Introduction
Osteosarcoma, the most common bone cancer in
children, accounting for about 5% of all childhood cancers, usually
presents in bones around the knee. About 80–90% of these tumors
develop in the ends of the long bones that form the knee. The
second most common site for these tumors is in the ends of the
upper arm bone close to the shoulder; they can also be in other
places, like the pelvis, shoulder and skull. Osteosarcoma usually
occurs in teenagers, is twice as common in males and is diagnosed
around 15 years of age (1).
Approximately 20% of children diagnosed with osteosarcoma have an
advanced stage that has metastasized to the lungs, brain and other
bones (2). If metastases are
present when the osteosarcoma is first diagnosed, the 5-year
survival rate is about 15 to 30%. The survival rate is closer to
40% if the cancer has spread only to the lungs (as opposed to
having reached other organs), or if all of the tumors (including
metastases) can be removed with surgery (3).
Pediatric soft tissue sarcomas (STSs) are a
heterogeneous group of malignant tumors that originate from
primitive mesenchymal tissue and account for 7% of all childhood
tumors (4). Rhabdomyosarcoma, a
tumor of striated muscle, is the most common soft tissue sarcoma in
children aged 0 to 14 years and accounts for 50% of tumors in this
age group (4). The remaining STSs
are a heterogeneous group of tumors and include neoplasms of
connective tissue (fibrous and adipose), peripheral nervous system,
smooth muscle (leiomyosarcomas) and vascular tissue (4). Though rhabdomyosarcoma can appear at
various sites, it primarily presents in the head and neck (35%),
the genitourinary tract (22%) and the extremities (18%) (5). Of the two main histological types of
pediatric rhabdomyosarcoma, embryonic and alveolar, embryonal is
more prevalent, contributing to roughly 53% of all diagnosed cases;
it generally presents in children under fifteen in either the head
and neck regions or the genitourinary tract (6). Alveolar rhabdomyosarcoma generally
affects the muscles of the extremities or trunk and has been found
to be more resistant to treatment and more likely to spread to
regional lymph nodes than the embryonal type (7). At diagnosis, roughly 50% of
rhabdomyosarcoma cases consist of patients five and younger and 25%
of all patients have metastatic disease (8).
Metastasis occurs secondary to cancer cell
detachment from the primary tumor, invasion through degraded
basement membrane into the surrounding stroma, and entry into and
transport through the vascular or lymphatic system to distal sites
such as the liver, lungs and brain, and extravasation, tumor cell
proliferation and angiogenesis at distal sites (9–13).
Tumor cell invasion depends upon degradation of the extra-cellular
matrix (ECM), which, when intact, acts as a barrier to block cancer
cell invasion. The ECM is composed of collagen, proteoglycans,
fibronectin, laminin and other glycoproteins (14–16).
Two families of proteases, the matrix metalloproteinases (MMPs) and
urokinase plasminogen activators (u-PA) are involved in tumor
invasion and metastasis. Numerous clinical and experimental studies
have demonstrated that elevated levels of u-PA and MMPs are
associated with cancer progression, metastasis and shortened
survival (17–20).
MMPs, especially MMP-2 and MMP-9 play key roles in
tumor cell invasion and metastasis due to their ability to degrade
type IV collagen, a major component of the ECM (16,21,22).
MMP-2 and -9 are secreted as inactive pro-enzymes in their latent
zymogenic form, and activated by other MMPs or proteases.
Proteolytic activities of MMP-2 and MMP-9 are inhibited by specific
inhibitors, tissue inhibitors of metalloproteinases (TIMPs). Thus,
a critical determinant of net proteolytic degradation is the
balance between MMP and TIMP levels. Clinical studies note the high
levels of MMP-9 expression in the highly malignant bone tumor
osteosarcoma (23,24). Of patients with osteosarcoma
treated with excision alone, 80% develop pulmonary metastases,
which suggest that micrometastases are present at diagnosis
(23). Studies of soft tissue
sarcomas also note correlation of metastasis and poor prognosis
with elevated MMP-2 and or MMP-9 and lack of TIMP-2 expression
(19).
The serine protease u-PA converts plasminogen to
plasmin, which is capable of promoting tumor growth and
angiogenesis, degrading the ECM and basement membrane and
activating pro-MMPs (25).
Components of the u-PA system such as u-PA, plasminogen activator
inhibitor-1 (PAI-1), and urokinase-type plasminogen activator
receptor (u-PAR) are overexpressed in a variety of cancer types,
most notably in breast cancer (26), but also in sarcomas (18), and correlate with cancer
progression, metastasis and poor prognosis. Thus the u-PA system
represents a potential target for anticancer strategies.
Rath and Pauling (27) proposed using nutrients such as
lysine and ascorbic acid to target plasmin-mediated connective
tissue degradation as a universal approach to tumor growth and
expansion. Binding to plasminogen active sites, lysine blocks
plasminogen activation into plasmin by tissue plasminogen activator
(t-PA). Thus it modulates the plasmin-induced MMP activation
cascade (28). Subsequent studies
confirmed this approach and led to identifying a novel formulation
composed of lysine, ascorbic acid, proline and green tea extract
and other micronutrients (NM), which has shown significant
anticancer activity against a large number (∼40) of cancer cell
lines, blocking cancer growth, tissue invasion and MMP expression
both in vitro and in vivo (29). In this study, we focused on the
modulating effect of NM on the activities of MMP-2 and -9, TIMPs
and u-PA in pediatric human sarcomas: osteosarcoma and
rhabdomyosarcoma cell lines.
Materials and methods
Materials
Human pediatric sarcoma cell lines osteosarcoma
MNNG-HOS, osteosarcoma U-2OS and embryonal rhabdomyosarcoma RD,
along with their culture media were obtained from ATCC.
Antibiotics, penicillin and fetal bovine serum (FBS), were obtained
from Gibco-BRL (Long Island, NY). Twenty-four-well tissue culture
plates were obtained from Costar (Cambrdige, MA). Gelatinase
zymography was performed in 10% Novex pre-cast SDS polyacrylamide
gel (Invitrogen Inc.) with 0.1% gelatin in non-reducing conditions.
The nutrient mixture (NM), prepared by VitaTech (Hayward, CA) was
composed of the following ingredients in the relative amounts
indicated: vitamin C (as ascorbic acid and as Mg, Ca and palmitate
ascorbate) 700 mg; L-lysine 1,000 mg; L-proline 750 mg; L-arginine
500 mg; N-acetyl cysteine 200 mg; standardized green tea extract
(80% polyphenol) 1,000 mg; selenium 30 μg; copper 2 mg;
manganese 1 mg. All other reagents used were of high quality and
were obtained from Sigma, unless otherwise indicated.
Cell cultures
The sarcoma cell lines were grown in their
respective media: osteosarcoma MNNG-HOS in MEM, osteosarcoma U-2OS
in McCoy medium, and rhabdomyosarcoma in DME, supplemented with 10%
FBS, penicillin (100 U/ml), and streptomycin (100 μg/ml) in
24-well tissue culture plates. The cells were plated at a density
of 1×105 cells/ml and grown to confluency in a
humidified atmosphere at 5% CO2 at 37°C.
Serum-supplemented media were removed and the cell monolayer was
washed once with PBS with the recommended serum-free media. The
cells were treated with the nutrient mixture, dissolved in media
and tested at 0, 50, 100, 250, 500 and 1,000 μg/ml in
triplicate at each dose for u-PA and TIMP-2 studies. For MMP
analysis, cells were treated with NM at 0, 10, 50, 100, 500 and
1,000 μg/ml. Parallel sets of cultures were treated with PMA
(100 ng/ml) for induction of MMP-9. Control and PMA treatments were
done in triplicates. The plates were then returned to the
incubator. The conditioned media were collected separately, pooled,
and centrifuged at 40°C for 10 min at 3,000 rpm to remove cells and
cell debris. The supernatant was collected and used to assess for
u-PA activity (by fibrin zymography on 10% SDS-PAGE gels containing
fibrinogen and plasminogen), MMP-2 and -9 (by gelatinase
zymography), and TIMPs (by reverse zymography).
Fibrin zymography
Fibrin zymography was used to analyze u-PA activity
on 10% SDS-PAGE gels containing fibrinogen (5.5 mg/ml) and
plasminogen (50 μg/ml). After electrophoresis, the gels were
washed twice with 2.5% Triton X-100 for 30 min. The gels were then
incubated overnight at 37°C with 0.1% glycine buffer pH 7.5 and
then stained with 0.5% Coomassie Brilliant Blue R250 and destained.
Electrophoresis of u-PA and t-PA were conducted for comparison.
Fibrin zymograms were scanned using CanoScan 9950F Canon
Scanner.
Gelatinase zymography
Gelatinase zymography was performed in 10% NOVEX
Pre-Cast SDS Polyacrylamide Gel (Invitrogen Corporation) in the
presence of 0.1% gelatin under non-reducing conditions. Culture
media (20 μl) were mixed with sample buffer and loaded for
SDS-PAGE with tris glycine SDS buffer as suggested by the
manufacturer (Novex). Samples were not boiled before
electrophoresis. Following electrophoresis the gels were washed
twice in 2.5% Triton X-100 for 30 min at room temperature to remove
SDS. The gels were then incubated at 37°C overnight in substrate
buffer containing 50 mM Tris-HCl and 10 mM CaCl2 at pH
8.0 and stained with 0.5% Coomassie Blue R250 in 50% methanol and
10% glacial acetic acid for 30 min and destained. Upon renaturation
of the enzyme, the gelatinases digest the gelatin in the gel and
give clear bands against an intensely stained background. Protein
standards were run concurrently and approximate molecular weights
were determined by plotting the relative mobilities of known
proteins.
Reverse zymography
TIMPs were analyzed by reverse zymography on 15% SDS
gels containing serum-free conditioned medium from cells. After
electrophoresis the gels were washed twice with 2.5% Triton X-100
for 30 min at room temperature to remove SDS. The gels were then
incubated at 37°C overnight in 50 mM Tris-HCl and 10 mM
CaCl2 at pH 7.6 and stained with 0.5% Coomassie Blue
R25, destained and scanned.
Scanning of gelatinase, reverse and
fibrin zymograms
Gelatinase, reverse and fibrin zymograms were
scanned using CanoScan 9950F Canon scanner at 300 dpi. The
intensity of the bands was evaluated using the pixel-based
densitometer program Un-Scan-It, Version 5.1, 32-bit, by Silk
Scientific Corporation (Orem, UT, USA), at a resolution of 1
Scanner Unit (1/100 of an inch for an image that was scanned at 100
dpi). The pixel densitometer calculates the optical density of each
pixel (values 0 to 255) using the darkly stained background of the
gel as a pixel value of 0. A logarithmic optical density scale was
used since the optical density of film and gels is logarithmically
proportional to the concentration. The pixel densitometer sums the
optical density of each pixel to give the band density.
Statistical analysis
Pearson;s correlation coefficient was determined
between NM effect on mean MMP-2 or MMP-9, u-PA and TIMP-2
expressions of sarcoma cell lines using MedCalc Software
(Mariakerke, Belgium).
Results
Table I provides an
overview of the tested pediatric sarcoma cell line u-PA, MMP and
TIMP-2 activities.
 | Table I.Overview of MMP-2, MMP-9, u-PA and
TIMP-2 expression of pediatric sarcoma cell lines. |
Table I.
Overview of MMP-2, MMP-9, u-PA and
TIMP-2 expression of pediatric sarcoma cell lines.
| Cancer cell
line | MMP-2 | MMP-9 | u-PA | TIMP-2 |
|---|
| Osteosarcoma
MNNG-HOS | + | With PMA | + | + |
| Osteosarcoma
U-2OS | + | + | + | + |
| Rhabdomyosarcoma
RD | + | + | + | + |
Effect of NM on u-PA activity in human
pediatric sarcoma cell lines
Activity of u-PA was detected in osteosarcoma
MNNG-HOS and U-2OS and rhabdomyosarcoma RD cell lines. MNNG-HOS
showed one band corresponding to subunit 1 (55 kD) and U-2OS and RD
showed bands corresponding to subunits 1 and 2 at 55 and 33 kD. NM
exerted dose response inhibition with virtual block of u-PA 1
activity at 500 μg/ml in MNNG-HOS (linear trend
R2=0.899) and 500 μg/ml (linear trend
R2=0.878) for u-PA-1 and 250 μg/ml (linear trend
R2=0.658) u-PA-2 in U-2OS. NM inhibited u-PA subunits 1
and 2 in RD dose-dependently with virtual block of u-PA-1 at 250
μg/ml and u-PA-2 at 50 μg/ml (linear trends
R2=0.667 and 0.493 for subunits 1 and 2, respectively).
See Fig. 1 for respective fibrin
zymograms and densitometry analyses.
 | Figure 1.Effect of NM on osteosarcoma
MNNG-HOS, osteosarcoma U-2OS and rhabdomyosarcoma RD u-PA
expression. Fibrin zymograms of (A) MNNG-HOS, (B) U-2OS, and (C) RD
u-PA expression. Legend: 1, u-PA; 2, markers; 3, control; 4–8, NM
50, 100, 250, 500 and 1,000 μg/ml, respectively.
Densitometric analyses of (D) MNNG-HOS, (E) U-2OS and (F) RD u-PA
expression. |
Effect of NM on MMP-2 and MMP-9
expression by osteosarcoma cell line MNNG-HOS
On gelatinase zymography, MNNG-HOS cells
demonstrated strong expression of MMP-2 and induced MMP-9 with PMA
(100 ng/ml) treatment that were inhibited by NM in a dose-dependent
fashion with virtual total inhibition of MMP-2 and MMP-9 at 100
μg/ml (linear trends R2=0.675 and 0.559,
respectively) See Fig. 2 for
gelatinase zymograms and densitometry analyses.
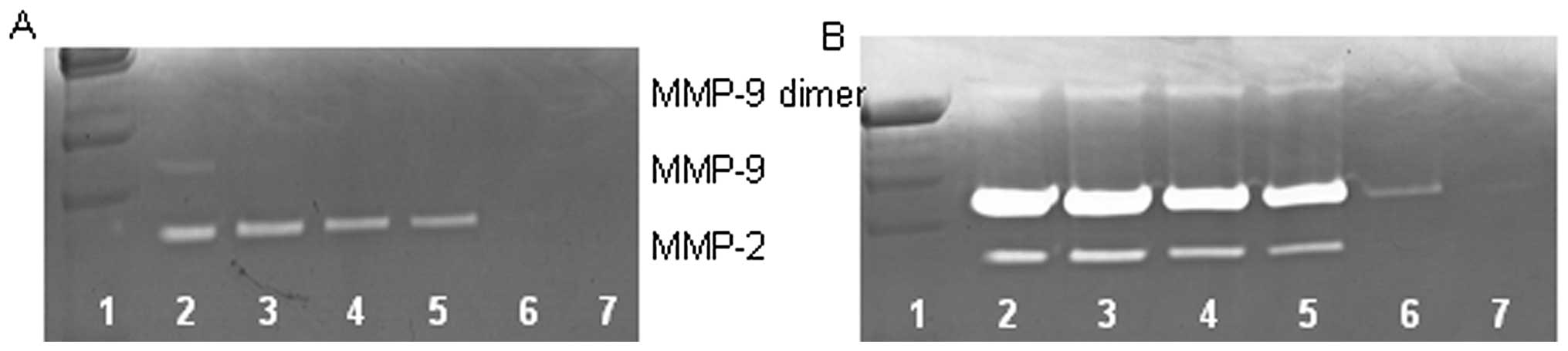
Effect of NM on MMP-2 and MMP-9
expression by osteosarcoma cell line U-2OS
On gelatinase zymography, U-2OS cells demonstrated
strong expression of MMP-9 and slight expression of MMP-2 with and
without PMA (100 ng/ml) treatment that were inhibited by NM in a
dose-dependent fashion with virtual total inhibition of MMP-2 at
500 μg/ml (linear trend R2=0.824) and MMP-9 at
1,000 μg/ml (linear trend R2=0.816). See Fig. 3 for gelatinase zymograms and
densitometry analyses.
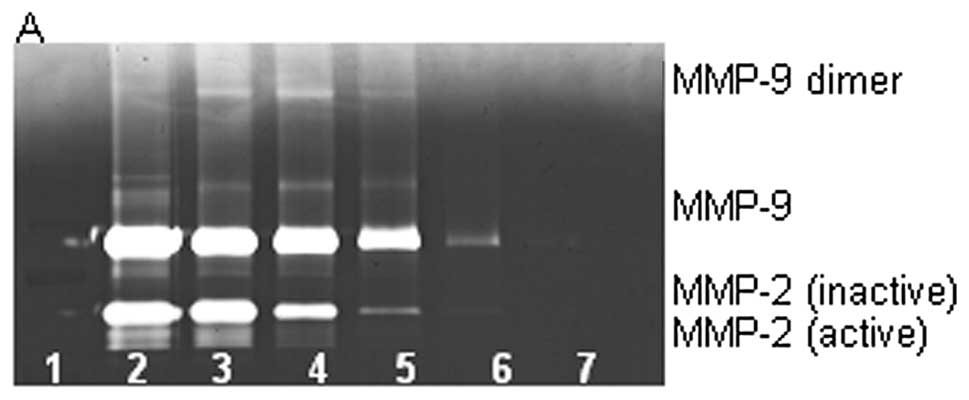
Effect of NM on MMP-2 and MMP-9
expression by rhabdomyosarcoma cell line RD
Zymography demonstrated strong expression of MMP-2
and slight expression of MMP-9 by RD cells that were inhibited by
NM in a dose-dependent fashion with virtual total inhibition of
MMP-2 at 500 μg/ml (linear trend R2=0.899) and
MMP-9 at 10 μg/ml (linear trend R2=0.429). PMA
(100 ng/ml) treatment profoundly enhanced MMP-9 expression and
MMP-9 dimer by RD cells and decreased MMP-2 expression; NM
inhibited MMP-2 and MMP-9 in a dose-dependent manner with total
block of MMP-2 and MMP-9 and MMP-9 dimer at 500 μg/ml
(linear trends R2=0.876, 0.769 and 0.800, respectively).
See Fig. 4 for gelatinase
zymograms and densitometry analyses.
Effect of NM on TIMP activity in
osteosarcoma MNNG-HOS and U-2OS and rhabdomyosarcoma RD
Reverse zymography revealed upregulation of TIMP-2
activity with NM treatment in all cancer cell lines in a
dose-dependent manner. Minimum activity was expressed at 50 and
maximum at 1,000 μg/ml NM. See Fig. 5 for respective reverse zymograms
and densitometry analyses.
 | Figure 5.Effect of NM on osteosarcoma
MNNG-HOS, osteosarcoma U-2OS and rhabdomyosarcoma RD TIMP-2
expression. Reverse zymograms of (A) MNNG-HOS, (B) U-2OS and (C) RD
TIMP-2 expression. Legend: 1, markers; 2, control; 3–7, NM 50, 100,
250, 500 and 1,000 μg/ml, respectively. Densitometric
analyses of (D) MNNG-HOS, (E) U-2OS and (F) RD TIMP-2
expression. |
Correlation between pediatric sarcoma
u-PA, TIMP-2 and MMP expressions
Analysis revealed a positive correlation between
u-PA and MMP-2 expressions of NM-treated osteosarcoma MNNG-HOS and
between u-PA and MMP-2 and MMP-9 expressions of osteosarcoma U-2OS
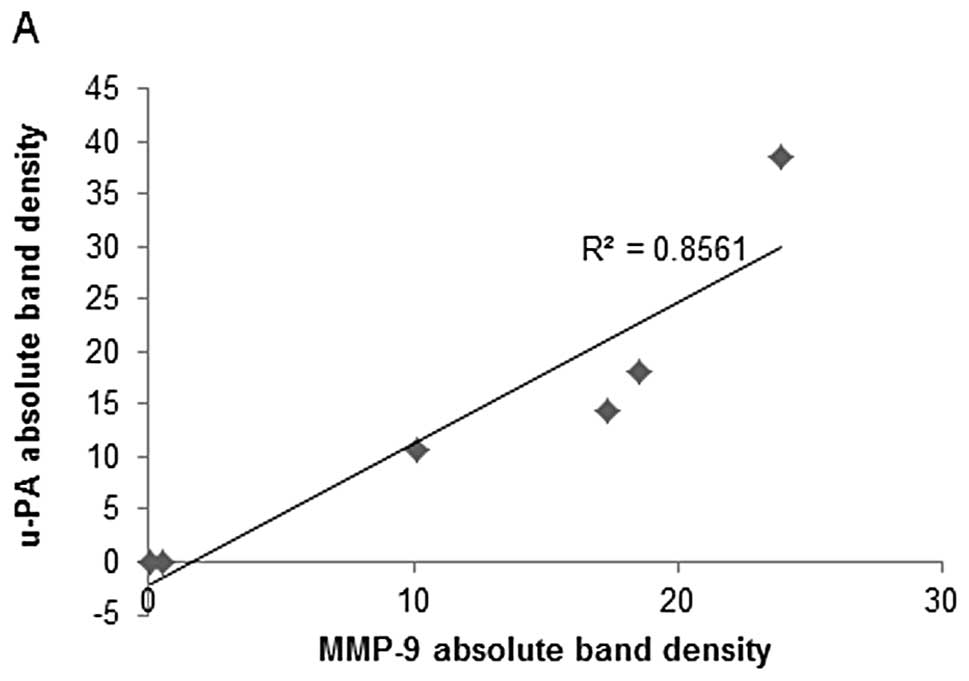
and rhabdomyosarcoma RD, as shown in Table II. Fig. 6A shows the correlation graph for
osteosarcoma U-2OS u-PA and MMP-2/MMP-9 with correlation
coefficients r=0.925 (MMP-9) and 0.877 (MMP-2). Negative
correlations were found between the expressions of MMP-2 or MMP-9
and TIMP-2 in all pediatric sarcoma cell lines treated with NM that
secreted TIMP-2, as shown in Table
II. The correlation (r=−0.740) between MMP-9 and TIMP-2 is
shown for osteosarcoma U-2OS in Fig.
6B. Negative correlations were found between expressions of
TIMP-2 and u-PA in all NM-treated sarcoma cell lines studied. The
correlation (r=−0.603) between u-PA and TIMP-2 is shown for
osteosarcoma U-2OS in Fig. 6C.
 | Table II.Correlation between effects of NM on
sarcoma cell u-PA, MMPs and TIMPs. |
Table II.
Correlation between effects of NM on
sarcoma cell u-PA, MMPs and TIMPs.
| Cell line | u-PA and MMPs | MMPs and TIMPs | u-PA and TIMPs |
|---|
| Osteosarcoma
MNNG-HOS | r=0.845
(MMP-2) | r=−0.696
(MMP-2) | r=−0.623 |
| Osteosarcoma
U-2OS | r=0.925
(MMP-9) | r=−0.740
(MMP-9) | r=−0.603 |
| r=0.877
(MMP-2) | | |
| Rhabdomyosarcoma
RD | r=0.511
(MMP-9) | r=−0.868
(MMP-2) | r=−0.423 |
| r=0.660
(MMP-2) | | |
Discussion
Two families of proteases, urokinase plasminogen
activators and matrix metalloproteinases, play key roles in tumor
cell invasion and metastasis. Urokinase plasminogen has a
demonstrated role as an initiator of ECM proteolysis and associated
tumor cell invasion (28). The
protease u-PA cleaves plasminogen to plasmin, which is capable of
promoting tumor growth and angiogenesis, degrading the ECM and
basement membrane and activating pro-MMPs (25,30).
The uPA-uPAR system is a key regulator of osteosarcoma invasion and
an inverse relationship has been demonstrated between u-PA levels
and survival time (18,30). Overexpression of u-PA in
soft-tissue sarcoma patients has also been correlated with cancer
progression, metastasis and poor prognosis (20). Matrix metalloproteinases,
especially MMP-2 and MMP-9, are also key regulators of tumor cell
invasion and metastasis due to their ability to degrade type IV
collagen, a major component of the ECM. Overproduction of MMPs,
especially MMP-2 and -9 and low levels of TIMPs have been shown to
be associated with a more aggressive behavior of sarcomas (19,23,31,32).
For example, increased expression of MMP-9 has been found to
correlate with osteosarcoma metastasis in patients and inhibitors
of MMPs, such as TIMP-1 have been shown to inhibit invasiveness of
osteosarcoma tumor cells in vitro (23,24,31).
A study of the immunohistochemical expression of MMPs and TIMPS in
human rhabdomyosarcoma demonstrated strong MMP-1, -3 and -9
expression in rhabdomyosarcoma, alveolar RMS greater than embryonal
RMS. Intratumor vessels and perivascular ECM were positive for
MMP-9 and negative for TIMPS in both types (33).
Our study demonstrated that the specific mixture of
nutrients tested significantly inhibited u-PA secretion in
osteosarcoma MNNG-HOS and U-2OS and in rhabdomyosarcoma RD cell
lines. Furthermore, the NM demonstrated dose-dependent decrease in
MMP secretion and increase in TIMP-2 secretion by all sarcoma cell
lines. As expected, a significant positive correlation was found
between the secretion of u-PA and MMP-2/MMP-9 and a significant
negative correlation between u-PA and TIMP-2 and between
MMP-2/MMP-9 and TIMP-2 secretion by NM treatment of osteosarcoma
and rhabdomyosarcoma cells. Furthermore, a previous study
demonstrated significant correlation between NM inhibition of
Matrigel invasion and NM modulation of the MMP-2 and -9 activities
of the sarcoma cell lines studied (34). A significant negative correlation
was found between NM modulation of Matrigel invasion inhibition and
MMP-2 secretion with osteosarcoma MNNG-HOS (r=−0.6531),
osteosarcoma U-2OS (r=−0.835) and rhabdomyosarcoma RD (r=−0.675). A
previous in vivo study of the effects of NM 0.5% on
xenograft tumor growth of osteosarcoma MNNG-HOS cells in nude mice
support these results in that it demonstrated significant
inhibition of xenograft tumor growth (53%, p=0.0001), tumor
vascularity and VEGF and MMP-9 tumor tissue staining compared to
tumors of mice in the control diet group (35).
The standard treatments for sarcomas include
surgery, chemotherapy and radiation with severe associated toxicity
and limited efficacy, clearly indicating a need for safe and
effective therapeutic approaches to control progression and
metastasis, especially in these young sarcoma patients. Extensive
research documents the efficacy and safety of dietary and botanical
natural compounds in cancer prevention (36). The nutrient mixture was formulated
by selecting nutrients that act on critical physiological targets
in cancer progression and metastasis, as documented in both
clinical and experimental studies. Combining these micronutrients
expands metabolic targets, maximizing biological impact with lower
doses of components. A previous study of the comparative effects of
NM, green tea extract and EGCG on inhibition of MMP-2 and MMP-9
secretion of different cancer cell lines with varying MMP secretion
patterns, revealed the superior potency of NM over GTE and EGCG at
equivalent doses (37). These
results can be understood from the more comprehensive treatment
offered by the combination of nutrients in NM over individual
components of NM since MMP-2 and MMP-9 are mediated by differential
pathways.
Optimal ECM structure depends upon adequate supplies
of ascorbic acid and the amino acids lysine and proline to ensure
proper synthesis and hydroxylation of collagen fibers. In addition,
lysine contributes to ECM stability as a natural inhibitor of
plasmin-induced proteolysis (27,38).
Manganese and copper are also essential for collagen formation.
There is considerable documentation of the potency of green tea
extract in modulating cancer cell growth, metastasis, angiogenesis,
and other aspects of cancer progression (39–45).
N-acetyl cysteine and selenium have demonstrated inhibition of
tumor cell MMP-9 and invasive activities, as well as migration of
endothelial cells through ECM (46–48).
Ascorbic acid demonstrates cytotoxic and antimetastatic actions on
malignant cell lines (49–53) and cancer patients have been found
to have low levels of ascorbic acid (54,55).
Low levels of arginine, a precursor of nitric oxide (NO), can limit
the production of NO, which has been shown to predominantly act as
an inducer of apoptosis (56).
In conclusion, the NM demonstrated potent anticancer
activity by targeting primary mechanisms responsible for the
aggressive spread of pediatric sarcomas. In this in vitro
study, the NM significantly inhibited secretion of u-PA and MMP-2
and/or -9 and increased secretion of TIMP-2 in osteosarcoma and
rhabdomyosarcoma cells, suggesting its potential in modulating
cancer invasion and metastasis. NM inhibition of MMP secretion was
found to be correlated significantly with Matrigel invasion of all
the sarcoma cell lines studied (34). Furthermore, use of the nutrient
mixture would not pose any toxic effect clinically, especially in
the relevant doses, as in vivo safety studies demonstrate.
An in vivo toxicology study (Roomi et al, J AM Coll
Nutr 22: abs. 86, 2003) showed that NM had no adverse effects on
vital organs (heart, liver and kidney), or on the associated
functional serum enzymes.
Acknowledgements
Mr. Monterrey provided assistance in
scanning the gels. The research study was funded by Dr Rath Health
Foundation (Santa Clara, CA, USA) a non-profit organization.
References
|
1.
|
National Institute of Health: PubMed
Health: Osteosarcoma. http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0002616/.
Accessed March 18, 2013.
|
|
2.
|
Kaste SC, Pratt CB, Cain AM, Jones-Wallace
DJ and Rao BN: Metastases detected at the time of diagnosis of
primary pediatric extremity osteosarcoma at diagnosis: imaging
features. Cancer. 86:1602–1608. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
American Cancer Society: Osteosarcoma:
What are the survival rates for osteosarcoma? http://www.cancer.org/cancer/osteosarcoma/detailedguide/osteosarcoma-survival-rates.
Accessed March 18, 2013.
|
|
4.
|
National Cancer Institute: Childhood Soft
Tissue Sarcoma Treatment (PDQ®). http://www.cancer.gov/cancertopics/pdq/treatment/child-soft-tissue-sarcoma/HealthProfessional/.
Accessed March 18, 2013.
|
|
5.
|
Barr FG: Molecular genetics and
pathogenesis of rhabdomyosarcoma. J Pediatr Hematol Oncol.
19:483–491. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Patham DM: Pathologic classification of
rhabdomyosarcoma and correlations with molecular studies. Med
Pathol. 14:506–514. 2001. View Article : Google Scholar
|
|
7.
|
Mandell L, Ghavinni F, LaQuaglia M and
Exelby P: Prognostic significance of regional lymph node
involvements in childhood extremity rhabdomyosarcoma. Med Pediatr
Oncol. 18:466–471. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Koscielniak E, Rodary C, Flamant F, Carli
M, Treuner J, Pinkerton CR and Grono P: Metastatic rhabdomyosarcoma
and histologically similar tumors in childhood: a retrospective
European multi-center analysis. Med Pediatr Oncol. 20:209–214.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Fidler IJ: Molecular biology of cancer:
invasion and metastasis. Cancer Principles and Practice of
Oncology. De Vita VT, Hellman S and Rosenberg SA: 5th edition.
Lippincott-Raven; Philadelphia, PA: pp. 135–152. 1997
|
|
10.
|
Egeblad M and Werb Z: New functions for
the matrix metalloproteinases in cancer progression. Nat Rev
Cancer. 2:161–174. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Folkman J: Role of angiogenesis in tumor
growth and metastasis. Semin Oncol. 29(Suppl 16): 15–18. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Chambers AF and Matrisian LM: Changing
views on the role of matrix metalloproteinases in metastasis. J
Natl Cancer Inst. 89:1260–1270. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Kleiner DL and Stetler-Stevenson WG:
Matrix metalloproteinases and metastasis. Cancer Chemother
Pharmacol. 43(Suppl): 42s–51s. 1999. View Article : Google Scholar
|
|
14.
|
Yurchenko PD and Schitny JC: Molecular
architecture of basement membranes. FASEB J. 4:1577–1590.
1990.PubMed/NCBI
|
|
15.
|
Barsky SH, Siegel GP, Jannotta F and
Liotta LA: Loss of basement membrane components by invasive tumors
but not by their benign counterparts. Lab Investig. 49:140–147.
1983.PubMed/NCBI
|
|
16.
|
Liotta LA, Tryggvason K, Garbisa A, Hart
I, Foltz CM and Shafie S: Metastatic potential correlates with
enzymatic degradation of basement membrane collagen. Nature.
284:67–68. 1980. View
Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Nelson AR, Fingleton B, Rothenberg ML and
Matrisian LM: Matrix metalloproteinases: biologic activity and
clinical implications. J Clin Oncol. 18:1135–1149. 2000.PubMed/NCBI
|
|
18.
|
Choong PF, Fernö M, Åkermans M, Willién H,
Lǻngström E, Gustafson P, Alvegǻrd T and Rydholm A:
Urokinaseplasminogen-activator levels and prognosis in 69
soft-tissue sarcomas. Int J Cancer. 69:268–272. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Benassi MS, Magagnoli G, Ponticelli F,
Pazzaglia L, Zanella L, Gamberti G, Ragazzini P, Ferrari C, Mercuri
M and Picci P: Tissue and serum loss of metalloproteinases
inhibitors in high-grade soft tissue sarcomas. Histol Histopathol.
18:1035–1040. 2003.PubMed/NCBI
|
|
20.
|
Taubert H, Würl P, Greither T, Kappler M,
Bache M, Lautenschläger C, Füssel S, Meye A, Eckert AW, Holzhausen
HJ, Magdolen V and Kotzsch M: Co-detection of members of the
urokinase plasminogen activator system in tumour tissue and serum
correlates with a poor prognosis for soft-tissue sarcoma patients.
Br J Cancer. 102:731–737. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Stetler-Stevenson WG: The role of matrix
metalloproteinases in tumor invasion, metastasis and angiogenesis.
Surg Oncol Clin N Am. 10:383–392. 2001.PubMed/NCBI
|
|
22.
|
Stetler-Stevenson WG: Type IV collagenases
in tumor invasion and metastasis. Cancer Metastasis Rev. 9:289–303.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Himelstein BP, Asada N, Carlton MR and
Collins MH: Matrix metalloproteinase-9 (MMP-9) expression in
childhood osseous osteosarcoma. Med Pediatr Oncol. 31:471–474.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Ferrari C, Benassi S, Ponticelli F,
Gamberi G, Ragazzini P, Pazzaglia L, Balladelli A, Bertoni F and
Picci P: Role of MMP-9 and its tissue inhibitor TIMP-1 in human
osteosarcoma: findings in 42 patients followed for 1–16 years. Acta
Orthop Scand. 75:487–491. 2004.PubMed/NCBI
|
|
25.
|
Dano K, Andreasen PA, Grondahl-Hansen J,
Kristensen P, Nielsen LS and Skriver L: Plasminogen activators,
tissue degradation and cancer. Adv Cancer Res. 44:139–266. 1985.
View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Duffy MJ, Duggan C, Mulcahy HE, McDermott
EW and O’Higgins NJ: Urokinase plasminogen activator: a prognostic
marker in breast cancer including patients with axillary
node-negative disease. Clin Chem. 44:1177–1183. 1998.PubMed/NCBI
|
|
27.
|
Rath M and Pauling L: Plasmin-induced
proteolysis and the role of apoprotein(a), lysine and synthetic
analogs. Orthomolecular Med. 7:17–23. 1992.
|
|
28.
|
Andreasen PA, Kjøller L, Christensen L and
Duffy MJ: The urokinase-type plasminogen activator system in cancer
metastasis: a review. Int J Cancer. 72:1–22. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Niedzwiecki A, Roomi MW, Kalinovsky T and
Rath M: Micronutrient synergy - a new tool in effective control of
metastasis and other key mechanisms of cancer. Cancer Metastasis
Rev. 29:529–543. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Choong PFM and Nadesapillai AP: Urokinase
plasminogen activator system: a multifunctional role in tumor
progression and metastasis. Clin Orthop Relat Res. 415:S46–S58.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Bjornland K, Flatmark K, Pettersen S,
Aaasen AO, Fodstad O and Maelandsmo GM: Matrix metalloproteinases
participate in osteosarcoma invasion. J Surg Res. 127:151–156.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Roebuck MM, Helliwell TR, Chaudhry IH,
Kalogrianitis S, Carter S, Kemp G, Ritchie DA, Jane MJ and Frostick
SP: Matrix metalloproteinase expression is related to angiogenesis
and histologic grade in spindle cell soft tissue neoplasms of the
extremities. Am J Clin Path. 123:405–414. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33.
|
Diomedi-Carnassei F, Boldrini R, Rava L,
Donfrancesco A, Boglino C, Messina E, Dominici C and Callea F:
Different patterns of matrix metalloproteinase expression in
alveolar versus embryonal rhabdomyosarcoma. J Pediatr Surg.
39:1673–1679. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Roomi MW, Monterrey JC, Kalinovsky T,
Niedzwiecki A and Rath M: Inhibition of invasion and MMPs by a
nutrient mixture in human cancer cell lines: a correlation study.
Exp Oncol. 32:243–248. 2010.PubMed/NCBI
|
|
35.
|
Roomi MW, Ivanov V, Kalinovsky T,
Niedzwiecki A and Rath M: Effect of ascorbic acid, lysine, proline
and green tea extract on human osteosarcoma cell line MNNG-HOS
xenografts in nude mice. Med Oncol. 23:411–417. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Amin ARMR, Kucek O, Khuri FR and Shin DM:
Perspectives for cancer prevention with natural compounds. J Clin
Oncol. 27:2712–2725. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Roomi MW, Monterrey JC, Kalinovsky T, Rath
M and Niedzwiecki A: Comparative effects of EGCG, green tea and a
nutrient mixture on the patterns of MMP-2 and MMP-9 expression in
cancer cell lines. Oncol Rep. 24:747–757. 2010.PubMed/NCBI
|
|
38.
|
Sun Z, Chen YH, Wang P, Zhang J, Gurewich
V, Zhang P and Liu JN: The blockage of high-affinity lysine binding
sites of plasminogen by EACA significantly inhibits
prourokinase-induced plasminogen activation. Biochem Biophys Acta.
1596:182–192. 2002.PubMed/NCBI
|
|
39.
|
Kemberling JK, Hampton JA, Keck RW, Gomez
MA and Selman SH: Inhibition of bladder tumor growth by the green
tea derivative epigallocatechin-3-gallate. J Urol. 170:773–776.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
40.
|
Sato D and Matsushima M: Preventive
effects of urinary bladder tumors induced by
N-butyl-N-(4-hydroxybutyl)-nitrosamine in rat by green tea leaves.
Int J Urol. 10:160–166. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
41.
|
Valcic S, Timmermann BN, Alberts DS,
Wachter GA, Krutzsch M, Wymer J and Guillen JM: Inhibitory effect
of six green tea catechins and caffeine on the growth of four
selected human tumor cell lines. Anticancer Drugs. 7:461–468. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
42.
|
Mukhtar H and Ahmed N: Tea polyphenols:
prevention of cancer and optimizing health. Am J Clin Nutr.
71:1698s–1702s. 2000.PubMed/NCBI
|
|
43.
|
Yang GY, Liao J, Kim K, Yurtow EJ and Yang
CS: Inhibition of growth and induction of apoptosis in human cancer
cell lines by tea polyphenols. Carcinogenesis. 19:611–616. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
44.
|
Taniguchi S, Fujiki H, Kobayashi H, Go H,
Miyado K, Sadano H and Shimikawa R: Effect of (−) epigallocatechin
gallate, the main constituent of green tea, on lung metastasis with
mouse B16 melanoma cell lines. Cancer Lett. 65:51–54. 1992.
|
|
45.
|
Hara Y: Green Tea: Health Benefits and
Applications. Marcel Dekker; New York, Basel: 2001, View Article : Google Scholar
|
|
46.
|
Kawakami S, Kageyama Y, Fujii Y, Kihara K
and Oshima H: Inhibitory effects of N-acetyl cysteine on invasion
and MMP 9 production of T24 human bladder cancer cells. Anticancer
Res. 21:213–219. 2001.PubMed/NCBI
|
|
47.
|
Morini M, Cai T, Aluigi MG, Noonan DM,
Masiello L, De Floro S, D’Agostinin F, Albini A and Fassima G: The
role of the thiol N-acetyl cysteine in the prevention of tumor
invasion and angiogenesis. Int J Biol Markers. 14:268–271.
1999.PubMed/NCBI
|
|
48.
|
Yoon SO, Kim MM and Chung AS: Inhibitory
effects of selenite on invasion of HT 1080 tumor cells. J Biol
Chem. 276:20085–20092. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
49.
|
Naidu KA, Karl RC and Coppola D:
Antiproliferative and proapoptotic effect of ascorbyl stearate in
human pancreatic cancer cells: association with decreased
expression of insulin-like growth factor 1 receptor. Dig Dis Sci.
48:230–237. 2003. View Article : Google Scholar
|
|
50.
|
Anthony HM and Schorah CJ: Severe
hypovitaminosis C in lung-cancer patients: the utilization of
vitamin C in surgical repair and lymphocyte-related host
resistance. Br J Cancer. 46:354–367. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
51.
|
Maramag C, Menon M, Balaji KC, Reddy PG
and Laxmanan S: Effect of vitamin C on prostate cancer cells in
vitro: effect on cell number, viability and DNA synthesis.
Prostate. 32:188–195. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
52.
|
Koh WS, Lee SJ, Lee H, Park C, Park MH,
Kim WS, Yoon SS, Park K, Hong SI, Chung MH and Park CH:
Differential effects and transport kinetics of ascorbate
derivatives in leukemic cell lines. Anticancer Res. 8:2487–2493.
1998.PubMed/NCBI
|
|
53.
|
Chen Q, Espey MG, Krishna MC, Mitchell JB,
Corpe CP, Buettner GR, Shacter E and Levine M: Pharmacologic
ascorbic acid concentrations selectively kill cancer cells: action
as a pro-drug to deliver hydrogen peroxide to tissues. Proc Natl
Acad Sci USA. 102:13604–13609. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
54.
|
Nunez C, Ortiz de Apodaca Y and Ruiz A:
Ascorbic acid in the plasma and blood cells of women with breast
cancer. The effect of consumption of food with an elevated content
of this vitamin. Nutr Hosp. 10:68–372. 1995.PubMed/NCBI
|
|
55.
|
Kurbacher CM, Wagner U, Kolster B,
Andreotti PE, Krebs D and Bruckner HW: Ascorbic acid (vitamin C)
improves the antineoplastic activity of doxorubicin, cisplatin and
paclitaxel in human breast carcinoma cells in vitro. Cancer Lett.
103:183–189. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
56.
|
Cooke JP and Dzau VJ: Nitric oxide
synthase: role in the genesis of vascular disease. Annu Rev Med.
48:489–509. 1997. View Article : Google Scholar : PubMed/NCBI
|