Introduction
As the fourth leading cause of cancer death
worldwide, pancreatic cancer is an aggressive malignant disease
with a median survival time of less than 6 months and a 5-year
survival rate of <6% (1). Due
to a lack of early symptoms, ~80% of pancreatic cancer patients are
advanced in unresectable stage at the time of diagnosis (2). Even patients with seemingly
resectable tumor are not always cured by surgery due to the
microscopic systemic spread of the pancreatic cancer that occurs
prior to the procedure (3).
Improvement of patient survival requires an increased understanding
of tumor metastasis mechanisms to allow for early disease detection
and the development of therapeutic strategy. Previously we
identified that superoxide dismutase (SOD)-dependent production of
reactive oxygen species (ROS) increased the invasive and migratory
ability of pancreatic cancer cells (4). However, little is known about the
specific mechanism underlying this linkage.
ROS generated by the mitochondrial respiratory
chain, consists of a number of chemically reactive molecules
derived from oxygen, including hydrogen peroxide
(H2O2). Accumulating evidence indicates that
the intracellular redox state plays an important role in cellular
signaling transduction and regulates multiple events, such as cell
cycle progression, apoptosis, migration, invasion and angiogenesis
in cancer (5,6). The main antioxidant enzymes include
SOD, which converts superoxide anion (O2−)
into H2O2, as well as catalase (CAT) and
glutathione peroxidase (GPX), which catabolize
H2O2 into water (7). Although they are sometimes classified
together, they should be considered separately because their
reactions are completely different. It has been demonstrated that
SOD can induce migration and invasion of cancer cells via its
metabolic product H2O2. Whereas, the
H2O2 scavenger, catalase inhibits
ROS-mediated tumor metastasis (4,8). In
pancreatic cancer, the antioxidant activity of SOD is increased in
the tumor cells cultured from ascites and metastatic tumor cell
lines compared to primary pancreatic tumor cells, which indicates
that SOD is intimately related with tumor metastasis potential
(9).
Epithelial-mesenchymal transition (EMT) has been
recognized not only as a physiological mechanism for development
and tissue remodeling, but also as a pathological mechanism in
cancer progression, during which cells lose their polarized
epithelial traits and acquire mesenchymal characteristics such as
the downregulation of E-cadherin and the upregulation of N-cadherin
and vimentin that in turn induce an aggressive phenotype (10,11).
This event not only facilitates the aggressiveness of the disease
but also promotes resistance to current treatments (12). Elevated amounts of ROS have been
proven to promote several cellular migratory processes, including
EMT, angiogenesis and metastasis in different cancer types
(13). Various stimulators such as
epidermal growth factor and transforming growth factor-β could also
induce EMT through ROS generation (14). As one of the most classical
signaling pathway, extracellular signal-regulated kinase (ERK)
pathway is an important signaling cascade downstream of ROS that is
involved in tumor migration and invasion (15).
In the present study, we investigated the production
of H2O2 in pancreatic cancer cells in
response to SOD and CAT. We also tested the hypothesis that
H2O2 mediates SOD-induced activation of ERK
signaling pathway, which further regulates the invasive and
migratory activity of pancreatic cancer cells through EMT. Results
from this study suggest that elevation of
H2O2 contributes to the SOD induced EMT
through ERK/NF-κB signaling pathway.
Materials and methods
Preparation of chemicals
Dulbecco’s modified Eagle’s medium (DMEM) and fetal
bovine serum (FBS) were from Gibco (Grand Island, NY, USA). SOD and
CAT were from Sigma-Aldrich (St. Louis, MO, USA). The hydrogen
peroxide assay kit was from Beyotime (Jinan, China). Millicell
culture plate inserts were from Millipore (Bedford, MA, USA).
Matrigel was from BD (Biosciences, Bedford, MA, USA). The ERK
inhibitor PD 98059 was obtained from Sigma-Aldrich. Primary
antibodies against E-cadherin, N-cadherin and vimentin were from
Santa Cruz Biotechnology (Santa Cruz, CA, USA). The anti-ERK,
anti-phospho-ERK (Thr202/Tyr204), anti-NF-κB and anti-phospho-NF-κB
(Ser468) antibodies were from Cell Signaling Technology (Beverly,
MA, USA). Nitrocellulose membranes were from Millipore. The BCA
assay kit and the chemiluminescence kit were from Pierce (Rockford,
IL, USA). Other reagents were purchased from common commercial
sources. All drug solutions were freshly prepared on the day of
testing.
Cell cultures and treatments
The human pancreatic cancer cell lines, BxPC-3 and
Panc-1, were obtained from the American Type Culture Collection
(Manassas, VA, USA). The cells were cultured in DMEM medium
containing 10% dialyzed heat-inactivated FBS, 100 U/ml penicillin,
and 100 μg/ml streptomycin in a humidified atmosphere of 5%
CO2 at 37°C. SOD (400 U/ml) was used to convert
superoxide anions into molecular oxygen and hydrogen peroxide, CAT
(400 U/ml) was also added to the in vitro model in
combination with SOD to decompose the hydrogen peroxide.
Hydrogen peroxide assay
The level of intracellular
H2O2 was measured using hydrogen peroxide
assay kit according to the manufacturer’s instructions. In this
kit, the ferrous ions (Fe2+) were oxidized to ferric
ions (Fe3+) by H2O2, which further
formed a complex with the indicator dye xylenol orange and produced
a visible purple-colored complex, that could then be measured using
a microplate reader at a wavelength of 560–590 nm (Bio-Rad, CA,
USA).
Transwell Matrigel invasion assay
The invasive ability of the pancreatic cancer cells
was analyzed using Matrigel invasion chambers in 24-well plates.
The 8.0-μm pore inserts were coated with 25 μl of Matrigel. After
serum starvation for 24 h, the BxPC-3 and Panc-1 cells were
suspended in DMEM containing 1% FBS in the top chamber at a
concentration of 5×104 in the absence or presence of
SOD, SOD accompanied with CAT at concentrations of 400 U/ml each
and SOD accompanied with PD 98059 (50 μmol/l). Simultaneously, 500
ml of DMEM containing 20% FBS was placed in the lower chambers. The
Matrigel invasion chamber was then incubated for 48 h in a
humidified tissue culture incubator. The non-invading cells were
removed from the upper surface by scraping with a wet cotton swab.
After rinsing with PBS, the filter was fixed and stained with
crystal violet. The invasion ability was determined by counting the
stained cells on the bottom surface. Three random fields were
captured at ×20 magnification (n=3).
Wound healing assay
Cell migratory ability was detected by a
wound-healing assay. Pancreatic cancer cells were seeded in 24-well
plates (1.0 ×105 cells/500 μl). After the cells grew to
90–100% confluence, a sterile pipette tip was used to produce a
wound line between the cells. Cellular debris was removed by
washing with PBS and then allowed to migrate for 24 h. Images were
taken at time 0 and 24 h post-wounding under a Nikon Diaphot TMD
inverted microscope (x10). The relative distance traveled by the
leading edge from 0 to 24 h was assessed using Photoshop software
(n=5).
Real-time quantitative PCR (QT-PCR)
Total RNA was extracted from the pancreatic cancer
cells using the Fastgen 200 RNA isolation system (Fastgen,
Shanghai, China) according to the manufacturer’s protocol. Total
RNA was reverse-transcribed into cDNA using the Fermentas
RevertAid™ kit (MBI Fermentas, Canada). The primer sequences were
as follows: E-cadherin-F, 5′-ATTCTGATTCTGCTGCTCTTG-3′ and
E-cadherin-R, 5′-AGTCCTGGTCCTCTTCTCC-3′; N-cadherin-F,
5′-TGTTTGACTATGAAGGCAGTGG-3′ and N-cadherin-R,
5′-TCAGTCATCACCTCCACCAT-3′; vimentin-F, 5′-AATGACCGCTTCGCCAAC-3′
and vimentin-R, 5′-CCG CATCTCCTCCTCGTAG-3′; β-actin-F, 5′-GACTTAGTT
GCGTTACACCCTTTCT-3′ and β-actin-R, 5′-GAACGGT GAAGGTGACAGCAGT-3′.
The PCR reactions consisted of 30 sec at 95°C, followed by 40
cycles of 95°C for 5 sec, 60°C for 30 sec and 72°C for 30 sec.
After each QT-PCR experiment, a dissociation curve analysis was
conducted. The relative gene expression was calculated using the
previously described 2−ΔΔCt method (16).
Protein extraction and western
blotting
Total protein was extracted from cultured cells in
radio-immunoprecipitation assay (RIPA) lysis buffer on ice for 25
min. Insoluble materials were removed by centrifugation at 4°C with
15,000 × g for 15 min. Subsequently, supernatants were collected
and total protein concentrations were measured using the BCA assay
kit. Proteins (30–80 μg) were electrophoretically resolved on a
denaturing SDS-polyacrylamide gel and electrotransferred onto
nitrocellulose membranes. The membranes were initially blocked with
5% non-fat dry milk in Tris-buffered saline (TBS) for 2 h and then
probed with antibodies against E-cadherin, N-cadherin, vimentin,
ERK, phospho-ERK, NF-κB, phospho-NF-κB and β-actin. After
incubation with the primary antibodies at 4°C overnight, the
membranes were hybridized with secondary goat anti-mouse or goat
anti-rabbit antibodies (Sigma-Aldrich) for 2 h at room temperature.
Immunopositive bands were developed using an enhanced
chemiluminescence (ECL) detection system (Amersham, Piscataway, NJ,
USA). All analyses were conducted in triplicate.
Statistical analysis
Statistical analysis was performed using SPSS
software (version 17.0, SPSS Inc., Chicago, IL, USA). Data are
presented as the means ± SEM of three replicate assays. Differences
between the groups were analyzed by analysis of variance (ANOVA).
Statistical significance was set at P<0.05. All experiments were
repeated independently at least three times.
Results
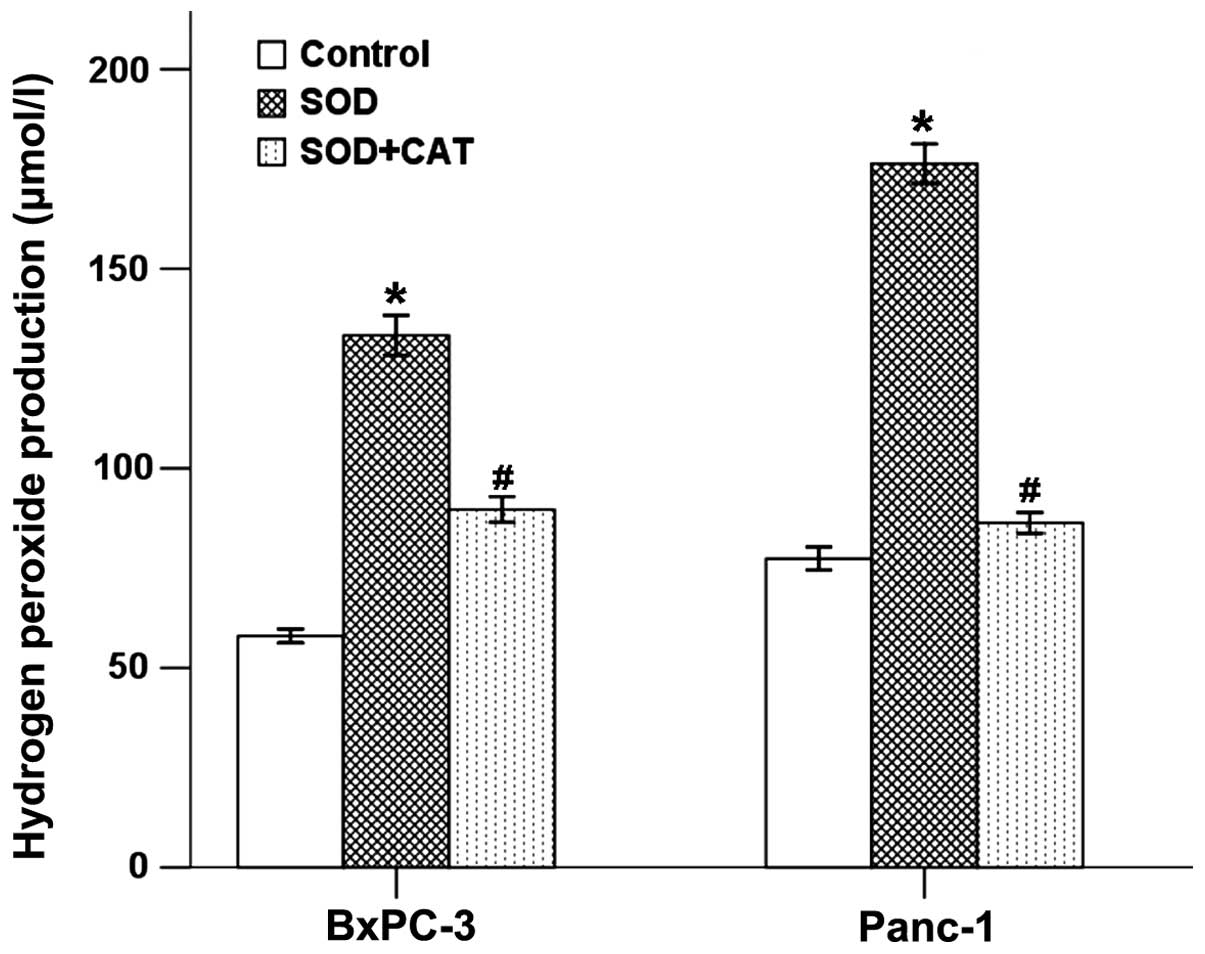
SOD increases the production of
H2O2 in pancreatic cancer cells
The intracellular H2O2 in
BxPC-3 and Panc-1 cells treated with SOD or SOD accompanied with
CAT was determined using a hydrogen peroxide assay kit detected by
a microplate reader. As shown in Fig.
1, the levels of H2O2 in both the BxPC-3
and Panc-1 cells were upregulated in response to SOD stimulation.
CAT could counter-balance the effect of SOD in both cancer cell
types.
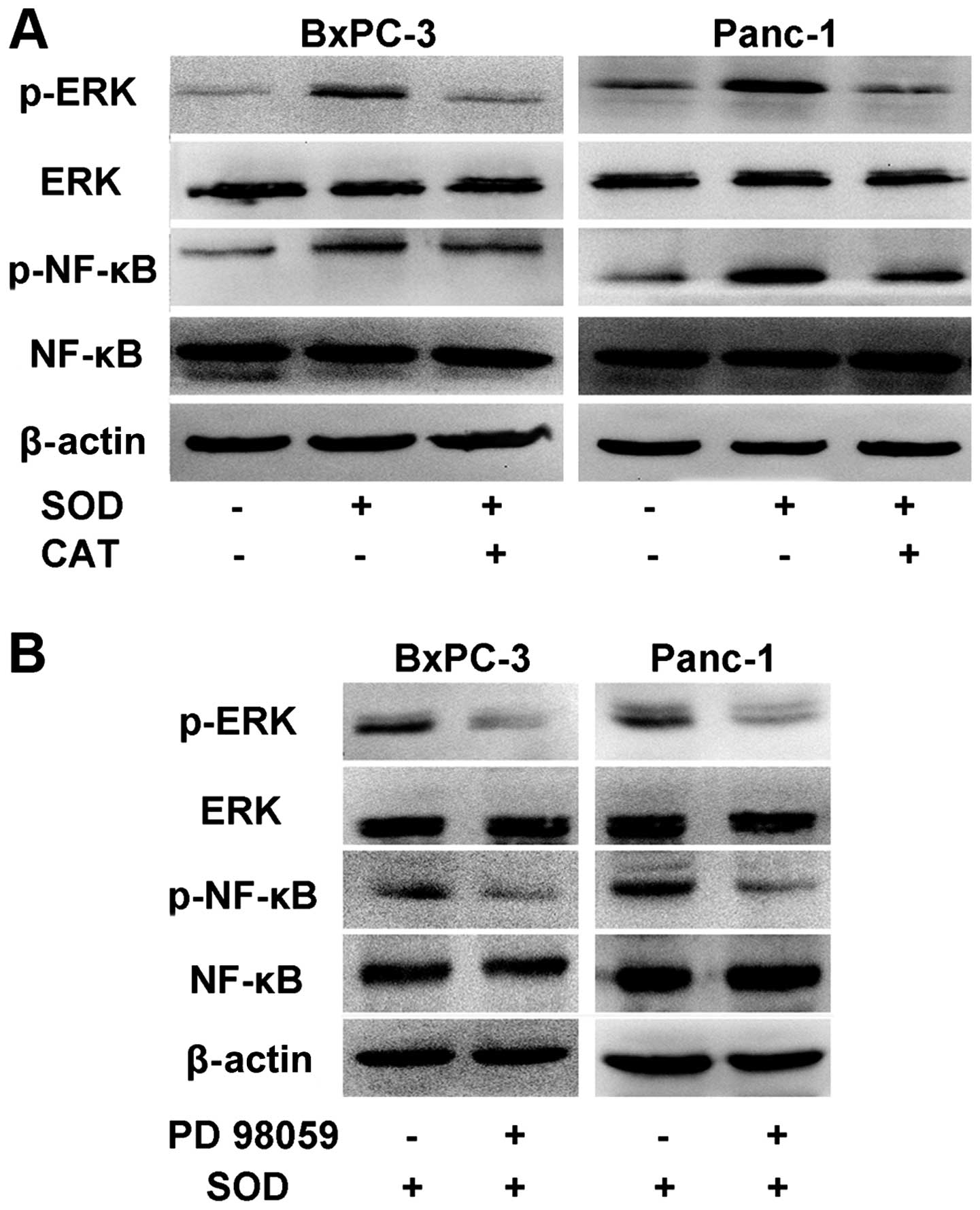
SOD upregulates the activation of the
ERK/NF-κB signaling pathway
As an important member of mitogen activated protein
kinase (MAPK) signaling pathway, ERK pathway is downstream of ROS
which is involved in tumor migration and invasion (17). A recent study demonstrated that ERK
pathway induces activation of NF-κB transcription factor, and is
associated with cell migration activity (18).
In the present study, we observed that both ERK and
NF-κB phosphorylation were strongly increased with the addition of
SOD (Fig. 2A). When we added SOD
with CAT together into the cell culture, the expression of
phospho-ERK and phospho-NF-κB decreased significantly, indicating
that the activation of ERK/NF-κB signaling pathway in BxPC-3 and
Panc-1 cells was hydrogen peroxide-dependent. In addition the ERK
inhibitor PD 98059 was able to inhibit the expression of
phospho-ERK and phospho-NF-κB, indicating that the NF-κB
transcription factor is modulated by the ERK pathway (Fig. 2B).
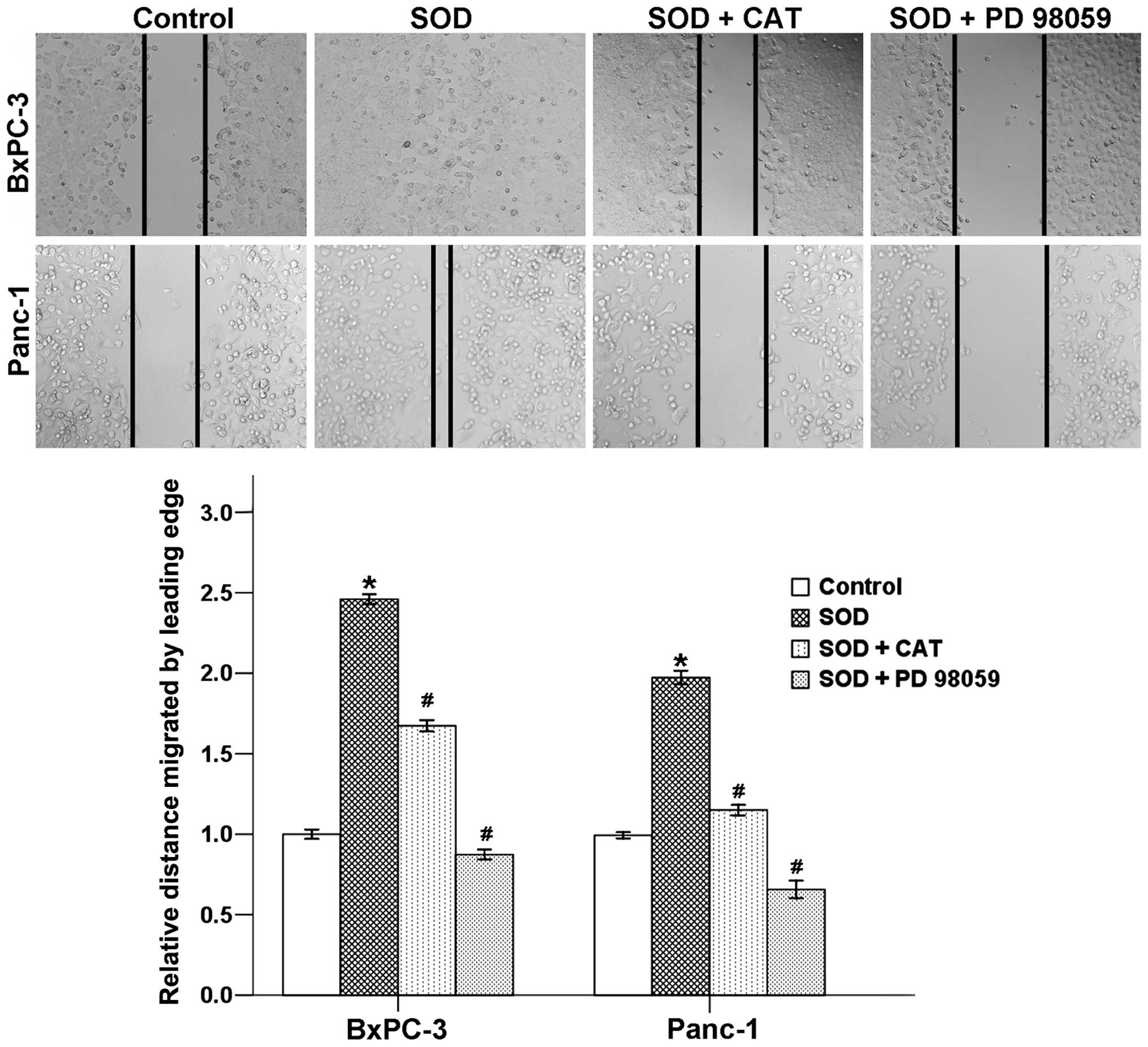
SOD promotes wound closure via the
activation of H2O2/ERK axis
Migration and invasion are two important aspects
that lead to the ability of cancer cells to form metastases. In
order to further characterize the role of SOD in promoting
metastasis, we investigated the role of SOD in migration using a
classic wound healing assay. Results showed that SOD caused a
significant increase in the migration of both BxPC-3 and Panc-1
cells after incubation for 24 h. Both CAT and PD 98059 suppressed
these effects which proved that H2O2/ERK axis
mediated SOD-induced cell migratory ability (Fig. 3).
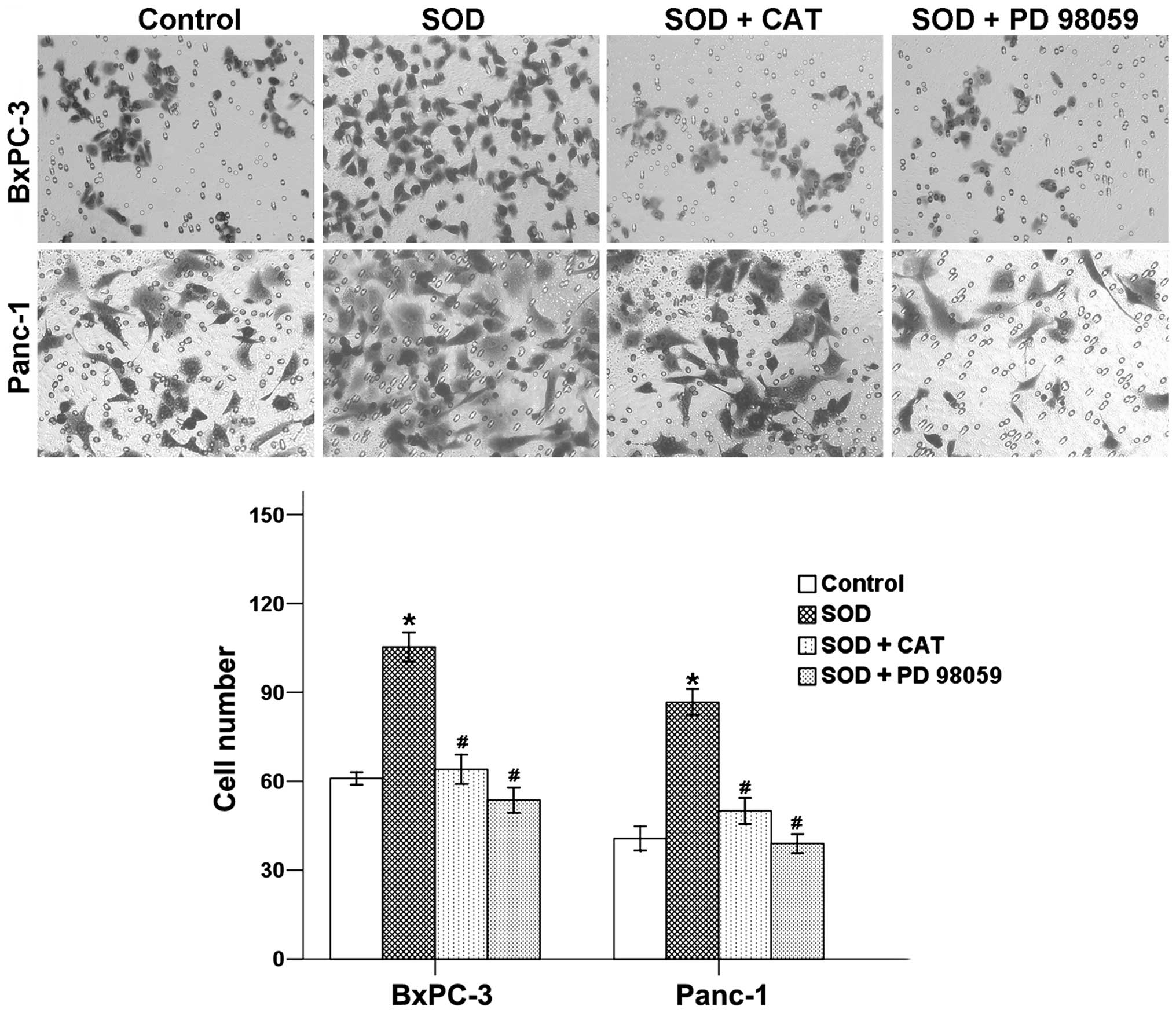
Activation of
H2O2/ERK axis is responsible for the
promotion of cancer cell invasion
To gain more insight into the role of
H2O2/ERK axis in cancer progression, BxPC-3
and Panc-1 cells were treated with SOD in the absence or presence
of CAT and PD 98059. As shown in Fig.
4, the average cell numbers that invaded into the lower chamber
increased with the addition of SOD after incubation for 48 h. This
increase was reversed by co-treating with both CAT and PD 98059.
These finding revealed that H2O2/ERK axis
might be an effective inhibitor of the migration and invasion of
pancreatic cancer cells.
SOD induces EMT via
H2O2 production in pancreatic cancer
cells
EMT, a critical character of tumor metastasis,
contains three essential processes: first, alterations of cell-cell
and cell-extracellular matrix (ECM) interactions occur releasing
the epithelial cells from the surrounding tissue. Then the
cyto-skeleton is reorganized so that the cells can gain the ability
to move through ECM. After that, a new transcriptional program is
induced to acquire morphological and functional characteristics of
mesenchymal-like cells (6). In our
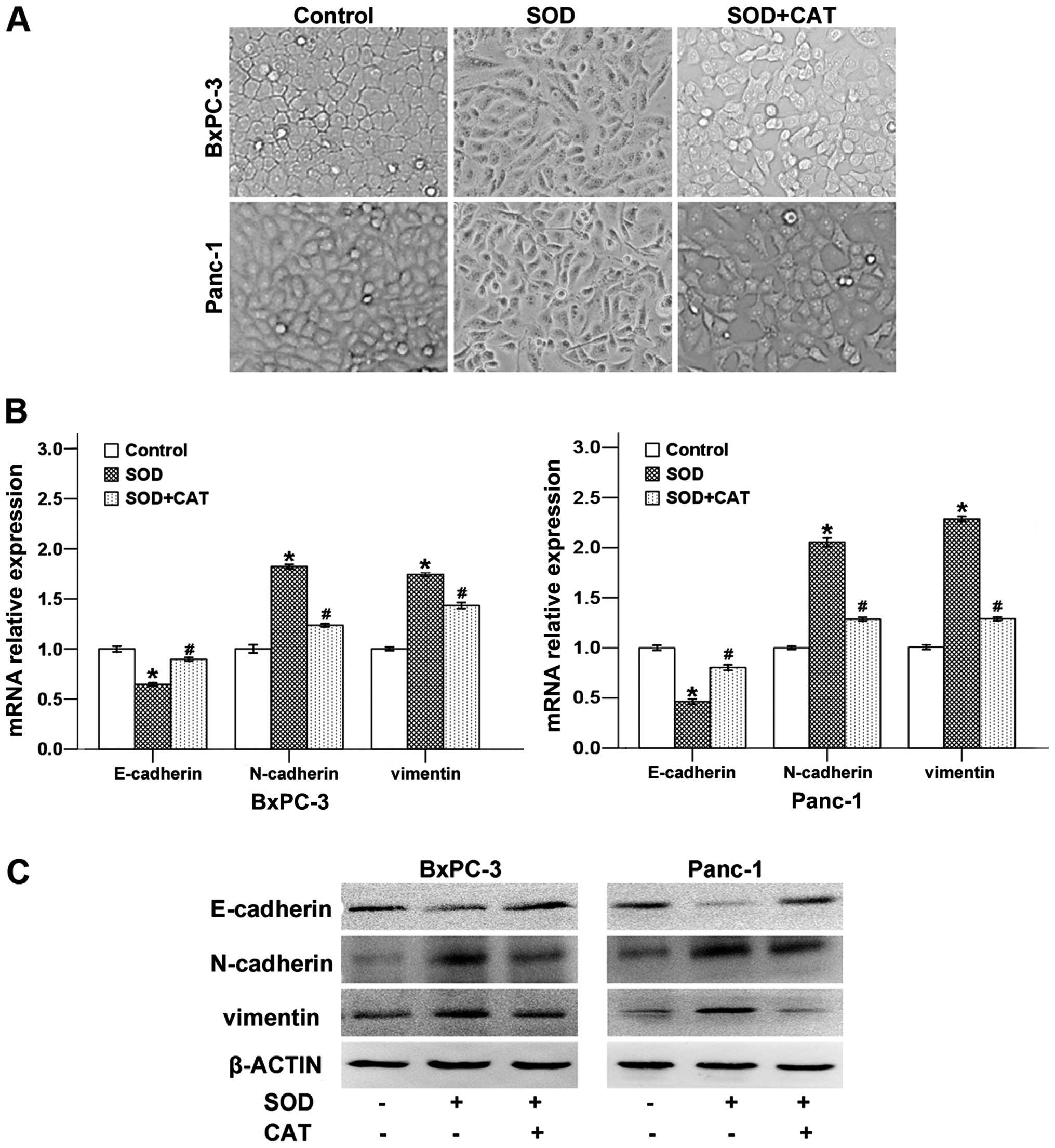
study, we demonstrated that after treated with SOD for 48 h, the
cellular morphology of both BxPC-3 and Panc-1 cells was changed
from an epithelial phenotype to a classical mesenchymal phenotype.
Cells treated with SOD and CAT displayed classical epithelial
morphology (Fig. 5A).
To further confirm the effect of
H2O2 on SOD-induced EMT, we determined the
expression levels of EMT-related genes after the cells were SOD
treated with or without CAT. As shown in Fig. 5B, SOD downregulated the mRNA level
of the epithelial marker E-cadherin, while the expression of
mesenchymal markers N-cadherin and vimentin were strongly
increased. CAT was able to significantly reverse these SOD-induced
effects.
To evaluate the effects of SOD on the expression of
E-cadherin, N-cadherin and vimentin at protein level, we determined
these proteins in BxPC-3 and Panc-1 cells with or without CAT using
western blotting. As shown in Fig.
5C, CAT counter-balanced the SOD-induced, EMT-related factors
at the protein level, and the trend was consistent with the mRNA
results. Taken together, our results demonstrate that SOD induces
EMT progression via the production of H2O2 in
both BxPC-3 and Panc-1 cells.
Discussion
As a fatal step in solid cancer progression, distant
metastasis is responsible for approximately 90% of cancer-related
deaths (19). It is commonly
believed that the poor prognosis of pancreatic cancer is due to
both the inherently aggressive biology of the disease and its late
diagnosis in most cases (20). In
recent years, EMT has received significant attention in cancer
metastasis. Emerging evidence has suggested that EMT is the first
step by which cancer cells invade and metastasize to other organs.
Cancer cells undergoing EMT are able to obtain invasive properties
to penetrate the surrounding tissue, leading to the creation of a
suitable microenvironment for cancer proliferation and metastasis
(13,21). Our previous study has demonstrated
that SOD-dependent production of ROS was able to increase the
invasive and migratory ability of pancreatic cancer cells. In this
study, we focus on the underlying mechanisms through which SOD
promotes the metastasis ability in pancreatic cancer cell lines
BxPC-3 and Panc-1.
Our data showed that SOD could significantly
increase the production of intracellular H2O2
in BxPC-3 and Panc-1 cells, which further enhanced the capacity of
the pancreatic cancer cells to migrate and invade the extracellular
matrix. As the scavenger of H2O2, CAT was
able to terminate these effects of SOD. ERK pathway has long been
recognized as an important signaling cascade downstream of ROS that
is involved in tumor migration and invasion (15). In order to determine whether the
cell invasion and migration potential as well as the increased
H2O2 production is related to ERK pathway, we
tested the effects of SOD and CAT on the activation of phospho-ERK
and the relative transcriptional factor NF-κB. With SOD alone, both
ERK and NF-κB phosphorylation was strongly increased in pancreatic
cancer cells, whereas the addition of CAT along with SOD to the
cell culture resulted in a decrease of phospho-ERK and
phospho-NF-κB. This phenomenon suggested that the activation of
ERK/NF-κB pathway mediated by SOD was
H2O2-dependent. In addition, the present
study also showed that SOD was able to modulate cancer cell
morphology as well as the expression of EMT-related factors, which
could be counter-balanced by both CAT treatment and the ERK
inhibitor PD 98059.
ROS, particularly, serve as regulators or secondary
messengers of signal transduction pathways for cell proliferation,
survival, apoptosis and migration (22). It has been proven that
H2O2 is diffusible and thus capable of
traveling across plasma membranes into the extracellular space to
exert a paracrine role (23).
Intrinsic antioxidant enzymes are vital to the regulation of
oxidative stress within cells. SOD, one of the primary cellular
antioxidants, catalyzes the conversion of superoxide anion to
H2O2, which can be removed by CAT. Three
forms of SOD exist in mammals that serve the same effect:
copper-zinc SOD (CuZnSOD), manganese SOD (MnSOD) and extracellular
SOD (EC-SOD). CuZnSOD is located in the cytoplasm, mitochondrial
intermembrane space and the nucleus. MnSOD is located exclusively
in the mitochondrial matrix, whereas EC-SOD is primary located in
the extracellular matrix (ECM) (23).
Both epidemiologic and experimental evidence
indicate that the levels of SOD increase in many tumor types as
they progress from early stage non-invasive disease to late stage
metastatic disease (24,25). Increased SOD levels have been
associated with poor prognosis and resistance to therapy of various
tumors in the central nervous system, gastrointestinal tract, and
head and neck (26). Toh et
al (27) reported 2.19-and
3.72-fold increases in SOD mRNA expression relative to normal
tissue in gastric and colorectal cancers, respectively.
Epidemiologic evidence has also linked a single nucleotide
polymorphism in SOD, which increases its activity, to risk of
developing cancers (28). The
SOD2-1221G>A AA genotype carriers had a significantly increased
risk for pancreatic cancer among those with a low dietary vitamin E
intake (29). Several authors have
reported that SOD is involved in the invasive properties of cancer
cells. It has been proven that metastatic pancreatic cancer cell
line Capan-1, contain elevated levels of SOD and decreased levels
of CAT, and that this change in steady-state levels of hydrogen
peroxide correlates with increased metastasis, angiogenesis,
proliferation and resistance to apoptosis (9,24).
Mice injected with MnSOD-GFP-transfected HT-1080 cells presented
with multiple intrapulmonary metastatic lesions surrounding
pulmonary blood vessels (30).
Migration and invasion of the SOD-expressing HT-1080 fibrosarcoma
and 253J transitional bladder carcinoma cells were inhibited
following overexpression of CAT, indicating that the promigratory
invasive phenotype of SOD-expressing cells is
H2O2-dependent (30).
Increasing number of researchers have focused on the
role of ROS in EMT-related cancer in recent years. Mori and
colleagues first established a direct link between extracellular
generation of ROS and EMT (31).
In their study, normal mouse mammary gland epithelial cells were
exposed to a low dose of H2O2 for periods of
2–4 days. A phenotypic conversion of mouse mammary epithelial cells
from an epithelial to a fibroblast-like phenotype was observed,
which was associated with the dissolution of cell-cell contacts,
redistribution of E-cadherin in the cytoplasm, and upegulation of a
set of integrin family members and matrix metalloproteinases
(MMPs). Kim et al (32)
reported that H2O2 could promote the EMT
program, which was mediated through HIF-1α and TGF-β1 in human
malignant mesothelioma (HMM) cells. Simultaneously, the
H2O2 also increased the expression of stem
cell-related genes, suggesting the enhanced potential of survival
and proliferation of the HMM cells metastasis. Our results confirm
that SOD-induced H2O2 influences the
migration and invasion via EMT in the pancreatic cancer cell lines
BxPC-3 and Panc-1.
Members of the MAPK family include the extracellular
ERK, c-jun NH-2 terminal kinase (JNK) and p38 MAPK. Numerous
studies have proven the relationship between ERK signaling pathway
and cancer progression. Lee et al (33) showed that hepatocyte growth factor
(HGF) regulates H2O2 production, which
further activates the ERK pathway and regulates uPA production,
eventually increasing the invasive potential of stomach cancer
cells. Liu et al (34) also
demonstrated that bone morphogenetic protein 2-induced cellular
invasiveness and MMP-2 activity is ROS/ERK-dependent in pancreatic
cancer. Recent studies proved that the metastasis-mediated effect
of the ERK signaling pathway might be modulated via the NF-κB
transcription factor, which is associated with cell proliferation,
cell migration and angiogenesis (35). Our results indicated that SOD was
able to increase the activation of phospho-ERK and phospho-NF-κB
which was counter-balanced by CAT. After suppressing the ERK
signaling pathway by PD 98059, the expression of phospho-ERK and
phospho-NF-κB as well as the invasion and migration ability of both
BxPC-3 and Panc-1 pancreatic cancer cells were decreased.
In conclusion, the results of the present study
suggest that SOD-induced H2O2 production can
promote EMT in pancreatic cancer, leading to increased motility and
invasion via activation of ERK signaling pathway. Managing
H2O2/ERK/NF-κB axis might be a novel strategy
for the treatment of this severe malignancy. Our findings warrant
further investigation of this hypothesis.
Acknowledgements
This study was supported by grant from National
Natural Science Foundation of China (grant no. 81301846).
References
|
1
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sarkar FH, Banerjee S and Li Y: Pancreatic
cancer: Pathogenesis, prevention and treatment. Toxicol Appl
Pharmacol. 224:326–336. 2007. View Article : Google Scholar
|
|
3
|
Castellanos EH, Cardin DB and Berlin JD:
Treatment of early-stage pancreatic cancer. Oncology (Williston
Park). 25:182–189. 2011.
|
|
4
|
Li W, Ma Q, Li J, Guo K, Liu H, Han L and
Ma G: Hyperglycemia enhances the invasive and migratory activity of
pancreatic cancer cells via hydrogen peroxide. Oncol Rep.
25:1279–1287. 2011.PubMed/NCBI
|
|
5
|
Lee DJ and Kang SW: Reactive oxygen
species and tumor metastasis. Mol Cells. 35:93–98. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li W, Ma Q, Liu J, Han L, Ma G, Liu H,
Shan T, Xie K and Wu E: Hyperglycemia as a mechanism of pancreatic
cancer metastasis. Front Biosci (Landmark Ed). 17:1761–1774. 2012.
View Article : Google Scholar
|
|
7
|
Costa A, Scholer-Dahirel A and
Mechta-Grigoriou F: The role of reactive oxygen species and
metabolism on cancer cells and their microenvironment. Semin Cancer
Biol. 25:23–32. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liu Z, Li S, Cai Y, Wang A, He Q, Zheng C,
Zhao T, Ding X and Zhou X: Manganese superoxide dismutase induces
migration and invasion of tongue squamous cell carcinoma via
H2O2-dependent Snail signaling. Free Radic
Biol Med. 53:44–50. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lewis A, Du J, Liu J, Ritchie JM, Oberley
LW and Cullen JJ: Metastatic progression of pancreatic cancer:
Changes in antioxidant enzymes and cell growth. Clin Exp
Metastasis. 22:523–532. 2005. View Article : Google Scholar
|
|
10
|
Duan W, Li R, Ma J, Lei J, Xu Q, Jiang Z,
Nan L, Li X, Wang Z, Huo X, et al: Overexpression of Nodal induces
a metastatic phenotype in pancreatic cancer cells via the Smad2/3
pathway. Oncotarget. 6:1490–1506. 2015.PubMed/NCBI
|
|
11
|
Inumaru J, Nagano O, Takahashi E, Ishimoto
T, Nakamura S, Suzuki Y, Niwa S, Umezawa K, Tanihara H and Saya H:
Molecular mechanisms regulating dissociation of cell-cell junction
of epithelial cells by oxidative stress. Genes Cells. 14:703–716.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Barnett P, Arnold RS, Mezencev R, Chung
LW, Zayzafoon M and Odero-Marah V: Snail-mediated regulation of
reactive oxygen species in ARCaP human prostate cancer cells.
Biochem Biophys Res Commun. 404:34–39. 2011. View Article : Google Scholar :
|
|
13
|
Das TP, Suman S and Damodaran C: Induction
of reactive oxygen species generation inhibits
epithelial-mesenchymal transition and promotes growth arrest in
prostate cancer cells. Mol Carcinog. 53:537–547. 2014. View Article : Google Scholar
|
|
14
|
Cho KH, Choi MJ, Jeong KJ, Kim JJ, Hwang
MH, Shin SC, Park CG and Lee HY: A ROS/STAT3/HIF-1α signaling
cascade mediates EGF-induced TWIST1 expression and prostate cancer
cell invasion. Prostate. 74:528–536. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wu WS, Wu JR and Hu CT: Signal cross talks
for sustained MAPK activation and cell migration: The potential
role of reactive oxygen species. Cancer Metastasis Rev. 27:303–314.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
17
|
Li F, Wang H, Huang C, Lin J, Zhu G, Hu R
and Feng H: Hydrogen peroxide contributes to the manganese
superoxide dismutase promotion of migration and invasion in glioma
cells. Free Radic Res. 45:1154–1161. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hsieh HL, Wang HH, Wu WB, Chu PJ and Yang
CM: Transforming growth factor-β1 induces matrix
metalloproteinase-9 and cell migration in astrocytes: Roles of
ROS-dependent ERK- and JNK-NF-κB pathways. J Neuroinflammation.
7:882010. View Article : Google Scholar
|
|
19
|
Wu Y and Zhou BP: New insights of
epithelial-mesenchymal transition in cancer metastasis. Acta
Biochim Biophys Sin (Shanghai). 40:643–650. 2008. View Article : Google Scholar
|
|
20
|
Wray CJ, Ahmad SA, Matthews JB and Lowy
AM: Surgery for pancreatic cancer: Recent controversies and current
practice. Gastroenterology. 128:1626–1641. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Iwatsuki M, Mimori K, Yokobori T, Ishi H,
Beppu T, Nakamori S, Baba H and Mori M: Epithelial-mesenchymal
transition in cancer development and its clinical significance.
Cancer Sci. 101:293–299. 2010. View Article : Google Scholar
|
|
22
|
Chiu WT, Shen SC, Chow JM, Lin CW, Shia LT
and Chen YC: Contribution of reactive oxygen species to
migration/invasion of human glioblastoma cells U87 via
ERK-dependent COX-2/PGE(2) activation. Neurobiol Dis. 37:118–129.
2010. View Article : Google Scholar
|
|
23
|
Chaiswing L, Zhong W, Cullen JJ, Oberley
LW and Oberley TD: Extracellular redox state regulates features
associated with prostate cancer cell invasion. Cancer Res.
68:5820–5826. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hempel N, Carrico PM and Melendez JA:
Manganese super-oxide dismutase (Sod2) and redox-control of
signaling events that drive metastasis. Anticancer Agents Med Chem.
11:191–201. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chen PM, Wu TC, Wang YC, Cheng YW, Sheu
GT, Chen CY and Lee H: Activation of NF-κB by SOD2 promotes the
aggressiveness of lung adenocarcinoma by modulating NKX2-1-mediated
IKKβ expression. Carcinogenesis. 34:2655–2663. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kinnula VL and Crapo JD: Superoxide
dismutases in malignant cells and human tumors. Free Radic Biol
Med. 36:718–744. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Toh Y, Kuninaka S, Oshiro T, Ikeda Y,
Nakashima H, Baba H, Kohnoe S, Okamura T, Mori M and Sugimachi K:
Overexpression of manganese superoxide dismutase mRNA may correlate
with aggressiveness in gastric and colorectal adenocarcinomas. Int
J Oncol. 17:107–112. 2000.PubMed/NCBI
|
|
28
|
Wang S, Wang F, Shi X, Dai J, Peng Y, Guo
X, Wang X, Shen H and Hu Z: Association between manganese
superoxide dismutase (MnSOD) Val-9Ala polymorphism and cancer risk
- A meta-analysis. Eur J Cancer. 45:2874–2881. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tang H, Dong X, Day RS, Hassan MM and Li
D: Antioxidant genes, diabetes and dietary antioxidants in
association with risk of pancreatic cancer. Carcinogenesis.
31:607–613. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Connor KM, Hempel N, Nelson KK, Dabiri G,
Gamarra A, Belarmino J, Van De Water L, Mian BM and Melendez JA:
Manganese superoxide dismutase enhances the invasive and migratory
activity of tumor cells. Cancer Res. 67:10260–10267. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mori K, Shibanuma M and Nose K: Invasive
potential induced under long-term oxidative stress in mammary
epithelial cells. Cancer Res. 64:7464–7472. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kim MC, Cui FJ and Kim Y: Hydrogen
peroxide promotes epithelial to mesenchymal transition and stemness
in human malignant mesothelioma cells. Asian Pac J Cancer Prev.
14:3625–3630. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lee KH, Kim SW and Kim JR: Reactive oxygen
species regulate urokinase plasminogen activator expression and
cell invasion via mitogen-activated protein kinase pathways after
treatment with hepatocyte growth factor in stomach cancer cells. J
Exp Clin Cancer Res. 28:732009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu J, Ben QW, Yao WY, Zhang JJ, Chen DF,
He XY, Li L and Yuan YZ: BMP2 induces PANC-1 cell invasion by MMP-2
over-expression through ROS and ERK. Front Biosci (Landmark Ed).
17:2541–2549. 2012. View
Article : Google Scholar
|
|
35
|
Gilmore TD: Introduction to NF-kappaB:
Players, pathways, perspectives. Oncogene. 25:6680–6684. 2006.
View Article : Google Scholar : PubMed/NCBI
|