Introduction
In recent years, research on the relation between
genetic mutation and cancer treatment efficiency is making
progress, which is being applied to the development of new drugs,
especially molecular target drugs. Along with the popularization of
molecular target drugs, diagnosis before medication has been
growing rapidly in practical use for drug selection and/or decision
making on the treatment strategy (1–3).
There are many types of molecular target drugs to target epidermal
growth factor receptor (EGFR) such as tyrosine kinase inhibitor; a
low-molecular compound, and antibody drugs. Oncogenic mutation that
lies downstream of EGFR target drugs is a signal transduction
molecule, and it is extremely important to check this mutation for
predicting drug efficacy (4).
Anti-EGFR antibody, cetuximab or panitumumab is a treatment for
colon cancer that is highly effective to patients with expression
of EGFR protein; however, it has been reported that patients with
KRAS gene mutations that lie downstream acquire resistance against
therapy (5–7).
KRAS is a signal transduction molecule that is
playing a part in mitogen-activated protein kinase (MAPK) pathway
that lies downstream of EGFR and is related to cell proliferation
and angiogenesis (8). KRAS gene
mutation is known for inducing constitutive activation of KRAS and
stimulating cancer growth, and it is found in various organs, such
as the colon, pancreas and lungs. In Japanese population, KRAS gene
mutation is found in 30–42% of colon cancer patients (9). The mutations are found mainly in
codons 12 and 13. Due to single nucleotide or dinucleotide
mutations, some amino acids are substituted with other amino acids.
It is known that resistance against anti-EGFR antibodies will be
acquired when there is a mutation in KRAS codon 12/13. However,
there is an interesting report that mutation in codon 13 (p.G13D)
has lower resistance against anti-EGFR antibodies compared with
other mutations and extends the overall survival and
progression-free survival time of the patient (10,11).
Thus, there is a high possibility that detection of p.G13D apart
from other mutations will have clinical importance in the
future.
The entire drug efficacy cannot be predicted just
from KRAS gene mutation itself and other factors are likely be
involved. One of the factors is the BRAF gene mutation (V600E) that
lies downstream of EGFR, similarly to KRAS (9). BRAF V600E mutation has been found in
~4.7% of the colon cancer patients in Japan. Again, similarly to
KRAS mutation, constant self-activation is considered to induce the
activity of signal pathway and stimulate canceration (9). There has also been a report that it
has resistance against treatment with anti-EGFR antibodies
(12). It is invaluable to give
appropriate therapeutic opportunity to patients to whom treatment
will be effective. Thus, checking these items for diagnosis prior
to drug administration is highly beneficial.
In order to spread these genetic mutation tests with
great clinical significance, we have established a measurement
system that allows simultaneous measurements of KRAS codon 12/13,
p.G13D and BRAF. This system uses QProbe (QP) method and can detect
these mutations quickly and relatively easily. In this study, the
accuracy of the novel system was compared to that of the
conventional Direct Sequence (DS) method. For gene analysis, we
have also studied a revolutionary pre-treatment that does not
require any complicated operations such as DNA purification, which
is also reported herein.
Materials and methods
Specimens
Tissues collected from 182 colon cancer patients who
received surgery between 2009 and 2010 at The Cancer Institute
Hospital of JFCR were used as specimens. Purified DNA was extracted
from the specimens using QIAamp DNA FFPE Tissue kit (Qiagen,
Hilden, Germany). Also, frozen biopsy specimens (32 specimens)
collected at the same hospital in 2011 were used for measurement
without extracting DNA. All the patients enrolled in this study
were approved by the Institutional Review Board at the Cancer
Institute Hospital of the Japanese Foundation for Cancer Research
(JFCR). Written informed consent was obtained from each
patient.
Detection of KRAS mutation
KRAS mutation was detected using the DS method and
QP method (13). For DS method
based on nested polymerase chain reaction (PCR), the primer
sequences listed below and PCR reaction conditions were used in the
measurement.
Primers used in the first round PCR reaction were
5′-ggagtatttgatagtgtattaacct-3′ (sense) and 5′-gaaaatggtcagagaaacc
tttatc-3′ (antisense). Conditions of PCR reaction were a cycle of 1
min at 94°C, 10 sec at 98°C, 5 sec at 55°C and 30 sec at 72°C,
repeated 40 times. After reaction, they were stored at 4°C. For the
second round PCR reaction, primers 5′-gtgtgacatgttctaatatagtca-3′
(sense) and 5′-gtcctgcaccagtaatatgc-3′ (antisense) were used.
Conditions of PCR reaction were a cycle of 1 min at 94°C, 10 sec at
98°C, 15 sec at 55°C and 30 sec at 72°C, repeated 30 times. Reagent
used for PCR reaction was Prime STAR HS (Premix) (Takara Bio Inc.,
Shiga, Japan) and instrument was GeneAmp PCR System 9700 (Applied
Biosystems, Foster City, CA, USA). Amplified product was purified
using QIAquik PCR Purification kit (Qiagen). Sequencing reaction
was performed using BigDye Terminator v3.1 Cycle Sequencing kit
(Applied Biosystems), and primers of the same sort used in the
second round PCR reaction were used for each reaction. Then, they
were purified using BigDye XTerminator Purification kit (Applied
Biosystems) for checking the sequence information with Applied
Biosystems 3130x/Genetic Analyzer (Applied Biosystems).
The following primer and QProbe sequences were used
for QP method measurement: primer sequences for detecting KRAS
mutations are 5′-aaggcctgctgaaaatgactg-3′ (sense) and
5′-ggtcctgcaccagtaatatgca-3′ (antisense). QProbe (Nippon Steel
& Sumikin Eco-Tech Corp., Tokyo, Japan) sequences are
5′-(BODIPY FL)-ctcttgcctacgccaccagctccaact-3′ for detecting KRAS
codon 12/13 and 5′-(Pacific Blue)-cttgcc tacgtca-3′ for detecting
p.G13D. In QP method, existence of KRAS codon 12/13 mutation can be
detected; however, codon 12 and 13 cannot be distinguished since
probes are designed for wild-type of KRAS codon 12/13 (14,15).
However, G13D can be distinguished by matching specific probes with
p.G13D.
Moreover, KRAS and BRAF mutation were detected
simultaneously in a single measurement by combining primers and
probes for BRAF V600E mutation detection. The following primer and
QProbe sequences were used for measuring BRAF mutation:
Primer sequence for detecting BRAF V600E mutation is
5′-tgcttgctctgataggaaaatgagatctac-3′ (sense) and 5′-aaact
gatgggacccactccat-3′ (antisense). QProbe sequence is 5′-gctaca
gAgaaatctc-(TAMRA)-3′. For the QP method, PCR and detection
processes were performed automatically using fully automated gene
analyzer, i-densy™ IS-5320 (Arkray Inc., Kyoto, Japan). Before the
measurement, the necessary number of tips, reaction tubes, reagent
packs and specimens are set in the instrument. The results become
available in 90 min just by pressing the START button (16). Since up to three mutations can be
run in parallel with a single reagent pack, we have tested the KRAS
codon 12/13, p.G13D and BRAF V600E mutations all at once. After 1
min of initial degeneration at 95°C, i-densy™ repeats 60 cycles of
PCR: heat degeneration for 1 se at 95°C and annealing for 30 sec at
62°C. After the completion of PCR, mutation is detected through Tm
analysis. Mutations are identified by the differences in melting
temperature. Specimens that gave different results in DS method and
QP method were checked by the Scorpion-ARMS method.
Simple pre-treatment without DNA
extraction
Biopsy specimen (≤1-mm3) was diluted to
10 μl with purified water and heat processed for 5 min at 95°C.
When biopsy specimen was large, it was lightly homogenized to
become <1-mm3. This method was used for the PCR
reactions and detections described below.
Results
Detection of KRAS mutations using the DS
and QP methods
DNA extracted from tissues collected from 182 colon
cancer patients was used to check the existence of KRAS mutation
(Table I). As a result, DS method
screened 121 cases out of 182 as KRAS mutation-negative, 47 cases
as KRAS codon 12 mutation-positive and 14 cases as KRAS codon 13
(p.G13D)-positive. On the other hand, QP method screened 92 cases
out of 182 as KRAS mutation-negative, 72 cases as KRAS codon 12
mutation-positive, 13 cases as p.G13D-positive and 5 cases as
undeterminable. The concordance rate of the two methods regarding
KRAS-negative, codon 12-positive and p.G13D-positive results were
75.2, 93.6 and 92.3%, respectively, and 81.4% on average (excluding
undeterminable specimens). There were as many as 33 specimens with
diverging results. Putting aside the 5 undeterminable cases with QP
method, among the 117 cases that became KRAS mutation-negative with
DS method, specimens with diverging results from QP method were as
many as 29 cases (28 cases for codon 12 mutation-positive and one
case of p.G13D-positive). In addition, 153 cases of BRAF V600E
mutation were also measured in parallel with KRAS mutation.
Although the measurement was only done with the QP method, 6 cases
became positive and the frequency of occurrence was 3.9% (data not
shown).
 | Table IComparison of KRAS codon 12/13
mutation between the DS and QP methods. |
Table I
Comparison of KRAS codon 12/13
mutation between the DS and QP methods.
| QProbe method | | |
|---|
|
| | |
|---|
| KRAS mutationa | Wild-type | Codon 12 | p.G13D | Undeterminable | Concordance
ratec |
|---|
| Wild-type | 88 | 28 | 1 | 4 | 72.7% (75.2%) |
| Codon 12b | 3 | 44 | 0 | 0 | 93.6% |
| p.G13D | 1 | 0 | 12 | 1 | 85.7% (92.3%) |
| | | | | Total 79.1% (81.4
%) |
QP method has higher capacity for KRAS
mutation detection compared to the DS method
Among 33 specimens with diverging results between DS
method and QP method, 12 specimens could be retested (Table II). In order to search for the
cause of the divergence, 12 specimens were retested with
Scorpion-ARMS method (Table
III). As a result, among the 10 cases where codon 12 mutation
became negative with DS method and positive with QP method, 7 cases
became positive with Scorpion-ARMS method. Also, 2 cases where the
results were negative with DS method became negative with both QP
method and Scorpion-ARMS method.
 | Table IIComparison of KRAS codon 12/13
mutation between the DS and QP methods. |
Table II
Comparison of KRAS codon 12/13
mutation between the DS and QP methods.
| No. | DS | QProbe |
|---|
| 1 | p.G12N | Codon 12 |
| 2 | p.G12V | Codon 12 |
| 3 | p.G12V | Codon 12 |
| 4 | p.G12V | Codon 12 |
| 5 | p.G12D | Codon 12 |
| 6 | p.G12D | Codon 12 |
| 7 | p.G12D | Codon 12 |
| 8 | p.G12V | Codon 12 |
| 9 | p.G12D | Codon 12 |
| 10 | p.G12D | Codon 12 |
| 11 | p.G12D | Codon 12 |
| 12 | p.G12D | Codon 12 |
| 13 | p.G12S | Codon 12 |
| 14 | p.G12D | Codon 12 |
| 15 | p.G12D | Codon 12 |
| 16 | p.G12S | Codon 12 |
| 17 | p.G12D | Codon 12 |
| 18 | p.G12D | Codon 12 |
| 19 | p.G12D | Codon 12 |
| 20 | p.G12D | Codon 12 |
| 21 | p.G12D | Codon 12 |
| 22 | p.G12V | Codon 12 |
| 23 | p.G12V | Codon 12 |
| 24 | p.G12S | Codon 12 |
| 25 | p.G12S | Codon 12 |
| 26 | p.G12D | Codon 12 |
| 27 | p.G12C | Codon 12 |
| 28 | p.G12D | Codon 12 |
| 29 | p.G12S | Codon 12 |
| 30 | p.G12D | Codon 12 |
| 31 | p.G12C | Codon 12 |
| 32 | p.G12V | Codon 12 |
| 33 | p.G12D | Codon 12 |
| 34 | p.G12D | Codon 12 |
| 35 | p.G12D | Codon 12 |
| 36 | p.G12V | Codon 12 |
| 37 | p.G12D | Codon 12 |
| 38 | p.G12D | Codon 12 |
| 39 | p.G12D | Codon 12 |
| 40 | p.G12V | Codon 12 |
| 41 | p.G12D | Codon 12 |
| 42 | p.G12D | Codon 12 |
| 43 | p.G12S | Codon 12 |
| 44 | p.G12A | Codon 12 |
| 45 | p.G12V/D | Wild-type |
| 46a | p.G12D | Wild-type |
| 47 | p.G12D | Wild-type |
| 48 | p.G13D | p.G13D |
| 49 | p.G13D | p.G13D |
| 50 | p.G13D | p.G13D |
| 51 | p.G13D | p.G13D |
| 52 | p.G13D | p.G13D |
| 53 | p.G13D | p.G13D |
| 54 | p.G13D | p.G13D |
| 55 | p.G13D | p.G13D |
| 56 | p.G13D | p.G13D |
| 57 | p.G13D | p.G13D |
| 58 | p.G13D | p.G13D |
| 59 | p.G13D | p.G13D |
| 60a | p.G13D | Wild-type |
| 61 | p.G13D | - |
| 62 | Wild-type | p.G13D |
| 63a | Wild-type | Codon 12 |
| 64a | Wild-type | Codon 12 |
| 65a | Wild-type | Codon 12 |
| 66 | Wild-type | Codon 12 |
| 67 | Wild-type | Codon 12 |
| 68 | Wild-type | Codon 12 |
| 69a | Wild-type | Codon 12 |
| 70 | Wild-type | Codon 12 |
| 71 | Wild-type | Codon 12 |
| 72a | Wild-type | Codon 12 |
| 73a | Wild-type | Codon 12 |
| 74 | Wild-type | Codon 12 |
| 75 | Wild-type | Codon 12 |
| 76 | Wild-type | Codon 12 |
| 77 | Wild-type | Codon 12 |
| 78 | Wild-type | Codon 12 |
| 79a | Wild-type | Codon 12 |
| 80a | Wild-type | Codon 12 |
| 81a | Wild-type | Codon 12 |
| 82a | Wild-type | Codon 12 |
| 83 | Wild-type | Codon 12 |
| 84 | Wild-type | Codon 12 |
| 85 | Wild-type | Codon 12 |
| 86 | Wild-type | Codon 12 |
| 87 | Wild-type | Codon 12 |
| 88 | Wild-type | Codon 12 |
| 89 | Wild-type | Codon 12 |
| 90 | Wild-type | Codon 12 |
| 91 | Wild-type | Wild-type |
| 92 | Wild-type | Wild-type |
| 93 | Wild-type | Wild-type |
| 94 | Wild-type | Wild-type |
| 95 | Wild-type | Wild-type |
| 96 | Wild-type | Wild-type |
| 97 | Wild-type | Wild-type |
| 98 | Wild-type | Wild-type |
| 99 | Wild-type | Wild-type |
| 100 | Wild-type | Wild-type |
| 101 | Wild-type | Wild-type |
| 102 | Wild-type | Wild-type |
| 103 | Wild-type | Wild-type |
| 104 | Wild-type | - |
| 105 | Wild-type | Wild-type |
| 106 | Wild-type | Wild-type |
| 107 | Wild-type | Wild-type |
| 108 | Wild-type | Wild-type |
| 109 | Wild-type | - |
| 110 | Wild-type | Wild-type |
| 111 | Wild-type | Wild-type |
| 112 | Wild-type | Wild-type |
| 113 | Wild-type | Wild-type |
| 114 | Wild-type | Wild-type |
| 115 | Wild-type | Wild-type |
| 116 | Wild-type | Wild-type |
| 117 | Wild-type | Wild-type |
| 118 | Wild-type | Wild-type |
| 119 | Wild-type | - |
| 120 | Wild-type | Wild-type |
| 121 | Wild-type | Wild-type |
| 122 | Wild-type | |
| 123 | Wild-type | Wild-type |
| 124 | Wild-type | Wild-type |
| 125 | Wild-type | Wild-type |
| 126 | Wild-type | Wild-type |
| 127 | Wild-type | Wild-type |
| 128 | Wild-type | Wild-type |
| 129 | Wild-type | Wild-type |
| 130 | Wild-type | Wild-type |
| 131 | Wild-type | Wild-type |
| 132 | Wild-type | Wild-type |
| 133 | Wild-type | Wild-type |
| 134 | Wild-type | Wild-type |
| 135 | Wild-type | Wild-type |
| 136 | Wild-type | Wild-type |
| 137 | Wild-type | Wild-type |
| 138 | Wild-type | Wild-type |
| 139 | Wild-type | Wild-type |
| 140 | Wild-type | Wild-type |
| 141 | Wild-type | Wild-type |
| 142 | Wild-type | Wild-type |
| 143 | Wild-type | Wild-type |
| 144 | Wild-type | Wild-type |
| 145 | Wild-type | Wild-type |
| 146 | Wild-type | Wild-type |
| 147 | Wild-type | Wild-type |
| 148 | Wild-type | Wild-type |
| 149 | Wild-type | Wild-type |
| 150 | Wild-type | Wild-type |
| 151 | Wild-type | Wild-type |
| 152 | Wild-type | Wild-type |
| 153 | Wild-type | Wild-type |
| 154 | Wild-type | Wild-type |
| 155 | Wild-type | Wild-type |
| 156 | Wild-type | Wild-type |
| 157 | Wild-type | Wild-type |
| 158 | Wild-type | Wild-type |
| 159 | Wild-type | Wild-type |
| 160 | Wild-type | Wild-type |
| 161 | Wild-type | Wild-type |
| 162 | Wild-type | Wild-type |
| 163 | Wild-type | Wild-type |
| 164 | Wild-type | Wild-type |
| 165 | Wild-type | - |
| 166 | Wild-type | Wild-type |
| 167 | Wild-type | Wild-type |
| 168 | Wild-type | Wild-type |
| 169 | Wild-type | Wild-type |
| 170 | Wild-type | Wild-type |
| 171 | Wild-type | Wild-type |
| 172 | Wild-type | Wild-type |
| 173 | Wild-type | Wild-type |
| 174 | Wild-type | Wild-type |
| 175 | Wild-type | Wild-type |
| 176 | Wild-type | Wild-type |
| 177 | Wild-type | Wild-type |
| 178 | Wild-type | Wild-type |
| 179 | Wild-type | Wild-type |
| 180 | Wild-type | Wild-type |
| 181 | Wild-type | Wild-type |
| 182 | Wild-type | Wild-type |
| | Wild-type |
 | Table IIIComparison of KRAS mutation results
with methods other than QP or DS for samples that diverged between
the two methods. |
Table III
Comparison of KRAS mutation results
with methods other than QP or DS for samples that diverged between
the two methods.
| No. | DS | QProbe | Scorpion-ARMS |
|---|
| 64 | Wild-type | Codon 12 | p.G12D |
| 69 | Wild-type | Codon 12 | p.G12V |
| 72 | Wild-type | Codon 12 | p.G12V |
| 73 | Wild-type | Codon 12 | p.G12V |
| 80 | Wild-type | Codon 12 | p.G12D |
| 81 | Wild-type | Codon 12 | p.G12C |
| 82 | Wild-type | Codon 12 | p.G12D |
| 63 | Wild-type | Codon 12 | Wild-type |
| 65 | Wild-type | Codon 12 | Wild-type |
| 79 | Wild-type | Codon 12 | Wild-type |
| 46 | p.G12D | Wild-type | Wild-type |
| 60 | p.G13D | Wild-type | |
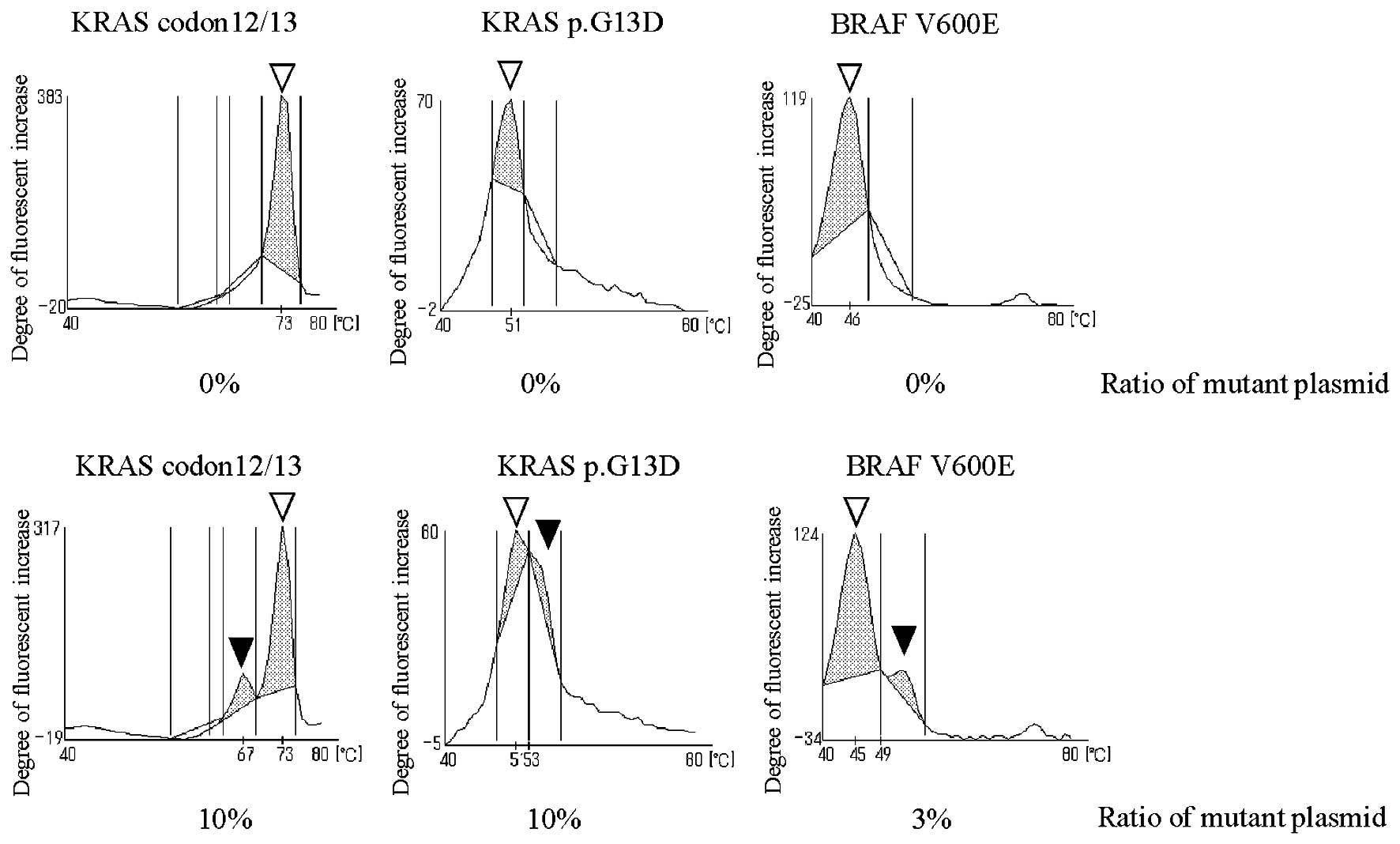
The QP method determines the existence of mutation
by the existence of peaks. In this system, clear peaks were found
in specimens with mutation plasmid content at 10% for KRAS codon
12/13 mutation, 10% for p.G13D and 3% for BRAF V600E mutation
(Fig. 1). A peak area can be
calculated by specifying the temperature range where the peak
appears (Table IV). In this
evaluation, cut-off values were set at a range where a clear
mutation peak can be obtained (KRAS codon 12/13: mt 10%, BRAF
V600E: mt 3%) in order to prevent false detection (Table V). Plasmid is being used for the
criteria of both the DS method and QP method. The results of the DS
method are considered to be positive when the mutation base
waveforms are confirmed by visual inspection in both sense and
antisense results. The detection sensitivity of KRAS codon 12/13
mutation using the DS method is estimated to be ~10%. From the
above, KRAS mutation detectability with QP method in this
evaluation using the actual sample is better than the DS method and
is equivalent to or better than Scorpion-ARMS method that have
relatively high sensitivity.
 | Table IVThe range of temperature for peak
area to detection of KRAS and BRAF mutation using the QP
method. |
Table IV
The range of temperature for peak
area to detection of KRAS and BRAF mutation using the QP
method.
| The range of
temperature for the peak area |
|---|
|
|
|---|
| KRAS codon
12/13 | KRAS p.G13D | BRAF V600E |
|---|
| wt peak area | 69–75°C | 47–52°C | 40–48°C |
| mt peak area 1 | 62–69°C | 52–57°C | 48–57°C |
| mt peak area 2 | 56–64°C | - | - |
 | Table VCriteria of KRAS and BRAF
mutation. |
Table V
Criteria of KRAS and BRAF
mutation.
| KRAS codon
12/13 |
|
Undeterminable | When reagent
reactivitya is ≤0.8 |
| With mutation | When mt peak area
1/wt peak area or mt peak area 2/wt peak area is ≥0.1b |
| No mutation | When mt peak area
1/wt peak area or mt peak area 2/wt peak area is <0.1 |
| KRAS p.G13D |
|
Undeterminable | When reagent
reactivity is ≤0.8 |
| With mutation | When mt peak
(~54°C) can be seen visually |
| No mutation | When mt peak
(~54°C) cannot be seen visually |
| BRAF V600E |
|
Undeterminable | When reagent
reactivity is ≤0.8 |
| With mutation | When mt peak area
1/wt peak area is ≤0.1 |
| No mutation | When mt peak area
1/wt peak area is <0.1 |
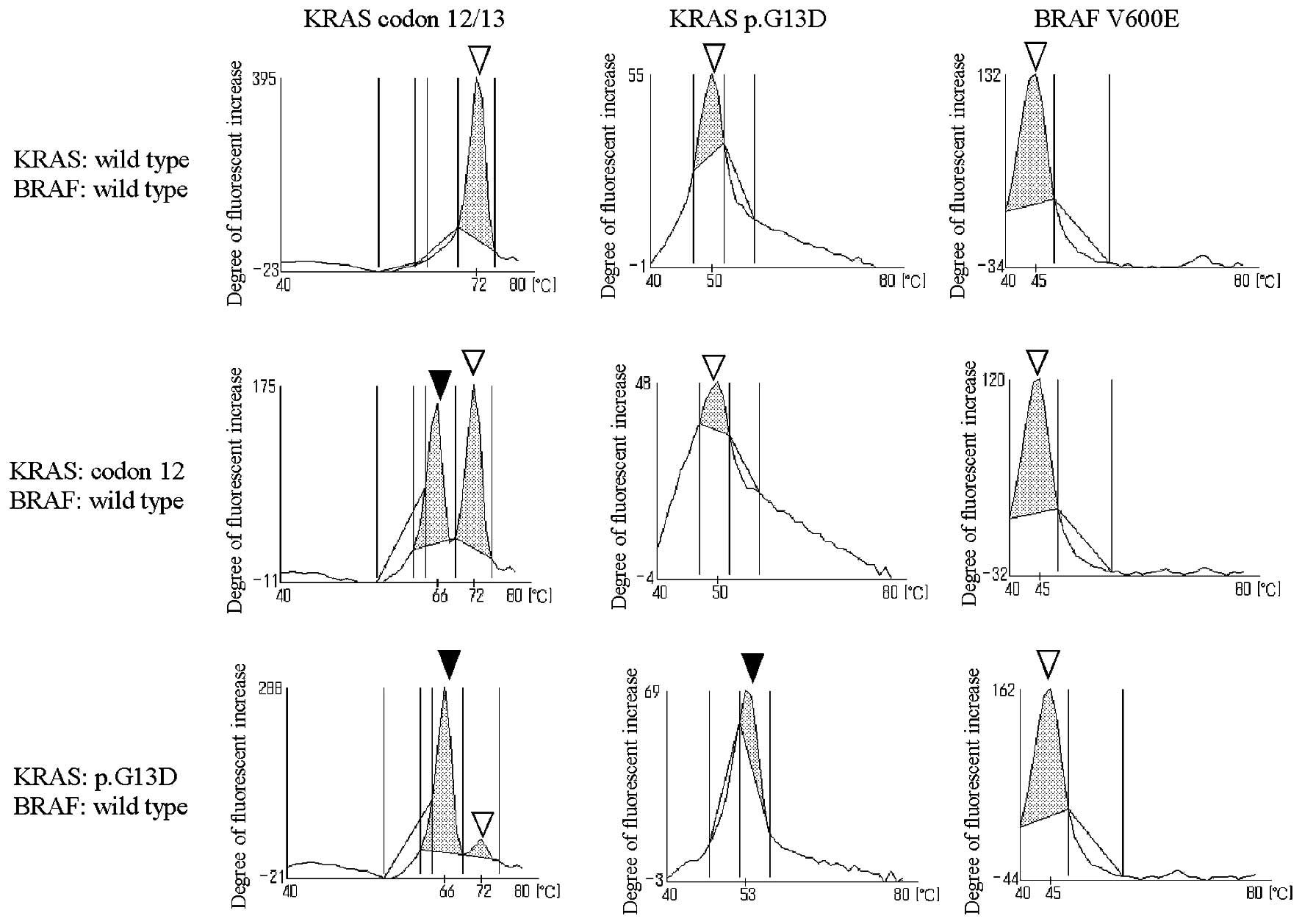
KRAS mutation can be detected directly
from specimen tissue without DNA extraction
Gene analyzer i-densy IS-5320 uses QP method and can
analyze genes from whole blood in full automation by using special
reagents. In this evaluation, a new protocol was evaluated with 32
biopsy specimens collected from colon cancer patients, which was
heated at 95°C by i-densy as pretreatment and used for gene
analysis. Biopsy specimens of 1-mm3 were used. KRAS
mutation in all of these specimens was analyzed from purified DNA
in advance by Luminex method. As a result of testing with i-densy,
good reaction was seen in 31 out of 32 cases in perfect match with
Luminex method (Table VI).
Specimens that gave results had clear peaks, and most of the mt
peaks were evidently higher than wt peaks (Fig. 2). False reaction was seen in one
case, and the possible cause of this was the large biopsy specimen
(~3 mm3) causing insufficient homogenization before heat
treatment. Since Multiplex reagent was used for this measurement,
BRAF V600E mutation was tested at the same time, and all 32 cases
gave negative results.
 | Table VIMeasurement results of KRAS codon1
2/13 mutation in biopsy samples without DNA extraction. |
Table VI
Measurement results of KRAS codon1
2/13 mutation in biopsy samples without DNA extraction.
| QProbe method | | |
|---|
|
| | |
|---|
| KRAS
mutationa | Wild-type | Codon 12 | p.G13D | Undeterminable | Concordance
ratec |
|---|
| Wild-type | 18 | 0 | 0 | 1 | 94.7% (100.0%) |
| Codon 12b | 0 | 10 | 0 | 0 | 100.0% |
| p.G13D | 0 | 0 | 3 | 0 | 100.0% |
| | | | | Total 96.9%
(100.0%) |
Discussion
In recent years, development of molecular target
drugs for cancer treatment is making progress, and gene test before
drug administration is becoming indispensable. Anti-EGFR antibody
is a drug targeting EGFR and is widely applied in the clinical
field. There have been many reports on the acquisition of drug
resistance when there is a mutation in KRAS codon 12/13 or BRAF
V600 that lie downstream of EGFR (17–20).
On the other hand, anti-EGFR antibodies may have drug efficacy for
p.G13D mutation among KRAS codon 12/13 mutation, so it will be
useful in the future to determine the existence of KRAS mutation by
distinguishing this mutation (10,11).
In this study, we have established a measurement system that can
detect the KRAS codon 12/13, p.G13D and BRAF V600E at the same
time. Compared to the system introduced in the previous studies
(14,15), the new system has improved
detection sensitivity and capability to identify p.G13D. Using QP
method, this system made it possible to detect these mutations in
high sensitivity compared to DS method based on nested PCR.
Moreover, the use of gene analyzer i-densy led to the successful
gene analysis from specimen tissue just with simple pre-treatment
without going through troublesome DNA extraction.
This system was compared with the conventional DS
method to check its accuracy, and divergence was seen in 33 out of
177 cases (Tables I and II). In particular, as many as 29
specimens became negative with DS method, but positive with the QP
method. Among 10 positive specimens with QP method that became
negative with DS method, 7 out of 10 became positive with
Scorpion-ARMS method, which suggest a high possibility that DS
method is overlooking KRAS mutation (Table III). Although QP and DS methods
were equivalent in mutation detection test using plasmid, many
specimens with inconsistent results were found in this study, which
leads to the following two causes to be considered. i) Difference
in copy-count sensitivity: QP method can amplify and detect plasmid
with only two copies, which means the copy-count sensitivity is
extremely high (data not shown). Thus, this method can measure DNA
with mutation even if the absolute quantity is very small. For
example, even if the content rate of DNA mutation is >10%,
pathological tissue specimen of poor quality will not be detected
with DS method if PCR cannot be amplified up to the volume required
to detect DNA quantity. Even if the results are equivalent in
mutation detectivity test with sufficient copies of plasmid (8,000
copies), the results may be inconsistent when testing actual
samples due to the possible shortage of copy-count. ii) Wild-type
inhibition system of QP method: KRAS measurement series of QP
method have the probes designed to be in perfect match with
wild-type and mismatch with mutant type. Wild inhibition system can
intentionally amplify mutant type sequence by adjusting the probe
sequence and temperature conditions to inhibit amplification of
wild-type sequence during PCR. The mutation detection of this
system improves under the condition of low enzyme activity where
PCR efficiency decreases. Unlike plasmid, the actual samples
contain foreign substances such as protein, which reduce the PCR
efficiency. Moreover, when samples contain DNA in advanced
fragmentation stage due to formalin fixation, fragmented DNA is
assumed to cause PCR inhibition as a foreign substance. Thus PCR
efficiency is expected to drop compared to tests using plasmid,
resulting in a possibility of mutation detection to be better than
10%, which is the expected sensitivity of this system. Furthermore,
2 specimens that were positive with DS method became negative with
the other two methods. Therefore, contamination from DS method
technique and erroneous decisions were called into question.
Although DS method is a proven method with stable results, there
are a few risks due to the manual work involved, leading to
contamination or mix-up of specimens. Moreover, the decision is
made by visual inspection, so there could be a possibility of
mistaking a noise waveform for a small mutation waveform. On the
other hand, QP method and Scorpion-ARMS method have clear criteria
and less manual work involved before the measurement, reducing such
risk. These results indicate that the system we have developed
offers highly sensitive and accurate gene analysis using DS method
with sensitivity equivalent to or better than Scorpion-ARMS
method.
BRAF V600E mutation was measured only with the QP
method, and the frequency of occurrence was 3.9% in this study. The
lower detection limit of BRAF V600E mutation was 3%, adjusting
mutant plasmid to this level. This measurement system clearly had
higher sensitivity compared to the DS method. Moreover, this test
result matched with the general report that the frequency of BRAF
mutation occurrence in Japanese colon cancer patients is ~4–5%
(9). As of now, definite treatment
is not established for BRAF mutation-positive cases, and the
primary goal of the measurement is to avoid unnecessary
administration of anti-EGFR antibody drug. Vemurafenib is an
effective treatment drug for malignant melanoma that is BRAF
mutation-positive; however, it is known to have little effect on
BRAF mutation-positive colon cancer. Activation of EGFR is known to
be a mechanism of vemurafenib-resistance (21). Therefore, there is a high degree of
expectation in combination therapy of BRAF inhibitor and anti-EGFR
antigen, etc. for BRAF mutation-positive colon cancer, and the
outcome of future clinical trials is eagerly expected. As indicated
above, studies for new treatment methods are advancing. They will
gain recognition in the future as screening test items before drug
administration for colon cancer treatment along with BRAF and
KRAS.
Purified DNA is typically used for gene analysis;
however, we have tried eliminating DNA purification process to
perform gene analysis with simple pre-treatment. Thirty-two frozen
biopsy specimens were measured with simple pre-treatment without
extracting DNA. All 31 cases that gave results fully matched with
the results of Luminex method (Table
VI). Since special reagent for i-densy used for the analysis
was developed for the measurement from whole blood, it is not prone
to being affected by foreign substances such as protein. There have
been many reports on gaining good test results from whole blood
(22–25). The special quality of this reagent
was most likely the major reason for good results even without DNA
purification from sample tissue. In addition, all 12 specimens
measured directly from the tissue that became KRAS
mutation-positive could be clearly identified as positive because
the mutation peaks were equivalent to or larger than that of
wild-type. This is likely to be caused by the high content rate of
cancer cells in biopsy specimen used for measurement because the
tissues are collected with endoscope directly from cancer tissues.
There was one PCR failure out of 32 cases; however, this is
presumed to be caused by excess sample volume. Although the reagent
composition is robust over foreign substances, sample volume must
be controlled strictly in order to secure good results. When using
formalin fixation tissues, it is known that tissue amount >1
mm3 will likely cause reaction failure, and all
reactions with tissue amount >2 mm3 will result in a
failure. Tissue specimens of ≤1 mm would be sufficient for good
analysis results. When the sample volume exceeds this level, it is
desirable to adjust the sample volume by homogenizing before use.
Although existence of cancer cells was not examined by pathologic
testing for this study, it is desirable to check for cancer cells
for instance by hematoxylin and eosin stain before genetic testing
in order to improve mutation detectivity. However, obtaining a
large tissue from an elderly or patient with metastatic cancer
would be difficult. The fact that genetic testing is possible by
using micro amount of specimen collected with an endoscope is one
of the merits of this method. Since the sample volume required for
the genetic testing is very small, sufficient amount of specimens
can be sent for pathologic testing. Moreover, deparaffinization of
paraffin-embedded tissues (FFPE) allows measurements to be
performed with the same protocol as frozen tissues, and liquid
cytological specimens can be used for measurement, making this
method widely applicable to examinations of micro amount of
specimens. This pre-treatment protocol allows eliminating DNA
purification process that typically takes place and performing
simple, quick and accurate gene analysis. By using this protocol,
complicated operations, labor time and reagent costs can be
reduced.
In order to predict the drug efficacy of molecular
target medicine targeting EGFR, it is important to check the
mutation of KRAS and BRAF genes, which are the signals that lie
downstream of EGFR. In addition, p.G13D among KRAS codon 12/13
mutations may likely have a different drug efficacy compared to
other mutations, thus further clinical validation would be
desirable. Today, there is not enough evidence supporting the drug
efficacy of p.G13D, which is why it is not actively considered at
the time of drug administration. In a case of KRAS p.G13D-positive,
FOLFOX or FOLFIRI ± bevacizumab treatment is administered in 1st
and 2nd lines, and chemotherapy such as regorafenib or TAS-102
monotherapy are considered for the 3rd and 4th lines (26,27).
However, when the treatment reaches a stage beyond that point,
anti-EGFR antibodies may be selected, thus it is helpful to
identify p.G13D beforehand.
Measurement using i-densy with QP method allows
quick and simple detection of gene mutations simultaneously from a
single reaction system, with sensitivity better than DS method.
Furthermore, innovative and simplified pre-treatment protocol
reduces the operation process and realizes simplified and accurate
gene analysis that can be performed by any user. By changing PCR
primer and QProbe, this versatile instrument can be used to detect
other gene mutations that may affect the efficacy of molecular
target treatment. Recently, NRAS has been reported as a
drug-resistant mutation similar to KRAS, which detecting system
needs to be established soon (28–30).
We sincerely hope that this study will drive the gene analysis to
spread to further clinical sites and to contribute to personalized
medicine in the future.
References
|
1
|
Borden EC and Raghavan D: Personalizing
medicine for cancer: The next decade. Nat Rev Drug Discov.
9:343–344. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Evans WE and Relling MV: Pharmacogenomics:
Translating functional genomics into rational therapeutics.
Science. 286:487–491. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ong FS, Das K, Wang J, Vakil H, Kuo JZ,
Blackwell WL, Lim SW, Goodarzi MO, Bernstein KE, Rotter JI, et al:
Personalized medicine and pharmacogenetic biomarkers: Progress in
molecular oncology testing. Expert Rev Mol Diagn. 12:593–602. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gazdar AF: Epidermal growth factor
receptor inhibition in lung cancer: The evolving role of
individualized therapy. Cancer Metastasis Rev. 29:37–48. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lièvre A, Bachet JB, Boige V, Cayre A, Le
Corre D, Buc E, Ychou M, Bouché O, Landi B, Louvet C, et al: KRAS
mutations as an independent prognostic factor in patients with
advanced colorectal cancer treated with cetuximab. J Clin Oncol.
26:374–379. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Siena S, Sartore-Bianchi A, Di
Nicolantonio F, Balfour J and Bardelli A: Biomarkers predicting
clinical outcome of epidermal growth factor receptor-targeted
therapy in metastatic colorectal cancer. J Natl Cancer Inst.
101:1308–1324. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Karapetis CS, Khambata-Ford S, Jonker DJ,
O’Callaghan CJ, Tu D, Tebbutt NC, Simes RJ, Chalchal H, Shapiro JD,
Robitaille S, et al: K-ras mutations and benefit from cetuximab in
advanced colorectal cancer. N Engl J Med. 359:1757–1765. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Baines AT, Xu D and Der CJ: Inhibition of
Ras for cancer treatment: The search continues. Future Med Chem.
3:1787–1808. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yokota T: Are KRAS/BRAF mutations potent
prognostic and/or predictive biomarkers in colorectal cancers?
Anticancer Agents Med Chem. 12:163–171. 2012. View Article : Google Scholar :
|
|
10
|
De Roock W, Jonker DJ, Di Nicolantonio F,
Sartore-Bianchi A, Tu D, Siena S, Lamba S, Arena S, Frattini M,
Piessevaux H, et al: Association of KRAS p.G13D mutation with
outcome in patients with chemotherapy-refractory metastatic
colorectal cancer treated with cetuximab. JAMA. 304:1812–1820.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mao C, Huang YF, Yang ZY, Zheng DY, Chen
JZ and Tang JL: KRAS p.G13D mutation and codon 12 mutations are not
created equal in predicting clinical outcomes of cetuximab in
metastatic colorectal cancer: A systematic review and
meta-analysis. Cancer. 119:714–721. 2013. View Article : Google Scholar
|
|
12
|
Yokota T, Ura T, Shibata N, Takahari D,
Shitara K, Nomura M, Kondo C, Mizota A, Utsunomiya S, Muro K, et
al: BRAF mutation is a powerful prognostic factor in advanced and
recurrent colorectal cancer. Br J Cancer. 104:856–862. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kurata S, Kanagawa T, Yamada K, Torimura
M, Yokomaku T, Kamagata Y and Kurane R: Fluorescent quenching-based
quantitative detection of specific DNA/RNA using a BODIPY((R))
FL-labeled probe or primer. Nucleic Acids Res. 29:E342001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ureshino N, Sueoka-Aragane N, Nakamura T,
Sato A, Komiya K, Iwanaga K, Mitsuoka M, Takeda Y, Hayashi S,
Sueoka E, et al: A fully integrated, automated and rapid detection
system for KRAS mutations. Oncol Rep. 26:609–613. 2011.PubMed/NCBI
|
|
15
|
Akagi K and Arai Y: Tm analysis method
using a quenching probe is a simple and rapid way to simultaneously
detect KRAS and BRAF mutations. Rinsho Byori. 59:757–762. 2011.(In
Japanese). PubMed/NCBI
|
|
16
|
Suzuki S, Komori M, Hirai M, Ureshino N
and Kimura S: Development of a novel, fully-automated genotyping
system: Principle and applications. Sens Basel. 12:16614–16627.
2012. View Article : Google Scholar
|
|
17
|
Jiang Y, Mackley H, Cheng H and Ajani JA:
Use of K-Ras as a predictive biomarker for selecting anti-EGF
receptor/pathway treatment. Biomarkers Med. 4:535–541. 2010.
View Article : Google Scholar
|
|
18
|
Benvenuti S, Sartore-Bianchi A, Di
Nicolantonio F, Zanon C, Moroni M, Veronese S, Siena S and Bardelli
A: Oncogenic activation of the RAS/RAF signaling pathway impairs
the response of metastatic colorectal cancers to anti-epidermal
growth factor receptor antibody therapies. Cancer Res.
67:2643–2648. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Heinemann V, Stintzing S, Kirchner T,
Boeck S and Jung A: Clinical relevance of EGFR- and KRAS-status in
colorectal cancer patients treated with monoclonal antibodies
directed against the EGFR. Cancer Treat Rev. 35:262–271. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Berg M and Soreide K: EGFR and downstream
genetic alterations in KRAS/BRAF and PI3K/AKT pathways in
colorectal cancer: Implications for targeted therapy. Discov Med.
14:207–214. 2012.PubMed/NCBI
|
|
21
|
Prahallad A, Sun C, Huang S, Di
Nicolantonio F, Salazar R, Zecchin D, Beijersbergen RL, Bardelli A
and Bernards R: Unresponsiveness of colon cancer to BRAF(V600E)
inhibition through feedback activation of EGFR. Nature.
483:100–103. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tanaka R, Kimura S, Ashihara E, Yoshimura
M, Takahashi N, Wakita H, Itoh K, Nishiwaki K, Suzuki K, Nagao R,
et al: Rapid automated detection of ABL kinase domain mutations in
imatinib-resistant patients. Cancer Lett. 312:228–234. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ureshino N, Aragane N, Nakamura T, Ide M,
Mochinaga S, Fukushima N, Hayashi S, Sueoka E and Kimura S: A fully
integrated and automated detection system for single nucleotide
polymorphisms of UGT1A1 and CYP2C19. Oncol Res. 19:111–114. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tanaka R, Kuroda J, Stevenson W, Ashihara
E, Ishikawa T, Taki T, Kobayashi Y, Kamitsuji Y, Kawata E, Takeuchi
M, et al: Fully automated and super-rapid system for the detection
of JAK2V617F mutation. Leuk Res. 32:1462–1467. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Takahashi H, Mizuta T, Oeda S, Isoda H,
Nakashita S, Kawaguchi Y, Izumi N, Hirai M, Kurose K, Iwane S, et
al: An automated rapid detection system using the quenching probe
method for detecting interleukin 28B and inosine triphosphatase
single nucleotide polymorphisms in chronic hepatitis C. J Viral
Hepat. 20:e124–e126. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yoshino T, Mizunuma N, Yamazaki K, Nishina
T, Komatsu Y, Baba H, Tsuji A, Yamaguchi K, Muro K, Sugimoto N, et
al: TAS-102 monotherapy for pretreated metastatic colorectal
cancer: A double-blind, randomised, placebo-controlled phase 2
trial. Lancet Oncol. 13:993–1001. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Grothey A, van Cutsem E, Sobrero A, Siena
S, Falcone A, Ychou M, Humblet Y, Bouché O, Mineur L, Barone C, et
al; CORRECT Study Group. Regorafenib monotherapy for previously
treated metastatic colorectal cancer (CORRECT): An international,
multicentre, randomised, placebo-controlled, phase 3 trial. Lancet.
381:303–312. 2013. View Article : Google Scholar
|
|
28
|
De Stefano A and Carlomagno C: Beyond
KRAS: Predictive factors of the efficacy of anti-EGFR monoclonal
antibodies in the treatment of metastatic colorectal cancer. World
J Gastroenterol. 20:9732–9743. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bando H, Yoshino T, Shinozaki E, Nishina
T, Yamazaki K, Yamaguchi K, Yuki S, Kajiura S, Fujii S, Yamanaka T,
et al: Simultaneous identification of 36 mutations in KRAS codons
61 and 146, BRAF, NRAS, and PIK3CA in a single reaction by
multiplex assay kit. BMC Cancer. 13:4052013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Patel GS and Karapetis CS: Personalized
treatment for advanced colorectal cancer: KRAS and beyond. Cancer
Manag Res. 5:387–400. 2013.PubMed/NCBI
|