Introduction
Among childhood cancers, brain tumors account for
almost one-third of all cancer-related deaths. Medulloblastomas
(MBs) demonstrate a poor prognosis despite advanced therapies
consisting of surgery, radiotherapy and chemotherapy, sometimes
followed by stem cell transplantation (1,2). The
long-term survival (>5 years) rate of MB patients with high-risk
disease is 60–65% (3). MB patients
often experience complications related to aggressive treatment
regimens, including developmental, neurological and psychosocial
deficits. There is thus an unmet medical need for the development
of new and effective treatments with minimal damage to the
developing brain in these children (4). MBs develop in the cerebellum or
brainstem and spread through the cerebrospinal fluid along the
neuroaxis, but rarely to organs outside the central nervous system
(CNS). The etiology of MB is not fully understood but the impact of
environmental factors such as diet, pathogens, exposure to
radiation and hereditary genetic defects have previously been
reported (5). Together with
clinical factors (e.g. subtotal tumor resection), receptor tyrosine
kinase (RTK) expression and loss of genetic material at chromosome
17p have been associated with a poor prognosis.
Two of the four currently recognized clinical
variants of MB involve the WNT/β-catenin and the Sonic hedgehog
(SHH) pathways (6). SHH signaling
synergizes with the insulin-like growth factor 2 (IGF-II) to
promote MB as well as cerebellar neural precursor cell
proliferation (7). The two
non-WNT/SHH MB variants, with particularly unfavorable prognosis,
are designated as group 3 and 4 tumors, respectively, and exhibit
frequent expression of follistatin (FSTL5) (8), elevated Myc (9) and GFI1 (10) expression. Although a rare finding,
recent data have demonstrated that MB presents with
subgroup-related mutations, some of which have direct epigenetic
effects such as regulation of histone H3K27 and H3K4 methylation as
evident for group 3 and 4 MBs (11). These data underscore the importance
of epigenetic mechanisms and the notion that environmental factors
(also during fetal life) working through epigenetic mechanisms may
be the underlying factors for the disease etiology. Indeed, recent
comprehensive epigenetic analyses of MBs emphasize the importance
of epigenetic alterations in the pathogenesis of the various MB
subtypes (12,13).
Specific changes in DNA methylation is a hallmark of
many cancers and can cause genomic instability and/or changes in
gene expression (14,15). In vertebrates, DNA methylation is
mediated by DNA methyltransferase enzymes (DNMTs), through transfer
of a methyl group from S-adenosylmethionine (SAM) to the C-5
position of cytosine residues in CpG dinucleotide sequences
(16,17). In mammals, the DNMT family has
three active members: DNMT-1, which is required for the maintenance
of methylation in the genome through the replication process, and
DNMT3A and DNMT3B, which are regarded as de novo
methyltransferases (18).
During recent years HCMV protein and nucleic acid
have been detected in several types of tumor tissues, including
glioblastoma, neuroblastoma and medulloblastoma (19–22).
HCMV is a widespread β-herpesvirus carried by 60-95% of adults
worldwide in both developing and developed countries and
establishes a lifelong latency and persistence in myeloid lineage
cells with periodic reactivation (23). HCMV infection is generally
asymptomatic in immunocompetent hosts, but active viral infection
is of considerable clinical importance in immunocompromised
individuals such as organ transplant recipients, AIDS patients, and
in congenitally infected newborns (24,25).
There is currently a poor understanding of the epigenetic
mechanisms of host cells involved in HCMV infection. It has been
reported that herpesvirus chromatin undergoes dynamic changes in
structure and histone modifications during different stages of
viral replication, latent infection and reactivation. Epigenetic
events and states may hence dictate the outcome of infection in
permissive cells (26,27). Furthermore, previous studies have
suggested a role for epigenetic alterations in HCMV latency by
histone deacetylation and chromatin condensation (26). We previously reported that HCMV
infection alters the DNA methylation machinery in susceptible
non-tumor cells and changes the intracellular localization of DNMTs
in vitro, resulting in a profound reduction in DNA
methylation capacity (27).
In the present study, we sought to explore the
effect of HCMV infection on DNMT-1 in MB cells and tumor biopsies
as well as in human endothelial cells since vessels within the MB
tumors most often express HCMV proteins and are important for
tumorigenesis. Our data demonstrated that cytoplasmic localization
of DNMT-1 in HCMV-infected MB and endothelial cells was associated
with expression of HCMV late gene UL-55 (glycoprotein B, gB). An
increased number of cells expressing viral proteins in
5-Azacytidine (5AZA)-treated infected cells suggest that HCMV
replication may benefit from inhibition of the host cell nuclear
methylation machinery.
Materials and methods
Cell culture and HCMV infection
The human medulloblastoma cell line D324 (American
Type Culture Collection (ATCC); LGC Standards, Teddington, UK) was
cultured in Dulbecco's modified Eagle's medium (DMEM) supplemented
with 10% heat-inactivated fetal bovine serum (FBS), 100 units
penicillin and 100 µg streptomycin (all from Gibco BRL;
Thermo Fisher Scientific, Inc., Waltham, MA, USA) at 37°C in a
humidified 5% CO2 atmosphere. Human umbilical vein
endothelial cells (HUVECs, ATCC) were grown in EBM2 endothelial
basal medium supplemented with EGM-2 SingleQuots®
(Clonetics™; Lonza, Walkersville, MD, USA). Both D324 cells and
HUVECs were infected at 80% confluency with the HCMV clinical
strain VR1814 (a kind gift from Dr Giuseppe Gerna, University of
Pavia, Pavia, Italy) at a multiplicity of infection (MOI) of 5, for
72 h. All experiments in this study were performed as three
independent experiments.
5-Azacytidine treatment (5AZA)
D324 cells and HUVECs were seeded in 8-well chamber
slides (VWR, Radnor, PA, USA), and at ~80% confluency, the cells
were left untreated or treated with 5AZA (10 µM for D324
cells and 2 µM for HUVECs) (Sigma, Stockholm, Sweden) for 3
days. Untreated and 5AZA-treated cells were left uninfected or
infected with HCMV-VR1814 at MOI 5, for 72 h. At 3 days
post-infection (3 dpi), the cells were fixed in ice-cold
acetone:methanol (1:1) for 10 min and kept at −20°C.
Ganciclovir treatment
Uninfected and HCMV-infected D324 cells and HUVECs
were left untreated or treated with 2 mM ganciclovir
(Cymevene®, Roche, Basel, Switzerland) starting at the
time of infection of the cells. Cells were fixed as described
above.
Immunofluorescence staining
Immunofluorescence staining of the cells was
performed as previously described, with minor modifications
(27). After blocking of
non-specific binding with protein blocker (Dako, Glostrup,
Denmark), the cells were incubated with primary antibodies for 1 h
at 4°C. Binding of the primary antibodies was detected using
fluorochrome-conjugated secondary antibodies incubated for 45 min
at room temperature (RT). The nuclei of the cells were visualized
using 4′,6-diamidino-2-phenylindole (DAPI, Vectashield, Vector
Laboratories, Burlingame, CA, USA), and coverslips were mounted
using Dako Fluorescence Mounting Medium (Agilent Technologies,
Inc., Santa Clara, CA, USA). The following primary and secondary
antibodies were used for detection of specific target proteins:
mouse anti-immediate early monoclonal antibody (at dilution 1:100,
11-003, Argene, Verniolle, France); mouse monoclonal antibody to gB
(dilution 1:50, P1216, Virusys Corporation, Taneytown, MD, USA);
rabbit antibody to DNMT-1 (dilution 1:500, 19905, Abcam, Cambridge,
UK); Alexa Fluor-488-conjugated goat anti-mouse (dilution 1:500,
A11001, Life Technologies, Eugene, OR, USA); and Texas red
conjugated goat anti-rabbit (dilution 1:500; Life Technologies,
T2767). Omitting primary antibodies was used as the negative
control.
Evaluation of the staining was performed with
confocal microscope (Leica TCS SP5) equipped with Leica Application
Suite Advanced Fluorescence software (Leica Microsystems, Wetzlar,
Germany).
RNA extraction and quantitative TaqMan
PCR (qPCR)
RNA was isolated from the cultured cells using
Allprep DNA/RNA/protein mini kit and/or RNeasy kit (Qiagen,
Stockholm, Sweden) according to the manufacturer's instructions.
The concentrations of RNA were measured using NanoDrop ND-1000
spectrophotometer (Thermo Scientific). Isolated RNA (500 ng) was
converted to cDNA using Superscripts III First-Strand Synthesis
System (Invitrogen, Stockholm, Sweden) using random hexamers
according to the manufacturer's application manual.
TaqMan Fast Universal PCR Master Mix (Life
Technologies; Thermo Fisher Scientific) was used with the following
primers/probes: HCMV-IE (forward primer, TGACGAGGGCCCTTCCT; reverse
primer, CCTTGGTCACGGGTGTCT and probes, FAM-AAGGTGCCACGGCCCG-NFQ)
and HCMV-gB (forward primer, GCTACCGCCCTACCTCAAG; reverse primer,
CGCCAACGGCCTTTCC and probes, FAM-CCCAGGCCGCTCATG-NFQ) and DNMT-1
(assay ID, Hs00945875_m1, Life Technologies; Thermo Fisher
Scientific). RNase P (assay ID, 4316844, Life Technologies, Thermo
Fisher Scientific) and β2-microglobulin (B2M, assay ID,
Hs00984230_m1, Thermo Fisher Scientific) were used for
normalization. The qRT-PCR was performed using an Applied
Biosystems 7900HT fast real-time PCR system (Thermo Fisher
Scientific) and the results were analyzed with SDS 2.4 software
(Applied Biosystems; Thermo Fisher Scientific). The ΔCT method was
used for calculation of CT values for different transcripts. The
2−ΔΔCT method was used to quantify relative fold
changes.
Quantification of DNMT-1
The concentration of DNMT-1 in cell lysates was
measured using the DNMT-1 human ELISA kit (SKU: K4195, BioVision
Incorporated, Milpitas, CA, USA) in accordance with the
manufacturer's instructions.
Immunohistochemical staining (IHC)
Paraffin-embedded human MB tumor tissue sections
from 5 patients were obtained from the Department of Pathology at
Rigshospitalet, Copenhagen, Denmark. Information regarding the
tumor characteristics is documented in Table I.
 | Table ICharacteristics of the
medulloblastoma tumors. |
Table I
Characteristics of the
medulloblastoma tumors.
| Patients | Initial
diagnosis | New classification
- immunoreactivity | New classification
- subgroup type |
|---|
| Patient 1 | Classical MB, that
recurred in a supratentorial supratentorial location the year
after. A primitive neuroectodermal tumor is considered | Membranous
β-catenin reactivity. YAP-1 negative. P53 wild-type | Non-WNT/non-SHH
(group 3/4) |
| Patient 2 | Recurring classical
MB. No information about location | Membranous and
nuclear β-catenin reactivity. YAP-1 positivity in areas. P53
wild-type | WNT-activated |
| Patient 3 | Autopsy:
Metastasizing classical MB which had spread to the cerebrum, in the
areas around the lateral ventricles and in the pons and
cerebellum | Membranous
β-catenin reactivity. YAP-1 negative. P53 wild-type | Non-WNT/non-SHH
(group 3/4) |
| Patient 4 | Classical MB | Membranous and
nuclear β-catenin reactivity. YAP-1 positivity in areas. P53
wild-type | WNT-activated |
| Patient 5 | Classical MB. No
loss of chromosome 1p/19q | Membranous
β-catenin reactivity. YAP-1 positivity. P53 wild-type | SHH-activated, p53
wild-type |
Ethical permission for the present study protocol
was granted by the Local Ethics Committee at Region Hovedstaden,
Denmark (H-6-2014-010) and the Local Ethics Committee at Karolinska
Institutet, Sweden (Dnr. 2008/628-31).
Immunohistochemical staining of the tissues was
performed as previously described (19,20).
Briefly, all sections were first deparaffinized in xylene and
rehydrated in a series of decreasing concentrations of ethanol and
were then treated with pepsin (BioSite, Täby, Sweden) and citrate
buffer (Biogenex, CA, USA) to retrieve the epitopes. Endogenous
peroxidase activity was blocked with 3% H2O2
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany). In addition, the
sections were treated with Avidin/Biotin blocker (Dako
Avidin/Biotin Blocking kit; Agilent Technologies), Fc receptor
blocker and Background Buster (Innovex Biosciences, Richmond, CA,
USA) to eliminate non-specific binding. The slides were incubated
with the primary antibodies: HCMV, IE (MAB810R, Merck, Stockholm,
Sweden), gB (a kind gift from Dr William Britt, University of
Alabama, AL, USA) or DNMT-1 (19905, Abcam), at 4°C overnight and
after washing the specific epitopes were detected with ImmPRESS
reagent kits (MP-7401 and MP-7402, Vector Laboratories) and
diaminobenzidine (DAB, NB314D, Innovex Biosciences). Omitting the
primary antibodies was used as the negative control. Hamamatsu Nano
Zoomer-XR Digital slide scanner C12000 with visualization using
Nano Zoomer Digital Pathology (NDP) viewer software (U12388-01;
NDP.view2 Viewing) was used for scanning of the sections.
Statistical analysis
All analyses were performed using GraphPad Prism
(GraphPad Software, San Diego, CA, USA). P<0.05 was considered
to indicate a statistically significant difference. Unpaired t-test
and Mann-Whitney U test were used to assess the statistical
significance between different variables.
Results
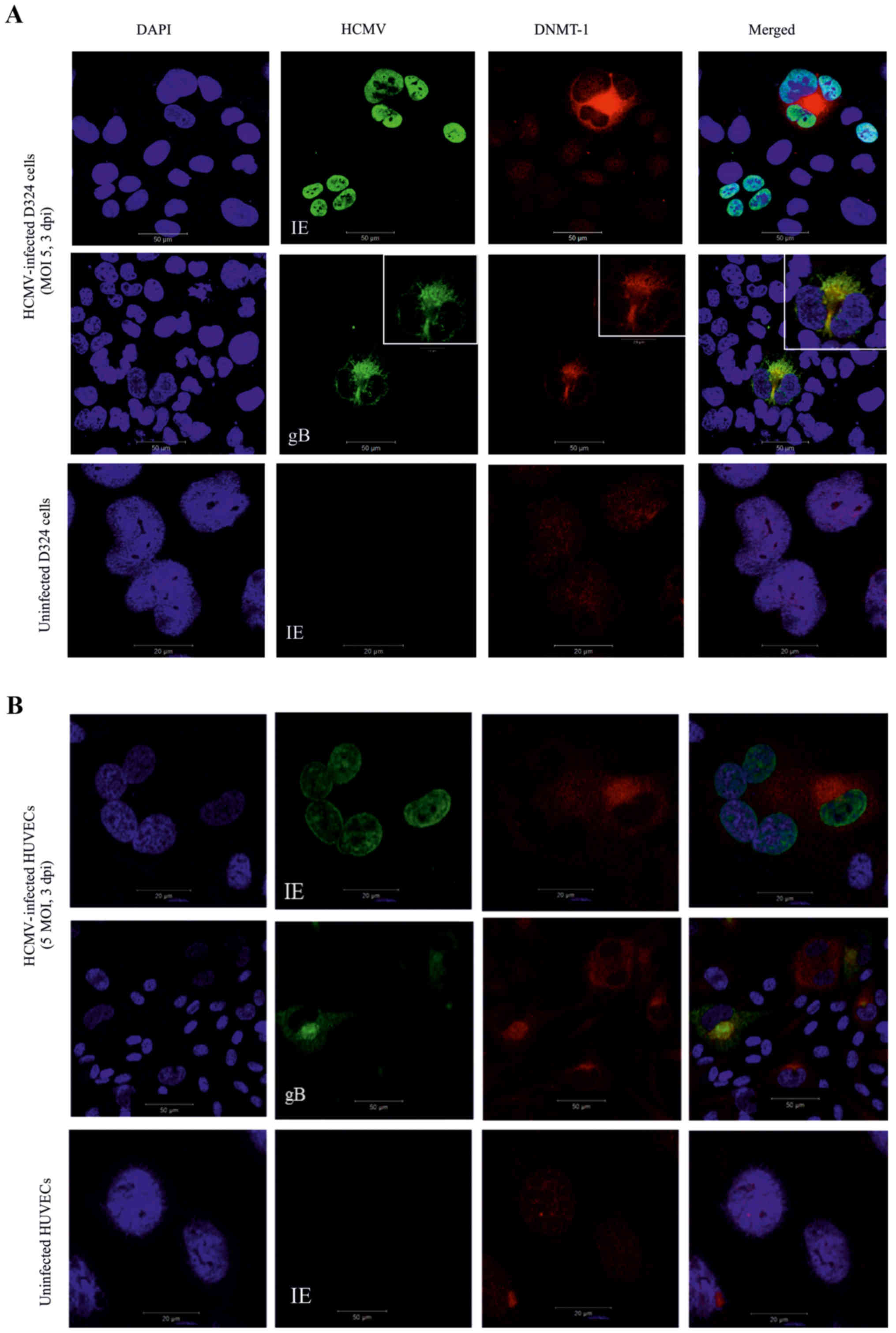
Cytoplasmic location of DNMT-1 in the
HCMV-gB-expressing cells
We examined DNMT-1 protein expression and
localization in the uninfected and HCMV-infected MB D324 cells and
HUVECs. HUVECs were included in this study since endothelial cells
of blood vessels are a major reservoir of HCMV with the capacity to
influence hemostasis and co-infection of other cells through
cell-cell contact. DNMT-1 was weakly visible in the nucleus of all
uninfected cells and in infected cells that expressed only HCMV-IE,
but was almost exclusively localized in the cytoplasm of
HCMV-gB-positive cells (Fig. 1A and
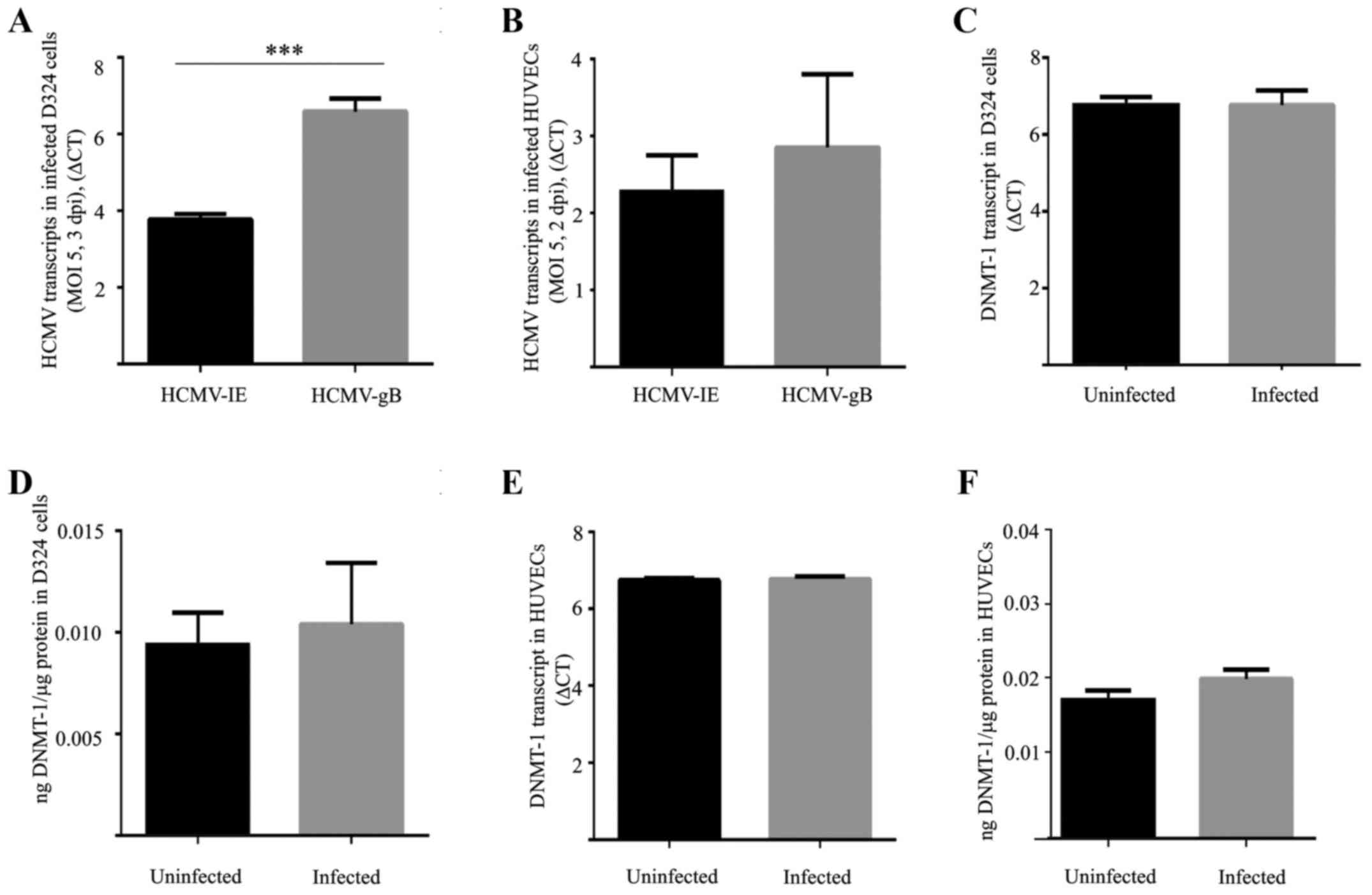
B). qPCR analysis revealed a significantly higher number of
HCMV-gB transcripts compared to HCMV-IE transcripts in infected
D324 cells (P=0.0002, Fig. 2A).
Equal numbers of transcripts of HCMV-IE and HCMV-gB were detected
in HUVECs (Fig. 2B). For DNMT-1,
neither the transcripts nor the protein levels of DNMT-1 differed
between uninfected and HCMV-infected cells (Fig. 2C–F).
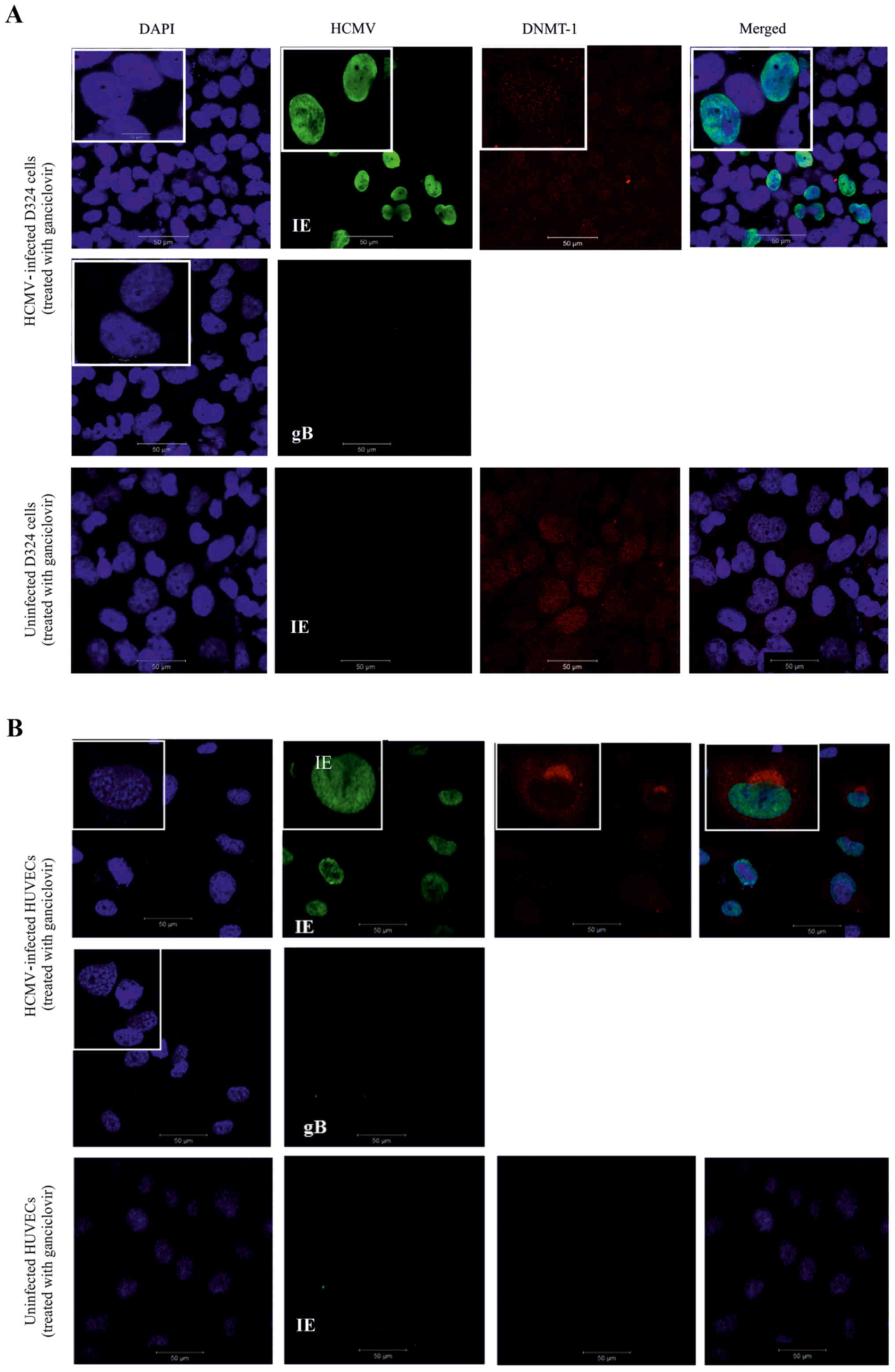
Maintenance of DNMT-1 nuclear
localization in ganciclovir-treated HCMV-infected D324 cells but
not in HUVECs
HCMV-infected D324 cells and HUVECs were treated
with ganciclovir. As expected, HCMV-IE protein was expressed while
HCMV late gene expression (represented by HCMV-gB) was inhibited in
the treated cells (Fig. 3A and B).
In HCMV-infected and ganciclovir-treated D324 cells, DNMT-1 was
only expressed in the nucleus of the cells (Fig. 3A). In contrast, the HCMV-infected
HUVECs did not respond to ganciclovir in the same way, but
displayed a cytoplasmic DNMT-1 localization similar to the
untreated cells (Fig. 3B).
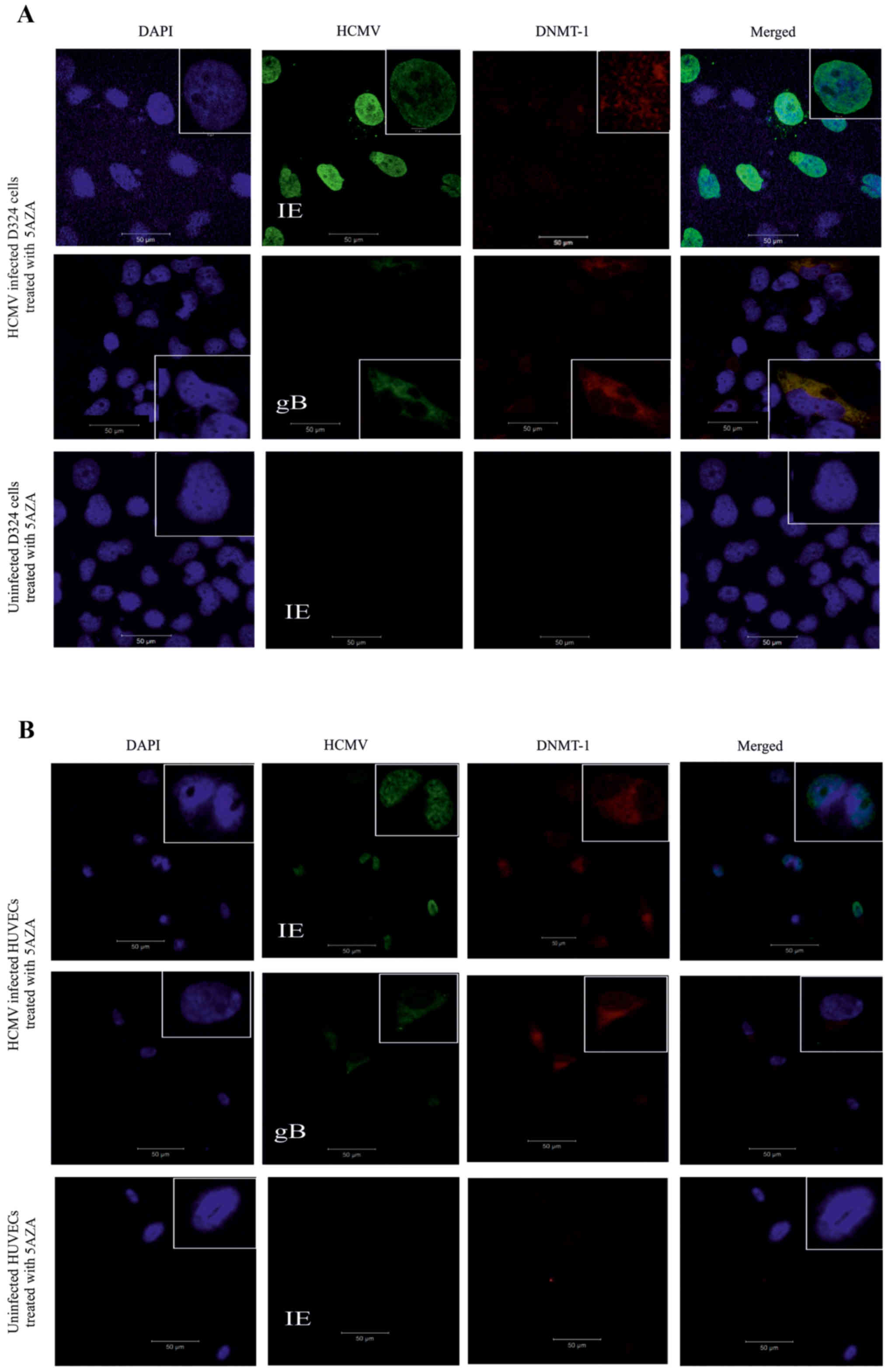
HCMV and DNMT-1 gene and protein
expression in the 5AZA-treated D324 cells and HUVECs
Uninfected and HCMV-infected D324 cells and HUVECs
were treated with 5AZA and analyzed for the expression of HCMV and
DNMT-1 proteins and transcripts (Fig.
4). In HCMV-infected and 5AZA -treated D324 cells the
proportion of HCMV-IE-positive cells was significantly increased
compared to the untreated cells (P=0.01) (Fig. 4C) while the proportion of
HCMV-gB-positive cells was not significantly increased (P=0.09)
(Fig. 4C). Based on qPCR analysis,
the expression of HCMV-IE and HCMV-gB transcripts was significantly
increased by 5AZA treatment in the D324 cells compared to those in
the untreated cells (P=0.0002, P=0.0003, respectively) (Fig. 4D). The number of HCMV-IE and
HCMV-gB protein-positive HUVECs were significantly increased
following 5AZA treatment, indicating enhanced infection efficiency
(P=0.0005 and P=0.04, respectively) (Fig. 4E). HCMV-IE and HCMV-gB transcripts
were significantly increased in the 5AZA-treated HCMV-infected
HUVECs compared to those in the untreated cells (P=0.004, P=0.008,
respectively) (Fig. 4F).
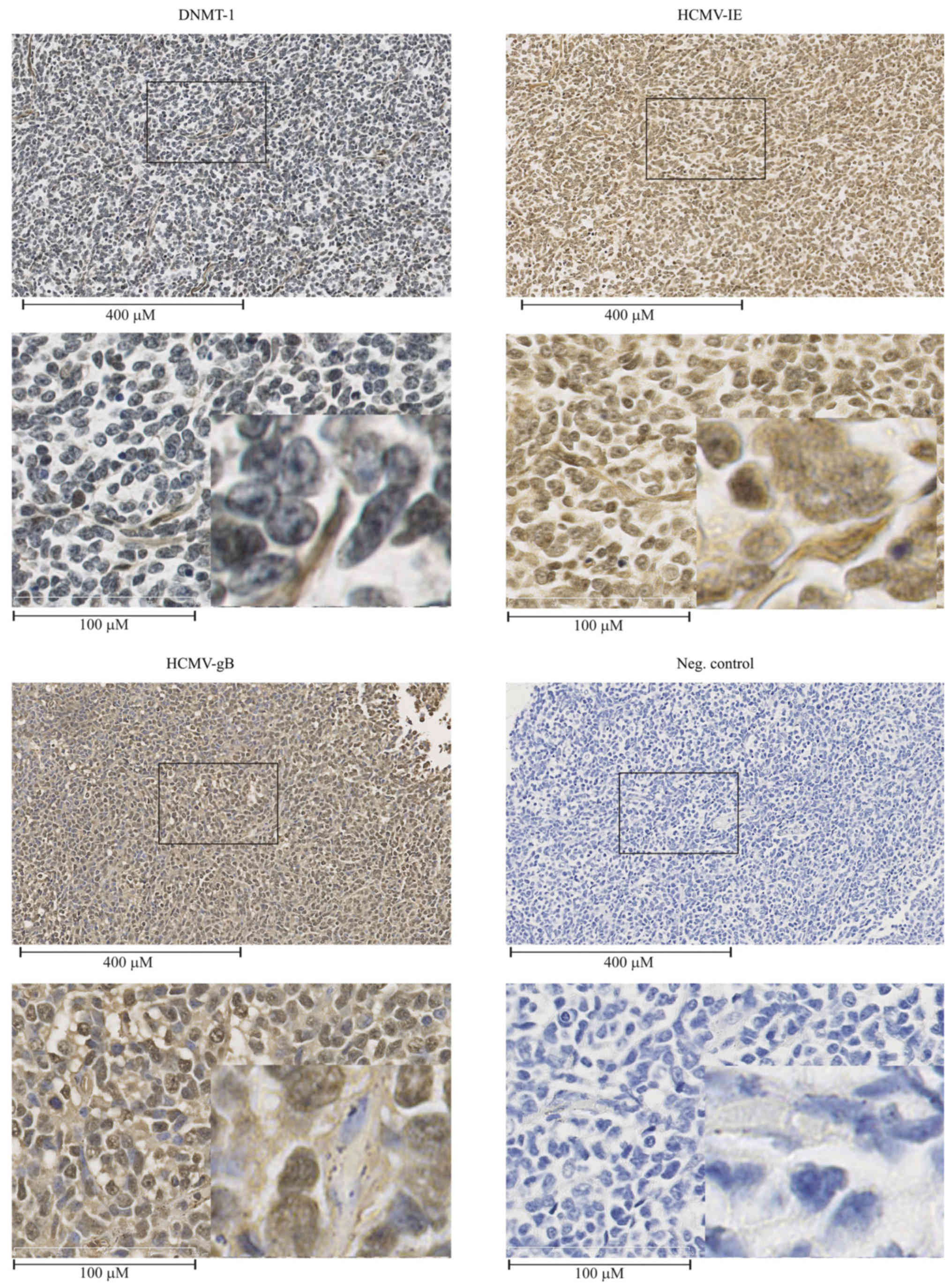
Frequent detection of HCMV-IE, HCMV-gB
and DNMT-1 in medulloblastoma tissue specimens
MB tissue sections were immunohistochemically
analyzed for HCMV-IE, HCMV-gB and DNMT-1. HCMV-IE and HCMV-gB
proteins were frequently detected at different levels in all the
examined MB tissues. Fig. 5
depicts one of the cases (patient no. 5, Table I). While DNMT-1 was detected in the
nucleus of the majority of the tumor cells, cytoplasmic expression
of DNMT-1 was detected in vessel walls within the tumors and in a
few tumor cells within the tissues (Fig. 5).
Discussion
In the present study, we examined the
interdependence of HCMV-IE, HCMV-gB proteins and DNMT-1 in MB
cells. In concordance with our previous report of human fibroblasts
(27), HCMV-infected D324 cells
and HUVECs exhibited cytoplasmic localization of DNA
methyltransferase DNMT-1, which correlated with the expression of
HCMV late protein gB (UL55). Interestingly, while not all cells
expressing IE genes had DNMT-1 located in the cytoplasm, all cells
expressing the late gene product gB displayed cytoplasmic DNMT-1.
Furthermore, ganciclovir treatment (which inhibits expression of
late viral proteins) attenuated the cytoplasmic localization of
DNMT-1 in HCMV-infected D324 cells, indicating an involvement of
HCMV late genes/products in the extranuclear accumulation.
Conversely, HCMV-infected HUVECs treated with ganciclovir still
maintained a cytoplasmic distribution of DNMT-1, implying the
involvement of different mechanisms in this process. We hence
suggest an association between HCMV infection in medulloblastoma
(MB) and endothelial cells and the epigenetic modifier enzyme
DNMT-1.
Tumor cells are generally characterized by global
hypomethylation, and local hypermethylation of certain gene
promoters such as tumor-suppressor genes (28). The relationship between DNA
methylation alteration and human diseases, and its role in
carcinogenesis has been established (15). DNMT-1 is required for the
maintenance of DNA methylation in the genome (29). Its cytoplasmic localization in
HCMV-infected cells might thus prevent normal gene methylation
maintenance, which may influence the cellular behavior important
for tumorigenesis and tumor development. During the past decade the
impact of epigenetic modification in MB has been intensively
discussed and investigators have reported epigenetic alterations in
candidate tumor-suppressor genes including RASSF1A, CASP8, and HIC1
in >30% of MB (30). Epigenetic
silencing of tumor-suppressor genes as a contributor to
tumorigenesis and development of MB thus emphasizes the importance
of epigenetic mechanisms for novel diagnostic and therapeutic
approaches (12).
Epigenetic mechanisms, such as methylation of DNA
cytosine residues and post-translational modifications of histone
proteins associated with DNA, control the structure and
transcriptional permissiveness of chromatin. The crosstalk between
DNA methylation and histone modification is a basis for regulation
of the functional genome. If these mechanisms run rogue, as in
malignant cells, they may force cells into fixed de-differentiated
states. The initial epigenetic errors may arise through alterations
of mechanisms triggered by external impact, such as chemical
agents, inflammation, or pathogens such as viruses, as we and
others have previously reported (27,31,32).
It is thus of great importance to understand which external factors
may manipulate the epigenome in such a way that the target cells
respond abnormally to their environment or become reprogrammed. In
addition, the original notion that DNA methylation is a guardian
against invading foreign nucleic acids provides a logical
explanation to a viral mechanism keeping newly replicated virus DNA
methylation-free by preventing DNMTs from functioning in the
nucleus.
In agreement with the idea that DNA methylation
might prevent viral replication and/or transcription, the
inhibition of methylation by 5AZA is expected to influence
active viral infection. Indeed, the number of HCMV-positive cells
was increased following 5AZA treatment in both D324 cells
and HUVECs, suggesting the benefits of a hypomethylated milieu for
viral production. This may have important implications for the
treatment of MB with epigenetic drugs, since an increased number of
HCMV-IE- and HCMV-gB-positive cells could contribute to the
oncomodulatory effects of HCMV infection in MB. HCMV-IE proteins
are viral regulatory proteins and transcription factors that act as
oncomodulatory proteins through different mechanisms such as
causing instability in chromosomes 1q42 and 1q21 (33,34),
interfering with p53, controlling p21 degradation and
phosphorylated retinoblastoma (pRb) proteins. These oncomodulatory
activities result in uncontrolled cellular proliferation and
transition to the S phase of the cell cycle (35,36).
A still unanswered question is whether 5AZA treatment alters
the host methylome and thereby its gene regulation. Although host
cell replication is attenuated by HCMV infection and passive
demethylation of the host genome cannot take place, as we
demonstrated for non-tumor cell types infected with HCMV (27), it is still possible that
5AZA can induce active demethylation through the induction
of Tet enzymes, and could therefore cause changes to the methylome
of the host cells. This issue will be further investigated.
Inflammation is one of the recently established
hallmarks of cancer, and in theory the relocation of DNMT-1 into
the cytoplasm of HCMV-infected endothelial cells could contribute
to increased induction of inflammatory factors important for
vascularization and angiogenesis, such as VEGF, FGF, IL-8 and IL-6.
Inflammation is in fact one of the key factors in the progression
of MB, and anti-inflammatory drugs including the COX2 inhibitor
celecoxib has been suggested to be used in these patients (37). Importantly, HCMV infection of the
cells in vitro leads to increased induction of COX2, and
COX2 inhibitors reduce viral replication (38). Furthermore, we previously reported
substantial reduction in tumor growth by the antiviral drug
Valcyte® and by celecoxib, both in vitro and
in vivo (19).
In conclusion, DNMT-1 localized to the cytoplasm in
HCMV-infected MB and endothelial cells expressing HCMV late genes.
Increased viral replication in 5AZA-treated infected cells suggests
that HCMV replication benefits from a lack of DNA methylation
activity in host cells. The concept of epigenetic therapy is
currently under consideration for MB (39), and since our findings raise the
possibility that epigenetic modulation can directly impact the
level of viral replication, this should be considered in the design
of epigenetic MB therapies.
Acknowledgments
The authors would like to acknowledge the Swedish
Cancer Foundation, the Swedish Children Cancer Foundation, the
Swedish Society for Medical Research (SLS), Goljes Memory
Foundation, Magnus Bergvalls Foundation, Swedish Society for
Medical Research (SSMF), the Karolinska Institutet Foundations and
Tore Nilsson's Foundation.
Notes
[1] Competing
interests
The authors declare that they have no competing
interests.
References
|
1
|
Mulhern RK, Merchant TE, Gajjar A, Reddick
WE and Kun LE: Late neurocognitive sequelae in survivors of brain
tumours in childhood. Lancet Oncol. 5:399–408. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rood BR, Macdonald TJ and Packer RJ:
Current treatment of medulloblastoma: Recent advances and future
challenges. Semin Oncol. 31:666–675. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
American Society of Clinical Oncology
(ASCO): Medulloblastoma - Childhood: Statistics. https://www.cancer.net/cancer-types/medulloblastoma-childhood/statistics.
Accessed Aug, 2016.
|
|
4
|
Oeffinger KC and Hudson MM: Long-term
complications following childhood and adolescent cancer:
Foundations for providing risk-based health care for survivors. CA
Cancer J Clin. 54:208–236. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
de Bont JM, Packer RJ, Michiels EM, den
Boer ML and Pieters R: Biological background of pediatric
medulloblastoma and ependymoma: A review from a translational
research perspective. Neuro-oncol. 10:1040–1060. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Thompson MC, Fuller C, Hogg TL, Dalton J,
Finkelstein D, Lau CC, Chintagumpala M, Adesina A, Ashley DM,
Kellie SJ, et al: Genomics identifies medulloblastoma subgroups
that are enriched for specific genetic alterations. J Clin Oncol.
24:1924–1931. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hartmann W, Koch A, Brune H, Waha A,
Schüller U, Dani I, Denkhaus D, Langmann W, Bode U, Wiestler OD, et
al: Insulin-like growth factor II is involved in the proliferation
control of medulloblastoma and its cerebellar precursor cells. Am J
Pathol. 166:1153–1162. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Remke M, Hielscher T, Korshunov A,
Northcott PA, Bender S, Kool M, Westermann F, Benner A, Cin H,
Ryzhova M, et al: FSTL5 is a marker of poor prognosis in
non-WNT/non-SHH medulloblastoma. J Clin Oncol. 29:3852–3861. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kawauchi D, Robinson G, Uziel T, Gibson P,
Rehg J, Gao C, Finkelstein D, Qu C, Pounds S, Ellison DW, et al: A
mouse model of the most aggressive subgroup of human
medulloblastoma. Cancer Cell. 21:168–180. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Northcott PA, Lee C, Zichner T, Stütz AM,
Erkek S, Kawauchi D, Shih DJ, Hovestadt V, Zapatka M, Sturm D, et
al: Enhancer hijacking activates GFI1 family oncogenes in
medulloblastoma. Nature. 511:428–434. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Robinson G, Parker M, Kranenburg TA, Lu C,
Chen X, Ding L, Phoenix TN, Hedlund E, Wei L, Zhu X, et al: Novel
mutations target distinct subgroups of medulloblastoma. Nature.
488:43–48. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hovestadt V, Jones DT, Picelli S, Wang W,
Kool M, Northcott PA, Sultan M, Stachurski K, Ryzhova M, Warnatz
HJ, et al: Decoding the regulatory landscape of medulloblastoma
using DNA methylation sequencing. Nature. 510:537–541. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lin CY, Erkek S, Tong Y, Yin L, Federation
AJ, Zapatka M, Haldipur P, Kawauchi D, Risch T, Warnatz HJ, et al:
Active medulloblastoma enhancers reveal subgroup-specific cellular
origins. Nature. 530:57–62. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Watanabe Y and Maekawa M: Methylation of
DNA in cancer. Adv Clin Chem. 52:145–167. 2010. View Article : Google Scholar
|
|
15
|
Feinberg AP, Koldobskiy MA and Göndör A:
Epigenetic modulators, modifiers and mediators in cancer aetiology
and progression. Nat Rev Genet. 17:284–299. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lister R, Pelizzola M, Dowen RH, Hawkins
RD, Hon G, Tonti-Filippini J, Nery JR, Lee L, Ye Z, Ngo QM, et al:
Human DNA methylomes at base resolution show widespread epigenomic
differences. Nature. 462:315–322. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ziller MJ, Gu H, Müller F, Donaghey J,
Tsai LT, Kohlbacher O, De Jager PL, Rosen ED, Bennett DA, Bernstein
BE, et al: Charting a dynamic DNA methylation landscape of the
human genome. Nature. 500:477–481. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rondelet G and Wouters J: Human DNA
(cytosine-5)-methyltransferases: A functional and structural
perspective for epigenetic cancer therapy. Biochimie. 139:137–147.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Baryawno N, Rahbar A, Wolmer-Solberg N,
Taher C, Odeberg J, Darabi A, Khan Z, Sveinbjörnsson B, FuskevÅg
OM, Segerström L, et al: Detection of human cytomegalovirus in
medulloblastomas reveals a potential therapeutic target. J Clin
Invest. 121:4043–4055. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rahbar A, Orrego A, Peredo I, Dzabic M,
Wolmer-Solberg N, Strååt K, Stragliotto G and Söderberg-Nauclér C:
Human cytomegalovirus infection levels in glioblastoma multiforme
are of prognostic value for survival. J Clin Virol. 57:36–42. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wolmer-Solberg N, Baryawno N, Rahbar A,
Fuchs D, Odeberg J, Taher C, Wilhelmi V, Milosevic J, Mohammad AA,
Martinsson T, et al: Frequent detection of human cytomegalovirus in
neuro-blastoma: A novel therapeutic target? Int J Cancer.
133:2351–2361. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bartek J Jr, Fornara O, Merchut-Maya JM,
Maya-Mendoza A, Rahbar A, Stragliotto G, Broholm H, Svensson M,
Sehested A, Söderberg Naucler C, et al: Replication stress, DNA
damage signalling, and cytomegalovirus infection in human
medulloblastomas. Mol Oncol. 11:945–964. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Taylor-Wiedeman J, Sissons JG, Borysiewicz
LK and Sinclair JH: Monocytes are a major site of persistence of
human cytomegalovirus in peripheral blood mononuclear cells. J Gen
Virol. 72:2059–2064. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Emery VC: Investigation of CMV disease in
immunocompromised patients. J Clin Pathol. 54:84–88. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gaytant MA, Steegers EA, Semmekrot BA,
Merkus HM and Galama JM: Congenital cytomegalovirus infection:
Review of the epidemiology and outcome. Obstet Gynecol Surv.
57:245–256. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu XF, Wang X, Yan S, Zhang Z, Abecassis
M and Hummel M: Epigenetic control of cytomegalovirus latency and
reactivation. Viruses. 5:1325–1345. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Esteki-Zadeh A, Karimi M, Strååt K,
Ammerpohl O, Zeitelhofer M, Jagodic M, Mehrab-Mohseni M, Sjöholm L,
Rahbar A, Söderberg-Nauclér C, et al: Human cytomegalovirus
infection is sensitive to the host cell DNA methylation state and
alters global DNA methylation capacity. Epigenetics. 7:585–593.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Estécio MR and Issa JP: Dissecting DNA
hypermethylation in cancer. FEBS Lett. 585:2078–2086. 2011.
View Article : Google Scholar
|
|
29
|
Kanherkar RR, Bhatia-Dey N and Csoka AB:
Epigenetics across the human lifespan. Front Cell Dev Biol.
2:492014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lindsey JC, Lusher ME, Anderton JA, Bailey
S, Gilbertson RJ, Pearson AD, Ellison DW and Clifford SC:
Identification of tumour-specific epigenetic events in
medulloblastoma development by hypermethylation profiling.
Carcinogenesis. 25:661–668. 2004. View Article : Google Scholar
|
|
31
|
Bayarsaihan D: Epigenetic mechanisms in
inflammation. J Dent Res. 90:9–17. 2011. View Article : Google Scholar :
|
|
32
|
Gómez-Díaz E, Jordà M, Peinado MA and
Rivero A: Epigenetics of host-pathogen interactions: The road ahead
and the road behind. PLoS Pathog. 8:e10030072012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cinatl J Jr, Vogel JU, Kotchetkov R and
Wilhelm Doerr H: Oncomodulatory signals by regulatory proteins
encoded by human cytomegalovirus: A novel role for viral infection
in tumor progression. FEMS Microbiol Rev. 28:59–77. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Fortunato EA, Dell'Aquila ML and Spector
DH: Specific chromosome 1 breaks induced by human cytomegalovirus.
Proc Natl Acad Sci USA. 97:853–858. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Prichard MN, Sztul E, Daily SL, Perry AL,
Frederick SL, Gill RB, Hartline CB, Streblow DN, Varnum SM, Smith
RD, et al: Human cytomegalovirus UL97 kinase activity is required
for the hyperphosphorylation of retinoblastoma protein and inhibits
the formation of nuclear aggresomes. J Virol. 82:5054–5067. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Herbein G and Kumar A: The oncogenic
potential of human cytomegalovirus and breast cancer. Front Oncol.
4:2302014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Baryawno N, Sveinbjörnsson B, Eksborg S,
Orrego A, Segerström L, Oqvist CO, Holm S, Gustavsson B, Kågedal B,
Kogner P and Johnsen JI: Tumor-growth-promoting cyclooxygenase-2
prostaglandin E2 pathway provides medulloblastoma therapeutic
targets. Neuro Oncol. 10:661–674. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhu H, Cong JP, Yu D, Bresnahan WA and
Shenk TE: Inhibition of cyclooxygenase 2 blocks human
cytomegalovirus replication. Proc Natl Acad Sci USA. 99:3932–3937.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
MacDonald TJ, Aguilera D and Castellino
RC: The rationale for targeted therapies in medulloblastoma.
Neuro-oncol. 16:9–20. 2014. View Article : Google Scholar
|