis progression.
By contrast with the pro-resorptive effects of LPS,
SCFAs exert bone-protective effects. SCFAs enhance intestinal
barrier integrity and enter the systemic circulation, where they
activate GPR41 and GPR43 on osteoblasts and their precursors
(67,85,86). This promotes osteoblast
differentiation, mineralization and bone formation. Concurrently,
SCFAs inhibit osteoclastogenesis by modulating immune responses,
such as promoting Treg function and suppressing pro-inflammatory
signaling pathways (60,85,87). Furthermore, SCFAs influence
systemic endocrine factors, including IGF-1, thereby supporting
osteoblast activity (88-94).
Dietary interventions that increase SCFA levels, such as high fiber
intake or specific probiotics, have also been demonstrated to
support bone mass accrual and mitigate bone loss (3,95-97). This pathway underscores the
beneficial role of specific microbial metabolites in maintaining
bone anabolism.
Considered together, the LPS/NF-κB and SCFA/GPR41/43
pathways illustrate how inflammatory and anabolic signals
originating from the gut microbiota converge within the bone
microenvironment to orchestrate remodeling balance. These
interconnected mechanisms highlight the integrative role of
microbial metabolites in coupling immune and metabolic homeostasis
with skeletal function, and form the molecular foundation of the
gut-bone signaling axis by which intestinal microbiota dynamically
influence bone health and disease.
A growing body of evidence demonstrates the
association between gut microbiota and osteoporosis (22,98-100). Recent findings have proposed
that changes in the diversity, structure and metabolites produced
by gut microbial communities, including trimethylamine N-oxide
(TMAO) and bile acids, have potential both as diagnostic biomarkers
for osteoporosis and as indicators of disease severity (22,99,100). Due to its key role in the
pathogenesis of osteoporosis, the gut microbiota has emerged as a
promising therapeutic target.
The composition of the gut microbiota and its
metabolites impact osteoporosis, serving as potential diagnostic
and prognostic indicators. Although regional variations exist,
previous studies have shown distinct bacterial profiles in patients
with osteoporosis, with certain beneficial taxa
(Lactobacillus, Ruminococcus and Bacteroides)
typically reduced, while changes in broader groups such as
Firmicutes are more variable and context-dependent (22). Beneficial gut microbes,
especially those that produce SCFAs, have a positive association
with bone quality (101). In
metabolic terms, elevated levels of TMAO in postmenopausal patients
contribute to an increased susceptibility to osteoporosis and
fracture risk (102), whereas
higher levels of bile acids improve bone mineral content and
decrease bone degradation (as determined by the β-C-terminal
telopeptide of type I collagen (β-CTX) test) (103). These gut-derived factors
represent crucial elements in the gut-bone axis, offering
therapeutic avenues for osteoporosis.
Estrogen deficiency has been shown to both induce
gut microbiota dysbiosis and increase intestinal permeability,
thereby promoting osteoclast differentiation through upregulated
cytokine secretion, leading to bone loss (104). This mechanism involves gut
microbiota-mediated systemic inflammation, as evidenced by the
prevention of bone loss following antibiotic treatment (104). Germ-free and
strain-supplemented mouse models demonstrate the key role of gut
microbiota in skeletal development (105-107). Compared with conventional mice,
germ-free mice exhibit impaired bone development and decreased bone
mass. However, supplementation with specific probiotic strains such
as Lactobacillus, or fecal microbiota transplantation,
restores normal bone metabolism and promote skeletal development.
Although studies have reported inconsistent results, potentially
due to methodological differences, the majority of the accrued
evidence has confirmed that a stable gut microbiota is key for
normal bone homeostasis, highlighting the importance of microbial
composition in regulating the gut-bone axis (22,101-107).
Bone metabolic homeostasis relies on a coordinated
equilibrium between osteoblast-mediated bone formation and
osteoclast-driven bone degradation, and this is regulated by
endocrine mediators such as estrogen, vitamin D, parathyroid
hormone and inflammatory cytokines. An imbalance, characterized by
increased osteoclast activity relative to osteoblast function,
accelerates bone resorption, consequently leading to osteoporosis
(2). Gut microbiota regulate
bone homeostasis through specific pathways: The LPS-NF-κB axis
promotes osteoclastogenesis (76), whereas the SCFA-GPR41/43 axis
enhances osteoblast activity and inhibits osteoclastogenesis
(59,60). Additional mechanisms exist, which
involve bile acids activating TGR5/FXR to stimulate osteoblast
differentiation (61,62,71-73), and TMAO promoting
osteoclastogenesis via the NF-κB signaling pathway (78-80,108). Gut microbiota dysbiosis impairs
BMSC osteogenic potential through oxidative stress (109), whereas probiotics restore BMSC
function (110). Gut microbiota
and their metabolites influence osteoporosis progression by
regulating the differentiation, proliferation and apoptosis of
osteoblasts, osteoclasts and BMSCs (108-111).
Gut microbiota dysbiosis promotes osteoclastogenesis
through an increase in levels of harmful metabolites, including LPS
and TMAO, which activate NF-κB signaling to drive osteoclast
differentiation and bone resorption. Multiple interventions have
demonstrated therapeutic potential in terms of suppressing
osteoclast activity through distinct mechanisms (75-77,111). For example, quercetin and
Inonotus hispidus polypeptide restore gut barrier integrity
and suppress NF-κB activation, thereby mitigating osteoclast
formation (82,84,111), whereas chondroitin sulfate,
bone-strengthening granules and milk-derived extracellular vesicles
increase SCFA production and rebalance the Treg/Th17 cell immune
responses to inhibit osteoclast activity (75,83). These findings establish the
gut-bone axis as a key regulatory pathway, where microbial
metabolites differentially modulate osteoclast function. Therefore,
gut microbial metabolites such as LPS and TMAO promote
osteoclastogenesis and bone loss, whereas SCFAs counteract these
effects through immunomodulation and the suppression of bone
resorption. Therapeutic strategies that target the
microbiota-metabolite axis may effectively ameliorate osteoporotic
processes. Collectively, these findings highlight gut microbiota
and SCFAs as critical regulators of bone-immune crosstalk,
rendering them potential therapeutic targets for osteoporosis. A
summary of the interventions targeting osteoclasts via the gut
microbiota is shown in Table
I.
Gut microbiota influences osteoblast function
through multiple pathways where an impaired intestinal barrier
permits LPS translocation that inhibits bone formation, whereas gut
microbiota dysbiosis dysregulate the hypothalamic-pituitary-adrenal
axis, leading to elevated cortisol levels that negatively impact
osteoblast activity (112,113). Various interventions
demonstrate osteogenic potential through modulation of the gut
microbiota: Cinnamic acid increases microbial diversity and
upregulates osteoblast-specific transcription factors through
BMP/TGF-β signaling pathway activation (114,115). Furthermore, SCFAs, notably
butyrate, enhance osteoblast differentiation and function by
activating the SCFA-GPR41/43 signaling axis and downstream
pathways, such as the Wnt/β-catenin pathway, which collectively
promote bone formation (87,116-118). By contrast, gut microbiota
dysbiosis disrupts serotonin metabolism, elevating bone resorption,
whereas inhibition of gut tryptophan hydroxylase 1 decreases levels
of 5-hydroxytryptamine and promotes bone formation (119). Collectively, these mechanisms
establish the gut-bone axis as essential for maintaining osteoblast
function.
In summary, gut microbiota and their metabolites
regulate osteoblast differentiation and survival through
inflammatory, endocrine and signaling pathways (Table II). Dysbiosis disrupts these
processes, whereas restoration of the microbial balance promotes
bone formation. These findings underscore the gut-bone axis as a
key target for osteoporosis prevention and therapy.
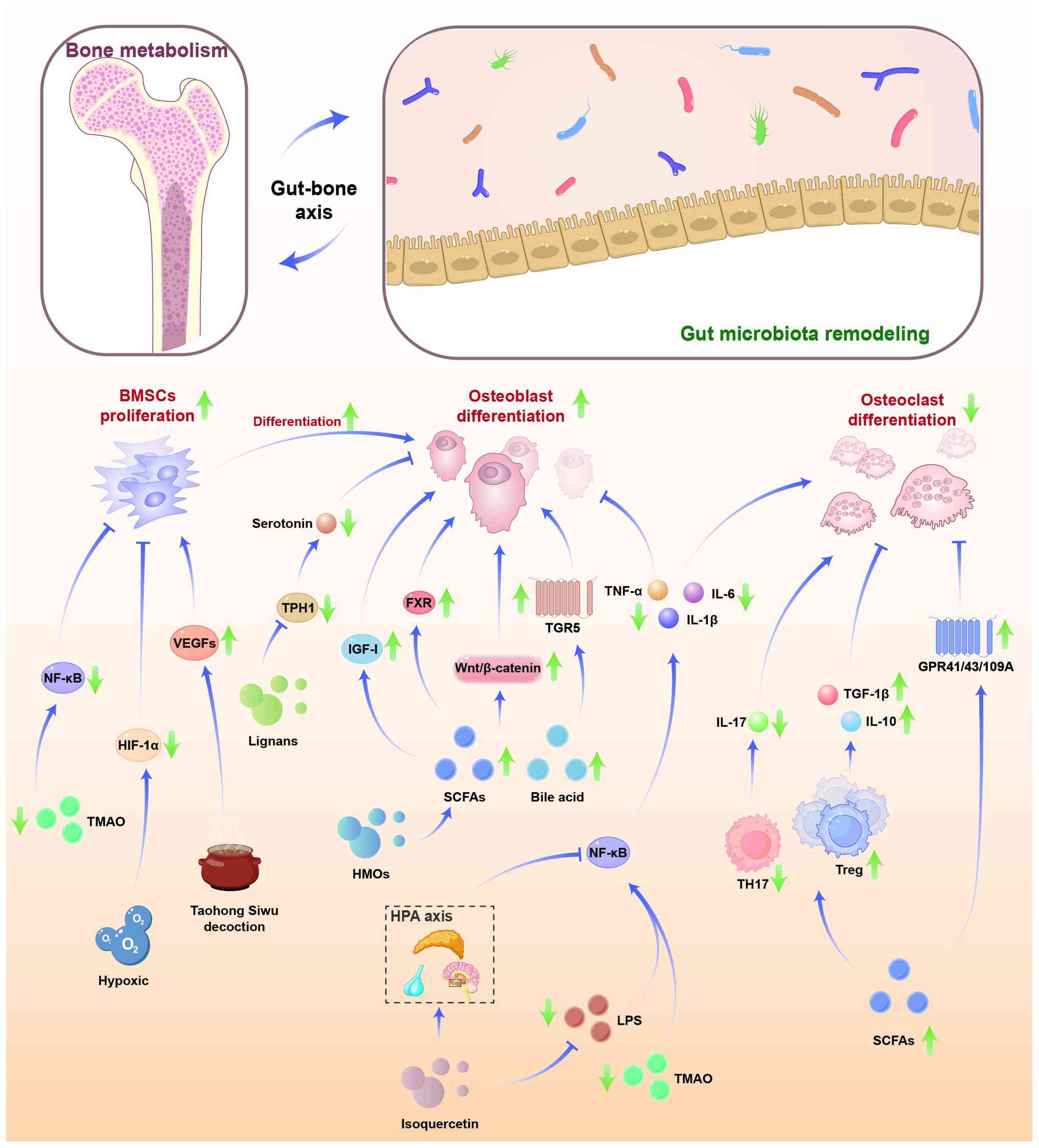
BMSCs differentiate into multiple lineages,
including osteoblasts, and their migration is key for bone
remodeling and regeneration (120,121). Under hypoxic conditions, the
adaptation of BMSCs occurs via hypoxia-inducible factor-1α (HIF-1α)
activation and exosome secretion, which modulates gut microbiota
and systemic inflammation, alleviating osteopenia (122). HIF-1α promotes glycolysis and
angiogenesis, whereas exosomes deliver regulatory microRNAs to
intestinal cells, thereby influencing microbial composition and
metabolic activity. On the other hand, dysbiosis and excessive
levels of TMAO activate NF-κB signaling, induce oxidative stress,
promote premature senescence and impair osteogenic differentiation
(109). Furthermore,
Lactobacillus animalis-derived extracellular vesicles
stimulate angiogenesis, enhance BMSC osteogenic differentiation and
inhibit osteoblast apoptosis (110), whereas Taohong Siwu decoction
was demonstrated to both regulate gut microbiota and activate
osteogenic pathways (123).
Collectively, these findings underscore gut microbiota modulation
as a promising strategy against osteoporosis.
In summary, gut microbiota and metabolites influence
BMSC function, with dysbiosis promoting senescence and impairing
osteogenesis. Interventions such as probiotic vesicles and herbal
formulations restore microbial balance and enhance bone formation.
Maintaining gut microbiota homeostasis and regulating microbial
metabolites preserve BMSC osteogenic potential, inhibit senescence
and improve bone remodeling, underscoring the gut-bone axis as a
promising therapeutic target. The interplay between gut microbiota,
their metabolites and bone cell activity is illustrated in Fig. 2.
Aerobic and resistance exercise reshape the gut
microbiota by enhancing its α-diversity and enriching the relative
abundance of beneficial bacterial genera such as Lactobacillus,
Bifidobacterium, and Akkermansia. This exercise-induced microbial
shift promotes the production of key metabolites like short-chain
fatty acids (SCFAs), thereby supporting microbiota-mediated
metabolic functions and contributing to host bone health through
the gut-bone axis (124-145).
Aerobic exercise exerts beneficial effects on gut
microbiota composition and host health. Short-term voluntary wheel
running significantly increase the abundance of Lactobacillus and
Bifidobacterium and intestinal butyrate concentrations in mice
(124). Subsequently, longer
interventions, including treadmill or jump rope exercise, enhance
microbial diversity, enrich beneficial taxa such as Firmicutes,
Lachnospiraceae, Butyricimonas, Prevotella, Akkermansia and
SCFA-producing bacteria, and modulate metabolites including
L-citrulline and L-arginine, thereby promoting bone formation and
metabolic health (125-129). In addition, human studies have
corroborated these findings: Positive correlations were identified
between cardiopulmonary fitness and microbiota diversity, with
exercise increasing the numbers of Roseburia, Lactobacillaceae and
Erysipelotrichaceae bacteria, while also improving insulin
sensitivity, decreasing endotoxemia and preventing bone loss via
microbiota-mediated mechanisms (130-134). Exercise-induced microbial
alterations are dynamic, typically being reversed upon the
cessation of activity, and this may vary with age, hormonal status
and intervention type (131,135,136). These findings suggest that
aerobic exercise promotes host health, bone metabolism by
remodeling gut microbiota, highlighting its critical role in the
gut-bone axis.
Anaerobic exercise, including resistance training
and high-intensity interval training (HIIT), has been shown to
significantly modulate gut microbiota diversity and function
(137-144). Interventions over a period of
12 weeks enhance microbial α-diversity, also adjusting the ratio of
Bacteroides/Firmicutes bacteria and protecting intestinal
integrity, although the effects vary according to genotype, obesity
and metabolic status (137-139). In addition, human studies have
confirmed that resistance training and combined HIIT improve the
microbiota composition, with synergistic responses observed between
bacterial and fungal communities (140,141). Furthermore, shorter or more
moderate intervention yields limited effects, highlighting the
importance of exercise duration and individual factors, such as
baseline fitness status, body composition, age, sex and underlying
metabolic conditions (142-144).
In summary, anaerobic exercise, including resistance
training and HIIT, modulates gut microbiota diversity and improves
beneficial microbial populations, with fungi potentially serving as
biomarkers; these microbiota-mediated effects may support metabolic
and bone health via the gut-bone axis.
Evidence from human studies provides support for
the exercise-mediated gut-bone axis (132-134). Observational studies
consistently demonstrate distinct gut microbial profiles in
physically active individuals, with athletes showing enhanced
microbial diversity and abundance of beneficial taxa such as
Akkermansia and Faecalibacterium (132,145). Cardiorespiratory fitness levels
are strongly and positively correlated with gut microbial diversity
and SCFA production capacity across various populations (130). Intervention studies have
further substantiated these findings, revealing that structured
aerobic and resistance training regimens notably modify gut
microbiota composition (129,140). These exercise-induced changes
are characterized by an increased abundance of SCFA-producing
bacteria and a decrease in pro-inflammatory taxa. Moreover, the
observed microbial alterations are associated with improved
metabolic parameters, including enhanced insulin sensitivity and
decreased systemic inflammation, both of which are key mediators of
bone metabolism (133,134). However, the extent to which
these modifications occur may be influenced by individual
characteristics, such as baseline fitness status and dietary
patterns (146).
Although available evidence has established an
exercise-microbiota connection in humans, only limited evidence
directly demonstrates its bone-protective effects (129,146). Future longitudinal studies
designed to track the complete pathway, from exercise intervention,
through microbial changes, to bone health outcomes, are essential
to validate the therapeutic potential of the gut-bone axis in
humans.
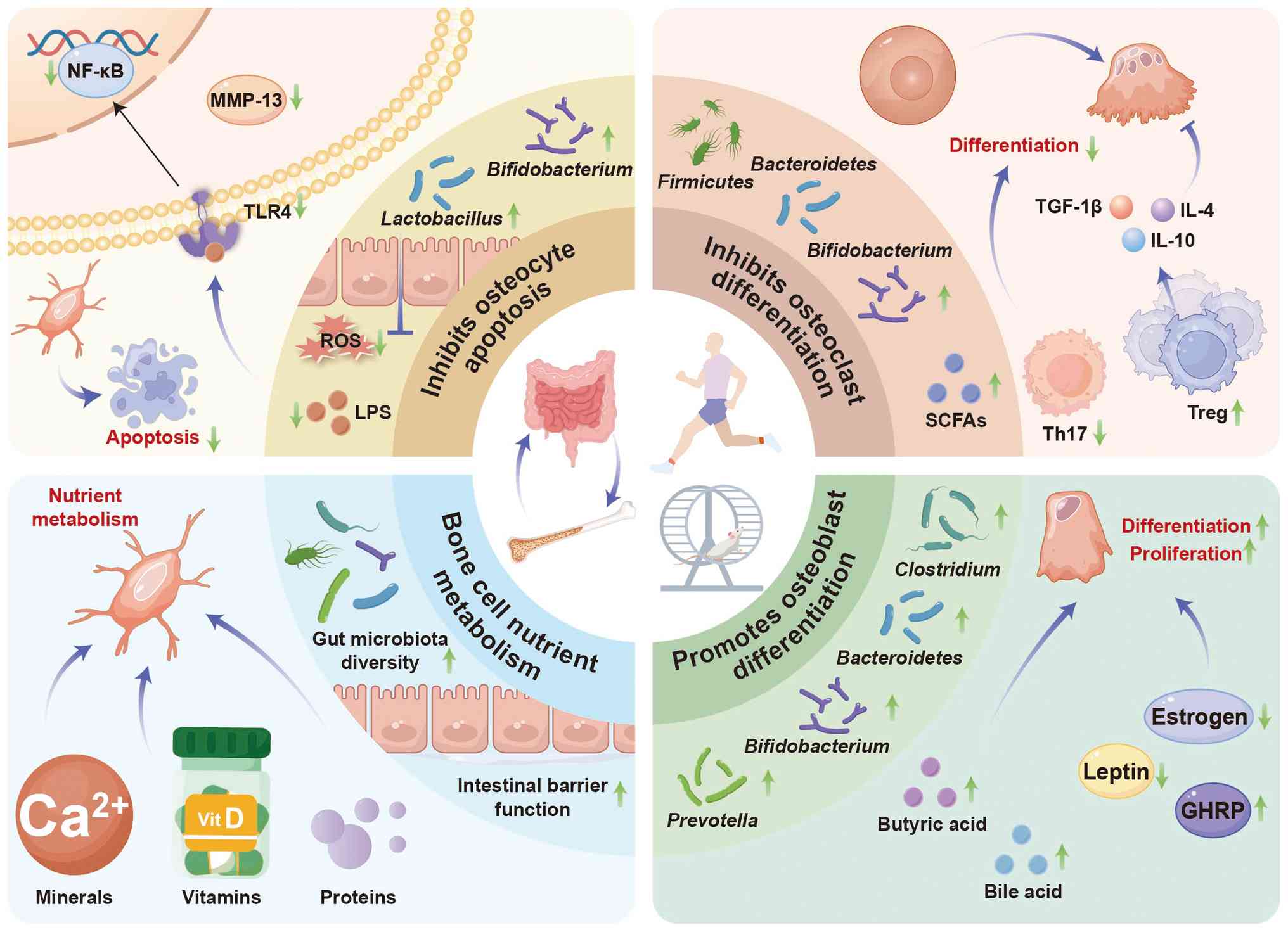
Regular exercise benefit skeletal health. A number
of studies have shown that exercise leads to an improvement in bone
health that is mediated via relevant signaling pathways, including
NF-κB, apelin, AMPK, bile acid/SCFA-mediated,
leptin/ghrelin/estrogen hormonal and Treg/Th17 immunoregulatory
pathways, and various effects have been observed, including
regulating the gut microbiota to inhibit osteocyte apoptosis,
suppressing osteoclast differentiation, promoting osteoblast
differentiation and modulating osteocyte nutrient metabolism
(147-175). The potential mechanisms by
which exercise-mediated changes in gut microbiota may improve bone
metabolism are shown in Fig. 3
and Table III.
Osteocyte apoptosis contributes to bone metabolic
imbalance and osteoporosis, making inhibition of apoptosis key for
bone health. Probiotics-fermented dairy products regulate gut
microbiota, reduce serum LPS levels and normalize both NF-κB
signaling and osteocyte apoptosis that is associated with gene
expression (Bcl-2, Bax, Caspase-3), thereby mitigating osteoporosis
(147). Exercise similarly
modulates the gut microbiota, eliciting a range of effects,
including enhancing intestinal barrier integrity, accelerating LPS
clearance and suppressing the LPS/TLR4/NF-κB inflammatory signaling
cascade, which decreases oxidative stress and osteocyte apoptosis
(84,148). Furthermore, voluntary wheel
running and high-intensity exercise increase the abundance of
beneficial microbes such as Lactobacillus and Bifidobacterium,
stabilize microbial homeostasis and enhance enzymatic and
non-enzymatic antioxidant levels, collectively alleviating
oxidative stress and protecting bone tissue (149-152). Therefore, exercise and
probiotics decrease osteocyte apoptosis by modulating gut
microbiota, thereby suppressing LPS-induced inflammation and
oxidative stress. Taken together, these findings suggest that
aerobic and high-intensity exercise attenuate osteocyte apoptosis
via gut microbiota modulation, improving redox balance and
inflammatory regulation. Although the majority of the studies that
have been performed to date are preclinical, they highlight the
gut-bone axis as a mechanistic pathway through which exercise
supports bone health, thereby providing a promising strategy for
osteoporosis prevention and therapy (84,148).
Inhibiting osteoclast proliferation and
differentiation is key for reducing bone resorption and preventing
osteoporosis (153). Exercise
modulates gut microbiota to achieve this effect through multiple
mechanisms. Desai et al (154) found that the combination of
specific cyclooxygenase-2 inhibitors and loaded exercise decreases
joint swelling in arthritis without interfering with the
bone-protective effects of loaded exercise, although this does not
directly inhibit osteoclast activity or number. In other studies,
resistance training suppressed osteoclast activation via the
inhibition of tartrate-resistant acid phosphatase and modulation of
the Fos/Fosb and calcitonin receptor pathways (155), whereas voluntary wheel running
was shown to decrease the ratio of Firmicutes/Bacteroidetes
bacteria, enrich the population of Bifidobacteriaceae and enhance
bone biomechanical properties (156,157). In addition, exercise regulates
the Treg/Th17 cell balance through multiple molecular and
immunological mechanisms. Physical activity increases the numbers
of circulating SCFAs, especially butyrate, which promote Treg
differentiation through histone deacetylase inhibition and GPR43
signaling (74,158). Concurrently, exercise decreases
the populations of pro-inflammatory cytokines, including IL-6 and
IL-23, thereby suppressing Th17 cell differentiation (74,159). Additionally, exercise-induced
metabolic adaptations enhance mitochondrial function in T cells,
favoring Treg development over Th17 lineage commitment through
AMPK-mediated metabolic reprogramming (159). These coordinated mechanisms
contribute to the maintenance of immune homeostasis and bone
health. Simultaneously, exercise promotes osteoblast
differentiation by enhancing bile acid and butyrate production,
modulating hormone secretion (for example, leptin, ghrelin and
estrogen), and activating signaling pathways, such as the apelin
signaling pathway (134,160-167).
In summary, exercise modulates gut microbiota to
suppress osteoclastogenesis and promote osteoblast activity through
immunomodulatory, metabolic and endocrine signaling pathways.
Diverse exercise modalities simultaneously inhibit osteoclast
differentiation and promote osteoblast activity, mediated via the
gut microbiota-mediated modulation of immune, endocrine and
metabolic pathways. These findings underscore the gut-bone axis as
a key therapeutic target for osteoporosis prevention.
Exercise has been shown to modulate gut microbial
homeostasis and endocrine hormone secretion, thereby activating
osteoblast functions and promoting proliferation (124,160-167). A 6-month walking intervention
combined with isoflavone supplementation in postmenopausal patients
was found to improve their body composition, lipid metabolism and
osteoblast activity (133,160,161). In ovariectomized mice, 8 weeks
of treadmill exercise was shown to increase the abundance of
Firmicutes bacteria and levels of bile acid metabolites and to
activate the apelin signaling pathway, collectively enhancing
osteoblast differentiation while suppressing adipocyte formation
(20). Aerobic exercise
regulates leptin, ghrelin and estrogen levels, thereby restoring
gut microbiota equilibrium, improving insulin sensitivity and
decreasing endotoxin levels, which all contribute to bone and
metabolic health (124,162,163). Microbial metabolites, including
bile acids and butyrate, are also enriched by exercise; this
stimulates osteoblast differentiation through SCFA-producing
bacteria such as Ruminococcus and Akkermansia (131,164-167).
In summary, exercise modulates gut microbiota
composition and endocrine function, thereby enhancing osteoblast
activity and bone formation. Through interventions such as walking
and treadmill running, exercise increases the levels of beneficial
microbial metabolites such as bile acids and SCFAs, which activate
osteogenic signaling pathways and suppress adipogenesis.
Collectively, these findings underscore the role of
exercise-induced gut microbiota modulation in promoting osteoblast
differentiation, highlighting the gut-bone axis as a key mediator
of skeletal health.
Adequate intake of vitamin D and calcium is key for
skeletal integrity, whereas deficiencies impair bone growth and
development (168). Exercise
modulates gut microbiota composition, strengthens the intestinal
barrier and optimizes nutrient absorption, which is critical for
osteogenesis (169).
Furthermore, resistance training combined with synbiotic
supplementation increases microbiota diversity, enhances SCFA
production, improves immune regulation and promotes calcium uptake,
thereby benefiting bone health (170). In addition, prolonged
moderate-intensity treadmill or swimming exercise improves the gut
microbial balance, thereby upregulating antimicrobial peptides and
tight junction proteins, decreasing inflammation and mitigating
intestinal barrier damage caused by high-fat diet or stress
(129,171,172). SCFAs, secretion of which is
promoted by exercise, increase the rate of calcium absorption by
bone (132,173,174).
In summary, exercise enhances gut barrier integrity
and microbiota diversity, thereby improving the absorption of key
nutrients such as calcium and vitamin D. Considered altogether,
these findings demonstrated that appropriately dosed exercise
supports gut microbiota-mediated nutrient absorption, reinforcing
bone cell function and skeletal health.
The gut-bone axis is a key regulator of skeletal
homeostasis. Changes in gut microbial composition and metabolite
output influence osteoclastogenesis, osteoblast function and MSC
differentiation via intertwined immune, endocrine and metabolic
pathways. Exercise modifies the intestinal microbiota and its
metabolites, thereby exerting multifaceted effects on bone through
the enhancement of barrier integrity, attenuation of systemic
inflammation and promotion of osteogenic signaling. Nevertheless,
gaps in knowledge are impeding clinical translation. Much of the
mechanistic evidence has been derived from animal models and the
corresponding human data are relatively sparse and heterogeneous.
Inter-individual differences (for example, those associated with
age, sex, diet, comorbidity and medication use) and methodological
variability (for example, microbiome assay, metabolite
quantification, exercise protocols and bone outcome measures)
impede cross-study comparisons and causal inference. Longitudinal
human cohort studies that combine metagenomics, metabolomics and
immunophenotyping with validated bone endpoints should be
undertaken to identify reproducible microbial and metabolite
biomarkers. Randomized, well-powered intervention trials testing
microbiota-directed strategies (for example, precision probiotics,
prebiotics, dietary fiber intervention and structured exercise
regimens) with standardized bone outcomes and stratification by
baseline microbiome features should be performed. Translational
pipelines that iterate between human observational signals and
mechanistic validations in controlled models to establish causality
are also required. In addition, methodological harmonization and
data sharing may improve reproducibility and clinical application.
In short, integrating microbiome science with exercise physiology
and bone biology offers a realistic route towards novel,
non-pharmacological approaches for osteoporosis prevention and
treatment. Focused translational research and standardized clinical
testing are key to realize the therapeutic potential of the
gut-bone axis.
Not applicable.
JiW and YR wrote and revised the manuscript. JuW
and SZ conceived and designed the study. YL, XY, BX, YW, YS and YX
revised the manuscript. Data authentication is not applicable. All
authors have read and approved the final manuscript.
Not applicable.
Not applicable.
The authors declare they have are no competing
interests.
Not applicable.
The present study was supported by the National Natural Science
Foundation of China (grant nos. 32101964, 32372302 and 82405210),
The Natural Science Foundation of Jiangsu Province (grant no.
BK20241755), Top Talent Support Program for Young and Middle-Aged
People of Wuxi Health Committee (grant no. BJ2023046) and Wuxi
Taihu Lake Talent Plan, Supports for Leading Talents in Medical and
Health Profession.
|
1
|
Lane NE: Epidemiology, etiology, and
diagnosis of osteoporosis. Am J Obstet Gynecol. 194(2 Suppl):
S3–S11. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kendler DL, Marin F, Zerbini C, Russo LA,
Greenspan SL, Zikan V, Bagur A, Malouf-Sierra J, Lakatos P,
Fahrleitner-Pammer A, et al: Effects of teriparatide and
risedronate on new fractures in post-menopausal women with severe
osteoporosis (VERO): A multicentre, double-blind, double-dummy,
randomised controlled trial. Lancet. 391:230–240. 2018. View Article : Google Scholar
|
|
3
|
Zhang YW, Song PR, Wang SC, Liu H, Shi ZM
and Su JC: Diets intervene osteoporosis via gut-bone axis. Gut
Microbes. 16:22954322024. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Inoue S, Hatakeyama J, Aoki H, Kuroki H,
Niikura T, Oe K, Fukui T, Kuroda R, Akisue T and Moriyama H:
Effects of ultrasound, radial extracorporeal shock waves, and
electrical stimulation on rat bone defect healing. Ann N Y Acad
Sci. 1497:3–14. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Fuggle N, Laslop A, Rizzoli R, Al-Daghri
N, Alokail M, Balkowiec-Iskra E, Beaudart C, Bruyère O, Bemden AB,
Burlet N, et al: Treatment of osteoporosis and osteoarthritis in
the oldest old. Drugs. 85:343–360. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Eastell R, Rosen CJ, Black DM, Cheung AM,
Murad MH and Shoback D: Pharmacological management of osteoporosis
in postmenopausal women: An Endocrine Society* clinical practice
guideline. J Clin Endocrinol Metab. 104:1595–1622. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen ZH, Wu JJ, Guo DY, Li YY, Chen MN,
Zhang ZY, Yuan ZD, Zhang KW, Chen WW, Tian F, et al: Physiological
functions of podosomes: From structure and function to therapy
implications in osteoclast biology of bone resorption. Ageing Res
Rev. 85:1018422023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Heffler E, Madeira L, Ferrando M, Puggioni
F, Racca F, Malvezzi L, Passalacqua G and Canonica GW: Inhaled
corticosteroids safety and adverse effects in patients with asthma.
J Allergy Clin Immunol Pract. 6:776–781. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hesari E, Sanjari M, Mansourzadeh MJ,
Fahimfar N, Khalagi K, Ghazbani A, Ostovar A and Fotouhi A:
Osteoporosis medication adherence tools: A systematic review.
Osteoporos Int. 34:1535–1548. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Waldbaum J, Xhumari J, Akinsuyi OS,
Arjmandi B, Anton S and Roesch L: Association between dysbiosis in
the gut microbiota of primary osteoporosis patients and bone loss.
Aging Dis. 14:2081–2095. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li Y, Yang P, Ye J, Xu Q, Wu J and Wang Y:
Updated mechanisms of MASLD pathogenesis. Lipids Health Dis.
23:1172024. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tang WH, Kitai T and Hazen SL: Gut
microbiota in cardiovascular health and disease. Circ Res.
120:1183–1196. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tao H, Li W, Zhang W, Yang C, Zhang C,
Liang X, Yin J, Bai J, Ge G, Zhang H, et al: Urolithin A suppresses
RANKL-induced osteoclastogenesis and postmenopausal osteoporosis
by, suppresses inflammation and downstream NF-κB activated
pyroptosis pathways. Pharmacol Res. 174:1059672021. View Article : Google Scholar
|
|
14
|
Jiang T, Yang X, Wu B, Tao R, Chen R, Jin
L, Sun D and Weng H: Gut microbiota in hypothyroidism: Pathogenic
mechanisms and opportunities for precision microbiome
interventions. Front Microbiol. 16:16612112025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Xie X, Song J, Wu Y, Li M, Guo W, Li S and
Li Y: Study on gut microbiota and metabolomics in postmenopausal
women. BMC Womens Health. 24:6082024. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Birg A and Lin HC: The role of
bacteria-derived hydrogen sulfide in multiple axes of disease. Int
J Mol Sci. 26:33402025. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Akinsuyi OS and Roesch L: Meta-analysis
reveals compositional and functional microbial changes associated
with osteoporosis. Microbiol Spectr. 11:e00322232023. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu JH, Chen CY, Liu ZZ, Luo ZW, Rao SS,
Jin L, Wan TF, Yue T, Tan YJ, Yin H, et al: Extracellular vesicles
from child gut microbiota enter into bone to preserve bone mass and
strength. Adv Sci (Weinh). 8:20048312021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li JY, Chassaing B, Tyagi AM, Vaccaro C,
Luo T, Adams J, Darby TM, Weitzmann MN, Mulle JG, Gewirtz AT, et
al: Sex steroid deficiency-associated bone loss is microbiota
dependent and prevented by probiotics. J Clin Invest.
126:2049–2063. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yu C, Sun R, Yang W, Gu T, Ying X, Ye L,
Zheng Y, Fan S, Zeng X and Yao S: Exercise ameliorates osteopenia
in mice via intestinal microbial-mediated bile acid metabolism
pathway. Theranostics. 15:1741–1759. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Inchingolo AM, Gargiulo Isacco C,
Inchingolo AD, Nguyen K, Cantore S, Santacroce L, Scacco S, Cirulli
N, Corriero A, Puntillo F, et al: The human microbiota key role in
the bone metabolism activity. Eur Rev Med Pharmacol Sci.
27:2659–2670. 2023.PubMed/NCBI
|
|
22
|
Huang R, Liu P, Bai Y, Huang J, Pan R, Li
H, Su Y, Zhou Q, Ma R, Zong S, et al: Changes in the gut microbiota
of osteoporosis patients based on 16S rRNA gene sequencing: A
systematic review and meta-analysis. J Zhejiang Univ Sci B.
23:1002–1013. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Guadalupe-Grau A, Fuentes T, Guerra B and
Calbet JA: Exercise and bone mass in adults. Sports Med.
39:439–468. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang L, Zeng C, Huang J, Yan H, Jiang Y
and Li R: Exploration of the miR-187-3p/CNR2 pathway in modulating
osteoblast differentiation and treating postmenopausal osteoporosis
through mechanical stress. FASEB J. 38:e237762024. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liao HW, Huang TH, Chang YH, Liou HH, Chou
YH, Sue YM, Hung PH, Chang YT, Ho PC and Tsai KJ: Exercise
alleviates osteoporosis in rats with mild chronic kidney disease by
decreasing sclerostin production. Int J Mol Sci. 20:20442019.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gao Y, Wang H, Shi L, Cao M, Liu X, Zhang
Y, Chen X, Rong Y, Han B, Lu P, et al: Gut-bone axis mediates
exercise modality-dependent suppression of inflammatory
osteoclastogenesis in ovariectomy-induced bone loss. Mediators
Inflamm. 2025:57153322025. View Article : Google Scholar
|
|
27
|
Barcik W, Boutin R, Sokolowska M and
Finlay BB: The role of lung and gut microbiota in the pathology of
asthma. Immunity. 52:241–255. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gillman SJ, McKenney EA and Lafferty D:
Wild black bears harbor simple gut microbial communities with
little difference between the jejunum and colon. Sci Rep.
10:207792020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Oswald IP: Role of intestinal epithelial
cells in the innate immune defence of the pig intestine. Vet Res.
37:359–368. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Pedersini P, Turroni S and Villafañe JH:
Gut microbiota and physical activity: Is there an evidence-based
link. Sci Total Environ. 727:1386482020. View Article : Google Scholar
|
|
31
|
Fernandes I, Pérez-Gregorio R, Soares S,
Mateus N and de Freitas V: Wine flavonoids in health and disease
prevention. Molecules. 22:2922017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bezirtzoglou E and Stavropoulou E:
Immunology and probiotic impact of the newborn and young children
intestinal microflora. Anaerobe. 17:369–374. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yang Y, Chen J, Gao H, Cui M, Zhu M, Xiang
X and Wang Q: Characterization of the gut microbiota and fecal and
blood metabolomes under various factors in urban children from
Northwest China. Front Cell Infect Microbiol. 14:13745442024.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zang Y, Lai X, Li C, Ding D, Wang Y and
Zhu Y: The Role of gut microbiota in various neurological and
psychiatric disorders-an evidence mapping based on quantified
evidence. Mediators Inflamm. 2023:51271572023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Heravi FS, Naseri K and Hu H: Gut
Microbiota composition in patients with neurodegenerative disorders
(Parkinson's and Alzheimer's) and healthy controls: A systematic
review. Nutrients. 15:43652023. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Singhal R, Donde H, Ghare S, Stocke K,
Zhang J, Vadhanam M, Reddy S, Gobejishvili L, Chilton P,
Joshi-Barve S, et al: Decrease in acetyl-CoA pathway utilizing
butyrate-producing bacteria is a key pathogenic feature of
alcohol-induced functional gut microbial dysbiosis and development
of liver disease in mice. Gut Microbes. 13:19463672021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Xing P, Hahnke RL, Unfried F, Markert S,
Huang S, Barbeyron T, Harder J, Becher D, Schweder T, Glöckner FO,
et al: Niches of two polysaccharide-degrading Polaribacter isolates
from the North Sea during a spring diatom bloom. ISME J.
9:1410–1422. 2015. View Article : Google Scholar
|
|
38
|
Pacoud M, Mandon K, Cazareth J, Pierre O,
Frendo P and Alloing G: Redox-sensitive fluorescent biosensors
detect Sinorhizobium meliloti intracellular redox changes under
free-living and symbiotic lifestyles. Free Radic Biol Med.
184:185–195. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Dierick M, Ongena R, Vanrompay D,
Devriendt B and Cox E: Exploring the modulatory role of bovine
lactoferrin on the microbiome and the immune response in healthy
and Shiga toxin-producing E. coli challenged weaned piglets. J Anim
Sci Biotechnol. 15:392024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kang EJ, Kim JH, Kim YE, Lee H, Jung KB,
Chang DH, Lee Y, Park S, Lee EY, Lee EJ, et al: The secreted
protein Amuc_1409 from Akkermansia muciniphila improves gut health
through intestinal stem cell regulation. Nat Commun. 15:29832024.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Thacharodi A, Hassan S, Ahmed Z, Singh P,
Maqbool M, Meenatchi R, Pugazhendhi A and Sharma A: The ruminant
gut microbiome vs enteric methane emission: The essential microbes
may help to mitigate the global methane crisis. Environ Res.
261:1196612024. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gao Z, Kang Y, Yu J and Ren L: Human
pharyngeal microbiome may play a protective role in respiratory
tract infections. Genomics Proteomics Bioinformatics. 12:144–150.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hu Q, Huang L, Yang Y, Xiang Y and Liu J:
Essential phage component induces resistance of bacterial
community. Sci Adv. 10:eadp50572024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kaye DM, Shihata WA, Jama HA, Tsyganov K,
Ziemann M, Kiriazis H, Horlock D, Vijay A, Giam B, Vinh A, et al:
Deficiency of prebiotic fiber and insufficient signaling through
gut metabolite-sensing receptors leads to cardiovascular disease.
Circulation. 141:1393–1403. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ridlon JM, Harris SC, Bhowmik S, Kang DJ
and Hylemon PB: Consequences of bile salt biotransformations by
intestinal bacteria. Gut Microbes. 7:22–39. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ma L, Ni Y, Wang Z, Tu W, Ni L, Zhuge F,
Zheng A, Hu L, Zhao Y, Zheng L, et al: Spermidine improves gut
barrier integrity and gut microbiota function in diet-induced obese
mice. Gut Microbes. 12:1–19. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Lalonde R and Strazielle C: Probiotic
effects on anxiety-like behavior in animal models. Rev Neurosci.
33:691–701. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wan C, Liu ZB, Tan H, Zhang Z, Zhou Q, Yao
LH, Meng W and Wang S: Effect of voluntary wheel running on gut
microbiota composition in developing mice. Nutr Hosp. 39:896–904.
2022.PubMed/NCBI
|
|
49
|
Leigh SJ, Kaakoush NO, Escorihuela RM,
Westbrook RF and Morris MJ: Treadmill exercise has minimal impact
on obesogenic diet-related gut microbiome changes but alters
adipose and hypothalamic gene expression in rats. Nutr Metab
(Lond). 17:712020. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Hendesi H, Godfrey DA, Ruble AF, Tran AM,
Villani DA, Landgrave SH, Hasan NA, Adams DJ and Zuscik MJ:
Intermittent fasting alleviates obesity-associated impairments in
bone fracture healing: Exploring the role of gut microbiome. Bone
Rep. 27:1018762025. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Okoro PC, Orwoll ES, Huttenhower C, Morgan
X, Kuntz TM, McIver LJ, Dufour AB, Bouxsein ML, Langsetmo L,
Farsijani S, et al: A two-cohort study on the association between
the gut microbiota and bone density, microarchitecture, and
strength. Front Endocrinol (Lausanne). 14:12377272023. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Barbu AC, Stoleru S, Zugravu A, Poenaru E,
Dragomir A, Costescu M, Aurelian SM, Shhab Y, Stoleru CM, Coman OA,
et al: Dopamine and the gut Microbiota: Interactions within the
Microbiota-gut-brain axis and therapeutic perspectives. Int J Mol
Sci. 27:2712025. View Article : Google Scholar
|
|
53
|
Ren H, Wen J, Liu J and Wang L: Gut
microbiota in immunomodulation and infection prevention among
multiple myeloma patients after chemotherapy: Current evidence and
clinical prospects. Am J Cancer Res. 15:4621–4638. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Kawaratani H, Moriya K, Namisaki T, Uejima
M, Kitade M, Takeda K, Okura Y, Kaji K, Takaya H, Nishimura N, et
al: Therapeutic strategies for alcoholic liver disease: Focusing on
inflammation and fibrosis (Review). Int J Mol Med. 40:263–270.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zhao Z, Ning J, Bao XQ, Shang M, Ma J, Li
G and Zhang D: Fecal microbiota transplantation protects
rotenone-induced Parkinson's disease mice via suppressing
inflammation mediated by the lipopolysaccharide-TLR4 signaling
pathway through the microbiota-gut-brain axis. Microbiome.
9:2262021. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Yokota K, Sato K, Miyazaki T, Aizaki Y,
Tanaka S, Sekikawa M, Kozu N, Kadono Y, Oda H and Mimura T:
Characterization and function of tumor necrosis factor and
interleukin-6-induced osteoclasts in rheumatoid arthritis.
Arthritis Rheumatol. 73:1145–1154. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Yin Y, Sichler A, Ecker J, Laschinger M,
Liebisch G, Höring M, Basic M, Bleich A, Zhang XJ, Kübelsbeck L, et
al: Gut microbiota promote liver regeneration through hepatic
membrane phospholipid biosynthesis. J Hepatol. 78:820–835. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Liu YJ, Tang B, Wang FC, Tang L, Lei YY,
Luo Y, Huang SJ, Yang M, Wu LY, Wang W, et al: Parthenolide
ameliorates colon inflammation through regulating Treg/Th17 balance
in a gut microbiota-dependent manner. Theranostics. 10:5225–5241.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Montalvany-Antonucci CC, Duffles LF, de
Arruda J, Zicker MC, de Oliveira S, Macari S, Garlet GP, Madeira M,
Fukada SY, Andrade I, et al: Short-chain fatty acids and FFAR2 as
suppressors of bone resorption. Bone. 125:112–121. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Song C, Yan Q, Ma Y, Li P, Yang Y, Wang Y,
Li W, Wan X, Li Y, Zhu R, et al: Modified Zuo Gui wan ameliorates
ovariectomy-induced osteoporosis in rats by regulating the
SCFA-GPR41-p38MAPK signaling pathway. Drug Des Devel Ther.
18:6359–6377. 2024. View Article : Google Scholar :
|
|
61
|
Carson MD, Warner AJ, Hathaway-Schrader
JD, Geiser VL, Kim J, Gerasco JE, Hill WD, Lemasters JJ,
Alekseyenko AV, Wu Y, et al: Minocycline-induced disruption of the
intestinal FXR/FGF15 axis impairs osteogenesis in mice. JCI
Insight. 8:e1605782023. View Article : Google Scholar :
|
|
62
|
Lyu Y, Hu J, Wang X, Zhang J, Li X, Cui M,
Tang X and Zhou P: Lactopontin regulates gut microbiota and calcium
absorption to promote bone growth in growing rats. Int J Biol
Macromol. 302:1405572025. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Yan J, Herzog JW, Tsang K, Brennan CA,
Bower MA, Garrett WS, Sartor BR, Aliprantis AO and Charles JF: Gut
microbiota induce IGF-1 and promote bone formation and growth. Proc
Natl Acad Sci USA. 113:E7554–E7563. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Chen Q, Shou P, Zheng C, Jiang M, Cao G,
Yang Q, Cao J, Xie N, Velletri T, Zhang X, et al: Fate decision of
mesenchymal stem cells: Adipocytes or osteoblasts. Cell Death
Differ. 23:1128–1139. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Shay JW, Homma N, Zhou R, Naseer MI,
Chaudhary AG, Al-Qahtani M, Hirokawa N, Goudarzi M, Fornace AJ Jr,
Baeesa S, et al: Abstracts from the 3rd International Genomic
Medicine Conference (3rd IGMC 2015): Jeddah, Kingdom of Saudi
Arabia. 30 November-3 December 2015. BMC Genomics. 17(Suppl 6):
S4872016. View Article : Google Scholar
|
|
66
|
Liu A, Liang X, Wang W, Wang C, Song J,
Guo J, Sun D, Wang D, Song M, Qian J, et al: Human umbilical cord
mesenchymal stem cells ameliorate colon inflammation via modulation
of gut microbiota-SCFAs-immune axis. Stem Cell Res Ther.
14:2712023. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Hao H, Liu Q, Zheng T, Li J, Zhang T, Yao
Y, Liu Y, Lin K, Liu T, Gong P, et al: Oral Milk-derived
extracellular vesicles inhibit osteoclastogenesis and ameliorate
bone loss in ovariectomized mice by improving gut microbiota. J
Agric Food Chem. 72:4726–4736. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Wei F, Hughes M, Omer M, Ngo C,
Pugazhendhi AS, Kolanthai E, Aceto M, Ghattas Y, Razavi M, Kean TJ,
et al: A multifunctional therapeutic strategy using P7C3 as A
countermeasure against bone loss and fragility in an ovariectomized
rat model of postmenopausal osteoporosis. Adv Sci (Weinh).
11:e23086982024. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Ding K, Hua F and Ding W: Gut microbiome
and osteoporosis. Aging Dis. 11:438–447. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Xiao L, Zhou Y, Bokoliya S, Lin Q and
Hurley M: Bone loss is ameliorated by fecal microbiota
transplantation through SCFA/GPR41/IGF1 pathway in sickle cell
disease mice. Sci Rep. 12:206382022. View Article : Google Scholar
|
|
71
|
Li Z, Huang J, Wang F, Li W, Wu X, Zhao C,
Zhao J, Wei H, Wu Z, Qian M, et al: Dual targeting of bile acid
receptor-1 (TGR5) and Farnesoid X receptor (FXR) prevents
estrogen-dependent bone loss in mice. J Bone Miner Res. 34:765–776.
2019. View Article : Google Scholar
|
|
72
|
Wang Q, Wang G, Wang B and Yang H:
Activation of TGR5 promotes osteoblastic cell differentiation and
mineralization. Biomed Pharmacother. 108:1797–1803. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Cho SW, An JH, Park H, Yang JY, Choi HJ,
Kim SW, Park YJ, Kim SY, Yim M, Baek WY, et al: Positive regulation
of osteogenesis by bile acid through FXR. J Bone Miner Res.
28:2109–2121. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zhu L, Hua F, Ding W, Ding K, Zhang Y and
Xu C: The correlation between the Th17/Treg cell balance and bone
health. Immun Ageing. 17:302020. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Sun P, Zhang C, Huang Y, Yang J, Zhou F,
Zeng J and Lin Y: Jiangu granule ameliorated OVX rats bone loss by
modulating gut microbiota-SCFAs-Treg/Th17 axis. Biomed
Pharmacother. 150:1129752022. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Liu S, Li G, Xu H, Wang Q, Wei Y, Yang Q,
Xiong A, Yu F, Weng J and Zeng H: 'Cross-talk' between gut
microbiome dysbiosis and osteoarthritis progression: A systematic
review. Front Immunol. 14:11505722023. View Article : Google Scholar
|
|
77
|
Li C, Huang Q, Yang R, Dai Y, Zeng Y, Tao
L, Li X, Zeng J and Wang Q: Gut microbiota composition and bone
mineral loss-epidemiologic evidence from individuals in Wuhan,
China. Osteoporos Int. 30:1003–1013. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Fata JE, Kong YY, Li J, Sasaki T,
Irie-Sasaki J, Moorehead RA, Elliott R, Scully S, Voura EB, Lacey
DL, et al: The osteoclast differentiation factor
osteoprotegerin-ligand is essential for mammary gland development.
Cell. 103:41–50. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Han D, Wang W, Gong J, Ma Y and Li Y:
Microbiota metabolites in bone: Shaping health and confronting
disease. Heliyon. 10:e284352024. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Wang N, Hao Y and Fu L:
Trimethylamine-N-Oxide promotes osteoclast differentiation and bone
loss via activating ROS-dependent NF-κB signaling pathway.
Nutrients. 14:39552022. View Article : Google Scholar
|
|
81
|
Zhao Y, Wang C, Qiu F, Liu J, Xie Y, Lin
Z, He J and Chen J: Trimethylamine-N-oxide promotes osteoclast
differentiation and oxidative stress by activating NF-κB pathway.
Aging (Albany NY). 16:9251–9263. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Feng R, Wang Q, Yu T, Hu H, Wu G, Duan X,
Jiang R, Xu Y and Huang Y: Quercetin ameliorates bone loss in OVX
rats by modulating the intestinal flora-SCFAs-inflammatory
signaling axis. Int Immunopharmacol. 136:1123412024. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Liu T, Yu H, Wang S, Li H, Du X and He X:
Chondroitin sulfate alleviates osteoporosis caused by calcium
deficiency by regulating lipid metabolism. Nutr Metab (Lond).
20:62023. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Li K, Liu A, Zong W, Dai L, Liu Y, Luo R,
Ge S and Dong G: Moderate exercise ameliorates osteoarthritis by
reducing lipopolysaccharides from gut microbiota in mice. Saudi J
Biol Sci. 28:40–49. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Yang X, Wang Y, Han X, Shu R, Chen T, Zeng
H, Xu X, Huang L, Ren A, Song J, et al: Effects of TGF-β1 on
OPG/RANKL expression of cementoblasts and osteoblasts are similar
without stress but different with mechanical compressive stress.
ScientificWorldJournal. 2015:7181802015. View Article : Google Scholar
|
|
86
|
Guo M, Liu H, Yu Y, Zhu X, Xie H, Wei C,
Mei C, Shi Y, Zhou N, Qin K, et al: Lactobacillus rhamnosus GG
ameliorates osteoporosis in ovariectomized rats by regulating the
Th17/Treg balance and gut microbiota structure. Gut Microbes.
15:21903042023. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Hu H, Yao Y, Liu F, Luo L, Liu J, Wang X
and Wang Q: Integrated microbiome and metabolomics revealed the
protective effect of baicalin on alveolar bone inflammatory
resorption in aging. Phytomedicine. 124:1552332024. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Beaudart C, Dawson A, Shaw SC, Harvey NC,
Kanis JA, Binkley N, Reginster JY, Chapurlat R, Chan DC, Bruyère O,
et al: Nutrition and physical activity in the prevention and
treatment of sarcopenia: Systematic review. Osteoporos Int.
28:1817–1833. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Angulo J, El Assar M, Álvarez-Bustos A and
Rodríguez-Mañas L: Physical activity and exercise: Strategies to
manage frailty. Redox Biol. 35:1015132020. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Lucas S, Omata Y, Hofmann J, Böttcher M,
Iljazovic A, Sarter K, Albrecht O, Schulz O, Krishnacoumar B,
Krönke G, et al: Short-chain fatty acids regulate systemic bone
mass and protect from pathological bone loss. Nat Commun. 9:552018.
View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Li X, Han Y, Guan Y, Zhang L, Bai C and Li
Y: Aluminum induces osteoblast apoptosis through the oxidative
stress-mediated JNK signaling pathway. Biol Trace Elem Res.
150:502–508. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Ding M, Li B, Chen H, Liang D, Ross RP,
Stanton C, Zhao J, Chen W and Yang B: Human breastmilk-derived
Bifidobacterium longum subsp. infantis CCFM1269 regulates bone
formation by the GH/IGF axis through PI3K/AKT pathway. Gut
Microbes. 16:22903442024. View Article : Google Scholar :
|
|
93
|
Guan Z, Xiao W, Guan Z, Xiao J, Jin X, Liu
S, Qin Y and Luo L: Intermittent fasting triggers interorgan
communication to improve the progression of diabetic osteoporosis.
Gut Microbes. 17:25556192025. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Li R, Miao Z, Liu Y, Chen X, Wang H, Su J
and Chen J: The brain-gut-bone axis in neurodegenerative diseases:
Insights, challenges, and future prospects. Adv Sci (Weinh).
11:e23079712024. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Lorenzo D, GianVincenzo Z, Carlo Luca R,
Karan G, Jorge V, Roberto M and Javad P: Oral-gut microbiota and
arthritis: Is there an evidence-based axis. J Clin Med.
8:02019.
|
|
96
|
Wu M, Chen C, Lei H, Cao Z, Zhang C, Du R,
Zhang C, Song Y, Qin M, Zhou J, et al: Dietary isoquercetin
ameliorates bone loss via restoration of the gut microbiota and
lipopolysaccharide-triggered inflammatory status in ovariectomy
mice. J Agric Food Chem. 71:15981–15990. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Li H, Hu S, Wu R, Zhou H, Zhang K, Li K,
Lin W, Shi Q, Chen H and Lv S: 11β-Hydroxysteroid dehydrogenase
type 1 facilitates osteoporosis by turning on Osteoclastogenesis
through Hippo Signaling. Int J Biol Sci. 19:3628–3639. 2023.
View Article : Google Scholar
|
|
98
|
Bai J, Si G, Wang R, Su S, Fan J, He X, Lv
Y, Gao S and Zhou F: Gut metabolite indoleacrylic acid suppresses
osteoclast formation by AHR mediated NF-κB signaling pathway. Int J
Biol Sci. 22:951–969. 2026. View Article : Google Scholar
|
|
99
|
Chen Y, Yang C, Dai Q, Tan J, Dou C and
Luo F: Gold-nanosphere mitigates osteoporosis through regulating
TMAO metabolism in a gut microbiota-dependent manner. J
Nanobiotechnology. 21:1252023. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Yadav S, Sapra L and Srivastava RK:
Polysaccharides to postbiotics: Nurturing bone health via
modulating 'gut-immune axis'. Int J Biol Macromol. 278:1346552024.
View Article : Google Scholar
|
|
101
|
Czernik PJ, Golonka RM, Chakraborty S,
Yeoh BS, Abokor AA, Saha P, Yeo JY, Mell B, Cheng X, Baroi S, et
al: Reconstitution of the host holobiont in germ-free born male
rats acutely increases bone growth and affects marrow cellular
content. Physiol Genomics. 53:518–533. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Liu Y, Guo YL, Meng S, Gao H, Sui LJ, Jin
S, Li Y and Fan SG: Gut microbiota-dependent Trimethylamine N-Oxide
are related with hip fracture in postmenopausal women: A matched
case-control study. Aging (Albany NY). 12:10633–10641. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Zhao YX, Song YW, Zhang L, Zheng FJ, Wang
XM, Zhuang XH, Wu F and Liu J: Association between bile acid
metabolism and bone mineral density in postmenopausal women.
Clinics (Sao Paulo). 75:e14862020. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Schwarzer M, Makki K, Storelli G,
Machuca-Gayet I, Srutkova D, Hermanova P, Martino ME, Balmand S,
Hudcovic T, Heddi A, et al: Lactobacillus plantarum strain
maintains growth of infant mice during chronic undernutrition.
Science. 351:854–857. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Quach D, Collins F, Parameswaran N, McCabe
L and Britton RA: Microbiota reconstitution does not cause bone
loss in germ-free mice. mSphere. 3:e00545–00517. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Li Z, Wang Q, Huang X, Wu Y and Shan D:
Microbiome's role in musculoskeletal health through the gut-bone
axis insights. Gut Microbes. 16:24104782024. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Sjögren K, Engdahl C, Henning P, Lerner
UH, Tremaroli V, Lagerquist MK, Bäckhed F and Ohlsson C: The gut
microbiota regulates bone mass in mice. J Bone Miner Res.
27:1357–1367. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Hartmann K, Lenz A, Letko G, Schulz HU,
Sokolowski A, Spormann H and Zühlke H: Isolated cells in suspension
for biological research-Part II. Structure and functional
properties of cells from pancreatic islets and acini. Exp Pathol.
34:1–22. 1988. View Article : Google Scholar
|
|
109
|
Lin H, Liu T, Li X, Gao X, Wu T and Li P:
The role of gut microbiota metabolite trimethylamine N-oxide in
functional impairment of bone marrow mesenchymal stem cells in
osteoporosis disease. Ann Transl Med. 8:10092020. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Chen CY, Rao SS, Yue T, Tan YJ, Yin H,
Chen LJ, Luo MJ, Wang Z, Wang YY, Hong CG, et al:
Glucocorticoid-induced loss of beneficial gut bacterial
extracellular vesicles is associated with the pathogenesis of
osteonecrosis. Sci Adv. 8:eabg83352022. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Wu Z, Chen J, Kong F, Zhang Y, Yi J, Li Y,
Hu M and Wang D: Polypeptide of Inonotus hispidus extracts
alleviates periodontitis through suppressing inflammatory bone
loss. Int J Biol Macromol. 287:1383502025. View Article : Google Scholar
|
|
112
|
Sudo N, Chida Y, Aiba Y, Sonoda J, Oyama
N, Yu XN, Kubo C and Koga Y: Postnatal microbial colonization
programs the hypothalamic-pituitary-adrenal system for stress
response in mice. J Physiol. 558:263–275. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Cooper MS: Sensitivity of bone to
glucocorticoids. Clin Sci (Lond). 107:111–123. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Zhou RX, Zhang YW, Cao MM, Liu CH, Rui YF
and Li YJ: Linking the relation between gut microbiota and
glucocorticoid-induced osteoporosis. J Bone Miner Metab.
41:145–162. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Hong S, Cha KH, Park JH, Jung DS, Choi JH,
Yoo G and Nho CW: Cinnamic acid suppresses bone loss via induction
of osteoblast differentiation with alteration of gut microbiota. J
Nutr Biochem. 101:1089002022. View Article : Google Scholar
|
|
116
|
Dong J, Shu G, Yang J, Wang B, Chen L,
Gong Z and Zhang X: Mechanistic study on the alleviation of
postmenopausal osteoporosis by Lactobacillus acidophilus through
butyrate-mediated inhibition of osteoclast activity. Sci Rep.
14:70422024. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Li JY, Yu M, Pal S, Tyagi AM, Dar H, Adams
J, Weitzmann MN, Jones RM and Pacifici R: Parathyroid
hormone-dependent bone formation requires butyrate production by
intestinal microbiota. J Clin Invest. 130:1767–1781. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Hu HT, Zhang ZY, Luo Z, Ti HB, Wu JJ, Nie
H, Yuan ZD, Wu X, Zhang KY, Shi SW, et al: Emerging regulated cell
death mechanisms in bone remodeling: Decoding ferroptosis,
cuproptosis, disulfidptosis, and PANoptosis as therapeutic targets
for skeletal. Cell Death Discov. 11:3352025. View Article : Google Scholar
|
|
119
|
Xiao HH, Zhu YX, Lu L, Zhou LP, Poon CC,
Chan CO, Wang LJ, Cao S, Yu WX, Wong KY, et al: The Lignan-rich
fraction from sambucus williamsii hance exerts bone protective
effects via altering circulating serotonin and gut microbiota in
rats. Nutrients. 14:47182022. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Wei X, Yang X, Han ZP, Qu FF, Shao L and
Shi YF: Mesenchymal stem cells: A new trend for cell therapy. Acta
Pharmacol Sin. 34:747–754. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Ozasa R, Matsugaki A, Isobe Y, Saku T, Yun
HS and Nakano T: Construction of human induced pluripotent stem
cell-derived oriented bone matrix microstructure by using in vitro
engineered anisotropic culture model. J Biomed Mater Res A.
106:360–369. 2018. View Article : Google Scholar :
|
|
122
|
Xing J, Ying Y, Mao C, Liu Y, Wang T, Zhao
Q, Zhang X, Yan F and Zhang H: Hypoxia induces senescence of bone
marrow mesenchymal stem cells via altered gut microbiota. Nat
Commun. 9:20202018. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Li W, Li T, Tang Z, Qi X, Zhou Y, Tang X,
Xu W and Xiong H: Taohong Siwu decoction promotes the process of
fracture healing by activating the VEGF-FAK signal pathway and
systemically regulating the gut microbiota. J Appl Microbiol.
133:1363–1377. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Queipo-Ortuño MI, Seoane LM, Murri M,
Pardo M, Gomez-Zumaquero JM, Cardona F, Casanueva F and Tinahones
FJ: Gut microbiota composition in male rat models under different
nutritional status and physical activity and its association with
serum leptin and ghrelin levels. PLoS One. 8:e654652013. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Wang D, Cai J, Pei Q, Yan Z, Zhu F, Zhao
Z, Liu R, Guo X, Sun T, Liu J, et al: Gut microbial alterations in
arginine metabolism determine bone mechanical adaptation. Cell
Metab. 36:1252–1268.e8. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Ye X, Sun P, Lao S, Wen M, Zheng R, Lin Y,
Gan L, Fan X, Wang P, Li Z, et al: Fgf21-dubosiella axis mediates
the protective effects of exercise against NAFLD development. Life
Sci. 334:1222312023. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Lambert JE, Myslicki JP, Bomhof MR, Belke
DD, Shearer J and Reimer RA: Exercise training modifies gut
microbiota in normal and diabetic mice. Appl Physiol Nutr Metab.
40:749–752. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Liu Z, Liu HY, Zhou H, Zhan Q, Lai W, Zeng
Q, Ren H and Xu D: Moderate-intensity exercise affects gut
microbiome composition and influences cardiac function in
myocardial infarction mice. Front Microbiol. 8:16872017. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Wang J, Zhang Q, Xia J and Sun H: Moderate
treadmill exercise modulates gut microbiota and improves intestinal
barrier in high-fat-diet-induced obese mice via the AMPK/CDX2
signaling pathway. Diabetes Metab Syndr Obes. 15:209–223. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Estaki M, Pither J, Baumeister P, Little
JP, Gill SK, Ghosh S, Ahmadi-Vand Z, Marsden KR and Gibson DL:
Cardiorespiratory fitness as a predictor of intestinal microbial
diversity and distinct metagenomic functions. Microbiome. 4:422016.
View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Allen JM, Mailing LJ, Niemiro GM, Moore R,
Cook MD, White BA, Holscher HD and Woods JA: Exercise alters gut
microbiota composition and function in lean and obese humans. Med
Sci Sports Exerc. 50:747–757. 2018. View Article : Google Scholar
|
|
132
|
Barton W, Penney NC, Cronin O,
Garcia-Perez I, Molloy MG, Holmes E, Shanahan F, Cotter PD and
O'Sullivan O: The microbiome of professional athletes differs from
that of more sedentary subjects in composition and particularly at
the functional metabolic level. Gut. 67:625–633. 2018. View Article : Google Scholar
|
|
133
|
Motiani KK, Collado MC, Eskelinen JJ,
Virtanen KA, Löyttyniemi E, Salminen S, Nuutila P, Kalliokoski KK
and Hannukainen JC: Exercise training modulates gut microbiota
profile and improves endotoxemia. Med Sci Sports Exerc. 52:94–104.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Wu J, Oka J, Higuchi M, Tabata I, Toda T,
Fujioka M, Fuku N, Teramoto T, Okuhira T, Ueno T, et al:
Cooperative effects of isoflavones and exercise on bone and lipid
metabolism in postmenopausal Japanese women: A randomized
placebo-controlled trial. Metabolism. 55:423–433. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Lin Z, Zhang X, Wu M, Ming Y, Wang X, Li
H, Huang F, Gao F and Zhu Y: High-fiber diet and rope-skipping
benefit cardiometabolic health and modulate gut microbiota in young
adults: A randomized controlled trial. Food Res Int.
173:1134212023. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Zhang K, Guo H, Zhang X, Yang H, Yuan G,
Zhu Z, Lu X, Zhang J, Du J, Shi H, et al: Effects of aerobic
exercise or Tai Chi Chuan interventions on problematic mobile phone
use and the potential role of intestinal flora: A multi-arm
randomized controlled. J Psychiatr Res. 170:394–407. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Wu WD, Wang Y and Wei JX: Effects of
MICT/HIIT on the ultrastructure of myocardium and soleus in rats
with high-fat diet and its mechanisms. Zhongguo Ying Yong Sheng Li
Xue Za Zhi. 38:708–713. 2022.In Chinese.
|
|
138
|
Castro AP, Silva KKS, Medeiros CSA, Alves
F, Araujo RC and Almeida JA: Effects of 12 weeks of resistance
training on rat gut microbiota composition. J Exp Biol.
224:jeb2425432021. View Article : Google Scholar
|
|
139
|
Wang L, Lavier J, Hua W, Wang Y, Gong L,
Wei H, Wang J, Pellegrin M, Millet GP and Zhang Y: High-intensity
interval training and moderate-intensity continuous training
attenuate oxidative damage and promote myokine response in the
skeletal muscle. Antioxidants (Basel). 10:9922021. View Article : Google Scholar
|
|
140
|
Denou E, Marcinko K, Surette MG, Steinberg
GR and Schertzer JD: High-intensity exercise training increases the
diversity and metabolic capacity of the mouse distal gut microbiota
during diet-induced obesity. Am J Physiol Endocrinol Metab.
310:E982–E993. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Jäger R, Kerksick CM, Campbell BI, Cribb
PJ, Wells SD, Skwiat TM, Purpura M, Ziegenfuss TN, Ferrando AA,
Arent SM, et al: International society of sports nutrition position
stand: Protein and exercise. J Int Soc Sports Nutr. 14:202017.
View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Donnelly JE, Blair SN, Jakicic JM, Manore
MM, Rankin JW and Smith BK; American College of Sports Medicine:
American College of Sports Medicine Position Stand. Appropriate
physical activity intervention strategies for weight loss and
prevention of weight regain for adults. Med Sci Sports Exerc.
41:459–471. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Locatelli JC, Costa JG, Haynes A, Naylor
LH, Fegan PG, Yeap BB and Green DJ: Incretin-based weight loss
pharmacotherapy: Can resistance exercise optimize changes in body
composition. Diabetes Care. 47:1718–1730. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Rettedal EA, Cree JME, Adams SE, MacRae C,
Skidmore PML, Cameron-Smith D, Gant N, Blenkiron C and Merry TL:
Short-term high-intensity interval training exercise does not
affect gut bacterial community diversity or composition of lean and
overweight men. Exp Physiol. 105:1268–1279. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Hung WC, Hung WW, Tsai HJ, Chang CC, Chiu
YW, Hwang SJ, Kuo MC, Chen SC, Dai CY and Tsai YC: The association
of targeted gut microbiota with body composition in type 2 diabetes
mellitus. Int J Med Sci. 18:511–519. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Mackay D, Mollard RC, Granger M, Bruce S,
Blewett H, Carlberg J, Duhamel T, Eck P, Faucher P, Hamm NC, et al:
The Manitoba personalized lifestyle research (TMPLR) study
protocol: A multicentre bidirectional observational cohort study
with administrative health record linkage investigating the
interactions between lifestyle and health in Manitoba, Canada. BMJ
Open. 9:e0233182019. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Nilsson AG, Sundh D, Bäckhed F and
Lorentzon M: Lactobacillus reuteri reduces bone loss in older women
with low bone mineral density: A randomized, placebo-controlled,
double-blind, clinical trial. J Intern Med. 284:307–317. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Zhang C and Lin Y, Li H, Hu H, Chen Y,
Huang Y, Huang Z, Fang X, Zhang W and Lin Y: Fatty acid binding
protein 4 (FABP4) induces chondrocyte degeneration via activation
of the NF-κb signaling pathway. FASEB J. 38:e233472024. View Article : Google Scholar
|
|
149
|
Strasser B, Wolters M, Weyh C, Krüger K
and Ticinesi A: The effects of lifestyle and diet on gut microbiota
composition, inflammation and muscle performance in our aging
society. Nutrients. 13:20452021. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Zhang L, Wang P, Huang J, Xing Y, Wong FS,
Suo J and Wen L: Gut microbiota and therapy for obesity and type 2
diabetes. Front Endocrinol (Lausanne). 15:13337782024. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
O'Brien MT, O'Sullivan O, Claesson MJ and
Cotter PD: The athlete gut microbiome and its relevance to health
and performance: A review. Sports Med. 52(Suppl 1): S119–S128.
2022. View Article : Google Scholar
|
|
152
|
Zhang Z, Liu L, Ti H, Chen M, Chen Y, Du
D, Zhan W, Wang T, Wu X, Wu J, et al: Synovial fibroblast derived
small extracellular vesicles miRNA15-29148 promotes articular
chondrocyte apoptosis in rheumatoid arthritis. Bone Res. 13:612025.
View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Yuan Y, Yang J, Zhuge A, Li L and Ni S:
Gut microbiota modulates osteoclast glutathione synthesis and
mitochondrial biogenesis in mice subjected to ovariectomy. Cell
Prolif. 55:e131942022. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Desai S, Wu J, Horkeby K, Norgård M,
Ohlsson C, Windahl SH and Engdahl C: A COX-2 inhibitor does not
interfere with the bone-protective effects of loading in male mice
with arthritis. JBMR Plus. 7:e107512023. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Wang Q, Weng H, Xu Y, Ye H, Liang Y, Wang
L, Zhang Y, Gao Y, Wang J, Xu Y, et al: Anti-osteoporosis mechanism
of resistance exercise in ovariectomized rats based on
transcriptome analysis: A pilot study. Front Endocrinol (Lausanne).
14:11624152023. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
McCabe LR, Irwin R, Tekalur A, Evans C,
Schepper JD, Parameswaran N and Ciancio M: Exercise prevents high
fat diet-induced bone loss, marrow adiposity and dysbiosis in male
mice. Bone. 118:20–31. 2019. View Article : Google Scholar
|
|
157
|
Yuan FL, Li X, Lu WG, Xu RS, Zhao YQ, Li
CW, Li JP and Chen FH: Regulatory T cells as a potent target for
controlling bone loss. Biochem Biophys Res Commun. 402:173–176.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Li M, van Esch B, Wagenaar G, Garssen J,
Folkerts G and Henricks P: Pro- and anti-inflammatory effects of
short chain fatty acids on immune and endothelial cells. Eur J
Pharmacol. 831:52–59. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Chen H, Shen L, Liu Y, Ma X, Long L, Ma X,
Ma L, Chen Z, Lin X, Si L and Chen X: Strength exercise confers
protection in central nervous system autoimmunity by altering the
gut microbiota. Front Immunol. 12:6286292021. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Li XX, Hara I and Matsumiya T: Effects of
osthole on postmenopausal osteoporosis using ovariectomized rats;
comparison to the effects of estradiol. Biol Pharm Bull.
25:738–742. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Xu L, Zhu J, Rong L, Yang H, Wang B, Lu S,
Zhang L, Li F, Yang S, Wang Z, et al: Osteoblast-specific
down-regulation of NLRP3 inflammasome by aptamer-functionalized
liposome nanoparticles improves bone quality in postmenopausal.
Theranostics. 14:3945–3962. 2024. View Article : Google Scholar
|
|
162
|
Cornish J, Callon KE, Bava U, Lin C, Naot
D, Hill BL, Grey AB, Broom N, Myers DE, Nicholson GC and Reid IR:
Leptin directly regulates bone cell function in vitro and reduces
bone fragility in vivo. J Endocrinol. 175:405–415. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Rios JL, Bomhof MR, Reimer RA, Hart DA,
Collins KH and Herzog W: Protective effect of prebiotic and
exercise intervention on knee health in a rat model of diet-induced
obesity. Sci Rep. 9:38932019. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Hong S, Nguyen BN, Min H, Youn HY, Choi S,
Hitayezu E, Cha KH, Park YT, Lee CG, Yoo G, et al: Host-specific
effects of Eubacterium species on Rg3-mediated modulation of
osteosarcopenia in a genetically diverse mouse population.
Microbiome. 12:2512024. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Meissner M, Lombardo E, Havinga R, Tietge
UJ, Kuipers F and Groen AK: Voluntary wheel running increases bile
acid as well as cholesterol excretion and decreases atherosclerosis
in hypercholesterolemic mice. Atherosclerosis. 218:323–329. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Chen See JR, Amos D, Wright J, Lamendella
R and Santanam N: Synergistic effects of exercise and catalase
overexpression on gut microbiome. Environ Microbiol. 24:4220–4235.
2022. View Article : Google Scholar
|
|
167
|
Munukka E, Ahtiainen JP, Puigbó P,
Jalkanen S, Pahkala K, Keskitalo A, Kujala UM, Pietilä S, Hollmén
M, Elo L, et al: Six-week endurance exercise alters gut metagenome
that is not reflected in systemic metabolism in over-weight women.
Front Microbiol. 9:23232018. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Uday S and Högler W: Nutritional rickets
and osteomalacia in the twenty-first century: Revised concepts,
public health, and prevention strategies. Curr Osteoporos Rep.
15:293–302. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Méndez-Sánchez L, Clark P, Winzenberg TM,
Tugwell P, Correa-Burrows P and Costello R: Calcium and vitamin D
for increasing bone mineral density in premenopausal women.
Cochrane Database Syst Rev. 1:CD0126642023.PubMed/NCBI
|
|
170
|
Ilesanmi-Oyelere BL, Roy NC and Kruger MC:
Modulation of bone and joint biomarkers, gut microbiota, and
inflammation status by synbiotic supplementation and weight-bearing
exercise: Human study protocol for a randomized controlled trial.
JMIR Res Protoc. 10:e301312021. View
Article : Google Scholar : PubMed/NCBI
|
|
171
|
Luo B, Xiang D, Nieman DC and Chen P: The
effects of moderate exercise on chronic stress-induced intestinal
barrier dysfunction and antimicrobial defense. Brain Behav Immun.
39:99–106. 2014. View Article : Google Scholar
|
|
172
|
Fu R, Niu R, Zhao F, Wang J, Cao Q, Yu Y,
Liu C, Zhang D and Sun Z: Exercise alleviated intestinal damage and
microbial disturbances in mice exposed to fluoride. Chemosphere.
288:1326582022. View Article : Google Scholar
|
|
173
|
Donohoe DR, Garge N, Zhang X, Sun W,
O'Connell TM, Bunger MK and Bultman SJ: The microbiome and butyrate
regulate energy metabolism and autophagy in the mammalian colon.
Cell Metab. 13:517–526. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Huang WC, Tung CL, Yang YSH, Lin IH, Ng XE
and Tung YT: Endurance exercise ameliorates Western diet-induced
atherosclerosis through modulation of microbiota and its
metabolites. Sci Rep. 12:36122022. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Saito T, Ono R, Kono S, Asano M, Fukuta A,
Tanaka Y, Takao S and Sakai Y: Physical activity among patients
with breast cancer receiving aromatase inhibitors is associated
with bone health: A cross-sectional observational study. Breast
Cancer Res Treat. 182:187–193. 2020. View Article : Google Scholar : PubMed/NCBI
|