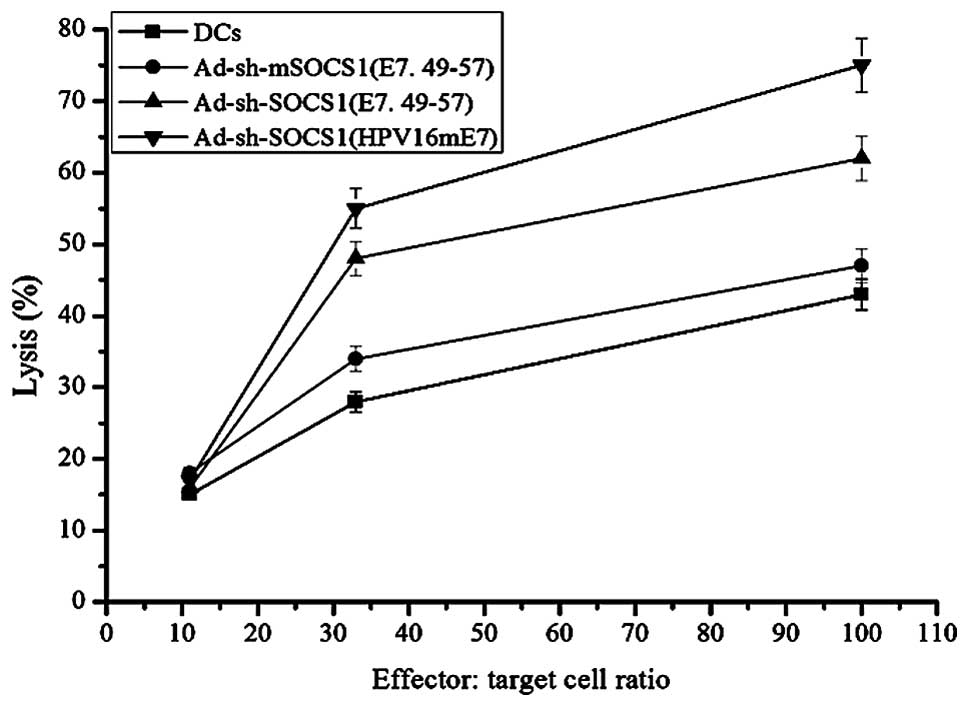
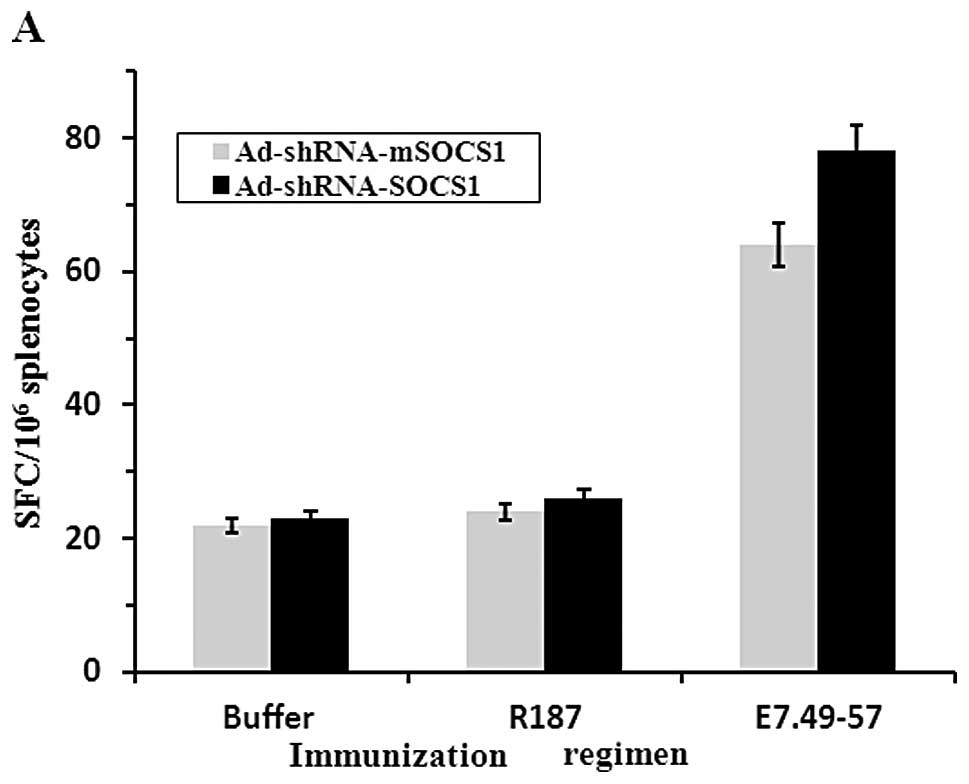
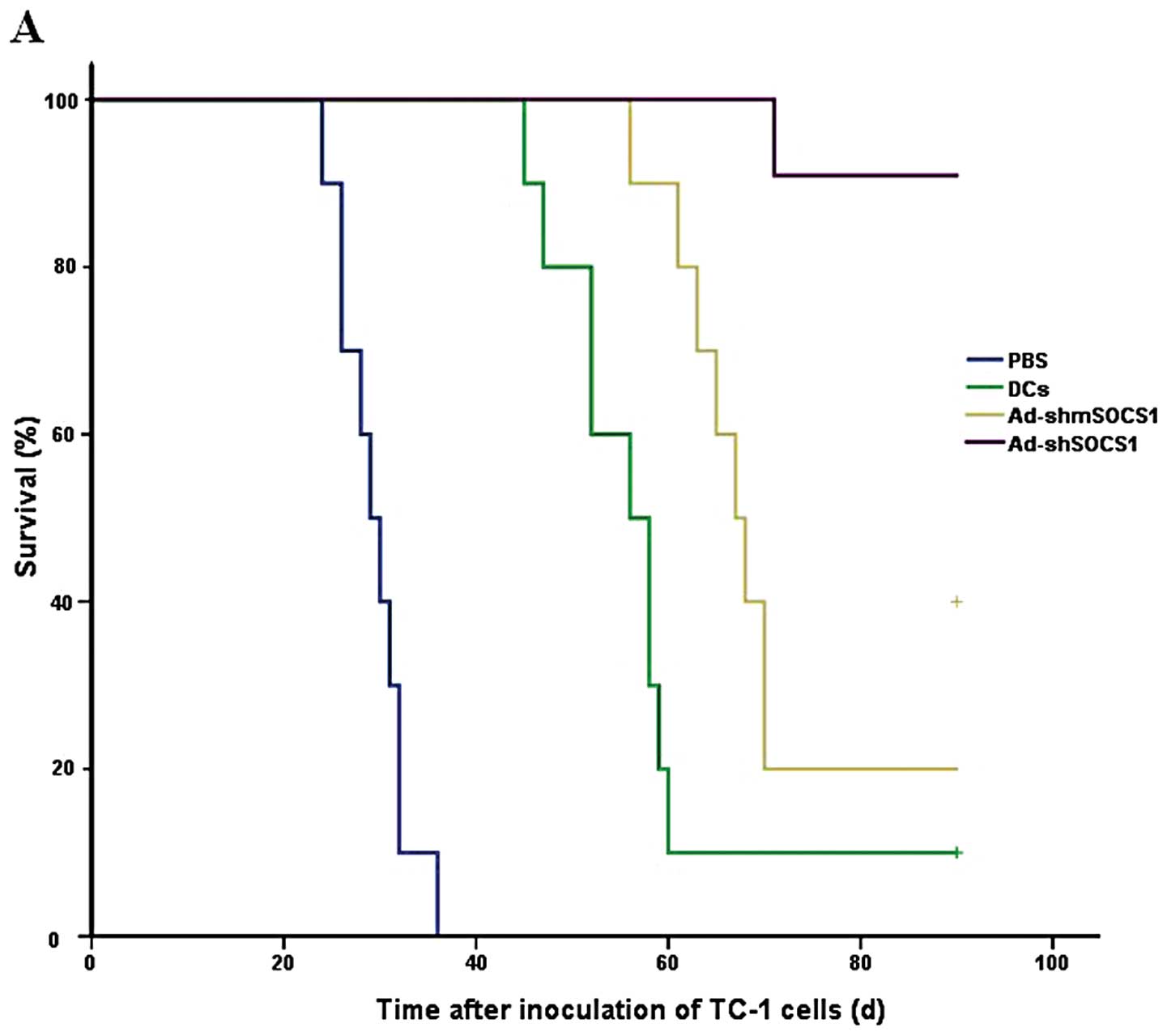
|
1.
|
Jemal A, Bray F and Center MM: Global
cancer statistics. CA Cancer J Clin. 61:69–90. 2011. View Article : Google Scholar
|
|
2.
|
Berzofsky JA, Terabe M, Oh SK, Belyakov
IM, Ahlers JD, Janik JE and Morris JC: Progress on new vaccine
strategies for the immunotherapy and prevention of cancer. J Clin
Invest. 113:1515–1525. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Ali M and Monk BJ: Vaccines against human
papillomavirus and cervical cancer: promises and challenges.
Oncologist. 10:528–538. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Kanodia S, Da Silva DM and Kast WM: Recent
advances in strategies for immunotherapy of human
papillomavirus-induced lesions. Int J Cancer. 122:247–259. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Psyrri A and Daniel D: Human
papillomavirus in cervical and head-and-neck cancer. Nat Clin Pract
Oncol. 5:24–31. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Brinton LA: Epidemiology of cervical
cancer: overview. IARC Sci Publ. 119:3–23. 1992.PubMed/NCBI
|
|
7.
|
Zur Hausen H: Papillomaviruses and cancer:
from basic studies to clinical application. Nat Rev Cancer.
2:342–350. 2002.PubMed/NCBI
|
|
8.
|
Kobayashi T and Yoshimura A: Keeping DCs
awake by putting SOCS1 to sleep. Trends Immunol. 26:177–179. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Santin AD, Hermonat PL, Ravaggi A,
Chiriva-Internati M, Zhan D and Pecorelli S: Induction of human
papillomavirus-specific CD4(+) and CD8(+) lymphocytes by E7-pulsed
autologous dendritic cells in patients with human papillomavirus
type 16- and 18-positive cervical cancer. J Virol. 73:5402–5410.
1999.PubMed/NCBI
|
|
10.
|
Kim TW, Lee JH, He L, Boyd DA, Hardwick JM
and Hung CF: Modification of professional antigen-presenting cells
with small interfering RNA in vivo to enhance cancer vaccine
potency. Cancer Res. 65:309–316. 2005.PubMed/NCBI
|
|
11.
|
Kelvin EK, Song XT, Aldrich M, Huang XF
and Chen SY: SOCS1 restricts dendritic cells’ ability to break self
tolerance and induce antitumor immunity by regulating IL-12
production and signaling. J Clin Invest. 116:90–100. 2006.
|
|
12.
|
Song XT, Evel-Kabler K, Rollins L, Aldrich
M, Gao F, Huang XF and Chen SY: An alternative and effective HIV
vaccination approach based on inhibition of antigen presentation
attenuators in dendritic cells. PLoS Med. 3:76–93. 2006.PubMed/NCBI
|
|
13.
|
Akita H, Kogure K, Moriguchi R, Nakamura Y
and Higashi T: Nanoparticles for ex vivo siRNA delivery to
dendritic cells for cancer vaccines: Programmed endosomal escape
and dissociation. J Control Release. 143:311–317. 2010. View Article : Google Scholar
|
|
14.
|
Kubo M, Hanada T and Yoshimura A:
Suppressors of cytokine signaling and immunity. Nat Immunol.
4:1169–1176. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Alexander WS and Hilton DJ: The role of
suppressors of cytokine signaling (SOCS) proteins in regulation of
the immune response. Annu Rev Immunol. 22:503–529. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Shen L, Evel-Kabler K, Strube R and Chen
SY: Silencing of SOCS1 enhances antigen presentation by dendritic
cells and antigen-specific anti-tumor immunity. Nat Biotechnol.
22:1546–1553. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Hong B, Ren W, Song XT and Evel-Kabler K:
Human suppressor of cytokine signaling 1 controls immunostimulatory
activity of monocyte-derived dendritic cells. Cancer Res.
69:8076–8084. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Hanada T, Yoshida H, Kato S, Tanaka K and
Masutani K: Suppressor of cytokine signaling-1 is essential for
suppressing dendritic cell activation and systemic autoimmunity.
Immunity. 19:437–450. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Guenterberg KD, Lesinski GB, Mundy-Bosse
BL and Karpa VI: Enhanced anti-tumor activity of interferon-alpha
in SOCS1-deficient mice is mediated by CD4+ and
CD8+ T cells. Cancer Immunol Immunother. 60:1281–1288.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Carow B, Ye Xq, Gavier-Widén D, Bhuju S
and Oehlmann W: Silencing suppressor of cytokine signaling-1
(SOCS1) in macrophages improves Mycobacterium tuberculosis
control in an IFN-γ-dependent. J Biol Chem. 286:26873–26887. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Münger K, Basile JR, Duensing S, Eichten A
and Gonzalez SL: Biological activities and molecular targets of the
human papillomavirus E7 oncoprotein. Oncogene. 20:7888–7898.
2001.PubMed/NCBI
|
|
22.
|
Cassetti MC, McElhiney SP, Shahabi V,
Pullen JK and Le Poole IC: Antitumor efficacy of Venezuelan equine
encephalitis virus replicon particles encoding mutated HPV16E6 and
E7 genes. Vaccine. 22:520–527. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Brinkman JA, Xu XM and Kast WM: The
efficacy a DNA vaccine containing inserted and replicated regions
of the E7 gene for treatment of HPV 16 induced tumors. Vaccine.
25:3437–3444. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Feltkamp MC, Smits HL, Vierboom MP,
Minnaar RP, de Jongh BM and Drijfhout JW: Vaccination with
cytotoxic T lymphocyte epitope containing peptide protects against
a tumor induced by human papillomavirus type 16-transformed cells.
Eur J Immunol. 23:2242–2249. 1993. View Article : Google Scholar
|
|
25.
|
Liu HG, Fan ZP, Chen WW, Yang HY, Liu QF
and Zhang H: A mutant HBs antigen (HbsAg) 183–191 epitope elicits
specific cytotoxic T lymphocytes in acute hepatitis B patients.
Clin Exp Immunol. 151:441–447. 2008.PubMed/NCBI
|
|
26.
|
Zheng Y, Zhang Y, Ma Y, Wan J and Shi C:
Enhancement of immunotherapeutic effects of HPV16E7 on cervical
cancer by fusion with CTLA4 extracellular region. J Microbiol.
46:728–736. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Brinkman JA, Caffrey AS and Muderspach LI:
The impact of anti HPV vaccination on cervical cancer incidence and
HPV induced cervical lesions: consequences for clinical management.
Eur J Gynaecol Oncol. 26:129–142. 2005.PubMed/NCBI
|
|
28.
|
Münger K, Phelps WC, Bubb V and Howley PM:
The E6 and E7 genes of the human papillomavirus type 16 together
are necessary and sufficient for transformation of primary human
keratinocytes. J Virol. 63:4417–4421. 1989.PubMed/NCBI
|
|
29.
|
Phelps WC, Yee CL and Munger K: The human
papillomavirus type 16 E7 gene encodes transactivation and
transformation functions similar to those of adenovirus E1A. Cell.
53:539–547. 1988. View Article : Google Scholar
|
|
30.
|
Nonn M, Schinz M, Zumbach K, Pawlita M and
Schneider A: Dendritic cell-based tumor vaccine for cervical cancer
I: in vitro vaccination with recombinant protein pulsed
dendritic cells induces specific T cells to HPV16 E7 and HPV18 E7.
J Cancer Res Clin Oncol. 129:511–520. 2003.PubMed/NCBI
|
|
31.
|
Santin AD, Bellone S, Gokden M and Cannon
MJ: Vaccination with HPV-18 E7-pulsed dendritic cells in a patient
with meta-static cervical cancer. N Engl J Med. 346:1752–1753.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Santin AD, Bellone S, Palmieri M, Zanolini
A, Ravaggi A and Siegel ER: Human papillomavirus type 16 and 18
E7-pulsed dendritic cell vaccination of stage IB or IIA cervical
cancer patients: a phase I escalating-dose trial. J Virol.
82:1968–1979. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33.
|
Ferrara A, Nonn M, Sehr P, Schreckenberger
C and Pawlita M: Dendritic cell-based tumor vaccine for cervical
cancer II: results of a clinical pilot study in 15 individual
patients. J Cancer Res Clin Oncol. 129:521–530. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Hashimoto M, Ayada T, Kinjyo I, Hiwatashi
K and Yoshida H: Silencing of SOCS1 in macrophages suppresses tumor
development by enhancing antitumor inflammation. Cancer Sci.
100:730–736. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35.
|
Judge AD, Sood V, Shaw JR, Fang D and
McClintock K: Sequence-dependent stimulation of the mammalian
innate immune response by synthetic siRNA. Nat Biotechnol.
23:457–462. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Hornung V, Guenthner-Biller M, Bourquin C
and Ablasser A: Sequence-specific potent induction of IFN-alpha by
short interfering RNA in plasmacytoid dendritic cells through TLR7.
Nat Med. 11:263–270. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Sledz CA, Holko M, de Veer MJ and
Silverman RH: Activation of the interferon system by
short-interfering RNAs. Nat Cell Biol. 5:834–839. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
38.
|
Basak SK, Kiertscher SM and Harui A:
Modifying adenoviral vectors for use as gene-based cancer vaccines.
Viral Immunol. 17:182–196. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
39.
|
Benihoud K, Yeh P and Perricaudet M:
Adenovirusvectors for gene delivery. Biotechnology. 10:440–447.
1999.PubMed/NCBI
|