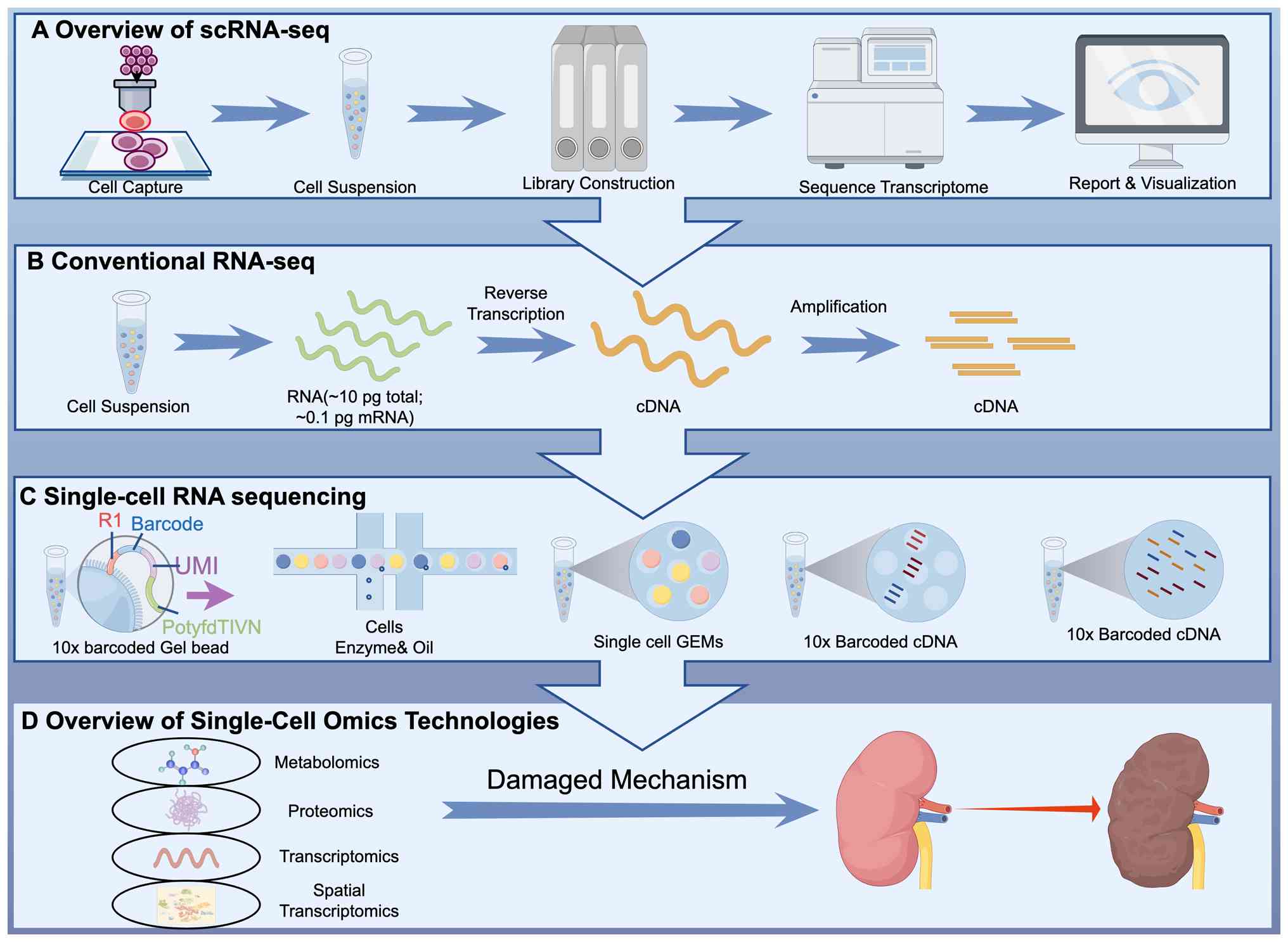
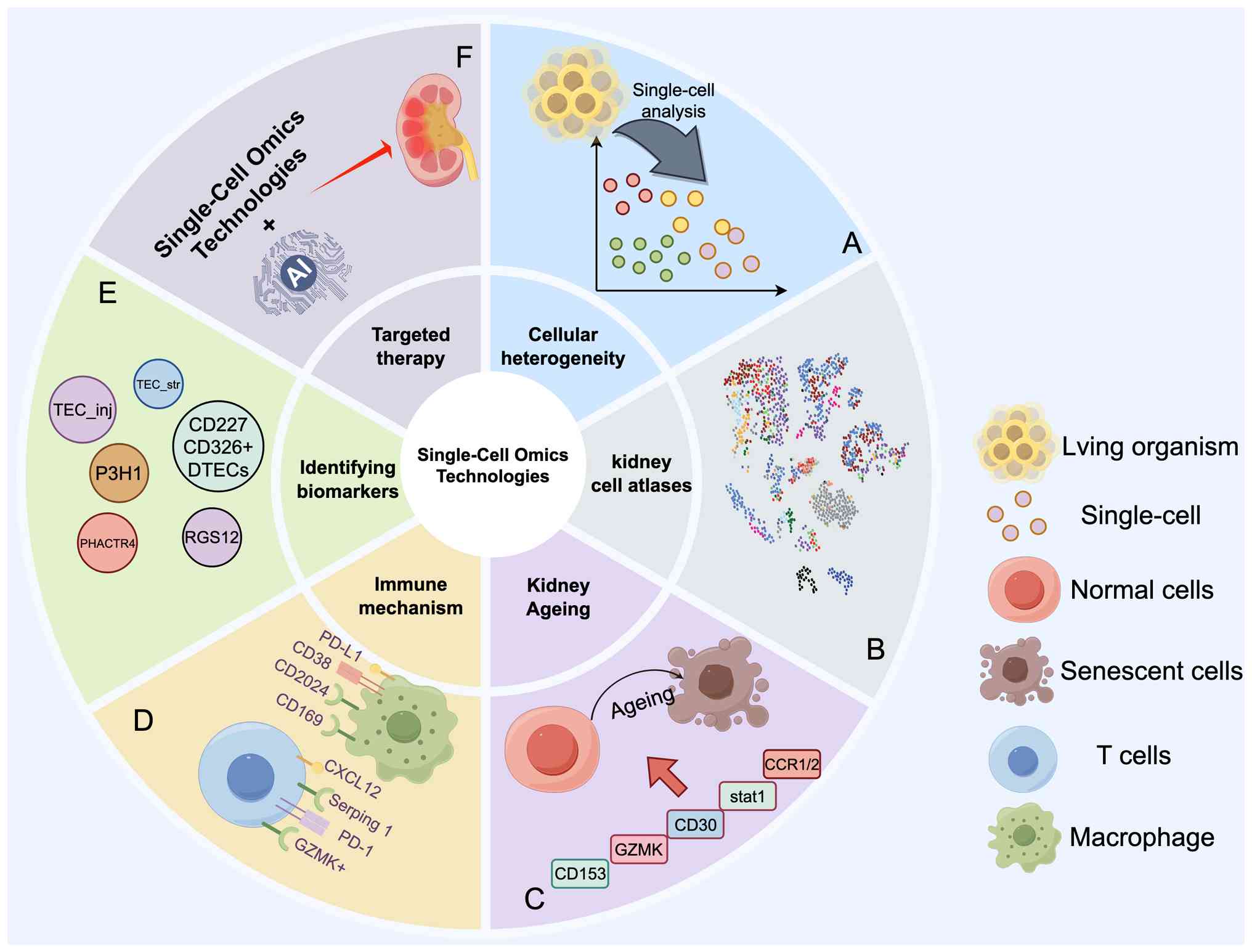
|
1
|
Francis A, Harhay MN, Ong ACM,
Tummalapalli SL, Ortiz A, Fogo AB, Fliser D, Roy-Chaudhury P,
Fontana M, Nangaku M, et al: Chronic kidney disease and the global
public health agenda: an international consensus. Nat Rev Nephrol.
20:473–485. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhang Y, Shi M, Li M, Qin S, Miao D and
Bai Y: Dynamic single-cell metabolomics reveals cell-cell
interaction between tumor cells and macrophages. Nat Commun.
16:45822025. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sharma K, Zhang G, Hansen J, Bjornstad P,
Lee HJ, Menon R, Hejazi L, Liu JJ, Franzone A, Looker HC, et al:
Endogenous adenine mediates kidney injury in diabetic models and
predicts diabetic kidney disease in patients. J Clin Invest.
133:e1703412023. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Alaterre E, Ovejero S, Bret C, Dutrieux L,
Sika D, Fernandez Perez R, Espéli M, Fest T, Cogné M, Martin-Subero
JI, et al: Integrative single-cell chromatin and transcriptome
analysis of human plasma cell differentiation. Blood. 144:496–509.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Potter SS: Single-cell RNA sequencing for
the study of development, physiology and disease. Nat Rev Nephrol.
14:479–492. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cheng C, Chen W, Jin H and Chen X: A
review of single-cell RNA-Seq annotation, integration, and
cell-cell communication. Cells. 12:19702023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lee J, Hyeon DY and Hwang D: Single-cell
multiomics: Technologies and data analysis methods. Exp Mol Med.
52:1428–1442. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Salcher S, Heidegger I, Untergasser G,
Fotakis G, Scheiber A, Martowicz A, Noureen A, Krogsdam A, Schatz
C, Schäfer G, et al: Comparative analysis of 10X Chromium vs BD
Rhapsody whole transcriptome single-cell sequencing technologies in
complex human tissues. Heliyon. 10:e283582024. View Article : Google Scholar
|
|
9
|
Zheng GXY, Terry JM, Belgrader P, Ryvkin
P, Bent ZW, Wilson R, Ziraldo SB, Wheeler TD, McDermott GP, Zhu J,
et al: Massively parallel digital transcriptional profiling of
single cells. Nat Commun. 8:140492017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim YJ and Takahashi K: Emerging
technologies of single-cell multi-omics. Haematologica.
110:1269–1277. 2025.PubMed/NCBI
|
|
11
|
Wang X, He Y, Zhang Q, Ren X and Zhang Z:
Direct comparative analyses of 10X genomics chromium and
smart-seq2. Genomics Proteomics Bioinformatics. 19:253–266. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mahat DB, Tippens ND, Martin-Rufino JD,
Waterton SK, Fu J, Blatt SE and Sharp PA: Single-cell nascent RNA
sequencing unveils coordinated global transcription. Nature.
631:216–223. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lundberg E and Borner GHH: Spatial
proteomics: A powerful discovery tool for cell biology. Nat Rev Mol
Cell Biol. 20:285–302. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Demichev V, Szyrwiel L, Yu F, Teo GC,
Rosenberger G, Niewienda A, Ludwig D, Decker J, Kaspar-Schoenefeld
S, Lilley KS, et al: dia-PASEF data analysis using FragPipe and
DIA-NN for deep proteomics of low sample amounts. Nat Commun.
13:39442022. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Budnik B, Levy E, Harmange G and Slavov N:
SCoPE-MS: Mass spectrometry of single mammalian cells quantifies
proteome heterogeneity during cell differentiation. Genome Biol.
19:1612018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ye Z, Sabatier P, Van Der Hoeven L,
Lechner MY, Phlairaharn T, Guzman UH, Liu Z, Huang H, Huang M, Li
X, et al: Enhanced sensitivity and scalability with a Chip-Tip
workflow enables deep single-cell proteomics. Nat Methods.
22:499–509. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Vander Heiden MG and DeBerardinis RJ:
Understanding the intersections between metabolism and cancer
biology. Cell. 168:657–669. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Petrova B and Guler AT: Recent
developments in single-cell metabolomics by mass spectrometry-a
perspective. J Proteome Res. 24:1493–1518. 2025. View Article : Google Scholar :
|
|
19
|
Tasdogan A, Faubert B, Ramesh V,
Ubellacker JM, Shen B, Solmonson A, Murphy MM, Gu Z, Gu W, Martin
M, et al: Metabolic heterogeneity confers differences in melanoma
metastatic potential. Nature. 577:115–120. 2020. View Article : Google Scholar :
|
|
20
|
Santos AA, Delgado TC, Marques V,
Ramirez-Moncayo C, Alonso C, Vidal-Puig A, Hall Z, Martínez-Chantar
ML and Rodrigues CMP: Spatial metabolomics and its application in
the liver. Hepatology. 79:1158–1179. 2024. View Article : Google Scholar :
|
|
21
|
Zenobi R: Single-cell metabolomics:
Analytical and biological perspectives. Science. 342:12432592013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhu X, Xu T, Peng C and Wu S: Advances in
MALDI mass spectrometry imaging single cell and tissues. Front
Chem. 9:7824322022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang G, Heijs B, Kostidis S, Rietjens RGJ,
Koning M, Yuan L, Tiemeier GL, Mahfouz A, Dumas SJ, Giera M, et al:
Spatial dynamic metabolomics identifies metabolic cell fate
trajectories in human kidney differentiation. Cell Stem Cell.
29:1580–1593.e7. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Qin S, Zhang Y, Shi M, Miao D, Lu J, Wen L
and Bai Y: In-depth organic mass cytometry reveals differential
contents of 3-hydroxybutanoic acid at the single-cell level. Nat
Commun. 15:43872024. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ståhl PL, Salmén F, Vickovic S, Lundmark
A, Navarro JF, Magnusson J, Giacomello S, Asp M, Westholm JO, Huss
M, et al: Visualization and analysis of gene expression in tissue
sections by spatial transcriptomics. Science. 353:78–82. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rodriques SG, Stickels RR, Goeva A, Martin
CA, Murray E, Vanderburg CR, Welch J, Chen LM, Chen F and Macosko
EZ: Slide-seq: A scalable technology for measuring genome-wide
expression at high spatial resolution. Science. 363:1463–1467.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Vickovic S, Eraslan G, Salmén F,
Klughammer J, Stenbeck L, Schapiro D, Äijö T, Bonneau R,
Bergenstråhle L, Navarro JF, et al: High-definition spatial
transcriptomics for in situ tissue profiling. Nat Methods.
16:987–990. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu L, Chen A, Li Y, Mulder J, Heyn H and
Xu X: Spatiotemporal omics for biology and medicine. Cell.
187:4488–4519. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sampath Kumar A, Tian L, Bolondi A,
Hernández AA, Stickels R, Kretzmer H, Murray E, Wittler L, Walther
M, Barakat G, et al: Spatiotemporal transcriptomic maps of whole
mouse embryos at the onset of organogenesis. Nat Genet.
55:1176–1185. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Moffitt JR, Lundberg E and Heyn H: The
emerging landscape of spatial profiling technologies. Nat Rev
Genet. 23:741–759. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rao A, Barkley D, França GS and Yanai I:
Exploring tissue architecture using spatial transcriptomics.
Nature. 596:211–220. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hsieh WC, Budiarto BR, Wang YF, Lin CY,
Gwo MC, So DK, Tzeng YS and Chen SY: Spatial multi-omics analyses
of the tumor immune microenvironment. J Biomed Sci. 29:962022.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu Y, Yang M, Deng Y, Su G, Enninful A,
Guo CC, Tebaldi T, Zhang D, Kim D, Bai Z, et al:
High-spatial-resolution multi-omics sequencing via deterministic
barcoding in tissue. Cell. 183:1665–1681.e18. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wu Y, Cheng Y, Wang X, Fan J and Gao Q:
Spatial omics: Navigating to the golden era of cancer research.
Clin Transl Med. 12:e6962022. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Park J, Shrestha R, Qiu C, Kondo A, Huang
S, Werth M, Li M, Barasch J and Suszták K: Single-cell
transcriptomics of the mouse kidney reveals potential cellular
targets of kidney disease. Science. 360:758–763. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Abedini A, Levinsohn J, Klötzer KA,
Dumoulin B, Ma Z, Frederick J, Dhillon P, Balzer MS, Shrestha R,
Liu H, et al: Single-cell multi-omic and spatial profiling of human
kidneys implicates the fibrotic microenvironment in kidney disease
progression. Nat Genet. 56:1712–1724. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Li H, Li D, Ledru N, Xuanyuan Q, Wu H,
Asthana A, Byers LN, Tullius SG, Orlando G, Waikar SS and Humphreys
BD: Transcriptomic, epigenomic, and spatial metabolomic cell
profiling redefines regional human kidney anatomy. Cell Metab.
36:1105–1125.e10. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
López-Otín C, Blasco MA, Partridge L,
Serrano M and Kroemer G: The hallmarks of aging. Cell.
153:1194–1217. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Fang Y, Gong AY, Haller ST, Dworkin LD,
Liu Z and Gong R: The ageing kidney: Molecular mechanisms and
clinical implications. Ageing Res Rev. 63:1011512020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Antonioli L, Fornai M, Pellegrini C, Masi
S, Puxeddu I and Blandizzi C: Ectopic lymphoid organs and
immune-mediated diseases: Molecular basis for pharmacological
approaches. Trends Mol Med. 26:1021–1033. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Sato Y, Oguchi A, Fukushima Y, Masuda K,
Toriu N, Taniguchi K, Yoshikawa T, Cui X, Kondo M, Hosoi T, et al:
CD153/CD30 signaling promotes age-dependent tertiary lymphoid
tissue expansion and kidney injury. J Clin Invest. 132:e1460712022.
View Article : Google Scholar :
|
|
42
|
Mogilenko DA, Shpynov O, Andhey PS, Arthur
L, Swain A, Esaulova E, Brioschi S, Shchukina I, Kerndl M,
Bambouskova M, et al: Comprehensive profiling of an aging immune
system reveals clonal GZMK+ CD8+ T cells as
conserved hallmark of inflammaging. Immunity. 54:99–115.e12. 2021.
View Article : Google Scholar
|
|
43
|
Yu X, Li S, Zhong J, Ji X, Xu H, Chen Q
and Li Q: Single-cell transcriptome sequencing partially revealed
the changes of T cells in the early stage of aging kidney. Mol
Immunol. 173:61–70. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Rex N, Melk A and Schmitt R: Cellular
senescence and kidney aging. Clin Sci (Lond). 137:1805–1821. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wu L, Lin H, Li S, Huang Y, Sun Y, Shu Y,
Luo T, Liang T, Lai W, Rao J, et al: Macrophage iron dyshomeostasis
promotes aging-related renal fibrosis. Aging Cell. 23:e142752024.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Medina-Ruiz L, Bartolini R, Mathie H,
Halawa HA, Cunningham M and Graham GJ: CCR1 and CCR2 coexpression
on monocytes is nonredundant and delineates a distinct monocyte
subpopulation. J Immunol. 213:214–225. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Kimmel JC, Penland L, Rubinstein ND,
Hendrickson DG, Kelley DR and Rosenthal AZ: Murine single-cell
RNA-seq reveals cell-identity- and tissue-specific trajectories of
aging. Genome Res. 29:2088–2103. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Zhu T, Shen Q, Shen L, Wang Y, Zhu B, Ma
L, Feng S, Wang C, Yan S, Li J, et al: Senescence-induced
p21high macrophages contributed to CD8+ T
cells-related immune hyporesponsiveness in kidney transplantation
via Zfp36/IL-27 axis. Cell Discov. 11:382025. View Article : Google Scholar
|
|
49
|
Mehta RL, Cerdá J, Burdmann EA, Tonelli M,
García-García G, Jha V, Susantitaphong P, Rocco M, Vanholder R,
Sever MS, et al: International society of nephrology's 0by25
initiative for acute kidney injury (zero preventable deaths by
2025): A human rights case for nephrology. Lancet. 385:2616–2643.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Liu J, Zeng D, Wang Y, Deng F, Wu S and
Deng Z: Identification of druggable targets in acute kidney injury
by proteome- and transcriptome-wide Mendelian randomization and
bioinformatics analysis. Biol Direct. 20:382025. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Klocke J, Kim SJ, Skopnik CM, Hinze C,
Boltengagen A, Metzke D, Grothgar E, Prskalo L, Wagner L, Freund P,
et al: Urinary single-cell sequencing captures kidney injury and
repair processes in human acute kidney injury. Kidney Int.
102:1359–1370. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hasegawa S: Urinary single-cell
transcriptomics: A promising noninvasive method for assessing acute
kidney injury. Kidney Int. 102:1219–1221. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Wagner L, Kujat J, Langhans V, Prskalo L,
Metzke D, Grothgar E, Freund P, Goerlich N, Brand H, Timm S, et al:
Flow-cytometric quantification of urine kidney epithelial cells
specifically reflects tubular damage in acute kidney diseases.
Kidney Int Rep. 10:1260–1273. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Li J, Shi X, Xu J, Wang K, Hou F, Luan X
and Chen L: Aldehyde dehydrogenase 2 lactylation aggravates
mitochondrial dysfunction by disrupting PHB2 mediated mitophagy in
acute kidney injury. Adv Sci (Weinh). 12:e24119432025. View Article : Google Scholar :
|
|
55
|
Polonsky M, Gerhardt LMS, Yun J, Koppitch
K, Colón KL, Amrhein H, Wold B, Zheng S, Yuan GC, Thomson M, et al:
Spatial transcriptomics defines injury specific microenvironments
and cellular interactions in kidney regeneration and disease. Nat
Commun. 15:70102024. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Cui N, Liu C, Tang X, Song L, Xiao Z, Wang
C, Wu Y, Zhou Y, Peng C, Liu Y, et al: ISG15 accelerates acute
kidney injury and the subsequent AKI-to-CKD transition by promoting
TGFβR1 ISGylation. Theranostics. 14:4536–4553. 2024. View Article : Google Scholar
|
|
57
|
Zhang YL, Tang TT, Wang B, Wen Y, Feng Y,
Yin Q, Jiang W, Zhang Y, Li ZL, Wu M, et al: Identification of a
novel ECM remodeling macrophage subset in AKI to CKD transition by
integrative spatial and single-cell analysis. Adv Sci (Weinh).
11:e23097522024. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Cai X, Li X, Shi J, Tang L, Yang J, Yu R,
Wang Z and Wang D: S100A8/A9 high-expression macrophages mediate
renal tubular epithelial cell damage in acute kidney injury
following acute type A aortic dissection surgery. Front Mol Biosci.
12:15307412025. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Yao W, Chen Y, Li Z, Ji J, You A, Jin S,
Ma Y, Zhao Y, Wang J, Qu L, et al: Single cell RNA sequencing
identifies a unique inflammatory macrophage subset as a druggable
target for alleviating acute kidney injury. Adv Sci (Weinh).
9:e21036752022. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Kato M and Natarajan R: Epigenetics and
epigenomics in diabetic kidney disease and metabolic memory. Nat
Rev Nephrol. 15:327–345. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Ogurtsova K, Da Rocha Fernandes JD, Huang
Y, Linnenkamp U, Guariguata L, Cho NH, Cavan D, Shaw JE and
Makaroff LE: IDF diabetes atlas: Global estimates for the
prevalence of diabetes for 2015 and 2040. Diabetes Res Clin Pract.
128:40–50. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Hu X, Chen S, Ye S, Chen W and Zhou Y: New
insights into the role of immunity and inflammation in diabetic
kidney disease in the omics era. Front Immunol. 15:13428372024.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Fan X, Yang M, Lang Y, Lu S, Kong Z, Gao
Y, Shen N, Zhang D and Lv Z: Mitochondrial metabolic reprogramming
in diabetic kidney disease. Cell Death Dis. 15:4422024. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Qi W, Keenan HA, Li Q, Ishikado A, Kannt
A, Sadowski T, Yorek MA, Wu IH, Lockhart S, Coppey LJ, et al:
Pyruvate kinase M2 activation may protect against the progression
of diabetic glomerular pathology and mitochondrial dysfunction. Nat
Med. 23:753–762. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Jiang L, Yu H, Jian J, Sai X, Wang Y,
Zhang Y and Wu X: Landscape analysis of m6A modification regulators
reveals LRPPRC as a key modulator in tubule cells for DKD: A
multi-omics study. Front Pharmacol. 16:15068962025. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Hou G, Dong Y, Jiang Y, Zhao W, Zhou L,
Cao S and Li W: Immune inflammation and metabolic interactions in
the pathogenesis of diabetic nephropathy. Front Endocrinol
(Lausanne). 16:16025942025. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Fu J, Sun Z, Wang X, Zhang T, Yuan W,
Salem F, Yu SMW, Zhang W, Lee K and He JC: The single-cell
landscape of kidney immune cells reveals transcriptional
heterogeneity in early diabetic kidney disease. Kidney Int.
102:1291–1304. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Wang Y, Feng X, Li Y, Niu S, Li J, Shi H,
Wang G and Wang L: Targeting inflammation and necroptosis in
diabetic kidney disease: A novel approach via PPARα modulation. Int
Immunopharmacol. 154:1145622025. View Article : Google Scholar
|
|
69
|
Ji C, Zhang J, Shi H, Chen B, Xu W, Jin J
and Qian H: Single-cell RNA transcriptomic reveal the mechanism of
MSC derived small extracellular vesicles against DKD fibrosis. J
Nanobiotechnology. 22:3392024. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Chen J, Zhang Q, Guo J, Gu D, Liu J, Luo
P, Bai Y, Chen J, Zhang X, Nie S, et al: Single-cell
transcriptomics reveals the ameliorative effect of rosmarinic acid
on diabetic nephropathy-induced kidney injury by modulating
oxidative stress and inflammation. Acta Pharm Sin B. 14:1661–1676.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Park PG, Hwang J, Kim Y, Hong M, Yun D,
Yoon H, Kang C, Bae S, Kwak SH, Kim YC, et al: Inflammatory milieu
by crosstalk between glomerulus and proximal tubular cells in type
2 diabetes mellitus kidney disease. Diabetes Metab J. 49:1024–1039.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Floege J, Rauen T and Tang SCW: Current
treatment of IgA nephropathy. Semin Immunopathol. 43:717–728. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Stamellou E, Seikrit C, Tang SCW, Boor P,
Tesař V, Floege J, Barratt J and Kramann R: IgA nephropathy. Nat
Rev Dis Primers. 9:672023. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zheng Y, Lu P, Deng Y, Wen L, Wang Y, Ma
X, Wang Z, Wu L, Hong Q, Duan S, et al: Single-cell transcriptomics
reveal immune mechanisms of the onset and progression of IgA
nephropathy. Cell Rep. 33:1085252020. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Zhang J, Fang Q, Huang Y, Qu Y, Liu Q, Li
R, Zhou Y, Cui S, Liu R, Wang X, et al: CX3CR1+
monocytes/macrophages promote regional immune injury in mesangial
proliferative glomerulonephritis through crosstalk with activated
mesangial cells. Research (Wash DC). 8:07162025.
|
|
76
|
Zambrano S, He L, Kano T, Sun Y, Charrin
E, Lal M, Betsholtz C, Suzuki Y and Patrakka J: Molecular insights
into the early stage of glomerular injury in IgA nephropathy using
single-cell RNA sequencing. Kidney Int. 101:752–765. 2022.
View Article : Google Scholar
|
|
77
|
Hasegawa K, Kawashima N, Kawabata A,
Sakakura M, Onoda N, Sano T, Urakawa I, Matsubara M and Naito S:
Inflammation-associated molecules in the glomerular-endothelium in
mild IgA-nephropathy patients identified by single-cell and spatial
transcriptome. Commun Biol. 8:9152025. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Chen L, Chen X, Cai G, Jiang H, Chen X and
Zhang M: An inflammatory cytokine signature predicts IgA
nephropathy severity and progression. MedComm (2020). 5:e7832024.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Anders HJ, Saxena R, Zhao M, Parodis I,
Salmon JE and Mohan C: Lupus nephritis. Nat Rev Dis Primers.
6:72020. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Mok CC: Towards new avenues in the
management of lupus glomerulonephritis. Nat Rev Rheumatol.
12:221–234. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Mok CC, Teng YKO, Saxena R and Tanaka Y:
Treatment of lupus nephritis: Consensus, evidence and perspectives.
Nat Rev Rheumatol. 19:227–238. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Parikh SV, Almaani S, Brodsky S and Rovin
BH: Update on lupus nephritis: Core curriculum 2020. Am J Kidney
Dis. 76:265–281. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Tanaka Y, O'Neill S, Li M, Tsai I and Yang
Y: Systemic lupus erythematosus: Targeted literature review of the
epidemiology, current treatment, and disease burden in the asia
pacific region. Arthritis Care Res (Hoboken). 74:187–198. 2022.
View Article : Google Scholar
|
|
84
|
Li Y, Tang C, Vanarsa K, Thai N, Castillo
J, Lea GAB, Lee KH, Kim S, Pedroza C, Wu T, et al: Proximity
extension assay proteomics and renal single cell transcriptomics
uncover novel urinary biomarkers for active lupus nephritis. J
Autoimmun. 143:1031652024. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Su C, Cong A, Wu H, Zhou Z, Hu Z, Luo J,
Cui S, Xu D, Zhou Z, Huang Z, et al: Glomerular CD68+
macrophages infiltration at initial biopsy predicts response to
standard immunosuppression in proliferative lupus nephritis:
CD68+ Mø predicts LN treatment response. J Autoimmun.
152:1033922025. View Article : Google Scholar
|
|
86
|
Tang C, Fang M, Tan G, Zhang S, Yang B, Li
Y, Zhang T, Saxena R, Mohan C and Wu T: Discovery of novel
circulating immune complexes in lupus nephritis using
immunoproteomics. Front Immunol. 13:8500152022. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Zhang Y, Gan L, Tang J, Liu D, Chen G and
Xu B: Metabolic profiling reveals new serum signatures to
discriminate lupus nephritis from systemic lupus erythematosus.
Front Immunol. 13:9673712022. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Danaher P, Hasle N, Nguyen ED, Roberts JE,
Rosenwasser N, Rickert C, Hsieh EWY, Hayward K, Okamura DM, Alpers
CE, et al: Childhood-onset lupus nephritis is characterized by
complex interactions between kidney stroma and infiltrating immune
cells. Sci Transl Med. 16:eadl16662024. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Wei S, Shen H, Zhang Y, Liu C, Li S, Yao
J, Jin Z and Yu H: Integrative analysis of single-cell and bulk
transcriptome data reveal the significant role of macrophages in
lupus nephritis. Arthritis Res Ther. 26:842024. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Richoz N, Tuong ZK, Loudon KW,
Patiño-Martínez E, Ferdinand JR, Portet A, Bashant KR, Thevenon E,
Rucci F, Hoyler T, et al: Distinct pathogenic roles for resident
and monocyte-derived macrophages in lupus nephritis. JCI Insight.
7:e1597512022. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Chen W, Jin B, Cheng C, Peng H, Zhang X,
Tan W, Tang R, Lian X, Diao H, Luo N, et al: Single-cell profiling
reveals kidney CD163+ dendritic cell participation in
human lupus nephritis. Ann Rheum Dis. 83:608–623. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Gartshteyn Y, Geraldino-Pardilla L,
Khalili L, Bukhari S, Lerrer S, Winchester RJ, Askanase AD and Mor
A: SAP-expressing T peripheral helper cells identify systemic lupus
erythematosus patients with lupus nephritis. Front Immunol.
15:13274372024. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Wu C, Jiang S, Chen Z, Li T, Gu X, Dai M,
Du F, Ye Y, Tang L, Wang M, et al: Single-cell transcriptomics
reveal potent extrafollicular B cell response linked with granzyme
K+ CD8 T cell activation in lupus kidney. Ann Rheum Dis.
84:451–466. 2025. View Article : Google Scholar
|
|
94
|
Yamaguchi J, Isnard P, Robil N, de la
Grange P, Hoguin C, Schmitt A, Hummel A, Megret J, Goudin N, Luka
M, et al: PIK3CA inhibition in models of proliferative
glomerulonephritis and lupus nephritis. J Clin Invest.
134:e1764022024. View Article : Google Scholar : PubMed/NCBI
|