|
1
|
Shan B, Huo Y, Guo Z, Li Q, Pan Z, Li Q
and Du X: miR-184, a downregulated ovary-elevated miRNA
transcriptionally activated by SREBF2, exerts anti-apoptotic
properties in ovarian granulosa cells through inducing SMAD3
expression. Cell Death Dis. 15:8922024. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wu H, Nguyen H, Hashim PH, Fogelgren B,
Duncan FE and Ward WS: Oocyte-specific EXOC5 expression is required
for mouse oogenesis and folliculogenesis. Mol Hum Reprod.
30:gaae0262024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang CH, Liu XY and Wang J: Essential
role of granulosa cell glucose and lipid metabolism on oocytes and
the potential metabolic imbalance in polycystic ovary syndrome. Int
J Mol Sci. 24:162472023. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rooda I, Hassan J, Hao J, Wagner M,
Moussaud-Lamodiere E, Jääger K, Otala M, Knuus K, Lindskog C,
Papaikonomou K, et al: In-depth analysis of transcriptomes in
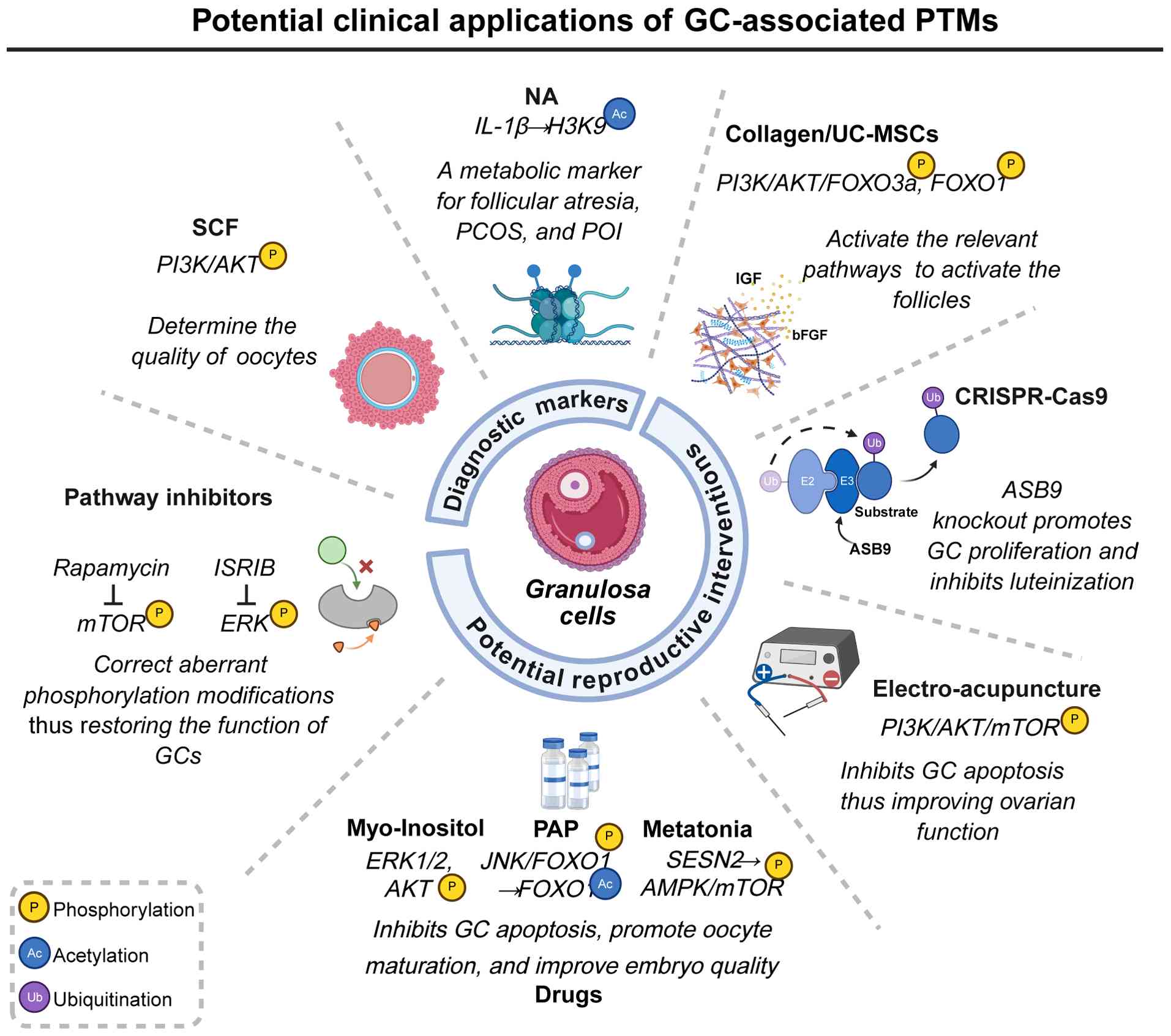
ovarian cortical follicles from children and adults reveals
interfollicular heterogeneity. Nat Commun. 15:69892024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Almeida CP, Ferreira MCF, Silveira CO,
Campos JR, Borges IT, Baeta PG, Silva FHS, Reis FM and Del Puerto
HL: Clinical correlation of apoptosis in human granulosa cells-a
review. Cell Biol Int. 42:1276–1281. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Vander Borght M and Wyns C: Fertility and
infertility: Definition and epidemiology. Clin Biochem. 62:2–10.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Habara O, Logan CY, Kanai-Azuma M, Nusse R
and Takase HM: WNT signaling in pre-granulosa cells is required for
ovarian folliculogenesis and female fertility. Development.
148:dev1988462021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sreerangaraja Urs DB, Wu WH, Komrskova K,
Postlerova P, Lin YF, Tzeng CR and Kao SH: Mitochondrial function
in modulating human granulosa cell steroidogenesis and female
fertility. Int J Mol Sci. 21:35922020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lin X, Dai Y, Tong X, Xu W, Huang Q, Jin
X, Li C, Zhou F, Zhou H, Lin X, et al: Excessive oxidative stress
in cumulus granulosa cells induced cell senescence contributes to
endometriosis-associated infertility. Redox Biol. 30:1014312020.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu Y, Liu H, Li Z, Fan H, Yan X, Liu X,
Xuan J, Feng D and Wei X: The release of peripheral immune
inflammatory cytokines promote an inflammatory cascade in PCOS
patients via altering the follicular microenvironment. Front
Immunol. 12:6857242021. View Article : Google Scholar : PubMed/NCBI
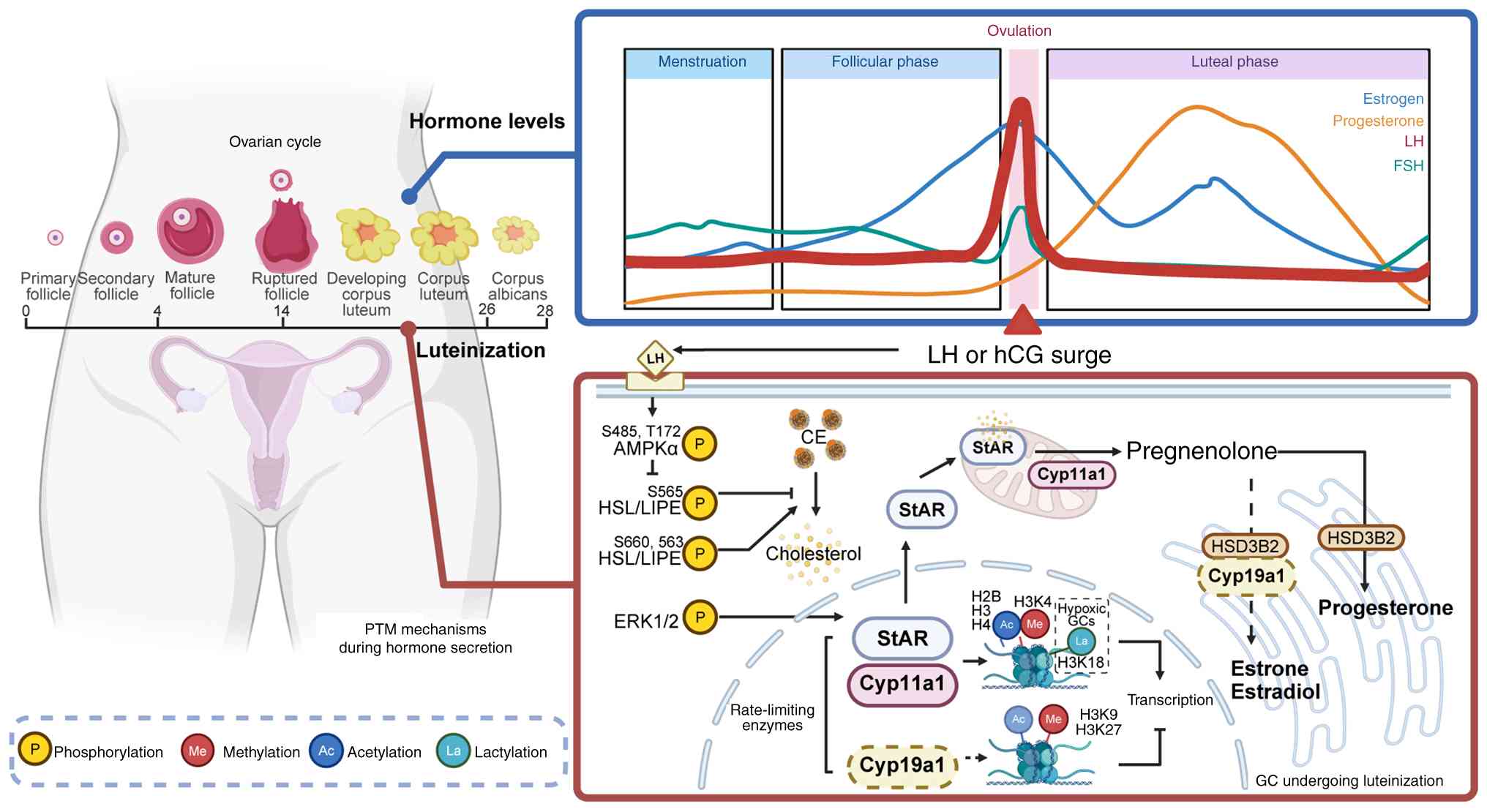
|
|
11
|
Kunitomi C, Harada M, Takahashi N, Azhary
JMK, Kusamoto A, Nose E, Oi N, Takeuchi A, Wada-Hiraike O, Hirata
T, et al: Activation of endoplasmic reticulum stress mediates
oxidative stress-induced apoptosis of granulosa cells in ovaries
affected by endometrioma. Mol Hum Reprod. 26:40–52. 2020.
View Article : Google Scholar
|
|
12
|
Ford EA, Frost ER, Beckett EL, Roman SD,
McLaughlin EA and Sutherland JM: Transcriptomic profiling of
neonatal mouse granulosa cells reveals new insights into primordial
follicle activation†. Biol Reprod. 106:503–514. 2022. View Article : Google Scholar :
|
|
13
|
Xing J, Qiao G, Luo X, Liu S, Chen S, Ye
G, Zhang C and Yi J: Ferredoxin 1 regulates granulosa cell
apoptosis and autophagy in polycystic ovary syndrome. Clin Sci
(Lond). 137:453–468. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Turathum B, Gao EM and Chian RC: The
function of cumulus cells in oocyte growth and maturation and in
subsequent ovulation and fertilization. Cells. 10:22922021.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Meldi KM, Gaconnet GA and Mayo KE: DNA
methylation and histone modifications are associated with
repression of the inhibin α promoter in the rat corpus luteum.
Endocrinology. 153:4905–4917. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pieroni L, Iavarone F, Olianas A, Greco V,
Desiderio C, Martelli C, Manconi B, Sanna MT, Messana I, Castagnola
M and Cabras T: Enrichments of post-translational modifications in
proteomic studies. J Sep Sci. 43:313–336. 2020. View Article : Google Scholar
|
|
17
|
Wang S, Osgood AO and Chatterjee A:
Uncovering posttranslational modification-associated
protein-protein interactions. Curr Opin Struct Biol. 74:1023522022.
View Article : Google Scholar
|
|
18
|
Walsh CT, Garneau-Tsodikova S and Gatto GJ
Jr: Protein posttranslational modifications: The chemistry of
proteome diversifications. Angew Chem Int Ed Engl. 44:7342–7372.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen M, Liu Y, Zuo M, Zhang M, Wang Z, Li
X, Yuan D, Xu H, Yu G and Li M: Integrated analysis reveals the
regulatory mechanism of the neddylation inhibitor MLN4924 on the
metabolic dysregulation in rabbit granulosa cells. BMC Genomics.
25:2542024. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lee L, Asada H, Kizuka F, Tamura I,
Maekawa R, Taketani T, Sato S, Yamagata Y, Tamura H and Sugino N:
Changes in histone modification and DNA methylation of the StAR and
Cyp19a1 promoter regions in granulosa cells undergoing
luteinization during ovulation in rats. Endocrinology. 154:458–470.
2013. View Article : Google Scholar
|
|
21
|
Bai X and Wang S: Signaling pathway
intervention in premature ovarian failure. Front Med (Lausanne).
9:9994402022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wu G, Pan Y, Chen M, Liu Z, Li C, Sheng Y,
Li H, Shen M and Liu H: Lactylation drives hCG-triggered
luteinization in hypoxic granulosa cells. Int J Biol Macromol.
280:1355802024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhou C, Zeng H, Xiao X, Wang L, Jia L, Shi
Y, Zhang M, Fang C, Zeng Y, Wu T, et al: Global crotonylome
identifies EP300-regulated ANXA2 crotonylation in cumulus cells as
a regulator of oocyte maturation. Int J Biol Macromol.
259:1291492024. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Qin X, Dang W, Yang X, Wang K, Kebreab E
and Lyu L: Neddylation inactivation affects cell cycle and
apoptosis in sheep follicular granulosa cells. J Cell Physiol.
237:3278–3291. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Le M, Li J, Zhang D, Yuan Y, Zhou C, He J,
Huang J, Hu L, Luo T and Zheng L: The emerging role of lysine
succinylation in ovarian aging. Reprod Biol Endocrinol. 21:382023.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sangalli JR, Nociti RP, Del Collado M,
Sampaio RV, da Silveira JC, Perecin F, Smith LC, Ross PJ and
Meirelles FV: Characterization of histone lysine
β-hydroxybutyrylation in bovine tissues, cells, and cumulus-oocyte
complexes. Mol Reprod Dev. 89:375–398. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Goud PT, Goud AP, Joshi N, Puscheck E,
Diamond MP and Abu-Soud HM: Dynamics of nitric oxide, altered
follicular microenvironment, and oocyte quality in women with
endometriosis. Fertil Steril. 102:151–159.e5. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Faramarzi A, Khalili MA and Jahromi MG: Is
there any correlation between apoptotic genes expression in cumulus
cells with embryo morphokinetics? Mol Biol Rep. 46:3663–3670. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zheng M, Liu M and Zhang C: Melatonin
ameliorates ovarian hyperstimulation syndrome (OHSS) through SESN2
regulated antiapoptosis. Obstet Gynecol Int. 2023:11212272023.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tabatabaie M, Amiri S, Golestan Jahromi M,
Sene AA, Zandieh Z, Mehdizadeh M and Amjadi F: The effect of
Myo-Inositol supplement on molecular regulation of
folliculogenesis, steroidogenesis, and assisted reproductive
technique outcomes in patients with polycystic ovarian syndrome.
Mol Biol Rep. 49:875–884. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang H, Qin F, Liu A, Sun Q, Wang Q, Xie
S, Lu S, Zhang D and Lu Z: Electro-acupuncture attenuates the mice
premature ovarian failure via mediating PI3K/AKT/mTOR pathway. Life
Sci. 217:169–175. 2019. View Article : Google Scholar
|
|
32
|
Chen D, Feng Y, Wu J, Zhou J, Li Z, Qiao
M, Chen T, Xu Z, Peng X and Mei S: Post-translational modifications
in mammalian folliculogenesis and ovarian pathologies. Cells.
14:12922025. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wu Y, Li M and Yang M: Post-translational
modifications in oocyte maturation and embryo development. Front
Cell Dev Biol. 9:6453182021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang L, Zhang Y and Sun H: Protein
modifications during early embryo development. Am J Reprod Immunol.
92:e700072024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang J, Sun X, Yang Z, Li S, Wang Y, Ren
R, Liu Z and Yu D: Epigenetic regulation in premature ovarian
failure: A literature review. Front Physiol. 13:9984242023.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gao Q, Ren J, Niu D, Guo L and Feng X:
Role of protein post-translational modifications in unexplained
recurrent pregnancy loss. Zhong Nan Da Xue Xue Bao Yi Xue Ban.
49:1495–1502. 2024.In English, Chinese. PubMed/NCBI
|
|
37
|
Lai H, Yang Y and Zhang J: Advances in
post-translational modifications and recurrent spontaneous
abortion. Gene. 927:1487002024. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Senthilkumar H, Chauhan SC and Arumugam M:
Unraveling the multifactorial pathophysiology of polycystic ovary
syndrome: exploring lifestyle, prenatal influences, neuroendocrine
dysfunction, and post-translational modifications. Mol Biol Rep.
52:9802025. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wei H, Huo P, Liu S, Huang H and Zhang S:
Posttranslational modifications in pathogenesis of PCOS. Front
Endocrinol (Lausanne). 13:10243202022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Siva AB, Srivastava P and Shivaji S:
Understanding the pathogenesis of endometriosis through proteomics:
Recent advances and future prospects. Proteomics Clin Appl.
8:86–98. 2014. View Article : Google Scholar
|
|
41
|
Huang JC, Duan CC, Jin S, Sheng CB, Wang
YS, Yue ZP and Guo B: HB-EGF induces mitochondrial dysfunction via
estrogen hypersecretion in granulosa cells dependent on
cAMP-PKA-JNK/ERK-Ca2+-FOXO1 pathway. Int J Biol Sci.
18:2047–2059. 2022. View Article : Google Scholar
|
|
42
|
Maucieri AM and Townson DH: Evidence and
manipulation of O-GlcNAcylation in granulosa cells of bovine antral
follicles†. Biol Reprod. 104:914–923. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Xue Y, Cheng X, Ma ZQ, Wang HP, Zhou C, Li
J, Zhang DL, Hu LL, Cui YF, Huang J, et al: Polystyrene
nanoplastics induce apoptosis, autophagy, and steroidogenesis
disruption in granulosa cells to reduce oocyte quality and
fertility by inhibiting the PI3K/AKT pathway in female mice. J
Nanobiotechnology. 22:4602024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Yang W, Yu S, Peng J, Chang P and Chen X:
FGF12 regulates cell cycle gene expression and promotes follicular
granulosa cell proliferation through ERK phosphorylation in geese.
Poult Sci. 102:1029372023. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhao S, Gu T, Weng K, Zhang Y, Cao Z,
Zhang Y, Zhao W, Chen G and Xu Q: Phosphoproteome reveals
extracellular regulated protein kinase phosphorylation mediated by
mitogen-activated protein kinase kinase-regulating granulosa cell
apoptosis in broody geese. Int J Mol Sci. 24:122782023. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Liu C, Pan B, Yang L, Wang B and Li J:
Beta defensin 3 enhances ovarian granulosa cell proliferation and
migration via ERK1/2 pathway in vitro†. Biol Reprod. 100:1057–1065.
2019. View Article : Google Scholar
|
|
47
|
Pan B, Zhan X and Li J: MicroRNA-574
impacts granulosa cell estradiol production via targeting TIMP3 and
ERK1/2 signaling pathway. Front Endocrinol (Lausanne).
13:8521272022. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Cottom J, Salvador LM, Maizels ET,
Reierstad S, Park Y, Carr DW, Davare MA, Hell JW, Palmer SS, Dent
P, et al: Follicle-stimulating hormone activates extracellular
signalregulated kinase but not extracellular signal-regulated
kinase kinase through a 100-kDa phosphotyrosine phosphatase. J Biol
Chem. 278:7167–7179. 2003. View Article : Google Scholar
|
|
49
|
Yan MQ, Zhu BH, Liu XH, Yang YM, Duan XY,
Wang Y, Sun H, Feng M, Li T and Liu XM: Mitoguardin 1 and 2 promote
granulosa cell proliferation by activating AKT and regulating the
Hippo-YAP1 signaling pathway. Cell Death Dis. 14:7792023.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Song A, Zhang S, Zhao X, Wu S, Qi X, Gao
S, Qi J, Li P and Tan J: Exosomes derived from menstrual blood
stromal cells ameliorated premature ovarian insufficiency and
granulosa cell apoptosis by regulating SMAD3/AKT/MDM2/P53 pathway
via delivery of thrombospondin-1. Biomed Pharmacother.
166:1153192023. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Alam H, Maizels ET, Park Y, Ghaey S,
Feiger ZJ, Chandel NS and Hunzicker-Dunn M: Follicle-stimulating
hormone activation of hypoxia-inducible factor-1 by the
phosphatidylinositol 3-kinase/AKT/Ras homolog enriched in brain
(Rheb)/mammalian target of rapamycin (mTOR) pathway is necessary
for induction of select protein markers of follicular
differentiation. J Biol Chem. 279:19431–19440. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hu J, Jin J, Qu Y, Liu W, Ma Z, Zhang J
and Chen F: ERO1α inhibits cell apoptosis and regulates
steroidogenesis in mouse granulosa cells. Mol Cell Endocrinol.
511:1108422020. View Article : Google Scholar
|
|
53
|
Zhang W, Gao L, Zhang X, Weng Y, Du Y, Sun
YL, Wei H, Hao T, Chen Y, Liang X and Zhang M: Theophylline
derivatives promote primordial follicle activation via
cAMP-PI3K/Akt pathway and ameliorate fertility deficits in
naturally aged mice. Int J Biol Sci. 20:5312–5329. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Tong C, Wu Y, Zhang L and Yu Y: Insulin
resistance, autophagy and apoptosis in patients with polycystic
ovary syndrome: Association with PI3K signaling pathway. Front
Endocrinol (Lausanne). 13:10911472022. View Article : Google Scholar :
|
|
55
|
Wu G, Chen M, He T, Pan Y, Li C, Liu Z, Li
H, Sheng Y, Dai W, Shen M and Liu H: Lactylation of CREB is
required for FSH-induced proliferation and differentiation of
ovarian granulosa cells. Nucleic Acids Res. 53:gkaf8822025.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Lanfranchi B, Rubia RF, Gassmann M,
Schuler G and Kowalewski MP: Transcriptional regulation of
HIF1α-mediated STAR expression in murine KK1 granulosa cell line
involves cJUN, CREB and CBP-dependent pathways. Gen Comp
Endocrinol. 315:1139232022. View Article : Google Scholar
|
|
57
|
Zhang X, Zhang W, Wang Z, Zheng N, Yuan F,
Li B, Li X, Deng L, Lin M, Chen X and Zhang M: Enhanced glycolysis
in granulosa cells promotes the activation of primordial follicles
through mTOR signaling. Cell Death Dis. 13:872022. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Wu G, Song D, Wu H, Zhao F, Ding W, Wang
Z, Shi F and Wei Q: Genistein ameliorates starvation-induced
porcine follicular granulosa cell apoptosis. Reproduction.
166:451–458. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Lin M, Hua R, Ma J, Zhou Y, Li P, Xu X, Yu
Z and Quan S: Bisphenol A promotes autophagy in ovarian granulosa
cells by inducing AMPK/mTOR/ULK1 signalling pathway. Environ Int.
147:1062982021. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Liu G, Wang Y, Zheng Y, Lv J, Li Y, Liu N,
Gao H, Ran H, Tang H and Jiang Z: PHB2 binds to ERβ to induce the
autophagy of porcine ovarian granulosa cells through mTOR
phosphorylation. Theriogenology. 198:114–122. 2023. View Article : Google Scholar
|
|
61
|
Liu J and Wang C: Lysophosphatidic acid is
associated with oocyte maturation by enhancing autophagy via
PI3K-AKT-mTOR signaling pathway in granulosa cells. J Ovarian Res.
16:1372023. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Tang X, Ma L, Guo S, Liang M and Jiang Z:
High doses of FSH induce autophagy in bovine ovarian granulosa
cells via the AKT/mTOR pathway. Reprod Domest Anim. 56:324–332.
2021. View Article : Google Scholar
|
|
63
|
Hu H, Zhang J, Xin X, Jin Y, Zhu Y, Zhang
H, Fan R, Ye Y, Jiang Y and Li D: Bushen jianpi tiaoxue decoction
(BJTD) inhibits the LIF-mTOR signaling axis to regulate
mitochondrial function and alleviate cyclophosphamide-induced
diminished ovarian reserve. Apoptosis. 30:1331–1350. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Ji R, Zhang Z, Yang Z, Chen X, Yin T and
Yang J: BOP1 contributes to the activation of autophagy in
polycystic ovary syndrome via nucleolar stress response. Cell Mol
Life Sci. 81:1012024. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Li C, Wu G, Ning C, Liu Z, Tao J, Lu X,
Shen M and Liu H: FOXO1-mediated nuclear sequestration of STAT3 and
AKT1 triggers FOXO3-dependent autophagic death in hypoxic granulosa
cells. Int J Biol Sci. 20:5939–5958. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Sun Z, Li P, Wang X, Lai S, Qiu H, Chen Z,
Hu S, Yao J and Shen J: GLP-1/GLP-1R signaling regulates ovarian
PCOS-associated granulosa cells proliferation and antiapoptosis by
modification of forkhead box protein O1 phosphorylation sites. Int
J Endocrinol. 2020:14843212020. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Lei Z, Ali I, Yang M, Yang C, Li Y and Li
L: Non-esterified fatty acid-induced apoptosis in bovine granulosa
cells via ROS-activated PI3K/AKT/FoxO1 pathway. Antioxidants
(Basel). 12:4342023. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Yan C, Ou Y, Sun X, Sun Y, Zhao J, Qin N
and Xu R: FSH-induced nuclear exclusion of FOXO1 mediated by
PI3K/Akt signaling pathway in granulosa cells is associated with
follicle selection and growth of the Hen Ovary. Cells. 14:18642025.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Yang F, Chen Y, Liu Q, Dai S and Zeng S:
Dynamics and regulations of BimEL Ser65 and Thr112 phosphorylation
in porcine granulosa cells during follicular atresia. Cells.
9:4022020. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Ling XM, Zhang XH, Tan Y, Yang JJ, Ji B,
Wu XR, Yi YK and Liang L: Protective effects of Oviductus
Ranae-containing serum on oxidative stress-induced apoptosis in rat
ovarian granulosa cells. J Ethnopharmacol. 208:138–148. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Przygrodzka E, Hou X, Zhang P, Plewes MR,
Franco R and Davis JS: PKA and AMPK signaling pathways
differentially regulate luteal steroidogenesis. Endocrinology.
162:bqab0152021. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Hou X, Arvisais EW and Davis JS:
Luteinizing hormone stimulates mammalian target of rapamycin
signaling in bovine luteal cells via pathways independent of AKT
and mitogen-activated protein kinase: Modulation of glycogen
synthase kinase 3 and AMP-activated protein kinase. Endocrinology.
151:2846–2857. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Tsui KH and Li CJ: Mitoquinone shifts
energy metabolism to reduce ROS-induced oxeiptosis in female
granulosa cells and mouse oocytes. Aging (Albany NY). 15:246–260.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zareifard A, Beaudry F and Ndiaye K: Janus
Kinase 3 phosphorylation and the JAK/STAT pathway are positively
modulated by follicle-stimulating hormone (FSH) in bovine granulosa
cells. BMC Mol Cell Biol. 24:212023. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Ardito F, Giuliani M, Perrone D, Troiano G
and Lo Muzio L: The crucial role of protein phosphorylation in cell
signaling and its use as targeted therapy (review). Int J Mol Med.
40:271–280. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Gershon E and Dekel N: Newly identified
regulators of ovarian folliculogenesis and ovulation. Int J Mol
Sci. 21:45652020. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Wang C, Lv X, Jiang C, Cordes CM, Fu L,
Lele SM and Davis JS: Transforming growth factor alpha (TGFα)
regulates granulosa cell tumor (GCT) cell proliferation and
migration through activation of multiple pathways. PLoS One.
7:e482992012. View Article : Google Scholar
|
|
78
|
Li H, Jing Y, Qu X, Yang J, Pan P, Liu X,
Gao H, Pei X, Zhang C and Yang Y: The activation of reticulophagy
by ER stress through the ATF4-MAP1LC3A-CCPG1 pathway in ovarian
granulosa cells is linked to apoptosis and necroptosis. Int J Mol
Sci. 24:27492023. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Lonergan P: Influence of progesterone on
oocyte quality and embryo development in cows. Theriogenology.
76:1594–1601. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Anthonsen MW, Rönnstrand L, Wernstedt C,
Degerman E and Holm C: Identification of novel phosphorylation
sites in hormone-sensitive lipase that are phosphorylated in
response to isoproterenol and govern activation properties in
vitro. J Biol Chem. 273:215–221. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Sha QQ, Jiang Y, Yu C, Xiang Y, Dai XX,
Jiang JC, Ou XH and Fan HY: CFP1-dependent histone H3K4
trimethylation in murine oocytes facilitates ovarian follicle
recruitment and ovulation in a cell-nonautonomous manner. Cell Mol
Life Sci. 77:2997–3012. 2020. View Article : Google Scholar
|
|
82
|
Maekawa R, Lee L, Okada M, Asada H,
Shinagawa M, Tamura I, Sato S, Tamura H and Sugino N: Changes in
gene expression of histone modification enzymes in rat granulosa
cells undergoing luteinization during ovulation. J Ovarian Res.
9:152016. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Sugino N: Molecular mechanisms of
luteinization. Obstet Gynecol Sci. 57:93–101. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Hosseini E, Mehraein F, Shahhoseini M,
Karimian L, Nikmard F, Ashrafi M, Afsharian P and Aflatoonian R:
Epigenetic alterations of CYP19A1 gene in cumulus cells and its
relevance to infertility in endometriosis. J Assist Reprod Genet.
33:1105–1113. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Hosseini E, Shahhoseini M, Afsharian P,
Karimian L, Ashrafi M, Mehraein F and Afatoonian R: Role of
epigenetic modifications in the aberrant CYP19A1 gene expression in
polycystic ovary syndrome. Arch Med Sci. 15:887–895. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Okada M, Lee L, Maekawa R, Sato S,
Kajimura T, Shinagawa M, Tamura I, Taketani T, Asada H, Tamura H
and Sugino N: Epigenetic changes of the Cyp11a1 promoter region in
granulosa cells undergoing luteinization during ovulation in female
rats. Endocrinology. 157:3344–3354. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Seneda MM, Godmann M, Murphy BD, Kimmins S
and Bordignon V: Developmental regulation of histone H3 methylation
at lysine 4 in the porcine ovary. Reproduction. 135:829–838. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Meunier H, Cajander SB, Roberts VJ, Rivier
C, Sawchenko PE, Hsueh AJ and Vale W: Rapid changes in the
expression of inhibin alpha-, beta A-, and beta B-subunits in
ovarian cell types during the rat estrous cycle. Mol Endocrinol.
2:1352–1363. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Shinagawa M, Tamura I, Maekawa R, Sato S,
Shirafuta Y, Mihara Y, Okada-Matsumoto M, Taketani T, Asada H,
Tamura H and Sugino N: C/EBPβ regulates Vegf gene expression in
granulosa cells undergoing luteinization during ovulation in female
rats. Sci Rep. 9:7142019. View Article : Google Scholar
|
|
90
|
Cui LX, Tian YQ, Hao HS, Zou HY, Pang YW,
Zhao SJ, Zhao XM, Zhu HB and Du WH: Knockdown of ASH1L
methyltransferase induced apoptosis inhibiting proliferation and
H3K36 methylation in bovine cumulus cells. Theriogenology.
161:65–73. 2021. View Article : Google Scholar
|
|
91
|
Zhong Y, Li L, He Y, He B, Li Z, Zhang Z,
Zhang H, Yuan X and Li J: Activation of steroidogenesis,
anti-apoptotic activity, and proliferation in porcine granulosa
cells by RUNX1 is negatively regulated by H3K27me3 transcriptional
repression. Genes (Basel). 11:4952020. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Chen W, Dong L, Wei C and Wu H: Role of
epigenetic regulation in diminished ovarian reserve. J Assist
Reprod Genet. 42:389–403. 2025. View Article : Google Scholar :
|
|
93
|
Eslami H, Eslami A, Favaedi R, Asadpour U,
Zari Moradi S, Eftekhari-Yazdi P, Madani T, Shahhoseini M and
Mohseni Meybodi A: Epigenetic aberration of FMR1 gene in infertile
women with diminished ovarian reserve. Cell J. 20:78–83.
2018.PubMed/NCBI
|
|
94
|
Wang D, Lu X, Jiang Y, Pan L, Zhu F, Yu A,
Zhao M, Yang M, Bi J, He X, et al: The chromatin remodeling protein
BPTF mediates cell cycle, proliferation and apoptosis in porcine
ovarian granulosa cells. Theriogenology. 211:172–181. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Yang Y, Cai Y, Guo J, Dai K, Liu L, Chen
Z, Wang F and Deng M: Knockdown of KDM5B leads to DNA damage and
cell cycle arrest in granulosa cells via MTF1. Curr Issues Mol
Biol. 45:3219–3237. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Glanzner WG, Gutierrez K, Rissi VB, de
Macedo MP, Lopez R, Currin L, Dicks N, Baldassarre H, Agellon LB
and Bordignon V: Histone lysine demethylases KDM5B and KDM5C
modulate genome activation and stability in porcine embryos. Front
Cell Dev Biol. 8:1512020. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Xiong X, Ma H, Min X, Su F, Xiong Y and Li
J: Effects of demethylase KDM4B on the biological characteristics
and function of yak cumulus cells in vitro. Theriogenology.
174:85–93. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Chen M, Dong F, Chen M, Shen Z, Wu H, Cen
C, Cui X, Bao S and Gao F: PRMT5 regulates ovarian follicle
development by facilitating Wt1 translation. Elife. 10:e689302021.
View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Bhat KP, Ümit Kaniskan H, Jin J and Gozani
O: Epigenetics and beyond: Targeting writers of protein lysine
methylation to treat disease. Nat Rev Drug Discov. 20:265–286.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Jambhekar A, Dhall A and Shi Y: Roles and
regulation of histone methylation in animal development. Nat Rev
Mol Cell Biol. 20:625–641. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Gong F and Miller KM: Histone methylation
and the DNA damage response. Mutat Res Rev Mutat Res. 780:37–47.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Bilmez Y, Talibova G and Ozturk S:
Expression of the histone lysine methyltransferases SETD1B, SETDB1,
SETD2, and CFP1 exhibits significant changes in the oocytes and
granulosa cells of aged mouse ovaries. Histochem Cell Biol.
158:79–95. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Karkhanis V, Hu YJ, Baiocchi RA, Imbalzano
AN and Sif S: Versatility of PRMT5-induced methylation in growth
control and development. Trends Biochem Sci. 36:633–641. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Zhao F, Lan Y, Chen T, Xin Z, Liang Y, Li
Y, Wang S, Zhang J and Yang X: Live birth rate comparison of three
controlled ovarian stimulation protocols for in vitro
fertilization-embryo transfer in patients with diminished ovarian
reserve after endometrioma cystectomy: A retrospective study. J
Ovarian Res. 13:232020. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Olsen KW, Castillo-Fernandez J, Chan AC,
la Cour Freiesleben N, Zedeler A, Bungum M, Cardona A, Perry JRB,
Skouby SO, Hoffmann ER, et al: Identification of a unique
epigenetic profile in women with diminished ovarian reserve. Fertil
Steril. 115:732–741. 2021. View Article : Google Scholar
|
|
106
|
Jin J, Ren P, Li X, Zhang Y, Yang W, Ma Y,
Lai M, Yu C, Zhang S and Zhang YL: Ovulatory signal-triggered
chromatin remodeling in ovarian granulosa cells by HDAC2
phosphorylation activation-mediated histone deacetylation.
Epigenetics Chromatin. 16:112023. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Ye Q, Zeng X, Wang S, Zeng X, Yang G, Ye
C, Cai S, Chen M, Li S and Qiao S: Butyrate drives the acetylation
of histone H3K9 to activate steroidogenesis through PPARγ and PGC1α
pathways in ovarian granulosa cells. FASEB J. 35:e213162021.
View Article : Google Scholar
|
|
108
|
Zhou X, He Y, Quan H, Yang J, Li S, Jiang
Y, Li J and Yuan X: Exposure to nicotine regulates prostaglandin E2
secretion and autophagy of granulosa cells to retard follicular
maturation in mammals. Ecotoxicol Environ Saf. 277:1163582024.
View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Zhang YL, Xia Y, Yu C, Richards JS, Liu J
and Fan HY: CBP-CITED4 is required for luteinizing
hormone-triggered target gene expression during ovulation. Mol Hum
Reprod. 20:850–860. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Min Z, Long X, Zhao H, Zhen X, Li R, Li M,
Fan Y, Yu Y, Zhao Y and Qiao J: Protein lysine acetylation in
ovarian granulosa cells affects metabolic homeostasis and clinical
presentations of women with polycystic ovary syndrome. Front Cell
Dev Biol. 8:5670282020. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Zhao S, Cui H, Fang X, Xia W, Tao C and Li
J: Increased DNMT1 acetylation leads to global DNA methylation
suppression in follicular granulosa cells during reproductive aging
in mammals. BMC Genomics. 25:10302024. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Zhang M, Zhang Q, Hu Y, Xu L, Jiang Y,
Zhang C, Ding L, Jiang R, Sun J, Sun H and Yan G: miR-181a
increases FoxO1 acetylation and promotes granulosa cell apoptosis
via SIRT1 downregulation. Cell Death Dis. 8:e30882017. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Kong C, Su J, Wang Q, Liu K, Fu R and Sui
S: Signaling pathways of periplaneta americana peptide resist
H2O2-induced apoptosis in pig-ovary granulosa
cells through FoxO1. Theriogenology. 183:108–119. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Guo B, Zhang S, Wang S, Zhang H, Fang J,
Kang N, Zhen X, Zhang Y, Zhou J, Yan G, et al: Decreased HAT1
expression in granulosa cells disturbs oocyte meiosis during mouse
ovarian aging. Reprod Biol Endocrinol. 21:1032023. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Park SA, Joo NR, Park JH and Oh SM: Role
of the SIRT1/p53 regulatory axis in oxidative stress-mediated
granulosa cell apoptosis. Mol Med Rep. 23:202021.
|
|
116
|
Joo NR, Park SA, Park JH and Oh SM: TOPK
inhibits TNF-α-induced granulosa cell apoptosis via regulation of
SIRT1/p53. Biochem Biophys Res Commun. 664:128–135. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Park JH, Park SA, Lee YJ, Joo NR, Shin J
and Oh SM: TOPK inhibition accelerates oxidative stress-induced
granulosa cell apoptosis via the p53/SIRT1 axis. Int J Mol Med.
46:1923–1937. 2020.PubMed/NCBI
|
|
118
|
Shvedunova M and Akhtar A: Modulation of
cellular processes by histone and non-histone protein acetylation.
Nat Rev Mol Cell Biol. 23:329–349. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Xu Y and Wan W: Acetylation in the
regulation of autophagy. Autophagy. 19:379–387. 2023. View Article : Google Scholar :
|
|
120
|
Narita T, Weinert BT and Choudhary C:
Functions and mechanisms of non-histone protein acetylation. Nat
Rev Mol Cell Biol. 20:156–174. 2019. View Article : Google Scholar
|
|
121
|
Shen M, Jiang Y, Guan Z, Cao Y, Li L, Liu
H and Sun SC: Protective mechanism of FSH against oxidative damage
in mouse ovarian granulosa cells by repressing autophagy.
Autophagy. 13:1364–1385. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Chen C, Zhou M, Ge Y and Wang X: SIRT1 and
aging related signaling pathways. Mech Ageing Dev. 187:1112152020.
View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Wang X, Lin C, Zhao X, Liu A, Zhu J, Li X
and Song L: Acylglycerol kinase promotes cell proliferation and
tumorigenicity in breast cancer via suppression of the FOXO1
transcription factor. Mol Cancer. 13:1062014. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Xu G, Dong Y, Wang Z, Ding H, Wang J, Zhao
J, Liu H and Lv W: Melatonin attenuates oxidative stress-induced
apoptosis of bovine ovarian granulosa cells by promoting mitophagy
via SIRT1/FoxO1 signaling pathway. Int J Mol Sci. 24:128542023.
View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Sun L, Ye H, Tian H, Xu L, Cai J, Zhang C,
Wang R, Yang H, Zhao S, Zhang J and Gao S: The E3 ubiquitin ligase
SYVN1 plays an antiapoptotic role in polycystic ovary syndrome by
regulating mitochondrial fission. Oxid Med Cell Longev.
2022:36393022022. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Ma J, Chen S, Liu J, Liao Y, Li L, Wang
CC, Song S, Feng R, Hu H and Quan S: Cryptochrome 1 regulates
ovarian granulosa cell senescence through NCOA4-mediated
ferritinophagy. Free Radic Biol Med. 217:1–14. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Ma LZ, Wang A, Lai YH, Zhang J, Zhang XF,
Chen SL and Zhou XY: USP14 inhibition promotes DNA damage repair
and represses ovarian granulosa cell senescence in premature
ovarian insufficiency. J Transl Med. 22:8342024. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Shi S, Chu G, Zhang L, Yuan H, Madaniyati
M, Zhou X, Wang L, Cai C, Pang W, Gao L and Yang G: Deubiquitinase
UCHL1 regulates estradiol synthesis by stabilizing
voltage-dependent anion channel 2. J Biol Chem. 299:1053162023.
View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Liu X, Sun C, Zou K, Li C, Chen X, Gu H,
Zhou Z, Yang Z, Tu Y, Qin N, et al: Novel PGK1 determines
SKP2-dependent AR stability and reprograms granular cell glucose
metabolism facilitating ovulation dysfunction. EBioMedicine.
61:1030582020. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Tang H, Jiang X, Hua Y, Li H, Zhu C, Hao
X, Yi M and Li L: NEDD4L facilitates granulosa cell ferroptosis by
promoting GPX4 ubiquitination and degradation. Endocr Connect.
12:e2204592023. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Han S, Yu C, Qiu M, Xiong X, Peng H, Song
X, Hu C, Zhang Z, Xia B, Yang L, et al: USP13 regulates ferroptosis
in chicken follicle granulosa cells by deubiquitinating ATG7. Poult
Sci. 103:1042092024. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Zhang Y, Wang H, Zhu Y, Hou X, Li X, Zhou
X, Ge L, Xu J and Su Y: The novel peptide PFAP1 promotes primordial
follicle activation by binding to MCM5. FASEB J. 37:e229312023.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Yang L, Wang S, Pan Z, Du X and Li Q:
TGFBR2 is a novel substrate and indirect transcription target of
deubiquitylase USP9X in granulosa cells. J Cell Physiol.
237:2969–2979. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Wang Y and Zeng S: Melatonin promotes
ubiquitination of phosphorylated pro-apoptotic protein
Bcl-2-interacting mediator of cell death-extra long
(BimEL) in porcine granulosa cells. Int J Mol Sci.
19:34312018. View Article : Google Scholar
|
|
135
|
Kwon YT and Ciechanover A: The ubiquitin
code in the ubiquitin-proteasome system and autophagy. Trends
Biochem Sci. 42:873–886. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Zhao T, He M, Zhu Z, Zhang T, Zheng W, Qin
S, Gao M, Wang W, Chen Z, Han J, et al: P62 promotes FSH-induced
antral follicle formation by directing degradation of ubiquitinated
WT1. Cell Mol Life Sci. 81:2212024. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Wang Y, Zhuo A, Yang Y, Wang Q, Xie J, Ma
W, Chen Y, Gao M, Tang L and Fu X: Regulatory T cells
overexpressing Peli1 show better efficacy in repairing ovarian
endocrine function in autoimmune premature ovarian insufficiency.
Lab Invest. 103:1000052023. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Wang TF, Feng ZQ, Sun YW, Zhao SJ, Zou HY,
Hao HS, Du WH, Zhao XM, Zhu HB and Pang YW: Disruption of
O-GlcNAcylation homeostasis induced ovarian granulosa cell injury
in bovine. Int J Mol Sci. 23:78152022. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Lyu L, Qin X, Xiu H, Qu Y, Wang Y, Yang X,
Dang W and Kebreab E: Neddylation inhibition affects cell
proliferation and steroidogenesis in sheep follicular granulosa
cells. Theriogenology. 237:99–109. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Zhang Y, He F, Cai N, Chen G, Wang Y, Bai
W and Guo P: SIRT5 regulates granulosa cell proliferation and
apoptosis in polycystic ovarian syndrome via desuccinylation of
GLI1. Gynecol Endocrinol. 41:25155162025. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Wang T, Ye Z, Li Z, Jing DS, Fan GX, Liu
MQ, Zhuo QF, Ji SR, Yu XJ, Xu XW and Qin Y: Lactate-induced protein
lactylation: A bridge between epigenetics and metabolic
reprogramming in cancer. Cell Prolif. 56:e134782023. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Tan M, Luo H, Lee S, Jin F, Yang JS,
Montellier E, Buchou T, Cheng Z, Rousseaux S, Rajagopal N, et al:
Identification of 67 histone marks and histone lysine crotonylation
as a new type of histone modification. Cell. 146:1016–1028. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Yang P, Qin Y, Zeng L, He Y, Xie Y, Cheng
X, Huang W and Cao L: Crotonylation and disease: Current progress
and future perspectives. Biomed Pharmacother. 165:1151082023.
View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Wan J, Liu H, Chu J and Zhang H: Functions
and mechanisms of lysine crotonylation. J Cell Mol Med.
23:7163–7169. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Enchev RI, Schulman BA and Peter M:
Protein neddylation: Beyond cullin-RING ligases. Nat Rev Mol Cell
Biol. 16:30–44. 2015. View Article : Google Scholar
|
|
146
|
Wu D, Cai Y and Jin J: Potential
coordination role between O-GlcNAcylation and epigenetics. Protein
Cell. 8:713–723. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Roy S, Gandra D, Seger C, Biswas A,
Kushnir VA, Gleicher N, Kumar TR and Sen A: Oocyte-derived factors
(GDF9 and BMP15) and FSH regulate AMH expression via modulation of
H3K27AC in granulosa cells. Endocrinology. 159:3433–3445. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Schreiber NB and Spicer LJ: Effects of
fibroblast growth factor 9 (FGF9) on steroidogenesis and gene
expression and control of FGF9 mRNA in bovine granulosa cells.
Endocrinology. 153:4491–4501. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Guo Y, Zhang Y, Wang Y, Chen Q, Sun Y,
Kang L and Jiang Y: Phosphorylation of LSD1 at serine 54 regulates
genes involved in follicle selection by enhancing demethylation
activity in chicken ovarian granulosa cells. Poult Sci.
103:1038502024. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Kimura N, Hoshino Y, Totsukawa K and Sato
E: Cellular and molecular events during oocyte maturation in
mammals: Molecules of cumulus-oocyte complex matrix and signalling
pathways regulating meiotic progression. Soc Reprod Fertil Suppl.
63:327–342. 2007.PubMed/NCBI
|
|
151
|
Liao HQ and Zhou J, Cao Y, Nie YL, Li MQ
and Zhou J: Vigilin interacts with ER-β to protect against palmitic
acid-induced granulosa cells apoptosis via inhibiting
calcineurin-mediated Drp1 signaling pathway. Steroids.
163:1086992020. View Article : Google Scholar
|
|
152
|
Wan J and Liu S: Rosiglitazone ameliorates
palmitic acid-induced endoplasmic reticulum stress and
steroidogenic capacity in granulosa cells. Reprod Biol. 20:293–299.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Shibahara H, Ishiguro A, Inoue Y, Koumei
S, Kuwayama T and Iwata H: Mechanism of palmitic acid-induced
deterioration of in vitro development of porcine oocytes and
granulosa cells. Theriogenology. 141:54–61. 2020. View Article : Google Scholar
|
|
154
|
Benoit G, Warma A, Lussier JG and Ndiaye
K: Gonadotropin regulation of ankyrin-repeat and SOCS-box protein 9
(ASB9) in ovarian follicles and identification of binding partners.
PLoS One. 14:e02125712019. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Wang X, Zhou L, Xu A, Nima D and Dong Z:
Effects of stem cell factor in follicular fluid and granulosa cells
on oocyte maturity and clinical pregnancy. Medicine (Baltimore).
102:e367492023. View Article : Google Scholar :
|
|
156
|
Yang X, Liu P, He H, Qi D and Yan L:
Comprehensive analysis of ovarian granulosa cell proteomics and
phosphoproteomics in PCOS patients without insulin resistance. Mol
Hum Reprod. 30:gaae0052024. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Wang M, Chen T, Zheng J, Shen C, He G,
Zhang J, Zhang B and Zheng L: The role of CLDN11 in promotion of
granulosa cell proliferation in polycystic ovary syndrome via
activation of the PI3K-AKT signalling pathway. Sci Rep.
15:35332025. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Yang F, Liu Q, Chen Y, Ye H, Wang H and
Zeng S: Integrative Proteomic and phosphoproteomic analyses of
granulosa cells during follicular atresia in porcine. Front Cell
Dev Biol. 8:6249852021. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Wang Y, Zhang J, Liang J, Jia L, Niu S,
Cheng K, Yang C, Lu Z, Mu L, Yang X, et al: In vivo promotion of
primordial follicle activation by stem cell factor treatment in
mice with premature ovarian insufficiency and advanced age. Mol Hum
Reprod. 29:gaac0412022. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Li Q, Chen J, Yu X and Gao JM: A mini
review of nervonic acid: Source, production, and biological
functions. Food Chem. 301:1252862019. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Zeng X, Fan X, Yu H, Cai S, Zhou L, Wu H,
Zhang Z, Quan S, Li S, Wang X, et al: Nervonic acid triggered
ovarian inflammation by inducing mitochondrial oxidative stress to
activate NLRP3/IL-1β pathway. J Adv Res. 73:73–91. 2025. View Article : Google Scholar :
|
|
162
|
Fotovati A, Abu-Ali S, Nakayama K and
Nakayama KI: Impaired ovarian development and reduced fertility in
female mice deficient in Skp2. J Anat. 218:668–677. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Lv S, Liu M, Xu L and Zhang C:
Downregulation of decidual SKP2 is associated with human recurrent
miscarriage. Reprod Biol Endocrinol. 19:882021. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Ding L, Yan G, Wang B, Xu L, Gu Y, Ru T,
Cui X, Lei L, Liu J, Sheng X, et al: Transplantation of UC-MSCs on
collagen scaffold activates follicles in dormant ovaries of POF
patients with long history of infertility. Sci China Life Sci.
61:1554–1565. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Nosratpour S and Ndiaye K: Ankyrin-repeat
and SOCS box-containing protein 9 (ASB9) regulates ovarian
granulosa cells function and MAPK signaling. Mol Reprod Dev.
88:830–843. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Wei Y, Lin Z, Huang Q, Wu H, Wang R and
Wang J: Burden of female infertility in 204 countries and
territories, 1990-2021: Results from the global burden of disease
study 2021. J Psychosom Obstet Gynaecol. 46:24596182025. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Zhu M, Miao S, Zhou W, Elnesr SS, Dong X
and Zou X: MAPK, AKT/FoxO3a and mTOR pathways are involved in
cadmium regulating the cell cycle, proliferation and apoptosis of
chicken follicular granulosa cells. Ecotoxicol Environ Saf.
214:1120912021. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Han Z, Liu J, Liang T, Yin J, Wei J, Zeng
Q, Cao W, Liu C and Sun S: Exposure to ambient particulate matter
and ovarian reserve impairment among reproductive age women in
China. J Hazard Mater. 480:1362122024. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Fauque P, De Mouzon J, Devaux A, Epelboin
S, Gervoise-Boyer MJ, Levy R, Valentin M, Viot G, Bergère A, De
Vienne C, et al: Reproductive technologies, female infertility, and
the risk of imprinting-related disorders. Clin Epigenetics.
12:1912020. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Dummer R, Ascierto PA, Gogas HJ, Arance A,
Mandala M, Liszkay G, Garbe C, Schadendorf D, Krajsova I, Gutzmer
R, et al: Encorafenib plus binimetinib versus vemurafenib or
encorafenib in patients with BRAF-mutant melanoma (COLUMBUS): A
multicentre, open-label, randomised phase 3 trial. Lancet Oncol.
19:603–615. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Kopp KO, Greer ME, Glotfelty EJ, Hsueh SC,
Tweedie D, Kim DS, Reale M, Vargesson N and Greig NH: A new
generation of IMiDs as treatments for neuroinflammatory and
neurodegenerative disorders. Biomolecules. 13:7472023. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Sun X, Zhang Q, Lin X, Shu P, Gao X and
Shen K: Imatinib induces ferroptosis in gastrointestinal stromal
tumors by promoting STUB1-mediated GPX4 ubiquitination. Cell Death
Dis. 14:8392023. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Wang F, Jin Y, Wang M, Luo HY, Fang WJ,
Wang YN, Chen YX, Huang RJ, Guan WL, Li JB, et al: Combined
anti-PD-1, HDAC inhibitor and anti-VEGF for MSS/pMMR colorectal
cancer: a randomized phase 2 trial. Nat Med. 30:1035–1043. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Nguyen TTA and Demeestere I: A journey to
reach the ovary using next-generation technologies. Int J Mol Sci.
24:165932023. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Shi M, Li X, Xing L, Li Z, Zhou S, Wang Z,
Zou X, She Y, Zhao R and Qin D: Polycystic ovary syndrome and the
potential for nanomaterial-based drug delivery in therapy of this
disease. Pharmaceutics. 16:15562024. View Article : Google Scholar :
|
|
176
|
Chen Y, Xie M, Wu S, Deng Z, Tang Y, Guan
Y, Ye Y, He Q and Li L: Multi-omics approach to reveal follicular
metabolic changes and their effects on oocyte competence in PCOS
patients. Front Endocrinol (Lausanne). 15:14265172024. View Article : Google Scholar : PubMed/NCBI
|