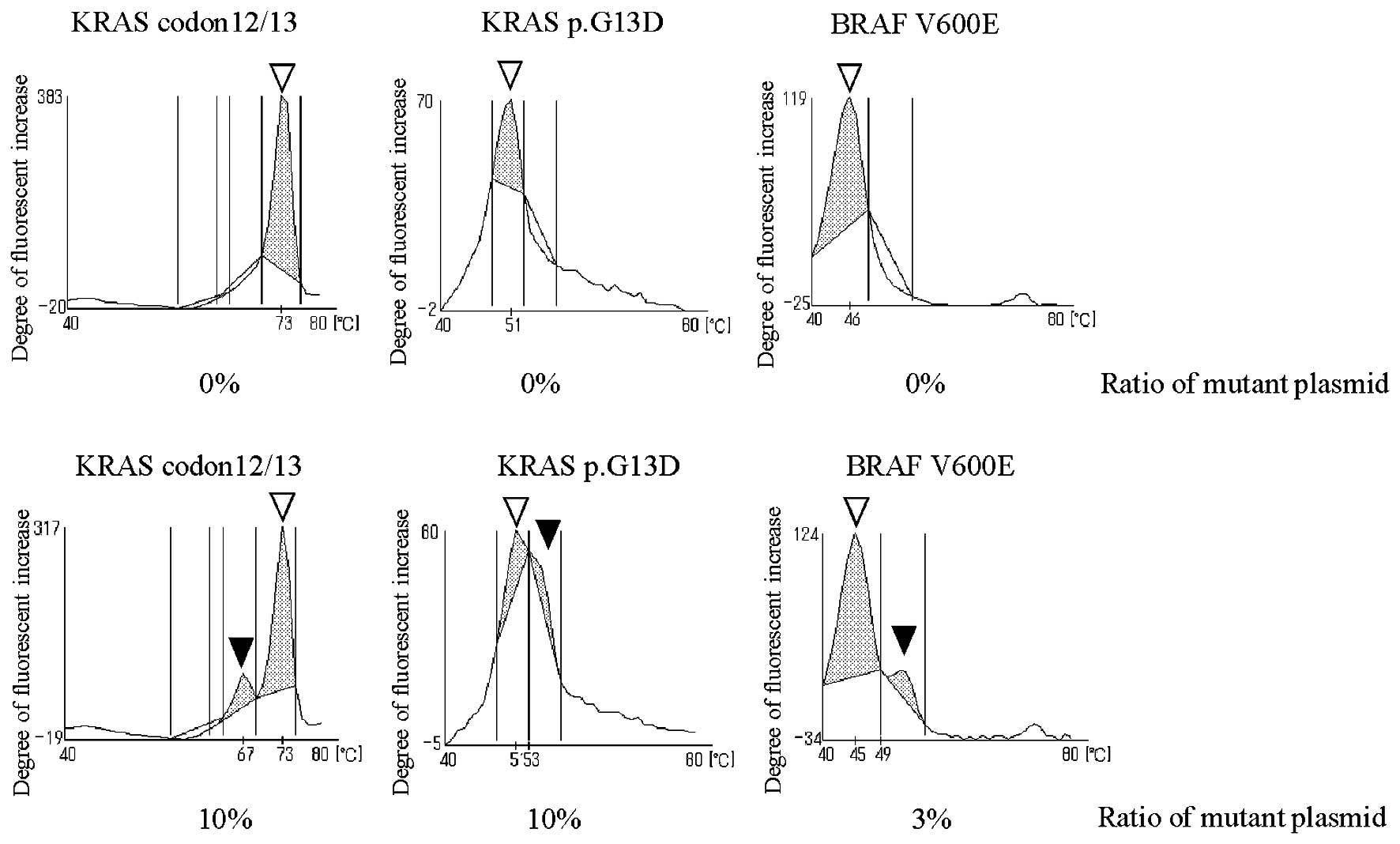
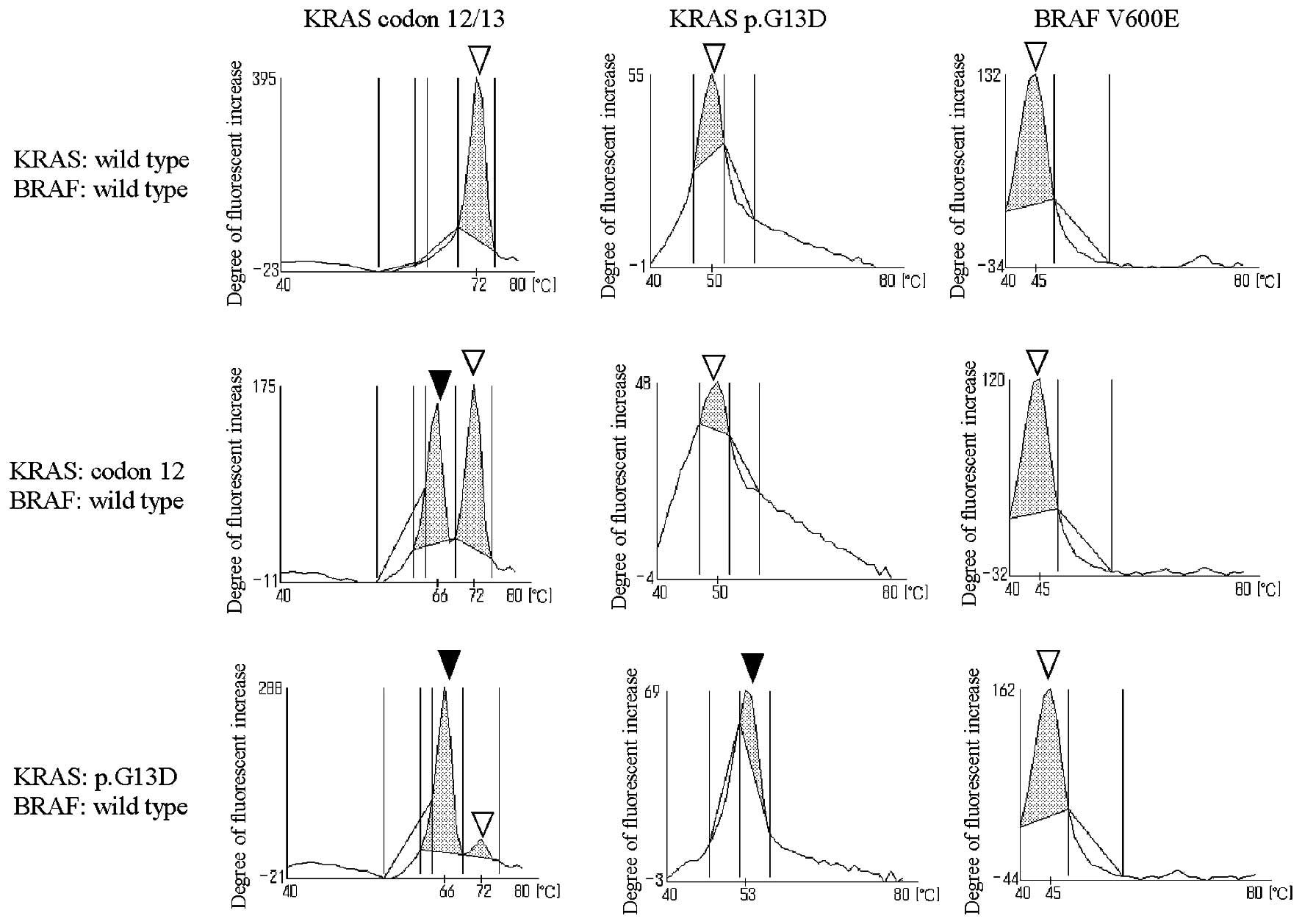
|
1
|
Borden EC and Raghavan D: Personalizing
medicine for cancer: The next decade. Nat Rev Drug Discov.
9:343–344. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Evans WE and Relling MV: Pharmacogenomics:
Translating functional genomics into rational therapeutics.
Science. 286:487–491. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ong FS, Das K, Wang J, Vakil H, Kuo JZ,
Blackwell WL, Lim SW, Goodarzi MO, Bernstein KE, Rotter JI, et al:
Personalized medicine and pharmacogenetic biomarkers: Progress in
molecular oncology testing. Expert Rev Mol Diagn. 12:593–602. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gazdar AF: Epidermal growth factor
receptor inhibition in lung cancer: The evolving role of
individualized therapy. Cancer Metastasis Rev. 29:37–48. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lièvre A, Bachet JB, Boige V, Cayre A, Le
Corre D, Buc E, Ychou M, Bouché O, Landi B, Louvet C, et al: KRAS
mutations as an independent prognostic factor in patients with
advanced colorectal cancer treated with cetuximab. J Clin Oncol.
26:374–379. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Siena S, Sartore-Bianchi A, Di
Nicolantonio F, Balfour J and Bardelli A: Biomarkers predicting
clinical outcome of epidermal growth factor receptor-targeted
therapy in metastatic colorectal cancer. J Natl Cancer Inst.
101:1308–1324. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Karapetis CS, Khambata-Ford S, Jonker DJ,
O’Callaghan CJ, Tu D, Tebbutt NC, Simes RJ, Chalchal H, Shapiro JD,
Robitaille S, et al: K-ras mutations and benefit from cetuximab in
advanced colorectal cancer. N Engl J Med. 359:1757–1765. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Baines AT, Xu D and Der CJ: Inhibition of
Ras for cancer treatment: The search continues. Future Med Chem.
3:1787–1808. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yokota T: Are KRAS/BRAF mutations potent
prognostic and/or predictive biomarkers in colorectal cancers?
Anticancer Agents Med Chem. 12:163–171. 2012. View Article : Google Scholar :
|
|
10
|
De Roock W, Jonker DJ, Di Nicolantonio F,
Sartore-Bianchi A, Tu D, Siena S, Lamba S, Arena S, Frattini M,
Piessevaux H, et al: Association of KRAS p.G13D mutation with
outcome in patients with chemotherapy-refractory metastatic
colorectal cancer treated with cetuximab. JAMA. 304:1812–1820.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mao C, Huang YF, Yang ZY, Zheng DY, Chen
JZ and Tang JL: KRAS p.G13D mutation and codon 12 mutations are not
created equal in predicting clinical outcomes of cetuximab in
metastatic colorectal cancer: A systematic review and
meta-analysis. Cancer. 119:714–721. 2013. View Article : Google Scholar
|
|
12
|
Yokota T, Ura T, Shibata N, Takahari D,
Shitara K, Nomura M, Kondo C, Mizota A, Utsunomiya S, Muro K, et
al: BRAF mutation is a powerful prognostic factor in advanced and
recurrent colorectal cancer. Br J Cancer. 104:856–862. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kurata S, Kanagawa T, Yamada K, Torimura
M, Yokomaku T, Kamagata Y and Kurane R: Fluorescent quenching-based
quantitative detection of specific DNA/RNA using a BODIPY((R))
FL-labeled probe or primer. Nucleic Acids Res. 29:E342001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ureshino N, Sueoka-Aragane N, Nakamura T,
Sato A, Komiya K, Iwanaga K, Mitsuoka M, Takeda Y, Hayashi S,
Sueoka E, et al: A fully integrated, automated and rapid detection
system for KRAS mutations. Oncol Rep. 26:609–613. 2011.PubMed/NCBI
|
|
15
|
Akagi K and Arai Y: Tm analysis method
using a quenching probe is a simple and rapid way to simultaneously
detect KRAS and BRAF mutations. Rinsho Byori. 59:757–762. 2011.(In
Japanese). PubMed/NCBI
|
|
16
|
Suzuki S, Komori M, Hirai M, Ureshino N
and Kimura S: Development of a novel, fully-automated genotyping
system: Principle and applications. Sens Basel. 12:16614–16627.
2012. View Article : Google Scholar
|
|
17
|
Jiang Y, Mackley H, Cheng H and Ajani JA:
Use of K-Ras as a predictive biomarker for selecting anti-EGF
receptor/pathway treatment. Biomarkers Med. 4:535–541. 2010.
View Article : Google Scholar
|
|
18
|
Benvenuti S, Sartore-Bianchi A, Di
Nicolantonio F, Zanon C, Moroni M, Veronese S, Siena S and Bardelli
A: Oncogenic activation of the RAS/RAF signaling pathway impairs
the response of metastatic colorectal cancers to anti-epidermal
growth factor receptor antibody therapies. Cancer Res.
67:2643–2648. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Heinemann V, Stintzing S, Kirchner T,
Boeck S and Jung A: Clinical relevance of EGFR- and KRAS-status in
colorectal cancer patients treated with monoclonal antibodies
directed against the EGFR. Cancer Treat Rev. 35:262–271. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Berg M and Soreide K: EGFR and downstream
genetic alterations in KRAS/BRAF and PI3K/AKT pathways in
colorectal cancer: Implications for targeted therapy. Discov Med.
14:207–214. 2012.PubMed/NCBI
|
|
21
|
Prahallad A, Sun C, Huang S, Di
Nicolantonio F, Salazar R, Zecchin D, Beijersbergen RL, Bardelli A
and Bernards R: Unresponsiveness of colon cancer to BRAF(V600E)
inhibition through feedback activation of EGFR. Nature.
483:100–103. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tanaka R, Kimura S, Ashihara E, Yoshimura
M, Takahashi N, Wakita H, Itoh K, Nishiwaki K, Suzuki K, Nagao R,
et al: Rapid automated detection of ABL kinase domain mutations in
imatinib-resistant patients. Cancer Lett. 312:228–234. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ureshino N, Aragane N, Nakamura T, Ide M,
Mochinaga S, Fukushima N, Hayashi S, Sueoka E and Kimura S: A fully
integrated and automated detection system for single nucleotide
polymorphisms of UGT1A1 and CYP2C19. Oncol Res. 19:111–114. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tanaka R, Kuroda J, Stevenson W, Ashihara
E, Ishikawa T, Taki T, Kobayashi Y, Kamitsuji Y, Kawata E, Takeuchi
M, et al: Fully automated and super-rapid system for the detection
of JAK2V617F mutation. Leuk Res. 32:1462–1467. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Takahashi H, Mizuta T, Oeda S, Isoda H,
Nakashita S, Kawaguchi Y, Izumi N, Hirai M, Kurose K, Iwane S, et
al: An automated rapid detection system using the quenching probe
method for detecting interleukin 28B and inosine triphosphatase
single nucleotide polymorphisms in chronic hepatitis C. J Viral
Hepat. 20:e124–e126. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yoshino T, Mizunuma N, Yamazaki K, Nishina
T, Komatsu Y, Baba H, Tsuji A, Yamaguchi K, Muro K, Sugimoto N, et
al: TAS-102 monotherapy for pretreated metastatic colorectal
cancer: A double-blind, randomised, placebo-controlled phase 2
trial. Lancet Oncol. 13:993–1001. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Grothey A, van Cutsem E, Sobrero A, Siena
S, Falcone A, Ychou M, Humblet Y, Bouché O, Mineur L, Barone C, et
al; CORRECT Study Group. Regorafenib monotherapy for previously
treated metastatic colorectal cancer (CORRECT): An international,
multicentre, randomised, placebo-controlled, phase 3 trial. Lancet.
381:303–312. 2013. View Article : Google Scholar
|
|
28
|
De Stefano A and Carlomagno C: Beyond
KRAS: Predictive factors of the efficacy of anti-EGFR monoclonal
antibodies in the treatment of metastatic colorectal cancer. World
J Gastroenterol. 20:9732–9743. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bando H, Yoshino T, Shinozaki E, Nishina
T, Yamazaki K, Yamaguchi K, Yuki S, Kajiura S, Fujii S, Yamanaka T,
et al: Simultaneous identification of 36 mutations in KRAS codons
61 and 146, BRAF, NRAS, and PIK3CA in a single reaction by
multiplex assay kit. BMC Cancer. 13:4052013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Patel GS and Karapetis CS: Personalized
treatment for advanced colorectal cancer: KRAS and beyond. Cancer
Manag Res. 5:387–400. 2013.PubMed/NCBI
|