Introduction
Stroke remains the second primary cause of global
death (~7.3 million/year), with ~85% of stroke cases being ischemic
strokes (IS) (1-3). IS is a clinical disease due to a
cerebral artery or its branch occlusion causing impaired regional
cerebral blood supply, leading to localized cerebral tissue
ischemia and hypoxic necrosis, which results in neurological
deficits (4,5). Cerebral ischemia-reperfusion injury
(CIRI) poses a notable challenge in IS management and severely
impacts patient outcomes (6).
When cerebral blood vessels become blocked, localized brain tissue
experiences ischemia and hypoxia. When blood flow is restored, it
triggers a series of complex pathological mechanisms (7). Neuroinflammation serves as the key
driver of the CIRI pathological process. Under hypoxic-ischemic
conditions, microglia undergo rapid activation, releasing large
quantities of pro-inflammatory cytokines including tumor necrosis
factor-α (TNF-α) and IL-1β, thereby intensifying the inflammatory
cascade (8,9). Pyroptosis serves a pivotal role in
CIRI-mediated neuronal injury. It further damages the neural
microenvironment, exacerbating neuronal death and neurological
dysfunction (10,11). Therefore, inhibiting neuronal
pyroptosis to decrease neuroinflammation may be an effective
approach to enhance the prognosis of CIRI.
Propofol (PPF) is a γ-aminobutyric acid receptor
agonist that produces sedative and hypnotic effects by inhibiting
neuronal excitability. Its structure contains a phenolic hydroxyl
group, thereby conferring antioxidant activity (12,13). In recent years, its research in
neuroprotection has garnered notable attention (12,14). One study indicated that PPF not
only alleviates inflammatory responses in microglia but also
provides neuroprotection by improving mitochondrial respiratory
chain function and glycolytic status (14). Additionally, PPF downregulates
the expression of proinflammatory genes induced by
lipopolysaccharide (LPS), suppresses neuroinflammatory responses
and inhibits excessive activation of microglia (15). In a traumatic brain injury mouse
model, Wang et al (16)
found that PPF downregulates gasdermin (GSDM)D-N and caspase-1
expression, thus inhibiting neuronal pyroptosis. PPF treatment
decreases cerebral infarction volume in CIRI mice, decreases the
extent of cortical tissue damage and improves neurological function
scores (17). Overall, the
neuroprotective effects of PPF may involve multiple pathways,
including its antioxidant activity, decreased inflammatory
responses and inhibition of pyroptosis. However, the specific
molecular mechanisms remain unclear.
As a multifunctional glycoprotein, milk fat
globule-epidermal growth factor 8 (MFG-E8) enhances phagocytic
function and increases the clearance rate of apoptotic cells,
thereby alleviating inflammatory responses (18,19). Changes in MFG-E8 expression in
the nervous system are associated with neuroprotection and injury
repair (20). The NF-κB
p/NOD-like receptor protein 3 (NLRP3) pathway is a key signaling
pathway mediating neuroinflammatory responses in CIRI (21,22). NF-κB regulates expression of
numerous inflammation-related genes, including classic
proinflammatory cytokines (IL-1β, IL-6, TNF-α). NLRP3 inflammasome
activation promotes the maturation and secretion of proinflammatory
cytokines, thus generating a strong inflammatory response (23-25). Excessive activation of the
NF-κB/NLRP3 pathway in rat models of intracerebral hemorrhage leads
to microglia M1 polarization and induces pyroptosis, thus
aggravating neuroinflammation (26). Notably, PPF regulates MFG-E8
expression in microglia (27)
and MFG-E8 can downregulate NF-κB expression in microglia and
inhibit their M1 polarization (28). To the best of our knowledge,
however, whether PPF inhibits the NF-κB/NLRP3 pathway by regulating
MFG-E8 expression, exerting neuroprotective effects in CIRI, has
not been reported. The present study utilized oxygen-glucose
deprivation/reoxygenation (OGD/R) BV2 microglial cell and transient
middle cerebral artery occlusion (tMCAO) mouse models to
investigate whether PPF inhibits pyroptosis mediated by the
NF-κB/NLRP3 pathway by upregulating MFG-E8, thereby improving
neuronal damage in CIRI.
Materials and methods
Construction of OGD/R cell models
Mouse microglial BV-2 (cat. no. SNL-155) and
hippocampal neuronal HT22 (cat. no. SNL-202) cells were obtained
from Wuhan Sunncell Biotechnology. Cells were cultured in BV-2
cell-specific medium (cat. no. SNLM-155) and HT22 cell-specific
medium (SNLM-202; both Wuhan Sunncell Biotechnology, respectively.
The cultures were maintained at 37°C with 5% CO2, and
the medium was refreshed every 2 days.
Following normal culture for 24 h at 37°C, BV-2 and
HT22 cells were placed in a glucose- and serum-free basal medium
(cat. no. PM150270, Wuhan Pricella Biotechnology) and placed in a
95% N2, 5% CO2 hypoxic incubator at 37°C for
2 h. Cells were transferred to normal medium and maintained at 37°C
with 95% oxygen and 5% CO2 for 24 h to construct an
OGD/R cell model (29). In
addition, for the PPF group, PPF (2, 4, 8, 16, 32, 64 and 128
μM, cat. no. HY-B0649, MedChemExpress) was added to the
culture medium during reoxygenation (30). In subsequent experiments, cells
were treated with 8, 16 and 32 μM PPF during reoxygenation
to evaluate its neuroprotective effects. According to the method
described by Beaulieu et al (31), OGD-treated HT22 (in the lower
chamber) and BV2 cells (in the upper chamber) were co-cultured at
37°C in a Transwell chamber (0.4 μm, Corning, Inc.) for 24 h
at a 1:3 inoculation ratio of HT22:BV2 cells (Fig. 1A).
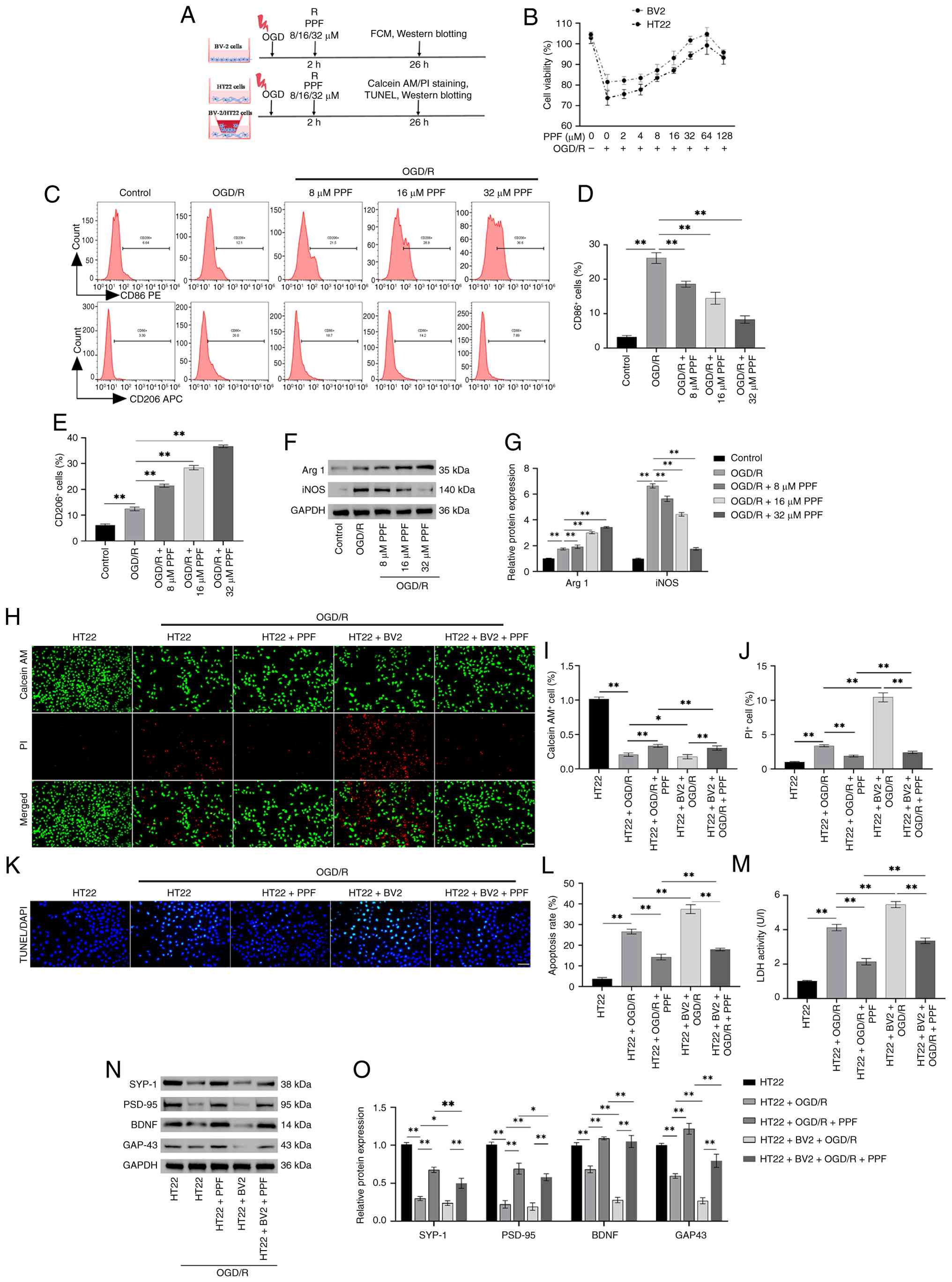 | Figure 1PPF suppresses excessive activation
of BV-2 cells and ameliorates damage in HT22 cells following OGD/R.
(A) Operational flowchart. (B) Cell Counting Kit-8 assay was
performed to determine the viability of BV2 and HT22 cells after 24
h treatment with PPF. (C) Flow cytometry revealed that PPF
decreased the number of CD86-positive and increased the number of
CD206-positive cells. (D) Quantitative analysis of flow cytometry
confirmed that PPF significantly decreased the proportion of
CD86-positive cells and (E) significantly increased the proportion
of CD206-positive cells. (F) Western blotting of iNOS, Arg 1, and
GAPDH (internal reference). (G) Western blotting revealed that PPF
decreased iNOS and increased Arg 1 levels in BV-2 cells. (H)
Calcein AM/PI staining revealed decreased viability in
OGD/R-treated HT22 cells, which was diminished upon co-culture with
BV-2 cells, whereas PPF enhanced cell survival (magnification, ×20;
scale bar, 100 μm). (I) Quantitative analysis of Calcein
AM/PI staining showed that PPF enhanced the survival ability of
HT22 cells in the OGD/R+BV2 co-culture system and (J) decreased the
death rate of HT22 cells in the OGD/R+BV2 co-culture system. (K)
TUNEL staining to assess TUNEL positivity in HT22 cells (green
fluorescence) (magnification, ×40; scale bar, 50 μm). (L)
TUNEL staining indicated increased apoptosis in OGD/R-treated HT22
cells. Co-culture with BV-2 cells elevated apoptosis rates, whereas
PPF reduced apoptosis. (M) Following OGD/R, LDH release increased
in HT22 cells and was further elevated after co-culture with BV-2
cells, while PPF treatment decreased LDH release. (N) Western
blotting protein bands for SYP-1, PSD-95, BDNF, GAP-43, and GAPDH.
(O) Quantitative analysis of western blotting revealed that PPF
induced elevated SYP-1, PSD-95, BDNF and GAP-43 levels in HT22
cells. *P<0.05, **P<0.01. PPF,
Propofol; OGD/R, Oxygen-glucose deprivation/reoxygenation; iNOS,
inducible nitric oxide synthase; Arg, arginase 1; LDH, lactate
dehydrogenase; SYP, synapsin; PSD, postsynaptic density protein;
BDNF, brain-derived neurotrophic factor; GAP, growth-associated
protein; FCM, flow cytometry. |
Cell transfection
MFG-E8 small interfering (si)RNA (forward,
5'-CCAAUGUCUGGUGACUUUTT-3' and reverse,
5'-AAAGUCACCAGACAUUUGGTT-3'), MFG-E8 overexpression (OE) plasmid
and negative control plasmids (si-NC: Forward,
5'-GAUGGAGAAGCUCGCUGATTT-3' and reverse, 5'-AAAUCAGCGAGCUUCUCCATT';
OE-NC) were synthesized by Sangon Biotech Co., Ltd. Plasmids (2
μg) were transfected into BV-2 cells for 48 h at 37°C using
Lipofectamine 3000 (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacturer's instructions. At 48 h
post-transfection, BV-2 cells were fully lysed with RIPA lysis
buffer (cat. no. 20-188, Sigma-Aldrich; Merck KGaA) and MFG-E8
protein expression was analyzed by western blotting to evaluate the
transfection efficiency. For the OGD/R + OE-MFG-E8 + LPS (NF-κB
agonist) group, cells were transfected with OE-MFG-E8, treated with
OGD and exposed to LPS (1 μg/ml; cat. no. HY-D1056,
MedChemExpress) during reoxygenation (32). For the OGD/R + si-MFG-E8 + PPF
group, BV-2 cells were first transfected with si-MFG-E8, then
subjected to OGD and treated with PPF during reoxygenation.
Cell Counting Kit-8 (CCK-8) assay
BV-2 and HT22 cells were inoculated into 96-well
plates (1×104 cells/well, 100 μl). When the cells
were completely adherent (12-16 h), PPF (2, 4, 8, 16, 32, 64 and
128 μM) or LPS (0.125, 0.250, 0.500, 1.000 and 2.000
μg/ml) were added for another 24 h at 37°C. The wells
received 10% CCK-8 reagent (cat. no. HY-K0301, MedChemExpress),
which was then thoroughly mixed and incubated in the dark for 2 h.
Finally, the optical density at 450 nm (OD450) was
assessed by SpectraMax Mini microplate reader (Molecular Devices,
LLC) to calculate cell viability.
Flow cytometry
BV-2 cells were resuspended with PBS to prepare a
single cell suspension with a concentration of 1.0×107
cells/ml. Anti-CD86 (1:100, cat. no. MA5-52361), anti-CD11b (1:100,
cat. no. PA5-79533) and anti-CD206 antibody (1:400, cat. no.
17-2061-82; all Invitrogen; Thermo Fisher Scientific, Inc.) were
added for 30 min under dark conditions at 4°C. Samples were
detected with BD FACSCalibur flow cytometer and the FlowJo software
(v10.8l; both BD Biosciences) for analysis.
Calcein AM/PI staining
The viability of HT22 cells was assessed by Calcein
AM/PI detection kit (cat. no. C2015M, Beyotime Biotechnology). HT22
cells were washed with PBS, mixed with Calcein AM/PI detection
working solution (500 μl) and incubated at 37°C for 30 min
in a dark environment. Following incubation, staining was observed
by fluorescence microscope (cat. no. BZ-X810, Keyence Corporation).
Live (green) and dead (red) cell counts were analyzed using ImageJ
1.54h software (National Institutes of Health).
TUNEL staining
HT22 cells were plated into 12-well plates
(1.0×105 cells/well, built-in sterile coverslip). Cells
were exposed to 4% paraformaldehyde (cat. no. 441244,
Sigma-Aldrich; Merck KGaA) to fix the cells for 30 min at 25°C.
0.3% Triton X-100 solution (cat. no. T824275, Shanghai Macklin
Biochemical Co., Ltd.) was added at 25°C for 10 min to increase
permeability. TUNEL detection solution (cat. no. C1086, Beyotime
Biotechnology) was added for 60 min at 37°C in a dark environment,
then rinsed three times with PBS. The cells were incubated with
DAPI (1 μg/ml, cat. no. D669617, Shanghai Macklin
Biochemical Co., Ltd.) at 25°C for 5 min in the dark to stain cell
nuclei, followed by three additional rinses with PBS. Once sealed
with Anti-fade Mounting Medium (cat. no. HY-K1047, MedChemExpress),
samples were observed using a fluorescence microscope, and 5
non-overlapping random fields of view were captured per sample to
assess the apoptosis rate using the ImageJ 1.54 h software.
Cell morphology observation
BV2 cells were mixed with glutaraldehyde solution
(2.5%, cat. no. G6257, Sigma-Aldrich; Merck KGaA) and fixed in the
dark at 4°C for 4 h. After being washed twice with sterile PBS to
remove glutaraldehyde, the samples were dehydrated with ethanol
solution (30, 50, 70, 80, 90%) for 10 min each, then dehydrated
with anhydrous ethanol. Finally, isoamyl acetate (cat. no. 79857,
Sigma-Aldrich; Merck KGaA) was used to replace anhydrous ethanol
twice, for 10 min each. After centrifugation at 1,000 × g, 4°C for
5 min, the liquid was mixed with the cell pellet, and the mixture
was dropped onto a silicon wafer, followed by drying at 37°C. The
dried sample was sputter-coated with gold for 150 sec and
morphological observations and image capture were performed by
scanning electron microscopy (SEM; cat. no. S-4800, Hitachi
Ltd.).
Yo-Pro-1 and Hoechst 33342 staining
BV2 cells were seeded into a 12-well plate at a
density of 1.0×105 cells/well (built-in sterile
coverslip). Cells were exposed to 4% paraformaldehyde for 30 min at
25°C. Hoechst 33342 (cat. no. C1025) and Yo-Pro-1 staining solution
(500 μl; cat. no. C1356S; both Beyotime Institute of
Biotechnology) were added at 37°C for 30 min. Cells were rinsed
with PBS and photographed under a fluorescence microscope.
Caspase-1 enzyme activity assay
Caspase-1 enzyme activity was detected using the
Caspase-1 Activity Assay kit (cat. no. E-CK-A381, Wuhan Elabscience
Biotechnology) according to the manufacturer's instructions. BV2
cells (~1×106) were collected and washed with PBS. A
total of 100 μl Cell Lysis Buffer was added in an ice bath
for 30 min. The lysed samples were centrifuged at 8,000 × g for 10
min at 4°C and the protein concentration in the supernatant was
assayed by the BCA Protein Quantification kit. A total of 50
μl lysate was added to 45 μl Reaction Buffer and 5
μl Ac-YVAD-pNA were added for 2 h at 37°C. OD405
was measured using a microplate reader and enzyme activity was
calculated.
Dual-luciferase reporter assay
Wild-type (WT) and mutant-type (MUT) dual luciferase
reporter plasmids for the NF-κB 3' untranslated region were
synthesized by Sangon Biotech Co., Ltd. NF-κB-WT and si-MFG-E8,
NF-κB-WT and si-NC, NF-κB-MUT, si-MFG-E8, and NF-κB-MUT and
si-MFG-E8, respectively, were co-transfected into BV2 cells using
Lipofectamine 3000 as aforementioned. Cells were collected after 48
h and assayed for luciferase activity using the Dual-Lucy Assay kit
(cat. no. RG027, Beyotime Biotechnology) as previously described
(33). Firefly luciferase
activity was normalized to Renilla luciferase activity.
Construction of tMCAO mouse model
A total of 63 male C57BL/6 mice (age, 3 months;
weight, 21-24 g) was obtained from Beijing Vital River Laboratory
Animal Technology Co., Ltd. and raised at 22°C, humidity of 45-50%,
12/12-h light/dark cycle and food and water ad libitum. The mice
were randomly divided into Sham (n=9) and experimental group (n=54)
according to the random number table method. According to the
method described by Liu et al (34), the tMCAO model was established.
The mice received anesthesia with 2% isoflurane, followed by 1.5%
isoflurane to maintain anesthesia and the surgical area was
sterilized with 75% ethanol and 10% iodophor. In the experimental
group, a small incision was made 4 mm from the bifurcation of right
common carotid artery (CCA), with a suture placed around the
internal carotid artery (ICA). The 0.16-mm diameter suture (cat.
no. 1618-50, Beijing Cinontech Co., Ltd.) was gently pushed with
ophthalmic tweezers, reaching an insertion depth of ~20 mm. The
suture at the distal end of ICA and CCA was fastened to fix the
suture. The incision was sutured and disinfected and a 6 mm suture
was retained in vitro for reperfusion. After the blood
supply was blocked for 45 min, the suture was removed. Blood flow
in the mouse brain was monitored using Laser Speckle Doppler
Flowmetry (PeriCam PSI Z; Perimed AB) (35). The body temperature of the mice
was maintained at 37.0±0.5°C using a homoeothermic blanket
throughout the surgical process. Sham group mice were sutured and
disinfected, but tMCAO was not performed. To reduce post-operative
pain in mice, buprenorphine (0.1 mg/kg; cat. no. PHR8512;
Sigma-Aldrich; Merck KGaA) was administered subcutaneously 15 min
before and 12 h and once 24 h after surgery.
At 45 min after tMCAO, mice were injected
intraperitoneally with normal saline or 50, 100 or 200 mg/kg PPF
(n=9 mice/group) (36). In
addition, for the tMCAO + 200 mg/kg PPF + si-MFG-E8 group or the
tMCAO + 200 mg/kg PPF + si-NC group, mice were microinjected with
si-MFG-E8 or si-NC (166.67 μM) intracerebrally 2 days prior
to surgery and were injected intraperitoneally 45 min
postoperatively with 200 mg/kg PPF (37). Mice were sacrificed 1 day after
PPF administration. A double-blind method was used during the
animal experiments; none of the researchers responsible for feeding
the mice, behavioral scoring or histological analysis were aware of
the grouping information.
Assessment of neurological deficit
At 24 h after tMCAO, Longa scoring method was used
to determine whether the modeling was successful (38). The Longa score was determined as
follows: 0, normal, no neurological deficit; 1, inability to fully
extend the contralateral forepaw, mild neurological deficit; 2,
turning to the paralyzed side when walking, moderate neurological
deficit; 3, body tilting toward the paralyzed side when walking,
severe neurological deficit; 4, inability to walk spontaneously,
loss of consciousness. A score of 4 represents mice with
irreversible severe cerebral ischemic damage or loss of
consciousness, which prevented subsequent behavioral scoring
(corner test). Therefore, a score of 1-3 points was judged as
successful modeling.
At 24 h after tMCAO, the sensorimotor and postural
asymmetry neurological functions of mice were detected by corner
test (39). The mice were placed
at the corner entrance between two plates connected at a 30° angle.
Once the mouse reaches the corner, it will retreat and turn right
or left. Each mouse was tested 20 times. If the mice did not
retreat when turning around, the experiment was abandoned. The
number of right and left turns was recorded.
Triphenyltetrazolium chloride (TTC)
staining
Following behavioral testing, mice were sacrificed
by intraperitoneal injection of sodium pentobarbital (100 mg/kg)
for 1 min, with cardiac and respiratory arrest used to determine
death. Mouse brain tissue was frozen at −80°C for 20 min and sliced
into 2-mm sections. Subsequently, the tissue sections were exposed
to TTC solution (2%, cat. no. C0652, Beyotime Institute of
Biotechnology) preheated to 37°C and incubated in a dark
environment for 15 min. Images were captured and the cerebral
infarction volume was evaluated through ImageJ 1.54h software.
Brain water content detection
Brain tissue was washed with pre-cooled PBS, the
surface liquid was dried by filter paper and immediately weighed.
The weighed tissue was placed in a drying tube and placed in an
oven at 100°C for 24 h to dry (until the weight was constant) and
weighed after cooling to room temperature. Brain water content (%)
was calculated as follows: (Wet weight-dry weight)/wet weight
×100%.
Hematoxylin and eosin (HE) staining
Brain tissue was exposed to 4% paraformaldehyde for
48 h at 4°C, paraffin-embedded and serially sectioned (4
μm). The sections were dried and deparaffinized using
xylene, then rehydrated using gradient ethanol. Following 10 min
staining with hematoxylin, the sections were exposed to
differentiation solution (cat. no. C0161s, Beyotime Institute of
Biotechnology) for 30 sec. Sections were dyed with 1% eosin (cat.
no. C0109, Beyotime Institute of Biotechnology) for 60 sec,
dehydrated with gradient ethanol, transparentized with xylene and
covered with neutral balsam. The ischemic penumbra was evaluated
using a light microscope (Leica GmbH).
Nissl staining
Brain tissue was exposed to 4% paraformaldehyde for
48 h at 4°C, paraffin-embedded and serially sectioned (4
μm), and then placed in warm water to expand. The slices
were placed in a drying oven at 65°C for 90 min, dewaxed with
xylene for 20 min and rehydrated in gradient ethanol. The liquid
was removed, and sections were exposed to Nissl staining solution
(cat. no. C0117, Beyotime Institute of Biotechnology) for 10 min at
25°C. After rinsing with tap water, tissue was immersed in 95%
ethanol for dehydration for 4 min, then xylene. Following sealing
with neutral balsam, samples were visualized with an inverted light
microscope.
Fluoro-Jade C (FJC) staining
As described by Liu et al (34), the FJC degenerated neuron
staining kit (cat. no. TR-100-FJT, Wuhan Anjiekai Biological
Medicine Technology Co., Ltd.) was used to evaluate the neuronal
degeneration in mice. The frozen sections of mouse brain tissue
were treated with NaOH/ethanol solution (1:10) for dehydration for
4 min at 25°C, then treated with potassium permanganate (1:10
dilution) for 10 min at 25°C to enhance reagent permeability.
Sections were then stained with FJC solution (1:10) in darkness for
10 min at 25°C. Sections were sealed with Anti-fade mounting
medium, observed by fluorescence microscope and the proportion of
FJC-positive cells (degenerated neurons) was counted using ImageJ
1.54h software (National Institutes of Health).
Lactate dehydrogenase (LDH) assay
LDH is present in the cytoplasm and cannot penetrate
the intact cell membrane. Therefore, the detection of LDH release
can evaluate cell membrane damage (40). The release of LDH from HT22 cells
was assessed using Cytotoxicity LDH assay kit (cat. no. HY-K1090,
MedChemExpress). HT22 cells were centrifuged at 1,000 × g for 5 min
at 4°C, and 50 μl supernatant was mixed thoroughly with 50
μl LDH working solution. Following incubation in dark for 30
min at 25°C, 50 μl Stop Solution was applied and
OD490 value was detected immediately by microplate
reader. In addition, the fresh brain tissue of mice was cut into
pieces (~1 mm3), mixed with PBS, homogenized,
centrifuged at 8,000 × g for 10 min at 4°C, and supernatant was
mixed with LDH working solution. The LDH release in brain tissue
was determined as aforementioned.
Immunofluorescence
Brain tissue was exposed to 4% paraformaldehyde for
48 h at 4°C, paraffin-embedded and serially sectioned (4
μm). The paraffin sections of mouse brain tissue were
dewaxed with xylene, hydrated in a descending anhydrous ethanol
series. For antigen retrieval, sections were immersed in 0.01 M
citrate buffer (pH 6.0) and heated in a microwave oven at 95°C for
15 min, then naturally cooled to 25°C and rinsed three times with
PBS. Then, 0.3% Triton X-100 was dropped on the surface for 10 min
and 5% bovine serum albumin (BSA; cat. no. ST023; Beyotime
Biotechnology) was added for 1 h at 25°C. Sections were exposed
overnight to primary antibodies against Ionized calcium binding
adaptor molecule 1 (Iba1, cat. no. PA5-27436, 1:500) and CD86 (cat.
no. PA5-114995, 1:100) at 4°C. The sections were rinsed in PBS and
incubated with FITC- (cat. no. F-2765, 1:100) or Cy3-conjugated
goat anti-rabbit IgG (cat. no. A10520, 1:100) in darkness for 1.5 h
at 25°C. All antibodies were obtained from Invitrogen (Thermo
Fisher Scientific, Inc.). Following sealing with Anti-fade Mounting
Medium, sections were observed by a fluorescence microscope and the
fluorescence intensity was quantified using ImageJ 1.54h
software.
BV2 cells were seeded in a 12-well plate
(1.0×105 cells/well, coverslips were placed in the
plate) for 24 h to make cell climbing slides. The slides were
exposed to paraformaldehyde for 20 min, rinsed with PBS and then
incubated with 0.3% Triton X-100 at 25°C for 10 min, blocked with
BSA and exposed to primary antibodies against NLRP3 (cat. no.
MA5-32255, 1:1,000) and apoptosis-associated speck-like protein
containing a CARD (ASC, cat. no. PA5-50915, 1:200, Invitrogen;
Thermo Fisher Scientific, Inc.) overnight at 4°C. Subsequent
immunofluorescence analysis was performed as aforementioned.
ELISA
Mouse TNF-α (cat. no. PT512), IL-10 (cat. no.
PI522), IL-6 (cat. no. PI326), transforming growth factor-β (TGF-β;
cat. no. PT878), IL-1β (cat. no. PI301), IL-18 (cat. no. PI553) and
IFN-γ (cat. no.) PI508; all Beyotime Biotechnology) ELISA kits were
used to evaluate inflammatory factors levels in mouse brain tissue
and BV2 cells, according to the manufacturer's instructions. Mouse
Claudin-5 (Cla; cat. no. CSB-EL005507MO; Cusabio Technology LLC),
Occludin (Occ; cat. no. ml063481) and zona occludens 1 (ZO-1, cat.
no. ml037693; both Shanghai Enzyme-linked Biotechnology Co., Ltd.)
ELISA kits were used to evaluate the tight junction proteins in
mouse brain tissue, according to the manufacturer's instructions.
The target areas of brain tissue (ischemic penumbra and core area
and non-ischemic area) were separated by a blade. The brain tissue
homogenate or cell supernatant and corresponding antibody were
added to the ELISA plate at 37°C for 90 min. HRP-labeled
streptavidin was introduced and incubated in the dark at 37°C for
20 min. Chromogenic agent A and B were incubated for 15 min in a
dark environment at 37°C. Finally, 50 μl termination
solution was introduced and mixed well to determine
OD450 value.
Western blotting
The fresh brain tissue of the mice was cut into
pieces and then RIPA lysis buffer (cat. no. 20-188, Sigma-Aldrich;
Merck KGaA) was added. For BV2 cells, pre-cooled PBS was used for
gentle washing twice and then RIPA lysis buffer was added for
lysis. After lysis, the protein concentration was detected by the
BCA protein quantification kit. A total of 30 μg
protein/lane was loaded and separated by 10% SDS-PAGE, transferred
onto PVDF membranes and blocked with 5% BSA at room temperature for
2 h. Following rinsing with TBST containing 0.1% Tween-20, the
membrane underwent overnight incubation with primary antibodies
against synapsin-1 (SYP-1, cat. no. 51-5200), inducible nitric
oxide synthase (iNOS; cat. no. PA5-17106l; both 1:1,000), arginase
1 (Arg 1, cat. no. PA5-29645, 1:5,000), postsynaptic density
protein-95 (PSD-95; cat. no. 51-6900), brain-derived neurotrophic
factor (BDNF; cat. no. PA5-85730; both 1:500), growth-associated
protein 43 (GAP-43; cat. no. PA5-34943, 1:5,000), GSDMD (cat. no.
PA5-116815; all Invitrogen; Thermo Fisher Scientific, Inc.),
cleaved-caspase-1 (cat. no. HY-P80622; both 1:500, MedChemExpress),
pro-caspase-1 (cat. no. PA5-87536, 1:2,000, Invitrogen; Thermo
Fisher Scientific, Inc.), ASC (cat. no. ab283684), GSDMD-N (cat.
no. ab215203; both 1:1,000; both Abcam), NLRP3 (cat. no. MA5-32255,
1:500), IL-1β (cat. no. P420B, 1:1,000), IL-18 (cat. no. PA5-79479,
1:2,000), phosphorylated (p-)NF-κB-p65 (cat. no. 44-711G, 1:1,000),
MFG-E8 (cat. no. PA5-109955; all Invitrogen; Thermo Fisher
Scientific, Inc.) and NF-κB-p65 (cat. no. ab76311; both 1:2,000,
Abcam) at 4°C. The membrane underwent incubation with goat
anti-rabbit antibody (cat. no. 31460, 1:10,000, Invitrogen; Thermo
Fisher Scientific, Inc.) for 2 h at 25°C. Subsequently, the ECL
working solution (cat. no. HY-K1005, MedChemExpress) was prepared
and uniformly dropped onto the membrane, which was scanned using
iBright CL1500 gel imaging system (Invitrogen; Thermo Fisher
Scientific, Inc.). After scanning, the gray value was determined
with ImageJ 1.54h software and normalized to GAPDH (cat. no.
PA1-987, 1:1,000, Invitrogen; Thermo Fisher Scientific, Inc.).
Statistical analysis
All data are presented as the mean ± standard
deviation of ≥3 independent experimental repeats. SPSS 26.0
software (IBM Corp.) was employed for statistical analysis.
P<0.05 was considered to indicate a statistically significant
difference. Prism software (GraphPad 9.0; Dotmatics) was used for
plotting. The results were analyzed via unpaired t-test or one-way
ANOVA with post hoc Tukey's test.
Results
PPF suppresses excessive activation of
BV-2 cells and ameliorates damage in HT22 cells following
OGD/R
The present study reoxygenated BV-2/HT22 cells in
the presence of PPF following OGD. BV-2 and HT22 cell viability
markedly declined following OGD/R, as indicated by CCK-8 assay
(Fig. 1B). PPF (2-64 μM)
increased cell viability, but viability decreased at 128 μM
PPF (Fig. 1B). In subsequent
experiments, cells were treated with 8, 16 and 32 μM PPF to
evaluate its neuroprotective effects. Flow cytometry revealed that
following OGD/R, CD86 (a marker of M1-type microglia) positivity
increased in BV-2 cells, accompanied by a significant rise in
CD206-positive cells (a marker of M2-type microglia) Following PPF
treatment, the number of CD86-positive cells decreased
significantly, while CD206-positive cells increased significantly,
suggesting that PPF regulated the phenotypic conversion of BV-2
cells (Fig. 1C-E). Western
blotting verified that PPF decreased iNOS (an M1-type marker)
protein levels in BV-2 cells while upregulating Arg 1 (an M2-type
marker), consistent with flow cytometry findings (Fig. 1F and G). OGD/R-treated HT22 cells
were co-cultured with BV-2 cells to simulate neuroglial
interactions. Calcein AM/PI dual staining demonstrated that HT22
cell viability decreased notably following OGD/R treatment. When
HT22 cells were co-cultured with BV-2 cells, their survival rate
decreased further. PPF treatment effectively reversed this trend,
increasing the proportion of viable HT22 cells (Fig. 1H-J). TUNEL staining revealed that
OGD/R treatment significantly increased apoptosis in HT22 cells,
with further elevation following co-culture with BV-2 cells
(Fig. 1K and L). PPF treatment
significantly decreased apoptosis in HT22 cells (Fig. 1K and L). Following OGD/R, LDH
release in HT22 cells significantly increased. Co-culture with BV-2
cells further elevated LDH release, indicating exacerbated cell
damage. PPF treatment markedly decreased LDH release, suggesting
its protective effect on HT22 cells (Fig. 1M). In addition, PPF increased the
levels of SYP-1, PSD-95, BDNF and GAP-43 in HT22 cells, suggesting
synaptic function maintenance and neural repair in HT22 cells
(Fig. 1N and O). The results
indicate that PPF suppressed OGD/R-induced excessive activation in
BV-2 cells, mitigated damage to HT22 cells and upregulated the
expression of neurofunction-related proteins.
PPF suppresses NLRP3-mediated pyroptosis
in BV2 cells following OGD/R
SEM revealed that following OGD/R, BV2 cells
exhibited typical pyroptotic features, such as marked cellular
swelling, irregular morphology, membrane pores and cytoplasmic
contents leakage. Following PPF, the aforementioned morphological
abnormalities in BV2 cells were attenuated (Fig. 2A). The levels of pyroptosis were
assessed using Yo-Pro-1 and Hoechst 33342 staining. Following
OGD/R, Yo-Pro-1-positive staining in BV2 cells was markedly
increased, while PPF treatment decreased the number of
Yo-Pro-1-positive cells (Fig. 2B and
C). Immunofluorescence analysis revealed increased fluorescence
intensity of NLRP3 and ASC following OGD/R, whereas PPF decreased
the fluorescence intensity of both proteins in BV2 cells (Fig. 2D-F). Caspase-1 activity was
significantly elevated in BV2 cells following OGD/R, whereas PPF
decreased its activity (Fig.
2G). Moreover, following OGD/R, the protein levels of
cleaved-caspase-1/pro-caspase-1, GSDMD-N/GSDMD, NLRP3, IL-1β, ASC,
and IL-18 were markedly elevated in BV2 cells. By contrast, PPF
lowered protein levels, suggesting that PPF inhibited NLRP3
inflammasome activation (Fig.
2H-K). ELISA showed that the levels of IL-1β and IL-18 in BV2
cells were significantly elevated following OGD/R, and PPF
treatment decreased the levels of both (Fig. 2L). These results indicated that
PPF suppresses NLRP3 inflammasome activation following OGD/R,
thereby inhibiting pyroptosis in BV2 cells.
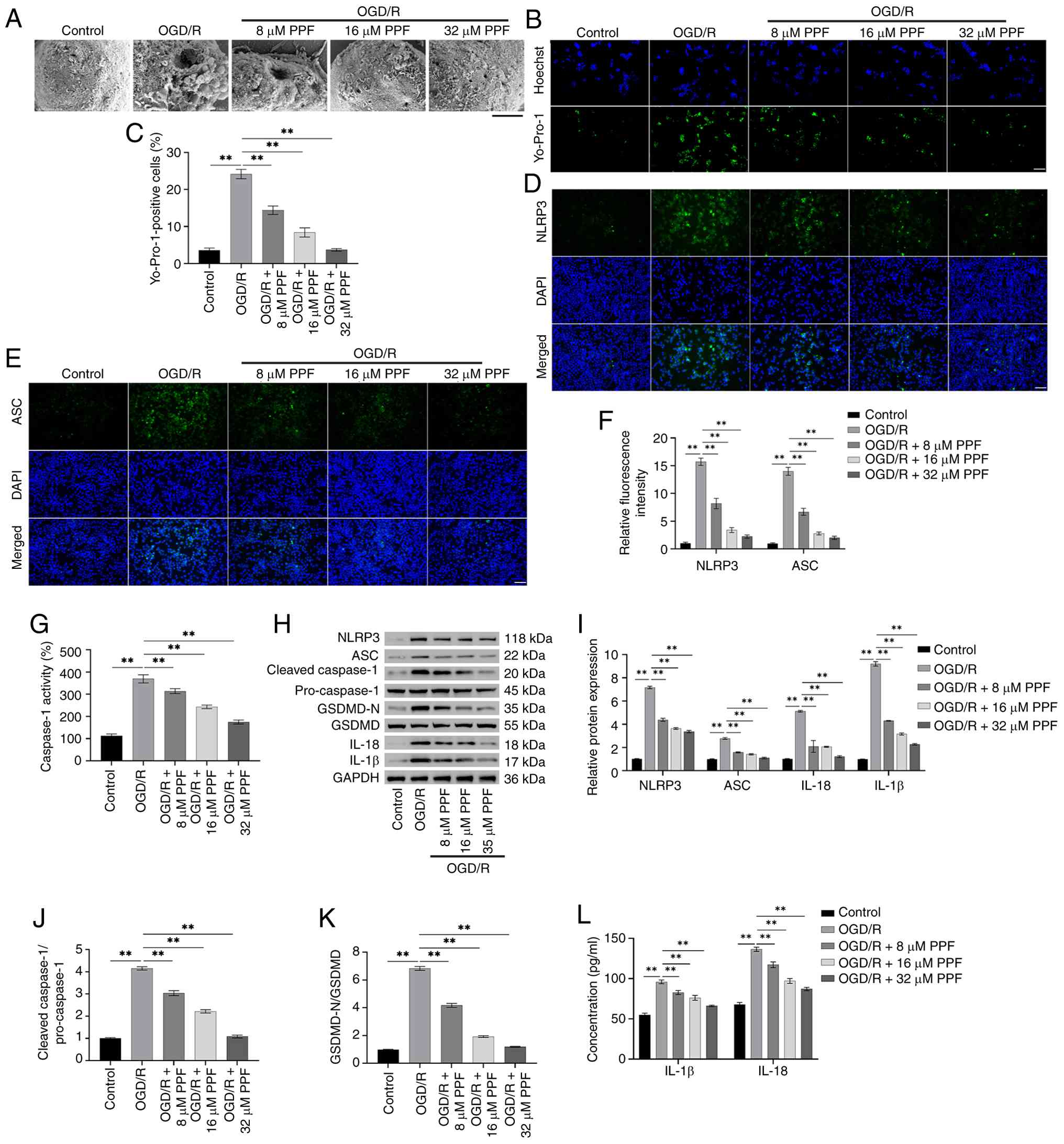 | Figure 2PPF suppresses NLRP3-mediated
pyroptosis in BV2 cells following OGD/R. (A) SEM observation of BV2
cell morphology (magnification, ×20,000; scale bar, 2 μm).
(B) Yo-Pro-1 and Hoechst 33342 staining for assessing the
pyroptosis levels in BV-2 cells. (C) Quantitative analysis of
Yo-Pro-1 and Hoechst 33342 staining revealed that PPF deceased
pyroptosis levels in BV-2 cells. (D) Immunofluorescence staining of
NLRP3 in BV2 cells (magnification, ×40; scale bar, 50 μm).
(E) Immunofluorescence staining of ASC in BV2 cells (magnification,
×40; scale bar, 50 μm). (F) Quantitative analysis of
immunofluorescence staining revealed that NLRP3 and ASC
fluorescence intensity increased in OGD/R-treated BV2 cells, while
PPF decreased the fluorescence intensity of both proteins. (G)
Caspase-1 activity was detected in BV2 cells following OGD/R using
Caspase-1 Activity Assay kit. (H) Western blotting of NLRP3, ASC,
cleaved-caspase-1, pro-caspase-1, GSDMD-N, GSDMD, IL-18, IL-1β, and
GAPDH (internal reference). (I) Western blot analysis revealed
elevated NLRP3, ASC, IL-1β and IL-18,
cleaved-caspase-1/pro-caspase-1 levels (J), and GSDMD-N/GSDMD
levels (K) in OGD/R-treated BV2 cells, PPF treatment reduced these
proteins levels. (L) ELISA showed that PPF decreased IL-1β and
IL-18 levels. **P<0.01. PPF, Propofol; OGD/R,
Oxygen-glucose deprivation/reoxygenation; ASC, apoptosis-associated
speck-like protein containing a CARD; GSDMD, gasdermin D. |
OE-MFG-E8 suppresses OGD/R-induced NF-κB
and NLRP3 activation in BV2 cells
Previous studies have revealed that MFG-E8 is key in
microglial inflammation (18,41). To investigate the impact of
MFG-E8 on NF-κB signaling and NLRP3 activation, OE-MFG-E8 was
transfected into BV2 cells, followed by OGD/R. Following OGD/R,
MFG-E8 expression decreased in BV2 cells, while p-NF-κB/NF-κB
levels increased, suggesting that OGD/R may downregulate MFG-E8 and
activate NF-κB signaling (Fig.
3A-C). Transfection of BV2 cells with OE-MFG-E8 significantly
elevated MFG-E8 protein levels, confirming successful establishment
of the model (Fig. 3D and E).
Following transfection with OE-MFG-E8, p-NF-κB/NF-κB, NLRP3 and ASC
expression notably declined in BV2 cells (Fig. 3F-H). The present study examined
the effects of different concentrations of NF-κB activator LPS on
the protein levels of the NF-κB pathway in microglia and found that
0.25-2.00 μg/ml LPS effectively reduced p-NF-κB/NF-κB ratio
(Fig. S1A and B). Additionally,
LPS at 0.125-1.000 μg/ml had no adverse effect on BV-2 cell
viability (Fig. S1C), therefore
1 μg/ml LPS was selected for subsequent experiments. The
p-NF-κB/NF-κB, NLRP3 and ASC levels were significantly elevated in
BV-2 cells following LPS treatment (Fig. 3I-K). Overexpression of MFG-E8
significantly decreased p-NF-κB/NF-κB levels in OGD/R-treated BV2
cells, whereas LPS increased p-NF-κB/NF-κB levels (Fig. 3L and M). Following OGD/R,
immunofluorescence indicated increased fluorescence intensity of
ASC and NLRP3 in BV2 cells. OE-MFG-E8 decreased the fluorescence
intensity of both proteins, whereas LPS attenuated the effect of
OE-MFG-E8 (Fig. 3N-P). In
addition, following MFG-E8 overexpression,
cleaved-caspase-1/pro-caspase-1, NLRP3, ASC, IL-1β, GSDMD-N/GSDMD
and IL-18 protein levels were reduced notably in OGD/R-treated BV2
cells. However, LPS inhibited the impact of OE-MFG-E8
overexpression (Fig. 3Q-T). The
results indicated that OE-MFG-E8 suppresses NF-κB signaling and
NLRP3 activation in OGD/R-induced BV2 cells.
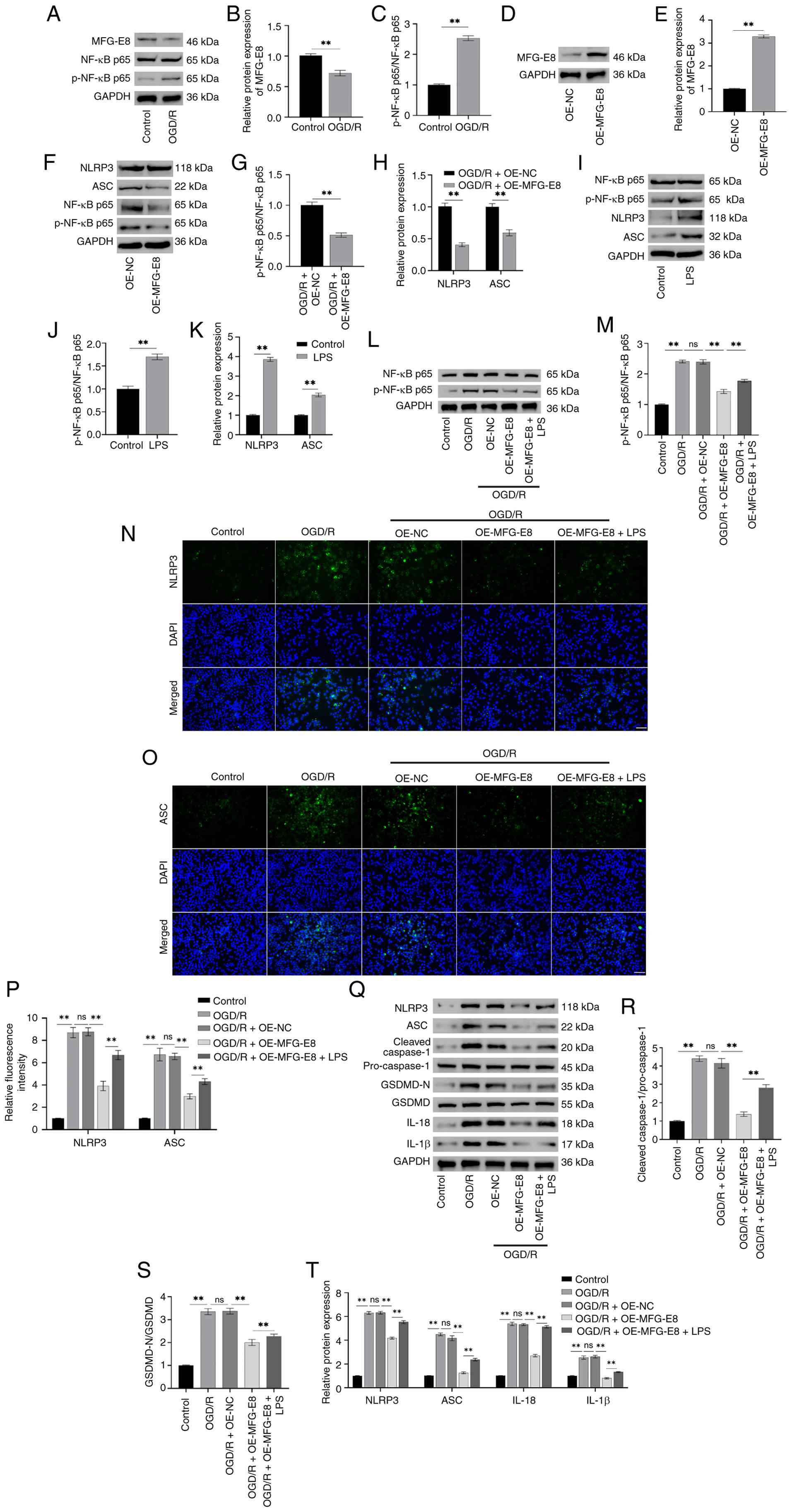 | Figure 3Overexpression of MFG-E8 suppresses
NF-κB and NLRP3 activation in OGD/R-induced BV2 cells. (A) Western
blotting protein bands for MFG-E8, p-NF-κB, NF-κB, and GAPDH
(internal reference). (B and C) Following OGD/R, western blotting
revealed decreased MFG-E8 expression (B) and elevated p-NF-κB/NF-κB
levels (C) in BV2 cells. (D) Western blotting protein bands for
MFG-E8 and GAPDH (internal reference). (E) Following transfection
of OE-MFG-E8 into BV2 cells, MFG-E8 expression was elevated
markedly. (F) Western blotting protein bands for NLRP3, ASC,
p-NF-κB, NF-κB, and GAPDH (internal reference). (G and H) Following
transfection with OE-MFG-E8, p-NF-κB/NF-κB levels (G), and NLRP3
and ASC levels (H) were detected using western blotting. (I)
Western blotting protein bands for NLRP3, ASC, p-NF-κB, NF-κB, and
GAPDH (internal reference). Following treatment with the NF-κB
activator LPS, p-NF-κB/NF-κB (J), and NLRP3 and ASC levels (K) were
detected by western blotting. (L) Western blotting protein bands
for p-NF-κB, NF-κB, and GAPDH (internal reference). (M) Following
transfection with OE-MFG-E8, p-NF-κB/NF-κB expression was
decreased, whereas treatment with LPS increased p-NF-κB/NF-κB
levels. (N) Immunofluorescence staining of NLRP3 in BV2 cells
(magnification, ×40; scale bar, 50 μm). (O)
Immunofluorescence staining of ASC in BV2 cells (magnification,
×40; scale bar, 50 μm). (P) Immunofluorescence revealed
OE-MFG-E8 reduced the fluorescence intensity of NLRP3 and ASC,
while LPS diminished the effect of MFG-E8 overexpression. (Q)
Western blotting protein bands for NLRP3, ASC, cleaved-caspase-1,
pro-caspase-1, GSDMD-N, GSDMD, IL-1β, IL-18, and GAPDH (internal
reference). (R-T) Western blotting revealed that OE-MFG-E8 reduced
cleaved-caspase-1/pro-caspase-1 levels (R), GSDMD-N/GSDMD levels
(S), NLRP3, ASC, IL-1β, and IL-18 levels (T) in OGD/R-induced BV2
cells, LPS attenuated the impact of OE-MFG-E8.
**P<0.01. PPF, Propofol; OGD/R, Oxygen-glucose
deprivation/reoxygenation; ASC, apoptosis-associated speck-like
protein containing a CARD; MFG-E8, milk fat globule-EGF factor 8;
GSDM, gasdermin; OE, overexpression; p-, phosphorylated; LPS,
lipopolysaccharide; NC, negative control; ns, not significant. |
PPF suppresses pyroptosis induced by
NF-κB/NLRP3 signaling by upregulating MFG-E8
The present study investigated whether PPF inhibits
pyroptosis by regulating MFG-E8 in BV-2 cells. si-MFG-E8 was
transfected into BV-2 cells, which were subjected to OGD/R and
exposed to PPF for 24 h. PPF treatment significantly increased
MFG-E8 expression in BV2 cells, while simultaneously decreasing
p-NF-κB/NF-κB levels, suggesting that PPF may inhibit NF-κB
activation by upregulating MFG-E8 (Fig. 4A-C). Following transfection with
si-MFG-E8, MFG-E8 protein expression markedly declined in BV2
cells, indicating successful establishment of MFG-E8 silencing
(Fig. 4D and E). Silencing
MFG-E8 significantly increased the luciferase activity of NF-κB-WT
but had no significant effect on NF-κB-MUT, confirming that NF-κB
was a downstream target of MFG-E8 (Fig. 4F). si-MFG-E8 attenuated the
impact of PPF, causing a notable rise in p-NF-κB/NF-κB levels. This
indicated that the inhibitory impact of PPF on NF-κB activation was
dependent on MFG-E8 (Fig. 4G and
H). PPF treatment significantly reduced the number of
Yo-Pro-1-positive BV2 cells following OGD/R, whereas MFG-E8
silencing led to an increase in Yo-Pro-1 positivity, indicating
that si-MFG-E8 attenuated the inhibitory effect of PPF on
pyroptosis (Fig. 4I and J).
ELISA revealed that levels of proinflammatory factors TNF-α, IL-1β
and IL-6 were markedly raised in BV2 cells following OGD/R, while
IL-10 levels decreased. PPF treatment reversed this effect, whereas
silencing MFG-E8 attenuated the anti-inflammatory effect of PPF
(Fig. 4K-N). Additionally, PPF
markedly decreased cleaved-caspase-1/pro-caspase-1, NLRP3, ASC,
IL-1β, GSDMD-N/GSDMD and IL-18 levels in OGD/R-treated BV2 cells,
whereas si-MFG-E8 markedly increased these protein levels (Fig. 4O-R). These results suggested that
PPF suppresses pyroptosis by upregulating MFG-E8 in BV2 cells,
thereby inhibiting NF-κB/NLRP3 pathway activation.
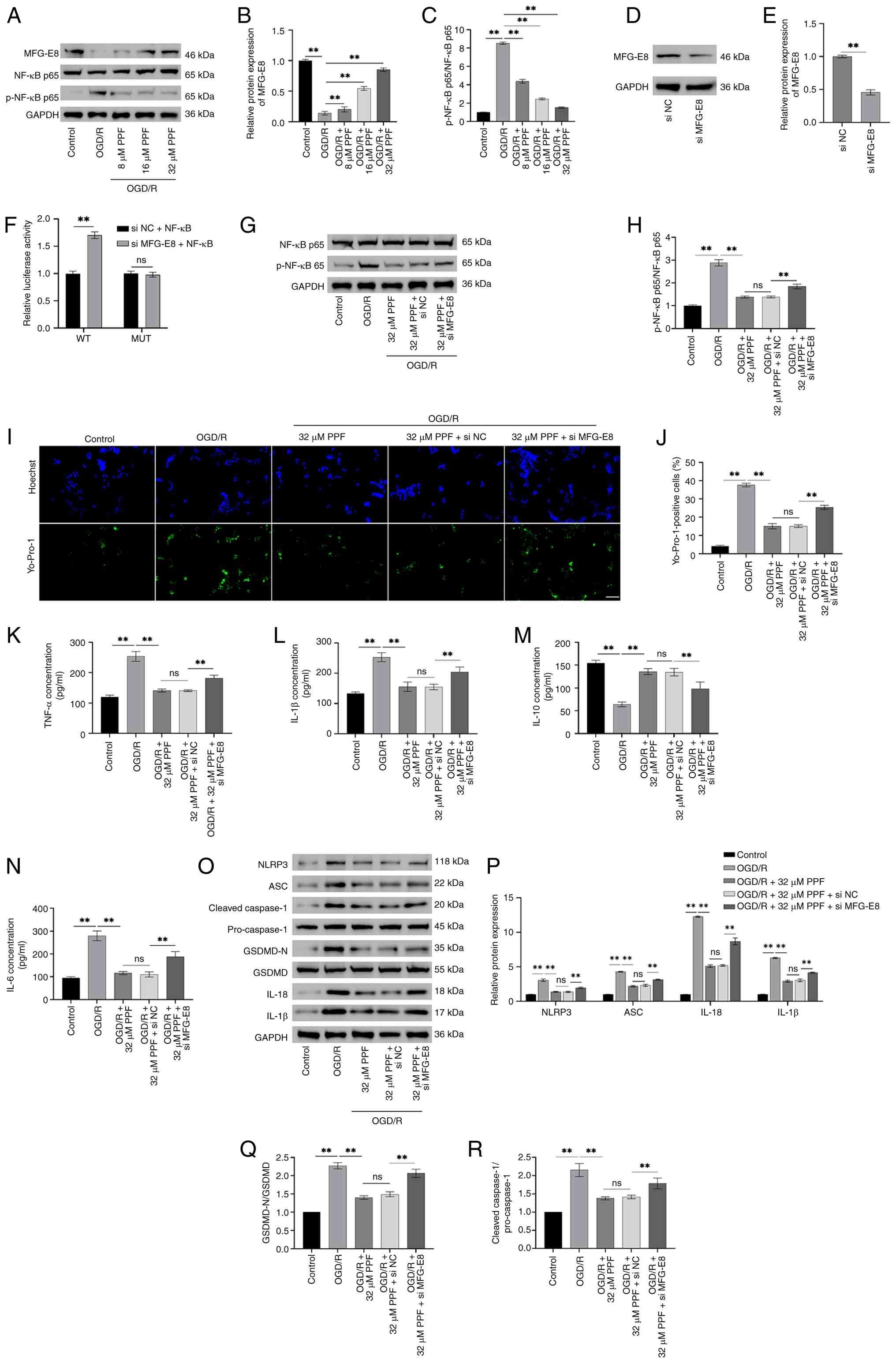 | Figure 4PPF suppresses pyroptosis caused by
NF-κB/NLRP3 signaling by upregulating MFG-E8. (A) Western blotting
protein bands for MFG-E8, p-NF-κB, NF-κB, and GAPDH (internal
reference) in BV2 cells. (B) Western blotting revealed that PPF
increased MFG-E8 and decreased p-NF-κB/NF-κB levels (C) in BV2
cells. (D) Western blotting for MFG-E8 and GAPDH in BV2 cells. (E)
Following transfection of si-MFG-E8 into BV2 cells, MFG-E8 protein
levels were significantly decreased. (F) Dual luciferase reporter
gene assay confirmed that NF-κB is the target of MFG-E8. (G)
Western blotting protein bands for p-NF-κB, NF-κB, and GAPDH
(internal reference) in BV2 cells. (H) si-MFG-E8 attenuated the
effects of PPF, resulting in elevated p-NF-κB/NF-κB levels. (I)
Yo-Pro-1 and Hoechst 33342 staining for assessing the pyroptosis
levels in BV-2 cells (magnification, ×40; scale bar, 50 μm).
(J) Yo-Pro-1 and Hoechst 33342 staining showed that PPF decreased
Yo-Pro-1 positivity, while silencing MFG-E8 increased Yo-Pro-1
positivity. ELISA showed that PPF decreased TNF-α (K) and IL-1β (L)
levels, silencing MFG-E8 reversed this effect. (M and N) ELISA
showed that PPF raised IL-10 levels (M) and decreased IL-6 levels
(N), silencing MFG-E8 reversed this effect. (O) Western blotting
protein bands for NLRP3, ASC, cleaved-caspase-1, pro-caspase-1,
GSDMD-N, GSDMD, IL-1β, IL-18, and GAPDH (internal reference) in BV2
cells. (P-R) Western blot analysis indicated that PPF decreased
NLRP3, ASC, IL-1β, and IL-18 levels (P), GSDMD-N/GSDMD levels (Q),
cleaved-caspase-1/pro-caspase-1 levels (R), silencing MFG-E8
increased these protein levels. **P<0.01. PPF,
propofol; MFG-E8, milk fat globule-EGF factor 8; OGD/R,
Oxygen-glucose deprivation/reoxygenation; ASC, apoptosis-associated
speck-like protein containing a CARD; GSDM, gasdermin; OE,
overexpression; p-, phosphorylated; LPS, lipopolysaccharide; NC,
negative control; ns, not significant; si, small interfering; WT,
wild-type; MUT, mutant. |
PPF improves neurological deficit and
reduces brain tissue pathology in tMCAO mice
The present study established a tMCAO mouse model to
investigate whether PPF effectively improves neuronal injury in
vivo (Fig. 5A). The Longa
scoring system results demonstrated that neurological deficit
scores were significantly lower in the PPF-treated group compared
with the tMCAO group, indicating that PPF improved neurological
function in tMCAO mice (Fig.
5B). The corner test demonstrated that PPF effectively
mitigated sensorimotor deficits in tMCAO mice, reduced turning bias
and enhanced limb coordination and spatial perception (Fig. 5C). Moreover, PPF significantly
reduced brain tissue water content in tMCAO mice, indicating that
PPF alleviated ischemia-induced cerebral edema (Fig. 5D). TTC staining revealed
well-defined white infarct lesions in the cerebral cortex of tMCAO
mice. Following PPF treatment, the white infarct areas
significantly decreased (Fig. 5E and
F). HE staining revealed notable brain tissue damage in the
penumbral region of tMCAO mice, characterized by disorganized cell
arrangement and pronounced interstitial edema. PPF mitigated the
extent of brain tissue damage (Fig.
5G). Nissl staining demonstrated a significant reduction in
Nissl-positive cells within ischemic penumbra of tMCAO mice,
indicating severe neuronal damage. PPF markedly increased the
number of Nissl-positive cells (Fig.
5H and I). Additionally, the number of FJC-positive
degenerating neurons in the ischemic penumbra of tMCAO mice was
markedly elevated. Following PPF treatment, the number of
FJC-positive cells markedly declined, indicating that PPF
suppressed neuronal degeneration following ischemia (Fig. 5J and K). The results indicate
that PPF improves neurological deficits in tMCAO mice and
effectively mitigated pathological damage in brain tissue.
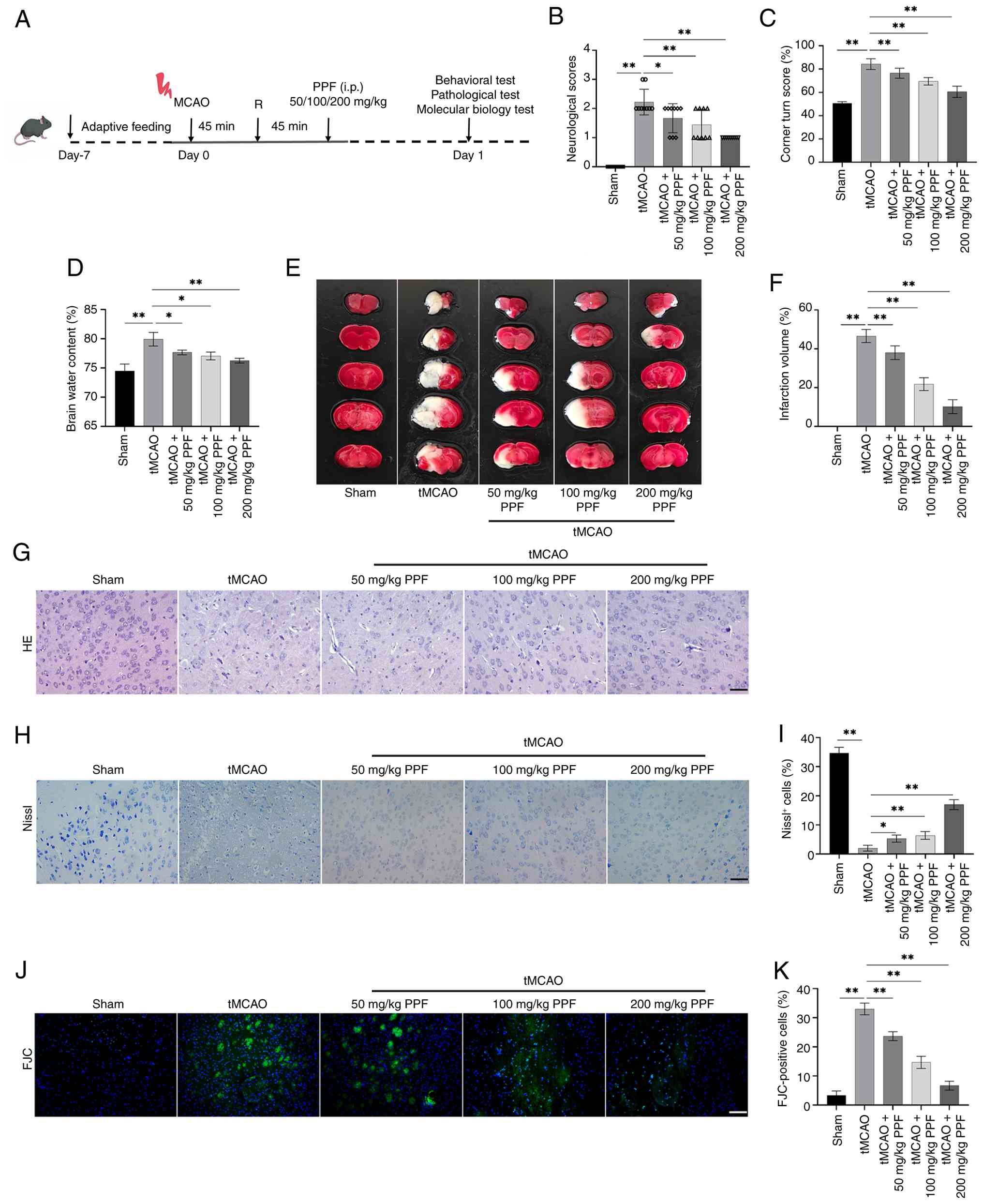 | Figure 5PPF improves neurological deficit and
decreases brain tissue pathology in tMCAO mice. (A) Operational
flowchart. (B) Longa scoring indicated that PPF decreased
neurological deficit scores in tMCAO mice (n=9). (C) Corner test
results demonstrated that PPF decreased sensorimotor deficits in
tMCAO mice. (D) PPF decreased brain water content in tMCAO mice.
(E) TTC staining to assess cerebral infarct lesions in tMCAO mice.
(F) TTC staining revealed well-defined infarct lesions in the
cerebral cortex of tMCAO mice, with PPF diminishing infarct volume.
(G) HE staining was used to assess the histopathological brain
damage in tMCAO mice (magnification, ×40; scale bar, 50 μm).
(H) Nissl staining was used to assess the proportion of
Nissl-positive cells in the brain tissue of tMCAO mice
(magnification, ×40; scale bar, 50 μm). (I) Nissl staining
revealed a decrease in Nissl-positive cell count in tMCAO mice. PPF
increased the number of Nissl-positive cells. (J) Number of
degenerative neurons in tMCAO mice was observed by FJC staining
(magnification, ×20; scale bar, 100 μm). (K) FJC staining
indicated PPF decreased the number of degenerating neurons in the
ischemic penumbra of tMCAO mice. *P<0.05,
**P<0.01. PPF, Propofol; tMCAO, transient middle
cerebral artery occlusion; HE, hematoxylin and eosin; FJC,
Fluoro-Jade C; i.p., intraperitoneal injection. |
PPF suppresses glial cell activation and
neuronal injury in tMCAO mice
Immunofluorescence colocalization revealed a notably
raised proportion of CD86+ Iba1+ cells in the
ischemic penumbra of tMCAO mouse brains, suggesting a shift of
microglia toward a proinflammatory phenotype (M1 type). Following
PPF treatment, the proportion of CD86+ Iba1+
cells significantly decreased, indicating PPF suppressed
proinflammatory activation of microglia in tMCAO mice after
ischemia (Fig. 6A and B). In
tMCAO mouse brain tissue, levels of proinflammatory factors (TNF-α,
IFN-γ) were markedly raised in the ischemic core (Fig. 6C), penumbra (Fig. 6D) and non-ischemic regions
(Fig. 6E), while
anti-inflammatory factor (IL-10, TGF-β) levels notably decreased.
PPF treatment reversed this phenomenon. LDH levels were markedly
elevated in the ischemic penumbra of tMCAO mice, while PPF
effectively decreased these levels, suggesting that PPF mitigated
the extent of cellular damage (Fig.
6F). tMCAO mice exhibited significantly decreased levels of
Cla, Occ, and ZO-1, whereas PPF markedly increased these tight
junction protein levels, confirming its protective effect on the
blood-brain barrier (BBB; Fig.
6G). Moreover, tMCAO mice exhibited significantly decreased
levels of SYP-1 and PSD-95 in brain tissue and elevated levels of
BDNF and GAP-43; PPF treatment reversed these effects (Fig. 6H and I). These results indicated
that PPF alleviates neuronal injury by suppressing microglial
activation in tMCAO mice, preserving BBB integrity and modulating
the expression of proteins associated with neuronal function.
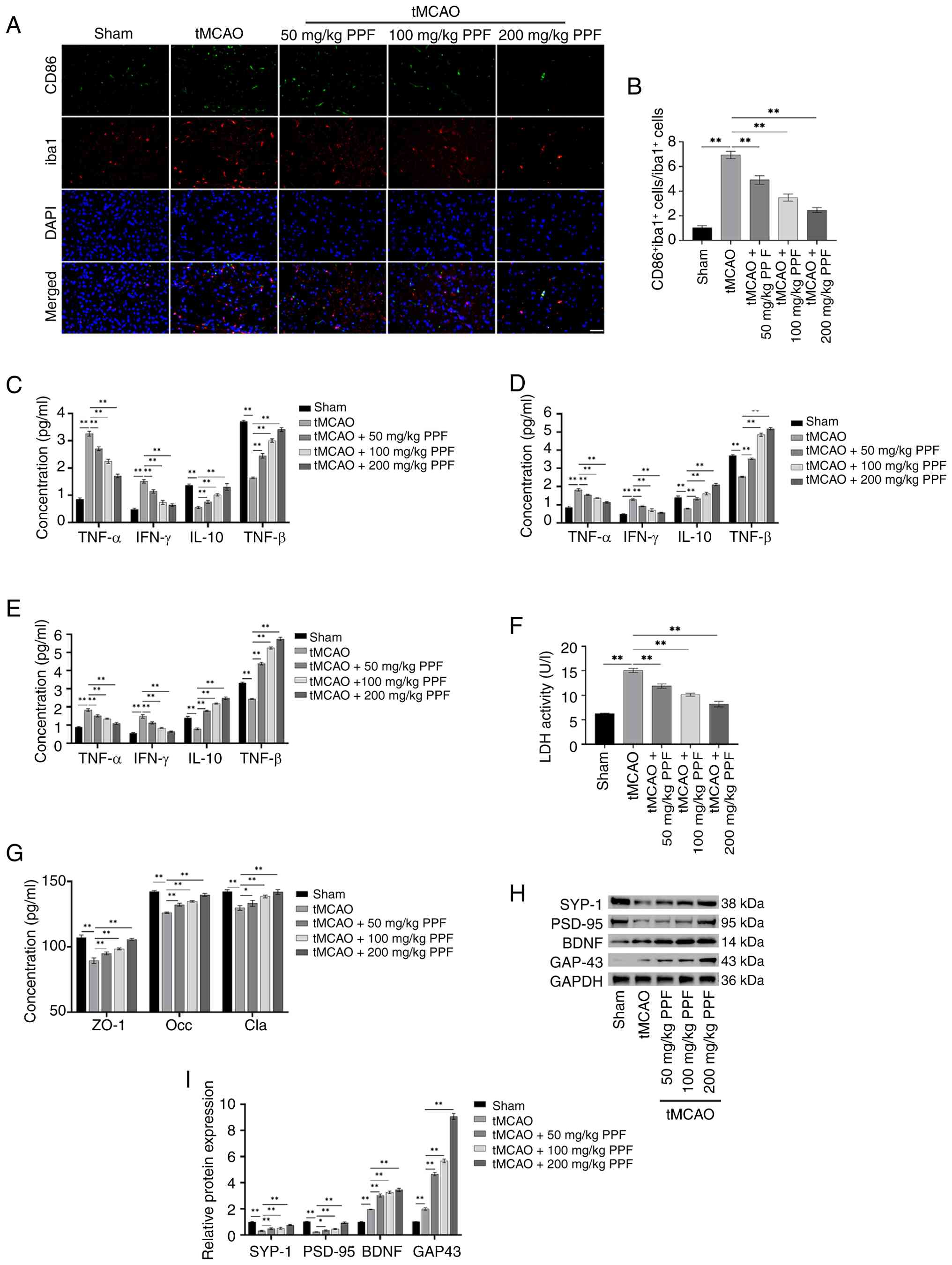 | Figure 6PPF suppresses microglial activation
and neuronal injury in tMCAO mice. (A) Co-localization of Iba1 and
CD86 was detected by immunofluorescence. (magnification, ×40; scale
bar, 50 μm). (B) Immunofluorescence colocalization revealed
an increased proportion of CD86+ Iba1+ cells
in tMCAO mice, which was decreased by PPF treatment. ELISA
detection of TNF-α, IL-10, IFN-γ, and TGF-β levels in the ischemic
(C) core and (D) penumbra and (E) non-ischemic zone of brain
tissue. (F) Ischemic penumbra of tMCAO mice exhibited heightened
LDH levels. PPF reduced LDH levels. (G) ELISA revealed decreased
Cla, Occ and ZO-1 levels in tMCAO mice, while PPF elevated these
tight junction-associated protein levels. (H) Western blotting of
SYP-1, PSD-95, BDNF, GAP-43, and GAPDH (internal reference). (I)
Western blotting revealed decreased SYP-1 and PSD-95 levels, along
with increased BDNF and GAP-43 levels in tMCAO mice; these effects
were reversed by PPF. *P<0.05,
**P<0.01. PPF, Propofol; tMCAO, transient middle
cerebral artery occlusion; Iba, Ionized calcium binding adaptor
molecule; LDH, lactate dehydrogenase; Cla, Claudin; Occ, Occludin;
ZO-1, zona occludens 1; PSD, postsynaptic density protein; BDNF,
brain-derived neurotrophic factor; GAP, growth-associated protein;
SYP, synapsin. |
PPF upregulates MFG-E8 to hinder
activation of the NF-κB/NLRP3 pathway
The present study investigated whether PPF exerts
neuroprotective effects by regulating MFG-E8 expression, thereby
identifying the key target of PPF intervention. MFG-E8 protein
expression was notably decreased in tMCAO mice, whereas
p-NF-κB/NF-κB levels were significantly elevated. Furthermore,
NLRP3 inflammasome and pyroptosis-associated protein levels were
markedly elevated. Following PPF treatment, MFG-E8 levels markedly
elevated in the ischemic penumbra, while p-NF-κB/NF-κB and
pyroptosis-associated protein levels significantly decreased. This
indicated that PPF may suppress NF-κB/NLRP3 pathway activation and
hinder pyroptosis by upregulating MFG-E8 (Fig. 7A-F). At 3 days after
intracerebroventricular injection of si-MFG-E8, the expression of
MFG-E8 protein in mouse brain tissue was significantly decreased
(Fig. 7G and H). Additionally,
MFG-E8 silencing markedly attenuated the protective impact of PPF
on the ischemic penumbra in tMCAO mice. Compared with tMCAO + 200
mg/kg PPF +si NC group, p-NF-κB/NF-κB levels were elevated and
cleaved-caspase-1/pro-caspase-1, NLRP3, ASC, IL-1β, GSDMD-N/GSDMD
and IL-18 levels were markedly increased in tMCAO + 200 mg/kg PPF
+si MFG-E8 group, confirming MFG-E8 as a key molecular target for
PPF-mediated neuroprotective effects (Fig. 7I-N). These findings suggest that
PPF suppressed abnormal activation of the NF-κB/NLRP3 pathway by
upregulating MFG-E8, thereby decreasing pyroptosis. This may
represent a key molecular mechanism underlying its neuroprotective
effects.
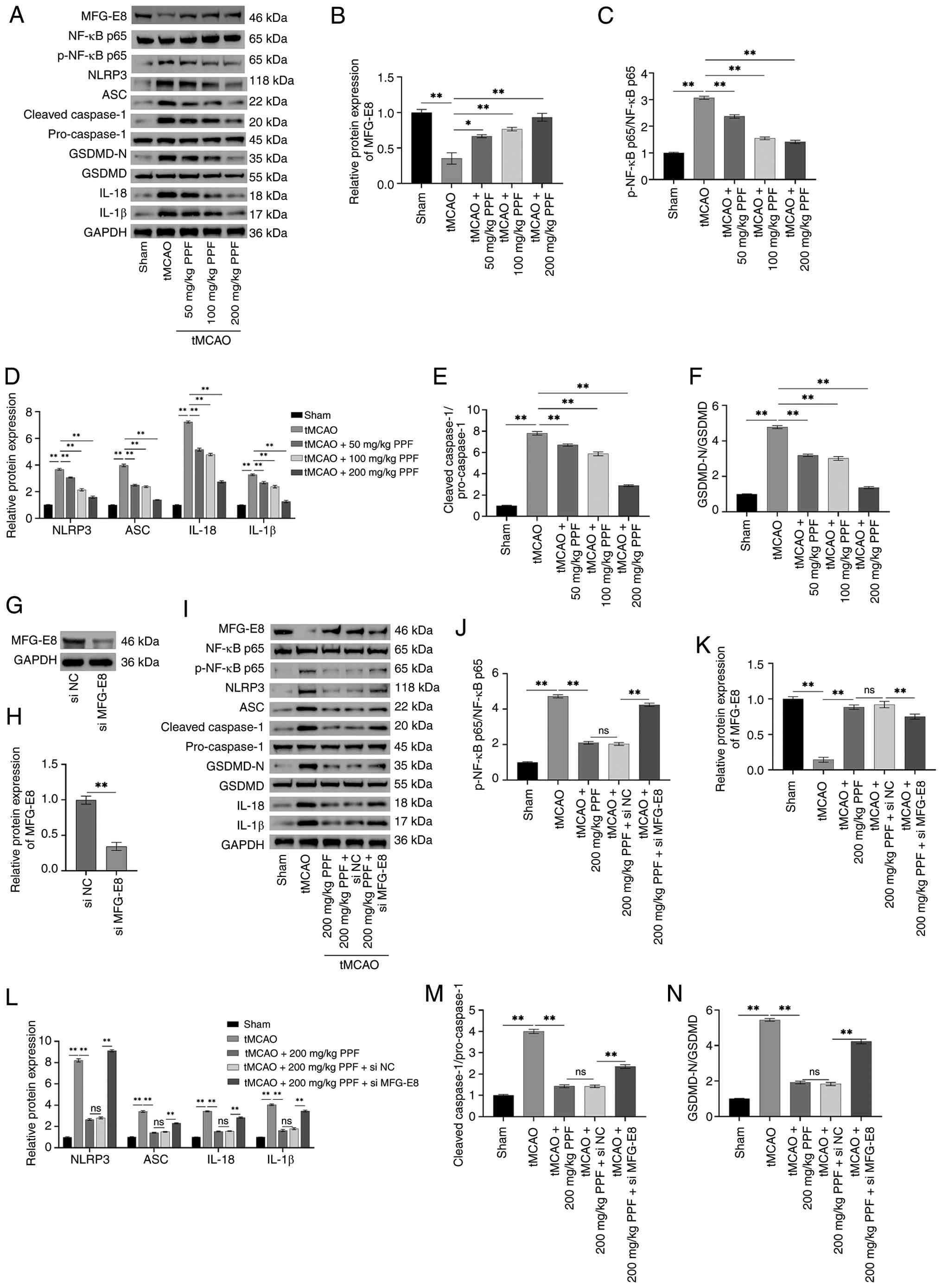 | Figure 7PPF upregulates MFG-E8 to inhibit
activation of NF-κB/NLRP3 pathway. (A) Western blotting of MFG-E8,
p-NF-κB, NF-κB, NLRP3, ASC, cleaved-caspase-1, pro-caspase-1,
GSDMD-N, GSDMD, IL-18, IL-1β, and GAPDH (internal reference). (B-F)
Western blot analysis revealed decreased MFG-E8 (B) levels in tMCAO
mice, along with elevated p-NF-κB/NF-κB (C), NLRP3, ASC, IL-1β,
IL-18 (D), cleaved-caspase-1/pro-caspase-1 (E), GSDMD-N/GSDMD (F)
levels. PPF reduced these protein levels. (G) Western blotting
protein bands for MFG-E8 and GAPDH (internal reference). (H)
Western blotting detected the expression of MFG-E8 in the brain
tissue of mice after injection of si-MFG-E8/si-NC. (I) Western
blotting protein bands for MFG-E8, p-NF-κB, NF-κB, NLRP3, ASC,
cleaved-caspase-1, pro-caspase-1, GSDMD-N, GSDMD, IL-18, IL-1β, and
GAPDH (internal reference). (J-N) Western blot analysis revealed
that si-MFG-E8 diminished the therapeutic efficacy of PPF, leading
to deceased MFG-E8 level (J) and elevated p-NF-κB/NF-κB (K), NLRP3,
ASC, IL-1β, and IL-18 levels (L), and
cleaved-caspase-1/pro-caspase-1 (M) and GSDMD-N/GSDMD (N) levels in
tMCAO mice. *P<0.05, **P<0.01. PPF,
Propofol; tMCAO, transient middle cerebral artery occlusion;
MFG-E8, milk fat globule-EGF factor 8; p-, phosphorylated; ASC,
apoptosis-associated speck-like protein containing a CARD; ns, not
significant; GSDMD, gasdermin D; si, small interfering; NC,
negative control. |
Discussion
CIRI-mediated neuroinflammation is a key
pathological mechanism in secondary brain injury. As crucial immune
cells in the central nervous system, microglia are rapidly
activated and polarized toward a pro-inflammatory phenotype (M1
type) following IRI. They release numerous pro-inflammatory
factors, triggering an inflammatory storm that exacerbates neuronal
damage (42,43). BV-2 cells are widely used as a
classical microglia model in vitro to study the inflammatory
mechanisms associated with ischemic brain injury (44,45). The key pathology of IRI is that
ischemia-induced interruption of oxygen and glucose supply triggers
cell hypoxic stress, which activates downstream inflammatory
signaling pathways (46). The
in vitro OGD/R model simulates this key pathological
process, laying the foundation for in vivo experiments to
explore the IR pathological process. The suture-based tMCAO model
is considered the most representative surgical model for simulating
human IS, offering advantages such as avoiding craniotomy, ease of
operation and stable, controllable reperfusion (47,48). Based on the methods described by
Liu et al (34) and Xu
et al (29), the present
study established OGD/R glial (BV-2) and hippocampal neuronal cell
(HT22) models, as well as a tMCAO mouse model, to elucidate the
neuroprotective effects of PPF in IS. Following OGD/R, BV2 cell
viability decreased, M1 markers were highly expressed and their M1
polarization exacerbated apoptosis in HT22 cells following OGD/R,
confirming the successful establishment of the OGD/R cell model.
Furthermore, mice following tMCAO surgery exhibited pronounced
neurological deficit and characteristic pathological lesions,
including elevated neurological deficit score, enlarged infarct
volume and increased numbers of degenerating neurons, indicating
successful establishment of the tMCAO mouse model. PPF enhanced BV2
and HT22 cell viability following OGD/R, decreased LDH release,
inhibited M1 polarization in BV2 cells and apoptosis in HT22 cells
and decreased neurological deficit scores and cerebral infarct
volume in tMCAO mice. Sun et al (17) confirmed that PPF can mitigate
cortical damage in MCAO mice and decrease apoptosis in HT22 cells.
These experimental findings collectively demonstrate that PPF
improves neuronal injury, thus establishing a basis for deeper
investigation into its neuroprotective effects. When the
anti-inflammatory ability is enhanced, the expression of CD206
increases, while in a pro-inflammatory environment, the expression
of CD86 increases. However, the OGD/R-induced double elevation in
the present study suggests an imbalance between the over-activation
of the M1 pathway and the pseudo-activation of the M2 pathway: On
the one hand, OGD/R continuously activates the NF-κB/NLRP3 pathway
through oxidative stress, leading to high CD86 expression and
release of pro-inflammatory factors, triggering an excessive
inflammatory response, whereas elevated CD206 is a compensatory
response for the body to initiate endogenous repair, but the repair
effect of the M2 phenotype in this state is weak, as evidenced by
the secretion of insufficient anti-inflammatory factors to inhibit
inflammation or promote tissue repair (49). Under complex pathological stimuli
such as hypoxia and trauma, microglia show co-expression of M1/M2
markers, but a pro-inflammatory phenotype (M1 type) predominates
(9,50). In conclusion, the elevation of
CD86 and CD206 after OGD/R is a phenotypic feature of microglia
imbalance.
The ischemic brain injury zone is divided into two
primary regions: Ischemic core and the penumbra (51). The ischemic core represents the
area of most severe ischemic damage where brain cells have
undergone irreversible necrotic death (52). The ischemic penumbra signifies
the brain tissue located between the ischemic core and the normal
area. If reperfusion therapy is administered promptly following
ischemic injury, the neurons in the penumbra retain the potential
for recovery (53,54). Therefore, preserving the survival
of neurons and delaying damage progression are key strategies for
treating IS. Given the clinical significance of the ischemic
penumbra, the present study focused on the protective impact of PPF
on neurons in the ischemic penumbra of tMCAO mice and its
underlying mechanisms. Histopathological staining revealed that PPF
increased the number of Nissl-positive neurons, decreased the
number of degenerating neurons and suppressed pro-inflammatory
microglia activation in the penumbra. This suggested that PPF
effectively protected neurons in the ischemic penumbra. The BBB, as
a key physiological barrier, typically restricts the migration of
peripheral immune cells into brain parenchyma through its tight
junction structure. Cerebral ischemia compromises the BBB,
triggering immune cell infiltration and causing neuronal damage
(55-57). Therefore, the present study
evaluated the protective effect of PPF on the BBB. PPF effectively
increased the tight junction-associated protein (Cla, Occ, and
ZO-1) levels in the ischemic penumbra, indicating that PPF repaired
the damaged BBB by upregulating these proteins. This may represent
a key mechanism by which PPF decreases peripheral immune cell
infiltration, alleviates neuroinflammation and protects neurons.
Additionally, tMCAO-induced inflammatory response resulted in a
significant decrease in SYP-1 and PSD-95 expression, reflecting the
disruption of synaptic structure and loss of neurotransmission in
the ischemic area. At the same time, the endogenous neuroprotective
host self-protection mechanism is activated, which increases the
expression of endogenous BDNF and promotes the repair of damaged
neurons. However, this compensatory effect does not eliminate the
neuronal damage caused by ischemia (58,59). The axonal regeneration marker
GAP-43 also fails to form functional growth cones at damaged axon
terminals to complete nerve loop reconstruction (60). Therefore, it was hypothesized
that the increase in protein levels of BDNF and GAP-43 during the
acute phase of tMCAO was a compensatory stress response.
MFG-E8, as a multifunctional glycoprotein, has drawn
more interest in recent years for its role in nerve injury repair
(61,62). Its key functions are in immune
regulation, and maintenance of tissue homeostasis (61). Cheyuo et al (63) demonstrated that MFG-E8 expression
decreases in the brains of MCAO rats 24 h after ischemia, and
injection of rhMFG-E8 decreases cerebral infarct volume and levels
of necrotic neurons. In a hypoxic-ischemic encephalopathy rat
model, MFG-E8 exhibits abnormal downregulation, treatment with
MFG-E8-carrying exosomes decreases cerebral infarction volume by
inhibiting autophagy and ferroptosis and alleviates cerebral edema
in rats (62). The present study
further validated this phenomenon: MFG-E8 levels notably declined
in the ischemic penumbra of tMCAO mice and in BV2 cells following
OGD/R. Conversely, PPF treatment induced a concentration-dependent
upregulation of MFG-E8, suggesting that MFG-E8 may act as a key
target for the neuroprotective effects of PPF. Cai et al
(27) investigated resting
microglia and found that PPF attenuates phagocytosis in resting BV2
cells by downregulating MFG-E8 expression. This suggests the
regulatory effect of PPF on MFG-E8 may dynamically adjust according
to the state of microglia. Future studies should investigate the
differential regulatory mechanisms of the PPF/MFG-E8 pathway across
different pathological stages or cell states.
Pyroptosis, also known as inflammatory necrosis, is
distinguished by the breaking of the cell membrane and notable
release of cell contents, thereby triggering a severe inflammatory
response (64,65). IRI activates the NLRP3
inflammasome in microglia, triggering pyroptosis cascades that
exacerbate neuroinflammation (66). Ruan et al (67) found that PPF protects lung tissue
by suppressing proinflammatory factor secretion via downregulation
of NLRP3. The present study also found that PPF inhibited NLRP3
activation, thereby suppressing microglial pyroptosis. In addition,
the NF-κB pathway participates in NLRP3 activation and is
associated with microglia M1 polarization; inhibiting NF-κB/NLRP3
pathway activation alleviates CIRI (68). Notably, OE-MFG-E8 in BV2 cells
inhibited NF-κB signaling and suppressed NLRP3 activation. By
contrast, following MFG-E8 silencing, the inhibitory effect of PPF
on NF-κB/NLRP3 activation was diminished and both
pyroptosis-associated marker and proinflammatory factor levels
significantly increased. This implies that PPF may exert its
neuroprotective effects by upregulating MFG-E8. In tMCAO mice,
silencing MFG-E8 decreased the suppressive impact of PPF on
NF-κB/NLRP3 activation, demonstrating that PPF blocks NF-κB/NLRP3
pathway activation by upregulating MFG-E8, thereby mitigating
pyroptosis.
PPF serves a role in modulating the NF-κB/NLRP3
pathway in post-operative cognitive dysfunction rat model (69), and PPF has been shown to activate
MFG-E8 (27). The present study
demonstrated that PPF may play a role in attenuating CIRI by
upregulating MFG-E8, thus modulating the NF-κB/NLRP3 pathway. More
importantly, the present study revealed the activation pattern of
microglia pyroptosis and polarization in CIRI, which provides a new
theoretical basis for understanding the functional imbalance of
microglia in complex pathological microenvironments and
demonstrated that PPF has a dual protective effect: It directly
acts on HT22 neurons to inhibit their apoptosis and regulate
microglia pyroptosis and polarization and block the damage to
neurons The present study provides more precise molecular targets
for the development of IS therapeutic strategies targeting
microglia.
The present study has certain limitations. Microglia
and neurons from IRI model mice were not used in the in
vitro experiments. Subsequent studies should isolate primary
cells and validate them in combination with the in vivo
tMCAO animal model to improve the reliability and translational
value of findings. In addition, LPS induces the activation of
multiple inflammatory pathways and validation experiments using
TNF-α or Phorbol 12-myristate 13-acetate/ionomycin as NF-κB
agonists should be conducted in the future. NLRP3 or GSDMD genes
should be knocked down in vitro to directly validate their
roles in the pyroptosis pathway.
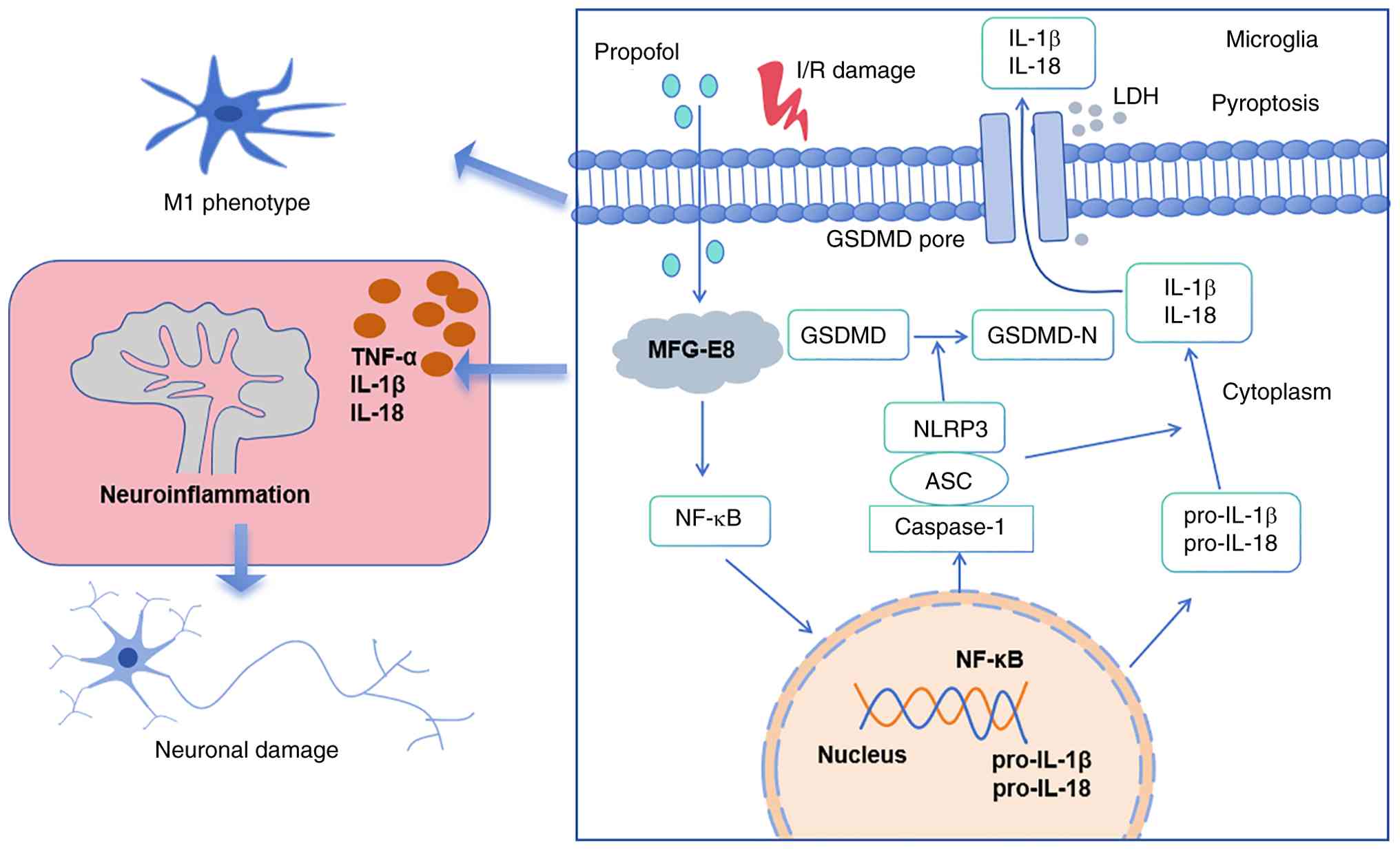
PPF suppresses NF-κB pathway activation and
downstream NLRP3-mediated pyroptosis by upregulating MFG-E8
expression, thereby alleviating CIRI (Fig. 8). PPF suppresses OGD/R-induced M1
activation and pyroptosis in BV2 cells, reduces proinflammatory
factor release and improves HT22 neuronal survival. PPF alleviated
neurological deficit in tMCAO mice, decreased infarct volume,
protected the BBB and diminished neuronal degeneration. The present
study revealed that PPF alleviates neuronal injury in CIRI by
modulating the MFG-E8/NF-κB/NLRP3 pathway, providing potential
therapeutic targets for CIRI. Future studies should investigate the
role of PPF in CIRI animal models to elucidate its neuroprotective
mechanisms.
Supplementary Data
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
SG designed the study, performed experiments,
interpreted data and edited the manuscript. YZ, GZ and ZZ
interpreted data. All authors have read and approved the final
manuscript. SG and ZZ confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
All animal experimental protocols were approved by
the First Affiliated Hospital of Zhengzhou University Animal
Welfare Committee (approval no. 2021090201; Henan, China).
Patient consent for publication
Not applicable.
Competing interests
The authors confirm that they have no competing
interests.
Acknowledgements
Not applicable.
Funding
The present study was supported by National Key Research and
Development Program Research on Prevention and Treatment of Common
and High-Incidence Diseases Key Special Project 2021 Annual Project
(grant no. 2021YFC2501100).
References
|
1
|
Feigin VL, Brainin M, Norrving B, Martins
SO, Pandian J, Lindsay P, F Grupper M and Rautalin I: World stroke
organization: Global stroke fact sheet 2025. Int J Stroke.
20:132–144. 2025. View Article : Google Scholar :
|
|
2
|
Tan KS, Pandian JD, Liu L, Toyoda K, Leung
TWH, Uchiyama S, Kuroda S, Suwanwela NC, Aaron S, Chang HM and
Venketasubramanian N: Stroke in Asia. Cerebrovasc Dis Extra.
14:58–75. 2024.PubMed/NCBI
|
|
3
|
Li XY, Kong XM, Yang CH, Cheng ZF, Lv JJ,
Guo H and Liu XH: Global, regional, and national burden of ischemic
stroke, 1990-2021: An analysis of data from the global burden of
disease study 2021. EClinicalMedicine. 75:1027582024. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Herpich F and Rincon F: Management of
acute ischemic stroke. Crit Care Med. 48:1654–1663. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Feske SK: Ischemic stroke. Am J Med.
134:1457–1464. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kapanova G, Tashenova G, Akhenbekova A,
Tokpınar A and Yılmaz S: Cerebral ischemia reperfusion injury: From
public health perspectives to mechanisms. Folia Neuropathol.
60:384–389. 2022. View Article : Google Scholar
|
|
7
|
Li M, Tang H, Li Z and Tang W: Emerging
treatment strategies for cerebral ischemia-reperfusion injury.
Neuroscience. 507:112–124. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yu L, Zhang Y, Chen Q, He Y, Zhou H, Wan H
and Yang J: Formononetin protects against inflammation associated
with cerebral ischemia-reperfusion injury in rats by targeting the
JAK2/STAT3 signaling pathway. Biomed Pharmacother. 149:1128362022.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li L, Jiang W, Yu B, Liang H, Mao S, Hu X,
Feng Y, Xu J and Chu L: Quercetin improves cerebral
ischemia/reperfusion injury by promoting microglia/macrophages M2
polarization via regulating PI3K/Akt/NF-κB signaling pathway.
Biomed Pharmacother. 168:1156532023. View Article : Google Scholar
|
|
10
|
Zhang Q, Jia M, Wang Y, Wang Q and Wu J:
Cell death mechanisms in cerebral ischemia-reperfusion injury.
Neurochem Res. 47:3525–3542. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shi M, Chen J, Liu T, Dai W, Zhou Z, Chen
L and Xie Y: Protective effects of remimazolam on cerebral
ischemia/reperfusion injury in rats by inhibiting of NLRP3
inflammasome-dependent pyroptosis. Drug Des Devel Ther. 16:413–423.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhu XN, Li J, Qiu GL, Wang L, Lu C, Guo
YG, Yang KX, Cai F, Xu T, Yuan TF and Hu J: Propofol exerts
anti-anhedonia effects via inhibiting the dopamine transporter.
Neuron. 111:1626–1636.e6. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Viderman D, Nabidollayeva F, Bilotta F and
Abdildin YG: Comparison of dexmedetomidine and propofol for
sedation in awake craniotomy: A meta-analysis. Clin Neurol
Neurosurg. 226:1076232023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Guan S, Sun L, Wang X, Huang X and Luo T:
Propofol inhibits neuroinflammation and metabolic reprogramming in
microglia in vitro and in vivo. Front Pharmacol. 14:11618102023.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu J, Ai P, Sun Y, Yang X, Li C, Liu Y,
Xia X and Zheng JC: Propofol inhibits microglial activation via
miR-106b/Pi3k/Akt axis. Front Cell Neurosci. 15:7683642021.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang D, Sun H, Hai K, Li N, Gu Y and Ma Z:
Propofol alleviates traumatic brain injury through regulating
Th17/Treg balance by activation of the AMPK/SIRT1 pathway. Toxicol
Mech Methods. 35:644–654. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sun B, Ou H, Ren F, Guan Y, Huan Y and Cai
H: Propofol protects against cerebral ischemia/reperfusion injury
by down-regulating long noncoding RNA SNHG14. ACS Chem Neurosci.
12:3002–3014. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gao YY, Tao T, Wu D, Zhuang Z, Lu Y, Wu
LY, Liu GJ, Zhou Y, Zhang DD, Wang H, et al: MFG-E8 attenuates
inflammation in subarachnoid hemorrhage by driving microglial M2
polarization. Exp Neurol. 336:1135322021. View Article : Google Scholar
|
|
19
|
Dong X, Zhang Z, Shu X, Zhuang Z, Liu P,
Liu R, Xia S, Bao X, Xu Y and Chen Y: MFG-E8 alleviates cognitive
impairments induced by chronic cerebral hypoperfusion by
phagocytosing myelin debris and promoting remyelination. Neurosci
Bull. 40:483–499. 2024. View Article : Google Scholar :
|
|
20
|
Li D, Lu W, Wang R and Ma Y: MFG-E8 in
microglial regulation: A review of basic research in the central
nervous system. J Food Sci. 90:e702352025. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cai Q, Zhao C, Xu Y, Lin H, Jia B, Huang
B, Lin S, Chen D, Jia P, Wang M, et al: Qingda granule alleviates
cerebral ischemia/reperfusion injury by inhibiting TLR4/NF-κB/NLRP3
signaling in microglia. J Ethnopharmacol. 324:1177122024.
View Article : Google Scholar
|
|
22
|
Ou Z, Zhao M, Xu Y, Wu Y, Qin L, Fang L,
Xu H and Chen J: Huangqi Guizhi Wuwu decoction promotes M2
microglia polarization and synaptic plasticity via
Sirt1/NF-κB/NLRP3 pathway in MCAO rats. Aging (Albany NY).
15:10031–10056. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xue K, Qi M, She T, Jiang Z, Zhang Y, Wang
X, Wang G, Xu L, Peng B, Liu J, et al: Argon mitigates post-stroke
neuroinflammation by regulating M1/M2 polarization and inhibiting
NF-κB/NLRP3 inflammasome signaling. J Mol Cell Biol.
14:mjac0772023. View Article : Google Scholar
|
|
24
|
Meimei C, Fei Z, Wen X, Huangwei L,
Zhenqiang H, Rongjun Y, Qiang Z, Qiuyang L, Xiaozhen L, Yuan Y, et
al: Taxus chinensis (Pilg.) Rehder fruit attenuates aging behaviors
and neuroinflammation by inhibiting microglia activation via
TLR4/NF-κB/NLRP3 pathway. J Ethnopharmacol. 337:1189432025.
View Article : Google Scholar
|
|
25
|
Ashrafizadeh M: Cell death mechanisms in
human cancers: Molecular pathways, therapy resistance and
therapeutic perspective. J Cancer Biomol Ther. 1:17–40. 2024.
View Article : Google Scholar
|
|
26
|
Lei P, Li Z, Hua Q, Song P, Gao L, Zhou L
and Cai Q: Ursolic acid alleviates neuroinflammation after
intracerebral hemorrhage by mediating microglial pyroptosis via the
NF-κB/NLRP3/GSDMD pathway. Int J Mol Sci. 24:147712023. View Article : Google Scholar
|
|
27
|
Cai X, Li Y, Zheng X, Hu R, Li Y, Xiao L
and Wang Z: Propofol suppresses microglial phagocytosis through the
downregulation of MFG-E8. J Neuroinflammation. 18:182021.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Shi X, Cai X, Di W, Li J, Xu X, Zhang A,
Qi W, Zhou Z and Fang Y: MFG-E8 selectively inhibited Aβ-induced
microglial M1 polarization via NF-κB and PI3K-Akt pathways. Mol
Neurobiol. 54:7777–7788. 2017. View Article : Google Scholar
|
|
29
|
Xu X, Gao W, Li L, Hao J, Yang B, Wang T,
Li L, Bai X, Li F, Ren H, et al: Annexin A1 protects against
cerebral ischemia-reperfusion injury by modulating
microglia/macrophage polarization via FPR2/ALX-dependent AMPK-mTOR
pathway. J Neuroinflammation. 18:1192021. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tao W, Zhang X, Ding J, Yu S, Ge P, Han J,
Luo X, Cui W and Chen J: The effect of propofol on hypoxia- and
TNF-α-mediated BDNF/TrkB pathway dysregulation in primary rat
hippocampal neurons. CNS Neurosci Ther. 28:761–774. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Beaulieu J, Costa G, Renaud J, Moitié A,
Glémet H, Sergi D and Martinoli MG: The neuroinflammatory and
neurotoxic potential of palmitic acid is mitigated by oleic acid in
microglial cells and microglial-neuronal co-cultures. Mol
Neurobiol. 58:3000–3014. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xian M, Cai J, Zheng K, Liu Q, Liu Y, Lin
H, Liang S and Wang S: Aloe-emodin prevents nerve injury and
neuroinflammation caused by ischemic stroke via the PI3K/AKT/mTOR
and NF-κB pathway. Food Funct. 12:8056–8067. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhao R, Zhao D, Zhu X, Li F, Xiong P, Li S
and Liu J: The influence of miR-3149 on the malignancy progression
of gastric cancer by negatively regulating CEACAM5. J Cancer Biomol
Ther. 1:1–10. 2024. View Article : Google Scholar
|
|
34
|
Liu J, Zhang X, Guo J, Zhang Y, Fan J, Liu
J, Chen J, Jiang J, Yu B, Zhang K and Zhou B: Ursolic acid
ameliorates cerebral ischemia-reperfusion injury by inhibiting
NF-κB/NLRP3-mediated microglia pyroptosis and neuroinf lammation.
Front Pharmacol. 16:16221312025. View Article : Google Scholar
|
|
35
|
Kong L, Li W, Chang E, Wang W, Shen N, Xu
X, Wang X, Zhang Y, Sun W, Hu W, et al: mtDNA-STING axis mediates
microglial polarization via IRF3/NF-κB signaling after ischemic
stroke. Front Immunol. 13:8609772022. View Article : Google Scholar
|
|
36
|
Zhang Q, Wang L, Chen B, Zhuo Q, Bao C and
Lin L: Propofol inhibits NF-κB activation to ameliorate airway
inflammation in ovalbumin (OVA)-induced allergic asthma mice. Int
Immunopharmacol. 51:158–164. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Gao YY, Zhang ZH, Zhuang Z, Lu Y, Wu LY,
Ye ZN, Zhang XS, Chen CL, Li W and Hang CH: Recombinant milk fat
globule-EGF factor-8 reduces apoptosis via integrin β3/FAK/PI3K/AKT
signaling pathway in rats after traumatic brain injury. Cell Death
Dis. 9:8452018. View Article : Google Scholar
|
|
38
|
Longa EZ, Weinstein PR, Carlson S and
Cummins R: Reversible middle cerebral artery occlusion without
craniectomy in rats. Stroke. 20:84–91. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lu J, Li Z, Zhao Q, Liu D and Mei YA:
Neuritin improves the neurological functional recovery after
experimental intracerebral hemorrhage in mice. Neurobiol Dis.
156:1054072021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Pandarathodiyil AK, Ramanathan A, Garg R,
Doss JG, Rahman FBA, Ghani WMN and Vijayan SP: Lactate
dehydrogenase: The beacon of hope? J Pharm Bioallied Sci. 14(Suppl
1): S1090–S1092. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhang L, Dai X, Li D, Wu J, Gao S, Song F,
Liu L, Zhou Y, Liu D and Mei W: MFG-E8 ameliorates nerve
injury-induced neuropathic pain by regulating microglial
polarization and neuroinflammation via integrin β3/SOCS3/STAT3
pathway in mice. J Neuroimmune Pharmacol. 19:492024. View Article : Google Scholar
|
|
42
|
Zhong Y, Gu L, Ye Y, Zhu H, Pu B, Wang J,
Li Y, Qiu S, Xiong X and Jian Z: JAK2/STAT3 axis intermediates
microglia/macrophage polarization during cerebral
ischemia/reperfusion injury. Neuroscience. 496:119–128. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Liao J, Wei M, Wang J, Zeng J, Liu D, Du
Q, Ge J and Mei Z: Naotaifang formula attenuates OGD/R-induced
inflammation and ferroptosis by regulating microglial M1/M2
polarization through BMP6/SMADs signaling pathway. Biomed
Pharmacother. 167:1154652023. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Chen S, Pan J, Gong Z, Wu M, Zhang X, Chen
H, Yang D, Qi S, Peng Y and Shen J: Hypochlorous acid derived from
microglial myeloperoxidase could mediate high-mobility group box 1
release from neurons to amplify brain damage in cerebral
ischemia-reperfusion injury. J Neuroinflammation. 21:702024.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lan Z, Tan F, He J, Liu J, Lu M, Hu Z,
Zhuo Y, Liu J, Tang X, Jiang Z, et al: Curcumin-primed olfactory
mucosa-derived mesenchymal stem cells mitigate cerebral
ischemia/reperfusion injury-induced neuronal PANoptosis by
modulating microglial polarization. Phytomedicine. 129:1556352024.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Gong L, Liang J, Xie L, Zhang Z, Mei Z and
Zhang W: Metabolic reprogramming in gliocyte post-cerebral
ischemia/reperfusion: From pathophysiology to therapeutic
potential. Curr Neuropharmacol. 22:1672–1696. 2024. View Article : Google Scholar
|
|
47
|
Gao J, Su G, Liu J, Song J, Chen W, Chai
M, Xie X, Wang M, Liu J and Zhang Z: A novel compound ligusticum
cycloprolactam alleviates neuroinflammation after ischemic stroke
via the FPR1/NLRP3 signaling axis. CNS Neurosci Ther.
30:e701582024. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Zeng L, Hu S, Zeng L, Chen R, Li H, Yu J
and Yang H: Animal models of ischemic stroke with different forms
of middle cerebral artery occlusion. Brain Sci. 13:10072023.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Luo L, Liu M, Fan Y, Zhang J, Liu L, Li Y,
Zhang Q, Xie H, Jiang C, Wu J, et al: Intermittent theta-burst
stimulation improves motor function by inhibiting neuronal
pyroptosis and regulating microglial polarization via
TLR4/NFκB/NLRP3 signaling pathway in cerebral ischemic mice. J
Neuroinflammation. 19:1412022. View Article : Google Scholar
|
|
50
|
Xu H, Ma Y, Shao L, Fu Y, Luo B, Xia C and
Min X: Pan-cancer analysis of DLAT reveals it as a prognostic
biomarker involved in immune infiltration of liver hepatocellular
carcinoma. J Cancer. 16:2167–2180. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ermine CM, Bivard A, Parsons MW and Baron
JC: The ischemic penumbra: From concept to reality. Int J Stroke.
16:497–509. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Biagioni F, Mastroiacovo F, Lenzi P,
Puglisi-Allegra S, Busceti CL, Ryskalin L, Ferese R, Bucci D, Frati
A, Nicoletti F and Fornai F: The autophagy-related organelle
autophagoproteasome is suppressed within ischemic penumbra. Int J
Mol Sci. 22:103642021. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Ji W, Ren Y, Wei X, Ding X, Dong Y and
Yuan B: Ischemic stroke protected by ISO-1 inhibition of apoptosis
via mitochondrial pathway. Sci Rep. 13:27882023. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Yang SH and Liu R: Four decades of
ischemic penumbra and its implication for ischemic stroke. Transl
Stroke Res. 12:937–945. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Candelario-Jalil E, Dijkhuizen RM and
Magnus T: Neuroinflammation, stroke, blood-brain barrier
dysfunction, and imaging modalities. Stroke. 53:1473–1486. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Anthony S, Cabantan D, Monsour M and
Borlongan CV: Neuroinflammation, stem cells, and stroke. Stroke.
53:1460–1472. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Wang H, Zhang S, Xie L, Zhong Z and Yan F:
Neuroinflammation and peripheral immunity: Focus on ischemic
stroke. Int Immunopharmacol. 120:1103322023. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Dmitrieva VG, Povarova OV, Skvortsova VI,
Limborska SA, Myasoedov NF and Dergunova LV: Semax and Pro-Gly-Pro
activate the transcription of neurotrophins and their receptor
genes after cerebral ischemia. Cell Mol Neurobiol. 30:71–79. 2010.
View Article : Google Scholar
|
|
59
|
Liu K, Li L, Liu Z, Li G, Wu Y, Jiang X,
Wang M, Chang Y, Jiang T, Luo J, et al: Acute administration of
metformin protects against neuronal apoptosis induced by cerebral
ischemia-reperfusion injury via regulation of the AMPK/CREB/BDNF
pathway. Front Pharmacol. 13:8326112022. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Guo L, Huang Z, Huang L, Liang J, Wang P,
Zhao L and Shi Y: Surface-modified engineered exosomes attenuated
cerebral ischemia/reperfusion injury by targeting the delivery of
quercetin towards impaired neurons. J Nanobiotechnology.
19:1412021. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Li D, Rongchun W, Lu W and Ma Y: Exploring
the potential of MFG-E8 in neurodegenerative diseases. Crit Rev
Food Sci Nutr. 65:5587–5601. 2025. View Article : Google Scholar
|
|
62
|
Zhao M, Wu Y, Huang L, Wang J and Zhang A:
The umbilical cord blood exosome MFG-E8 alleviates hypoxic-ischemic
encephalopathy brain injury in neonatal rats by restoring autophagy
flux and inhibiting ferroptosis through GSK3β/β-catenin signaling.
Regen Ther. 30:321–332. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Cheyuo C, Jacob A, Wu R, Zhou M, Qi L,
Dong W, Ji Y, Chaung WW, Wang H, Nicastro J, et al: Recombinant
human MFG-E8 attenuates cerebral ischemic injury: Its role in
anti-inflammation and anti-apoptosis. Neuropharmacology.
62:890–900. 2012. View Article : Google Scholar
|
|
64
|
Han YH, Liu XD, Jin MH, Sun HN and Kwon T:
Role of NLRP3 inflammasome-mediated neuronal pyroptosis and
neuroinflammation in neurodegenerative diseases. Inflamm Res.
72:1839–1859. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Fu J and Wu H: Structural mechanisms of
NLRP3 inflammasome assembly and activation. Annu Rev Immunol.
41:301–316. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Li X, Yao M, Li L, Ma H, Sun Y, Lu X, Jing
W and Nie S: Aloe-emodin alleviates cerebral ischemia-reperfusion
injury by regulating microglial polarization and pyroptosis through
inhibition of NLRP3 inflammasome activation. Phytomedicine.
129:1555782024. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ruan H, Li W, Wang J, Chen G, Xia B, Wang
Z and Zhang M: Propofol alleviates ventilator-induced lung injury
through regulating the Nrf2/NLRP3 signaling pathway. Exp Mol
Pathol. 114:1044272020. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Shen G, Lou C, Li Q, Zhao B, Luo Y, Wu F,
Jiao D, Fang M and Geng Y: Edaravone dexborneol alleviates cerebral
ischemia-reperfusion injury through NF-κB/NLRP3 signal pathway.
Anat Rec (Hoboken). 307:372–384. 2024. View Article : Google Scholar
|
|
69
|
Liu PF, Gao T, Li TZ, Yang YT, Xu YX, Xu
ZP and Mi WD: Repeated propofol exposure-induced neuronal damage
and cognitive impairment in aged rats by activation of NF-κB
pathway and NLRP3 inflammasome. Neurosci Lett. 740:1354612021.
View Article : Google Scholar
|