|
1.
|
Bruera G, Santomaggio A, Cannita K,
Lanfiuti Baldi P, Tudini M, De Galitiis F, Mancini M, Marchetti P,
Antonucci A, Ficorella C and Ricevuto E: ‘Poker’ association of
weekly alternating 5-Fluorouracil, Irinotecan, Bevacizumab and
Oxaliplatin (FIr-B/FOx) in first line treatment of metastatic
colorectal cancer: a phase II study. BMC Cancer. 10:672010.
|
|
2.
|
Bruera G and Ricevuto E: Intensive
chemotherapy of metastatic colorectal cancer: weighing between
safety and clinical efficacy. Evaluation of Masi G, Loupakis F,
Salvatore L, et al. Bevacizumab with FOLFOXIRI (irinotecan,
oxaliplatin, fluorouracil, and folinate) as first-line treatment
for metastatic colorectal cancer: a phase 2 trial. Lancet Oncol.
2010.11:845–52, Expert Opin Biol Ther 11: 821–824, 2011.
|
|
3.
|
Bruera G, Cannita K, Giuliante F, Lanfiuti
Baldi P, Vicentini R, Marchetti P, Nuzzo G, Antonucci A, Ficorella
C and Ricevuto E: Effectiveness of liver metastasectomies in
patients with metastatic colorectal cancer treated with FIr-B/FOx
triplet chemotherapy plus bevacizumab. Clin Colorectal Cancer.
11:119–126. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Ficorella C, Bruera G, Cannita K, Porzio
G, Lanfiuti Baldi P, Tinari N, Natoli C and Ricevuto E: Triplet
chemotherapy in patients with metastatic colorectal cancer: toward
the best way to safely administer a highly active regimen in
clinical practice. Clin Colorectal Cancer. 11:229–237. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Bruera G, Cannita K, Di Giacomo D, Lamy A,
Troncone G, Dal Mas A, Coletti G, Frébourg T, Sabourin JC, Tosi M,
Ficorella C and Ricevuto E: Prognostic value of KRAS genotype in
metastatic colorectal cancer (MCRC) patients treated with intensive
triplet chemotherapy plus bevacizumab (FIr-B/FOx) according to
extension of metastatic disease. BMC Med. 10:1352012. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Masi G, Vasile E, Loupakis F, Cupini S,
Fornaro L, Baldi G, Salvatore L, Cremolini C, Stasi I, Brunetti I,
Fabbri MA, Pugliesi M, Trenta P, Granetto C, Chiara S, Fioretto L,
Allegrini G, Crinò L, Andreuccetti M and Falcone A: Randomized
trial of two induction chemotherapy regimens in metastatic
colorectal cancer: an updated analysis. J Natl Cancer Inst.
103:21–30. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Tournigand C, André T, Achille E, Lledo G,
Flesh M, Mery-Mignard D, Quinaux E, Couteau C, Buyse M, Ganem G,
Landi B, Colin P, Louvet C and de Gramont A: FOLFIRI followed by
FOLFOX6 or the reverse sequence in advanced colorectal cancer: a
randomized GERCOR study. J Clin Oncol. 22:229–237. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Grothey A, Sargent D, Goldberg RM and
Schmoll HJ: Survival of patients with advanced colorectal cancer
improves with the availability of fluorouracil-leucovorin,
irinotecan, and oxaliplatin in the course of treatment. J Clin
Oncol. 22:1209–1214. 2004. View Article : Google Scholar
|
|
9.
|
Grothey A, Sugrue MM, Purdie DM, Dong W,
Sargent D, Hedrick E and Kozloff M: Bevacizumab beyond first
progression is associated with prolonged overall survival in
metastatic colorectal cancer: results from a large observational
cohort study (BRiTE). J Clin Oncol. 26:5326–5334. 2008. View Article : Google Scholar
|
|
10.
|
Masi G, Marcucci L, Loupakis F, Cerri E,
Barbara C, Bursi S, Allegroni G, Brunetti IM, Murr R, Ricci S,
Cupini S, Andreuccetti M and Falcone A: First-line
5-fluorouracil/folinic acid, oxaliplatin and irinotecan (FOLFOXIRI)
does not impair the feasibility and the activity of second line
treatments in meta-static colorectal cancer. Ann Oncol.
17:1249–1254. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Fornaro L, Vasile E, Masi G, Loupakis F,
Baldi GG, Allegrini G, Salvatore L, Cremolini C, Cupini S, Cortesi
E, Tuzi A, Granetto C, Brunetti IM, Ricci S and Falcone A: Outcome
of second-line treatment after first-line chemotherapy with the
GONO FOLFOXIRI regimen. Clin Colorectal Cancer. 11:71–76. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Cunningham D, Pyrhönen S, James RD, Punt
CJ, Hickish TF, Heikkila R, Johannesen TB, Starkhammar H, Topham
CA, Awad L, Jacques C and Herait P: Randomised trial of irinotecan
plus supportive care versus supportive care alone after
fluorouracil failure for patients with metastatic colorectal
cancer. Lancet. 352:1413–1418. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Fuchs CS, Moore MR, Harker G, Villa L,
Rinaldi D and Hecht JR: Phase III comparison of two irinotecan
dosing regimens in second-line therapy of metastatic colorectal
cancer. J Clin Oncol. 21:807–814. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Kim GP, Sargent DJ, Mahoney MR, Rowland KM
Jr, Philip PA, Mitchell E, Mathews AP, Fitch TR, Goldberg RM,
Alberts SR and Pitot HC: Phase III noninferiority trial comparing
irinotecan with oxaliplatin, fluorouracil, and leucovorin in
patients with advanced colorectal carcinoma previously treated with
fluorouracil: N9841. J Clin Oncol. 27:2848–2854. 2009. View Article : Google Scholar
|
|
15.
|
Haller DG, Rothenberg ML, Wong AO,
Koralewski PM, Miller WH Jr, Bodoky G, Habboubi N, Garay C and
Olivato LO: Oxaliplatin plus irinotecan compared with irinotecan
alone as second-line treatment after single-agent fluoropyrimidine
therapy for metastatic colorectal carcinoma. J Clin Oncol.
26:4544–4550. 2008. View Article : Google Scholar
|
|
16.
|
Giantonio BJ, Catalano PJ, Meropol NJ,
O’Dwyer PJ, Mitchell EP, Alberts SR, Schwartz MA and Benson AB III:
Bevacizumab in combination with oxaliplatin, fluorouracil, and
leucovorin (FOLFOX4) for previously treated metastatic colorectal
cancer: results from the Eastern Cooperative Oncology Group Study
E3200. J Clin Oncol. 25:1539–1544. 2007. View Article : Google Scholar
|
|
17.
|
Jonker DJ, O’Callaghan CJ, Karapetis CS,
Zalcberg JR, Tu D, Au HJ, Berry SR, Krahn M, Price T, Simes RJ,
Tebbutt NC, van Hazel G, Wierzbicki R, Langer C and Moore MJ:
Cetuximab for the treatment of colorectal cancer. N Engl J Med.
357:2040–2048. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Saltz LB, Meropol NJ, Loehrer PJ, Needle
MN, Kopit J and Mayer RJ: Phase II trial of cetuximab in patients
with refractory colorectal cancer that expresses the epidermal
growth factor receptor. J Clin Oncol. 22:1201–1208. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Saltz LB, Meropol NJ, Loehrer PJ, Needle
MN, Kopit J and Mayer RJ: Cetuximab monotherapy and Cetuximab plus
Irinotecan in Irinotecan refractory metastatic colorectal cancer. N
Engl J Med. 351:337–345. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Sobrero AF, Maurel J, Fehrenbacher L,
Scheithauer W, Abubakr YA, Lutz MP, Vega-Villegas ME, Eng C,
Steinhauer EU, Prausova J, Lenz HJ, Borg C, Middleton G, Kroning H,
Luppi G, Kisker O, Zubel A, Langer C, Kopit J and Burris HA III:
EPIC: phase III trial of cetuximab plus irinotecan after
fluoropyrimidine and oxaliplatin failure in patients with
metastatic colorectal cancer. J Clin Oncol. 26:2311–2319. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Karapetis CS, Khambata-Ford S, Jonker DJ,
O’Callaghan CJ, Tu D, Tebbutt NC, Simes RJ, Chalchal H, Shapiro JD,
Robitaille S, Price TJ, Shepherd L, Au HJ, Langer C, Moore MJ and
Zalcberg JR: K-ras mutations and benefit from Cetuximab in advanced
colorectal cancer. N Engl J Med. 359:1757–1765. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Van Cutsem E, Peeters M, Siena S, Humblet
Y, Hendlisz A, Neyns B, Canon JL, Van Laethem JL, Maurel J,
Richardson G, Wolf M and Amado RG: Open-label phase III trial of
panitumumab plus best supportive care compared with best supportive
care alone in patients with chemotherapy-refractory metastatic
colorectal cancer. J Clin Oncol. 25:1658–1664. 2007.PubMed/NCBI
|
|
23.
|
Amado RG, Wolf M, Peeters M, Van Cutsem E,
Siena S, Freeman DJ, Juan T, Sikorski R, Suggs S, Radinsky R,
Patterson SD and Chang DD: Wild-type KRAS is required for
panitumumab efficacy in patients with metastatic colorectal cancer.
J Clin Oncol. 26:1626–1634. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Peeters M, Price YJ, Cervantes A, Sobrero
AF, Ducreux M, Hotko Y, André T, Chan E, Lordick F, Punt CJA,
Strickland AH, Wilson G, Ciuleanu TE, Roman L, Van Cutsem E,
Tzekova V, Collins S, Oliner KS, Rong A and Gansert J: Randomized
phase III study of panitumumab with fluorouracil, leucovorin, and
irinotecan (FOLFIRI) compared with FOLFIRI alone as second-line
treatment in patients with metastatic colorectal cancer. J Clin
Oncol. 28:4706–4713. 2010. View Article : Google Scholar
|
|
25.
|
Masi G, Loupakis F, Salvatore L, Fornaro
L, Cremolini C, Cupini S, Ciarlo A, Del Monte F, Cortesi E, Amoroso
D, Granetto C, Fontanini G, Sensi E, Lupi C, Andreuccetti M and
Falcone A: Bevacizumab with FOLFOXIRI (irinotecan, oxaliplatin,
fluorouracil, and folinate) as first-line treatment for metastatic
colorectal cancer: a phase 2 trial. Lancet Oncol. 11:845–852. 2010.
View Article : Google Scholar
|
|
26.
|
Garufi C, Torsello A, Tumolo S, Ettorre
GM, Zeuli M, Campanella C, Vennarecci G, Mottolese M, Sperduti I
and Cognetti F: Cetuximab plus chronomodulated irinotecan,
5-fluorouracil, leucovorin and oxaliplatin as neoadiuvant
chemotherapy in colorectal liver metastases: POCHER trial. Br J
Cancer. 103:1542–1547. 2010. View Article : Google Scholar
|
|
27.
|
Ficorella C, Ricevuto E, Morelli MF,
Morese R, Cannita K, Cianci G, Di Rocco ZC, De Galitiis F, De Tursi
M, Tinari N, Iacobelli S and Marchetti P: Increased tolerability of
bimonthly 12-hour timed flat infusion 5-fluorouracil/irinotecan
regimen in advanced colorectal cancer: a dose-finding study. Oncol
Rep. 5:1345–1350. 2006.PubMed/NCBI
|
|
28.
|
Morelli MF, Santomaggio A, Ricevuto E,
Cannita K, De Galitiis F, Tudini M, Bruera G, Mancini M,
Pelliccione M, Calista F, Guglielmi F, Martella F, Lanfiuti Baldi
P, Porzio G, Russo A, Gebbia N, Iacobelli S, Marchetti P and
Ficorella C: Triplet schedule of weekly 5-fluorouracil and
alternating irinotecan or oxaliplatin in advanced colorectal
cancer: a dose-finding and phase II study. Oncol Rep. 23:1635–1640.
2010.PubMed/NCBI
|
|
29.
|
Therasse P, Arbuck SG, Eisenhauer EA,
Wanders J, Kaplan RS, Rubinstein L, Verweij J, Glabbeke MV, van
Oosterom AT, Christian MC and Gwyther SG: New guidelines to
evaluate the response to treatment in solid tumors: European
Organization for Research and Treatment of Cancer, National Cancer
Institute of the United States, National Cancer Institute of
Canada. J Natl Cancer Inst. 92:205–216. 2000. View Article : Google Scholar
|
|
30.
|
Kaplan EL and Meier P: Nonparametric
estimation of incomplete observations. J Am Statist Assoc.
53:457–481. 1958. View Article : Google Scholar
|
|
31.
|
Peto R and Peto J: Asymptomatically
efficient rank invariant test procedures. J Roy Statist Soc A.
135:185–206. 1972. View Article : Google Scholar
|
|
32.
|
Di Fiore F, Blanchard F, Charbonnier F, Le
Pessot F, Lamy A, Galais MP, Bastit L, Killian A, Sesboué R, Tuech
JJ, Queniet AM, Paillot B, Sabouirin JC, Michot F, Michel P and
Frebourg T: Clinical relevance of KRAS mutation detection in
metastatic colorectal cancer treated by cetuximab plus
chemotherapy. Br J Cancer. 96:1166–1169. 2007.PubMed/NCBI
|
|
33.
|
Lamy A, Blanchard F, Le Pessot F, Sesboué
R, Di Fiore F, Bossut J, Fiant E, Frébourg T and Sabourin JC:
Metastatic colorectal cancer KRAS genotyping in routine practice:
results and pitfalls. Mod Pathol. 24:1090–1100. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Van Cutsem E, Tabernero J, Lakomy R,
Prener H, Prausova J, Macarulla T, Ruff P, Van Hazel GA, Moiseyenko
V, Ferry D, McKendrick J, Polikoff J, Tellier A, Castan R and
Allegra C: Addition of aflibercept to fluorouracil, leucovorin, and
irinotecan improves survival in a phase III randomized trial in
patients with metastatic colorectal cancer previously treated with
an oxaliplatin-based regimen. J Clin Oncol. 30:3499–3506. 2012.
|
|
35.
|
Arnold D, Andre T, Bennouna J, Sastre J,
Osterlund PJ, Greil R, Van Cutsem E, Von Moos R, Reyes-Rivera I,
Bendahmane B and Kubicka S: Bevacizumab (BEV) plus chemotherapy
(CT) continued beyond first progression in patients with metastatic
colorectal cancer (mCRC) previously treated with BEV plus CT:
results of a randomized phase III intergroup study (TML study). J
Clin Oncol. 30(Suppl): CRA35032012.
|
|
36.
|
Ince WL, Jubb AM, Holden SN, Holmgren EB,
Tobin P, Sridhar M, Hurwitz HI, Kabbinavar F, Novotny WF, Hillan KJ
and Koeppen H: Association of k-ras, b-raf, and p53 status with the
treatment effect of bevacizumab. J Natl Cancer Inst. 97:981–989.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Hurwitz HI, Yi J, Ince W, Novotny WF and
Rosen O: The clinical benefit of bevacizumab in metastatic
colorectal cancer is independent of K-ras mutation status: analysis
of a phase III study of bevacizumab with chemotherapy in previously
untreated metastatic colorectal cancer. Oncologist. 14:22–28. 2009.
View Article : Google Scholar
|
|
38.
|
Guerrero S, Casanova I, Farrè L, Mazo A,
Capellà G and Mangues R: K-ras codon 12 mutation induces higher
level of resistance to apoptosis and predisposition to
anchorage-independent growth than codon 13 mutation or
proto-oncogene overexpression. Cancer Res. 60:6750–6756. 2000.
|
|
39.
|
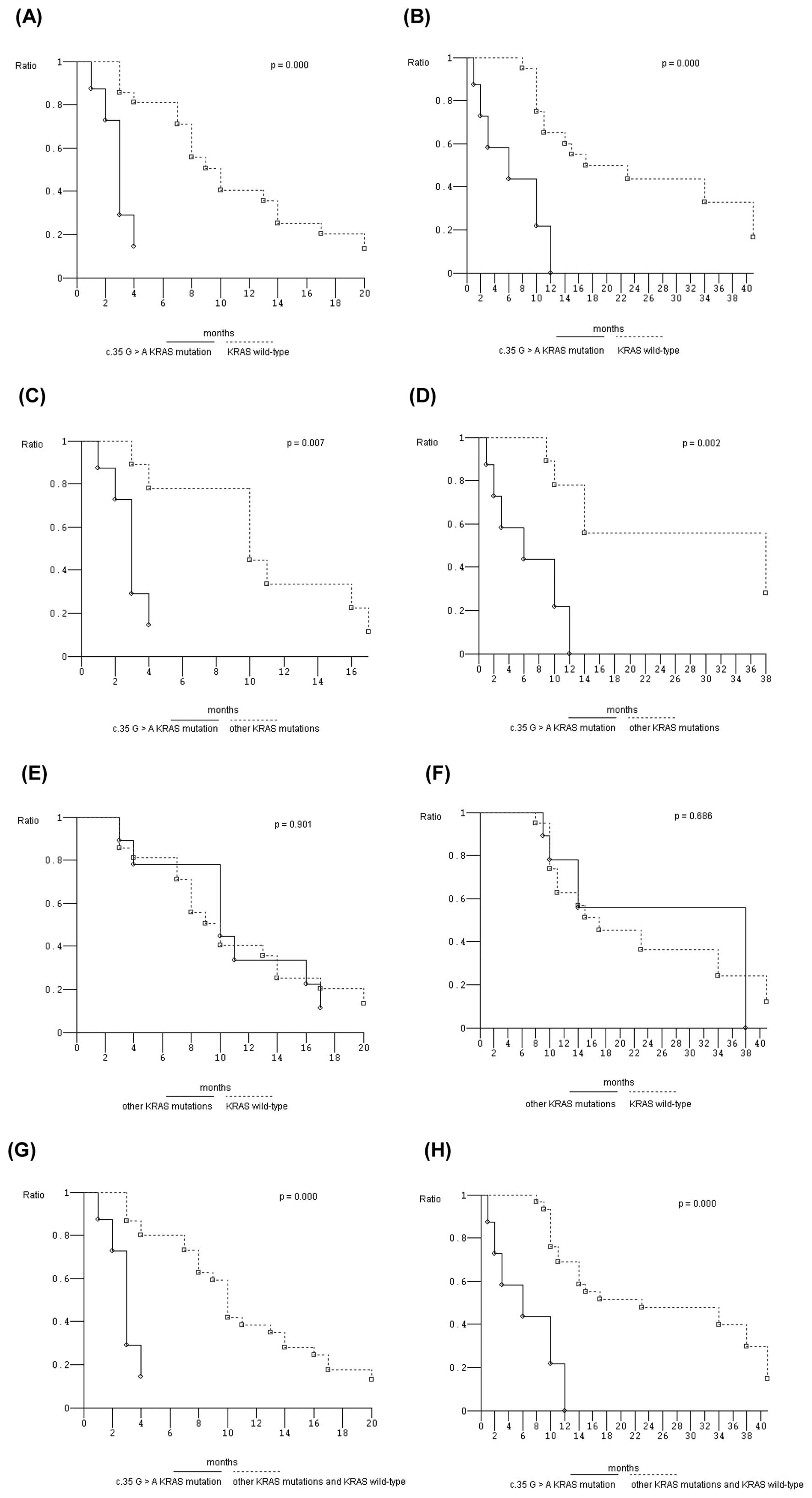
Bruera G, Cannita K, Di Giacomo D, Lamy A,
Frébourg T, Sabourin JC, Tosi M, Ficorella C and Ricevuto E: Worse
prognosis of KRAS c.35 G>A mutant metastatic colorectal cancer
(MCRC) patients treated with intensive triplet chemotherapy plus
bevacizumab (FIr-B/FOx). BMC Med. 11:592013.
|