Introduction
Lipid droplets (LDs), typically considered inert
reservoirs of neutral lipids, are recognized as dynamic and
metabolically active organelles central to cell energy balance,
lipid metabolism, proteostasis and stress adaptation (1,2).
Structurally, LDs consist of a hydrophobic core enriched in
triacylglycerols (TAGs) and cholesteryl esters (CEs), encased by a
phospholipid monolayer that is uniquely enriched in proteins such
as perilipin (PLIN), lipase and endoplasmic reticulum
(ER)-associated scaffolding proteins (3). The formation of LDs begins at
specific subdomains of the ER, where neutral lipid phase separation
is initiated and nucleated by key regulatory proteins including
seipin (SEIP-1) (4,5).
While extensively studied in metabolically active
tissue such as adipocytes and hepatocytes, LDs have garnered
growing attention in non-adipose cells where their functions extend
beyond energy storage (2,6,7).
In the central nervous system, immune cells and particularly in
reproductive cells, LDs regulate cell signaling, redox homeostasis,
steroidogenesis and embryonic morphogenesis (8-10). Reproductive cells exhibit notable
plasticity in energy demand and metabolic activity, particularly
during oocyte maturation, fertilization and preimplantation embryo
development, periods marked by high biosynthetic and proliferative
requirements.
Oocytes accumulate substantial quantities of LDs
during proliferation and maturation, forming lipid-rich stores that
support the metabolic needs of the embryo during early cleavage
divisions (11,12). In lipid-rich species such as
pigs, cows and humans, this cytoplasmic lipid reserve provides
energy via β-oxidation and supports membrane biogenesis and redox
buffering (13). Dysregulation
of LD content in oocytes, either excessive or deficient, is linked
to impaired maturation, fertilization failure and reduced
developmental competence (14).
By contrast, mature spermatozoa are devoid of
prominent LDs, but their development is dependent on lipid
mobilization and LD dynamics in precursor germ and Sertoli cells
(15). Sertoli cells exhibit
cyclic changes in LD number and composition, reflecting their roles
in nutrient provision, phagocytosis of residual bodies and energy
buffering during spermatogenesis (16,17). Testicular Leydig cells also use
LDs as platforms for storing and mobilizing cholesterol esters in
response to luteinizing hormone (LH) stimulation, enabling rapid
testosterone synthesis (18,19).
Granulosa and luteal cells in the ovary similarly
use LDs for estrogen and progesterone production (20). These cells respond to
follicle-stimulating hormone (FSH) or LH by activating
hormone-sensitive lipase (HSL) to hydrolyze CE within LDs,
liberating free cholesterol for steroidogenesis within mitochondria
(21,22). Proteomic studies have identified
key steroidogenic enzymes, such as CYP11A1 and 3β-hydroxysteroid
dehydrogenase, localizing to LDs in these cells, suggesting that
LDs not only serve as storage platforms but also scaffold sites for
enzymatic reactions (20,23,24).
The nematode Caenorhabditis elegans is a
powerful genetic model for studying LD function in reproduction.
Its transparency, short generation time and well-mapped
reproductive system enable in vivo tracking of LD dynamics
during gametogenesis and embryogenesis (25). In C. elegans, SEIP-1
regulates a subpopulation of LDs that contribute to lipid layer
assembly in the embryonic eggshell, which is key for embryo
viability (25). Mutants lacking
SEIP-1 exhibit disrupted permeability barriers and embryonic
lethality, phenotypes that can be partially rescued by modulating
PLIN-1 or Ras-related protein Rab-18 function, revealing parallel
compensatory pathways (25,26).
Despite these advances, major questions remain
unresolved. The temporal coordination of LD biogenesis and
degradation during fertilization or implantation requires further
elucidation. Molecular cues that determine LD targeting by
lipophagy in reproductive cells remain to be identified. LD
imbalance contributes to reproductive pathologies such as
infertility or polycystic ovary syndrome (PCOS) (27-30), however the mechanisms that
warrant deeper exploration. Understanding these questions is key
given the metabolic sensitivity of reproductive cells and their
susceptibility to lipid imbalance.
Coherent anti-Stokes Raman scattering (CARS) and
stimulated Raman scattering (SRS) microscopy enable label-free,
real-time imaging of LD dynamics in living oocytes and embryos
(31,32). Lipidomics and metabolomics, even
at single-embryo resolution, have uncovered lipid profile shifts
associated with in vitro maturation or developmental arrest
(33). Genome editing tools such
as clustered regularly interspaced short palindromic repeats
(CRISPR)-CRISPR-associated protein 9 (CRISPR-Cas9) and RNA
interference (RNAi) screens in C. elegans or mammalian
systems have revealed novel regulators of LD size, turnover and
localization (34,35). Additionally, single-cell
transcriptomics has revealed cell type-specific expression of
LD-associated genes [PLIN2, Diacylglycerol O-acyltransferase DGAT2,
adipose triglyceride lipase (ATGL) across the testis, ovary and
early embryo (36).
LD dysfunction is increasingly linked to
reproductive disorder (28,37). Obesity and metabolic syndrome
alter lipid composition and increase oxidative stress in oocytes
and sperm, decreasing fertility and embryo quality (38,39). In PCOS, altered lipid metabolism
in granulosa cells impairs steroid hormone production, oocyte
competence and follicular development (30). Genetic disorders affecting LD
regulators, such as mutations in SEIP-1, the protein encoded by
Berardinelli-Seip congenital lipodystrophy 2, in congenital
lipodystrophy, typically involve hypogonadism and infertility
(40).
The present review aimed to summarize the roles of
LDs in reproduction across species and cell types, including
oocytes, sperm and early embryos, and how supporting somatic
Sertoli, Leydig, granulosa and luteal cells use LDs for metabolic
coordination and hormonal output, as well as findings from C.
elegans that uncover conserved regulatory mechanisms. The
present review aimed to highlight key proteins such as ATGL, HSL,
PLIN, DGATs, SEIP-1 and lipophagy-associated factors and assess how
cutting-edge technologies are advancing the study of LD biology in
reproductive physiology and how LD dysfunction contributes to
reproductive disease.
Biological landscape and core functions of
LDs in reproduction
Lipid availability in reproductive systems is
heterogeneous, varying across cell types, developmental stages, and
species. Oocytes, particularly in lipid-rich species such as pigs,
cows and humans, accumulate abundant LDs during growth, whereas
mature spermatozoa contain few if any visible lipid stores. By
contrast, reproductive somatic cells, including Sertoli, Leydig,
granulosa, and luteal cells, display dynamic LD populations that
fluctuate in response to developmental cues and hormonal
stimulation (24,41-43).
Beyond spatial heterogeneity, LDs also exhibit
notable temporal dynamics. Their abundance increases during oocyte
maturation and steroidogenic activation, is remodeled following
fertilization and progressively declines during early embryonic
development as stored lipids are mobilized (37,41,44). These spatial and temporal
patterns establish the foundational context in which LDs serve not
only as energy reserves, but as platforms that support metabolic
coordination and signaling processes (Fig. 1).
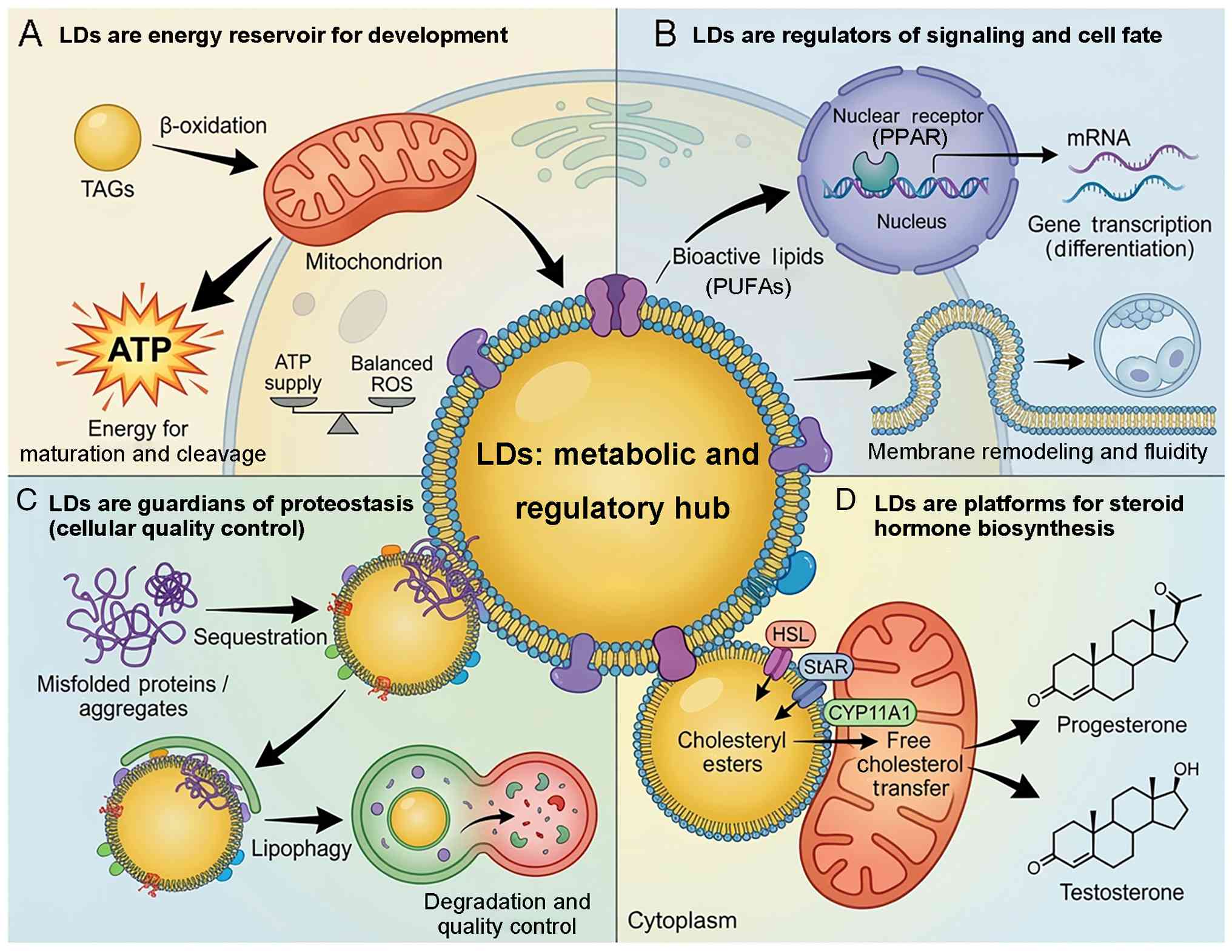 | Figure 1Roles of LDs as metabolic and
regulatory hubs in reproductive cells. (A) LDs are energy reservoir
for development. LDs store TAGs that are mobilized via lipolysis to
provide fatty acids for mitochondrial β-oxidation. This generates
ATP, which is key for oocyte maturation, fertilization and early
cleavage, while maintaining a balanced redox state (low ROS), which
is critical for embryo viability. (B) LDs are regulators of
signaling and cell fate. LDs release bioactive lipids (such as
PUFAs) that act as signaling ligands for nuclear receptors (such as
PPARs) to drive gene transcription and differentiation. LD-derived
lipids contribute to membrane remodeling, influencing fluidity and
morphogenetic events during cell division. (C) LDs are guardians of
proteostasis (cell quality control). LDs function as sequestration
sites for misfolded or aggregated proteins, acting as transient
detoxification zones. These protein-laden LDs are cleared via
lipophagy (autophagic degradation) or lysosomal pathways, which is
key for maintaining proteostasis during high-stress periods such as
spermatogenesis. (D) LDs are platforms for steroid hormone
biosynthesis. In steroidogenic cells (Leydig, granulosa and luteal
cells), LDs store cholesteryl esters. Following hormonal
stimulation, these esters are hydrolyzed to free cholesterol, which
is transported to mitochondria (facilitated by proteins such as
StAR and HSL at the LD-mitochondria interface) to serve as the
substrate for the synthesis of steroid hormones such as
progesterone and testosterone. HSL, hormone-sensitive lipase; LD,
lipid droplet; PPAR, peroxisome proliferator-activated receptor;
PUFA, polyunsaturated fatty acid; ROS, reactive oxygen species;
StAR, steroidogenic acute regulatory protein; TAG,
triacylglycerol. |
Spatial distribution of lipids and LDs in
reproductive systems
Lipid availability in reproductive systems exhibits
marked heterogeneity, with distinct patterns across germ and
somatic cells as well as species-specific adaptations. For
instance, while oocytes in lipid-rich species like pigs, cows, and
humans amass numerous cytoplasmic LDs that confer opacity and fuel
early embryogenesis through β-oxidation and membrane synthesis,
murine oocytes maintain lower lipid reserves, highlighting
metabolic divergences (9-12).
In somatic support cells, LD profiles are tailored to functional
demands: Sertoli cells show cyclic LD accumulation tied to
phagocytosis and spermatogenic cycles (15-17), whereas granulosa and luteal cells
prioritize cholesteryl ester storage for sustained steroidogenesis
under hormonal regulation (20-22).
Temporal dynamics of LDs across
reproductive stages
In addition to spatial heterogeneity, LDs exhibit
notable temporal regulation throughout reproductive processes.
During oocyte growth, LD content increases as neutral lipids are
synthesized or imported and stored in preparation for fertilization
and early embryonic development (11,12). Following fertilization,
maternally inherited LDs undergo redistribution and partial
consumption during cleavage divisions, reflecting a shift from
lipid storage to utilization (13,31).
Similar temporal patterns are evident in
reproductive somatic cells: In Sertoli cells, LD abundance
fluctuates across the spermatogenic cycle, increasing during
periods of active germ cell turnover and phagocytosis of residual
bodies (16,17,45,46). In granulosa cells, LD
accumulation intensifies during follicular maturation and peaks
following luteinization, coinciding with maximal steroidogenic
activity (24,47,48). These dynamic changes indicate
that LDs are not static lipid depots but responsive organelles
whose formation and turnover are associated with developmental
timing and hormonal cues.
Metabolic use of stored lipids in
reproductive cells
Once accumulated, lipids stored within LDs serve as
key metabolic substrates. Triacylglycerols can be hydrolyzed to
release fatty acids (FAs) that fuel mitochondrial β-oxidation,
providing ATP during energetically demanding processes such as
oocyte maturation, early embryonic cleavage and spermatogenic
support by Sertoli cells (11,13,49). In parallel, LD-derived lipids
contribute to membrane biogenesis, ensuring sufficient phospholipid
supply during rapid cell division and cellular remodeling (50).
The balance between lipid storage and mobilization
is regulated. Excessive lipid accumulation leads to lipotoxicity
and oxidative stress, whereas insufficient lipid reserves
compromise energy availability and developmental competence
(14,51). Reproductive cells therefore rely
on coordinated control of lipid synthesis, lipolysis and oxidation
to maintain metabolic homeostasis across fluctuating physiological
demands (49,50).
LDs as regulatory and signaling hubs
Beyond their metabolic roles, LDs integrate lipid
metabolism with signaling and regulatory pathways. The controlled
release of bioactive lipid species from LDs influences nuclear
receptor activation, including peroxisome proliferator-activated
receptors (PPARs), thereby modulating transcriptional programs
associated with cell differentiation and developmental progression
(52,53). In steroidogenic cells, LDs store
cholesteryl esters that are rapidly mobilized in response to
gonadotropic stimulation, coupling lipid storage directly to
hormone biosynthesis (20-22).
LDs also engage in notable physical and functional
interactions with other organelles. Increasing evidence supports
the existence of membrane contact sites between LDs and
mitochondria or the ER, enabling efficient lipid transfer,
metabolic channeling and coordination of redox homeostasis
(54,55). Ultrastructural evidence supports
the concept of LDs as metabolic hubs: Electron microscopy studies
have revealed tight membrane contact sites between LDs and
mitochondria or the ER, indicating that these organelles are
physically connected rather than randomly juxtaposed (54-57). Such contacts are hypothesized to
facilitate efficient lipid transfer, metabolic channeling and
coordinated regulation of energy production and lipid metabolism,
providing a structural basis for the functional interactions
illustrated in schematic models (58,59). Through these interactions, LDs
serve not merely as passive reservoirs but as dynamic hubs that
synchronize energy metabolism, signaling and cell adaptation during
reproduction (2,10).
Accumulating evidence indicates that the
LD-mitochondria interface represents a physically tethered unit
rather than a transient or stochastic association (58,60). Ultrastructural analyses have
revealed well-defined membrane contact sites that anchor LDs to
mitochondria, thereby establishing stable platforms for lipid
transfer and metabolic coordination (54,55,61,62). These contact sites are mediated
by specific tethering proteins that physically link the organelles.
Among these, PLIN5 is a LD-associated protein that promotes
sustained LD-mitochondria coupling and facilitates the channeling
of FAs from LDs to mitochondria for β-oxidation. In parallel,
mitoguardin 2, a mitochondrial outer membrane protein, forms
physical bridges between mitochondria and LDs, coordinating lipid
trafficking and mitochondrial energy metabolism (54,63,64). Together, these tethering
mechanisms support a model in which LDs and mitochondria serve as
integrated metabolic units, providing a structural and molecular
basis for the metabolic hub concept in reproductive cells.
The diverse and sometimes contradictory functions of
LDs can be reconciled by viewing them as organelles with multiphase
activities that are dynamically regulated across developmental and
physiological contexts. In a storage phase, LDs primarily
accumulate neutral lipids, serving as reservoirs that buffer energy
availability and protect cells from lipid overload. During
metabolic phases, LDs undergo controlled lipolysis, releasing FAs
that fuel mitochondrial β-oxidation and support membrane
biosynthesis. In signaling phases, LD-derived lipid species serve
as bioactive molecules that engage nuclear receptors, such as
PPARs, thereby influencing transcriptional programs associated with
cell fate decisions and developmental progression (2,60,65).
The transition between these phases is not fixed but
context-dependent, shaped by developmental timing, hormonal cues
and cellular energy demands. This multiphase framework provides a
conceptual basis for understanding how the same LD population
alternately serves as a protective storage depot, a metabolic fuel
source or a signaling platform during reproduction (2,41,48,66).
LDs in oocyte maturation and competence
The oocyte is a metabolically unique cell,
characterized by large size, prolonged growth phase and dependence
on stored reserves to sustain early embryogenesis. Among these
reserves, LDs serve a key role in determining oocyte quality and
developmental competence (37,41,49). While their role as energy depots
is well-established, evidence highlights a broader regulatory role
for LDs in shaping the oocyte redox balance, signaling landscape
and cytoplasmic remodeling capacity, which are indispensable for
meiotic progression and post-fertilization events (67-69) (Fig. 2).
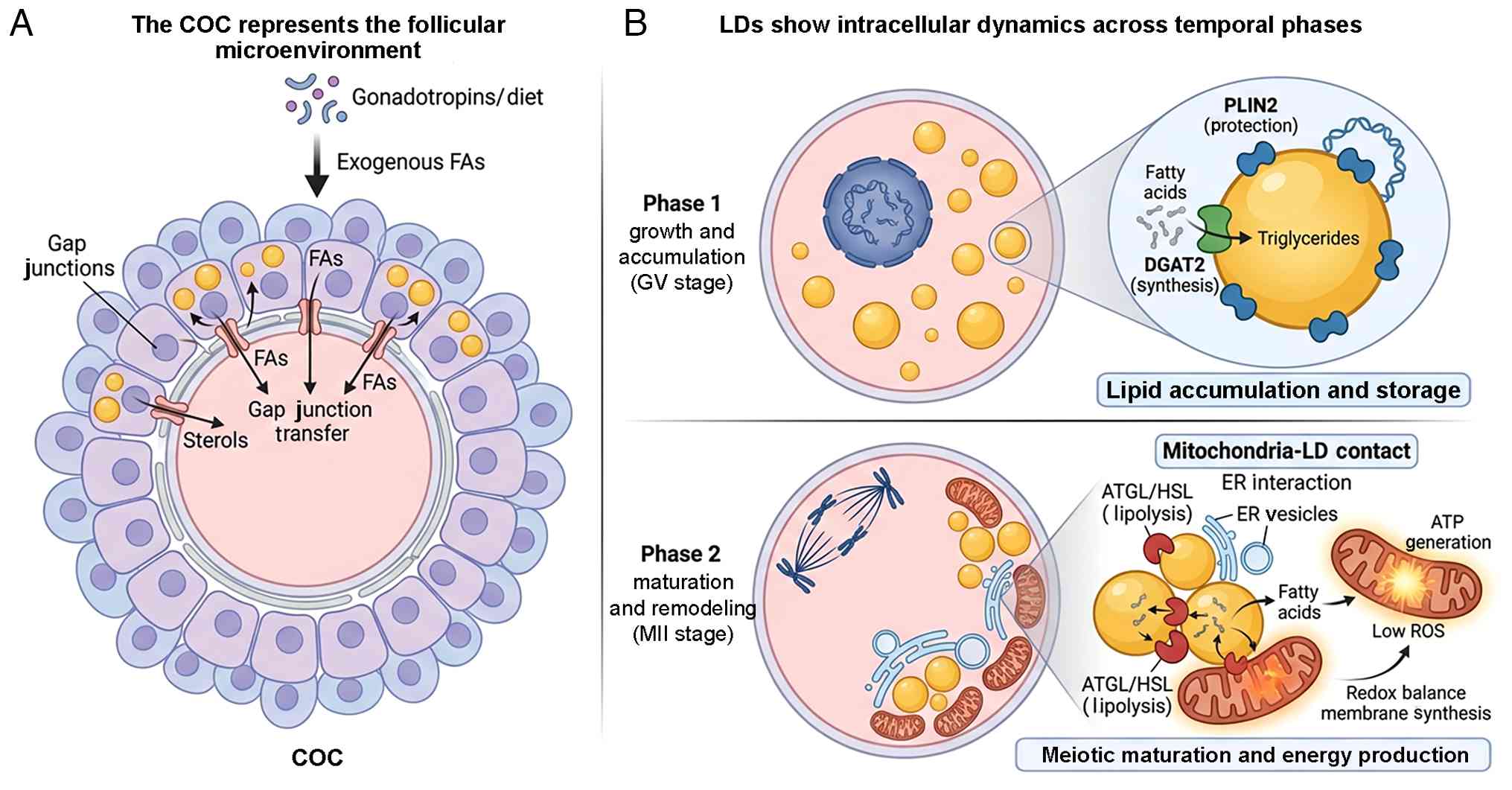 | Figure 2LD dynamics and regulation during
oocyte maturation. (A) COC (the follicular microenvironment). The
oocyte is surrounded by cumulus cells, forming the COC. Cumulus
cells modulate the oocyte lipid profile by transferring FAs and
sterols through gap junctions. This supply chain is influenced by
external factors such as gonadotropins and dietary intake
(exogenous FAs). Cumulus cells maintain their own LD reserves to
support this metabolic coupling. (B) LDs show intracellular
dynamics across temporal phases. During the GV stage, the oocyte
actively synthesizes and stores lipids. DGAT2 catalyzes
triacylglycerol synthesis and PLIN2 facilitates coating of the LD
surface to promote stability and prevent premature lipolysis,
resulting in the accumulation of dispersed LDs. Following meiotic
resumption (MII transition), LDs undergo spatial reorganization and
functional activation. At mitochondria-LD contact sites, lipolytic
enzymes (ATGL/HSL) mobilize stored lipids. The released FAs are
directed into mitochondria for β-oxidation, generating ATP while
maintaining redox balance (low ROS) and supporting membrane
synthesis essential for meiotic progression and fertilization
competence. COC, cumulus-oocyte complex; DGAT2, diacylglycerol
acyltransferase 2; ER, endoplasmic reticulum; GV, germinal vesicle;
HSL, hormone-sensitive lipase; MII, metaphase II; PLIN2, perilipin
2; ROS, reactive oxygen species; FA, fatty acid; LD, lipid droplet;
ATGL, adipose triglyceride lipase. |
LD accumulation is temporally regulated
during oocyte growth
LD biogenesis in oocytes is a tightly orchestrated
process that coincides with folliculogenesis. Throughout the
growing phase, oocytes accumulate neutral lipids via both de
novo synthesis and uptake of exogenous FAs, which are
esterified and stored in LDs (37,68). During oocyte growth, LDs
primarily operate in a storage phase, ensuring sufficient lipid
reserves for subsequent developmental transitions. In large antral
follicles, LDs become prominent cytoplasmic features (68). Their abundance and distribution
vary across species: Porcine and bovine oocytes are lipid-rich and
visibly opaque, whereas murine and human oocytes have fewer LDs and
a clearer cytoplasm (41,42,70,71).
These differences reflect fundamental differences in lipid
metabolism, sensitivity to in vitro conditions and
developmental strategies (49,72). For example, lipid-rich oocytes in
porcine and bovine species rely heavily on β-oxidation of stored
lipids for energy during early embryogenesis, as shown by reduced
developmental rates when β-oxidation inhibitors are applied in
culture (11). In contrast,
murine oocytes exhibit greater dependence on glycolysis, making
them less sensitive to lipid perturbations but more vulnerable to
glucose fluctuations in vitro (73). Human oocytes, while
lipid-moderate, show intermediate sensitivity, with in vitro
maturation success influenced by media supplements that mitigate
oxidative stress from lipid peroxidation (74). These variations underscore
species-specific reproductive adaptations, where lipid-rich
strategies buffer against nutrient scarcity post-fertilization,
whereas lipid-poor ones prioritize rapid external nutrient uptake
(49).
Oocyte capacity to accumulate and mobilize LDs is
associated with the ability to resume meiosis and support embryo
development (72,75). Disruptions in LD formation,
either through inhibition of DGAT1/2 or alterations in FA
composition, impair nuclear maturation and decrease blastocyst
yield, underscoring the role of LDs in establishing developmental
competence (49,69,76).
LD dynamics and cytoplasmic
remodeling
LDs undergo dynamic spatial reorganization during
meiotic maturation. In many species, LDs are dispersed throughout
the oocyte cytoplasm at the germinal vesicle (GV) stage, then
undergo clustering or partial consumption during GV breakdown and
metaphase II transition (31,37,49,77). These changes may reflect a
metabolic switch: As the oocyte transitions from quiescence to a
highly active biosynthetic state, lipid oxidation increases,
supported by mitochondrial redistribution and enhanced FA flux
(67,68).
Moreover, LD remodeling is typically coordinated
with organelle positioning. In mammalian oocytes, LDs have been
observed in proximity to mitochondria and ER-derived vesicles,
suggesting metabolic crosstalk and potential transfer of lipid
species (54,55,61,62). These interactions may be key for
shaping mitochondrial function, as lipid overload or
misdistribution is associated with increased ROS production and
mitochondrial dysfunction, which are detrimental to fertilization
and embryo cleavage (63,64,71).
Regulatory mechanisms: Enzymes and
LD-coating proteins
The functional integrity of LDs in oocytes is
governed by a tightly regulated network of enzymes and structural
proteins (37,69). DGAT2, which catalyzes TAG
synthesis, is enriched in growing oocytes, and its inhibition leads
to decreased lipid storage and impaired oocyte maturation (69,76). Similarly, the LD surface protein
PLIN2 is abundantly expressed in lipid-rich oocytes, and is
hypothesized to stabilize LDs by preventing premature lipolysis
(78-80). Knockdown or pharmacological
interference with PLIN2 results in dysregulated lipid metabolism
and altered oocyte developmental trajectories (69,81).
In addition to synthesis and stabilization, lipid
mobilization is precisely timed. Lipolytic enzymes such as ATGL and
HSL are activated in peri-ovulatory periods, allowing controlled
release of FAs for β-oxidation and membrane synthesis (82-85). An imbalance in this process,
either via excessive lipid accumulation or hyperactive lipolysis,
compromises oocyte viability (86).
Paracrine influences and somatic-oocyte
interaction
LD content and composition in the oocyte are not
solely determined by intrinsic metabolic programs. Surrounding
cumulus and granulosa cells contribute to the oocyte lipid profile
through paracrine signaling and metabolite transfer (41,87,88). Cumulus-oocyte complexes exhibit
extensive gap junction communication, allowing transfer of small
lipophilic molecules such as FAs and sterols (89-92). Cumulus cells express lipoprotein
receptors, FA transporters and lipogenic enzymes, and can modulate
the lipid environment of the oocyte in response to gonadotropic
stimulation or dietary lipid availability (41,91,93).
Alterations in cumulus cell metabolism, such as
those seen in high-fat diet models or PCOS, lead to excessive lipid
accumulation in oocytes and are associated with lower fertilization
and blastocyst rates (71,77,93). These findings emphasize that LD
metabolism in the oocyte must be understood in the context of the
follicular microenvironment (41,87,88).
LD metabolism as a marker and modulator
of oocyte quality
Because LD content reflects metabolic history and
readiness for fertilization, it is increasingly studied as a
biomarker of oocyte competence (31,94,95). Non-invasive imaging modalities,
such as CARS microscopy, have made it possible to quantify LD
content in live oocytes and demonstrate its association with
subsequent embryo development (31,96-99). Moreover, interventions aimed at
modifying LD metabolism through culture media supplementation with
oleic acid or antioxidants have shown potential to rescue
poor-quality oocytes by rebalancing lipid profiles (96-99).
Altogether, LDs in oocytes serve as more than static
energy stores; they are dynamic organelles whose content,
composition and spatial behavior are critical determinants of
oocyte maturation and developmental success (37,69). Their regulation is
multifactorial, involving intrinsic enzyme systems, extrinsic
paracrine input and inter-organelle coordination. Understanding and
manipulating LD biology in the oocyte may thus represent a
promising avenue for improving assisted reproduction outcomes
(96,100,101).
LDs in spermatogenesis and sperm
function
Spermatogenesis is a complex, highly regulated
process that requires precise coordination of energy metabolism,
membrane remodeling and quality control. While mature spermatozoa
lack prominent LDs, increasing evidence suggests that LDs play key
upstream roles during the earlier stages of germ cell development
and in the metabolic crosstalk between developing spermatogenic
cells and their supporting Sertoli cells (15,43,102) (Fig. 3).
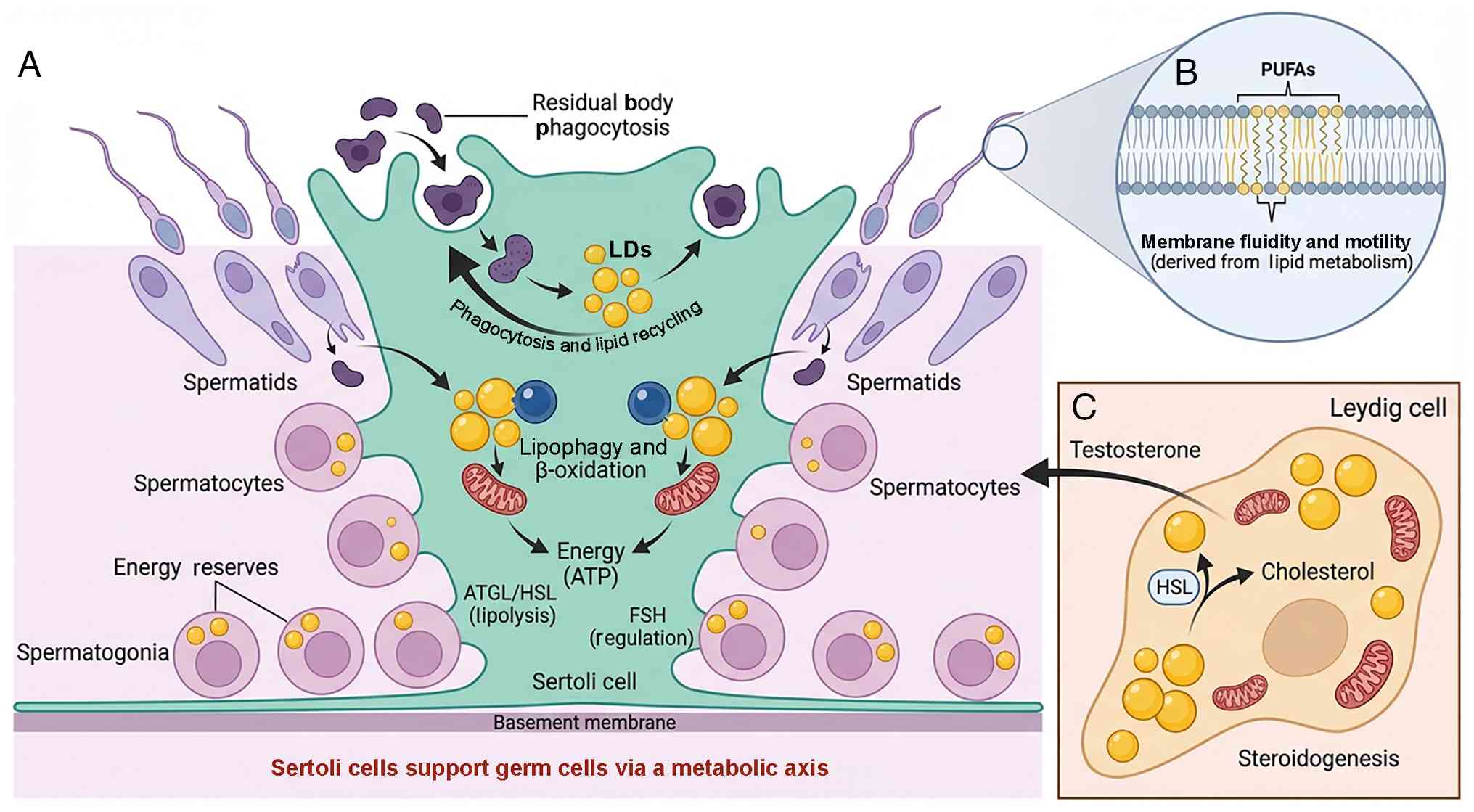 | Figure 3LD dynamics and metabolic crosstalk
in the testis. (A) Sertoli cells support germ cells via a metabolic
axis. Within the seminiferous tubule, Sertoli cells support the
developing germ cells. As spermatids elongate, they shed excess
cytoplasm as residual bodies, which are phagocytosed by Sertoli
cells. These lipid-rich remnants are sequestered into LDs
(phagocytosis and lipid recycling). Stored LDs in Sertoli cells are
catabolized via lipophagy (lysosomal degradation) and mitochondrial
β-oxidation to generate ATP, fueling the high energy demands of
spermatogenesis. Early germ cells (spermatogonia/cytes) contain
transient LDs as energy reserves, which decline as cells
differentiate into mature spermatozoa. (B) Sperm undergo membrane
remodeling. Although mature spermatozoa lack LDs, their plasma
membranes are enriched with PUFAs derived from upstream lipid
metabolism. Integration of PUFAs into the sperm membrane is a
critical factor for maintaining membrane fluidity, motility and
fertilization capacity. FSH, follicle-stimulating hormone; HSL,
hormone-sensitive lipase; LD, lipid droplet; PUFA, polyunsaturated
fatty acid. (C) Leydig cells regulate interstitial processes. In
the interstitial space, Leydig cells use LDs as reservoirs for
cholesteryl esters. Under the regulation of LH, HSL mobilizes
cholesterol from these LDs to synthesize Testosterone, which is key
for maintaining spermatogenesis. |
LDs in early germ cells and spermatogenic
progression
During the early phases of spermatogenesis,
including in spermatogonia and early spermatocytes, LDs are readily
observed and serve as temporary energy reserves and platforms for
lipid remodeling (15,102,103). As germ cells differentiate
toward the elongated spermatid stage, LD content typically
declines, which is associated with cytoplasmic condensation and
organelle removal (15,102). However, disturbances in lipid
storage and LD homeostasis during these early stages impair germ
cell differentiation and decrease sperm output (43,104,105).
Model organisms such as Drosophila and C.
elegans are key in dissecting LD function during
spermatogenesis (43). For
example, the Drosophila homolog of ATGL, Brummer, localizes
to LDs in testicular germ cells and its deletion results in massive
LD accumulation and spermatogenic arrest (43). Similarly, in mammals, ATGL and
HSL are functionally important in lipid mobilization during
spermatogenic progression (43,106). Deficiency in these enzymes
leads to abnormal lipid accumulation, disrupted spermatid
elongation and reduced fertility (106).
While studies in C. elegans have uncovered
genetically conserved roles of LD in embryonic integrity (25,107,108), mammalian systems exhibit
additional layers of complexity, particularly in steroidogenesis
and hormonal regulation. For instance, LDs in mammalian
reproductive cells integrate with hormone-responsive lipases to
mobilize cholesterol for steroid hormone synthesis and interact
with organelles such as mitochondria for enhanced metabolic
channeling, features modulated by endocrine signals absent in
simpler models (Table I).
 | Table IConserved and divergent roles of LDs
in reproduction. |
Table I
Conserved and divergent roles of LDs
in reproduction.
| Biological
process | C.
elegans | Mammals | Conservation | (Refs.) |
|---|
| LD biogenesis | Regulated by SEIP-1
at ER-LD junctions | Conserved role of
SEIP-1 in LD formation | Conserved | (215,216) |
| LD spatial
distribution | Prominent LDs in
germline and early embryos | LD-rich oocytes and
steroidogenic cells | Conserved | (217,218) |
| Role in
embryogenesis | SEIP-1-dependent
LDs support eggshell lipid barrier formation | LDs support early
embryonic energy supply and membrane biosynthesis | Functionally
conserved | (37,219) |
|
Steroidogenesis | Absent | LDs store
cholesteryl esters for steroid hormone synthesis | Divergent | (220,221) |
| LD-mitochondria
interaction | Functional coupling
inferred genetically | Physical tethering
via PLIN5/MIGA2-mediated contact sites | Partially
conserved | (222,223) |
| Lipid
mobilization | Lipolysis supports
embryonic viability | β-oxidation fuels
oocyte maturation and embryo development | Conserved | (215,224,225) |
| Regulatory
signaling | Genetic pathways
linking LDs to development | Nuclear receptor
signaling (PPARs) linked to LD-derived lipids | Divergent | (215,226) |
| Clinical
relevance | Model for conserved
mechanisms | Direct relevance to
infertility and ART outcomes | Divergent | (35,219) |
Sertoli cell-LD axis in supporting
spermatogenesis
Sertoli cells provide structural and metabolic
support to developing germ cells (46,109-111). Sertoli cells contain abundant
LDs, the composition and abundance of which fluctuate in response
to the spermatogenic cycle and phagocytic activity (45,46,110). One notable source of LDs in
Sertoli cells is the engulfment of residual bodies (cytoplasmic
fragments shed by spermatids during final maturation) (17,46,110,112). Lipid-rich components of these
residual bodies are internalized and sequestered into LDs within
Sertoli cells, where they may be catabolized via β-oxidation or
lipophagy to fuel Sertoli cell metabolism (15,17,46).
The dynamic balance between LD formation and
degradation in Sertoli cells is key for maintaining testicular
homeostasis (103,113,114). Exposure to toxicants such as
lead or cadmium disrupts lysosomal and autophagic pathways in
Sertoli cells, leading to impaired LD clearance, lipid overload and
testicular dysfunction (113).
Furthermore, hormonal regulation, particularly by FSH, modulates LD
content in Sertoli cells by stimulating lipid uptake and lipogenic
gene expression (109). This
endocrine-metabolic axis ensures that Sertoli cells are
metabolically equipped to support the rapid turnover of lipids
during active spermatogenesis (109,111,114).
While rodent models have provided insights into the
metabolic coupling between Sertoli and germ cells, key differences
exist between human and rodent spermatogenesis (115,116). Human spermatogenesis is
characterized by a longer developmental timeline, distinct
seminiferous epithelial organization and differences in hormonal
regulation and metabolic demands compared with commonly used rodent
models (117,118). Moreover, the dynamics of lipid
metabolism and LD turnover in human Sertoli cells are less well
characterized, in part due to limited access to human testicular
tissue (115,117,119).
These species-specific features suggest that,
although core principles of Sertoli-germ cell metabolic support are
potentially conserved, direct extrapolation from rodent studies to
human reproductive physiology should be approached with caution.
Integrating findings from human tissue analyses, organoid systems
and single-cell profiling is key to define the relevance of
LD-mediated mechanisms in human spermatogenesis (120-123).
Lipids and membrane remodeling in sperm
maturation
Although mature spermatozoa lack classical LDs
(typical cytoplasmic organelles characterized by a hydrophobic core
of neutral lipids encased in a phospholipid monolayer), lipid
metabolism is key for their structural and functional integrity
(43,124). The plasma membrane of sperm is
rich in polyunsaturated FAs (PUFAs), which provide membrane
fluidity necessary for motility and capacitation (124-130). The composition of these lipids
is tightly regulated during epididymal maturation and influenced by
prior LD metabolism in germ and Sertoli cells (43,103,124,131).
Enzymes such as acyl-CoA synthetase long-chain
family members, which participate in FA activation and
incorporation into complex lipids, are key for proper sperm
development (75). Genetic
disruption of these enzymes leads to altered lipid composition and
defective sperm morphology (75). These findings suggest that early
LD metabolism indirectly shapes sperm functionality by controlling
the availability and remodeling of lipid precursors (43, 75,124).
LD-associated defects and male
infertility
Aberrant lipid metabolism in the testis is
increasingly implicated in male reproductive disorder (43,104,105,132). In both mouse and rat models and
human patients, disturbances in lipid homeostasis, manifested as
altered LD dynamics, defective lipolysis or accumulation of
cholesteryl esters, are associated with low sperm count, impaired
motility and hormonal imbalance (43,104-106,132). Notably, mouse models with HSL
knockout exhibit lipid-laden Leydig cells, decreased testosterone
levels and oligospermia, underscoring the importance of intact LD
mobilization for androgen synthesis and spermatogenic support
(106,133,134).
Lipidomics and histological analyses of human
testicular biopsies have revealed elevated LD accumulation in
Sertoli and Leydig cells of infertile patients, often accompanied
by disrupted mitochondrial morphology and increased oxidative
stress markers (119,135-137). These observations further
reinforce the idea that LD dysregulation contributes to male
infertility not only through energy imbalance, but also by
disrupting redox homeostasis, membrane remodeling and hormone
production (43,104,106,132,138).
LDs in reproductive support cells
Reproductive support cells (Sertoli cells in the
testis and granulosa cells in the ovary) serve key roles in
nurturing germ cells and regulating the hormonal environment of the
gonads (139). Both cell types
are metabolically active, highly responsive to hormonal cues and
rely on tightly regulated lipid metabolism to perform their
functions (140). LDs within
these cells serve as key metabolic and regulatory hubs,
participating not only in lipid storage and mobilization but also
in steroidogenesis, phagocytic recycling and paracrine signaling
(6) (Fig. 4).
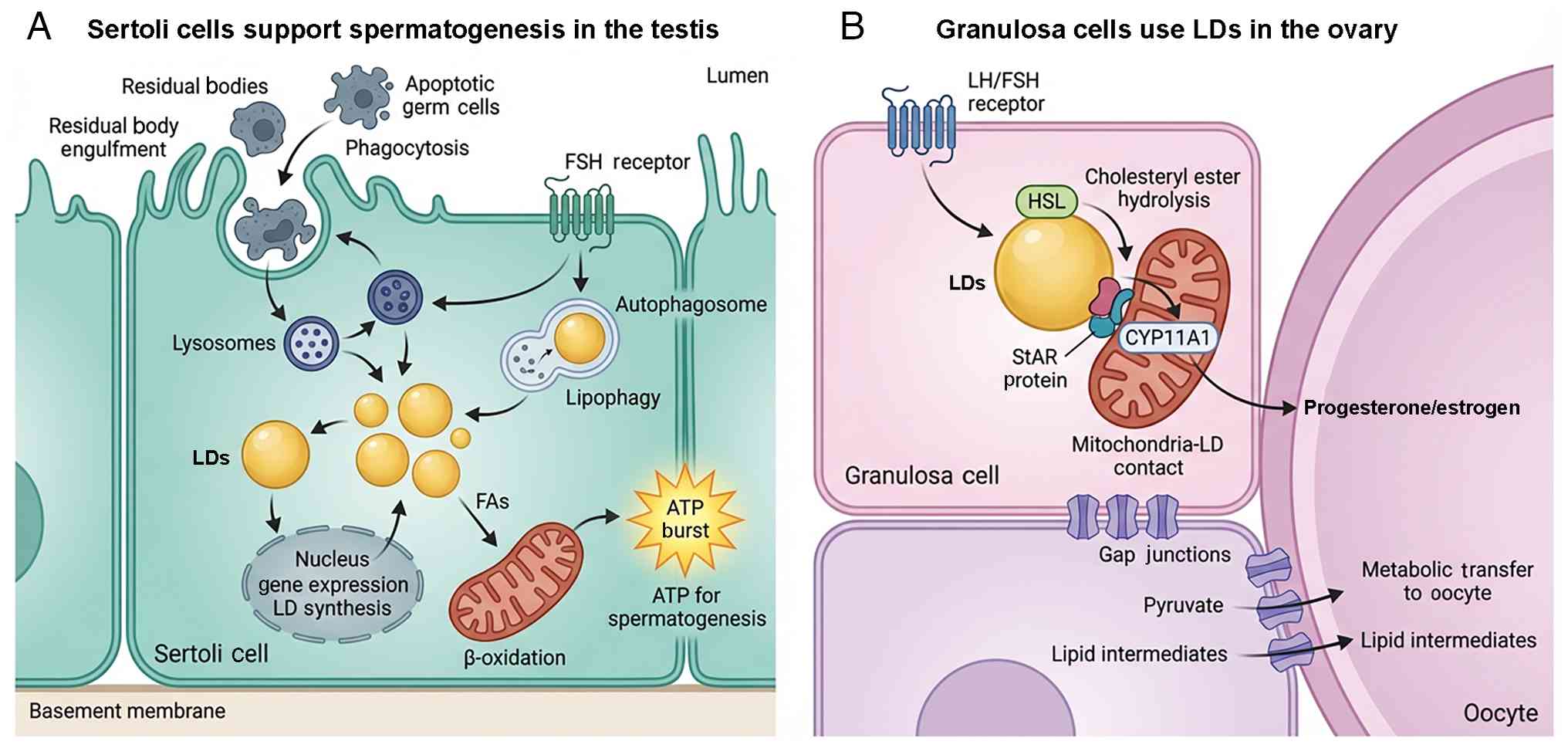 | Figure 4Comparative functions of LDs in
reproductive support cells. (A) In the seminiferous epithelium,
Sertoli cells support spermatogenesis through a recycling
mechanism. Sertoli cells engulf residual bodies (cytoplasmic
remnants) and apoptotic germ cells via phagocytosis. These
internalized materials are processed in lysosomes and their lipid
content is recycled into LDs. Stored lipids are subsequently
mobilized via lipophagy (autophagic degradation) and mitochondrial
β-oxidation to generate ATP, providing the energy required for
Sertoli cell metabolism and germ cell support. This process is
regulated by FSH, which stimulates lipid uptake and storage gene
expression. (B) In the ovarian follicle, granulosa cells use LDs
primarily as substrate reservoirs for hormone synthesis. Upon
stimulation by gonadotropins (LH/FSH), HSL hydrolyzes stored
cholesteryl esters into free cholesterol. Cholesterol is
transported to mitochondria via the StAR protein at LD-mitochondria
contact sites. Inside the mitochondria, CYP11A1 initiates the
conversion of cholesterol into steroid hormones
(progesterone/estrogen). Granulosa cells also metabolically support
the oocyte by transferring pyruvate and lipid intermediates through
gap junctions, a process associated with the metabolic status of
their own LDs. CYP11A1, cytochrome P450 family 11 subfamily A
member 1; FSH, follicle-stimulating hormone; HSL, hormone-sensitive
lipase; LD, lipid droplet; LH, luteinizing hormone; StAR,
steroidogenic acute regulatory protein; FA, fatty acid. |
LDs in Sertoli cells: Nutrient buffering
and phagocytic recycling
Sertoli cells are the central architectural and
metabolic support for spermatogenesis, forming the blood-testis
barrier, providing nutrients to developing germ cells and clearing
apoptotic or senescent spermatocytes (139). These cells contain prominent
LDs, particularly during periods of active spermatogenic turnover
or high phagocytic activity (15).
A notable source of lipids for LD formation in
Sertoli cells is the internalization of residual bodies and
degenerating germ cells (17).
These engulfed materials, rich in membrane and cytoplasmic lipids,
are processed in lysosomes, with a portion of the liberated FAs and
sterols re-esterified and stored in LDs. These droplets are
mobilized for energy production via β-oxidation, allowing Sertoli
cells to meet their metabolic demands while supporting adjacent
spermatogenic cells (141).
The degradation of LDs in Sertoli cells involves
not only cytosolic lipases (HSL) but also selective autophagy of
LDs, known as lipophagy (142).
This process is regulated by nutrient availability and endocrine
factors. Under physiological stress or toxicant exposure (lead,
cadmium), lysosomal dysfunction or autophagy impairment leads to LD
accumulation, disrupted lipid homeostasis and impaired Sertoli cell
function, contributing to testicular atrophy and infertility
(143).
Furthermore, hormonal regulation serves a key role
in LD dynamics. FSH promotes lipid uptake and storage in Sertoli
cells by upregulating lipoprotein receptors and lipogenic gene
expression. This hormonally mediated lipid buffering ensures an
adequate energy reserve during active spermatogenesis and
facilitates metabolic synchronization between Sertoli and germ
cells (109,144-146).
LDs in granulosa and luteal cells:
Platforms for steroidogenesis
Granulosa cells, which surround and support the
developing oocyte, undergo extensive metabolic reprogramming as
follicles mature (147). In the
pre-ovulatory phase, these cells proliferate, increase
steroidogenic activity and accumulate LDs rich in cholesteryl
esters (49). Following
ovulation, granulosa cells differentiate into luteal cells, which
are among the most steroidogenically active cells in the body
(147). In both phases, LDs
serve as central platforms for steroid hormone biosynthesis
(148).
LDs in granulosa and luteal cells store cholesteryl
esters that serve as substrates for estrogen and progesterone
synthesis, respectively (149).
Upon gonadotropin (FSH or LH) stimulation, HSL is activated and
hydrolyzes cholesteryl esters into free cholesterol (148). The cholesterol is transported
to mitochondria via steroidogenic acute regulatory protein, where
it is converted to pregnenolone by CYP11A1, initiating the
steroidogenic cascade (149).
Several studies have demonstrated that LD content
and associated enzyme expression levels vary depending on
follicular stage and endocrine environment (150-153). For example, mature preovulatory
granulosa cells contain larger and more numerous LDs than their
early antral counterparts, consistent with enhanced steroidogenic
readiness (23). Disruption of
lipid mobilization pathways via HSL inhibition, PLIN dysregulation
or excessive lipid accumulation impairs estrogen and progesterone
synthesis, follicular rupture and corpus luteum formation (150).
Proteomic and lipidomic profiling of granulosa cell
LDs has identified key components of the steroid biosynthetic
machinery localized to or enriched around LDs, including enzymes of
the P450 family and mitochondrial contact proteins (90). These findings suggest that LDs
are not passive lipid depots, but serve as biochemical scaffolds
that facilitate rapid and localized hormone production (148).
LD-oocyte communication: The somatic-germ
cell interface
Beyond their intrinsic functions, support cell LDs
also influence germ cell development through metabolic coupling and
paracrine interactions (154).
Cumulus granulosa cells, which form the innermost layer of
follicular somatic cells directly surrounding the oocyte, are
metabolically connected to the oocyte via transzonal projections
and gap junctions (155). These
cells actively metabolize glucose and FAs, generating pyruvate,
amino acids and lipid intermediates that are transferred to the
oocyte to support growth and maturation (28,154).
LDs within cumulus cells reflect this metabolic
activity (156). Their number
and size increase with gonadotropin stimulation and they show
dynamic responses to oxidative stress, FA exposure and endocrine
disruption (157). Excess
accumulation of saturated FAs in cumulus cell LDs, such as under
high-fat diet or PCOS, alter oocyte lipid composition, increase ER
stress and reduce developmental competence (156).
Therefore, somatic cell LDs contribute indirectly
to oocyte quality by modulating the follicular lipid environment,
buffering toxic lipid species and fine-tuning substrate
availability for oocyte maturation (157). Disruption of these functions
leads to broader metabolic dysfunction and subfertility,
highlighting the need to consider support cell lipid metabolism in
the assessment of reproductive health (89).
LDs in early embryo development and
pluripotency
Early embryonic development is an energy-intensive
process that depends on maternally derived reserves, since
transcriptional activity is minimal until zygotic genome activation
(37). Among the maternally
supplied nutrients, LDs serve as critical reservoirs of neutral
lipids and bioactive lipids that sustain cleavage division, lineage
specification and metabolic reprogramming (69). Beyond their role as passive
energy stores, LDs are increasingly recognized as dynamic
organelles that integrate metabolic and signaling pathways,
influencing not only embryo viability but also the maintenance of
pluripotency (37,69,75,158) (Fig. 5).
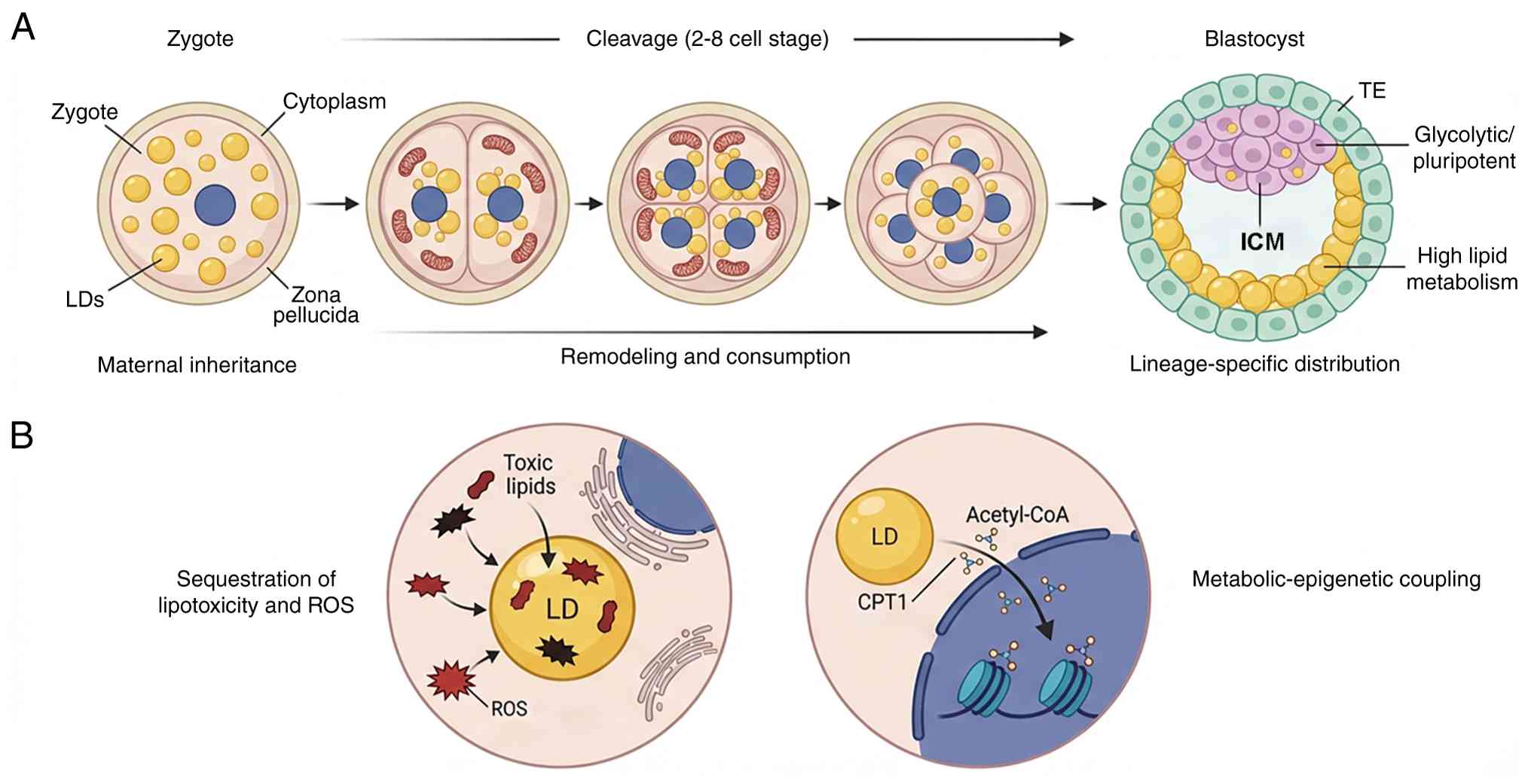 | Figure 5Spatiotemporal dynamics and
functional roles of LDs during preimplantation development. (A)
Developmental timeline (zygote to blastocyst). Following
fertilization, the zygote inherits a pool of maternal LDs. During
early cleavage divisions, LDs undergo dynamic clustering (typically
perinuclear) and partial lipolysis. They interact with mitochondria
to provide fatty acids for β-oxidation, fueling the
energy-intensive process of rapid cell division. At the blastocyst
stage, LDs exhibit asymmetric distribution. TE cells (outer layer)
contain larger and more numerous LDs, supporting their high lipid
metabolic needs for implantation and steroidogenesis. By contrast,
the ICM (inner cluster) contains fewer LDs, consistent with a
glycolytic, pluripotent metabolic state. (B) Functional mechanisms.
LDs act as detoxification sinks by sequestering toxic saturated
fatty acids and peroxidized lipids, thereby protecting the embryo
from lipotoxicity and oxidative stress (ROS) when antioxidant
defenses are developing. In pluripotent cells, LD-derived
acetyl-CoA enters the nucleus to serve as a substrate for histone
acetylation. This metabolic-epigenetic link influences chromatin
structure and gene expression, thereby regulating the maintenance
of pluripotency and stem cell fate decisions. ICM, inner cell mass;
LD, lipid droplet; ROS, reactive oxygen species; TE, trophectoderm;
CPT, carnitine palmitoyltransferase. |
LD inheritance and remodeling
post-fertilization
Upon fertilization, the zygote inherits LDs from
the oocyte, which vary in size, number and lipid composition across
species (72). Following
fertilization, LDs transition toward a metabolic phase
characterized by enhanced lipolysis and FA oxidation (FAO). In
lipid-rich species such as pigs and cows, zygotes contain abundant
LDs that are readily visualized by light microscopy, while in mice
and humans LDs are smaller (159). These maternal LDs are
progressively redistributed during the first cleavage divisions
(42). Time-lapse imaging
studies reveal that LDs undergo dynamic clustering, dispersion and
partial lipolysis in synchrony with cell cycle progression
(158,160-162). This remodeling reflects a shift
from maternal lipid storage toward active use, enabling the embryo
to meet energetic demands of rapid mitosis (31). Beyond serving as lipid
reservoirs, LDs in early embryos also participate in the storage
and regulation of non-lipid macromolecules. Johnson et al
(126) demonstrated that LDs in
early embryos serve as storage depots for maternal histones and
selectively recruit RNA-binding proteins involved in
post-transcriptional regulation. Through this mechanism, LDs
contribute to the temporal control of mRNA translation during early
embryogenesis, a developmental window characterized by limited
zygotic transcription. These findings expand the functional scope
of LDs beyond energy metabolism, positioning them as organizational
platforms that coordinate lipid storage with proteostasis and
translational control in the early embryo.
The extent of LD remodeling is sensitive to culture
conditions and external nutrient availability (163). Embryos cultured in vitro
often display altered LD number and distribution compared with
in vivo counterparts, which has been linked to reduced
developmental potential (164).
These findings highlight LDs as sensitive indicators of embryo
metabolic state (71).
LD metabolism and early energy
supply
During cleavage, embryos rely primarily on pyruvate
and lactate metabolism, but FAO becomes increasingly important as
development proceeds to the blastocyst stage (49). LDs provide a readily accessible
pool of FAs through controlled lipolysis (165). Inhibition of FAO enzymes such
as carnitine palmitoyltransferase 1 (CPT1) leads to developmental
arrest, underscoring the importance of LD-derived FAs in sustaining
blastocyst formation (166).
Mitochondrial-LD interactions are central to this
process (37). In mouse and
bovine embryos, LDs are typically found close to mitochondria,
facilitating efficient FA transfer and oxidation (13). This spatial proximity suggests
that LDs and mitochondria form functional units that coordinate
energy production and redox regulation during early development
(167).
An additional context in which LD-mitochondria
interactions may be relevant is ovarian aging. Advanced maternal
age is associated with progressive mitochondrial dysfunction in
oocytes, including decreased oxidative capacity, altered
mitochondrial dynamics and increased oxidative stress (168,169). Emerging evidence suggests that
aged oocytes often display abnormal LD accumulation and altered
lipid distribution, raising the possibility that impaired
mitochondrial FA utilization contributes to LD dysregulation during
aging (69,170,171).
Whether aberrant LD accumulation in aged oocytes
represents a compensatory response to mitochondrial insufficiency
or a maladaptive process that compromises oocyte quality remains
unresolved. Clarifying how mitochondrial dysfunction intersects
with LD storage, mobilization and oxidative stress control during
ovarian aging is key for understanding age-associated declines in
oocyte competence and embryo developmental potential.
In addition to their roles in energy metabolism and
signaling, LDs have recently been implicated in protecting embryos
from lipid peroxidation and ferroptotic cell death (172-174). PUFAs, while essential for
membrane synthesis and developmental signaling, are susceptible to
oxidative damage. Sequestration of PUFAs within LDs limits their
availability for uncontrolled lipid peroxidation, thereby
decreasing oxidative stress and ferroptosis during early embryonic
development (174-177).
Recent studies have highlighted LDs as key buffers
that spatially compartmentalize PUFAs away from pro-oxidant
environments, particularly under conditions of heightened
mitochondrial activity and reactive oxygen species production
(175,176,178). This protective function may be
key during early embryogenesis, when antioxidant capacity is
limited and metabolic demands rapidly increase. Together, these
findings position LDs not only as metabolic hubs but also as key
guardians of redox homeostasis and embryo survival.
Carnitine shuttle system as the
rate-limiting step of LD-derived FAO
While FA β-oxidation is a notable pathway through
which LD-derived lipids support reproductive processes, its flux is
constrained by a rate-limiting step: The transport of long-chain
FAs into mitochondria (179-181). This process is mediated by the
carnitine shuttle system, which consists of CPT1 on the outer
mitochondrial membrane and CPT2 on the inner membrane (2,179,182,183). CPT1 catalyzes the conversion of
long-chain acyl-CoAs into acyl-carnitines, enabling their
translocation across the mitochondrial membranes, whereas CPT2
reconverts acyl-carnitines into acyl-CoAs within the mitochondrial
matrix for β-oxidation (180,184-186).
In oocytes and early embryos where mitochondrial
oxidative capacity is tightly coupled with developmental
competence, disruption of carnitine-dependent FA transport leads to
impaired energy production and developmental arrest, underscoring
the importance of this regulatory checkpoint in lipid utilization
(49,166). Notably, supplementation with
L-carnitine or acetyl-L-carnitine improves mitochondrial function,
enhances FAO and increases blastocyst yield in multiple assisted
reproduction settings (187,188). These findings suggest that the
carnitine shuttle not only represents a biochemical bottleneck of
LD-derived energy metabolism, but also constitutes a clinically
actionable node linking LD biology to gamete quality and embryo
developmental outcomes.
LDs as regulators of redox homeostasis
and stress responses
Beyond energy supply, LDs also protect early
embryos from lipotoxicity and oxidative stress (2). Excess accumulation of saturated FAs
in culture or maternal metabolic disorders can trigger ER stress
and apoptosis in embryos (69).
By sequestering potentially toxic lipids into LDs, embryos buffer
against lipotoxic insult (189). Moreover, LDs can serve as sinks
for peroxidized lipids, thereby limiting propagation of oxidative
damage (190). This
detoxification role is key during preimplantation, when embryonic
antioxidant defenses are developing (161).
LD dynamics and lineage
specification
As embryos reach the blastocyst stage, LD
distribution becomes asymmetric between inner cell mass (ICM) and
trophectoderm (TE) (42). TE
cells, which contribute to the placenta, contain more and larger
LDs than ICM cells, suggesting lineage-specific metabolic
requirements (42,158,191). This differential LD allocation
may reflect the TE role in steroidogenesis, nutrient transport and
implantation, which demand robust lipid metabolism (158).
In pluripotent ICM cells, LDs are fewer but
metabolically active, supporting a glycolysis-dominant phenotype
typical of stem cells (192).
Manipulating LD metabolism in vitro influences stem cell
fate: Pharmacological activation of lipolysis promotes
differentiation, while stabilizing LDs maintains pluripotency
(193). These findings position
LDs as regulators of cell fate transitions in the embryo (194).
Implications for assisted reproduction
and stem cell biology
Understanding LD biology in embryos has practical
implications (72). In assisted
reproductive technology, embryo selection is typically based on
morphology, yet LD content and dynamics may provide more sensitive
biomarkers of viability (31).
Non-invasive imaging techniques such as CARS microscopy have been
applied to quantify LDs in live embryos, revealing an association
between lipid distribution and implantation success (160).
In stem cell biology, insights into LD regulation
in early embryos inform strategies to maintain pluripotency in
vitro and direct differentiation (49). For example, modulating lipid
availability or LD turnover influences epigenetic programming,
since acetyl-CoA and lipid-derived metabolites serve as cofactors
for chromatin-modifying enzymes. Thus, LDs not only support
metabolic needs but also contribute to epigenetic landscapes that
govern developmental trajectories (195).
Key regulators of LD biology in reproductive
cells
The biology of LDs in reproductive systems is
orchestrated by a complex network of enzymes, structural proteins,
transcriptional factors and signaling pathways (6). These regulators not only govern LD
biogenesis and turnover but also link lipid metabolism to the
unique demands of gametogenesis, steroidogenesis and embryogenesis
(49). Their coordinated
activity ensures that lipid reserves are properly balanced between
storage and mobilization, safeguarding reproductive success
(69) (Fig. 6).
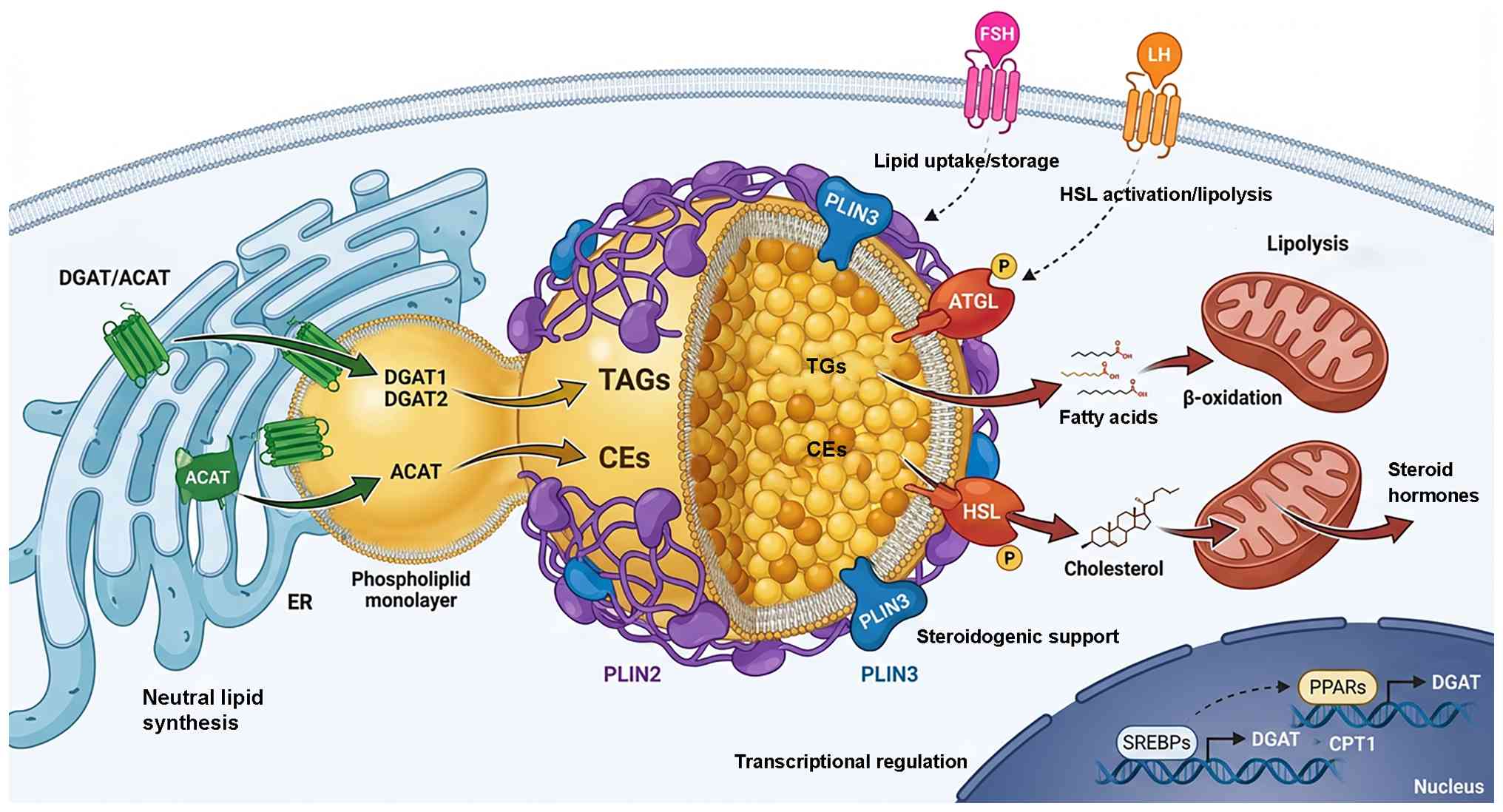 | Figure 6Molecular landscape of LD regulation
in reproductive cells. Integrated network of enzymes, structural
proteins and signaling pathways orchestrate LD biology, ensuring
the balance between lipid storage and utilization. LD formation
originates at the ER. DGAT1/2 catalyze the synthesis of TAGs, while
ACATs synthesize CEs. These neutral lipids are packaged into the
nascent LD core, establishing the metabolic reserves required for
gametogenesis and steroidogenesis. The LD surface is coated by PLIN
family proteins. PLIN2 stabilizes the LD by preventing uncontrolled
lipolysis (crucial for oocyte lipid retention), while PLIN3
supports cholesterol storage in steroidogenic cells. These proteins
serve as gatekeepers, regulating access to the lipid core.
Controlled lipid breakdown is mediated by lipases. ATGL initiates
TAG hydrolysis to release fatty acids for mitochondrial
β-oxidation. HSL, activated via phosphorylation, hydrolyzes TAGs
and CEs, liberating cholesterol for steroid hormone synthesis. The
entire system is governed by upstream regulators. Hormones such as
FSH promote lipid uptake/storage, whereas LH triggers lipolysis by
activating HSL. At the nuclear level, transcription factors SREBP
and PPAR modulate the expression of lipogenic and oxidative genes,
respectively, adapting cell metabolism to developmental demands.
ACAT, acyl-CoA cholesterol acyltransferase; ATGL, adipose
triglyceride lipase; DGAT, diacylglycerol acyltransferase; ER,
endoplasmic reticulum; FSH, follicle-stimulating hormone; HSL,
hormone-sensitive lipase; LH, luteinizing hormone; PLIN, perilipin;
PPAR, peroxisome proliferator-activated receptor; SREBP, sterol
regulatory element-binding protein; LD, lipid droplet; TAG,
triacylglycerol; CPT, carnitine palmitoyltransferase; CE,
cholesteryl Ester. |
Enzymes of neutral lipid synthesis
LD biogenesis begins with the synthesis of neutral
lipids within the ER, a process primarily mediated by enzymes such
as DGAT1 and DGAT2 for triacylglycerols and acyl-CoA cholesterol
acyltransferases (ACATs) for cholesteryl esters (140). In reproductive cells, these
enzymes facilitate metabolic readiness: In oocytes, DGAT2 activity
underlies the accumulation of lipid stores necessary for meiotic
progression (196), while in
granulosa and luteal cells, ACAT-mediated cholesterol
esterification establishes a pool of precursors for steroid hormone
synthesis (149). Disruption of
these synthetic pathways reduces oocyte competence and compromises
hormone production, highlighting their key role in reproductive
physiology (150).
LD-coating and stabilizing proteins
Once formed, LDs are stabilized and functionally
tuned by coat proteins, most prominently the PLIN family (197). PLIN2, highly abundant in
lipid-rich oocytes and granulosa cells, prevents uncontrolled
lipolysis and ensures lipids remain available for developmental
cues (37,48,68). PLIN3, enriched in steroidogenic
cells, contributes to cholesterol storage and supports rapid
progesterone synthesis following LH stimulation (22,24,47). These surface proteins not only
regulate LD size and stability but also serve as molecular
scaffolds that recruit lipases or tether LDs to partner organelles,
integrating storage with utilization (2).
Lipolytic enzymes
Mobilization of LD lipids is mediated by lipolytic
enzymes, whose activity is coupled to reproductive events (148). ATGL initiates triacylglycerol
breakdown, providing FAs for mitochondrial β-oxidation, which is
key for oocyte maturation and embryonic cleavage (166). HSL plays a dual role,
hydrolyzing both TAGs and cholesteryl esters. In luteal cells,
LH-induced phosphorylation of HSL rapidly liberates cholesterol for
progesterone synthesis, directly linking lipolysis to endocrine
function (147). Failure of
these pathways, as seen in mouse knockout models of ATGL or HSL
deficiency, results in lipid accumulation, impaired gametogenesis
and subfertility (106,133).
Transcriptional and hormonal
regulators
The transcriptional and hormonal control of LD
regulators provides another layer of coordination (143). Sterol regulatory
element-binding proteins activate the expression of lipogenic
enzymes, ensuring reproductive cells adapt to energy demand and
steroidogenic flux (198).
PPARs fine-tune lipid storage and oxidation, with PPARγ promoting
lipid accumulation in granulosa cells and PPARα enhancing FA use in
embryos (199). Endocrine
signals, particularly FSH and LH, further dictate LD dynamics: FSH
promotes lipid uptake and storage in Sertoli and granulosa cells,
while LH triggers lipolysis and cholesterol mobilization,
exemplifying how systemic hormonal cues converge on LD regulation
(20,109,200).
LD dysfunction and reproductive
disorders
The regulation of LD biology is key for
reproductive competence and its disruption is increasingly
recognized as a pathogenic factor in infertility and reproductive
disease (6,37,41). Aberrant LD accumulation, altered
lipid composition or defective mobilization disturb gametogenesis,
hormone production and embryonic development, ultimately
compromising reproductive outcomes (49) (Fig. 7).
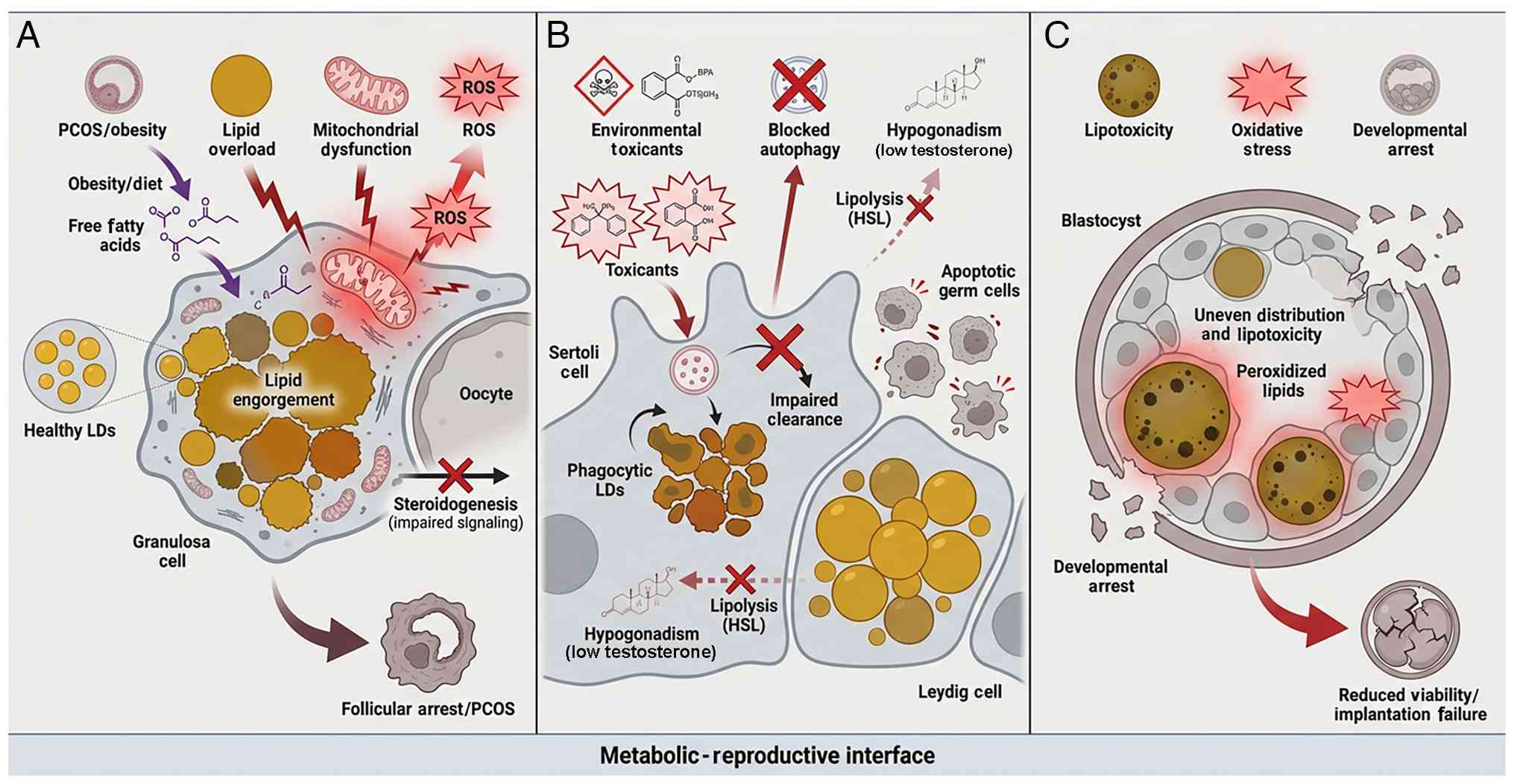 | Figure 7LD dysfunction as a driver of
reproductive pathology. (A) Obesity causes lipid overload in
granulosa cells and oocytes. Under conditions of obesity or dietary
excess, high levels of free fatty acids cause lipid overload in
granulosa cells and oocytes. This leads to mitochondrial
dysfunction and the generation of ROS. The resulting metabolic
stress impairs steroidogenic signaling and disrupts follicle
development, culminating in follicular arrest and phenotypes
associated with PCOS. (B) Toxicants disrupt LD homeostasis in the
testis. Exposure to environmental toxicants (BPA, phthalates)
disrupts LD homeostasis in the testis. In Sertoli cells, toxicants
block lysosomal/autophagic pathways, preventing the clearance of
phagocytosed lipids. This leads to the accumulation of phagocytic
LDs and impairs the nutritional support provided to germ cells. In
Leydig cells, disruption of lipolysis (HSL inhibition) prevents
cholesterol mobilization, resulting in decreased testosterone
synthesis (hypogonadism) and germ cell apoptosis. (C) Lipotoxicity
leads to developmental arrest in embryos. In the context of IVF or
maternal metabolic disorder, embryos may exhibit uneven
distribution of LDs. The accumulation of peroxidized lipids creates
a state of lipotoxicity and oxidative stress. These cell insults
compromise blastocyst quality, leading to developmental arrest,
decreased viability and implantation failure. BPA, bisphenol A;
HSL, hormone-sensitive lipase; IVF, in vitro fertilization;
LD, lipid droplet; PCOS, polycystic ovary syndrome; ROS, reactive
oxygen species. |
Excessive lipid storage or impaired lipid
utilization is associated with ovarian pathologies (150). Patients with PCOS, one of the
most common causes of infertility, display abnormal LD accumulation
and dysregulated lipid metabolism in granulosa cells (156). This metabolic imbalance alters
steroidogenic signaling, impairs follicle development and reduces
oocyte competence (89).
Similarly, maternal obesity and diabetes are associated with
excessive LD deposition in oocytes and embryos, resulting in
lipotoxicity, mitochondrial dysfunction and decreased embryo
viability (201). These
findings suggest that LD mismanagement contributes to the
metabolic-reproductive interface that underlies certain female
infertility syndromes (38).
In the male gonad, LD dysfunction also exerts
notable effects (143). Sertoli
cells, which provide structural and metabolic support for
spermatogenesis, rely on LD turnover to supply energy substrates to
developing germ cells (139).
Disruption of lipolysis or autophagic clearance in these cells
leads to LD accumulation, impaired nutrient transfer and defective
sperm maturation (141). Leydig
cells depend on LDs for cholesterol ester storage and rapid
mobilization following LD stimulation; defects in this system
decrease testosterone synthesis, contributing to hypogonadism and
subfertility (202). Moreover,
environmental toxicants such as phthalates and bisphenol A perturb
LD homeostasis in testicular cells, linking lipid dysregulation to
environmentally induced reproductive disorder (203).
Beyond the gonads, LD abnormality influences early
developmental competence (69).
In vitro fertilization studies reveal that embryo with
excessive or uneven LD distribution exhibit lower developmental
potential, potentially due to oxidative stress and impaired energy
regulation (31,41,69,204). Defects in LD-associated
proteins, including PLIN and SEIP-1, cause embryonic lethality or
reduced implantation success, underscoring the importance of
regulated LD dynamics for reproductive success (205).
Together, these findings position LD dysfunction as
both a biomarker and a mechanistic driver of reproductive pathology
(2). By disrupting lipid
homeostasis, LD abnormality compromises gamete quality, hormone
production and embryo viability (37). Clarifying the molecular
underpinnings of LD dysfunction in reproductive disorders holds
promise not only for understanding infertility but also for
identifying novel diagnostic markers and therapeutic targets
(206).
Technological advances in studying LDs in
reproduction
The study of LDs in reproductive biology has
benefited from technological innovations that allow improved
resolution, sensitivity and functional insight (6). Traditional staining approaches,
such as Oil Red O and BODIPY dyes, provided the first visualization
of LDs in oocytes and embryos (207), but advances in imaging and
molecular profiling permit dynamic and quantitative analyses of LD
biology within reproductive contexts (69).
High-resolution microscopy has been key to these
advances (13). Confocal and
two-photon microscopy enable three-dimensional imaging of LD
distribution in intact oocytes and embryos (31), while super-resolution techniques
such as stimulated emission depletion and structured illumination
microscopy reveal nanoscale details of LD-organelle interactions.
Live-cell imaging with fluorescently tagged LD-associated proteins
makes it possible to track LD dynamics during meiotic maturation,
fertilization and early embryogenesis, providing functional insight
into how LD turnover couples with developmental transition
(160).
Complementing imaging, mass spectrometry-based
lipidomics has transformed the characterization of LD composition
in reproductive tissue (208).
Shotgun and targeted lipidomics approaches identify neutral lipid
species, cholesterol esters and signaling lipids within LDs,
enabling the detection of metabolic alterations in pathological
states such as PCOS or maternal obesity. Single-cell and spatial
lipidomics approaches have been applied to oocytes and embryos,
resolving metabolic heterogeneity that underlies differences in
developmental competence (209-212).
Genetic and molecular tools have also advanced
functional studies (37,213,214). CRISPR-Cas9 editing in mice and
C. elegans allows precise manipulation of LD-associated
genes, demonstrating their roles in gametogenesis and embryonic
development (205). Fluorescent
reporters, such as PLIN or SEIP-1 fusion proteins, permit in
vivo visualization of LD subsets and their dynamic responses to
hormonal or metabolic cues (196). In parallel, optogenetic and
inducible systems are powerful approaches to manipulate LD
formation or lipolysis in real time, offering a causal
understanding of LD function in reproductive processes (167).
Finally, integrative approaches that combine
imaging, lipidomics and systems biology are beginning to redefine
the landscape of LD research in reproduction (2). Spatial multi-omics platforms map
LDs in the context of transcriptional and metabolic states
(42), while computational
models help predict how LD dysfunction develops into cell stress
and developmental failure (192). Such integrative technologies
not only provide mechanistic understanding but also hold promise
for translational applications, such as developing LD-based
biomarkers of oocyte quality or embryo viability in assisted
reproductive technology (89).
In sum, methodological advances have propelled LD
research from descriptive observations to mechanistic and
translational insight (49). By
coupling dynamic visualization with molecular profiling and
functional manipulation, these technologies reshape understanding
of how LDs support reproductive success and how their dysfunction
contributes to infertility (150).
Conclusion
LDs are multifaceted organelles that are key for
reproductive physiology. Beyond their traditional role as passive
lipid stores, LDs serve as dynamic hubs integrating energy supply,
membrane biosynthesis, steroidogenesis and stress adaptation. From
oocyte maturation to early embryonic development, their regulated
formation, turnover and organelle interactions ensure reproductive
success. Dysregulation of LD biology, conversely, contributes to a
number of disorders including PCOS, male infertility, and embryonic
developmental defects, underscoring their centrality to
reproductive health.
Mechanistically, it is not fully understood how LD
subpopulations are formed or differentiated (based on size, lipid
composition or functional specialization) in reproductive cells and
how their interactions with mitochondria, ER and lysosomes are
spatiotemporally regulated. The contribution of lipid signaling
molecules released from LDs to epigenetic programming and lineage
specification during early embryogenesis also remains largely
unexplored. Moreover, the interplay between systemic metabolic
disorders, such as obesity and diabetes, and LD dysfunction in
gametes and embryos requires investigation to explain the
intergenerational transmission of reproductive risk.
Future research may benefit from continued
integration of cutting-edge technologies. Advances in live-cell
super-resolution microscopy, spatial and single-cell lipidomics and
CRISPR-based gene editing provide powerful opportunities to dissect
LD biology. LD-based biomarkers hold promise for assessing oocyte
and embryo quality in assisted reproductive technology, while
therapeutic strategies targeting LD metabolism may emerge as novel
approaches to treat infertility associated with metabolic or
endocrine dysfunction. Importantly, comparative studies across
species, from C. elegans and mice to humans, may demonstrate
the evolutionary conservation and divergence of LD functions in
reproduction.
In conclusion, LDs represent a dynamic interface
between lipid metabolism and reproductive biology. By bridging
basic mechanistic insights with clinical applications, LD research
may deepen understanding of fundamental cell biology but also to
transform reproductive medicine.
Availability of data and materials
Not applicable.
Authors' contributions
LP conducted the literature review and wrote the
manuscript. ZW constructed figures. YJ wrote the manuscript and
provided supervision. All authors have read and approved the final
manuscript. Data authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, the authors
used artificial intelligence-assisted tools (including Grok) to
improve the readability and language of the manuscript, to draft
the cover letter and to propose a running title. No artificial
intelligence tool was used to generate scientific content, analyse
or interpret data, draw scientific conclusions or create figures.
After using these tools, the authors reviewed and edited the
content as needed and take full responsibility for the content of
the publication.
Acknowledgements
Not applicable.
Funding
The present study was supported by Shandong Provincial Medical
and Health Science and Technology Project (grant no. 202402081168),
Ji Nan Health High-Caliber Talent Project (grant no. 202512),
Science and Technology Development Plan Project of Jinan Municipal
Health Commission (grant no. 2024302002), the National Natural
Science Foundation of China (grant no. 82401009), Shandong
Provincial Natural Science Foundation (grant no. ZR2025MS1456),
Science and Technology Development Program of Jinan Municipal
Health Commission (grant no. 2024202001) and the Research Start-up
Fee for Introducing Talents to Jinan Central Hospital (grant nos.
YJRC2023001 and YJRC2023005).
References
|
1
|
Farese RV Jr and Walther TC: Lipid
droplets finally get a little R-E-S-P-E-C-T. Cell. 139:855–860.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Olzmann JA and Carvalho P: Dynamics and
functions of lipid droplets. Nat Rev Mol Cell Biol. 20:137–155.
2019. View Article : Google Scholar
|
|
3
|
Walther TC, Chung J and Farese RV Jr:
Lipid droplet biogenesis. Annu Rev Cell Dev Biol. 33:491–510. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Salo VT, Belevich I, Li S, Karhinen L,
Vihinen H, Vigouroux C, Magré J, Thiele C, Hölttä-Vuori M, Jokitalo
E and Ikonen E: Seipin regulates ER-lipid droplet contacts and
cargo delivery. EMBO J. 35:2699–2716. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cartwright BR and Goodman JM: Seipin: From
human disease to molecular mechanism. J Lipid Res. 53:1042–1055.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Welte MA and Gould AP: Lipid droplet
functions beyond energy storage. Biochim Biophys Acta Mol Cell Biol
Lipids. 1862:1260–1272. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bosch M, Sanchez-Alvarez M, Fajardo A,
Kapetanovic R, Steiner B, Dutra F, Moreira L, López JA, Campo R,
Marí M, et al: Mammalian lipid droplets are innate immune hubs
integrating cell metabolism and host defense. Science.
370:eaay80852020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lass A, Zimmermann R, Oberer M and Zechner
R: Lipolysis-a highly regulated multi-enzyme complex mediates the
catabolism of cellular fat stores. Prog Lipid Res. 50:14–27. 2011.
View Article : Google Scholar :
|
|
9
|
Cruz ALS, Barreto EA, Fazolini NPB, Viola
JPB and Bozza PT: Lipid droplets: Platforms with multiple functions
in cancer hallmarks. Cell Death Dis. 11:1052020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Henne WM, Reese ML and Goodman JM: The
assembly of lipid droplets and their roles in challenged cells.
EMBO J. 37:e989472018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sturmey RG, Reis A, Leese HJ and McEvoy
TG: Role of fatty acids in energy provision during oocyte
maturation and early embryo development. Reprod Domest Anim.
44(Suppl 3): S50–S58. 2009. View Article : Google Scholar
|
|
12
|
Brusentsev EY, Mokrousova VI, Igonina TN,
Rozhkova IN and Amstislavsky SY: Role of lipid droplets in the
development of oocytes and preimplantation embryos in mammals. Russ
J Dev Biol. 50:230–237. 2019. View Article : Google Scholar
|
|
13
|
Bradley J and Swann K: Mitochondria and
lipid metabolism in mammalian oocytes and early embryos. Int J Dev
Biol. 63:93–103. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Aardema H, Vos PL, Lolicato F, Roelen BA,
Knijn HM, Vaandrager AB, Helms JB and Gadella BM: Oleic acid
prevents detrimental effects of saturated fatty acids on bovine
oocyte developmental competence. Biol Reprod. 85:62–69. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ahmed N, Liu Y, Chen H, Yang P, Waqas Y,
Liu T, Gandahi JA, Huang Y, Wang L, Song X, et al: Novel cellular
evidence of lipophagy within the Sertoli cells during
spermatogenesis in the turtle. Aging (Albany NY). 9:41–51. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Staggenborg S, Koch R, Rode K, Hüneke H,
Tiedje L, Wirth G, Langeheine M, Blume I, Rohn K, Wrede C, et al:
Connexin43 represents an important regulator for Sertoli cell
morphology, Sertoli cell nuclear ultrastructure, and Sertoli cell
maturation. Sci Rep. 12:128982022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang H, Wang H, Xiong W, Chen Y, Ma Q, Ma
J, Ge Y and Han D: Evaluation on the phagocytosis of apoptotic
spermatogenic cells by Sertoli cells in vitro through detecting
lipid droplet formation by Oil Red O staining. Reproduction.
132:485–492. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yamaguchi T, Fujikawa N, Nimura S, Tokuoka
Y, Tsuda S, Aiuchi T, Kato R, Obama T and Itabe H: Characterization
of lipid droplets in steroidogenic MLTC-1 Leydig cells: Protein
profiles and the morphological change induced by hormone
stimulation. Biochim Biophys Acta. 1851:1285–1295. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gao F, Li G, Liu C, Gao H, Wang H, Liu W,
Chen M, Shang Y, Wang L, Shi J, et al: Autophagy regulates
testosterone synthesis by facilitating cholesterol uptake in Leydig
cells. J Cell Biol. 217:2103–2119. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Plewes MR, Talbott HA, Saviola AJ, Woods
NT, Schott MB and Davis JS: Luteal lipid droplets: A novel platform
for steroid synthesis. Endocrinology. 164:bqad1242023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Przygrodzka E, Hou X, Zhang P, Plewes MR,
Franco R and Davis JS: PKA and AMPK signaling pathways
differentially regulate luteal steroidogenesis. Endocrinology.
162:bqab0152021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Plewes MR, Krause C, Talbott HA,
Przygrodzka E, Wood JR, Cupp AS and Davis JS: Trafficking of
cholesterol from lipid droplets to mitochondria in bovine luteal
cells: Acute control of progesterone synthesis. FASEB J.
34:10731–10750. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Khor VK, Ahrends R, Lin Y, Shen WJ, Adams
CM, Roseman AN, Cortez Y, Teruel MN, Azhar S and Kraemer FB: The
proteome of cholesteryl-ester-enriched versus
triacylglycerol-enriched lipid droplets. PLoS One. 9:e1050472014.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shen WJ, Azhar S and Kraemer FB: Lipid
droplets and steroidogenic cells. Exp Cell Res. 340:209–214. 2016.
View Article : Google Scholar
|
|
25
|
Cao Z, Fung CW and Mak HY: A flexible
network of lipid droplet associated proteins support embryonic
integrity of C. elegans. Front Cell Dev Biol. 10:8564742022.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bai X, Huang LJ, Chen SW, Nebenfuehr B,
Wysolmerski B, Wu JC, Olson SK, Golden A and Wang CW: Loss of the
seipin gene perturbs eggshell formation in Caenorhabditiselegans.
Development. 147:dev1929972020. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tao A, Wu T, Han X, Niu D and Feng X:
Lipidomics reveals common mechanisms in polycystic ovarian
syndrome, recurrent spontaneous abortion, and infertility: A
genetic-based analysis. Int J Womens Health. 17:1055–1065. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Raviv S, Hantisteanu S, Sharon SM, Atzmon
Y, Michaeli M and Shalom-Paz E: Lipid droplets in granulosa cells
are correlated with reduced pregnancy rates. J Ovarian Res.
13:42020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu Q, Xie YJ, Qu LH, Zhang MX and Mo ZC:
Dyslipidemia involvement in the development of polycystic ovary
syndrome. Taiwan J Obstet Gynecol. 58:447–453. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang CH, Liu XY and Wang J: Essential
role of granulosa cell glucose and lipid metabolism on oocytes and
the potential metabolic imbalance in polycystic ovary Syndrome. Int
J Mol Sci. 24:162472023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bradley J, Pope I, Masia F, Sanusi R,
Langbein W, Swann K and Borri P: Quantitative imaging of lipids in
live mouse oocytes and early embryos using CARS microscopy.
Development. 143:2238–2247. 2016.PubMed/NCBI
|
|
32
|
Zhang C: Coherent Raman scattering
microscopy of lipid droplets in cells and tissues. J Raman
Spectrosc. 54:988–1000. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Inoue N, Nishida Y, Harada E, Sakai K and
Narahara H: GC-MS/MS analysis of metabolites derived from a single
human blastocyst. Metabolomics. 17:172021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Schwartz HT and Sternberg PW: A
sequencing-based screening method identifies regulators of EGFR
signaling from nonviable mutants in Caenorhabditis elegans. Sci
Signal. 18:eadp93772025. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
An L, Fu X, Chen J and Ma J: Application
of caenorhabditis elegans in lipid metabolism research. Int J Mol
Sci. 24:11732023. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wamaitha SE, Nie X, Pandolfi EC, Wang X,
Yang Y, Stukenborg JB, Cairns BR, Guo J and Clark AT: Single-cell
analysis of the developing human ovary defines distinct insights
into ovarian somatic and germline progenitors. Dev Cell.
58:2097–2111 e3. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ibayashi M, Aizawa R, Mitsui J and
Tsukamoto S: Homeostatic regulation of lipid droplet content in
mammalian oocytes and embryos. Reproduction. 162:R99–R109. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Purcell SH and Moley KH: The impact of
obesity on egg quality. J Assist Reprod Genet. 28:517–524. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Leary C, Leese HJ and Sturmey RG: Human
embryos from overweight and obese women display phenotypic and
metabolic abnormalities. Hum Reprod. 30:122–132. 2015. View Article : Google Scholar
|
|
40
|
Jiang M, Gao M, Wu C, He H, Guo X, Zhou Z,
Yang H, Xiao X, Liu G and Sha J: Lack of testicular seipin causes
teratozoospermia syndrome in men. Proc Natl Acad Sci USA.
111:7054–7059. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Khan R, Jiang X, Hameed U and Shi Q: Role
of lipid metabolism and signaling in mammalian oocyte maturation,
quality, and acquisition of competence. Front Cell Dev Biol.
9:6397042021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lipinska P, Pawlak P and Warzych E:
Species and embryo genome origin affect lipid droplets in
preimplantation embryos. Front Cell Dev Biol. 11:11878322023.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chao CF, Pesch YY, Yu H, Wang C,
Aristizabal MJ, Huan T, Tanentzapf G and Rideout E: An important
role for triglyceride in regulating spermatogenesis. Elife.
12:RP875232024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Aizawa R, Ibayashi M, Mitsui J and
Tsukamoto S: Lipid droplet formation is spatiotemporally regulated
in oocytes during follicular development in mice. J Reprod Dev.
70:18–24. 2024. View Article : Google Scholar :
|
|
45
|
Kerr JB and De Kretser DM: Cyclic
variations in Sertoli cell lipid content throughout the
spermatogenic cycle in the rat. J Reprod Fertil. 43:1–8. 1975.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Regueira M, Gorga A, Rindone GM,
Pellizzari EH, Cigorraga SB, Galardo MN, Riera MF and Meroni SB:
Apoptotic germ cells regulate Sertoli cell lipid storage and fatty
acid oxidation. Reproduction. 156:515–525. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Talbott HA, Plewes MR, Krause C, Hou X,
Zhang P, Rizzo WB, Wood JR, Cupp AS and Davis JS: Formation and
characterization of lipid droplets of the bovine corpus luteum. Sci
Rep. 10:112872020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Plewes MR, Talbott HA, Schott MB, Wood JR,
Cupp AS and Davis JS: Unraveling the role of lipid droplets and
perilipin 2 in bovine luteal cells. FASEB J. 38:e237102024.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Dunning KR, Russell DL and Robker RL:
Lipids and oocyte developmental competence: The role of fatty acids
and beta-oxidation. Reproduction. 148:R15–R27. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Schindler M, Pendzialek M, Grybel KJ,
Seeling T, Gürke J, Fischer B and Navarrete Santos A: Adiponectin
stimulates lipid metabolism via AMPK in rabbit blastocysts. Hum
Reprod. 32:1382–1392. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ma W, Yang X and Liang X: Obesity does not
aggravate vitrification injury in mouse embryos: A prospective
study. Reprod Biol Endocrinol. 10:682012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hallberg M, Morganstein DL, Kiskinis E,
Shah K, Kralli A, Dilworth SM, White R, Parker MG and Christian M:
A functional interaction between RIP140 and PGC-1alpha regulates
the expression of the lipid droplet protein CIDEA. Mol Cell Biol.
28:6785–6795. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Ning Z, Deng X, Li L, Feng J, Du X, Amevor
FK, Tian Y, Li L, Rao Y, Yi Z, et al: miR-128-3p regulates chicken
granulosa cell function via 14-3-3β/FoxO and PPAR-ү/LPL signaling
pathways. Int J Biol Macromol. 241:1246542023. View Article : Google Scholar
|
|
54
|
Cui L and Liu P: Two types of contact
between lipid droplets and mitochondria. Front Cell Dev Biol.
8:6183222020. View Article : Google Scholar :
|
|
55
|
Ma X, Qian H, Chen A, Ni HM and Ding WX:
Perspectives on mitochondria-ER and mitochondria-lipid droplet
contact in hepatocytes and hepatic lipid metabolism. Cells.
10:22732021. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Latchman NR, Stevens TL, Bedi KC, Prosser
BL, Margulies KB and Elrod JW: Ultrastructure analysis of
mitochondria, lipid droplet and sarcoplasmic reticulum apposition
in human heart failure. J Mol Cell Cardiol Plus.
13:1004612025.PubMed/NCBI
|
|
57
|
Liu L, Yang S, Liu Y, Li X, Hu J, Xiao L
and Xu T: DeepContact: High-throughput quantification of membrane
contact sites based on electron microscopy imaging. J Cell Biol.
221:e2021061902022. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Benador IY, Veliova M, Liesa M and
Shirihai OS: Mitochondria bound to lipid droplets: Where
mitochondrial dynamics regulate lipid storage and utilization. Cell
Metab. 29:827–835. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Freyre CAC, Rauher PC, Ejsing CS and Klemm
RW: MIGA2 links mitochondria, the ER, and lipid droplets and
promotes de novo lipogenesis in adipocytes. Mol Cell. 76:811–825
e14. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Herker E, Vieyres G, Beller M, Krahmer N
and Bohnert M: Lipid droplet contact sites in health and disease.
Trends Cell Biol. 31:345–358. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Fan H and Tan Y: Lipid
droplet-mitochondria contacts in health and disease. Int J Mol Sci.
25:68782024. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Monteiro-Cardoso VF and Giordano F:
Emerging functions of the mitochondria-ER-lipid droplet three-way
junction in coordinating lipid transfer, metabolism, and storage in
cells. FEBS Lett. 598:1252–1273. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Benador IY, Veliova M, Mahdaviani K,
Petcherski A, Wikstrom JD, Assali EA, Acín-Pérez R, Shum M,
Oliveira MF, Cinti S, et al: Mitochondria bound to lipid droplets
have unique bioenergetics, composition, and dynamics that support
lipid droplet expansion. Cell Metab. 27:869–885 e6. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Talari NK, Mattam U, Meher NK, Paripati
AK, Mahadev K, Krishnamoorthy T and Sepuri NBV: Lipid-droplet
associated mitochondria promote fatty-acid oxidation through a
distinct bioenergetic pattern in male Wistar rats. Nat Commun.
14:7662023. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Flores-Romero H, Ros U and Garcia-Saez AJ:
A lipid perspective on regulated cell death. Int Rev Cell Mol Biol.
351:197–236. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Danielsen ET, Moeller ME, Yamanaka N, Ou
Q, Laursen JM, Soenderholm C, Zhuo R, Phelps B, Tang K, Zeng J, et
al: A Drosophila genome-wide screen identifies regulators of
steroid hormone production and developmental timing. Dev Cell.
37:558–570. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Prates EG, Nunes JT and Pereira RM: A role
of lipid metabolism during cumulus-oocyte complex maturation:
impact of lipid modulators to improve embryo production. Mediators
Inflamm. 2014:6920672014. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Liu T, Qu J, Tian M, Yang R, Song X, Li R,
Yan J and Qiao J: Lipid metabolic process involved in oocyte
maturation during folliculogenesis. Front Cell Dev Biol.
10:8068902022. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Li T, Jin Y, Wu J and Ren Z: Beyond energy
provider: Multifunction of lipid droplets in embryonic development.
Biol Res. 56:382023. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Kajdasz A, Warzych E, Derebecka N, Madeja
ZE, Lechniak D, Wesoly J and Pawlak P: Lipid stores and lipid
metabolism associated gene expression in porcine and bovine
parthenogenetic embryos revealed by fluorescent staining and
RNA-seq. Int J Mol Sci. 21:64882020. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
de Andrade Melo-Sterza F and Poehland R:
Lipid metabolism in bovine oocytes and early embryos under in vivo,
in vitro, and stress conditions. Int J Mol Sci. 22:34212021.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Arena R, Bisogno S, Gasior L, Rudnicka J,
Bernhardt L, Haaf T, Zacchini F, Bochenek M, Fic K, Bik E, et al:
Lipid droplets in mammalian eggs are utilized during embryonic
diapause. Proc Natl Acad Sci USA. 118:e20183621182021. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Downs SM: Nutrient pathways regulating the
nuclear maturation of mammalian oocytes. Reprod Fertil Dev.
27:572–582. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Agarwal A, Gupta S and Sharma R: Oxidative
stress and its implications in female infertility-a clinician's
perspective. Reprod Biomed Online. 11:641–650. 2005. View Article : Google Scholar
|
|
75
|
Aizawa R, Ibayashi M, Tatsumi T, Yamamoto
A, Kokubo T, Miyasaka N, Sato K, Ikeda S, Minami N and Tsukamoto S:
Synthesis and maintenance of lipid droplets are essential for mouse
preimplantation embryonic development. Development.
146:dev1819252019. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Zhang JF, Choi SH, Li Q, Wang Y, Sun B,
Tang L, Wang EZ, Hua H and Li XZ: Overexpression of DGAT2
stimulates lipid droplet formation and triacylglycerol accumulation
in bovine satellite cells. Animals (Basel). 12:18472022. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Malyszka N, Pawlak P, Cieslak A,
Szkudelska K and Lechniak D: Distinct dynamics of lipid
accumulation by porcine cumulus cells during in vitro maturation
with follicular fluid of low and high fatty acid contents.
Theriogenology. 195:93–102. 2023. View Article : Google Scholar
|
|
78
|
Zhang RN, Fu XW, Jia BY, Liu C, Cheng KR
and Zhu SE: Expression of perilipin 2 (PLIN2) in porcine oocytes
during maturation. Reprod Domest Anim. 49:875–880. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Sowinska N, Lechtanska J, Greczka K,
Lechniak D and Pawlak P: Feline cumulus cells and oocytes show
massive accumulation of lipid droplets and upregulation of PLIN2
expression after in vitro maturation. Theriogenology. 232:70–78.
2025. View Article : Google Scholar
|
|
80
|
Monks J, Orlicky DJ, Libby AE,
Dzieciatkowska M, Ladinsky MS and McManaman JL: Perilipin-2
promotes lipid droplet-plasma membrane interactions that facilitate
apocrine lipid secretion in secretory epithelial cells of the mouse
mammary gland. Front Cell Dev Biol. 10:9585662022. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Xiang X, Chen J and Che L: Emerging roles
and therapeutic implications of lipid droplet protein perilipin 2
in liver disease. Genes Dis. 13:1017122025. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Morak M, Schmidinger H, Riesenhuber G,
Rechberger GN, Kollroser M, Haemmerle G, Zechner R, Kronenberg F
and Hermetter A: Adipose triglyceride lipase (ATGL) and
hormone-sensitive lipase (HSL) deficiencies affect expression of
lipolytic activities in mouse adipose tissues. Mol Cell Proteomics.
11:1777–1789. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Miyoshi H, Perfield JW II, Obin MS and
Greenberg AS: Adipose triglyceride lipase regulates basal lipolysis
and lipid droplet size in adipocytes. J Cell Biochem.
105:1430–1436. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Schweiger M, Schreiber R, Haemmerle G,
Lass A, Fledelius C, Jacobsen P, Tornqvist H, Zechner R and
Zimmermann R: Adipose triglyceride lipase and hormone-sensitive
lipase are the major enzymes in adipose tissue triacylglycerol
catabolism. J Biol Chem. 281:40236–40241. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Lass A, Zimmermann R, Haemmerle G,
Riederer M, Schoiswohl G, Schweiger M, Kienesberger P, Strauss JG,
Gorkiewicz G and Zechner R: Adipose triglyceride lipase-mediated
lipolysis of cellular fat stores is activated by CGI-58 and
defective in Chanarin-Dorfman Syndrome. Cell Metab. 3:309–319.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Gaidhu MP, Anthony NM, Patel P, Hawke TJ
and Ceddia RB: Dysregulation of lipolysis and lipid metabolism in
visceral and subcutaneous adipocytes by high-fat diet: role of
ATGL, HSL, and AMPK. Am J Physiol Cell Physiol. 298:C961–C971.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Turathum B, Gao EM and Chian RC: The
function of cumulus cells in oocyte growth and maturation and in
subsequent ovulation and fertilization. Cells. 10:22922021.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Richani D, Poljak A, Wang B, Mahbub SB,
Biazik J, Campbell JM, Habibalahi A, Stocker WA, Marinova MB, Nixon
B, et al: Oocyte and cumulus cell cooperativity and metabolic
plasticity under the direction of oocyte paracrine factors. Am J
Physiol Endocrinol Metab. 326:E366–E381. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Dumesic DA, Meldrum DR, Katz-Jaffe MG,
Krisher RL and Schoolcraft WB: Oocyte environment: Follicular fluid
and cumulus cells are critical for oocyte health. Fertil Steril.
103:303–316. 2015. View Article : Google Scholar
|
|
90
|
Xie J, Xu X and Liu S: Intercellular
communication in the cumulus-oocyte complex during
folliculogenesis: A review. Front Cell Dev Biol. 11:10876122023.
View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Uhde K, van Tol HTA, Stout TAE and Roelen
BAJ: Metabolomic profiles of bovine cumulus cells and
cumulus-oocyte-complex-conditioned medium during maturation in
vitro. Sci Rep. 8:94772018. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Russell DL, Gilchrist RB, Brown HM and
Thompson JG: Bidirectional communication between cumulus cells and
the oocyte: Old hands and new players? Theriogenology. 86:62–68.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Chen M, Yang W, Guo Y, Hou X, Zhu S, Sun
H, Guo X, Chen M and Wang Q: Multi-omics reveal the metabolic
patterns in mouse cumulus cells during oocyte maturation. J Ovarian
Res. 16:1562023. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Lawson GLG: Composition of Lipid Droplets
and Vesicles and their Relation to Oocyte Development Competency.
Reader K: BBiomedSc (Hons). University of Otago; 2023
|
|
95
|
Ordonez-Leon EA, Merchant H, Medrano A,
Kjelland M and Romo S: Lipid droplet analysis using in vitro bovine
oocytes and embryos. Reprod Domest Anim. 49:306–314. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Budani MC and Tiboni GM: Effects of
supplementation with natural antioxidants on oocytes and
preimplantation embryos. Antioxidants (Basel). 9:6122020.
View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Fayezi S, Leroy JLMR, Ghaffari Novin M and
Darabi M: Oleic acid in the modulation of oocyte and
preimplantation embryo development. Zygote. 26:1–13. 2018.
View Article : Google Scholar
|
|
98
|
Rakha SI, Elmetwally MA, El-Sheikh Ali H,
Balboula A, Mahmoud AM and Zaabel SM: Importance of antioxidant
supplementation during in vitro maturation of mammalian oocytes.
Vet Sci. 9:4392022.PubMed/NCBI
|
|
99
|
Guseva O, Kan N, Chekmareva V, Kokorev D,
Ilyasov P and Zhu YQ: The impact of antioxidant supplements on
oocytes and preimplantation embryos of humans and mammals, and
their potential application for mitigating the consequences of
oxidative stress in vitro: A review. Reproductive and Developmental
Medicine. 8:252–263. 2024. View Article : Google Scholar
|
|
100
|
Dubeibe Marin DF, da Costa NN, di Paula
Bessa Santana P, de Souza EB and Ohashi OM: Importance of lipid
metabolism on oocyte maturation and early embryo development: Can
we apply what we know to buffalo? Anim Reprod Sci. 211:1062202019.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Catandi GD, LiPuma L, Obeidat YM,
Maclellan LJ, Broeckling CD, Chen T, Chicco AJ and Carnevale EM:
Oocyte metabolic function, lipid composition, and developmental
potential are altered by diet in older mares. Reproduction.
163:183–198. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Chen H, Huang Y, Yang P, Shi Y, Ahmed N,
Liu T, Bai X, Haseeb A and Chen Q: Autophagy enhances lipid droplet
development during spermiogenesis in Chinese soft-shelled turtle,
Pelodiscus sinensis. Theriogenology. 147:154–165. 2020. View Article : Google Scholar
|
|
103
|
Oresti GM, Ayuza Aresti PL, Gigola G,
Reyes LE and Aveldano MI: Sequential depletion of rat testicular
lipids with long-chain and very long-chain polyenoic fatty acids
after X-ray-induced interruption of spermatogenesis. J Lipid Res.
51:2600–2610. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Liu M, Qi L, Zeng Y, Yang Y, Bi Y, Shi X,
Zhu H, Zhou Z and Sha J: Transient scrotal hyperthermia induces
lipid droplet accumulation and reveals a different ADFP expression
pattern between the testes and liver in mice. PLoS One.
7:e456942012. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Furland NE, Luquez JM, Oresti GM and
Aveldano MI: Mild testicular hyperthermia transiently increases
lipid droplet accumulation and modifies sphingolipid and
glycerophospholipid acyl chains in the rat testis. Lipids.
46:443–454. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Osuga J, Ishibashi S, Oka T, Yagyu H,
Tozawa R, Fujimoto A, Shionoiri F, Yahagi N, Kraemer FB, Tsutsumi O
and Yamada N: Targeted disruption of hormone-sensitive lipase
results in male sterility and adipocyte hypertrophy, but not in
obesity. Proc Natl Acad Sci USA. 97:787–792. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Zhang J, Hu Y, Wang Y, Fu L, Xu X, Li C,
Xu J, Li C, Zhang L, Yang R, et al: mmBCFA C17iso ensures
endoplasmic reticulum integrity for lipid droplet growth. J Cell
Biol. 220:e2021021222021. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Wang C, Wang B, Pandey T, Long Y, Zhang J,
Oh F, Sima J, Guo R, Liu Y, Zhang C, et al: A conserved
megaprotein-based molecular bridge critical for lipid trafficking
and cold resilience. Nat Commun. 13:68052022. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Dasso ME, Centola CL, Galardo MN, Riera MF
and Meroni SB: FSH increases lipid droplet content by regulating
the expression of genes related to lipid storage in Rat Sertoli
cells. Mol Cell Endocrinol. 595:1124032025. View Article : Google Scholar
|
|
110
|
Gorga A, Rindone GM, Regueira M,
Pellizzari EH, Camberos MC, Cigorraga SB, Riera MF, Galardo MN and
Meroni SB: PPARү activation regulates lipid droplet formation and
lactate production in rat Sertoli cells. Cell Tissue Res.
369:611–624. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Chen ZF, Shen YF, Gao DW, Lin DF, Ma WZ
and Chang DG: Metabolic pathways and male fertility: Exploring the
role of Sertoli cells in energy homeostasis and spermatogenesis. Am
J Physiol Endocrinol Metab. 329:E160–E178. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Breucker H, Schafer E and Holstein AF:
Morphogenesis and fate of the residual body in human
spermiogenesis. Cell Tissue Res. 240:303–309. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Guo C, Wang L, Cui K, Zhang G, Tan Y, Chen
W, Wang Y, Liu J, Liu W, Zhang G and Zhou Z: Lead causes lipid
droplet accumulation by impairing lysosomal function and autophagic
flux in testicular sertoli cells. Toxics. 13:1752025. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Rindone GM, Dasso ME, Centola CL,
Pellizzari EH, Camberos MDC, Toneatto J, Galardo MN, Meroni SB and
Riera MF: Sertoli cell adaptation to glucose deprivation: Potential
role of AMPK in the regulation of lipid metabolism. J Cell Biochem.
124:716–730. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Bush SJ, Nikola R, Han S, Suzuki S,
Yoshida S, Simons BD and Goriely A: Adult human, but not rodent,
spermatogonial stem cells retain states with a foetal-like
signature. Cells. 13:7422024. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Wang J, Chen M, Yao Y, Zhu M, Jiang Y,
Duan J, Yuan Y, Li L, Chen M and Sha J: Characterization of
metabolic patterns in mouse spermatogenesis and its clinical
implications in humans. Int J Mol Sci. 26:10012025. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Plant TM and Marshall GR: The functional
significance of FSH in spermatogenesis and the control of its
secretion in male primates. Endocr Rev. 22:764–786. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Guo J, Grow EJ, Mlcochova H, Maher GJ,
Lindskog C, Nie X, Guo Y, Takei Y, Yun J, Cai L, et al: The adult
human testis transcriptional cell atlas. Cell Res. 28:1141–1157.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Paniagua R, Nistal M, Saez FJ and Fraile
B: Ultrastructure of the aging human testis. J Electron Microsc
Tech. 19:241–260. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Li N, Mruk DD, Mok KW, Li MW, Wong CK, Lee
WM, Han D, Silvestrini B and Cheng CY: Connexin 43 reboots meiosis
and reseals blood-testis barrier following toxicant-mediated
aspermatogenesis and barrier disruption. FASEB J. 30:1436–1452.
2016. View Article : Google Scholar
|
|
121
|
Vo BT, Morton D Jr, Komaragiri S, Millena
AC, Leath C and Khan SA: TGF-beta effects on prostate cancer cell
migration and invasion are mediated by PGE2 through activation of
PI3K/AKT/mTOR pathway. Endocrinology. 154:1768–1779. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Sahin MB, Schwartz RE, Buckley SM,
Heremans Y, Chase L, Hu WS and Verfaillie CM: Isolation and
characterization of a novel population of progenitor cells from
unmanipulated rat liver. Liver Transpl. 14:333–345. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Richburg JH, Myers JL and Bratton SB: The
role of E3 ligases in the ubiquitin-dependent regulation of
spermatogenesis. Semin Cell Dev Biol. 30:27–35. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Zalata AA, Christophe AB, Depuydt CE,
Schoonjans F and Comhaire FH: The fatty acid composition of
phospholipids of spermatozoa from infertile patients. Mol Hum
Reprod. 4:111–118. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Fusaro I, Parrillo S, Buonaiuto G,
Prasinou P, Gramenzi A, Bucci R, Cavallini D, Carosi A, Carluccio A
and De Amicis I: Effects of hemp-based polyunsaturated fatty acid
supplementation on membrane lipid profiles and reproductive
performance in Martina Franca jacks. Front Vet Sci. 12:15532182025.
View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Johnson MR, Stephenson RA, Ghaemmaghami S
and Welte MA: Developmentally regulated H2Av buffering via dynamic
sequestration to lipid droplets in Drosophila embryos. Elife.
7:e360212018. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Byrne CJ, Fair S, English AM, Holden SA,
Dick JR, Lonergan P and Kenny DA: Dietary polyunsaturated fatty
acid supplementation of young post-pubertal dairy bulls alters the
fatty acid composition of seminal plasma and spermatozoa but has no
effect on semen volume or sperm quality. Theriogenology.
90:289–300. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Prochowska S, Bonarska-Kujawa D, Bobak L,
Eberhardt M and Nizanski W: Fatty acid composition and biophysical
characteristics of the cell membrane of feline spermatozoa. Sci
Rep. 14:102142024. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Liu Q, Zhou YF, Duan RJ, Wei HK, Peng J
and Jiang SW: Dietary n-6:n-3 ratio and Vitamin E improve motility
characteristics in association with membrane properties of boar
spermatozoa. Asian J Androl. 19:223–229. 2017. View Article : Google Scholar :
|
|
130
|
Tran LV, Malla BA, Sharma AN, Kumar S,
Tyagi N and Tyagi AK: Effect of omega-3 and omega-6 polyunsaturated
fatty acid enriched diet on plasma IGF-1 and testosterone
concentration, puberty and semen quality in male buffalo. Anim
Reprod Sci. 173:63–72. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Schlegel RA, Hammerstedt R, Cofer GP,
Kozarsky K, Freidus D and Williamson P: Changes in the organization
of the lipid bilayer of the plasma membrane during spermatogenesis
and epididymal maturation. Biol Reprod. 34:379–391. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Jabarineitapeh M, Naderi N, Tavalaee M and
Nasr-Esfahani MH: Effects of L-carnitine over-supplementation on
spermatogenesis and sperm function in healthy NMRI mice. Tissue
Cell. 96:1030142025. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Casado ME, Huerta L, Marcos-Diaz A, Ortiz
AI, Kraemer FB, Lasunción MA, Busto R and Martín-Hidalgo A:
Hormone-sensitive lipase deficiency affects the expression of
SR-BI, LDLr, and ABCA1 receptors/transporters involved in cellular
cholesterol uptake and efflux and disturbs fertility in mouse
testis. Biochim Biophys Acta Mol Cell Biol Lipids. 1866:1590432021.
View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Casado ME, Huerta L, Ortiz AI,
Pérez-Crespo M, Gutiérrez-Adán A, Kraemer FB, Lasunción MÁ, Busto R
and Martín-Hidalgo A: HSL-knockout mouse testis exhibits class B
scavenger receptor upregulation and disrupted lipid raft
microdomains. J Lipid Res. 53:2586–2597. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Nistal M, Jimenez F and Paniagua R:
Sertoli cell types in the Sertoli-cell-only syndrome: Relationships
between Sertoli cell morphology and aetiology. Histopathology.
16:173–180. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Chung KW and Hamilton JB: Testicular
lipids in mice with testicular feminization. Cell Tissue Res.
160:69–80. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Weinbauer GF, Respondek M, Themann H and
Nieschlag E: Reversibility of long-term effects of GnRH agonist
administration on testicular histology and sperm production in the
nonhuman primate. J Androl. 8:319–329. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Wang T, Xiao Y, Hu Z, Gu J, Hua R, Hai Z,
Chen X, Zhang JV, Yu Z, Wu T, et al: MFN2 deficiency impairs
mitochondrial functions and PPAR pathway during spermatogenesis and
meiosis in mice. Front Cell Dev Biol. 10:8625062022. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Griswold MD: The central role of Sertoli
cells in spermatogenesis. Semin Cell Dev Biol. 9:411–416. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Walther TC and Farese RV Jr: Lipid
droplets and cellular lipid metabolism. Annu Rev Biochem.
81:687–714. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Hermo L, Pelletier RM, Cyr DG and Smith
CE: Surfing the wave, cycle, life history, and genes/proteins
expressed by testicular germ cells. Part 1: Background to
spermatogenesis, spermatogonia, and spermatocytes. Microsc Res
Tech. 73:241–278. 2010. View Article : Google Scholar
|
|
142
|
Singh R, Kaushik S, Wang Y, Xiang Y, Novak
I, Komatsu M, Tanaka K, Cuervo AM and Czaja MJ: Autophagy regulates
lipid metabolism. Nature. 458:1131–1135. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Rato L, Alves MG, Socorro S, Duarte AI,
Cavaco JE and Oliveira PF: Metabolic regulation is important for
spermatogenesis. Nat Rev Urol. 9:330–338. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Regueira M, Riera MF, Galardo MN, Camberos
Mdel C, Pellizzari EH, Cigorraga SB and Meroni SB: FSH and bFGF
regulate the expression of genes involved in Sertoli cell energetic
metabolism. Gen Comp Endocrinol. 222:124–133. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Shi JF, Li YK, Ren K, Xie YJ, Yin WD and
Mo ZC: Characterization of cholesterol metabolism in Sertoli cells
and spermatogenesis (Review). Mol Med Rep. 17:705–713. 2018.
|
|
146
|
Guma FC, Wagner M, Martini LH and Bernard
EA: Effect of FSH and insulin on lipogenesis in cultures of Sertoli
cells from immature rats. Braz J Med Biol Res. 30:591–597. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Stocco C: Aromatase expression in the
ovary: hormonal and molecular regulation. Steroids. 73:473–487.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Kraemer FB and Shen WJ: Hormone-sensitive
lipase: control of intracellular tri-(di-)acylglycerol and
cholesteryl ester hydrolysis. J Lipid Res. 43:1585–1594. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Miller WL and Auchus RJ: The molecular
biology, biochemistry, and physiology of human steroidogenesis and
its disorders. Endocr Rev. 32:81–151. 2011. View Article : Google Scholar
|
|
150
|
Wu LL, Dunning KR, Yang X, Russell DL,
Lane M, Norman RJ and Robker RL: High-fat diet causes lipotoxicity
responses in cumulus-oocyte complexes and decreased fertilization
rates. Endocrinology. 151:5438–5445. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Yuan X, Zhang X, Lin Y, Xie H, Wang Z, Hu
X, Hu S, Li L, Liu H, He H, et al: Proteome of granulosa cells
lipid droplets reveals mechanisms regulating lipid metabolism at
hierarchical and pre-hierarchical follicle in goose. Front Vet Sci.
12:15447182025. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Ibayashi M and Tsukamoto S: Lipid droplet
biogenesis in the ovary. Reprod Med Biol. 23:e126182024. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Gao S, Gan X, He H, Hu S, Deng Y, Chen X,
Li L, Hu J, Li L and Wang J: Dynamic characteristics of lipid
metabolism in cultured granulosa cells from geese follicles at
different developmental stages. Biosci Rep. 39:BSR201921882019.
View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Su YQ, Sugiura K and Eppig JJ: Mouse
oocyte control of granulosa cell development and function:
Paracrine regulation of cumulus cell metabolism. Semin Reprod Med.
27:32–42. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Kidder GM and Vanderhyden BC:
Bidirectional communication between oocytes and follicle cells:
Ensuring oocyte developmental competence. Can J Physiol Pharmacol.
88:399–413. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Robker RL, Wu LL and Yang X: Inflammatory
pathways linking obesity and ovarian dysfunction. J Reprod Immunol.
88:142–148. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Russell DL and Robker RL: Molecular
mechanisms of ovulation: Co-ordination through the cumulus complex.
Hum Reprod Update. 13:289–312. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Mau KHT, Karimlou D, Barneda D, Brochard
V, Royer C, Leeke B, de Souza RA, Pailles M, Percharde M, Srinivas
S, et al: Dynamic enlargement and mobilization of lipid droplets in
pluripotent cells coordinate morphogenesis during mouse
peri-implantation development. Nat Commun. 13:38612022. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Kikuchi K, Ekwall H, Tienthai P, Kawai Y,
Noguchi J, Kaneko H and Rodriguez-Martinez H: Morphological
features of lipid droplet transition during porcine oocyte
fertilisation and early embryonic development to blastocyst in vivo
and in vitro. Zygote. 10:355–366. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Watanabe T, Thayil A, Jesacher A, Grieve
K, Debarre D, Wilson T, Booth M and Srinivas S: Characterisation of
the dynamic behaviour of lipid droplets in the early mouse embryo
using adaptive harmonic generation microscopy. BMC Cell Biol.
11:382010. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Welte MA: As the fat flies: The dynamic
lipid droplets of Drosophila embryos. Biochim Biophys Acta.
1851:1156–1185. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Li Z, Thiel K, Thul PJ, Beller M, Kuhnlein
RP and Welte MA: Lipid droplets control the maternal histone supply
of Drosophila embryos. Curr Biol. 22:2104–2113. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Crocco MC, Kelmansky DM and Mariano MI:
Does serum cause lipid-droplet accumulation in bovine embryos
produced in vitro, during developmental days 1 to 4? J Assist
Reprod Genet. 30:1377–1388. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Abe H, Yamashita S, Satoh T and Hoshi H:
Accumulation of cytoplasmic lipid droplets in bovine embryos and
cryotolerance of embryos developed in different culture systems
using serum-free or serum-containing media. Mol Reprod Dev.
61:57–66. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
van der Weijden VA, Stotzel M, Iyer DP,
Fauler B, Gralinska E, Shahraz M, Meierhofer D, Vingron M, Rulands
S, Alexandrov T, et al: FOXO1-mediated lipid metabolism maintains
mammalian embryos in dormancy. Nat Cell Biol. 26:181–193. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Dunning KR, Cashman K, Russell DL,
Thompson JG, Norman RJ and Robker RL: Beta-oxidation is essential
for mouse oocyte developmental competence and early embryo
development. Biol Reprod. 83:909–918. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Herms A, Bosch M, Reddy BJ, Schieber NL,
Fajardo A, Rupérez C, Fernández-Vidal A, Ferguson C, Rentero C,
Tebar F, et al: AMPK activation promotes lipid droplet dispersion
on detyrosinated microtubules to increase mitochondrial fatty acid
oxidation. Nat Commun. 6:71762015. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Babayev E and Seli E: Oocyte mitochondrial
function and reproduction. Curr Opin Obstet Gynecol. 27:175–181.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
May-Panloup P, Boucret L, Chao de la Barca
JM, Desquiret-Dumas V, Ferré-L'Hotellier V, Morinière C, Descamps
P, Procaccio V and Reynier P: Ovarian ageing: The role of
mitochondria in oocytes and follicles. Hum Reprod Update.
22:725–743. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Steinhauser CB, Askelson K, Lambo CA,
Hobbs KC, Bazer FW and Satterfield MC: Lipid metabolism is altered
in maternal, placental, and fetal tissues of ewes with small for
gestational age fetusesdagger. Biol Reprod. 104:170–180. 2021.
View Article : Google Scholar :
|
|
171
|
Wu LL, Russell DL, Wong SL, Chen M, Tsai
TS, St John JC, Norman RJ, Febbraio MA, Carroll J and Robker RL:
Mitochondrial dysfunction in oocytes of obese mothers: Transmission
to offspring and reversal by pharmacological endoplasmic reticulum
stress inhibitors. Development. 142:681–691. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Naowarojna N, Wu TW, Pan Z, Li M, Han JR
and Zou Y: Dynamic regulation of ferroptosis by lipid metabolism.
Antioxid Redox Signal. 39:59–78. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Motamedi S, Ravoet N, Dehairs J,
Vanderhoydonc F, Escamilla-Ayala A, Sliwinska MA, Wang S, Idkowiak
J, Soenen S, Agostinis P, et al: AMP-activated protein
kinase-driven lipid droplet dynamics govern melanoma sensitivity to
polyunsaturated fatty acid and iron-induced ferroptosis. Nat
Commun. 16:112522025. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Lange M, Wolk M, Li VW, Doubravsky CE,
Hendricks JM, Kato S, Otoki Y, Styler B, Johnson SL, Harris CA, et
al: FSP1-mediated lipid droplet quality control prevents neutral
lipid peroxidation and ferroptosis. Nat Cell Biol. 27:1902–1913.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Danielli M, Perne L, Jarc Jovicic E and
Petan T: Lipid droplets and polyunsaturated fatty acid trafficking:
Balancing life and death. Front Cell Dev Biol. 11:11047252023.
View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Lee H, Horbath A, Kondiparthi L, Meena JK,
Lei G, Dasgupta S, Liu X, Zhuang L, Koppula P, Li M, et al: Cell
cycle arrest induces lipid droplet formation and confers
ferroptosis resistance. Nat Commun. 15:792024. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Obaseki E, Adebayo D, Bandyopadhyay S and
Hariri H: Lipid droplets and fatty acid-induced lipotoxicity: In a
nutshell. FEBS Lett. 598:1207–1214. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Chen Z, Ho IL, Soeung M, Yen EY, Liu J,
Yan L, Rose JL, Srinivasan S, Jiang S, Edward Chang Q, et al: Ether
phospholipids are required for mitochondrial reactive oxygen
species homeostasis. Nat Commun. 14:21942023. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Virmani A, Pinto L, Bauermann O, Zerelli
S, Diedenhofen A, Binienda ZK, Ali SF and van der Leij FR: The
carnitine palmitoyl transferase (CPT) system and possible relevance
for neuropsychiatric and neurological conditions. Mol Neurobiol.
52:826–836. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Qu Q, Zeng F, Liu X, Wang QJ and Deng F:
Fatty acid oxidation and carnitine palmitoyltransferase I: Emerging
therapeutic targets in cancer. Cell Death Dis. 7:e22262016.
View Article : Google Scholar : PubMed/NCBI
|
|
181
|
De Paula IF, Santos-Araujo S, Majerowicz
D, Ramos I and Gondim KC: Knockdown of carnitine
palmitoyltransferase I (CPT1) reduces fat body lipid mobilization
and resistance to starvation in the insect vector Rhodnius
prolixus. Front Physiol. 14:12016702023. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Choi J, Smith DM, Scafidi S, Riddle RC and
Wolfgang MJ: Carnitine palmitoyltransferase 1 facilitates fatty
acid oxidation in a non-cell-autonomous manner. Cell Rep.
43:1150062024. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Dissanayake LV, Smith BA, Zietara A,
Levchenko V, Lowe M, Kravtsova O, Shapiro A, Upadhyay G, Halade GV,
Geurts AM, et al: The role of carnitine palmitoyl transferase 2 in
the progression of salt-sensitive hypertension. Am J Physiol Cell
Physiol. 329:C1188–C1202. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Xiang F, Zhang Z, Xie J, Xiong S, Yang C,
Liao D, Xia B and Lin L: Comprehensive review of the expanding
roles of the carnitine pool in metabolic physiology: Beyond fatty
acid oxidation. J Transl Med. 23:3242025. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Liang K: Mitochondrial CPT1A: Insights
into structure, function, and basis for drug development. Front
Pharmacol. 14:11604402023. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Knottnerus SJG, Bleeker JC, Wust RCI,
Ferdinandusse S, IJ L 1st, Wijburg FA, Wanders RJA, Visser G and
Houtkooper RH: Disorders of mitochondrial long-chain fatty acid
oxidation and the carnitine shuttle. Rev Endocr Metab Disord.
19:93–106. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Reader KL, Cox NR, Stanton JA and Juengel
JL: Effects of acetyl-L-carnitine on lamb oocyte blastocyst rate,
ultrastructure, and mitochondrial DNA copy number. Theriogenology.
83:1484–1492. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Knitlova D, Hulinska P, Jeseta M,
Hanzalova K, Kempisty B and Machatkova M: Supplementation of
l-carnitine during in vitro maturation improves embryo development
from less competent bovine oocytes. Theriogenology. 102:16–22.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Kilwein MD, Dao TK and Welte MA:
Drosophila embryos allocate lipid droplets to specific lineages to
ensure punctual development and redox homeostasis. PLoS Genet.
19:e10108752023. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Bailey AP, Koster G, Guillermier C, Hirst
EM, MacRae JI, Lechene CP, Postle AD and Gould AP: Antioxidant role
for lipid droplets in a stem cell niche of Drosophila. Cell.
163:340–353. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Kim K, Park S and Roh S: Lipid-rich
blastomeres in the two-cell stage of porcine parthenotes show bias
toward contributing to the embryonic part. Anim Reprod Sci.
130:91–98. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Zhang J, Khvorostov I, Hong JS, Oktay Y,
Vergnes L, Nuebel E, Wahjudi PN, Setoguchi K, Wang G, Do A, et al:
UCP2 regulates energy metabolism and differentiation potential of
human pluripotent stem cells. EMBO J. 30:4860–4873. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Folmes CD, Nelson TJ, Martinez-Fernandez
A, Arrell DK, Lindor JZ, Dzeja PP, Ikeda Y, Perez-Terzic C and
Terzic A: Somatic oxidative bioenergetics transitions into
pluripotency-dependent glycolysis to facilitate nuclear
reprogramming. Cell Metab. 14:264–271. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Yanes O, Clark J, Wong DM, Patti GJ,
Sánchez-Ruiz A, Benton HP, Trauger SA, Desponts C, Ding S and
Siuzdak G: Metabolic oxidation regulates embryonic stem cell
differentiation. Nat Chem Biol. 6:411–417. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Shiraki N, Shiraki Y, Tsuyama T, Obata F,
Miura M, Nagae G, Aburatani H, Kume K, Endo F and Kume S:
Methionine metabolism regulates maintenance and differentiation of
human pluripotent stem cells. Cell Metab. 19:780–794. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Dunning KR, Anastasi MR, Zhang VJ, Russell
DL and Robker RL: Regulation of fatty acid oxidation in mouse
cumulus-oocyte complexes during maturation and modulation by PPAR
agonists. PLoS One. 9:e873272014. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Brasaemle DL: Thematic review series:
Adipocyte biology. The perilipin family of structural lipid droplet
proteins: Stabilization of lipid droplets and control of lipolysis.
J Lipid Res. 48:2547–2559. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Horton JD, Goldstein JL and Brown MS:
SREBPs: Activators of the complete program of cholesterol and fatty
acid synthesis in the liver. J Clin Invest. 109:1125–1131. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Fan W and Evans R: PPARs and ERRs:
Molecular mediators of mitochondrial metabolism. Curr Opin Cell
Biol. 33:49–54. 2015. View Article : Google Scholar
|
|
200
|
Sayers NS, Anujan P, Yu HN, Palmer SS,
Nautiyal J, Franks S and Hanyaloglu AC: Follicle-stimulating
hormone induces lipid droplets via Galphai/o and β-Arrestin in an
endometrial cancer cell line. Front Endocrinol (Lausanne).
12:7988662021. View Article : Google Scholar
|
|
201
|
Igosheva N, Abramov AY, Poston L, Eckert
JJ, Fleming TP, Duchen MR and McConnell J: Maternal diet-induced
obesity alters mitochondrial activity and redox status in mouse
oocytes and zygotes. PLoS One. 5:e100742010. View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Shen WJ, Patel S, Natu V, Hong R, Wang J,
Azhar S and Kraemer FB: Interaction of hormone-sensitive lipase
with steroidogenic acute regulatory protein: Facilitation of
cholesterol transfer in adrenal. J Biol Chem. 278:43870–43876.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Ludovic, Le, Corre, et al: Bisphenol A
disrupts the intestinal lipid metabolism. Toxicology Letters.
211:S2102012. View Article : Google Scholar
|
|
204
|
Vit FF, Sangalli JR, Fiorenza MF, de
Almeida Saraiva HFR, Ferst JG, Perecin F, Meirelles FV, de la Torre
LG and da Silveira JC: Dynamic microdevice culture during bovine
oocyte maturation decreases lipid accumulation and improve
blastocyst cell numbers. Sci Rep. 16:2322025. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Zhu J, Lam SM, Yang L, Liang J, Ding M,
Shui G and Huang X: Reduced phosphatidylcholine synthesis
suppresses the embryonic lethality of seipin deficiency. Life
Metab. 1:175–189. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Dumesic DA, Padmanabhan V and Abbott DH:
Polycystic ovary syndrome and oocyte developmental competence.
Obstet Gynecol Surv. 63:39–48. 2008. View Article : Google Scholar
|
|
207
|
Sturmey RG and Leese HJ: Energy metabolism
in pig oocytes and early embryos. Reproduction. 126:197–204. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Horn PJ, Ledbetter NR, James CN, Hoffman
WD, Case CR, Verbeck GF and Chapman KD: Visualization of lipid
droplet composition by direct organelle mass spectrometry. J Biol
Chem. 286:3298–3306. 2011. View Article : Google Scholar
|
|
209
|
Ding Y, Jiang Y, Zhu M, Zhu Q, He Y, Lu Y,
Wang Y, Qi J, Feng Y, Huang R, et al: Follicular fluid lipidomic
profiling reveals potential biomarkers of polycystic ovary
syndrome: A pilot study. Front Endocrinol (Lausanne).
13:9602742022. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Zhang M, Wang Y, Di J, Zhang X, Liu Y,
Zhang Y, Li B, Qi S, Cao X, Liu L, et al: High coverage of targeted
lipidomics revealed lipid changes in the follicular fluid of
patients with insulin-resistant polycystic ovary syndrome and a
positive correlation between plasmalogens and oocyte quality. Front
Endocrinol (Lausanne). 15:14142892024. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Ruebel ML, Piccolo BD, Mercer KE, Pack L,
Moutos D, Shankar K and Andres A: Obesity leads to distinct
metabolomic signatures in follicular fluid of women undergoing in
vitro fertilization. Am J Physiol Endocrinol Metab. 316:E383–E396.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Gao J, Liu M, Liu J, Shi P, Cui H, Zhao S,
Zhang X and Tao C: Effect of high-fat diet on the lipid profile of
ovarian granulosa cells and female reproduction in mice. PLoS One.
18:e02875342023. View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Cao Z, Hao Y, Fung CW, Lee YY, Wang P, Li
X, Xie K, Lam WJ, Qiu Y, Tang BZ, et al: Dietary fatty acids
promote lipid droplet diversity through seipin enrichment in an ER
subdomain. Nat Commun. 10:29022019. View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Schmiedel JM, Klemm SL, Zheng Y, Sahay A,
Blüthgen N, Marks DS and van Oudenaarden A: Gene expression.
MicroRNA control of protein expression noise. Science. 348:128–132.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Mak HY: Lipid droplets as fat storage
organelles in Caenorhabditis elegans: Thematic review series: Lipid
droplet synthesis and metabolism: From Yeast To Man. J Lipid Res.
53:28–33. 2012. View Article : Google Scholar
|
|
216
|
Kumari RM, Khatri A, Chaudhary R and
Choudhary V: Concept of lipid droplet biogenesis. Eur J Cell Biol.
102:1513622023. View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Meister P, Towbin BD, Pike BL, Ponti A and
Gasser SM: The spatial dynamics of tissue-specific promoters during
C. elegans development. Genes Dev. 24:766–782. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Frezal L and Felix MA: C. elegans outside
the Petri dish. Elife. 4:e058492015. View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Schmokel V, Memar N, Wiekenberg A,
Trotzmuller M, Schnabel R and Doring F: Genetics of lipid-storage
management in caenorhabditis elegans embryos. Genetics.
202:1071–1083. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Vrablik TL, Petyuk VA, Larson EM, Smith RD
and Watts JL: Lipidomic and proteomic analysis of Caenorhabditis
elegans lipid droplets and identification of ACS-4 as a lipid
droplet-associated protein. Biochim Biophys Acta. 1851:1337–1345.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Liu Y, Xu S, Zhang C, Zhu X, Hammad MA,
Zhang X, Christian M, Zhang H and Liu P: Hydroxysteroid
dehydrogenase family proteins on lipid droplets through bacteria,
C. elegans, and mammals. Biochim Biophys Acta Mol Cell Biol Lipids.
1863:881–894. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Mondal M, Scifo E, Ciliberti RE, Wischhof
L, Abbariki TN, Jackson J, Menegatou IM, Zeisler-Diehl V, Riemer J,
Jussila B, et al: Dietary lipid content modifies
wah-1/AIFM1-associated phenotypes via LRK-1 and DRP-1 expression in
C. elegans. Nat Commun. 16:108172025. View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Campbell D and Zuryn S: The mechanisms and
roles of mitochondrial dynamics in C. elegans. Semin Cell Dev Biol.
156:266–275. 2024. View Article : Google Scholar
|
|
224
|
Mullaney BC and Ashrafi K: C. elegans fat
storage and metabolic regulation. Biochim Biophys Acta.
1791:474–478. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Lynn DA, Dalton HM, Sowa JN, Wang MC,
Soukas AA and Curran SP: Omega-3 and -6 fatty acids allocate
somatic and germline lipids to ensure fitness during nutrient and
oxidative stress in Caenorhabditis elegans. Proc Natl Acad Sci USA.
112:15378–15383. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Li Q, Zhou X, Zhang X, Zhang C and Zhang
SO: Nuclear receptor signaling regulates compartmentalized
phosphatidylcholine remodeling to facilitate thermosensitive lipid
droplet fusion. Nat Commun. 16:39552025. View Article : Google Scholar : PubMed/NCBI
|





















