GCs are the specialized somatic cells of the sex
cord-stromal lineage found within the mammalian ovarian follicle,
where they typically form either a monolayer or a multilayer around
the oocyte to facilitate follicular development and steroid hormone
synthesis (1). During follicular
development, GCs differentiate into two functionally distinct
subpopulations: Mural GCs, which align with the basal lamina to
constitute the follicular wall, and cumulus cells, which maintain
communication with the central oocyte (2). These cells also establish gap
junction-mediated communication with the oocyte, providing
essential nutrient support and regulating the microenvironment
necessary for oocyte maturation (3,4).
The proper proliferation and differentiation of GCs are imperative
for the correct formation of follicles and the subsequent quality
of the embryo, both of which are key determinants of female
fertility (5). Infertility has
increasingly been acknowledged as a considerable global public
health issue. Female factors contribute to >50% of infertility
cases, predominantly driven by environmental degradation and
adverse lifestyle conditions (6). Dysfunctions in GCs include a range
of pathophysiological processes, such as imbalances in
proliferation and differentiation, abnormalities in cellular
senescence and apoptosis, and disturbances in hormone synthesis
(7,8). The mechanisms contributing to these
dysfunctions are complexly associated with excessive oxidative
stress, mitochondrial dysfunction, abnormal inflammatory responses
and endoplasmic reticulum stress (ERS) (8-11). Such dysfunctions can markedly
compromise female fertility through various molecular pathways,
including the premature depletion of the primordial follicle pool
(12), disrupted
folliculogenesis (13),
ovulation disorders (14) and
embryo implantation failures (15). Given the pivotal role of GCs in
folliculogenesis, a comprehensive understanding of their regulatory
mechanisms is indispensable. Acquiring this knowledge is key for
identifying potential biomarkers for the early diagnosis of
infertility and could lay the groundwork for the development of
GC-targeted therapeutic strategies, thereby advancing precision
medicine in the management of clinical infertility.
PTMs of proteins refer to the process of adding or
removing chemical groups from amino acid residues in the
polypeptide chains of proteins. These modifications, defined as the
side chain modification of amino acids that occur after protein
synthesis (16,17), provide a powerful means to
augment and regulate protein function. PTMs are essential cellular
mechanisms that regulate protein activity, stability and
subcellular localization through reversible covalent modifications.
As fundamental regulatory mechanisms, PTMs carry out a pivotal role
in orchestrating diverse biological processes (18). Within GCs, PTMs meticulously
regulate cellular proliferation, differentiation, apoptosis and
hormone secretion by modulating signaling pathways, epigenetic
landscapes, proteostasis and metabolic modifications (7,19,20). Canonical PTMs, including
phosphorylation, methylation, ubiquitination and acetylation, are
important in regulating the functions of GCs. For example,
PTM-mediated activation of the PI3K/AKT/FOXO3 signaling pathway is
essential for primordial follicle activation, underscoring the
fundamental role of PTMs in folliculogenesis (21). Additionally, dynamic histone
modifications, particularly H3K4me3, H3K9me and H3K27me3, act as
epigenetic switches that precisely regulate progesterone production
during luteinization (20).
Advancements in high-resolution mass spectrometry have revealed
several novel PTMs including lactylation (22), crotonylation (23), neddylation (24), lysine succinylation (25) and lysine β-hydroxybutyrylation
(26), which notably influence
GC. Accumulating evidence suggests that aberrant PTM patterns in
GCs are a principal factor contributing to infertility. Thus,
elucidating the PTM profiles in GCs not only enhances the
understanding of the pathogenesis of infertility but also aids in
the development of therapeutic strategies targeting specific PTM
enzymes.
Assisted reproductive technology (ART) serves as an
important intervention for fertility preservation. Dysfunctional
GCs impair oocyte quality, maturation and embryo viability, thereby
reducing the success rates of ART (5,27,28). Almeida et al (5) suggested that a pre-ART evaluation
of GC quality could enhance the selection of oocytes and embryos.
Notably, ovarian hyperstimulation syndrome (OHSS), a severe
complication of ovarian stimulation during ART, may benefit from
therapeutic approaches involving PTMs in the future. Zheng et
al (29) demonstrated that
melatonin mitigates reactive oxygen species (ROS)-induced apoptosis
in GCs via the phosphorylation regulation of the SESN2-AMPK-mTOR
axis, suggesting considerable clinical potential for OHSS therapy.
Furthermore, a clinical study on polycystic ovary syndrome (PCOS)
indicated that Myo-Inositol supplementation improves
steroidogenesis, oocyte maturation, fertilization rates and embryo
quality through phosphorylation-dependent modulation of the ERK1/2
and AKT pathway in cumulus cells (30). Concurrently, Zhang et al
(31) confirmed that
electro-acupuncture improves ovarian function in a premature
ovarian failure (POF) mouse model through phosphorylation-mediated
regulation of the PI3K/AKT/mTOR signaling cascade. Collectively,
these studies underscore that PTM-targeted interventions in GCs
represent a promising therapeutic approach for treating female
infertility.
However, current reviews on PTMs in female
infertility focus on physiological processes such as
folliculogenesis (32), oocyte
meiotic maturation and embryonic development (33,34) or are restricted to one specific
pathological context, such as POF (35), unexplained recurrent pregnancy
loss (36), recurrent
spontaneous abortion (37), PCOS
(38,39) and endometriosis (40). Although PTMs have been confirmed
to be involved in GCs, the majority of studies are fragmented,
focusing largely on a single class of modifications, without
integrating how different PTM pathways interact (41-43). Comprehensive elucidation of the
dynamic interplay between the full spectrum of PTMs and GC
physiology remains limited. From the perspective of GCs, the
present review integrates studies on how various PTMs regulate
diverse vital activities of GCs, including proliferation,
differentiation, apoptosis and steroid hormone secretion, evaluates
their potential for therapeutic targeting and ultimately aims to
establish a novel framework for understanding the regulatory
networks of GCs, thereby facilitating PTM-based clinical
interventions for infertility.
Phosphorylation is one of the most common PTMs of
proteins. Protein phosphorylation refers to the addition of a
phosphate group to proteins, mainly serine, threonine and tyrosine,
and activates/inactivates numerous enzymes and receptors through
phosphorylation and dephosphorylation to regulate the function and
localization of proteins, which is an important cellular regulatory
mechanism (75). Protein
phosphorylation serves as a fundamental regulatory mechanism in
folliculogenesis. This reversible modification dynamically controls
the fate of GCs during follicular development through precisely
regulated signal transduction cascades. Dysregulated
phosphorylation disrupts key physiological processes in GCs,
including proliferation, autophagy, differentiation, apoptosis and
hormone secretion. It also impairs the bidirectional metabolic
exchange between GCs and oocytes, ultimately leading to aberrant
follicular atresia and diminished female fertility (21,76).
The regulation of GC proliferation is meticulously
governed by phosphorylation-dependent signaling pathways, notably
the ERK, Hippo/YAP1 and EGFR/PI3K/AKT/mTOR pathways (77). For example, FGF12 has been shown
to promote GC proliferation by enhancing the phosphorylation of
ERK1/2 (44). Additionally, the
mitochondrial proteins MIGA1/2 support the proliferation of GCs
through dual mechanisms that involve the phosphorylation of both
AKT and Hippo/YAP1 signaling, specifically through the
phosphorylation of YAP1 at Ser127 (49). Elevated levels of ROS induce
pathological oxidative stress, leading to disruptions in
mitochondrial homeostasis and ERS, which are pivotal in driving
apoptosis in GCs. It was demonstrated that exposure to ROS in mouse
GCs decreases phosphorylation at Ser637 while increasing
phosphorylation at Ser616 on the mitochondrial fission protein Drp
1, a dysregulated phosphorylation pattern that exacerbates
mitochondrial dysfunction and ultimately triggers oxeiptosis in GCs
(73). Xue et al
(43) found that an imbalance in
ROS suppresses AKT phosphorylation, deactivating the PI3K/AKT
pathway and initiating autophagy via reduced mTOR inhibition.
Additionally, GLP-1/GLP-1R signaling influences GC proliferation
and apoptosis through the phosphorylation of FOXO1 at both Ser256
and Ser319 (66). Concurrently,
excessive ERS triggers reticulophagy as a compensatory response,
mitigating GC apoptosis-mediated follicular atresia in GCs
(78).
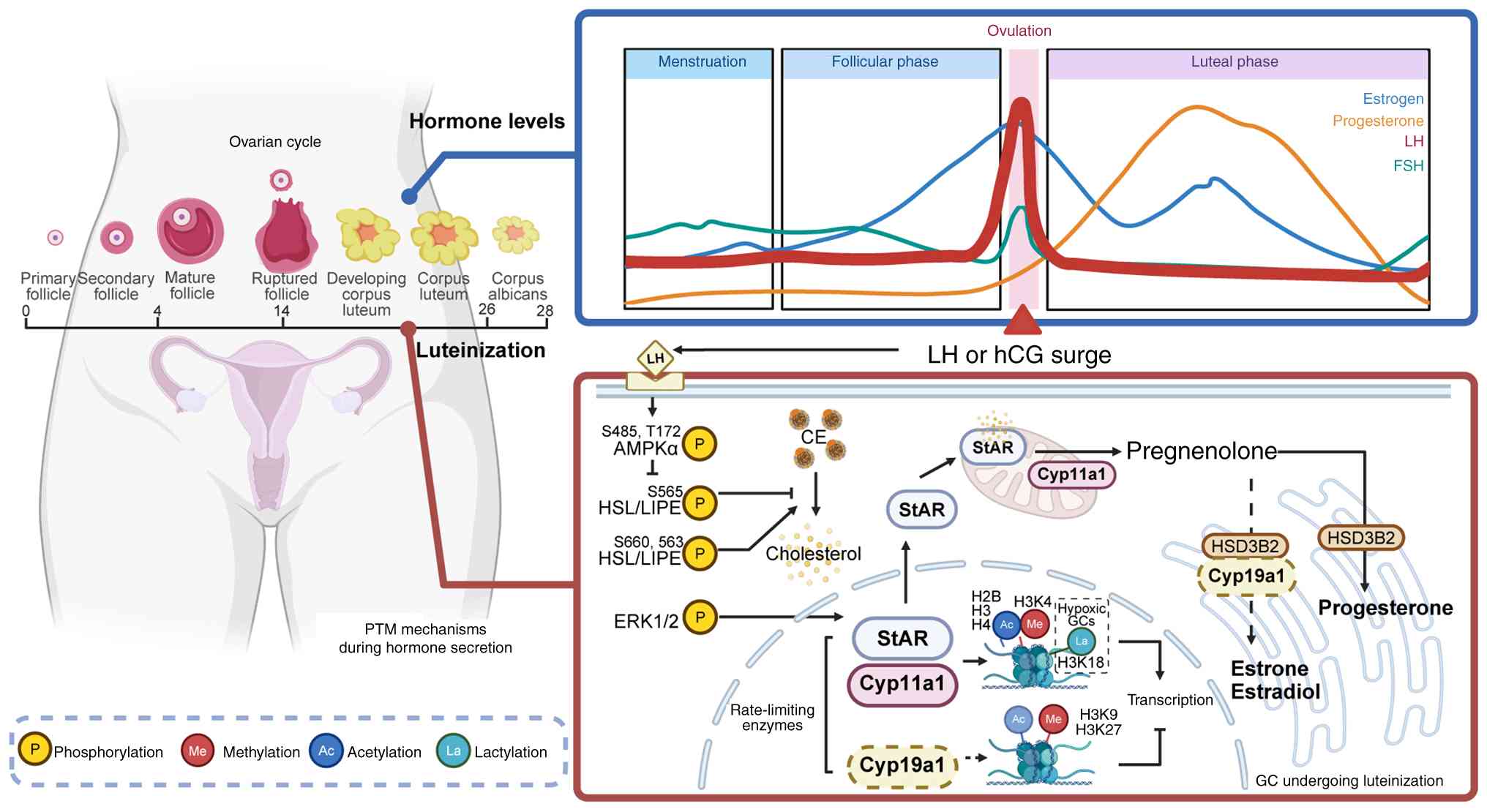
Progesterone (P4) carries out a key role in
embryonic development and uterine implantation. Large luteal cells,
which differentiate from GCs, synthesize P4 using cholesterol as
the substrate (71,79). Luteinizing hormone (LH) enhances
the efficiency of cholesterol mobilization through a dual
regulatory mechanism mediated by the protein kinase A signaling
pathway, which phosphorylates both AMP-activated protein kinase and
hormone-sensitive lipase to optimize P4 biosynthesis (71,72,80). The production of P4 induced by LH
remains unaffected by the mTOR inhibitor rapamycin, suggesting that
LH may regulate P4 synthesis via non-canonical molecular pathways;
thus, the precise mechanisms remain to be elucidated (72).
In summary, aberrant phosphorylation modifications
within key signaling pathways contribute to the dysfunction of GCs.
Some targeted inhibitors, such as the mTOR inhibitor rapamycin
(77) and the ERK pathway
antagonist ISRIB (44), have
proven effective in restoring GC function. Consequently, the
development of precision therapeutics that modulate GC
phosphorylation networks offers a promising novel strategy. Future
research should focus on the rational design of drugs that utilize
established phosphorylation regulatory networks, coupled with a
comprehensive mapping of phosphorylation dynamics across various
stages of follicular development (43).
Protein methylation is an important PTM that occurs
primarily on lysine and arginine residues and modulates histone and
non-histone functions (99,100). Methylation represents a key
determinant of the fate of GCs. During luteinization, the LH surge
induces chromatin remodeling through histone methylation, thus
orchestrating GC luteinization and hormone synthesis, including P4
and estrogen. Histone modifications such as trimethylation of
lysine 4 on histone H3 (H3K4me3) and lysine 36 on histone H3
generally facilitate transcriptional activation, while
trimethylation of lysine 9 on histone H3 (H3K9me3) and lysine 27 on
histone H3 (H3K27me3) are associated with transcriptional
repression (91,101). The enzymes StAR and CYP11A1 are
key in P4 synthesis. Following the LH surge, the ERK1/2 signaling
pathway is activated, increasing methylation at the H3K4me3 site
within the promoter regions of these genes, while concurrently
reducing methylation at the H3K9me3 and H3K27me3 sites (20,82,86). This coordinated alteration
enhances transcriptional activation and supports the expression of
steroidogenic genes. Concurrently, the sustained expression of
lysine-specific demethylase 1A (LSD1), an H3K4 demethylase, ensures
precise epigenetic regulation over the P4 biosynthesis pathway
(87). Moreover, modifications
of H3K9me3 and H3K27me3 carry out roles in regulating reproductive
physiology by maintaining normal estrous cycles and follicular
recruitment through the regulation of the inhibin α promoter
(15,88), and by promoting luteal
angiogenesis via vascular endothelial growth factor transcriptional
regulation (89). Beyond P4
regulation, histone methylation also controls estrogen secretion by
modulating the expression of CYP19A1, the rate-limiting enzyme in
estrogen synthesis, through modifications such as H3K4me3 and
H3K9me3 (83-85). Specifically, dysregulation of
H3K9me3 at the Cyp19a1 gene locus may disrupt endocrine
function and contribute to the pathogenesis of endometriosis
(84). Collectively, these
findings highlight that histone methylation acts as a bidirectional
epigenetic switch, dynamically balancing transcriptional activation
and repression to precisely regulate luteal function and
reproductive homeostasis.
Aberrant elevations of H3K36me1/2/3 and H3K27me3
have been demonstrated to markedly impair the proliferative
capacity of GCs and promote their apoptosis (90,91). Conversely, reduced levels of
H3K4me2/3 induce G2/M phase cell cycle arrest (94). Moreover, deficiencies in
KDM4B/5B/5C demethylases lead to the accumulation of DNA
double-strand breaks, resulting in S phase arrest and promoting
apoptosis in GCs (95-97). During ovarian aging, decreased
activity of methyltransferase is associated with the dysregulated
redistribution of H3K9me2/3 and H3K4me2/3, suggesting that aberrant
PTMs serve as one of the fundamental drivers of reproductive
senescence (92,93,102).
In addition to modifications involving lysine
methylation, arginine methylation also carries out a key role in
the developmental regulation of GCs. Protein arginine
methyltransferase 5 (PRMT5), the major type II enzyme, is
responsible for the symmetric dimethylation of arginine, which
facilitates internal ribosome entry site (IRES)-dependent
translation of WT1 mRNA by methylating HnRNPA1 (98,103). In PRMT5-deficient GCs, the
expression of steroidogenic genes was overactivated by WT1
downregulation. This dysregulation drives the premature
differentiation of GCs into a luteinized-like state. Such
precocious differentiation disrupts the essential physical and
nutritional support between GCs and the oocyte, ultimately leading
to follicular developmental arrest, structural disorganization,
atresia and female infertility (98,104). Clinically, the repressive
chromatin in mural GCs exhibits considerable enrichment of H3K27me3
methylation during folliculogenesis in patients with diminished
ovarian reserve, validating the pivotal role of histone methylation
in determining the fate and function of GCs (105).
Protein acetylation can be broadly categorized into
two types: Histone acetylation and non-histone acetylation. Both
types involve the transfer of acetyl groups to lysine residues via
an enzyme-catalyzed reaction (118). This reversible modification is
tightly regulated by lysine acetyltransferases, histone
deacetylases (HDACs) and the sirtuin family of deacetylases
(119).
During human Chorionic Gonadotropin (hCG)-induced
ovulation, H3K27ac rapidly undergoes deacetylation within 1 h,
followed by a re-establishment of acetylation at elevated levels
across specific genomic regions. This 'erase-and-rewrite' kinetic
pattern is essential for the activation of transcription in genes
associated with ovulation (106). Consequently, targeting this
dynamic process of erasure and re-establishment offers notable
therapeutic potential for the treatment of ovulatory disorders and
merits further exploration. In the context of steroidogenesis,
butyric acid promotes the acetylation of H3K9 by inhibiting HDACs,
while the activity of HDACs themselves synergistically augments
hormone production through dual stimulation of the PPARγ/PGC1α
pathway (107). Notably,
exposure to nicotine leads to the enrichment of HDAC3 at the
cyclooxygenase 1 (COX1) promoter region in GCs, resulting in
histone hypoacetylation that suppresses COX1 transcription and
consequently inhibits PGE2 biosynthesis. These nicotine-induced
epigenetic modifications trigger apoptosis and autophagy in GCs,
ultimately impairing follicular maturation (108). As depicted in Fig. 1, beyond methylation, the dynamic
alterations in histone acetylation are important in pre-ovulatory
GCs. Upon LH induction, the dynamic acetylation of H2BK5, H3K9 and
H4 facilitates chromatin remodeling, thereby regulating the
expression of CYP19A1 and inhibin α and coordinating key processes
such as estrogen synthesis, follicular development and ovulation
(15,20,85,109). Moreover, the transcription of
StAR, a rate-limiting enzyme in P4 synthesis, is dynamically
regulated through acetylation of H3 and H4 at its promoter during
this period (20). Thus, histone
acetylation carries out a pivotal role in gene expression by
mediating dynamic changes in chromatin structure, serving as a
fundamental regulator in physiological processes including
steroidogenesis, folliculogenesis and ovulation.
In addition to modifications in histone acetylation,
previous studies have unveiled a key role for non-histone protein
acetylation in various key cellular processes in GCs, such as
proliferation, apoptosis, hormonal signaling, DNA methylation, cell
cycle progression, autophagy and metabolism (110-112,116,120,121). Silencing information regulator
2 related enzyme 1 (SIRT1), an NAD+-dependent
deacetylase, deacetylates FOXO1 and regulates its activity,
especially under conditions of stress (112,122). This dysregulation facilitates
the nuclear translocation of FOXO1, leading to the activation of
pro-apoptotic genes, such as Puma, Bim, Trail
and Fas ligand, and thus inducing apoptosis in GCs (112,113,123). Conversely, the activation of
SIRT1-mediated deacetylation has been shown to rescue GCs from
oxidative stress through the JNK/FOXO1 signaling pathway (113,124). Another study has shown that
acetylation of FOXO1 in GCs is associated with ovarian competence.
A decrease in FOXO1 acetylation levels enhances apoptosis in GCs,
thereby increasing meiotic defects and aneuploidy in oocytes
(114). These findings
collectively highlight the dual role of FOXO1 acetylation in stress
adaptation, although further research is needed to elucidate the
mechanisms underlying this dual functionality.
In addition to FOXO1, the tumor suppressor protein
p53 also carries out a pivotal role in promoting GC apoptosis,
particularly under conditions of oxidative stress. SIRT1 inhibits
p53 activity through deacetylation at the K382 site, and its
downregulation leads to the accumulation of acetylated p53, thereby
exacerbating H2O2 or TNF-α induced apoptosis
in GCs (115,116). Furthermore, the inhibition of
T-LAK cell-originated protein kinase amplifies this pathway by
diminishing SIRT1 expression, resulting in sustained activation of
p53 acetylation and subsequent caspase-dependent apoptotic cascades
(117). Although strategies
targeting SIRT1 activation (for example, NAD+ boosters)
or modulation of p53 acetylation (for example, HDAC inhibitors)
have demonstrated potential in protecting ovarian function, a
comprehensive evaluation of their specificity and potential
off-target effects remains essential (117).
In summary, acetylation modifications targeting both
histone and non-histone proteins play integral roles in regulating
GC proliferation, hormonal synthesis and follicular development.
Dysregulation of acetylation is associated with infertility,
presenting promising therapeutic targets for related reproductive
disorders.
Ubiquitination is a broad PTM that falls into two
main types, called monoubiquitin and polyubiquitin, whose states
are regulated by ubiquitination and deubiquitination systems,
usually triggering degradation through proteasome and autophagy
pathways. The coordinated action of two predominant protein
degradation pathways is key for GC differentiation, steroidogenesis
and follicular development (135,136).
A study demonstrated that the E3 ligase SYVN1
suppresses mitochondrial fission and apoptosis, and delays
follicular atresia by targeting Drp 1 for degradation (125). Ma et al (126) discovered that Cry1
deficiency impairs NCOA4 degradation due to the downregulation of
the E3 enzyme HERC2, which triggers iron overload and senescence
via the ferritin-lysosome pathway. Additionally, USP14, the
deubiquitinating enzyme (DUB) impairs DNA repair mechanisms through
its deubiquitination activity, contributing to the pathogenesis of
POF (127). By contrast,
another DUB, UCHL1 promotes follicular development by stabilizing
voltage-dependent anion channel 2, which enhances cholesterol
transport and estradiol synthesis (128).
Moreover, ubiquitination is also involved in the
regulation of metabolic and stress response pathways. Liu et
al (129) found that
SKP2-mediated ubiquitination of the glycolytic enzyme
phosphoglycerate kinase 1 (PGK1) stabilizes the androgen receptor,
thereby associating aberrant glucose metabolism with ovulation
disorders in PCOS. Similarly, neuronal precursor cells expressed
developmentally down-regulated 4-like (NEDD4L) directly promotes
glutathione peroxidase 4 ubiquitination, inducing ferroptosis in
GCs, a process considerably exacerbated in PCOS and ultimately
leads to follicular dysfunction (130). Regarding autophagy regulation,
GCs resist apoptosis through two distinct mechanisms:
Deubiquitination-dependent stabilization of TGFβR2/SMAD4 signaling
or melatonin-induced proteasomal degradation of BimEl (133,134).
Previously, therapeutic strategies targeting
ubiquitinating enzymes have shown promising potential for improving
ovarian function. Zhang et al (132) revealed that primordial
follicular activation peptide 1 (PFAP1) stabilizes minichromosome
maintenance complex component 5 to promote primordial follicle
activation. Furthermore, lentiviral overexpression of the E3 ligase
Peli1 in regulatory T cells enhances GC survival and promotes
recovery of ovarian function, offering a novel approach for the
treatment of POF (137).
To summarize, E3 ligases and DUBs act as molecular
switches that finely regulate GC responses to hormonal and
environmental signals. The aforementioned studies demonstrate that
ubiquitination modification serves as a fundamental regulatory
mechanism essential for maintaining proteome stability and signal
transduction within GCs, offering novel strategies and therapeutic
avenues for ovarian diseases.
Beyond the four canonical PTMs previously discussed,
recent research has identified several novel PTMs that carry out
key roles as regulators in steroidogenesis, cell proliferation and
apoptosis (22,25,139,140). The dysregulation of these PTMs
has been associated with ovarian pathologies.
Lactylation, a protein modification induced by
lactate accumulation under conditions of hypoxic or metabolic
stress, carries out a pivotal role in the regulation of GCs
(141). Wu et al
(22) demonstrated that hypoxia
accelerated hCG-induced GC luteinization, which could be inhibited
by blocking lactate production or lactylation. Mechanistically, hCG
selectively increases H3K18la, thereby augmenting the transcription
of CYP11A1 and STAR, and consequently stimulating P4 production
during GC luteinization. Additionally, the non-histone protein CREB
at K136 has been identified as a potential lactylation site that
mediates hCG-induced luteinization, which may activate
proliferative signaling pathways and contribute to GC function and
survival (22,55).
Neddylation is a PTM in which the ubiquitin-like
protein neural precursor cell expressed developmentally
downregulated protein 8 is covalently conjugated to target proteins
via a dedicated enzymatic cascade. This process regulates
fundamental cellular functions, most notably by activating
Cullin-RING Ligases E3 (CRL) and thereby controlling protein
stability and signaling (145).
Chen et al (19) found
that MLN4924-mediated inhibition of Cullin protein neddylation
disrupts the activity of the CRL complex and downregulates the
expression of PPARα/γ. This results in the suppression of
anti-apoptotic genes while paradoxically activating proliferative
pathways in GCs. Notably, MLN4924 also inhibits the neddylation of
enzymes involved in lipid synthesis, leading to disrupted energy
metabolism. These dual effects suggest its potential utility as a
therapeutic target for ovarian protection.
Research has also clarified that O-GlcNAcylation and
lysine succinylation (Ksuc) are not merely apoptotic regulators but
pivotal modulators of GC physiology. O-GlcNAcylation occurs through
the attachment of an O-GlcNAc group to serine or threonine residues
on protein substrates, dynamically controlled by O-linked
β-N-acetylglucosamine transferase and O-GlcNAcase (146). Disruption in O-G lcNAc
modification homeostasis impairs energy metabolism in GCs,
affecting glycolysis, mitochondrial function and the tricarboxylic
acid cycle, ultimately leading to cellular dysfunction and
apoptosis (138). Concurrently,
succinylation occurs by the transfer of succinyl groups from
succinyl-CoA to amino acid residues of the protein to be modified
by succinyltransferases, with lysine being the most easily-modified
amino acid. Le et al (25) identified Ksuc as a potential
driver of ovarian aging, revealing that aberrant Ksuc accumulation
compromises ovarian reserve markers, (such as anti-Mullerian
hormone (AMH) and estrogen (E2), and promotes apoptosis through the
upregulation of the aging marker P21. Together, these emerging PTMs
represent promising biomarkers for assessing ovarian quality and
offer novel therapeutic targets for fertility preservation.
The regulation of GCs involves not only individual
PTMs but also crosstalk and synergistic interactions among various
modifications, which together form a dynamic regulatory network
within GCs.
Phosphorylation and acetylation demonstrate
synergistic effects in the modulation of AMH expression. The
histone acetyltransferase p300 is dually activated through
PI3K/AKT-mediated phosphorylation and direct interaction with
SMAD2/3. These cooperative interactions considerably enhance
H3K27ac, promoting AMH expression, which is important for
restricting the recruitment of primordial follicles and maintaining
the ovarian reserve. However, this activation can be counteracted
by the recruitment of HDAC2 induced by follicle-stimulating
hormone, creating a dynamic regulatory balance in GCs, thereby
strictly regulating the timing of follicle selection and preventing
premature ovarian exhaustion (147).
Fibroblast growth factor 9 (FGF9) functions as a key
regulator of follicular kinetics and GC proliferation (148). The efficacy of FGF9 signaling
is contingent upon the levels of histone H3K4me2 in GCs. An
enrichment of H3K4me2 enhances FGF9-mediated proliferation and
steroidogenesis in GCs, whereas its depletion results in the
downregulation of FGF9, which consequently triggers GC
differentiation and follicular selection. This epigenetic
regulation is mediated by LSD1, which undergoes considerable
enhancement following phosphorylation (149).
Cumulus cells, specialized GCs connected with
oocytes by transzonal projections, form a structure known as the
cumulus-oocyte complex (150).
A study showed that increased lysine crotonylation of ANXA2
enhances its binding to EGFR, thereby activating the EGFR pathway.
This activation triggers a subsequent phosphorylation cascade,
modulating the phosphorylation of AKT and ERK, which in turn
promotes cumulus cell proliferation and suppresses apoptosis,
ultimately supporting cumulus cell-dependent maturation and
influencing oocyte maturation (23).
Several studies have shown that palmitic acid (PA)
induces ERS and even causes apoptosis in GCs (151,152). Shibahara et al (153) discovered that PA induces
apoptosis in GCs through a mechanism involving triple PTMs.
Specifically, the inhibition of AKT Ser473 phosphorylation by PA
leads to the suppression of the PI3K/AKT pathway, which in turn
activates apoptotic effectors such as caspase-3. Additionally, PA
treatment results in the accumulation of ceramide in GCs, which
directly inhibits cell proliferation. In oocytes, PA suppresses the
AKT signaling pathway while concurrently upregulating the
activities of histone deacetylase and methyltransferase. These
alterations lead to hypoacetylation at H4K12 and hyperdimethylation
at H3K9, culminating in epigenetic dysregulation that compromises
oocyte quality.
PTMs carry out key roles in follicular function by
dynamically regulating protein activity, localization and
interaction networks (32).
Proteomic advances have enabled systematic profiling of PTM
alterations in GCs linked to reproductive diseases, offering
mechanistic insights into infertility (Fig. 2). These PTM patterns,
particularly phosphorylation, acetylation, and ubiquitination, hold
clinical potential as diagnostic biomarkers and therapeutic targets
(154-156).
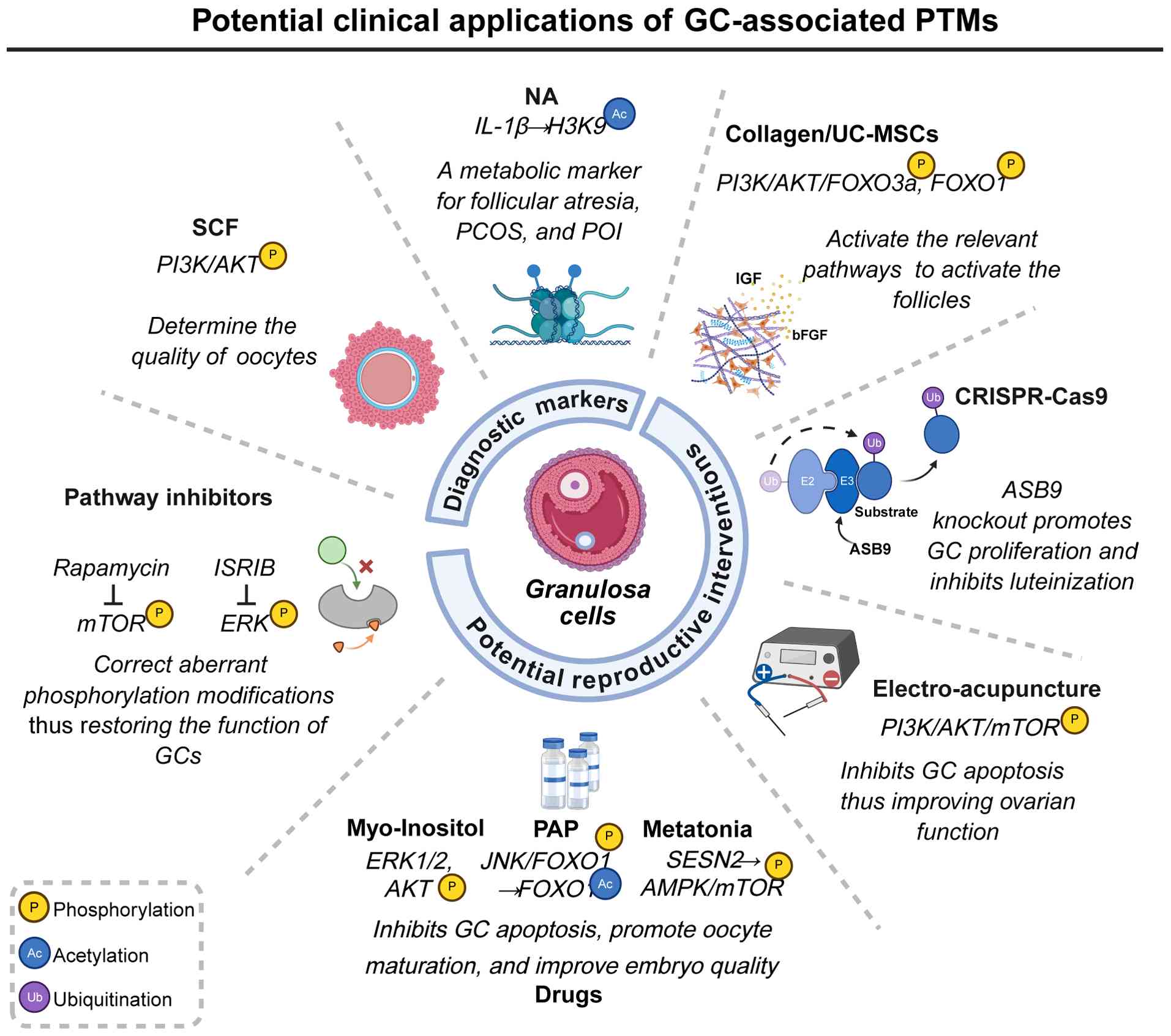
PTMs, which critically regulate the function of GCs,
demonstrate notable potential as precise diagnostic biomarkers for
female reproductive disorders (156-158).
In GCs, the stem cell factor (SCF) stimulates the
activation and maturation of primordial follicles and enhances
oocyte quality through modulation of the PI3K/AKT signaling
pathway. Additionally, SCF in follicular fluid regulates oxidative
stress and facilitates bidirectional communication between the
oocyte and GCs. Given its multifaceted roles, SCF is recognized not
only as a potential therapeutic target for conditions such as
ovarian aging, infertility due to poor ovarian response and
compromised oocyte quality but also as a non-invasive biomarker for
assessing oocyte maturity and predicting pregnancy outcomes,
particularly under antiretroviral therapy (155,159).
Nervonic acid (NA), primarily involved in
sphingolipid metabolism and cell membrane structure formation
(160), exhibits dual effects
on GCs. Aberrant accumulation of NA markedly alters H3K9
acetylation through two mechanisms. Firstly, NA upregulates the
deacetylase SIRT6, markedly reducing H3K9ac levels and
transcriptionally repressing key steroidogenic genes, thereby
disrupting estradiol synthesis and impairing luteal function.
Secondly, NA enhances the recruitment of the transcription factor
activator protein-1 (AP-1) to the IL-1β promoter region,
specifically increasing H3K9ac levels at this site, which promotes
the overexpression of IL-1β and exacerbates the ovarian
inflammatory environment. Consequently, serum levels of NA may
serve as valuable metabolic biomarkers for reproductive disorders
such as PCOS, POI and follicular atresia (161).
Precise modulation of targeted PTMs presents an
innovative strategy for enhancing the follicular
microenvironment.
A three-dimensional biomaterial, collagen/umbilical
cord mesenchymal stem cells (UC-MSCs), has been developed. This
biomaterial embeds UC-MSCs within a collagen scaffold, replicating
the physicochemical properties of the native extracellular matrix.
This configuration markedly enhances UC-MSC adhesion, proliferation
and paracrine secretion, thereby improving the targeting efficiency
and sustainability of stem cell-based therapies. Through paracrine
signaling, collagen/UC-MSCs secrete insulin like growth factor and
basic fibroblast growth factor, which activate the PI3K-AKT pathway
in GCs. This activation leads to the phosphorylation of FOXO3a and
FOXO1, facilitating their nuclear export and mitigating their
inhibitory impact on primordial follicle activation. This mechanism
rejuvenates the ovarian niche and provides a novel therapeutic
opportunity for patients with POF (164).
CRISPR-Cas9 technology has emerged as a potent tool
for epigenetic editing. ASB9, identified as a specific substrate
recognition component of the E3 ligase, is differentially expressed
in the GCs of ovulatory follicles. Previous studies employing
CRISPR-Cas9-mediated ASB9 knockout have demonstrated an increase in
GC number, providing robust evidence for the role of ASB9 as a
regulator of GC function that limits GC proliferation and
contributes to GC luteinization (154,165).
In conclusion, PTM-targeted interventions in GCs
meld molecular mechanisms with clinical innovation, defining the
forefront of reproductive medicine. Future research should focus on
rigorous translational and clinical validation to ensure the safety
and efficacy of these precision strategies for treating
infertility.
Infertility is increasingly recognized as a
considerable global challenge affecting female reproductive health
(166). As a fundamental
regulatory mechanism of protein function, PTMs meticulously
orchestrate protein networks within GCs and carry out a key role in
reproductive pathologies. Emerging evidence suggests that PTM
dysregulation in GCs is a key determinant in the pathogenesis of
female infertility, offering novel mechanistic insights and
therapeutic targets for clinical intervention. Notably, with the
rise in global environmental pollution, exposure to toxins such as
heavy metals, radiation and hazardous chemicals may disrupt
physiological PTMs in GCs, leading to autophagy or apoptosis of
these cells, ultimately exacerbating infertility (43,167,168). However, the molecular
mechanisms underlying the interactions between environmental
factors and reproductive health remain elusive, representing a
notable area of scientific and clinical interest for the prevention
and treatment of infertility amid increasing environmental
pollution.
As the primary treatment modality for infertility,
the long-term safety of ART necessitates further evaluation.
Current evidence suggests that offspring conceived via ART may be
at increased risk for rare imprinting disorders, potentially
associated with aberrant epigenetic reprogramming in gametes or
embryos (169). With the
widespread adoption of ART, understanding its intergenerational
health impacts is imperative. Notably, dynamic PTMs in GCs during
ART have been shown to markedly influence the success of clinical
pregnancies (30). However, few
studies have explored whether in vitro manipulations disrupt
embryonic epigenetic programming via PTM dysregulation in GCs.
Thus, elucidating the molecular causality in the 'ART/PTM
remodeling/embryonic development' pathway is crucial for optimizing
ART procedures and ensuring intergenerational health.
In conclusion, the present review systematically
assessed the dynamic regulatory networks of both canonical and
novel PTMs in the proliferation, differentiation, apoptosis and
hormone synthesis of GCs. It elucidates the pathological mechanisms
of aberrant PTMs in female infertility, and comprehensively
assesses the translational medical value of targeted PTM therapy
for GCs. By integrating epigenetic regulation with clinical
applications, this work aims to provide novel insights into
precision diagnosis and treatment strategies for female
infertility.
Not applicable.
YZh designed the concept, performed the analysis,
designed the figures and contributed to the writing of the original
draft. YZo contributed to the writing of the original draft and
translated the manuscript. ZY critically revised the manuscript.
JW, ZP and JF supervised the work, provided expert knowledge and
critically revised the manuscript. All authors critically revised
the manuscript and all authors read and approved the final
manuscript. Data authentication is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
During the preparation of this work, AI tools were
used to improve the readability and language of the manuscript, and
subsequently, the authors revised and edited the content produced
by the AI tools as necessary, taking full responsibility for the
ultimate content of the present manuscript.
Not applicable.
This work was supported by the National Natural Science
Foundation of China (grant no. 81960236).
|
1
|
Shan B, Huo Y, Guo Z, Li Q, Pan Z, Li Q
and Du X: miR-184, a downregulated ovary-elevated miRNA
transcriptionally activated by SREBF2, exerts anti-apoptotic
properties in ovarian granulosa cells through inducing SMAD3
expression. Cell Death Dis. 15:8922024. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wu H, Nguyen H, Hashim PH, Fogelgren B,
Duncan FE and Ward WS: Oocyte-specific EXOC5 expression is required
for mouse oogenesis and folliculogenesis. Mol Hum Reprod.
30:gaae0262024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang CH, Liu XY and Wang J: Essential
role of granulosa cell glucose and lipid metabolism on oocytes and
the potential metabolic imbalance in polycystic ovary syndrome. Int
J Mol Sci. 24:162472023. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rooda I, Hassan J, Hao J, Wagner M,
Moussaud-Lamodiere E, Jääger K, Otala M, Knuus K, Lindskog C,
Papaikonomou K, et al: In-depth analysis of transcriptomes in
ovarian cortical follicles from children and adults reveals
interfollicular heterogeneity. Nat Commun. 15:69892024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Almeida CP, Ferreira MCF, Silveira CO,
Campos JR, Borges IT, Baeta PG, Silva FHS, Reis FM and Del Puerto
HL: Clinical correlation of apoptosis in human granulosa cells-a
review. Cell Biol Int. 42:1276–1281. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Vander Borght M and Wyns C: Fertility and
infertility: Definition and epidemiology. Clin Biochem. 62:2–10.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Habara O, Logan CY, Kanai-Azuma M, Nusse R
and Takase HM: WNT signaling in pre-granulosa cells is required for
ovarian folliculogenesis and female fertility. Development.
148:dev1988462021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sreerangaraja Urs DB, Wu WH, Komrskova K,
Postlerova P, Lin YF, Tzeng CR and Kao SH: Mitochondrial function
in modulating human granulosa cell steroidogenesis and female
fertility. Int J Mol Sci. 21:35922020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lin X, Dai Y, Tong X, Xu W, Huang Q, Jin
X, Li C, Zhou F, Zhou H, Lin X, et al: Excessive oxidative stress
in cumulus granulosa cells induced cell senescence contributes to
endometriosis-associated infertility. Redox Biol. 30:1014312020.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu Y, Liu H, Li Z, Fan H, Yan X, Liu X,
Xuan J, Feng D and Wei X: The release of peripheral immune
inflammatory cytokines promote an inflammatory cascade in PCOS
patients via altering the follicular microenvironment. Front
Immunol. 12:6857242021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kunitomi C, Harada M, Takahashi N, Azhary
JMK, Kusamoto A, Nose E, Oi N, Takeuchi A, Wada-Hiraike O, Hirata
T, et al: Activation of endoplasmic reticulum stress mediates
oxidative stress-induced apoptosis of granulosa cells in ovaries
affected by endometrioma. Mol Hum Reprod. 26:40–52. 2020.
View Article : Google Scholar
|
|
12
|
Ford EA, Frost ER, Beckett EL, Roman SD,
McLaughlin EA and Sutherland JM: Transcriptomic profiling of
neonatal mouse granulosa cells reveals new insights into primordial
follicle activation†. Biol Reprod. 106:503–514. 2022. View Article : Google Scholar :
|
|
13
|
Xing J, Qiao G, Luo X, Liu S, Chen S, Ye
G, Zhang C and Yi J: Ferredoxin 1 regulates granulosa cell
apoptosis and autophagy in polycystic ovary syndrome. Clin Sci
(Lond). 137:453–468. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Turathum B, Gao EM and Chian RC: The
function of cumulus cells in oocyte growth and maturation and in
subsequent ovulation and fertilization. Cells. 10:22922021.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Meldi KM, Gaconnet GA and Mayo KE: DNA
methylation and histone modifications are associated with
repression of the inhibin α promoter in the rat corpus luteum.
Endocrinology. 153:4905–4917. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pieroni L, Iavarone F, Olianas A, Greco V,
Desiderio C, Martelli C, Manconi B, Sanna MT, Messana I, Castagnola
M and Cabras T: Enrichments of post-translational modifications in
proteomic studies. J Sep Sci. 43:313–336. 2020. View Article : Google Scholar
|
|
17
|
Wang S, Osgood AO and Chatterjee A:
Uncovering posttranslational modification-associated
protein-protein interactions. Curr Opin Struct Biol. 74:1023522022.
View Article : Google Scholar
|
|
18
|
Walsh CT, Garneau-Tsodikova S and Gatto GJ
Jr: Protein posttranslational modifications: The chemistry of
proteome diversifications. Angew Chem Int Ed Engl. 44:7342–7372.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen M, Liu Y, Zuo M, Zhang M, Wang Z, Li
X, Yuan D, Xu H, Yu G and Li M: Integrated analysis reveals the
regulatory mechanism of the neddylation inhibitor MLN4924 on the
metabolic dysregulation in rabbit granulosa cells. BMC Genomics.
25:2542024. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lee L, Asada H, Kizuka F, Tamura I,
Maekawa R, Taketani T, Sato S, Yamagata Y, Tamura H and Sugino N:
Changes in histone modification and DNA methylation of the StAR and
Cyp19a1 promoter regions in granulosa cells undergoing
luteinization during ovulation in rats. Endocrinology. 154:458–470.
2013. View Article : Google Scholar
|
|
21
|
Bai X and Wang S: Signaling pathway
intervention in premature ovarian failure. Front Med (Lausanne).
9:9994402022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wu G, Pan Y, Chen M, Liu Z, Li C, Sheng Y,
Li H, Shen M and Liu H: Lactylation drives hCG-triggered
luteinization in hypoxic granulosa cells. Int J Biol Macromol.
280:1355802024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhou C, Zeng H, Xiao X, Wang L, Jia L, Shi
Y, Zhang M, Fang C, Zeng Y, Wu T, et al: Global crotonylome
identifies EP300-regulated ANXA2 crotonylation in cumulus cells as
a regulator of oocyte maturation. Int J Biol Macromol.
259:1291492024. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Qin X, Dang W, Yang X, Wang K, Kebreab E
and Lyu L: Neddylation inactivation affects cell cycle and
apoptosis in sheep follicular granulosa cells. J Cell Physiol.
237:3278–3291. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Le M, Li J, Zhang D, Yuan Y, Zhou C, He J,
Huang J, Hu L, Luo T and Zheng L: The emerging role of lysine
succinylation in ovarian aging. Reprod Biol Endocrinol. 21:382023.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sangalli JR, Nociti RP, Del Collado M,
Sampaio RV, da Silveira JC, Perecin F, Smith LC, Ross PJ and
Meirelles FV: Characterization of histone lysine
β-hydroxybutyrylation in bovine tissues, cells, and cumulus-oocyte
complexes. Mol Reprod Dev. 89:375–398. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Goud PT, Goud AP, Joshi N, Puscheck E,
Diamond MP and Abu-Soud HM: Dynamics of nitric oxide, altered
follicular microenvironment, and oocyte quality in women with
endometriosis. Fertil Steril. 102:151–159.e5. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Faramarzi A, Khalili MA and Jahromi MG: Is
there any correlation between apoptotic genes expression in cumulus
cells with embryo morphokinetics? Mol Biol Rep. 46:3663–3670. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zheng M, Liu M and Zhang C: Melatonin
ameliorates ovarian hyperstimulation syndrome (OHSS) through SESN2
regulated antiapoptosis. Obstet Gynecol Int. 2023:11212272023.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tabatabaie M, Amiri S, Golestan Jahromi M,
Sene AA, Zandieh Z, Mehdizadeh M and Amjadi F: The effect of
Myo-Inositol supplement on molecular regulation of
folliculogenesis, steroidogenesis, and assisted reproductive
technique outcomes in patients with polycystic ovarian syndrome.
Mol Biol Rep. 49:875–884. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang H, Qin F, Liu A, Sun Q, Wang Q, Xie
S, Lu S, Zhang D and Lu Z: Electro-acupuncture attenuates the mice
premature ovarian failure via mediating PI3K/AKT/mTOR pathway. Life
Sci. 217:169–175. 2019. View Article : Google Scholar
|
|
32
|
Chen D, Feng Y, Wu J, Zhou J, Li Z, Qiao
M, Chen T, Xu Z, Peng X and Mei S: Post-translational modifications
in mammalian folliculogenesis and ovarian pathologies. Cells.
14:12922025. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wu Y, Li M and Yang M: Post-translational
modifications in oocyte maturation and embryo development. Front
Cell Dev Biol. 9:6453182021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang L, Zhang Y and Sun H: Protein
modifications during early embryo development. Am J Reprod Immunol.
92:e700072024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang J, Sun X, Yang Z, Li S, Wang Y, Ren
R, Liu Z and Yu D: Epigenetic regulation in premature ovarian
failure: A literature review. Front Physiol. 13:9984242023.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gao Q, Ren J, Niu D, Guo L and Feng X:
Role of protein post-translational modifications in unexplained
recurrent pregnancy loss. Zhong Nan Da Xue Xue Bao Yi Xue Ban.
49:1495–1502. 2024.In English, Chinese. PubMed/NCBI
|
|
37
|
Lai H, Yang Y and Zhang J: Advances in
post-translational modifications and recurrent spontaneous
abortion. Gene. 927:1487002024. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Senthilkumar H, Chauhan SC and Arumugam M:
Unraveling the multifactorial pathophysiology of polycystic ovary
syndrome: exploring lifestyle, prenatal influences, neuroendocrine
dysfunction, and post-translational modifications. Mol Biol Rep.
52:9802025. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wei H, Huo P, Liu S, Huang H and Zhang S:
Posttranslational modifications in pathogenesis of PCOS. Front
Endocrinol (Lausanne). 13:10243202022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Siva AB, Srivastava P and Shivaji S:
Understanding the pathogenesis of endometriosis through proteomics:
Recent advances and future prospects. Proteomics Clin Appl.
8:86–98. 2014. View Article : Google Scholar
|
|
41
|
Huang JC, Duan CC, Jin S, Sheng CB, Wang
YS, Yue ZP and Guo B: HB-EGF induces mitochondrial dysfunction via
estrogen hypersecretion in granulosa cells dependent on
cAMP-PKA-JNK/ERK-Ca2+-FOXO1 pathway. Int J Biol Sci.
18:2047–2059. 2022. View Article : Google Scholar
|
|
42
|
Maucieri AM and Townson DH: Evidence and
manipulation of O-GlcNAcylation in granulosa cells of bovine antral
follicles†. Biol Reprod. 104:914–923. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Xue Y, Cheng X, Ma ZQ, Wang HP, Zhou C, Li
J, Zhang DL, Hu LL, Cui YF, Huang J, et al: Polystyrene
nanoplastics induce apoptosis, autophagy, and steroidogenesis
disruption in granulosa cells to reduce oocyte quality and
fertility by inhibiting the PI3K/AKT pathway in female mice. J
Nanobiotechnology. 22:4602024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Yang W, Yu S, Peng J, Chang P and Chen X:
FGF12 regulates cell cycle gene expression and promotes follicular
granulosa cell proliferation through ERK phosphorylation in geese.
Poult Sci. 102:1029372023. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhao S, Gu T, Weng K, Zhang Y, Cao Z,
Zhang Y, Zhao W, Chen G and Xu Q: Phosphoproteome reveals
extracellular regulated protein kinase phosphorylation mediated by
mitogen-activated protein kinase kinase-regulating granulosa cell
apoptosis in broody geese. Int J Mol Sci. 24:122782023. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Liu C, Pan B, Yang L, Wang B and Li J:
Beta defensin 3 enhances ovarian granulosa cell proliferation and
migration via ERK1/2 pathway in vitro†. Biol Reprod. 100:1057–1065.
2019. View Article : Google Scholar
|
|
47
|
Pan B, Zhan X and Li J: MicroRNA-574
impacts granulosa cell estradiol production via targeting TIMP3 and
ERK1/2 signaling pathway. Front Endocrinol (Lausanne).
13:8521272022. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Cottom J, Salvador LM, Maizels ET,
Reierstad S, Park Y, Carr DW, Davare MA, Hell JW, Palmer SS, Dent
P, et al: Follicle-stimulating hormone activates extracellular
signalregulated kinase but not extracellular signal-regulated
kinase kinase through a 100-kDa phosphotyrosine phosphatase. J Biol
Chem. 278:7167–7179. 2003. View Article : Google Scholar
|
|
49
|
Yan MQ, Zhu BH, Liu XH, Yang YM, Duan XY,
Wang Y, Sun H, Feng M, Li T and Liu XM: Mitoguardin 1 and 2 promote
granulosa cell proliferation by activating AKT and regulating the
Hippo-YAP1 signaling pathway. Cell Death Dis. 14:7792023.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Song A, Zhang S, Zhao X, Wu S, Qi X, Gao
S, Qi J, Li P and Tan J: Exosomes derived from menstrual blood
stromal cells ameliorated premature ovarian insufficiency and
granulosa cell apoptosis by regulating SMAD3/AKT/MDM2/P53 pathway
via delivery of thrombospondin-1. Biomed Pharmacother.
166:1153192023. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Alam H, Maizels ET, Park Y, Ghaey S,
Feiger ZJ, Chandel NS and Hunzicker-Dunn M: Follicle-stimulating
hormone activation of hypoxia-inducible factor-1 by the
phosphatidylinositol 3-kinase/AKT/Ras homolog enriched in brain
(Rheb)/mammalian target of rapamycin (mTOR) pathway is necessary
for induction of select protein markers of follicular
differentiation. J Biol Chem. 279:19431–19440. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hu J, Jin J, Qu Y, Liu W, Ma Z, Zhang J
and Chen F: ERO1α inhibits cell apoptosis and regulates
steroidogenesis in mouse granulosa cells. Mol Cell Endocrinol.
511:1108422020. View Article : Google Scholar
|
|
53
|
Zhang W, Gao L, Zhang X, Weng Y, Du Y, Sun
YL, Wei H, Hao T, Chen Y, Liang X and Zhang M: Theophylline
derivatives promote primordial follicle activation via
cAMP-PI3K/Akt pathway and ameliorate fertility deficits in
naturally aged mice. Int J Biol Sci. 20:5312–5329. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Tong C, Wu Y, Zhang L and Yu Y: Insulin
resistance, autophagy and apoptosis in patients with polycystic
ovary syndrome: Association with PI3K signaling pathway. Front
Endocrinol (Lausanne). 13:10911472022. View Article : Google Scholar :
|
|
55
|
Wu G, Chen M, He T, Pan Y, Li C, Liu Z, Li
H, Sheng Y, Dai W, Shen M and Liu H: Lactylation of CREB is
required for FSH-induced proliferation and differentiation of
ovarian granulosa cells. Nucleic Acids Res. 53:gkaf8822025.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Lanfranchi B, Rubia RF, Gassmann M,
Schuler G and Kowalewski MP: Transcriptional regulation of
HIF1α-mediated STAR expression in murine KK1 granulosa cell line
involves cJUN, CREB and CBP-dependent pathways. Gen Comp
Endocrinol. 315:1139232022. View Article : Google Scholar
|
|
57
|
Zhang X, Zhang W, Wang Z, Zheng N, Yuan F,
Li B, Li X, Deng L, Lin M, Chen X and Zhang M: Enhanced glycolysis
in granulosa cells promotes the activation of primordial follicles
through mTOR signaling. Cell Death Dis. 13:872022. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Wu G, Song D, Wu H, Zhao F, Ding W, Wang
Z, Shi F and Wei Q: Genistein ameliorates starvation-induced
porcine follicular granulosa cell apoptosis. Reproduction.
166:451–458. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Lin M, Hua R, Ma J, Zhou Y, Li P, Xu X, Yu
Z and Quan S: Bisphenol A promotes autophagy in ovarian granulosa
cells by inducing AMPK/mTOR/ULK1 signalling pathway. Environ Int.
147:1062982021. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Liu G, Wang Y, Zheng Y, Lv J, Li Y, Liu N,
Gao H, Ran H, Tang H and Jiang Z: PHB2 binds to ERβ to induce the
autophagy of porcine ovarian granulosa cells through mTOR
phosphorylation. Theriogenology. 198:114–122. 2023. View Article : Google Scholar
|
|
61
|
Liu J and Wang C: Lysophosphatidic acid is
associated with oocyte maturation by enhancing autophagy via
PI3K-AKT-mTOR signaling pathway in granulosa cells. J Ovarian Res.
16:1372023. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Tang X, Ma L, Guo S, Liang M and Jiang Z:
High doses of FSH induce autophagy in bovine ovarian granulosa
cells via the AKT/mTOR pathway. Reprod Domest Anim. 56:324–332.
2021. View Article : Google Scholar
|
|
63
|
Hu H, Zhang J, Xin X, Jin Y, Zhu Y, Zhang
H, Fan R, Ye Y, Jiang Y and Li D: Bushen jianpi tiaoxue decoction
(BJTD) inhibits the LIF-mTOR signaling axis to regulate
mitochondrial function and alleviate cyclophosphamide-induced
diminished ovarian reserve. Apoptosis. 30:1331–1350. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Ji R, Zhang Z, Yang Z, Chen X, Yin T and
Yang J: BOP1 contributes to the activation of autophagy in
polycystic ovary syndrome via nucleolar stress response. Cell Mol
Life Sci. 81:1012024. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Li C, Wu G, Ning C, Liu Z, Tao J, Lu X,
Shen M and Liu H: FOXO1-mediated nuclear sequestration of STAT3 and
AKT1 triggers FOXO3-dependent autophagic death in hypoxic granulosa
cells. Int J Biol Sci. 20:5939–5958. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Sun Z, Li P, Wang X, Lai S, Qiu H, Chen Z,
Hu S, Yao J and Shen J: GLP-1/GLP-1R signaling regulates ovarian
PCOS-associated granulosa cells proliferation and antiapoptosis by
modification of forkhead box protein O1 phosphorylation sites. Int
J Endocrinol. 2020:14843212020. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Lei Z, Ali I, Yang M, Yang C, Li Y and Li
L: Non-esterified fatty acid-induced apoptosis in bovine granulosa
cells via ROS-activated PI3K/AKT/FoxO1 pathway. Antioxidants
(Basel). 12:4342023. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Yan C, Ou Y, Sun X, Sun Y, Zhao J, Qin N
and Xu R: FSH-induced nuclear exclusion of FOXO1 mediated by
PI3K/Akt signaling pathway in granulosa cells is associated with
follicle selection and growth of the Hen Ovary. Cells. 14:18642025.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Yang F, Chen Y, Liu Q, Dai S and Zeng S:
Dynamics and regulations of BimEL Ser65 and Thr112 phosphorylation
in porcine granulosa cells during follicular atresia. Cells.
9:4022020. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Ling XM, Zhang XH, Tan Y, Yang JJ, Ji B,
Wu XR, Yi YK and Liang L: Protective effects of Oviductus
Ranae-containing serum on oxidative stress-induced apoptosis in rat
ovarian granulosa cells. J Ethnopharmacol. 208:138–148. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Przygrodzka E, Hou X, Zhang P, Plewes MR,
Franco R and Davis JS: PKA and AMPK signaling pathways
differentially regulate luteal steroidogenesis. Endocrinology.
162:bqab0152021. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Hou X, Arvisais EW and Davis JS:
Luteinizing hormone stimulates mammalian target of rapamycin
signaling in bovine luteal cells via pathways independent of AKT
and mitogen-activated protein kinase: Modulation of glycogen
synthase kinase 3 and AMP-activated protein kinase. Endocrinology.
151:2846–2857. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Tsui KH and Li CJ: Mitoquinone shifts
energy metabolism to reduce ROS-induced oxeiptosis in female
granulosa cells and mouse oocytes. Aging (Albany NY). 15:246–260.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zareifard A, Beaudry F and Ndiaye K: Janus
Kinase 3 phosphorylation and the JAK/STAT pathway are positively
modulated by follicle-stimulating hormone (FSH) in bovine granulosa
cells. BMC Mol Cell Biol. 24:212023. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Ardito F, Giuliani M, Perrone D, Troiano G
and Lo Muzio L: The crucial role of protein phosphorylation in cell
signaling and its use as targeted therapy (review). Int J Mol Med.
40:271–280. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Gershon E and Dekel N: Newly identified
regulators of ovarian folliculogenesis and ovulation. Int J Mol
Sci. 21:45652020. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Wang C, Lv X, Jiang C, Cordes CM, Fu L,
Lele SM and Davis JS: Transforming growth factor alpha (TGFα)
regulates granulosa cell tumor (GCT) cell proliferation and
migration through activation of multiple pathways. PLoS One.
7:e482992012. View Article : Google Scholar
|
|
78
|
Li H, Jing Y, Qu X, Yang J, Pan P, Liu X,
Gao H, Pei X, Zhang C and Yang Y: The activation of reticulophagy
by ER stress through the ATF4-MAP1LC3A-CCPG1 pathway in ovarian
granulosa cells is linked to apoptosis and necroptosis. Int J Mol
Sci. 24:27492023. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Lonergan P: Influence of progesterone on
oocyte quality and embryo development in cows. Theriogenology.
76:1594–1601. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Anthonsen MW, Rönnstrand L, Wernstedt C,
Degerman E and Holm C: Identification of novel phosphorylation
sites in hormone-sensitive lipase that are phosphorylated in
response to isoproterenol and govern activation properties in
vitro. J Biol Chem. 273:215–221. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Sha QQ, Jiang Y, Yu C, Xiang Y, Dai XX,
Jiang JC, Ou XH and Fan HY: CFP1-dependent histone H3K4
trimethylation in murine oocytes facilitates ovarian follicle
recruitment and ovulation in a cell-nonautonomous manner. Cell Mol
Life Sci. 77:2997–3012. 2020. View Article : Google Scholar
|
|
82
|
Maekawa R, Lee L, Okada M, Asada H,
Shinagawa M, Tamura I, Sato S, Tamura H and Sugino N: Changes in
gene expression of histone modification enzymes in rat granulosa
cells undergoing luteinization during ovulation. J Ovarian Res.
9:152016. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Sugino N: Molecular mechanisms of
luteinization. Obstet Gynecol Sci. 57:93–101. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Hosseini E, Mehraein F, Shahhoseini M,
Karimian L, Nikmard F, Ashrafi M, Afsharian P and Aflatoonian R:
Epigenetic alterations of CYP19A1 gene in cumulus cells and its
relevance to infertility in endometriosis. J Assist Reprod Genet.
33:1105–1113. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Hosseini E, Shahhoseini M, Afsharian P,
Karimian L, Ashrafi M, Mehraein F and Afatoonian R: Role of
epigenetic modifications in the aberrant CYP19A1 gene expression in
polycystic ovary syndrome. Arch Med Sci. 15:887–895. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Okada M, Lee L, Maekawa R, Sato S,
Kajimura T, Shinagawa M, Tamura I, Taketani T, Asada H, Tamura H
and Sugino N: Epigenetic changes of the Cyp11a1 promoter region in
granulosa cells undergoing luteinization during ovulation in female
rats. Endocrinology. 157:3344–3354. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Seneda MM, Godmann M, Murphy BD, Kimmins S
and Bordignon V: Developmental regulation of histone H3 methylation
at lysine 4 in the porcine ovary. Reproduction. 135:829–838. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Meunier H, Cajander SB, Roberts VJ, Rivier
C, Sawchenko PE, Hsueh AJ and Vale W: Rapid changes in the
expression of inhibin alpha-, beta A-, and beta B-subunits in
ovarian cell types during the rat estrous cycle. Mol Endocrinol.
2:1352–1363. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Shinagawa M, Tamura I, Maekawa R, Sato S,
Shirafuta Y, Mihara Y, Okada-Matsumoto M, Taketani T, Asada H,
Tamura H and Sugino N: C/EBPβ regulates Vegf gene expression in
granulosa cells undergoing luteinization during ovulation in female
rats. Sci Rep. 9:7142019. View Article : Google Scholar
|
|
90
|
Cui LX, Tian YQ, Hao HS, Zou HY, Pang YW,
Zhao SJ, Zhao XM, Zhu HB and Du WH: Knockdown of ASH1L
methyltransferase induced apoptosis inhibiting proliferation and
H3K36 methylation in bovine cumulus cells. Theriogenology.
161:65–73. 2021. View Article : Google Scholar
|
|
91
|
Zhong Y, Li L, He Y, He B, Li Z, Zhang Z,
Zhang H, Yuan X and Li J: Activation of steroidogenesis,
anti-apoptotic activity, and proliferation in porcine granulosa
cells by RUNX1 is negatively regulated by H3K27me3 transcriptional
repression. Genes (Basel). 11:4952020. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Chen W, Dong L, Wei C and Wu H: Role of
epigenetic regulation in diminished ovarian reserve. J Assist
Reprod Genet. 42:389–403. 2025. View Article : Google Scholar :
|
|
93
|
Eslami H, Eslami A, Favaedi R, Asadpour U,
Zari Moradi S, Eftekhari-Yazdi P, Madani T, Shahhoseini M and
Mohseni Meybodi A: Epigenetic aberration of FMR1 gene in infertile
women with diminished ovarian reserve. Cell J. 20:78–83.
2018.PubMed/NCBI
|
|
94
|
Wang D, Lu X, Jiang Y, Pan L, Zhu F, Yu A,
Zhao M, Yang M, Bi J, He X, et al: The chromatin remodeling protein
BPTF mediates cell cycle, proliferation and apoptosis in porcine
ovarian granulosa cells. Theriogenology. 211:172–181. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Yang Y, Cai Y, Guo J, Dai K, Liu L, Chen
Z, Wang F and Deng M: Knockdown of KDM5B leads to DNA damage and
cell cycle arrest in granulosa cells via MTF1. Curr Issues Mol
Biol. 45:3219–3237. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Glanzner WG, Gutierrez K, Rissi VB, de
Macedo MP, Lopez R, Currin L, Dicks N, Baldassarre H, Agellon LB
and Bordignon V: Histone lysine demethylases KDM5B and KDM5C
modulate genome activation and stability in porcine embryos. Front
Cell Dev Biol. 8:1512020. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Xiong X, Ma H, Min X, Su F, Xiong Y and Li
J: Effects of demethylase KDM4B on the biological characteristics
and function of yak cumulus cells in vitro. Theriogenology.
174:85–93. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Chen M, Dong F, Chen M, Shen Z, Wu H, Cen
C, Cui X, Bao S and Gao F: PRMT5 regulates ovarian follicle
development by facilitating Wt1 translation. Elife. 10:e689302021.
View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Bhat KP, Ümit Kaniskan H, Jin J and Gozani
O: Epigenetics and beyond: Targeting writers of protein lysine
methylation to treat disease. Nat Rev Drug Discov. 20:265–286.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Jambhekar A, Dhall A and Shi Y: Roles and
regulation of histone methylation in animal development. Nat Rev
Mol Cell Biol. 20:625–641. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Gong F and Miller KM: Histone methylation
and the DNA damage response. Mutat Res Rev Mutat Res. 780:37–47.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Bilmez Y, Talibova G and Ozturk S:
Expression of the histone lysine methyltransferases SETD1B, SETDB1,
SETD2, and CFP1 exhibits significant changes in the oocytes and
granulosa cells of aged mouse ovaries. Histochem Cell Biol.
158:79–95. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Karkhanis V, Hu YJ, Baiocchi RA, Imbalzano
AN and Sif S: Versatility of PRMT5-induced methylation in growth
control and development. Trends Biochem Sci. 36:633–641. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Zhao F, Lan Y, Chen T, Xin Z, Liang Y, Li
Y, Wang S, Zhang J and Yang X: Live birth rate comparison of three
controlled ovarian stimulation protocols for in vitro
fertilization-embryo transfer in patients with diminished ovarian
reserve after endometrioma cystectomy: A retrospective study. J
Ovarian Res. 13:232020. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Olsen KW, Castillo-Fernandez J, Chan AC,
la Cour Freiesleben N, Zedeler A, Bungum M, Cardona A, Perry JRB,
Skouby SO, Hoffmann ER, et al: Identification of a unique
epigenetic profile in women with diminished ovarian reserve. Fertil
Steril. 115:732–741. 2021. View Article : Google Scholar
|
|
106
|
Jin J, Ren P, Li X, Zhang Y, Yang W, Ma Y,
Lai M, Yu C, Zhang S and Zhang YL: Ovulatory signal-triggered
chromatin remodeling in ovarian granulosa cells by HDAC2
phosphorylation activation-mediated histone deacetylation.
Epigenetics Chromatin. 16:112023. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Ye Q, Zeng X, Wang S, Zeng X, Yang G, Ye
C, Cai S, Chen M, Li S and Qiao S: Butyrate drives the acetylation
of histone H3K9 to activate steroidogenesis through PPARγ and PGC1α
pathways in ovarian granulosa cells. FASEB J. 35:e213162021.
View Article : Google Scholar
|
|
108
|
Zhou X, He Y, Quan H, Yang J, Li S, Jiang
Y, Li J and Yuan X: Exposure to nicotine regulates prostaglandin E2
secretion and autophagy of granulosa cells to retard follicular
maturation in mammals. Ecotoxicol Environ Saf. 277:1163582024.
View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Zhang YL, Xia Y, Yu C, Richards JS, Liu J
and Fan HY: CBP-CITED4 is required for luteinizing
hormone-triggered target gene expression during ovulation. Mol Hum
Reprod. 20:850–860. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Min Z, Long X, Zhao H, Zhen X, Li R, Li M,
Fan Y, Yu Y, Zhao Y and Qiao J: Protein lysine acetylation in
ovarian granulosa cells affects metabolic homeostasis and clinical
presentations of women with polycystic ovary syndrome. Front Cell
Dev Biol. 8:5670282020. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Zhao S, Cui H, Fang X, Xia W, Tao C and Li
J: Increased DNMT1 acetylation leads to global DNA methylation
suppression in follicular granulosa cells during reproductive aging
in mammals. BMC Genomics. 25:10302024. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Zhang M, Zhang Q, Hu Y, Xu L, Jiang Y,
Zhang C, Ding L, Jiang R, Sun J, Sun H and Yan G: miR-181a
increases FoxO1 acetylation and promotes granulosa cell apoptosis
via SIRT1 downregulation. Cell Death Dis. 8:e30882017. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Kong C, Su J, Wang Q, Liu K, Fu R and Sui
S: Signaling pathways of periplaneta americana peptide resist
H2O2-induced apoptosis in pig-ovary granulosa
cells through FoxO1. Theriogenology. 183:108–119. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Guo B, Zhang S, Wang S, Zhang H, Fang J,
Kang N, Zhen X, Zhang Y, Zhou J, Yan G, et al: Decreased HAT1
expression in granulosa cells disturbs oocyte meiosis during mouse
ovarian aging. Reprod Biol Endocrinol. 21:1032023. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Park SA, Joo NR, Park JH and Oh SM: Role
of the SIRT1/p53 regulatory axis in oxidative stress-mediated
granulosa cell apoptosis. Mol Med Rep. 23:202021.
|
|
116
|
Joo NR, Park SA, Park JH and Oh SM: TOPK
inhibits TNF-α-induced granulosa cell apoptosis via regulation of
SIRT1/p53. Biochem Biophys Res Commun. 664:128–135. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Park JH, Park SA, Lee YJ, Joo NR, Shin J
and Oh SM: TOPK inhibition accelerates oxidative stress-induced
granulosa cell apoptosis via the p53/SIRT1 axis. Int J Mol Med.
46:1923–1937. 2020.PubMed/NCBI
|
|
118
|
Shvedunova M and Akhtar A: Modulation of
cellular processes by histone and non-histone protein acetylation.
Nat Rev Mol Cell Biol. 23:329–349. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Xu Y and Wan W: Acetylation in the
regulation of autophagy. Autophagy. 19:379–387. 2023. View Article : Google Scholar :
|
|
120
|
Narita T, Weinert BT and Choudhary C:
Functions and mechanisms of non-histone protein acetylation. Nat
Rev Mol Cell Biol. 20:156–174. 2019. View Article : Google Scholar
|
|
121
|
Shen M, Jiang Y, Guan Z, Cao Y, Li L, Liu
H and Sun SC: Protective mechanism of FSH against oxidative damage
in mouse ovarian granulosa cells by repressing autophagy.
Autophagy. 13:1364–1385. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Chen C, Zhou M, Ge Y and Wang X: SIRT1 and
aging related signaling pathways. Mech Ageing Dev. 187:1112152020.
View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Wang X, Lin C, Zhao X, Liu A, Zhu J, Li X
and Song L: Acylglycerol kinase promotes cell proliferation and
tumorigenicity in breast cancer via suppression of the FOXO1
transcription factor. Mol Cancer. 13:1062014. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Xu G, Dong Y, Wang Z, Ding H, Wang J, Zhao
J, Liu H and Lv W: Melatonin attenuates oxidative stress-induced
apoptosis of bovine ovarian granulosa cells by promoting mitophagy
via SIRT1/FoxO1 signaling pathway. Int J Mol Sci. 24:128542023.
View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Sun L, Ye H, Tian H, Xu L, Cai J, Zhang C,
Wang R, Yang H, Zhao S, Zhang J and Gao S: The E3 ubiquitin ligase
SYVN1 plays an antiapoptotic role in polycystic ovary syndrome by
regulating mitochondrial fission. Oxid Med Cell Longev.
2022:36393022022. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Ma J, Chen S, Liu J, Liao Y, Li L, Wang
CC, Song S, Feng R, Hu H and Quan S: Cryptochrome 1 regulates
ovarian granulosa cell senescence through NCOA4-mediated
ferritinophagy. Free Radic Biol Med. 217:1–14. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Ma LZ, Wang A, Lai YH, Zhang J, Zhang XF,
Chen SL and Zhou XY: USP14 inhibition promotes DNA damage repair
and represses ovarian granulosa cell senescence in premature
ovarian insufficiency. J Transl Med. 22:8342024. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Shi S, Chu G, Zhang L, Yuan H, Madaniyati
M, Zhou X, Wang L, Cai C, Pang W, Gao L and Yang G: Deubiquitinase
UCHL1 regulates estradiol synthesis by stabilizing
voltage-dependent anion channel 2. J Biol Chem. 299:1053162023.
View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Liu X, Sun C, Zou K, Li C, Chen X, Gu H,
Zhou Z, Yang Z, Tu Y, Qin N, et al: Novel PGK1 determines
SKP2-dependent AR stability and reprograms granular cell glucose
metabolism facilitating ovulation dysfunction. EBioMedicine.
61:1030582020. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Tang H, Jiang X, Hua Y, Li H, Zhu C, Hao
X, Yi M and Li L: NEDD4L facilitates granulosa cell ferroptosis by
promoting GPX4 ubiquitination and degradation. Endocr Connect.
12:e2204592023. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Han S, Yu C, Qiu M, Xiong X, Peng H, Song
X, Hu C, Zhang Z, Xia B, Yang L, et al: USP13 regulates ferroptosis
in chicken follicle granulosa cells by deubiquitinating ATG7. Poult
Sci. 103:1042092024. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Zhang Y, Wang H, Zhu Y, Hou X, Li X, Zhou
X, Ge L, Xu J and Su Y: The novel peptide PFAP1 promotes primordial
follicle activation by binding to MCM5. FASEB J. 37:e229312023.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Yang L, Wang S, Pan Z, Du X and Li Q:
TGFBR2 is a novel substrate and indirect transcription target of
deubiquitylase USP9X in granulosa cells. J Cell Physiol.
237:2969–2979. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Wang Y and Zeng S: Melatonin promotes
ubiquitination of phosphorylated pro-apoptotic protein
Bcl-2-interacting mediator of cell death-extra long
(BimEL) in porcine granulosa cells. Int J Mol Sci.
19:34312018. View Article : Google Scholar
|
|
135
|
Kwon YT and Ciechanover A: The ubiquitin
code in the ubiquitin-proteasome system and autophagy. Trends
Biochem Sci. 42:873–886. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Zhao T, He M, Zhu Z, Zhang T, Zheng W, Qin
S, Gao M, Wang W, Chen Z, Han J, et al: P62 promotes FSH-induced
antral follicle formation by directing degradation of ubiquitinated
WT1. Cell Mol Life Sci. 81:2212024. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Wang Y, Zhuo A, Yang Y, Wang Q, Xie J, Ma
W, Chen Y, Gao M, Tang L and Fu X: Regulatory T cells
overexpressing Peli1 show better efficacy in repairing ovarian
endocrine function in autoimmune premature ovarian insufficiency.
Lab Invest. 103:1000052023. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Wang TF, Feng ZQ, Sun YW, Zhao SJ, Zou HY,
Hao HS, Du WH, Zhao XM, Zhu HB and Pang YW: Disruption of
O-GlcNAcylation homeostasis induced ovarian granulosa cell injury
in bovine. Int J Mol Sci. 23:78152022. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Lyu L, Qin X, Xiu H, Qu Y, Wang Y, Yang X,
Dang W and Kebreab E: Neddylation inhibition affects cell
proliferation and steroidogenesis in sheep follicular granulosa
cells. Theriogenology. 237:99–109. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Zhang Y, He F, Cai N, Chen G, Wang Y, Bai
W and Guo P: SIRT5 regulates granulosa cell proliferation and
apoptosis in polycystic ovarian syndrome via desuccinylation of
GLI1. Gynecol Endocrinol. 41:25155162025. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Wang T, Ye Z, Li Z, Jing DS, Fan GX, Liu
MQ, Zhuo QF, Ji SR, Yu XJ, Xu XW and Qin Y: Lactate-induced protein
lactylation: A bridge between epigenetics and metabolic
reprogramming in cancer. Cell Prolif. 56:e134782023. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Tan M, Luo H, Lee S, Jin F, Yang JS,
Montellier E, Buchou T, Cheng Z, Rousseaux S, Rajagopal N, et al:
Identification of 67 histone marks and histone lysine crotonylation
as a new type of histone modification. Cell. 146:1016–1028. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Yang P, Qin Y, Zeng L, He Y, Xie Y, Cheng
X, Huang W and Cao L: Crotonylation and disease: Current progress
and future perspectives. Biomed Pharmacother. 165:1151082023.
View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Wan J, Liu H, Chu J and Zhang H: Functions
and mechanisms of lysine crotonylation. J Cell Mol Med.
23:7163–7169. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Enchev RI, Schulman BA and Peter M:
Protein neddylation: Beyond cullin-RING ligases. Nat Rev Mol Cell
Biol. 16:30–44. 2015. View Article : Google Scholar
|
|
146
|
Wu D, Cai Y and Jin J: Potential
coordination role between O-GlcNAcylation and epigenetics. Protein
Cell. 8:713–723. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Roy S, Gandra D, Seger C, Biswas A,
Kushnir VA, Gleicher N, Kumar TR and Sen A: Oocyte-derived factors
(GDF9 and BMP15) and FSH regulate AMH expression via modulation of
H3K27AC in granulosa cells. Endocrinology. 159:3433–3445. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Schreiber NB and Spicer LJ: Effects of
fibroblast growth factor 9 (FGF9) on steroidogenesis and gene
expression and control of FGF9 mRNA in bovine granulosa cells.
Endocrinology. 153:4491–4501. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Guo Y, Zhang Y, Wang Y, Chen Q, Sun Y,
Kang L and Jiang Y: Phosphorylation of LSD1 at serine 54 regulates
genes involved in follicle selection by enhancing demethylation
activity in chicken ovarian granulosa cells. Poult Sci.
103:1038502024. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Kimura N, Hoshino Y, Totsukawa K and Sato
E: Cellular and molecular events during oocyte maturation in
mammals: Molecules of cumulus-oocyte complex matrix and signalling
pathways regulating meiotic progression. Soc Reprod Fertil Suppl.
63:327–342. 2007.PubMed/NCBI
|
|
151
|
Liao HQ and Zhou J, Cao Y, Nie YL, Li MQ
and Zhou J: Vigilin interacts with ER-β to protect against palmitic
acid-induced granulosa cells apoptosis via inhibiting
calcineurin-mediated Drp1 signaling pathway. Steroids.
163:1086992020. View Article : Google Scholar
|
|
152
|
Wan J and Liu S: Rosiglitazone ameliorates
palmitic acid-induced endoplasmic reticulum stress and
steroidogenic capacity in granulosa cells. Reprod Biol. 20:293–299.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Shibahara H, Ishiguro A, Inoue Y, Koumei
S, Kuwayama T and Iwata H: Mechanism of palmitic acid-induced
deterioration of in vitro development of porcine oocytes and
granulosa cells. Theriogenology. 141:54–61. 2020. View Article : Google Scholar
|
|
154
|
Benoit G, Warma A, Lussier JG and Ndiaye
K: Gonadotropin regulation of ankyrin-repeat and SOCS-box protein 9
(ASB9) in ovarian follicles and identification of binding partners.
PLoS One. 14:e02125712019. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Wang X, Zhou L, Xu A, Nima D and Dong Z:
Effects of stem cell factor in follicular fluid and granulosa cells
on oocyte maturity and clinical pregnancy. Medicine (Baltimore).
102:e367492023. View Article : Google Scholar :
|
|
156
|
Yang X, Liu P, He H, Qi D and Yan L:
Comprehensive analysis of ovarian granulosa cell proteomics and
phosphoproteomics in PCOS patients without insulin resistance. Mol
Hum Reprod. 30:gaae0052024. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Wang M, Chen T, Zheng J, Shen C, He G,
Zhang J, Zhang B and Zheng L: The role of CLDN11 in promotion of
granulosa cell proliferation in polycystic ovary syndrome via
activation of the PI3K-AKT signalling pathway. Sci Rep.
15:35332025. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Yang F, Liu Q, Chen Y, Ye H, Wang H and
Zeng S: Integrative Proteomic and phosphoproteomic analyses of
granulosa cells during follicular atresia in porcine. Front Cell
Dev Biol. 8:6249852021. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Wang Y, Zhang J, Liang J, Jia L, Niu S,
Cheng K, Yang C, Lu Z, Mu L, Yang X, et al: In vivo promotion of
primordial follicle activation by stem cell factor treatment in
mice with premature ovarian insufficiency and advanced age. Mol Hum
Reprod. 29:gaac0412022. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Li Q, Chen J, Yu X and Gao JM: A mini
review of nervonic acid: Source, production, and biological
functions. Food Chem. 301:1252862019. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Zeng X, Fan X, Yu H, Cai S, Zhou L, Wu H,
Zhang Z, Quan S, Li S, Wang X, et al: Nervonic acid triggered
ovarian inflammation by inducing mitochondrial oxidative stress to
activate NLRP3/IL-1β pathway. J Adv Res. 73:73–91. 2025. View Article : Google Scholar :
|
|
162
|
Fotovati A, Abu-Ali S, Nakayama K and
Nakayama KI: Impaired ovarian development and reduced fertility in
female mice deficient in Skp2. J Anat. 218:668–677. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Lv S, Liu M, Xu L and Zhang C:
Downregulation of decidual SKP2 is associated with human recurrent
miscarriage. Reprod Biol Endocrinol. 19:882021. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Ding L, Yan G, Wang B, Xu L, Gu Y, Ru T,
Cui X, Lei L, Liu J, Sheng X, et al: Transplantation of UC-MSCs on
collagen scaffold activates follicles in dormant ovaries of POF
patients with long history of infertility. Sci China Life Sci.
61:1554–1565. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Nosratpour S and Ndiaye K: Ankyrin-repeat
and SOCS box-containing protein 9 (ASB9) regulates ovarian
granulosa cells function and MAPK signaling. Mol Reprod Dev.
88:830–843. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Wei Y, Lin Z, Huang Q, Wu H, Wang R and
Wang J: Burden of female infertility in 204 countries and
territories, 1990-2021: Results from the global burden of disease
study 2021. J Psychosom Obstet Gynaecol. 46:24596182025. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Zhu M, Miao S, Zhou W, Elnesr SS, Dong X
and Zou X: MAPK, AKT/FoxO3a and mTOR pathways are involved in
cadmium regulating the cell cycle, proliferation and apoptosis of
chicken follicular granulosa cells. Ecotoxicol Environ Saf.
214:1120912021. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Han Z, Liu J, Liang T, Yin J, Wei J, Zeng
Q, Cao W, Liu C and Sun S: Exposure to ambient particulate matter
and ovarian reserve impairment among reproductive age women in
China. J Hazard Mater. 480:1362122024. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Fauque P, De Mouzon J, Devaux A, Epelboin
S, Gervoise-Boyer MJ, Levy R, Valentin M, Viot G, Bergère A, De
Vienne C, et al: Reproductive technologies, female infertility, and
the risk of imprinting-related disorders. Clin Epigenetics.
12:1912020. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Dummer R, Ascierto PA, Gogas HJ, Arance A,
Mandala M, Liszkay G, Garbe C, Schadendorf D, Krajsova I, Gutzmer
R, et al: Encorafenib plus binimetinib versus vemurafenib or
encorafenib in patients with BRAF-mutant melanoma (COLUMBUS): A
multicentre, open-label, randomised phase 3 trial. Lancet Oncol.
19:603–615. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Kopp KO, Greer ME, Glotfelty EJ, Hsueh SC,
Tweedie D, Kim DS, Reale M, Vargesson N and Greig NH: A new
generation of IMiDs as treatments for neuroinflammatory and
neurodegenerative disorders. Biomolecules. 13:7472023. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Sun X, Zhang Q, Lin X, Shu P, Gao X and
Shen K: Imatinib induces ferroptosis in gastrointestinal stromal
tumors by promoting STUB1-mediated GPX4 ubiquitination. Cell Death
Dis. 14:8392023. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Wang F, Jin Y, Wang M, Luo HY, Fang WJ,
Wang YN, Chen YX, Huang RJ, Guan WL, Li JB, et al: Combined
anti-PD-1, HDAC inhibitor and anti-VEGF for MSS/pMMR colorectal
cancer: a randomized phase 2 trial. Nat Med. 30:1035–1043. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Nguyen TTA and Demeestere I: A journey to
reach the ovary using next-generation technologies. Int J Mol Sci.
24:165932023. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Shi M, Li X, Xing L, Li Z, Zhou S, Wang Z,
Zou X, She Y, Zhao R and Qin D: Polycystic ovary syndrome and the
potential for nanomaterial-based drug delivery in therapy of this
disease. Pharmaceutics. 16:15562024. View Article : Google Scholar :
|
|
176
|
Chen Y, Xie M, Wu S, Deng Z, Tang Y, Guan
Y, Ye Y, He Q and Li L: Multi-omics approach to reveal follicular
metabolic changes and their effects on oocyte competence in PCOS
patients. Front Endocrinol (Lausanne). 15:14265172024. View Article : Google Scholar : PubMed/NCBI
|