Introduction
Malignant gliomas are the most common and highly
aggressive primary brain tumors. Even using multiple modalities of
treatment, including maximal safe resection, radiotherapy, and
chemotherapy, the prognosis of patients with malignant gliomas
remains dismal (1,2). Therefore, novel therapeutic
strategies and drugs for treatment of malignant gliomas are
urgently required.
A large number of chemopreventive and
chemotherapeutic agents have been discovered from natural products
and provided promising approaches to treat and prevent cancer
(3,4). Res
(3,5,4′-trihydroxy-trans-stilbene), a naturally polyphenolic
compound, has been identified in the skin of red grapes, peanuts
and various food products. It has a variety of important biological
effects such as suppression of platelet aggregation,
anti-inflammatory, anti-oxidant, and vasorelaxant activities
(5–7). Res also exhibits antitumor properties
by blocking the three stages of carcinogenesis, initiation,
promotion and progression (8). Res
has exerted anticancer effects against prostate, breast, leukemia
and other epithelial cancer cells. There are some reports on the
studies of the treatment of gliomas with Res (9–12)
but the molecular mechanism of its antitumorigenic or
chemopreventive activities is complex and not fully understood.
The objective of the present study was to
investigate the anti-proliferative effect of Res on glioma cells
using both in vitro and in vivo models. It is well
known that small non-coding regulatory RNAs - microRNAs (miRs) are
found to be dysregulated in almost all types of cancers and play
important roles in cancer development and progression (13–17).
Recent studies have demonstrated that miRs can be regulated by
natural agents such as curcumin and Res resulting in suppression of
tumor growth, drug resistance and metastasis (18–21).
Our previous studies have confirmed that specific miRs, including
miR-21, miR19 and miR-30a-5p are upregulated in GBMs with
significant oncogenic activity and their major targeted genes
involved in gliomagenesis (22–24).
Antisense oligonucleotides of these miRs significantly suppress the
glioma cell growth either in vitro or in vivo
(25,26). We examined the alteration of these
specific miRs in GBM cells after treatment with Res, and also the
expression of their targeted genes and the genes related to glioma
formation and progression.
Materials and methods
Cell culture
Rat C6 glioma cells were obtained from the Institute
of Biochemistry and Cell Biology, Chinese Academy of Sciences.
Cells were cultured at 37°C with 5% CO2 in Dulbecco’s
modified Eagle’s medium (Invitrogen, USA) supplemented with 10%
heat-inactivated fetal calf serum, 4 mM glutamine, 50 U/ml
penicillin and 50 μg/ml streptomycin (Invitrogen). Cells were
subcultured every other day.
Cell proliferation examined by MTT
(3-(4.5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide)
assay
MTT were purchased from Sigma-Aldrich (St. Louis,
MO, USA). Cells were implanted in 96-well plates (4,000 cells/well)
and allowed to grow overnight, then treated with 50, 100 and 150 μM
of Res or 0.3% DMSO for 24, 48, 72 and 96 h, and exposed to 20 μl
MTT (5 mg/ml) at each time-point, then incubated for additional 4
h. The resulting formazan crystals were solubilized with DMSO and
optical density was measured in triplicate wells at the wavelength
of 570 nm. Results were expressed as percentage of control.
Cell cycle kinetic analysis
C6 glioma cells (6×105 cells/well) were
plated in 10-cm dishes and incubated for 24 h. DMSO (0.3%) or
different concentration of Res (50, 100 and 150 μM) were added to
the dishes and incubated for additional 48 h. Cells were
trypsinized, washed with PBS, fixed with 70% ethanol for a minimum
of 30 min, and incubated with RNase A (100 μg/ml) for 30 min at
37°C. Cells were then stained with propidium iodide in PBS (50
μg/ml) for 30 min at room temperature. Samples were analyzed by
FACSCalibur flow cytometer and Modifit software (Becton-Dickinson,
USA).
Detection of apoptotic cell death
The extent of apoptotic cell death was analyzed by
using Annexin V-FITC Apoptosis Detection kit (BD Biosciences, USA).
Cells (6×105 cells/dish) were cultured in 10-cm dishes
for 24 h, treated with 0.3% DMSO and 50–150 μM of Res for 48 h.
Then both adherent and floating cells were harvested,
double-labeled with Annexin V-PE and 7-amino-actinomycin, examined
with FACSCalibur flow cytometer and analyzed by CellQuest software
(Becton-Dickinson).
For xenograft tumor specimens, apoptosis was
detected using TUNEL method with an in situ cell death kit
(Roche, USA) according to the supplier’s instructions. Stained
cells were visualized using FluoView Confocal Laser Scanning
Microscopes-FV1000 (Olympus, Tokyo, Japan) and analyzed using
IPP5.1 (Olympus).
qRT-PCR
Total RNA from cells treated with 0.3% DMSO and
different concentration of Res (50, 100 and 150 μM) for 48 h were
extracted using TRIzol reagent (Invitrogen). For analysis of miR
expression, real-time RT-PCR analyses were carried out using
Hairpin-it™ miRNAs qPCR Quantitation kit (Shanghai GenePharma Co.,
Ltd., China) according to the manufacturer’s instructions.
Amplification reaction was performed with MJ-real-time PCR
(Bio-Rad, Hercules, CA, USA). Relative expression was calculated
using the ΔΔCT method and normalized to the expression of U6
endogenous control RNA and analyzed initially using Opticon Monitor
Analysis V2.02 software (MJ Research, Waltham, MA, USA). All
qRT-PCRs were performed in triplicate, and the data are presented
as means ± standard errors of the means (SEM).
Western blot analysis
Total proteins were extracted from cells treated
with 0.3% DMSO or 50, 100 and 150 μM of Res for 48 h. The protein
concentration was determined by Lowry method. Protein (40 μg) was
subjected to SDS-PAGE. Separated proteins were transferred to PVDF
membranes (Millipore, USA), followed by incubation for 1 h in
blocking buffer. The membranes were then incubated separately with
primary antibodies against EGFR, PI3K, p-AKT, mTOR, PTEN, VEGF,
PCNA, Cyclin A, cyclin E, Bcl-2, Caspase-3, GFAP, NF-κB, Stat3,
COX-2 (1:500 dilution, Santa Cruz Biotech, USA), followed by
incubation with HRP-conjugated secondary antibodies (1:500
dilution, Zymed, USA). The specific proteins were detected using a
SuperSignal protein detection kit (Pierce, USA). After washing with
stripping buffer, the membranes were reprobed with an antibody
against β-actin (1:500 dilution, Santa Cruz) following the
procedures described above. The relative quantification was
expressed as the density of specific proteins divided by the
density of β-actin.
Immunohistochemical staining
After Res treatment, cells were immunostained for
MMP-9 or NF-κB by ABC-peroxidase method. Briefly, cells grown on
the coverslips for 24 h were treated with DMSO (0.3%) or Res (100
μM) for 48 h, then fixed with 4% paraformaldehyde, treated with 3%
hydrogen peroxide and incubated with appropriate antibodies
(1:1,000 dilution, Santa Cruz) overnight at 4°C. After incubation,
biotinylated secondary antibody (1:1,000 dilution, Zymed, USA) was
added and incubated for 1 h at room temperature, followed by
incubation with ABC-peroxidase for additional 1 h. Then washing
with Tris-buffer, the cells were incubated with DAB (3,3′
diaminbenzidine, 30 mg dissolved in 100 ml Tris-buffer containing
0.03% hydrogen peroxide) for 5 min, rinsed in water and
counterstained with hematoxylin.
Tumor growth in vivo
Rat models bearing intracranial C6 glioma were
established as described previously (25). All animal experiments were approved
by the Animal Care and Use Committee of Tianjin Medical University
(Tianjin, China). Rats were randomly divided into two groups:
control (10 rats) and treated (8 rats). In treated group, rats were
given Res 8 mg/kg/day by oral administration until the end of
observation period. The general behavior and survival of the rats
were observed. The tumor growth in each group of rats were
monitored at regular intervals using high-resolution MRI in order
to compare the tumor volume at different periods in each individual
animal. Whenever the rats in the control or Res treated group died
naturally or were sacrificed at various time-points, their brains
were removed and prepared as paraffin-embedded sections for
histopathological examination and immunohistochemical staining. The
expression of EGFR, GFAP, PCNA, MMP-9, NF-κB, COX-2 and VEGF was
detected by immunohistochemistry. The primary antibodies (1:1,000
dilution, Santa Cruz) used and the procedures were the same as
described above.
Statistical analysis
The results are expressed as mean ± SD. A Student’s
t-test was used to compare individual data with control value. A
probability of p<0.05 was considered to be significantly
different from control data.
Results
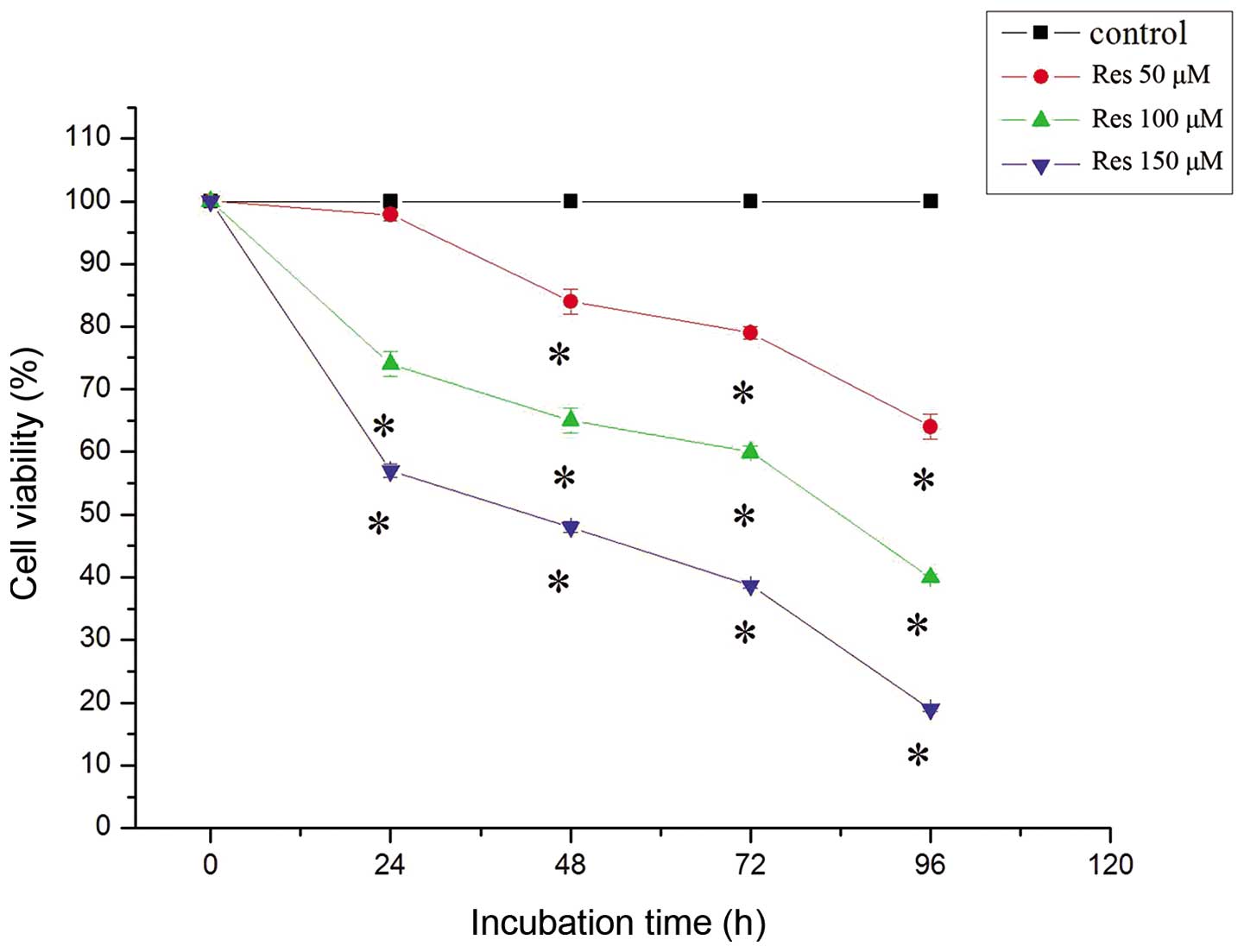
Res inhibits glioma cell growth
The survival rate of C6 glioma cells treated with
Res was measured by MTT assay. As shown in Fig. 1, the suppressive effect of Res on
the proliferation of C6 cells was dose- and time-dependent. After
treatment with 150 μM Res for 24 and 96 h, the survival rate of C6
glioma cells declined to 58±1.26 and 19±1.52%, respectively, as
compared to the control.
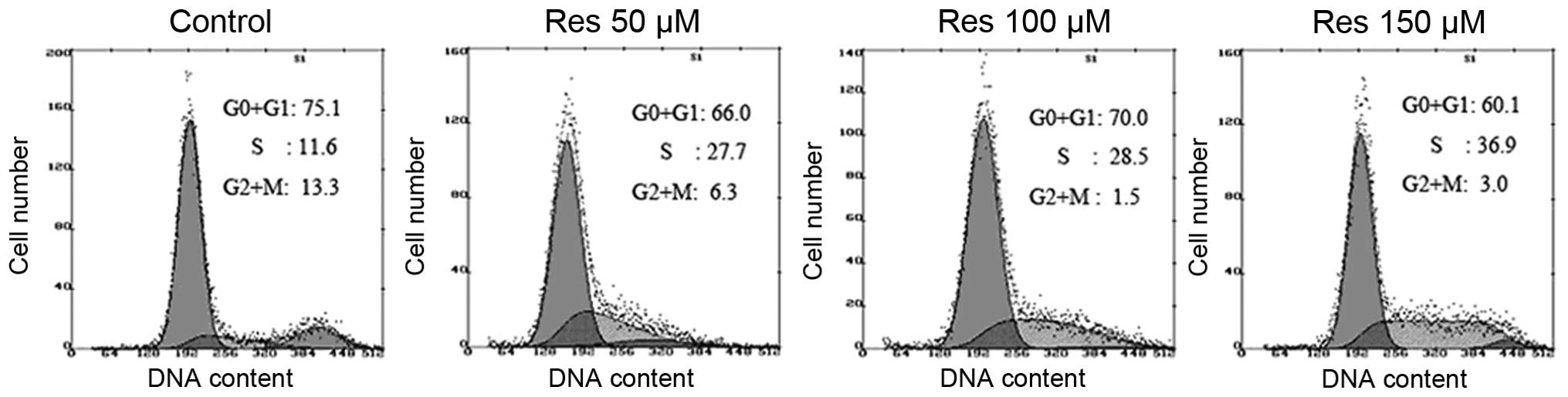
Res arrests the cell cycle
The effect of Res on cell cycle kinetics was
analyzed by flow cytometry. The data obtained from cells treated
with 0.3% DMSO and 50, 100 and 150 μM of Res for 48 h are
summarized in Fig. 2. Two-to-three
fold increase of S phase fraction was demonstrated in C6 cells
treated with different concentration of Res as compared to the
control. These results suggested that Res was able to delay the
cell cycle progression and inhibited the cell proliferation by
arresting the cells cycle at S phase.
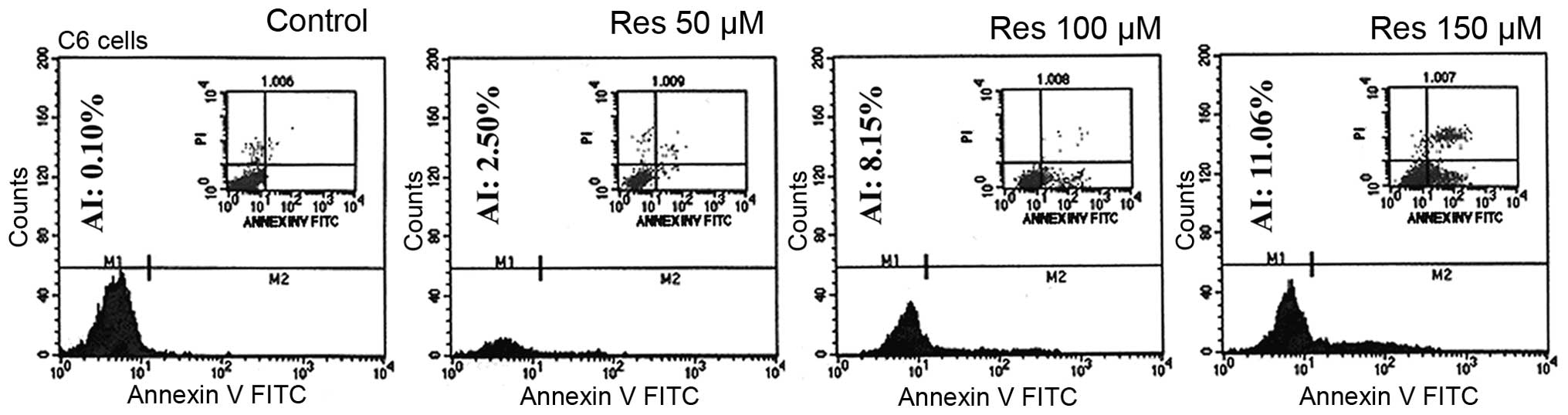
Res induces apoptotic cell death
The effect of Res on the apoptosis of glioma cells
was determined by Annexin V-PE immunofluorescence staining.
Apoptosis in glioma cells treated with Res was induced and the
population of apoptotic cells was significantly increased in a
dose-dependent manner. The percentage of apoptotic cells increased
from 2.50 to 11.06% in C6 cells treated with 50–150 μM of Res
(Fig. 3).
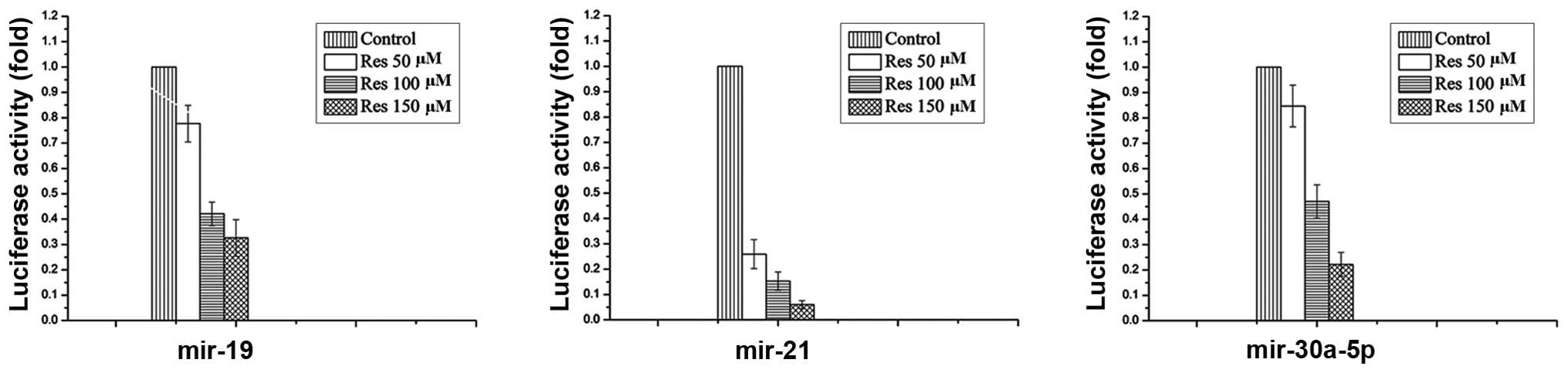
Res downregulates the expression of
miR-21, miR-30a-5p, miR-19 and their targeted or related
components
qRT-PCR and the comparative Ct (ΔΔCt) method were
used to determine the change of miR-21, miR-19 and miR30a-5p
expression in C6 glioma cells before and after treatment with
different concentration of Res. It was found that the expression of
all these miRs was significantly downregulated in Res treated cells
and also in a dose-dependent manner (Fig. 4).
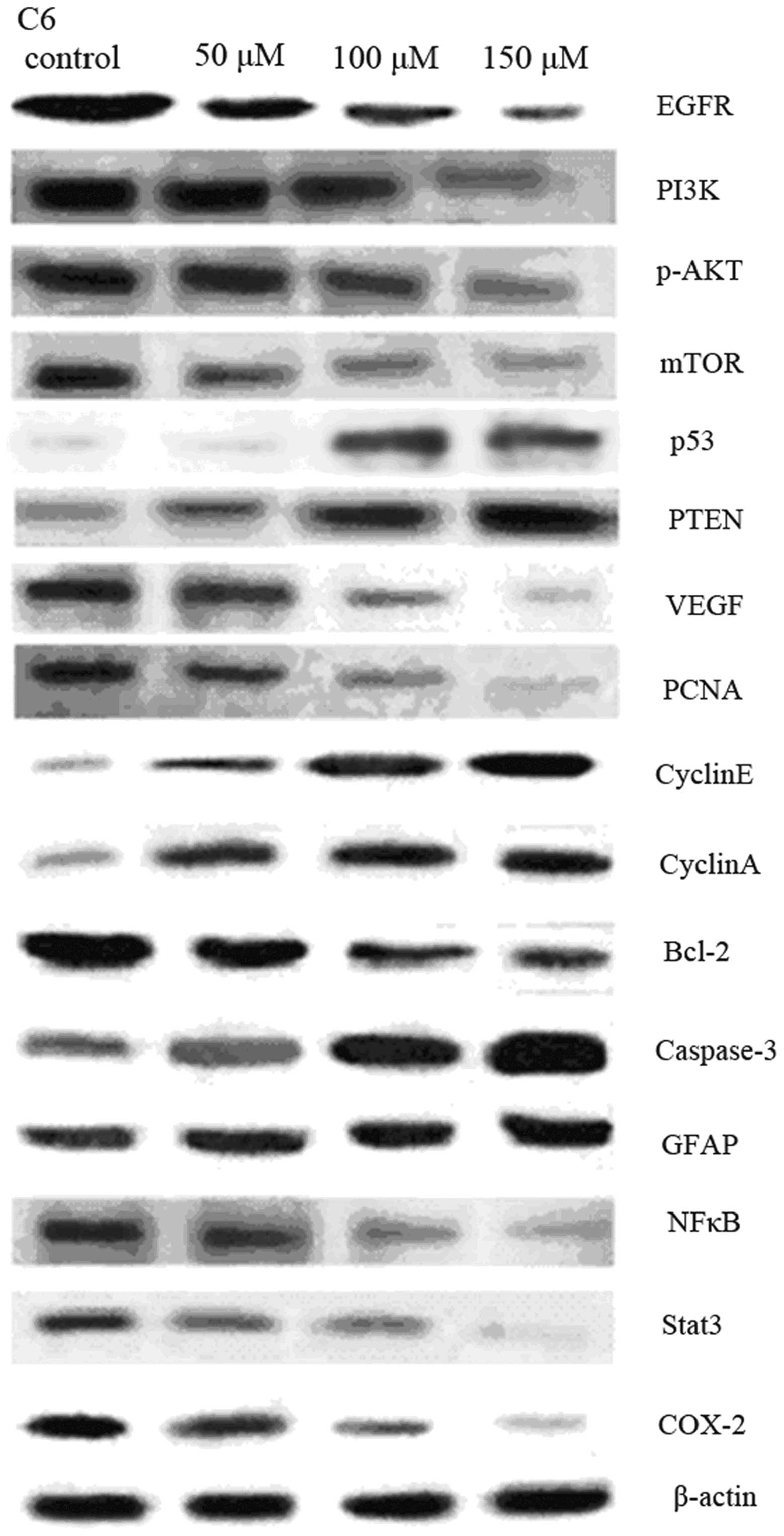
Western blot analysis demonstrated that the
expression of EGFR, PI3K, p-AKT, mTOR, VEGF, PCNA, Bcl-2, NF-κB,
STAT3, COX-2 was downregulated, whereas the expression of p53,
PTEN, Cyclin A, cyclin E, GFAP and Caspase-3 was upregulated in
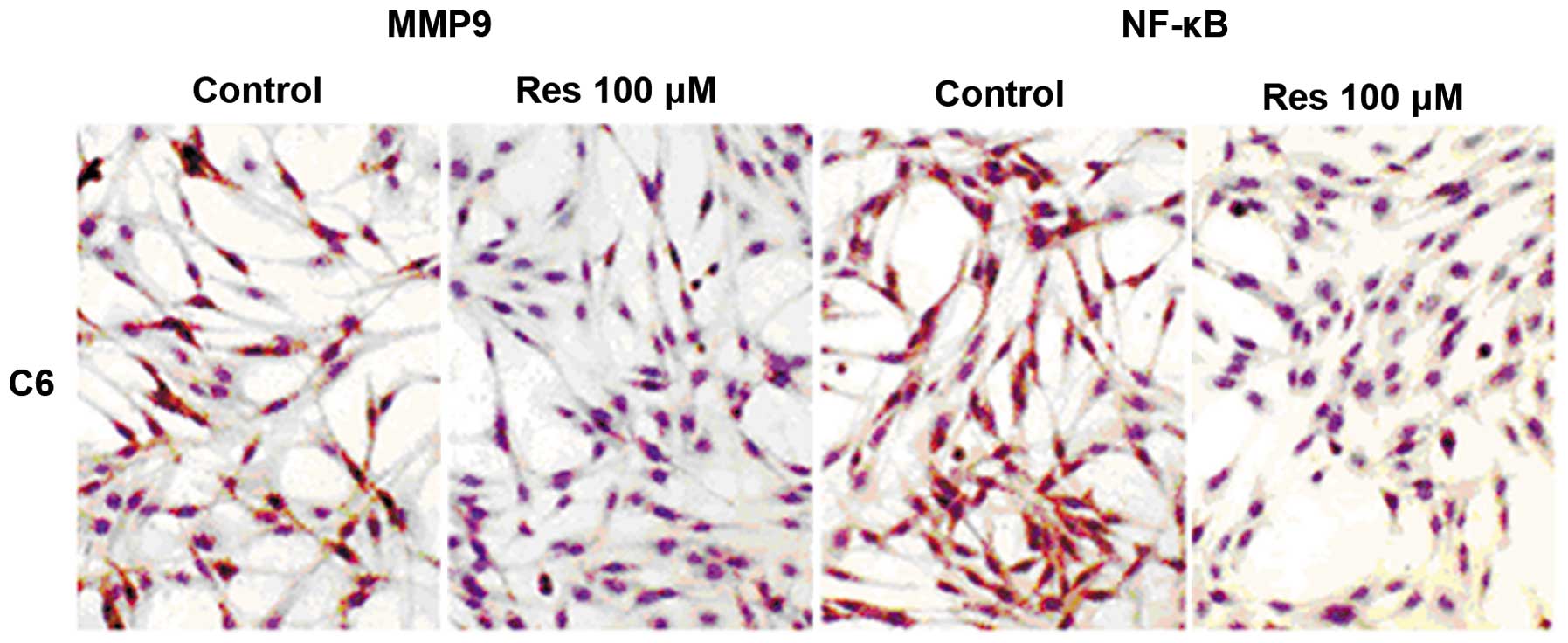
glioma cells treated with 50, 100 and 150 μM of Res (Fig. 5). In addition, MMP-9 and NF-κB
expression were decreased as shown by immunohistochemical staining
(Fig. 6).
Antitumor effect of Res in rat C6 glioma
xenograft
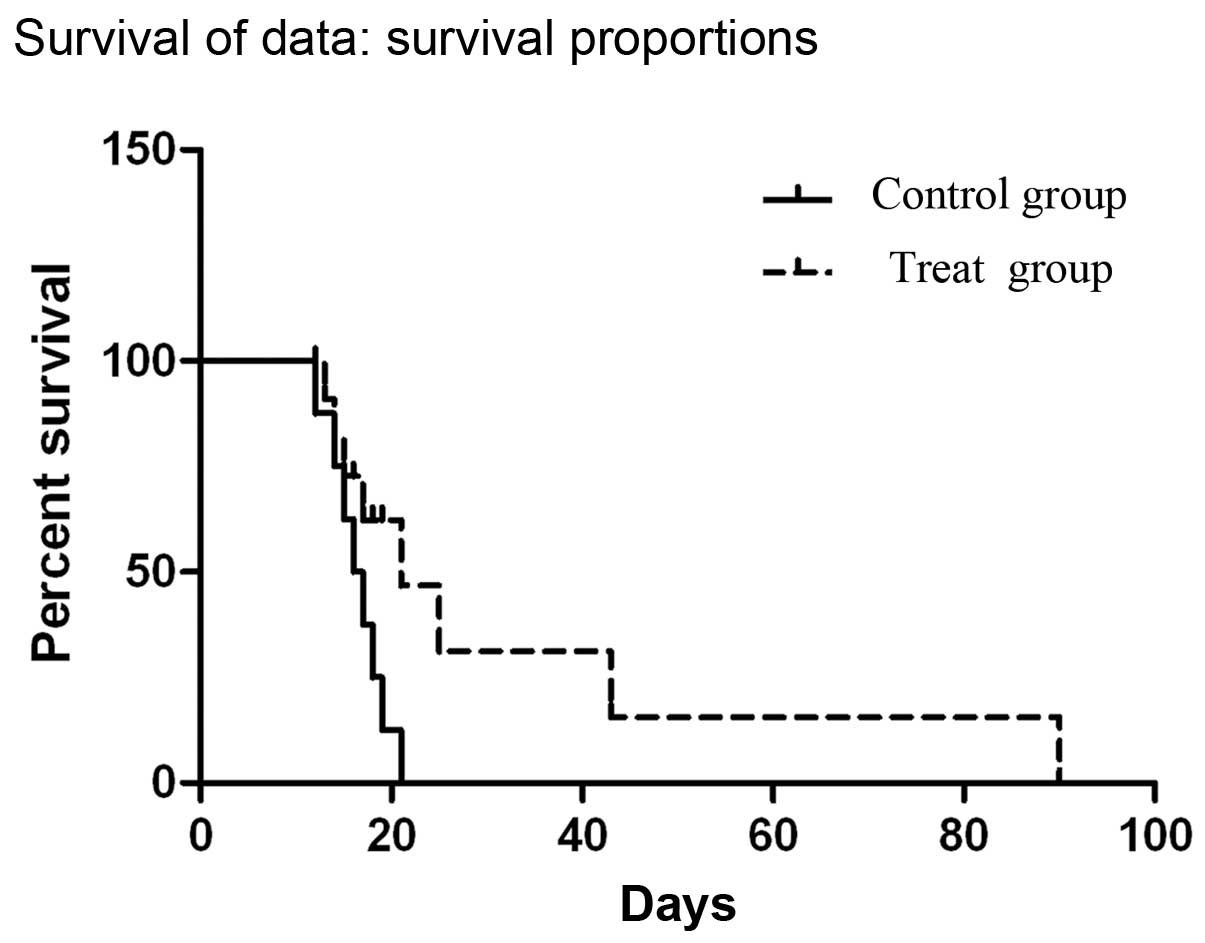
In vivo experiment showed that the general
condition of rats was deteriorated at one week after injection of
parental C6 glioma cells. The mean survival time of rats in control
group was 15.80±0.93 days (10 rats died on days 12, 13, 13, 14, 15,
16, 17, 18, 19 and 21, respectively). Eight rats receiving Res
treatment had a longer survival than the control rats, with a mean
survival of 29.75±9.27 days (8 rats died on days 13, 14, 15, 17,
21, 25, 43 and 90, respectively). There was a statistical
difference between the mean survival time of the treatment group
and the control group (p<0.05) (Fig. 7).
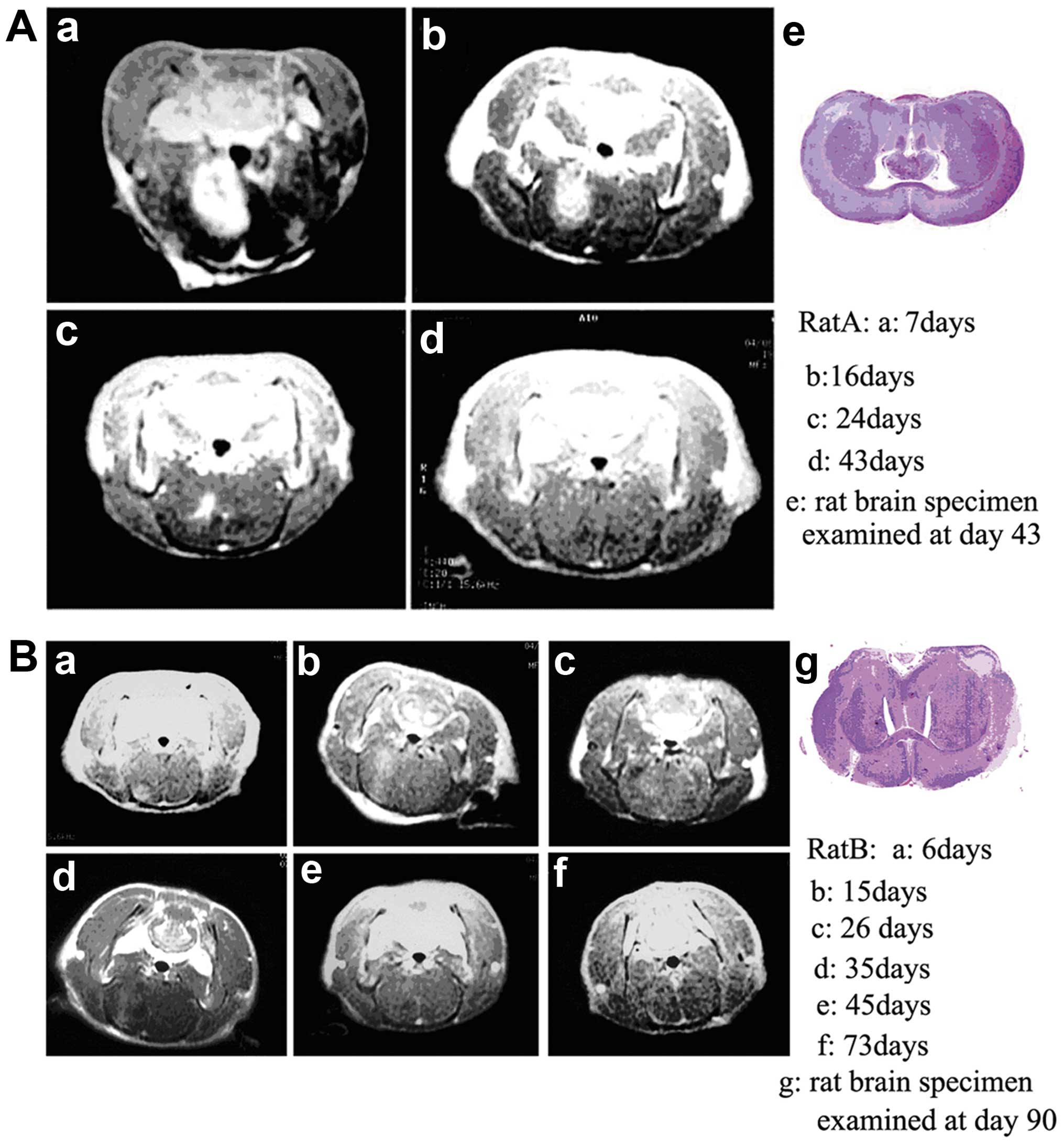
Two out of eight rats treated with Res had more
favorable response. Their general conditions returned to normal at
~4 weeks after treatment. In one of them the primary tumor focus
disappeared on day 73 and in another longer survival rat the tumor
disappeared on day 35 as shown by MRI (the rats were sacrificed on
days 90 and 43, respectively). Histopathological examination of
their brains showed no macroscopic gliomas but only a tiny amounts
of tumor cell residue in the original tumor region (Fig. 8). These results indicated that Res
could inhibit tumor growth and prolong the survival in a fraction
of the rats bearing intracranial C6 gliomas.
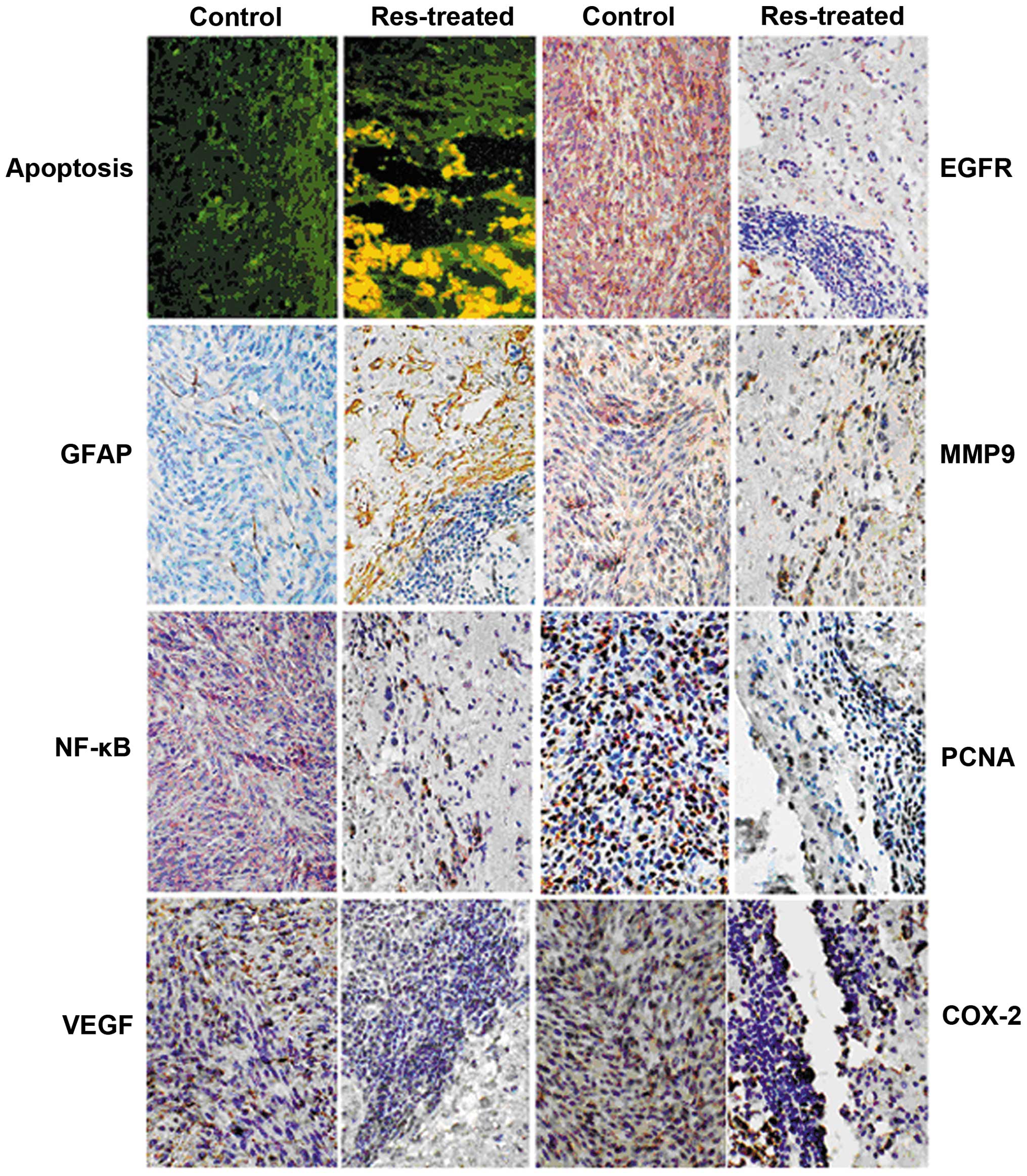
Besides, there were no apoptotic cells found in the
tumor specimens of control rats. However, increased number of
apoptotic cells could be observed in the tumors of Res-treated rats
as detected by TUNEL method (Fig.
9).
The immunohistochemical staining of rat brain tumors
treated with Res also demonstrated that the expression of EGFR,
MMP-9 NF-κB, PCNA, COX-2 and VEGF was decreased, whereas the
expression of GFAP was increased as compared with those in control
rats (Fig. 9). These results
suggested that multiple signaling molecules might be modulated by
Res.
Discussion
The present study investigated anti-glioma effect of
Res and its underlying molecular mechanism. Our results demonstrate
that Res exerts a significant inhibitory effect on C6 glioma cell
growth in a dose- and time-dependent manner. This
anti-proliferative effect appears to result from the cell cycle
arrest at S-phase and induction of apoptotic cell death. These
results are in accordance with the findings from previously
reported studies (27–29). Limited information is available on
the anti-glioma effect of Res in vivo. In this study, we
found that the survival of a fraction of rats bearing intracranial
gliomas treated with Res is prolonged. Among them, two rats had
more favorable response to Res. These results indicate that Res is
able to inhibit glioma growth both in vitro and in
vivo, which is similar to the study of the suppressive effect
of Res on the tumor growth of RT-2 glioma in vitro and in
vivo (11). In addition, Gao
et al reported that Res has strong anti-proliferative and
pro-apoptotic effects against 32Dp210 leukemic cells in
vitro, but Res at doses of 8 mg/kg/day and 40 mg/kg/day (oral
administration for 5 days/week throughout the experiment) is
ineffective in slowing down the progression of leukemia, as the
doses escalated to 80 mg/kg/day, Res can prevent a small fraction
of mice from leukemia-induced death (7). Based on our previous experiments on
the rat C6 glioma models, the intracranial C6 gliomas shown by MRI
could not regress spontaneously without appropriate treatment. It
is conceivable to suggest that Res could inhibit tumor growth in a
fraction of rat C6 gliomas. However, the dose of Res we used for
the treatment of xenografts in vivo is low, whether the
escalated doses of Res will enhance the suppression of glioma
growth should be further studied.
Res has been shown to have antioxidant,
anti-inflammatory, cell cycle arrest and cell death-inducing
properties. Therefore, it is predictable that multiple targets and
complex molecular events may be involved in Res-mediated inhibiton
of glioma cell growth.
Alterations of a number of miRNAs play an oncogenic
or tumor suppressor role in glioma formation and progression. Our
previous studies have shown that miR-21, miR-19 and miR-30a-5p are
overexpressed in gliomas and glioma cell lines, and involved in
gliomagenesis as oncogenic miRs through their targeting genes.
Antisense oligonucleotides of these miRs significantly suppress the
glioma cell growth either in vitro or in vivo. The
first study of Res affecting endogenous miRNAs was conducted in a
human colon cancer cell line, Res was identified to decrease the
levels of oncogenic miRNAs, including miR-17, -21, -23a/b, -146a
and -103-1/2, and increase the level of tumor suppressor miR-663
(30). We also found that Res is
able to inhibit the expression of oncogenic miR-21, miR-19 and
miR-30a-5p in glioma cells.
MiR-21 overexpression in gliomas and glioma cell
lines is a general consensus. It has been identified as a
predominant microRNA altered in gliomas and play a crucial role in
a myriad of biological processes. To our current knowledge, miR-21
targets sets of genes associated with tumor suppressive and
pro-apoptotic function, such as PTEN, p53, TGF-β, PDCD4, TPM1 and
Reck (31–34). MiR-21 expression is also correlated
with downstream factors such as Bcl-2 and MMP2, which may represent
indirect targets affected by the expression levels of miR-21, since
there is no evidence of direct interaction between miR-21 and 3′UTR
of their mRNA.
The expression and function of miR-19 and miR-30a-5p
in gliomas have not been reported before. However, PTEN, the target
of miR-19 we identified in glioma cells (23), and also reported as the target of
miR19a/b in gastric cancer (35).
Besides, miR-30 family members have been found to regulate cell
apoptosis through the p53-Drp1 (dynamin-related protein 1)
pathway.
PTEN is the common target of miR-21 and miR-19 and
the critical tumor suppressor in GBM (24). As shown in Fig. 6, Res suppresses miR-21 and miR-19
may induce PTEN activation and results in repression of the major
oncogenic PI3K/AKT/mTOR signaling pathway in glioma cells, leading
towards inhibition of tumor cell proliferation, invasion and
induction of apoptosis. p53 is also the major target of miR-21, and
tumor suppressor p53 deletion and mutation is also an important
molecular event in gliomas. As shown in the present study, we
demonstrated that p53 expression is significantly upregulated after
treatment with Res. So that Res suppresses glioma cell growth, at
least in part, through downregulation miR-21 and miR-19 and
consequently, upregulating PTEN and p53.
EGFR overexpression is one of the most important
genetic aberrations in malignant gliomas. A number of studies have
demonstrated that EGFR is overexpressed in 60–90% of glioblastomas
(36), as well as in C6 glioma
cells (25). Hyperactivated EGFR
signaling promotes cell growth and inhibits apoptosis via its major
downstream and crosstalk signaling pathways, such as PI3K/AKT/mTOR
and RAS/MAPK, which can regulate cell survival, proliferation,
invasion and angiogenesis (37–39).
Our previous studies have demonstrated that antisense and
dominant-negative EGFR cDNA or siRNA targeting EGFR effectively
inhibit glioma cell growth (25,40).
It has also been found that there is a significant correlation
between EGFR and miR-21 levels in glioma cells. Antisense miR-21
markedly reduced EGFR expression in our previous study (24). Thus, the overexpression of EGFR is
decreased in C6 glioma cells after treatment with Res, may be
partially through the inhibition of miR-21.
STAT3 overactivation is associated with a variety of
cancers, including gliomas, and is identified as a master regulator
of biological processes leading to the cancer formation (41–43).
The present study demonstrates that Res significantly represses the
expression of STAT3. This is one of the major reversions of
aberrant molecular events in glioma cells following treatment with
Res. It has been reported that Res suppresses constitutively active
STAT3 in all the NK cell lines by inhibiting JAK2 phosphorylation
but not other upstream mediators of STAT3 activation.
We have found previously that STAT3 binds to the
miR-21 promoter by CHIP analysis and the STAT3 inhibitor
significantly reduced miR-21 expression (44). Thus, STAT3 activation suppressed by
Res may contribute to the reduced expression of miR-21.
The anti-inflammatory property of Res results from
its inhibitory effect on the synthesis of pro-inflammatory mediator
COX-2, and downregulation of COX-2 is also mediated by the
inhibitory effects of Res on the important nuclear transcription
factor NF-κB. COX-2 overexpression has been documented in malignant
gliomas and strongly correlated with the poor prognosis of
glioblastomas (45). COX-2
catalyzes the conversion of free arachidonic acid to
prostaglandins. Prostaglandins can stimulate tumor cell
proliferation, promote angiogenesis, and suppress apoptosis. Res
significantly inhibits the expression of COX2 and NF-κB either
in vitro or in vivo. The suppressive effect of Res on
COX-2 is also suggested as one of the molecular mechanisms
responsible for its antiglioma activity. Additionally, NF-κB is
constitutively activated in a number of cancers, including gliomas,
Res can downregulate the expression of NF-κB-regulated genes,
including Bcl-2, VEGF, and MMP-9, through which it may also affect
apoptosis and invasion of glioma cells. Banerjee et al
showed that Res inhibits the activation of NF-κB in breast cancer
cells and dietary administration of Res in rats bearing mammary
tumors inhibits tumor growth and decreases the expression of NF-κB,
COX-2 and MMP-9 in tumor tissues (46). It is consistent with our findings
that the expression of NF-κB, COX-2, MMP-9 and VEGF is decreased in
glioma cells after Res treatment.
Recently, it has been reported that Res can
downregulate lncRNA (long non-coding RNA) MALAT-1 that results in
the inhibition of Wnt/β-catenin signaling pathway and suppression
of human colorectal cancer cell invasion and metastasis (47), whereas Wnt/β-catenin signaling
pathway is also an important oncogenic pathway to promote glioma
cell growth as we reported before (48). Thus the effect of Res on non-coding
RNAs should be studied in more detail.
In addition, the preclinical studies have shown that
Res enhances the antiglioma effect of temozolomide (TMZ) via
ROS-dependent AMPK-TSC-mTOR signaling pathway and inhibiting
autophagy, or Res reverses TMZ resistance via downregulation of
MGMT. It has also been reported that Res sensitizes GBM tumor
initiating cells to radiotherapy by inhibition of STAT3 axis
(49–52).
Taken together, the results from the present study
demonstrate that Res could inhibit cellular proliferation and
potentiate apoptosis in rat C6 glioma cells both in vitro
and in vivo. Based on the interfering with the expression of
several oncogenic miRs and a panel of genes of signaling pathways
involved in the processes of gliomagenesis and possibly sensitizing
chemo- and radiotherapy for gliomas after treatment with Res, it is
conceivable that Res could be classified as a multi-targeting
agent.
Since Res is able to cross the blood-brain barrier
and incorporate into brain tissue, and its toxicity is minimal even
high doses used long-term in rats and mice (3,53).
Therefore, clinical trials in human gliomas are urgently required.
Currently, analogs of Res with improved bioviability are being
developed as potential anticancer agents, and clinical trials of
Res against some other cancers are being conducted (54,55).
It is expected that Res will provide at least an adjuvant for
currently available combined therapy against malignant gliomas.
Acknowledgements
This study was supported by the Natural Science
Foundation of China (no. 81101915).
References
|
1
|
Kevin P, Becker, Yu J, et al: Status quo -
standard-of-care medical and radiation therapy for glioblastoma.
Cancer J. 18:12–19. 2012. View Article : Google Scholar
|
|
2
|
Yang P, Wang Y, Peng X, et al: Management
and survival rates in patients with glioma in China (2004–2010): a
retrospective study from a single-institution. J Neurooncol.
2:259–266. 2013. View Article : Google Scholar
|
|
3
|
Aggarwal BB, Bhardwaj A, Aggarwal RS, et
al: Role of resveratrol in prevention and therapy of cancer:
preclinical and clinical studies. Anticancer Res. 24:2783–2840.
2004.PubMed/NCBI
|
|
4
|
Cao Y, Wang F, Liu HY, et al: Resveratrol
induces apoptosis and differentiation in acute promyelocytic
leukemia (NB4) cells. J Asian Nat Prod Res. 7:633–641. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
De la Lastra CA and Villegas I:
Resveratrol as an anti-inflammatory and anti-aging agent:
mechanisms and clinical implications. Mol Nutr Food Res.
49:405–430. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Castello L and Tessitore L: Resveratrol
inhibits cell cycle progression in U937 cells. Oncol Rep.
13:133–137. 2005.
|
|
7
|
Gao X, Xu YX, Divine G, et al: Disparate
in vitro and in vivo antileukemic effects of resveratrol, a natural
polyphenolic compound found in grapes. J Nutr. 132:2076–2081.
2002.PubMed/NCBI
|
|
8
|
Jang M, Cai L, Udeani GO, et al: Cancer
chemopreventive activity of resveratrol, a natural product derived
from grapes. Science. 275:218–220. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gagliano N, Moscheni C, Torri C, et al:
Effect of resveratrol on matrix metalloproteinase-2 (MMP-2) and
Secreted Protein Acidic and Rich in Cysteine (SPARC) on human
cultured glioblastoma cells. Biomed Pharmacother. 59:359–364. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jiang H, Zhang L, Kuo J, et al:
Resveratrol-induced apoptotic death in human U251 glioma cells. Mol
Cancer Ther. 4:554–561. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tseng SH, Lin SM, Chen JC, et al:
Resveratrol suppresses the angiogenesis and tumor growth of gliomas
in rats. Clin Cancer Res. 10:2190–2202. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yang YP, Chang YL, Huang PI, et al:
Resveratrol suppresses tumorigenicity and enhances radiosensitivity
in primary glioblastoma tumor initiating cells by inhibiting the
STAT3 axis. J Cell Physiol. 227:976–993. 2012. View Article : Google Scholar
|
|
13
|
Zhang W, Wang YE, Zhang Y, et al: Global
epigenetic regulation of microRNAs in multiple myeloma. PLoS One.
9:e1109732014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chan JA, Krichevsky AM and Kosik KS:
MicroRNA-21 Is an Antiapoptotic Factor in Human Glioblastoma Cells.
Cancer Res. 65:6029–6033. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tuomarila M, Luostari K, Soini Y, et al:
Overexpression of microRNA-200c predicts poor outcome in patients
with PR-negative breast cancer. PLoS One. 9:e1095082014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang KY, Ma J, Zhang FX, et al:
MicroRNA-378 inhibits cell growth and enhances l-OHP-induced
apoptosis in human colorectal cancer. IUBMB Life. 66:645–654. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen Q, Yang L, Xiao Y, et al: Circulating
microRNA-182 in plasma and its potential diagnostic and prognostic
value for pancreatic cancer. Med Oncol. 31:2252014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li Yiwei, Kong Dejuan, Wang Zhiwei, et al:
Regulation of microRNAs by natural agents: an emerging field in
chemoprevention and chemotherapy research. Pharm Res. 27:1027–1041.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Melkamu T, Zhang X, Tan J, et al:
Alteration of microRNA expression in vinyl-carbamateinduced mouse
lung tumors and modulation by the chemopreventive agent
indole-3-carbinol. Carcinogenesis. 31:252–258. 2010. View Article : Google Scholar
|
|
20
|
Li Y, VandenBoom TG, Kong D, et al:
Up-regulation of miR-200 and let-7 by natural agents leads to the
reversal of epithelial-to-mesenchymal transition in
gemcitabine-resistant pancreatic cancer cells. Cancer Res.
69:6704–6712. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sun M, Estrov Z, Ji Y, et al: Curcumin
(diferuloylmethane) alters the expression profiles of microRNAs in
human pancreatic cancer cells. Mol Cancer Ther. 7:464–473. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jia Z, Wang K, Wang G, et al: MiR-30a-5p
antisense oligonucleotide suppresses glioma cell growth by
targeting SEPT7. PLoS One. 8:e550082013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jia Z, Wang K, Zhang A, et al: MiR-19a and
miR-19b overexpression in gliomas. Pathol Oncol Res. 19:847–853.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhou X, Ren Y, Moore L, et al:
Downregulation of miR-21 inhibits EGFR pathway and suppresses the
growth of human glioblastoma cells independent of PTEN status. Lab
Invest. 90:144–155. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Pu P, Liu X, Liu A, et al: Inhibitory
effect of antisense epidermal growth factor receptor RNA on the
proliferation of rat C6 glioma cells in vitro and in vivo. J
Neurosurg. 92:132–139. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Pu P, Kang C, Li J, et al: The effects of
antisense AKT2 RNA on the inhibition of malignant glioma cell
growth in vitro and in vivo. J Neurooncol. 76:1–11. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bove K, Lincoln DW and Tsan MF: Effect of
resveratrol on growth of 4T1 breast cancer cells in vitro and in
vivo. Biochem Biophys Res Commun. 291:1001–1005. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kuwajerwala N, Cifuentes E, Gautam S, et
al: Resveratrol induces prostate cancer cell entry into S phase and
inhibits DNA synthesis. Cancer Res. 62:2488–2492. 2002.PubMed/NCBI
|
|
29
|
Kim YA, Lim SY, Rhee SH, et al:
Resveratrol inhibits inducible nitric oxide synthase and
cyclooxygenase-2 expression in beta-amyloid-treated C6 glioma
cells. Int J Mol Med. 17:1069–1075. 2006.PubMed/NCBI
|
|
30
|
Tili E, Michaille JJ, Alder H, et al:
Resveratrol modulates the levels of microRNAs targeting genes
encoding tumor-suppressors and effectors of TGFβ signaling pathway
in SW480 cells. Biochem Pharmacol. 80:2057–2065. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sheth S, Jajoo S, Kaur T, et al:
Resveratrol reduces prostate cancer growth and metastasis by
inhibiting the Akt/MicroRNA-21 pathway. PLoS One. 7:e516552012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Krichevsky AM and Gabriely G: miR-21: a
small multi-faceted RNA. J Cell Mol Med. 13:39–53. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Papagiannakopoulos T, Shapiro A and Kosik
KS: MicroRNA-21 targets a network of key tumor-suppressive pathways
in glioblastoma cells. Cancer Res. 68:8164–8172. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhu S, Wu H, Wu F, et al: MicroRNA-21
targets tumor suppressor genes in invasion and metastasis. Cell
Res. 18:350–359. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang F, Li T, Zhang B, et al:
MicroRNA-19a/b regulates multidrug resistance in human gastric
cancer cells by targeting PTEN. Biochem Biophys Res Commun.
434:688–694. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zawrocki A and Biernat W: Epidermal growth
factor receptor in glioblastoma. Folia Neuropathol. 43:123–132.
2005.PubMed/NCBI
|
|
37
|
Pu P, Kang C, Zhang Z, et al:
Downregulation of PIK3CB by siRNA suppresses malignant glioma cell
growth in vitro and in vivo. Technol Cancer Res Treat. 5:271–280.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Gürsel DB, Connell-Albert YS, Tuskan RG,
et al: Control of proliferation in astrocytoma cells by the
receptor tyrosine kinase/PI3K/AKT signaling axis and the use of
PI-103 and TCN as potential anti-astrocytoma therapies. Neuro
Oncol. 13:610–621. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ruano Y, Mollejo M, Camacho FI, et al:
Identification of survival-related genes of the
phosphatidylinositol 3′-kinase signaling pathway in glioblastoma
multiforme. Cancer. 112:1575–1584. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kang CS, Pu PY, Li YH, et al: An in vitro
study on the suppressive effect of glioma cell growth induced by
plasmid-based small interference RNA (siRNA) targeting human
epidermal growth factor receptor. J Neurooncol. 74:267–273. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Brantley EC and Benveniste EN: Signal
transducer and activator of transcription-3: a molecular hub for
signaling pathways in gliomas. Mol Cancer Res. 6:675–684. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Germain D and Frank DA: Targeting the
cytoplasmic and nuclear functions of signal transducers and
activators of transcription 3 for cancer therapy. Clin Cancer Res.
13:5665–5669. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Quoc Trung L, Espinoza JL, Takami A, et
al: Resveratrol induces cell cycle arrest and apoptosis in
malignant NK cells via JAK2/STAT3 pathway inhibition. PLoS One.
8:e551832013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Han L, Yue X, Zhou X, et al: MicroRNA-21
expression is regulated by β-catenin/STAT3 pathway and promotes
glioma cell invasion by direct targeting RECK. CNS Neurosci Ther.
18:573–583. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Sminia P, Stoter TR, van der Valk P, et
al: Expression of cyclooxygenase-2 and epidermal growth factor
receptor in primary and recurrent glioblastoma multiforme. J Cancer
Res Clin Oncol. 131:653–661. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Banerjee S, Bueso-Ramos C and Aggarwal BB:
Suppression of 7,12-dimethylbenz(a)anthracene-induced mammary
carcinogenesis in rats by resveratrol: role of nuclear
factor-kappaB, cyclooxygenase-2, and matrix metalloprotease 9.
Cancer Res. 62:4945–4954. 2002.PubMed/NCBI
|
|
47
|
Ji Q, Liu X, Fu X, et al: Resveratrol
inhibits invasion and metastasis of colorectal cancer cells via
MALAT1 mediated Wnt/β-catenin signal pathway. PLoS One.
8:e787002013. View Article : Google Scholar
|
|
48
|
Pu P, Zhang Z, Kang C, et al:
Downregulation of Wnt2 and beta-catenin by siRNA suppresses
malignant glioma cell growth. Cancer Gene Ther. 16:351–361. 2009.
View Article : Google Scholar
|
|
49
|
Filippi-Chiela EC, Thomé MP, Bueno e Silva
MM, et al: Resveratrol abrogates the temozolomide-induced G2 arrest
leading to mitotic catastrophe and reinforces the
temozolomide-induced senescence in glioma cells. BMC Cancer.
22:1472013. View Article : Google Scholar
|
|
50
|
Nakada M, Furuta T, Hayashi Y, et al: The
strategy for enhancing temozolomide against malignant glioma. Front
Oncol. 2:982012. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Huang H, Lin H, Zhang X, et al:
Resveratrol reverses temozolomide resistance by downregulation of
MGMT in T98G glioblastoma cells by the NF-κB-dependent pathway.
Oncol Rep. 27:2050–2056. 2012.PubMed/NCBI
|
|
52
|
Yuan Y, Xue X, Guo RB, et al: Resveratrol
enhances the antitumor effects of temozolomide in glioblastoma via
ROS-dependent AMPK-TSC-mTOR signaling pathway. CNS Neurosci Ther.
18:536–546. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Gagliano N, Aldini G, Colombo G, et al:
The potential of resveratrol against human gliomas. Anticancer
Drugs. 21:140–150. 2010. View Article : Google Scholar
|
|
54
|
Iguchi K, Toyama T, Ito T, et al:
Antiandrogenic activity of resveratrol analogs in prostate cancer
LNCaP cells. J Androl. 33:1208–1215. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Szekeres T, Saiko P and Fritzer-Szekeres
M: Chemopreventive effects of resveratrol and resveratrol
derivatives. Ann NY Acad Sci. 1215:89–95. 2011. View Article : Google Scholar : PubMed/NCBI
|