Introduction
Gliomas are the most common primary tumors
originating in the central nervous system (CNS) (1). They are graded from I to IV based on
their degrees of malignancy. The grade IV glioblastoma (GB) is the
most common and most malignant type of glioma which is highly
invasive, and currently there exists no cure. This could in part be
due to the existence of so-called brain tumor stem cells (BTSCs), a
cellular subfraction within GB which contribute to recurrent tumor
growth and resistance to drugs and irradiation (2). BTSCs exhibit the ability to
self-renew as well as give rise to differentiated tissue cells,
which are responsible for the progress of a tumor.
Several studies have shown that angiogenesis is
essential for glioma growth and metastases (3,4).
Vascular endotheli al growth factor (VEGF) is the most important
stimulant factor in regulating angiogenesis. VEGF, a diffusible
36-46-kDa glycoprotein (5,6), and has been shown to be a potent
mediator of brain tumor angiogenesis, vascular permeability and
gliomas growth (7). Several lines
of direct evidence show that gliomas and BTSCs express high levels
of VEGF (8–11). VEGF secreted by tumor cells
interacts with VEGF receptors (VEGFRs) and stimulates downstream
signaling molecules such as Akt and mitogen-activated protein
kinases (MAPKs) to promote the migration, growth and survival of
endothelial cells (12–14).
VEGFRs include two tyrosine kinase receptors,
VEGFR-1 and VEGFR-2 (15–18). Traditionally, VEGFRs were thought
to be expressed on the surface of tumor endothelial cells, but not
on tumor cells (18–20). However, recent studies suggest that
tumor-derived VEGF provides not only paracrine survival cues for
endothelial cells, but may also fuel autocrine processes in gliomas
cells (tumor-secreted VEGF providing prosurvival signaling through
VEGFRs expressed by tumor cells or BTSCs themselves) and play a
role in tumor resistance to existing therapies (21–24).
However, the direct effect of VEGF on biological
behavior of BTSCs has not been completely elucidated. In this
study, we established C6 glioma stem cells (C6GSCs), and then the
VEGF expression of C6GSCs was downregulated by lentiviral
vector-VEGF short hairpin RNA (shRNA). The proliferation,
differentiation and invasion of C6GSCs were detected by an inverted
phase contrast microscope, flow cytometry and
immunofluorescence.
Materials and methods
Isolation and identification of
C6GSCs
C6 glioma cells were purchased from the Shanghai
Institute of Biochemistry and Cell Biology (Shanghai, China). The
cells were cultured in the serum containing medium composed of
Dulbecco’s modified Eagle’s medium (DMEM) with high glucose and 10%
fetal bovine serum. Cells that were in the exponential growth phase
were then collected and transplanted to a new culture flask with an
equal volume of serum-free neural stem cell (NSC) medium containing
DMEM/F12 with 2% B27, epidermal growth factor (EGF, 10 ng/ml) and
basic fibroblast growth factor (bFGF, 10 ng/ml). Cells were
incubated at 37°C with 95% air, 5% CO2 and 100%
humidity. Two weeks later, floating primary tumor spheres were
collected. The sphere cells were harvested, dissociated into single
cells, and plated into a 96-well plate for the subsphere-forming
assay by limiting dilution as described previously (25). In brief, the cells in single-cell
suspension were diluted to one cell per microliter and plated at
one to two cells per well. Cells were fed with 200 μl
serum-free media with growth factors, changing half of the medium
every 5 days. After plating, the cells were observed, and only
wells containing a single cell were considered.
Subspheres from one mother cell were characterized
by immunocytochemistry against anti-CD133 antibody (1:400, Roche,
Germany) or anti-nestin antibody (1:1,000, Chemicon) as described
previously. In brief, the subspheres were incubated with primary
antibodies: rat anti-CD133, mouse monoclonal anti-nestin overnight
at 4°C, followed by incubation with Alexa Fluor 568-conjugated goat
anti-rat (1:1,000, Molecular Probes), 488-conjugated goat
anti-mouse (1:800, invitrogen). Cell nuclei were counter-stained
with Hoechst 33342 for 30 min at room temperature (RT).
Immunopositive cells were observed using a fluorescent
microscope.
Construction and identification of shRNA
lentiviral vector targeting rat VEGF
Four self-complementary hairpin DNA oligos targeting
rat VEGF mRNA were designed by using Dharmagon siDESIGN Center
algorithrm (http://www.dhar-macon.com/DesignCenter/DesignCenterPage.aspx)
as follows: VEGF shRNA1 is 5′-TGCGGATCAAACCTCACC AAA-3′; VEGF
shRNA2 is 5′-GAGCGGAGAAAGCATTTG TTT-3′; VEGF shRNA3 is
5′-GCGAGGCAGCTTGAGTTA AAC-3′; VEGF shRNA4 is 5′-GCCTCTGAAACCATGAAC
TTT-3′, including a 9-nt loop sequences. Also, a mock sequence that
lacks any similarity to the rat sequence was designed and
synthetized. Lentiviral vectors for rat VEGF shRNA encoding a green
fluorescent protein (GFP) sequence and a puromycin resistance gene
was constructed by GenePharma (Shanghai, China), named as
pGLVH1/GFP/PURO. DNA oligos were annealed and inserted in the rat
H1 promoter site of the PGLVH1-GFP vector. Then, the RNA
interference (RNAi) cassette was cloned into the latter vector and
lentiviral vectors expressing shRNA were constructed. They were
then confirmed by DNA sequencing identification.
Lentiviral vector DNAs and packaging vectors
(pHelper1.0, pHelper2.0) (Qiagen) were then transfected into 293T
cells. After transfection, the cells were incubated at 32°C to
increase viral titer. Forty-eight hours later, the supernatant
containing the retroviral particles was collected, filtered through
the 0.45-μm low protein binding syringe filter and the titer
of lentivirus was determined.
C6GSCs were maintained in above mentioned medium and
were plated into 6-well plates at 4×105 cells/well.
Twenty-four hours later, the cells were transfected with viral
supernatants in the presence of polybrene (6 μg/ml final
concentration) for 12 h at a multiplicity of infection (MOI) of 10
and 20, then replaced with a fresh medium containing 5 μg/ml
puromycin. After 72 h, the cells had been transfected for the
subsequent study.
Semiquantitative RT-PCR analysis
Total RNA was extracted from 1×106
transfected cells in each group using TRIzol reagent (Invitrogen,
Carlsbad, CA, USA), and 2 μg of total RNA was reverse
transcribed into cDNA with oligo dT primers using Omniscript
reverse transcriptase (Qiagen, Hilden, Germany) according to the
manufacture’s protocol. Hot start PCR was performed to analyze
expression levels with the following primers. VEGF: sense
5′-GACCCTGGTGGAC ATCTTCCAGGA-3′ and antisense 5′-GGTGAGAGGTCTAG
TTCCCGA-3′. GAPDH: sense 5′-TGATGACATCAAGAAG GTGGTGAAG-3′ and
antisense 5′-TCCTTGGAGGCCAT GTGGGCCAT-3′.
The PCR products were subjected to agarose gel
electrophoresis followed by staining with ethidium bromide. The
band optical density of VEGF, relative to that of GAPDH (internal
control) was quantified by image analysis system (Leica Q550I W,
Cambridge, UK).
Western blot analysis
Western blot analysis was performed for whole cell
lysate. Aliquots of total protein (50 μg per lane) were
electrophoresed on a 12% SDS-polyacrylamide gradient gel and
transferred to nitrocellulose membranes (Millipore). Washed in
rinse buffer at RT and incubated in blocking buffer (5% fat-free
milk in rinse buffer) for 30 min, the membranes were incubated for
2 h at RT with antibody. Further washed with rinse buffer, the
membranes were incubated with 1:1,000 diluted HRP-conjugated
secondary antibody (Santa Cruz) for 2 h at RT, followed by
developing with enhanced chemiluminescence reagents (Amershame,
Little Chalfont Buckinghamshire, UK). In addition, β-actin was used
as a reference protein. The optical densities were analyzed by
using ImageMaster™2D Platinum (Version 5.0, Amersham Biosciences,
Piscataway, NJ, USA).
Proliferation assay of C6GSCs
The non-transfected C6GSCs (control group), mock
transfected C6GSCs (mock group) and lentiviral vector-VEGF shRNA1
transfected C6GSCs (silence group) were cultured in the above
mentioned medium. Photomicrographs were taken by an inverted phase
contrast microscope and the size of cell spheres was analyzed. At 7
days in vitro (DIV), some cell spheres of three groups were
dissociated into single-cell suspension mechanically by trypsin
digestion. Collected cells were washed once in cold
phosphate-buffered saline (PBS). The cells were fixed in 70%
ethanol, treated with 100 mg/l RNase at 37°C for 30 min and stained
with 50 mg/l propidium iodide (Sigma) for 30 min. The cells were
analyzed using flow cytometry (Epics XL; Beckman Coulter, Fullerton
CA, USA).
Differentiation assay of C6GSCs
C6GSCs of three groups (1×105 cells/ml)
were plated onto poly-D-lysine coated cover slips in 24-well plates
containing serum-free medium, respectively. After 7 DIV, cells were
fixed with 4% paraformaldehyde (PFA) in 0.1 M phosphate buffer (PB)
for 1 h, blocked in 5% goat serum for 30 min at RT and then
incubated for 12 h at RT with primary antibodies. After washing
three times in PBS, the cells were incubated in secondary
antibodies for 6 h at RT. Antibodies were as follows: the primary
monoclonal antibodies were against mouse microtubule associated
proteins-2 (anti-MAP-2, 1:200, Chemicon), mouse glial fibrillary
acidic protein (anti-GFAP, 1:200, Sigma), rat anti-nestin (1:2,000,
Millipore). Secondary antibodies were Alexa Fluor 568-conjugated
(red) goat anti-mouse IgG (1:500, Invitrogen) or FITC-conjugated
(green) goat anti-rat IgG (1:200, Millipore). Cell nuclei were
counter-stained with Hoechst 33342 for 30 min at RT. Immunopositive
cells were observed using fluorescent microscopy.
Invasion assay of C6GSCs
C6GSCs of three groups (1×105 cells/ml)
were added to the upper chamber of the Transwell. The lower chamber
was filled with DMEM/F12 medium. After incubation for 12 h at 37°C,
the cells were fixed with 4% PFA for 30 min and stained with
Hoechst 33342 for 30 min at RT. The filters were then rinsed
thoroughly in distilled water and checked by bright field
microscopy to ensure that the cells were adherent and had migrated.
The non-migrating cells were then removed from the upper surface
(inside) of the Transwell with a wet cotton swab.
Statistical analysis
Images were analyzed by Leica Qwin image processing
and analysis software (Leica imaging system, Cambridge, UK).
Statistical analysis was performed using statistics package for
social science 16.0 (SPSS 16.0). Data are presented as mean ±
standard deviation (M ± SD). Statistical comparisons were performed
using one-way analysis of variance (ANOVA) and differences at
P<0.05 were considered statistically significant.
Results
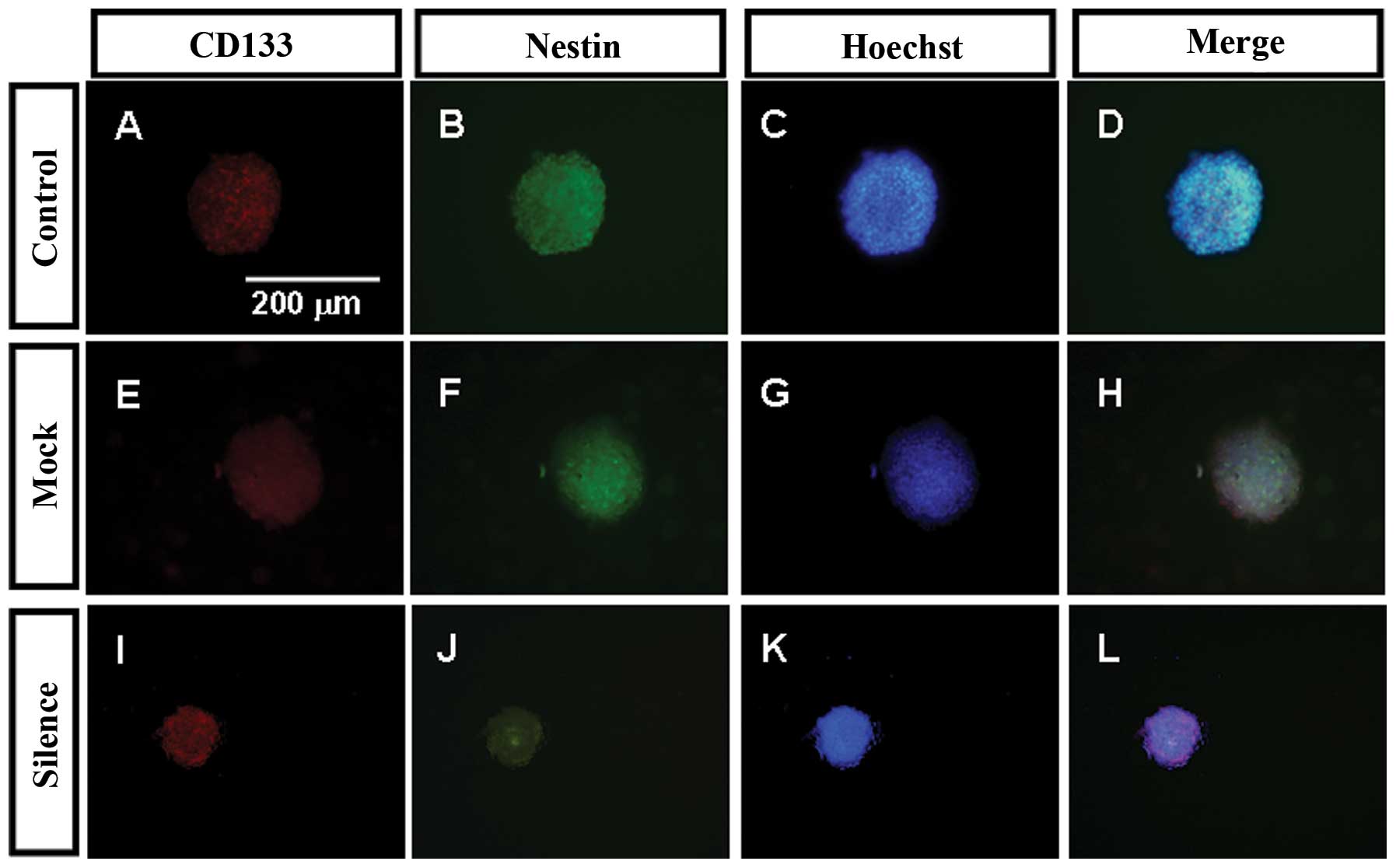
Identification of C6GSCs
The C6 cells were cultured in serum-free NSC medium,
and many formed floating primary tumor spheres. Subspheres from one
mother cell were CD133 and nestin double-positive. The nestin
fluorescence intensity of the C6GSC sphere of the silence group was
lower than that of the other two groups (Fig. 1).
Lentivirus-mediated RNAi inhibits VEGF
expression in C6GSCs
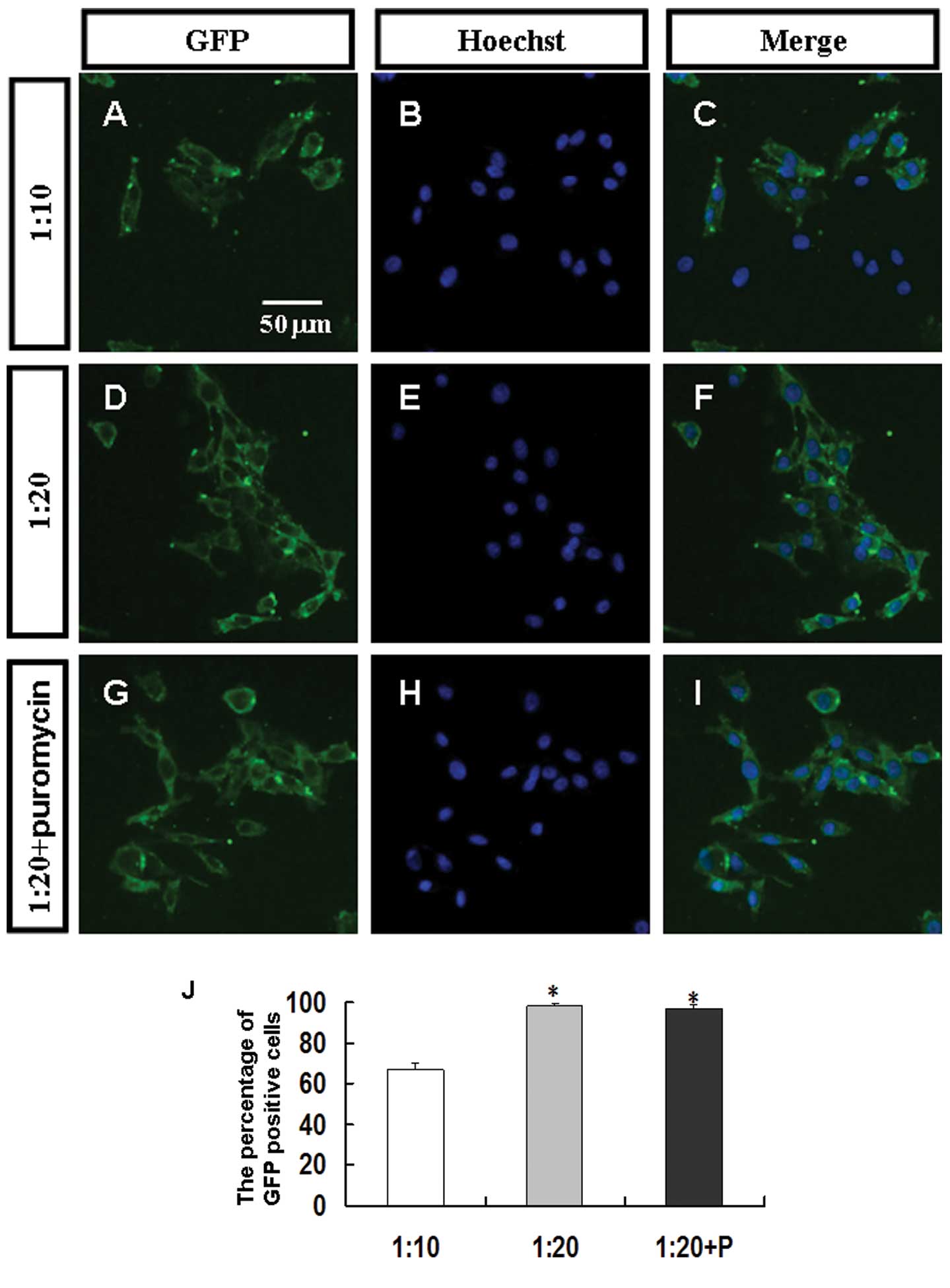
In order to exclude an off-target silencing effect
mediated by specific shRNA, we employed 4 different sequences of
VEGF shRNA. Lentiviral infection efficiency was calculated by GFP
expression under a fluorescence microscope. The infection
efficiency of lentiviral vector-shRNA was 98.02±1.37% when MOI was
1:20 (Fig. 2). To evaluate the
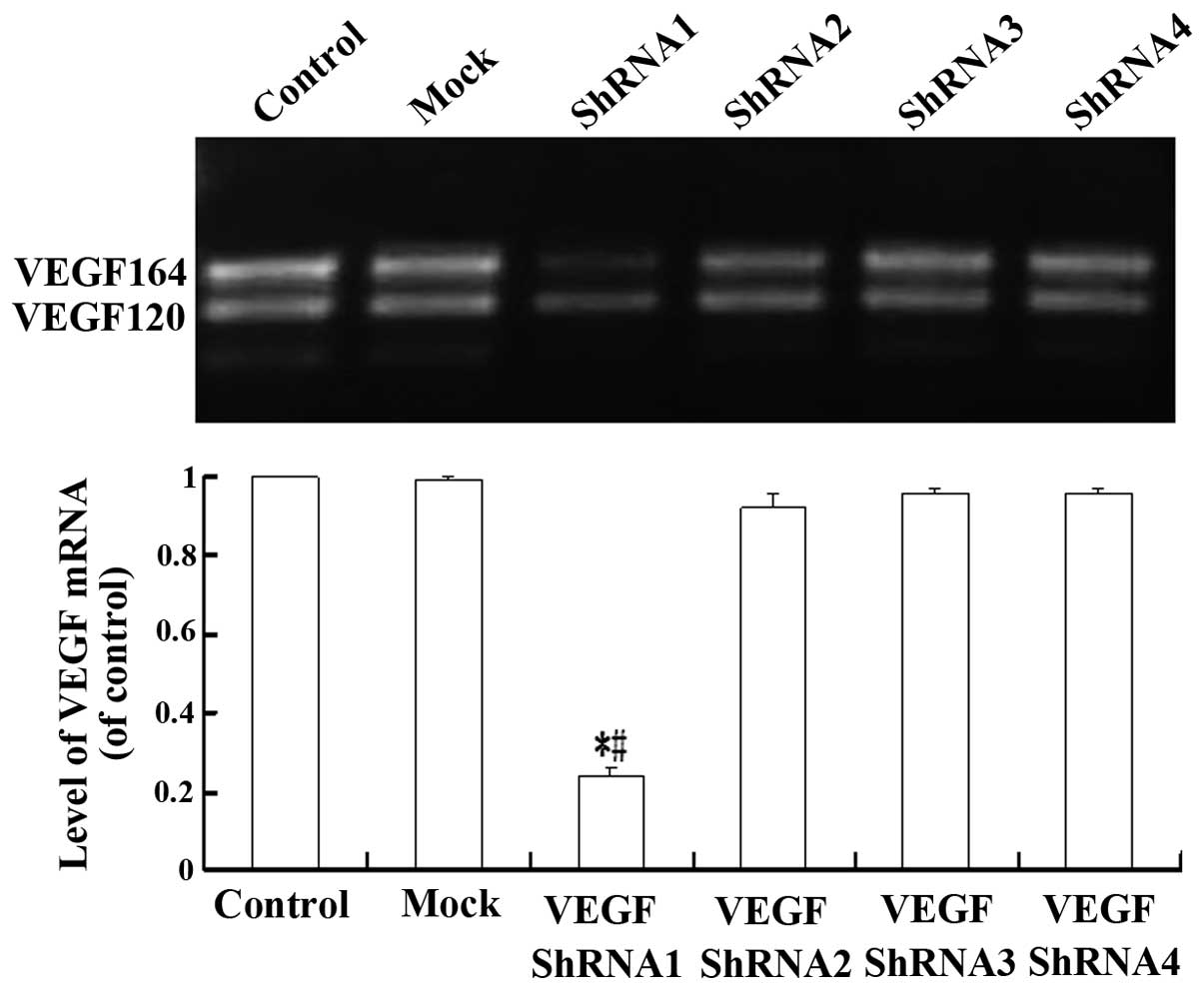
inhibition of VEGF mRNA expression, semi-quantitative RT-PCR was
performed 72 h after infection, the VEGF mRNA expression in
lentiviral vector-VEGF shRNA1 transfected C6GSCs was reduced by
∼75%, as compared with the non-transfected and mock transfected
ones (P<0.05). In addition, no difference was observed among the
non-transfected cells, the mock-infected cells, and VEGF shRNA2, 3
and 4 transfected cells (Fig. 3),
indicating that the corresponding mRNA sequence for VEGF shRNA1,
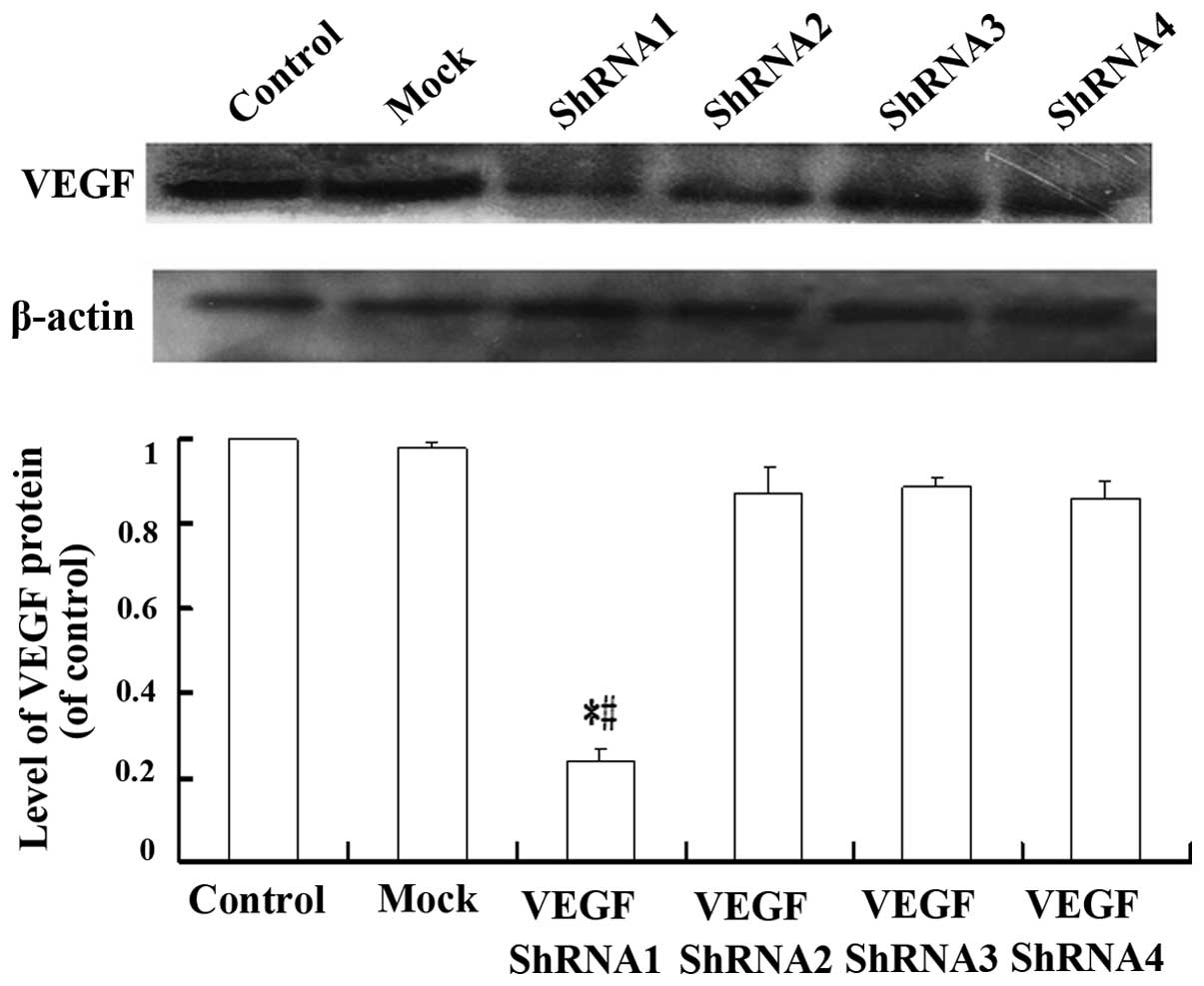
not VEGF shRNA2, 3 and 4 is specific RNAi target. Western blot
analysis was also performed 72 h after infection. The VEGF protein
expression demonstrated a significant reduction in lentiviral
vector-VEGF shRNA1 transfected C6GSCs, as compared with
non-transfected cells and mock transfected cells (P<0.05)
(Fig. 4), suggesting that VEGF
shRNA1 strongly blocked VEGF expression, whereas no obvious
inhibition of VEGF protein was observed in VEGF shRNA2, 3 and 4
transfected cells.
Proliferation of C6GSCs
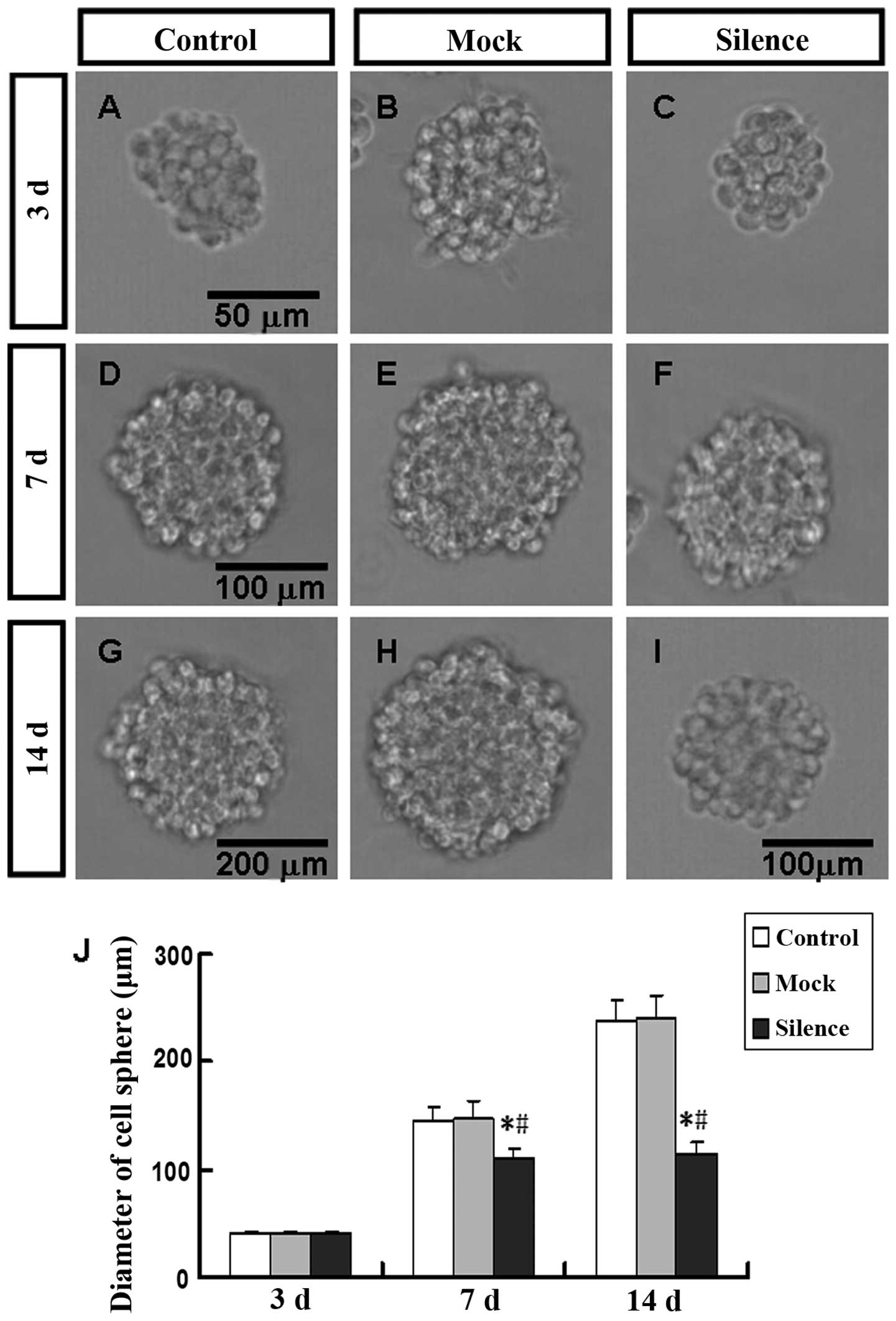
The C6GSCs of 3 groups were cultured under the same
conditions outlined in Materials and methods. The appearance of the
sphere of three kinds of stem cells is similar at the beginning,
showing a small and uniform cell sphere (Fig. 5A–C). In the following days in
culture, nontransfected C6GSCs and mock transfected C6GSC spheres
grew significantly faster and reached a larger size than lentiviral
vector-VEGF shRNA1 transfected C6GSC spheres (Fig. 5D–I). At 7 DIV, the proliferation of
lentiviral vector-VEGF shRNA1 transfected C6GSCs reached the peak.
On the contrary, the other two groups proliferated continually and
the diameter of a cell sphere was up to ∼236.6820.43 and
240.8520.52 μm (Fig. 5J).
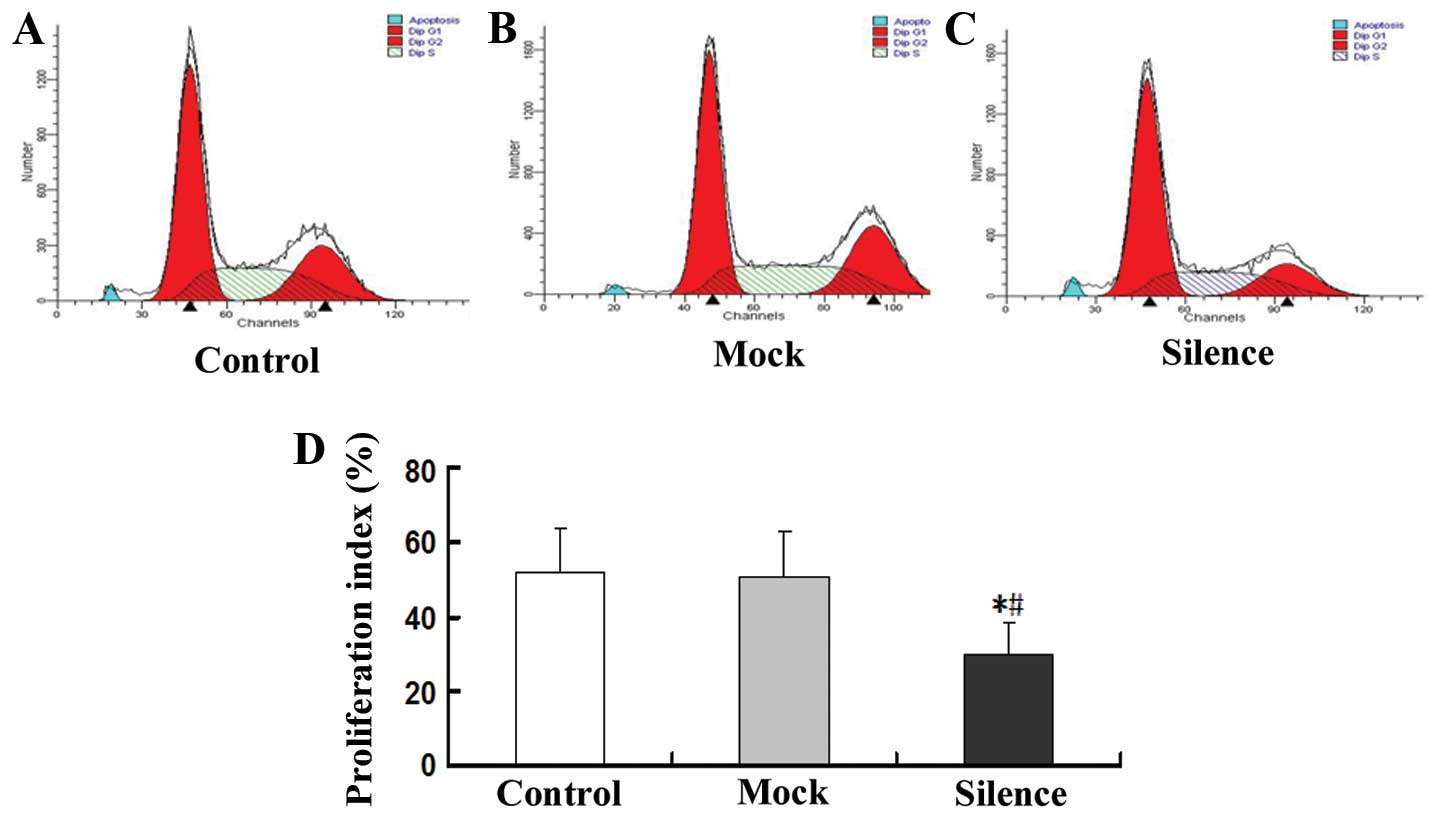
Flow cytometry also indicated that proliferation index of
lentiviral vector-VEGF shRNA1 transfected C6GSCs were lower than
that of the other two groups (Fig.
6).
Differentiation of C6GSCs
Differentiation culture of three kinds of stem cells
was performed in the conditions outlined in Materials and methods.
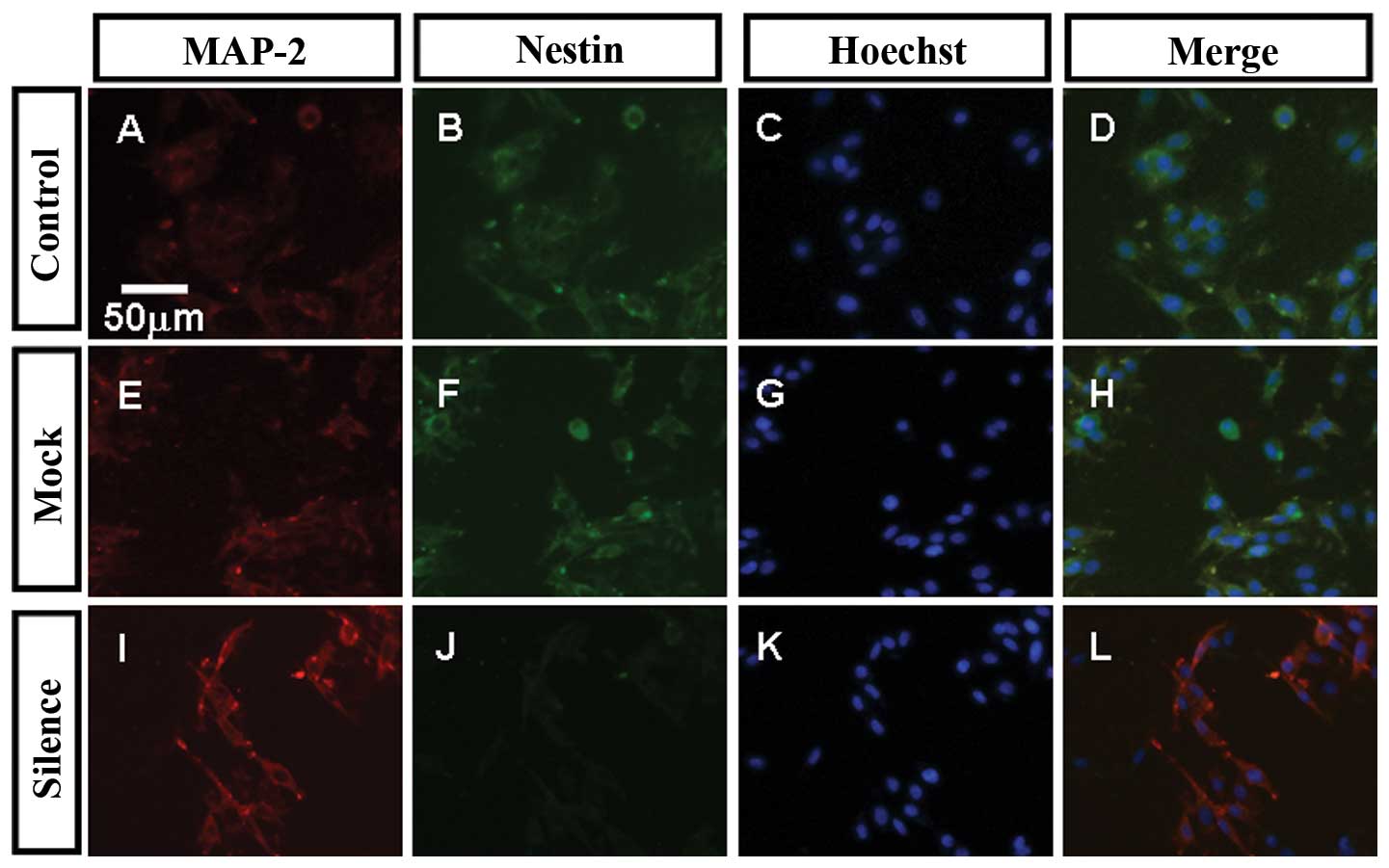
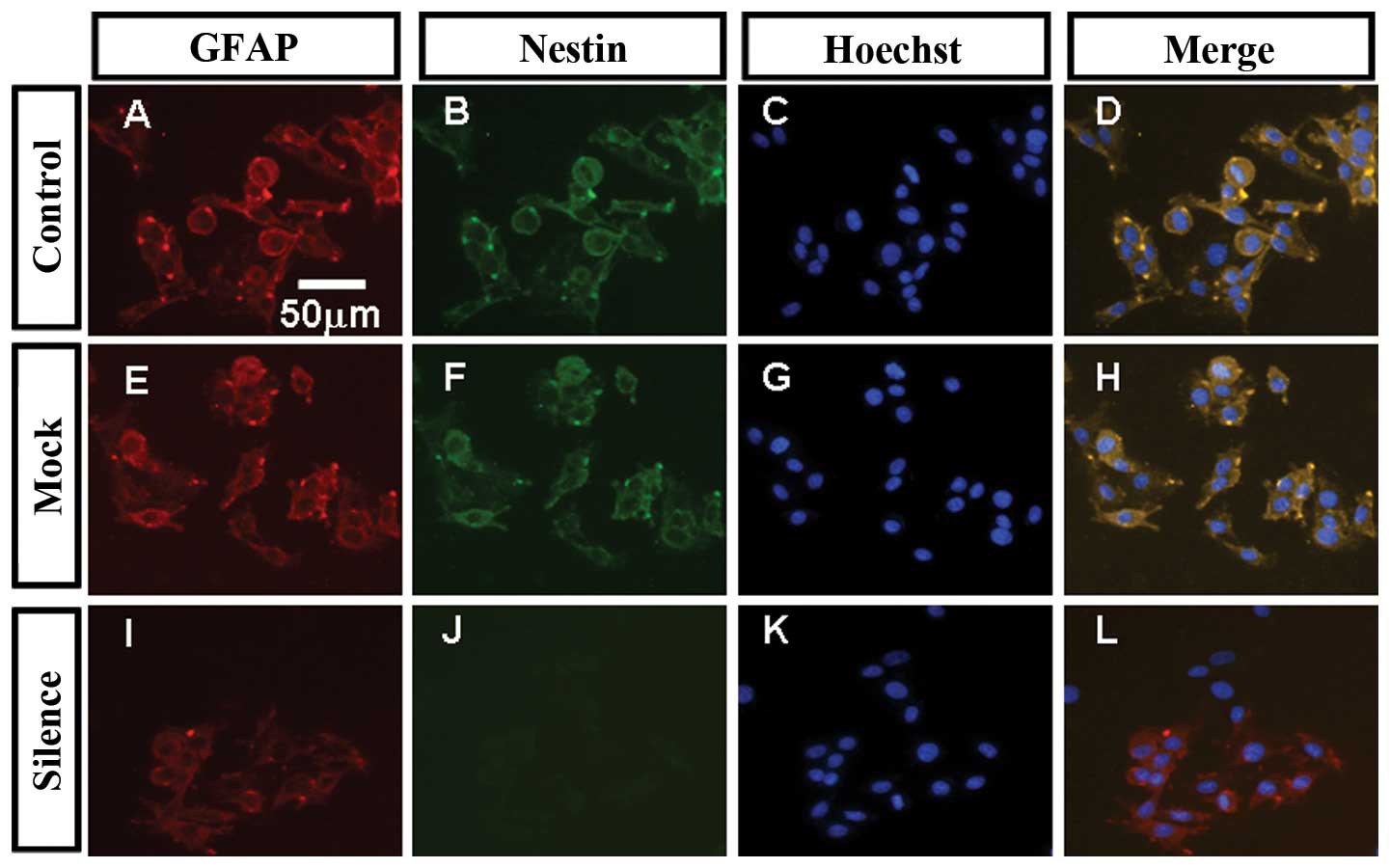
After 7 DIV, three kinds of stem cells exhibited multi-directional
differentiation, all of them differentiated into MAP-2-positive
neurons and GFAP-positive astrocytes. MAP-2-positive neurons and
GFAP-positive astrocytes derived from lentiviral vector-VEGF shRNA1
transfected C6GSCs were nestin-negative, cell morphology was good,
showing extensive processes and rounded bodies. MAP-2-positive
neurons and GFAP-positive astrocytes derived from the other two
groups were nestin-positive, cell morphology was less distinct, the
differentiated cells were poorly differentiated (Figs. 7 and 8).
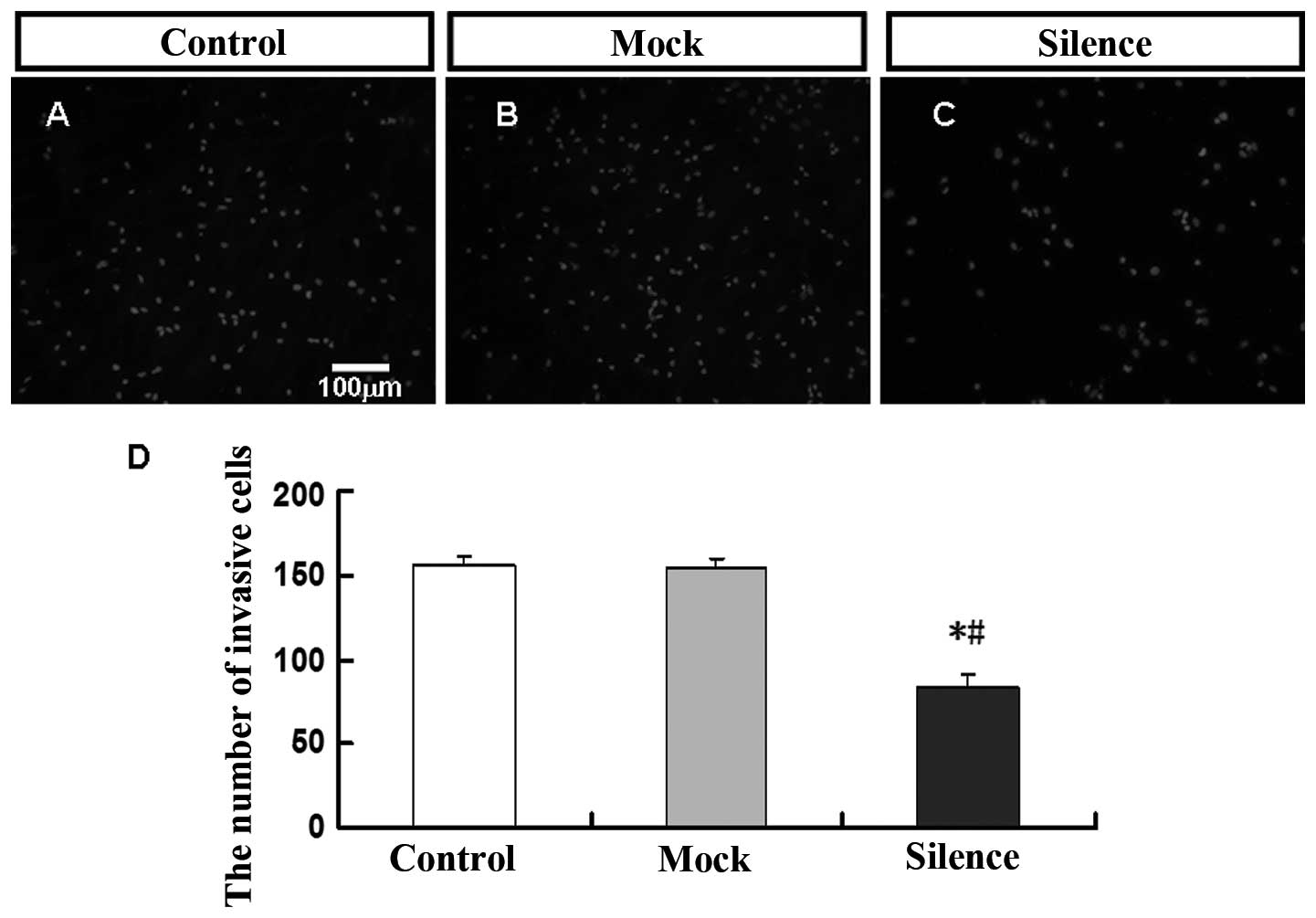
Invasion of C6GSCs
Cell invasion is a characteristic feature of
carcinoma cells. To investigate the role of VEGF in tumor cell
invasion, we measured the ability of C6GSCs to invade through
Matrigel-coated filters in a Transwell chamber assay. After
transfected with VEGF shRNA1, the invasive cells were obviously
decreased compared with the other two groups. However, there were
no significant differences between the other two groups (Fig. 9).
Discussion
Gliomas are characterized by high morphological,
genetic, and phenotypic heterogeneity. However, studies performed
simply on bulk glioma cells yield limited information on its origin
and the mechanisms which mediate its growth and progression.
Encouragingly, by applying the methods used to study normal NSCs to
brain tumor cells, many researcher have reported there were BTSCs
in brain gliomas of different grades from both children and adults
(26–31). BTSCs possess the ability to
self-renew and to give rise to a variety of proliferating and
differentiated cells that make up the brain tumor mass. BTSCs play
an important role in resistance to radiotherapy and chemotherapy
(32,33), angiogenesis (34) and metastasis (35). However, their source and molecular
signal pathways are still not fully understood.
There have been only a few successful isolations of
BTSCs from established cell lines such as the human U373, A172 and
U87 glioma cell lines (36–38).
Although the rat glioma cell line C6 is widely used in the study of
gliomas, there is still controversy over the culture methods for
BTSCs. The pioneering studies suggested cell surface markers such
as CD133 (a cell surface marker for normal NSCs) and nestin (a
cytoskeleton protein associated with NSCs and progenitor cells in
central nervous system development) are recommended for the
specific identification of BTSCs (28,30,39,40).
In this study, the C6 cell line was cultured in a simplified
serum-free NSC medium, in which a fraction of C6 cells could form
stem cell spheres. The C6GSC spheres were CD133 and nestin
double-positive.
Direct evidence shows that gliomas and BTSCs express
high levels of VEGF (8–11). In this study, RT-PCR and western
blot analysis showed that also the C6GSCs could express VEGF.
Traditionally, it is thought VEGF secreted by tumor cells interacts
with VEGFRs expressed on the surface of tumor endothelial cells,
but not on tumor cells (18–20)
and promote the migration, growth and survival of endothelial cells
(12–14). Recent studies suggest that tumor
cells or BTSCs could express VEGFRs themselves (21–24).
However, the direct effect of VEGF on biological behavior of BTSCs
has not been completely elucidated.
We suggest that VEGF may be a key factor influencing
the characteristics of C6GSCs. RNAi is highly specific and
efficient in inhibiting target genes and is the focus of recent
studies to make it a new tool for curing tumor, infectious and
hereditary diseases (41). In this
study, lentiviral vector-VEGF shRNA was constructed to target the
VEGF genes in the C6GSCs. After 72-h transfection, the VEGF mRNA
expression in lentiviral vector-VEGF shRNA1 transfected C6GSCs was
reduced by ∼75%. Compared to control and mock groups, the ability
of cell proliferation of lentiviral vector-VEGF shRNA1 transfected
C6GSCs was decreased significantly and GFAP, MAP-2-positive cells
were nestin-negative, which indicated that the ability of
differentiation was enhanced. After transfected with VEGF shRNA1,
the invasive cells were obviously decreased compared with the other
two groups through Matrigel-coated filters in a Transwell chamber
assay. All the data demonstrate that downregulation of VEGF
expression attenuates malignant biological behavior of C6GSCs. RNAi
of VEGF expression implies an effective anti-glioma strategy.
References
|
1.
|
Louis DN: Molecular pathology of malignant
gliomas. Annu Rev Pathol. 1:97–117. 2006. View Article : Google Scholar
|
|
2.
|
Rasper M, Schafer A, Piontek G, et al:
Aldehyde dehydrogenase 1 positive glioblastoma cells show brain
tumor stem cell capacity. Neuro Oncol. 12:1024–1033. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Folkman J: Role of angiogenesis in tumor
growth and metastasis. Semin Oncol. 29:15–18. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Verhoeff JJ, van Tellingen O, Claes A, et
al: Concerns about anti-angiogenic treatment in patients with
glioblastoma multiforme. BMC Cancer. 9:4442009. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Keck PJ, Hauser SD, Krivi G, et al:
Vascular permeability factor, an endothelial cell mitogen related
to PDGF. Science. 246:1309–1312. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Leung DW, Cachianes G, Kuang WJ, Goeddel
DV and Ferrara N: Vascular endothelial growth factor is a secreted
angiogenic mitogen. Science. 246:1306–1309. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Takano S, Yoshii Y, Kondo S, et al:
Concentration of vascular endothelial growth factor in the serum
and tumor tissue of brain tumor patients. Cancer Res. 56:2185–2190.
1996.PubMed/NCBI
|
|
8.
|
Nishikawa R, Cheng SY, Nagashima R, Huang
HJ, Cavenee WK and Matsutani M: Expression of vascular endothelial
growth factor in human brain tumors. Acta Neuropathol. 96:453–462.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Berkman RA, Merrill MJ, Reinhold WC, et
al: Expression of the vascular permeability factor/vascular
endothelial growth factor gene in central nervous system neoplasms.
J Clin Invest. 91:153–159. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Hamerlik P, Lathia JD, Rasmussen R, et al:
Autocrine VEGFVEGFR2-Neuropilin-1 signaling promotes glioma
stem-like cell viability and tumor growth. J Exp Med. 209:507–520.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
He H, Niu CS and Li MW: Correlation
between glioblastoma stem-like cells and tumor vascularization.
Oncol Rep. 27:45–50. 2012.PubMed/NCBI
|
|
12.
|
Gu Q, Wang D, Wang X, et al: Basic
fibroblast growth factor inhibits radiation-induced apoptosis of
HUVECs. I. The PI3K/AKT pathway and induction of phosphorylation of
BAD. Radiat Res. 161:692–702. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Gingis-Velitski S, Zetser A, Flugelman MY,
Vlodavsky I and Ilan N: Heparanase induces endothelial cell
migration via protein kinase B/Akt activation. J Biol Chem.
279:23536–23541. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Gerber HP, McMurtrey A, Kowalski J, et al:
Vascular endothelial growth factor regulates endothelial cell
survival through the phosphatidylinositol 3′-kinase/Akt signal
transduction pathway. Requirement for Flk-1/KDR activation. J Biol
Chem. 273:30336–30343. 1998.
|
|
15.
|
de Vries C, Escobedo JA, Ueno H, Houck K,
Ferrara N and Williams LT: The fms-like tyrosine kinase, a receptor
for vascular endothelial growth factor. Science. 255:989–991.
1992.
|
|
16.
|
Millauer B, Wizigmann-Voos S, Schnurch H,
et al: High affinity VEGF binding and developmental expression
suggest Flk-1 as a major regulator of vasculogenesis and
angiogenesis. Cell. 72:835–846. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Terman BI, Dougher-Vermazen M, Carrion ME,
et al: Identification of the KDR tyrosine kinase as a receptor for
vascular endothelial cell growth factor. Biochem Biophys Res
Commun. 187:1579–1586. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Plate KH, Breier G, Weich HA, Mennel HD
and Risau W: Vascular endothelial growth factor and glioma
angiogenesis: coordinate induction of VEGF receptors, distribution
of VEGF protein and possible in vivo regulatory mechanisms. Int J
Cancer. 59:520–529. 1994. View Article : Google Scholar
|
|
19.
|
Norden AD, Drappatz J and Wen PY:
Antiangiogenic therapies for high-grade glioma. Nat Rev Neurol.
5:610–620. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Iwamoto FM and Fine HA: Bevacizumab for
malignant gliomas. Arch Neurol. 67:285–288. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Gorski DH, Beckett MA, Jaskowiak NT, et
al: Blockage of the vascular endothelial growth factor stress
response increases the antitumor effects of ionizing radiation.
Cancer Res. 59:3374–3378. 1999.
|
|
22.
|
Graeven U, Fiedler W, Karpinski S, et al:
Melanoma-associated expression of vascular endothelial growth
factor and its receptors FLT-1 and KDR. J Cancer Res Clin Oncol.
125:621–629. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Knizetova P, Ehrmann J, Hlobilkova A, et
al: Autocrine regulation of glioblastoma cell cycle progression,
viability and radioresistance through the VEGF-VEGFR2 (KDR)
interplay. Cell Cycle. 7:2553–2561. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Hlobilkova A, Ehrmann J, Knizetova P,
Krejci V, Kalita O and Kolar Z: Analysis of VEGF, Flt-1, Flk-1,
nestin and MMP-9 in relation to astrocytoma pathogenesis and
progression. Neoplasma. 56:284–290. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Kabos P, Ehtesham M, Kabosova A, Black KL
and Yu JS: Generation of neural progenitor cells from whole adult
bone marrow. Exp Neurol. 178:288–293. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Ignatova TN, Kukekov VG, Laywell ED,
Suslov ON, Vrionis FD and Steindler DA: Human cortical glial tumors
contain neural stem-like cells expressing astroglial and neuronal
markers in vitro. Glia. 39:193–206. 2002. View Article : Google Scholar
|
|
27.
|
Hemmati HD, Nakano I, Lazareff JA, et al:
Cancerous stem cells can arise from pediatric brain tumors. Proc
Natl Acad Sci USA. 100:15178–15183. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Singh SK, Clarke ID, Terasaki M, et al:
Identification of a cancer stem cell in human brain tumors. Cancer
Res. 63:5821–5828. 2003.PubMed/NCBI
|
|
29.
|
Galli R, Binda E, Orfanelli U, et al:
Isolation and characterization of tumorigenic, stem-like neural
precursors from human glioblastoma. Cancer Res. 64:7011–7021. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Singh SK, Hawkins C, Clarke ID, et al:
Identification of human brain tumour initiating cells. Nature.
432:396–401. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Yuan X, Curtin J, Xiong Y, et al:
Isolation of cancer stem cells from adult glioblastoma multiforme.
Oncogene. 23:9392–9400. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Beier D, Hau P, Proescholdt M, et al:
CD133(+) and CD133(−) glioblastoma-derived cancer stem cells show
differential growth characteristics and molecular profiles. Cancer
Res. 67:4010–4015. 2007.
|
|
33.
|
Bao S, Wu Q, McLendon RE, et al: Glioma
stem cells promote radioresistance by preferential activation of
the DNA damage response. Nature. 444:756–760. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Dean M, Fojo T and Bates S: Tumour stem
cells and drug resistance. Nat Rev Cancer. 5:275–284. 2005.
View Article : Google Scholar
|
|
35.
|
Bao S, Wu Q, Sathornsumetee S, et al: Stem
cell-like glioma cells promote tumor angiogenesis through vascular
endothelial growth factor. Cancer Res. 66:7843–7848. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Kang SK, Park JB and Cha SH: Multipotent,
dedifferentiated cancer stem-like cells from brain gliomas. Stem
Cells Dev. 15:423–435. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Patrawala L, Calhoun T,
Schneider-Broussard R, Zhou J, Claypool K and Tang DG: Side
population is enriched in tumorigenic, stem-like cancer cells,
whereas ABCG2+ and ABCG2− cancer cells are similarly tumorigenic.
Cancer Res. 65:6207–6219. 2005.
|
|
38.
|
Yu SC, Ping YF, Yi L, et al: Isolation and
characterization of cancer stem cells from a human glioblastoma
cell line U87. Cancer Lett. 265:124–134. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
39.
|
Shen G, Shen F, Shi Z, et al:
Identification of cancer stem-like cells in the C6 glioma cell line
and the limitation of current identification methods. In Vitro Cell
Dev Biol Anim. 44:280–289. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
40.
|
Zhou XD, Wang XY, Qu FJ, et al: Detection
of cancer stem cells from the C6 glioma cell line. J Int Med Res.
37:503–510. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
41.
|
Jain RK: Normalizing tumor vasculature
with anti-angiogenic therapy: a new paradigm for combination
therapy. Nat Med. 7:987–989. 2001. View Article : Google Scholar : PubMed/NCBI
|