Traditional cell lines and animal model systems have
demonstrated notable success in biomedical research throughout the
late twentieth and early twenty-first centuries, advancing the
present understanding of cellular signaling pathways, identifying
potential drug targets, and guiding therapeutic development for
conditions including cancer and infectious diseases. The widespread
adoption of these model systems in modern biomedical research
validates their substantial contributions. The traditional pathway
for studying disease mechanisms in animal models involves initial
genetic screening in invertebrates, followed by verification of
evolutionary conservation in mammalian systems, and ultimate
translation to human clinical applications. This systematic
approach has enabled a detailed mechanistic understanding of
numerous human diseases.
Acute liver failure (ALF) represents a rare and
complex clinical condition, with liver transplantation serving as
the most effective treatment. However, limited donor availability
restricts its clinical application (1-3).
Novel therapeutic approaches, including hepatocellular cell
transplantation, bioartificial liver support systems and stem cell
transplantation, require evaluation in animal models before
clinical implementation (4-6).
Consequently, animal models constitute a fundamental component in
therapeutic development.
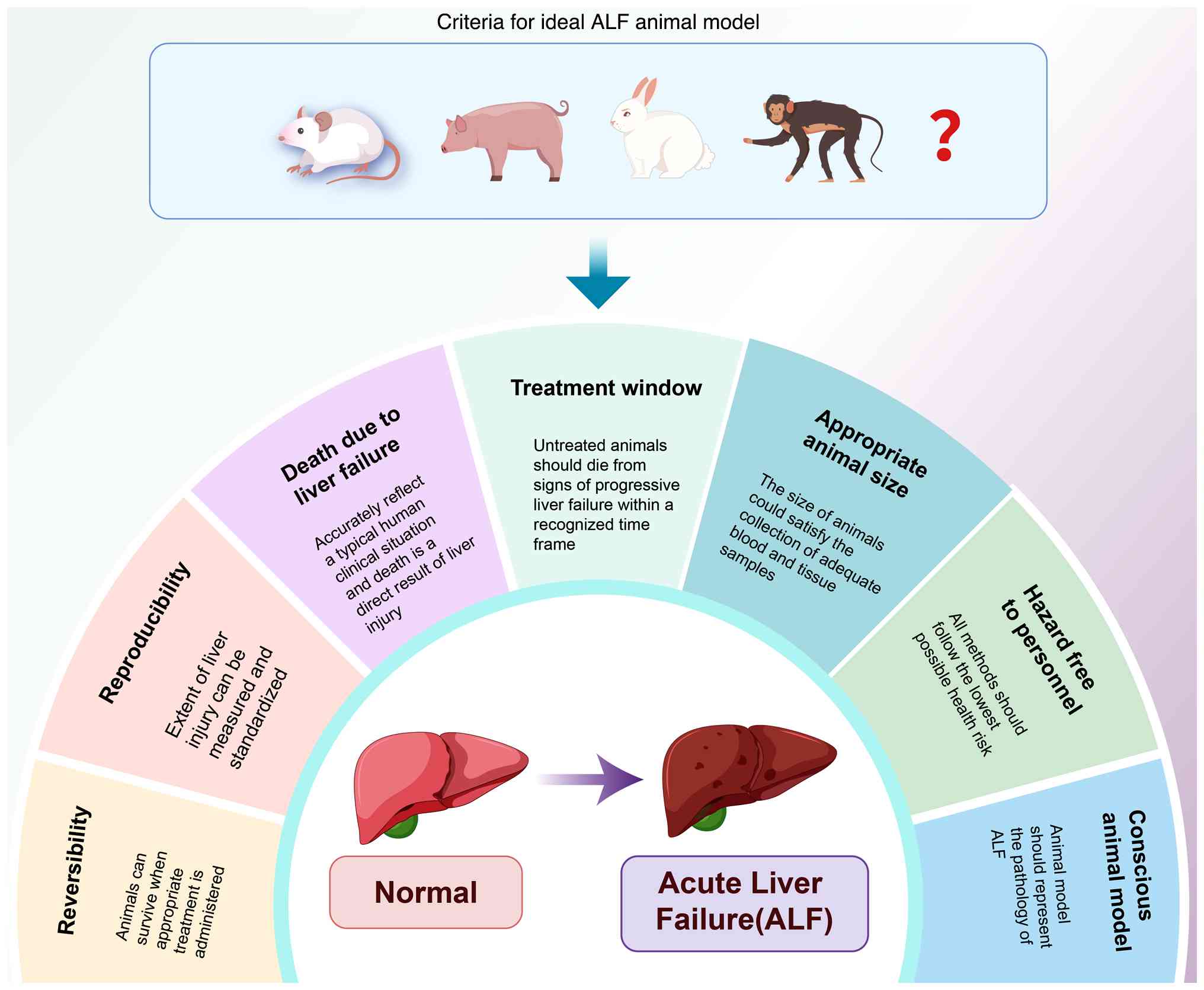
The ideal ALF model incorporates the following seven
criteria: i) Reversibility: Some animals can survive the process if
appropriate treatment is administered; ii) reproducibility: Death
occurs at recognized time intervals and the extent of liver injury
can be measured and standardized; iii) death due to liver failure:
Complications arising from the injury need to accurately reflect a
typical human clinical situation and death is a direct result of
liver injury; iv) treatment window: Untreated animals should die
from signs of progressive liver failure within a recognized time
frame; v) appropriate animal size: Animals used need to be of a
size that permits the collection of adequate blood and tissue
samples during treatment; vi) minimal hazard to personnel: All
methods used should represent the lowest possible health risk; and
vii) conscious animal model: The use of a conscious animal model to
assess the development of hepatic encephalopathy, which is an
important component of ALF pathology (Fig. 1) (7).
The mouse serves as an effective experimental model,
with developmental processes closely resembling human biology.
Despite having a less comprehensive metabolic system compared with
larger animal models, mouse models have gained widespread adoption
and demonstrated notable utility (8). The C57BL/6J mouse strain from The
Jackson Laboratory has emerged as a predominant choice in medical
research, exhibiting gene regulation patterns similar to humans and
strong concordance in immune and inflammatory pathways, thus
providing a representative model of human disease (9). However, research has revealed
substantial variations in drug and chemical dosages used in
pharmacologically-induced and chemically-induced ALF models using
C57BL/6J mice, impeding standardized research under consistent
pathologic characteristics and hindering progress in ALF
therapeutic development.
The present review examines C57BL/6J-derived acute
liver injury (ALI)/ALF models induced by pharmacological and
chemical injuries that demonstrate potential in modeling ALF onset,
development and disease progression. While previous reviews have
comprehensively described various model systems (10,11), the present analysis focuses on
distinguishing between ALI and ALF models from existing research to
establish uniform standards for drug/chemical usage in animal model
development. The present review examines variations in drug and
chemical applications for mouse models, assesses dosage
similarities and limitations between animal models and human
conditions, and highlights current dosage recommendations to
promote standardized approaches in ALF pathophysiology
research.
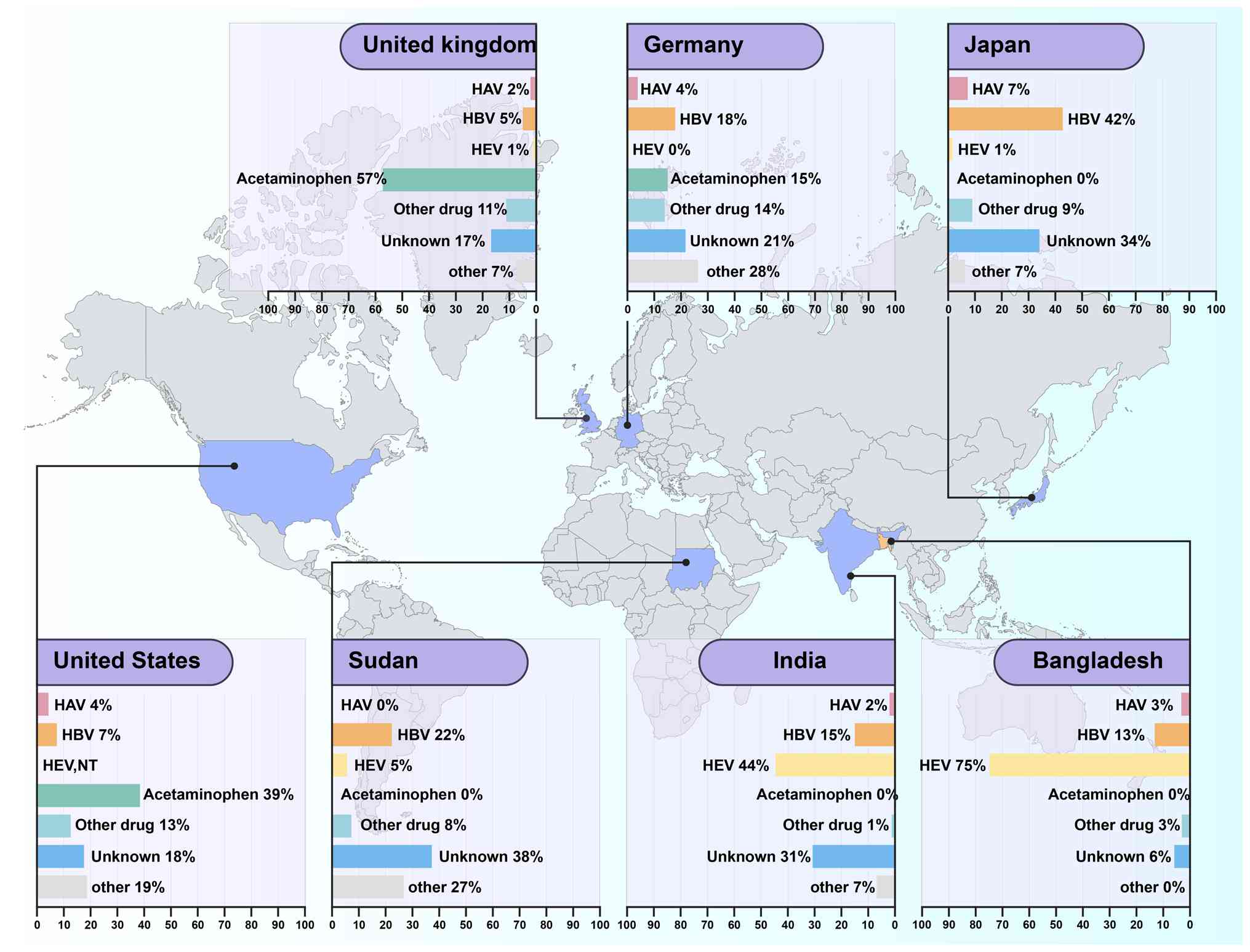
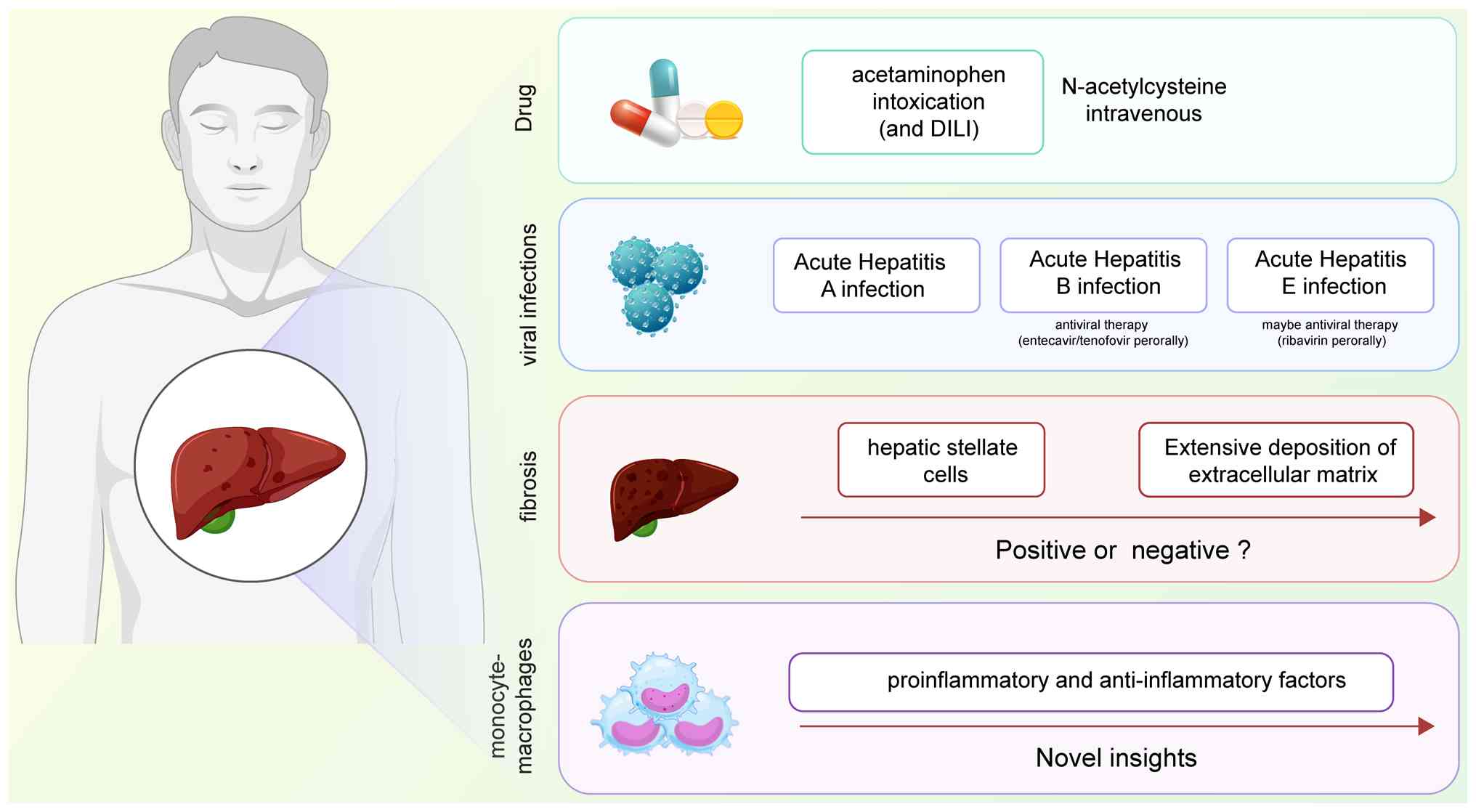
ALF primarily results from poisoning, viral
infection, autoimmune deficiency and hereditary diseases,
predominantly affecting healthy adults that are ~30 years of age.
The clinical manifestations include hepatic insufficiency, liver
biochemical abnormalities and coagulation dysfunction (21). In developing nations, acute viral
hepatitis remains the primary cause of ALF (20), whilst hepatitis B virus (HBV)
infection constitutes the most common cause in Asian countries and
the Mediterranean region (21,22). However, with increased HBV
vaccination rates and declining HBV carrier populations,
pharmacologic injury-induced ALF is becoming more prevalent
(23), particularly in Western
countries (Fig. 3).
ALF develops from rapid liver function loss
triggered by various acute injuries, including hepatotoxic drugs,
immune-mediated attacks or viral infection-induced rapid
hepatocellular necrosis (24,25). The condition manifests when
hepatocellular death exceeds the regenerative capacity of the
liver, primarily through necrosis, apoptosis and necrotizing
apoptosis (26). Apoptotic
hepatocytes interact with Kupffer cells (KCs), newly infiltrated
bone marrow-derived macrophages and hepatic stellate cells (HSCs).
However, extensive hepatocyte death overwhelms the clearance
capacity, preventing liver function restoration (27). The inflammatory response
distinguishes necrotic from apoptotic cell death. Necrotic cell
rupture triggers inflammation through the release of intracellular
contents, notably the NLR family pyrin domain containing 3 (NLRP3)
inflammasome. This well-established human inflammasome, when
aberrantly activated, associates with various ALF types and induces
different programmed cell death forms (Fig. 4) (28).
The predominant mechanism of ALF cell death induced
by viral infection involves the activation of death receptors.
Blood samples from patients with ALF showed significantly elevated
levels of death receptors and ligands, including CD95L, TNF-α and
TNF receptor (29-32). Studies using in
vitro-constructed TNF-α and anti-CD95 mouse models have
demonstrated toxicity patterns similar to those observed in
patients with ALF (33,34). Additionally, HCV-infected human
livers demonstrated increased TRAIL receptor expression and
enhanced susceptibility to TRAIL-induced apoptosis (31).
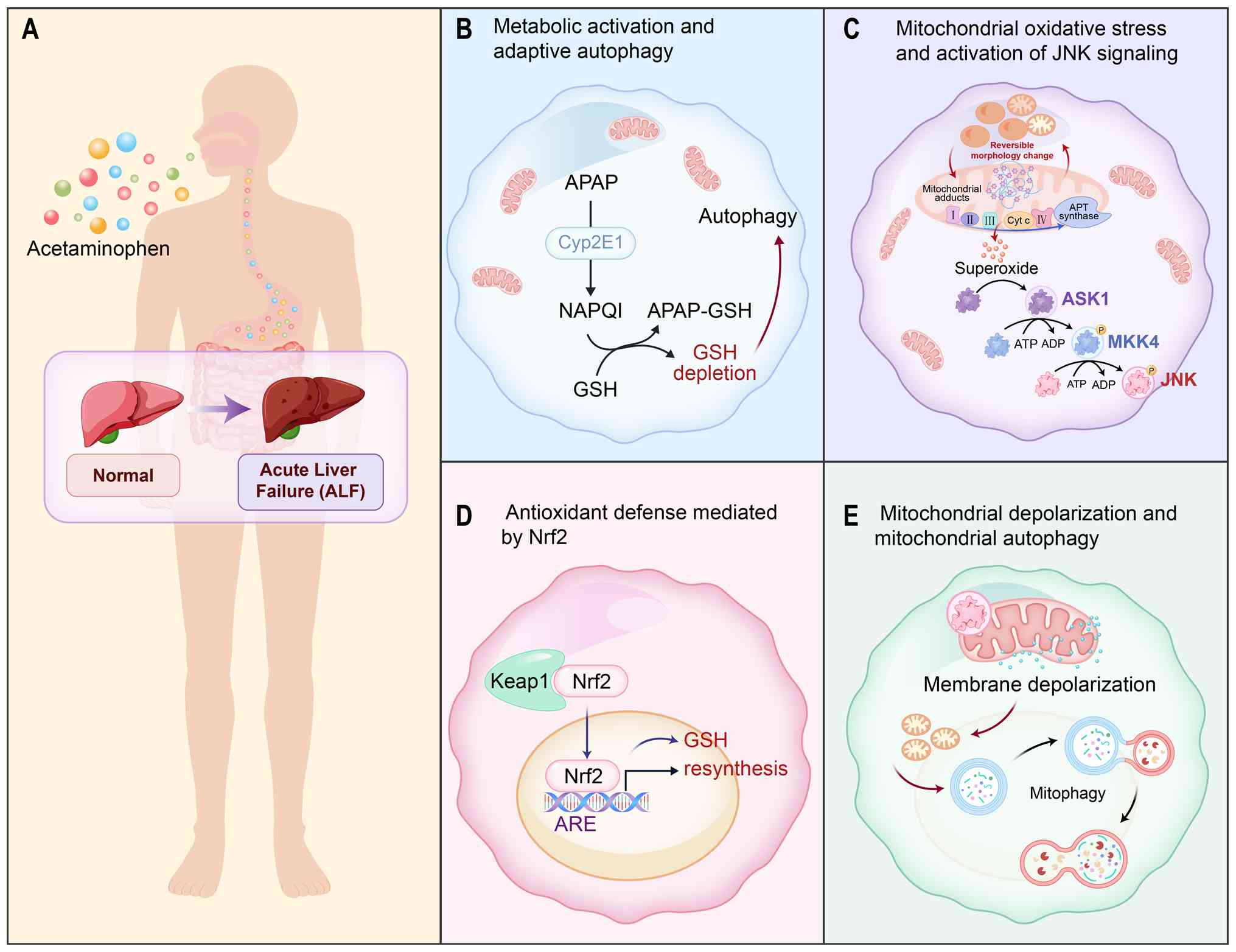
Unlike viral infection, drug-induced liver injury
primarily involves signaling pathways originating from mitochondria
(35). The mechanism of
acetaminophen (APAP)-induced ALF cell death primarily involves
metabolic and adaptive mechanisms regulating toxicity in the
cytochrome P450 system (36).
Mitochondrial autophagy, a selective form of autophagy, eliminates
damaged mitochondria through the interaction between mitochondrial
and autophagic mechanisms. This process occurs through
ubiquitin-dependent or ubiquitin-independent pathways,
characterized primarily by PINK/Parkin-mediated and light chain 3
interacting region-domain receptor-mediated pathways, respectively
(37).
Mitochondrial autophagy serves to attenuate local
inflammatory responses and tissue necrosis by eliminating damaged
or excess mitochondria, maintaining mitochondrial homeostasis, and
inhibiting pro-inflammatory factor secretion triggered by
inflammatory vesicles such as NLRP3 (38,39). During ALF onset, injured
hepatocyte mitochondria release damage-associated molecular
patterns and secrete inflammatory factors IL-1β and TNF-α. This
leads to increased reactive oxygen species (ROS) production and ATP
depletion, followed by mTOR activity inhibition and enhanced
autophagic flux. Autophagic vesicles accumulate around necrotic
foci, limiting necrosis expansion into normal hepatic areas.
Mitochondrial autophagy proteins, activated by ectopic or
ubiquitinated mitochondrial proteins, facilitate damaged
mitochondria removal, ROS reduction, inflammation decrease and
hepatocyte repair (40).
However, insufficient activation of mitochondrial autophagy during
disease onset impairs timely removal of damaged mitochondria and
necrotic hepatocyte repair, resulting in increased local and
systemic inflammation and ultimately irreparable massive liver
parenchyma necrosis (41). In
APAP-induced ALF, Parkin protein inhibition/knockdown significantly
reduces mitochondrial autophagy, leading to NLRP3 inflammatory
vesicle accumulation and increased APAP-induced hepatotoxicity
(42). By contrast,
mitochondrial autophagy activation removes ROS-damaged
mitochondria, inhibits IL-1β release and alleviates APAP-induced
ALF symptoms (43). This
evidence demonstrates the crucial role of mitochondrial autophagy
in liver protection and homeostasis maintenance, while its
dysfunction leads to damaged mitochondria accumulation, disrupted
mitochondrial homeostasis and ALF progression (Fig. 5) (41).
Previous evidence has suggested that extensive
extracellular matrix (ECM) deposition and scarring from persistent
HSC activation impairs tissue regeneration by hepatocytes (44). However, another study showed that
preventing fibrosis through activated HSC depletion in ALF mouse
models results in more severe liver injury and reduced survival
(45). Fibrosis is an intrinsic
injury response that maintains organ integrity during extensive
necrosis or apoptosis, suggesting its beneficial role during acute
tissue injury (46). Previous
studies have proposed that ALF-related fibrosis may possess a
protective function. The acute production of collagen and fibrosis
onset in ALF may represent an intentional physiological process
(47-49). Further investigation is needed to
determine whether fibrosis formation could serve as an intervention
target in ALF animal models.
From a clinical perspective, the primary cause of
mortality in ALF is systemic inflammatory response syndrome, which
results from excessive inflammatory responses, leading to
concentrative renal anemia and multi-organ failure (50). Disruptions in the innate immune
system constitute the principal mechanism leading to ALF (51). Notably, monocyte-macrophages,
serving as crucial effector cells in both natural and intrinsic
immunity, generate numerous proinflammatory and anti-inflammatory
factors, playing a central role in ALF initiation and progression.
As hepatic macrophages, both KCs and monocyte-derived macrophages
regulate the hepatic inflammatory response. During ALF onset,
hepatic KCs become activated by injury-associated molecular
patterns released from damaged hepatocyte mitochondria,
subsequently activating the NF-κB signaling pathway to produce
inflammatory cytokines and reactive oxygen clusters (52). This leads to a substantial
increase in hepatic KCs, partially through KC proliferation and
division, and partially through circulating monocytes that migrate
to injured hepatic areas via monocyte chemoattractant protein-1
receptor signaling. These monocytes differentiate into KCs,
secreting additional inflammatory mediators and promoting
neutrophil infiltration, thereby intensifying local inflammation
and tissue necrosis (53). The
resulting local and systemic inflammatory responses rapidly
amplify, ultimately causing irreversible inflammation and tissue
death. This evidence indicated that KCs represent a potential
therapeutic target for ALF, where timely inhibition of KC
activation may reduce liver injury and enhance liver regeneration.
This area warrants further investigation, given its significance in
ALF progression and outcomes (54,55).
Since the recognition of Mendel's work in the early
20th century, researchers have investigated numerous organisms in
laboratories worldwide (56).
However, relatively few have emerged as 'model organisms', which
typically exhibit characteristics such as robustness, brief growth
cycles, rapid production of numerous offspring and cost-effective
laboratory cultivation (57).
Established model organisms including Saccharomyces
cerevisiae (brewer's yeast), Drosophila melanogaster
(fruit fly) and mice possess extensive research histories and have
notably influenced various biological fields. Subsequently, the
development of novel model systems, such as nematodes, zebrafish
and African clawed toads, has further expanded scientific
understanding. For example, Caenorhabditis elegans
demonstrates rapid growth and maintains its cell lineage, enabling
precise analysis of cell fate determination mechanisms (58). Zebrafish larvae are particularly
advantageous for studying vertebrate organ development due to their
transparent bodies (59,60). The African clawed toad exhibits
regenerative capabilities exclusively during its tadpole stage
(through developmental stage, 50-54), losing this ability upon
developing into a juvenile frog (stages, 56-66). The sole exception
is the optic nerve, which retains lifelong regenerative capacity,
offering valuable insights for regeneration research (61).
The most commonly used model organisms currently
include non-mammalian models (brewer's yeast, C. elegans,
Drosophila melanogaster and Danio rerio), and
mammalian models (Mus musculus). Patient-derived xenograft
models and cell lines are also used in cancer research. In
addition, in situ tissues and stem cell-derived organoids
provide a new platform for basic research, each with unique
advantages and limitations.
However, these non-mammalian species exhibit notable
differences from humans in their growth and development processes,
including their physiological characteristics, genomes and disease
pathogenesis.
Among mammalian models, mice represent closer
analogues to human genetics, development and disease compared with
Drosophila or worms. The mouse serves as the preferred animal model
for biomedical research, enabling researchers to identify
interventions for neurological disorders (69,70), validate initial stem cell origin
hypotheses (71,72) and advance emerging fields such as
'fertility preservation' (73)
and reversal of epigenetic changes induced by assisted reproduction
techniques (74). Researchers
selected the mouse as a model organism for three primary reasons:
i) The mouse genome is more thoroughly characterized than other
animal models, with sophisticated gene editing tools and germline
totipotent embryonic stem cells, facilitating genetic modification
through evolving tools for genetics, genotyping and phenotyping;
ii) mouse populations can be readily expanded, enabling large-scale
experiments to derive statistically significant conclusions
regarding developmental and physiological mechanisms and systemic
properties; and iii) feeding management and environmental
conditions can be precisely controlled, allowing systematic
multi-omics studies at molecular, cellular and organ levels
(75-100). Although studies using identical
mouse models may yield different findings, such variations often
result from differences in technical analysis methods, a
reproducibility challenge also present in human studies. This
necessitates enhanced optimization of husbandry environments,
stringent verification of mouse strain identity and standardization
of basic experimental conditions, which, with appropriate mouse
models, can provide accurate insights into human conditions,
facilitate biological principle discovery, prioritize human studies
and validate related hypotheses.
Prior to the emergence of organoid technology,
various approaches to simulate human organ biology have been
explored, including 2D human stem cell differentiation with or
without 3D matrices, bio-3D printing of human cells and cell
culture in microfluidic devices ('organ-on-a-chip') (101). Organoid generation from human
patient-derived cells or human-induced pluripotent stem cells has
demonstrated notable value in studying mechanisms related to
differentiation, morphogenesis, pathogenesis and drug action, while
also facilitating biomarker discovery for diseases at cellular and
tissue levels (102).
However, organoids, being closed structures, lack
tissue-tissue interfaces, vascular flow, circulating immune cells
and physiologically relevant mechanical cues, limiting their
capacity to fully capture organ-level responses or study drug
effects under pharmacologically relevant conditions dependent on
dynamic drug pharmacokinetic exposure profiles. The incorporation
of self-organizing capillary networks in 3D organoid cultures
(103) and immune cell addition
to surrounding ECM gel may address some limitations (104). Nevertheless, substantial
constraints persist, including the inability to control vascular
structure and flow dynamics, and challenges in understanding
chemical, inflammatory molecule, drug and immune cell movement
within living vascularized organs. Due to their closed structure
surrounded by dense ECM, organoids present difficulties in
measuring nutrient, chemical or drug transport and uptake in
epithelial tissues, sampling inner lumen contents, maintaining
microbiome co-cultures and integrating sensors for on-line
functional measurements (105).
Previous research indicates that organoids
demonstrate notable potential in complementing existing model
systems and advancing basic biology, medical research and drug
discovery within physiologically relevant human environments
(101). However, organoid
technology remains in its early developmental stages compared with
established cell lines and animal models, with numerous challenges
yet to be addressed.
The utilization of distinct animal models, 2D human
cell lines and 3D organoids throughout the past three decades has
substantially enhanced the understanding of disease pathogenesis,
while simultaneously revealing the limitations of these systems in
replicating human pathophysiology. The mouse model maintains its
position as the predominant model organism for disease research,
with particular validation coming from disease-related genes and
pathological features initially discovered in mouse studies being
subsequently confirmed in humans (106,107), establishing the mouse model as
the preferred animal model for biomedical research.
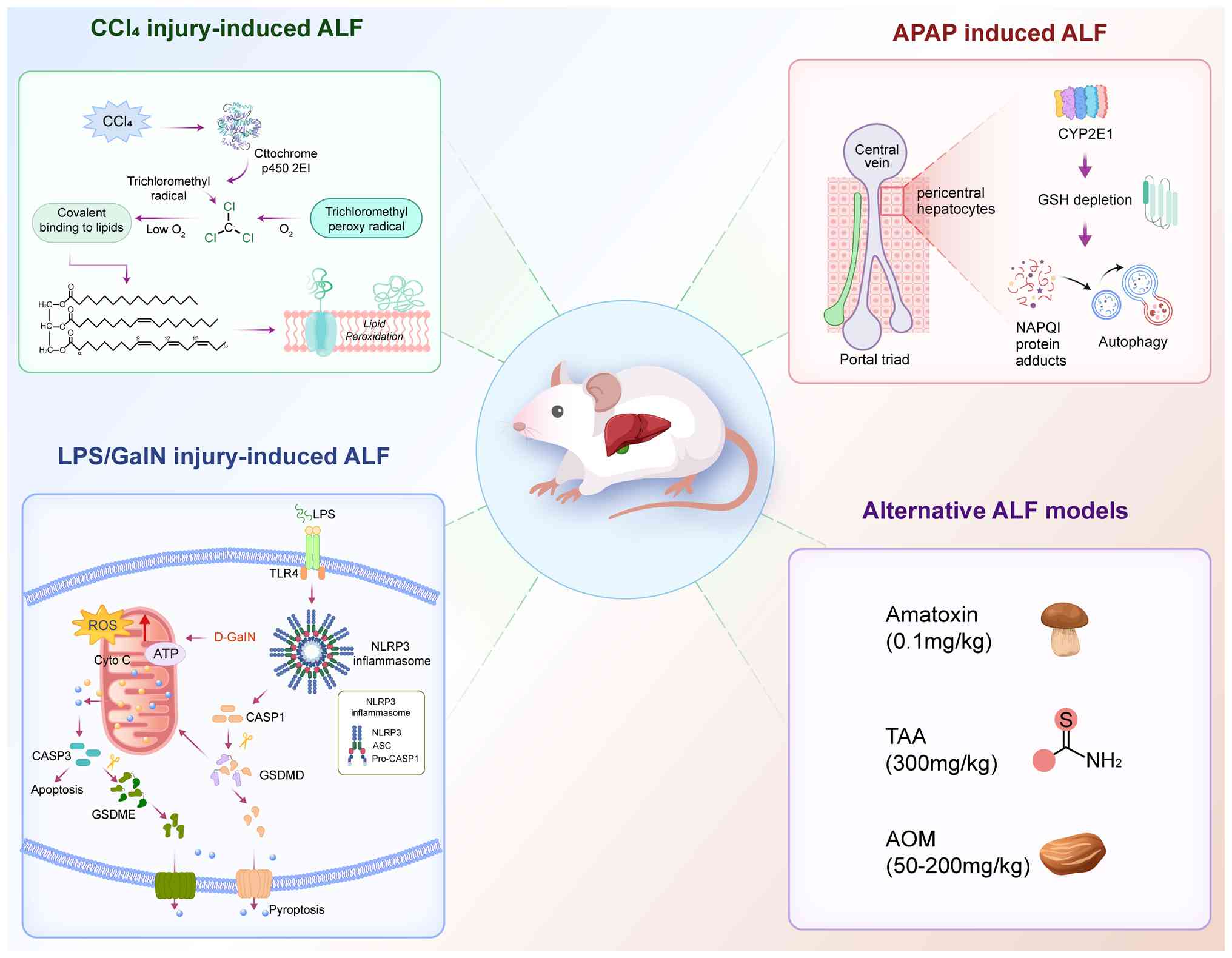
An ideal animal model is essential for disease
research. Currently, to more accurately replicate the
characteristics of ALF in vivo, the primary construction
methods are categorized into APAP-based pharmacological injury and
chemical injury-based fibrosis models (Fig. 6).
For ALI modeling in C57BL/6J mice, APAP doses
typically range from 300 to 600 mg/kg, with 600-750 mg/kg
considered the lethal dose for ALF modeling. However, a
standardized dose for ALI/ALF modeling remains undefined, with
researchers selecting different APAP doses for various mechanistic
studies. APAP overdose triggers excessive production of
reactive-free radicals and n-acetyl-p-benzoquinone imine (NAPQI)
(113). NAPQI rapidly depletes
cellular glutathione and forms complexes with biologically active
molecules, inducing oxidative stress, causing mitochondrial damage
and ultimately resulting in hepatocellular injury (114).
Herbal extracts demonstrate particular promise as
therapeutic agents for ALI/liver failure, functioning through
antioxidants. Platanoside (PTS), isolated from American sycamore
(Platanus occidentalis), represents a potential novel
tetramolecular phytopharmaceutical antibiotic effective against
drug-resistant infectious diseases and exhibits antioxidant
properties. A previous study demonstrated reduced inducible nitric
oxide synthase expression and c-Jun-N terminal kinase (JNK)
activation when APAP-overdosed mice (300 mg/kg) received PTS
treatment (115).
Atractylenolide I (AO-I), a phytochemical derived from
Atractylodes macrocephala Koidz., demonstrates established
antioxidant properties. Li et al (116) administered AO-I to C57BL/6 mice
following 500 mg/kg APAP-induced hepatotoxicity, revealing that
AO-I provided protection against APAP-induced hepatotoxicity
through the TLR4/MAPKs/NF-κB pathway (117). Research utilizing an ALF mouse
model constructed with high-dose APAP (700 mg/kg) demonstrated that
mannose-binding lectin regulates CYP2E1 expression through the
ROS-dependent JNK/SP1 pathway and mitigates APAP-induced
hepatotoxicity (116).
Mitochondrial damage plays a crucial role in
APAP-induced hepatocyte necrosis and liver dysfunction. Research
demonstrates that APAP activates mitophagy, evidenced by increased
Parkin translocation to mitochondria and ubiquitination of
mitochondrial proteins, along with concurrent sequestration of
damaged mitochondria in autophagosomes and degradation of
mitochondrial proteins in primary hepatocytes from mouse livers
(118). In studies examining
the relationship between mitochondrial autophagy and ALF
occurrence, researchers predominantly established ALI models
through intraperitoneal injection of 500 mg/kg APAP for 24 h
(119-124). The initial implementation of
the APAP-ALI model was conducted by Ni et al (125), who utilized the 500 mg/kg APAP
C57BL/6J mouse model in 2012 and demonstrated that treatment with
the autophagy inducer rapamycin inhibited APAP-induced
hepatotoxicity. Subsequently, in 2015, the authors identified
distinct responses to mitochondrial autophagy and hepatic injury in
mice induced by chronic deletion of Parkin and acute knockdown of
APAP using the same model (126). In 2019, their research revealed
that PINK1 and dual deletion of Parkin compromised hepatic
mitochondrial autophagy and intensified APAP-induced hepatic injury
in mice (127). Studies
investigating autophagy through amelioration of inflammatory
response and apoptosis-related research typically employ a lower
dose of 300 mg/kg APAP (42,128). Chen et al (129) administered 400 mg/kg APAP
coenzyme Q10 to C57BL/6J mice to activate mitochondrial autophagy
and prevent APAP-induced hepatic injury. APAP doses >700 mg/kg
are considered lethal, with C57BL/6J mice survival rates <60 h,
paralleling ALF disease progression (130).
The ER facilitates a specialized intracellular
environment for protein processing, folding and sorting for
intracellular and extracellular transport. ER stress (ERS) occurs
when the ER structure sustains damage or when protein synthesis
exceeds the functional capacity of the ER (131). Transient ERS serves as a
protective mechanism for normal cellular function under stress.
However, prolonged ERS may trigger apoptosis, potentially
compromising organ function (132). Sustained ERS can enhance ROS
production, intensifying oxidative stress (133), or initiate an inflammatory
response, further aggravating cellular damage (134). In 2019, Torres et al
(135) first established that
APAP induces ALI through ERS, utilizing C57BL/6J 300 mg/kg APAP for
ALI modeling. By contrast, studies examining rhodiola rosea
glycosides for APAP-induced ALI prevention employed C57BL/6J 500
mg/kg APAP (136). This dosage
also revealed the transcription factor CHOP as a key regulator of
APAP-induced hepatotoxicity (137). The investigation of
hepatotoxicity alleviation through ERS remains in its early stages,
leaving uncertainty about future optimal APAP model
development.
The primary administration method for the ALI/ALF
model in C57BL/6J mice involves mixing CCl4 solution
with peanut, corn or olive oil at specific ratios, followed by
intraperitoneal injection for 24 h. Based on varying mixing ratios
and intraperitoneal injection doses, the constructed models are
primarily categorized into ALI and ALF models, with some
researchers defining specific lethal doses of CCl4
injection. For ALI model construction (Table I), CCl4 is mixed with
co-solvents at ratios of 0.3-50%, with injection doses ranging from
0.5-10 ml/kg. At this dose, extensive hepatocyte injury and intense
inflammatory responses occur, triggering repair pathways while
preserving sufficient residual hepatocytes and regenerative
potential. Animals survive severe injury and undergo gradual
repair. This model is suitable for studying liver injury,
inflammatory responses, repair initiation and early reversible
lesions such as drug-induced liver injury and acute hepatitis
(140-150).
Research indicates that severe ALI may progress to
ALF with notable mortality rates if not properly treated (156). While most current studies refer
to the constructed model as 'ALI' rather than 'ALF', the ALI model
has not successfully replicated ALF characteristics in terms of
survival or disease progression. Consequently, additional research
is necessary to analyze the effects of varying CCl4
doses on mouse survival and liver pathology, aiming to establish
reference standards for ALF research.
LPS/D-galactosamine (D-GalN)-induced ALF represents
a well-established animal model extensively utilized to investigate
the pathogenesis of fulminant hepatitis in humans (Fig. 6) (157). LPS (also termed endotoxin), a
macromolecule present in the outer membrane of Gram-negative
bacteria, stimulates inflammation. D-GalN molecules, when
metabolized through the galactose pathway in the liver, induce
severe metabolic alterations and hepatic necrosis by depleting
various intracellular uridine mediators (11). Additionally, D-GalN heightens the
sensitivity of the liver to the lethal effects of LPS and
hepatocellular cell death (158). The ALF model developed using
LPS/D-GalN demonstrates pathophysiological characteristics more
closely aligned with the progression of ALF triggered by ALI.
However, current studies lack a uniform standard, and even when
utilizing the same animal model, C57BL/6J, for ALF modeling, the
dosages employed by different researchers exhibit considerable
variation (159-174) (Table III). Notable variations in LPS
dosage exist across different studies, ranging from 2.5 to 250
μg/kg. However, independent studies across various LPS
dosage groups consistently demonstrate different degrees of liver
injury. For instance, serum AST and ALT levels both reached 2,000
U/l within 12 h, with a 60% mortality rate in ALF models following
administration of 300 mg/kg D-GalN and 5 μg/kg LPS to
C57BL/6J mice (162). However,
mice injected with high doses of drugs such as 800 mg/kg D-GalN and
100 μg/kg LPS exhibited biochemical indicators similar to
those observed at lower doses, yet their mortality rate reached as
high as 100% (174). To date,
existing studies cannot conclusively determine whether a
dose-dependent effect exists when LPS and D-GalN are combined.
However, current research indicates that the ALF induced by the
combination of LPS and D-GalN is primarily used to study the
hepatoprotective effects of drugs.
Azoxymethane (AOM) is a chemical compound found in
the nuts of the Guam soursop palm, that exhibits hepatotoxic and
carcinogenic properties (175).
Matkowskyj et al (176)
demonstrated that intraperitoneal injection of AOM in mice at doses
ranging from 50-200 mg/kg induced ALF and hepatic encephalopathy.
In studies of ALF hepatic encephalopathy using C57BL/6J mice as a
model, researchers administered a single intraperitoneal injection
of AOM at a uniform dose of 100 mg/kg (177-179) (Table IV). However, the AOM mouse model
is unique as it is a progressive liver injury model that does not
spontaneously recover; the survival rate of this mouse model
remains unclear.
Thioacetamide (TAA) is a sulfur-containing compound
that induces hepatocellular necrosis through monooxygenase
biotransformation (180). This
compound is frequently utilized in the development of induced
fulminant hepatic failure models (181). The primary mechanism of
TAA-induced hepatic injury involves the generation of ROS,
triggering oxidative damage (182). Consequently, TAA-based ALF
mouse models are widely employed to investigate antioxidant
protective effects (183), free
radical scavenging in damaged tissues (184) and pro-hepatic tissue
regeneration (185-189). The model construction in
C57BL/6J mice or other mouse strains typically involves a single
intraperitoneal injection of 300 mg/kg TAA for 24 h, resulting in a
3/24 survival rate (Table
IV).
As animal models constitute the primary research
subjects in ALF studies, establishing a standardized method for ALF
construction would notably advance ALF research. Current models
predominantly focus on developing ALI models. However, not all ALIs
progress to ALF, limiting precise therapeutic interventions. In the
APAP-induced pharmacological C57BL/6J mouse hepatocyte injury
model, doses >600 mg/kg resulted in a 48-h survival rate.
Similarly, single intraperitoneal doses of CCl4 at ≥5
mg/kg (1:1) demonstrated a 48-h survival rate. While these survival
cycles may reflect ALF progression more accurately, establishing
uniform ALF standards requires additional pathological and
physiological characterization. Recent advances in single-cell RNA
sequencing and spatial transcriptomics enable precise tracking of
disease onset and progression, potentially facilitating the
assessment of disease feature remodeling efficiency by various drug
doses or chemical inducers. For instance, after obtaining spatial
transcriptomic data, by calculating 'cell adjacency relationships',
whether the probability of cytotoxic T cells directly adjoining
dying hepatocytes in pathological regions significantly increases
with rising drug doses can be quantified, thereby measuring the
'spatial intensity' of immune attacks. Similarly, by defining
'niche composition', the co-localization ratio of pro-reparative
macrophages, activated stellate cells and hepatic progenitor cells
within a 100-μm radius of regenerating nodules can be
analyzed. This ratio serves as an indicator of 'regenerative
microenvironment health'. Ultimately, these mathematical vectors
extracted from spatial coordinates will form the objective,
quantitative foundation for comparing whether tissue
microenvironments undergo 'orderly remodeling' or 'disruptive
chaos' under different dose treatments.
Although LPS/D-GalN offers an alternative approach
for ALF model construction, the dosing protocols lack
standardization and exhibit greater variability compared with APAP
and CCl4 protocols, despite utilizing the same C57BL/6J
mouse strain. The existing literature does not adequately explain
these dose variations in ALF studies, necessitating further
consultation with investigators to understand the rationale behind
the different dose combinations crucial for ALF model
development.
Notably, ALF may exhibit patient-specific
heterogeneity, which could potentially explain the current lack of
harmonization in animal models. Addressing this challenge requires
enhanced collaboration between hepatologists and biologists. Tools
such as humanized mouse models or patient-derived organoids offer a
promising path toward accurately capturing the diversity of human
ALF. By inducing liver injury and implanting a mixture of human
fetal liver cells with hepatic progenitor cells and liver stem
cells, humanized mice with human-mouse liver chimeras
(human-reconstructed liver mice, HEP mice) have been successfully
established. These mice can be used to study human hepatotropic
pathogens (such as hepatitis B and C virus infections) and liver
diseases, providing a research platform for replicating the human
immune system (194). On the
other hand, patient-derived liver organoids hold immense potential
for reproducing disease characteristics, offering a unique platform
for identifying biomarkers of disease progression and inferring
personalized treatment strategies. For instance, iPSC-derived liver
organoids retain innate immune responses and maintain hepatocyte
polarity, reproducing the natural entry process of HBV and HCV and
enabling their intercellular transmission, faithfully recreating
host-virus interactions (195).
Constructing 'immunized' or 'vascularized' organoids that better
reflect the in vivo microenvironment by introducing HSCs,
endothelial cells, and immune cells may aid in reproducing the
pathological features of human ALF (196,197).
Model organisms provide essential support for
analyzing human health and disease mechanisms. The precise
development of model organisms with standardized feeding
environments, genetic backgrounds and biological interventions
facilitates both the collection of biological samples from cases
and controls throughout disease progression and the establishment
of a comprehensive understanding of molecular, cellular,
developmental and physiological properties, representing
fundamental elements of organismal biology research.
Not applicable.
SL designed the scope and structure of the review
and wrote the manuscript. FW and XH performed structured literature
searches. YJ performed the literature review and designed the
figures. DL and BT provided expert knowledge and critically revised
the manuscript. All authors read and approved the final manuscript.
Data authentication is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
The present review was supported by grants from the Science and
Technology Support Project of Yinchuan City (grant no. 2024SF045),
and the Special Talent Introduction Project of Ningxia Autonomous
Region Key R&D Programs (grant no. 2024BEH04106).
|
1
|
Newsome PN, Plevris JN, Nelson LJ and
Hayes PC: Animal models of fulminant hepatic failure: A critical
evaluation. Liver Transpl. 6:21–31. 2000.PubMed/NCBI
|
|
2
|
Nussler A, Konig S, Ott M, Sokal E, Christ
B, Thasler W, Brulport M, Gabelein G, Schormann W, Schulze M, et
al: Present status and perspectives of cell-based therapies for
liver diseases. J Hepatol. 45:144–159. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rifaie N and Saner FH: Critical care
management in patients with acute liver failure. Best Pract Res
Clin Anaesthesiol. 34:89–99. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Adam R, Cailliez V, Majno P, Karam V,
McMaster P, Caine RY, O'Grady J, Pichlmayr R, Neuhaus P, Otte JB,
et al: Normalised intrinsic mortality risk in liver
transplantation: European liver transplant registry study. Lancet.
356:621–627. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lidofsky SD, Bass NM, Prager MC,
Washington DE, Read AE, Wright TL, Ascher NL, Roberts JP,
Scharschmidt BF and Lake JR: Intracranial pressure monitoring and
liver transplantation for fulminant hepatic failure. Hepatology.
16:1–7. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
de Rave S, Tilanus HW, van der Linden J,
de Man RA, van der Berg B, Hop WC, Ijzermans JN, Zondervan PE and
Metselaar HJ: The importance of orthotopic liver transplantation in
acute hepatic failure. Transpl Int. 15:29–33. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Terblanche J and Hickman R: Animal models
of fulminant hepatic failure. Dig Dis Sci. 36:770–774. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mooney SM, Pjetri E, Friday WB and Smith
SM: Growth and behavioral differences in a C57BL/6J mouse model of
prenatal alcohol exposure. Alcohol. 97:51–57. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bishop CR, Caten FT, Nakaya HI and
Suhrbier A: Chikungunya patient transcriptional signatures
faithfully recapitulated in a C57BL/6J mouse model. Front Immunol.
13:10923702022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhou P, Xia J, Guo G, Huang ZX, Lu Q, Li
L, Li HX, Shi YJ and Bu H: A Macaca mulatta model of fulminant
hepatic failure. World J Gastroenterol. 18:435–444. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tuñón MJ, Alvarez M, Culebras JM and
González-Gallego J: An overview of animal models for investigating
the pathogenesis and therapeutic strategies in acute hepatic
failure. World J Gastroenterol. 15:3086–3098. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shingina A, Mukhtar N, Wakim-Fleming J,
Alqahtani S, Wong RJ, Limketkai BN, Larson AM and Grant L: Acute
liver failure guidelines. Am J Gastroenterol. 118:1128–1153. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Trey C and Davidson CS: The management of
fulminant hepatic failure. Prog Liver Dis. 3:282–298.
1970.PubMed/NCBI
|
|
14
|
European Association for the Study of the
Liver: Electronic address easloffice@easloffice.eu; Clinical
practice guidelines panel; Wendon J; Panel members; Cordoba J,
Dhawan A, Larsen FS, Manns M, Samuel D, et al: EASL clinical
practical guidelines on the management of acute (fulminant) liver
failure. J Hepatol. 66:1047–1081. 2017. View Article : Google Scholar
|
|
15
|
Lee WM, Stravitz RT and Larson AM:
Introduction to the revised American association for the study of
liver diseases position paper on acute liver failure 2011.
Hepatology. 55:965–967. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sarin SK, Choudhury A, Sharma MK, Maiwall
R, Al Mahtab M, Rahman S, Saigal S, Saraf N, Soin AS, Devarbhavi H,
et al: Acute-on-chronic liver failure: Consensus recommendations of
the Asian Pacific association for the study of the liver (APASL):
An update. Hepatol Int. 13:353–390. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kim A, Niu B, Woreta T and Chen PH:
Clinical considerations of coagulopathy in acute liver failure. J
Clin Transl Hepatol. 8:407–413. 2020. View Article : Google Scholar
|
|
18
|
Liver Failure and Artificial Liver Group;
Chinese Society of Infectious Diseases; Chinese Medical
Association; Severe Liver Disease and Artificial Liver Group;
Chinese Society of Hepatology; Chinese Medical Association:
Guideline for diagnosis and treatment of liver failure. Zhonghua
Gan Zang Bing Za Zhi. 27:18–26. 2019.In Chinese.
|
|
19
|
Rakela JL, Karvellas CJ, Koch DG, Vegunta
S and Lee WM: Acute liver failure: Biomarkers evaluated by the
acute liver failure study group. Clin Transl Gastroenterol.
14:e005652023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Stravitz RT and Lee WM: Acute liver
failure. Lancet. 394:869–881. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kumar R, Shalimar, Bhatia V, Khanal S,
Sreenivas V, Gupta SD, Panda SK and Acharya SK: Antituberculosis
therapy-induced acute liver failure: magnitude, profile, prognosis,
and predictors of outcome. Hepatology. 51:1665–1674. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Torres HA and Davila M: Reactivation of
hepatitis B virus and hepatitis C virus in patients with cancer.
Nat Rev Clin Oncol. 9:156–166. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhao P, Wang C, Liu W, Chen G, Liu X, Wang
X, Wang B, Yu L, Sun Y, Liang X, et al: Causes and outcomes of
acute liver failure in China. PLoS One. 8:e809912013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bernal W and Wendon J: Acute liver
failure. N Engl J Med. 369:2525–2534. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang YH, Wu DB, Chen B, Chen EQ and Tang
H: Progress in mesenchymal stem cell-based therapy for acute liver
failure. Stem Cell Res Ther. 9:2272018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bantel H and Schulze-Osthoff K: Mechanisms
of cell death in acute liver failure. Front Physiol. 3:792012.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lefkowitch JH: The pathology of acute
liver failure. Adv Anat Pathol. 23:144–158. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yu C, Chen P, Miao L and Di G: The role of
the NLRP3 inflammasome and programmed cell death in acute liver
injury. Int J Mol Sci. 24:30672023. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ryo K, Kamogawa Y, Ikeda I, Yamauchi K,
Yonehara S, Nagata S and Hayashi N: Significance of Fas
antigen-mediated apoptosis in human fulminant hepatic failure. Am J
Gastroenterol. 95:2047–2055. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Streetz K, Leifeld L, Grundmann D,
Ramakers J, Eckert K, Spengler U, Brenner D, Manns M and Trautwein
C: Tumor necrosis factor alpha in the pathogenesis of human and
murine fulminant hepatic failure. Gastroenterology. 119:446–460.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Tokushige K, Yamaguchi N, Ikeda I,
Hashimoto E, Yamauchi K and Hayashi N: Significance of soluble TNF
receptor-I in acute-type fulminant hepatitis. Am J Gastroenterol.
95:2040–2046. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Volkmann X, Anstaett M, Hadem J, Stiefel
P, Bahr MJ, Lehner F, Manns MP, Schulze-Osthoff K and Bantel H:
Caspase activation is associated with spontaneous recovery from
acute liver failure. Hepatology. 47:1624–1633. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Keppler D, Lesch R, Reutter W and Decker
K: Experimental hepatitis induced by D-galactosamine. Exp Mol
Pathol. 9:279–290. 1968. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
El-Mofty SK, Scrutton MC, Serroni A,
Nicolini C and Farber JL: Early, reversible plasma membrane injury
in galactosamine-induced liver cell death. Am J Pathol. 79:579–596.
1975.PubMed/NCBI
|
|
35
|
Hargreaves IP, Al Shahrani M, Wainwright L
and Heales SJ: Drug-induced mitochondrial toxicity. Drug Safety.
39:661–674. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jaeschke H and Ramachandran A:
Acetaminophen hepatotoxicity: Paradigm for understanding mechanisms
of drug-induced liver injury. Annu Rev Pathol. 19:453–478. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang C and Wang Y: The role and mechanism
of action of mitophagy in various liver diseases. Antioxid Redox
Signal. 38:529–549. 2023. View Article : Google Scholar
|
|
38
|
Wu KKL, Long K, Lin H, Siu PMF, Hoo RLC,
Ye D, Xu A and Cheng KKY: The APPL1-Rab5 axis restricts NLRP3
inflammasome activation through early endosomal-dependent mitophagy
in macrophages. Nat Commun. 12:66372021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang H, Zheng Y, Huang J and Li J:
Mitophagy in antiviral immunity. Front Cell Dev Biol. 9:7231082021.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chao X and Ding WX: Role and mechanisms of
autophagy in alcohol-induced liver injury. Adv Pharmacol.
85:109–131. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Lu X, Xuan W and Li J, Yao H, Huang C and
Li J: AMPK protects against alcohol-induced liver injury through
UQCRC2 to up-regulate mitophagy. Autophagy. 17:3622–3643. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Shan S, Shen Z, Zhang C, Kou R, Xie K and
Song F: Mitophagy protects against acetaminophen-induced acute
liver injury in mice through inhibiting NLRP3 inflammasome
activation. Biochem Pharmacol. 169:1136432019. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Jiang Z, Yang X, Han Y, Li J, Hu C, Liu C
and Xiao W: Sarmentosin promotes USP17 and regulates Nrf2-mediated
mitophagy and cellular oxidative stress to alleviate APAP-induced
acute liver failure. Phytomedicine. 104:1543372022. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Sowa JP, Gerken G and Canbay A: Acute
liver failure-it's just a matter of cell death. Dig Dis.
34:423–428. 2016. View Article : Google Scholar
|
|
45
|
Shen K, Chang W, Gao X, Wang H, Niu W,
Song L and Qin X: Depletion of activated hepatic stellate cell
correlates with severe liver damage and abnormal liver regeneration
in acetaminophen-induced liver injury. Acta Biochim Biophys Sin
(Shanghai). 43:307–315. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Mohammed OS, Attia HG, Mohamed BMSA,
Elbaset MA and Fayed HM: Current investigations for liver fibrosis
treatment: Between repurposing the FDA-approved drugs and the other
emerging approaches. J Pharm Pharm Sci. 26:118082023. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Issa R, Zhou X, Trim N, Millward-Sadler H,
Krane S, Benyon C and Iredale J: Mutation in collagen-1 that
confers resistance to the action of collagenase results in failure
of recovery from CCl4-induced liver fibrosis, persistence of
activated hepatic stellate cells, and diminished hepatocyte
regeneration. FASEB J. 17:47–49. 2003. View Article : Google Scholar
|
|
48
|
He Y, Jin L, Wang J, Yan Z, Chen T and
Zhao Y: Mechanisms of fibrosis in acute liver failure. Liver Int.
35:1877–1885. 2015. View Article : Google Scholar
|
|
49
|
Zhang Q, Yu T, Tan H and Shi H: Hepatic
recruitment of myeloid-derived suppressor cells upon liver injury
promotes both liver regeneration and fibrosis. BMC Gastroenterol.
24:1632024. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Antoniades CG, Berry PA, Wendon JA and
Vergani D: The importance of immune dysfunction in determining
outcome in acute liver failure. J Hepatol. 49:845–861. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Liaskou E, Wilson DV and Oo YH: Innate
immune cells in liver inflammation. Mediators Inflamm.
2012:9491572012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Yang Q, Shi Y, He J and Chen Z: The
evolving story of macrophages in acute liver failure. Immunol Lett.
147:1–9. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Lo GH: The role of imaging techniques in
clinical decision making for the management of hepatocellular
carcinoma. Hepatology. 56:7862012. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Abd El-Rahman SS and Fayed HM: Targeting
AngII/AT1R signaling pathway by perindopril inhibits ongoing liver
fibrosis in rat. J Tissue Eng Regen Med. 13:2131–2141. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Elbaset MA, Mohamed BMSA, Hessin A, Abd
El-Rahman SS, Esatbeyoglu T, Afifi SM and Fayed HM: Nrf2/HO-1,
NF-κB and PI3K/Akt signalling pathways decipher the therapeutic
mechanism of pitavastatin in early phase liver fibrosis in rats. J
Cell Mol Med. 28:e181162024. View Article : Google Scholar
|
|
56
|
Irion U and Nüsslein-Volhard C:
Developmental genetics with model organisms. Proc Natl Acad Sci
USA. 119:e21221481192022. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Kim J, Koo BK and Knoblich JA: Human
organoids: Model systems for human biology and medicine. Nat Rev
Mol Cell Biol. 21:571–584. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Sulston JE, Schierenberg E, White JG and
Thomson JN: The embryonic cell lineage of the nematode
Caenorhabditis elegans. Dev Biol. 100:64–119. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Mullins MC, Hammerschmidt M, Haffter P and
Nüsslein-Volhard C: Large-scale mutagenesis in the zebrafish: In
search of genes controlling development in a vertebrate. Curr Biol.
4:189–202. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Haffter P and Nüsslein-Volhard C: Large
scale genetics in a small vertebrate, the zebrafish. Int J Dev
Biol. 40:221–227. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Lee-Liu D, Méndez-Olivos EE, Muñoz R and
Larraín J: The African clawed frog Xenopus laevis: A model organism
to study regeneration of the central nervous system. Neurosci Lett.
652:82–93. 2017. View Article : Google Scholar
|
|
62
|
Brenner S: The genetics of Caenorhabditis
elegans. Genetics. 77:71–94. 1974. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Ellis HM and Horvitz HR: Genetic control
of programmed cell death in the nematode C. elegans. Cell.
44:817–829. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Grunwald DJ and Eisen JS: Headwaters of
the zebrafish-emergence of a new model vertebrate. Nat Rev Genet.
3:717–724. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
65
|
Streisinger G, Walker C, Dower N, Knauber
D and Singer F: Production of clones of homozygous diploid zebra
fish (Brachydanio rerio). Nature. 291:293–296. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Nüsslein-Volhard C: The zebrafish issue of
development. Development. 139:4099–4103. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Driever W, Solnica-Krezel L, Schier AF,
Neuhauss SC, Malicki J, Stemple DL, Stainier DY, Zwartkruis F,
Abdelilah S, Rangini Z, et al: A genetic screen for mutations
affecting embryogenesis in zebrafish. Development. 123:37–46. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Haffter P, Granato M, Brand M, Mullins MC,
Hammerschmidt M, Kane DA, Odenthal J, van Eeden FJ, Jiang YJ,
Heisenberg CP, et al: The identification of genes with unique and
essential functions in the development of the zebrafish, Danio
rerio. Development. 123:1–36. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Tan Q and Zoghbi HY: Mouse models as a
tool for discovering new neurological diseases. Neurobiol Learn
Mem. 165:1069022019. View Article : Google Scholar
|
|
70
|
Sztainberg Y and Zoghbi HY: Lessons
learned from studying syndromic autism spectrum disorders. Nat
Neurosci. 19:1408–1417. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Kleinsmith LJ and Pierce GB Jr:
Multipotentiality of single embryonal carcinoma cells. Cancer Res.
24:1544–1551. 1964.PubMed/NCBI
|
|
72
|
Stevens LC: Experimental production of
testicular teratomas in mice. Proc Natl Acad Sci USA. 52:654–661.
1964. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Zhang Z, Shao S and Meistrich ML:
Irradiated mouse testes efficiently support spermatogenesis derived
from donor germ cells of mice and rats. J Androl. 27:365–375. 2006.
View Article : Google Scholar
|
|
74
|
de Waal E, Yamazaki Y, Ingale P,
Bartolomei M, Yanagimachi R and McCarrey JR: Primary epimutations
introduced during intracytoplasmic sperm injection (ICSI) are
corrected by germline-specific epigenetic reprogramming. Proc Natl
Acad Sci USA. 109:4163–4168. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Winston F and Koshland D: Back to the
future: Mutant hunts are still the way to go. Genetics.
203:1007–1010. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Brown SD and Nolan PM: Mouse
mutagenesis-systematic studies of mammalian gene function. Hum Mol
Genet. 7:1627–1633. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Hrabé de Angelis MH, Flaswinkel H, Fuchs
H, Rathkolb B, Soewarto D, Marschall S, Heffner S, Pargent W,
Wuensch K, Jung M, et al: Genome-wide, large-scale production of
mutant mice by ENU mutagenesis. Nat Genet. 25:444–447. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Balling R: ENU mutagenesis: Analyzing gene
function in mice. Annu Rev Genomics Hum Genet. 2:463–492. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Beutler B: Finding new components of the
mammalian immune system. Rambam Maimonides Med J. 7:e00182016.
View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Gordon JW, Scangos GA, Plotkin DJ, Barbosa
JA and Ruddle FH: Genetic transformation of mouse embryos by
microinjection of purified DNA. Proc Natl Acad Sci USA.
77:7380–7384. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Brinster RL, Chen HY, Trumbauer M, Senear
AW, Warren R and Palmiter RD: Somatic expression of herpes
thymidine kinase in mice following injection of a fusion gene into
eggs. Cell. 27:223–231. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Costantini F and Lacy E: Introduction of a
rabbit beta-globin gene into the mouse germ line. Nature.
294:92–94. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Wagner EF, Stewart TA and Mintz B: The
human beta-globin gene and a functional viral thymidine kinase gene
in developing mice. Proc Natl Acad Sci USA. 78:5016–5020. 1981.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Smithies O, Gregg RG, Boggs SS, Koralewski
MA and Kucherlapati RS: Insertion of DNA sequences into the human
chromosomal beta-globin locus by homologous recombination. Nature.
317:230–234. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Capecchi MR: The new mouse genetics:
Altering the genome by gene targeting. Trends Genet. 5:70–76. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Johnson MP, Drugan A, Miller OJ and Evans
MI: Genetic correction of hereditary disease. Fetal Ther. 4(Suppl
1): S28–S39. 1989. View Article : Google Scholar
|
|
87
|
Cong L, Ran FA, Cox D, Lin S, Barretto R,
Habib N, Hsu PD, Wu X, Jiang W, Marraffini LA and Zhang F:
Multiplex genome engineering using CRISPR/Cas systems. Science.
339:819–823. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Gilbert LA, Larson MH, Morsut L, Liu Z,
Brar GA, Torres SE, Stern-Ginossar N, Brandman O, Whitehead EH,
Doudna JA, et al: CRISPR-mediated modular RNA-guided regulation of
transcription in eukaryotes. Cell. 154:442–451. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Mali P, Yang L, Esvelt KM, Aach J, Guell
M, DiCarlo JE, Norville JE and Church GM: RNA-guided human genome
engineering via Cas9. Science. 339:823–826. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Austin CP, Battey JF, Bradley A, Bucan M,
Capecchi M, Collins FS, Dove WF, Duyk G, Dymecki S, Eppig JT, et
al: The knockout mouse project. Nat Genet. 36:921–924. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Koutnikova H, Cock TA, Watanabe M, Houten
SM, Champy MF, Dierich A and Auwerx J: Compensation by the muscle
limits the metabolic consequences of lipodystrophy in PPAR gamma
hypomorphic mice. Proc Natl Acad Sci USA. 100:14457–14462. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Shultz LD, Brehm MA, Garcia-Martinez JV
and Greiner DL: Humanized mice for immune system investigation:
Progress, promise and challenges. Nat Rev Immunol. 12:786–798.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Holash J, Davis S, Papadopoulos N, Croll
SD, Ho L, Russell M, Boland P, Leidich R, Hylton D, Burova E, et
al: VEGF-Trap: A VEGF blocker with potent antitumor effects. Proc
Natl Acad Sci USA. 99:11393–11398. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Rongvaux A, Willinger T, Takizawa H,
Rathinam C, Auerbach W, Murphy AJ, Valenzuela DM, Yancopoulos GD,
Eynon EE, Stevens S, et al: Human thrombopoietin knockin mice
efficiently support human hematopoiesis in vivo. Proc Natl Acad Sci
USA. 108:2378–2383. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Vaughan AM, Pinapati RS, Cheeseman IH,
Camargo N, Fishbaugher M, Checkley LA, Nair S, Hutyra CA, Nosten
FH, Anderson TJ, et al: Plasmodium falciparum genetic crosses in a
humanized mouse model. Nat Methods. 12:631–633. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Martin FH, Suggs SV, Langley KE, Lu HS,
Ting J, Okino KH, Morris CF, McNiece IK, Jacobsen FW, Mendiaz EA,
et al: Primary structure and functional expression of rat and human
stem cell factor DNAs. Cell. 63:203–211. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Willinger T, Rongvaux A, Takizawa H,
Yancopoulos GD, Valenzuela DM, Murphy AJ, Auerbach W, Eynon EE,
Stevens S, Manz MG and Flavell RA: Human IL-3/GM-CSF knock-in mice
support human alveolar macrophage development and human immune
responses in the lung. Proc Natl Acad Sci USA. 108:2390–2395. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Macdonald LE, Karow M, Stevens S, Auerbach
W, Poueymirou WT, Yasenchak J, Frendewey D, Valenzuela DM,
Giallourakis CC, Alt FW, et al: Precise and in situ genetic
humanization of 6 Mb of mouse immunoglobulin genes. Proc Natl Acad
Sci USA. 111:5147–5152. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Ussar S, Griffin NW, Bezy O, Fujisaka S,
Vienberg S, Softic S, Deng L, Bry L, Gordon JI and Kahn CR:
Interactions between gut microbiota, host genetics and diet
modulate the predisposition to obesity and metabolic syndrome. Cell
Metab. 22:516–530. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Champy MF, Selloum M, Piard L, Zeitler V,
Caradec C, Chambon P and Auwerx J: Mouse functional genomics
requires standardization of mouse handling and housing conditions.
Mamm Genome. 15:768–783. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Rossi G, Manfrin A and Lutolf MP: Progress
and potential in organoid research. Nat Rev Genet. 19:671–687.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Clevers H: Modeling development and
disease with organoids. Cell. 165:1586–1597. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Homan KA, Gupta N, Kroll KT, Kolesky DB,
Skylar-Scott M, Miyoshi T, Mau D, Valerius MT, Ferrante T,
Bonventre JV, et al: Flow-enhanced vascularization and maturation
of kidney organoids in vitro. Nat Methods. 16:255–262. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Neal JT, Li X, Zhu J, Giangarra V,
Grzeskowiak CL, Ju J, Liu IH, Chiou SH, Salahudeen AA, Smith AR, et
al: Organoid modeling of the tumor immune microenvironment. Cell.
175:1972–1988.e16. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Ingber DE: Is it time for reviewer 3 to
request human organ chip experiments instead of animal validation
studies? Adv Sci (Weinh). 7:20020302020. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Collins AL, Levenson JM, Vilaythong AP,
Richman R, Armstrong DL, Noebels JL, David Sweatt J and Zoghbi HY:
Mild overexpression of MeCP2 causes a progressive neurological
disorder in mice. Hum Mol Genet. 13:2679–2689. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Han K, Holder JL Jr, Schaaf CP, Lu H, Chen
H, Kang H, Tang J, Wu Z, Hao S, Cheung SW, et al: SHANK3
overexpression causes manic-like behaviour with unique
pharmacogenetic properties. Nature. 503:72–77. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Hassan NH, Kamel GM, Fayed HM, Korany RMS
and Ramadan A: Dapagliflozin alleviates thioacetamide induced-liver
fibrosis in rats via controlling the Nrf2/HO-1 and TLR4/TGF-β1/PI3K
signaling pathways. Immunopharmacol Immunotoxicol. 47:392–405.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Mitchell JR, Jollow DJ, Potter WZ, Davis
DC, Gillette JR and Brodie BB: Acetaminophen-induced hepatic
necrosis. I. Role of drug metabolism. J Pharmacol Exp Ther.
187:185–194. 1973. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Jaeschke H and Mitchell JR: Use of
isolated perfused organs in hypoxia and ischemia/reperfusion
oxidant stress. Methods Enzymol. 186:752–759. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Knight TR, Kurtz A, Bajt ML, Hinson JA and
Jaeschke H: Vascular and hepatocellular peroxynitrite formation
during acetaminophen toxicity: Role of mitochondrial oxidant
stress. Toxicol Sci. 62:212–220. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Agarwal R, MacMillan-Crow LA, Rafferty TM,
Saba H, Roberts DW, Fifer EK, James LP and Hinson JA:
Acetaminophen-induced hepatotoxicity in mice occurs with inhibition
of activity and nitration of mitochondrial manganese superoxide
dismutase. J Pharmacol Exp Ther. 337:110–116. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Masubuchi Y, Suda C and Horie T:
Involvement of mitochondrial permeability transition in
acetaminophen-induced liver injury in mice. J Hepatol. 42:110–116.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Yan M, Huo Y, Yin S and Hu H: Mechanisms
of acetaminophen-induced liver injury and its implications for
therapeutic interventions. Redox Biol. 17:274–283. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Samuvel DJ, Nguyen NT, Jaeschke H,
Lemasters JJ, Wang X, Choo YM, Hamann MT and Zhong Z: Platanosides,
a potential botanical drug combination, decrease liver injury
caused by acetaminophen overdose in mice. J Nat Prod. 85:1779–1788.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Li H, Liu Y, Li J, Liu Y, Dong L, Yin Y,
Yu Y, Zhou J, Zhang L, Lu X, et al: Mannan-binding lectin
attenuates acetaminophen-induced hepatotoxicity by regulating
CYP2E1 expression via ROS-dependent JNK/SP1 pathway. Eur J Immunol.
49:564–575. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Du Z, Ma Z, Lai S, Ding Q, Hu Z, Yang W,
Qian Q, Zhu L, Dou X and Li S: Atractylenolide I ameliorates
acetaminophen-induced acute liver injury via the TLR4/MAPKs/NF-κB
signaling pathways. Front Pharmacol. 13:7974992022. View Article : Google Scholar
|
|
118
|
Ma X, McKeen T, Zhang J and Ding WX: Role
and mechanisms of mitophagy in liver diseases. Cells. 9:8372020.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Kim HY, Yoon HS, Heo AJ, Jung EJ, Ji CH,
Mun SR, Lee MJ, Kwon YT and Park JW: Mitophagy and endoplasmic
reticulum-phagy accelerated by a p62 ZZ ligand alleviates
paracetamol-induced hepatotoxicity. Br J Pharmacol. 180:1247–1266.
2023. View Article : Google Scholar
|
|
120
|
Cai J, Kong D, Long Z, Liu J, Liu R and
Hai C: Targeting PARK7 improves acetaminophen-induced acute liver
injury by orchestrating mitochondrial quality control and metabolic
reprogramming. Antioxidants (Basel). 11:21282022. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Baulies A, Ribas V, Núñez S, Torres S,
Alarcón-Vila C, Martínez L, Suda J, Ybanez MD, Kaplowitz N,
García-Ruiz C and Fernández-Checa JC: Lysosomal cholesterol
accumulation sensitizes to acetaminophen hepatotoxicity by
impairing mitophagy. Sci Rep. 5:180172015. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Chen Y, Park HJ, Park J, Song HC, Ryter
SW, Surh YJ, Kim UH, Joe Y and Chung HT: Carbon monoxide
ameliorates acetaminophen-induced liver injury by increasing
hepatic HO-1 and Parkin expression. FASEB J. 33:13905–13919. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Ni HM, McGill MR, Chao X, Du K, Williams
JA, Xie Y, Jaeschke H and Ding WX: Removal of acetaminophen protein
adducts by autophagy protects against acetaminophen-induced liver
injury in mice. J Hepatol. 65:354–362. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Lin Z, Wu F, Lin S, Pan X, Jin L, Lu T,
Shi L, Wang Y, Xu A and Li X: Adiponectin protects against
acetaminophen-induced mitochondrial dysfunction and acute liver
injury by promoting autophagy in mice. J Hepatol. 61:825–831. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Ni HM, Bockus A, Boggess N, Jaeschke H and
Ding WX: Activation of autophagy protects against
acetaminophen-induced hepatotoxicity. Hepatology. 55:222–232. 2012.
View Article : Google Scholar
|
|
126
|
Williams JA, Ni HM, Haynes A, Manley S, Li
Y, Jaeschke H and Ding WX: Chronic deletion and acute knockdown of
parkin have differential responses to acetaminophen-induced
mitophagy and liver injury in mice. J Biol Chem. 290:10934–10946.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Wang H, Ni HM, Chao X, Ma X, Rodriguez YA,
Chavan H, Wang S, Krishnamurthy P, Dobrowsky R, Xu DX, et al:
Double deletion of PINK1 and Parkin impairs hepatic mitophagy and
exacerbates acetaminophen-induced liver injury in mice. Redox Biol.
22:101482019. View Article : Google Scholar
|
|
128
|
Hu B, Li J, Gong D, Dai Y, Wang P, Wan L
and Xu S: Long-term consumption of food-derived chlorogenic acid
protects mice against acetaminophen-induced hepatotoxicity via
promoting PINK1-dependent mitophagy and inhibiting apoptosis.
Toxics. 10:6652022. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Zhang P, Chen S, Tang H, Fang W, Chen K
and Chen X: CoQ10 protects against acetaminophen-induced liver
injury by enhancing mitophagy. Toxicol Appl Pharmacol.
410:1153552021. View Article : Google Scholar
|
|
130
|
Wu Y, Li W, Zhang J, Lin J, You L, Su J,
Zheng C, Gao Y, Kong X and Sun X: Shaoyao-Gancao Decoction, a
famous Chinese medicine formula, protects against APAP-induced
liver injury by promoting autophagy/mitophagy. Phytomedicine.
135:1560532024. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Di Conza G and Ho PC: ER stress responses:
An emerging modulator for innate immunity. Cells. 9:6952020.
View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Hu H, Tian M, Ding C and Yu S: The C/EBP
homologous protein (CHOP) transcription factor functions in
endoplasmic reticulum stress-induced apoptosis and microbial
infection. Front Immunol. 9:30832019. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Feng K, Chen Z, Pengcheng L, Zhang S and
Wang X: Quercetin attenuates oxidative stress-induced apoptosis via
SIRT1/AMPK-mediated inhibition of ER stress in rat chondrocytes and
prevents the progression of osteoarthritis in a rat model. J Cell
Physiol. 234:18192–18205. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Sprenkle NT, Sims SG, Sánchez CL and
Meares GP: Endoplasmic reticulum stress and inflammation in the
central nervous system. Mol Neurodegener. 12:422017. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Torres S, Baulies A, Insausti-Urkia N,
Alarcón-Vila C, Fucho R, Solsona-Vilarrasa E, Núñez S, Robles D,
Ribas V, Wakefield L, et al: Endoplasmic reticulum stress-induced
upregulation of STARD1 promotes acetaminophen-induced acute liver
failure. Gastroenterology. 157:552–568. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Xu J, Zhao L, Zhang X, Ying K, Zhou R, Cai
W, Wu X, Jiang H, Xu Q, Miao D, et al: Salidroside ameliorates
acetaminophen-induced acute liver injury through the inhibition of
endoplasmic reticulum stress-mediated ferroptosis by activating the
AMPK/SIRT1 pathway. Ecotoxicol Environ Saf. Aug 7–2023.Epub ahead
of print. View Article : Google Scholar
|
|
137
|
Uzi D, Barda L, Scaiewicz V, Mills M,
Mueller T, Gonzalez-Rodriguez A, Valverde AM, Iwawaki T, Nahmias Y,
Xavier R, et al: CHOP is a critical regulator of
acetaminophen-induced hepatotoxicity. J Hepatol. 59:495–503. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Zhang JQ, Shi L, Xu XN, Huang SC, Lu B, Ji
LL and Wang ZT: Therapeutic detoxification of quercetin against
carbon tetrachloride-induced acute liver injury in mice and its
mechanism. J Zhejiang Univ Sci B. 15:1039–1047. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Recknagel RO, Glende EA Jr, Dolak JA and
Waller RL: Mechanisms of carbon tetrachloride toxicity. Pharmacol
Ther. 43:139–154. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Liu J, Zhang QY, Yu LM, Liu B, Li MY and
Zhu RZ: Phycocyanobilin accelerates liver regeneration and reduces
mortality rate in carbon tetrachloride-induced liver injury mice.
World J Gastroenterol. 21:5465–5472. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Wang DH, Wang YN, Ge JY, Liu HY, Zhang HJ,
Qi Y, Liu ZH and Cui XL: Role of activin A in carbon
tetrachloride-induced acute liver injury. World J Gastroenterol.
19:3802–3809. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Chen Y, Wu Y, Sun H, Zhang H, Tang D, Yuan
T, Chen C, Huang W, Zhou X, Wu H, et al: Human liver
progenitor-like cells-derived extracellular vesicles promote liver
regeneration during acute liver failure. Cell Biol Toxicol.
40:1062024. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Kakizaki M, Yamamoto Y, Nakayama S, Kameda
K, Nagashima E, Ito M, Suyama T, Matsuzaki Y, Chiba T, Sumiyoshi H,
et al: Human hepatocyte-derived extracellular vesicles attenuate
the carbon tetrachloride-induced acute liver injury in mice. Cell
Death Dis. 12:10102021. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Zhou J, Feng X, Zhu J, Feng B, Yao Q, Pan
Q, Yu J, Yang J, Li L and Cao H: Mesenchymal stem cell treatment
restores liver macrophages homeostasis to alleviate mouse acute
liver injury revealed by single-cell analysis. Pharmacol Res.
179:1062292022. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Hu D, Huang S, Ding Y, Zhao X, Zhang W,
Chen H and Wang J: Specnuezhenide reduces carbon
tetrachloride-induced liver injury in mice through inhibition of
oxidative stress and hepatocyte apoptosis. J Pharm Pharmacol.
74:191–199. 2022. View Article : Google Scholar
|
|
146
|
Jia S, Chen Q, Wu J, Yao X, Shao J, Cheng
X, Zhang C, Cen D, Wang Y, Shen Z, et al: Danshensu derivative ADTM
ameliorates CCl4-induced acute liver injury in mice
through inhibiting oxidative stress and apoptosis. Pathol Res
Pract. 228:1536562021. View Article : Google Scholar
|
|
147
|
Zhang X, Kuang G, Wan J, Jiang R, Ma L,
Gong X and Liu X: Salidroside protects mice against CCl4-induced
acute liver injury via down-regulating CYP2E1 expression and
inhibiting NLRP3 inflammasome activation. Int Immunopharmacol.
85:1066622020. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Yukawa H, Noguchi H, Oishi K, Takagi S,
Hamaguchi M, Hamajima N and Hayashi S: Cell transplantation of
adipose tissue-derived stem cells in combination with heparin
attenuated acute liver failure in mice. Cell Transplant.
18:611–618. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Zhang F, Wang X, Qiu X, Wang J, Fang H,
Wang Z, Sun Y and Xia Z: The protective effect of Esculentoside A
on experimental acute liver injury in mice. PLoS One.
9:e1131072014. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Lu S, Shi G, Xu X, Wang G, Lan X, Sun P,
Li X, Zhang B, Gu X, Ichim TE and Wang H: Human endometrial
regenerative cells alleviate carbon tetrachloride-induced acute
liver injury in mice. J Transl Med. 14:3002016. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Dai C, Xiao X, Li D, Tun S, Wang Y, Velkov
T and Tang S: Chloroquine ameliorates carbon tetrachloride-induced
acute liver injury in mice via the concomitant inhibition of
inflammation and induction of apoptosis. Cell Death Dis.
9:11642018. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Nautiyal N, Maheshwari D, Tripathi DM,
Kumar D, Kumari R, Gupta S, Sharma S, Mohanty S, Parasar A, Bihari
C, et al: Establishment of a murine model of acute-on-chronic liver
failure with multi-organ dysfunction. Hepatol Int. 15:1389–1401.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Liang H, Huang K, Su T, Li Z, Hu S, Dinh
PU, Wrona EA, Shao C, Qiao L, Vandergriff AC, et al: Mesenchymal
stem cell/red blood cell-inspired nanoparticle therapy in mice with
carbon tetrachloride-induced acute liver failure. ACS Nano.
12:6536–6544. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Wang CH, Chen CY, Wang KH, Kao AP, Chen
YJ, Lin PH, Chen M, Wu TY, Cheng JJ, Lee KD and Chuang KH:
Comparing the therapeutic mechanism and immune response of human
and mouse mesenchymal stem cells in immunocompetent mice with acute
liver failure. Stem Cells Transl Med. 12:39–53. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Li F, Wei H, Jin Y, Xue T, Xu Y, Wang H,
Ju E, Tao Y and Li M: Microfluidic fabrication of MicroRNA-induced
hepatocyte-like cells/human umbilical vein endothelial cells-laden
microgels for acute liver failure treatment. ACS Nano.
17:25243–25256. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Bernal W, Auzinger G, Dhawan A and Wendon
J: Acute liver failure. Lancet. 376:190–201. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Huang S, Wang Y, Xie S, Lai Y, Mo C, Zeng
T, Kuang S, Deng G, Zhou C, Chen Y, et al: Hepatic TGFβr1
deficiency attenuates lipopolysaccharide/D-galactosamine-induced
acute liver failure through inhibiting GSK3β-Nrf2-mediated
hepatocyte apoptosis and ferroptosis. Cell Mol Gastroenterol
Hepatol. 13:1649–1672. 2022. View Article : Google Scholar
|
|
158
|
Shashikumar A, Roy R and Dessai SN:
Hepatocyte growth factor and fish oil facilitated reversal of
D-galactosamine-induced toxicity in primary hepatocyte cultures of
albino mice. Nutrition. 91-92:1114662021. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Zhang H, Gao M, Wang H, Zhang J, Wang L,
Dong G, Ma Q, Li C, Dai J, Li Z, et al: Atractylenolide I prevents
acute liver failure in mouse by regulating M1 macrophage
polarization. Sci Rep. 15:40152025. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Wang Z, Ling J, You S and Zhu B: Exosomes
derived from liver failure patients' plasma stimulated mesenchymal
stem cells alleviate acute liver failure. Stem Cell Res Ther.
16:482025. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Ding W, Fan YY, Zhang C, Fu L, Chen X and
Xu DX: Obeticholic acid differentially regulates hepatic injury and
inflammation at different stages of
D-galactosamine/lipopolysaccharide-evoked acute liver failure. Eur
J Pharmacol. 850:150–157. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Wang W, Lu H, Lu X, Wang D, Wang Z, Dai W,
Wang J and Liu P: Effect of tumor necrosis factor-α on the
expression of the ammonia transporter Rhcg in the brain in mice
with acute liver failure. J Neuroinflammation. 15:2342018.
View Article : Google Scholar
|
|
163
|
Zheng J, Xiao J, Fan Y, Zheng H, Liu H,
Xiang J, Hai L, Wang Y and Zhang X: CD24 regulates liver immune
response and ameliorates acute hepatic injury through controlling
hepatic macrophages. Eur J Immunol. 54:e24511782024. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Zhou Z, Wang B, Pan X, Lv J, Lou Z, Han Y,
Yao Y, Chen J, Wang Q and Li L: Microbial metabolites indole
derivatives sensitize mice to D-GalN/LPS induced-acute liver
failure via the Tlr2/NF-κB pathway. Front Microbiol.
13:11039982023. View Article : Google Scholar
|
|
165
|
Wang Y, Shi C, Guo J, Zhang D, Zhang Y,
Zhang L and Gong Z: IDH1/MDH1 deacetylation promotes acute liver
failure by regulating NETosis. Cell Mol Biol Lett. 29:82024.
View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Zhang WB, Zhang HY, Wang Y, Jiao FZ, Wang
LW and Gong ZJ: Quantitative proteomic analysis reveals the sites
related to acetylation and mechanism of ACY-1215 in acute liver
failure mice. Front Pharmacol. 10:6532019. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Xu FF, Li ZC, Zhang WJ, Li Q, Li DJ, Meng
HB, Shen FM and Fu H: Activation of α7 nicotinic acetylcholine
receptors inhibits hepatic necroptosis and ameliorates acute liver
injury in mice. Anesthesiology. 141:1119–1138. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Sun F, Wang J, Ji X, Wang Z, Gao S and
Wang K: CCL25 contributes to the pathogenesis of D-Gal/LPS-induced
acute liver failure. J Gastroenterol Hepatol. 39:2880–2891. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Zhang X, Hu Y, Wang W, Ji R, Li Z, Yu W,
Wu Z, Xiao Y, Guo T, Qi Z, et al: IRGM/Irgm1 increases autophagy to
inhibit activation of NLRP3 inflammasome in inflammatory injury
induced acute liver failure. Cell Death Discov. 10:2722024.
View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Ji XF, Fan YC, Sun F, Wang JW and Wang K:
Noncanonical Wnt5a/JNK signaling contributes to the development of
D-Gal/LPS-induced acute liver failure. Inflammation. 45:1362–1373.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Fan XG, Pei SY, Zhou D, Zhou PC, Huang Y,
Hu XW, Li T, Wang Y, Huang ZB and Li N: Melittin ameliorates
inflammation in mouse acute liver failure via inhibition of
PKM2-mediated Warburg effect. Acta Pharmacol Sin. 42:1256–1266.
2021. View Article : Google Scholar
|
|
172
|
Kong D, Xu H, Chen M, Yu Y, Qian Y, Qin T,
Tong Y, Xia Q and Hang H: Co-encapsulation of HNF4α overexpressing
UMSCs and human primary hepatocytes ameliorates mouse acute liver
failure. Stem Cell Res Ther. 11:4492020. View Article : Google Scholar
|
|
173
|
Ma J, Xu Y, Zhang M and Li Y: Geraniol
ameliorates acute liver failure induced by
lipopolysaccharide/D-galactosamine via regulating macrophage
polarization and NLRP3 inflammasome activation by PPAR-γ
methylation Geraniol alleviates acute liver failure. Biochem
Pharmacol. 210:1154672023. View Article : Google Scholar
|
|
174
|
Nakama T, Hirono S, Moriuchi A, Hasuike S,
Nagata K, Hori T, Ido A, Hayashi K and Tsubouchi H: Etoposide
prevents apoptosis in mouse liver with
D-galactosamine/lipopolysaccharide-induced fulminant hepatic
failure resulting in reduction of lethality. Hepatology.
33:1441–1450. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Ward JM: Dose response to a single
injection of azoxymethane in rats. Induction of tumors in the
gastrointestinal tract, auditory sebaceous glands, kidney, liver
and preputial gland. Vet Pathol. 12:165–177. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Matkowskyj KA, Marrero JA, Carroll RE,
Danilkovich AV, Green RM and Benya RV: Azoxymethane-induced
fulminant hepatic failure in C57BL/6J mice: Characterization of a
new animal model. Am J Physiol. 277:G455–G462. 1999.PubMed/NCBI
|
|
177
|
Jefferson B, Ali M, Grant S, Frampton G,
Ploof M, Andry S, DeMorrow S and McMillin M: Thrombospondin-1
exacerbates acute liver failure and hepatic encephalopathy
pathology in mice by activating transforming growth factor β1. Am J
Pathol. 190:347–357. 2020. View Article : Google Scholar
|
|
178
|
Xie G, Wang X, Jiang R, Zhao A, Yan J,
Zheng X, Huang F, Liu X, Panee J, Rajani C, et al: Dysregulated
bile acid signaling contributes to the neurological impairment in
murine models of acute and chronic liver failure. EBioMedicine.
37:294–306. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Bhattarai SM, Jhawer A, Frampton G,
Troyanovskaya E, DeMorrow S and McMillin M: Characterization of
hepatic pathology during azoxymethane-induced acute liver failure.
World J Gastroenterol. 31:1039522025. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Chieli E and Malvaldi G: Role of the
microsomal FAD-containing monooxygenase in the liver toxicity of
thioacetamide S-oxide. Toxicology. 31:41–52. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Bruck R, Aeed H, Schey R, Matas Z, Reifen
R, Zaiger G, Hochman A and Avni Y: Pyrrolidine dithiocarbamate
protects against thioacetamide-induced fulminant hepatic failure in
rats. J Hepatol. 36:370–377. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Pallottini V, Martini C, Bassi AM, Romano
P, Nanni G and Trentalance A: Rat HMGCoA reductase activation in
thioacetamide-induced liver injury is related to an increased
reactive oxygen species content. J Hepatol. 44:368–374. 2006.
View Article : Google Scholar
|
|
183
|
Shapiro H, Ashkenazi M, Weizman N,
Shahmurov M, Aeed H and Bruck R: Curcumin ameliorates acute
thioacetamide-induced hepatotoxicity. J Gastroenterol Hepatol.
21:358–366. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Harputluoglu MM, Demirel U, Ciralik H,
Temel I, Firat S, Ara C, Aladag M, Karincaoglu M and Hilmioglu F:
Protective effects of Gingko biloba on thioacetamide-induced
fulminant hepatic failure in rats. Hum Exp Toxicol. 25:705–713.
2006. View Article : Google Scholar
|
|
185
|
Margeli AP, Papadimitriou L, Ninos S,
Manolis E, Mykoniatis MG and Theocharis SE: Hepatic stimulator
substance administration ameliorates liver regeneration in an
animal model of fulminant hepatic failure and encephalopathy. Liver
Int. 23:171–178. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Chu CJ, Hsiao CC, Wang TF, Chan CY, Lee
FY, Chang FY, Chen YC, Huang HC, Wang SS and Lee SD: Prostacyclin
inhibition by indomethacin aggravates hepatic damage and
encephalopathy in rats with thioacetamide-induced fulminant hepatic
failure. World J Gastroenterol. 11:232–236. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Avraham Y, Zolotarev O, Grigoriadis NC,
Poutahidis T, Magen I, Vorobiav L, Zimmer A, Ilan Y, Mechoulam R
and Berry EM: Cannabinoids and capsaicin improve liver function
following thioacetamide-induced acute injury in mice. Am J
Gastroenterol. 103:3047–3056. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Xu R, Ni B, Wang L, Shan J, Pan L, He Y,
Lv G, Lin H, Chen W and Zhang Q: CCR2-overexpressing mesenchymal
stem cells targeting damaged liver enhance recovery of acute liver
failure. Stem Cell Res Ther. 13:552022. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Kolodziejczyk AA, Federici S, Zmora N,
Mohapatra G, Dori-Bachash M, Hornstein S, Leshem A, Reuveni D,
Zigmond E, Tobar A, et al: Acute liver failure is regulated by MYC-
and microbiome-dependent programs. Nat Med. 26:1899–1911. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Xue J, Lou X, Ning D, Shao R and Chen G:
Mechanism and treatment of α-amanitin poisoning. Arch Toxicol.
97:121–131. 2023. View Article : Google Scholar
|
|
191
|
Takada Y, Ishiguro S, Fukunaga K, Gu M,
Taniguchi H, Seino KI, Yuzawa K, Otsuka M, Todoroki T and Fukao K:
Increased intracranial pressure in a porcine model of fulminant
hepatic failure using amatoxin and endotoxin. J Hepatol.
34:825–831. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Wang H, Zhang H, Miao L, Wang C, Teng H,
Li X, Zhang X, Yang G, Wang S and Zeng X: α-amanitin induces
hepatotoxicity via PPAR-γ inhibition and NLRP3 inflammasome
activation. Ecotoxicol Environ Saf. 290:1177492025. View Article : Google Scholar
|
|
193
|
Wang H, Yang G, Zhang X, Zhang H, Liu Y,
Wang C, Miao L, Li Y, Huang Y, Teng H, et al: Cannabidiol protects
the liver from α-Amanitin-induced apoptosis and oxidative stress
through the regulation of Nrf2. Food Chem Toxicol. 182:1141962023.
View Article : Google Scholar
|
|
194
|
Ye W and Chen Q: Potential applications
and perspectives of humanized mouse models. Annu Rev Anim Biosci.
10:395–417. 2022. View Article : Google Scholar
|
|
195
|
Prior N, Inacio P and Huch M: Liver
organoids: From basic research to therapeutic applications. Gut.
68:2228–2237. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Salih ARC, Peirsman A, Khorsandi D, Ferrao
R, Ferreira L, Kamaraj M, John JV, Baquerizo A and Jucaud V: Liver
tissueoid on-a-chip modeling liver regeneration and allograft
rejection. Adv Mater. e211782026.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Balachander GM, Ng IC, Pai RR, Mitra K,
Tasnim F, Lim YS, Kwok R, Song Y, Yaw LP, Quah CB, et al: LEADS-a
comprehensive human liver-on-a-chip for non-alcoholic
steatohepatitis (NASH) drug testing. Lab Chip. 25:3444–3466. 2025.
View Article : Google Scholar : PubMed/NCBI
|