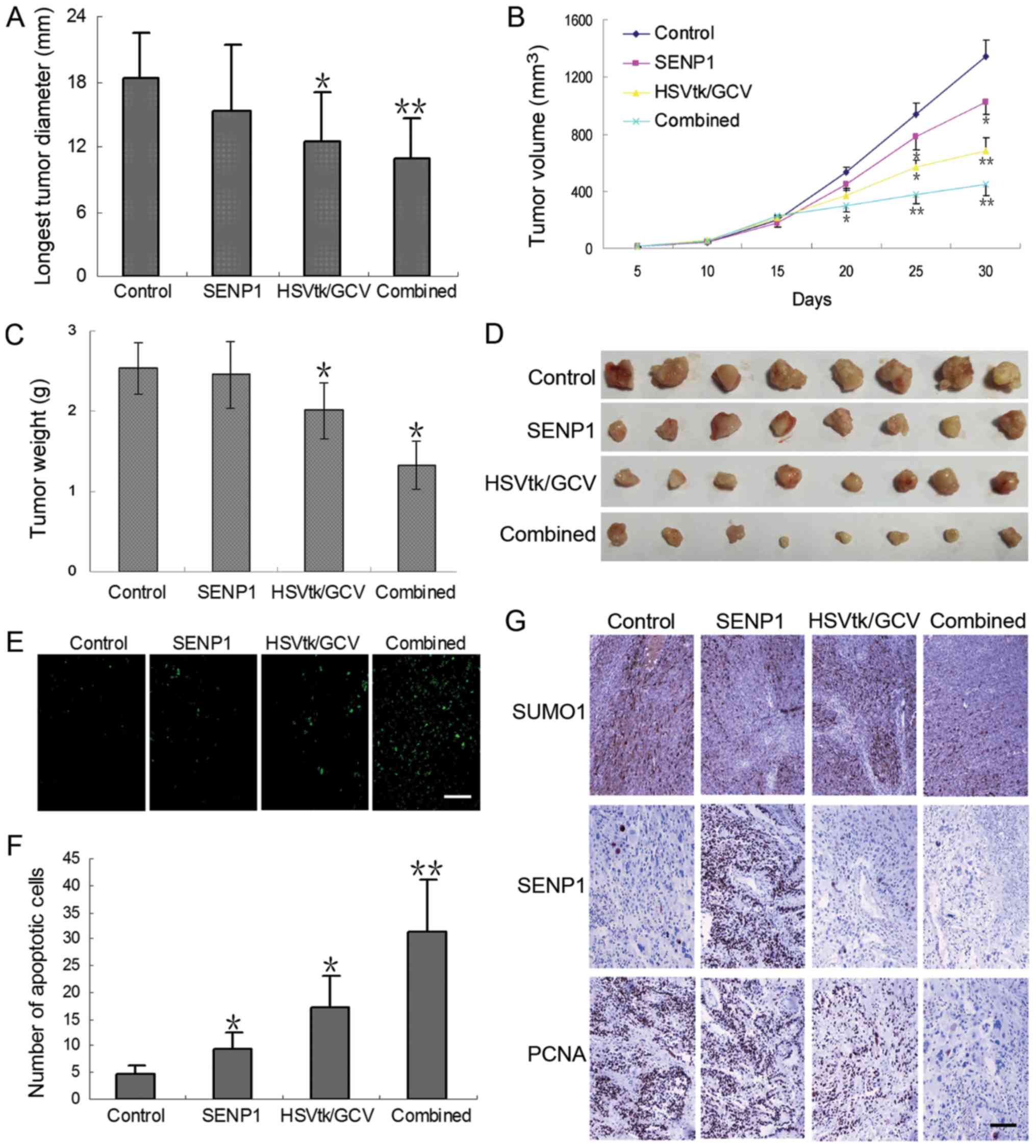
|
1
|
Adamopoulos C, Gargalionis AN, Piperi C
and Papavassiliou AG: Recent advances in mechanobiology of
osteosarcoma. J Cell Biochem. 118:232–236. 2017. View Article : Google Scholar
|
|
2
|
Abarrategi A, Tornin J, Martinez-Cruzado
L, Hamilton A, Martinez-Campos E, Rodrigo JP, González MV, Baldini
N, Garcia-Castro J and Rodriguez R: Osteosarcoma: Cells-of-origin,
cancer stem cells, and targeted therapies. Stem Cells Int.
2016.3631764:2016.
|
|
3
|
Abed R and Grimer R: Surgical modalities
in the treatment of bone sarcoma in children. Cancer Treat Rev.
36:342–347. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Andreou D, Hardes J, Gosheger G, Henrichs
MP, Nottrott M and Streitbürger A: Interdisciplinary diagnostic and
treatment of bone sarcomas of the extremities and trunk. Handchir
Mikrochir Plast Chir. 47:90–99. 2015.In German. PubMed/NCBI
|
|
5
|
Claude L, Rousmans S, Carrie C, Breteau N,
Dijoud F, Gentet JC, Giammarile F, Jouve JL, Kind M, Marec-Bérard
P, et al: Fédération nationale des centres de lutte contre le
cancer (FNCLCC); Fédération hospitalière de France (FHF);
Fédération nationale de cancérologie des CHRU (FNCCHRU); Fédération
française de cancérologie des CHG (FFCCHG); centres régionaux de
lutte contre le cancer (CRLCC); Société française de lutte contre
les cancers de l′ enfant et de l′ adolescent (SFCE): Standards and
Options for the use of radiation therapy in the management of
patients with osteosarcoma. Update 2004. Cancer Radiother.
9:104–121. 2005.In French. PubMed/NCBI
|
|
6
|
Liebner DA: The indications and efficacy
of conventional chemotherapy in primary and recurrent sarcoma. J
Surg Oncol. 111:622–631. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Biazzo A and De Paolis M:
Multidisciplinary approach to osteosarcoma. Acta Orthop Belg.
82:690–698. 2016.
|
|
8
|
Yan GN, Lv YF and Guo QN: Advances in
osteosarcoma stem cell research and opportunities for novel
therapeutic targets. Cancer Lett. 370:268–274. 2016. View Article : Google Scholar
|
|
9
|
Zeng W, Wan R, Zheng Y, Singh SR and Wei
Y: Hypoxia, stem cells and bone tumor. Cancer Lett. 313:129–136.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gibbs CP Jr, Levings PP and Ghivizzani SC:
Evidence for the osteosarcoma stem cell. Curr Orthop Pract.
22:322–326. 2011. View Article : Google Scholar :
|
|
11
|
Brown HK, Tellez-Gabriel M and Heymann D:
Cancer stem cells in osteosarcoma. Cancer Lett. 386:189–195. 2017.
View Article : Google Scholar
|
|
12
|
Seeliger C, Balmayor ER and van Griensven
M: miRNAs related to skeletal diseases. Stem Cells Dev.
25:1261–1281. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bishop MW, Janeway KA and Gorlick R:
Future directions in the treatment of osteosarcoma. Curr Opin
Pediatr. 28:26–33. 2016. View Article : Google Scholar :
|
|
14
|
Shang D, Wu J, Guo L, Xu Y, Liu L and Lu
J: Metformin increases sensitivity of osteosarcoma stem cells to
cisplatin by inhibiting expression of PKM2. Int J Oncol.
50:1848–1856. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mongre RK, Sodhi SS, Ghosh M, Kim JH, Kim
N, Park YH, Kim SJ, Heo YJ, Sharma N and Jeong DK: The novel
inhibitor BRM270 downregulates tumorigenesis by suppression of
NF-κB signaling cascade in MDR-induced stem like cancer-initiating
cells. Int J Oncol. 46:2573–2585. 2015. View Article : Google Scholar
|
|
16
|
Lee OK, Coathup MJ, Goodship AE and Blunn
GW: Use of mesenchymal stem cells to facilitate bone regeneration
in normal and chemotherapy-treated rats. Tissue Eng. 11:1727–1735.
2005. View Article : Google Scholar
|
|
17
|
Bayat M, Virdi A, Rezaei F and Chien S:
Comparison of the in vitro effects of low-level laser therapy and
low-intensity pulsed ultrasound therapy on bony cells and stem
cells. Prog Biophys Mol Biol. 133:36–48. 2018. View Article : Google Scholar
|
|
18
|
Liu B, Ma W, Jha RK and Gurung K: Cancer
stem cells in osteosarcoma: Recent progress and perspective. Acta
Oncol. 50:1142–1150. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen JN, Wu DW, Li T, Yang KJ, Cheng L,
Zhou ZP, Pu SM and Lin WH: Arylurea Derivatives: A Class of
Potential Cancer Targeting Agents. Curr Top Med Chem. 17:3099–3130.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gong L, Qi R and Li DW: Sumoylation
pathway as potential therapeutic targets in cancer. Curr Mol Med.
16:900–905. 2017. View Article : Google Scholar
|
|
21
|
Cheng CT, Kuo CY and Ann DK: KAPtain in
charge of multiple missions: Emerging roles of KAP1. World J Biol
Chem. 5:308–320. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yang XJ and Chiang CM: Sumoylation in gene
regulation, human disease, and therapeutic action. F1000Prime Rep.
5:452013. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hannoun Z, Greenhough S, Jaffray E, Hay RT
and Hay DC: Post-translational modification by SUMO. Toxicology.
278:288–293. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang J and Schwartz RJ: Sumoylation and
regulation of cardiac gene expression. Circ Res. 107:19–29. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Butler JT, Hall LL, Smith KP and Lawrence
JB: Changing nuclear landscape and unique PML structures during
early epigenetic transitions of human embryonic stem cells. J Cell
Biochem. 107:609–621. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhu C, Chen C, Huang J, Zhang H, Zhao X,
Deng R, Dou J, Jin H, Chen R, Xu M, et al: SUMOylation at K707 of
DGCR8 controls direct function of primary microRNA. Nucleic Acids
Res. 43:7945–7960. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jones MC, Fusi L, Higham JH, Abdel-Hafiz
H, Horwitz KB, Lam EW and Brosens JJ: Regulation of the SUMO
pathway sensitizes differentiating human endometrial stromal cells
to progesterone. Proc Natl Acad Sci USA. 103:16272–16277. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cheng JK: SUMO-specific protease 1 and
cancer. Ai Zheng. 27:771–774. 2008.In Chinese. PubMed/NCBI
|
|
29
|
Fasci D, Anania VG, Lill JR and Salvesen
GS: SUMO deconjugation is required for arsenic-triggered
ubiquitylation of PML. Sci Signal. 8:ra562015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
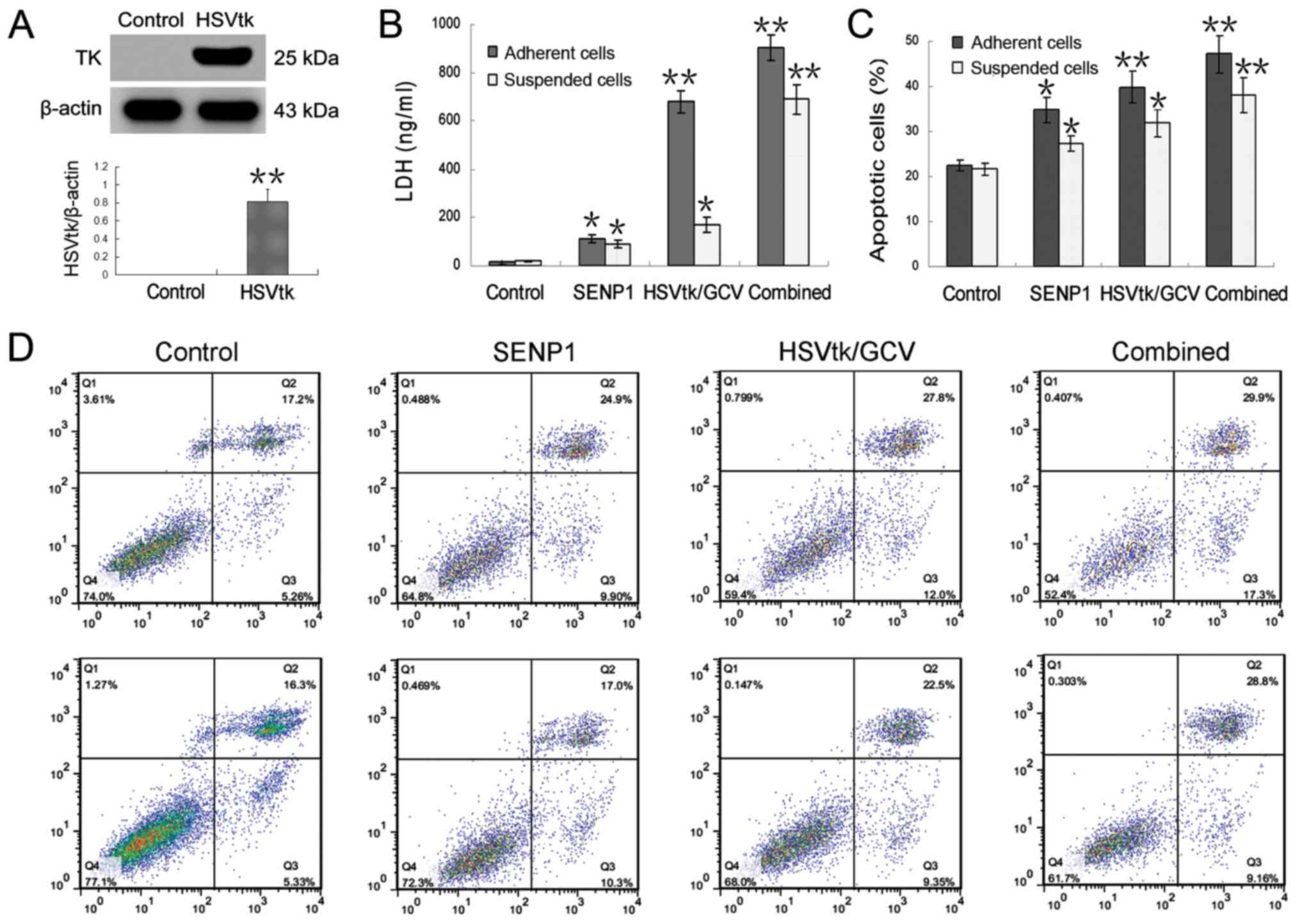
Fillat C, Carrió M, Cascante A and Sangro
B: Suicide gene therapy mediated by the Herpes Simplex virus
thymidine kinase gene/Ganciclovir system: Fifteen years of
application. Curr Gene Ther. 3:13–26. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Grignet-Debrus C, Noël A, Foidart JM and
Calberg-Bacq CM: Identification of factors important for the
success of suicide gene therapy after a comparative study of
Varicella zoster and Herpes simplex viral thymidine kinases
efficacy on breast cancer cells. Cell Mol Biol (Noisy-le-grand).
51:37–48. 2005.
|
|
32
|
Marples B, Greco O, Joiner MC and Scott
SD: Molecular approaches to chemo-radiotherapy. Eur J Cancer.
38:231–239. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shibata MA, Morimoto J, Ito Y, Kusakabe K
and Otsuki Y: Experimental gene therapy in mammary and urinary
bladder cancer using electrogene transfer. Med Electron Microsc.
37:216–224. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Smythe WR: Prodrug/drug sensitivity gene
therapy: Current status. Curr Oncol Rep. 2:17–22. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ghanbari M, Saberfar E, Goodarzi Z,
Lashini H, Ghanbari S, Karamimanesh M and Baesi K: Regulation of
HSVtk gene by endogenous microRNA-122a in liver cell lines as
suicide gene therapy. Gastroenterol Hepatol Bed Bench. 10:202–207.
2017.PubMed/NCBI
|
|
36
|
Cerella C, Grandjenette C, Dicato M and
Diederich M: Roles of apoptosis and cellular senescence in cancer
and aging. Curr Drug Targets. 17:405–415. 2016. View Article : Google Scholar
|
|
37
|
Liu Z, Jiang Z, Huang J, Huang S, Li Y,
Sheng F, Yu S, Yu S and Liu X: Mesenchymal stem cells show little
tropism for the resting and differentiated cancer stem cell-like
glioma cells. Int J Oncol. 44:1223–1232. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang J and Liu X, Jiang Z, Li L, Cui Z,
Gao Y, Kong D and Liu X: A novel method to limit breast cancer stem
cells in states of quiescence, proliferation or differentiation:
Use of gel stress in combination with stem cell growth factors.
Oncol Lett. 12:1355–1360. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Liu Z, Jiang Z, Huang J, Huang S, Li Y, Yu
S, Yu S and Liu X: miR-7 inhibits glioblastoma growth by
simultaneously interfering with the PI3K/ATK and Raf/MEK/ERK
pathways. Int J Oncol. 44:1571–1580. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Liu X, Li G, Su Z, Jiang Z, Chen L, Wang
J, Yu S and Liu Z: Poly(amido amine) is an ideal carrier of miR-7
for enhancing gene silencing effects on the EGFR pathway in U251
glioma cells. Oncol Rep. 29:1387–1394. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Li G, Liu X, Liu Z and Su Z: Interactions
of connexin 43 and aquaporin-4 in the formation of glioma-induced
brain edema. Mol Med Rep. 11:1188–1194. 2015. View Article : Google Scholar
|
|
42
|
Orsi GA, Naughtin M and Almouzni G: The
epigenome and cancer stem cell fate: Connected by a linker histone
variant. Cell Stem Cell. 19:567–568. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Cui CP, Wong CC, Kai AK, Ho DW, Lau EY,
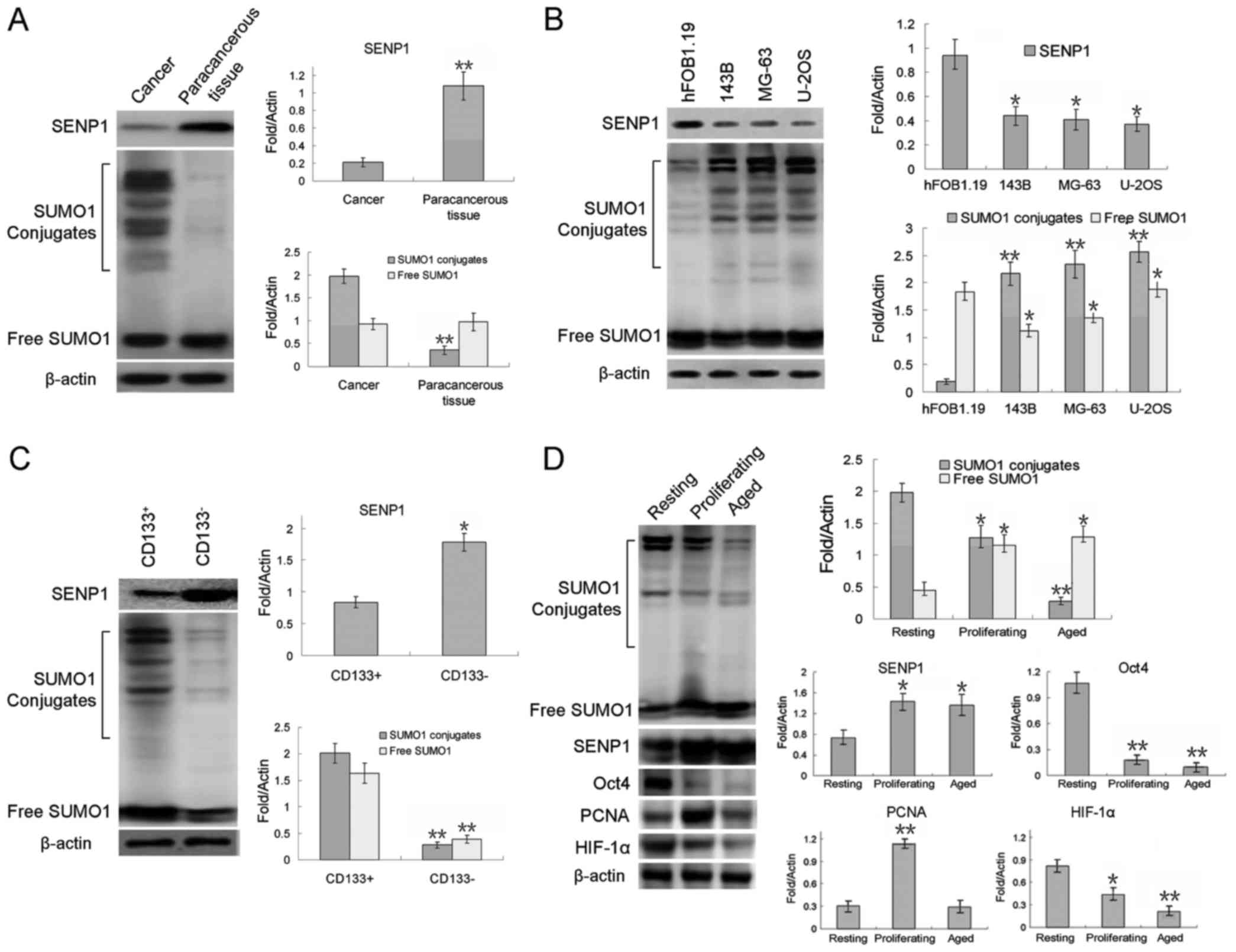
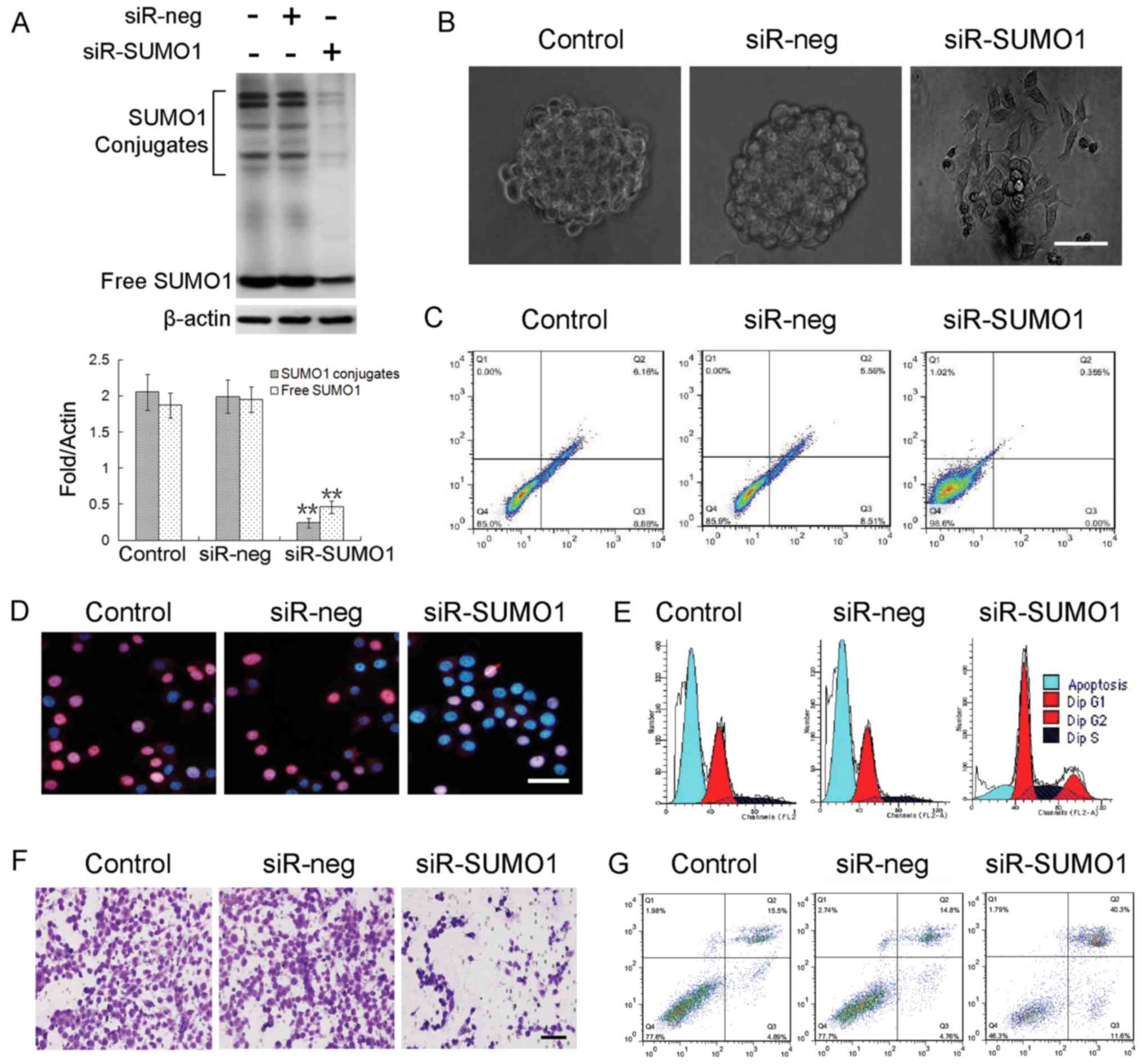
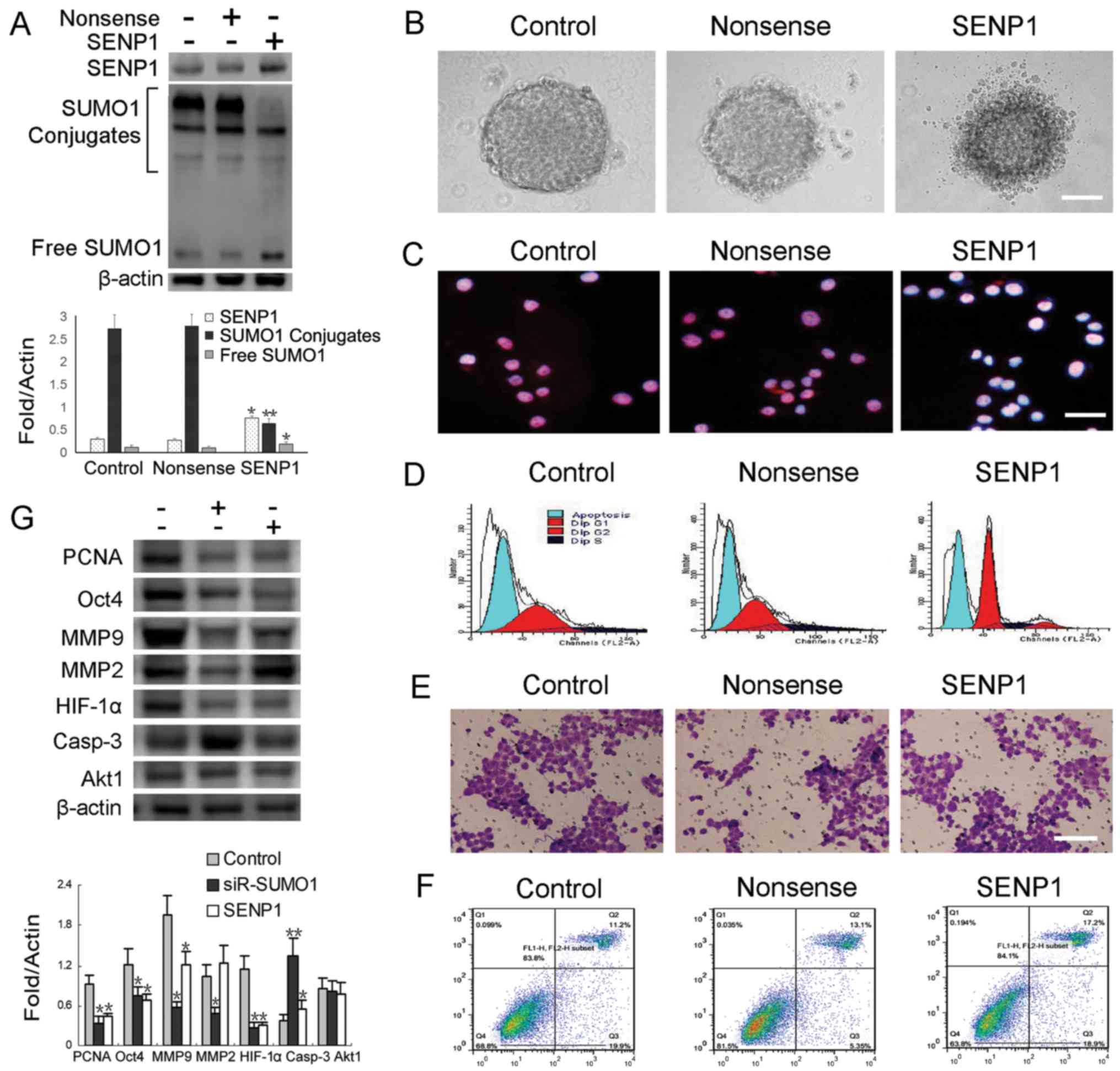
Tsui YM, Chan LK, Cheung TT, Chok KS, Chan ACY, et al: SENP1
promotes hypoxia-induced cancer stemness by HIF-1α deSUMOylation
and SENP1/HIF-1α positive feedback loop. Gut. 66:2149–2159. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Mattoscio D and Chiocca S: SUMO pathway
components as possible cancer biomarkers. Future Oncol.
11:1599–1610. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Nuro-Gyina PK and Parvin JD: Roles for
SUMO in pre-mRNA processing. Wiley Interdiscip Rev RNA. 7:105–112.
2016. View Article : Google Scholar
|
|
46
|
Yang W and Paschen W: SUMO proteomics to
decipher the SUMO-modified proteome regulated by various diseases.
Proteomics. 15:1181–1191. 2015. View Article : Google Scholar :
|
|
47
|
Villodre ES, Kipper FC, Pereira MB and
Lenz G: Roles of OCT4 in tumorigenesis, cancer therapy resistance
and prognosis. Cancer Treat Rev. 51:1–9. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wei F, Schöler HR and Atchison ML:
Sumoylation of Oct4 enhances its stability, DNA binding, and
transactivation. J Biol Chem. 282:21551–21560. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Tsutakawa SE, Yan C, Xu X, Zhuang Z, Todd
Washington M, Tainer JA and Ivanov I: 112 Structurally distinct
ubiquitin- and SUMO-modified PCNA: Implications for their distinct
roles in the DNA damage response. J Biomol Struct Dyn. 1:70–71.
2015. View Article : Google Scholar
|
|
50
|
Burkovics P, Sebesta M, Sisakova A, Plault
N, Szukacsov V, Robert T, Pinter L, Marini V, Kolesar P, Haracska
L, et al: Srs2 mediates PCNA-SUMO-dependent inhibition of DNA
repair synthesis. EMBO J. 32:742–755. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Leach CA and Michael WM: Ubiquitin/SUMO
modification of PCNA promotes replication fork progression in
Xenopus laevis egg extracts. J Cell Biol. 171:947–954. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Han X, Wang XL, Li Q, Dong XX, Zhang JS
and Yan QC: HIF-1α SUMOylation affects the stability and
transcriptional activity of HIF-1α in human lens epithelial cells.
Graefes Arch Clin Exp Ophthalmol. 253:1279–1290. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Gu J, Fan Y, Liu X, Zhou L, Cheng J, Cai R
and Xue S: SENP1 protects against myocardial ischaemia/reperfusion
injury via a HIF1α-dependent pathway. Cardiovasc Res. 104:83–92.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
de la Cruz-Herrera CF, Campagna M, Lang V,
del Carmen González-Santamaría J, Marcos-Villar L, Rodríguez MS,
Vidal A, Collado M and Rivas C: SUMOylation regulates AKT1
activity. Oncogene. 34:1442–1450. 2015. View Article : Google Scholar
|
|
55
|
Li R, Wei J, Jiang C, Liu D, Deng L, Zhang
K and Wang P: Akt SUMOylation regulates cell proliferation and
tumorigenesis. Cancer Res. 73:5742–5753. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Liu X, Ren W, Jiang Z, Su Z, Ma X, Li Y,
Jiang R, Zhang J and Yang X: Hypothermia inhibits the proliferation
of bone marrow-derived mesenchymal stem cells and increases
tolerance to hypoxia by enhancing SUMOylation. Int J Mol Med.
40:1631–1638. 2017.PubMed/NCBI
|
|
57
|
Shen Y, Li Y, Ma X, Wan Q, Jiang Z, Liu Y,
Zhang D, Liu X and Wu W: Connexin 43 SUMOylation improves gap
junction functions between liver cancer stem cells and enhances
their sensitivity to HSVtk/GCV. Int J Oncol. 52:872–880.
2018.PubMed/NCBI
|