Introduction
Despite therapeutic and diagnostic advances, oral
squamous cell carcinoma (OSCC) patients are often diagnosed at
advanced stages, and therefore mortality rates remain high
(1). OSCC carcinogenesis appears
to evolve through a multistep process involving biomolecular
changes, which results in the development of premalignant lesions
and, subsequently, an invasive cancer. The identification of the
molecular alterations associated with these events could yield
insight into the mechanisms underlying the initiation and
progression of this neoplasia, and provide new tools for the
diagnosis, treatment, and prevention of OSCCs. To address this
issue, we recently developed a strategy, using proteomics
technologies to search for significant molecular biomarkers for
oral carcinogenesis (2). Among the
proteins identified was the interleukin-1 receptor antagonist
(IL1RN), whose expression was found to be significantly
downregulated in OSCC-derived cell lines compared to normal human
oral keratinocytes.
It is generally accepted that inflammation is a
strong risk factor for tumor development (3,4). In
oral cancer, chronic inflammation induced by prolonged exposure to
alcohol, tobacco and pathogenic agents, has been considered to
represent a potential common denominator in the development of
tumors (5,6). Interleukin-1 (IL-1) is a major
proinflammatory cytokine, responsible for various acute and chronic
inflammatory conditions. Evidence suggests that IL-1 may contribute
to the promotion of tumor growth, angiogenesis, and metastasis in
various human malignancies (7–9).
IL1RN was initially characterized as a naturally occurring
antagonist for IL-1. It shares 70% sequence homology with IL-1, and
has the ability to bind to the membrane-anchored IL-1 receptor
without initiating the IL-1 intracellular signaling cascade
(10–12). Maintenance of an equilibrium
between the IL-1 and IL1RN levels in local tissues influences the
relative inflammatory effects of IL-1, and as reviewed by Arend in
2002, alteration of the balance predisposes cells to the
development of a variety of diseases, including cancer (13).
Alterations in IL1RN expression could have an
important role in oral carcinogenesis, however, little is known
about the significance of IL1RN in OSCC. Therefore, the aim of the
current study was to investigate the expression levels of IL1RN in
a series of human primary OSCCs and oral premalignant lesions
(OPLs), in order to establish a link between IL1RN expression and
oral malignancies.
Materials and methods
Tissue specimens
Thirty-nine pairs of primary OSCC samples and
corresponding normal oral epithelium tissue samples were obtained
at the time of surgery performed at Chiba University Hospital
between 2005 and 2009. The clinicopathological characteristics of
the OSCC cases in this series are summarized in Table I. In addition, the 18 cases of
advanced OPLs pathologically diagnosed as leukoplakia with
epithelial dysplasia, i.e., mild (n=1), moderate (n=10), and severe
(n=7), in a high-risk oral site such as the ventral-lateral tongue
or gingiva, were obtained in the same manner. The resected tissues
were divided into two parts: one part was frozen immediately after
removal of the surrounding normal tissue, and was stored at −80°C
until extraction of its RNA was performed, and the second part was
fixed in 10% buffered formaldehyde solution in preparation for
pathological diagnosis and immunohistochemical (IHC) staining. The
histopathological diagnosis of each tumor specimen was performed
according to the International Histological Classification of
Tumors, by the Department of Pathology, Chiba University Hospital.
Clinicopathologic staging was determined by the TNM classification
system of the International Union against Cancer.
 | Table IProfiles of OSCC cases. |
Table I
Profiles of OSCC cases.
| Factors | Data |
|---|
| Gender |
| Male | 26 |
| Female | 13 |
| Age |
| Min | 27 years |
| Max | 87 years |
| Average | 64.4 years |
| SD | 13.1 |
| Lesion sites |
| Tongue | 20 |
| Gingiva | 8 |
| Buccal mucosa | 4 |
| Oral floor | 3 |
| Fauces | 2 |
| Lip | 1 |
| Mandibular
bone | 1 |
|
Differentiation |
| Well | 23 |
| Moderately | 15 |
| Poorly | 1 |
| Primary tumor
size |
| T1 | 1 |
| T2 | 11 |
| T3 | 17 |
| T4 | 10 |
| Regional lymph node
metastasis |
| N0 | 18 |
| N1 | 8 |
| N2 | 15 |
| Distant
metastasis |
| No | 27 |
| Unknown | 12 |
| Stage |
| I | 1 |
| II | 7 |
| III | 10 |
| IV | 21 |
| pNa |
| No | 22 |
| Yes | 17 |
Ethics statement
All patients provided written informed consent, in
accordance with the protocol that was reviewed and approved by the
Ethical Committee of Graduate School of Medicine, Chiba University
(approval number, 236). The study was performed in accordance with
the ethical standards of the Declaration of Helsinki.
Preparation of RNA
Total RNA was extracted using the TRIzol reagent
(Invitrogen Life Technologies, Carlsbad, CA, USA), according to the
manufacturer’s instructions. Each extracted RNA or protein sample
was stored separately at −80°C until use.
mRNA expression analysis
Following RNA extraction and conversion of the
extracted RNA to cDNA by reverse transcription, the mRNA expression
levels of IL1RN were examined by real-time quantitative
polymerase chain reaction methods (qRT-PCR) in OSCC specimens, and
were compared with corresponding normal tissues. Before the cDNA
synthesis, residual genomic DNA was removed from the total RNA by
DNase I treatment (DNA-free kit; Ambion, Austin, TX, USA). The
nucleotide sequences of the gene-specific primers for qRT-PCR
amplification of IL1RN were: forward 5′-TCCAAG
CTCCATCTCCACTC-3′, reverse 5′-GCTGAGTACCTGCCAAGAGC-3′. The
glyceraldehyde-3-phosphate dehydrogenase gene (GAPDH; forward
5′-TTGGTATCGTGGA AGGACTGA-3′, reverse 5′-TGTCATGATATTTGGCAG
GTTT-3′) was used as an internal control. The sequences of specific
primers were checked before use by the Primer3 program (available
at http:www-genome.wi.mit.edu/cgi-bin/primer/primer3_www.cgi),
to avoid amplification of genomic DNA or pseudogenes. Amplified
products were analyzed by 3% agarose gel electrophoresis to
ascertain their size and purity.
Real-time qRT-PCR was performed using the
LightCycler FastStart DNA Master SYBR Green I kit (Roche,
Indianapolis, IN, USA). To prepare the standard curve, 3 μg of
total RNA from normal oral tissue was reverse-transcribed with
Superscript RT (Life Technologies) and oligo-d(T)12–18 primer,
after which, serial dilutions were made corresponding to cDNA
transcribed from 300, 30, 3.0 and 0.3 ng of total RNA. PCRs using
the LightCycler apparatus were carried out in a final volume of 20
μl of reaction mixture, consisting of 2 μl of FirstStart DNA Master
SYBR Green I mix, 3 mM MgCl2 and 0.2 μl of primers,
according to the manufacturer’s instructions. The reaction mixture
was then loaded into glass capillary tubes. For real-time qRT-PCR,
the DNA was subjected to an initial denaturation at 95°C for 10
min, followed by 45 rounds of amplification at 95°C (10 sec) for
denaturation, 58°C (10 sec) for annealing and 72°C for extension,
with a temperature slope of 20°C/sec. The transcript levels of
IL1RN were estimated from the respective standard curves and
normalized to the GAPDH transcript in corresponding
samples.
Immunohistochemistry (IHC)
Immunohistochemical staining was performed using a
series of OSCC specimens, which included 39 OSCCs with
corresponding normal tissues, and 18 OPLs that were diagnosed
histopathologically as leukoplakia with epithelial dysplasia.
Considering that evidence has shown that the malignant
transformation rate of oral leukoplakia with dysplasia is higher
than that of oral leukoplakia without dysplasia (14), patients with advanced OPLs, defined
as leukoplakia exhibiting epithelial dysplasia, were considered to
be eligible for this study. IHC staining was carried out on 4 μm
sections of paraffin-embedded specimens. Briefly, after
deparaffinization and hydration, the slides were pretreated in 10
mM sodium citrate buffer (pH 6.0) in a microwave oven for 5 min at
95°C.
The endogenous peroxidase activity was quenched by a
30-min incubation in a mixture of 0.3% hydrogen peroxide solution
in 100% methanol. After washing with PBS buffer, the sections then
were incubated with the primary antibody (biotinylated goat
anti-human IL1RN antibody (1:100 dilution; R&D Systems, Inc.,
Minneapolis, MN, USA) at room temperature and in a humidified
chamber for 2 h. After washing with PBS buffer, the slides were
treated with peroxidase-labeled secondary antibody for 1 h,
followed by color development in 3,3′-diaminobenzidine
tetrahydro-chloride (Dako Japan Inc.). Finally, the slides were
lightly counterstained with hematoxylin. As negative controls,
slides were incubated with PBS instead of the primary antibody. To
quantify the IL1RN protein expression, we used the Histo-score
(H-score) system as previously described (15,16).
In brief, the mean percentage of epithelial cells exhibiting a
persistent IL1RN signal was determined in at least five distinct
fields using ×400 magnification in each section. The intensity of
the immunoreaction was scored as follows: 1+, weak; 2+, moderate
and 3+, intense. Three target cell types: normal, premalignant and
malignant epithelial cells, were identified for scoring.
In the present study, IL1RN immunoreactivity was
detected mainly on the plasma membrane of target cells, and
heterogeneously in the cytoplasm. The plasma membrane IL1RN
immunoreaction was selected for the scoring. The percentage of
IL1RN-positive cells and the staining intensity were then
multiplied to produce the IL1RN H-score. The H-scoring of each
sample was performed by two independent pathologists, neither of
whom had knowledge of the clinical status of the patients.
Statistical analysis
The statistical analyses of the expression levels of
IL1RN mRNA and IL1RN protein were performed using unpaired
or paired Student’s t-test. P<0.05 was considered to be
statistically significant.
Results
IL1RN mRNA expression in OSCC
tissues
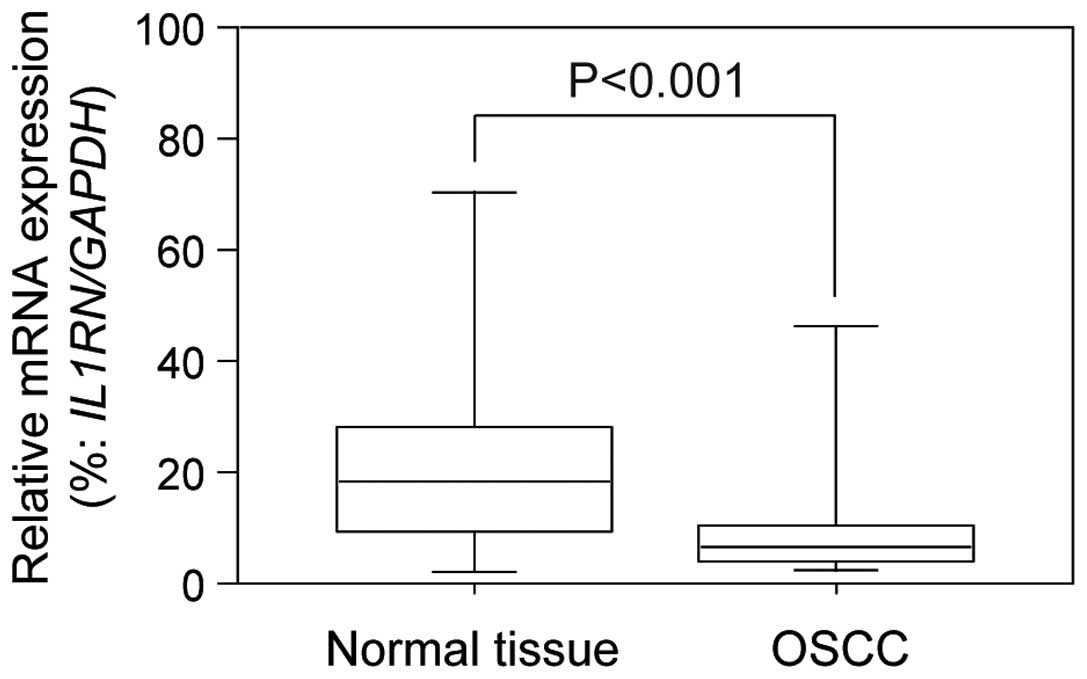
According to real-time qRT-PCR data, the
IL1RN mRNA expression in OSCC specimens was significantly
(P<0.001) lower than that of the corresponding normal mucosa
(Fig. 1). The relative
IL1RN mRNA expression levels in normal and tumor sites
ranged from 0.022 to 4.616 (median, 0.397) and 0.050 to 8.565
(median, 1.335), respectively.
IL1RN expression in OSCCs and OPLs
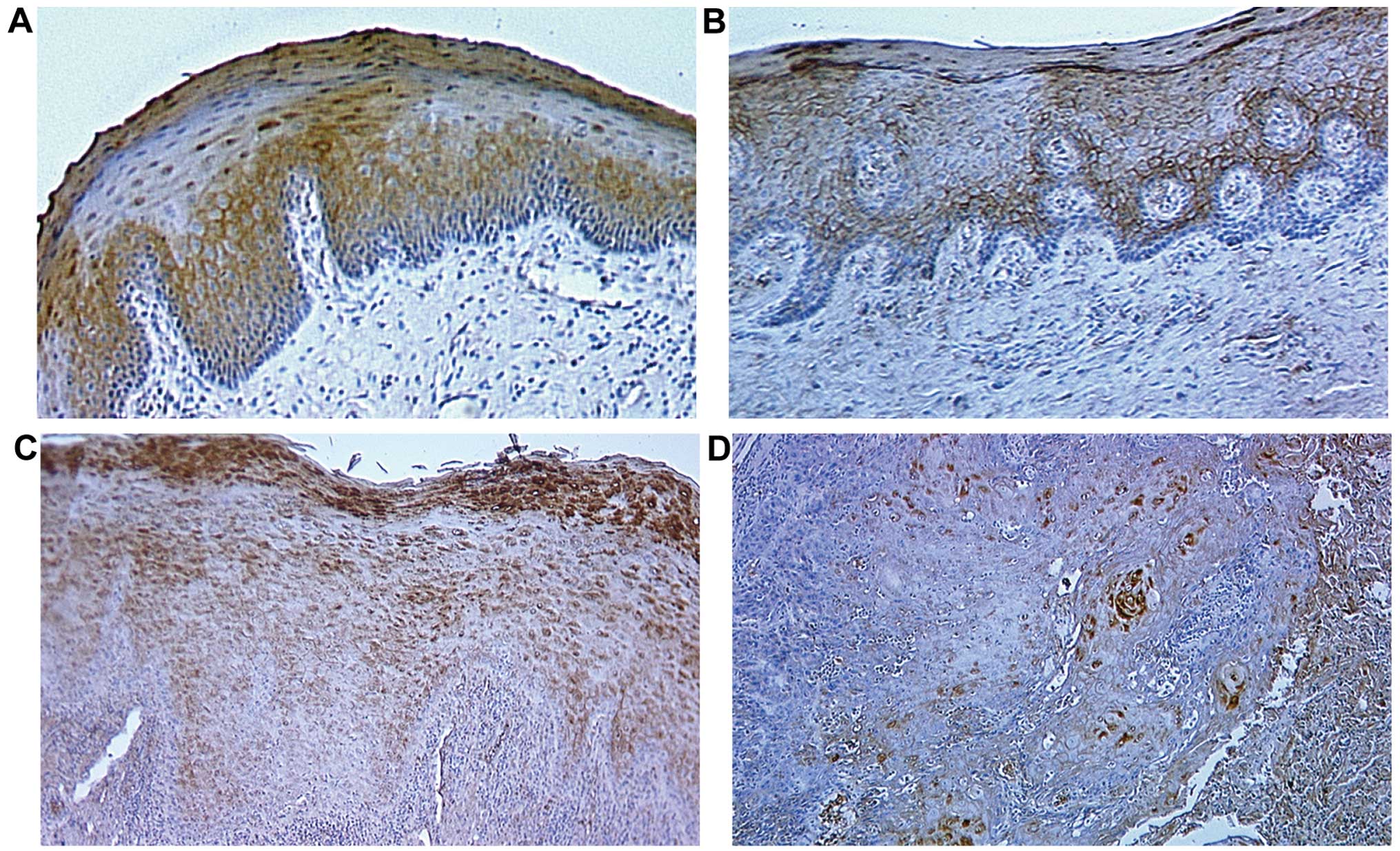
Representative results for IL1RN protein expression
in normal oral tissues, OPLs and primary OSCCs are shown in
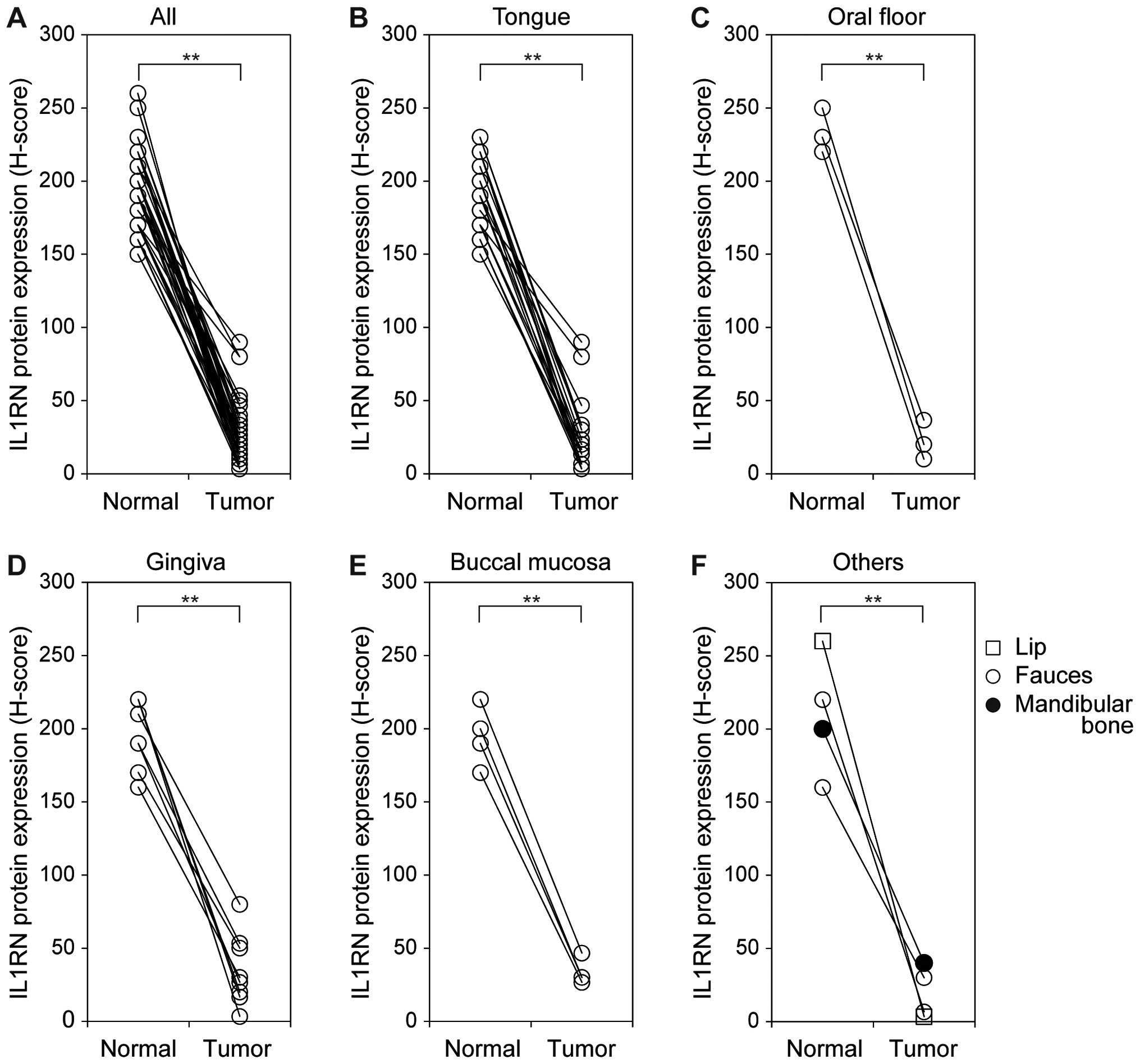
Fig. 2. Normal oral mucosal
specimens exhibited consistently strong IL1RN immunoreactivity on
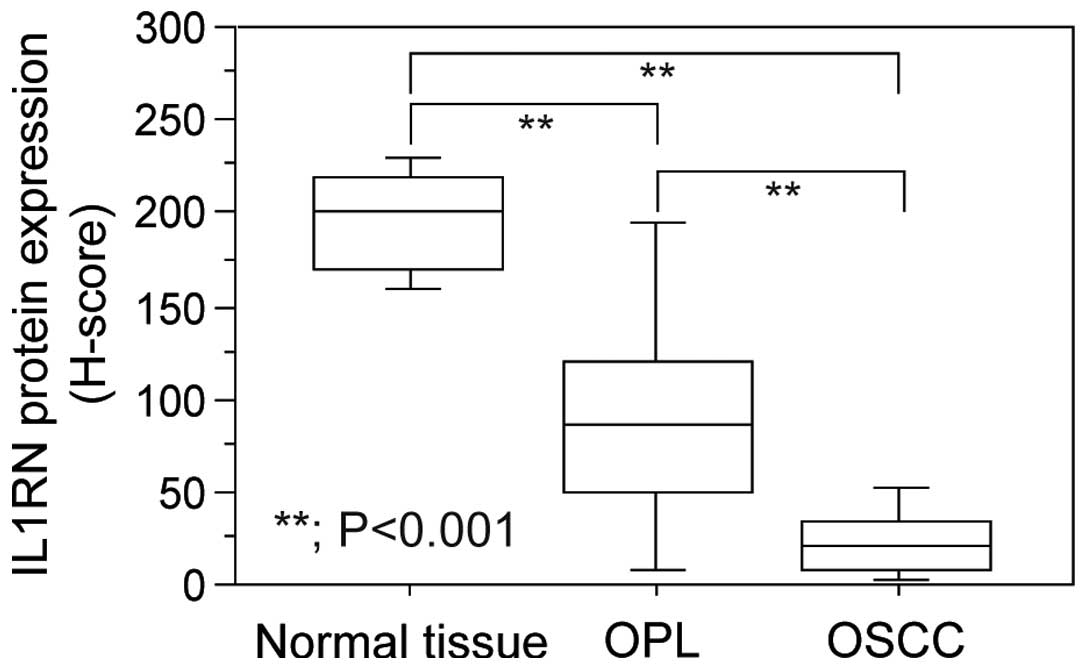
the plasma membrane of cells. According to the H-scores, the
expression levels of plasma membranous IL1RN were significantly
reduced not only in OSCCs, but also in OPLs, in comparison to those
of normal tissues (Fig. 3). In
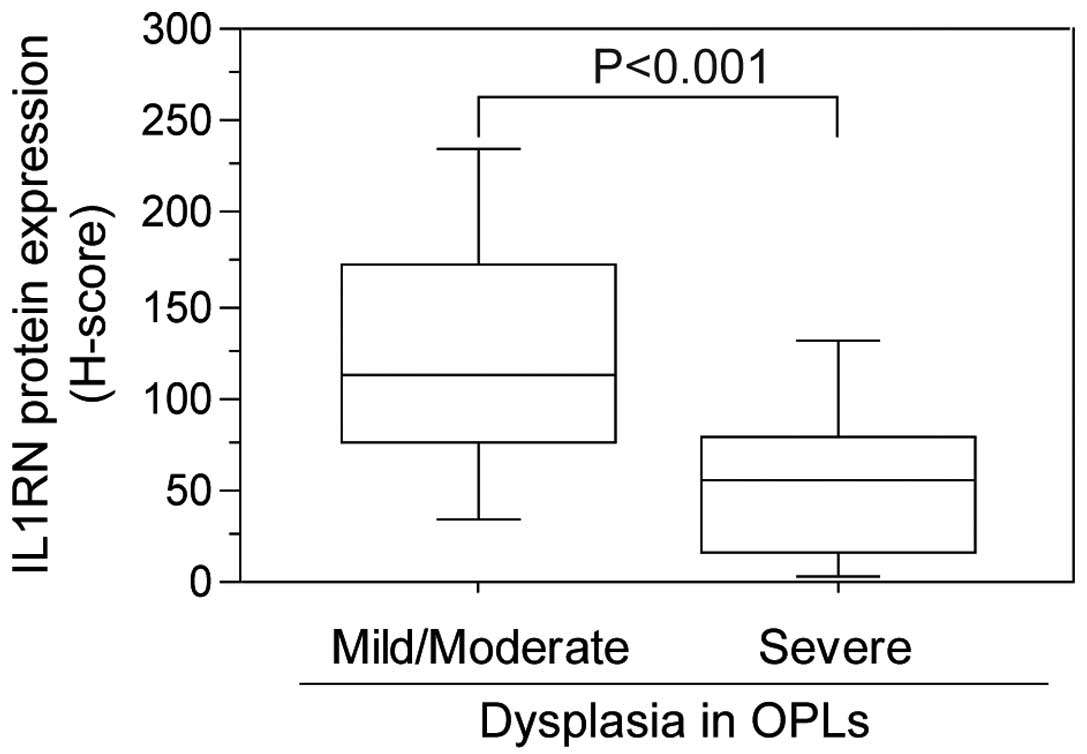
OPLs, although a case with mild dysplasia showed positive plasma
membranous immunostaining for IL1RN, the staining signals decreased
gradually in moderate through to severe dysplasia cases.
Significant differences were observed between mild/moderate
dysplasia cases and severe dysplasia cases (Fig. 4). Notably, IL1RN staining was also
detected heterogeneously in the cytoplasm of OPL cells in some
cases.
In OSCCs, the IL1RN immunoreactivity was largely
lost in the specimens examined. Significantly downregulated IL1RN
expression was observed in all lesion sites examined when compared
with the matched normal tissues (P<0.001) (Fig. 5). However, the decreased level of
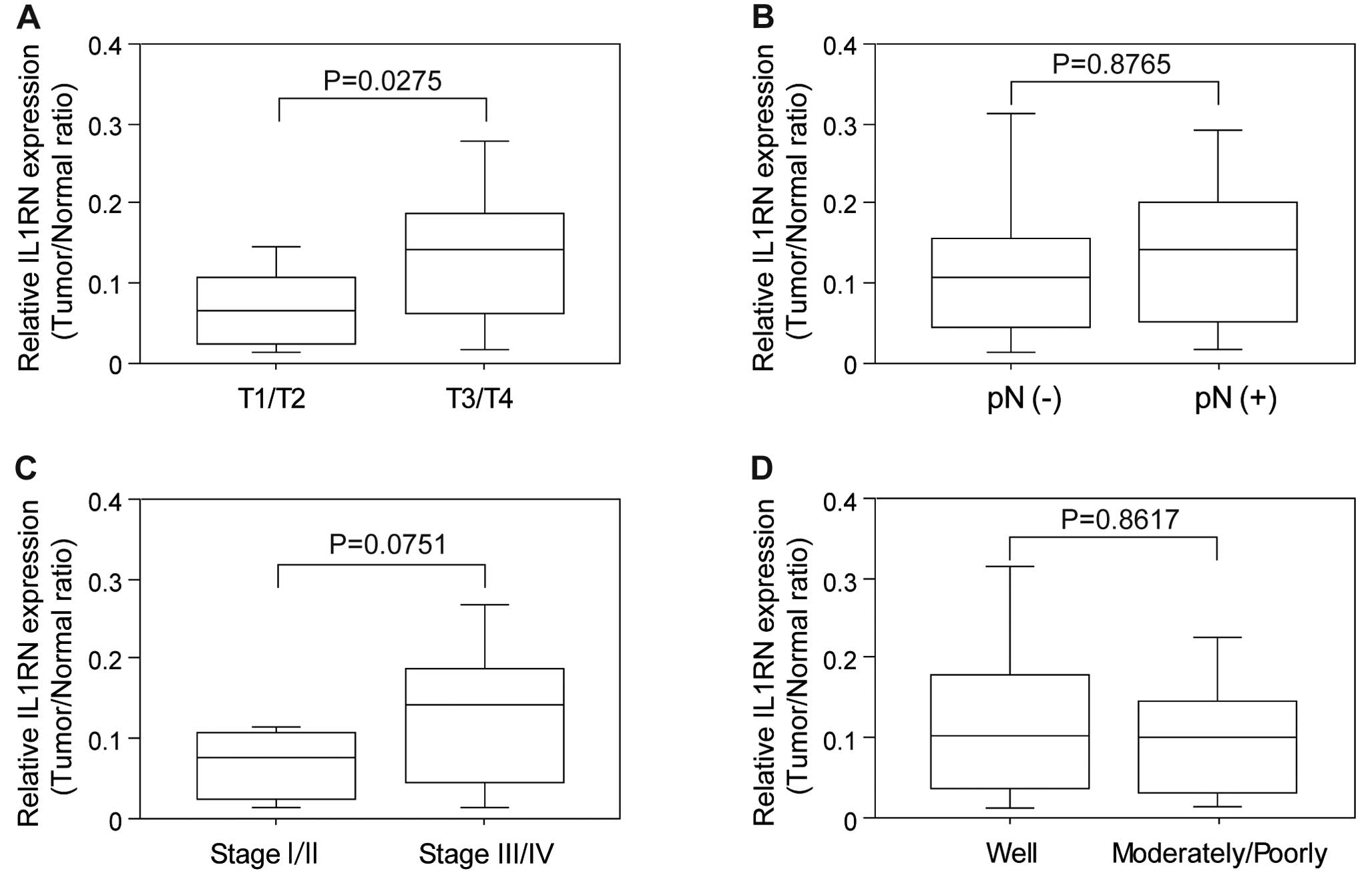
IL1RN expression did not correspond with tumor progression
(Fig. 6). Relative IL1RN
expression levels, determined as relative values of H-scores in
tumor samples compared with normal tissues, were analyzed alongside
clinicopathological factors. Noteworthy, IL1RN expression was
higher in the advanced OSCC cases (T3/T4) in comparison to the
early cases (T1/T2) (P=0.0275) (Fig.
6A). Significant alteration of IL1RN associated with regional
lymph node metastasis (Fig. 6B),
TNM staging (Fig. 6C),
differentiation (Fig. 6D) was not
observed. OSCC cases were divided into two groups according to
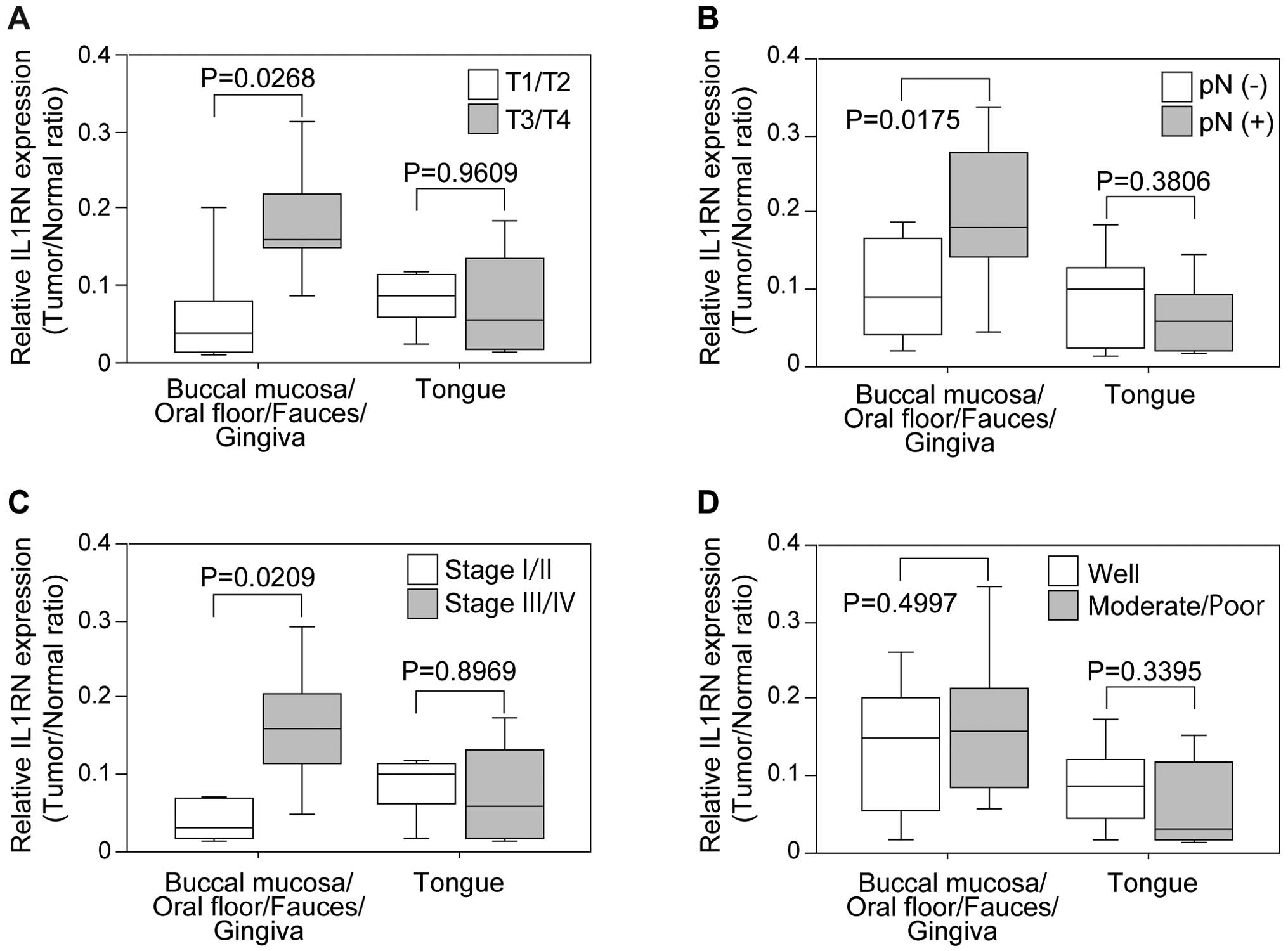
lesion sites: buccal mucosa/oral floor/fauces/gingival, and tongue.
IL1RN expression associated with the clinicopathological factors
was also analyzed separately, and the data revealed that higher
IL1RN expression was associated with active tumor development:
tumor size (Fig. 7A), regional
lymph node metastasis (Fig. 7B),
TNM staging (Fig. 7C), in the OSCC
in buccal mucosa, oral floor, fauces and gingiva but not in the
tongue. No significant difference was found between well and
moderately/poorly differentiated OSCCs (Fig. 7D).
Discussion
A real-time qRT-PCR assay revealed that the relative
IL1RN mRNA expression levels were significantly lower in
OSCCs than in their normal counterparts, which agrees with the
recent study by Lallemant et al (17). Similarly, the IL1RN expression was
significantly downregulated in the OSCC specimens examined compared
to the normal oral epithelium. Yamamoto et al showed that in
OSCCs, a significant loss of heterozygosity occurred at alleles in
chromosome band 2q14, where the IL1RN gene is located (18), and that genetic variations of IL1RN
were strongly associated with the IL1RN expression level (19,20).
This suggested that altered IL1RN expression at the mRNA and
protein levels could result from gene mutations in OSCC.
Low levels of IL1RN have been associated with
greater disease severity in a variety of human malignancies,
including leukemia (21), myeloma
(22), colorectal cancer (23) and prostate cancer (24). In addition, experimental studies
have shown that IL1RN decreases tumor growth or aggressive behavior
by inhibiting IL-1-mediated activities in the cancer cells.
Enhanced expression of IL1RN inhibits tumor growth of skin
carcinoma cells in vitro and in vivo through blocking
the transcriptional effect of IL-1 on cyclooxygenase-2, which is
thought to play a pivotal role in tumor development (25). Elaraj et al showed that
recombinant IL1RN significantly inhibited the tumor growth and
metastatic potential of a human melanoma xenograft that
constitutively secreted IL-1 (26). They also detected that IL1RN had
anti-angiogenic effects in tumors, where it decreased the
IL-1-mediated induction of angiogenic molecules, including
interleukin-8 (IL-8) and vascular endothelial growth factor (VEGF).
Similar findings were also observed for human breast cancer
(27). These findings and our
present results indicate that IL1RN may exhibit tumor-suppressing
activity.
In contrast, there have been some contradictory
observations that suggest IL1RN could increase tumorigenesis. For
example, IL1RN was significantly overexpressed in cervical
carcinomas compared with their normal counterparts (28). Additionally, IL1RN upregulation was
also detected in gastric cancer, and was associated with a high
incidence of metastases (29).
Furthermore, in vitro studies showed that IL1RN enhances the
growth and proliferation of human glioblastoma (30), prostatic (31) and hepatic cancer cells (32). Considering that IL-1 plays multiple
biological roles in various tissues, the effect of IL1RN may be
cell or tissue type-specific, or microenvironment-specific; for
example, it may enhance cytotoxic T cell activity, and the
tumoricidal capacities of natural killer (NK) cells (33,34),
and alterations in the properties of IL1RN are likely due to
changes in the local IL-1-dependent pathways. Although the details
of the biological mechanisms responsible for IL1RN alterations in
tumors have not been determined, our results suggest that IL1RN may
be critical for maintaining the normal condition of cells, and that
a loss of the expression of the protein could result in an enhanced
risk of cancer development in the oral cavity.
The data described in the present study also showed
that the IL1RN expression was gradually downregulated in accordance
with the degree of dysplasia. As oral dysplasia shows a significant
rate of transformation to cancer (35,36),
there is an urgent need to identify better ways to predict which
patients with dysplastic precursor lesions will develop OSCC. The
present results suggest that IL1RN may suppress the early
carcinogenic events and that the expression level could represent a
useful biomarker for the early diagnosis of OSCC. However, our data
revealed that relatively higher expression of IL1RN is found in the
developed OSCC samples, especially in the OSCC occurring in buccal
mucosa, oral floor, fauces and gingiva but not in the tongue.
In conclusion, IL1RN may exhibit conflicting
characteristics in controlling oral malignancies; namely,
suppressive activity in the early carcinogenic events, but also
lesion site-dependent tumor development promotion. Thus, IL1RN
should be a reliable biomarker for the early diagnosis of OSCC.
However, IL1RN may have unknown and complicated functions in the
developed OSCC, suggesting that further investigation is necessary
before considering IL1RN as a therapeutic target in oral
cancer.
Abbreviations:
|
IL1RN
|
interleukin-1 receptor antagonist
|
|
OSCC
|
oral squamous cell carcinoma
|
|
OPL
|
oral premalignant lesion
|
References
|
1
|
La Vecchia C, Lucchini F, Negri E and Levi
F: Trends in oral cancer mortality in Europe. Oral Oncol.
40:433–439. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Koike H, Uzawa K, Nakashima D, Shimada K,
Kato Y, Higo M, Kouzu Y, Endo Y, Kasamatsu A and Tanzawa H:
Identification of differentially expressed proteins in oral
squamous cell carcinoma using a global proteomic approach. Int J
Oncol. 27:59–67. 2005.PubMed/NCBI
|
|
3
|
Sethi G, Shanmugam MK, Ramachandran L,
Kumar AP and Tergaonkar V: Multifaceted link between cancer and
inflammation. Biosci Rep. 32:1–15. 2012. View Article : Google Scholar
|
|
4
|
Aggarwal BB and Gehlot P: Inflammation and
cancer: How friendly is the relationship for cancer patients? Curr
Opin Pharmacol. 9:351–369. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Patel MM and Pandya AN: Relationship of
oral cancer with age, sex, site distribution and habits. Indian J
Pathol Microbiol. 47:195–197. 2004.
|
|
6
|
Znaor A, Brennan P, Gajalakshmi V, Mathew
A, Shanta V, Varghese C and Boffetta P: Independent and combined
effects of tobacco smoking, chewing and alcohol drinking on the
risk of oral, pharyngeal and esophageal cancers in Indian men. Int
J Cancer. 105:681–686. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Matsuo Y, Sawai H, Ma J, Xu D, Ochi N,
Yasuda A, Takahashi H, Funahashi H and Takeyama H: IL-1alpha
secreted by colon cancer cells enhances angiogenesis: The
relationship between IL-1alpha release and tumor cells’ potential
for liver metastasis. J Surg Oncol. 99:361–367. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Matsuo Y, Sawai H, Ochi N, Yasuda A,
Takahashi H, Funahashi H, Takeyama H and Guha S: Interleukin-1alpha
secreted by pancreatic cancer cells promotes angiogenesis and its
therapeutic implications. J Surg Res. 153:274–281. 2009. View Article : Google Scholar
|
|
9
|
Ma J, Sawai H, Matsuo Y, et al:
Interleukin-1alpha enhances angiogenesis and is associated with
liver metastatic potential in human gastric cancer cell lines. J
Surg Res. 148:197–204. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dinarello CA: Interleukin-1 and its
biologically related cytokines. Adv Immunol. 44:153–205. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dinarello CA: Interleukin-1, interleukin-1
receptors and interleukin-1 receptor antagonist. Int Rev Immunol.
16:457–499. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Arend WP: Interleukin 1 receptor
antagonist. A new member of the interleukin 1 family. J Clin
Invest. 88:1445–1451. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Arend WP: The balance between IL-1 and
IL-1Ra in disease. Cytokine Growth Factor Rev. 13:323–340. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Amagasa T, Yamashiro M and Ishikawa H:
Yamashiro M and and Ishikawa H: Oral leukoplakia related to
malignant transformation. Oral Sci Int. 3:45–55. 2006. View Article : Google Scholar
|
|
15
|
Bilalovic N, Sandstad B, Golouh R, Nesland
JM, Selak I and Torlakovic EE: CD10 protein expression in tumor and
stromal cells of malignant melanoma is associated with tumor
progression. Mod Pathol. 17:1251–1258. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
McCarty KS Jr, Szabo E, Flowers JL, et al:
Use of a monoclonal anti-estrogen receptor antibody in the
immunohistochemical evaluation of human tumors. Cancer Res.
46(Suppl 8): S4244–S4248. 1986.
|
|
17
|
Lallemant B, Evrard A, Combescure C, et
al: Clinical relevance of nine transcriptional molecular markers
for the diagnosis of head and neck squamous cell carcinoma in
tissue and saliva rinse. BMC Cancer. 9:3702009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yamamoto N, Mizoe J, Numasawa H, Tsujii H,
Shibahara T and Noma H: Allelic loss on chromosomes 2q, 3p and 21q:
Possibly a poor prognostic factor in oral squamous cell carcinoma.
Oral Oncol. 39:796–805. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Dewberry R, Holden H, Crossman D and
Francis S: Interleukin-1 receptor antagonist expression in human
endothelial cells and atherosclerosis. Arterioscler Thromb Vasc
Biol. 20:2394–2400. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rafiq S, Stevens K, Hurst AJ, et al:
Common genetic variation in the gene encoding
interleukin-1-receptor antagonist (IL-1RA) is associated with
altered circulating IL-1RA levels. Genes Immun. 8:344–351. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kurzrock R: Cytokine deregulation in
cancer. Biomed Pharmacother. 55:543–547. 2001. View Article : Google Scholar
|
|
22
|
Gherardi RK, Bélec L, Soubrier M, Malapert
D, Zuber M, Viard JP, Intrator L, Degos JD and Authier FJ:
Overproduction of proinflammatory cytokines imbalanced by their
antagonists in POEMS syndrome. Blood. 87:1458–1465. 1996.PubMed/NCBI
|
|
23
|
Iwagaki H, Hizuta A and Tanaka N:
Interleukin-1 receptor antagonists and other markers in colorectal
cancer patients. Scand J Gastroenterol. 32:577–581. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Parekh DJ, Ankerst DP, Baillargeon J,
Higgins B, Platz EA, Troyer D, Hernandez J, Leach RJ, Lokshin A and
Thompson IM: Assessment of 54 biomarkers for biopsy-detectable
prostate cancer. Cancer Epidemiol Biomarkers Prev. 16:1966–1972.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
La E, Rundhaug JE and Fischer SM: Role of
intracellular interleukin-1 receptor antagonist in skin
carcinogenesis. Mol Carcinog. 30:218–223. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Elaraj DM, Weinreich DM, Varghese S,
Puhlmann M, Hewitt SM, Carroll NM, Feldman ED, Turner EM and
Alexander HR: The role of interleukin 1 in growth and metastasis of
human cancer xenografts. Clin Cancer Res. 12:1088–1096. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lindahl G, Saarinen N, Abrahamsson A and
Dabrosin C: Tamoxifen, flaxseed, and the lignan enterolactone
increase stroma- and cancer cell-derived IL-1Ra and decrease tumor
angiogenesis in estrogen-dependent breast cancer. Cancer Res.
71:51–60. 2011. View Article : Google Scholar
|
|
28
|
Fujiwaki R, Iida K, Nakayama K, Kanasaki
H, Hata K, Katabuchi H, Okamura H and Miyazaki K: Clinical
significance of interleukin-1 receptor antagonist in patients with
cervical carcinoma. Gynecol Oncol. 89:77–83. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Iizuka N, Hazama S, Hirose K, Abe T,
Tokuda N, Fukumoto T, Tangoku A and Oka M: Interleukin-1 receptor
antagonist mRNA expression and the progression of gastric
carcinoma. Cancer Lett. 142:179–184. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Oelmann E, Kraemer A, Serve H, Reufi B,
Oberberg D, Patt S, Herbst H, Stein H, Thiel E and Berdel WE:
Autocrine interleukin-1 receptor antagonist can support malignant
growth of glioblastoma by blocking growth-inhibiting autocrine loop
of interleukin-1. Int J Cancer. 71:1066–1076. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hsieh TC and Chiao JW: Growth modulation
of human prostatic cancer cells by interleukin-1 and interleukin-1
receptor antagonist. Cancer Lett. 95:119–123. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yamada Y, Karasaki H, Matsushima K, Lee GH
and Ogawa K: Expression of an IL-1 receptor antagonist during mouse
hepato-carcinogenesis demonstrated by differential display
analysis. Lab Invest. 79:1059–1067. 1999.PubMed/NCBI
|
|
33
|
Dvorkin T, Song X, Argov S, White RM,
Zoller M, Segal S, Dinarello CA, Voronov E and Apte RN: Immune
phenomena involved in the in vivo regression of fibrosarcoma cells
expressing cell-associated IL-1alpha. J Leukoc Biol. 80:96–106.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Voronov E, Weinstein Y, Benharroch D, et
al: Antitumor and immunotherapeutic effects of activated invasive T
lymphoma cells that display short-term interleukin 1alpha
expression. Cancer Res. 59:1029–1035. 1999.PubMed/NCBI
|
|
35
|
Al-Dakkak I: Oral dysplasia and risk of
progression to cancer. Evid Based Dent. 11:91–92. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Liu W, Bao ZX, Shi LJ, Tang GY and Zhou
ZT: Malignant transformation of oral epithelial dysplasia:
Clinicopathological risk factors and outcome analysis in a
retrospective cohort of 138 cases. Histopathology. 59:733–740.
2011. View Article : Google Scholar : PubMed/NCBI
|