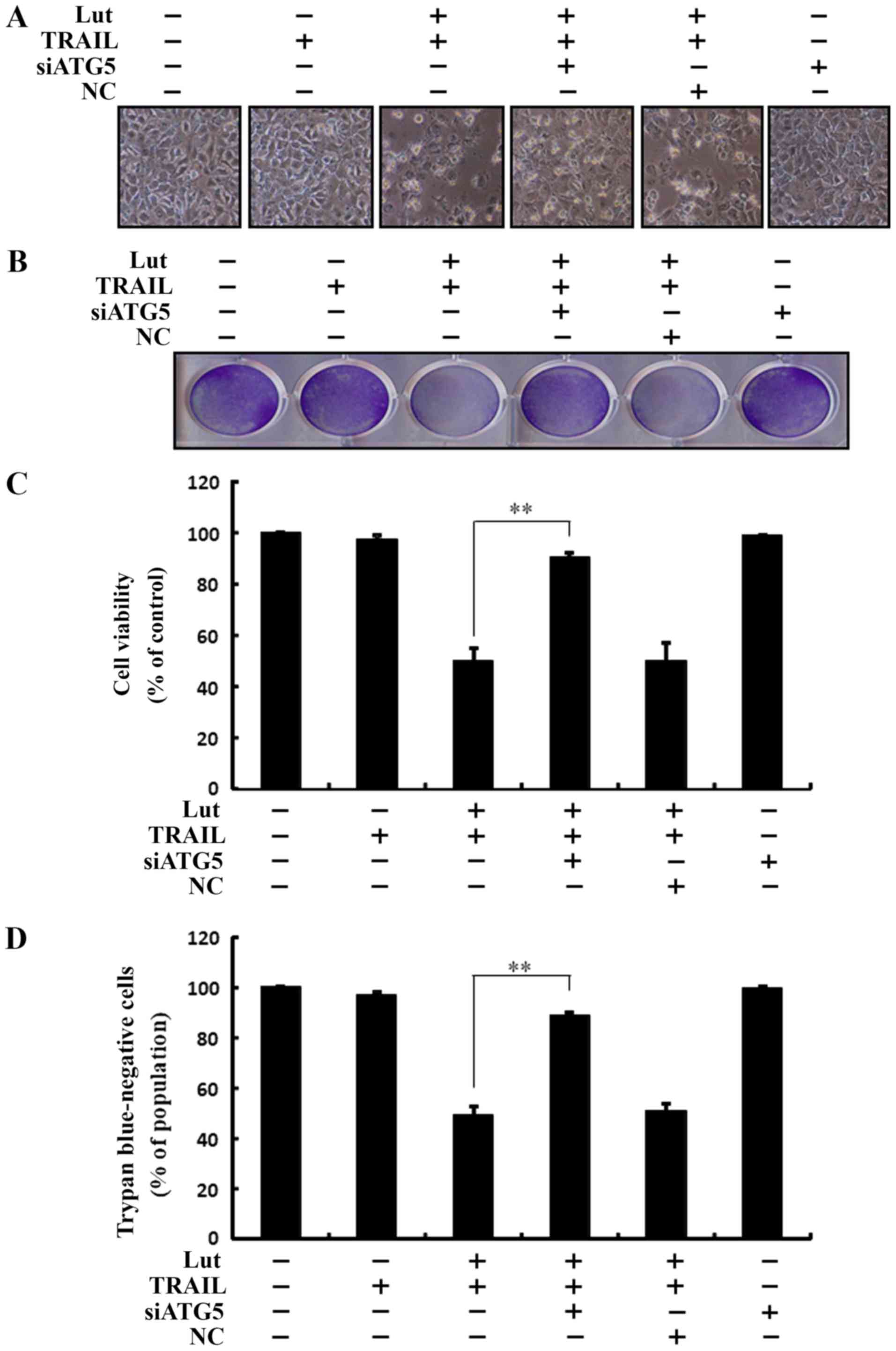
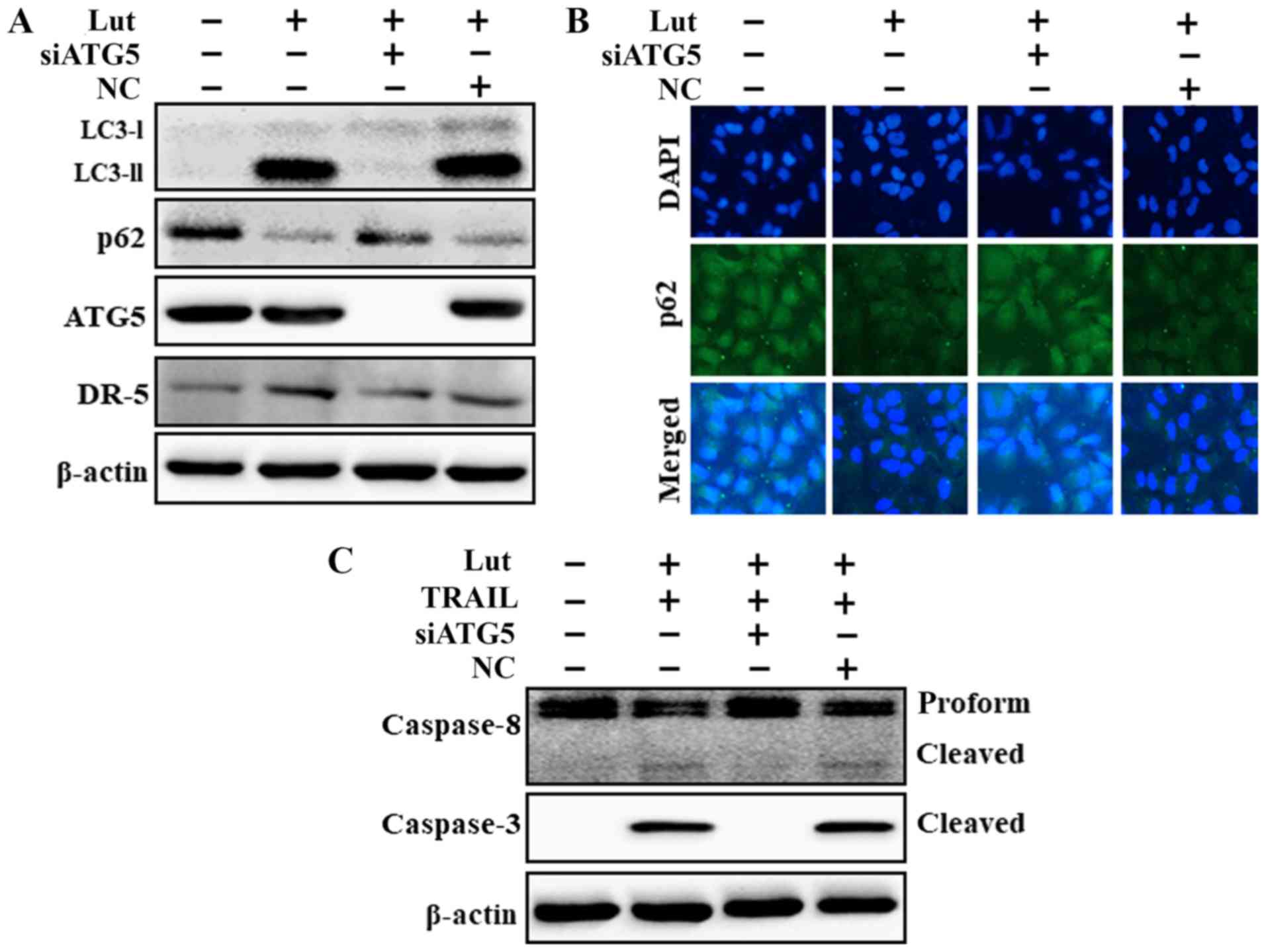
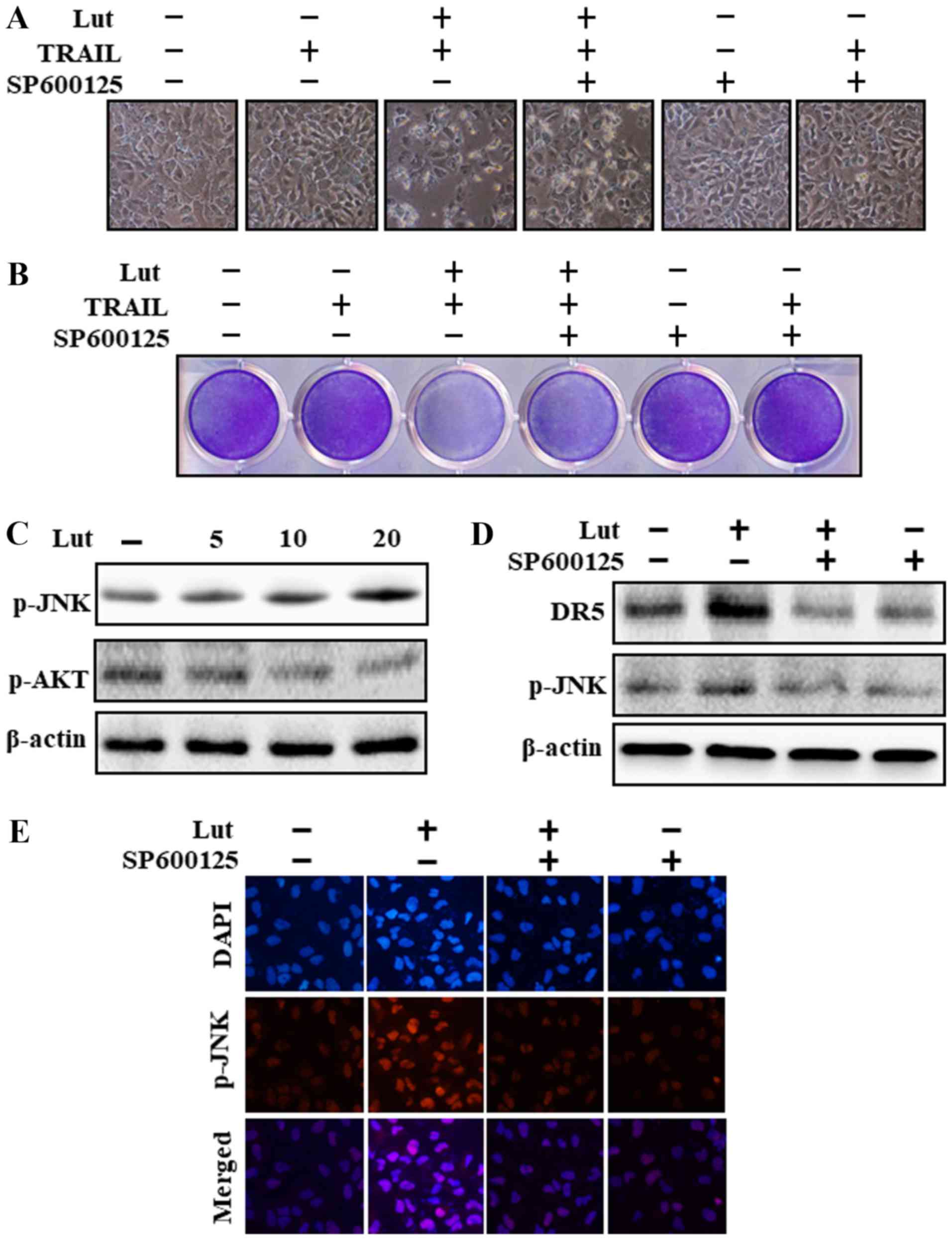
|
1
|
El-Serag HB and Rudolph KL: Hepatocellular
carcinoma: Epidemiology and molecular carcinogenesis.
Gastroenterology. 132:2557–2576. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sherman M: Epidemiology of hepatocellular
carcinoma. Oncology. 78(Suppl 1): 7–10. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Balogh J, Victor D III, Asham EH,
Burroughs SG, Boktour M, Saharia A, Li X, Ghobrial RM and Monsour
HP Jr: Hepatocellular carcinoma: A review. J Hepatocell Carcinoma.
3:41–53. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Arii S: Molecularly targeted therapy for
hepatocellular carcinoma from the basic and clinical aspects. Int J
Clin Oncol. 15:2342010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Llovet JM, Di Bisceglie AM, Bruix J,
Kramer BS, Lencioni R, Zhu AX, Sherman M, Schwartz M, Lotze M,
Talwalkar J, et al Panel of Experts in HCC-Design Clinical Trials:
Design and endpoints of clinical trials in hepatocellular
carcinoma. J Natl Cancer Inst. 100:698–711. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang S: The promise of cancer therapeutics
targeting the TNF-related apoptosis-inducing ligand and TRAIL
receptor pathway. Oncogene. 27:6207–6215. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mahmood Z and Shukla Y: Death receptors:
Targets for cancer therapy. Exp Cell Res. 316:887–899. 2010.
View Article : Google Scholar
|
|
8
|
Allen JE and El-Deiry WS: Regulation of
the human TRAIL gene. Cancer Biol Ther. 13:1143–1151. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Micheau O, Shirley S and Dufour F: Death
receptors as targets in cancer. Br J Pharmacol. 169:1723–1744.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen CY, Yiin SJ, Hsu JL, Wang WC, Lin SC
and Chern CL: Isoobtusilactone A sensitizes human hepatoma Hep G2
cells to TRAIL-induced apoptosis via ROS and CHOP-mediated
up-regulation of DR5. J Agric Food Chem. 60:3533–3539. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Pan G, O’Rourke K, Chinnaiyan AM, Gentz R,
Ebner R, Ni J and Dixit VM: The receptor for the cytotoxic ligand
TRAIL. Science. 276:111–113. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Walczak H, Degli-Esposti MA, Johnson RS,
Smolak PJ, Waugh JY, Boiani N, Timour MS, Gerhart MJ, Schooley KA,
Smith CA, et al: TRAIL-R2: A novel apoptosis-mediating receptor for
TRAIL. EMBO J. 16:5386–5397. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ashkenazi A, Pai RC, Fong S, Leung S,
Lawrence DA, Marsters SA, Blackie C, Chang L, McMurtrey AE, Hebert
A, et al: Safety and antitumor activity of recombinant soluble Apo2
ligand. J Clin Invest. 104:155–162. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ashokkumar P and Sudhandiran G: Protective
role of luteolin on the status of lipid peroxidation and
antioxidant defense against azoxymethane-induced experimental colon
carcinogenesis. Biomed Pharmacother. 62:590–597. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nishitani Y, Yamamoto K, Yoshida M, Azuma
T, Kanazawa K, Hashimoto T and Mizuno M: Intestinal
anti-inflammatory activity of luteolin: Role of the aglycone in
NF-κB inactivation in macrophages co-cultured with intestinal
epithelial cells. Biofactors. 39:522–533. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ashokkumar P and Sudhandiran G: Luteolin
inhibits cell proliferation during Azoxymethane-induced
experimental colon carcinogenesis via Wnt/β-catenin pathway. Invest
New Drugs. 29:273–284. 2011. View Article : Google Scholar
|
|
17
|
Huang X, Dai S, Dai J, Xiao Y, Bai Y, Chen
B and Zhou M: Luteolin decreases invasiveness, deactivates STAT3
signaling, and reverses interleukin-6 induced
epithelial-mesenchymal transition and matrix metalloproteinase
secretion of pancreatic cancer cells. OncoTargets Ther.
8:2989–3001. 2015. View Article : Google Scholar
|
|
18
|
Han K, Meng W, Zhang JJ, Zhou Y, Wang YL,
Su Y, Lin SC, Gan ZH, Sun YN and Min DL: Luteolin inhibited
proliferation and induced apoptosis of prostate cancer cells
through miR-301. OncoTargets Ther. 9:3085–3094. 2016. View Article : Google Scholar
|
|
19
|
Naso LG, Badiola I, Marquez Clavijo J,
Valcarcel M, Salado C, Ferrer EG and Williams PAM: Inhibition of
the metastatic progression of breast and colorectal cancer in vitro
and in vivo in murine model by the oxidovanadium(IV) complex with
luteolin. Bioorg Med Chem. 24:6004–6011. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Dia VP and Pangloli P:
Epithelial-to-Mesenchymal Transition in Paclitaxel-Resistant
Ovarian Cancer Cells Is Downregulated by Luteolin. J Cell Physiol.
232:391–401. 2017. View Article : Google Scholar
|
|
21
|
Youdim KA, Qaiser MZ, Begley DJ,
Rice-Evans CA and Abbott NJ: Flavonoid permeability across an in
situ model of the blood-brain barrier. Free Radic Biol Med.
36:592–604. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jang S, Kelley KW and Johnson RW: Luteolin
reduces IL-6 production in microglia by inhibiting JNK
phosphorylation and activation of AP-1. Proc Natl Acad Sci USA.
105:7534–7539. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Cao Z, Zhang H, Cai X, Fang W, Chai D, Wen
Y, Chen H, Chu F and Zhang Y: Luteolin promotes cell apoptosis by
inducing autophagy in hepatocellular carcinoma. Cell Physiol
Biochem. 43:1803–1812. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Levine B and Klionsky DJ: Development by
self-digestion: Molecular mechanisms and biological functions of
autophagy. Dev Cell. 6:463–477. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
El-Khattouti A, Selimovic D, Haikel Y and
Hassan M: Crosstalk between apoptosis and autophagy: Molecular
mechanisms and therapeutic strategies in cancer. J Cell Death.
6:37–55. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liang C: Negative regulation of autophagy.
Cell Death Differ. 17:1807–1815. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Giampietri C, Petrungaro S, Padula F,
D’Alessio A, Marini ES, Facchiano A, Filippini A and Ziparo E:
Autophagy modulators sensitize prostate epithelial cancer cell
lines to TNF-alpha-dependent apoptosis. Apoptosis. 17:1210–1222.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lorenzi PL, Claerhout S, Mills GB and
Weinstein JN: A curated census of autophagy-modulating proteins and
small molecules: Candidate targets for cancer therapy. Autophagy.
10:1316–1326. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yao D, Wang P, Zhang J, Fu L, Ouyang L and
Wang J: Deconvoluting the relationships between autophagy and
metastasis for potential cancer therapy. Apoptosis. 21:683–698.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kabeya Y, Mizushima N, Ueno T, Yamamoto A,
Kirisako T, Noda T, Kominami E, Ohsumi Y and Yoshimori T: LC3, a
mammalian homologue of yeast Apg8p, is localized in autopha-gosome
membranes after processing. EMBO J. 19:5720–5728. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Tanida I, Minematsu-Ikeguchi N, Ueno T and
Kominami E: Lysosomal turnover, but not a cellular level, of
endogenous LC3 is a marker for autophagy. Autophagy. 1:84–91. 2005.
View Article : Google Scholar
|
|
32
|
Nazim UM, Jeong JK and Park SY:
Ophiopogonin B sensitizes TRAIL-induced apoptosis through
activation of autophagy flux and downregulates cellular FLICE-like
inhibitory protein. Oncotarget. 9:4161–4172. 2017.
|
|
33
|
Kelley SK and Ashkenazi A: Targeting death
receptors in cancer with Apo2L/TRAIL. Curr Opin Pharmacol.
4:333–339. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Volkmann X, Fischer U, Bahr MJ, Ott M,
Lehner F, Macfarlane M, Cohen GM, Manns MP, Schulze-Osthoff K and
Bantel H: Increased hepatotoxicity of tumor necrosis factor-related
apoptosis-inducing ligand in diseased human liver. Hepatology.
46:1498–1508. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yee SB, Choi HJ, Chung SW, Park DH, Sung
B, Chung HY and Kim ND: Growth inhibition of luteolin on HepG2
cells is induced via p53 and Fas/Fas-ligand besides the TGF-β
pathway. Int J Oncol. 47:747–754. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Park SH, Ham S, Kwon TH, Kim MS, Lee DH,
Kang JW, Oh SR and Yoon DY: Luteolin induces cell cycle arrest and
apoptosis through extrinsic and intrinsic signaling pathways in
MCF-7 breast cancer cells. J Environ Pathol Toxicol Oncol.
33:219–231. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Sun DW, Zhang HD, Mao L, Mao CF, Chen W,
Cui M, Ma R, Cao HX, Jing CW and Wang Z: Luteolin inhibits breast
cancer development and progression in vitro and in vivo by
suppressing notch signaling and regulating miRNAs. Cell Physiol
Biochem. 37:1693–1711. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sato Y, Sasaki N, Saito M, Endo N, Kugawa
F and Ueno A: Luteolin attenuates doxorubicin-induced cytotoxicity
to MCF-7 human breast cancer cells. Biol Pharm Bull. 38:703–709.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sui JQ, Xie KP and Xie MJ: Inhibitory
effect of luteolin on the proliferation of human breast cancer cell
lines induced by epidermal growth factor. Sheng Li Xue Bao.
68:27–34. 2016.PubMed/NCBI
|
|
40
|
Mizushima N and Komatsu M: Autophagy:
Renovation of cells and tissues. Cell. 147:728–741. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Quan W, Jung HS and Lee MS: Role of
autophagy in the progression from obesity to diabetes and in the
control of energy balance. Arch Pharm Res. 36:223–229. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Szczesny B, Brunyánszki A, Ahmad A, Oláh
G, Porter C, Toliver-Kinsky T, Sidossis L, Herndon DN and Szabo C:
Time-dependent and organ-specific changes in mitochondrial
function, mitochondrial DNA integrity, oxidative stress and
mononuclear cell infiltration in a mouse model of burn injury. PLoS
One. 10:e01437302015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Li X, Xu HL, Liu YX, An N, Zhao S and Bao
JK: Autophagy modulation as a target for anticancer drug discovery.
Acta Pharmacol Sin. 34:612–624. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lamy L, Ngo VN, Emre NC, Shaffer AL III,
Yang Y, Tian E, Nair V, Kruhlak MJ, Zingone A, Landgren O, et al:
Control of autophagic cell death by caspase-10 in multiple myeloma.
Cancer Cell. 23:435–449. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Luo YH, Wu SB, Wei YH, Chen YC, Tsai MH,
Ho CC, Lin SY, Yang CS and Lin P: Cadmium-based quantum dot induced
autophagy formation for cell survival via oxidative stress. Chem
Res Toxicol. 26:662–673. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Yamanaka T, Shiraki K, Sugimoto K, Ito T,
Fujikawa K, Ito M, Takase K, Moriyama M, Nakano T and Suzuki A:
Chemotherapeutic agents augment TRAIL-induced apoptosis in human
hepatocellular carcinoma cell lines. Hepatology. 32:482–490. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Shankar S and Srivastava RK: Enhancement
of therapeutic potential of TRAIL by cancer chemotherapy and
irradiation: mechanisms and clinical implications. Drug Resist
Updat. 7:139–156. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Chen JJ, Chou CW, Chang YF and Chen CC:
Proteasome inhibitors enhance TRAIL-induced apoptosis through the
intronic regulation of DR5: involvement of NF-kappa B and reactive
oxygen species-mediated p53 activation. J Immunol. 180:8030–8039.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kim H, Kim EH, Eom YW, Kim WH, Kwon TK,
Lee SJ and Choi KS: Sulforaphane sensitizes tumor necrosis
factor-related apoptosis-inducing ligand (TRAIL)-resistant hepatoma
cells to TRAIL-induced apoptosis through reactive oxygen
species-mediated up-regulation of DR5. Cancer Res. 66:1740–1750.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Jung EM, Lim JH, Lee TJ, Park JW, Choi KS
and Kwon TK: Curcumin sensitizes tumor necrosis factor-related
apop-tosis-inducing ligand (TRAIL)-induced apoptosis through
reactive oxygen species-mediated upregulation of death receptor 5
(DR5). Carcinogenesis. 26:1905–1913. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Horinaka M, Yoshida T, Shiraishi T, Nakata
S, Wakada M and Sakai T: The dietary flavonoid apigenin sensitizes
malignant tumor cells to tumor necrosis factor-related
apoptosis-inducing ligand. Mol Cancer Ther. 5:945–951. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Chen L, Meng Y, Guo X, Sheng X, Tai G,
Zhang F, Cheng H and Zhou Y: Gefitinib enhances human colon cancer
cells to TRAIL-induced apoptosis of via autophagy- and JNK-mediated
death receptors upregulation. Apoptosis. 21:1291–1301. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Cheng H, Hong B, Zhou L, Allen JE, Tai G,
Humphreys R, Dicker DT, Liu YY and El-Deiry WS: Mitomycin C
poten-tiates TRAIL-induced apoptosis through p53-independent
upregulation of death receptors: Evidence for the role of c-Jun
N-terminal kinase activation. Cell Cycle. 11:3312–3323. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Thamkachy R, Kumar R, Rajasekharan KN and
Sengupta S: ERK mediated upregulation of death receptor 5 overcomes
the lack of p53 functionality in the diaminothiazole DAT1 induced
apoptosis in colon cancer models: Efficiency of DAT1 in Ras-Raf
mutated cells. Mol Cancer. 15:222016. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Shoeb M, Ramana KV and Srivastava SK:
Aldose reductase inhibition enhances TRAIL-induced human colon
cancer cell apoptosis through AKT/FOXO3a-dependent upregulation of
death receptors. Free Radic Biol Med. 63:280–290. 2013. View Article : Google Scholar : PubMed/NCBI
|