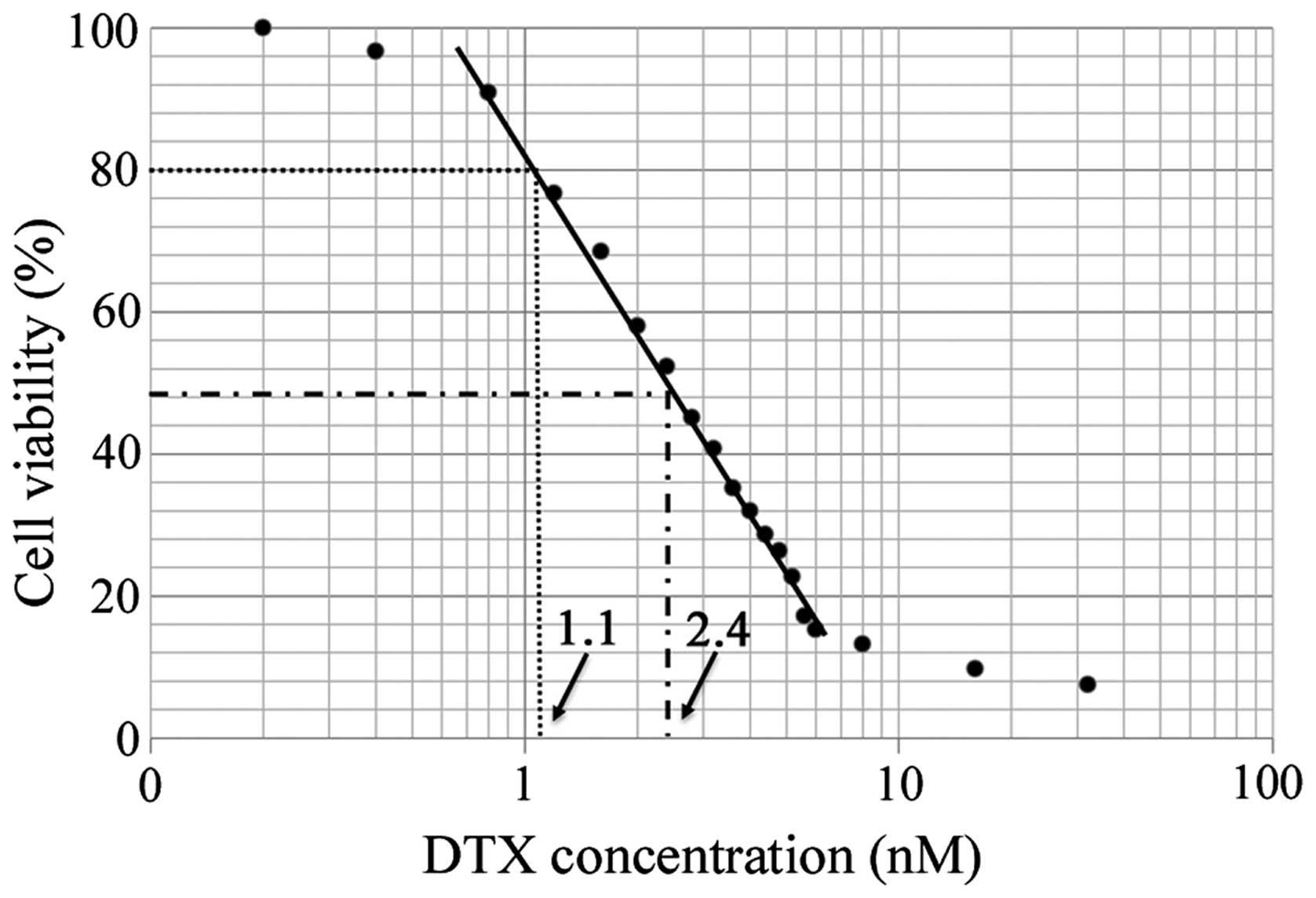
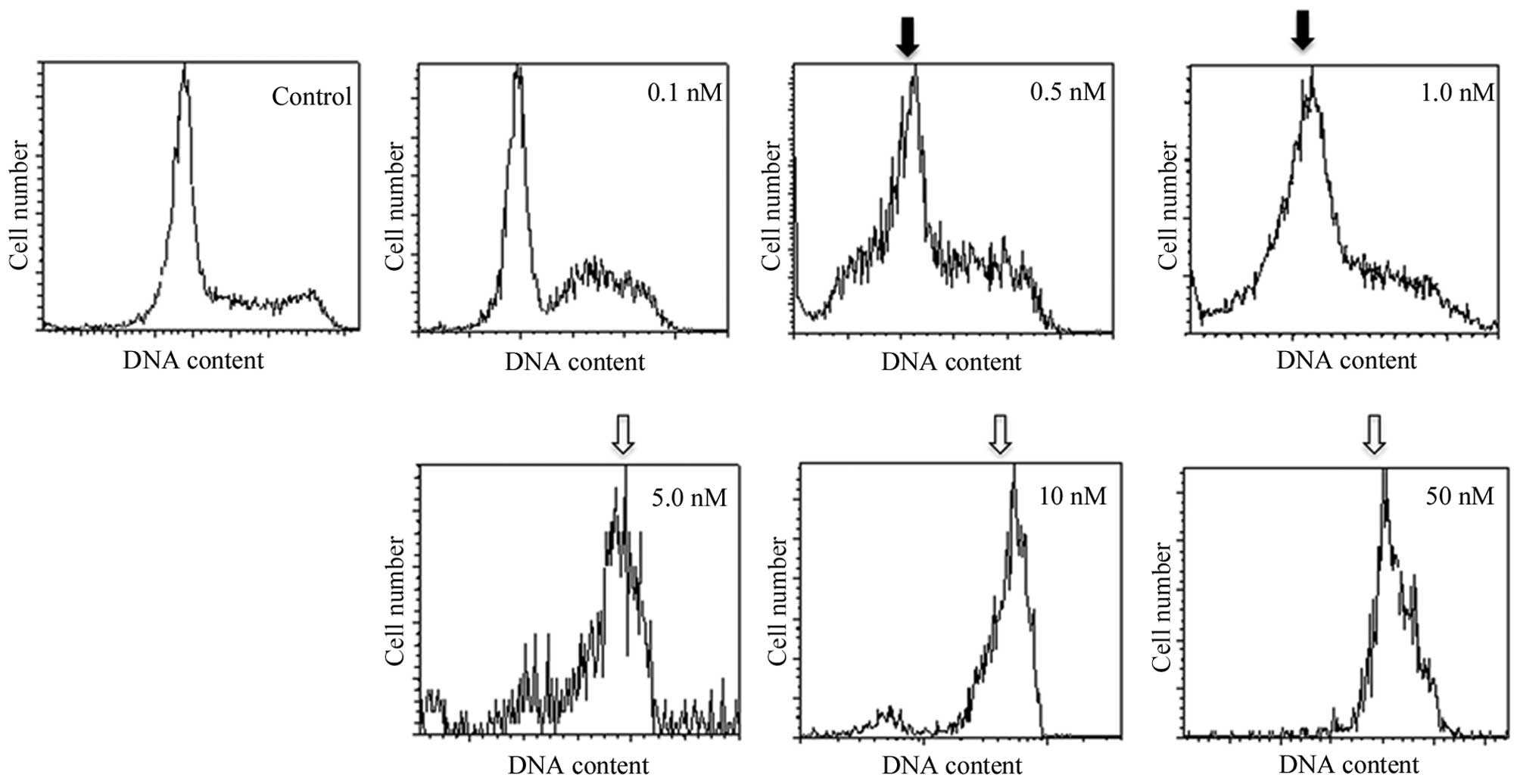
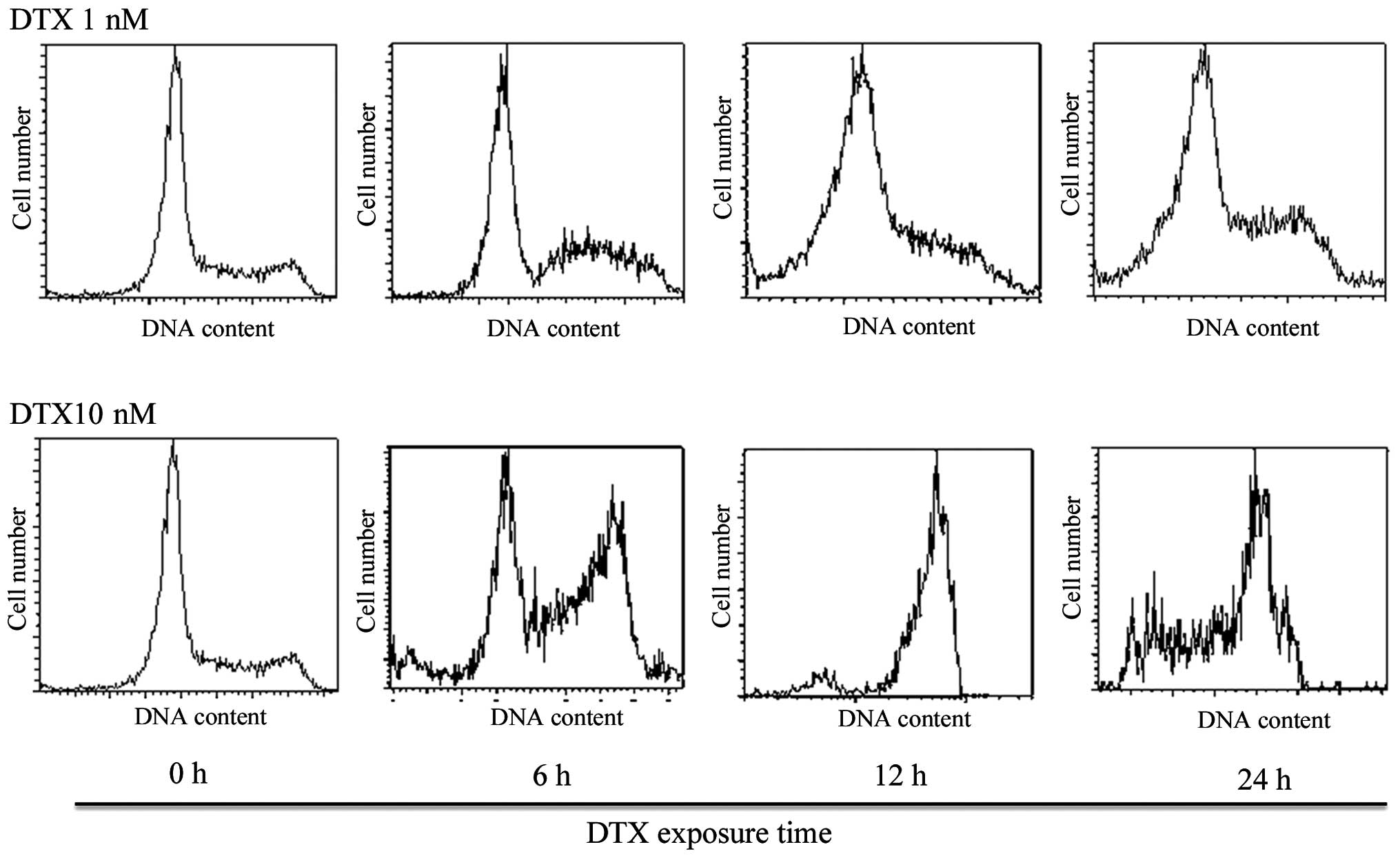
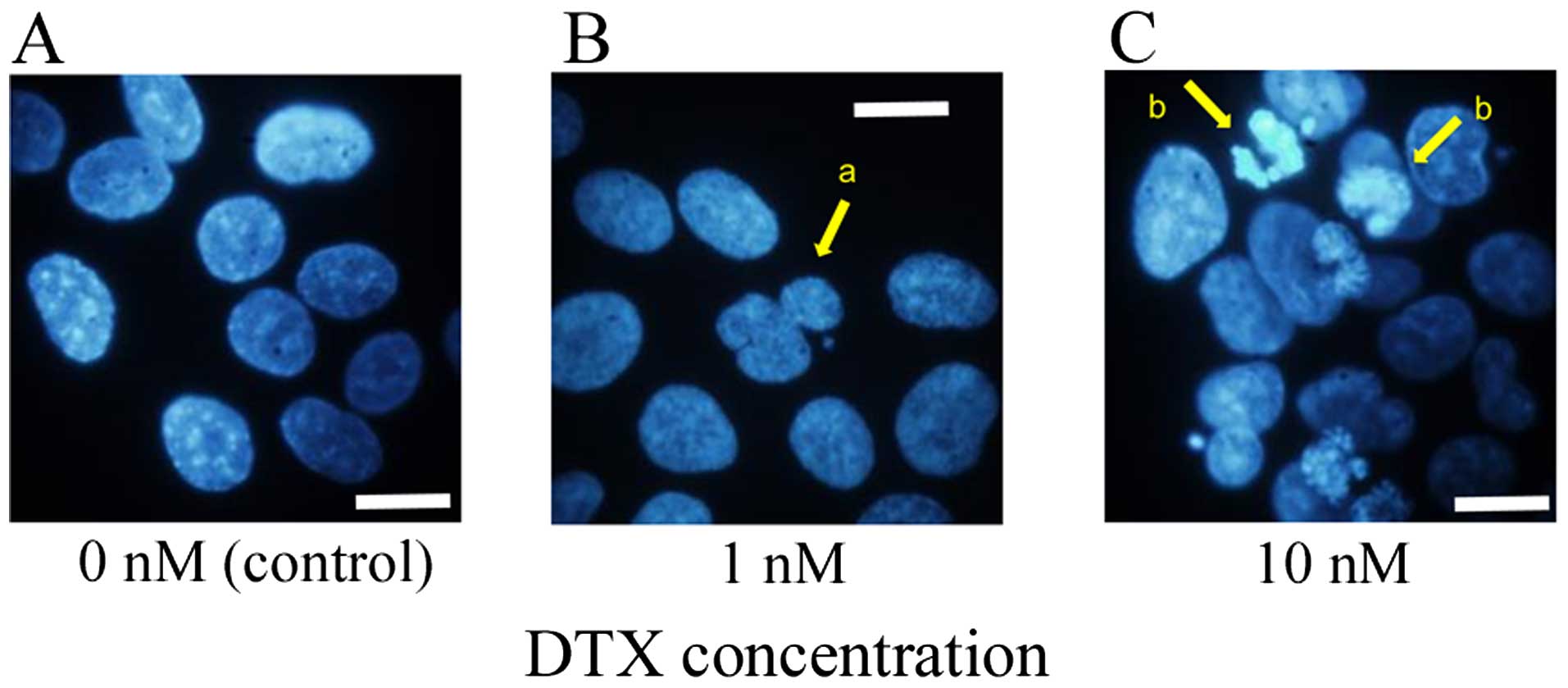
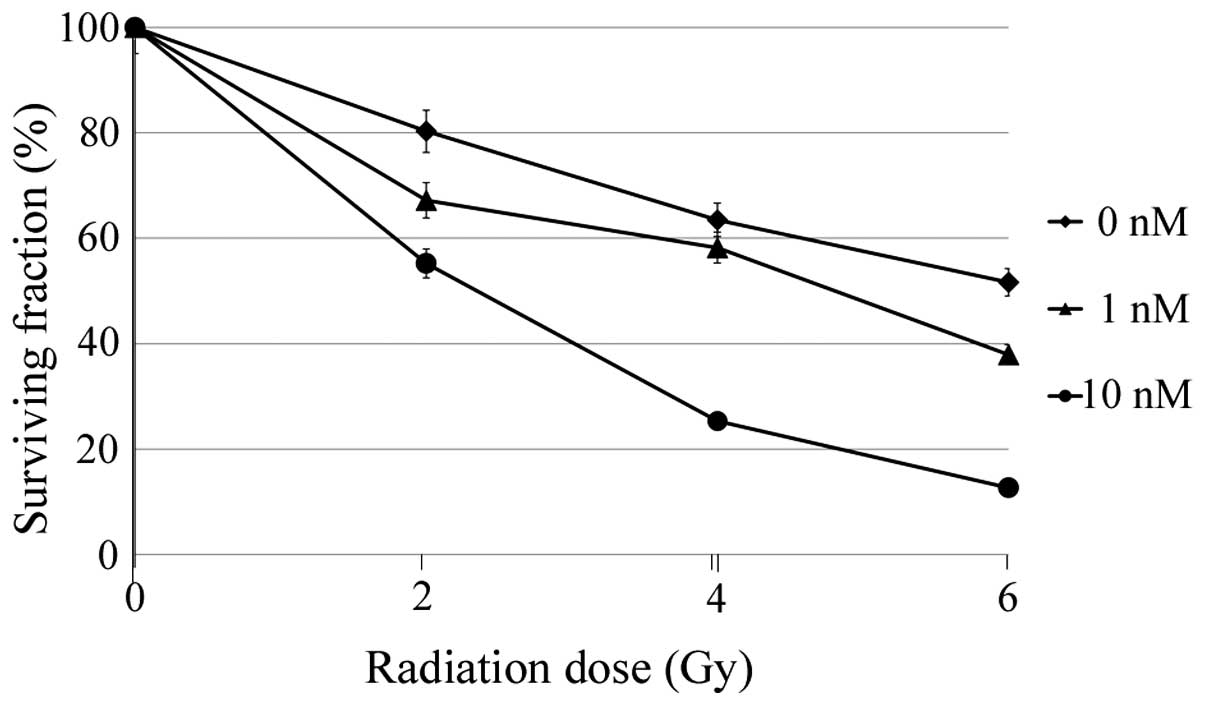
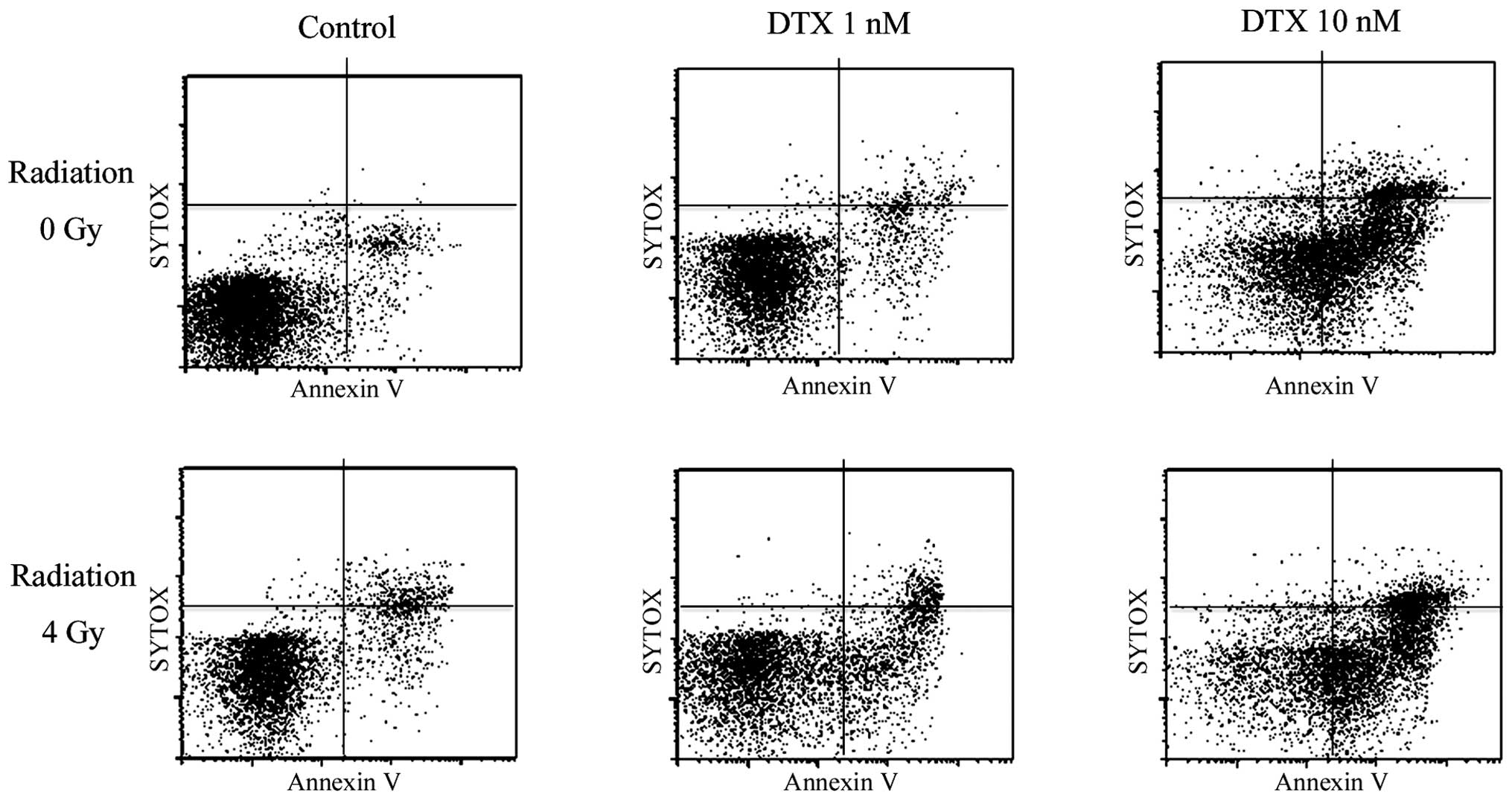
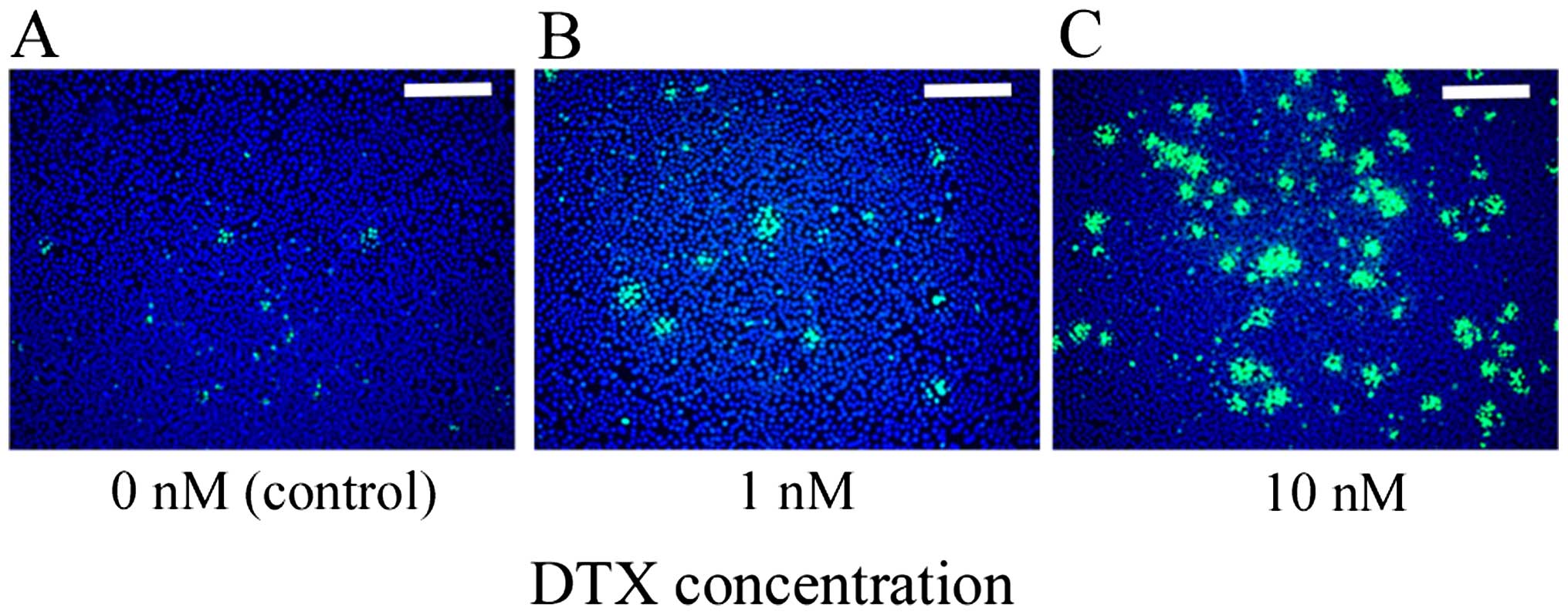
|
1
|
Castro C, Bosetti C, Malvezzi M, Bertuccio
P, Levi F, Negri E, La Vecchia C and Lunet N: Patterns and trends
in esophageal cancer mortality and incidence in Europe (1980–2011)
and predictions to 2015. Ann Oncol. 25:283–290. 2014. View Article : Google Scholar
|
|
2
|
Ohtsu A, Boku N, Muro K, Chin K, Muto M,
Yoshida S, Satake M, Ishikura S, Ogino T, Miyata Y, et al:
Definitive chemoradiotherapy for T4 and/or M1 lymph node squamous
cell carcinoma of the esophagus. J Clin Oncol. 17:2915–2921.
1999.PubMed/NCBI
|
|
3
|
Adelstein DJ, Li Y, Adams GL, Wagner H Jr,
Kish JA, Ensley JF, Schuller DE and Forastiere AA: An intergroup
phase III comparison of standard radiation therapy and two
schedules of concurrent chemoradiotherapy in patients with
unresectable squamous cell head and neck cancer. J Clin Oncol.
21:92–98. 2003. View Article : Google Scholar
|
|
4
|
Ishida K, Ando N, Yamamoto S, Ide H and
Shinoda M: Phase II study of cisplatin and 5-fluorouracil with
concurrent radiotherapy in advanced squamous cell carcinoma of the
esophagus: A Japan Esophageal Oncology Group (JEOG)/Japan Clinical
Oncology Group trial (JCOG9516). Jpn J Clin Oncol. 34:615–619.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Herskovic A, Martz K, al-Sarraf M,
Leichman L, Brindle J, Vaitkevicius V, Cooper J, Byhardt R, Davis L
and Emami B: Combined chemotherapy and radiotherapy compared with
radiotherapy alone in patients with cancer of the esophagus. N Engl
J Med. 326:1593–1598. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kaneko K, Ito H, Konishi K, Kurahashi T,
Ito T, Katagiri A, Yamamoto T, Kitahara T, Mizutani Y, Ohtsu A, et
al: Definitive chemoradiotherapy for patients with malignant
stricture due to T3 or T4 squamous cell carcinoma of the
oesophagus. Br J Cancer. 88:18–24. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Choy H: Taxanes in combined modality
therapy for solid tumors. Crit Rev Oncol Hematol. 37:237–247. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ngeow J, Lim WT, Leong SS, Ang MK, Toh CK,
Gao F, Chowbay B and Tan EH: Docetaxel is effective in heavily
pretreated patients with disseminated nasopharyngeal carcinoma. Ann
Oncol. 22:718–722. 2011. View Article : Google Scholar
|
|
9
|
Bang YJ, Kang WK, Kang YK, Kim HC, Jacques
C, Zuber E, Daglish B, Boudraa Y, Kim WS, Heo DS, et al: Docetaxel
75 mg/m(2) is active and well tolerated in patients with metastatic
or recurrent gastric cancer: A phase II trial. Jpn J Clin Oncol.
32:248–254. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Abbrederis K, Lorenzen S, von Weikersthal
LF, Vehling-Kaiser U, Schuster T, Rothling N, Peschel C and Lordick
F: Weekly docetaxel monotherapy for advanced gastric or
esophagogastric junction cancer. Results of a phase II study in
elderly patients or patients with impaired performance status. Crit
Rev Oncol Hematol. 66:84–90. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nuzzo F, Morabito A, Gravina A, Di Rella
F, Landi G, Pacilio C, Labonia V, Rossi E, De Maio E, Piccirillo
MC, et al: Effects on quality of life of weekly docetaxel-based
chemotherapy in patients with locally advanced or metastatic breast
cancer: Results of a single-centre randomized phase 3 trial. BMC
Cancer. 11:752011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gravis G, Bladou F, Salem N,
Macquart-Moulin G, Serment G, Camerlo J, Genre D, Bardou VJ,
Maraninchi D and Viens P: Weekly administration of docetaxel for
symptomatic metastatic hormone-refractory prostate carcinoma.
Cancer. 98:1627–1634. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Perng RP, Shih JF, Chen YM, Chou KC, Lee
YC and Tsai CM: A phase II study of single-agent docetaxel
chemotherapy for non-small cell lung cancer. Jpn J Clin Oncol.
30:429–434. 2000. View Article : Google Scholar
|
|
14
|
Kawada K, Kitagawa K, Kamei S, Inada M,
Mitsuma A, Sawaki M, Kikumori T, Fujimoto Y, Arima H, Imai T, et
al: The feasibility study of docetaxel in patients with anaplastic
thyroid cancer. Jpn J Clin Oncol. 40:596–599. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Muro K, Hamaguchi T, Ohtsu A, Boku N, Chin
K, Hyodo I, Fujita H, Takiyama W and Ohtsu T: A phase II study of
single-agent docetaxel in patients with metastatic esophageal
cancer. Ann Oncol. 15:955–959. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hennequin C, Giocanti N and Favaudon V:
Interaction of ionizing radiation with paclitaxel (Taxol) and
docetaxel (Taxotere) in HeLa and SQ20B cells. Cancer Res.
56:1842–1850. 1996.PubMed/NCBI
|
|
17
|
Balcer-Kubiczek EK, Attarpour M, Wang JZ
and Regine WF: The effect of docetaxel (taxotere) on human gastric
cancer cells exhibiting low-dose radiation hypersensitivity. Clin
Med Oncol. 2:301–311. 2008.PubMed/NCBI
|
|
18
|
Mason KA, Hunter NR, Milas M, Abbruzzese
JL and Milas L: Docetaxel enhances tumor radioresponse in vivo.
Clin Cancer Res. 3:2431–2438. 1997.
|
|
19
|
Yoshida T, Tokashiki R, Itoh H, Nakamura
K, Hiramatsu H, Tsukahara K, Shimizu S, Takada D, Okamoto I, Abe K,
et al: A phase I–II study of bi-weekly docetaxel combined with
radiation therapy for patients with cancer of the
larynx/hypopharynx. Jpn J Clin Oncol. 37:641–646. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li YQ, Shi AH, Li FH, Yu R and Zhu GY:
Phase I study to determine MTD of docetaxel and cisplatin with
concurrent radiation therapy for Stage III non-small cell lung
cancer. Chin J Cancer Res. 23:129–133. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kiura K, Ueoka H, Segawa Y, Tabata M,
Kamei H, Takigawa N, Hiraki S, Watanabe Y, Bessho A, Eguchi K, et
al; Okayama Lung Cancer Study Group. Phase I/II study of docetaxel
and cisplatin with concurrent thoracic radiation therapy for
locally advanced non-small-cell lung cancer. Br J Cancer.
89:795–802. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tishler RB, Norris CM Jr, Colevas AD, Lamb
CC, Karp D, Busse PM, Nixon A, Frankenthaler R, Lake-Willcutt B,
Costello R, et al: A Phase I/II trial of concurrent docetaxel and
radiation after induction chemotherapy in patients with poor
prognosis squamous cell carcinoma of the head and neck. Cancer.
95:1472–1481. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Fujii M, Tsukuda M, Satake B, Kubota A,
Kida A, Kohno N, Okami K and Inuyama Y; Japan Cooperative Head and
Neck Oncology Group (JCHNOG). Phase I/II trial of weekly docetaxel
and concomitant radiotherapy for squamous cell carcinoma of the
head and neck. Int J Clin Oncol. 9:107–112. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tishler RB, Posner MR, Norris CM Jr,
Mahadevan A, Sullivan C, Goguen L, Wirth LJ, Costello R, Case M,
Stowell S, et al: Concurrent weekly docetaxel and concomitant boost
radiation therapy in the treatment of locally advanced squamous
cell cancer of the head and neck. Int J Radiat Oncol Biol Phys.
65:1036–1044. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Clark JI, Eisner RM, Hofmeister C, Norton
J, Thomas S, Choudhury A, Petruzzelli G, Lathers D, Young MR, Lau
A, et al: Phase I adjuvant radiation with docetaxel in high-risk
head and neck cancer. Am J Clin Oncol. 32:396–400. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Fukada J, Shigematsu N, Takeda A, Ohashi
T, Tomita T, Shiotani A, Kunieda E, Kawaguchi O, Fujii M and Kubo
A: Weekly low-dose docetaxel-based chemoradiotherapy for locally
advanced oropharyngeal or hypopharyngeal carcinoma: A
retrospective, single-institution study. Int J Radiat Oncol Biol
Phys. 76:417–424. 2010. View Article : Google Scholar
|
|
27
|
Onishi H, Kuriyama K, Yamaguchi M,
Komiyama T, Tanaka S, Araki T, Nishikawa K and Ishihara H:
Concurrent two-dimensional radiotherapy and weekly docetaxel in the
treatment of stage III non-small cell lung cancer: A good local
response but no good survival due to radiation pneumonitis. Lung
Cancer. 40:79–84. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Brunsvig PF, Hatlevoll R, Berg R, Lauvvang
G, Owre K, Wang M and Aamdal S: Weekly docetaxel with concurrent
radiotherapy in locally advanced non-small cell lung cancer: A
phase I/II study with 5 years' follow-up. Lung Cancer. 50:97–105.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kunos CA, Sill MW, Buekers TE, Walker JL,
Schilder JM, Yamada SD, Waggoner SE, Mohiuddin M and Fracasso PM:
Low-dose abdominal radiation as a docetaxel chemosensitizer for
recurrent epithelial ovarian cancer: A phase I study of the
Gynecologic Oncology Group. Gynecol Oncol. 120:224–228. 2011.
View Article : Google Scholar :
|
|
30
|
Font A, Arellano A, Fernández-Llamazares
J, Casas D, Boix J, Cardenal J, Margelí M, Manzano JL, Abad A and
Rosell R: Weekly docetaxel with concomitant radiotherapy in
patients with inoperable oesophageal cancer. Clin Transl Oncol.
9:177–182. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Makino I, Okamoto K, Kinoshita J, Hayashi
H, Nakamura K, Oyama K, Nakagawara H, Fujita H, Tajima H, Takamura
H, et al: A pilot study of chemoradiotherapy with weekly docetaxel
for thoracic esophageal carcinoma with T4 and/or M1 lymph node
metastasis. World J Oncol. 2:252–258. 2011.
|
|
32
|
Kushida T, Nohara S, Yoshino K, Fujiwara
D, Ouchi K, Amano T, Isayama F, Tomita N, Iwanuma Y, Sasai K, et
al: Utility of weekly docetaxel combined with preoperative
radiotherapy for locally advanced esophageal cancer from
pathological analysis. Dis Esophagus. 27:368–373. 2014. View Article : Google Scholar
|
|
33
|
Moos PJ and Fitzpatrick FA: Taxanes
propagate apoptosis via two cell populations with distinctive
cytological and molecular traits. Cell Growth Differ. 9:687–697.
1998.PubMed/NCBI
|
|
34
|
Torres K and Horwitz SB: Mechanisms of
Taxol-induced cell death are concentration dependent. Cancer Res.
58:3620–3626. 1998.PubMed/NCBI
|
|
35
|
Hernández-Vargas H, Palacios J and
Moreno-Bueno G: Telling cells how to die: Docetaxel therapy in
cancer cell lines. Cell Cycle. 6:780–783. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang D, Yang R, Wang S and Dong Z:
Paclitaxel: New uses for an old drug. Drug Des Devel Ther.
8:279–284. 2014.PubMed/NCBI
|
|
37
|
Chaffey JT and Hellman S: Differing
responses to radiation of murine bone marrow stem cells in relation
to the cell cycle. Cancer Res. 31:1613–1615. 1971.PubMed/NCBI
|
|
38
|
Parshad R, Gantt R, Sanford KK and Jones
GM: Chromosomal radiosensitivity of human tumor cells during the G2
cell cycle period. Cancer Res. 44:5577–5582. 1984.PubMed/NCBI
|
|
39
|
Paoletti A, Giocanti N, Favaudon V and
Bornens M: Pulse treatment of interphasic HeLa cells with nanomolar
doses of docetaxel affects centrosome organization and leads to
catastrophic exit of mitosis. J Cell Sci. 110:2403–2415.
1997.PubMed/NCBI
|
|
40
|
Morse DL, Gray H, Payne CM and Gillies RJ:
Docetaxel induces cell death through mitotic catastrophe in human
breast cancer cells. Mol Cancer Ther. 4:1495–1504. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Pradier O, Rave-Fränk M, Lehmann J, Lücke
E, Boghun O, Hess CF and Schmidberger H: Effects of docetaxel in
combination with radiation on human head and neck cancer cells
(ZMK-1) and cervical squamous cell carcinoma cells (CaSki). Int J
Cancer. 91:840–845. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Clarke SJ and Rivory LP: Clinical
pharmacokinetics of docetaxel. Clin Pharmacokinet. 36:99–114. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Bruno R and Sanderink GJ: Pharmacokinetics
and metabolism of Taxotere (docetaxel). Cancer Surv. 17:305–313.
1993.PubMed/NCBI
|
|
44
|
Brunsvig PF, Andersen A, Aamdal S,
Kristensen V and Olsen H: Pharmacokinetic analysis of two different
docetaxel dose levels in patients with non-small cell lung cancer
treated with docetaxel as monotherapy or with concurrent
radiotherapy. BMC Cancer. 7:1972007. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Milas L, Hunter NR, Mason KA, Kurdoglu B
and Peters LJ: Enhancement of tumor radioresponse of a murine
mammary carcinoma by paclitaxel. Cancer Res. 54:3506–3510.
1994.PubMed/NCBI
|
|
46
|
Milas L, Hunter NR, Mason KA, Milross CG,
Saito Y and Peters LJ: Role of reoxygenation in induction of
enhancement of tumor radioresponse by paclitaxel. Cancer Res.
55:3564–3568. 1995.PubMed/NCBI
|
|
47
|
McIlwrath AJ, Vasey PA, Ross GM and Brown
R: Cell cycle arrests and radiosensitivity of human tumor cell
lines: Dependence on wild-type p53 for radiosensitivity. Cancer
Res. 54:3718–3722. 1994.PubMed/NCBI
|