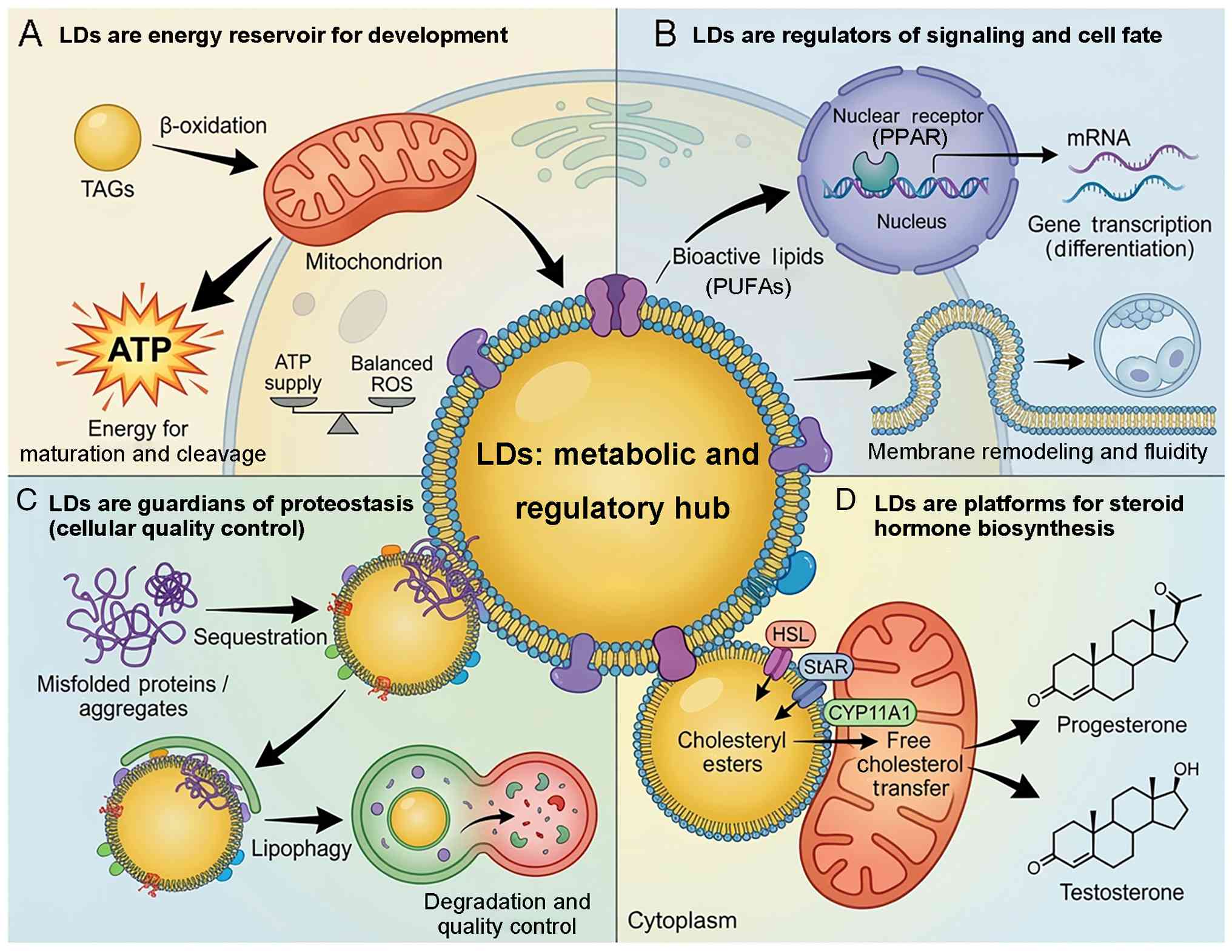
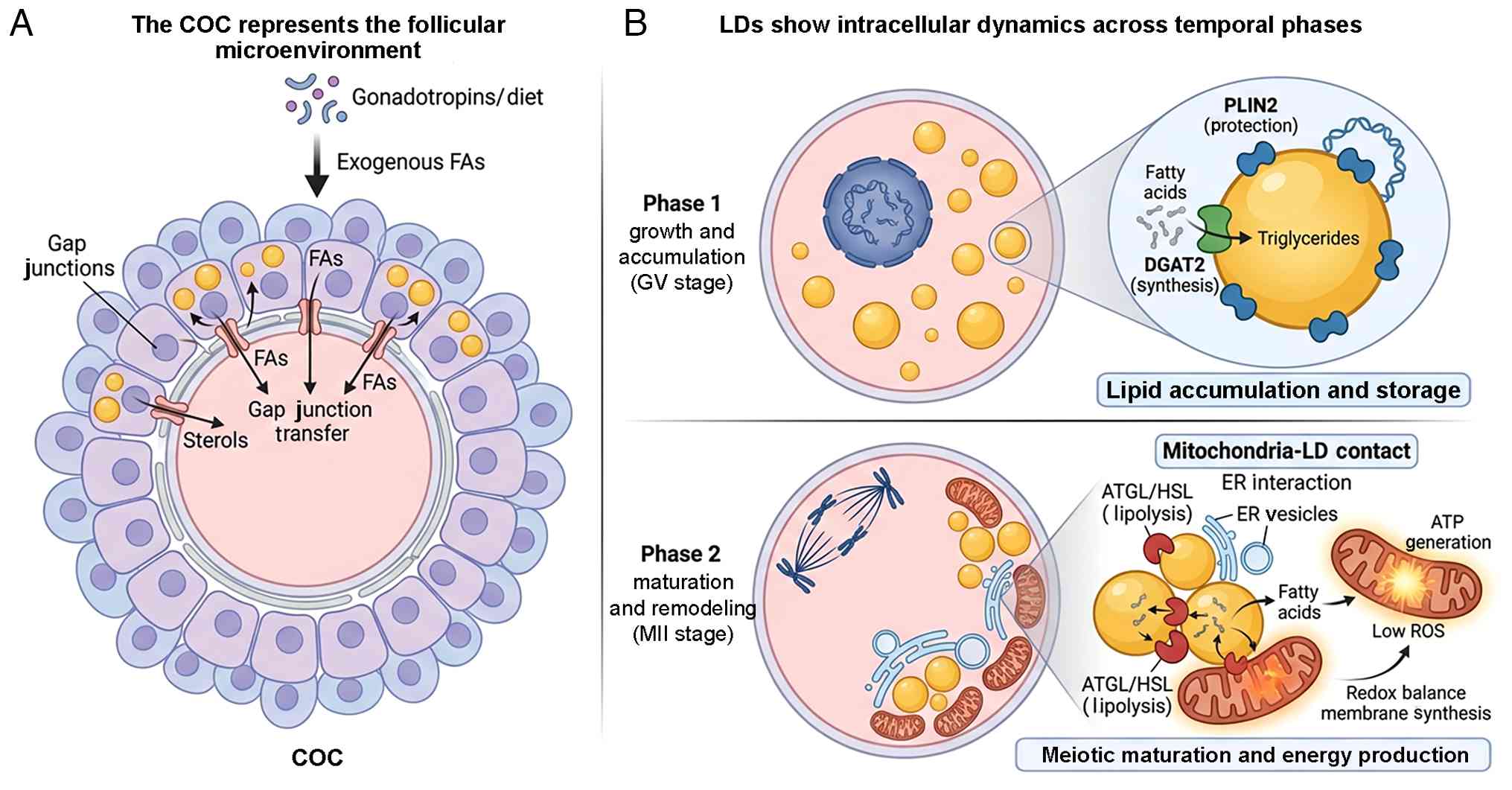
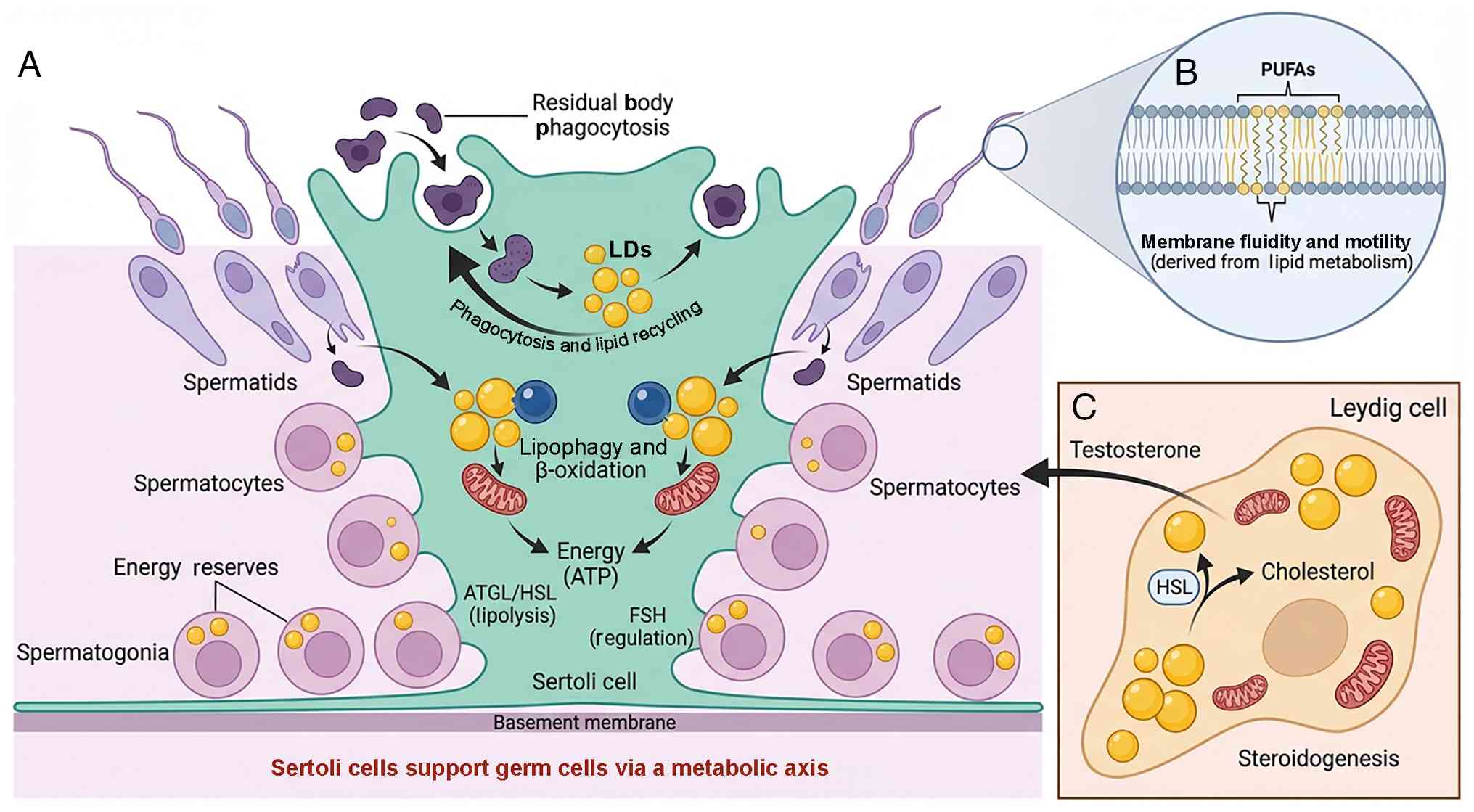
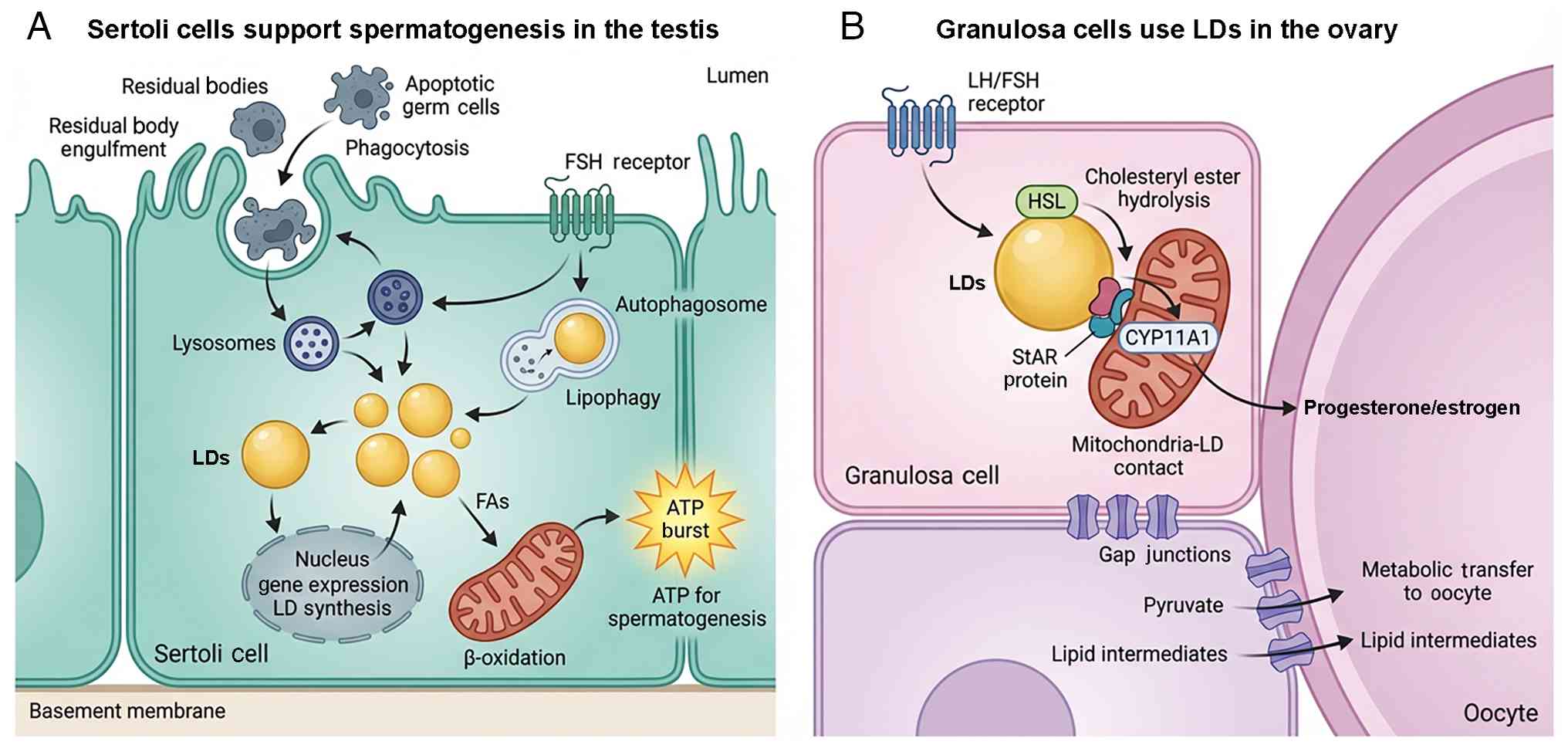
|
1
|
Farese RV Jr and Walther TC: Lipid
droplets finally get a little R-E-S-P-E-C-T. Cell. 139:855–860.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Olzmann JA and Carvalho P: Dynamics and
functions of lipid droplets. Nat Rev Mol Cell Biol. 20:137–155.
2019. View Article : Google Scholar
|
|
3
|
Walther TC, Chung J and Farese RV Jr:
Lipid droplet biogenesis. Annu Rev Cell Dev Biol. 33:491–510. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Salo VT, Belevich I, Li S, Karhinen L,
Vihinen H, Vigouroux C, Magré J, Thiele C, Hölttä-Vuori M, Jokitalo
E and Ikonen E: Seipin regulates ER-lipid droplet contacts and
cargo delivery. EMBO J. 35:2699–2716. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cartwright BR and Goodman JM: Seipin: From
human disease to molecular mechanism. J Lipid Res. 53:1042–1055.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Welte MA and Gould AP: Lipid droplet
functions beyond energy storage. Biochim Biophys Acta Mol Cell Biol
Lipids. 1862:1260–1272. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bosch M, Sanchez-Alvarez M, Fajardo A,
Kapetanovic R, Steiner B, Dutra F, Moreira L, López JA, Campo R,
Marí M, et al: Mammalian lipid droplets are innate immune hubs
integrating cell metabolism and host defense. Science.
370:eaay80852020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lass A, Zimmermann R, Oberer M and Zechner
R: Lipolysis-a highly regulated multi-enzyme complex mediates the
catabolism of cellular fat stores. Prog Lipid Res. 50:14–27. 2011.
View Article : Google Scholar :
|
|
9
|
Cruz ALS, Barreto EA, Fazolini NPB, Viola
JPB and Bozza PT: Lipid droplets: Platforms with multiple functions
in cancer hallmarks. Cell Death Dis. 11:1052020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Henne WM, Reese ML and Goodman JM: The
assembly of lipid droplets and their roles in challenged cells.
EMBO J. 37:e989472018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sturmey RG, Reis A, Leese HJ and McEvoy
TG: Role of fatty acids in energy provision during oocyte
maturation and early embryo development. Reprod Domest Anim.
44(Suppl 3): S50–S58. 2009. View Article : Google Scholar
|
|
12
|
Brusentsev EY, Mokrousova VI, Igonina TN,
Rozhkova IN and Amstislavsky SY: Role of lipid droplets in the
development of oocytes and preimplantation embryos in mammals. Russ
J Dev Biol. 50:230–237. 2019. View Article : Google Scholar
|
|
13
|
Bradley J and Swann K: Mitochondria and
lipid metabolism in mammalian oocytes and early embryos. Int J Dev
Biol. 63:93–103. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Aardema H, Vos PL, Lolicato F, Roelen BA,
Knijn HM, Vaandrager AB, Helms JB and Gadella BM: Oleic acid
prevents detrimental effects of saturated fatty acids on bovine
oocyte developmental competence. Biol Reprod. 85:62–69. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ahmed N, Liu Y, Chen H, Yang P, Waqas Y,
Liu T, Gandahi JA, Huang Y, Wang L, Song X, et al: Novel cellular
evidence of lipophagy within the Sertoli cells during
spermatogenesis in the turtle. Aging (Albany NY). 9:41–51. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Staggenborg S, Koch R, Rode K, Hüneke H,
Tiedje L, Wirth G, Langeheine M, Blume I, Rohn K, Wrede C, et al:
Connexin43 represents an important regulator for Sertoli cell
morphology, Sertoli cell nuclear ultrastructure, and Sertoli cell
maturation. Sci Rep. 12:128982022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang H, Wang H, Xiong W, Chen Y, Ma Q, Ma
J, Ge Y and Han D: Evaluation on the phagocytosis of apoptotic
spermatogenic cells by Sertoli cells in vitro through detecting
lipid droplet formation by Oil Red O staining. Reproduction.
132:485–492. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yamaguchi T, Fujikawa N, Nimura S, Tokuoka
Y, Tsuda S, Aiuchi T, Kato R, Obama T and Itabe H: Characterization
of lipid droplets in steroidogenic MLTC-1 Leydig cells: Protein
profiles and the morphological change induced by hormone
stimulation. Biochim Biophys Acta. 1851:1285–1295. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gao F, Li G, Liu C, Gao H, Wang H, Liu W,
Chen M, Shang Y, Wang L, Shi J, et al: Autophagy regulates
testosterone synthesis by facilitating cholesterol uptake in Leydig
cells. J Cell Biol. 217:2103–2119. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Plewes MR, Talbott HA, Saviola AJ, Woods
NT, Schott MB and Davis JS: Luteal lipid droplets: A novel platform
for steroid synthesis. Endocrinology. 164:bqad1242023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Przygrodzka E, Hou X, Zhang P, Plewes MR,
Franco R and Davis JS: PKA and AMPK signaling pathways
differentially regulate luteal steroidogenesis. Endocrinology.
162:bqab0152021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Plewes MR, Krause C, Talbott HA,
Przygrodzka E, Wood JR, Cupp AS and Davis JS: Trafficking of
cholesterol from lipid droplets to mitochondria in bovine luteal
cells: Acute control of progesterone synthesis. FASEB J.
34:10731–10750. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Khor VK, Ahrends R, Lin Y, Shen WJ, Adams
CM, Roseman AN, Cortez Y, Teruel MN, Azhar S and Kraemer FB: The
proteome of cholesteryl-ester-enriched versus
triacylglycerol-enriched lipid droplets. PLoS One. 9:e1050472014.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shen WJ, Azhar S and Kraemer FB: Lipid
droplets and steroidogenic cells. Exp Cell Res. 340:209–214. 2016.
View Article : Google Scholar
|
|
25
|
Cao Z, Fung CW and Mak HY: A flexible
network of lipid droplet associated proteins support embryonic
integrity of C. elegans. Front Cell Dev Biol. 10:8564742022.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bai X, Huang LJ, Chen SW, Nebenfuehr B,
Wysolmerski B, Wu JC, Olson SK, Golden A and Wang CW: Loss of the
seipin gene perturbs eggshell formation in Caenorhabditiselegans.
Development. 147:dev1929972020. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tao A, Wu T, Han X, Niu D and Feng X:
Lipidomics reveals common mechanisms in polycystic ovarian
syndrome, recurrent spontaneous abortion, and infertility: A
genetic-based analysis. Int J Womens Health. 17:1055–1065. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Raviv S, Hantisteanu S, Sharon SM, Atzmon
Y, Michaeli M and Shalom-Paz E: Lipid droplets in granulosa cells
are correlated with reduced pregnancy rates. J Ovarian Res.
13:42020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu Q, Xie YJ, Qu LH, Zhang MX and Mo ZC:
Dyslipidemia involvement in the development of polycystic ovary
syndrome. Taiwan J Obstet Gynecol. 58:447–453. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang CH, Liu XY and Wang J: Essential
role of granulosa cell glucose and lipid metabolism on oocytes and
the potential metabolic imbalance in polycystic ovary Syndrome. Int
J Mol Sci. 24:162472023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bradley J, Pope I, Masia F, Sanusi R,
Langbein W, Swann K and Borri P: Quantitative imaging of lipids in
live mouse oocytes and early embryos using CARS microscopy.
Development. 143:2238–2247. 2016.PubMed/NCBI
|
|
32
|
Zhang C: Coherent Raman scattering
microscopy of lipid droplets in cells and tissues. J Raman
Spectrosc. 54:988–1000. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Inoue N, Nishida Y, Harada E, Sakai K and
Narahara H: GC-MS/MS analysis of metabolites derived from a single
human blastocyst. Metabolomics. 17:172021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Schwartz HT and Sternberg PW: A
sequencing-based screening method identifies regulators of EGFR
signaling from nonviable mutants in Caenorhabditis elegans. Sci
Signal. 18:eadp93772025. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
An L, Fu X, Chen J and Ma J: Application
of caenorhabditis elegans in lipid metabolism research. Int J Mol
Sci. 24:11732023. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wamaitha SE, Nie X, Pandolfi EC, Wang X,
Yang Y, Stukenborg JB, Cairns BR, Guo J and Clark AT: Single-cell
analysis of the developing human ovary defines distinct insights
into ovarian somatic and germline progenitors. Dev Cell.
58:2097–2111 e3. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ibayashi M, Aizawa R, Mitsui J and
Tsukamoto S: Homeostatic regulation of lipid droplet content in
mammalian oocytes and embryos. Reproduction. 162:R99–R109. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Purcell SH and Moley KH: The impact of
obesity on egg quality. J Assist Reprod Genet. 28:517–524. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Leary C, Leese HJ and Sturmey RG: Human
embryos from overweight and obese women display phenotypic and
metabolic abnormalities. Hum Reprod. 30:122–132. 2015. View Article : Google Scholar
|
|
40
|
Jiang M, Gao M, Wu C, He H, Guo X, Zhou Z,
Yang H, Xiao X, Liu G and Sha J: Lack of testicular seipin causes
teratozoospermia syndrome in men. Proc Natl Acad Sci USA.
111:7054–7059. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Khan R, Jiang X, Hameed U and Shi Q: Role
of lipid metabolism and signaling in mammalian oocyte maturation,
quality, and acquisition of competence. Front Cell Dev Biol.
9:6397042021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lipinska P, Pawlak P and Warzych E:
Species and embryo genome origin affect lipid droplets in
preimplantation embryos. Front Cell Dev Biol. 11:11878322023.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chao CF, Pesch YY, Yu H, Wang C,
Aristizabal MJ, Huan T, Tanentzapf G and Rideout E: An important
role for triglyceride in regulating spermatogenesis. Elife.
12:RP875232024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Aizawa R, Ibayashi M, Mitsui J and
Tsukamoto S: Lipid droplet formation is spatiotemporally regulated
in oocytes during follicular development in mice. J Reprod Dev.
70:18–24. 2024. View Article : Google Scholar :
|
|
45
|
Kerr JB and De Kretser DM: Cyclic
variations in Sertoli cell lipid content throughout the
spermatogenic cycle in the rat. J Reprod Fertil. 43:1–8. 1975.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Regueira M, Gorga A, Rindone GM,
Pellizzari EH, Cigorraga SB, Galardo MN, Riera MF and Meroni SB:
Apoptotic germ cells regulate Sertoli cell lipid storage and fatty
acid oxidation. Reproduction. 156:515–525. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Talbott HA, Plewes MR, Krause C, Hou X,
Zhang P, Rizzo WB, Wood JR, Cupp AS and Davis JS: Formation and
characterization of lipid droplets of the bovine corpus luteum. Sci
Rep. 10:112872020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Plewes MR, Talbott HA, Schott MB, Wood JR,
Cupp AS and Davis JS: Unraveling the role of lipid droplets and
perilipin 2 in bovine luteal cells. FASEB J. 38:e237102024.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Dunning KR, Russell DL and Robker RL:
Lipids and oocyte developmental competence: The role of fatty acids
and beta-oxidation. Reproduction. 148:R15–R27. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Schindler M, Pendzialek M, Grybel KJ,
Seeling T, Gürke J, Fischer B and Navarrete Santos A: Adiponectin
stimulates lipid metabolism via AMPK in rabbit blastocysts. Hum
Reprod. 32:1382–1392. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ma W, Yang X and Liang X: Obesity does not
aggravate vitrification injury in mouse embryos: A prospective
study. Reprod Biol Endocrinol. 10:682012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hallberg M, Morganstein DL, Kiskinis E,
Shah K, Kralli A, Dilworth SM, White R, Parker MG and Christian M:
A functional interaction between RIP140 and PGC-1alpha regulates
the expression of the lipid droplet protein CIDEA. Mol Cell Biol.
28:6785–6795. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Ning Z, Deng X, Li L, Feng J, Du X, Amevor
FK, Tian Y, Li L, Rao Y, Yi Z, et al: miR-128-3p regulates chicken
granulosa cell function via 14-3-3β/FoxO and PPAR-ү/LPL signaling
pathways. Int J Biol Macromol. 241:1246542023. View Article : Google Scholar
|
|
54
|
Cui L and Liu P: Two types of contact
between lipid droplets and mitochondria. Front Cell Dev Biol.
8:6183222020. View Article : Google Scholar :
|
|
55
|
Ma X, Qian H, Chen A, Ni HM and Ding WX:
Perspectives on mitochondria-ER and mitochondria-lipid droplet
contact in hepatocytes and hepatic lipid metabolism. Cells.
10:22732021. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Latchman NR, Stevens TL, Bedi KC, Prosser
BL, Margulies KB and Elrod JW: Ultrastructure analysis of
mitochondria, lipid droplet and sarcoplasmic reticulum apposition
in human heart failure. J Mol Cell Cardiol Plus.
13:1004612025.PubMed/NCBI
|
|
57
|
Liu L, Yang S, Liu Y, Li X, Hu J, Xiao L
and Xu T: DeepContact: High-throughput quantification of membrane
contact sites based on electron microscopy imaging. J Cell Biol.
221:e2021061902022. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Benador IY, Veliova M, Liesa M and
Shirihai OS: Mitochondria bound to lipid droplets: Where
mitochondrial dynamics regulate lipid storage and utilization. Cell
Metab. 29:827–835. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Freyre CAC, Rauher PC, Ejsing CS and Klemm
RW: MIGA2 links mitochondria, the ER, and lipid droplets and
promotes de novo lipogenesis in adipocytes. Mol Cell. 76:811–825
e14. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Herker E, Vieyres G, Beller M, Krahmer N
and Bohnert M: Lipid droplet contact sites in health and disease.
Trends Cell Biol. 31:345–358. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Fan H and Tan Y: Lipid
droplet-mitochondria contacts in health and disease. Int J Mol Sci.
25:68782024. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Monteiro-Cardoso VF and Giordano F:
Emerging functions of the mitochondria-ER-lipid droplet three-way
junction in coordinating lipid transfer, metabolism, and storage in
cells. FEBS Lett. 598:1252–1273. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Benador IY, Veliova M, Mahdaviani K,
Petcherski A, Wikstrom JD, Assali EA, Acín-Pérez R, Shum M,
Oliveira MF, Cinti S, et al: Mitochondria bound to lipid droplets
have unique bioenergetics, composition, and dynamics that support
lipid droplet expansion. Cell Metab. 27:869–885 e6. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Talari NK, Mattam U, Meher NK, Paripati
AK, Mahadev K, Krishnamoorthy T and Sepuri NBV: Lipid-droplet
associated mitochondria promote fatty-acid oxidation through a
distinct bioenergetic pattern in male Wistar rats. Nat Commun.
14:7662023. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Flores-Romero H, Ros U and Garcia-Saez AJ:
A lipid perspective on regulated cell death. Int Rev Cell Mol Biol.
351:197–236. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Danielsen ET, Moeller ME, Yamanaka N, Ou
Q, Laursen JM, Soenderholm C, Zhuo R, Phelps B, Tang K, Zeng J, et
al: A Drosophila genome-wide screen identifies regulators of
steroid hormone production and developmental timing. Dev Cell.
37:558–570. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Prates EG, Nunes JT and Pereira RM: A role
of lipid metabolism during cumulus-oocyte complex maturation:
impact of lipid modulators to improve embryo production. Mediators
Inflamm. 2014:6920672014. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Liu T, Qu J, Tian M, Yang R, Song X, Li R,
Yan J and Qiao J: Lipid metabolic process involved in oocyte
maturation during folliculogenesis. Front Cell Dev Biol.
10:8068902022. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Li T, Jin Y, Wu J and Ren Z: Beyond energy
provider: Multifunction of lipid droplets in embryonic development.
Biol Res. 56:382023. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Kajdasz A, Warzych E, Derebecka N, Madeja
ZE, Lechniak D, Wesoly J and Pawlak P: Lipid stores and lipid
metabolism associated gene expression in porcine and bovine
parthenogenetic embryos revealed by fluorescent staining and
RNA-seq. Int J Mol Sci. 21:64882020. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
de Andrade Melo-Sterza F and Poehland R:
Lipid metabolism in bovine oocytes and early embryos under in vivo,
in vitro, and stress conditions. Int J Mol Sci. 22:34212021.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Arena R, Bisogno S, Gasior L, Rudnicka J,
Bernhardt L, Haaf T, Zacchini F, Bochenek M, Fic K, Bik E, et al:
Lipid droplets in mammalian eggs are utilized during embryonic
diapause. Proc Natl Acad Sci USA. 118:e20183621182021. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Downs SM: Nutrient pathways regulating the
nuclear maturation of mammalian oocytes. Reprod Fertil Dev.
27:572–582. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Agarwal A, Gupta S and Sharma R: Oxidative
stress and its implications in female infertility-a clinician's
perspective. Reprod Biomed Online. 11:641–650. 2005. View Article : Google Scholar
|
|
75
|
Aizawa R, Ibayashi M, Tatsumi T, Yamamoto
A, Kokubo T, Miyasaka N, Sato K, Ikeda S, Minami N and Tsukamoto S:
Synthesis and maintenance of lipid droplets are essential for mouse
preimplantation embryonic development. Development.
146:dev1819252019. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Zhang JF, Choi SH, Li Q, Wang Y, Sun B,
Tang L, Wang EZ, Hua H and Li XZ: Overexpression of DGAT2
stimulates lipid droplet formation and triacylglycerol accumulation
in bovine satellite cells. Animals (Basel). 12:18472022. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Malyszka N, Pawlak P, Cieslak A,
Szkudelska K and Lechniak D: Distinct dynamics of lipid
accumulation by porcine cumulus cells during in vitro maturation
with follicular fluid of low and high fatty acid contents.
Theriogenology. 195:93–102. 2023. View Article : Google Scholar
|
|
78
|
Zhang RN, Fu XW, Jia BY, Liu C, Cheng KR
and Zhu SE: Expression of perilipin 2 (PLIN2) in porcine oocytes
during maturation. Reprod Domest Anim. 49:875–880. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Sowinska N, Lechtanska J, Greczka K,
Lechniak D and Pawlak P: Feline cumulus cells and oocytes show
massive accumulation of lipid droplets and upregulation of PLIN2
expression after in vitro maturation. Theriogenology. 232:70–78.
2025. View Article : Google Scholar
|
|
80
|
Monks J, Orlicky DJ, Libby AE,
Dzieciatkowska M, Ladinsky MS and McManaman JL: Perilipin-2
promotes lipid droplet-plasma membrane interactions that facilitate
apocrine lipid secretion in secretory epithelial cells of the mouse
mammary gland. Front Cell Dev Biol. 10:9585662022. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Xiang X, Chen J and Che L: Emerging roles
and therapeutic implications of lipid droplet protein perilipin 2
in liver disease. Genes Dis. 13:1017122025. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Morak M, Schmidinger H, Riesenhuber G,
Rechberger GN, Kollroser M, Haemmerle G, Zechner R, Kronenberg F
and Hermetter A: Adipose triglyceride lipase (ATGL) and
hormone-sensitive lipase (HSL) deficiencies affect expression of
lipolytic activities in mouse adipose tissues. Mol Cell Proteomics.
11:1777–1789. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Miyoshi H, Perfield JW II, Obin MS and
Greenberg AS: Adipose triglyceride lipase regulates basal lipolysis
and lipid droplet size in adipocytes. J Cell Biochem.
105:1430–1436. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Schweiger M, Schreiber R, Haemmerle G,
Lass A, Fledelius C, Jacobsen P, Tornqvist H, Zechner R and
Zimmermann R: Adipose triglyceride lipase and hormone-sensitive
lipase are the major enzymes in adipose tissue triacylglycerol
catabolism. J Biol Chem. 281:40236–40241. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Lass A, Zimmermann R, Haemmerle G,
Riederer M, Schoiswohl G, Schweiger M, Kienesberger P, Strauss JG,
Gorkiewicz G and Zechner R: Adipose triglyceride lipase-mediated
lipolysis of cellular fat stores is activated by CGI-58 and
defective in Chanarin-Dorfman Syndrome. Cell Metab. 3:309–319.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Gaidhu MP, Anthony NM, Patel P, Hawke TJ
and Ceddia RB: Dysregulation of lipolysis and lipid metabolism in
visceral and subcutaneous adipocytes by high-fat diet: role of
ATGL, HSL, and AMPK. Am J Physiol Cell Physiol. 298:C961–C971.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Turathum B, Gao EM and Chian RC: The
function of cumulus cells in oocyte growth and maturation and in
subsequent ovulation and fertilization. Cells. 10:22922021.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Richani D, Poljak A, Wang B, Mahbub SB,
Biazik J, Campbell JM, Habibalahi A, Stocker WA, Marinova MB, Nixon
B, et al: Oocyte and cumulus cell cooperativity and metabolic
plasticity under the direction of oocyte paracrine factors. Am J
Physiol Endocrinol Metab. 326:E366–E381. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Dumesic DA, Meldrum DR, Katz-Jaffe MG,
Krisher RL and Schoolcraft WB: Oocyte environment: Follicular fluid
and cumulus cells are critical for oocyte health. Fertil Steril.
103:303–316. 2015. View Article : Google Scholar
|
|
90
|
Xie J, Xu X and Liu S: Intercellular
communication in the cumulus-oocyte complex during
folliculogenesis: A review. Front Cell Dev Biol. 11:10876122023.
View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Uhde K, van Tol HTA, Stout TAE and Roelen
BAJ: Metabolomic profiles of bovine cumulus cells and
cumulus-oocyte-complex-conditioned medium during maturation in
vitro. Sci Rep. 8:94772018. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Russell DL, Gilchrist RB, Brown HM and
Thompson JG: Bidirectional communication between cumulus cells and
the oocyte: Old hands and new players? Theriogenology. 86:62–68.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Chen M, Yang W, Guo Y, Hou X, Zhu S, Sun
H, Guo X, Chen M and Wang Q: Multi-omics reveal the metabolic
patterns in mouse cumulus cells during oocyte maturation. J Ovarian
Res. 16:1562023. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Lawson GLG: Composition of Lipid Droplets
and Vesicles and their Relation to Oocyte Development Competency.
Reader K: BBiomedSc (Hons). University of Otago; 2023
|
|
95
|
Ordonez-Leon EA, Merchant H, Medrano A,
Kjelland M and Romo S: Lipid droplet analysis using in vitro bovine
oocytes and embryos. Reprod Domest Anim. 49:306–314. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Budani MC and Tiboni GM: Effects of
supplementation with natural antioxidants on oocytes and
preimplantation embryos. Antioxidants (Basel). 9:6122020.
View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Fayezi S, Leroy JLMR, Ghaffari Novin M and
Darabi M: Oleic acid in the modulation of oocyte and
preimplantation embryo development. Zygote. 26:1–13. 2018.
View Article : Google Scholar
|
|
98
|
Rakha SI, Elmetwally MA, El-Sheikh Ali H,
Balboula A, Mahmoud AM and Zaabel SM: Importance of antioxidant
supplementation during in vitro maturation of mammalian oocytes.
Vet Sci. 9:4392022.PubMed/NCBI
|
|
99
|
Guseva O, Kan N, Chekmareva V, Kokorev D,
Ilyasov P and Zhu YQ: The impact of antioxidant supplements on
oocytes and preimplantation embryos of humans and mammals, and
their potential application for mitigating the consequences of
oxidative stress in vitro: A review. Reproductive and Developmental
Medicine. 8:252–263. 2024. View Article : Google Scholar
|
|
100
|
Dubeibe Marin DF, da Costa NN, di Paula
Bessa Santana P, de Souza EB and Ohashi OM: Importance of lipid
metabolism on oocyte maturation and early embryo development: Can
we apply what we know to buffalo? Anim Reprod Sci. 211:1062202019.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Catandi GD, LiPuma L, Obeidat YM,
Maclellan LJ, Broeckling CD, Chen T, Chicco AJ and Carnevale EM:
Oocyte metabolic function, lipid composition, and developmental
potential are altered by diet in older mares. Reproduction.
163:183–198. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Chen H, Huang Y, Yang P, Shi Y, Ahmed N,
Liu T, Bai X, Haseeb A and Chen Q: Autophagy enhances lipid droplet
development during spermiogenesis in Chinese soft-shelled turtle,
Pelodiscus sinensis. Theriogenology. 147:154–165. 2020. View Article : Google Scholar
|
|
103
|
Oresti GM, Ayuza Aresti PL, Gigola G,
Reyes LE and Aveldano MI: Sequential depletion of rat testicular
lipids with long-chain and very long-chain polyenoic fatty acids
after X-ray-induced interruption of spermatogenesis. J Lipid Res.
51:2600–2610. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Liu M, Qi L, Zeng Y, Yang Y, Bi Y, Shi X,
Zhu H, Zhou Z and Sha J: Transient scrotal hyperthermia induces
lipid droplet accumulation and reveals a different ADFP expression
pattern between the testes and liver in mice. PLoS One.
7:e456942012. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Furland NE, Luquez JM, Oresti GM and
Aveldano MI: Mild testicular hyperthermia transiently increases
lipid droplet accumulation and modifies sphingolipid and
glycerophospholipid acyl chains in the rat testis. Lipids.
46:443–454. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Osuga J, Ishibashi S, Oka T, Yagyu H,
Tozawa R, Fujimoto A, Shionoiri F, Yahagi N, Kraemer FB, Tsutsumi O
and Yamada N: Targeted disruption of hormone-sensitive lipase
results in male sterility and adipocyte hypertrophy, but not in
obesity. Proc Natl Acad Sci USA. 97:787–792. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Zhang J, Hu Y, Wang Y, Fu L, Xu X, Li C,
Xu J, Li C, Zhang L, Yang R, et al: mmBCFA C17iso ensures
endoplasmic reticulum integrity for lipid droplet growth. J Cell
Biol. 220:e2021021222021. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Wang C, Wang B, Pandey T, Long Y, Zhang J,
Oh F, Sima J, Guo R, Liu Y, Zhang C, et al: A conserved
megaprotein-based molecular bridge critical for lipid trafficking
and cold resilience. Nat Commun. 13:68052022. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Dasso ME, Centola CL, Galardo MN, Riera MF
and Meroni SB: FSH increases lipid droplet content by regulating
the expression of genes related to lipid storage in Rat Sertoli
cells. Mol Cell Endocrinol. 595:1124032025. View Article : Google Scholar
|
|
110
|
Gorga A, Rindone GM, Regueira M,
Pellizzari EH, Camberos MC, Cigorraga SB, Riera MF, Galardo MN and
Meroni SB: PPARү activation regulates lipid droplet formation and
lactate production in rat Sertoli cells. Cell Tissue Res.
369:611–624. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Chen ZF, Shen YF, Gao DW, Lin DF, Ma WZ
and Chang DG: Metabolic pathways and male fertility: Exploring the
role of Sertoli cells in energy homeostasis and spermatogenesis. Am
J Physiol Endocrinol Metab. 329:E160–E178. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Breucker H, Schafer E and Holstein AF:
Morphogenesis and fate of the residual body in human
spermiogenesis. Cell Tissue Res. 240:303–309. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Guo C, Wang L, Cui K, Zhang G, Tan Y, Chen
W, Wang Y, Liu J, Liu W, Zhang G and Zhou Z: Lead causes lipid
droplet accumulation by impairing lysosomal function and autophagic
flux in testicular sertoli cells. Toxics. 13:1752025. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Rindone GM, Dasso ME, Centola CL,
Pellizzari EH, Camberos MDC, Toneatto J, Galardo MN, Meroni SB and
Riera MF: Sertoli cell adaptation to glucose deprivation: Potential
role of AMPK in the regulation of lipid metabolism. J Cell Biochem.
124:716–730. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Bush SJ, Nikola R, Han S, Suzuki S,
Yoshida S, Simons BD and Goriely A: Adult human, but not rodent,
spermatogonial stem cells retain states with a foetal-like
signature. Cells. 13:7422024. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Wang J, Chen M, Yao Y, Zhu M, Jiang Y,
Duan J, Yuan Y, Li L, Chen M and Sha J: Characterization of
metabolic patterns in mouse spermatogenesis and its clinical
implications in humans. Int J Mol Sci. 26:10012025. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Plant TM and Marshall GR: The functional
significance of FSH in spermatogenesis and the control of its
secretion in male primates. Endocr Rev. 22:764–786. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Guo J, Grow EJ, Mlcochova H, Maher GJ,
Lindskog C, Nie X, Guo Y, Takei Y, Yun J, Cai L, et al: The adult
human testis transcriptional cell atlas. Cell Res. 28:1141–1157.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Paniagua R, Nistal M, Saez FJ and Fraile
B: Ultrastructure of the aging human testis. J Electron Microsc
Tech. 19:241–260. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Li N, Mruk DD, Mok KW, Li MW, Wong CK, Lee
WM, Han D, Silvestrini B and Cheng CY: Connexin 43 reboots meiosis
and reseals blood-testis barrier following toxicant-mediated
aspermatogenesis and barrier disruption. FASEB J. 30:1436–1452.
2016. View Article : Google Scholar
|
|
121
|
Vo BT, Morton D Jr, Komaragiri S, Millena
AC, Leath C and Khan SA: TGF-beta effects on prostate cancer cell
migration and invasion are mediated by PGE2 through activation of
PI3K/AKT/mTOR pathway. Endocrinology. 154:1768–1779. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Sahin MB, Schwartz RE, Buckley SM,
Heremans Y, Chase L, Hu WS and Verfaillie CM: Isolation and
characterization of a novel population of progenitor cells from
unmanipulated rat liver. Liver Transpl. 14:333–345. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Richburg JH, Myers JL and Bratton SB: The
role of E3 ligases in the ubiquitin-dependent regulation of
spermatogenesis. Semin Cell Dev Biol. 30:27–35. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Zalata AA, Christophe AB, Depuydt CE,
Schoonjans F and Comhaire FH: The fatty acid composition of
phospholipids of spermatozoa from infertile patients. Mol Hum
Reprod. 4:111–118. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Fusaro I, Parrillo S, Buonaiuto G,
Prasinou P, Gramenzi A, Bucci R, Cavallini D, Carosi A, Carluccio A
and De Amicis I: Effects of hemp-based polyunsaturated fatty acid
supplementation on membrane lipid profiles and reproductive
performance in Martina Franca jacks. Front Vet Sci. 12:15532182025.
View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Johnson MR, Stephenson RA, Ghaemmaghami S
and Welte MA: Developmentally regulated H2Av buffering via dynamic
sequestration to lipid droplets in Drosophila embryos. Elife.
7:e360212018. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Byrne CJ, Fair S, English AM, Holden SA,
Dick JR, Lonergan P and Kenny DA: Dietary polyunsaturated fatty
acid supplementation of young post-pubertal dairy bulls alters the
fatty acid composition of seminal plasma and spermatozoa but has no
effect on semen volume or sperm quality. Theriogenology.
90:289–300. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Prochowska S, Bonarska-Kujawa D, Bobak L,
Eberhardt M and Nizanski W: Fatty acid composition and biophysical
characteristics of the cell membrane of feline spermatozoa. Sci
Rep. 14:102142024. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Liu Q, Zhou YF, Duan RJ, Wei HK, Peng J
and Jiang SW: Dietary n-6:n-3 ratio and Vitamin E improve motility
characteristics in association with membrane properties of boar
spermatozoa. Asian J Androl. 19:223–229. 2017. View Article : Google Scholar :
|
|
130
|
Tran LV, Malla BA, Sharma AN, Kumar S,
Tyagi N and Tyagi AK: Effect of omega-3 and omega-6 polyunsaturated
fatty acid enriched diet on plasma IGF-1 and testosterone
concentration, puberty and semen quality in male buffalo. Anim
Reprod Sci. 173:63–72. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Schlegel RA, Hammerstedt R, Cofer GP,
Kozarsky K, Freidus D and Williamson P: Changes in the organization
of the lipid bilayer of the plasma membrane during spermatogenesis
and epididymal maturation. Biol Reprod. 34:379–391. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Jabarineitapeh M, Naderi N, Tavalaee M and
Nasr-Esfahani MH: Effects of L-carnitine over-supplementation on
spermatogenesis and sperm function in healthy NMRI mice. Tissue
Cell. 96:1030142025. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Casado ME, Huerta L, Marcos-Diaz A, Ortiz
AI, Kraemer FB, Lasunción MA, Busto R and Martín-Hidalgo A:
Hormone-sensitive lipase deficiency affects the expression of
SR-BI, LDLr, and ABCA1 receptors/transporters involved in cellular
cholesterol uptake and efflux and disturbs fertility in mouse
testis. Biochim Biophys Acta Mol Cell Biol Lipids. 1866:1590432021.
View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Casado ME, Huerta L, Ortiz AI,
Pérez-Crespo M, Gutiérrez-Adán A, Kraemer FB, Lasunción MÁ, Busto R
and Martín-Hidalgo A: HSL-knockout mouse testis exhibits class B
scavenger receptor upregulation and disrupted lipid raft
microdomains. J Lipid Res. 53:2586–2597. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Nistal M, Jimenez F and Paniagua R:
Sertoli cell types in the Sertoli-cell-only syndrome: Relationships
between Sertoli cell morphology and aetiology. Histopathology.
16:173–180. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Chung KW and Hamilton JB: Testicular
lipids in mice with testicular feminization. Cell Tissue Res.
160:69–80. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Weinbauer GF, Respondek M, Themann H and
Nieschlag E: Reversibility of long-term effects of GnRH agonist
administration on testicular histology and sperm production in the
nonhuman primate. J Androl. 8:319–329. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Wang T, Xiao Y, Hu Z, Gu J, Hua R, Hai Z,
Chen X, Zhang JV, Yu Z, Wu T, et al: MFN2 deficiency impairs
mitochondrial functions and PPAR pathway during spermatogenesis and
meiosis in mice. Front Cell Dev Biol. 10:8625062022. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Griswold MD: The central role of Sertoli
cells in spermatogenesis. Semin Cell Dev Biol. 9:411–416. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Walther TC and Farese RV Jr: Lipid
droplets and cellular lipid metabolism. Annu Rev Biochem.
81:687–714. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Hermo L, Pelletier RM, Cyr DG and Smith
CE: Surfing the wave, cycle, life history, and genes/proteins
expressed by testicular germ cells. Part 1: Background to
spermatogenesis, spermatogonia, and spermatocytes. Microsc Res
Tech. 73:241–278. 2010. View Article : Google Scholar
|
|
142
|
Singh R, Kaushik S, Wang Y, Xiang Y, Novak
I, Komatsu M, Tanaka K, Cuervo AM and Czaja MJ: Autophagy regulates
lipid metabolism. Nature. 458:1131–1135. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Rato L, Alves MG, Socorro S, Duarte AI,
Cavaco JE and Oliveira PF: Metabolic regulation is important for
spermatogenesis. Nat Rev Urol. 9:330–338. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Regueira M, Riera MF, Galardo MN, Camberos
Mdel C, Pellizzari EH, Cigorraga SB and Meroni SB: FSH and bFGF
regulate the expression of genes involved in Sertoli cell energetic
metabolism. Gen Comp Endocrinol. 222:124–133. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Shi JF, Li YK, Ren K, Xie YJ, Yin WD and
Mo ZC: Characterization of cholesterol metabolism in Sertoli cells
and spermatogenesis (Review). Mol Med Rep. 17:705–713. 2018.
|
|
146
|
Guma FC, Wagner M, Martini LH and Bernard
EA: Effect of FSH and insulin on lipogenesis in cultures of Sertoli
cells from immature rats. Braz J Med Biol Res. 30:591–597. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Stocco C: Aromatase expression in the
ovary: hormonal and molecular regulation. Steroids. 73:473–487.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Kraemer FB and Shen WJ: Hormone-sensitive
lipase: control of intracellular tri-(di-)acylglycerol and
cholesteryl ester hydrolysis. J Lipid Res. 43:1585–1594. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Miller WL and Auchus RJ: The molecular
biology, biochemistry, and physiology of human steroidogenesis and
its disorders. Endocr Rev. 32:81–151. 2011. View Article : Google Scholar
|
|
150
|
Wu LL, Dunning KR, Yang X, Russell DL,
Lane M, Norman RJ and Robker RL: High-fat diet causes lipotoxicity
responses in cumulus-oocyte complexes and decreased fertilization
rates. Endocrinology. 151:5438–5445. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Yuan X, Zhang X, Lin Y, Xie H, Wang Z, Hu
X, Hu S, Li L, Liu H, He H, et al: Proteome of granulosa cells
lipid droplets reveals mechanisms regulating lipid metabolism at
hierarchical and pre-hierarchical follicle in goose. Front Vet Sci.
12:15447182025. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Ibayashi M and Tsukamoto S: Lipid droplet
biogenesis in the ovary. Reprod Med Biol. 23:e126182024. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Gao S, Gan X, He H, Hu S, Deng Y, Chen X,
Li L, Hu J, Li L and Wang J: Dynamic characteristics of lipid
metabolism in cultured granulosa cells from geese follicles at
different developmental stages. Biosci Rep. 39:BSR201921882019.
View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Su YQ, Sugiura K and Eppig JJ: Mouse
oocyte control of granulosa cell development and function:
Paracrine regulation of cumulus cell metabolism. Semin Reprod Med.
27:32–42. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Kidder GM and Vanderhyden BC:
Bidirectional communication between oocytes and follicle cells:
Ensuring oocyte developmental competence. Can J Physiol Pharmacol.
88:399–413. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Robker RL, Wu LL and Yang X: Inflammatory
pathways linking obesity and ovarian dysfunction. J Reprod Immunol.
88:142–148. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Russell DL and Robker RL: Molecular
mechanisms of ovulation: Co-ordination through the cumulus complex.
Hum Reprod Update. 13:289–312. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Mau KHT, Karimlou D, Barneda D, Brochard
V, Royer C, Leeke B, de Souza RA, Pailles M, Percharde M, Srinivas
S, et al: Dynamic enlargement and mobilization of lipid droplets in
pluripotent cells coordinate morphogenesis during mouse
peri-implantation development. Nat Commun. 13:38612022. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Kikuchi K, Ekwall H, Tienthai P, Kawai Y,
Noguchi J, Kaneko H and Rodriguez-Martinez H: Morphological
features of lipid droplet transition during porcine oocyte
fertilisation and early embryonic development to blastocyst in vivo
and in vitro. Zygote. 10:355–366. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Watanabe T, Thayil A, Jesacher A, Grieve
K, Debarre D, Wilson T, Booth M and Srinivas S: Characterisation of
the dynamic behaviour of lipid droplets in the early mouse embryo
using adaptive harmonic generation microscopy. BMC Cell Biol.
11:382010. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Welte MA: As the fat flies: The dynamic
lipid droplets of Drosophila embryos. Biochim Biophys Acta.
1851:1156–1185. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Li Z, Thiel K, Thul PJ, Beller M, Kuhnlein
RP and Welte MA: Lipid droplets control the maternal histone supply
of Drosophila embryos. Curr Biol. 22:2104–2113. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Crocco MC, Kelmansky DM and Mariano MI:
Does serum cause lipid-droplet accumulation in bovine embryos
produced in vitro, during developmental days 1 to 4? J Assist
Reprod Genet. 30:1377–1388. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Abe H, Yamashita S, Satoh T and Hoshi H:
Accumulation of cytoplasmic lipid droplets in bovine embryos and
cryotolerance of embryos developed in different culture systems
using serum-free or serum-containing media. Mol Reprod Dev.
61:57–66. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
van der Weijden VA, Stotzel M, Iyer DP,
Fauler B, Gralinska E, Shahraz M, Meierhofer D, Vingron M, Rulands
S, Alexandrov T, et al: FOXO1-mediated lipid metabolism maintains
mammalian embryos in dormancy. Nat Cell Biol. 26:181–193. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Dunning KR, Cashman K, Russell DL,
Thompson JG, Norman RJ and Robker RL: Beta-oxidation is essential
for mouse oocyte developmental competence and early embryo
development. Biol Reprod. 83:909–918. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Herms A, Bosch M, Reddy BJ, Schieber NL,
Fajardo A, Rupérez C, Fernández-Vidal A, Ferguson C, Rentero C,
Tebar F, et al: AMPK activation promotes lipid droplet dispersion
on detyrosinated microtubules to increase mitochondrial fatty acid
oxidation. Nat Commun. 6:71762015. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Babayev E and Seli E: Oocyte mitochondrial
function and reproduction. Curr Opin Obstet Gynecol. 27:175–181.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
May-Panloup P, Boucret L, Chao de la Barca
JM, Desquiret-Dumas V, Ferré-L'Hotellier V, Morinière C, Descamps
P, Procaccio V and Reynier P: Ovarian ageing: The role of
mitochondria in oocytes and follicles. Hum Reprod Update.
22:725–743. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Steinhauser CB, Askelson K, Lambo CA,
Hobbs KC, Bazer FW and Satterfield MC: Lipid metabolism is altered
in maternal, placental, and fetal tissues of ewes with small for
gestational age fetusesdagger. Biol Reprod. 104:170–180. 2021.
View Article : Google Scholar :
|
|
171
|
Wu LL, Russell DL, Wong SL, Chen M, Tsai
TS, St John JC, Norman RJ, Febbraio MA, Carroll J and Robker RL:
Mitochondrial dysfunction in oocytes of obese mothers: Transmission
to offspring and reversal by pharmacological endoplasmic reticulum
stress inhibitors. Development. 142:681–691. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Naowarojna N, Wu TW, Pan Z, Li M, Han JR
and Zou Y: Dynamic regulation of ferroptosis by lipid metabolism.
Antioxid Redox Signal. 39:59–78. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Motamedi S, Ravoet N, Dehairs J,
Vanderhoydonc F, Escamilla-Ayala A, Sliwinska MA, Wang S, Idkowiak
J, Soenen S, Agostinis P, et al: AMP-activated protein
kinase-driven lipid droplet dynamics govern melanoma sensitivity to
polyunsaturated fatty acid and iron-induced ferroptosis. Nat
Commun. 16:112522025. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Lange M, Wolk M, Li VW, Doubravsky CE,
Hendricks JM, Kato S, Otoki Y, Styler B, Johnson SL, Harris CA, et
al: FSP1-mediated lipid droplet quality control prevents neutral
lipid peroxidation and ferroptosis. Nat Cell Biol. 27:1902–1913.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Danielli M, Perne L, Jarc Jovicic E and
Petan T: Lipid droplets and polyunsaturated fatty acid trafficking:
Balancing life and death. Front Cell Dev Biol. 11:11047252023.
View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Lee H, Horbath A, Kondiparthi L, Meena JK,
Lei G, Dasgupta S, Liu X, Zhuang L, Koppula P, Li M, et al: Cell
cycle arrest induces lipid droplet formation and confers
ferroptosis resistance. Nat Commun. 15:792024. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Obaseki E, Adebayo D, Bandyopadhyay S and
Hariri H: Lipid droplets and fatty acid-induced lipotoxicity: In a
nutshell. FEBS Lett. 598:1207–1214. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Chen Z, Ho IL, Soeung M, Yen EY, Liu J,
Yan L, Rose JL, Srinivasan S, Jiang S, Edward Chang Q, et al: Ether
phospholipids are required for mitochondrial reactive oxygen
species homeostasis. Nat Commun. 14:21942023. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Virmani A, Pinto L, Bauermann O, Zerelli
S, Diedenhofen A, Binienda ZK, Ali SF and van der Leij FR: The
carnitine palmitoyl transferase (CPT) system and possible relevance
for neuropsychiatric and neurological conditions. Mol Neurobiol.
52:826–836. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Qu Q, Zeng F, Liu X, Wang QJ and Deng F:
Fatty acid oxidation and carnitine palmitoyltransferase I: Emerging
therapeutic targets in cancer. Cell Death Dis. 7:e22262016.
View Article : Google Scholar : PubMed/NCBI
|
|
181
|
De Paula IF, Santos-Araujo S, Majerowicz
D, Ramos I and Gondim KC: Knockdown of carnitine
palmitoyltransferase I (CPT1) reduces fat body lipid mobilization
and resistance to starvation in the insect vector Rhodnius
prolixus. Front Physiol. 14:12016702023. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Choi J, Smith DM, Scafidi S, Riddle RC and
Wolfgang MJ: Carnitine palmitoyltransferase 1 facilitates fatty
acid oxidation in a non-cell-autonomous manner. Cell Rep.
43:1150062024. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Dissanayake LV, Smith BA, Zietara A,
Levchenko V, Lowe M, Kravtsova O, Shapiro A, Upadhyay G, Halade GV,
Geurts AM, et al: The role of carnitine palmitoyl transferase 2 in
the progression of salt-sensitive hypertension. Am J Physiol Cell
Physiol. 329:C1188–C1202. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Xiang F, Zhang Z, Xie J, Xiong S, Yang C,
Liao D, Xia B and Lin L: Comprehensive review of the expanding
roles of the carnitine pool in metabolic physiology: Beyond fatty
acid oxidation. J Transl Med. 23:3242025. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Liang K: Mitochondrial CPT1A: Insights
into structure, function, and basis for drug development. Front
Pharmacol. 14:11604402023. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Knottnerus SJG, Bleeker JC, Wust RCI,
Ferdinandusse S, IJ L 1st, Wijburg FA, Wanders RJA, Visser G and
Houtkooper RH: Disorders of mitochondrial long-chain fatty acid
oxidation and the carnitine shuttle. Rev Endocr Metab Disord.
19:93–106. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Reader KL, Cox NR, Stanton JA and Juengel
JL: Effects of acetyl-L-carnitine on lamb oocyte blastocyst rate,
ultrastructure, and mitochondrial DNA copy number. Theriogenology.
83:1484–1492. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Knitlova D, Hulinska P, Jeseta M,
Hanzalova K, Kempisty B and Machatkova M: Supplementation of
l-carnitine during in vitro maturation improves embryo development
from less competent bovine oocytes. Theriogenology. 102:16–22.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Kilwein MD, Dao TK and Welte MA:
Drosophila embryos allocate lipid droplets to specific lineages to
ensure punctual development and redox homeostasis. PLoS Genet.
19:e10108752023. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Bailey AP, Koster G, Guillermier C, Hirst
EM, MacRae JI, Lechene CP, Postle AD and Gould AP: Antioxidant role
for lipid droplets in a stem cell niche of Drosophila. Cell.
163:340–353. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Kim K, Park S and Roh S: Lipid-rich
blastomeres in the two-cell stage of porcine parthenotes show bias
toward contributing to the embryonic part. Anim Reprod Sci.
130:91–98. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Zhang J, Khvorostov I, Hong JS, Oktay Y,
Vergnes L, Nuebel E, Wahjudi PN, Setoguchi K, Wang G, Do A, et al:
UCP2 regulates energy metabolism and differentiation potential of
human pluripotent stem cells. EMBO J. 30:4860–4873. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Folmes CD, Nelson TJ, Martinez-Fernandez
A, Arrell DK, Lindor JZ, Dzeja PP, Ikeda Y, Perez-Terzic C and
Terzic A: Somatic oxidative bioenergetics transitions into
pluripotency-dependent glycolysis to facilitate nuclear
reprogramming. Cell Metab. 14:264–271. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Yanes O, Clark J, Wong DM, Patti GJ,
Sánchez-Ruiz A, Benton HP, Trauger SA, Desponts C, Ding S and
Siuzdak G: Metabolic oxidation regulates embryonic stem cell
differentiation. Nat Chem Biol. 6:411–417. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Shiraki N, Shiraki Y, Tsuyama T, Obata F,
Miura M, Nagae G, Aburatani H, Kume K, Endo F and Kume S:
Methionine metabolism regulates maintenance and differentiation of
human pluripotent stem cells. Cell Metab. 19:780–794. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Dunning KR, Anastasi MR, Zhang VJ, Russell
DL and Robker RL: Regulation of fatty acid oxidation in mouse
cumulus-oocyte complexes during maturation and modulation by PPAR
agonists. PLoS One. 9:e873272014. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Brasaemle DL: Thematic review series:
Adipocyte biology. The perilipin family of structural lipid droplet
proteins: Stabilization of lipid droplets and control of lipolysis.
J Lipid Res. 48:2547–2559. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Horton JD, Goldstein JL and Brown MS:
SREBPs: Activators of the complete program of cholesterol and fatty
acid synthesis in the liver. J Clin Invest. 109:1125–1131. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Fan W and Evans R: PPARs and ERRs:
Molecular mediators of mitochondrial metabolism. Curr Opin Cell
Biol. 33:49–54. 2015. View Article : Google Scholar
|
|
200
|
Sayers NS, Anujan P, Yu HN, Palmer SS,
Nautiyal J, Franks S and Hanyaloglu AC: Follicle-stimulating
hormone induces lipid droplets via Galphai/o and β-Arrestin in an
endometrial cancer cell line. Front Endocrinol (Lausanne).
12:7988662021. View Article : Google Scholar
|
|
201
|
Igosheva N, Abramov AY, Poston L, Eckert
JJ, Fleming TP, Duchen MR and McConnell J: Maternal diet-induced
obesity alters mitochondrial activity and redox status in mouse
oocytes and zygotes. PLoS One. 5:e100742010. View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Shen WJ, Patel S, Natu V, Hong R, Wang J,
Azhar S and Kraemer FB: Interaction of hormone-sensitive lipase
with steroidogenic acute regulatory protein: Facilitation of
cholesterol transfer in adrenal. J Biol Chem. 278:43870–43876.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Ludovic, Le, Corre, et al: Bisphenol A
disrupts the intestinal lipid metabolism. Toxicology Letters.
211:S2102012. View Article : Google Scholar
|
|
204
|
Vit FF, Sangalli JR, Fiorenza MF, de
Almeida Saraiva HFR, Ferst JG, Perecin F, Meirelles FV, de la Torre
LG and da Silveira JC: Dynamic microdevice culture during bovine
oocyte maturation decreases lipid accumulation and improve
blastocyst cell numbers. Sci Rep. 16:2322025. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Zhu J, Lam SM, Yang L, Liang J, Ding M,
Shui G and Huang X: Reduced phosphatidylcholine synthesis
suppresses the embryonic lethality of seipin deficiency. Life
Metab. 1:175–189. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Dumesic DA, Padmanabhan V and Abbott DH:
Polycystic ovary syndrome and oocyte developmental competence.
Obstet Gynecol Surv. 63:39–48. 2008. View Article : Google Scholar
|
|
207
|
Sturmey RG and Leese HJ: Energy metabolism
in pig oocytes and early embryos. Reproduction. 126:197–204. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Horn PJ, Ledbetter NR, James CN, Hoffman
WD, Case CR, Verbeck GF and Chapman KD: Visualization of lipid
droplet composition by direct organelle mass spectrometry. J Biol
Chem. 286:3298–3306. 2011. View Article : Google Scholar
|
|
209
|
Ding Y, Jiang Y, Zhu M, Zhu Q, He Y, Lu Y,
Wang Y, Qi J, Feng Y, Huang R, et al: Follicular fluid lipidomic
profiling reveals potential biomarkers of polycystic ovary
syndrome: A pilot study. Front Endocrinol (Lausanne).
13:9602742022. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Zhang M, Wang Y, Di J, Zhang X, Liu Y,
Zhang Y, Li B, Qi S, Cao X, Liu L, et al: High coverage of targeted
lipidomics revealed lipid changes in the follicular fluid of
patients with insulin-resistant polycystic ovary syndrome and a
positive correlation between plasmalogens and oocyte quality. Front
Endocrinol (Lausanne). 15:14142892024. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Ruebel ML, Piccolo BD, Mercer KE, Pack L,
Moutos D, Shankar K and Andres A: Obesity leads to distinct
metabolomic signatures in follicular fluid of women undergoing in
vitro fertilization. Am J Physiol Endocrinol Metab. 316:E383–E396.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Gao J, Liu M, Liu J, Shi P, Cui H, Zhao S,
Zhang X and Tao C: Effect of high-fat diet on the lipid profile of
ovarian granulosa cells and female reproduction in mice. PLoS One.
18:e02875342023. View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Cao Z, Hao Y, Fung CW, Lee YY, Wang P, Li
X, Xie K, Lam WJ, Qiu Y, Tang BZ, et al: Dietary fatty acids
promote lipid droplet diversity through seipin enrichment in an ER
subdomain. Nat Commun. 10:29022019. View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Schmiedel JM, Klemm SL, Zheng Y, Sahay A,
Blüthgen N, Marks DS and van Oudenaarden A: Gene expression.
MicroRNA control of protein expression noise. Science. 348:128–132.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Mak HY: Lipid droplets as fat storage
organelles in Caenorhabditis elegans: Thematic review series: Lipid
droplet synthesis and metabolism: From Yeast To Man. J Lipid Res.
53:28–33. 2012. View Article : Google Scholar
|
|
216
|
Kumari RM, Khatri A, Chaudhary R and
Choudhary V: Concept of lipid droplet biogenesis. Eur J Cell Biol.
102:1513622023. View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Meister P, Towbin BD, Pike BL, Ponti A and
Gasser SM: The spatial dynamics of tissue-specific promoters during
C. elegans development. Genes Dev. 24:766–782. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Frezal L and Felix MA: C. elegans outside
the Petri dish. Elife. 4:e058492015. View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Schmokel V, Memar N, Wiekenberg A,
Trotzmuller M, Schnabel R and Doring F: Genetics of lipid-storage
management in caenorhabditis elegans embryos. Genetics.
202:1071–1083. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Vrablik TL, Petyuk VA, Larson EM, Smith RD
and Watts JL: Lipidomic and proteomic analysis of Caenorhabditis
elegans lipid droplets and identification of ACS-4 as a lipid
droplet-associated protein. Biochim Biophys Acta. 1851:1337–1345.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Liu Y, Xu S, Zhang C, Zhu X, Hammad MA,
Zhang X, Christian M, Zhang H and Liu P: Hydroxysteroid
dehydrogenase family proteins on lipid droplets through bacteria,
C. elegans, and mammals. Biochim Biophys Acta Mol Cell Biol Lipids.
1863:881–894. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Mondal M, Scifo E, Ciliberti RE, Wischhof
L, Abbariki TN, Jackson J, Menegatou IM, Zeisler-Diehl V, Riemer J,
Jussila B, et al: Dietary lipid content modifies
wah-1/AIFM1-associated phenotypes via LRK-1 and DRP-1 expression in
C. elegans. Nat Commun. 16:108172025. View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Campbell D and Zuryn S: The mechanisms and
roles of mitochondrial dynamics in C. elegans. Semin Cell Dev Biol.
156:266–275. 2024. View Article : Google Scholar
|
|
224
|
Mullaney BC and Ashrafi K: C. elegans fat
storage and metabolic regulation. Biochim Biophys Acta.
1791:474–478. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Lynn DA, Dalton HM, Sowa JN, Wang MC,
Soukas AA and Curran SP: Omega-3 and -6 fatty acids allocate
somatic and germline lipids to ensure fitness during nutrient and
oxidative stress in Caenorhabditis elegans. Proc Natl Acad Sci USA.
112:15378–15383. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Li Q, Zhou X, Zhang X, Zhang C and Zhang
SO: Nuclear receptor signaling regulates compartmentalized
phosphatidylcholine remodeling to facilitate thermosensitive lipid
droplet fusion. Nat Commun. 16:39552025. View Article : Google Scholar : PubMed/NCBI
|