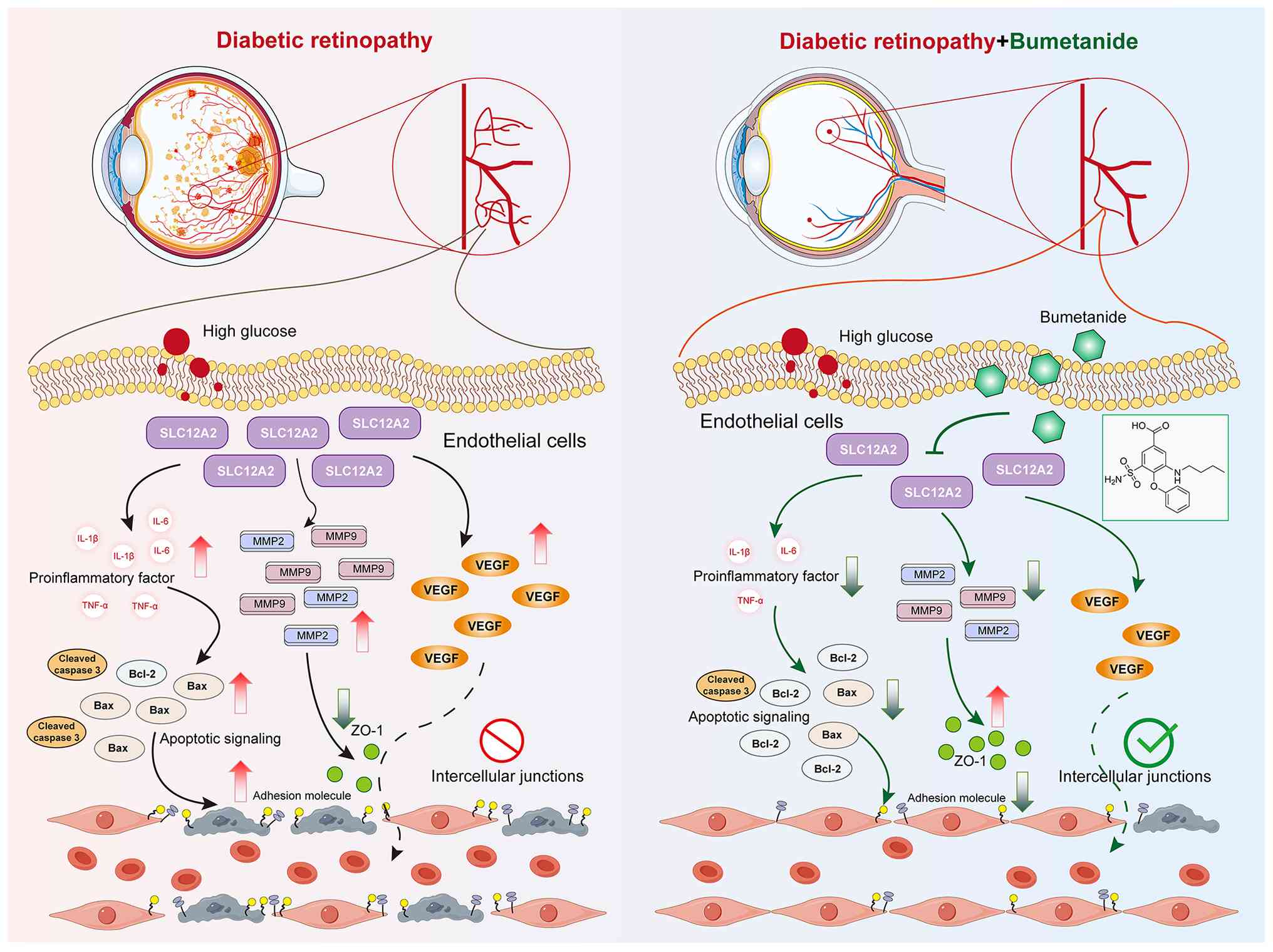
Introduction
Diabetic retinopathy (DR), a widespread
microvascular complication of diabetes, is a progressive disease
and a leading cause of vision loss (1). With the increasing prevalence of
diabetes, the number of patients with DR is rapidly increasing. The
global incidence of DR is projected to reach 160.5 million by 2045,
increasing the demand for ophthalmic care and treatment (2). Currently, treatment strategies for
DR are limited to advanced cases presenting with symptoms of
retinal damage, and mainly include laser photocoagulation, surgery
and intravitreal injections of anti-vascular endothelial growth
factor (VEGF) or corticosteroids (3). Although these methods can restrict
disease progression, they do not prevent side effects or treatment
resistance. Considering that most DR are preventable, there is an
urgent need to find new ways to treat DR. In recent years, the
identification of specific biomarkers can provide a scientific
basis for the development of DR therapeutic agents, such as RBP3
(4,5). However, the targets available for
clinical use are still lacking.
Bumetanide (PubChem CID: 2471) is a potent
circulating diuretic that acts by inhibiting Na-K-Cl co-transport
and is effective in the treatment of various inflammations or
ischemia-induced edema (6).
Recent evidence has uncovered the potential role of bumetanide in
the alleviation of retinopathy. For example, it exhibits
antiangiogenic and oxidative stress inhibitory properties in
oxygen-induced retinopathy (7).
It also reduced the number of apoptotic cells and the expression of
AQP4 (a factor involved in retinal edema) in a retinal
ischemia-reperfusion injury (8).
In addition, bumetanide has anti-inflammatory properties and low
cytotoxicity, which suppresses diabetic activity (9). However, it is unclear whether
bumetanide has effect in attenuating DR. To the best of the
authors' knowledge, the pharmacological effects of bumetanide
involve the modulation of NKCC1 (also known as SLC12A2) and KCC2
(also known as SLC12A4) (10,11). Moreover, there is a large gap
regarding the influence of SLC12A2 and SLC12A4 on the
pathophysiology of DR.
The emergence of single-cell RNA sequencing
(scRNA-seq) technology has provided an opportunity to dissect
complex pathological mechanisms at single-cell resolution (12). In 2020, Van Hove et al
(13) first applied scRNA-seq in
DR to reveal the cellular and molecular changes of the disease
(13). Since then, an increasing
number of studies have used scRNA-seq, which can be considered a
powerful tool for developing effective therapeutic targets, to
explore the underlying mechanisms of DR (14). Reportedly, dysfunction of some
endothelial cell subpopulations (specifically expressing IL-1β,
S100A8, S100A9) during DR enhanced the release of inflammatory
molecules and stimulation of adhesion molecules, leading to
increased neovascularization and permeability (15). The elimination of functional
endothelial cell subpopulations may delay the progression of DR.
Hence, scRNA-seq has great potential for identifying new
therapeutic strategies for DR.
In the present study, bioinformatics analysis and
biological approaches were employed to elucidate the protective
effects of bumetanide against DR and its underlying mechanisms. A
flow chart and the experimental design are depicted in Fig. 1. It was demonstrated that SLC12A2
and SLC12A4 play critical roles in mediating the protective effects
of bumetanide. Subsequently, utilizing scRNA-seq and RNA-seq data
from the Gene Expression Omnibus (GEO; https://www.ncbi.nlm.nih.gov/geo/) database, it was
found that SLC12A2 was highly expressed in endothelial cells, which
are the primary targets of hyperglycemic damage (16). Endothelial cell markers were
enriched in the angiogenesis regulation and adhesion
molecule-related pathways. In vitro and in vivo
experiments further revealed that under hyperglycemic conditions,
SLC12A2 deficiency reduced endothelial cell apoptosis and
inflammatory events by inhibiting cell adhesion molecules, thereby
contributing to improved DR. These findings provide novel insights
into the role of bumetanide in DR and its regulatory genes,
highlighting SLC12A2 as a promising therapeutic target for DR.
Materials and methods
Bioinformatics analysis Research
datasets
DR-related datasets were retrieved from the GEO
database using the keyword 'DR'. Ultimately, five datasets were
selected for the present study. Among them, there were three
scRNA-seq datasets [GSE165816 (17); 3 healthy samples; GSE248284
(18); 3 DR cases; GSE165784
(19); 5 DR cases] and two
RNA-seq datasets [GSE221521 (20); 69 DR and 50 healthy; and
GSE185011 (21); 5 DR and 5
healthy].
scRNA-seq data processing
First, data from the three scRNA-seq datasets were
merged using Seurat (version 4.4.0) in the R package (version
4.3.3; https://cran.r-project.org/), and
low-quality cells were removed, including those with gene counts
<500, mitochondrial genes >25%, and erythrocyte genes >3%.
Second, after normalizing the data with NormalizeData,
FindVariableFeatures was utilized to extract intercellular variable
genes, and 2,000 highly variable genes were screened for analysis.
The RunPCA function was applied to conduct principal component
analysis of the cells based on the expression levels of the
variable genes, and the top 20 PC with statistical significance
were selected for downstream analysis. Next, the cells were
analyzed by t-distributed Stochastic Neighbour Embedding (t-SNE)
non-linear dimensionality reduction using the FindCluster tool.
Finally, marker genes for each cell cluster were identified by the
Seurat package, followed by the annotation of cells using
CellMarker. Notably, the distribution of the key genes, SLC12A2 and
SLC12A4, was also demonstrated in different cells using t-SNE
maps.
Functional enrichment analysis
As both scRNA-seq analysis and literature reports
indicate the key role of endothelial cells in the pathogenesis of
DR, the major functions of the marker gene set were explored in
endothelial cells (22).
Specifically, the top 200 marker genes of endothelial cells were
included in Gene Ontology (GO; https://geneontology.org/) and Kyoto Encyclopedia of
Genes and Genomes (KEGG; https://www.genome.jp/kegg/) enrichment analyses.
Experimental procedures
Cell culture
Rat retinal microvascular endothelial cells (RRMECs;
cat. no. CP-R114) and 293T cells (cat. no. CL-0005) obtained from
Pricella Biotechnology Co., Ltd. were cultured in DMEM [containing
10% FBS (cat. no. 26-500-FBS; Ephraim), 1% penicillin/streptomycin]
and a humid atmosphere at 37°C/5% CO2. RRMECs were
exposed to normal glucose (5 mM) or high glucose (HG, 25 mM,
simulating an in vitro DR model) (23).
Cell treatment
In the present study, two experiments were designed
to explore the effects of bumetanide and SLC12A2 on cells. In
experiment 1, the cells were classified into control, HG, and HG +
Bumetanide groups. For drug treatment, cells were treated with 10
μM of bumetanide in the presence of HG for 24 h at 37°C.
In experiment 2, the cells were classified into
control, HG, HG + sh-NC and HG + sh-SLC12A2 groups. Specific
targeting of SLC12A2 (sh-SLC12A2, 5'-CCAAGTTCTTCTTACATTATA-3') and
negative control (sh-NC, 5'-CCTAAGGTTAAGTCGCCCTCG-3') sequences
were designed by using VectorBuilder platform (VectorBuilder Inc.).
For the NC and sh-SLC12A2 groups, 12 μg target plasmid
(sh-NC or sh-SLC12A2) and 12 μg packaging plasmids
(pMDLg/pRRE: pVSV-G: pRSV-Rev=5:3:2) were transfected into 293T
cells using 8 μl HighGene reagent (cat. no. RM09014;
ABclonal Biotech Co., Ltd.). After 48 and 72 h of transfection (at
37°C), the supernatant containing the virus was collected
separately for transduction of target cells (RRMECs). The collected
supernatants were mixed and centrifuged at 3,000 × g for 10 min at
4°C, and then filtered through a 0.45-μm membrane. During
transduction, lentiviral solution with a titer of 1×108
TU/ml was added to well-growing RRMECs (cell density 70-90%;
multiplicity of infection=20), and cells were infected for 18 h at
37°C. Subsequently, the viral medium was replaced with fresh
medium. After continuing the transduction for 72 h, the cells were
selected using 2.5 μg/ml puromycin. Stable SLC12A2 knockdown
cell line was established following 1 week of selection culture.
Cells were then expanded in medium containing puromycin at a
reduced concentration of 0.67 μg/ml. Reverse
transcription-quantitative PCR (RT-qPCR) and western blotting were
performed 1 week after initiation of this expansion phase to
examine knockdown efficiency. After confirming SLC12A2 knockdown,
cells were seeded in HG (25 mM) medium for 24 h.
Cell viability assessment
Cell viability was measured via Cell Counting Kit-8
(CCK-8) (24). After
transfection for 24 h, CCK-8 reaction solution (10 μl; cat.
no. C0037; Beyotime Institute of Biotechnology) was added to each
well and incubated for 2 h at 37°C. Cell viability was expressed as
the absorbance at 450 nm measured by the enzyme labeler (cat. no.
DR-3518G; Hiwell-Diatek Instruments Co., Ltd.).
Cell migration evaluation
Transwell was used to assess the cell migratory
capacity of cells (25). In
brief, 200 μl of cell suspension was added to the Transwell
chamber (8-μm pore size) at a density of
1×105/ml, while medium (containing 10% FBS) was added at
the bottom chamber, maintaining in the incubator for 24 h. Next,
the cells were fixed with methanol [30 min, room temperature (RT)]
and stained with 0.1% crystal violet (20 min, RT). After swabbing
the non-migratory cells, three fields were randomly selected for
observation and calculation of the number of migratory cells under
a light microscope (magnification, ×200).
Tube formation test
Tube formation test was performed according to a
previous study (26). After
trypsin digestion and re-suspension, cell suspensions were seeded
into 24-well plates coated with Matrigel gel with a density of
5×104/well and then incubated for 72 h. Matrigel was
polymerized at 37°C for 30 min before cell seeding. After that,
images of three fields were randomly selected to be captured with a
microscope (magnification, ×100), and the number of meshes was
quantified with ImageJ software (National Institutes of
Health).
Inflammatory factor detection
The secretion levels of VEGF (cat. no. ml002862),
IL-1β (cat. no. ml037361), IL-6 (cat. no. ml102828) and TNF-α (cat.
no. ml002859; all from Shanghai Enzyme-linked Biotechnology Co.,
Ltd.) in cell supernatant were estimated using commercial kits, in
accordance with the instructions.
ZO-1 detection
Changes in ZO-1 expression were evaluated by
immunofluorescence (IF) staining to reveal the permeability of the
blood-retinal barrier (BRB) (27). After fixation with formaldehyde
(4%, 15 min, RT) and permeabilization with Triton-X (1%), the cells
were blocked with a BSA solution (3%, 30 min, RT; Beijing Solarbio
Science & Technology Co., Ltd.). Subsequently, samples were
incubated with ZO-1 primary antibody (cat. no. AF5145; Affinity
Biosciences) at 4°C overnight and then reacted with fluorescent
Alexa Fluor® 488-conjugated goat anti-rabbit IgG H&L
secondary antibody (1:500, dilution; cat. no. ab150077; Abcam)/DAPI
(0.001 mg/ml) mixture for 30 min. Cell images were captured using a
fluorescence microscope.
Matrix metalloproteinases (MMPs)
examination
MMPs regulate various pathological processes and can
lead to the breakdown of tight junction proteins such as ZO-1 under
HG stimulation, thus destroying the integrity of the BRB (28). In the present study, kits for
MMP2 (cat. no. E-EL-R0618) and MMP9 (cat. no. E-EL-R3021; both from
Elabscience Biotechnology, Inc.) were employed to detect the
activity of these two factors in cell supernatants. The experiments
were performed according to the manufacturer's instructions.
Animal model establishment and
grouping
Animal experiments utilized 20 C57BL/6 (wild-type,
WT) and 20 SLC12A2−/− C57BL/6 (SLC12A2 specific
knockout, SLC12A2-KO) mice (age, 6 weeks; weight, 18-22 g), with
equal numbers of males and females (n=10 per sex) in each genotype
group. Genotyping of the mice was determined by PCR amplification.
Animal treatments followed the ARVO Statement for the Use of
Animals in Ophthalmic and Vision Research. All animal experiments
were conducted at Yangzhou University and were approved by the
Animal Care and Use Committee of Yangzhou University (approval no.
202410027; Yangzhou, China). WT mice were randomly divided into two
groups: control (WT) and diabetic (DR). Similarly, SLC12A2-KO mice
were classified into control (SLC12A2−/−) and diabetic
(DR-SLC12A2−/−) groups. There were 10 mice in each
group. All mice were housed in a pathogen-free facility with
standard conditions (circadian pattern: light hours 6:00-18:00;
temperature: 22-24°C; humidity: 30-50%). As described previously
(29), DR and
DR-SLC12A2−/− mice were intraperitoneally injected with
STZ (50 mg/kg) for 7 consecutive days. For the control groups (WT
and SLC12A2−/−), mice were intraperitoneally injected
with the same volume of citrate buffer. At the end of the
treatment, the body weight of the mice was recorded. Meanwhile,
blood glucose was measured at the tail by a glucose meter (OneTouch
Verio Vue, Johnson & Johnson Medical (China) Ltd.). Mice with
blood glucose levels greater than 16.7 mmol/l were considered
diabetic mice for the study. Venous blood (tail vein) was collected
to evaluate the glucolipid metabolism of mice. Subsequently, the
mice were deeply anesthetized by intraperitoneal injection of 2%
sodium pentobarbital (40 mg/kg), followed by euthanasia via
cervical dislocation. Death was confirmed by the absence of a
heart-beat and spontaneous respiration. The retinal specimens were
collected for further experiments.
Detection of blood biochemical
indexes
Venous blood was centrifuged at 1,000 × g, 4°C for
15 min, and serum samples were harvested. The concentrations of
triglyceride (TG), total cholesterol (TC), low-density lipoprotein
cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C)
and glycated hemoglobin (HbA1c) in serum were determined using a
biochemical autoanalyzer (30).
Moreover, commercial Enzyme-linked immunosorbent assay (ELISA) kits
were applied to detect the levels of serum inflammatory factors,
including VEGF (cat. no. PV957), IL-1β (cat. no. PI301), IL-6 (cat.
no. PI326) and TNF-α (cat. no. PT512; all from Beyotime Institute
of Biotechnology), followed by quantification of the concentrations
by measuring absorbance with a microplate reader. All experiments
were conducted following the manufacturer's provided protocol.
Hematoxylin and eosin (H&E)
staining
H&E staining was utilized to assess the
pathological changes in the retina, including ganglion cell layer
(GCL), inner plexiform layer, inner nuclear layer (INL), outer
plexiform layer and outer nuclear layer (ONL) (31). Briefly, retinal samples were
fixed in 4% paraformaldehyde at RT for 24 h, dehydrated with an
ethanol gradient, and embedded in paraffin. Slices with a thickness
of 5 μm were cut from the sample on a microtome, and then
H&E staining was performed according to the standard procedures
(32). Structural changes in the
tissue were observed under a light microscope, and retinal
thickness was quantified.
Apoptosis assay
Terminal deoxynucleotidyl transferase dUTP nick-end
labeling (TUNEL) approach was employed to assess apoptosis status
(33). For in vitro
observation, cells and nuclei were stained with TUNEL (cat. no.
C1082; 60 min, 37°C) and DAPI (cat. no. C1005; both from Beyotime
Institute of Biotechnology; 10 min, RT) reagents. For tissue
detection, retinal sections were deparaffinized/rehydrated, treated
with proteinase K for 30 min, and then washed three times with PBS.
Afterwards, the tissue sections were stained successively with
TUNEL (60 min, 37°C) and DAPI (0.001 mg/ml, 10 min, RT) reagents.
Finally, the staining of cells or tissue sections was observed
under a fluorescence microscope. Among them, blue and green
fluorescence represented nuclei and TUNEL-positive cells,
respectively.
CD31 measurement
To evaluate pathological changes in blood vessels,
the IF method was employed to detect the expression levels of CD31,
a specific marker for vascular endothelial cells (34). Retinal sections were incubated
with primary antibody anti-CD31 (cat. no. ab222783; Abcam)
overnight at 4°C. After washing with PBS, the samples were
incubated with secondary antibody for 1 h at 37°C, followed by
staining of nuclei with DAPI (0.001 mg/ml). Fluorescence images
were captured by confocal fluorescence microscopy.
RT-qPCR
RT-qPCR assay was undertaken as previously described
(35). Total RNA from cells or
retinal tissues was prepared by using TRIzol (cat. no. 15596018CN;
Thermo Fisher Scientific, Inc.), followed by reverse transcription
to cDNA using FastKing RT SuperMix (cat. no. KR118-02; Tiangen
Biotech Co., Ltd.) according to the manufacturer's protocol. The
mRNA was quantified using SYBR Green PCR Master Mix (cat. no.
A4004M; Xiamen Life Internet Technology Co., Ltd.) and a Real-time
PCR system (cat. no. CFX96 Touch; Bio-Rad Laboratories, Inc.).
RT-qPCR was conducted under the following thermocycling conditions:
An initial denaturation at 95°C for 10 min, followed by 40 cycles
of denaturation at 95°C for 12 sec, and annealing/extension at 60°C
for 40 sec. Finally, the relative gene expression was calculated
via 2−ΔΔcq (36),
with GAPDH as a stable reference. The primer sequences are
displayed in Table SI.
Western blot analysis
Western blot analysis was conducted as previously
described (35). Briefly, total
protein was collected from cultured cell or retinal tissues using
RIPA lysis buffer (cat. no. P0013B; Beyotime Institute of
Biotechnology), and protein concentration was determined by using
the BCA assay. The proteins (25 μg) separated by
electrophoresis were transferred to PVDF membrane and blocked by
skim milk powder (5%) for 60 min at RT. Furthermore, the membranes
were incubated overnight with the primary antibody at 4°C and
continued incubation with goat anti-rabbit IgG for 60 min at RT.
The bands were developed with ECL reagent (cat. no. P1000; Applygen
Technologies, Inc.), and the gray values were quantified with
ImageJ software (version 1.53k; National Institutes of Health). All
western blot data were normalized to GAPDH. Detailed information on
the antibodies used is provided in Table SII.
Statistics
Statistical analyses were done using R (version
4.3.3; bioinformatics analysis) or GraphPad Prism (version 8.0;
Dotmatics; experimental section). Measurement data were exhibited
as the mean ± SD, with experiments replicated at least three times.
Group differences were analyzed using one-way analysis of variance
(ANOVA) followed by Tukey's multiple comparison post-test.
P<0.05 was considered to indicate a statistically significant
difference.
Results
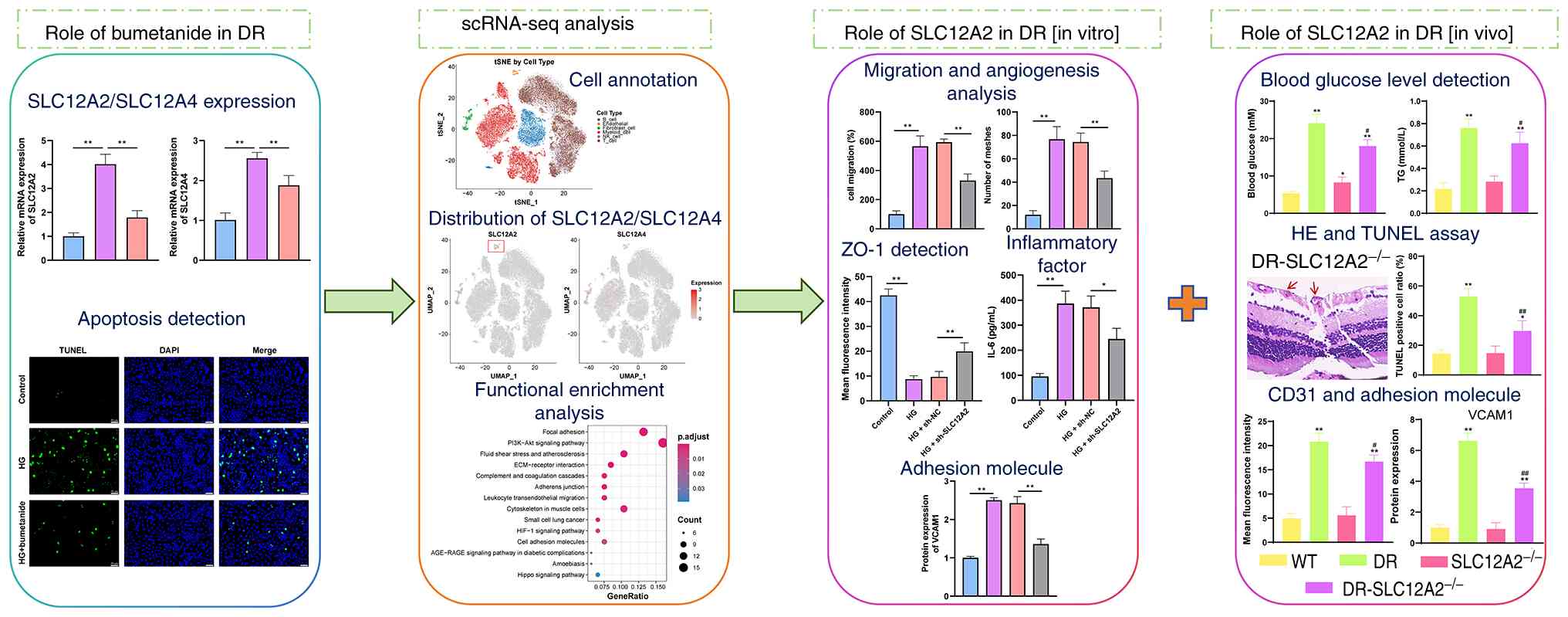
Bumetanide mitigates SLC12A2, SLC12A4 and
apoptosis in HG-treated RRMECs
Considering the protective effects of bumetanide
against retinopathy, the expression levels of these two genes were
determined using RT-qPCR. Results showed that the mRNA expression
levels of SLC12A2 and SLC12A4 in the HG group were obviously
increased compared with those in the control group. However,
bumetanide treatment significantly reduced their expression
compared with that in the HG (Fig.
2A). Retinal cell apoptosis is an early feature of DR (37), and apoptosis-related indices were
measured. TUNEL staining indicated that apoptosis was more serious
in the HG group than in the control group; however, it was
significantly alleviated in the HG group after treatment with
bumetanide (Fig. 2B).
Afterwards, western blotting was conducted to examine the
expression of apoptotic proteins. Compared with the HG group,
bumetanide significantly suppressed protein expression levels of
cleaved-caspase 3, Bax, TNF-α, IL-1β and VEGF, while promoting
BCL-2 protein expression, with no significant alteration in the
total caspase 3 level (Fig. 2C).
Taken together, bumetanide reversed retinal cell apoptosis, and its
therapeutic effects involved the modulation of SLC12A2 and SLC12A4
expression.
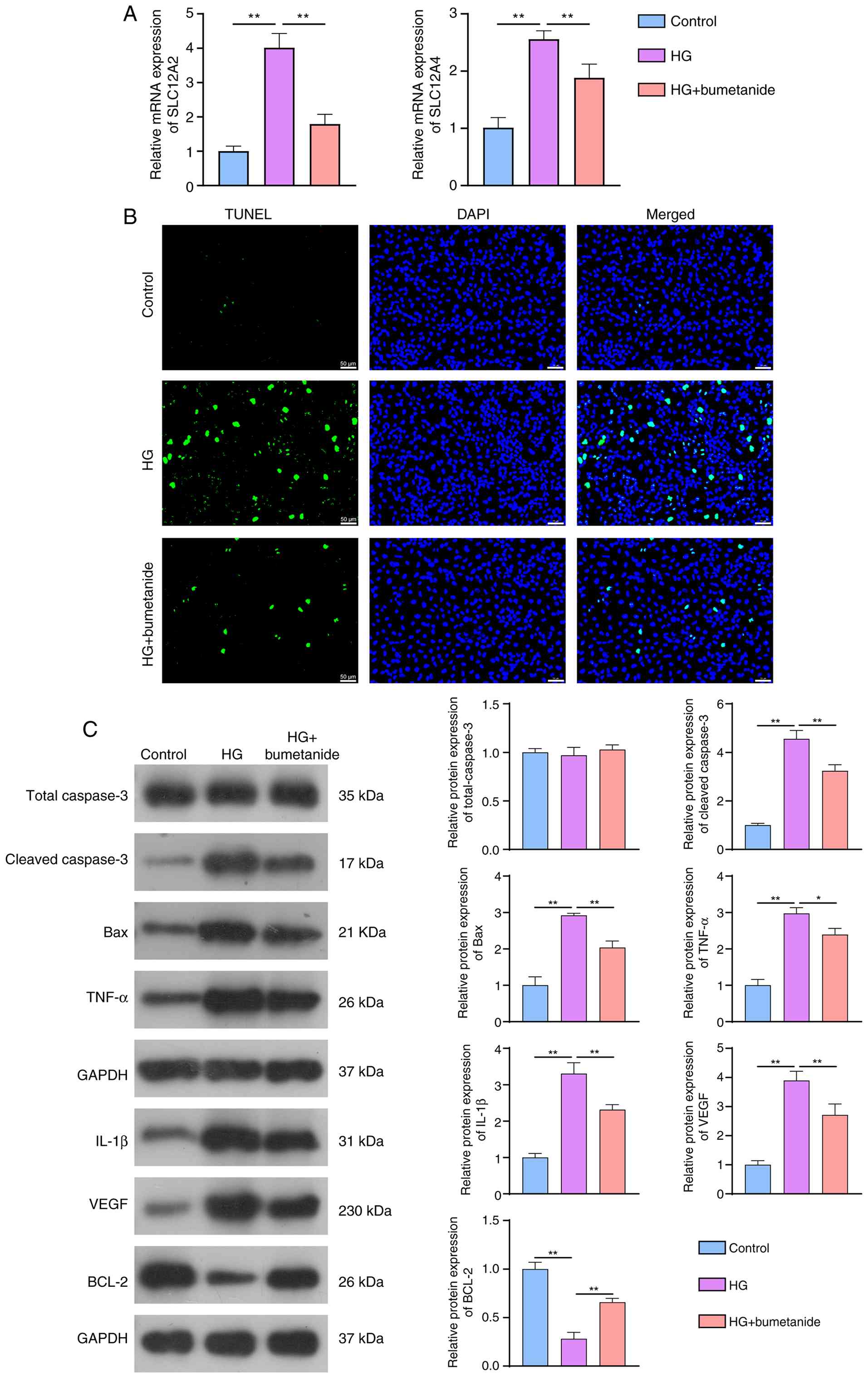
Single-cell landscapes
The three scRNA seq datasets used in the present
study were obtained from the GEO database. After quality control
and filtering of the data, 38,744 cells and 21,412 genes were
identified. Next, the three datasets were analyzed using
downscaling and de-batching. The samples from the different
datasets were well integrated without a significant batch effect,
making them suitable for subsequent cluster analysis (Fig. 3A). The top 2,000 highly variable
genes were selected for downstream analysis. Cluster analysis of
the cells was performed using the Seurat package, and 18 cell
clusters were obtained. Results were visualized by using a t-SNE
diagram (Fig. 3B). From these
clusters, the following six known cell types were identified with
reference to cell type marker genes recorded in the cell marker
database: T cells, NK cells, B cells, myeloid cells, endothelial
cells and fibroblast cells (Fig.
3C).
Next, focus was addressed on the expression
distribution of the key genes SLC12A2 and SLC12A4 in different
cells. Results revealed that SLC12A2 was mainly enriched in
endothelial cells, whereas SLC12A4 expression was enhanced in
fibroblast cell clusters (Fig. 3D
and E). Microvascular endothelial cells are the main targets of
hyperglycemic damage in the pathological microenvironment of
diabetes. Retinal endothelial cell dysfunction is closely
associated with DR (16). Hence,
the functional enrichment analysis of the top 200 marker genes in
endothelial cells was performed. GO analysis indicated that these
genes were primarily enriched in regulation of angiogenesis
(GO-BP), collagen-containing extracellular matrix (GO-CC) and
cadherin binding (GO-MF) (Fig.
3F). Furthermore, KEGG enrichment analysis suggested that these
genes were involved in adhesion molecule-related pathways, such as
focal adhesion, adherens junction and cell adhesion molecules
(Fig. 3G). Overall, the
contribution of SLC12A2 and endothelial cells to DR pathology
deserves further exploration.
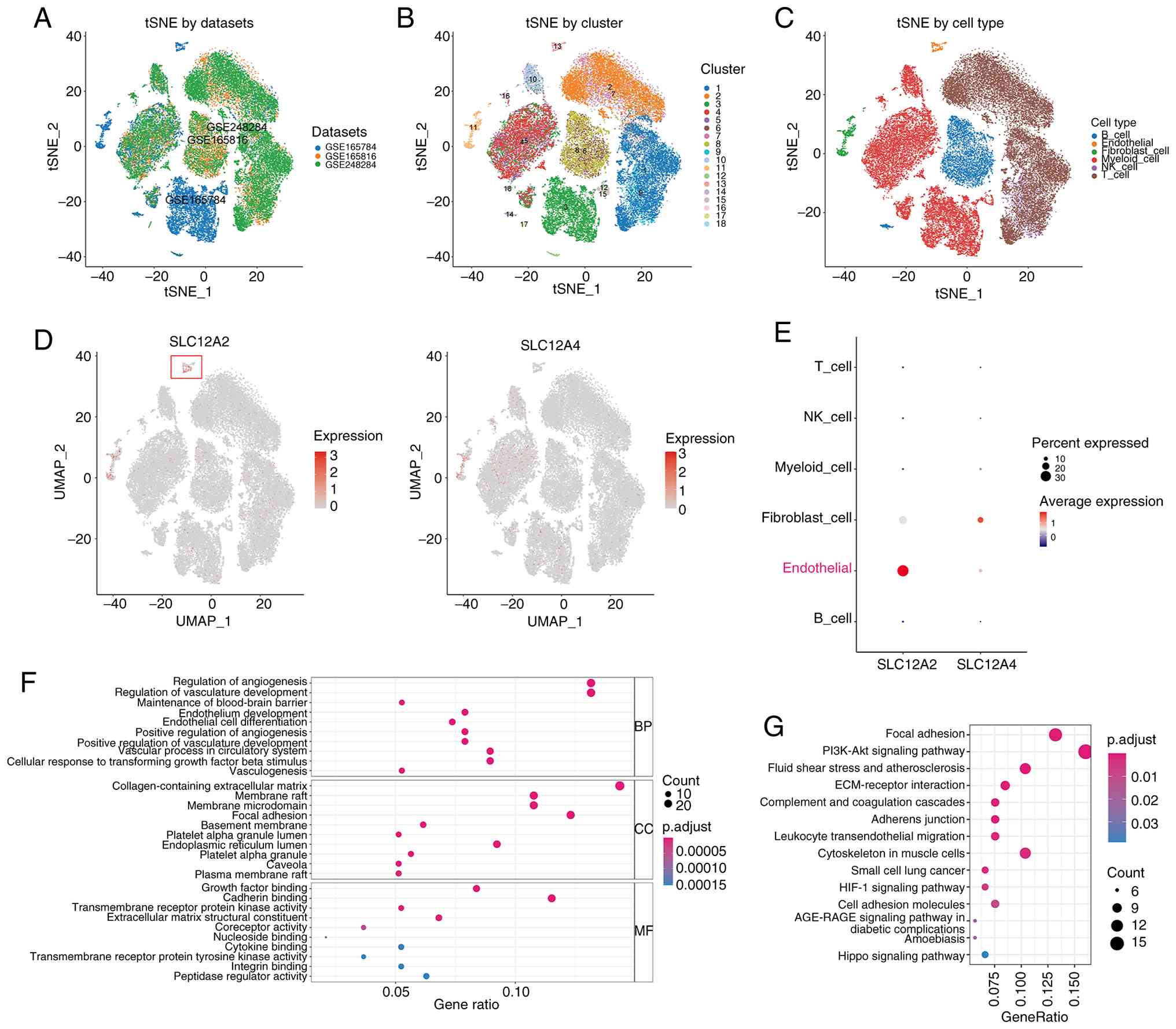
Knockdown of SLC12A2 alleviates
HG-induced abnormal migration and angiogenesis of HG-induced
RRMECs
To explore the engagement of SLC12A2 in the cell
model, RRMECs with SLC12A2 knockdown were generated via lentiviral
delivery of short hairpin RNA (Fig.
4A and B). Under HG conditions, CCK-8 analysis indicated that
the viability of RRMECs was significantly reduced; however, the
cell activity was restored after SLC12A2 knockdown (Fig. 4C). Cell migration and the number
of meshes were also measured. Exposure to HG increased the number
of migratory RRMECs, which was attenuated by the SLC12A2 knockdown
(Fig. 4D and E). HG observably
enhanced the tube-forming capacity of cells, but this effect was
weakened by downregulation of SLC12A2 (Fig. 4F and G). Overall, SLC12A2
knockdown protected cells from HG damage.
Knockdown of SLC12A2 inhibits
inflammatory factors and restores retinal endothelial barrier
function in HG-treated RRMECs
The pathophysiological mechanisms of DR are complex
and involve the increased secretion of VEGF and pro-inflammatory
mediators (38). As previously
reported, significant increases in VEGF and inflammatory factors
(IL-1β, IL-6 and TNF-α) were also observed in the HG group, which
were ameliorated by SLC12A2 knockdown (Fig. 5A). ZO-1 is a tight connexin, and
its abnormality can disrupt the BRB and increase the permeability
of the retinal vasculature (39). Additionally, MMPs are involved in
BRB disruption in DR. Therefore, the effect of SLC12A2 knockdown on
these indicators was explored. As demonstrated in Fig. 5B and C, compared with the control
group, the fluorescence intensity of ZO-1 was significantly
inhibited in the HG group, while the levels of MMP2 and MMP9 were
enhanced. Notably, the aberrant expression of ZO-1 and MMP2/9 in HG
was clearly reversed after SLC12A2 knockdown.
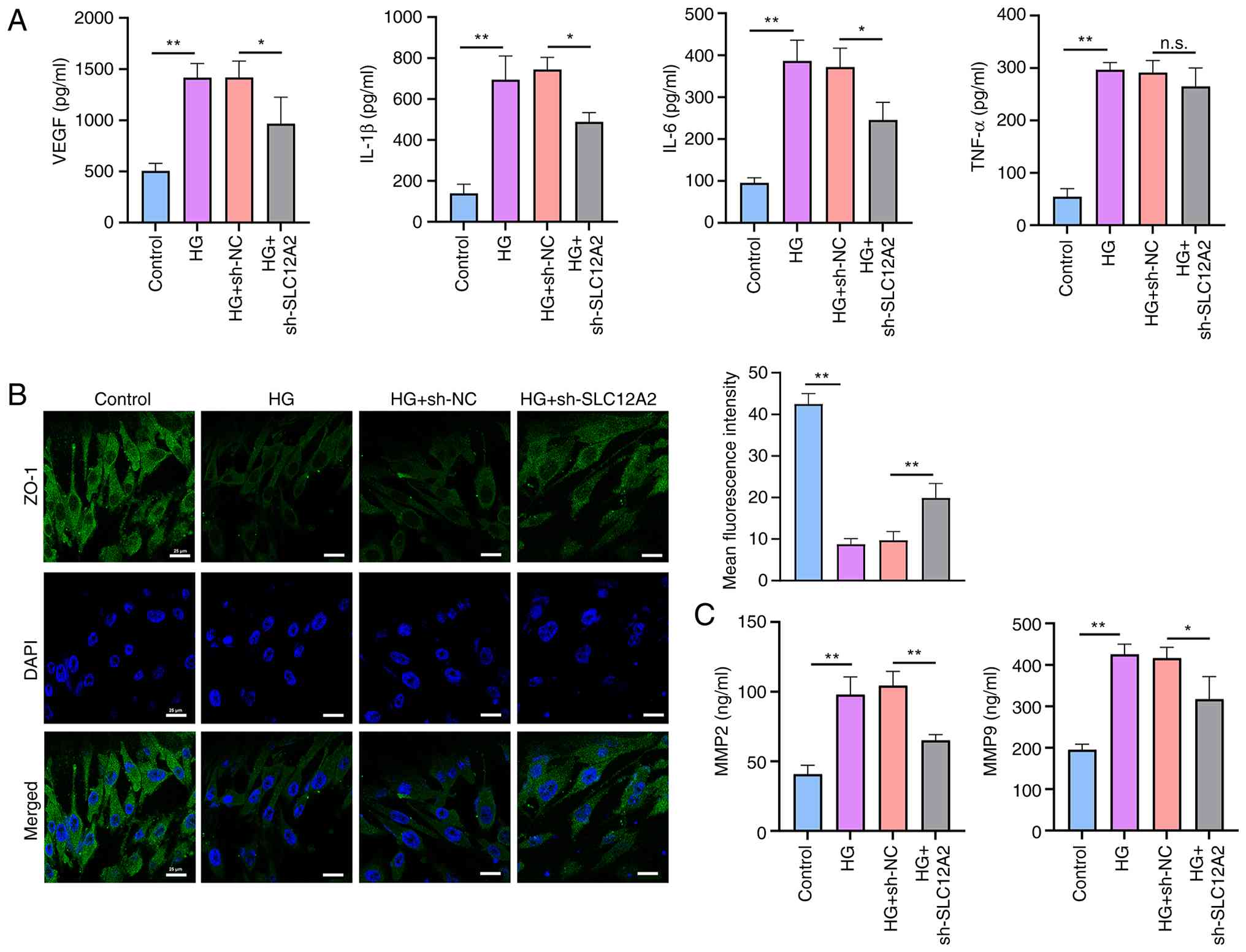 | Figure 5Effect of SLC12A2 knockdown on
inflammatory factor release and blood-retinal barrier in HG-treated
rat retinal microvascular endothelial cells. (A) Levels of VEGF,
IL-1β, IL-6 and TNF-α detected by ELISA. (B) Immunofluorescence
staining showing the ZO-1 expression in each group. (C) Levels of
MMP2 and MMP9 detected by commercial kits. Data from three
independent experiments are indicated as the mean ± SD (n=3).
*P<0.05 and **P<0.01. HG, high glucose;
VEGF, vascular endothelial growth factor; IL-1β, interleukin-1Beta;
IL-6, interleukin-6; TNF-α, tumor necrosis factor-alpha; ZO-1,
zonula occludens-1; MMP, matrix metalloproteinase; n.s., not
significant. |
Knockdown of SLC12A2 suppresses the
expression of cell adhesion-related molecules in HG-treated
RRMECs
Cell adhesion molecules have been found to be
involved in the regulation of diseases in scRNA-seq analyses, and
have been reported to be positively correlated with DR severity
(40). Thus, the regulatory role
of SLC12A2 in cell adhesion-related molecules was confirmed.
Compared with the control group, HG treatment caused a distinct
upregulation of mRNA and protein expression of multiple adhesion
molecules, such as VCAM1, ICAM1, PECAM-1, VE-cadherin, E-Selectin
and P-Selectin (Fig. 6A and B).
In addition, after the transfection of sh-SLC12A2 into HG-treated
cells, the expression levels of these factors displayed a downward
trend. Except for PECAM-1 and VE-cadherin, the expression changes
of the other molecules showed statistical differences.
Collectively, these results suggested that SLC12A2 knockdown
inhibited the enhanced expression of adhesion-related molecules
induced by HG stimulation.
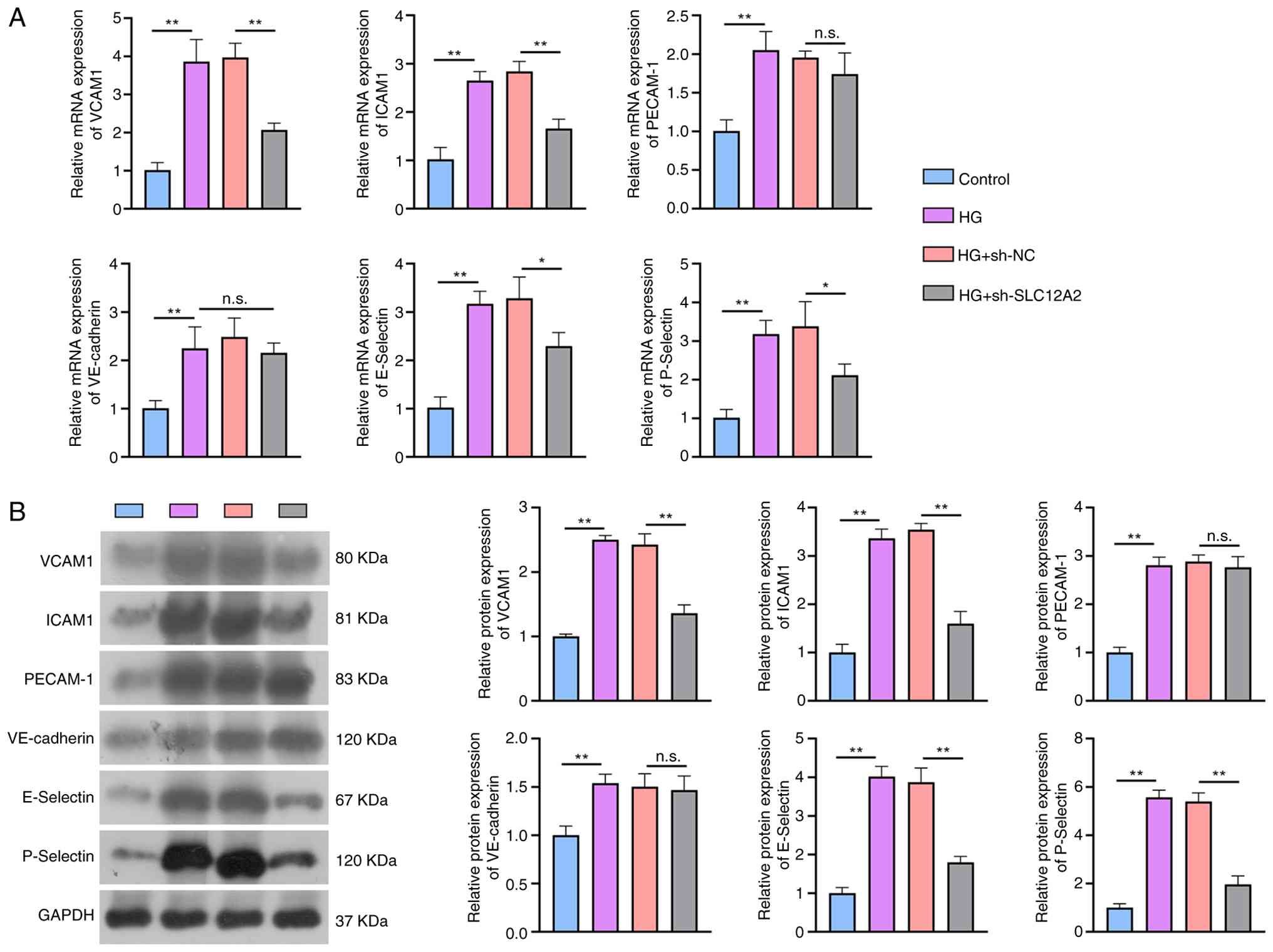 | Figure 6Effect of SLC12A2 knockdown on
adhesion molecules in HG-treated rat retinal microvascular
endothelial cells. (A) Reverse transcription-quantitative PCR and
(B) western blot assays for expression levels of adhesion
molecules, including VCAM1, ICAM1, PECAM-1, VE-cadherin, E-Selectin
and P-Selectin. Data from three independent experiments are
indicated as the mean ± SD (n=3). *P<0.05 and
**P<0.01. HG, high glucose; VCAM1, vascular cell
adhesion molecule-1; ICAM1, intercellular adhesion molecule-1;
PECAM-1, platelet endothelial cell adhesion molecule-1; VE,
vascular endothelial; sh-, short hairpin; NC, negative control. |
SLC12A2 deficiency ameliorates
hyperglycemic and inflammatory effects in STZ-induced diabetic
mice
Based on these results, SLC12A2 may be a promising
target for protecting retinal cells from hyperglycemic damage.
Hence, the association between SLC12A2 and pathological changes in
DR was evaluated by using an in vivo gene knockout. First,
RT-qPCR and western blot results revealed that SLC12A2 expression
levels in SLC12A2−/− mice were significantly reduced
compared with those in WT mice, confirming the successful
construction of the SLC12A2-KO model (Fig. S1A and B). In addition, there
were no significant differences in body weight, serum lipid
concentrations, or HbA1c levels between WT and
SLC12A2−/− mice. A total of 7 days after STZ injection,
DR and DR-SLC12A2−/− mice displayed increased body
weight and blood glucose levels compared with those in control
groups (WT and SLC12A2−/−), indicating that the mice
developed diabetes. Importantly, blood glucose levels, TG, TC and
HbA1c levels were significantly lower in the
DR-SLC12A2−/− group compared with the DR group (Fig. 7A-C). The secretion levels of
inflammatory factors in mouse serum were also measured via ELISA.
Results revealed that the diabetic mice had significantly elevated
levels of inflammatory and angiogenic factors, including VEGF,
IL-1β, IL-6 and TNF-α. However, SLC12A2 deficiency reduced the
secretion of proinflammatory factors in diabetic mice (Fig. 7D). These data further confirmed
that SLC12A2 may improve DR by controlling blood glucose levels and
suppressing inflammatory responses.
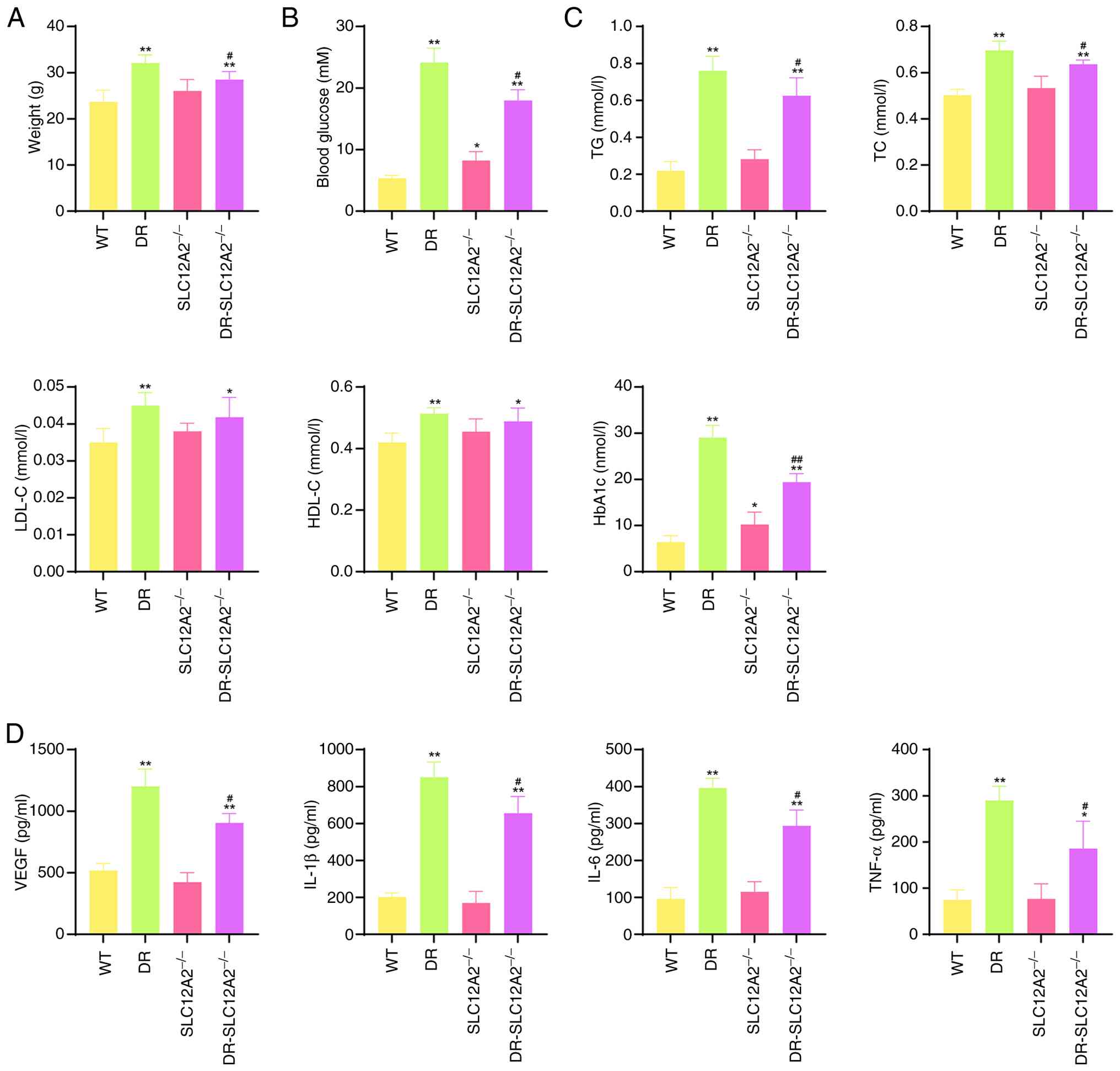 | Figure 7SLC12A2 deficiency ameliorates
hyperglycemic and inflammatory effects in streptozotocin-induced
diabetic mice. (A) Body weight and (B) blood glucose levels of mice
at the end of the experiment. (C) Serum lipid and HbA1c levels of
all mice. (D) Levels of inflammatory factors (VEGF, IL-1β, IL-6 and
TNF-α) detected by commercial ELISA kits. Results are expressed as
the mean ± SD (n=10/group). *P<0.05 and
**P<0.01 vs. the WT group; #P<0.05 and
##P<0.01 vs. the DR group. HbA1c, glycated
hemoglobin; VEGF, vascular endothelial growth factor; IL-1β,
interleukin-1Beta; IL-6, interleukin-6; TNF-α, tumor necrosis
factor-alpha; WT, wild-type; TG, triglyceride; TC, total
cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C,
high-density lipoprotein cholesterol; DR, diabetic retinopathy. |
SLC12A2 deficiency inhibits retinal
structural damage in STZ-induced diabetic mice
Following, H&E staining was utilized to observe
pathological changes in the retina (Fig. 8A). Results showed that the
structure of the retinal layers in the WT group was clear, with the
INL and ONL cells arranged in an orderly and tight manner. However,
in the DR group, a neovascularized cavity was observed in the
retina (red arrows), and its structure was markedly dilated and
vacuolated. The GCL appeared to be fractured, and the cells in each
layer were loosely arranged, indicating significant pathological
changes in the retinal tissue. Notably, the structure and shape of
the retina in the DR-SLC12A2−/− mice were clearly
improved compared with those in DR mice. Furthermore, statistical
analysis indicated that DR mice had an increased number of
neovascular nuclei, but a significantly thinner INL than WT mice.
SLC12A2 knockout significantly ameliorated these pathological
impairments in diabetic mice, as evidenced by increased retinal
thickness and a reduced number of neovascular nuclei. Subsequently,
TUNEL was employed to explore the effect of SLC12A2 deficiency on
retinal apoptosis in the diabetic state (Fig. 8B). The proportion of
TUNEL-positive cells in the DR group was significantly higher than
that in the WT group. As expected, SLC12A2 deficiency markedly
reduced the rate of apoptosis. Collectively, these results
suggested that SLC12A2 deficiency has a significant protective
effect against diabetic retinal damage.
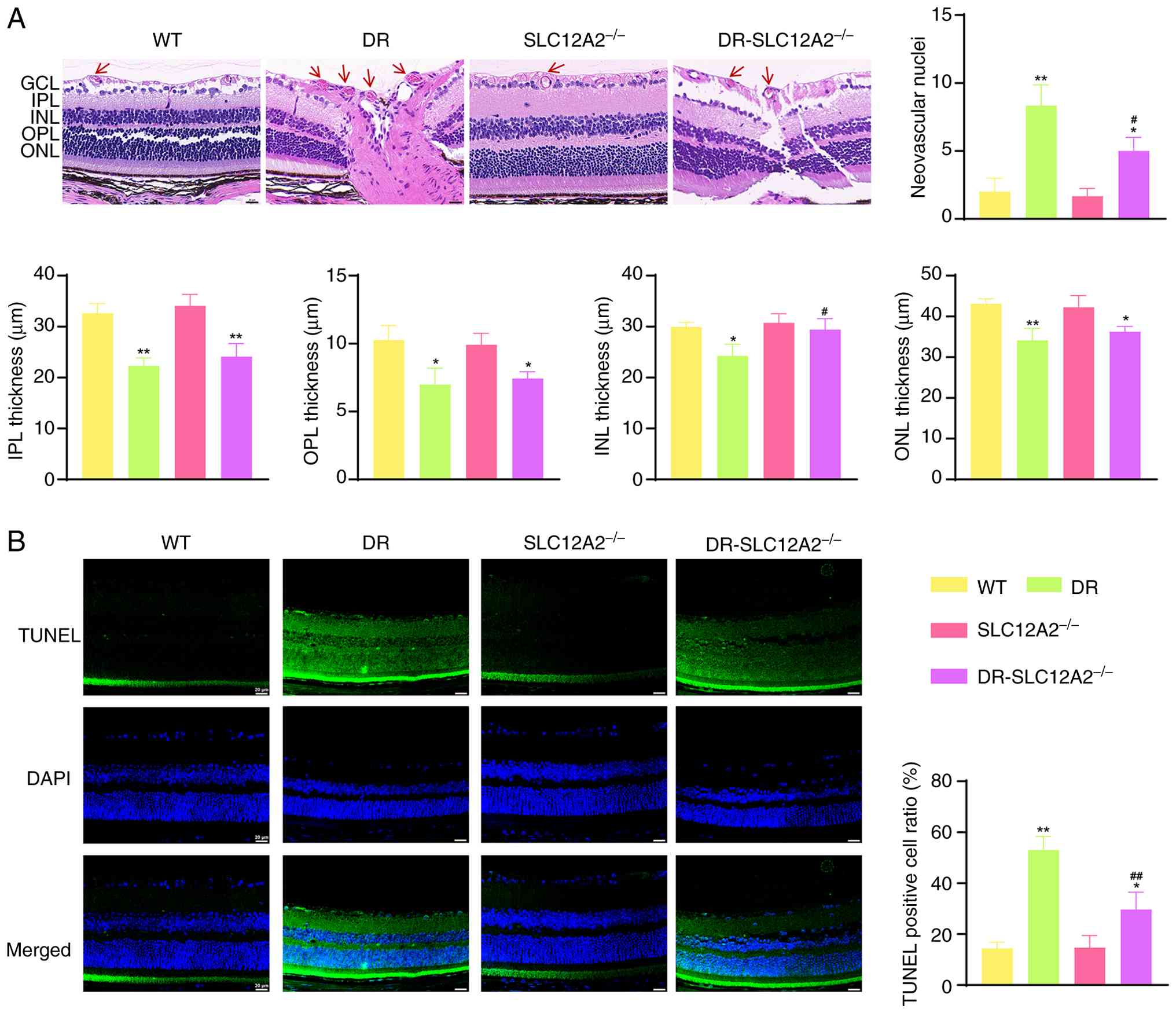 | Figure 8SLC12A2 deficiency inhibits retinal
structural damage in streptozotocin-induced diabetic mice. (A)
Representative H&E-stained images and retinal thickness
measurements in each group (magnification, ×40). The red arrow
represents neovascular nuclei. (B) Representative TUNEL assay
images in each group (magnification, ×40). Results are expressed as
the mean ± SD (n= 10/group). *P<0.05 and
**P<0.01 vs. the WT group; #P<0.05 and
##P<0.01 vs. the DR group. TUNEL, terminal
deoxynucleotidyl transferase dUTP nick-end labeling; GCL, ganglion
cell layer; IPL, inner plexiform layer; INL, inner nuclear layer;
OPL, outer plexiform layer; ONL, outer nuclear layer; WT,
wild-type; DR, diabetic retinopathy. |
SLC12A2 deficiency suppresses cell
adhesion molecules expression in STZ-induced diabetic mice
Considering that the protective effect of the
SLC12A2 knockdown in endothelial cells was achieved by suppressing
the expression of adhesion-related molecules, this finding was
further validated in vivo. As demonstrated in Fig. 9A, the protein expression level of
CD31 in the retinas of STZ-triggered DR mice significantly
increased, while SLC12A2 knockout suppressed its expression.
Besides, the mRNA and protein expression levels of multiple
adhesion molecules, including VCAM1, ICAM1, and VE-cadherin, were
significantly increased in the retinas of DR mice compared with
those of WT mice. SLC12A2 knockout showed the opposite changes in
the expression of these genes (Fig.
9B and C). Collectively, the aforementioned results suggested
that SLC12A2 deficiency alleviates the pathological changes in DR
by inhibiting the expression of cell adhesion-related
molecules.
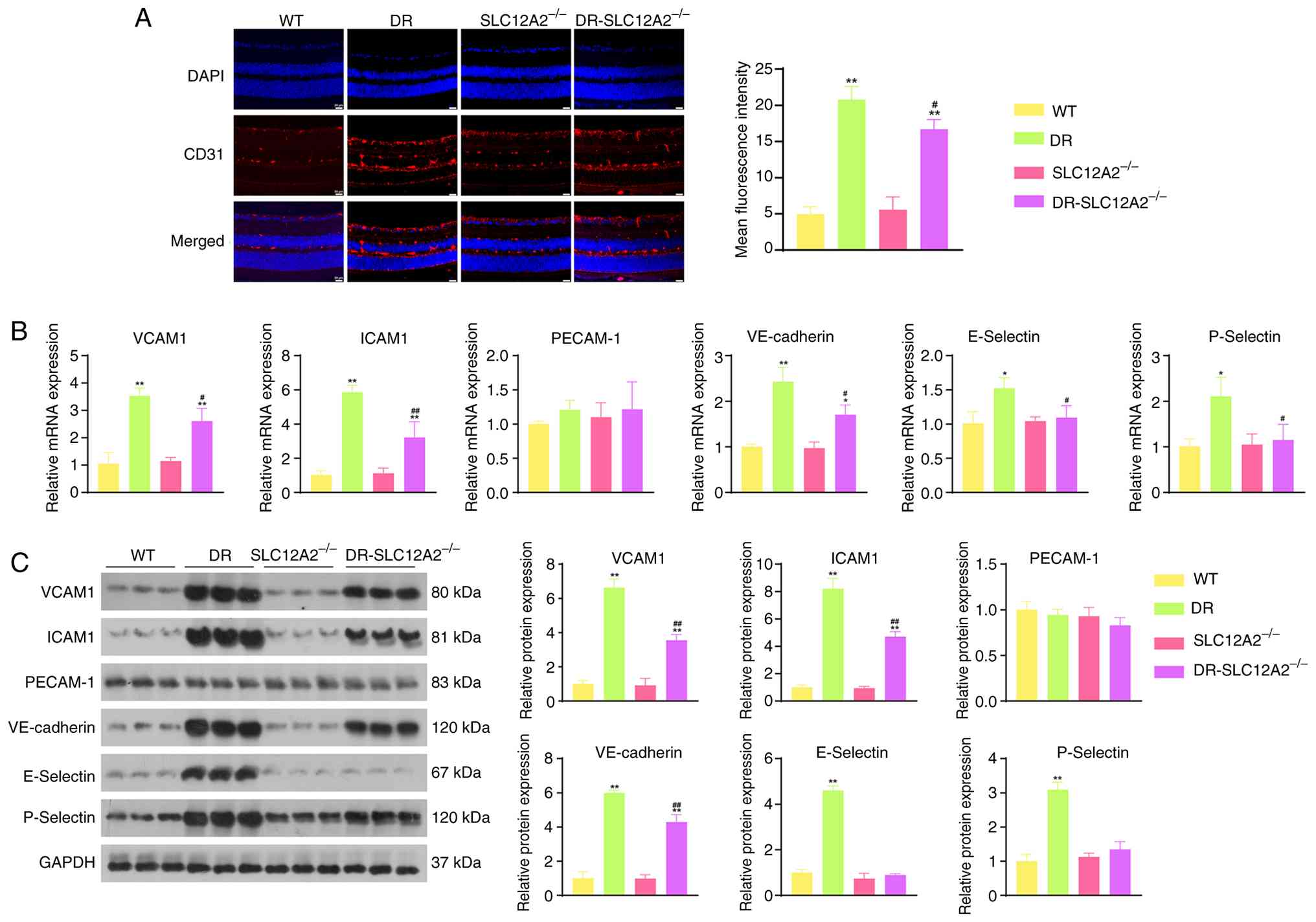 | Figure 9SLC12A2 deficiency suppresses
adhesion molecule expression in streptozotocin-induced diabetic
mice. (A) Immunofluorescence images of CD31 (magnification, ×40).
(B and C) Expression of VCAM1, ICAM1, PECAM-1, VE-cadherin,
E-Selectin and P-Selectin determined by reverse
transcription-quantitative PCR and western blotting. Results are
expressed as the mean ± SD (n=10/group). *P<0.05 and
**P<0.01 vs. the WT group; #P<0.05 and
##P<0.01 vs. the DR group. VCAM1, vascular cell
adhesion molecule-1; ICAM1, intercellular adhesion molecule-1;
PECAM-1, platelet endothelial cell adhesion molecule-1; VE,
vascular endothelial; WT, wild-type; DR, diabetic retinopathy. |
Discussion
Anti-VEGF approaches play a prominent role in the
treatment of DR (41). However,
treating patients who respond poorly to this approach remains a
clinical challenge, and therapeutic agents against novel
pathological targets may be required. In the present study,
scRNA-seq analysis revealed that DR was composed of six cell types,
and SLC12A2 was predominantly expressed in endothelial cells.
Endothelial cell markers were significantly involved in
angiogenesis regulation and adhesion molecule-related biological
functions. Importantly, it was found that bumetanide and its drug
target SLC12A2 inhibited abnormal apoptosis and angiogenesis in an
in vitro DR model. It was further demonstrated in
vivo that SLC12A2 deficiency suppressed hyperglycemia and
maintained normal retinal anatomy in diabetic mice, thereby
preventing the development of DR. Notably, the underlying vascular
protective mechanism of SLC12A2 may involve the inhibition of cell
adhesion molecules, leading to a reduction in apoptotic and
inflammatory events (Fig. 10).
These findings suggested that SLC12A2is a promising biomarker for
the development of DR-targeted drugs.
Bumetanide modulates retinal function by reducing
retinal swelling and damage (42). This mechanism involves
competition with Cl− for the second chloride-binding
site, which exerts the functions of inhibiting NKCC1 (SLC12A2)
(43). In the present study, for
the first time to the best of the authors' knowledge, the effect of
bumetanide on HG-treated RRMECs was observed. Bumetanide
significantly suppressed the expression of SLC12A2 and SLC12A4 in
HG-exposed endothelial cells (RRMECs). Endothelial cells, the
innermost layer of blood vessels, are the primary targets of damage
under HG conditions (44).
Similar to previous results (8),
in the HG-induced RRMECs injury model, bumetanide suppressed the
expression of apoptotic proteins (cleaved-caspase 3 and Bax) while
increasing the levels of anti-apoptotic proteins (BCL-2), thus
reducing the number of apoptotic cells. Importantly, inflammatory
molecules and neovascularization play crucial roles in the
pathogenesis and progression of DR (45-47). It was observed that bumetanide
treatment significantly reduced the expression of inflammatory
factors (TNF-α and IL-1β) and VEGF in HG-stimulated cells,
consistent with findings reported by Guzel et al (48). Taken together, bumetanide
ameliorates diabetes-induced retinal inflammation and vascular
dysfunction, thereby attenuating DR progression.
The protective effects of bumetanide were inspiring
and thus its underlying mechanisms were investigated. Two
pharmacological targets of bumetanide were explored. Bioinformatics
analysis of scRNA-seq and RNA-seq data revealed that SLC12A2 was
predominantly highly expressed in endothelial cells, whereas
SLC12A4 was primarily localized in fibroblasts. Microvascular
dysfunction is a central pathogenic hallmark of DR. As a
fundamental structural and functional unit of the retinal blood
vessels, endothelial cell stability is essential for maintaining
vascular integrity. In the early stages of DR, hyperglycemic
stimulation induces apoptosis, phenotypic alterations, and enhanced
migratory capacity in retinal endothelial cells, consequently
leading to increased vascular permeability and pathological
neovascularization. These changes ultimately contribute to the
development of microaneurysms, fluid leakage and tissue damage
(49,50). Furthermore, as DR progresses,
dysfunctional endothelial cells promote excessive accumulation of
extracellular matrix proteins in the retina and activate fibroblast
proliferation (51). Massive
proliferation of fibroblasts triggers retinal fibrosis, leading to
severe visual impairments such as tractional retinal detachment
(52). Taken together,
endothelial cell injury may represent a critical early event in DR,
allowing the invasion of pathogenic factors that occur before other
events (53). Therefore, the
protection of endothelial cells is a potential therapeutic target
for early-stage DR interventions. However, current treatment
modalities primarily address advanced DR, and the ability to
restore impaired vision remains limited (54). Based on this evidence, SLC12A2
was selected, which is highly expressed in endothelial cells, as
the target for functional experiments.
As expected, in HG-treated RRMECs, SLC12A2 knockdown
promoted cell proliferation, while attenuating cell migration and
pathological angiogenesis. These results suggested that SLC12A2
knockdown attenuates cell damage in DR, which is consistent with
the results of a previous study (35). The role of inflammatory processes
in the pathological changes associated with DR has received
extensive attention. There is crosstalk between inflammation and
neovascularization in the pathogenesis of DR, and inhibition of
inflammation may facilitate the control of retinal angiogenesis
(38). In the present study,
SLC12A2 knockdown markedly reduced the release of inflammatory
factors in HG-treated RRMECs, such as VEGF, IL-1β, IL-6 and TNF-α.
The internal BRB is a highly selective endothelial barrier that
maintains tissue homeostasis by regulating permeability and
molecular transport between the circulatory system and the neural
retina (55). In patients with
DR, inner BRB (iBRB) disruption leads to retinal neurodegeneration
and vision loss (56).
Therefore, the effects of SLC12A2 knockdown were examined on the
expression of core functional proteins essential for iBRB
integrity. Results showed that SLC12A2 knockdown maintained the
integrity of the iBRB by upregulating ZO-1 and downregulating
MMP2/MMP9. Reportedly, reduced ZO-1 levels disrupt tight junctions
in endothelial cells, leading to dysfunction of the endothelial
barrier (57). Activation of
MMPs, particularly MMP2 and MMP9, represents an early event in DR
that induces mitochondrial damage and promotes retinal apoptosis in
vascular cells (pericytes and endothelial cells) (28,58). Similarly, SLC12A2 deficiency
alleviated the progression of DR in vivo by improving
retinal thickness and pathological changes (inflammation and
angiogenesis) in STZ-induced diabetic mice. Consequently, SLC12A2
knockdown may exert a protective effect against DR by preventing
iBRB breakdown through suppression of proinflammatory factors and
VEGF expression.
Subsequently, it was found that endothelial cell
markers were primarily involved in pathways related to angiogenesis
and cell adhesion. The upregulation of adhesion molecules is a
critical step in the pathogenesis of DR. Hyperglycemia-induced
proinflammatory cytokines secrete intercellular adhesion molecules
such as ICAM-1 and VCAM-1, which mediate leukocyte-endothelial cell
interactions to promote leukocyte recruitment, facilitating a
persistent inflammatory response, thereby leading to increased
retinal capillary permeability and stimulating pathological retinal
neovascularization (59-61). Therefore, observing the changes
in adhesion molecules caused by SLC12A2 knockdown is important. It
was found that SLC12A2 knockdown significantly rescued the
HG-induced enhanced expression of adhesion factors, including
VCAM1, ICAM1, E-Selectin and P-Selectin. VCAM1 promotes adhesion
between leukocytes and vascular endothelial cells, which is
positively correlated with the severity of DR (62). Indeed, a reduction in VCAM1 has
been reported in DR remission (32). ICAM1 plays a similar role and its
expression is inhibited by DR therapeutic drugs such as finerenone
(63). E-Selectin is present in
activated endothelial cells and plays an important role in
leukocyte recruitment to inflammatory areas (64). Controlling blood glucose reduces
E-Selectin levels, which may play a role in preventing DR (65). P-Selectin is also involved in
leukocyte recruitment, and its high levels are potentially
associated with DR (66). Taken
together, the inhibition of SLC12A2 alleviates DR-associated
pathological symptoms, indicating that SLC12A2 may be a valuable
therapeutic candidate against DR.
To the best of the authors' knowledge, this is the
first study to elucidate the protective role of bumetanide against
endothelial cell injury in DR and its specific molecular
mechanisms. However, the limitations of the present study warrant
further investigation. First, it was concluded that bumetanide
prevents DR progression by downregulating SLC12A2 to reduce
inflammatory factors, growth factors, and adhesion molecules in
endothelial cells. Nevertheless, the specific downstream targets of
SLC12A2 remain unknown. Second, the current results have only been
validated in HG-induced RRMECs and STZ-induced diabetic mouse
models, which may not fully replicate the complex pathological
environment of human DR. Third, although focus was mainly addressed
on the regulatory role of SLC12A2 in DR, whether SLC12A4, another
pharmacological target of bumetanide, also exerts a similar effect
remains unexplored. Therefore, more comprehensive and in-depth
studies on endothelially expressed SLC12A2 and SLC12A4 in DR are
warranted.
In summary, the current data indicate that SLC12A2,
a key pharmacological target of bumetanide, is effective in
alleviating pathological alterations in DR, including the
attenuation of the inflammatory response, suppression of adhesion
molecule expression, reduction of cell apoptosis, and enhancement
of BRB integrity. The present study is the first to demonstrate the
protective effects of bumetanide against DR, suggesting that
targeting SLC12A2 may represent a promising preventive or
therapeutic strategy against DR.
Supplementary Data
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
YZ and HY conceived and designed the research. YZ,
YH, DH and ZL acquired data. YH, DH, XW, HY, QX and MN analyzed and
interpreted data. YZ, TX, XW, HY, QX and MN performed statistical
analysis. YZ and YH drafted the manuscript. HY, XW and QX revised
the manuscript for important intellectual content. All authors
confirm the authenticity of all the raw data. All authors read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
Animal procedures were performed in compliance with
the ARRIVE guidelines and were approved from the Ethics Committee
of the Experimental Animal Center of Yangzhou University (approval
no. 202410027; Yangzhou, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgements
Not applicable.
Funding
No funding was received.
References
|
1
|
Tan TE and Wong TY: Diabetic retinopathy:
Looking forward to 2030. Front Endocrinol (Lausanne).
13:10776692022. View Article : Google Scholar
|
|
2
|
Teo ZL, Tham YC, Yu M, Chee M, Rim TH,
Cheung N, Bikbov MM, Wang YX, Tang Y, Lu Y, et al: Global
prevalence of diabetic retinopathy and projection of burden through
2045: Systematic review and meta-analysis. Ophthalmology.
128:1580–1591. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Himasa FI, Singhal M, Ojha A and Kumar B:
Prospective for diagnosis and treatment of diabetic retinopathy.
Curr Pharm Des. 28:560–569. 2022. View Article : Google Scholar
|
|
4
|
Ren J, Zhang S, Pan Y, Jin M, Li J, Luo Y,
Sun X and Li G: Diabetic retinopathy: Involved cells, biomarkers,
and treatments. Front Pharmacol. 13:9536912022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kaushik V, Gessa L, Kumar N and Fernandes
H: Towards a new biomarker for diabetic retinopathy: Exploring RBP3
structure and retinoids binding for functional imaging of eyes in
vivo. Int J Mol Sci. 24:44082023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lam TI, Anderson SE, Glaser N and
O'donnell ME: Bumetanide reduces cerebral edema formation in rats
with diabetic ketoacidosis. Diabetes. 54:510–516. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Guzel S, Cai CL, Ahmad T, Quan M, Valencia
GB, Aranda JV and Beharry KD: Bumetanide suppression of
angiogenesis in a rat model of Oxygen-induced retinopathy. Int J
Mol Sci. 21:9872020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen C, Fan P, Zhang L, Xue K, Hu J, Huang
J, Lu W, Xu J, Xu S, Qiu G, et al: Bumetanide rescues Aquaporin-4
depolarization via suppressing β-dystroglycan cleavage and provides
neuroprotection in rat retinal ischemia-reperfusion injury.
Neuroscience. 510:95–108. 2022. View Article : Google Scholar
|
|
9
|
Navas A, Jannus F, Fernández B, Cepeda J,
Medina O'Donnell M, Díaz-Ruiz L, Sánchez-González C, Llopis J, Seco
JM, Rufino-Palomares E, et al: Designing Single-molecule magnets as
drugs with dual Anti-inflammatory and anti-diabetic effects. Int J
Mol Sci. 21:31462020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hampel P, Römermann K, Gailus B, Johne M,
Gericke B, Kaczmarek E and Löscher W: Effects of the NKCC1
inhibitors bumetanide, azosemide, and torasemide alone or in
combination with phenobarbital on seizure threshold in epileptic
and nonepileptic mice. Neuropharmacology. 185:1084492021.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rivera A, Nasburg JA, Shim H, Shmukler BE,
Kitten J, Wohlgemuth JG, Dlott JS, Snyder LM, Brugnara C, Wulff H
and Alper SL: The erythroid K-Cl cotransport inhibitor
[(dihydroindenyl)oxy]acetic acid blocks erythroid Ca2+-activated K+
channel KCNN4. Am J Physiol Cell Physiol. 323:C694–C705. 2022.
View Article : Google Scholar
|
|
12
|
Tang F, Barbacioru C, Wang Y, Nordman E,
Lee C, Xu N, Wang X, Bodeau J, Tuch BB, Siddiqui A, et al: mRNA-Seq
whole-transcriptome analysis of a single cell. Nat Methods.
6:377–382. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Van Hove I, De Groef L, Boeckx B, Modave
E, Hu TT, Beets K, Etienne I, Van Bergen T, Lambrechts D, Moons L,
et al: Single-cell transcriptome analysis of the Akimba mouse
retina reveals cell-type-specific insights into the pathobiology of
diabetic retinopathy. Diabetologia. 63:2235–2248. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang X, Zhang F and Xu X: Single-cell RNA
sequencing in exploring the pathogenesis of diabetic retinopathy.
Clin Transl Med. 14:e17512024. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sun L, Wang R, Hu G, Liu H, Lv K, Duan Y,
Shen N, Wu J, Hu J, Liu Y, et al: Single cell RNA sequencing
(scRNA-Seq) deciphering pathological alterations in
streptozotocin-induced diabetic retinas. Exp Eye Res.
210:1087182021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu K, Gao X, Hu C, Gui Y, Gui S, Ni Q,
Tao L and Jiang Z: Capsaicin ameliorates diabetic retinopathy by
inhibiting poldip2-induced oxidative stress. Redox Biol.
56:1024602022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Theocharidis G, Thomas BE, Sarkar D, Mumme
HL, Pilcher WJR, Dwivedi B, Sandoval-Schaefer T, Sîrbulescu RF,
Kafanas A, Mezghani I, et al: Single cell transcriptomic landscape
of diabetic foot ulcers. Nat Commun. 13:1812022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liao D, Fan W, Li N, Li R, Wang X, Liu J,
Wang H and Hou S: A single cell atlas of circulating immune cells
involved in diabetic retinopathy. iScience. 27:1090032024.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hu Z, Mao X, Chen M, Wu X, Zhu T, Liu Y,
Zhang Z, Fan W, Xie P, Yuan S and Liu Q: Single-cell
transcriptomics reveals novel role of microglia in fibrovascular
membrane of proliferative diabetic retinopathy. Diabetes.
71:762–773. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xiang ZY, Chen SL, Qin XR, Lin SL, Xu Y,
Lu LN and Zou HD: Changes and related factors of blood CCN1 levels
in diabetic patients. Front Endocrinol (Lausanne). 14:11319932023.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hui Z, Chen YM, Gong WK, Lai JB, Yao BB,
Zhao ZJ, Lu QK, Ye K, Ji LD and Xu J: Shared and specific
biological signalling pathways for diabetic retinopathy, peripheral
neuropathy and nephropathy by high-throughput sequencing analysis.
Diab Vasc Dis Res. 19:147916412211229182022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gui F, You Z, Fu S, Wu H and Zhang Y:
Endothelial dysfunction in diabetic retinopathy. Front Endocrinol
(Lausanne). 11:5912020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kaur G, Song Y, Xia K, Mccarthy K, Zhang
F, Linhardt RJ and Harris NR: Effect of high glucose on
glycosaminoglycans in cultured retinal endothelial cells and rat
retina. Glycobiology. 32:720–734. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shi J, Lv H, Tang C, Li Y, Huang J and
Zhang H: Mangiferin inhibits cell migration and angiogenesis via
PI3K/AKT/mTOR signaling in high glucose- and hypoxia-induced
RRCECs. Mol Med Rep. 23:4732021. View Article : Google Scholar
|
|
25
|
Justus CR, Marie MA, Sanderlin EJ and Yang
LV: Transwell in vitro cell migration and invasion assays. Methods
Mol Biol. 2644:349–359. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lin Y, Luo G, Liu Q, Yang R, Sol Reinach P
and Yan D: METTL3-mediated RNA m6A modification regulates the
angiogenic behaviors of retinal endothelial cells by methylating
MMP2 and TIE2. Invest Ophthalmol Vis Sci. 64:182023. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li J, Lu X, Wei L, Ye D, Lin J, Tang X,
Cui K, Yu S, Xu Y and Liang X: PHD2 attenuates high-glucose-induced
blood retinal barrier breakdown in human retinal microvascular
endothelial cells by regulating the Hif-1α/VEGF pathway. Inflamm
Res. 71:69–79. 2021. View Article : Google Scholar
|
|
28
|
Giebel SJ, Menicucci G, Mcguire PG and Das
A: Matrix metalloproteinases in early diabetic retinopathy and
their role in alteration of the blood-retinal barrier. Lab Invest.
85:597–607. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Huang Y, Wang Z, Ye B, Ma JH, Ji S, Sheng
W, Ye S, Ou Y, Peng Y, Yang X, et al: Sodium butyrate ameliorates
diabetic retinopathy in mice via the regulation of gut microbiota
and related short-chain fatty acids. J Transl Med. 21:4512023.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang L, Zhou X, Chen H, You L, Zhang T,
Cheng M, Yao Y, Pan X and Yang X: Mulberry extract ameliorates
T2DM-related symptoms via AMPK pathway in STZ-HFD-induced C57BL/6J
mice. J Ethnopharmacol. 313:1164752023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wu L, Li J, Zhao F and Xiang Y: MiR-340-5p
inhibits Müller cell activation and pro-inflammatory cytokine
production by targeting BMP4 in experimental diabetic retinopathy.
Cytokine. 149:1557452021. View Article : Google Scholar
|
|
32
|
Ai X, Yu P, Luo L, Sun J, Tao H, Wang X
and Meng X: Berberis Dictyophylla F. inhibits angiogenesis and
apoptosis of diabetic retinopathy via suppressing
HIF-1α/VEGF/DLL-4/Notch-1 pathway. J Ethnopharmacol.
296:1154532022. View Article : Google Scholar
|
|
33
|
Kyrylkova K, Kyryachenko S, Leid M and
Kioussi C: Detection of apoptosis by TUNEL assay. Methods Mol Biol.
887:41–47. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hu L, Lv X, Li D, Zhang W, Ran G, Li Q and
Hu J: The anti-angiogenesis role of FBXW7 in diabetic retinopathy
by facilitating the ubiquitination degradation of c-Myc to
orchestrate the HDAC2. J Cell Mol Med. 25:2190–2202. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shao J, Bai Z, Zhang L and Zhang F:
Ferrostatin-1 alleviates tissue and cell damage in diabetic
retinopathy by improving the antioxidant capacity of the Xc-GPX4
system. Cell Death Discov. 8:4262022. View Article : Google Scholar
|
|
36
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
37
|
Simó R, Simó-Servat O, Bogdanov P and
Hernández C: Diabetic retinopathy: Role of neurodegeneration and
therapeutic perspectives. Asia Pac J Ophthalmol (Phila).
11:160–167. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kaštelan S, Orešković I, Bišćan F,
Kaštelan H and Gverović Antunica A: Inflammatory and angiogenic
biomarkers in diabetic retinopathy. Biochem Med (Zagreb).
30:0305022020. View Article : Google Scholar
|
|
39
|
Gardner TW: Histamine, ZO-1 and increased
blood-retinal barrier permeability in diabetic retinopathy. Trans
Am Ophthalmol Soc. 93:583–621. 1995.PubMed/NCBI
|
|
40
|
Blum A, Pastukh N, Socea D and Jabaly H:
Levels of adhesion molecules in peripheral blood correlat with
stages of diabetic retinopathy and may serve as bio markers for
microvascular complications. Cytokine. 106:76–79. 2018. View Article : Google Scholar
|
|
41
|
Uludag G, Hassan M, Matsumiya W, Pham BH,
Chea S, Trong Tuong Than N, Doan HL, Akhavanrezayat A, Halim MS, Do
DV and Nguyen QD: Efficacy and safety of intravitreal anti-VEGF
therapy in diabetic retinopathy: What we have learned and what
should we learn further? Expert Opin Biol Ther. 22:1275–1291. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Crewther SG, Murphy MJ and Crewther DP:
Potassium channel and NKCC cotransporter involvement in ocular
refractive control mechanisms. PLoS One. 3:e28392008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Russell JM: Sodium-potassium-chloride
cotransport. Physiol Rev. 80:211–276. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Betts-Obregon BS, Vellanki S, Buikema J,
Tsin AT and Wright K: Effect of glucose on retinal endothelial cell
viability and VEGF secretion. HSOA J Cell Biol Cell Metabol.
3:0082016.PubMed/NCBI
|
|
45
|
Uemura A, Fruttiger M, D'amore PA, De
Falco S, Joussen AM, Sennlaub F, Brunck LR, Johnson KT, Lambrou GN,
Rittenhouse KD and Langmann T: VEGFR1 signaling in retinal
angiogenesis and microinflammation. Prog Retin Eye Res.
84:1009542021. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Tang L, Xu GT and Zhang JF: Inflammation
in diabetic retinopathy: Possible roles in pathogenesis and
potential implications for therapy. Neural Regen Res. 18:976–982.
2023. View Article : Google Scholar :
|
|
47
|
Panda SP, Reddy PH, Gorla US and Prasanth
D: Neuroinflammation and neovascularization in diabetic eye
diseases (DEDs): Identification of potential pharmacotherapeutic
targets. Mol Biol Rep. 50:1857–1869. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Guzel S, Cai CL, Aranda JV and Beharry KD:
Dose response of bumetanide on aquaporins and angiogenesis
biomarkers in human retinal endothelial cells exposed to
intermittent hypoxia. Pharmaceuticals (Basel). 14:9672021.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Yang S, Zhang J and Chen L: The cells
involved in the pathological process of diabetic retinopathy.
Biomed Pharmacother. 132:1108182020. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Rajendran S, Seetharaman S, Dharmarajan A
and Kuppan K: Microvascular cells: A special focus on heterogeneity
of pericytes in diabetes associated complications. Int J Biochem
Cell Biol. 134:1059712021. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Abu El-Asrar AM, De Hertogh G, Van Den
Eynde K, Alam K, Van Raemdonck K, Opdenakker G, Van Damme J, Geboes
K and Struyf S: Myofibroblasts in proliferative diabetic
retinopathy can originate from infiltrating fibrocytes and through
endothelial-to-mesenchymal transition (EndoMT). Exp Eye Res.
132:179–189. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Tuleta I and Frangogiannis NG: Diabetic
fibrosis. Biochim Biophys Acta Mol Basis Dis. 1867:1660442020.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Yao X, Zhao Z, Zhang W, Liu R, Ni T, Cui
B, Lei Y, Du J, Ai D, Jiang H, et al: Specialized retinal
endothelial cells modulate Blood-Retina barrier in diabetic
retinopathy. Diabetes. 73:225–236. 2024. View Article : Google Scholar
|
|
54
|
Yamato M, Kato N, Yamada KI and Inoguchi
T: The early pathogenesis of diabetic retinopathy and its
attenuation by sodium-glucose transporter 2 inhibitors. Diabetes.
73:1153–1166. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
O'Leary F and Campbell M: The blood-retina
barrier in health and disease. FEBS J. 290:878–891. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Maurissen TL, Spielmann AJ, Schellenberg
G, Bickle M, Vieira JR, Lai SY, Pavlou G, Fauser S, Westenskow PD,
Kamm RD and Ragelle H: Modeling early pathophysiological phenotypes
of diabetic retinopathy in a human inner blood-retinal
barrier-on-a-chip. Nat Commun. 15:13722024. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Wang N, Yao F, Xu W, Feng T, Li Z, Zhang
Q, Wang W, Zhang X, Lei W, Zheng G, et al: The transcription factor
Islet-1 regulates Diabetes-induced inner blood-retinal barrier
disruption. Invest Ophthalmol Vis Sci. 66:82025. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Kowluru RA, Zhong Q and Santos JM: Matrix
metalloproteinases in diabetic retinopathy: Potential role of
MMP-9. Expert Opin Investig Drugs. 21:797–805. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Qian HY, Wei XH and Huang JO: Inflammatory
mechanisms in diabetic retinopathy: Pathogenic roles and
therapeutic perspectives. Am J Transl Res. 17:6262–6274. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Joys S and Siddiqui K: Molecular and
pathophysiological mechanisms of diabetic retinopathy in relation
to adhesion molecules. Curr Diabetes Rev. 15:363–371. 2019.
View Article : Google Scholar
|
|
61
|
Yue T, Shi Y, Luo S, Weng J, Wu Y and
Zheng X: The role of inflammation in immune system of diabetic
retinopathy: Molecular mechanisms, pathogenetic role and
therapeutic implications. Front Immunol. 13:10550872022. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Xu Y, Hou H and Zhao L: The role of VCAM-1
in diabetic retinopathy: A systematic review and meta-analysis. J
Diabetes Complications. 37:1083802023. View Article : Google Scholar
|
|
63
|
Jerome JR, Deliyanti D, Suphapimol V,
Kolkhof P and Wilkinson-Berka JL: Finerenone, a Non-steroidal
mineralocorticoid receptor antagonist, reduces vascular injury and
increases regulatory T-Cells: Studies in rodents with diabetic and
neovascular retinopathy. Int J Mol Sci. 24:23342023. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Borgström P, Hughes GK, Hansell P,
Wolitsky BA and Sriramarao P: Leukocyte adhesion in angiogenic
blood vessels. Role of E-selectin, P-selectin, and beta2 integrin
in lymphotoxin-mediated leukocyte recruitment in tumor
microvessels. J Clin Invest. 99:2246–2253. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Kasza M, Meleg J, Vardai J, Nagy B, Szalai
E, Damjanovich J, Csutak A, Ujhelyi B and Nagy V: Plasma E-selectin
levels can play a role in the development of diabetic retinopathy.
Graefes Arch Clin Exp Ophthalmol. 255:25–30. 2017. View Article : Google Scholar
|
|
66
|
Penman A, Hoadley S, Wilson JG, Taylor HA,
Chen CJ and Sobrin L: P-selectin plasma levels and genetic variant
associated with diabetic retinopathy in african Americans. Am J
Ophthalmol. 159:1152–1160.e2. 2015. View Article : Google Scholar : PubMed/NCBI
|