Introduction
Most tumors consist of heterogeneous cell
populations, with varying capacities for proliferation and tumor
formation in immune-deficient mice, even when derived from a single
clone. Recently, cancer stem cells, or cancer-initiating cells
(CICs), were defined as a small population among tumor cells
possessing the ability to self-renew and generate heterogeneous
lineages that comprise the tumor (1,2).
Numerous investigations have demonstrated that CICs exist in a
variety of human tumors, including hematopoietic malignancies,
brain tumors, breast cancer, gastroenterological cancer and head
and neck cancer (3–7).
Oral squamous cell carcinoma (OSCC) is the most
common malignancy of the maxillofacial region, accounting for
nearly 3% of all cancer cases worldwide (8). Advances in therapy have improved
quality of life, but survival rates have remained unchanged over
the past decades. Mortality caused by OSCC remains high because of
the development of distant metastases and the emergence of local
and systemic recurrences resistant to chemo- and radiotherapy.
Local recurrence is a particularly powerful prognostic indicators
following curative resection, because OSCC recurs with a frequency
of 25–48% (9). It is therefore
essential to gain a deeper understanding of the biology of this
disease to develop more effective therapeutic approaches.
The aldehyde dehydrogenases (ALDH) are a family of
ubiquitous enzymes that catalyze the irreversible oxidation of
aldehydes to their corresponding carboxylic acids (10). Among the 17 ALDH isoforms, aldehyde
dehydrogenases 1 (ALDH1) oxidizes retinol to retinoic acid in the
early stages of stem cell differentiation and exhibits a high level
of activity in hematopoietic and neural stem cells (11,12).
Growing evidence suggests that elevated ALDH1 activity can define
cancer stem cell populations in many cancer types, including human
multiple myeloma, acute myeloid leukemia and cancers of the brain,
lung, pancreas and breast (11,13–15).
Therefore, ALDH1 activity could act as a common marker for both
normal and malignant stem cell populations (16). Furthermore, recent studies have
demonstrated that ALDH1 expression is a predictor of poor clinical
outcome in several types of malignant tumors (11,17–19).
We hypothesized that the level of ALDH1 enzymatic
activity and expression correlates with the biologic
characteristics of OSCC, including clinicopathological significance
and antitumor drug resistance. We aimed to investigate whether
alterations in the population of tumor cells exhibiting ALDH1
immunostaining predicted clinicopathological features of prognostic
importance for cancer progression and whether ALDH1 expression and
enzymatic activity contributed to the characteristics of CICs in
cisplatin-treated OSCC. Our results suggest that increased cell
populations exhibiting ALDH1 activity and expression may be
responsible for refractory events during OSCC.
Materials and methods
Ethics
During initial diagnosis, all patients provided
written consent for their tumor samples to be used for
investigational purposes. Approval from the Internal Review and the
Ethics Boards of the Fukuoka Dental College General Hospital
(Fukuoka, Japan) was obtained for this study. Data were anonymously
analyzed.
Patients and tumor specimens
Surgical specimens of primary OSCC were obtained
from 90 patients attending the Fukuoka Dental College Hospital
between January, 2005 and January, 2010. The study population
consisted of 51 men and 39 women [age range, 45–88 (median, 71.5)
years]. Clinical disease stage was determined based on the findings
of a preoperative diagnostic examination. Brief clinical findings
of the 90 patients are summarized in Table I. Resected specimens obtained from
patients who had not undergone preoperative chemoand radiotherapy
were fixed in 10% formalin and processed for paraffin embedding.
Histological sections, 4-μm thick, were used for hematoxylin
and eosin and immunoperoxidase staining.
 | Table I.Brief summary of 90 patients with
OSCC. |
Table I.
Brief summary of 90 patients with
OSCC.
| Characteristics | n |
|---|
| Age, years | |
| <60 | 24 |
| ≥60 | 66 |
| Gender | |
| Male | 51 |
| Female | 39 |
| Site | |
| Tongue | 24 |
| Gingiva | 42 |
| Others | 24 |
| Tumor size | |
| ≤4 cm | 35 |
| >4 cm | 55 |
| Pathologic T
classification | |
| T1 | 13 |
| T2 | 44 |
| T3 | 14 |
| T4 | 21 |
| Pathologic N
classification | |
| N0 | 71 |
| N1 | 19 |
| Pathologic stage
(pTNM) | |
| I | 13 |
| II | 36 |
| III | 14 |
| IV | 27 |
| Recurrence | |
| Negative | 73 |
| Positive | 17 |
Immunohistochemistry on tissue sections
of OSCC
Immunohistochemistry was performed using the
Histofine® Simple stain kit accoring to the
manufacturer's protocol (Nichirei Corp., Tokyo, Japan). ALDH1
antibody (1:250; BD Biosciences, San Jose, CA, USA) was incubated
overnight at 4°C. As a negative control, staining was performed
without a primary antibody. The immunoreaction was visualized with
3,3′-diaminobenzidine. Immunohistochemical staining for ALDH1 was
analyzed by two oral pathologists (K.T. and J.O.). Staining was
scored according to cytoplasmic staining and evaluated by the
criteria of Chang et al (18). Patients with <5% ALDH1-positive
cells were given a score of 0; those with 5–20% were given a score
of 1; those with 20–50% were given a score of 2; and those with
>50% were given a score of 3. For statistical analysis, we
divided patients into two groups: low expression (with scores of 0
or 1) and high expression (with scores of 2 or 3).
Cell line and culture
The OSCC cell line, HSC-3, was obtained from the
Japanese Collection of Research Bioresources Cell Bank (Osaka,
Japan). HSC-3 cells were cultured in Dulbecco's modified Eagle's
medium (DMEM; Wako, Tokyo, Japan) containing 10% (v/v) fetal bovine
serum (FBS; Invitrogen, Carlsbad, CA, USA) at 37°C with 5%
CO2. The medium was changed daily. When 85% of the cells
adhered, the medium was replaced and the cells were cultured for 48
h. When cells reached 95% confluence in the exponential growth
phase, they were used for subsequent experiments.
Induction of cisplatin surviving HSC-3
cells
Cisplatin [cisdiammineplatinum (II) dichloride,
0.1–50 μM; Sigma-Aldrich, St. Louis, MO, USA) was added to
the culture medium and HSC-3 cells were cultured for 24 h. The
culture media were removed along with unattached cells and the
cells were cultured in the same medium with cisplatin for 72 h.
Cisplatin-surviving cells (CiSCs) were derived from original
parental cells (PTCs) by continuous exposure to cisplatin.
Cell proliferation assay
HSC-3 cells were seeded in 96-well plates with 100
μl medium at a density of 1×104 and incubated at
37°C overnight to allow the adherent cells to attach to the wells.
The cultured cells were then incubated with cisplatin (0.1–50
μM) for 72 h. Next, 10 μl cell counting kit-8
solution (CCK-8; Dojindo Laboratories, Kumamoto, Japan) was added
to each well and the cultured cells were incubated at 37°C for 4 h.
Optical density was measured at 480 nm.
Immunocytochemistry
Cells were harvested from cell lines, resuspended in
medium, and cultured on coverslips for 48 h. HSC-3 cells were fixed
with 4% paraformaldehyde in phosphate-buffered saline (PBS) and
permeabilized by incubation with 0.2% Triton X-100 in PBS for 15
min. Anti-mouse immunoglobulin G conjugated with Alexa
Fluor® 488 (1:200; Molecular Probes, Eugene, OR, USA)
was over-laid on cells, followed by preincubation with anti-ALDH1
antibody (1:250). Immunostained cells were then counter-stained
with 4,6-diamidino-2-phenylindole (DAPI; Vector Laboratories Inc.,
Burlingame, CA, USA).
Aldefluor™ assay
ALDH1 enzymatic activity was detected using the
Aldefluor™ kit (Stem Cell Technologies Inc., Vancouver, Canada),
according to the manufacturer's instructions. In brief, cells
(1×106 cells/ml) were harvested from PTCs and CiSCs cell
lines and resuspended in Aldefluor assay buffer containing ALDH1
substrate and boron-dipyrromethene (BODIPY)-aminoacetaldehyde
(BAAA) and incubated for 40 min at 37°C. BAAA taken up by living
HSC-3 cells was converted by intracellular ALDH1 into
BODIPY-aminoacetate, causing the cells to bright fluorescence. As a
negative control, cells were stained under identical conditions
with a specific ALDH inhibitor, 4-diethylaminobenzaldehyde (DEAB).
Fluorescent ALDH1-positive cells were detected in the green
fluorescent channel of a fluorescent microscope or FACSCalibur™
flow cytometer (BD Biosciences). For fluorescent microscopic
observation, cell nuclei were stained with DAPI.
Western blot analysis
Total protein was extracted from PTCs and CiSCs
using ice-cold cell lysis buffer (20 mM Tris-HCl, pH 7.5; 150 mM
NaCl; 1 mM ethylenediaminetetraacetic acid (EDTA); 1 mM
Na2 EDTA; 1 mM ethylene glycol tetraacetic acid; 1%
(v/v) Triton X-100; 2.5 mM sodium pyrophosphate; 1 mM
β-glycerophosphate; 1 mM Na3VO4; and 1
μg/ml leupeptin and phenylmethyl sulfonyl fluoride). Equal
amounts of protein (20 μg) were separated by sodium dodecyl
sulfate-polyacrylamide gel electrophoresis (SDS-PAGE; 12%
separating gel). After electrophoresis, proteins were transferred
to polyvinylidene dif luoride membranes (Bio-Rad Laboratories,
Tokyo, Japan). The blots were blocked in 1% casein in Tris-buffered
saline (TBS) containing 0.1% Tween-20 (TBS-T) for 1 h at room
temperature and then incubated with primary antibodies overnight at
4°C. Primary antibodies against ALDH1 (described earlier), ABCG2
(Sigma-Aldrich), and β-actin (Sigma-Aldrich) were used. Membranes
were washed in TBS-T and incubated with secondary horseradish
peroxidase-labeled antibodies for 1 h at room temperature. Bound
antibody complexes were detected by enhanced chemiluminescence
(Bio-Rad Laboratories).
Matrigel™ invasion assay
Tumor cell invasion into Matrigel™ was examined
using BioCoat™ Matrigel invasion chambers (BD Biosciences)
according to the manufacturer's protocol. In brief, PTCs or CiSCs
were seeded in DMEM without FBS in the upper chamber and cultured
for 24 h. The lower chamber contained DMEM and 10% FBS. Invading
cells were stained using a Diff-Quik Staining kit (Siemens, Munich,
Germany). The number of invading cells was counted in four
microscopic fields per well at a ×20 magnification and the extent
of invasion was expressed as the average number of cells per square
millimeter.
Cancer sphere formation assay
Single CiSC and PTC cells were plated at
1×103 cells/100 μl on a low-cell binding culture
plate (96-well; Thermo Fisher Scientific, Tokyo, Japan) in
serum-free DMEM/F12 supplemented with B27 (Life Technologies,
Tokyo, Japan), 20 ng/ml epidermal growth factor (BD Biosciences),
0.4% bovine serum albumin (Sigma-Aldrich) and 4 μg/ml
insulin (Sigma-Aldrich). Sphere formation was assessed 24 h after
seeding the cells and the size of the sphere was analyzed.
Statistical analysis
Statistical analyses were performed using StatView
software (SAS Institute Inc., Cary, NC, USA). The χ2 and
Fisher's exact probability tests were used to analyze the
correlation between ALDH1 expression and clinicopathological
factors in OSCC. Statistical analyses of invasion and cancer sphere
formation assays were performed with the two-tailed Student's
t-test. Data are presented as the mean ± standard error and p-value
<0.05 was considered statistically significant.
Results
ALDH1 expression and localization in
tissue sections obtained from patients with OSCC
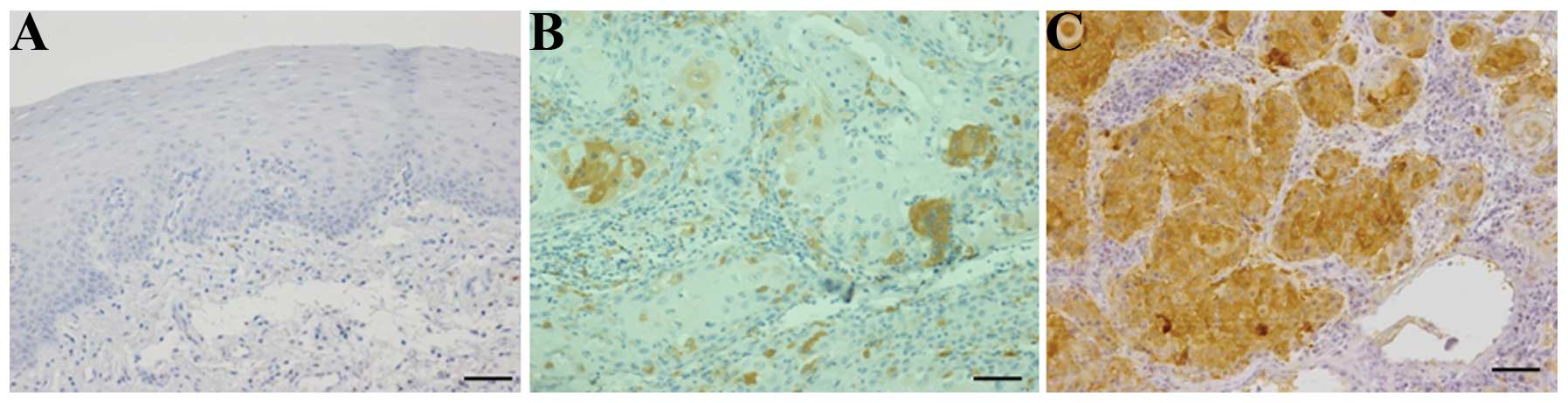
Normal-like oral mucosa showed no staining for
anti-ALDH1 antibody (Fig. 1A). In
tissue sections from patients with OSCC, diffuse,
moderate-intensity cytoplasmic staining for anti-ALDH1 antibody was
observed in a proportion of the tumor cells. ALDH1 expression in
epithelial cancer cells was scored and subjected to statistical
analysis. The percentage of positive cancer cells varied from
<5% to >50% in the population studied (Fig. 1B and C).
ALDH1 expression correlates with
recurrence in patients with OSCC
Results of immunostaining for ALDH1 in OSCC
specimens organized according to the clinicopathological
characteristics of patients are shown in Table II. No ALDH1 was expressed (score,
0) in 7 patients (7.8%); 1–5% of cells expressed ALDH1 in 20
patients (22.8%; score, 0); 6–20% of cells expressed ALDH1 in 16
patients (17.8%; score, 1); 21–50% of cells expressed ALDH1 in 37
patients (41.1%; score, 2); and >50% of cells expressed ALDH1 in
10 patients (11.1%; score, 3). No correlation existed between a
high level of expression of ALDH1 (>20% of cells) and age
(p=0.464), gender (p=0.876), tumor location (p=0.392), tumor size
(p=0.239), T classification (p=0.574), nodal status (p=0.577) or
pTNM score (p=0.694). Conversely, a positive correlation existed
between a high level of expression of ALDH1 (>20% of cells) and
local recurrence (p=0.006).
 | Table II.Clinicopathological correlation of
ALDH1 expression in OSSC. |
Table II.
Clinicopathological correlation of
ALDH1 expression in OSSC.
| Factors | No. of patients
| P-value |
|---|
| Low expression | High
expression |
|---|
| Age, years | | | 0.464 |
| <60 | 13 | 11 | |
| ≥60 | 30 | 36 | |
| Gender | | | 0.876 |
| Male | 24 | 27 | |
| Female | 19 | 20 | |
| Site | | | 0.392 |
| Tongue | 11 | 13 | |
| Gingiva | 23 | 19 | |
| Others | 9 | 15 | |
| Tumor size | | | 0.239 |
| ≤4 cm | 29 | 26 | |
| >4 cm | 14 | 21 | |
| Pathologic T
classification | | | 0.574 |
| T1 | 8 | 5 | |
| T2 | 21 | 21 | |
| T3 | 6 | 8 | |
| T4 | 8 | 13 | |
| Pathologic N
classification | | | 0.577 |
| N0 | 35 | 36 | |
| N+ | 8 | 11 | |
| Pathologic stage
(pTNM) | | | 0.694 |
| I | 7 | 6 | |
| II | 14 | 11 | |
| III | 6 | 8 | |
| IV | 17 | 23 | |
| Recurrence | | | 0.006 |
| Negative | 40 | 33 | |
| Positive | 3 | 14 | |
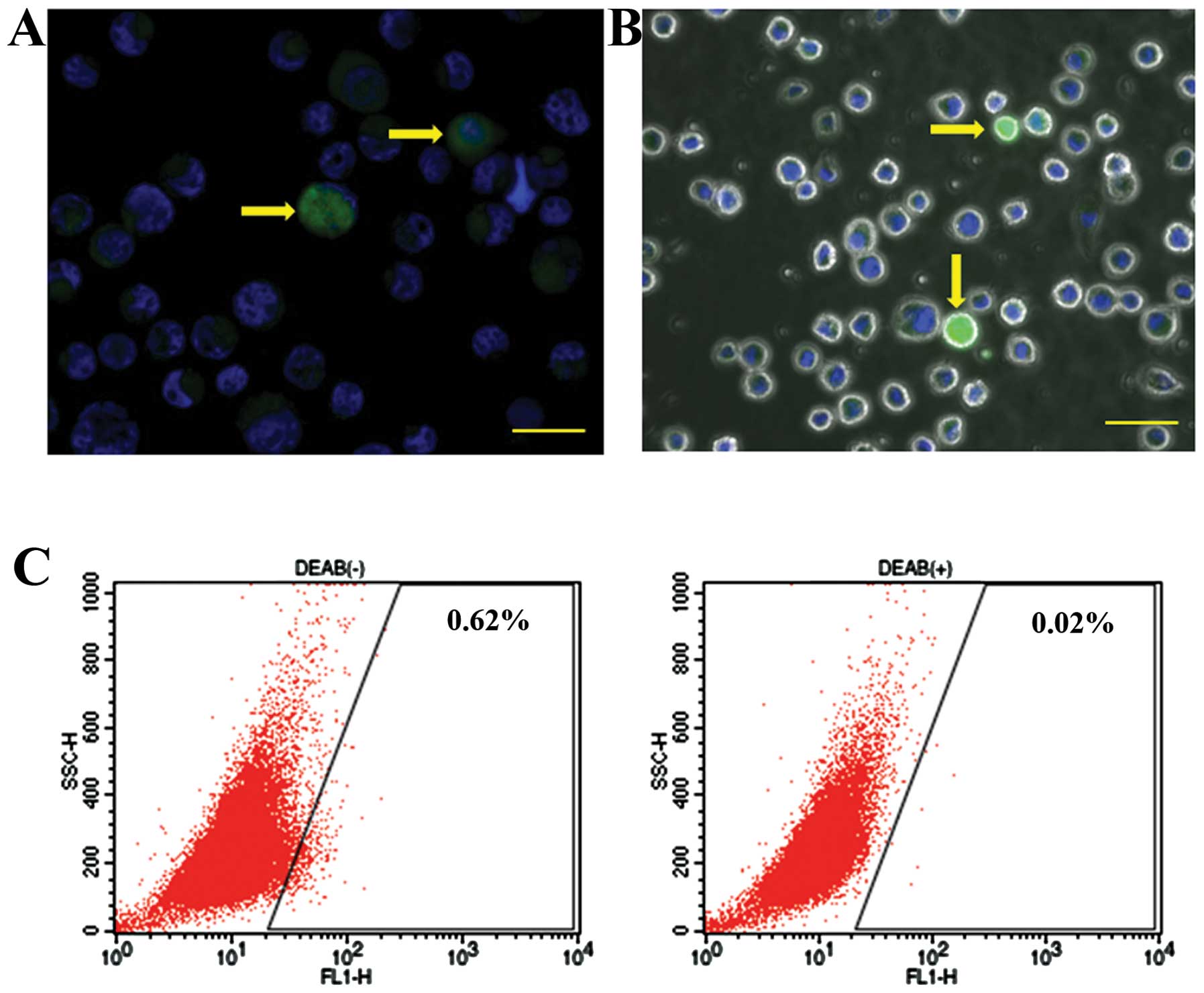
ALDH1 expression in an OSCC cell
line
Overexpression of ALDH1 has been reported in some
cancers (13,17,20).
We undertook immunocytochemical and enzyme-histochemical detection
of ALDH1 in the OSCC cell line HSC-3. During immunocytochemical
analysis, a few HSC-3 cells were stained with anti-ALDH1 antibody
(Fig. 2A). Similarly, in the
enzyme-histochemical analysis, ALDH1 enzymatic activity was
detected in a small population of HSC-3 cells using fluorescence
microscopy (Fig. 2B). Consistent
with this finding, we identified 0.62% of HSC-3 cells exhibiting
ALDH1 enzymatic activity using the flow cytometry (Fig. 2C).
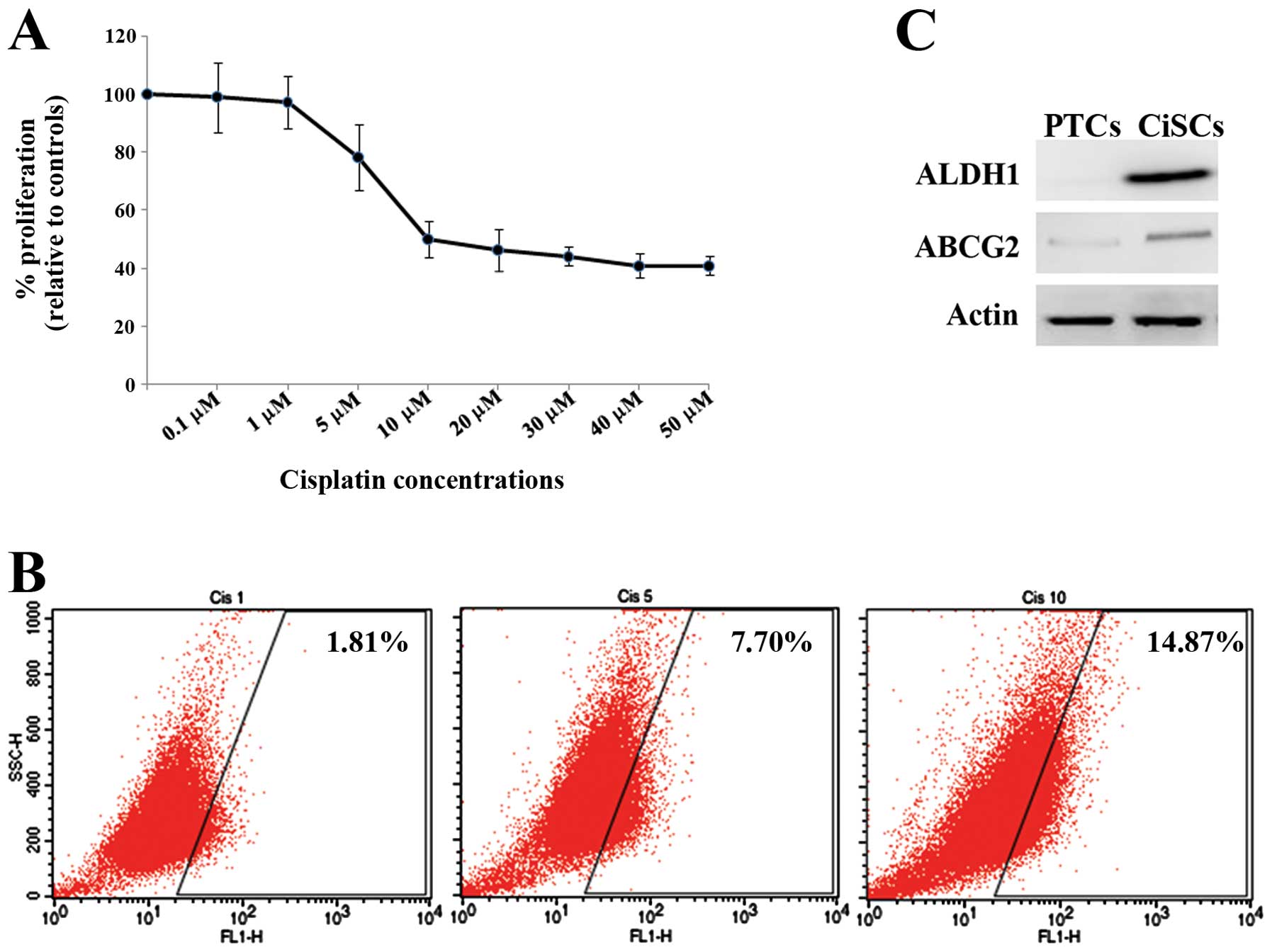
Effects of cisplatin on ALDH1 activity
and expression in HSC-3 cells
As cisplatin is commonly used to treat malignant
tumors, we examined the effect of ALDH1 on the resistance of HSC-3
cells to cisplatin. The effect of cisplatin on HSC-3 cell
proliferation was then examined using the CCK-8 assay. As shown in
Fig. 3A, treatment of HSC-3 cells
with 0.1 or 1 μM cisplatin for 72 h did not affect cell
proliferation, whereas treatment with >5 μM caused a
decrease in cell proliferation. This suggests that cisplatin
decreases cell proliferation in a dose-dependent manner (Fig. 3A). Because treatment with 10
μM cisplatin yielded a decrease in cell proliferation of
approximately 50%, we used 10 μM cisplatin for 72 h in
HSC-3-derived CiSCs for further experiments. The percentage of
HSC-3 cells exhibiting ALDH1 enzymatic activity was examined by
flow cytometry to determine the effect of cisplatin treatment. A
higher percentage of cells exhibiting ALDH1 activity was found
among CiSCs (14.8%) compared with cells treated with 1 μM
(1.81%) or 5 μM (7.70%) cisplatin (Fig. 3B). The percentage of ALDH1 activity
in CiSCs was 24-fold higher than that in PTCs, indicating that a
population of CiSCs shifts to become ALDH1-rich. We next examined
protein expression of ALDH1 and ABCG2, an ATF-binding cassette
(ABC) transporter (21), in CiSCs
and PTCs using western blot analysis. As shown in Fig. 3C, both ALDH1 and ABCG2 accumulation
was markedly increased in CiSCs. In contrast, expression of both
proteins was negative or low in PTCs. These results suggest that an
ALDH1-rich population of CiSCs retains a certain amount of both
ALDH1 and ABCG2 proteins.
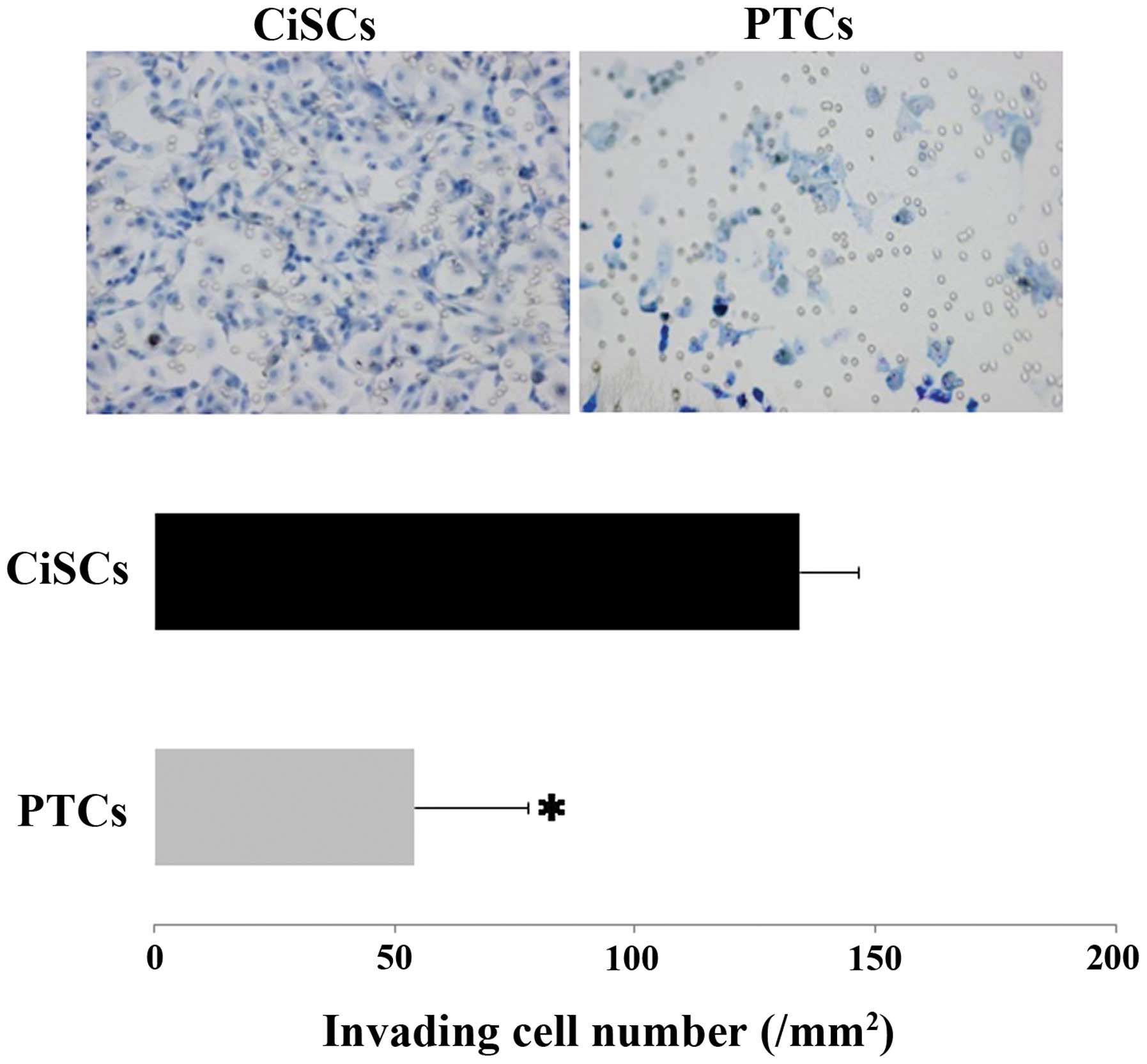
Invasive ability of CiSCs
Flow cytometric analysis of CiSCs and PTCs revealed
differences between populations of cells exhibiting ALDH1 enzymatic
activity, suggesting potential differences in their
characteristics. To test this hypothesis, we examined the invasive
ability of CiSCs compared with that of PTCs using Matrigel invasion
assay. The number of invading cells was higher among CiSCs
(134±12.1) than PTCs (54±23.4) at 24 h, indicating that CiSCs
possess enhanced invasive capability relative to PTCs (Fig. 4).
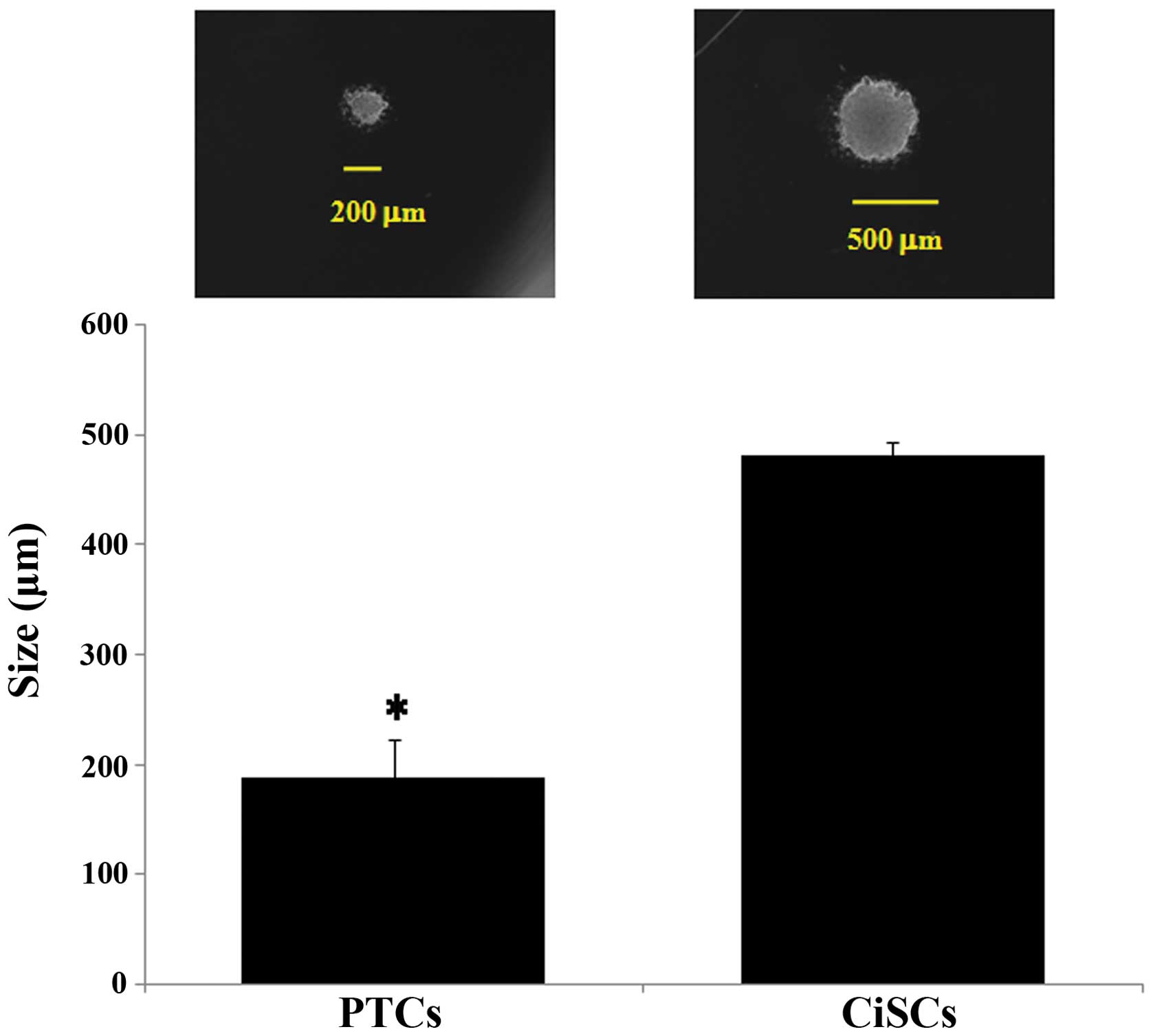
Cancer sphere formation in CiSCs
The abilities to self-renew and generate
differentiated progeny are fundamental properties of CICs. Cancer
sphere generation is an in vitro assay of self-renewal
potential (7). We assessed the
self-renewal properties of CiSCs by their ability to form tumor
spheres when cultured in serum-free medium under non-adherent
conditions. Both CiSCs and PTCs generated cancer spheres 24 h after
cells were seeded. However, the size of the spheres formed was
greater for CiSCs than PTCs (Fig.
5). The size of the sphere developed from PTCs matched that of
the sphere developed from CiSCs after 4 days. These results
indicate that the cancer spheres developed from CiSCs grew faster
and were larger than those developed from PTCs.
Discussion
In solid malignancies, CICs or cancer stem cells,
possess a defined population of cells that exhibit elevated ALDH1
activity and resistance to antitumor drugs and radiotherapy
(13,20,22–25).
We present clinicopathological and cellular experimental evidence
supporting the theory that increased ALDH1 immunostaining and
enzymatic activity contributes to the characteristics of CICs in
OSCC. First, an immunohistochemical approach using clinical
specimens confirmed that increased expression of ALDH1 is
associated with local recurrence. Second, in vitro cellular
experiments indicated that CiSCs, among which an increased
population exhibit elevated ALDH1 activity, represent refractory
characteristics of CICs, such as ABCG2 expression, invasive
capabilities and the ability to self-renew.
Our clinicopathological results showed that a high
level of ALDH1 expression correlated with local recurrence. This
finding is supported by the observation that a high percentage of
ALDH1-expressing cells in most types of epithelial tumors,
including breast, lung, pancreas, bladder, ovarian and prostate
tumors, is associated with poor outcome (13,22–25).
In this study, no association was found between ALDH1 expression
and prognostic factors in OSCC other than local recurrence,
although elevated ALDH1 expression is significantly more frequent
in tumors than in normal oral mucosa. This finding could be
explained by the small patient cohort in this study: >95% of
cancer cells were ALDH1-expressing and prognostic significance was
demonstrated in a cohort comprising >400 patients with other
epithelial cancers (17,18). However, ALDH1 may present a useful
marker for the prediction of local recurrence of OSCC because this
tumor exhibits a high incidence of local recurrence (9).
In clinical specimens of OSCC, ALDH1-high group
showed a diffuse staining pattern in cancer nests. This seems
incompatible with the concept that CICs comprise a small population
of cancer cells with multiple differentiation and long-term
repopulation capabilities. In some studies, CIC markers were
expressed in clinical samples of most tumor cells, e.g., breast and
endometrioid cancers (11,17). Tumor characteristics may become
more aggressive, when most tumor cells possess CIC characteristics.
Alternatively, ALDH1 may be a marker of undifferentiated cancer
cells rather than CICs.
Using flow cytometry, we identified that the
percentage of cells exhibiting ALDH1 enzymatic activity
significantly increased among CiSCs. In cellular experiments, the
presence of an increased population of cells exhibiting ALDH1
activity appears to contribute to the refractory characteristics of
CICs. By western blot analysis, we showed that ABCG2 expression is
upregulated in CiSCs compared with PTCs. These findings are
supported by previous reports (20,26).
ABCG2 is a member of the ABC-transporter superfamily and is known
to contribute to multidrug resistance to cancer chemotherapy
(27). Elevated expression of ABC
transport proteins in stem cells compared with non-stem cells
confers a relative resistance to the toxic effects of chemotherapy
drugs (28,29). Therefore, our results suggest that
increased ABCG2 expression in CiSCs may be responsible for drug
efflux, conferring resistance to cisplatin. In a Matrigel invasion
assay, CiSCs exhibited a significantly increased invasive capacity
compared to that of PTCs over a short time period (24 h). These
results support the idea that the ability to initiate and drive
primary tumor growth, invasion and metastasis is central to the
definition of CICs. Self-renewal and the ability to generate
differentiated progenitors are considered fundamental properties of
CICs (7,30). We found that CiSCs, in contrast to
PTCs, have an enhanced ability to generate cancer spheres under
selective culture conditions, indicating their increased potential
for self-renewal. Both in vivo and in vitro results
in this study suggest that ALDH1 could represent an effective
therapeutic target for CICs in OSCC. Future studies on ALDH1
regulation may yield novel therapeutic modalities for OSCC.
In this study, we demonstrated that an increased
population of cells exhibiting ALDH1 expression and enzymatic
activity contributed to the characteristics of CICs in OSCC. A high
level of expression of ALDH1 was associated with local recurrence
in a study of tissue sections from patients with OSCC. An increased
population of cells exhibiting ALDH1 activity appeared to
participate in antitumor drug efflux, invasive capacity, and the
potential for self-renewal. Our findings suggest the possibility of
a novel therapeutic tool against OSCC that targets CICs.
Acknowledgements
This study was supported in part by a
Grant-in-Aid from the Ministry of Education, Culture, Sports,
Science and Technology of Japan (nos. 24390422 and 25670802 to
J.O.). The authors would like to thank Enago (www.enago.jp) for the English language review.
References
|
1.
|
Al-Hajj M and Clarke MF: Self-renewal and
solid tumor stem cells. Oncogene. 23:7274–7282. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Clarke MF, Dick JE, Dirks PB, Eaves CJ,
Jamieson CH, Jones DL, Visvader J, Weissman IL and Wahl GM: Cancer
stem cells - perspectives on current status and future directions:
AACR Workshop on cancer stem cells. Cancer Res. 66:9339–9344. 2006.
View Article : Google Scholar
|
|
3.
|
Prince ME, Sivanandan R, Kaczorowski A,
Wolf GT, Kaplan MJ, Dalerba P, Weissman IL, Clarke MF and Ailles
LE: Identification of a subpopulation of cells with cancer stem
cell properties in head and neck squamous cell carcinoma. Proc Natl
Acad Sci USA. 104:973–978. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Bonnet D and Dick JE: Human acute myeloid
leukemia is organized as a hierarchy that originates from a
primitive hematopoietic cell. Nat Med. 3:730–737. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Singh SK, Clarke ID, Terasaki M, Bonn VE,
Hawkins C, Squire J and Dirks PB: Identification of a cancer stem
cell in human brain tumors. Cancer Res. 63:5821–5828.
2003.PubMed/NCBI
|
|
6.
|
Ricci-Vitiani L, Lombardi DG, Pilozzi E,
Biffoni M, Todaro M, Peschle C and De Maria R: Identification and
expansion of human colon-cancer-initiating cells. Nature.
445:111–115. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Ponti D, Costa A, Zaffaroni N, Pratesi G,
Petrangolini G, Coradini D, Pilotti S, Pierotti MA and Daidone MG:
Isolation and in vitro propagation of tumorigenic breast cancer
cells with stem/progenitor cell properties. Cancer Res.
65:5506–5511. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
9.
|
Schwartz GJ, Mehta RH, Wenig BL, Shaligram
C and Portugal LG: Salvage treatment for recurrent squamous cell
carcinoma of the oral cavity. Head Neck. 22:34–41. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Lindahl R: Aldehyde dehydrogenases and
their role in carcinogenesis. Cri Rev Biochem Mol Biol. 27:283–335.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Ginestier C, Hur MH, Charafe-Jauffret E,
Monville F, Dutcher J, Brown M, Jacquemier J, Viens P, Kleer CG,
Liu S, Schott A, Hayes D, Birnbaum D, Wicha MS and Dontu G: ALDH1
is a marker of normal and malignant human mammary stem cells and a
predictor of poor clinical outcome. Cell Stem Cell. 1:555–567.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Ibarra I, Erlich Y, Muthuswamy SK,
Sachidanandam R and Hannon GJ: A role for microRNAs in maintenance
of mouse mammary epithelial progenitor cells. Genes Dev.
21:3238–3243. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Jiang F, Qiu Q, Khanna A, Todd NW, Deepak
J, Xing L, Wang H, Liu Z, Su Y, Stass SA and Katz RL: Aldehyde
dehydrogenase 1 is a tumor stem cell-associated marker in lung
cancer. Mol Cancer Res. 7:330–338. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Balicki D: Moving forward in human mammary
stem cell biology and breast cancer prognostication using ALDH1.
Cell Stem Cell. 1:485–487. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Feldmann G, Dhara S, Fendrich V, Bedja D,
Beaty R, Mullendore M, Karikari C, Alvarez H, Iacobuzio-Donahue C,
Jimeno A, Gabrielson KL, Matsui W and Maitra A: Blockade of
hedgehog signaling inhibits pancreatic cancer invasion and
metastases: a new paradigm for combination therapy in solid
cancers. Cancer Res. 67:2187–2196. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Moreb JS: Aldehyde dehydrogenase as a
marker for stem cells. Curr Stem Cell Res Ther. 3:237–246. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Rahadiani N, Ikeda J, Mamat S, Matsuzaki
S, Ueda Y, Umehara R, Tian T, Wang Y, Enomoto T, Kimura T, et al:
Expression of aldehyde dehydrogenase 1 (ALDH1) in endometrioid
adenocarcinoma and its clinical implications. Cancer Sci.
102:903–908. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Chang B, Liu G, Xue F, Rosen DG, Xiao L,
Wang X and Liu J: ALDH1 expression correlates with favorable
prognosis in ovarian cancers. Mod Pathol. 22:817–823.
2009.PubMed/NCBI
|
|
19.
|
Marcato P, Dean CA, Pan D, Araslanova R,
Gillis M, Joshi M, Helyer L, Pan L, Leidal A, Gujar S, et al:
Aldehyde dehydrogenase activity of breast cancer stem cells is
primarily due to isoform ALDH1A3 and its expression is predictive
of metastasis. Stem Cells. 29:32–45. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Gong C, Yao H, Liu Q, Chen J, Shi J, Su F
and Song E: Markers of tumor-initiating cells predict
chemoresistance in breast cancer. PLoS One. 5:e156302010.
View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Zhou S, Schuetz JD, Bunting KD, Colapietro
AM, Sampath J, Morris JJ, Lagutina I, Grosveld GC, Osawa M,
Nakauchi H and Sorrentino BP: The ABC transporter Bcrp1/ABCG2 is
expressed in a wide variety of stem cells and is a molecular
determinant of the side-population phenotype. Nat Med. 7:1028–1034.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Rasheed ZA, Yang J, Wang Q, Kowalski J,
Freed I, Murter C, Hong SM, Koorstra JB, Rajeshkumar NV, He X, et
al: Prognostic significance of tumorigenic cells with mesenchymal
features in pancreatic adenocarcinoma. J Natl Cancer Inst.
102:340–351. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Su Y, Qiu Q, Zhang X, Jiang Z, Leng Q, Liu
Z, Stass SA and Jiang F: Aldehyde dehydrogenase 1 A1-positive cell
population is enriched in tumor-initiating cells and associated
with progression of bladder cancer. Cancer Epidemiol Biomarkers
Prev. 19:327–337. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Li T, Su Y, Mei Y, Leng Q, Leng B, Liu Z,
Stass SA and Jiang F: ALDH1A1 is a marker for malignant prostate
stem cells and predictor of prostate cancer patients' outcome. Lab
Invest. 90:234–244. 2010.PubMed/NCBI
|
|
25.
|
Dave B and Chang J: Treatment resistance
in stem cells and breast cancer. J Mammary Gland Biol Neoplasia.
14:79–82. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Lohberger B, Rinner B, Stuendl N, Absenger
M, Liegl-Atzwanger B, Walzer SM, Windhager R and Leithner A:
Aldehyde dehydrogenase 1, a potential marker for cancer stem cells
in human sarcoma. PLoS One. 7:e436642012. View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Mo W and Zhang JT: Human ABCG2: structure,
function, and its role in multidrug resistance. Int J Biochem Mol
Biol. 3:1–27. 2012.PubMed/NCBI
|
|
28.
|
Ding XW, Wu JH and Jiang CP: ABCG2: a
potential marker of stem cells and novel target in stem cell and
cancer therapy. Life Sci. 86:631–637. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
An Y and Ongkeko WM: ABCG2: the key to
chemoresistance in cancer stem cells? Expert Opin Drug Metab
Toxicol. 5:1529–1542. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Reya T, Morrison SJ, Clarke MF and
Weissman IL: Stem cells, cancer, and cancer stem cells. Nature.
414:105–111. 2001. View
Article : Google Scholar : PubMed/NCBI
|