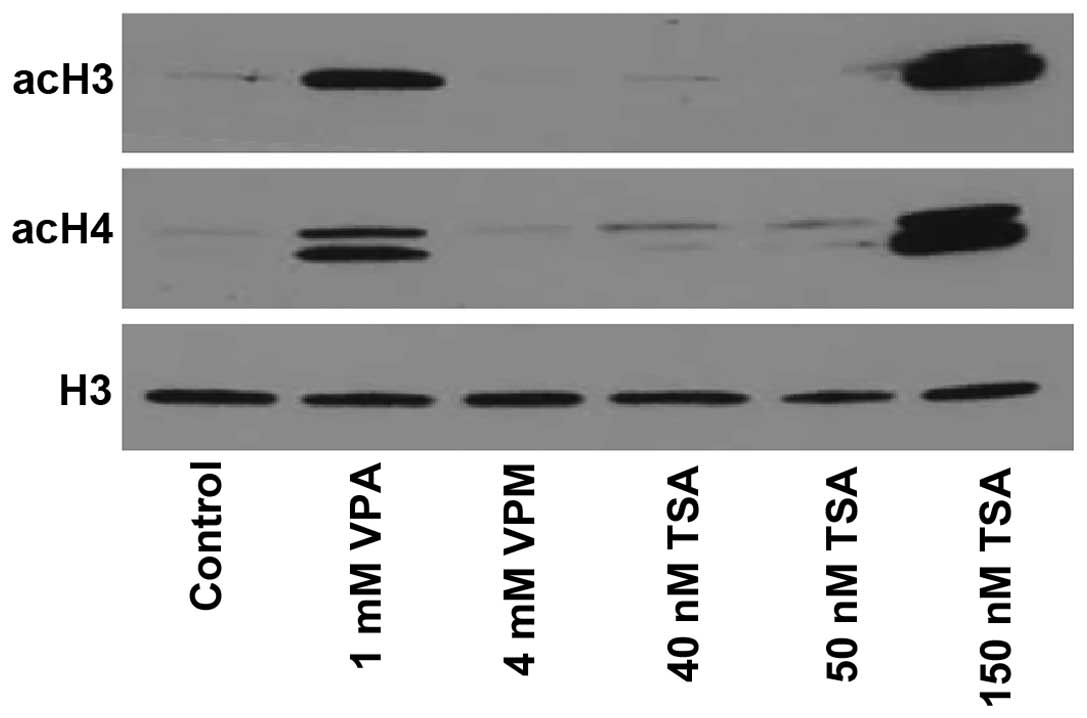
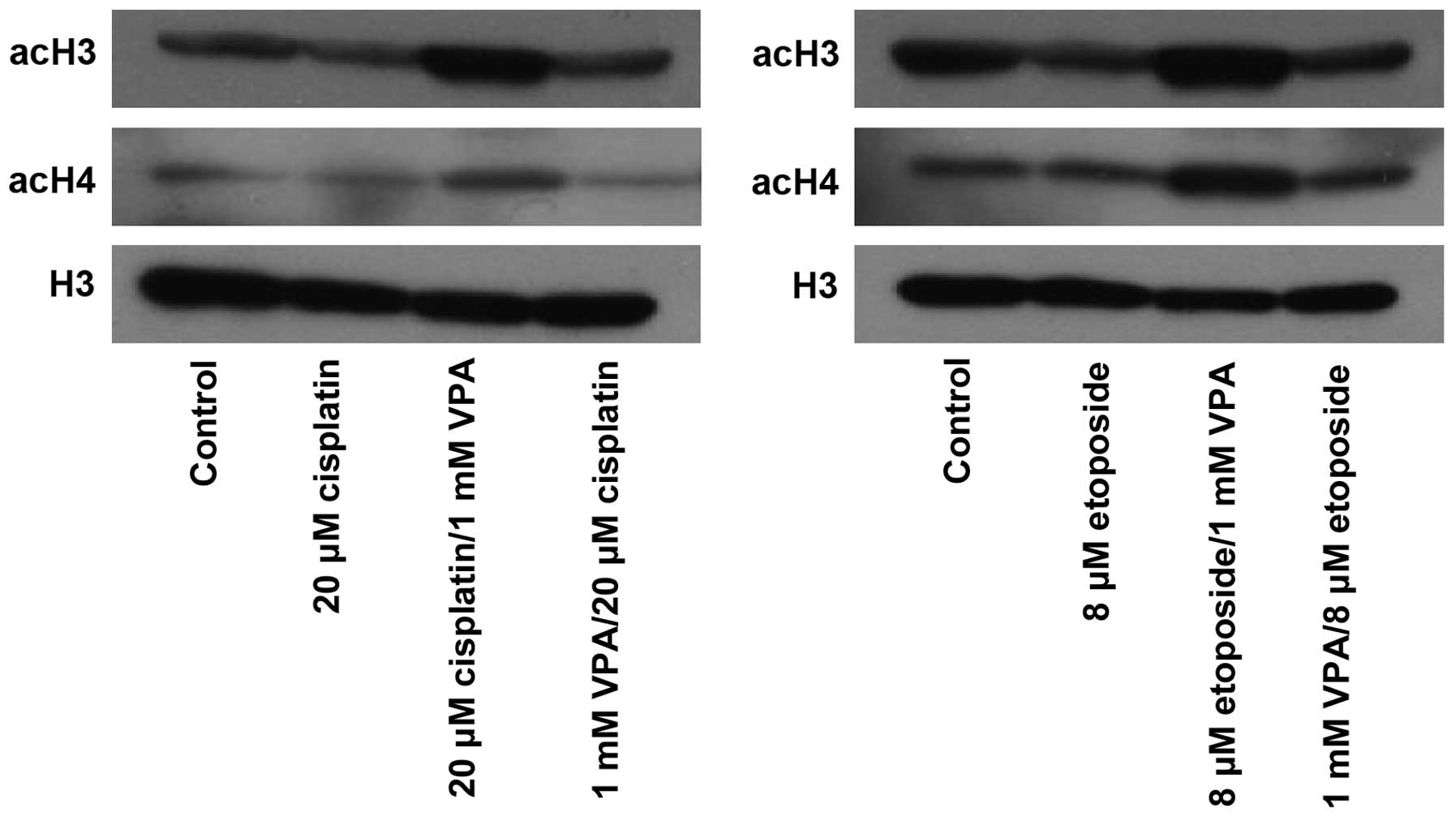
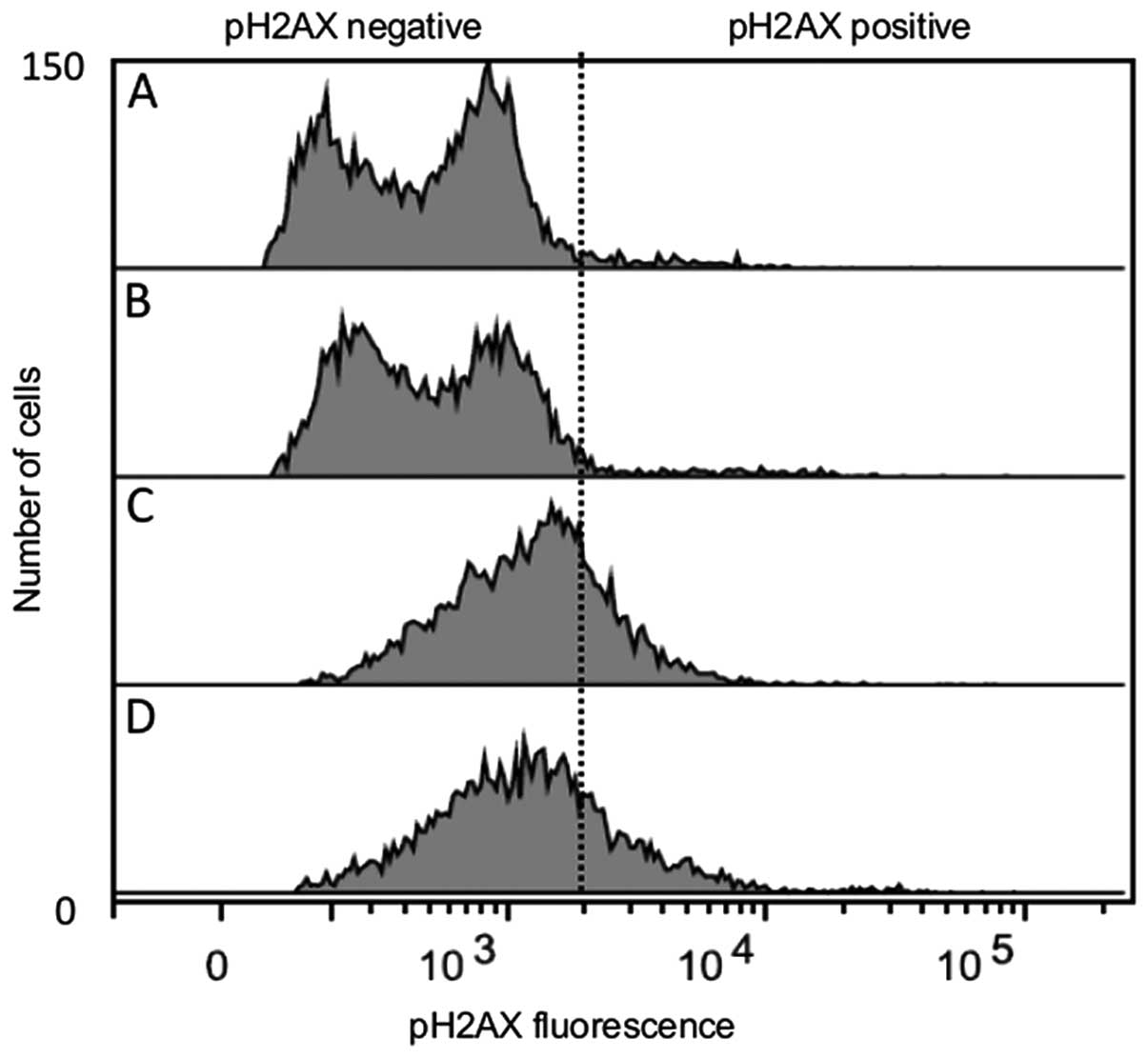
|
1
|
Brodeur GM: Neuroblastoma: Biological
insights into a clinical enigma. Nat Rev Cancer. 3:203–216. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Schwab M, Westermann F, Hero B and
Berthold F: Neuroblastoma: Biology and molecular and chromosomal
pathology. Lancet Oncol. 4:472–480. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Maris JM, Hogarty MD, Bagatell R and Cohn
SL: Neuroblastoma. Lancet. 369:2106–2120. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Furchert SE, Lanvers-Kaminsky C, Juürgens
H, Jung M, Loidl A and Frühwald MC: Inhibitors of histone
deacetylases as potential therapeutic tools for high-risk embryonal
tumors of the nervous system of childhood. Int J Cancer.
120:1787–1794. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Decock A, Ongenaert M, Vandesompele J and
Speleman F: Neuroblastoma epigenetics: From candidate gene
approaches to genome-wide screenings. Epigenetics. 6:962–970. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Santini V, Gozzini A and Ferrari G:
Histone deacetylase inhibitors: Molecular and biological activity
as a premise to clinical application. Curr Drug Metab. 8:383–393.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Portela A and Esteller M: Epigenetic
modifications and human disease. Nat Biotechnol. 28:1057–1068.
2010. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kouzarides T: Chromatin modifications and
their function. Cell. 128:693–705. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Glozak MA, Sengupta N, Zhang X and Seto E:
Acetylation and deacetylation of non-histone proteins. Gene.
363:15–23. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Stiborová M, Eckschlager T, Poljaková J,
Hraběta J, Adam V, Kizek R and Frei E: The synergistic effects of
DNA-targeted chemotherapeutics and histone deacetylase inhibitors
as therapeutic strategies for cancer treatment. Curr Med Chem.
19:4218–4238. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Atmaca A, Al-Batran SE, Maurer A, Neumann
A, Heinzel T, Hentsch B, Schwarz SE, Hövelmann S, Göttlicher M,
Knuth A, et al: Valproic acid (VPA) in patients with refractory
advanced cancer: a dose escalating phase I clinical trial. Br J
Cancer. 97:177–182. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Munster P, Marchion D, Bicaku E, Lacevic
M, Kim J, Centeno B, Daud A, Neuger A, Minton S and Sullivan D:
Clinical and biological effects of valproic acid as a histone
deacetylase inhibitor on tumor and surrogate tissues: Phase I/II
trial of valproic acid and epirubicin/FEC. Clin Cancer Res.
15:2488–2496. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rocca A, Minucci S, Tosti G, Croci D,
Contegno F, Ballarini M, Nolè F, Munzone E, Salmaggi A, Goldhirsch
A, et al: A phase I–II study of the histone deacetylase inhibitor
valproic acid plus chemoimmunotherapy in patients with advanced
melanoma. Br J Cancer. 100:28–36. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Vandermeers F, Hubert P, Delvenne P,
Mascaux C, Grigoriu B, Burny A, Scherpereel A and Willems L:
Valproate, in combination with pemetrexed and cisplatin, provides
additional efficacy to the treatment of malignant mesothelioma.
Clin Cancer Res. 15:2818–2828. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kim MS, Blake M, Baek JH, Kohlhagen G,
Pommier Y and Carrier F: Inhibition of histone deacetylase
increases cytotoxicity to anticancer drugs targeting DNA. Cancer
Res. 63:7291–7300. 2003.PubMed/NCBI
|
|
16
|
Munshi A, Kurland JF, Nishikawa T, Tanaka
T, Hobbs ML, Tucker SL, Ismail S, Stevens C and Meyn RE: Histone
deacetylase inhibitors radiosensitize human melanoma cells by
suppressing DNA repair activity. Clin Cancer Res. 11:4912–4922.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Das CM, Zage PE, Taylor P, Aguilera D,
Wolff JE, Lee D and Gopalakrishnan V: Chromatin remodelling at the
topoisomerase II-beta promoter is associated with enhanced
sensitivity to etoposide in human neuroblastoma cell lines. Eur J
Cancer. 46:2771–2780. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Poljakova J, Hrebackova J, Dvorakova M,
Moserova M, Eckschlager T, Hrabeta J, Göttlicherova M, Kopejtkova
B, Frei E, Kizek R, et al: Anticancer agent ellipticine combined
with histone deacetylase inhibitors, valproic acid and trichostatin
A, is an effective DNA damage strategy in human neuroblastoma.
Neuro Endocrinol Lett. 32(Suppl 1): 101–116. 2011.PubMed/NCBI
|
|
19
|
Cipro Š, Hřebačková J, Hraběta J,
Poljaková J and Eckschlager T: Valproic acid overcomes
hypoxia-induced resistance to apoptosis. Oncol Rep. 27:1219–1226.
2012.
|
|
20
|
Groh T, Hrabeta J, Poljakova J,
Eckschlager T and Stiborova M: Impact of histone deacetylase
inhibitor valproic acid on the anticancer effect of etoposide on
neuroblastoma cells. Neuro Endocrinol Lett. 33(Suppl 3): S16–S24.
2012.
|
|
21
|
Wang G, Edwards H, Caldwell JT, Buck SA,
Qing WY, Taub JW, Ge Y and Wang Z: Panobinostat synergistically
enhances the cytotoxic effects of cisplatin, doxorubicin or
etoposide on high-risk neuroblastoma cells. PLoS One. 8:e766622013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hrabeta J, Stiborova M, Adam V, Kizek R
and Eckschlager T: Histone deacetylase inhibitors in cancer
therapy. A review. Biomed Pap Med Fac Univ Palacky Olomouc Czech
Repub. 158:161–169. 2014.
|
|
23
|
Kartalou M and Essigmann JM: Mechanisms of
resistance to cisplatin. Mutat Res. 478:23–43. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Miyajima A, Nakashima J, Yoshioka K,
Tachibana M, Tazaki H and Murai M: Role of reactive oxygen species
in cis-dichlorodi-ammineplatinum-induced cytotoxicity on bladder
cancer cells. Br J Cancer. 76:206–210. 1997. View Article : Google Scholar
|
|
25
|
Huang H-L, Fang L-W, Lu S-P, Chou C-K, Luh
T-Y and Lai M-Z: DNA-damaging reagents induce apoptosis through
reactive oxygen species-dependent Fas aggregation. Oncogene.
22:8168–8177. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wozniak AJ and Ross WE: DNA damage as a
basis for
4′-demethy-lepipodophyllotoxin-9-(4,6-O-ethylidene-beta-D-glucopyranoside)
(etoposide) cytotoxicity. Cancer Res. 43:120–124. 1983.PubMed/NCBI
|
|
27
|
Hande KR: Etoposide: Four decades of
development of a topoisomerase II inhibitor. Eur J Cancer.
34:1514–1521. 1998. View Article : Google Scholar
|
|
28
|
Baldwin EL and Osheroff N: Etoposide,
topoisomerase II and cancer. Curr Med Chem Anticancer Agents.
5:363–372. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
New M, Olzscha H and La Thangue NB: HDAC
inhibitor-based therapies: Can we interpret the code? Mol Oncol.
6:637–656. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yoshida M, Kijima M, Akita M and Beppu T:
Potent and specific inhibition of mammalian histone deacetylase
both in vivo and in vitro by trichostatin A. J Biol Chem.
265:17174–17179. 1990.PubMed/NCBI
|
|
31
|
Yoon CY, Park MJ, Lee JS, Lee SC, Oh JJ,
Park H, Chung CW, Abdullajanov MM, Jeong SJ, Hong SK, et al: The
histone deacetylase inhibitor trichostatin A synergistically
resensitizes a cisplatin resistant human bladder cancer cell line.
J Urol. 185:1102–1111. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Meng J, Zhang H-H, Zhou C-X, Li C, Zhang F
and Mei Q-B: The histone deacetylase inhibitor trichostatin A
induces cell cycle arrest and apoptosis in colorectal cancer cells
via p53-dependent and -independent pathways. Oncol Rep. 28:384–388.
2012.PubMed/NCBI
|
|
33
|
Poljaková J, Eckschlager T, Hraběta J,
Hrebacková J, Smutný S, Frei E, Martínek V, Kizek R and Stiborová
M: The mechanism of cytotoxicity and DNA adduct formation by the
anticancer drug ellipticine in human neuroblastoma cells. Biochem
Pharmacol. 77:1466–1479. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Poljaková J, Groh T, Gudino ZO, Hraběta J,
Bořek-Dohalská L, Kizek R, Doktorová H, Eckschlager T and Stiborová
M: Hypoxia-mediated histone acetylation and expression of N-myc
transcription factor dictate aggressiveness of neuroblastoma cells.
Oncol Rep. 31:1928–1934. 2014.PubMed/NCBI
|
|
35
|
Shechter D, Dormann HL, Allis CD and Hake
SB: Extraction, purification and analysis of histones. Nat Protoc.
2:1445–1457. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lowry OH, Rosebrough NJ, Farr AL and
Randall RJ: Protein measurement with the Folin phenol reagent. J
Biol Chem. 193:265–275. 1951.PubMed/NCBI
|
|
37
|
Ke N, Wang X, Xu X and Abassi YA: The
xCELLigence system for real-time and label-free monitoring of cell
viability. Methods Mol Biol. 740:33–43. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Earnshaw WC, Martins LM and Kaufmann SH:
Mammalian caspases: Structure, activation, substrates, and
functions during apoptosis. Annu Rev Biochem. 68:383–424. 1999.
View Article : Google Scholar
|
|
39
|
Porter AG and Jänicke RU: Emerging roles
of caspase-3 in apoptosis. Cell Death Differ. 6:99–104. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Slee EA, Adrain C and Martin SJ:
Executioner caspase-3, −6, and −7 perform distinct, non-redundant
roles during the demolition phase of apoptosis. J Biol Chem.
276:7320–7326. 2001. View Article : Google Scholar
|
|
41
|
Atienza JM, Yu N, Kirstein SL, Xi B, Wang
X, Xu X and Abassi YA: Dynamic and label-free cell-based assays
using the real-time cell electronic sensing system. Assay Drug Dev
Technol. 4:597–607. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chou TC and Talalay P: Quantitative
analysis of dose-effect relationships: The combined effects of
multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 22:27–55.
1984. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chou T-C: Theoretical basis, experimental
design, and computerized simulation of synergism and antagonism in
drug combination studies. Pharmacol Rev. 58:621–681. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Luchenko VL, Salcido CD, Zhang Y, Agama K,
Komlodi-Pasztor E, Murphy RF, Giaccone G, Pommier Y, Bates SE and
Varticovski L: Schedule-dependent synergy of histone deacetylase
inhibitors with DNA damaging agents in small cell lung cancer. Cell
Cycle. 10:3119–3128. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Jordan MA and Wilson L: Microtubules as a
target for anticancer drugs. Nat Rev Cancer. 4:253–265. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Tu Y, Cheng S, Zhang S, Sun H and Xu Z:
Vincristine induces cell cycle arrest and apoptosis in SH-SY5Y
human neuroblastoma cells. Int J Mol Med. 31:113–119. 2013.
|
|
47
|
Phiel CJ, Zhang F, Huang EY, Guenther MG,
Lazar MA and Klein PS: Histone deacetylase is a direct target of
valproic acid, a potent anticonvulsant, mood stabilizer, and
teratogen. J Biol Chem. 276:36734–36741. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Sokolov MV, Dickey JS, Bonner WM and
Sedelnikova OA: gamma-H2AX in bystander cells: Not just a
radiation-triggered event, a cellular response to stress mediated
by intercellular communication. Cell Cycle. 6:2210–2212. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Bonner WM, Redon CE, Dickey JS, Nakamura
AJ, Sedelnikova OA, Solier S and Pommier Y: GammaH2AX and cancer.
Nat Rev Cancer. 8:957–967. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Nakamura AJ, Rao VA, Pommier Y and Bonner
WM: The complexity of phosphorylated H2AX foci formation and DNA
repair assembly at DNA double-strand breaks. Cell Cycle. 9:389–397.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yuan J, Adamski R and Chen J: Focus on
histone variant H2AX: To be or not to be. FEBS Lett. 584:3717–3724.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
dos Santos MP, Schwartsmann G, Roesler R,
Brunetto AL and Abujamra AL: Sodium butyrate enhances the cytotoxic
effect of antineoplastic drugs in human lymphoblastic T-cells. Leuk
Res. 33:218–221. 2009. View Article : Google Scholar
|
|
53
|
Kurz EU, Wilson SE, Leader KB, Sampey BP,
Allan WP, Yalowich JC and Kroll DJ: The histone deacetylase
inhibitor sodium butyrate induces DNA topoisomerase II alpha
expression and confers hypersensitivity to etoposide in human
leukemic cell lines. Mol Cancer Ther. 1:121–131. 2001.
|
|
54
|
Bruzzese F, Rocco M, Castelli S, Di
Gennaro E, Desideri A and Budillon A: Synergistic antitumor effect
between vorinostat and topotecan in small cell lung cancer cells is
mediated by generation of reactive oxygen species and DNA
damage-induced apoptosis. Mol Cancer Ther. 8:3075–3087. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Itoh T, Terazawa R, Kojima K, Nakane K,
Deguchi T, Ando M, Tsukamasa Y, Ito M and Nozawa Y: Cisplatin
induces production of reactive oxygen species via NADPH oxidase
activation in human prostate cancer cells. Free Radic Res.
45:1033–1039. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Yu C, Friday BB, Lai JP, McCollum A,
Atadja P, Roberts LR and Adjei AA: Abrogation of MAPK and Akt
signaling by AEE788 synergistically potentiates histone deacetylase
inhibitor-induced apoptosis through reactive oxygen species
generation. Clin Cancer Res. 13:1140–1148. 2007. View Article : Google Scholar : PubMed/NCBI
|