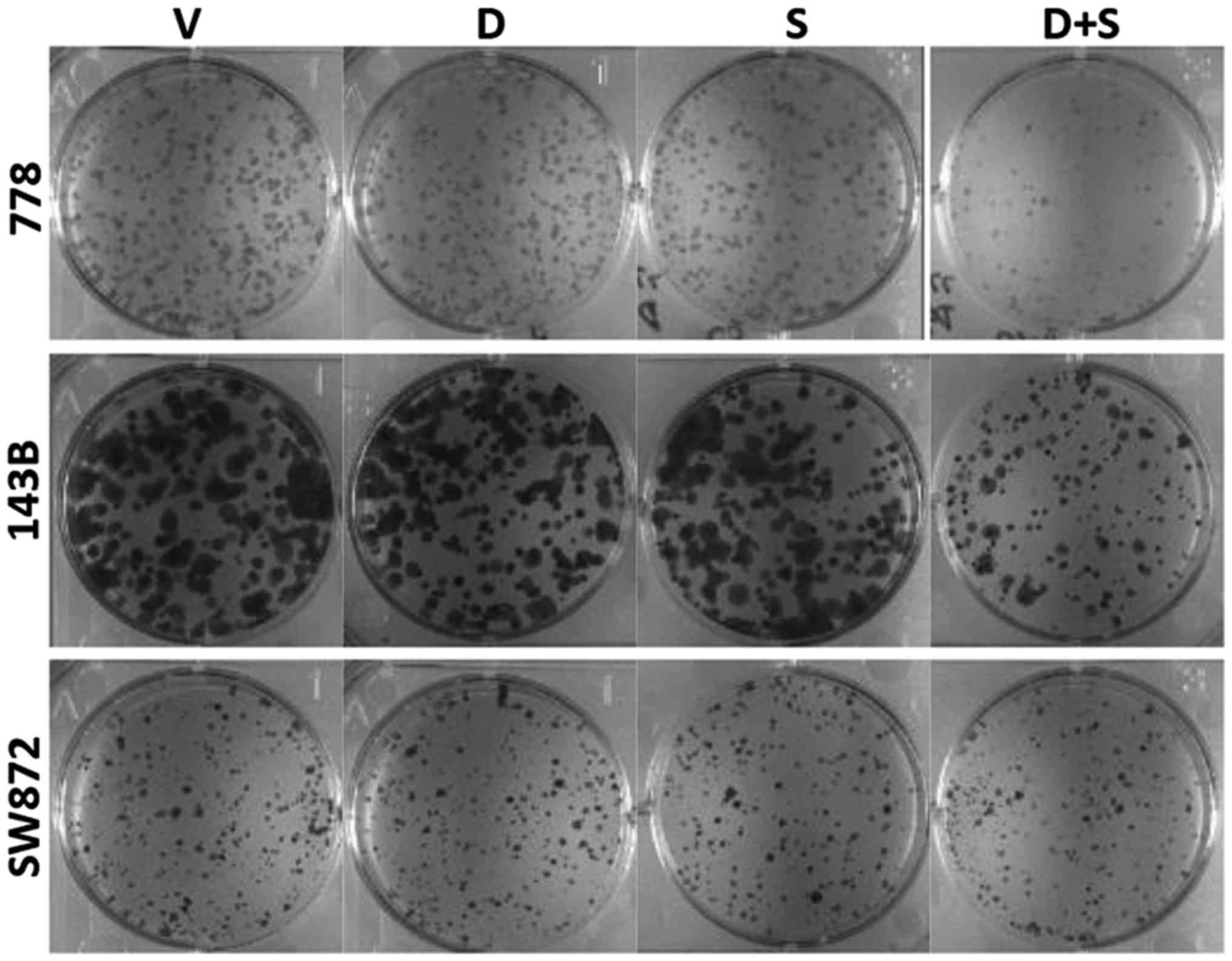
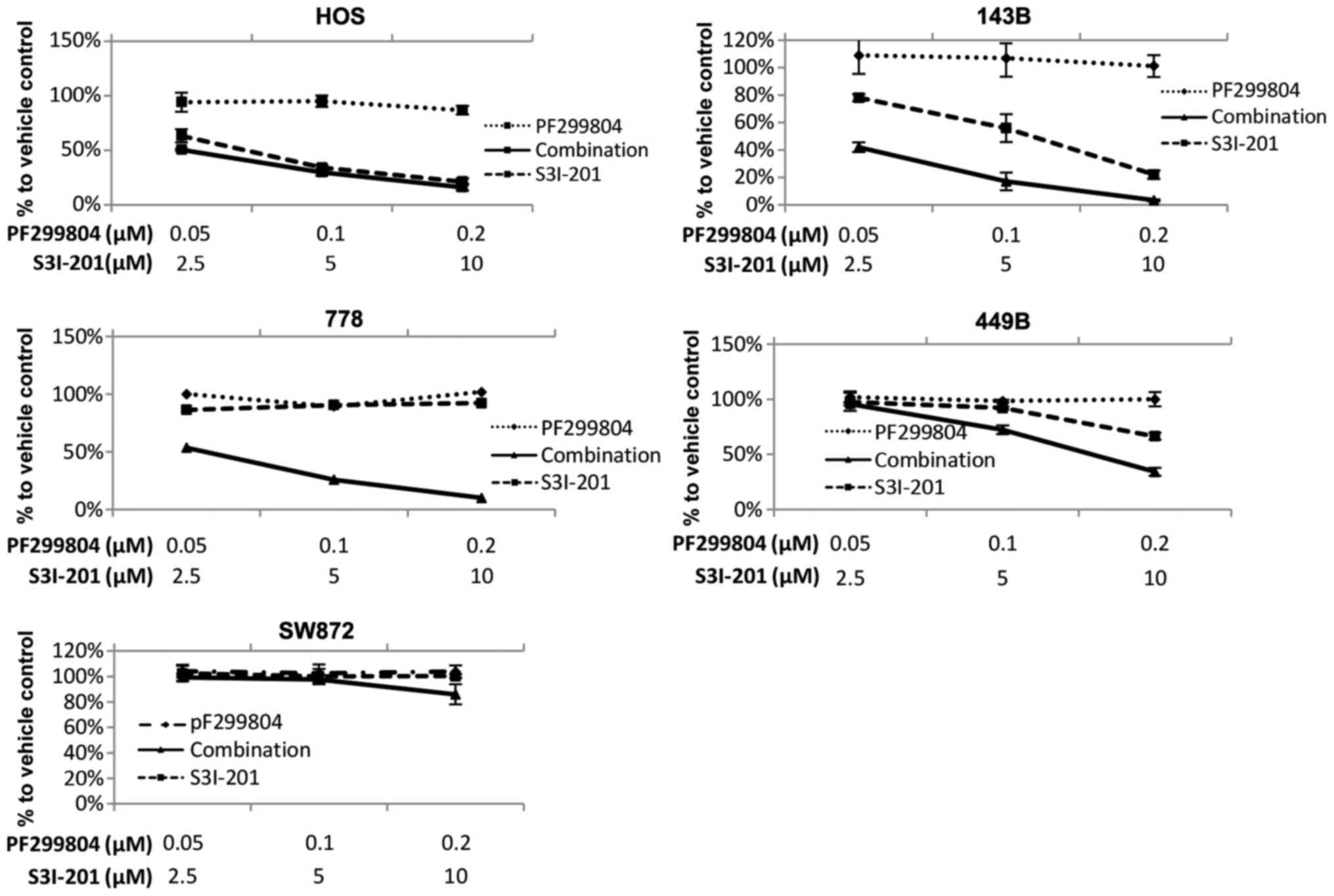
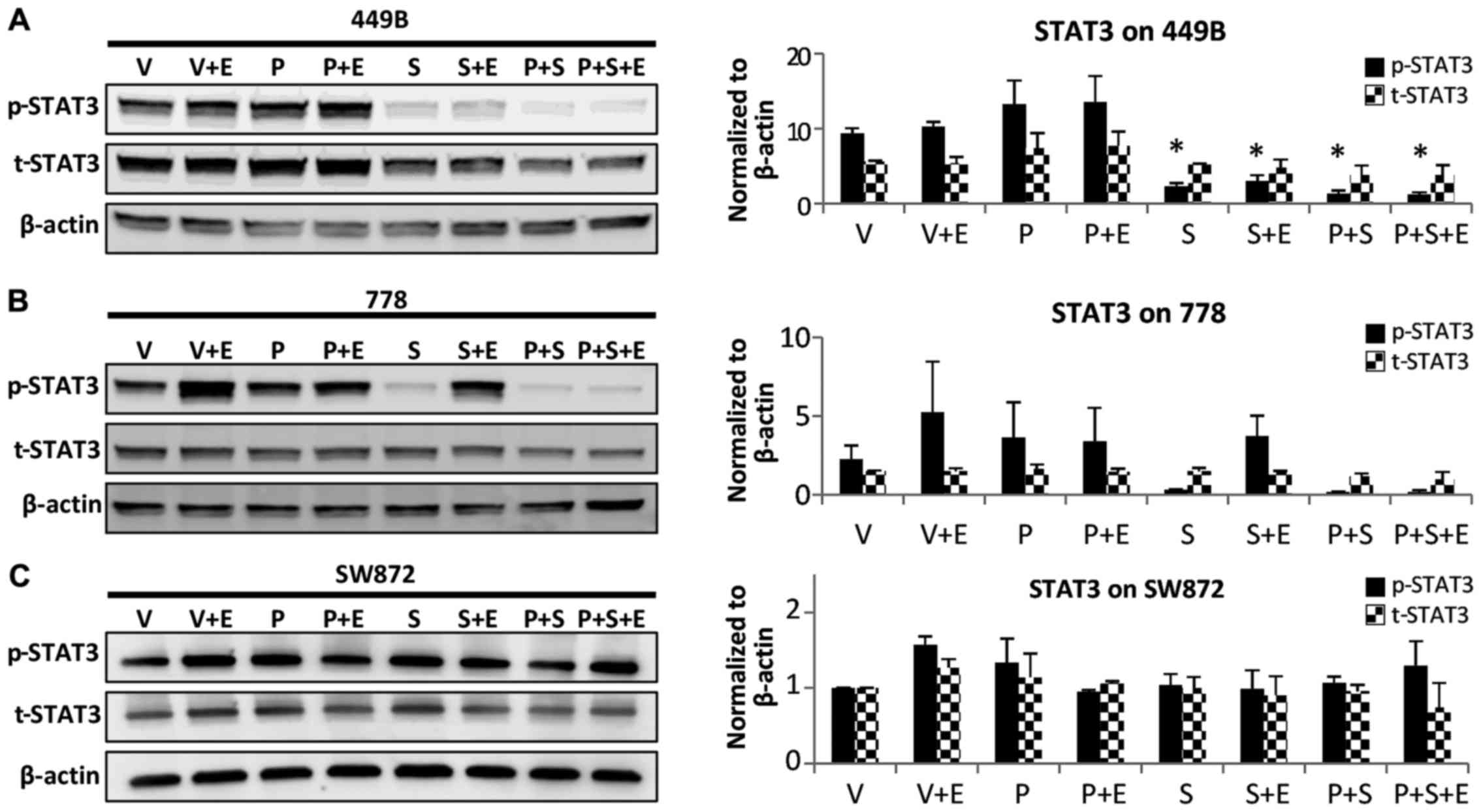
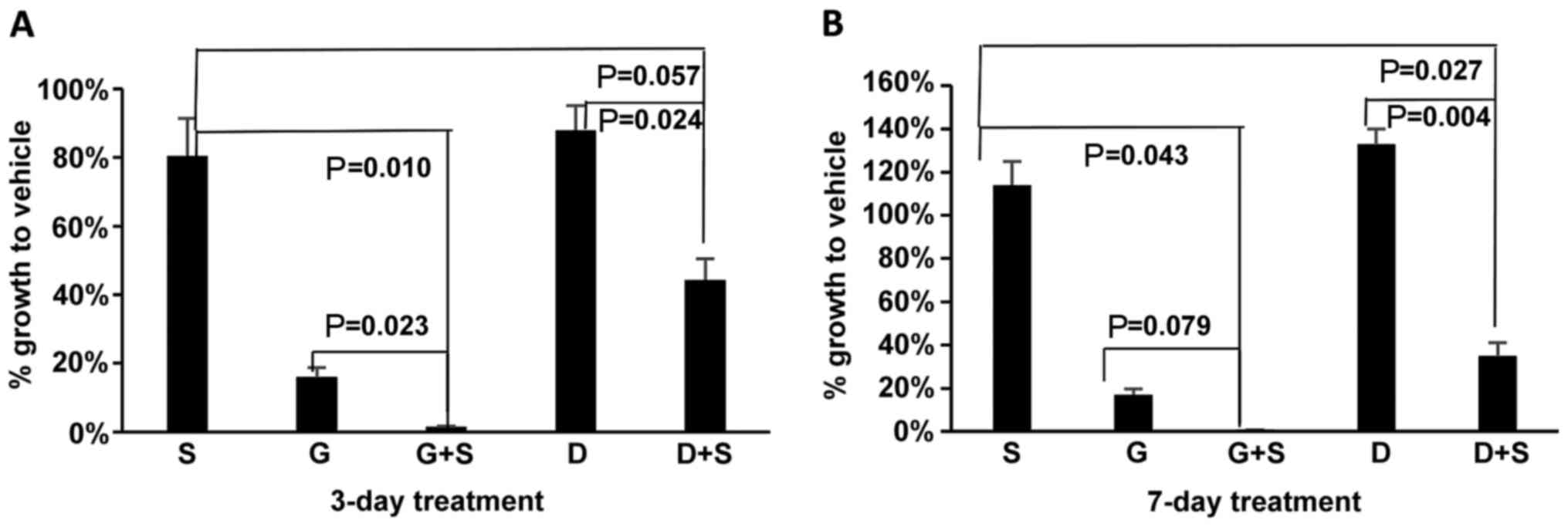
|
1
|
Sarcoma, Soft Tissue: Statistics. ASCO,
Cancer.Net Editorial Board; 2016, http://www.cancer.net/cancer-types/sarcoma-soft-tissue/statistics.
|
|
2
|
Baselga J and Arteaga CL: Critical update
and emerging trends in epidermal growth factor receptor targeting
in cancer. J Clin Oncol. 23:2445–2459. 2005. View Article : Google Scholar
|
|
3
|
Hynes NE and Lane HA: ERBB receptors and
cancer: The complexity of targeted inhibitors. Nat Rev Cancer.
5:341–354. 2005. View
Article : Google Scholar
|
|
4
|
Wang X, Batty KM, Crowe PJ, Goldstein D
and Yang JL: The potential of panHER inhibition in cancer. Front
Oncol. 5:22015. View Article : Google Scholar
|
|
5
|
Holbro T and Hynes NE: ErbB receptors:
Directing key signaling networks throughout life. Annu Rev
Pharmacol Toxicol. 44:195–217. 2004. View Article : Google Scholar
|
|
6
|
Yang JL, Hannan MT, Russell PJ and Crowe
PJ: Expression of HER1/EGFR protein in human soft tissue sarcomas.
Eur J Surg Oncol. 32:466–468. 2006. View Article : Google Scholar
|
|
7
|
Sato O, Wada T, Kawai A, Yamaguchi U,
Makimoto A, Kokai Y, Yamashita T, Chuman H, Beppu Y, Tani Y, et al:
Expression of epidermal growth factor receptor, ERBB2 and KIT in
adult soft tissue sarcomas: A clinicopathologic study of 281 cases.
Cancer. 103:1881–1890. 2005. View Article : Google Scholar
|
|
8
|
Teng HW, Wang HW, Chen WM, Chao TC, Hsieh
YY, Hsih CH, Tzeng CH, Chen PC and Yen CC: Prevalence and
prognostic influence of genomic changes of EGFR pathway markers in
synovial sarcoma. J Surg Oncol. 103:773–781. 2011. View Article : Google Scholar
|
|
9
|
Cascio MJ, O’Donnell RJ and Horvai AE:
Epithelioid sarcoma expresses epidermal growth factor receptor but
gene amplification and kinase domain mutations are rare. Mod
Pathol. 23:574–580. 2010. View Article : Google Scholar
|
|
10
|
Keizman D, Issakov J, Meller I, Maimon N,
Ish-Shalom M, Sher O and Merimsky O: Expression and significance of
EGFR in malignant peripheral nerve sheath tumor. J Neurooncol.
94:383–388. 2009. View Article : Google Scholar
|
|
11
|
Biscuola M, Van de Vijver K, Castilla MA,
Romero-Pérez L, López-García MÁ, Díaz-Martín J, Matias-Guiu X,
Oliva E and Palacios Calvo J: Oncogene alterations in endometrial
carcinosarcomas. Hum Pathol. 44:852–859. 2013. View Article : Google Scholar
|
|
12
|
Wang X, Goldstein D, Crowe PJ, Yang M,
Garrett K, Zeps N and Yang JL: Overcoming resistance of targeted
EGFR monotherapy by inhibition of STAT3 escape pathway in soft
tissue sarcoma. Oncotarget. 7:21496–21509. 2016.
|
|
13
|
Yang JL, Gupta RD, Goldstein D and Crowe
PJ: Significance of phosphorylated epidermal growth factor receptor
and its signal transducers in human soft tissue sarcoma. Int J Mol
Sci. 18:182017. View Article : Google Scholar
|
|
14
|
Ray-Coquard I, Le Cesne A, Whelan JS,
Schoffski P, Bui BN, Verweij J, Marreaud S, van Glabbeke M,
Hogendoorn P and Blay JY: A phase II study of gefitinib for
patients with advanced HER-1 expressing synovial sarcoma refractory
to doxorubicin-containing regimens. Oncologist. 13:467–473. 2008.
View Article : Google Scholar
|
|
15
|
Hawthorne VS, Huang WC, Neal CL, Tseng LM,
Hung MC and Yu D: ErbB2-mediated Src and signal transducer and
activator of transcription 3 activation leads to transcriptional
up-regulation of p21Cip1 and chemoresistance in breast cancer
cells. Mol Cancer Res. 7:592–600. 2009. View Article : Google Scholar
|
|
16
|
Proietti CJ, Rosemblit C, Beguelin W,
Rivas MA, Díaz Flaqué MC, Charreau EH, Schillaci R and Elizalde PV:
Activation of Stat3 by heregulin/ErbB-2 through the co-option of
progesterone receptor signaling drives breast cancer growth. Mol
Cell Biol. 29:1249–1265. 2009. View Article : Google Scholar
|
|
17
|
Béguelin W, Díaz Flaqué MC, Proietti CJ,
Cayrol F, Rivas MA, Tkach M, Rosemblit C, Tocci JM, Charreau EH,
Schillaci R, et al: Progesterone receptor induces ErbB-2 nuclear
translocation to promote breast cancer growth via a novel
transcriptional effect: ErbB-2 function as a coactivator of Stat3.
Mol Cell Biol. 30:5456–5472. 2010. View Article : Google Scholar
|
|
18
|
Gao L, Li F, Dong B, Zhang J, Rao Y, Cong
Y, Mao B and Chen X: Inhibition of STAT3 and ErbB2 suppresses tumor
growth, enhances radiosensitivity, and induces
mitochondria-dependent apoptosis in glioma cells. Int J Radiat
Oncol Biol Phys. 77:1223–1231. 2010. View Article : Google Scholar
|
|
19
|
Boyer MJ, Blackhall FH, Park K, Barrios
CH, Krzakowski MJ, Taylor I, Liang JQ, Denis LJ, O’Connell JP and
Ramalingam SS: Efficacy and safety of PF299804 versus erlotinib
(E): A glolbal, randomized phase II trial in patients (pts) with
advanced non-small cell lung cancer (NSCLC) after failure of
chemotherapy (CT). In: ASCO Annual Meeting 2010; J Clin Oncol;
Chicago, IL; 2010
|
|
20
|
Jänne PA, Boss DS, Camidge DR, Britten CD,
Engelman JA, Garon EB, Guo F, Wong S, Liang J, Letrent S, et al:
Phase I dose-escalation study of the pan-HER inhibitor, PF299804,
in patients with advanced malignant solid tumors. Clin Cancer Res.
17:1131–1139. 2011. View Article : Google Scholar
|
|
21
|
Engelman JA, Zejnullahu K, Gale C-M,
Lifshits E, Gonzales AJ, Shimamura T, Zhao F, Vincent PW, Naumov
GN, Bradner JE, et al: PF00299804, an irreversible pan-ERBB
inhibitor, is effective in lung cancer models with EGFR and ERBB2
mutations that are resistant to gefitinib. Cancer Res.
67:11924–11932. 2007. View Article : Google Scholar
|
|
22
|
Gonzales AJ, Hook KE, Althaus IW, Ellis
PA, Trachet E, Delaney AM, Harvey PJ, Ellis TA, Amato DM, Nelson
JM, et al: Antitumor activity and pharmacokinetic properties of
PF-00299804, a second-generation irreversible pan-erbB receptor
tyrosine kinase inhibitor. Mol Cancer Ther. 7:1880–1889. 2008.
View Article : Google Scholar
|
|
23
|
Nam H-J, Ching KA, Kan J, Kim HP, Han SW,
Im SA, Kim TY, Christensen JG, Oh DY and Bang YJ: Evaluation of the
antitumor effects and mechanisms of PF00299804, a pan-HER
inhibitor, alone or in combination with chemotherapy or targeted
agents in gastric cancer. Mol Cancer Ther. 11:439–451. 2012.
View Article : Google Scholar
|
|
24
|
Takahashi T, Boku N, Murakami H, Naito T,
Tsuya A, Nakamura Y, Ono A, Machida N, Yamazaki K, Watanabe J, et
al: Phase I and pharmacokinetic study of dacomitinib (PF-00299804),
an oral irreversible, small molecule inhibitor of human epidermal
growth factor receptor-1, -2, and -4 tyrosine kinases, in Japanese
patients with advanced solid tumors. Invest New Drugs.
30:2352–2363. 2012. View Article : Google Scholar
|
|
25
|
Park K, Cho BC, Kim DW, Ahn MJ, Lee SY,
Gernhardt D, Taylor I, Campbell AK, Zhang H, Giri N, et al: Safety
and efficacy of dacomitinib in korean patients with KRAS wild-type
advanced non-small-cell lung cancer refractory to chemotherapy and
erlotinib or gefitinib: A phase I/II trial. J Thorac Oncol.
9:1523–1531. 2014. View Article : Google Scholar
|
|
26
|
Reckamp KL, Giaccone G, Camidge DR,
Gadgeel SM, Khuri FR, Engelman JA, Koczywas M, Rajan A, Campbell
AK, Gernhardt D, et al: A phase 2 trial of dacomitinib
(PF-00299804), an oral, irreversible pan-HER (human epidermal
growth factor receptor) inhibitor, in patients with advanced
non-small cell lung cancer after failure of prior chemotherapy and
erlotinib. Cancer. 120:1145–1154. 2014. View Article : Google Scholar
|
|
27
|
Oh DY, Lee KW, Cho JY, Kang WK, Im SA, Kim
JW and Bang YJ: Phase II trial of dacomitinib in patients with
HER2-positive gastric cancer. Gastric Cancer. 19:1095–1103. 2016.
View Article : Google Scholar
|
|
28
|
Abdul Razak AR, Soulières D, Laurie SA,
Hotte SJ, Singh S, Winquist E, Chia S, Le Tourneau C, Nguyen-Tan
PF, Chen EX, et al: A phase II trial of dacomitinib, an oral
pan-human EGF receptor (HER) inhibitor, as first-line treatment in
recurrent and/or metastatic squamous-cell carcinoma of the head and
neck. Ann Oncol. 24:761–769. 2013. View Article : Google Scholar
|
|
29
|
Jänne PA, Ou SH, Kim DW, Oxnard GR,
Martins R, Kris MG, Dunphy F, Nishio M, O’Connell J, Paweletz C, et
al: Dacomitinib as first-line treatment in patients with clinically
or molecularly selected advanced non-small-cell lung cancer: A
multicentre, open-label, phase 2 trial. Lancet Oncol. 15:1433–1441.
2014. View Article : Google Scholar
|
|
30
|
Kris MG, Camidge DR, Giaccone G, Hida T,
Li BT, O’Connell J, Taylor I, Zhang H, Arcila ME, Goldberg Z, et
al: Targeting HER2 aberrations as actionable drivers in lung
cancers: Phase II trial of the pan-HER tyrosine kinase inhibitor
dacomitinib in patients with HER2-mutant or amplified tumors. Ann
Oncol. 26:1421–1427. 2015. View Article : Google Scholar
|
|
31
|
Ramalingam SS, Blackhall F, Krzakowski M,
Barrios CH, Park K, Bover I, Seog Heo D, Rosell R, Talbot DC, Frank
R, et al: Randomized phase II study of dacomitinib (PF-00299804),
an irreversible pan-human epidermal growth factor receptor
inhibitor, versus erlotinib in patients with advanced
non-small-cell lung cancer. J Clin Oncol. 30:3337–3344. 2012.
View Article : Google Scholar
|
|
32
|
Ellis PM, Shepherd FA, Millward M, Perrone
F, Seymour L, Liu G, Sun S, Cho BC, Morabito A, Leighl NB, et al
NCIC CTG; Australasian Lung Cancer Trials Group; NCI Naples
Clinical Trials Unit: Dacomitinib compared with placebo in
pretreated patients with advanced or metastatic non-small-cell lung
cancer (NCIC CTG BR.26): A double-blind, randomised, phase 3 trial.
Lancet Oncol. 15:1379–1388. 2014. View Article : Google Scholar
|
|
33
|
Ramalingam SS, Jänne PA, Mok T, O’Byrne K,
Boyer MJ, Von Pawel J, Pluzanski A, Shtivelband M, Docampo LI,
Bennouna J, et al: Dacomitinib versus erlotinib in patients with
advanced-stage, previously treated non-small-cell lung cancer
(ARCHER 1009): A randomised, double-blind, phase 3 trial. Lancet
Oncol. 15:1369–1378. 2014. View Article : Google Scholar
|
|
34
|
Ramalingam SS, O’Byrne K, Boyer M, Mok T,
Jänne PA, Zhang H, Liang J, Taylor I, Sbar EI and Paz-Ares L:
Dacomitinib versus erlotinib in patients with EGFR-mutated advanced
nonsmall-cell lung cancer (NSCLC): Pooled subset analyses from two
randomized trials. Ann Oncol. 27:423–429. 2016. View Article : Google Scholar
|
|
35
|
Wu YL, Cheng Y, Zhou X, Lee KH, Nakagawa
K, Niho S, Tsuji F, Linke R, Rosell R, Corral J, et al: Dacomitinib
versus gefitinib for the first-line treatment of advanced EGFR
mutation positive non-small cell lung cancer (ARCHER 1050): A
randomized, open-label phase III trial. Lancet Oncol. 18:1454–1466.
2017. View Article : Google Scholar
|
|
36
|
Wang X, Goldstein D, Crowe PJ and Yang
J-L: S3I-201, a Novel STAT3 Inhibitor, Inhibits Growth of Human
Soft Tissue Sarcoma Cell Lines. World J Cancer Res. 1:61–68. 2013.
View Article : Google Scholar
|
|
37
|
Wang X, Goldstein D, Crowe PJ and Yang JL:
Impact of STAT3 inhibition on survival of osteosarcoma cell lines.
Anticancer Res. 34:6537–6545. 2014.
|
|
38
|
Koizumi F, Shimoyama T, Taguchi F, Saijo N
and Nishio K: Establishment of a human non-small cell lung cancer
cell line resistant to gefitinib. Int J Cancer. 116:36–44. 2005.
View Article : Google Scholar
|
|
39
|
Muller PY and Milton MN: The determination
and interpretation of the therapeutic index in drug development.
Nat Rev Drug Discov. 11:751–761. 2012. View Article : Google Scholar
|
|
40
|
Ciardiello F and Normanno N: HER2
signaling and resistance to the anti-EGFR monoclonal antibody
cetuximab: A further step toward personalized medicine for patients
with colorectal cancer. Cancer Discov. 1:472–474. 2011. View Article : Google Scholar
|
|
41
|
Engelman JA, Zejnullahu K, Mitsudomi T,
Song Y, Hyland C, Park JO, Lindeman N, Gale CM, Zhao X, Christensen
J, et al: MET amplification leads to gefitinib resistance in lung
cancer by activating ERBB3 signaling. Science. 316:1039–1043. 2007.
View Article : Google Scholar
|
|
42
|
Bean J, Brennan C, Shih JY, Riely G, Viale
A, Wang L, Chitale D, Motoi N, Szoke J, Broderick S, et al: MET
amplification occurs with or without T790M mutations in EGFR mutant
lung tumors with acquired resistance to gefitinib or erlotinib.
Proc Natl Acad Sci USA. 104:20932–20937. 2007. View Article : Google Scholar
|
|
43
|
Nam H-J, Kim H-P, Yoon Y-K, Song SH, Min
AR, Han SW, Im SA, Kim TY, Oh DY and Bang YJ: The irreversible
pan-HER inhibitor PF00299804 alone or combined with gemcitabine has
an antitumor effect in biliary tract cancer cell lines. Invest New
Drugs. 30:2148–2160. 2012. View Article : Google Scholar
|
|
44
|
Kalous O, Conklin D, Desai AJ, O’Brien NA,
Ginther C, Anderson L, Cohen DJ, Britten CD, Taylor I, Christensen
JG, et al: Dacomitinib (PF-00299804), an irreversible Pan-HER
inhibitor, inhibits proliferation of HER2-amplified breast cancer
cell lines resistant to trastuzumab and lapatinib. Mol Cancer Ther.
11:1978–1987. 2012. View Article : Google Scholar
|
|
45
|
Ather F, Hamidi H, Fejzo MS, Letrent S,
Finn RS, Kabbinavar F, Head C and Wong SG: Dacomitinib, an
irreversible Pan-ErbB inhibitor significantly abrogates growth in
head and neck cancer models that exhibit low response to cetuximab.
PLoS One. 8:e561122013. View Article : Google Scholar
|
|
46
|
Ercan D, Zejnullahu K, Yonesaka K, Xiao Y,
Capelletti M, Rogers A, Lifshits E, Brown A, Lee C, Christensen JG,
et al: Amplification of EGFR T790M causes resistance to an
irreversible EGFR inhibitor. Oncogene. 29:2346–2356. 2010.
View Article : Google Scholar
|
|
47
|
Sriuranpong V, Park JI, Amornphimoltham P,
Patel V, Nelkin BD and Gutkind JS: Epidermal growth factor
receptor-independent constitutive activation of STAT3 in head and
neck squamous cell carcinoma is mediated by the autocrine/paracrine
stimulation of the interleukin 6/gp130 cytokine system. Cancer Res.
63:2948–2956. 2003.
|
|
48
|
Gao SP, Mark KG, Leslie K, Pao W, Motoi N,
Gerald WL, Travis WD, Bornmann W, Veach D, Clarkson B, et al:
Mutations in the EGFR kinase domain mediate STAT3 activation via
IL-6 production in human lung adenocarcinomas. J Clin Invest.
117:3846–3856. 2007. View Article : Google Scholar
|
|
49
|
Wang X, Crowe PJ, Goldstein D and Yang JL:
STAT3 inhibition, a novel approach to enhancing targeted therapy in
human cancers (Review). Int J Oncol. 41:1181–1191. 2012. View Article : Google Scholar
|
|
50
|
Kim SM, Kwon O-J, Hong YK, Kim JH, Solca
F, Ha SJ, Soo RA, Christensen JG, Lee JH and Cho BC: Activation of
IL-6R/JAK1/STAT3 signaling induces de novo resistance to
irreversible EGFR inhibitors in non-small cell lung cancer with
T790M resistance mutation. Mol Cancer Ther. 11:2254–2264. 2012.
View Article : Google Scholar
|