Introduction
Diabetic retinopathy (DR) and retinal vein occlusion
(RVO) are the leading causes of preventable vision loss worldwide
(1,2). Despite their differing etiologies,
both disorders are fundamentally characterized by dysfunction of
the blood-retinal barrier (BRB), which drives core pathological
processes, including increased vascular permeability, retinal
neurodegeneration and macular edema (3). In advanced stages, severe
complications such as vitreous hemorrhage and neovascular glaucoma
may emerge, culminating in irreversible visual impairment (4,5).
The existing therapeutic strategies, including administration of
intravitreal anti-VEGF agents and laser photocoagulation, are
typically initiated at advanced disease stages and yield limited
functional recovery (6,7). Therefore, elucidating the molecular
mechanisms underlying BRB disruption and its effects on
neurovascular homeostasis are crucial for the development of
effective early interventions.
The neurovascular unit (NVU) is an intricate
microanatomical and functional structure composed of vascular
endothelial cells, the basement membrane, pericytes, astrocytes,
neurons and microglia (8).
Within this unit, the BRB is mainly formed by endothelial cells,
the basement membrane and pericytes, which tightly regulate retinal
blood flow and the transport of essential substances (9). Analogous to the blood-brain
barrier, the BRB is a highly specialized structure that protects
the neural retina from potentially harmful circulating substances
(10). Anatomically, the BRB is
composed of two layers: The outer BRB, which is formed by retinal
pigment epithelial cells, and the inner BRB (iBRB), which is
primarily composed of endothelial cells connected by tight
junctions (11). Disruption of
the iBRB, particularly under ischemic or inflammatory conditions,
is a major contributor to disease progression (12). The functioning of the BRB relies
on coordinated interactions among endothelial cells, pericytes,
neurons and glial cells within the NVU, highlighting that vascular
integrity and neural health are tightly linked (13,14). Endothelial cells govern vascular
permeability and hemodynamic regulation, pericytes provide
structural and functional support to the microvasculature, and the
extracellular matrix (ECM) provides essential mechanical support
and biochemical signaling cues (15). Perturbation of any of these
constituent elements can precipitate neurovascular dysfunction and
compromise both vascular integrity and neuronal viability (16,17). Under physiological conditions,
endothelial permeability is tightly controlled; however,
pathological stimuli induce junctional disassembly and subsequent
vascular leakage (18).
Furthermore, pericyte loss destabilizes the microvasculature,
whereas ECM remodeling exacerbates BRB breakdown (19-21).
Unc-5 netrin receptor B (UNC5B), a member of the
UNC5 family of netrin receptors, is predominantly expressed in
endothelial cells and functions as a receptor for netrin-1, which
regulates neuronal migration, apoptosis and vascular development
(22-24). Notably, netrin-1 exerts
concentration-dependent effects on vascular cells. At low
concentrations, netrin-1 binds to CD146 to promote endothelial
proliferation and VEGF expression, whereas at high concentrations,
it preferentially interacts with UNC5B to inhibit proliferation and
suppress VEGF production (25-27). Genetic ablation or functional
impairment of UNC5B abolishes these regulatory actions,
highlighting its indispensable role in maintaining vascular
function and NVU homeostasis (28).
The BRB and the NVU serve essential roles in
maintaining retinal homeostasis. Disruption of BRB integrity and
NVU dysfunction are hallmarks of retinal vascular diseases such as
DR and RVO (29,30). UNC5B, an endothelial receptor,
has been implicated in vascular and neural regulation (31,32), but its role in BRB and NVU
homeostasis in these disease contexts remains unclear. The present
study aimed to investigate the function of endothelial UNC5B in
maintaining BRB integrity and NVU homeostasis, using both in
vitro cell cultures, and in vivo DR and RVO mouse
models.
Materials and methods
Single-cell RNA sequencing (scRNA-seq)
analysis
scRNA-seq data (GSE178121), including the raw count
matrix and metadata with cell type annotations and experimental
conditions, were downloaded from the Gene Expression Omnibus
database (https://www.ncbi.nlm.nih.gov/geo/) (33). The dataset included one control
retinal sample and one streptozotocin (STZ)-induced DR retinal
sample, each of which was generated by pooling retinas from three
mice. Data preprocessing was performed using the Seurat package
(v4.1.1; https://satijalab.org/seurat/) in R software (v4.5.1;
R Foundation for Statistical Computing). Low-quality cells and
genes were filtered out on the basis of the gene count (200-2,500)
and mitochondrial content (<5%). The data were log-normalized,
and 2,000 highly variable genes were selected for principal
component analysis. The top 50 principal components were used for
clustering and t-distributed stochastic neighbor embedding
visualization. Cell clusters were visualized using DimPlot, and
DotPlot was used to show the expression of selected genes across
clusters (both functions were provided in the Seurat package;
v4.1.1). Clusters were annotated into retinal cell types based on
the expression of known marker genes, by comparison with public
databases, including PanglaoDB (https://panglaodb.se/) and the Mouse Cell Atlas
(https://bis.zju.edu.cn/MCA/), as well as
relevant published literature (33,34). Following cell type annotation,
endothelial cells were selected and further divided into
UNC5B-positive and UNC5B-negative groups on the basis of UNC5B
expression (raw count >0). To assess differences in pathway
activity, gene set variation analysis (GSVA; v1.44.5; https://bioconductor.org/packages/release/bioc/html/GSVA.html)
was performed at the single-cell level using the Kyoto Encyclopedia
of Genes and Genomes (KEGG; https://www.genome.jp/kegg/) and Gene Ontology (GO;
https://geneontology.org/) Biological Process
(BP) gene sets. Pathway-level enrichment scores were compared
between groups using the Wilcoxon rank-sum test with the
Benjamini-Hochberg correction for multiple testing. Significantly
enriched pathways (adjusted P<0.05) were visualized using bubble
plots.
Cell culture
Immortalized human retinal microvascular endothelial
cells (HRMECs; cat. no. XY-H469; Shanghai Xinyu Biotechnology Co.,
Ltd.) were used for high-glucose stimulation and UNC5B knockdown
experiments, while immortalized human retinal microvascular
pericytes (HRMVPCs; cat. no. ZQY001; Shanghai Zhongqiao Xinzhou
Biotechnology Co., Ltd.) were used for UNC5B knockdown experiments
and co-culture experiments with endothelial cells. HRMECs and
HRMVPCs were cultured in DMEM (cat. no. 11965092; Gibco; Thermo
Fisher Scientific, Inc.) containing 10% FBS (cat. no. 10099141C;
Gibco; Thermo Fisher Scientific, Inc.) and 1%
antibiotic-antimycotic solution (penicillin-streptomycin; cat. no.
15140122; Gibco; Thermo Fisher Scientific, Inc.) at 37°C in a
humidified atmosphere containing 5% CO2. For
high-glucose treatment, HRMECs were exposed to 30 mM glucose for 24
or 48 h at 37°C to mimic hyperglycemic conditions. Cell function
assays and reverse transcription-quantitative PCR (RT-qPCR) were
performed using five biological replicates. Western blot
experiments were conducted using four biological replicates.
Transcriptome sequencing was performed in triplicates.
Transduction with UNC5B short hairpin RNA
(shRNA) in vitro
The lentiviral expression vectors for human UNC5B
shRNA were developed and produced by Shanghai GeneChem Co., Ltd.
The shRNA targeting sequence for UNC5B was
5'-CTACGAGATGTATCTACTCAT-3', while the scrambled control sequence
was 5'-TTCTCCGAACGTGTCACGT-3'. These sequences were cloned into the
GV112 lentiviral vector. Lentiviral particles were generated using
a three-plasmid, second-generation packaging system. Briefly, 293T
cells used for viral packaging were provided by the manufacturer as
part of this service, and were co-transfected with the transfer
vector and two helper plasmids (pHelper 1.0 and pHelper 2.0) at a
mass ratio of 4:3:2 (total 45 μg DNA per 10-cm dish).
Transfection was carried out at 37°C for 6 h before the medium was
replaced. Viral particles were harvested 48-72 h post-transfection
and concentrated via ultracentrifugation. Briefly, the supernatant
was cleared of debris by centrifugation at 4°C at 4,000 × g for 10
min, filtered through a 0.45-μm filter, and ultracentrifuged
at 100,000 × g for 2 h at 4°C. The resulting viral pellet was
resuspended in virus preservation solution and briefly centrifuged
at 10,000 × g at 4°C for 5 min before use. HRMECs or HRMVPCs were
seeded in six-well tissue culture plates at a subconfluent density
of 60% and transduced with lentiviral particles at an MOI of 50.
After incubation for 24 h at 37°C, the medium was refreshed. Stable
cell lines were selected using puromycin (1.0 μg/ml; cat.
no. ST551; Beyotime Biotechnology) for 7-8 days. Following the
establishment of stable colonies, the cells were maintained in
medium containing a maintenance concentration of 0.5 μg/ml
puromycin. Successful transduction was confirmed by the observation
of green fluorescence under a fluorescence microscope. The
expression levels of UNC5B in the stably transduced cells were
quantified by RT-qPCR. As a control, parallel cultures of cells
were infected with nonsense-scrambled shRNA lentiviral
particles.
Propidium iodide (PI) and calcein-AM
double staining
HRMECs or HRMVPCs with or without UNC5B knockdown
were seeded in 24-well plates and cultured at 37°C overnight in
DMEM containing 10% FBS to allow cell attachment. The cells were
stained with a 1:1 mixture of PI and calcein-AM dye (cat. no.
C1371; Beyotime Biotechnology) at 37°C for 10 min. Fluorescence
microscopy (Olympus Corporation) was then employed to visualize the
staining patterns, with viable cells emitting green fluorescence
under a 490-nm excitation filter and necrotic cells emitting red
fluorescence under a 545-nm excitation filter.
Uptake of bovine serum albumin (BSA) in
vitro
The albumin uptake assay was adapted from previously
published protocols (35,36)
with minor modifications. HRMECs were seeded in 24-well culture
plates at a density of 5×104 cells per well in 500
μl medium. Subsequently, the cells were incubated with 50
μg/ml rhodamine B isothiocyanate-labeled BSA (cat. no.
SR063; Beijing Solarbio Science & Technology Co., Ltd.) at 37°C
for 6 h in a medium containing 10% FBS but without antibiotics to
avoid interference. Following incubation, the culture medium was
removed, and the cells were gently washed with cold PBS (cat. no.
BL302A; Biosharp Life Sciences) to remove any unbound or excess
BSA. Subsequently, the cells were fixed at room temperature for 15
min in 4% paraformaldehyde (PFA; cat. no. BL539A; Biosharp Life
Sciences) to preserve their morphological integrity and
fluorescence properties for fluorescence imaging. After fixation,
the nuclei were stained with DAPI (cat. no. C1002; Beyotime
Biotechnology) for 5 min at room temperature, followed by three
washes with PBS before fluorescence imaging.
Permeability of HRMEC monolayers
Transwell multiplates containing cell culture
inserts with a polycarbonate membrane at the lower aperture (pore
size, 0.4 μm; diameter, 6.5 mm; cat. no. 353090; Corning,
Inc.) were used. HRMECs with or without UNC5B knockdown were seeded
at a density of 2.0×105 cells/cm2 on the
membranes (upper chambers) in standard culture medium containing
10% FBS and 1% antibiotic-antimycotic solution. The lower chambers
were filled with the same culture medium. The cells were cultured
at 37°C to form a confluent monolayer within 2 days of seeding. On
day 3 after seeding, 50 μg/ml FITC-conjugated dextran-40 kDa
(cat. no. FD40S; MilliporeSigma) was added to the upper chamber.
Dextran-40 kDa, a macromolecular tracer, was chosen because of its
ability to approximate the size of macromolecules commonly
encountered under physiological conditions (37). The cells were incubated with this
tracer at 37°C for 6 h to allow sufficient time for permeation
across the endothelial monolayer. Subsequently, the medium in the
lower chambers was carefully collected, and its absorbance was
measured using a microplate reader (Thermo Scientific Multiskan
SkyHigh Microplate Spectrophotometer; Thermo Fisher Scientific,
Inc.) as a quantitative indicator of monolayer permeability.
Matrigel co-culture assay
For the Matrigel co-culture assay, 24-well plates
were primed with Matrigel (cat. no. 356234; Corning, Inc.) and
subsequently incubated at 37°C for 30 min to facilitate the
polymerization of Matrigel matrices. HRMECs (6×104 cells
per well) and HRMVPCs (4×104 cells per well) were seeded
onto the 24-well plates pre-coated with Matrigel and covered with
DMEM supplemented with 10% FBS (38). After a culture period of 6 h at
37°C, the cells were fixed in 4% PFA at room temperature for 15
min. Subsequently, the endothelial cells were fluorescently labeled
with isolectin B4 (IB4; 1:50; cat. no. I21411; Thermo Fisher
Scientific, Inc.), whereas pericytes were stained with an
anti-neuron-glial antigen 2 (NG2) antibody (1:200; cat. no.
sc-53389; Santa Cruz Biotechnology, Inc.) at 4°C overnight. After
washing using PBS, appropriate fluorescent secondary antibodies
were applied at room temperature for 1 h. The antibodies used are
listed in Table SI. The
co-localization of these two fluorophores indicated the recruitment
of pericytes to endothelial cells, and was visualized under a
fluorescence microscope.
In vitro BRB permeability assay
An in vitro BRB model was established using
Transwell inserts with 0.4-μm polycarbonate membranes
(39). HRMVPCs were seeded at a
density of 3.0×104 cells/cm2 on the underside
of the membrane in inverted inserts and incubated for 1-2 h at 37°C
to allow adhesion. The inserts were then returned to the upright
position, and HRMECs were seeded on the upper side at a density of
9.0×104 cells/cm2. Co-cultures were
maintained for 48-72 h at 37°C until confluent monolayers formed on
both sides. To assess permeability, the upper chamber was washed
using PBS, and standard culture medium containing 50 μg/ml
FITC-dextran was added, while the lower chamber received medium
without dye. After 6 h of incubation at 37°C, medium (100
μl) from the lower chamber was collected, and the
fluorescence intensity was measured using a microplate reader to
evaluate FITC-dextran diffusion across the BRB model.
5-Ethynyl-2'-deoxyuridine (EdU)
staining
An EdU proliferation kit (cat. no. C0075; Beyotime
Biotechnology) was used to assess cell proliferation. HRMVPCs were
incubated with EdU solution (10 μM) at 37°C for 2 h.
Subsequently, the cells were fixed with 4% PFA at room temperature
for 15 min, washed using PBS, and co-stained with DAPI at room
temperature for 10 min. Finally, the cells were visualized under a
fluorescence microscope and analyzed using ImageJ software (v1.53;
National Institutes of Health).
In vitro cell migration assays
HRMVPCs (with or without UNC5B knockdown) seeded at
a density of 5×104 cells per chamber in serum-free DMEM
were added to the apical compartment of Transwell inserts featuring
a membrane with 8-μm pores (cat. no. 353097; Corning, Inc.).
The basal chamber was filled with complete medium containing 10%
FBS. Subsequently, the Transwell chambers were incubated in a
humidified environment maintained at 37°C with 5% CO2.
After 24 h, the migrated cells were fixed with 4% PFA for 15 min at
room temperature, stained with crystal violet at room temperature
for 1 min and imaged under a light microscope for quantitative
analysis.
RT-qPCR
Total cellular and tissue RNA was extracted using
the FastPure Cell/Tissue Total RNA Isolation Kit V2 (cat. no.
RC112-01; Vazyme Biotech Co., Ltd.). HiScript IV RT SuperMix for
qPCR (+gDNA wiper) (cat. no. R423-01; Vazyme Biotech Co., Ltd.) was
used to transcribe cDNA. Reverse transcription was performed at
50°C for 5 min, followed by 85°C for 5 sec, according to the
manufacturer's instructions. The reaction mixture contained 1
μl cDNA template, 1 μl of primers, 4 μl
diethyl pyrocarbonate-treated water and 4 μl 2X SYBR Green
PCR Mix (cat. no. Q711-02; Vazyme Biotech Co., Ltd.). RT-qPCR was
performed using a real-time PCR system (Thermo Fisher Scientific,
Inc.) with the following thermocycling conditions: Initial
denaturation at 95°C for 30 sec, followed by 40 cycles of 95°C for
10 sec and 60°C for 30 sec. Melting curves were generated to verify
the specificity. Relative mRNA expression levels were quantified
using the comparative Ct method (2−ΔΔCq) (40), with β-actin tested as the
reference gene. The primer sequences used for RT-qPCR are listed in
Table SII.
Western blot analyses
Cellular and tissue lysates (20-30 μg per
treatment) were prepared using RIPA lysis buffer (cat. no. P0013B;
Beyotime Biotechnology) supplemented with protease inhibitor
cocktail (cat. no. HY-K0011; MedChemExpress). Protein
concentrations were determined using the BCA assay (cat. no. 23225;
Thermo Fisher Scientific, Inc.). Equal amounts of protein (20-30
μg per lane) were separated by 6-15% SDS-PAGE and then
transferred to a PVDF membrane. The membranes were blocked with 5%
nonfat milk for 2 h at room temperature to prevent nonspecific
binding. Primary antibodies were then applied, and the membranes
were incubated overnight at 4°C to ensure optimal binding to target
proteins. After thorough washing with 1X Tris-Buffered Saline with
0.5% Tween-20 (cat. no. T1085; Beijing Solarbio Science &
Technology Co., Ltd.), the membranes were incubated with secondary
antibodies for 1 h at room temperature. Protein bands were detected
using an enhanced chemiluminescence substrate (cat. no. P0018AS;
Beyotime Biotechnology). Protein bands were visualized using a
Tanon 5200 series fully automated chemiluminescence imaging system
(Tanon Science and Technology Co., Ltd.), which allowed a clear and
quantifiable assessment of target protein expression. Band
intensities were semi-quantified using ImageJ software. The
antibodies used for western blotting are listed in Table SI.
Bulk RNA sequencing analysis
Bulk RNA sequencing analysis was performed using
transcriptomic data obtained from HRMECs with and without UNC5B
knockdown. HRMECs transduced with UNC5B shRNA (knockdown group) or
scramble shRNA (control group) were harvested at 80-90% confluence.
Cells were washed with cold PBS and lysed directly in TRIzol
Reagent (cat. no. 15596026; Thermo Fisher Scientific, Inc.).
Lysates were aliquoted into RNase-free tubes, snap-frozen in liquid
nitrogen, and stored at −80°C until shipment on dry ice to HaploX
Ltd. for library preparation and sequencing. RNA quality and
integrity were assessed using a NanoDrop™ One/OneC (OD260/280 and
OD260/230), Qubit® 3.0 Fluorometer (Thermo Fisher
Scientific, Inc.) with the Qubit™ RNA HS Assay Kit (cat. no.
Q32852; Thermo Fisher Scientific, Inc.) for precise quantification,
and Agilent 4200 TapeStation (Agilent Technologies, Inc.) for RNA
integrity number assessment. Libraries were prepared according to
the manufacturer's instructions. Library quality was validated
using an Agilent 2100 Bioanalyzer (Agilent Technologies, Inc.) to
check the insert size, and quantified via RT-qPCR to ensure an
effective concentration of >2 nM. Sequencing was performed on an
Illumina, Inc., platform with paired-end 150-bp reads. Raw
sequencing data were processed using HaploX Ltd.'s in-house
open-source software fastp (v0.23.0; https://github.com/OpenGene/fastp) (41). Processing steps included adaptor
removal, filtering of reads containing a high proportion of N bases
(default, 5 bp), removal of reads with >40% low-quality bases
(Phred score, ≤20) and sliding-window trimming (window size, 4 bp;
average quality threshold, 20). Differential expression analysis
was conducted using the limma package (v3.52.2; https://bioconductor.org/packages/release/bioc/html/limma.html)
in R, with genes filtered on the basis of adjusted P<0.05 and
|log2 fold change|>0.5. Significantly differentially
expressed genes were further subjected to GO and KEGG enrichment
analysis using clusterProfiler (v4.0.0; https://bioconductor.org/packages/release/bioc/html/clusterProfiler.html)
and the enrichment results were visualized using ggplot2 (v3.3.5;
https://cran.r-project.org/web/packages/ggplot2/index.html).
Animals
All animal experiments were approved by the
Institutional Animal Care and Use Committee of Nanjing Medical
University (approval no. IACUC-2407092; Nanjing, China), and
conducted in accordance with the Association for Research in Vision
and Ophthalmology Statement for the Use of Animals in Ophthalmic
and Vision Research. A total of 200 C57BL/6J male mice, aged 6-8
weeks and weighing 20-25 g, were used in the present study. Mice
were purchased from Nanjing Junke Biotechnology Co., Ltd. They were
bred in an air-conditioned room maintained at 22±2°C with a
relative humidity of 50-60%, under a 12-h light/dark cycle, with
free access to standard laboratory chow and water. Mice were
anesthetized with intraperitoneal injection of ketamine (90 mg/kg)
and xylazine (7.5 mg/kg) prior to any invasive procedures. For
terminal experiments, including enucleation of the eyes, mice were
humanely euthanized by cervical dislocation under anesthesia as
aforementioned. Mice were randomly assigned to experimental or
control groups according to the specific study design. Animals were
assigned to the following independent groups: Wild-type (20 mice),
scramble control shRNA (shC; 10 mice), UNC5B shRNA (shUNC5B; 10
mice), negative control (NC; 5 mice), UNC5B overexpression
(oeUNC5B; 5 mice), DR (20 mice), RVO (5 mice), DR + 5 ng/ml
Netrin-1 (5 mice), DR + 50 ng/ml Netrin-1 (5 mice), DR + 500 ng/ml
Netrin-1 (5 mice), DR + 1,000 ng/ml Netrin-1 (5 mice), DR + 5,000
ng/ml Netrin-1 (5 mice), DR + shC (20 mice), DR + shUNC5B (20
mice), DR + Netrin-1 (15 mice), DR + shUNC5B + Netrin-1 (15 mice),
DR + NC (15 mice) and DR + oeUNC5B (15 mice). Unless otherwise
specified, all animal experiments were performed using n=5 mice per
group. For western blot analysis, each group consisted of n=4
biological replicates.
STZ-induced DR in mice
C57BL/6J mice were fasted overnight and
intraperitoneally injected with STZ (50 mg/kg; cat. no. EZ66D1DFD8;
BioFroxx; neoFroxx) for 5 consecutive days. Starting 1 week after
the final STZ injection, blood glucose levels were measured using a
portable blood glucose meter once weekly using a drop of blood
collected from the tail tip. Mice with blood glucose levels ≥16.7
mmol/l were considered diabetic and were included in the study.
Endothelial-specific adeno-associated
virus (AAV)-mediated knockdown and overexpression of retinal
UNC5B
Endothelial-tropic recombinant AAV vectors (Shanghai
GeneChem Co., Ltd.) were administered by retro-orbital intravenous
injection to modulate UNC5B expression in the mouse retina. For
knockdown, an AAV carrying an shRNA against Unc5b driven by the
intercellular adhesion molecule 2 promoter (target sequence,
5'-GGATCATGAGGTCCTTCTGCA-3') with an enhanced green fluorescent
protein reporter was used. A non-targeting scramble shRNA AAV
(target sequence, 5'-TTCTCCGAACGTGTCACGT-3') with the same promoter
and reporter was used as the control. For overexpression,
full-length mouse Unc5b cDNA under the control of the tyrosine
kinase with immunoglobulin-like and EGF-like domains 2 promoter was
delivered in the same manner, with an empty vector as control. AAV
vectors were administered via retro-orbital injection at a total
dose of ~3×1011 viral genomes per mouse. Subsequent
experiments were conducted ≥4 weeks post-injection.
Endothelium-specific modulation of UNC5B expression was validated
by western blotting and immunofluorescence staining (Fig. S1).
Intravitreal injection
Mice were anesthetized via intraperitoneal injection
of ketamine (90 mg/kg) and xylazine (7.5 mg/kg). The pupils were
dilated with 0.5% tropicamide (Santen Pharmaceutical Co., Ltd.),
and 0.5% proparacaine (Alcon, Inc.) was used for topical
anesthesia. A 33G needle (cat. no. 80300; Hamilton Company) was
inserted through the inferotemporal sclera under a
stereomicroscope. Mice received a single intravitreal injection of
2 μl Netrin-1 (cat. no. ALX-522-124; Enzo Life Sciences,
Inc.) at various concentrations (5, 50, 500, 1,000 or 5,000 ng/ml),
with 1,000 ng/ml as the primary concentration used for subsequent
analyses. Control eyes received equal volumes of PBS as the vehicle
solution. After the injection, 0.3% ofloxacin eye ointment
(Shenyang Sinqi Pharmaceutical Co., Ltd.) was used to prevent
infection. The recovery, respiration and activity of mice were
monitored.
Retro-orbital venous injection
Mice were anesthetized via intraperitoneal injection
of ketamine (90 mg/kg) and xylazine (7.5 mg/kg), and positioned
with their eyes oriented upward. A microinjector needle was
inserted at a 45° angle through the medial canthus into the
retro-orbital venous sinus. The aforementioned AAVs were injected
slowly to avoid vascular and ocular injuries. After needle
withdrawal, gentle pressure was applied with a sterile gauze to
achieve hemostasis. The mice were monitored for immediate
post-procedure abnormalities before being returned to their
cages.
Retinal vascular permeability assay
Adult C57BL/6J mice were anesthetized via
intraperitoneal injection of ketamine (90 mg/kg) and xylazine (7.5
mg/kg). Subsequently, Evans Blue (EB; 2%; 45 mg/kg) dye (cat. no.
E104208; Shanghai Aladdin Biochemical Technology Co., Ltd.) was
injected into the femoral vein and allowed to circulate for 1 h
(42). This was a terminal
procedure; mice were euthanized immediately after EB circulation.
The eyes were then gently enucleated, and the cornea, sclera, lens
and vitreous humor were carefully removed to isolate the retina.
The retinas were fixed in 4% PFA for 30 min at room temperature to
preserve their structural integrity and ensure optimal staining
conditions. Finally, the retinas were carefully separated and
examined under a fluorescence microscope. This allowed the
visualization and quantification of EB leakage from the
vasculature, providing a direct assessment of retinal vascular
permeability.
Periodic acid-Schiff (PAS) staining of
retinal vasculature
The retinal vasculature was meticulously isolated
using a standardized trypsin digestion protocol (43). Freshly excised eyes were promptly
fixed in 10% buffered formalin overnight at 4°C to preserve
delicate vascular structures. After fixation, the retinas were
gently dissected and incubated in double-distilled water overnight
at room temperature on a shaking table to ensure the thorough
removal of any residual fixative or debris. Subsequently, the
retinas were transferred to a 2% trypsin solution and incubated at
37°C for 1 h, allowing controlled enzymatic digestion to gently
break down the surrounding tissue while preserving the vascular
network. After digestion, the retinas were thoroughly washed with
double-distilled water for 5 min to remove any residual trypsin.
The isolated retinal vasculature was then air-dried in a
well-ventilated environment to prevent unwanted morphological
changes. Finally, the vasculature was stained with PAS and
hematoxylin using a stain kit (cat. no. G1281; Beijing Solarbio
Science & Technology Co., Ltd.) according to the manufacturer's
instructions. Briefly, samples were incubated with periodic acid at
room temperature for 5-10 min, followed by Schiff reagent for 15
min at room temperature, and counterstained with hematoxylin for
1-2 min at room temperature. Retinas were imaged under a light
microscope, and quantitative analyses were performed using ImageJ
software, revealing the intricate patterns of blood vessels within
the retina.
Retinal whole-mount immunofluorescence
staining
Mouse eyes were enucleated and fixed in 4% PFA for 2
h at room temperature. Subsequently, the eyes were dissected to
isolate the retinas, which were carefully cut into four-leaf clover
patterns to facilitate flat mounting. The retinas were fixed in 4%
PFA for 15 min at room temperature. The tissues were permeabilized
and blocked simultaneously in 1% Triton X-100 (cat. no. 9002-93-1;
Shanghai Hushi Laboratory Equipment Co., Ltd.) and 5% BSA (cat. no.
4240; BioFroxx; neoFroxx) at 37°C for 1 h. The retinas were
incubated with primary antibodies overnight at 4°C. The following
day, the retinas were incubated with the appropriate
fluorophore-conjugated secondary antibodies for 1 h at room
temperature. The antibodies used in the present study are listed in
Table SI. Fluorescence images
were acquired using a fluorescence microscope. Quantitative
analysis was performed using ImageJ software.
Retinal cryosection and
immunofluorescence staining
Mouse eyes were immersed in 4% PFA and stored at 4°C
for 2 h immediately after enucleation. After removal of the cornea
and lens, the eyes were subjected to an additional fixation step in
4% PFA overnight at 4°C. Tissues were then dehydrated in 30%
sucrose for 48 h and embedded in optimal cutting temperature
compound (cat. no. 4583; Sakura Finetek USA, Inc.) for
cryosectioning. Sagittal retinal sections (10 μm thick) were
prepared using a cryostat and mounted onto microscope slides. The
sections were permeabilized with 0.3% Triton X-100 and blocked with
5% BSA simultaneously at 37°C for 1 h. Subsequently, the sections
were incubated overnight at 4°C with primary antibodies. After
three washes with PBS, the sections were incubated with
fluorophore-conjugated secondary antibodies appropriate for the
host species of the primary antibodies for 1 h at room temperature,
followed by counterstaining with DAPI for 5 min at room
temperature. A list of the antibodies used is provided in Table SI. Fluorescence images were
captured using a fluorescence microscope, and the fluorescence
intensity and number of positive cells were quantified using ImageJ
software.
Establishment of the RVO model
C57BL/6J mice received a tail-vein injection of 1%
Rose Bengal solution (cat. no. A17053; Thermo Fisher Scientific,
Inc.) prepared in saline (20 mg/kg) using an insulin syringe. The
mice were then monitored for adverse reactions. Within 20 min of
Rose Bengal injection, the mice were anesthetized via
intraperitoneal injection of ketamine (90 mg/kg) and xylazine (7.5
mg/kg). Furthermore, 0.5% tropicamide eye drops (Santen
Pharmaceutical Co., Ltd.) were used for pupil dilation and 0.5%
levofloxacin eye drops (Santen Pharmaceutical Co., Ltd.) were
administered to prevent infection. A 514-nm laser (100 mW; 0.1 sec
pulse; 50 μm spot size) was directed at the retinal veins to
induce RVO (44,45).
Optical coherence tomography (OCT)
imaging in mice
OCT imaging (Saris; Nanjing Robotrak Technologies
Co., Ltd.) was performed 1 h, 1 day and 8 days after laser
treatment to assess retinal structural changes. The mice were
anesthetized as described previously, and their pupils were dilated
with 0.5% tropicamide (Santen Pharmaceutical Co., Ltd.). To prevent
corneal dehydration, the ocular surface was lubricated throughout
the procedure. The anesthetized mice were positioned on the OCT
imaging platform to ensure proper eye alignment for retinal scans.
Cross-sectional (B-scan) and facial images were acquired for each
eye. Multiple scans were captured to ensure reproducibility, and
the most focused images were selected for analysis. Retinal
thickness and lesion morphology were quantified using the built-in
analysis software provided by the manufacturer.
Patients used for aqueous humor (AH)
study
All procedures involving human participants were
approved by The Ethics Committee of The Affiliated Eye Hospital of
Nanjing Medical University (approval no. 2023003; Nanjing, China)
and conducted in accordance with The Declaration of Helsinki. AH
samples were collected from patients diagnosed with
non-proliferative DR (NPDR), proliferative DR (PDR) or RVO, and
age-related cataract at The Affiliated Eye Hospital of Nanjing
Medical University (Nanjing, China) between April 2023 and April
2024. A total of 32 patients were included: Patients with NPDR
(n=8; age range, 61-75 years; mean age, 67.38±5.40 years; 3 male
and 5 female patients), patients with PDR (n=8; age range, 62-68
years; mean age, 65.13±2.30 years; 4 male and 4 female patients),
patients with RVO (n=8; age range, 62-69 years; mean age,
65.38±2.26 years; 4 male and 4 female patients) and cataract
controls (n=8; age range, 61-76 years; mean age, 70.38±4.81 years;
4 male and 4 female patients). There were no statistically
significant differences in age between the cataract control group
and the disease groups (NPDR, PDR and RVO) (P>0.05). The
inclusion criteria were as follows: A clinical diagnosis of NPDR,
PDR or RVO based on fundus examination and imaging, and
availability of intraocular samples. Patients with age-related
cataract without retinal or systemic vascular diseases were
enrolled as controls. The exclusion criteria included the presence
of other retinal diseases, ocular inflammation, prior intraocular
surgery or intravitreal injection within the past 6 months, and
systemic inflammatory or autoimmune diseases. Using a precision
30-gauge needle, 30-50 μl of AH was gently aspirated from
each patient, with the needle insertion performed through the
peripheral cornea to avoid any contact with the iris or lens
tissue. This procedure was conducted under meticulous observation
under a surgical microscope before cataract surgery or anti-VEGF
intravitreal injection. The AH samples were transferred into
sterile tubes and centrifuged at 12,000 × g for 5 min at 4°C to
separate the cellular components and clarify the supernatant. The
expression levels of UNC5B in the AH samples were detected using a
human UNC5B enzyme-linked immunosorbent assay kit (cat. no.
MM-7554761; MEIMIAN) in accordance with the manufacturer's
instructions.
Statistical analysis
All data are presented as the mean ± SD to provide a
comprehensive and quantitative representation of the experimental
outcomes. Statistical analyses were performed using GraphPad Prism
9 software (Dotmatics). Significant differences between groups were
determined using the unpaired two-tailed Student's t-test for
pairwise comparisons or one-way ANOVA followed by Tukey's or
Dunnett's multiple comparisons test for multiple-group comparisons,
depending on the specific experimental design. P<0.05 was
considered to indicate a statistically significant difference.
Results
Bioinformatics analysis revealed
downregulated UNC5B expression in the retina of DR model mice,
predominantly in endothelial cells
Initial bioinformatics analysis was performed to
determine the cellular localization and differential expression of
UNC5B in the diabetic retina. Following normalization,
dimensionality reduction and clustering of the scRNA-seq data, 19
distinct cell clusters were identified using the first 10 principal
components (Fig. 1A). These
clusters were annotated into 11 major retinal cell types (Fig. 1B and C). Subsequent analysis of
the expression profile revealed that UNC5B was predominantly
expressed in endothelial cells, and its expression in DR retinal
endothelial cells was lower than that in controls (Fig. 1D and E).
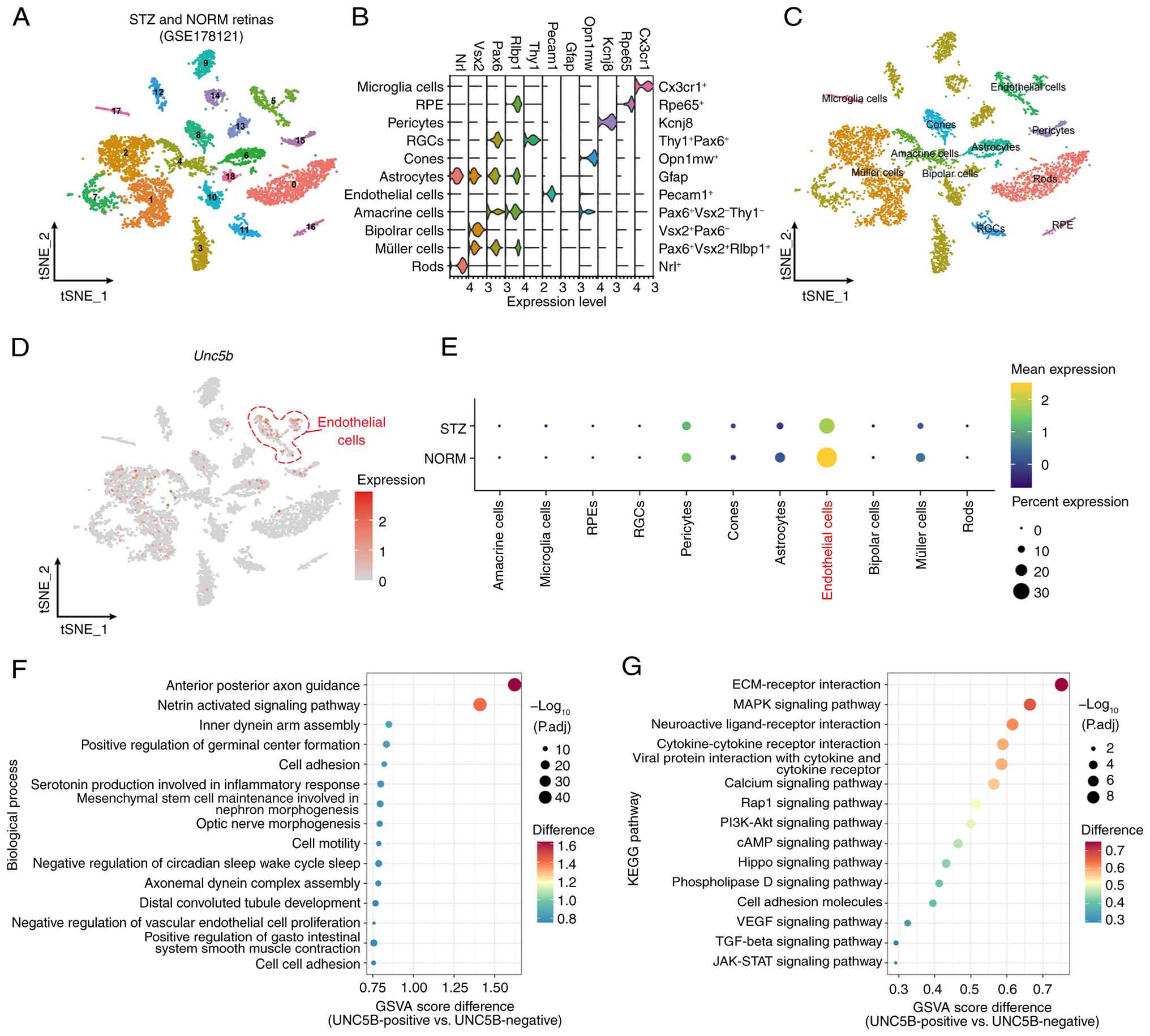 | Figure 1Bioinformatics analysis reveals
downregulated UNC5B expression in the retina of DR model mice,
predominantly in endothelial cells. The DR mice retinal single-cell
sequencing dataset (GSE178121) was obtained from the Gene
Expression Omnibus database. (A-C) Single-cell RNA sequencing data
were processed with Seurat software, showing (A) the tSNE
visualization of cell clusters, (B) the initial subpopulation
annotation based on known cell markers and (C) the final
identification of 11 retinal cell types after marker-based
annotation. (D) tSNE plot illustrating the global expression
pattern of UNC5B across retinal cells. (E) Dot plots comparing
UNC5B expression between control and DR mouse retinas. (F) GSVA
enrichment analysis of endothelial cells classified as
UNC5B-positive and UNC5B-negative, showing differentially enriched
biological processes. (G) GSVA enrichment analysis of endothelial
cells classified as UNC5B-positive and UNC5B-negative, showing
differentially enriched KEGG pathways. DR, diabetic retinopathy;
GSVA, gene set variation analysis; KEGG, Kyoto Encyclopedia of
Genes and Genomes; NORM, normal; p_adj, adjusted P-value; RGC,
retinal ganglion cell; RPE, retinal pigment epithelium; STZ,
streptozotocin; tSNE, t-distributed stochastic neighbor embedding;
UNC5B, unc-5 netrin receptor B. |
To further explore the potential functional role of
UNC5B in endothelial cells, endothelial cells were stratified into
UNC5B-positive and UNC5B-negative groups and subjected to GSVA
using KEGG and GO BP pathways. Pathway enrichment analysis
demonstrated that UNC5B expression in endothelial cells was
associated with pathways such as 'cell adhesion', 'ECM-receptor
interaction' and the 'Hippo signaling pathway' (Fig. 1F and G).
These findings suggested that UNC5B serves a crucial
role in maintaining retinal vascular homeostasis during the
pathological process of DR, particularly in preserving endothelial
cell function, and potentially contributing to BRB integrity.
UNC5B expression is downregulated in DR
and RVO models
To validate the results of bioinformatics analysis,
an STZ-induced DR mouse model was established using C57BL/6J mice.
Retinal tissues were collected from different mice at 2, 4 and 8
weeks post-modeling for RT-qPCR and western blot analyses. UNC5B
mRNA and protein levels in the retinas of 8-week DR mice were
significantly lower than those in normal mice (mRNA, P<0.001;
protein, P=0.004; Fig. 2A and
B). Furthermore, in cultured endothelial cells, high-glucose
stimulation significantly reduced UNC5B expression at 24 h (mRNA,
P=0.039; protein, P=0.005) and 48 h (mRNA, P<0.001; protein,
P<0.001) (Fig. 2C and D).
Similarly, significant reductions in retinal UNC5B levels were
observed in the RVO mouse model (mRNA, P=0.023; protein, P=0.003;
Fig. 2E and F).
Immunofluorescence staining of retinal whole mounts, using IB4 to
label the retinal vasculature, further confirmed the close
co-localization of UNC5B with the retinal vascular network
(Fig. 2G), thereby supporting
the scRNA-seq finding that UNC5B was predominantly expressed in
endothelial cells.
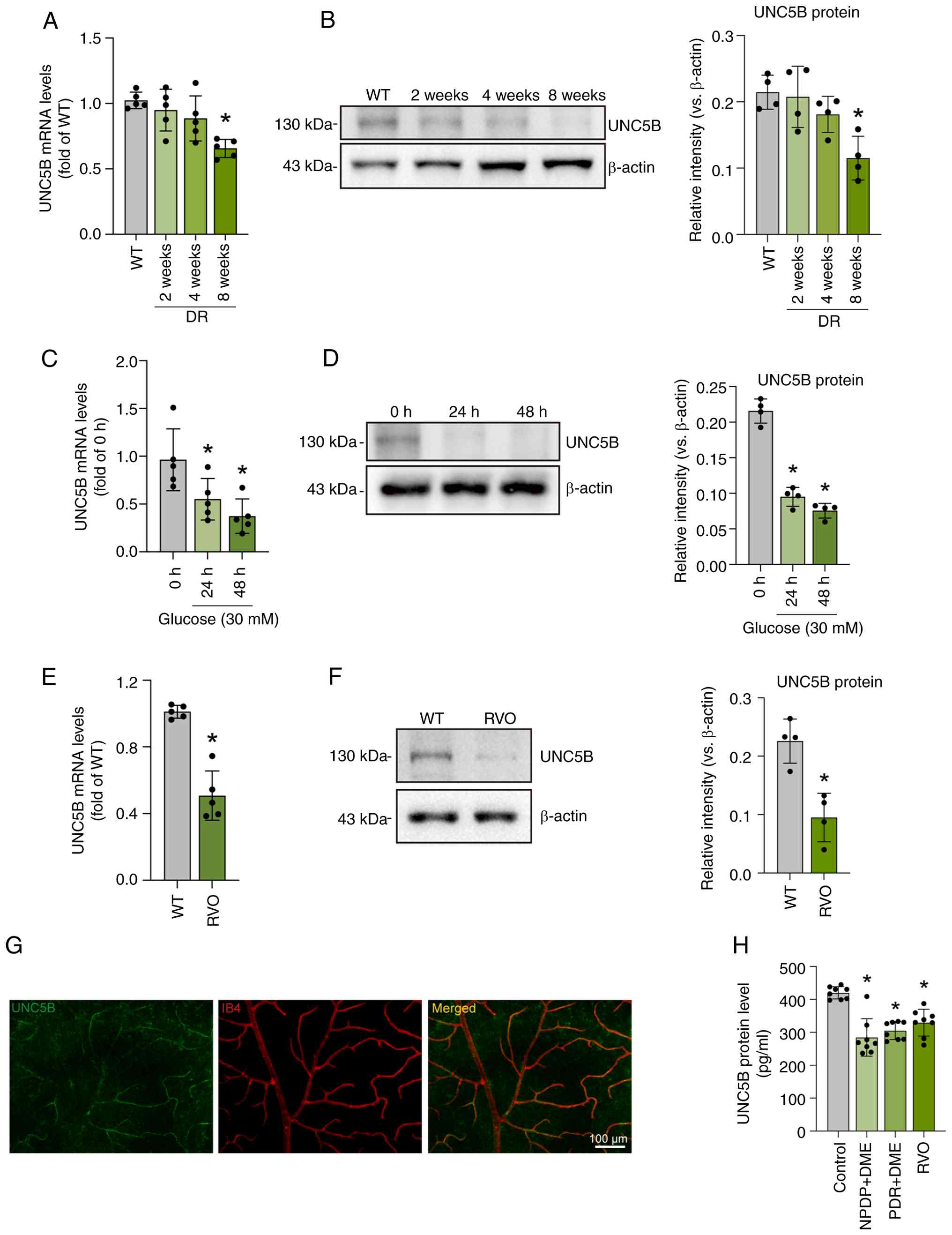 | Figure 2UNC5B expression is downregulated in
DR and RVO models. Retinal tissues from DR model mice (DR) were
collected at 2, 4 and 8 weeks post-induction and compared with
those from age-matched untreated mice (WT). (A) RT-qPCR and (B)
western blot analysis of UNC5B expression in the retinas of both
groups. Endothelial cells were stimulated with 30 mM glucose for 24
and 48 h and compared with untreated cells from the same batch (0
h). (C) RT-qPCR and (D) western blot analysis of UNC5B expression
in these three groups. Retinal tissues were collected from RVO
model mice 1 day after successful induction (RVO) and compared with
those from age-matched untreated mice (WT). (E) RT-qPCR and (F)
western blot analysis of UNC5B expression in the retinas of both
groups. (G) Immunofluorescence staining of whole-mounted retinas
from untreated normal mice to show UNC5B localization (green) and
IB4-labeled vasculature (red). Scale bar, 100 μm. Aqueous
humor samples were collected from patients diagnosed with senile
cataract, DR and RVO, with 8 individuals per group. (H) UNC5B
expression was assayed by ELISA in aqueous humor samples from
patients with DR and RVO, and age-related cataract controls. Data
are presented as the mean ± SD. n=5 per group, except for western
blot experiments (n=4) and ELISA (n=8). Multi-group comparisons
between the WT/0 h/Control group and each of the other groups were
performed using one-way ANOVA followed by Dunnett's multiple
comparisons test, while two-group comparisons between the WT and
RVO groups were performed using two-tailed unpaired Student's
t-tests. *P<0.05 vs. WT/0 h/Control. DME, diabetic
macular edema; DR, diabetic retinopathy; IB4, isolectin B4; NPDR,
non-proliferative DR; PDR, proliferative DR; RT-qPCR, reverse
transcription-quantitative PCR; RVO, retinal vein occlusion; UNC5B,
unc-5 netrin receptor B; w, weeks; WT, wild-type. |
To assess the clinical relevance, UNC5B levels were
analyzed in human AH samples. The AH was collected from patients
with DR showing diabetic macular edema (DME) ('NPDR + DME' and 'PDR
+ DME') and RVO who had not undergone intravitreal anti-VEGF
injection, using samples from patients with age-related cataract as
controls. ELISA results showed that the UNC5B expression in the AH
of patients with DR and RVO was significantly lower than that in
the control group (P<0.001 for all comparisons; Fig. 2H). These findings strongly
supported the crucial role of UNC5B in the maintenance of BRB
integrity.
UNC5B maintains barrier function of
endothelial cells
To further elucidate the role of UNC5B in
maintaining endothelial barrier integrity, UNC5B was knocked down
in HRMECs by transfection with lentivirus-encapsulated UNC5B shRNA.
Successful transfection was verified by fluorescence imaging
(Fig. 3A), and the knockdown
efficiency was confirmed at both the mRNA (P<0.001) and protein
levels (P=0.002) (Fig. 3B and
C). PI/calcein-AM staining showed that cell death was
significantly increased after UNC5B knockdown (P<0.001; Fig. 3D).
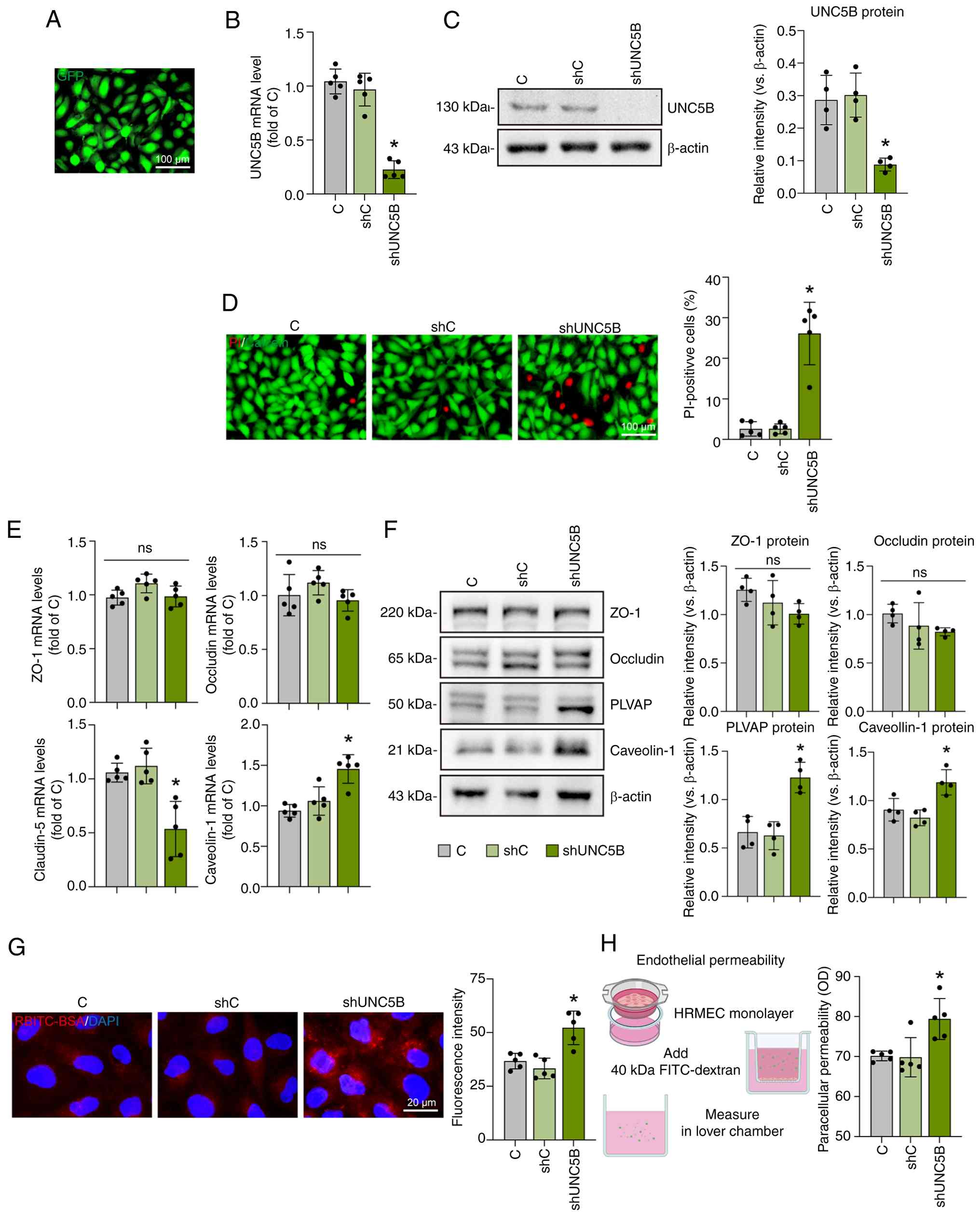 | Figure 3UNC5B maintains the normal barrier
function of endothelial cells. (A) Fluorescence microscopy image of
HRMECs transfected with green fluorescence-labeled
lentivirus-coated UNC5B shRNA. Scale bar, 100 μm. HRMECs
were transfected with lentiviral shUNC5B or shC, and puromycin was
used to select stably transfected HRMECs. UNC5B (B) mRNA and (C)
protein expression was examined by reverse
transcription-quantitative PCR and western blotting. (D) Live and
apoptotic cells were assessed using PI/Calcein-AM staining (green,
live cells; red, dead or dying cells). Scale bar, 100 μm.
(E) mRNA and (F) protein expression levels of barrier regulators
were examined. (G) HRMECs were incubated with RBITC-BSA for 6 h.
The internalized RBITC-BSA was imaged by microscopy and the
intensity of the internalized RBITC-BSA was quantitated using
ImageJ (red, RBITC-BSA; blue, DAPI). Scale bar, 20 μm. (H)
Schematic illustration of the experimental procedure for assessing
HRMEC monolayer permeability and the corresponding quantitative
results. Data are presented as the mean ± SD. n=5 per group, except
for western blot experiments (n=4). All statistical comparisons in
this figure were performed between the C group and each of the
other groups using one-way ANOVA followed by Dunnett's multiple
comparisons test. *P<0.05 vs. C. C, control; GFP,
green fluorescent protein; HRMEC, human retinal microvascular
endothelial cell; ns, not significant; OD, optical density; PLVAP,
plasmalemma vesicle-associated protein; RBITC, rhodamine B
isothiocyanate; shRNA, short hairpin RNA; shC, scramble control
shRNA; shUNC5B, UNC5B shRNA; UNC5B, unc-5 netrin receptor B; ZO-1,
zonula occludens 1. |
The present study subsequently investigated the
effects of UNC5B depletion on key regulators of the endothelial
barrier. RT-qPCR analysis showed no significant changes in the
expression of the paracellular junction proteins zonula occludens 1
and occludin, whereas the claudin-5 level was significantly
decreased (P=0.001). Concurrently, the expression levels of
caveolin-1, which is indicative of increased transcellular
transport (46), were markedly
upregulated (P<0.001) (Fig.
3E). Western blot analysis corroborated these findings,
verifying increased caveolin-1 expression (P=0.002) and elevated
levels of plasmalemma vesicle-associated protein (PLVAP), another
transcellular transport marker (P=0.014) (Fig. 3F).
Functional assays demonstrated that transcellular
transport, as assessed using labeled albumin, was elevated after
UNC5B knockdown (P=0.002; Fig.
3G), and the paracellular leakage of monolayer endothelial
cells was also increased (P=0.008; Fig. 3H). On the basis of these results,
it was inferred that UNC5B serves a key role in maintaining
endothelial cell barrier function.
UNC5B specifically regulates
endothelium-pericyte interactions with no significant effect on
pericyte function
To investigate the role of UNC5B in regulating
endothelium-pericyte crosstalk, the interactions between
endothelial cells and pericytes were assessed following UNC5B
knockdown in HRMECs. Immunofluorescence staining of IB4 and NG2
revealed a significant reduction in pericyte recruitment to the
endothelial tubes after UNC5B knockdown (P<0.001; Fig. 4A). BRB formation was then
simulated by co-culturing HRMECs with HRMVPCs (Fig. 4B). The results showed that UNC5B
knockdown led to increased leakage of the fluorescent tracer
(P<0.001; Fig. 4C). These
findings suggested that reduced UNC5B levels in endothelial cells
negatively affected pericyte function.
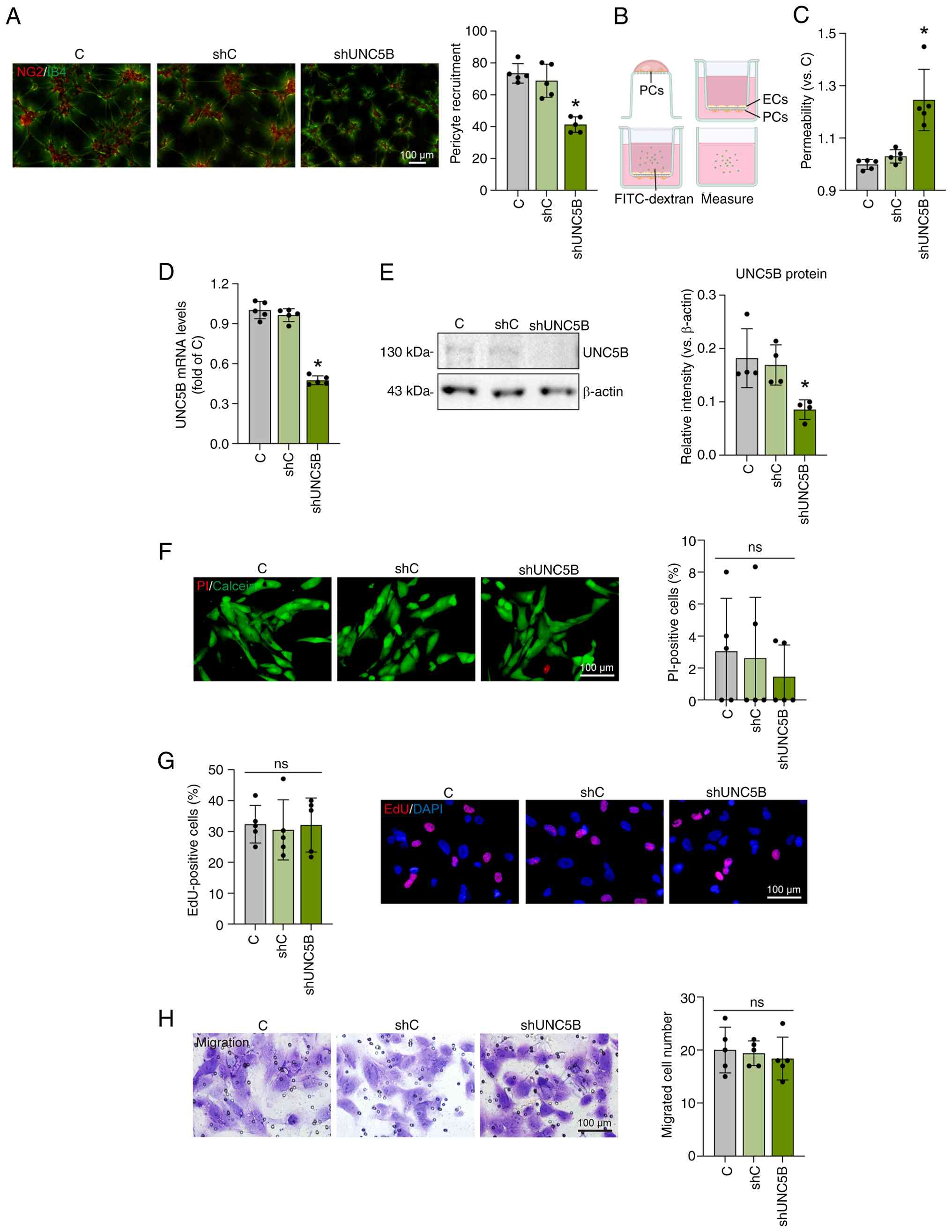 | Figure 4UNC5B specifically regulates
endothelium-pericyte interactions with no significant effect on
pericyte function. HRMECs were transfected with lentiviral shUNC5B
or shC and puromycin was used to select stably transfected HRMECs.
(A) Following transduction, HRMECs were co-cultured with HRMVPCs
for 6 h and then stained with NG2 (HRMVPCs) and IB4 (HRMECs) to
detect the recruitment of pericytes toward endothelial cells. Scale
bar, 100 μm. (B) For BRB model formation, HRMVPCs were
seeded in the lower compartment of the Transwell insert 1 h prior
to the addition of HRMECs. Co-cultures were maintained for 2 days
in complete DMEM. (C) Barrier properties of the BRB model were
assessed by permeability analysis. Subsequently, HRMVPCs were
transfected with lentiviral shUNC5B or shC, and puromycin was used
to select stably transfected HRMVPCs. UNC5B expression was examined
by (D) reverse transcription-quantitative PCR and (E) western
blotting. (F) Apoptosis (measured by PI incorporation), (G) cell
proliferation (measured by EdU incorporation) and (H) migration
were examined using the described assays, and the results were
quantified. Scale bar, 100 μm. Data are presented as the
mean ± SD. n=5 per group, except for western blot experiments
(n=4). All statistical comparisons in this figure were performed
between the C group and each of the other groups using one-way
ANOVA followed by Dunnett's multiple comparisons test.
*P<0.05 vs. C. BRB, blood-retinal barrier; C,
control; ECs, endothelial cells; EdU, 5-ethynyl-2'-deoxyuridine;
HRMEC, human retinal microvascular endothelial cell; HRMVPC, human
retinal microvascular pericyte; IB4, isolectin B4; NG2,
neuron-glial antigen 2; ns, not significant; PCs, pericytes; shRNA,
short hairpin RNA; shC, scramble control shRNA; shUNC5B, UNC5B
shRNA; UNC5B, unc-5 netrin receptor B. |
To investigate whether UNC5B directly affects
pericyte function, UNC5B was silenced in HRMVPCs using lentiviral
shRNA and a stable UNC5B-knockdown HRMVPC cell line was established
using puromycin selection. RT-qPCR and western blot analyses
confirmed the knockdown efficiency (mRNA, P<0.001; protein,
P=0.014; Fig. 4D and E).
Assessment of pericyte apoptosis, proliferation and migration
revealed no significant changes following UNC5B knockdown (Fig. 4F-H).
These results indicated that UNC5B served a crucial
role in regulating endothelium-pericyte interactions and
maintaining endothelial barrier function, with negligible direct
effects on pericytes. Silencing UNC5B in endothelial cells
simultaneously affected both transcellular and paracellular
transport as well as interactions with pericytes, thereby
disrupting BRB integrity.
UNC5B silencing in endothelial cells
disrupts BRB homeostasis in DR model mice and abrogates the
protective effect of high-concentration netrin-1
The concentration-dependent effects of netrin-1 on
vascular permeability were evaluated in the DR mouse model. The
results of EB staining showed that compared with DR mice, high
concentrations of netrin-1 (≥1,000 ng/ml) protected retinal
vascular integrity (1,000 ng/ml, P=0.016; 5,000 ng/ml, P<0.001),
whereas lower concentrations (<500 ng/ml) promoted vascular
leakage (5 ng/ml, P=0.038; 50 ng/ml, P<0.001) (Fig. 5A).
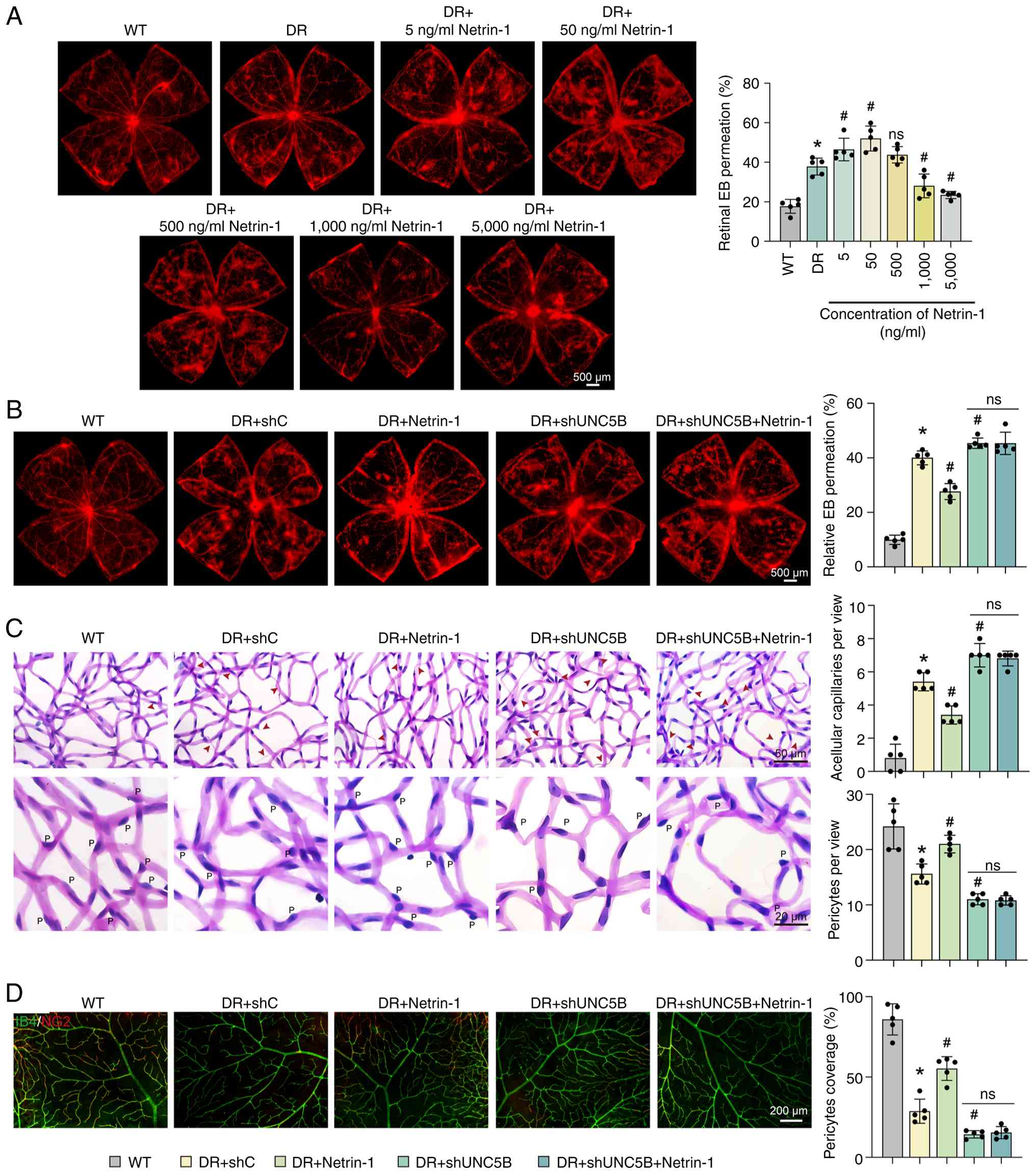 | Figure 5UNC5B silencing in endothelial cells
disrupts blood-retinal barrier homeostasis in DR model mice and
abrogates the protective effect of high-concentration netrin-1. At
week 4 after successful DR model induction, different
concentrations of netrin-1 (5, 50, 500, 1,000 and 5,000 ng/ml) were
injected intravitreally. Retinal tissues were collected after ≥12
weeks of DR model induction for subsequent analysis. (A) EB
staining was used to observe retinal vascular leakage in the
different groups. Scale bar, 500 μm. At week 4 after DR
model induction, control shRNA-AAV carrying an endothelial
cell-specific promoter sequence (DR + shC), 1,000 ng/ml netrin-1
(DR + Netrin-1), UNC5B shRNA-AAV carrying an endothelial
cell-specific promoter sequence (DR + shUNC5B) or a combination of
1,000 ng/ml netrin-1 and UNC5B shRNA-AAV (DR + shUNC5B + Netrin-1)
were injected. After ≥12 weeks of DR model induction, retinal
tissues were collected for analysis. (B) EB staining showed retinal
vascular leakage in the different groups. Scale bar, 500 μm.
(C) Periodic acid-Schiff staining revealed the formation of
acellular capillaries (arrows) and the number of pericytes ('P') in
the retinal tissues. Scale bar, 50 or 20 μm. (D) Whole-mount
retinal immunofluorescence staining showed the pericyte coverage in
the retinas of the different groups (red, NG2; green, IB4). Scale
bar, 200 μm. The data are presented as the mean ± SD. n=5
per group. All statistical comparisons in this figure were
performed among all groups using one-way ANOVA followed by Tukey's
multiple comparisons test. *P<0.05 vs. WT.
#P<0.05 vs. DR/DR + shC. AAV, adeno-associated virus;
DR, diabetic retinopathy; EB, Evans Blue; IB4, isolectin B4; NG2,
neuron-glial antigen 2; ns, not significant; shRNA, short hairpin
RNA; shC, scramble control shRNA; shUNC5B, UNC5B shRNA; UNC5B,
unc-5 netrin receptor B; WT, wild-type. |
Subsequently, the present study focused on the role
of UNC5B in mediating the protective effects of high-concentration
netrin-1 (1,000 ng/ml) in DR mice. Endothelial-specific silencing
of UNC5B was successfully achieved in vivo by AAV-mediated
gene targeting, as confirmed by reporter fluorescence (P<0.001)
and protein expression (P=0.003) (Fig. S1A and B). Compared with that in
DR mice treated with scrambled shRNA, intravitreal injection of
netrin-1 significantly reduced retinal vascular leakage
(P<0.001). Crucially, UNC5B silencing in DR mice not only
increased vascular leakage (P=0.042) but also prevented netrin-1
from exerting its protective effect (Fig. 5B).
Retinal vascular morphology was assessed by PAS
staining. In comparison with DR mice treated with scrambled shRNA,
mice that received netrin-1 treatment showed fewer acellular
capillaries (P<0.001) and increased pericyte coverage (P=0.007).
By contrast, endothelial UNC5B knockdown increased the number of
acellular capillaries (P=0.006) and decreased pericyte numbers
(P=0.026), preventing netrin-1 from exerting its protective effects
(Fig. 5C). Similar findings were
obtained using IB4 and NG2 immunofluorescence staining. Pericyte
coverage was significantly increased in DR mice injected with
netrin-1 compared with DR mice treated with scrambled shRNA
(P<0.001). By contrast, UNC5B knockdown reduced pericyte
coverage (P=0.022), and netrin-1 treatment did not improve coverage
in mice receiving UNC5B shRNA (Fig.
5D).
UNC5B overexpression in endothelial cells
maintains BRB homeostasis in DR model mice
Given the protective role of UNC5B in retinal
endothelial cells, it was evaluated whether endothelial-specific
overexpression preserved the integrity of the BRB in DR mice. UNC5B
overexpression was confirmed by immunofluorescence staining
(P<0.001) and western blot analysis (P<0.001) (Fig. S1C and D). The results showed
that specific UNC5B overexpression in the endothelial cells of DR
mice significantly reduced retinal vascular leakage (P<0.001;
Fig. 6A), alleviated the
formation of acellular capillaries (P<0.001; Fig. 6B) and preserved pericyte number
(P<0.001; Fig. 6B) and
coverage (P=0.004; Fig. 6C).
These findings highlighted the therapeutic potential of targeting
endothelial-cell UNC5B to maintain the BRB integrity in DR.
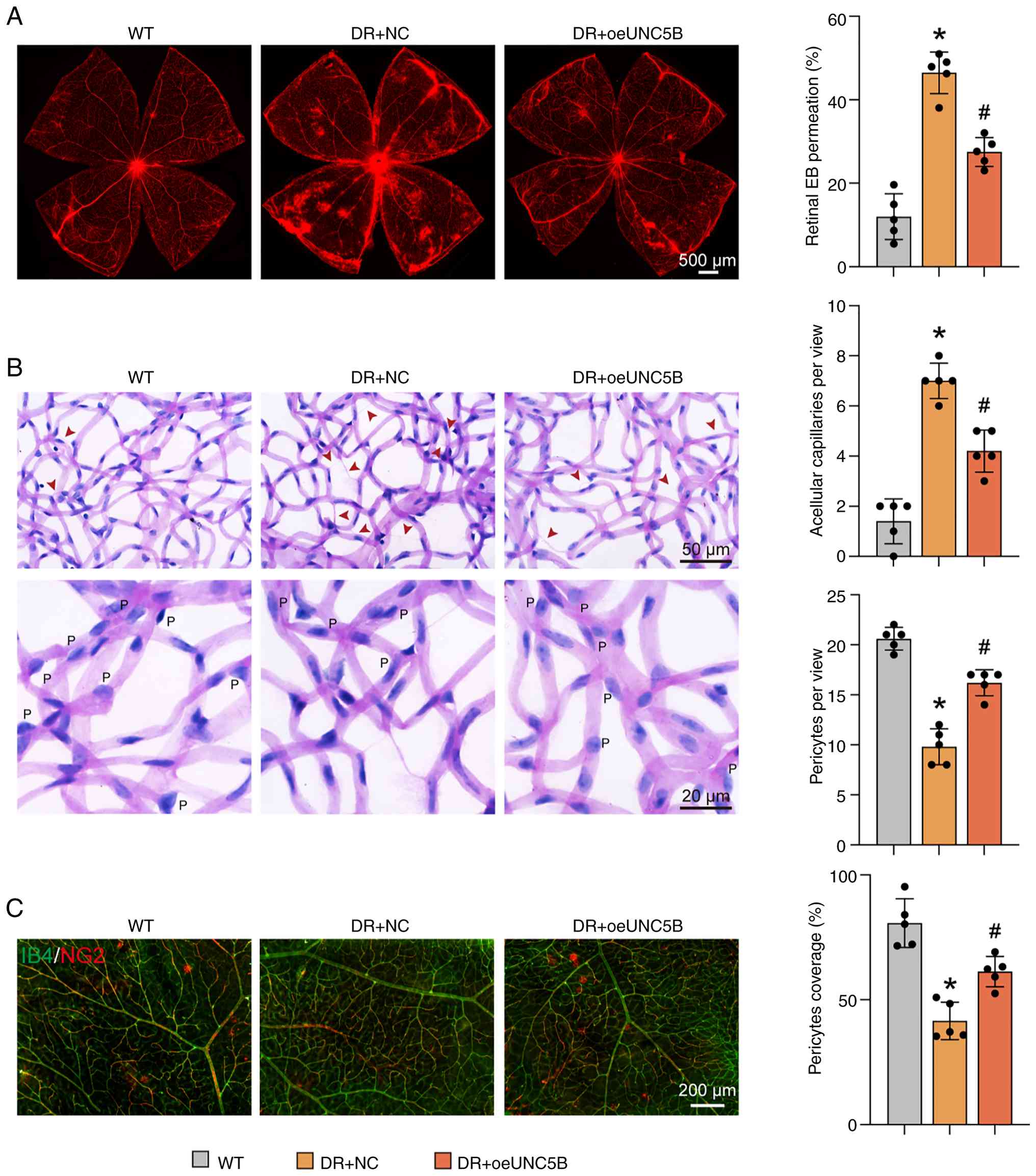 | Figure 6UNC5B overexpression in endothelial
cells maintains blood-retinal barrier homeostasis in DR model mice.
At week 4 after successful DR model induction, DR mice received a
single retro-orbital injection of adeno-associated virus carrying
an endothelial cell-specific promoter. Mice were assigned to two
groups: One with overexpression of UNC5B (DR + oeUNC5B) and the
other receiving an empty vector as a control (DR + NC). Retinal
tissues were collected after ≥12 weeks of DR model induction for
subsequent analysis. (A) EB staining showed retinal vascular
leakage in the different groups. Scale bar, 500 μm. (B)
Periodic acid-Schiff staining revealed acellular capillary
formation (arrows) and pericyte numbers ('P') in the retinas of the
different groups. Scale bar, 50 or 20 μm. (C) Whole-mount
retinal immunofluorescence staining showed pericyte coverage in the
retinas of the different groups (red, NG2; green, IB4). Scale bar,
200 μm. The data are presented as the mean ± SD. n=5 per
group. All statistical comparisons in this figure were performed
among all groups using one-way ANOVA followed by Tukey's multiple
comparisons test. *P<0.05 vs. WT.
#P<0.05 vs. DR + NC. DR, diabetic retinopathy; EB,
Evans Blue; IB4, isolectin B4; NC, negative control; NG2,
neuron-glial antigen 2; oe, overexpression; UNC5B, unc-5 netrin
receptor B; WT, wild-type. |
UNC5B affects neurodegeneration and glial
activation in the DR mouse model
To assess the role of UNC5B in DR-associated
neurodegeneration and gliosis, UNC5B was selectively silenced or
overexpressed in retinal endothelial cells using AAV delivery as
aforementioned (Fig. S1), and
analyses were conducted 14 weeks post-DR induction.
Immunofluorescence staining of retinal cryosections revealed a
significant decrease in neuron-specific nuclear protein
(NeuN)-positive ganglion cells and β-III tubulin (TUBB3)
fluorescence intensity in DR mice in comparison with the
corresponding findings in wild-type controls, confirming ganglion
cell loss. Endothelial-specific silencing of UNC5B further
exacerbated this neuronal loss (NeuN, P<0.001; TUBB3, P=0.031),
whereas UNC5B overexpression partially mitigated ganglion cell
degeneration (NeuN, P=0.020; TUBB3, P=0.022) (Fig. 7A and B). Retinal flat mounts
stained with class III β-tubulin further confirmed that modulation
of endothelial UNC5B expression influenced ganglion cell survival
in the DR model (DR + shUNC5B, P=0.014; DR + overexpression of
UNC5B, P=0.021; Fig. 7C).
Vimentin staining of retinal cryosections demonstrated reactive
gliosis in DR mice, with UNC5B silencing promoting glial
proliferation (P=0.020), whereas UNC5B overexpression significantly
attenuated this glial activation (P=0.004) (Fig. 7D). These findings suggested that
endothelial UNC5B expression influenced neuronal and glial outcomes
in DR by maintaining BRB integrity.
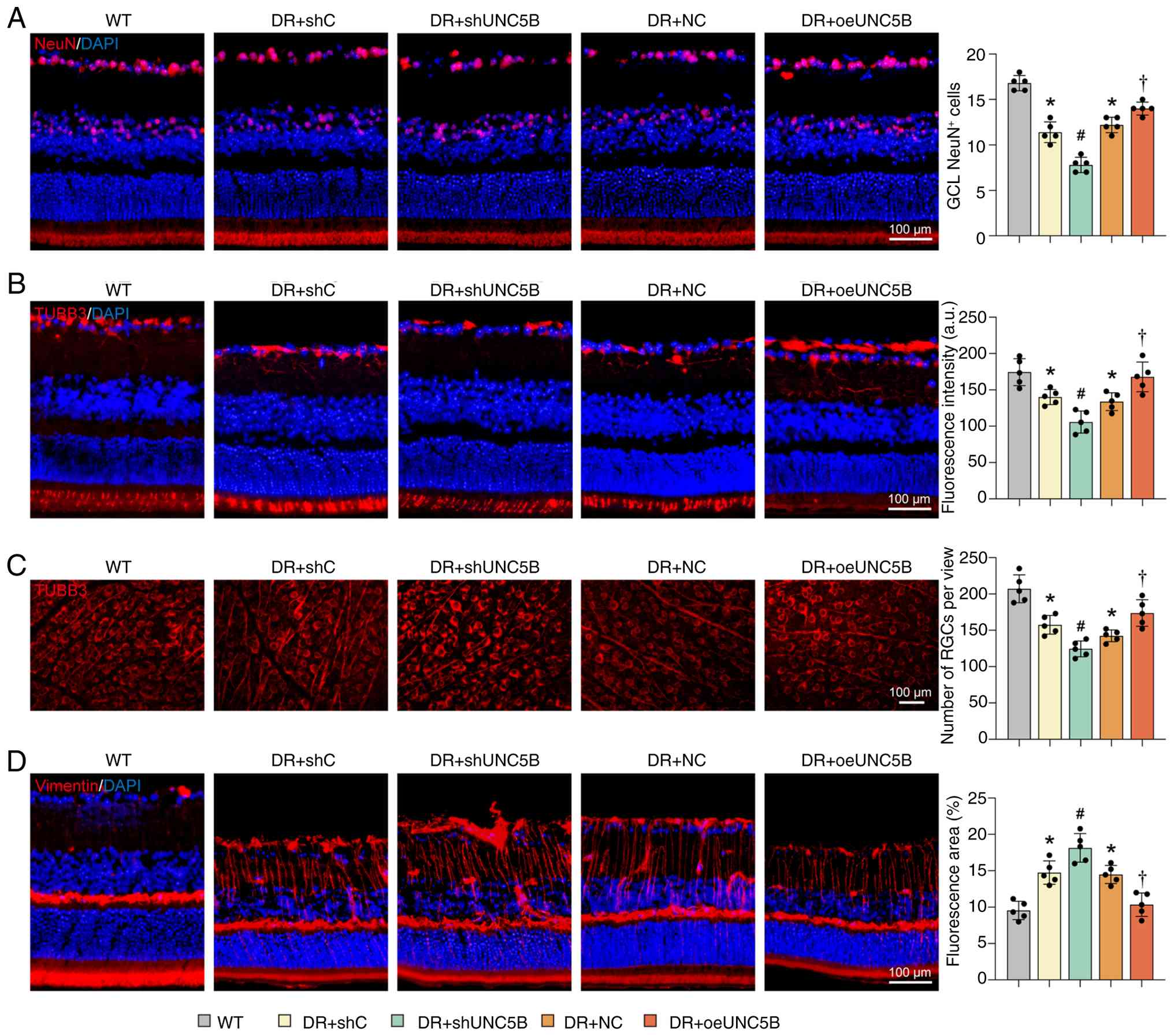 | Figure 7UNC5B affects neurodegeneration and
glial activation in the DR mouse model. After ≥14 weeks of DR model
induction, retinal tissues were collected for analysis. Mice were
divided into groups, including a WT group and four DR groups that
received different AAV treatments: DR + shC, DR + shUNC5B, DR + NC
and DR + oeUNC5B. Retinal cryosection immunofluorescence staining
showed (A) the number of NeuN-positive cells (red, NeuN; blue,
DAPI) and (B) TUBB3 fluorescence intensity (red, TUBB3; blue, DAPI)
in different groups. Scale bar, 100 μm. (C) Retinal
flat-mount TUBB3 immunofluorescence staining showed the number of
ganglion cells per field in different groups. Scale bar, 100
μm. (D) Retinal cryosection vimentin staining demonstrated
glial reactivity in different groups (red, vimentin; blue, DAPI).
Scale bar, 100 μm. The data are presented as the mean ± SD.
n=5 per group. All statistical comparisons in this figure were
performed among all groups using one-way ANOVA followed by Tukey's
multiple comparisons test. *P<0.05 vs. WT.
#P<0.05 vs. DR + shC. †P<0.05 vs. DR +
NC. DR, diabetic retinopathy; GCL, ganglion cell layer; NC,
negative control; NeuN, neuron-specific nuclear protein; oe,
overexpression; RGC, retinal ganglion cell; shRNA, short hairpin
RNA; shC, scramble control shRNA; shUNC5B, UNC5B shRNA; TUBB3,
β-III tubulin; UNC5B, unc-5 netrin receptor B; WT, wild-type. |
UNC5B silencing in endothelial cells
aggravates retinal edema in RVO model mice
Since both DR and RVO are characterized by BRB
disruption, which leads to macular edema (12), the protective role of UNC5B was
further investigated in an RVO mouse model. Endothelium-specific
UNC5B knockdown was achieved by AAV-mediated targeting as
aforementioned (Fig. S1A and
B). At least 4 weeks after AAV injection, RVO was induced by
tail-vein injection of Rose Bengal followed by laser treatment
(47).
Retinal edema and atrophy were dynamically monitored
using OCT 1 h, 1 day and 8 days after laser induction (Fig. 8A). The OCT results showed that
after endothelial UNC5B silencing, the peak retinal edema thickness
was significantly increased (day 1, P=0.015), and this subsequently
led to greater thinning of the retina by day 8 (P=0.037) (Fig. 8B).
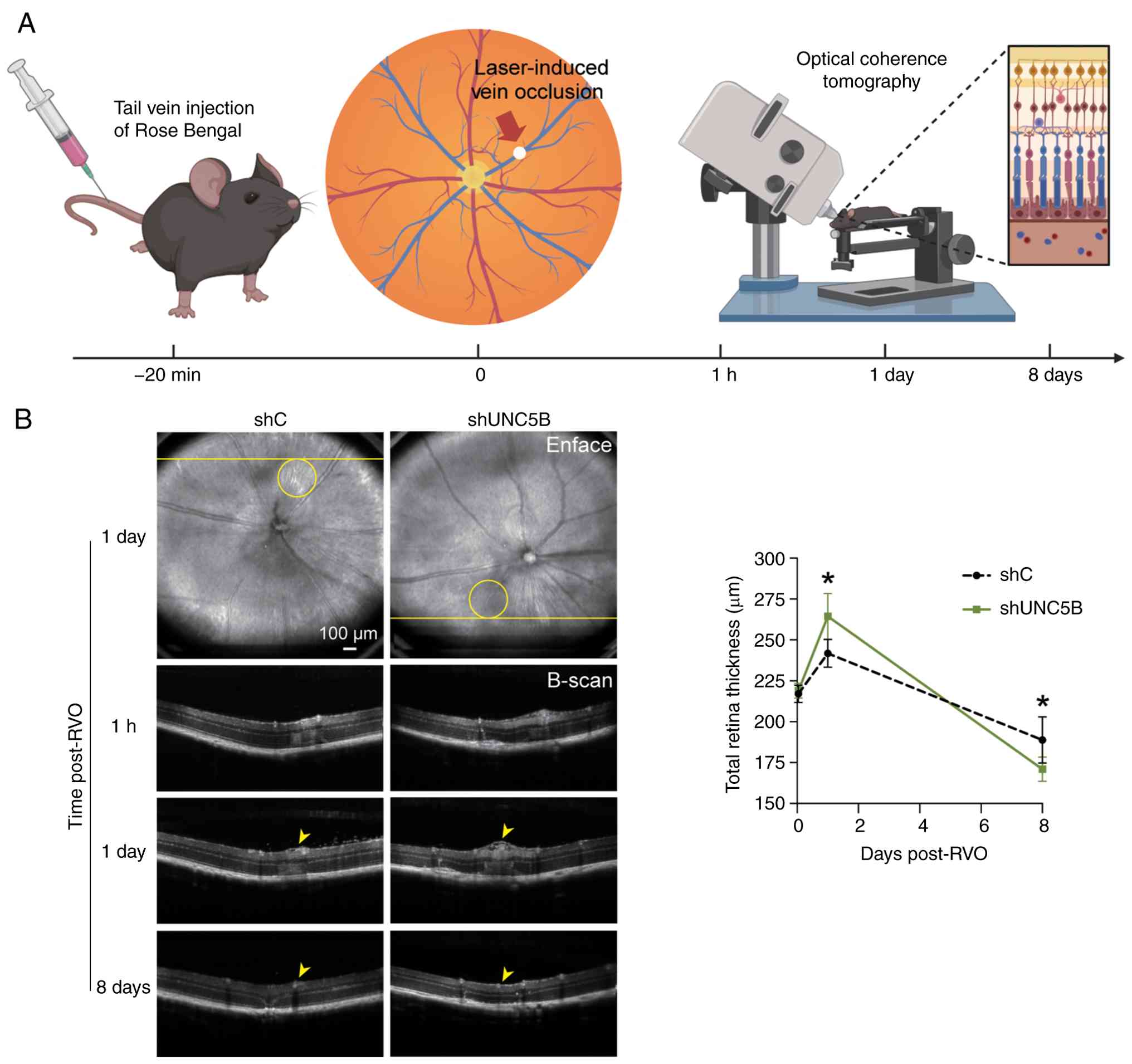 | Figure 8UNC5B silencing in endothelial cells
aggravates retinal edema in RVO model mice. In 6-8 week-old
wild-type mice, UNC5B shRNA-AAV (shUNC5B) or control shRNA-AAV
(shC), both carrying an endothelial cell-specific promoter
sequence, were administered via posterior orbital vein injection.
Following viral delivery, the mice were maintained for ≥4 weeks to
allow sufficient gene knockdown before subsequent procedures. (A)
Rose Bengal was injected via the tail vein, and retinal vein
occlusion was induced by laser treatment within 20 min
post-injection (created in BioRender; https://BioRender.com/g5aw0dv). (B) Optical coherence
tomography scans were performed 1 h, 1 day and 8 days after laser
induction to observe retinal edema and atrophy. The data are
presented as the mean ± SD. n=5 per group. Statistical comparisons
between the UNC5B knockdown and control groups at each time point
were performed using two-tailed unpaired Student's t-tests.
*P<0.05 vs. shC. AAV, adeno-associated virus; RVO,
retinal vein occlusion; shRNA, short hairpin RNA; shC, scramble
control shRNA; shUNC5B, UNC5B shRNA; UNC5B, unc-5 netrin receptor
B. |
Silencing of UNC5B promotes ECM-related
protein synthesis and inhibits the Hippo signaling pathway
To investigate the regulatory role of UNC5B in
endothelial cells, transcriptome sequencing analysis of HRMECs
transfected with UNC5B-shRNA lentivirus and control cells
transfected with scrambled control lentivirus was performed. The
volcano plot highlighted numerous significantly upregulated and
downregulated genes, indicating that UNC5B silencing markedly
altered the transcriptional landscape of endothelial cells
(Fig. S2A). GO and KEGG
enrichment analysis revealed that these differential genes were
closely related to 'extracellular matrix organization',
'ECM-receptor interaction' and the 'Hippo signaling pathway'
(Fig. 9A and B). Furthermore,
the differential genes were enriched in cellular components such as
'collagen-containing extracellular matrix' and 'cell-substrate
junction' (Fig. S2B). In terms
of molecular function, these genes were primarily involved in
'extracellular matrix structural constituent' and the binding of
key proteins such as integrin, fibronectin and laminin (Fig. S2C).
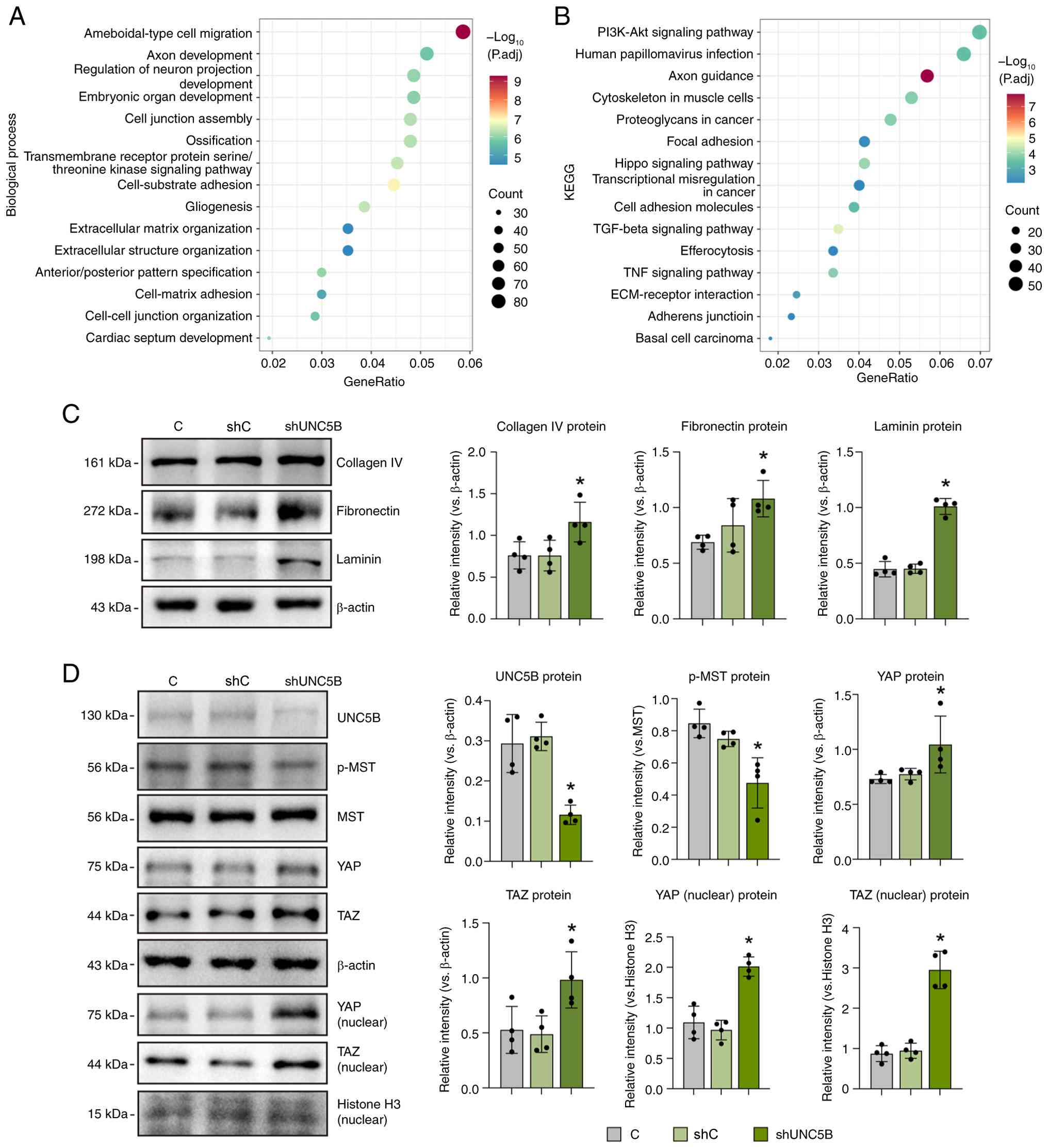 | Figure 9Silencing of UNC5B promotes
ECM-related protein synthesis and inhibits the Hippo signaling
pathway. HRMECs were transfected with lentiviral UNC5B shRNA, and
stable UNC5B-silenced cell lines (shUNC5B) were constructed using
puromycin selection. A control group was established by
transfection with negative control shRNA (shC). A total of three
samples from each group were used for transcriptome sequencing, and
differentially expressed genes were analyzed by Gene Ontology and
KEGG functional enrichment analysis. The lollipop plots displayed
the enrichment features of the two groups in terms of (A)
biological processes and (B) KEGG pathways. (C) Western blotting
was used to detect the expression of ECM-related proteins in HRMECs
after silencing of UNC5B. (D) Western blotting was also used to
assess the activation of the Hippo signaling pathway in HRMECs
after silencing of UNC5B. The data are presented as the mean ± SD.
n=4 per group. All statistical comparisons in this figure were
performed between the C group and each of the other groups using
one-way ANOVA followed by Dunnett's multiple comparisons test.
*P<0.05 vs. C. C, control; ECM, extracellular matrix;
HRMEC, human retinal microvascular endothelial cell; KEGG, Kyoto
Encyclopedia of Genes and Genomes; MST, mammalian Ste20-like
kinase; p-, phosphorylated; padj, adjusted P-value; shRNA, short
hairpin RNA; shC, scramble control shRNA; shUNC5B, UNC5B shRNA;
TAZ, transcriptional co-activator with PDZ-binding motif; UNC5B,
unc-5 netrin receptor B; YAP, yes-associated protein. |
These ECM findings were further validated at the
protein level. Western blotting confirmed that UNC5B knockdown
increased the expression of several core ECM components such as
collagen IV (P=0.034), fibronectin (P=0.019) and laminin
(P<0.001), suggesting that UNC5B may regulate the integrity of
the BRB by modulating ECM components (Fig. 9C).
The Hippo signaling pathway, which regulates cell
proliferation and apoptosis, also maintains vascular stability by
controlling the ECM composition and barrier protein expression
(48-50). Dysregulation of this pathway can
lead to excessive ECM accumulation, basement membrane thickening
and subsequent BRB dysfunction, contributing to retinal diseases
such as DR and RVO (51,52). Consistent with the observed
increase in ECM components, UNC5B silencing inhibited the
phosphorylation of the key Hippo signaling pathway kinase mammalian
STE20-like kinase (MST; P=0.002), leading to elevated protein
levels of the downstream effectors yes-associated protein (YAP;
P=0.033) and transcriptional co-activator with PDZ-binding motif
(TAZ; P=0.027). Analysis of nuclear protein extracts by western
blotting demonstrated significant nuclear accumulation of YAP and
TAZ in UNC5B-silenced cells compared with controls (P<0.001 for
all comparisons; Fig. 9D).
Collectively, these results indicated that UNC5B
influenced ECM synthesis and metabolism and inhibited the Hippo
signaling pathway, thereby participating in the maintenance of
endothelial cell homeostasis and vascular barrier function.
Discussion
Retinal vascular diseases, including DR and RVO,
share a common and devastating pathological feature: Disruption of
the BRB (53,54). Although this barrier is essential
for maintaining immune privilege and homeostasis of the neural
retina (55), earlier studies
have primarily examined UNC5B in developmental angiogenesis
(24,56). While more recent evidence has
implicated UNC5B in maintaining the mature BRB through the
regulation of barrier-associated proteins and Wnt signaling
(57-59), a comprehensive understanding of
its specific role in preserving vascular and neural homeostasis in
the pathological retina remains largely undefined. The present
study defined a previously unrecognized role of endothelial UNC5B
in maintaining BRB integrity and the coordinated functioning of the
NVU. By integrating cellular, animal and patient-derived data with
transcriptomic profiling, it was demonstrated that UNC5B occupied a
central position in the regulatory networks that preserve the
stability of the retinal microenvironment. These findings extend
the current understanding of barrier regulation in retinal vascular
diseases, and identified UNC5B as a mechanistically relevant target
for interventions aimed at sustaining vascular and neural
homeostasis. As illustrated in Fig.
10, endothelial UNC5B helps maintain BRB and NVU integrity
under normal conditions, while its downregulation in DR and RVO
leads to Hippo signaling suppression, and barrier and NVU
dysfunction.
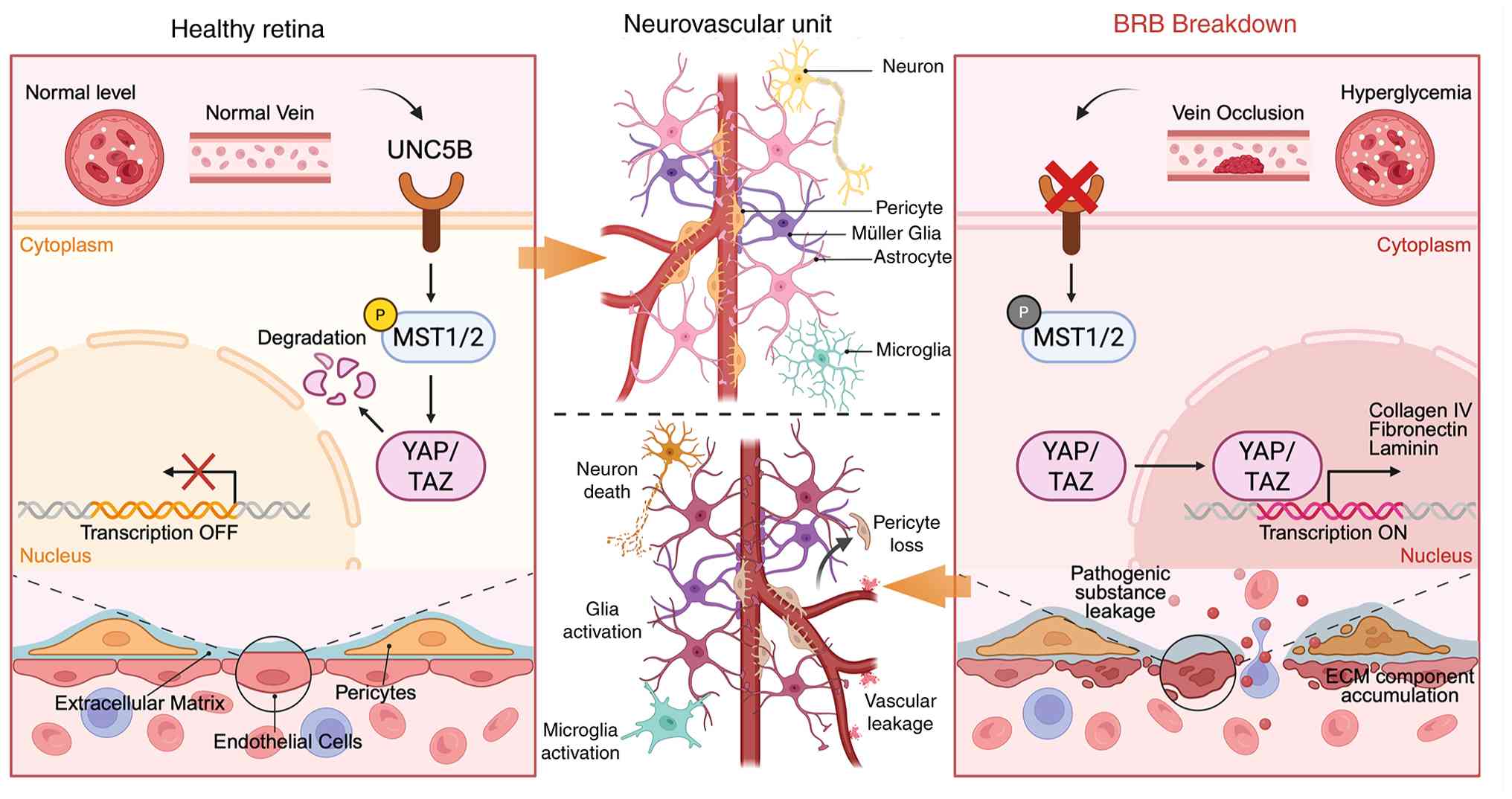 | Figure 10Role of endothelial UNC5B in
maintaining BRB and NVU integrity under normal and pathological
conditions. Normal UNC5B expression sustains BRB and NVU stability,
whereas its downregulation in diabetic retinopathy and retinal vein
occlusion is linked to Hippo signaling suppression, and impairment
of both BRB and NVU integrity. Created in BioRender; https://BioRender.com/xp1oos1. BRB,
blood-retinal barrier; ECM, extracellular matrix; MST, mammalian
Ste20-like kinase; NVU, neurovascular unit; P, phosphorylated; TAZ,
transcriptional co-activator with PDZ-binding motif; UNC5B, unc-5
netrin receptor B; YAP, yes-associated protein. |
The integrity of the BRB is maintained by a delicate
balance between paracellular tight junctions and transcellular
transport mechanisms (60). The
present data indicated that UNC5B functioned as a central regulator
of the normal low-permeability state of endothelial cells. By
selectively modulating tight junction proteins such as claudin-5
and suppressing vesicle-associated proteins such as PLVAP and
caveolin-1, UNC5B ensures a restrictive endothelial barrier. This
dual-regulatory capacity is important because it suggests that
UNC5B creates a comprehensive defense against vascular
hyperpermeability, suppressing both intercellular gaps and
unchecked transcytosis (57).
This finding indicates that the loss of this single receptor is
sufficient to trigger a broad breakdown of the endothelial barrier
function, offering a molecular explanation for the widespread
vascular leakage observed in ischemic retinopathies.
Furthermore, the present study advanced the
understanding of endothelium-pericyte crosstalk, which is a
cornerstone of vascular stability. Endothelium-pericyte
interactions are essential for vascular stability (61). The present study demonstrated
that endothelial UNC5B was essential for maintaining pericyte
coverage, supporting an 'inside-out' signaling paradigm in which
endothelial integrity governs pericyte retention. This implies that
endothelial dysfunction is an upstream initiator of mural cell
loss. Consequently, strategies aimed at preserving pericytes in DR
or RVO may prove ineffective unless the underlying endothelial
deficits, specifically the loss of UNC5B, are addressed
concurrently. This highlights the need to target the endothelium to
preserve the structural and functional coherence of the NVU.
Previous studies have reported that the netrin-1
signaling axis yields divergent vascular effects depending on
context and concentration (27,62). The present study confirmed that
the effect of netrin-1 on BRB integrity in the DR mouse model was
dose-dependent; concentrations ≥1,000 ng/ml reduced vascular
leakage, whereas lower concentrations exacerbated it, consistent
with previous findings (26,63). Crucially, the present in
vivo experiments established that this protective effect was
UNC5B-dependent, since endothelial UNC5B knockdown abolished the
stabilizing action of netrin-1, promoting DR-like pathology. This
extends the results of previous studies on UNC5B (56-59), from physiological barrier
maintenance to a definitive pathological context, thereby providing
a molecular basis for optimizing netrin-1-based therapeutic
interventions that maximize BRB protection. Consistent with this,
the protective role of endothelial UNC5B was validated in two
distinct retinal vascular pathologies. In DR and RVO model mice,
in vivo silencing aggravated vascular leakage, edema and
subsequent atrophy, whereas its overexpression in DR model mice
mitigated these pathological changes. Together, these findings
indicated that netrin-1-based therapies require careful dose
optimization and may need to be tailored according to the UNC5B
expression status. The findings further suggested that restoring
UNC5B expression may be a prerequisite for reestablishing retinal
responsiveness to ligand-based vascular protection.
The central mechanistic insight of the present study
was the presence of concurrent alterations in Hippo pathway
activity and ECM component expression after UNC5B silencing.
Transcriptomic analysis revealed that UNC5B silencing profoundly
altered the expression of genes associated with ECM organization
and Hippo signaling. Functional validation showed that UNC5B
knockdown was associated with increased deposition of core ECM
components. Since pathological basement membrane thickening
resulting from excessive ECM deposition is a classic
histopathological feature of DR (64), the present data indicated that
UNC5B deficiency may drive this pathological change. Furthermore,
UNC5B deficiency was accompanied by reduced phosphorylation of the
Hippo pathway kinase MST and increased nuclear accumulation of YAP
and TAZ. The Hippo signaling pathway is increasingly being
recognized as a critical regulator of vascular integrity and
mechanotransduction (65-68).
These observations indicated potential associations among UNC5B,
Hippo signaling and ECM remodeling in the context of BRB
disruption, although the precise causal relationships remain to be
determined. Further studies are warranted to determine whether
UNC5B directly modulates Hippo pathway components or ECM remodeling
acts upstream to influence pathway activity, thus providing
potential targets for therapeutic interventions.
Beyond vascular structural stability, the present
findings underscored the critical role of endothelial UNC5B in
preserving the functional integrity of the NVU. The retinal NVU
operates as an interdependent cellular ensemble (16,69,70), and the neuronal loss and glial
activation observed following endothelial-specific UNC5B depletion
indicated that disturbances in endothelial signaling could
propagate to neural components. Thus, neurodegeneration in ischemic
retinopathies is, at least in part, precipitated by the loss of
endothelial UNC5B and the subsequent breakdown of the homeostatic
microenvironment. Although the present study identified an
association between vascular dysfunction and neural stress, the
findings did not clarify whether the altered endothelial secretome,
driven by the Hippo/ECM axis, directly affects neuronal survival or
if the damage is secondary to metabolic mismatch caused by barrier
failure. Nevertheless, these data reinforce the idea that
therapeutic strategies targeting endothelial UNC5B offer two
benefits: Sealing the vasculature, and concurrently shielding the
neural retina from secondary degeneration.
The present study had several limitations. First,
the findings were primarily based on animal and cell models, and a
comprehensive validation in human retinal tissues is lacking.
Second, although the results suggested that UNC5B regulated ECM
remodeling and Hippo pathway activity, the direct downstream
effectors that mediate these processes remain unclear. Previous
studies have revealed that netrin-1-UNC5B signaling interacts with
the Hippo pathway in neuronal and tumor cells (71,72). However, whether and how this
crosstalk contributes to retinal endothelial function and BRB
homeostasis remains unclear. Further exploration of this
interaction may help clarify how UNC5B maintains vascular stability
and provide novel insights into barrier-protective strategies.
Future research should aim to identify these molecular targets
downstream of UNC5B that mediate ECM remodeling and Hippo pathway
activity, and explore whether pharmacological activation of UNC5B
or its signaling partners can be translated into clinical
therapies. In summary, the present findings revealed that
endothelial UNC5B was a key regulator of BRB integrity and
neurovascular stability, and that restoring its function may
represent a promising strategy for treating retinal vascular
diseases.
In summary, the present study established
endothelial UNC5B as a key regulator of BRB integrity and NVU
homeostasis in retinal vascular diseases. The loss of UNC5B
precipitated a cascade of pathological events, including barrier
leakage, pericyte detachment and aberrant ECM remodeling, which
were potentially linked to Hippo pathway dysregulation. Conversely,
restoration of UNC5B function preserved vascular stability and
mitigated neuroglial dysfunction. These findings deepen the
mechanistic understanding of DR and RVO pathogenesis, and position
UNC5B, together with its downstream signaling pathways, as a
promising and context-dependent therapeutic axis for preserving
retinal vascular and neural functions.
Supplementary Data
Availability of data and materials
The sequencing data generated in the present study
may be found in the China National GeneBank DataBase under
accession number CNP0008595 or at the following URL: https://db.cngb.org/data_resources/project/CNP0008595.
The other data generated in the present study may be requested from
the corresponding author.
Authors' contributions
YY, JL, QJia and KL designed the study. YY, SW,
QJin and ZC conducted the experiments. YY and SW analyzed the data
and wrote the manuscript. QJia, JL and KL supervised the research
and revised the manuscript. QJia and KL confirm the authenticity of
all the raw data. All authors read and approved the final version
of the manuscript.
Ethics approval and consent to
participate
All animal experiments were conducted according to
the guidelines of the Association for Research in Vision and
Ophthalmology Statement for the Use of Animals in Ophthalmic and
Vision Research, and approved by the Institutional Animal Care and
Use Committee of Nanjing Medical University (approval no.
IACUC-2407092; Nanjing, China). All procedures involving human
participants were conducted in accordance with The Declaration of
Helsinki and were approved by The Ethics Committee of The
Affiliated Eye Hospital of Nanjing Medical University (approval no.
2023003; Nanjing, China). Written informed consent was obtained
from all patients for the use of their clinical samples and related
data for scientific research. The Ethics Committee of The
Affiliated Eye Hospital of Nanjing Medical University (Nanjing,
China) confirmed that the in vitro experiments using
commercially available human cells qualified for exemption from
ethical review.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National Natural Science
Foundation of China (grant nos. 82571232 and 82171080), the Science
Fund for Excellent Young Scholars of Jiangsu Province (grant no.
BK20240122), and the Lang Shi Jie & Mu Guang Ming-Ophthalmology
Research Project for Young and Middle-Aged Scientists (grant no.
BCF-KH-YK-20230803-07).
References
|
1
|
Meng Y, Liu Y, Duan R, Liu B, Lin Z, Ma Y,
Jiang L, Qin Z and Li T: Global, regional, and national
epidemiology of vision impairment due to diabetic retinopathy among
working-age population, 1990-2021. J Diabetes. 17:e701212025.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen Y, Tang S, Huang Y, Deng J, Chen X,
Qi Y, Xiao H, Li Y, Li H and Guan H: Global burden of blindness or
visually impairment attributable to diabetic retinopathy in the
adults aged 70 years and older, 1990-2021: Results from the global
burden of disease study in 2021. Diabetes Res Clin Pract.
226:1123832025. View Article : Google Scholar
|
|
3
|
Antonetti DA, Silva PS and Stitt AW:
Current understanding of the molecular and cellular pathology of
diabetic retinopathy. Nat Rev Endocrinol. 17:195–206. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tang Y, Shi Y and Fan Z: The mechanism and
therapeutic strategies for neovascular glaucoma secondary to
diabetic retinopathy. Front Endocrinol (Lausanne). 14:11023612023.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yue T, Shi Y, Luo S, Weng J, Wu Y and
Zheng X: The role of inflammation in immune system of diabetic
retinopathy: Molecular mechanisms, pathogenetic role and
therapeutic implications. Front Immunol. 13:10550872022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kuroiwa DAK, Malerbi FK and Regatieri CVS:
New insights in resistant diabetic macular edema. Ophthalmologica.
244:485–494. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tatsumi T: Current treatments for diabetic
macular edema. Int J Mol Sci. 24:95912023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sweeney MD, Ayyadurai S and Zlokovic BV:
Pericytes of the neurovascular unit: Key functions and signaling
pathways. Nat Neurosci. 19:771–783. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhou L, Xu Z, Lu H, Cho H, Xie Y, Lee G,
Ri K and Duh EJ: Suppression of inner blood-retinal barrier
breakdown and pathogenic Müller glia activation in ischemia
retinopathy by myeloid cell depletion. J Neuroinflammation.
21:2102024. View Article : Google Scholar
|
|
10
|
Bora K, Kushwah N, Maurya M, Pavlovich MC,
Wang Z and Chen J: Assessment of inner blood-retinal barrier:
Animal models and methods. Cells. 12:24432023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
O'Leary F and Campbell M: The blood-retina
barrier in health and disease. FEBS J. 290:878–891. 2023.
View Article : Google Scholar
|
|
12
|
Klaassen I, Van Noorden CJF and
Schlingemann RO: Molecular basis of the inner blood-retinal barrier
and its breakdown in diabetic macular edema and other pathological
conditions. Prog Retin Eye Res. 34:19–48. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fletcher EL, Dixon MA, Mills SA and
Jobling AI: Anomalies in neurovascular coupling during early
diabetes: A review. Clin Exp Ophthalmol. 51:81–91. 2023. View Article : Google Scholar
|
|
14
|
Kugler EC, Greenwood J and MacDonald RB:
The 'neuro-glial-vascular' unit: The role of glia in neurovascular
unit formation and dysfunction. Front Cell Dev Biol. 9:7328202021.
View Article : Google Scholar
|
|
15
|
Grimes WN, Berson DM, Sabnis A, Hoon M,
Sinha R, Tian H and Diamond JS: Layer-specific anatomical and
physiological features of the retina's neurovascular unit. Curr
Biol. 35:109–120.e4. 2025. View Article : Google Scholar :
|
|
16
|
Ivanova E, Alam NM, Prusky GT and
Sagdullaev BT: Blood-retina barrier failure and vision loss in
neuron-specific degeneration. JCI Insight. 5:e1267472019.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sheng X, Zhang C, Zhao J, Xu J, Zhang P,
Ding Q and Zhang J: Microvascular destabilization and intricated
network of the cytokines in diabetic retinopathy: From the
perspective of cellular and molecular components. Cell Biosci.
14:852024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rudraraju M, Narayanan SP and Somanath PR:
Regulation of blood-retinal barrier cell-junctions in diabetic
retinopathy. Pharmacol Res. 161:1051152020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Daneman R, Zhou L, Kebede AA and Barres
BA: Pericytes are required for blood-brain barrier integrity during
embryogenesis. Nature. 468:562–566. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Resnikoff HA, Miller CG and Schwarzbauer
JE: Implications of fibrotic extracellular matrix in diabetic
retinopathy. Exp Biol Med (Maywood). 247:1093–1102. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Park DY, Lee J, Kim J, Kim K, Hong S, Han
S, Kubota Y, Augustin HG, Ding L, Kim JW, et al: Plastic roles of
pericytes in the blood-retinal barrier. Nat Commun. 8:152962017.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Leonardo ED, Hinck L, Masu M, Keino-Masu
K, Ackerman SL and Tessier-Lavigne M: Vertebrate homologues of C.
elegans UNC-5 are candidate netrin receptors. Nature. 386:833–838.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Serafini T, Colamarino SA, Leonardo ED,
Wang H, Beddington R, Skarnes WC and Tessier-Lavigne M: Netrin-1 is
required for commissural axon guidance in the developing vertebrate
nervous system. Cell. 87:1001–1014. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Navankasattusas S, Whitehead KJ, Suli A,
Sorensen LK, Lim AH, Zhao J, Park KW, Wythe JD, Thomas KR, Chien CB
and Li D: The netrin receptor UNC5B promotes angiogenesis in
specific vascular beds. Development. 135:659–667. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Larrivée B, Freitas C, Trombe M, Lv X,
Delafarge B, Yuan L, Bouvrée K, Bréant C, Del Toro R, Bréchot N, et
al: Activation of the UNC5B receptor by netrin-1 inhibits sprouting
angiogenesis. Genes Dev. 21:2433–2447. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tu T, Zhang C, Yan H, Luo Y, Kong R, Wen
P, Ye Z, Chen J, Feng J, Liu F, et al: CD146 acts as a novel
receptor for netrin-1 in promoting angiogenesis and vascular
development. Cell Res. 25:275–287. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yu Y, Zou J, Han Y, Quyang L, He H, Hu P,
Shao Y and Tu P: Effects of intravitreal injection of netrin-1 in
retinal neovascularization of streptozotocin-induced diabetic rats.
Drug Des Devel Ther. 9:6363–6377. 2015.PubMed/NCBI
|
|
28
|
Yang Y, Zou L, Wang Y, Xu KS, Zhang JX and
Zhang JH: Axon guidance cue netrin-1 has dual function in
angiogenesis. Cancer Biol Ther. 6:743–748. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Oshitari T: Neurovascular impairment and
therapeutic strategies in diabetic retinopathy. Int J Environ Res
Public Health. 19:4392021. View Article : Google Scholar
|
|
30
|
Ma J, Liu X, Qin Y, Qu Y, Liu Q and Li J:
Canagliflozin inhibits NLRP3 inflammasome activation to protect the
retinal neurovascular unit in a mouse retinal vein occlusion model.
Exp Eye Res. 263:1108032026. View Article : Google Scholar
|
|
31
|
Lu X, Le Noble F, Yuan L, Jiang Q, De
Lafarge B, Sugiyama D, Bréant C, Claes F, De Smet F, Thomas JL, et
al: The netrin receptor UNC5B mediates guidance events controlling
morphogenesis of the vascular system. Nature. 432:179–186. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kaur S, Abu-Asab MS, Singla S, Yeo SY and
Ramchandran R: Expression pattern for unc5b, an axon guidance gene
in embryonic zebrafish development. Gene Expr. 13:321–327. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sun L, Wang R, Hu G, Liu H, Lv K, Duan Y,
Shen N, Wu J, Hu J, Liu Y, et al: Single cell RNA sequencing
(scRNA-Seq) deciphering pathological alterations in
streptozotocin-induced diabetic retinas. Exp Eye Res.
210:1087182021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Binet F, Cagnone G, Crespo-Garcia S, Hata
M, Neault M, Dejda A, Wilson AM, Buscarlet M, Mawambo GT, Howard
JP, et al: Neutrophil extracellular traps target senescent
vasculature for tissue remodeling in retinopathy. Science.
369:eaay53562020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Siddiqui SS, Siddiqui ZK and Malik AB:
Albumin endocytosis in endothelial cells induces TGF-beta receptor
II signaling. Am J Physiol Lung Cell Mol Physiol. 286:L1016–L1026.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang N, Zhang D, Sun G, Zhang H, You Q,
Shao M and Yue Y: Lipopolysaccharide-induced caveolin-1
phosphorylation-dependent increase in transcellular permeability
precedes the increase in paracellular permeability. Drug Des Devel
Ther. 9:4965–4977. 2015.PubMed/NCBI
|
|
37
|
Adzraku SY, Wang G, Cao C, Bao Y, Wang Y,
Smith AO, Du Y, Wang H, Li Y, Xu K, et al: Robo4 inhibits gamma
radiation-induced permeability of a murine microvascular
endothelial cell by regulating the junctions. Cell Mol Biol Lett.
28:22023. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Xia M, Jiao L, Wang XH, Tong M, Yao MD, Li
XM, Yao J, Li D, Zhao PQ and Yan B: Single-cell RNA sequencing
reveals a unique pericyte type associated with capillary
dysfunction. Theranostics. 13:2515–2530. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Fresta CG, Fidilio A, Caruso G, Caraci F,
Giblin FJ, Leggio GM, Salomone S, Drago F and Bucolo C: A New human
blood-retinal barrier model based on endothelial cells, pericytes,
and astrocytes. Int J Mol Sci. 21:16362020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
41
|
Chen S, Zhou Y, Chen Y and Gu J: fastp: An
ultra-fast all-in-one FASTQ preprocessor. Bioinformatics.
34:i884–i890. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Oosuka S, Kida T, Oku H, Horie T,
Morishita S, Fukumoto M, Sato T and Ikeda T: Effects of an
aquaporin 4 inhibitor, TGN-020, on murine diabetic retina. Int J
Mol Sci. 21:23242020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chou JC, Rollins SD and Fawzi AA: Trypsin
digest protocol to analyze the retinal vasculature of a mouse
model. J Vis Exp. e504892013.PubMed/NCBI
|
|
44
|
Zhao Y, Ge Z, Guo T, Liu H, Zhou Y, Chen
J, Xu H and Chen Z: Complement C3/C3aR signaling pathway inhibition
ameliorates retinal damage in experimental retinal vein occlusion.
Invest Ophthalmol Vis Sci. 66:22025. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Avrutsky MI, Chen CW, Lawson JM, Snipas
SJ, Salvesen GS and Troy CM: Caspase-9 inhibition confers stronger
neuronal and vascular protection compared to VEGF neutralization in
a mouse model of retinal vein occlusion. Front Neurosci.
17:12095272023. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Minshall RD, Sessa WC, Stan RV, Anderson
RG and Malik AB: Caveolin regulation of endothelial function. Am J
Physiol Lung Cell Mol Physiol. 285:L1179–L1183. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Avrutsky MI, Ortiz CC, Johnson KV,
Potenski AM, Chen CW, Lawson JM, White AJ, Yuen SK, Morales FN,
Canepa E, et al: Endothelial activation of caspase-9 promotes
neurovascular injury in retinal vein occlusion. Nat Commun.
11:31732020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wei L, Gao J, Wang L, Tao Q and Tu C:
Hippo/YAP signaling pathway: A new therapeutic target for diabetes
mellitus and vascular complications. Ther Adv Endocrinol Metab.
14:204201882312201342023. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Lei Y, Liu Q, Chen B, Wu F, Li Y, Dong X,
Ma N, Wu Z, Zhu Y, Wang L, et al: Protein O-GlcNAcylation coupled
to Hippo signaling drives vascular dysfunction in diabetic
retinopathy. Nat Commun. 15:93342024. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Chaqour B: CCN-Hippo YAP signaling in
vision and its role in neuronal, glial and vascular cell function
and behavior. J Cell Commun Signal. 17:255–262. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ahmad US, Uttagomol J and Wan H: The
regulation of the Hippo pathway by intercellular junction proteins.
Life (Basel). 12:17922022.PubMed/NCBI
|
|
52
|
Park JA and Kwon YG: Hippo-YAP/TAZ
signaling in angiogenesis. BMB Rep. 51:157–162. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Gu Y, Hao S, Liu K, Gao M, Lu B, Sheng F,
Zhang L, Xu Y, Wu D, Han Y, et al: Airborne fine particulate matter
(PM2.5) damages the inner blood-retinal barrier by
inducing inflammation and ferroptosis in retinal vascular
endothelial cells. Sci Total Environ. 838:1565632022. View Article : Google Scholar
|
|
54
|
Haydinger CD, Ferreira LB, Williams KA and
Smith JR: Mechanisms of macular edema. Front Med (Lausanne).
10:11288112023. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zhou LY, Liu ZG, Sun YQ, Li YZ, Teng ZQ
and Liu CM: Preserving blood-retinal barrier integrity: A path to
retinal ganglion cell protection in glaucoma and traumatic optic
neuropathy. Cell Regen. 14:132025. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Raza Q, Nadeem T, Youn SW, Swaminathan B,
Gupta A, Sargis T, Du J, Cuervo H, Eichmann A, Ackerman SL, et al:
Notch signaling regulates UNC5B to suppress endothelial
proliferation, migration, junction activity, and retinal plexus
branching. Sci Rep. 14:136032024. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Boyé K, Geraldo LH, Furtado J,
Pibouin-Fragner L, Poulet M, Kim D, Nelson B, Xu Y, Jacob L, Maissa
N, et al: Endothelial Unc5B controls blood-brain barrier integrity.
Nat Commun. 13:11692022. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Furtado J, Geraldo LH, Leser FS, Poulet M,
Park H, Pibouin-Fragner L, Eichmann A and Boyé K: Netrin-1 binding
to Unc5B regulates blood-retina barrier integrity. bioRxiv
[Preprint]: 2023.01.21.525006. 2023.
|
|
59
|
Furtado J, Geraldo LH, Leser FS,
Bartkowiak B, Poulet M, Park H, Robinson M, Pibouin-Fragner L,
Eichmann A and Boyé K: Interplay between netrin-1 and Norrin
controls arteriovenous zonation of blood-retina barrier integrity.
Proc Natl Acad Sci USA. 121:e24086741212024. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Zhang CL, Ma JJ, Li X, Yan HQ, Gui YK, Yan
ZX, You MF and Zhang P: The role of transcytosis in the
blood-retina barrier: From pathophysiological functions to drug
delivery. Front Pharmacol. 16:15653822025. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Armulik A, Abramsson A and Betsholtz C:
Endothelial/pericyte interactions. Circ Res. 97:512–523. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Nabeel Mustafa A, Salih Mahdi M, Ballal S,
Chahar M, Verma R, Ali Al-Nuaimi AM, Kumar MR, Kadhim A, Al-Hussein
R, Adil M and Jasem Jawad M: Netrin-1: Key insights in neural
development and disorders. Tissue Cell. 93:1026782025. View Article : Google Scholar
|
|
63
|
Li Y, Chai JL, Shi X, Feng Y, Li JJ, Zhou
LN, Cao C and Li KR: Gαi1/3 mediate netrin-1-CD146-activated
signaling and angiogenesis. Theranostics. 13:2319–2336. 2023.
View Article : Google Scholar
|
|
64
|
Roy S and Kim D: Retinal capillary
basement membrane thickening: Role in the pathogenesis of diabetic
retinopathy. Prog Retin Eye Res. 82:1009032021. View Article : Google Scholar
|
|
65
|
Boopathy GTK and Hong W: Role of Hippo
pathway-YAP/TAZ signaling in angiogenesis. Front Cell Dev Biol.
7:492019. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Islam MS, Afrin S, Singh B, Jayes FL,
Brennan JT, Borahay MA, Leppert PC and Segars JH: Extracellular
matrix and Hippo signaling as therapeutic targets of antifibrotic
compounds for uterine fibroids. Clin Transl Med. 11:e4752021.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Lv H and Ai D: Hippo/yes-associated
protein signaling functions as a mechanotransducer in regulating
vascular homeostasis. J Mol Cell Cardiol. 162:158–165. 2022.
View Article : Google Scholar
|
|
68
|
Weiss EL: Hippo unleashed! Proteome-scale
analysis reveals new views of Hippo pathway biology. Sci Signal.
6:pe362013. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Ji L, Tian H, Webster KA and Li W:
Neurovascular regulation in diabetic retinopathy and emerging
therapies. Cell Mol Life Sci. 78:5977–5985. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Meng C, Gu C, He S, Su T, Lhamo T, Draga D
and Qiu Q: Pyroptosis in the retinal neurovascular unit: New
insights into diabetic retinopathy. Front Immunol. 12:7630922021.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Ahn EH, Kang SS, Qi Q, Liu X and Ye K:
Netrin1 deficiency activates MST1 via UNC5B receptor, promoting
dopaminergic apoptosis in Parkinson's disease. Proc Natl Acad Sci
USA. 117:24503–24513. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Qi Q, Li DY, Luo HR, Guan KL and Ye K:
Netrin-1 exerts oncogenic activities through enhancing
Yes-associated protein stability. Proc Natl Acad Sci USA.
112:7255–7260. 2015. View Article : Google Scholar : PubMed/NCBI
|
























