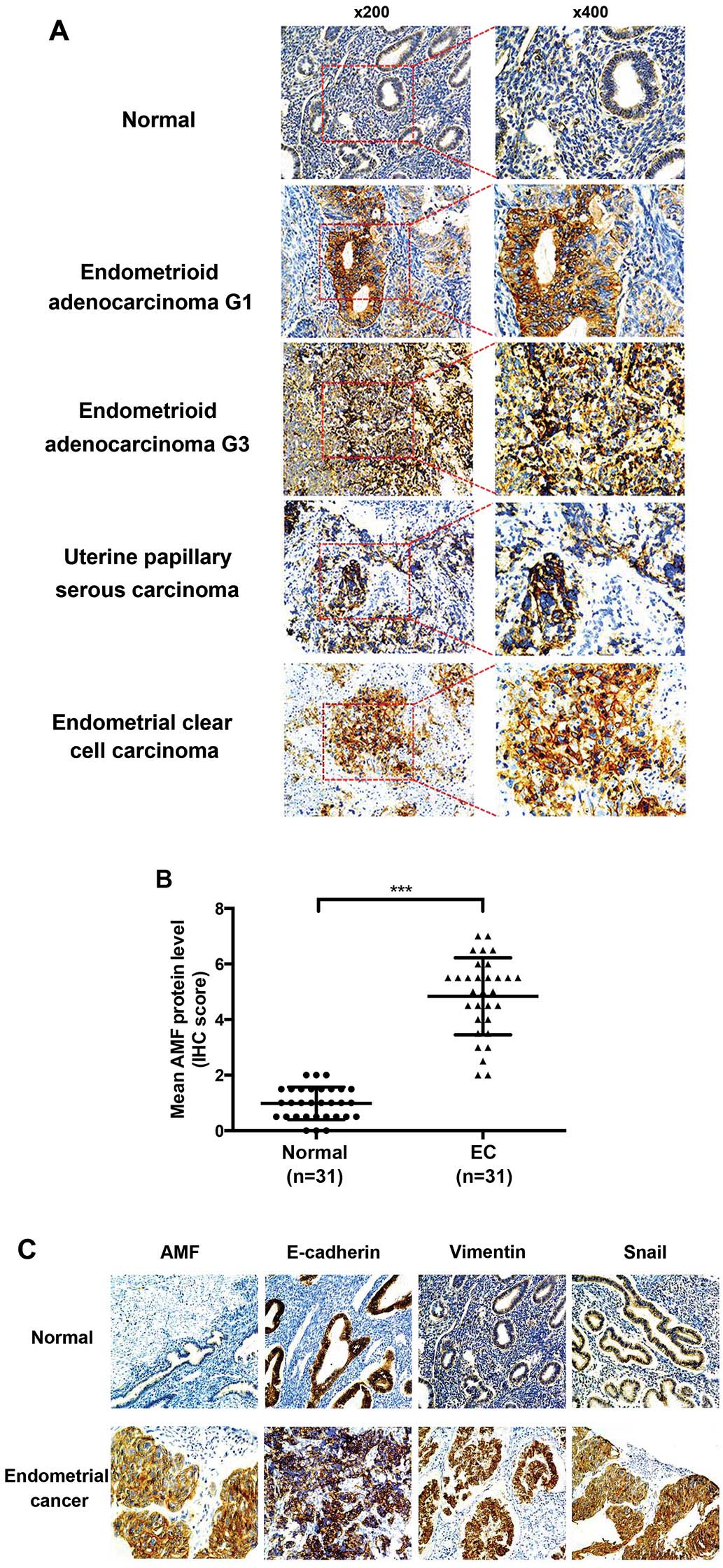
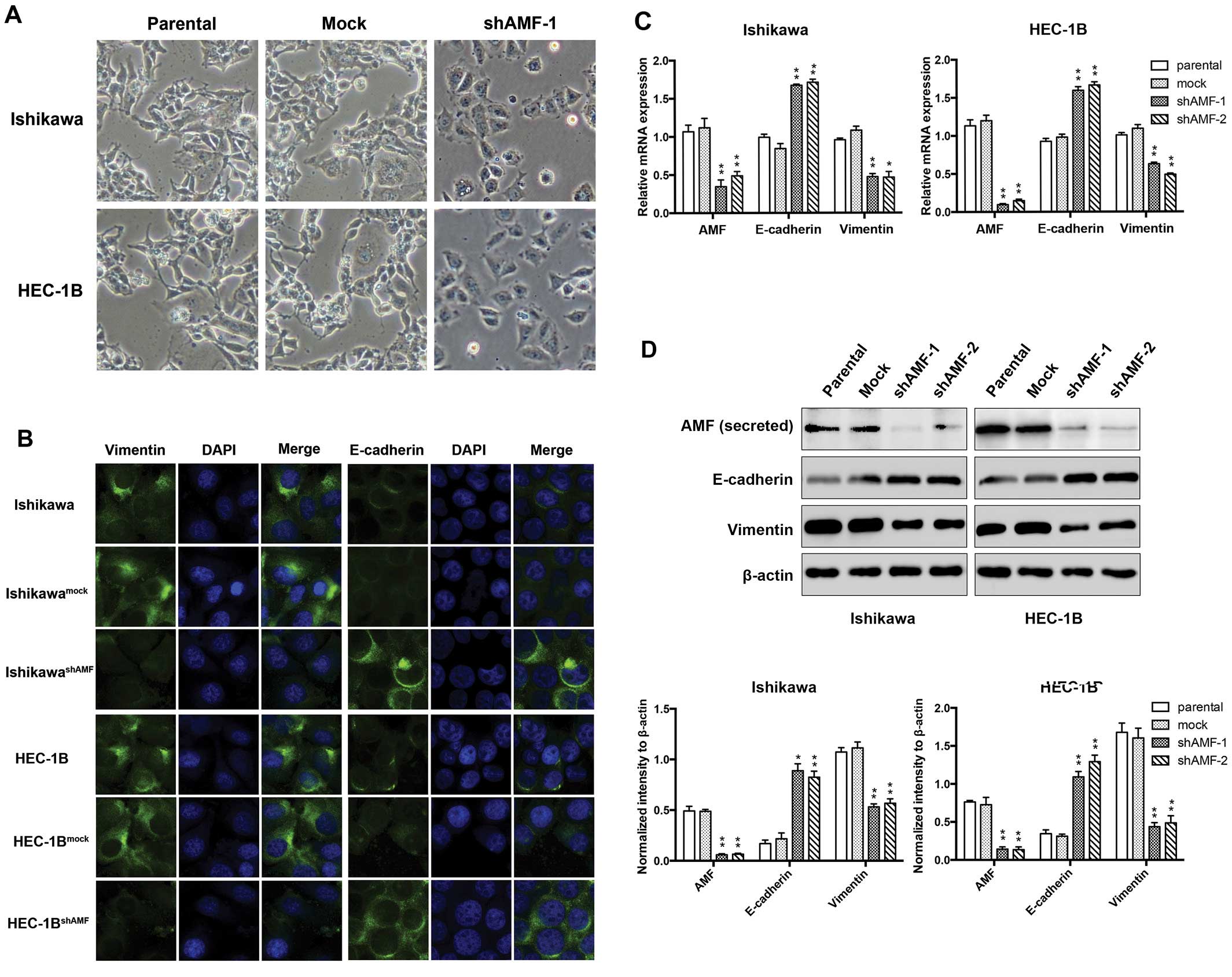
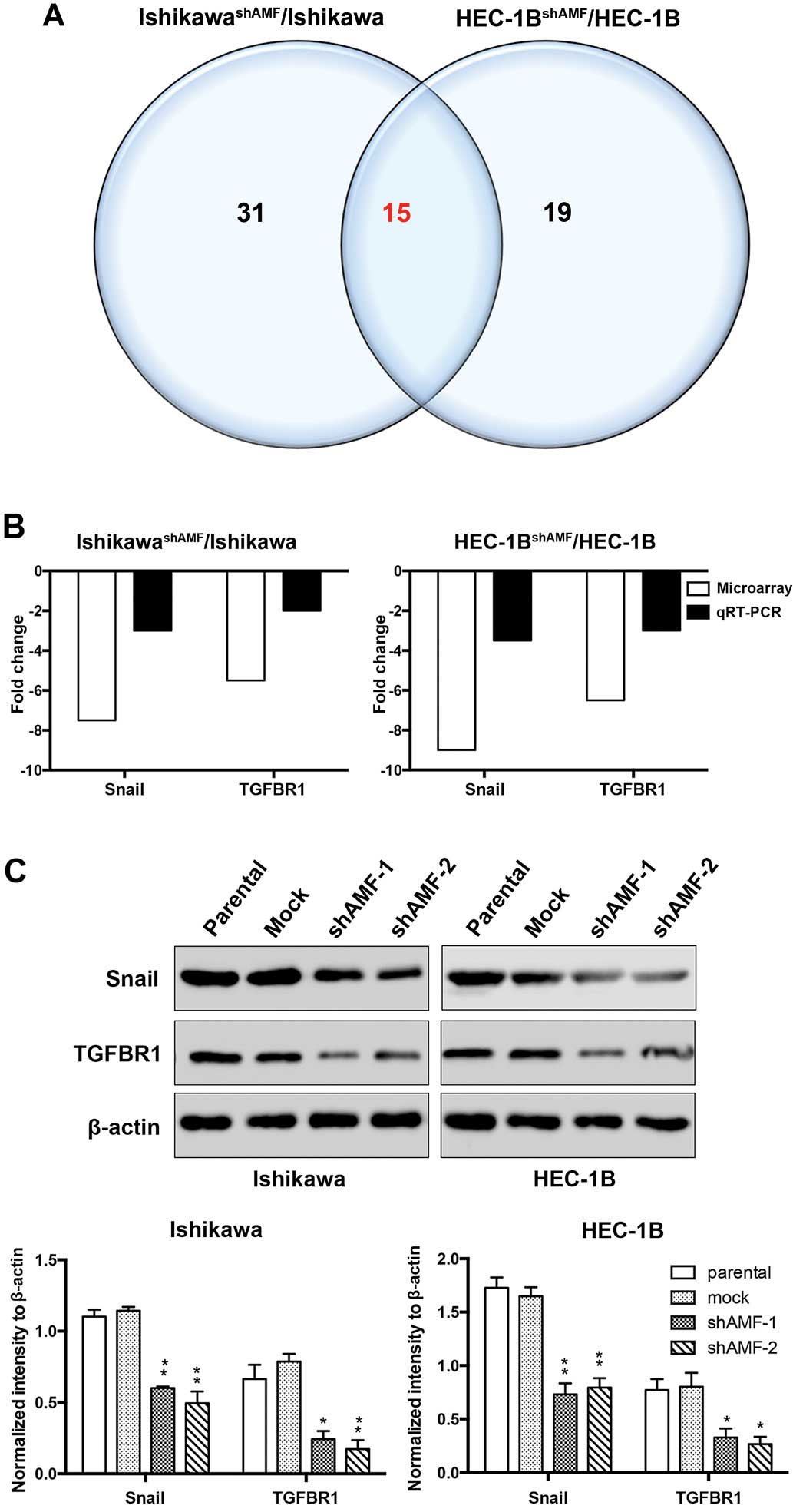
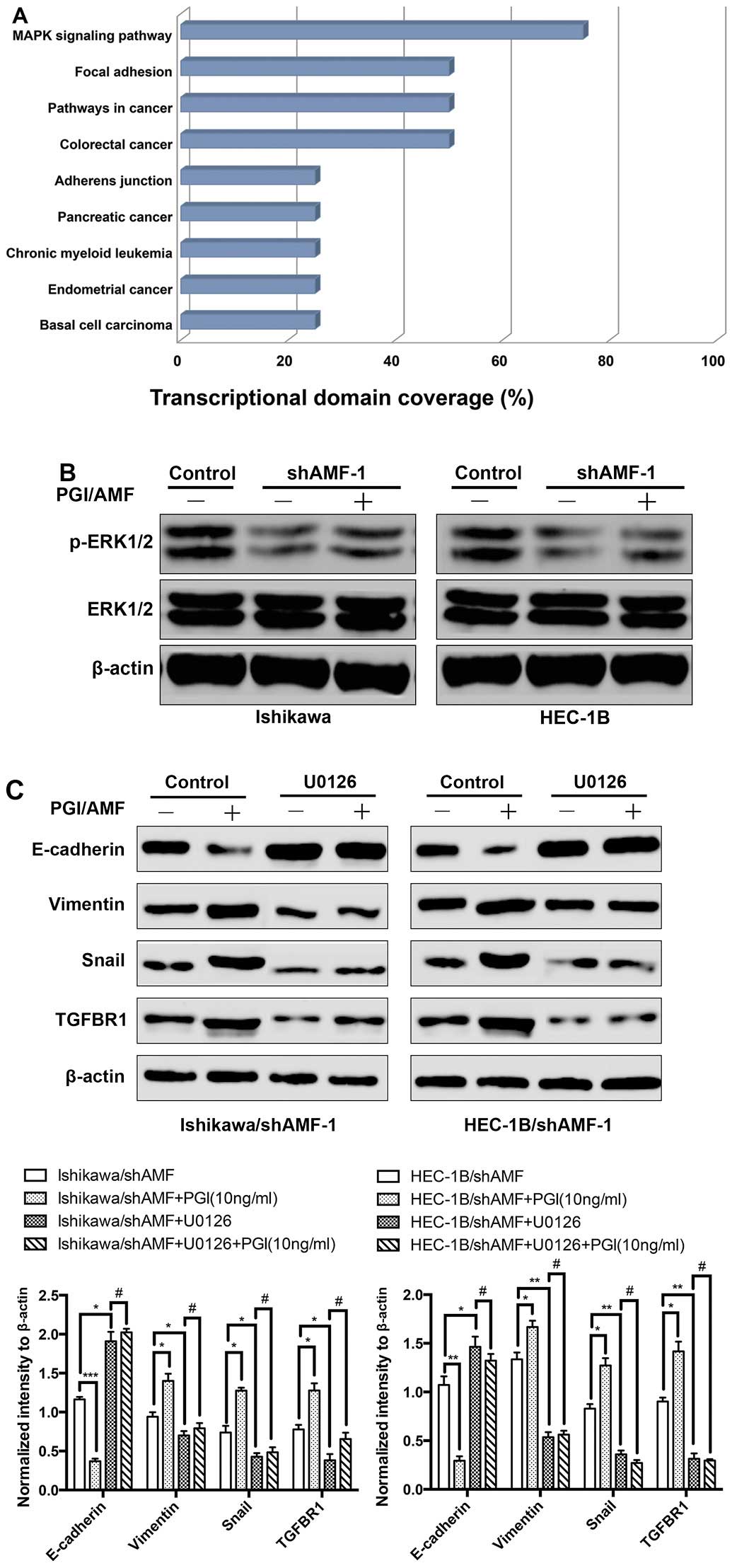
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2015. CA Cancer J Clin. 65:5–29. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Salvesen HB, Haldorsen IS and Trovik J:
Markers for individualised therapy in endometrial carcinoma. Lancet
Oncol. 13:e353–e361. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sorosky JI: Endometrial cancer. Obstet
Gynecol. 120:383–397. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Watanabe H, Takehana K, Date M, Shinozaki
T and Raz A: Tumor cell autocrine motility factor is the
neuroleukin/phosphohexose isomerase polypeptide. Cancer Res.
56:2960–2963. 1996.PubMed/NCBI
|
|
5
|
Niinaka Y, Paku S, Haga A, Watanabe H and
Raz A: Expression and secretion of neuroleukin/phosphohexose
isomerase/maturation factor as autocrine motility factor by tumor
cells. Cancer Res. 58:2667–2674. 1998.PubMed/NCBI
|
|
6
|
Kim JW and Dang CV: Multifaceted roles of
glycolytic enzymes. Trends Biochem Sci. 30:142–150. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gurney ME, Apatoff BR, Spear GT, Baumel
MJ, Antel JP, Bania MB and Reder AT: Neuroleukin: A lymphokine
product of lectin-stimulated T cells. Science. 234:574–581. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xu W, Seiter K, Feldman E, Ahmed T and
Chiao JW: The differentiation and maturation mediator for human
myeloid leukemia cells shares homology with neuroleukin or
phosphoglucose isomerase. Blood. 87:4502–4506. 1996.PubMed/NCBI
|
|
9
|
Kho DH, Zhang T, Balan V, Wang Y, Ha SW,
Xie Y and Raz A: Autocrine motility factor modulates EGF-mediated
invasion signaling. Cancer Res. 74:2229–2237. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kho DH, Nangia-Makker P, Balan V, Hogan V,
Tait L, Wang Y and Raz A: Autocrine motility factor promotes HER2
cleavage and signaling in breast cancer cells. Cancer Res.
73:1411–1419. 2013. View Article : Google Scholar :
|
|
11
|
Bayo J, Fiore E, Aquino JB, Malvicini M,
Rizzo M, Peixoto E, Andriani O, Alaniz L, Piccioni F, Bolontrade M,
et al: Increased migration of human mesenchymal stromal cells by
autocrine motility factor (AMF) resulted in enhanced recruitment
towards hepatocellular carcinoma. PLoS One. 9:e951712014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shih WL, Liao MH, Lin PY, Chang CI, Cheng
HL, Yu FL and Lee JW: PI 3-kinase/Akt and STAT3 are required for
the prevention of TGF-beta-induced Hep3B cell apoptosis by
autocrine motility factor/phosphoglucose isomerase. Cancer Lett.
290:223–237. 2010. View Article : Google Scholar
|
|
13
|
Niinaka Y, Harada K, Fujimuro M, Oda M,
Haga A, Hosoki M, Uzawa N, Arai N, Yamaguchi S, Yamashiro M, et al:
Silencing of autocrine motility factor induces
mesenchymal-to-epithelial transition and suppression of
osteosarcoma pulmonary metastasis. Cancer Res. 70:9483–9493. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Funasaka T, Hogan V and Raz A:
Phosphoglucose isomerase/autocrine motility factor mediates
epithelial and mesenchymal phenotype conversions in breast cancer.
Cancer Res. 69:5349–5356. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Thiery JP and Sleeman JP: Complex networks
orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell
Biol. 7:131–142. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Greenburg G and Hay ED: Epithelia
suspended in collagen gels can lose polarity and express
characteristics of migrating mesenchymal cells. J Cell Biol.
95:333–339. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hay ED: The mesenchymal cell, its role in
the embryo, and the remarkable signaling mechanisms that create it.
Dev Dyn. 233:706–720. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lee JM, Dedhar S, Kalluri R and Thompson
EW: The epithelial-mesenchymal transition: New insights in
signaling, development, and disease. J Cell Biol. 172:973–981.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
De Craene B and Berx G: Regulatory
networks defining EMT during cancer initiation and progression. Nat
Rev Cancer. 13:97–110. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dong P, Karaayvaz M, Jia N, Kaneuchi M,
Hamada J, Watari H, Sudo S, Ju J and Sakuragi N: Mutant p53
gain-of-function induces epithelial-mesenchymal transition through
modulation of the miR-130b-ZEB1 axis. Oncogene. 32:3286–3295. 2013.
View Article : Google Scholar :
|
|
22
|
Ahmad A, Aboukameel A, Kong D, Wang Z,
Sethi S, Chen W, Sarkar FH and Raz A: Phosphoglucose
isomerase/autocrine motility factor mediates epithelial-mesenchymal
transition regulated by miR-200 in breast cancer cells. Cancer Res.
71:3400–3409. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Funasaka T, Hu H, Yanagawa T, Hogan V and
Raz A: Down-regulation of phosphoglucose isomerase/autocrine
motility factor results in mesenchymal-to-epithelial transition of
human lung fibrosarcoma cells. Cancer Res. 67:4236–4243. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kyo S, Sakaguchi J, Ohno S, Mizumoto Y,
Maida Y, Hashimoto M, Nakamura M, Takakura M, Nakajima M and
Masutomi K: High Twist expression is involved in infiltrative
endometrial cancer and affects patient survival. Hum Pathol.
37:431–438. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Massagué J: TGFβ signalling in context.
Nat Rev Mol Cell Biol. 13:616–630. 2012. View Article : Google Scholar
|
|
26
|
Cano A, Pérez-Moreno MA, Rodrigo I,
Locascio A, Blanco MJ, del Barrio MG, Portillo F and Nieto MA: The
transcription factor snail controls epithelial-mesenchymal
transitions by repressing E-cadherin expression. Nat Cell Biol.
2:76–83. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Subarsky P and Hill RP: The hypoxic tumour
microenvironment and metastatic progression. Clin Exp Metastasis.
20:237–250. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tsutsumi S, Yanagawa T, Shimura T,
Fukumori T, Hogan V, Kuwano H and Raz A: Regulation of cell
proliferation by autocrine motility factor/phosphoglucose isomerase
signaling. J Biol Chem. 278:32165–32172. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang J and Weinberg RA:
Epithelial-mesenchymal transition: At the crossroads of development
and tumor metastasis. Dev Cell. 14:818–829. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Peinado H, Olmeda D and Cano A: Snail, Zeb
and bHLH factors in tumour progression: An alliance against the
epithelial phenotype? Nat Rev Cancer. 7:415–428. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Prindull G and Zipori D: Environmental
guidance of normal and tumor cell plasticity: Epithelial
mesenchymal transitions as a paradigm. Blood. 103:2892–2899. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Otto T, Birchmeier W, Schmidt U, Hinke A,
Schipper J, Rübben H and Raz A: Inverse relation of E-cadherin and
autocrine motility factor receptor expression as a prognostic
factor in patients with bladder carcinomas. Cancer Res.
54:3120–3123. 1994.PubMed/NCBI
|
|
33
|
Batlle E, Sancho E, Francí C, Domínguez D,
Monfar M, Baulida J and García De Herreros A: The transcription
factor snail is a repressor of E-cadherin gene expression in
epithelial tumour cells. Nat Cell Biol. 2:84–89. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Pickup M, Novitskiy S and Moses HL: The
roles of TGFβ in the tumour microenvironment. Nat Rev Cancer.
13:788–799. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View Article : Google Scholar : PubMed/NCBI
|