Introduction
A major problem in the treatment of leukemia is the
appearance of multi-drug resistance (1). Chemoresistance often is at the origin
of a major number of relapses which are mediated by the expulsion
activity of the ABC pumps.
The microenvironment is intimately related to the
development of drug resistance in cancer cells. However, the
precise nature of molecular events in action between the stromal
and leukemic cells remains still elusive. Chemoresistance of
leukemic cells, besides the microenvironment is also controlled by
the ATP binding cassette genes (ABC), notably ABCB1 (MDR1) which
code for the P-gp protein. In a rather unorthodox series of
experiments we identified recently the presence of a distinct
population of stromal cells in the tumor microenvironment which we
baptized ‘Hospicells’ (BMH). These cells were found to establish
very particular relationship with HL60 (acute leukemia cell lines),
MDA-MB-231-GFP (breast cancer cell line) and OVCAR-3GFP (ovarian
cancer cell line) (2–7). In solid tumors, the very presence of
Hospicells were found to confer drug resistance to cancer cells
during chemotherapy via oncologic trogocytosis (2,3), as
a result of upregulation of ATP binding cassette gene family
(4).
Hospicells by their presence promotes tumorigenicity
and angiogenesis (5) besides
influencing the immune response of tumor microenvironments
(6,7). A plethora of cell metabolic functions
such as vasculogenic mimicry, cancer cell proliferation, inhibition
of cell death and metastasis are known today to be influenced by
IGF-1 (8–20).
This is the first report concerning the effect of
IGF-I on the regulation of the ABC genes (MDR1, MRP1,
MRP2, MRP3, MRP5 and BCRP) in leukemic
cells when they are put in contact with a subset of bone marrow
derived stromal cell named Hospicells.
Materials and methods
Cell culture
Four cell lines were used in this study, HL-60,
(acute promyelocytic leukemia ATCC, CCL-240), K-562 (chronic
myelogenous leukemia ATCC, CCL 243), K562 and HL60 derived from
K562/Dox and HL60/Dnr, which were developed as daunorubicin and
doxorubicin resistant lines, respectively. These cells are
deposited at the Leukemia Tumor Bank, Hôpital Saint Antoine, Paris.
The phenotype for HL60 wild-type (CD13+,
CD15+, CD33+ CD3−,
CD14−, CD19−, HLA-Dr−) and for
K562 wild-type (BCRAbl+ ‘Philadelphia chromosome’,
CD13+, glycophorin+ and CD3−,
CD19−) were determined by flow cytometry. These two cell
lines are routinely used as reference cells for chromosome mutation
(K562-BCRAbl) and HL60 for leukemia cell endothelial protein-C
receptor. All cell lines were cultured using RPMI-1640 containing
10% fetal bovine serum (FBS), 1% L-glutamine 50 U/ml penicillin and
50 μg/ml streptomycin and incubated at 37°C in 5% CO2
atmosphere.
Bone marrow Hospicells
Hospicells were isolated from eight AML bone marrows
(Hôpital Hotel Dieu, Paris). The bone marrow-adherent mononuclear
cell population (BMMNCs) isolation was carried out by Ficoll
density gradient centrifugation as described (21). The contaminating monocytes in the
mononuclear cell population were excluded by adhesion on plastic
plates for 30 min. Then, the non-adherent cells were collected and
cultured for 2 weeks in wells coated with 0.2% gelatin in
endothelial cell basal medium MV2 (Promocell, Heidelberg, Germany)
supplemented with final concentrations of 1 g/ml ascorbic acid, 10
ng/ml b-FGF, 5 ng/ml EGF, 20 ng/ml insulin growth factor-1 (IGF-1),
0.5 ng/ml VEGF and 15% FBS. The bone marrow Hospicells were
isolated as described (2–7).
Adhesion assay
Adherent BMMNCs (105) were seeded in
6-well plates with their respective culture medium and left to
adhere. After completion of cell adhesion, they were washed twice
with PBS. HL60 (3×106) were plated onto the adherent
cell monolayer at 37°C for 24 min. Then, the supernatant containing
non-adherent cells was discarded and the cell monolayer was again
washed twice with PBS. The number of BMH in the adherent cell
population, obtained after 24 h culture was evaluated. When 5 or
more leukemic cells adhered to a single bone marrow stromal cell
the latter was considered to be a Hospicell.
Treatment of cells with IGF-I and IGF-IR
inhibitor
The leukemic cell lines were incubated with IGF-I
(200 ng/ml) and IGF-IR inhibitor PPP (picropodophyllin, 1 μM) from
Calbiochem (Paris).
Reverse transcription and polymerase
chain reaction (RT-PCR)
Total RNA was extracted with the Nucleospin RNA II
kit (Macherey-Nagel EURL, Hoerdt, France). Reverse transcription
was performed using M-MLV reverse transcriptase and oligo(dt)
primers (Gibco-BRL, Paisley, UK). The polymerase chain reaction
(PCR) was performed by Taq DNA polymerase (Gibco-BRL).
Specific primers for IGF-I (sense: 5′-AAA TCA GCA G TC TTC
CAA C-3′ and antisense: 5′-CTT CTG GGT CTT GGG CAT GT-3′);
IGF-II (sense: 5′-AGT CGA TGC TGG CTT CTC A-3′ and
antisense: 5′-GTG GGC GGG GTCT TGG GTG GGT AG-3′); IGF-IR
(sense: 5′-GAC ATC CGC AAC GAC TAT CAG-3′ and antisense: 5′-GTA GTT
ATT GGA CAC CGC ATC-3′); IGF-IIR (sense: 5′-TAC AAC TTC CGG
TGG TAC ACC A-3′ and antisense: 5′-CAT GGC ATA CCA GTT TCC TCC
A-3′); MDR1 (sense: 5′-GTT ATA GGA AGT TTG AGT TT-3′ and
antisense: 5′-AAA AAC TAT CCC ATA ATA AC-3′); MRP (sense:
5′-AAT GCG CCA AGA CTA GGA AG-3′ and antisense: 5′-ACG GGA GGA TGT
TGA ACA AG-3′); MRP2 (sense: 5′-CTG GTT GAT GAA GGC TCT
GA-3′ and antisense: 5′-CTG CCA TAA TGT CCA GGT TC-3′); MRP3
(sense: 5′-GCA GGT GAC ATT TGC TCT GA-3′ and antisense: 5′-CCC TCT
GAG CAC TGG AAG TC-3′); MRP5 (sense: 5′-GGA TAA CTT CTC AGT
GGG-3′ and antisense: 5′-GGA ATG GCA ATG CTC TAA AG-3′);
BCRP (sense: 5′-TTA GGA TTG AAG CCA AAG G-3′ and antisense:
5′-TAG GCA ATT GTG AGG AAA ATA-3′) and β2-microglubulin
(sense: 5′-CCA GCA GAG AAT GGA AAG TC-3′ and antisense: 5′-GAT GCT
GCT TAC ATG TCT CG-3′). The PCR products, along with a 100-bp DNA
ladder, were analysed by electrophoresis on agarose gels containing
ethidium bromide.
IGF expression by leukemic cells
The presence of proteins belonging to IGF family in
cell lines was revealed by immunocytochemistry. Cells were seeded
and fixed at 20,000 cells/well in glass bottom chamber slides
(Nunc, Lab-Tek, Naperville, IL, USA). They were then permeabilized
and incubated for 2 h at room temperature either with specific
primary antibodies (dilution 1/200) anti-IGF-I, -II, -IR or -IIR
(R&D Systems, Minneapolis, MN, USA). After several washes, the
cells were incubated successively with biotinylated secondary
antibody and streptavidine coupled to fluorescein isothiocyanate
(dilution 1/500), for 45 min. Isotypic controls were performed
concurrently and nuclei were DAPI-labeled. The cells were then
visualized by fluorescence microscopy.
Analysis of P-gp expression
P-gp was studied by using UIC2 (Immunotech, France),
monoclonal antibody, followed by labeling with a secondary antibody
conjugated with phycoerythrin. Cells (1×106) were fixed
and permeabilized using IntraPrep™ (Beckman Coulter, Villepinte,
France) as per the manufacturer’s instructions. Fluorescence was
measured and analyzed by flow cytometry. Protein expression for
each transporter was quantified as the mean fluorescence intensity
(MFI) shift (ratio of the MFI of antibody and isotype control). All
experiments were performed in triplicate.
Studies of drug resistance of HL60 and
HL60/Dnr cells in the presence of Hospicells
The Hospicells were seeded first at 60% confluency
in a 96-well flat-bottomed culture plate with complete medium
containing 10% FBS. After 18 h incubation, the leukemic cells
(10,000 cells/well) were added and co-cultured for 24 h with
Hospicells in the presence of IGF-I (200 μg/ml) or IGF-IR inhibitor
(1 μM before addition of daunorubicin). The effect of these
cytotoxic agents was evaluated by optical density (OD) measurement
using the multi-well plate reader (Wallac, Perkin-Elmer, Waltham,
MA, USA). The result is representative of three independent
experiments.
Statistical analyses
The results are presented as mean ± SE and data were
analyzed using Student’s t-test P-value (<0.05 was considered
significant).
Results
Expression of IGFs and IGF-R by leukemic
cells
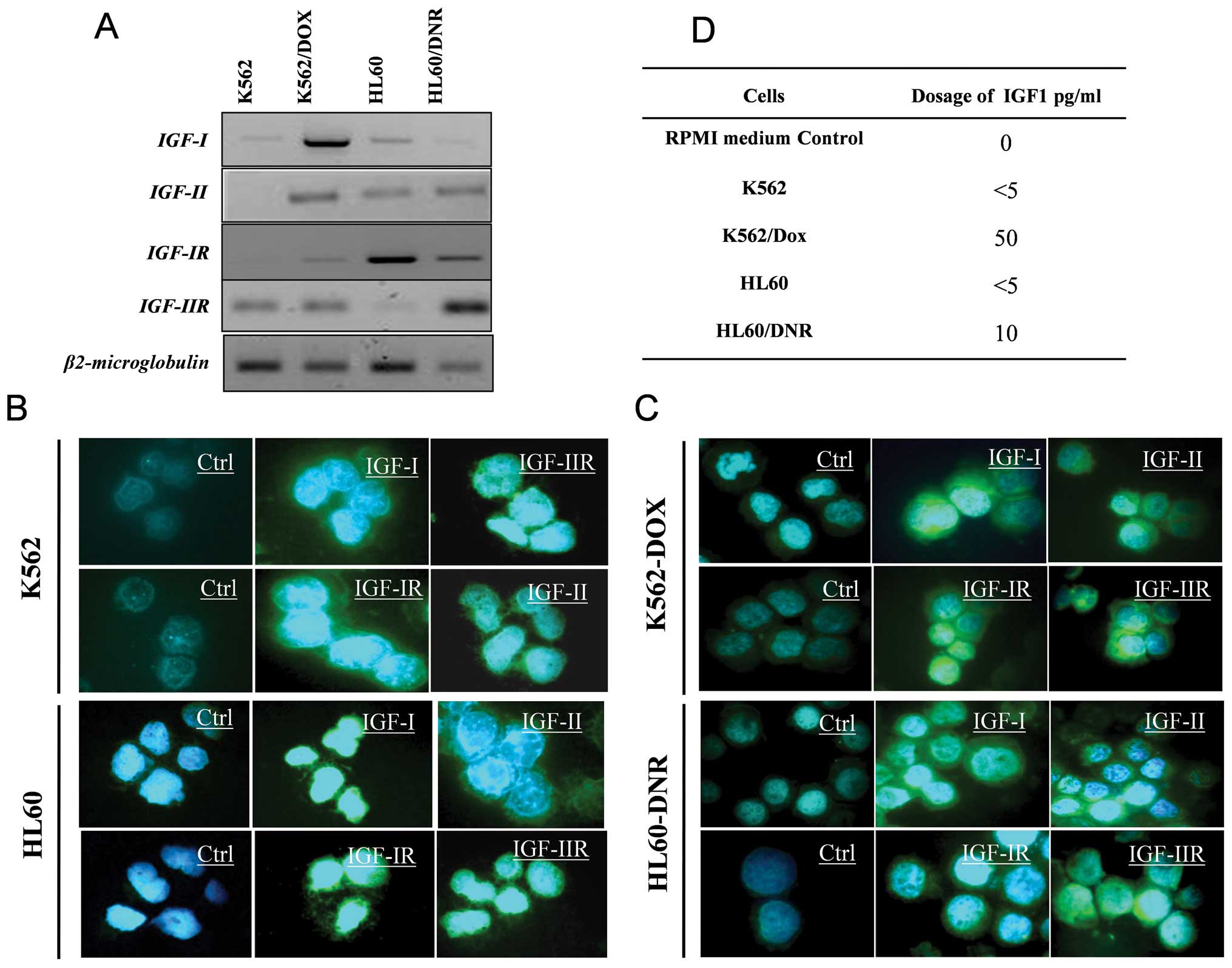
Fig. 1A shows the
four cell lines (HL60 and K562 sensitive and resistant) the
difference in transcription levels of IGFs and their receptor
genes. These results were confirmed at the protein level by
immunocytochemistry (Fig. 1B and
C). Fig. 1D indicates the
amount of IGF-I in the supernatants of the sensitive and resistant
cells. The results show that the resistant cells secrete more IGF-I
(50 pg/ml) than the sensitive cells (<5 pg/ml).
Expression of P-gp by the sensitive and
resistant cells
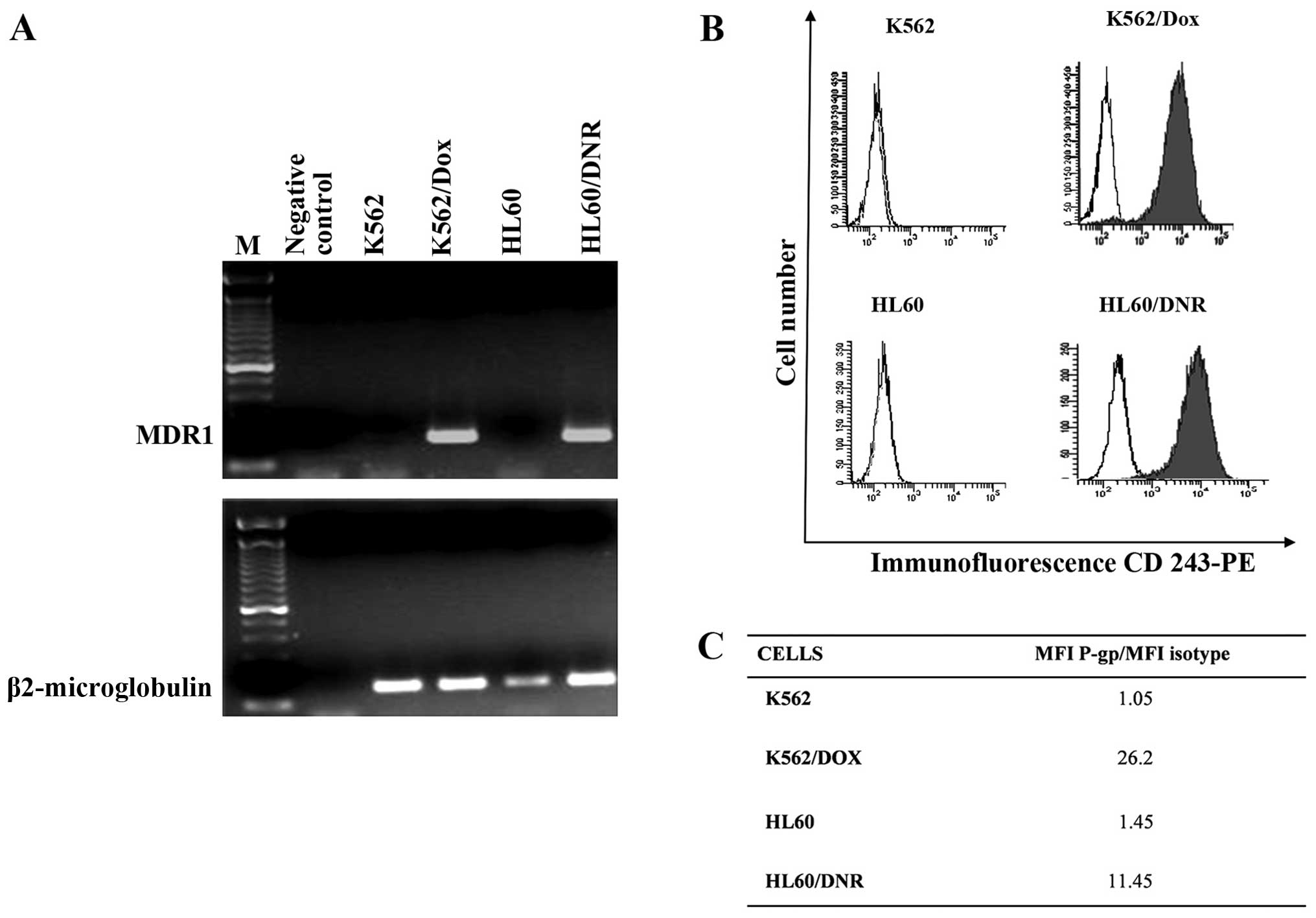
The MDR1 gene coding P-gp protein was studied
by the RT-PCR. Fig. 2A shows that
MDR1 mRNA is strongly expressed by resistant cells whereas the
sensitive cells not at all. These results were confirmed by flow
cytometry of P-gp protein (Fig.
2B). The ratios of the Mean Fluorescence Intensity (MFI) of
P-gp in resistant cells as against the value for controls are
presented in Fig. 2C. Thus it can
be inferred that the drug expulsion mechanism is active in the
resistance cells and not in the controls.
The effect of IGF-I on cell
proliferation
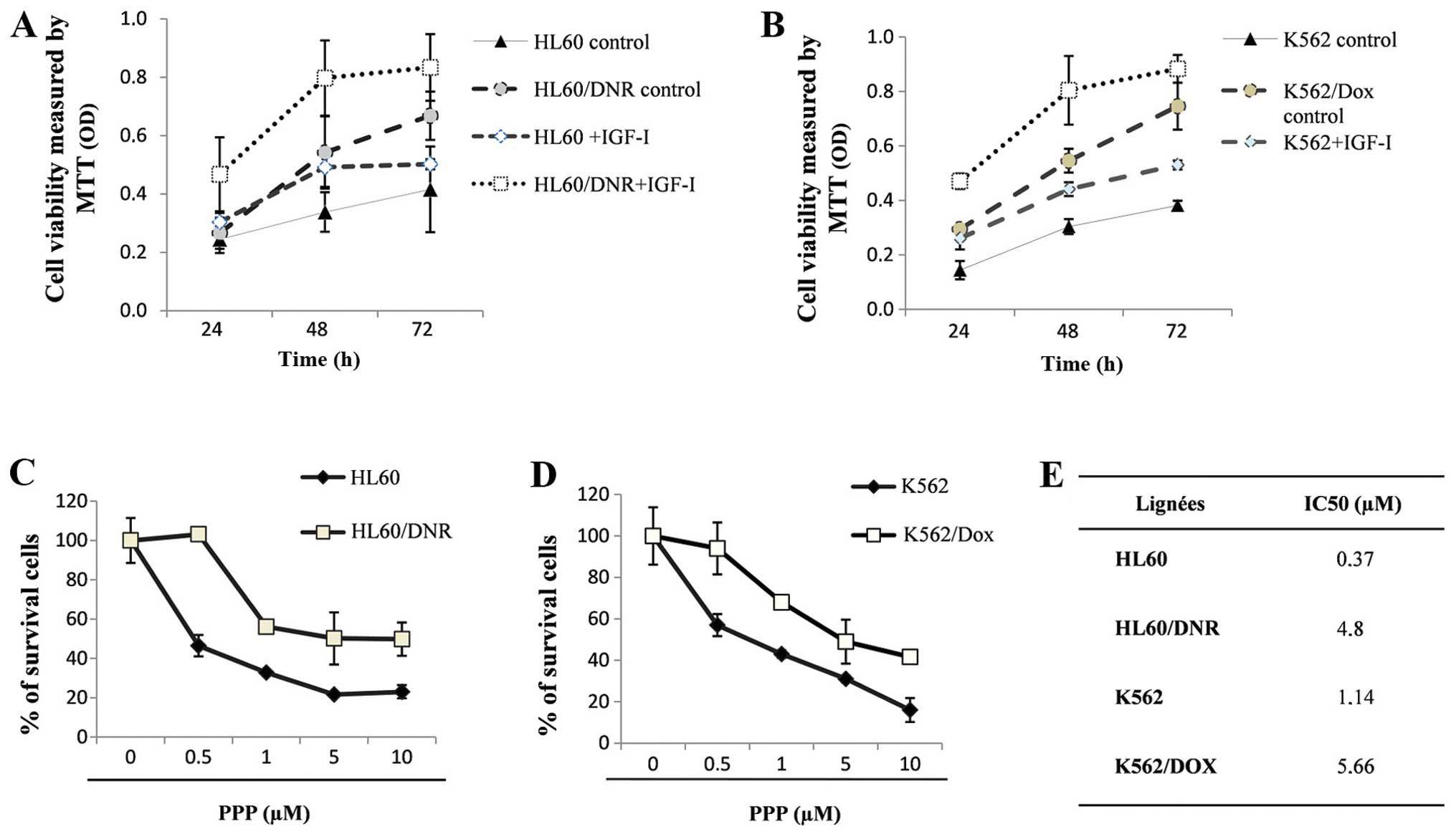
The effect of IGF-I on the proliferation rate of
sensitive K562 and HL60 and resistant K562/Dox and HL60/Dnr cells
are shown (Fig. 3A and B). The
rate of proliferation of the sensitive cells was lower than that of
the resistant ones. These results show that IGF-I promotes
proliferation, its effect being more important in the resistant
cells than in the sensitive cells.
We also determined the effect of the cyclolignan
PPP, an IGF-IR inhibitor (22), on
the resistant cells which express P-gp: HL60/Dnr (Fig. 3C) and K562/Dox (Fig. 3D) and sensitive cells K562 and HL60
which do not. The values of the IC50 of PPP in these
cells were evaluated by MTT tests. Fig. 3E (see table inserted) shows that
the value of the IC50 of PPP is higher in the resistant
cells as compared to sensitive cells.
IGF-I mediated expression of MDR1 mRNA
and P-gp in the sensitive and resistant cells
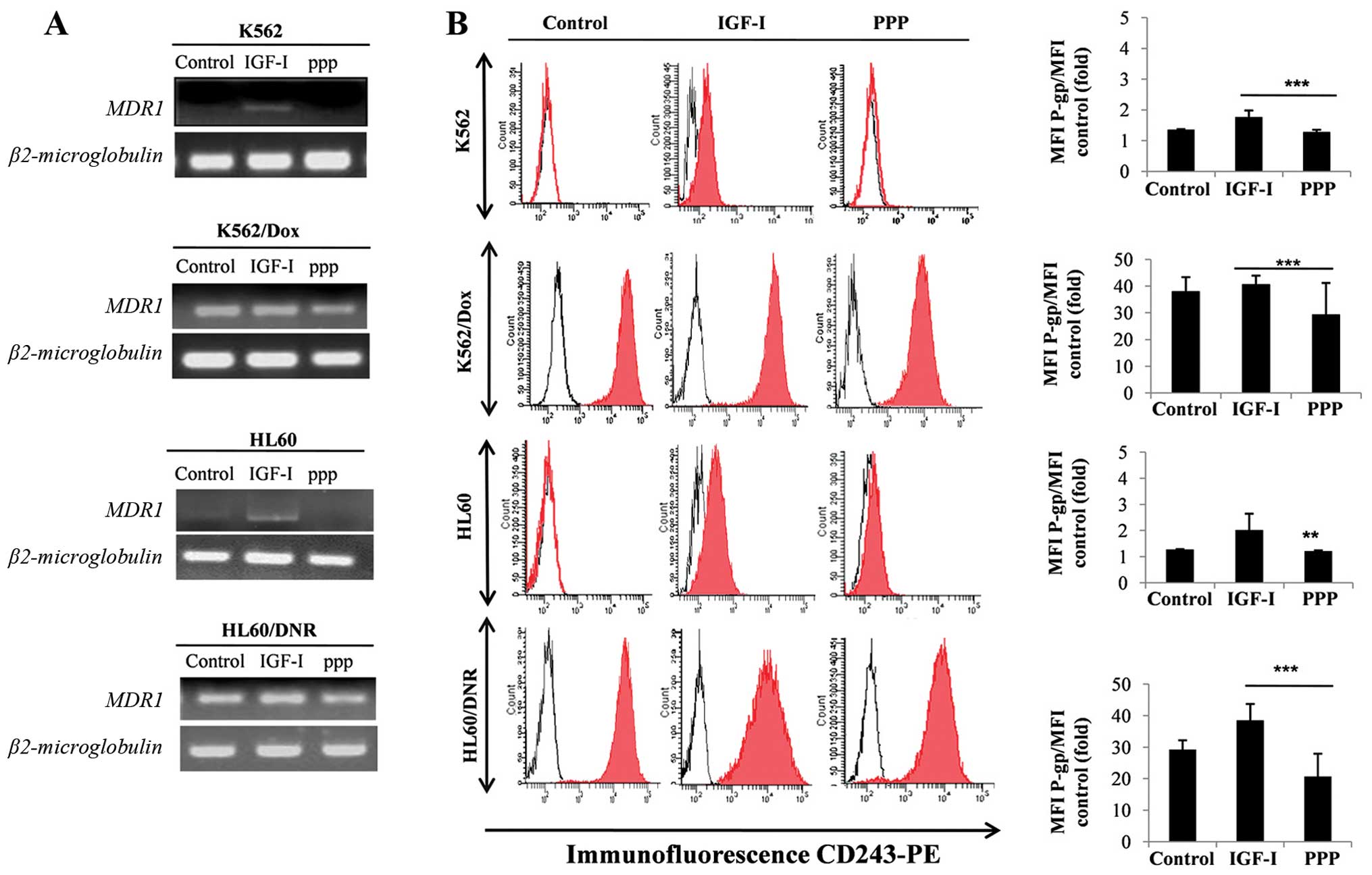
Fig. 4A shows
induction of MDR1 mRNA expression and its protein P-gp (Fig. 4B) in cells treated by IGF-1. This
expression of P-gp, induced by IGF-I, is inhibited by the presence
of PPP in both resistant and sensitive cells. The results suggest
that IGF-I, via IGF-IR, is able to induce and control the
expression of P-gp, the major protein implicated in
chemoresistance.
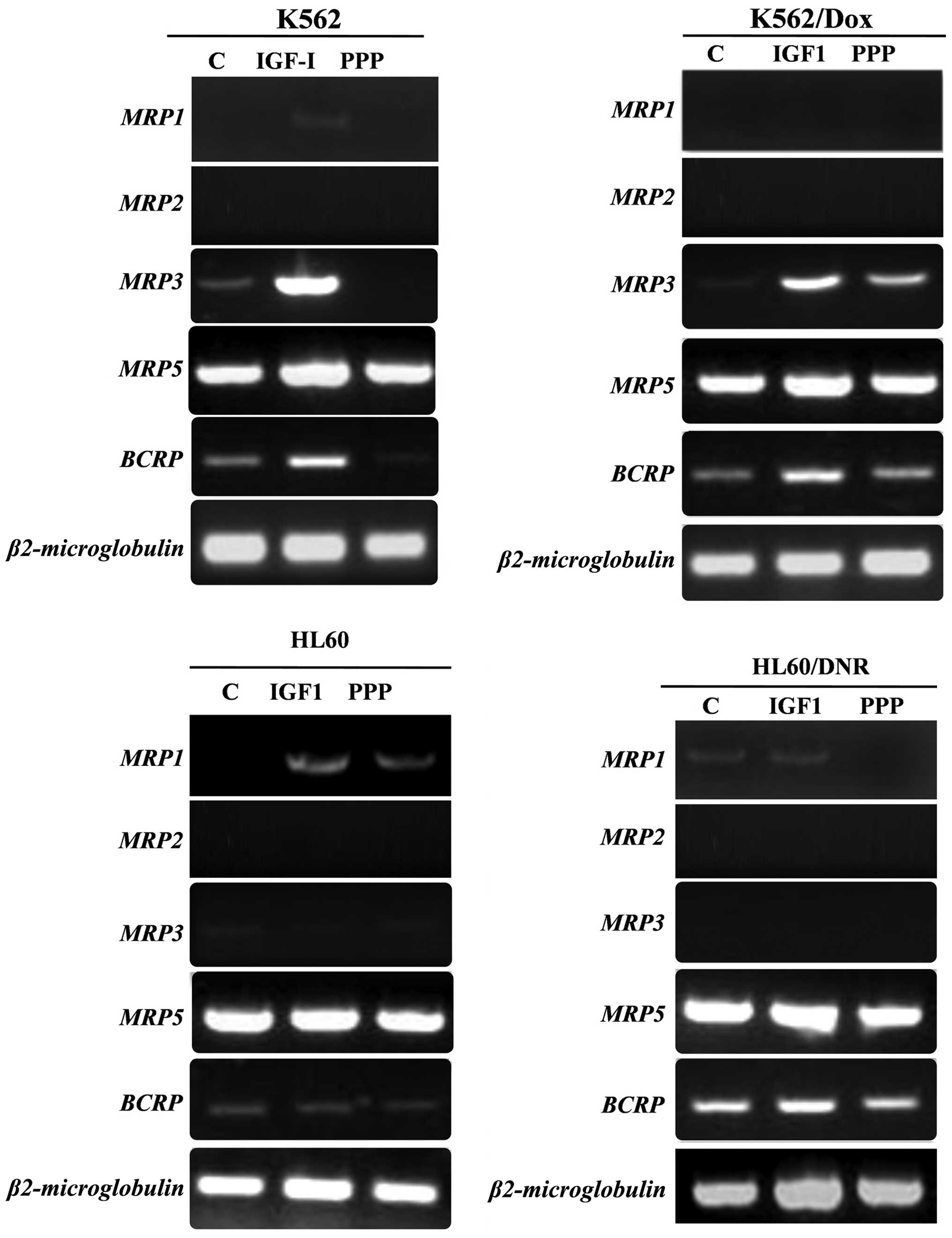
Regulation of ABC genes by IGF-I
Normally, the expression of MRP1,
MRP2, MRP3 and BCRP is low or non-existent in
the 4 cell lines (K562, K562/Dox, HL60 and HL60/Dnr), whereas
MRP5 is strongly expressed (Fig. 5). In the K562 and its derivative
K562/Dox cells, the expression of MRP3 and BCRP
increased significantly in the presence of IGF-I. On the other
hand, the expression of MRP3 and BCRP, in these same cells
decreases or disappears in the presence of PPP.
In HL60/Dnr, we found no significant difference in
the expression of MRP1 mRNA in the presence of IGF-I, however,
exposure to PPP in the medium led to the inhibition of MRP1
gene expression. We found no difference in the expression of BCRP
and MRP2 mRNAs in the HL60 cells and its derivative HL60/Dnr.
Whereas, the expression of MRP5 gene was not affected by
addition of IGF-I nor that of PPP.
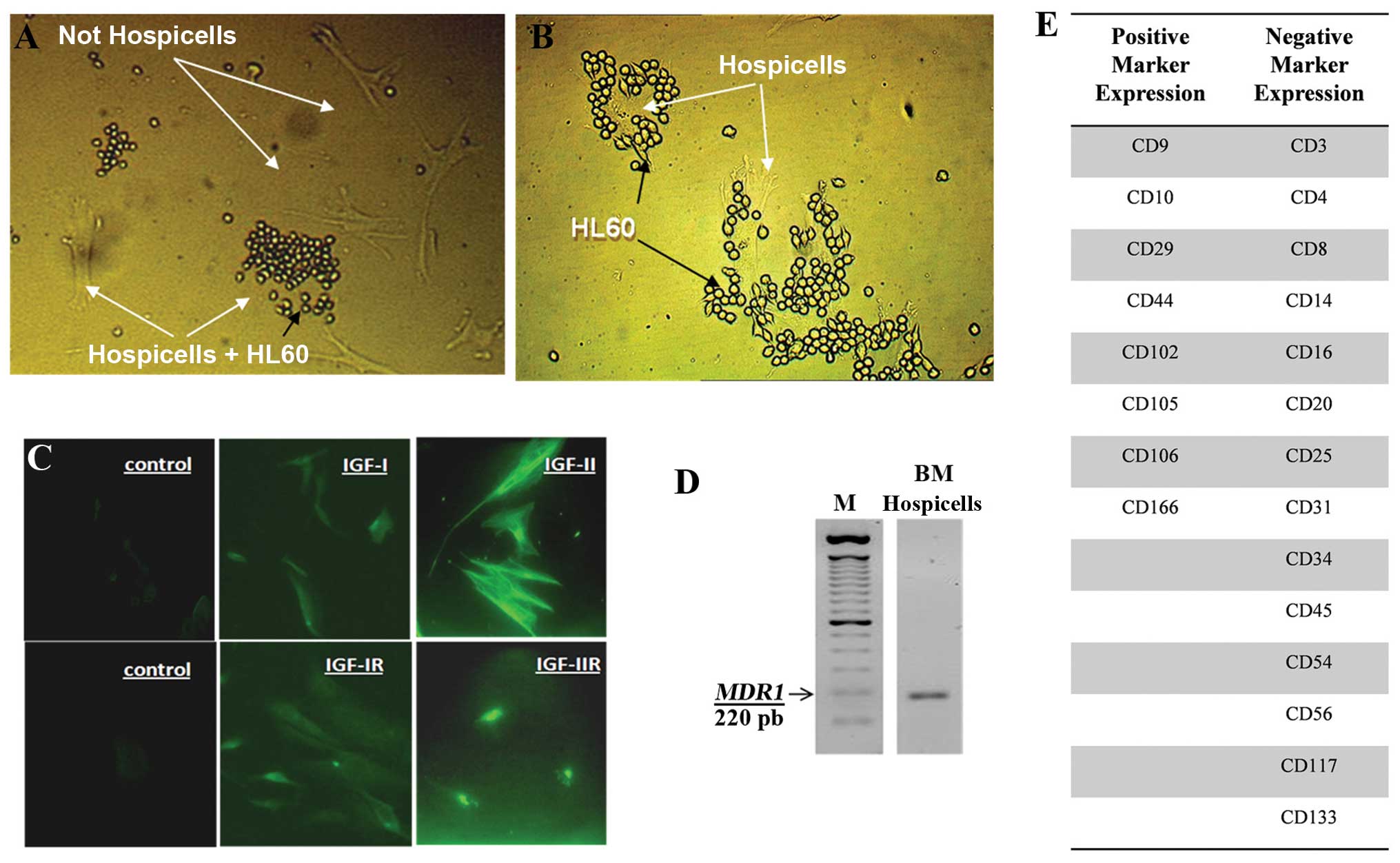
Expression of IGF, IGF-R and MDR1 by
Hospicells
Hospicells from the stroma were identified because
of their property to interact with HL60 cells. Fig. 6A shows HL60 interaction with a
subset of stromal cells (Hospicells) and Fig. 6B with the enriched population of
Hospicells. These cells express IGF-I, IGF-II, IGF-IR and IGF-IIR,
as studied by immunocytochemistry (Fig. 6C) and P-gp (MDR-1) mRNA by RT-PCR
(Fig. 6D).
Transcriptional regulation of the ABC
genes through the association of HL60 with Hospicells
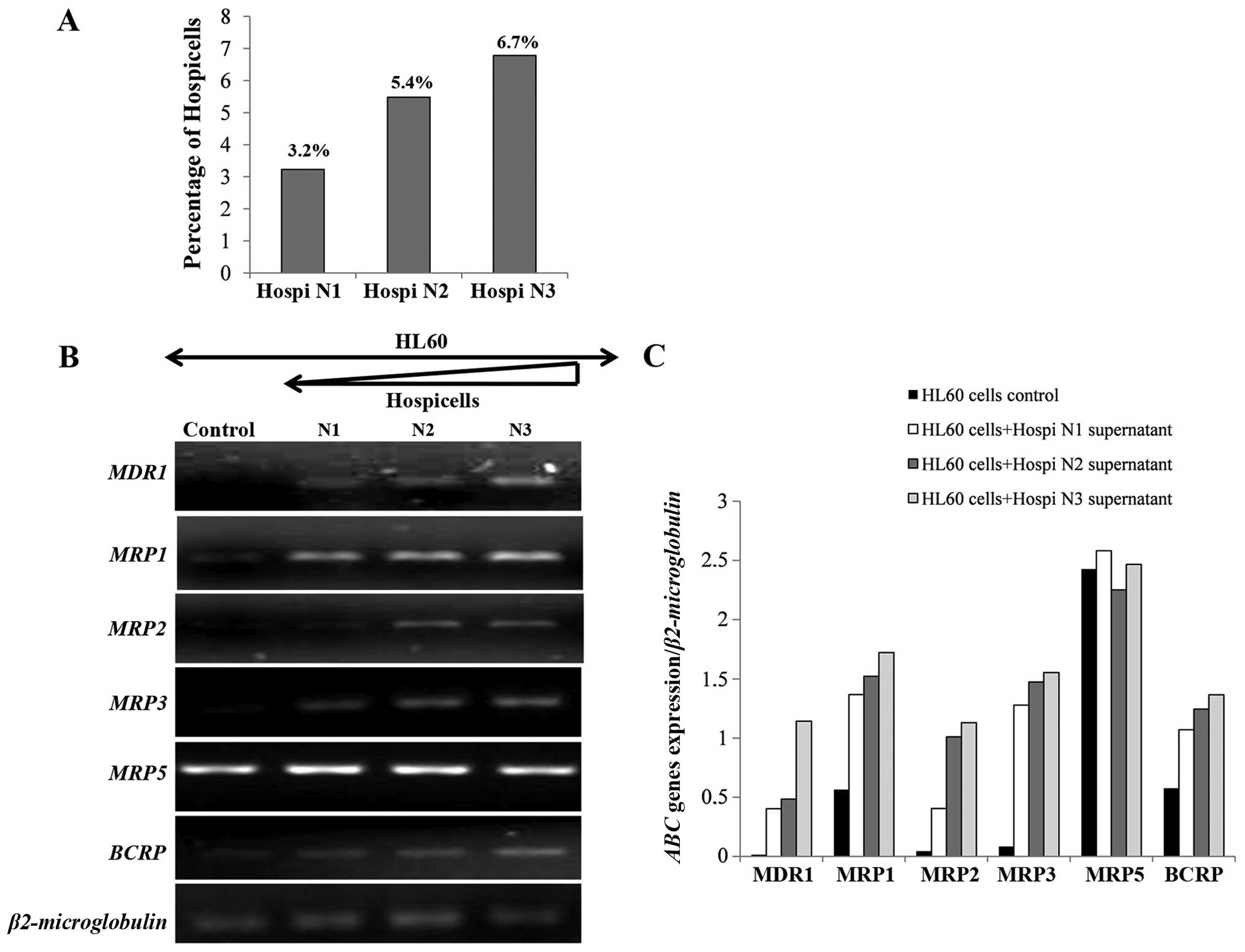
The number of Hospicells was counted in each bone
marrow stroma preparation. It varied from 3 to 9.7% of the total
stromal cells. In normal bone marrow only 0.7% of stromal cells are
Hospicells (data not shown). Three samples were chosen from AML
bone marrow depending on the number of Hospicells (Fig. 7A). Fig. 7B shows that HL60 cells express mRNA
for MDR1, MRP2 and MRP3 in the presence of the
Hospicells whereas it was not expressed in control cells. Moreover,
the level of expression of ABC genes was directly related to the
number of Hospicells present initially in the three BM samples.
However, no difference in the expression of MRP5 in the
presence of the Hospicells compared to the control was seen
(Fig. 7C).
Fig. 7D and E show
that HL60 cells co-cultivated with Hospicells from AML (N3 rich in
Hospicells), express the ABC genes. On the other hand, the addition
of PPP decreased the expression of MDR1 and MRP1 as
also the BCRP.
These results are interesting since it may suggest
that IGF-I was secreted by Hospicells or HL60 cells and acts
through autocrine and/or paracrine mechanisms on MDR1,
MRP1, MRP2 and BCRP genes.
Interaction between HL60 and Hospicells
in the presence of IGF-I and induction of chemoresistance
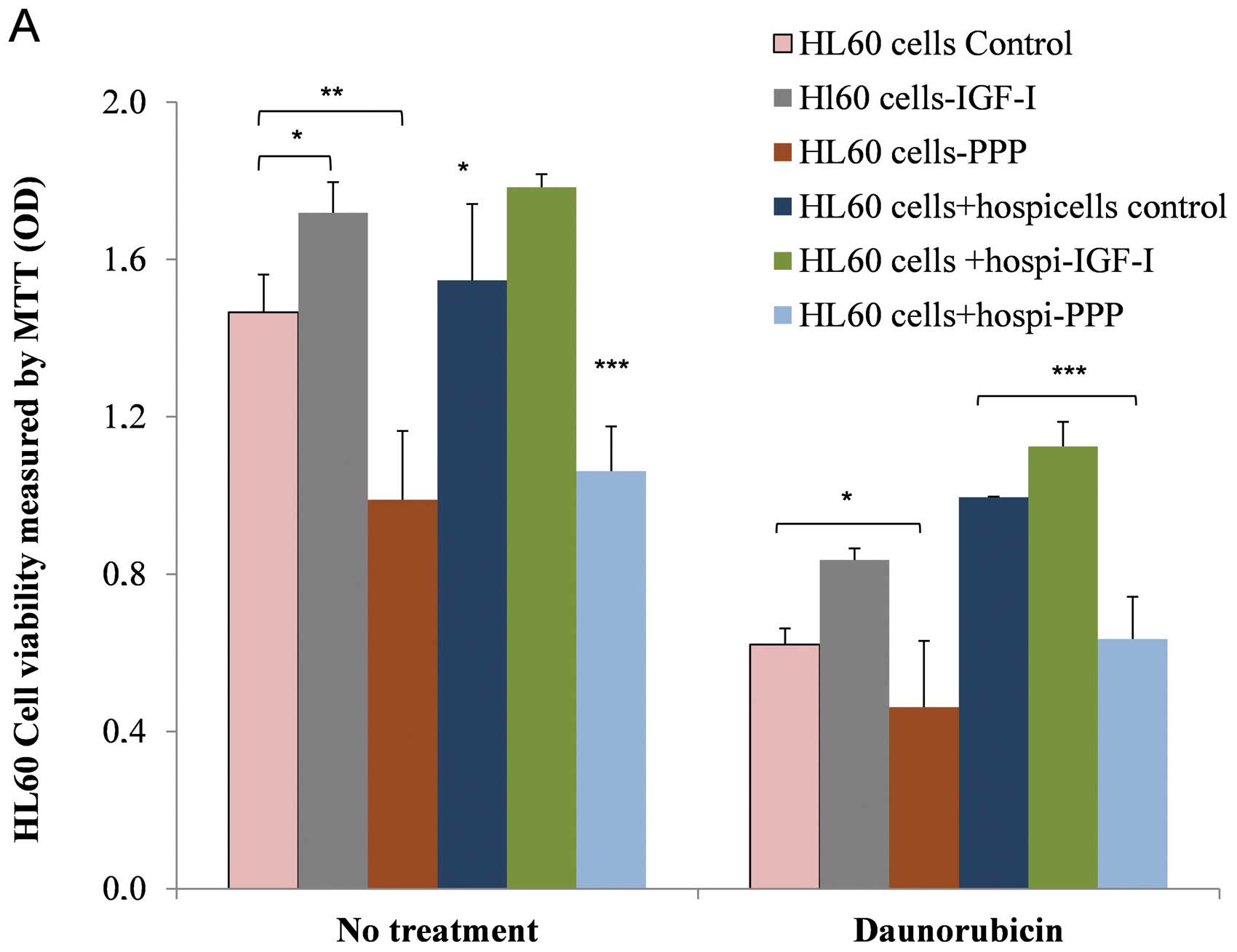
Our results show that under culture conditions
(without treatment), HL60 sensitive cell proliferation is
stimulated by IGF-I (P=0.02) whereas this strongly diminishes in
the presence of PPP (P=0.002). This indeed confirms our earlier
results (Fig. 8A). In the presence
of Hospicells, we observed a significant increased proliferative
activity of HL60 cells (P=0.03) as compared to controls. HL60 cells
when in physical contact with the Hospicells display a clear
resistance to daunorubicin. This resistance is reinforced further
by IGF-I (P=0.03) whereas it decreases in the presence of PPP
(P=0.02).
These observations confirm that, on one hand IGF-I
regulates ABC gene expression (mainly MDR1), whereas
Hospicells provide protection to HL60 leukemic cells against the
effect of chemotherapy. Fig. 8B
depicts the interaction between HL60/Dnr cells and Hospicells in
the presence of IGF-I and the enhancement of chemoresistance. It
shows that proliferation of these resistant cells is stimulated by
IGF-I (P=0.0002), whereas it decreases in the presence of PPP
(P=0.0003). Thus the interaction between Hospicells and HL60/Dnr
cells increases the proliferation of the resistance cells which is
further amplified by the presence of IGF-I. The inhibition of
IGF-IR by the addition of PPP reduces this drug resistance.
Discussion
Of the two insulin-like growth factors, IGF-I and
IGF-II, IGF-I binds to two distinct cell surface receptors named
IGF receptor-I (IGF-IR) and the insulin receptor (INSR). The
binding of IGF-I/IGF-IR occurs at a higher affinity than IGFI/INSR
binding. IGF-IR is a transmembrane tyrosine kinase receptor and is
activated by IGF-I and IGF-II (24–27).
IGF-II binds to the IGF-II receptor and acts as a
signaling antagonist. IGF-I and IGF-II form complexes with
insulin-like growth factor binding protein (IGFBP). Integrin αvβ3
also plays an important role in IGF-IR signaling and its biological
functions (28). Several studies
have shown that IGF-I is involved in the progression of malignant
hematologic diseases (23).
During this study, the characteristics of the
myeloid cell lines, both sensitive and resistant to the agents of
chemotherapy, were defined by studying the expression of
MDR1 and its protein P-gp. In addition, the proliferative
effect of IGF-I on sensitive or resistant cells and the effect of
the IGF-IR inhibitor (PPP) were ascertained. It was noted that the
basal level of proliferation of the resistant cells was well above
that of the sensitive cells. Exogenous addition of IGF-I stimulated
proliferation of the two cell lines without marked difference.
The degree of resistance displayed by HL60 (in
contact with Hospicells) was more pronounced when the proportion of
Hospicells in givens stroma was high. This was partially abolished
by the addition of PPP. However, MRP5 was an exception and remained
unchanged. The addition of IGF-I increased the expression of P-gP
which could be inhibited again by PPP. The co-cultures of
Hospicells with sensitive leukemic cells showed that the Hospicells
were able to transmit the character of drug resistance to the
sensitive cells. We had previously provided evidence that bone
marrow stromal cells secrete significant amounts of FGF (fibroblast
growth factor), SDF-1α (stromal cell-derived factor 1) and IGF-1
(8). In the present study we
report that bone marrow Hospicells also secrete IGF-1.
Consequently, IGF (via autocrine or paracrine pathway) could be
considered a strong candidate for the regulation of ABC genes and
chemoresistance of HL60 cells.
In an unpublished study we had observed that the
interaction of cancer cells with BMH was integrin-dependent.
Cell-cell interaction activates integrin signaling pathway which
play a crucial role in IGF signaling (28). The IGF-IR and its ligands may also
promote growth of metastatic cells in the bone, the preferred site
of metastases (29). Recently, we
reported that Hospicells from solid tumors can be involved in
chemoresistance via oncologic trogocytosis, i.e., through transfer
of MDR proteins onto the incoming cancer cells (2) or upregulation of the ABC genes
(3). The IGFs are among the more
abundant growth factors in bone tissue and are synthesized by
various bone marrow cells including stromal cells (8) and BMH. Within the microenvironment,
the stromal Hospicells, in concert with IGF-1, provide strong
synergetic effect for the maintenance and proliferation of cancer
cells. In addition, BMH may represent a niche for homing of cancer
cells and the secretion of IGF-1 which provide protection while
promoting their proliferation and chemoresistance.
Our results demonstrate that tumor stromal cells
contribute in the physiopathological response to IGF-1/IGF-1R.
Targeting IGF-1R in multiple myeloma (30) and breast cancer (31) indicated a decrease in the formation
of tumors and a diminuation of angiogenesis. In view of the scant
attention given so far to the role of microenvironment, in the
behavior and biology of the leukemic cells, we have focused
attention in the present study on the intimate relationships
between Hospicells, IGF-1 and malignant cells.
IGF and IGF1R expression levels are relevant
indicators of tumor stage and/or disease progression. Additional
studies will be necessary to further clarify the mode of action of
the ABC pumps as also other pumps such as LRP (lung
resistance-related protein). Curiously IGF seems not to have any
effect on MRP5 gene expression.
In conclusion our results suggest the importance of
the microenvironment and the IGF-I pathway in drug resistance of
leukemic cells. A better knowledge of this close relationship can
be helpful in the search for new openings in cancer therapy.
References
|
1
|
Baudard M, Beauchamp-Nicoud A, Delmer A,
Rio B, Blanc C, Zittoun R and Marie JP: Has the prognosis of adult
patients with acute myeloid leukemia improved over years? A single
institution experience of 784 consecutive patients over a 16-year
period. Leukemia. 13:1481–1489. 1999.
|
|
2
|
Rafii A, Mirshahi P, Poupot M, Faussat AM,
Simon A, Ducros E, Mery E, Couderc B, Lis R, Capdet J, Bergalet J,
Querleu D, Dagonnet F, Fournié JJ, Marie JP, Pujade-Lauraine E,
Favre G, Soria J and Mirshahi M: Oncologic trogocytosis of an
original stromal cells induces chemoresistance of ovarian tumours.
PLoS One. 3:e38942008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lis R, Capdet J, Mirshahi P, Lacroix-Triki
M, Dagonnet F, Klein C, Mirshahi M, Fournié JJ, Rafii A and Poupot
M: Oncologic trogocytosis with Hospicells induces the expression of
N-cadherin by breast cancer cells. Int J Oncol. 37:1453–1461.
2010.PubMed/NCBI
|
|
4
|
Benabbou N, Mirshahi P, Cadillon M, Soria
J, Therwath A and Mirshahi M: Hospicells promote upregulation of
the ATP binding cassette genes by insulin like growth factor-I via
JAK2- STAT3 signaling pathway in ovarian cancer cell line. Int J
Oncol. 43:685–694. 2013.PubMed/NCBI
|
|
5
|
Pasquet M, Golzio M, Mery E, Rafii A,
Benabbou N, Mirshahi P, Hennebelle I, Bourin P, Allal B, Teissie J,
Mirshahi M and Couderc B: Hospicells (ascites-derived stromal
cells) promote tumorigenicity and angiogenesis. Int J Cancer.
126:2090–2101. 2010.PubMed/NCBI
|
|
6
|
Martinet L, Poupot R, Mirshahi P, Rafii A,
Fournié JJ, Mirshahi M and Poupot M: Hospicells derived from
ovarian cancer stroma inhibit T-cell immune responses. Int J
Cancer. 126:2143–2152. 2010.PubMed/NCBI
|
|
7
|
Castells M, Thibault B, Mery E, Golzio M,
Pasquet M, Hennebelle I, Bourin P, Mirshahi M, Delord JP, Querleu D
and Couderc B: Ovarian ascites-derived Hospicells promote
angiogenesis via activation of macrophages. Cancer Lett. 326:59–68.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mirshahi P, Rafii A, Vincent L, Berthaut
A, Varin R, Kalantar G, Marzac C, Calandini OA, Marie JP, Soria C,
Soria J and Mirshahi M: Vasculogenic mimicry of acute leukemic bone
marrow stromal cells. Leukemia. 23:1039–1048. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wu X, Tortolero-Luna G, Zhao H, Phatak D,
Spitz MR and Follen M: Serum levels of insulin-like growth factor I
and risk of squamous intraepithelial lesions of the cervix. Clin
Cancer Res. 9:3356–3361. 2003.PubMed/NCBI
|
|
10
|
Yang SY and Winslet M: The IGF system in
carcinogenesis and its implication for cancer therapy. Curr Oncol.
18:301–302. 2011.PubMed/NCBI
|
|
11
|
Samani AA, Yakar S, Le Roith D and Brodt
P: The role ofhe IGF system in cancer growth and metastasis:
overview and recent insights. Endocr Rev. 28:20–47. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Héron-Milhavet L and LeRoith D:
Insulin-like growth factor I induces MDM2-dependent degradation of
p53 via the p38 MAPK pathway in response to DNA damage. J Biol
Chem. 277:15600–15606. 2002.PubMed/NCBI
|
|
13
|
He Y, Zhang J, Zheng J, Du W, Xiao H, Liu
W, Li X, Chen X, Yang L and Huang S: The insulin-like growth
factor-1 receptor kinase inhibitor, NVP-ADW742, suppresses survival
and resistance to chemotherapy in acute myeloid leukemia cells.
Oncol Res. 19:35–43. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Abe S, Funato T, Takahashi S, Yokoyama H,
Yamamoto J, Tomiya Y, Yamada-Fujiwara M, Ishizawa K, Kameoka J,
Kaku M, Harigae H and Sasaki T: Increased expression of
insulin-like growth factor 1 is associated with Ara-C resistance in
leukemia. Tohoku J Exp Med. 209:217–228. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kuhn DJ, Berkova Z, Jones RJ, Woessner R,
Bjorklund CC, Ma W, Davis RE, Lin P, Wang H, Madden TL, Wei C,
Baladandayuthapani V, et al: Targeting the insulin-like growth
factor-1 receptor to overcome bortezomib resistance in preclinical
models of multiple myeloma. Blood. 120:3260–3270. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Guo YS, Jin GF, Houston CW, Thompson JC
and Townsend CM Jr: Insulin-like growth factor-I promotes multidrug
resistance in MCLM colon cancer cells. J Cell Physiol. 175:141–148.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Schwarze CP, Neu S, Beck J, Mavridou K,
Ranke MB and Binder G: Influence of IGF-I and cell density on MDR1
expression in the T-lymphoblastoid cell line CCRF-CEM. Horm Res.
52:192–199. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shimon I and Shpilberg O: The insulin-like
growth factor system in regulation of normal and malignant
hematopoiesis. Leuk Res. 19:233–240. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Doepfner KT, Spertini O and Arcaro A:
Autocrine insulin-like growth factor-I signaling promotes growth
and survival of human acute myeloid leukemia cells via the
phosphoinositide 3-kinase/Akt pathway. Leukemia. 21:1921–1930.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Baier TG, Jenne EW, Blum W, Schönberg D
and Hartmann KK: Influence of antibodies against IGF-I, insulin or
their receptors on proliferation of human acute lymphoblastic
leukemia cell lines. Leuk Res. 16:807–814. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gilmore MJ, Prentice HG, Blacklock HA,
Janossy G and Hoffbrand AV: A technique for rapid isolation of bone
marrow mononuclear cells using Ficoll-Metrizoate and the IBM 2991
blood cell processor. Br J Haematol. 50:619–626. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Girnita A, Girnita L, del Prete F,
Bartolazzi A, Larsson O and Axelson M: Cyclolignans as inhibitors
of the insulin-like growth factor-1 receptor and malignant cell
growth. Cancer Res. 64:236–242. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Qiang YW, Kopantzev E and Rudikoff S:
Insulin like growth factor-I signaling in multiple myeloma:
downstream elements, functional correlates, and pathway cross-talk.
Blood. 99:4138–4146. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rodon J, DeSantos V, Ferry RJ Jr and
Kurzrock R: Early drug development of inhibitors of the
insulin-like growth factor-I receptor pathway: lessons from the
first clinical trials. Mol Cancer Ther. 7:2575–2588. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gregory CW, DeGeorges A and Sikes RA: The
IGF axis in the development and progression of prostate cancer.
Recent Res Dev Cancer. 3:437–462. 2001.
|
|
26
|
Macaulay VM: Insulin-like growth factors
and cancer. Br J Cancer. 65:311–320. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Burren CP, Berka JL, Edmondson SR, Werther
GA and Batch JA: Localization of mRNAs for insulin-like growth
factor-I (IGF-I), IGF-I receptor, and IGF binding proteins in rat
eye. Invest Ophthalmol Vis Sci. 37:1459–1468. 1996.PubMed/NCBI
|
|
28
|
Clemmons DR and Maile LA: Integral
membrane proteins that function coordinately with the insulin-like
growth factor I receptor to regulate intracellular signaling.
Endocrinology. 144:1664–1670. 2003. View Article : Google Scholar
|
|
29
|
Turner HE, Harris AL, Melmed S and Wass
JA: Angiogenesis in endocrine tumors. Endocr Rev. 24:600–632. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wu KD, Zhou L, Burtrum D, Ludwig DL and
Moore MA: Antibody targeting of the insulin like growth factor I
receptor enhances the anti-tumor response of multiple myeloma to
chemotherapy through inhibition of tumor proliferation and
angiogenesis. Cancer Immunol Immunother. 56:343–357. 2007.
|
|
31
|
Karamouzis MV and Papavassiliou AG:
Targeting insulin-like growth factor in breast cancer therapeutics.
Crit Rev Oncol Hematol. 84:8–17. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hopkins A, Crowe PJ and Yang JL: Effect of
type 1 insulin-like growth factor receptor targeted therapy on
chemotherapy in human cancer and the mechanisms involved. J Cancer
Res Clin Oncol. 136:639–650. 2010. View Article : Google Scholar : PubMed/NCBI
|