Introduction
Laboratory animals are indispensable for biomedical
research. They are used to develop disease models to better
understand the pathogenesis of human diseases and to develop novel
treatment options. In cancer research, transplanted syngeneic,
xenografted and orthotopic tumor models or chemically induced
tumors in mice or rats have been used for decades (1). Despite these widely used experimental
animal models, it has been overlooked in human oncology that
animals may suffer from spontaneously arising tumors in a
comparable fashion to human patients. These spontaneous animal
tumors are much closely associated with the situation in humans
compared with experimental tumor models, as they arise
spontaneously and are neither maintained by transplantation nor are
they induced by chemical carcinogens (2). Therefore, spontaneous veterinary
tumors represent an attractive, although underestimated,
opportunity to study novel treatment strategies prior to clinical
application in human patients with cancer. As the treatment of
spontaneous tumors in animals is also of importance in veterinary
medicine, studies on veterinary tumors are also of great impact for
veterinary oncology. The survival prognosis of malignant tumors in
small animals is far from satisfactory and the majority succumb to
the disease even after application of surgical, radio- or
chemotherapeutic interventions (3,4). The
clinical prognosis and clinical, pathological and biochemical
factors influencing the survival of pets have been reported for
dogs (5-9), cats (10-12),
canines (13) and other species
(14,15). Despite considerable progress in
treating both veterinary and human tumors, the situation remains
poor and numerous patients succumb to their disease. Therefore,
novel treatment options are urgently required.
The majority of clinically established drugs are
derived from natural products (16). Therefore, the search for novel
treatments appears to be most promising when using natural sources.
A recent example for the validity of this concept is artemisinin.
Artemisinin is a bioactive terpenoid isolated from Artemisia
annua L., which is a medicinal herb that has been used for
about two millennia in traditional Chinese medicine (17). The isolation of artemisinin from
A. annua led to a novel treatment option of eminent
importance for malaria. Artemisinin and its derivatives have been
responsible for the survival of millions of patients with malaria
(18,19). This achievement was appreciated in
2015 with the conferment of the Nobel Prize for Medicine or
Physiology to the Chinese scientist Youyou Tu (20). While artemisinin derivatives, such
as artemether and artesunate, are well-established as anti-malarial
drugs, herbal preparations of A. annua also inhibit
Plasmodia infections in patients with malaria (21). Notably, the bioactivity of
artemisinin is not restricted to malaria, and other diseases are
also susceptible to artemisinin and A. annua treatment, such
as schistosomiasis and trypano-somiasis (22-24),
diverse viral infections (25) and
diseases related to the metabolic syndromes, including obesity,
diabetes and atherosclerosis (26-28).
Artemisinin derivatives also inhibit human tumor
cell growth in vitro and in vivo (29-31).
This is relevant not only for cancer therapy, but also for cancer
prevention (32,33). Artemisinin derivatives exert
additive or synergistic interactions in combination with a wide
array of clinically established drugs (34-36).
This has also been demonstrated in veterinarian tumor cell lines
in vitro and veterinarian clinical trials (37-39).
Based on the anticancer activity in experimental tumor models, it
has been possible to investigate the anticancer activity in human
cancer patients in the form of compassionate uses (40,41)
and even to perform clinical phase I/II trials in human cancer
patients (42-44). Another previous clinical phase I/II
trial indicated the anticancer activity in a number of dogs with
tumors (39). Preliminary results
in three dogs and one cat revealed that a herbal preparation of
A. annua (Luparte®) may exhibit the potential to
prolong the survival time of animals with tumors (45). To substantiate these preliminary
results from the compassionate use of A. annua, the present
study has performed a retrospective analysis by evaluating survival
times of 25 pets treated with A. annua compared with 11
animals without A. annua treatment. Independent of A.
annua food supplementation, all animals were subjected to
standard treatment protocols. In addition, the expression of two
biomarkers, transferrin receptor (TfR) and the proliferation marker
Ki-67, was determined by immu-nohistochemistry analysis of tumor
biopsies.
Materials and methods
Artemisinin determination in A.
annua
Nuclear magnetic resonance (NMR) spectra were
recorded on a Bruker 300 NMR spectrometer (Bruker Corporation).
Reversed phase high-performance liquid chromatography (HPLC)-mass
spectrometry (MS) analysis was performed on a Waters Alliance 2695
LC (Waters Corporation) coupled to a Quattro Ultima triple
quadrupole MS (Waters Corporation) using the same separation
conditions as described previously (46). The separation conditions were
follow: Chromatogram column, XBridge™ column (4.6×150 mm, 5
µm); column temperature, 20°C; and injection volume, 1
µl. Elution was performed at a flow rate of 1 ml/min, using
as the mobile phase a mixture of water (A) and acetonitrile (B).
The samples were eluted using the following gradient: 0 min, 98.0%
A; 0-8 min, linear increase to 100% B; 100% A held for 2 min, 11-12
min, return to 98.0% A. The final optimization of ESI source
operation parameters were a nitrogen gas flow of 11 l/min,
nebulizer of 30 psi, capillary voltage of ±2.4 kV and a drying gas
temperature of 250°C (N2). The Quattro Ultima triple quadrupole MS
operated in multiple reaction monitoring mode with a resolution of
0.7 m/z. Electrospray ionization (positive mode) and photodiode
array detection at 254 nm were performed.
Different batches of A. annua
(Luparte®) were determined by the Department of
Pharmaceutical Biology, Johannes Gutenberg University (Mainz,
Germany). This blinded approach was used as a quality measure to
independently guarantee the presence of artemisinin in
Luparte®. Each batch of A. annua powder (2×10 g)
was extracted with two different polarity solvents, methylene
chloride and methanol, at room temperature for 24 h. The extract
was concentrated in vacuo to obtain a residue of 0.8 and 1.2
g for methylene chloride and methanol, respectively. Each extract
was transferred to a small vial and kept dry at room temperature.
Small crystals were formed, which were collected and washed with
n-hexane and methylene chloride to remove extract residues.
Then, 1H and 13C NMR analyses were
performed.
Tumors
The animals were treated between 2010 and 2017.
Briefly, 16 dogs (10 males and 6 females) and 4 cats (2 females, 1
male and 1 N/A) were treated with A. annua. The mean age of
the dogs was 10.31 years and the mean age of the cats was 12.25
years. The mean weight of the dogs was 30.125 kg and the mean
weight of the cats was 5.10 kg. Tissue samples were taken from all
animals and initially fixed at room temperature (~22°C) in 4%
formaldehyde solution for 48 h. Following fixation, the organs were
trimmed, processed and embedded in paraffin wax at approximately
60°C. Sections were cut to a thickness of 4 µm and routinely
stained with hematoxylin and eosin at room temperature.
Histopathological examination of the hematoxylin and eosin-stained
sections was performed under a light microscope in immersion oil
with a Zeiss Axioskop. After routine pathological diagnosis, the
paraffin blocks were used as excess material for subsequent
immunohistochemical studies. Furthermore, the histological
hematoxylin and eosin stained tumor sections were assessed for the
presence of the tumor-infiltrating lymphocytes (TILs). The clinical
data are presented in Tables I and
II. The owners of the animals
provided written informed consent for this retrospective study. The
signed written consent forms are deposited at the Department of
Pharmaceutical Biology, Johannes Gutenberg University (Mainz,
Germany) and can be inspected upon reasonable demand.
 | Table IClinical data of animals treated with
Artemisia annua after standard treatment. |
Table I
Clinical data of animals treated with
Artemisia annua after standard treatment.
A, Carcinoma
|
|---|
| Species | Breed | Sex | Weight, kg | Date of diagnosis
and start of treatment | Tumor type | Survival time,
months | Initial blood iron
content, μg/dl | Treatment before
A. annua | Age, years | Source of the
tumor |
|---|
| Dog | Doberman | F | 33 | May 2, 2014 | Adenocarcinoma | 1.0 | 74 | a | 8 | Subcutis |
| Dog | White Swiss
Shepherd | M | 32 | Jan 1, 2015 | Adenocarcinoma | 5.0 | 111 | a | 9 | Mammary glands |
| Dog | Pyrenean
Shepherd | F | 11 | Apr 28, 2015 | Adenocarcinoma | 3.0 | 95 | a | 13 | Subcutis |
| Dog | Poodle | Fcastr | 9 | Mar 17, 2011 | carcinoma | 48.0 | 134 | c | 14 | Mammary glands |
| Dog | Mix | F | 55 | N/A | carcinoma | 1.0 | 223 | a | 9 | Lung |
| Dog | Border Collie | M | 16 | Oct 17, 2016 | carcinoma | 2.5 | 106 | a | 1 | Lung |
| Cat | European
Shorthair | Fcastr | 4 | Dec 21, 2012 | Squamous
carcinoma | 5.0 | 45 | b | 16 | Gingiva |
| Cat | European
Shorthair | N/A | N/A | Sep 14, 2010 | Non-specified
carcinoma | 38.0 | 222 | b | 12 | Abdominal skin |
| Dog | Labrador | M | 42 | Aug 23, 2012 | Keratinizing
carcinoma | 4.0 | 93 | b | 9 | Nose skin |
| Dog | Mix | Mcastr | 35 | Jul 22, 2011 | Hepatoma | 8.0 | 164 | a | 13 | Liver |
|
B, Sarcoma
|
| Species | Breed | Sex | Weight, kg | Date of diagnosis
and start of treatment | Tumor type | Survival time,
months | Initial blood iron
content, μg/dl | Treatment before
A. annua | Age, years | Source of the
tumor |
|
| Cat | European
Shorthair | Mcastr | 5.5 | Feb 21, 2014 | Fibrosarcoma | 0.5 | 66 | c | 10 | Abdominal
subcutis |
| Dog | Gordon Setter | Fcastr | 26 | Aug 24, 2010 | Fibrosarcoma | 72.0 | 85 | c | 10 | Subcutis |
| Dog | Mix | M | 38 | Feb 04, 2010 | Fibrosarcoma | 13.0 | 107 | c | 13 | Subcutis |
| Cat | European
Shorthair | Fcastr | 5.8 | Jul 11, 2011 | Fibrosarcoma | >68 | 84 | c | 11 | Abdominal
subcutis |
| Dog | Rottweiler | M | 51 | Nov 16, 2011 | Fibrosarcoma | 1.0 | 97 | c | 8 | Subcutis |
| Dog | Mix | M | 26 | Jan 25, 2011 |
Hemangiosarcoma | >36 | 124 | c | 10 | Spleen |
| Dog | Mix | Fcastr | 27 | Jan 31, 2012 |
Hemangiosarcoma | >18 | 131 | c | 14 | Spleen |
| Dog | Labrador | Mcastr | 33 | Apr 05, 2013 |
Hemangiosarcoma | 41.0 | 81 | c | 10 | Spleen |
| Dog | Mix | M | 22 | Jan 22, 2014 |
Hemangiosarcoma | 12.0 | 354 | c | 13 | Spleen |
| Dog | Mix | Mcastr | 26 | Apr 02, 2014 | Lymphosarcoma | 13.0 | 116 | c | 11 | Lymph node, left
scapula |
 | Table IIClinical data of animals without
Artemisia annua treatment after standard treatment. |
Table II
Clinical data of animals without
Artemisia annua treatment after standard treatment.
A, Carcinoma
|
|---|
| Species | Breed | Sex | Weight, kg | Date of diagnosis
and start of treatment | Tumor type | Survival time,
months | Initial blood iron
content, µg/dl | Treatment before
A. annua | Age, years | Source of the
tumor |
|---|
| Dog | Mix | F | 6.8 | Jul 22, 2011 | Adenocarcinoma | N/A | 6 | c | 12 | Mammary glands |
| Cat | European
Shorthair | Fcastr | 3.8 | Aug 22, 2012 | Adenocarcinoma | 84.0 | 4 | c | 16 | Mammary glands |
| Dog | Labrador | Fcastr | 36 | Jan 21, 2011 | Adenocarnoma | 112.0 | 3 | a | 9 | Mammary glands |
| Dog | Mix | Fcastr | 8.6 | Dec 29, 2014 | Hepatoma | 107.0 | 2 | a | 7 | Liver |
| Cat | European
Shorthair | Mcastr | 4.2 | Nov 07, 2014 | Kidney
carcinoma | 69.0 | 4 | c | 13 | Kidney |
|
B, Sarcoma
|
| Species | Breed | Sex | Weight, kg | Date of diagnosis
and start of treatment | Tumor type | Survival time,
months | Initial blood iron
content, µg/dl | Treatment before
A. annua | Age, years | Source of the
tumor |
|
| Cat | European
Shorthair | Mcastr | 6.0 | Aug 06, 2012 | Fibrosarcoma | 62.0 | 7 | c | 14 | Abdominal
subcutis |
| Dog | Mix | Mcastr | 11.8 | May 27, 2014 |
Hemangiosarcoma | 220.0 | 7 | c | 13 | Spleen |
| Dog | Mix | Mcastr | 38.6 | Sep 18, 2012 |
Hemangiosarcoma | 136.0 | 10 | c | 13 | Spleen |
| Dog | Dachshund | Fcastr | 14.2 | Jan 31, 2012 | Liposarcoma | N/A | 17 | c | 11 | Eye lid |
| Dog | Mix | F | 42.0 | Aug 27, 2012 |
Rhabdomyosarcoma | 83.0 | 0.1 | a | 11 | Thigh skeletal
muscles with metastases in liver and lymph nodes |
| Cat | European
Shorthair | N/A | N/A | Apr 16, 2015 |
Hemangiopericytoma | 80.0 | 16 | c | 13 | Abdominal
subcutis |
Treatment protocol
The serum iron content was determined following
collection of blood samples; a normal range is between 140 and 170
µg/dl (47). After initial
iron content determination using ferrozine color test (48), the tumors were surgically removed
with safety margins by a standard protocol after visual inspection
and where required after lung radiography. Between blood collection
and determination of the serum iron results from the clinical
diagnosis laboratory, iron was administered orally
[Ferrosanol® capsules (100 mg/capsule or 40
mg/capsule)]. The dosage was 100 mg/30 kg BW twice daily mixed into
vegetable-poor food to avoid iron binding plant molecules, such as
phytane, oxalates and/or phosphates. Alternatively, iron was
subcutaneously injected (Myofer®/Ursoferran®;
100 mg/ml) at a dose of 100 mg/10 kg BW daily, until the serum iron
results were obtained back from the clinical laboratory. The iron
substitution was individually continued until an iron content of
250±30 µg/dl (~43±5 µmol/l) was stably reached in
blood serum. From the fourth day of the initial iron substitution
onward, the animals were orally treated with Luparte®
capsules at a dosage of 1,400 mg/m2 body surface divided
to three fractions per day (Table
III). The capsules were administered 1-2 h before the next
meal. The serum iron content was regularly monitored and where
necessary adapted to 250±30 µg/dl.
 | Table IIIConversion table for
Luparte® treatment. |
Table III
Conversion table for
Luparte® treatment.
| Body weight,
kg | Body surface,
m2 |
|---|
| 0.5 | 0.06 |
| 1 | 0.1 |
| 2 | 0.16 |
| 5 | 0.29 |
| 10 | 0.46 |
| 15 | 0.61 |
| 20 | 0.74 |
| 30 | 0.97 |
| 40 | 1.17 |
| 50 | 1.36 |
| 60 | 1.63 |
| 70 | 1.7 |
The treatment details and the follow-up information,
including survival times, have been recorded in the clinical
documentation files. All ethical issues have been discussed prior
to evaluation of clinical and experimental data with the
Regierungspräsidium (Government Presidium) Freiburg
(Germany). According to the design of this retrospective study, the
Regierungspräsidium (Government Presidium) Freiburg gave
written permission for the present study (Az. 35-9185.81/1, dated
from February 4th, 2019).
Cell lines
The D-GBM cell line was generated by Dr George
Stoica (Department of Veterinary Pathobiology, Texas A&M
University, College Station, TX, USA) from a brain tumor of an
8-year-old male Boxer dog (49).
The D-GBM cells used in the present study were obtained from Dr
Pablo Steinberg (Institute for Food Toxicology and Analytical
Chemistry, University of Veterinary Medicine Hannover, Hannover,
Germany), who obtained it from Dr G. Stoica.
The histiocytic DH82 cell line was generated by Dr
M.L. Wellman (Department of Veterinary Pathobiology, College of
Veterinary Medicine, Ohio State University, CO, USA) (50). The DH82 cell line used in the
present study was also obtained from Dr Pablo Steinberg, who
purchased it from the European Tissue Culture Collection (catalog
no. 94062922).
A panel of 54 human tumor cell lines of the
Developmental Therapeutics Program (DTP) of the National Cancer
Institute consisted of cell lines derived from leukemia, melanoma,
non-small cell lung cancer, colon cancer, renal cancer, ovarian
cancer, breast cancer and prostate carcinoma, as well as central
nervous system tumors (51).
Previously, the cytotoxicity of cells treated with artemisinin for
48 h was evaluated using a sulforho-damine B assay (52). The log10 IC50
values for artemisinin and mRNA expressions for Ki-67 and TfR were
available publicly in the DTP database (https://dtp.cancer.gov/default.htm).
Cytotoxicity assay
A resazurin reduction assay (Promega Corporation)
was performed as previously described (53) to assess the cytotoxicity of
artemisinin, artesunate and dihydro-artemisinin towards the dog
tumor cell lines DH82 and DGBM. The concept of the assay is based
on the metabolic reduction of the non-fluorescent dye by living
cells to the strongly-fluorescent dye resorufin (54). IC50 values were
calculated from dose-response curves using a non-linear regression
analysis tool in GraphPad Prism 7 software (GraphPad Prism, Inc.).
All IC50 values are expressed as the mean ± standard
deviation. Each assay was performed thrice independently, with six
replicates each.
Immunohistochemistry
A total of 17 tumor tissues representing different
tumor types in pets were collected to evaluate the expression of
Ki-67 and TfR in animal tumors (Table
V). Briefly, tumor tissues were obtained from 15 dogs (7 males
and 8 females) and 2 cats (both female). The mean age of the dogs
was 9.33 years and the mean age of the cats was 4 years. The mean
weight of the dogs was 27.266 kg and the mean weight of the cats
was 4.00 kg. All the chosen animals had been kept in pet animal
housing conditions. The immuno-histochemical staining of tumor
tissues was performed as described previously (55). The slides were washed twice with
xylene (98.5% xylene for 5 min each at room temperature) to remove
paraffin. Then, sample tissues were rehydrated through graded
washes with isopropanol in water. Heat-induced epitope retrieval
was performed using a pressure cooker as a heating device.
Ultra-vision protein block and UltraVision Hydrogen Peroxide Block
(catalog nos. TA-060-UB and TA-060-H2O2Q, respectively; Thermo
Fisher Scientific, Inc.) were added to block endogenous proteins
and endogenous peroxidase activity, respectively, to avoid
non-specific background staining. Overnight incubation at 4°C was
performed following the addition of monoclonal primary antibodies.
For the detection of Ki-67, the SP6 clone (catalog no. ab16667;
Abcam) was used at a dilution of 1:200. For the determination of
TfR expression, the H68.4 clone (catalog. no. 136800; Thermo Fisher
Scientific, Inc.) was applied at a dilution of 1:100. Subsequently,
horseradish peroxidase-labeled polymers conjugated with secondary
antibodies specific for both mouse and rabbit primary antibodies
were added (catalog nos. TL-060-QPH and TL-060-QPB; Thermo Fisher
Scientific, Inc.) at room temperature for 1 h, according to the
manufacturer's protocol. The final staining reaction was performed
with diaminobenzidine and slides were counterstained with
hematoxylin for 3 min at room temperature.
 | Table VExpression of TfR and the
proliferation marker Ki-67 in dog tumors according to
immunohistochemistry. |
Table V
Expression of TfR and the
proliferation marker Ki-67 in dog tumors according to
immunohistochemistry.
| Case | Species | Breed | Weight, Sex | kg | Age, years | Percentage of
TfR-positive cells | Percentage-of Ki-76
postive cells | Histology |
|---|
| 1 | Cat | European
shorthair | Fcastr | 4 | 3 | 94.3±9.0 | 85.7±4.7 | Tubulopapillary
breast adenocarcinoma |
| 2 | Dog | Mix | Fcastr | 17 | 13 | 95.1±3.6 | 84.8±5.9 | Tubulopapillary
breast adenocarcinoma |
| 3 | Dog | Weimarian | M | 28 | 8 | 99.7±0.1 | 72.5±4.4 | Soft tissue
sarcoma, Grade 1 |
| 4 | Dog |
Deutsch-Adrahthaar | Fcastr | 28 | 9 | 87.2±9.6 | 71.6±9.7 | Tubulopapillary
breast adenocarcinoma with lymph node metastasis |
| 5 | Dog | French bulldog | Mcastr | 11 | 7 | 83.9±6.8 | 53.5±18.7 | Soft tissue
sarcoma, Grade 1 |
| 6 | Dog | American
Staffordshire Terrier | Mcastr | 33 | 3 | 98.6±0.5 | 49.1±4.9 | Mastocytoma, Type
2 |
| 7 | Dog | Mix | M | 21 | 12 | 52.1±32.1 | 43.5±3.1 |
Hemangiosarcoma |
| 8 | Dog | Podenco | Fcastr | 24 | 12 | 99.9±0.1 | 41.1±16.3 | Osteosarcoma |
| 9 | Dog | Blue lacy | M | 26 | 11 | 99.8±0.2 | 33.9±5.0 | Mastocytoma, Type
2 |
| 10 | Dog | Mix | F | 14 | 2 | 43.5±37.9 | 33.2±13.8 | Mastocytoma, Type
2 |
| 11 | Cat | European
shorthair | Fcastr | 4 | 5 | 97.4±1.8 | 31.3±8.3 | Neuroendocrine
carcinoma |
| 12 | Dog | Podenco | Fcastr | 25 | 12 | 84.6±9.5 | 30.5±10.7 | Osteosarcoma,
highly malignant |
| 13 | Dog | Rottweiler | Fcastr | 51 | 8 | 99.0±0.1 | 30.1±14.6 | Adenocarcinoma
metastasis |
| 14 | Dog | Mix | Mcastr | 27 | 10 | 99.1±1.4 | 29.5±8.4 | Teleangiectatic
osteosarcoma |
| 15 | Dog | Labrador mix | F | 37 | 12 | 95.2±3.9 | 28.6±7.4 | Carcinoma |
| 16 | Dog | Labrador | M | 43 | 9 | 99.2±0.6 | 21.3±9.7 | Squamous cell
carcinoma |
| 17 | Dog | Podenco | Fcastr | 24 | 12 | 53.3±17.8 | 6.6±1.8 | Breast carcinoma,
complex/myxoid |
The immunostaining of DH82 and DGBM cell lines was
performed without the rehydration or epitope retrieval steps.
Briefly, six cover slips were placed in a 6-well plate, then
5×105 cells/well were seeded over the cover slips and
incubated in humidified 5% CO2 atmosphere at 37°C
overnight to let the cells attach to coverslips. Subsequently, the
cells were fixed in 4% paraformaldehyde for 15 min at room
temperature, and the blocking and staining steps were performed as
described for the tissue section. However, the Ki-67 and TfR
antibodies were diluted at 1:500 and 1:1,000, respectively. The
immunostained slides were scanned using Pannoramic Desk (3DHISTECH
Ltd.) and the amount of Ki-67- or TfR-expressing cells was
quantified by Pannoramic Viewer software version 1.15 (3DHISTECH
Ltd.).
Statistical analysis. Significance values and
correlation coefficients were calculated using Pearson's
correlation coefficient and Fisher's exact test with the WinSTAT
software program version 2012.1 (www.winstat.com/). Linear regression was performed
using Excel 2016 (Microsoft Corporation) for the log10
IC50 values for artemisinin and mRNA expressions for
Ki-67 and TfR. P<0.05 was considered to indicate a statistically
significant difference.
Results
Identification of artemisinin. As
presented in Fig. 1A, the
1H NMR spectrum of a colorless crystal
contained signals typical of artemisinin. The spectrum displayed
signals for three methyl groups: Two secondary at δH
1.00 (d, J=6.5, H-14), 1.17 (d, J=7.2, H-13) and one
tertiary methyl signal at δH 1.40 (s, H-15).
Additionally, two aliphatic methylene signals were observed at
δH 2.12 (brddd, J=14.9,4.2,2.6) and 2.40 (ddd,
J=14.9, 13.1,4.1) for H-3a and H-3b, respectively. A
characteristic downfield singlet signal at δH 6.01
indicated the presence of oxygenated proton for H-5. As presented
in Fig. 1B, The 13C NMR
spectrum displayed 15 major carbon signals, including significant
carbons signals confirming the presence for artemisinin as the
following: δC 173.3 (s, C-12), 105.2 (s, C-4), 94.3 (d,
C-5) and 79.5 (s, C-6) (56).
Reversed phase HPLC-MS was used to quantitatively
analyze the presence of artemisinin. Fig. 1C presents the chro-matogram
obtained after dissolving 2 mg/ml MeOH extract, which indicated
that artemisinin represents 3.46% of the A. annua
(Luparte®) extract.
Clinical treatment
In total, 23 dogs and 8 cats were included in the
present study. Among these, 20 were treated with A. annua in
addition to their standard therapy (Table I) and 11 were subjected to standard
treatment alone (Table II). The
A. annua-treated group consisted of 10 carcinomas (8 dogs, 2
cats) and 10 sarcomas (8 dogs, 2 cats). All animals were treated
between 2010 and 2017 (Table I).
In the group of animals without A. annua treatment, 5
animals presented with carcinoma (3 dogs, 2 cats) and 6 animals had
sarcoma (4 dogs, 2 cats) (Table
II). The blood iron content and the survival times of all pets
were recorded.
The effect of A. annua treatment on overall
survival time was assessed. In total, 13 A. annua-treated
animals and 11 non-treated animals exhibited a survival time of
<18 months following therapy. Whereas, 7 animals in the A.
annua-treated group had a survival time of >18 months after
A. annua treatment, but no animals in the non-treated group
survived >18 months. This difference in survival times between
the groups was statistically significant (P=0.033; two-tailed
Fisher's exact test; Table
IV).
 | Table IVCorrelation of Luparte®
treatment and survival time. |
Table IV
Correlation of Luparte®
treatment and survival time.
| Survival time,
(months) | Treatment | No treatment |
|---|
| <18 | 13 | 11 |
| ≥18 | 7 | 0 |
Histology and immunohistochemistry
Using a second collection of 17 tumors, the
histology of the tumors was determined by hematoxylin and eosin
staining of formalin-fixed and paraffin-embedded tumor sections.
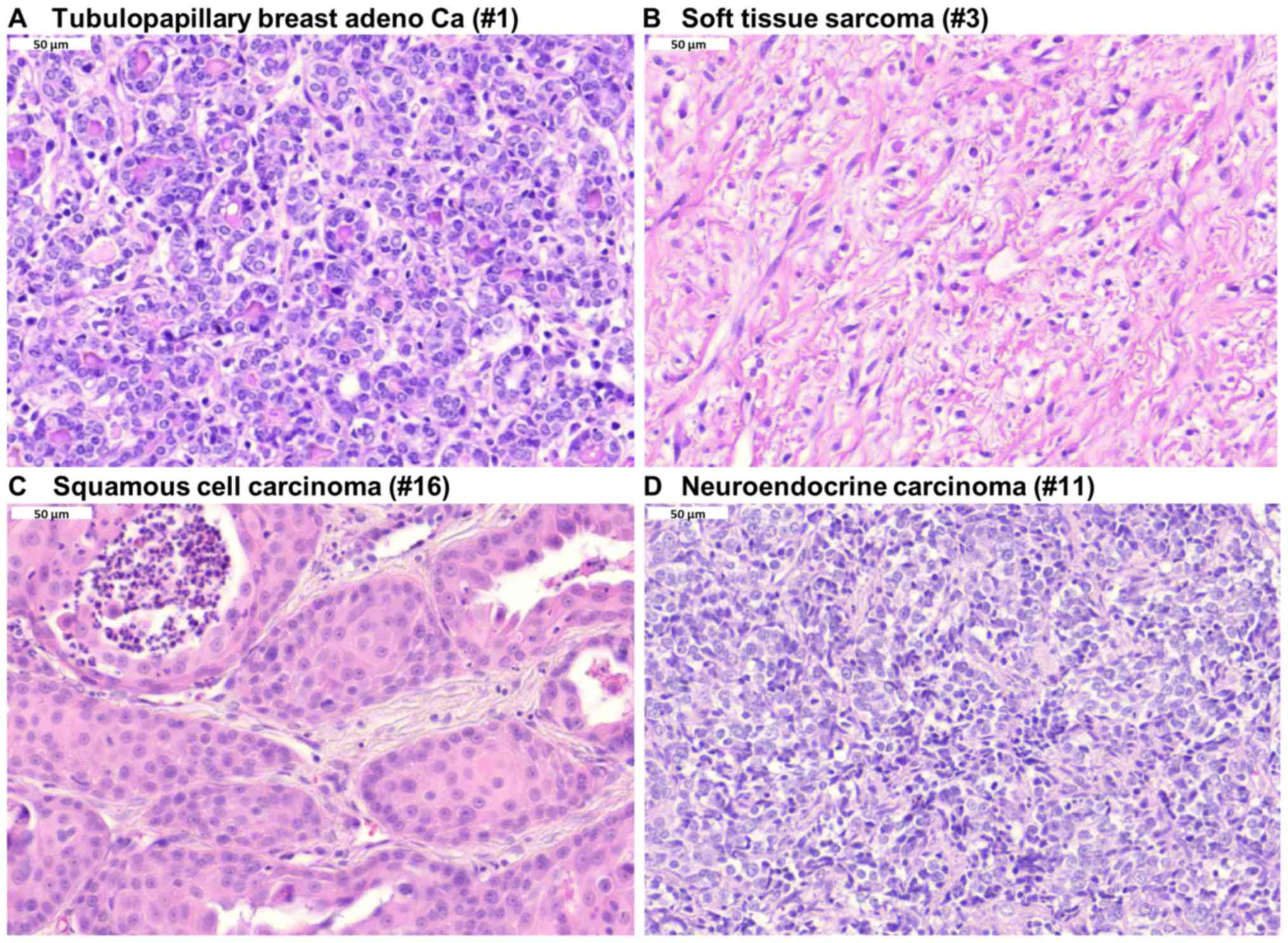
Representative photographs of different tumor histology are
presented in Fig. 2, including a
tubulopapillary breast adenocarcinoma, soft tissue sarcoma,
squamous cell carcinoma and neuroendocrine carcinoma.
Furthermore, immunohistochemical analyses were
performed. The membrane-bound expression of TfR (CD71) and the
nuclear expression of the proliferation marker Ki-67 were
determined. The percentage of stained cells was quantified using a
computer-based quantification system. Six different areas were
selected from each tumor section to provide representative
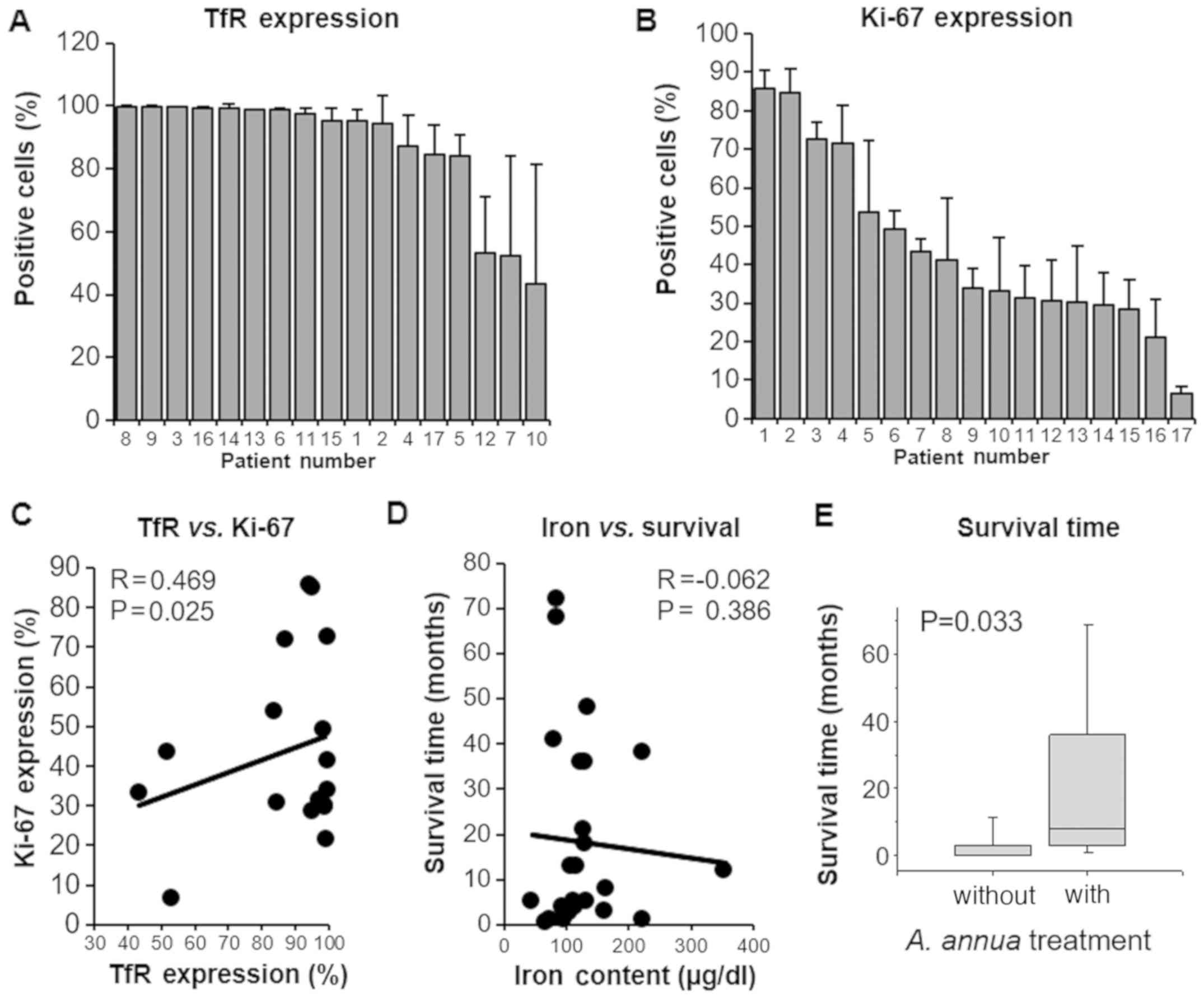
expression values. The percentage of TfR-expressing cells ranged
between 43.5 (±37.9%) and 99.2% (±0.2%), whereas the percentage of
Ki-67-positive cells was in a range between 6.6 (±1.8%) and 85.7%
(±4.7%) (Fig. 3A and B). As tumor
heterogeneity plays an important role in the response to anticancer
therapy, the present study also focused on heterogeneous and
homogeneous staining patterns among the different tumor biopsies.
Certain tumors were very heterogeneous in the immunohistochemical
staining, while others revealed uniform staining. This can be seen
in the standard deviations, which ranged between 0.1 (cases no. 3,
8 and 13) and 37.9% (case 10) for TfR, and between 1.8 (case 17)
and 16.3% (case 8) for Ki-67 (Table
V). Subsequently, the correlation between the expression of TfR
and Ki-67 with the set of tumor biopsies was investigated, which
revealed a statistically significant correlation (P=0.025; r=0.469;
Fig. 3C). No significant
correlation was identified between blood iron content and survival
time of the animals (P=0.386; r=-0.062; Fig. 3D). Notable, the association between
survival time and A. annua treatment was significant. Pets
treated with standard therapy plus A. annua has
significantly longer survival times compared with those treated
with standard therapy alone (P=0.033; Fig. 3E).
As the direct association between TfR and Ki-67
expression and the cytotoxicity of artemisinin could not be
assessed in these tumor samples, the TfR and Ki-67 microarray-based
mRNA expression in 54 human tumor cell lines was analyzed compared
with the log10 IC50 values, as determined by
sulforhodamine assay. Both TfR and Ki-67 expression were
significantly negatively correlated with the cytotoxicity of
artemisinin in these cell lines (P<0.05; r<-0.20), which
indicates a higher expression of these two markers was associated
with a higher sensitivity of the cell lines to arte-misinin
(Table VI). The expression levels
of TfR and Ki-67 were positively correlated (P=0.008; r=0.317).
This panel of 54 human tumor lines consisted of cell lines derived
from eight tumor types (leukemia, melanoma and brain tumor, and
carcinoma of colon, breast, ovary, kidney and prostate). The number
of cell lines of each tumor type (n=1-9) was too low to reveal
significant correlations except for the three following results. In
lung cancer cell lines, the log10 IC50 values
of arte-misinin were inversely correlated with the Ki-67 expression
(P=0.009; r=-0.759). In melanoma cell lines, the expression levels
of Ki-67 and TfR were significantly correlated (P=0.010; r=0.787).
In renal cancer cell lines, the expression levels of Ki-67 and TfR
were also significantly correlated (P=0.019; r=0.781) (data not
shown). Since all other associations between artemisinin response
and the expression of Ki-67 and TfR were not statistically
significant, reliable conclusions on the role of Ki-67 and TfR may
be drawn from the cell line panel as a whole, but not from tumor
type-specific subsets.
 | Table VICorrelation of artemisinin
(log10 IC50 values) with microarray-based TfR
and Ki-67 mRNA expression in 54 human tumor cell lines. |
Table VI
Correlation of artemisinin
(log10 IC50 values) with microarray-based TfR
and Ki-67 mRNA expression in 54 human tumor cell lines.
| Marker | Artemisinin
cytotoxicity | Ki-67
expression |
|---|
| TfR expression | | |
| r value | -0.282 | 0.317 |
| P-value | 0.019 | 0.008 |
| Ki-67
expression | | |
| r value | -0.239 | |
| P-value | 0.041 | |
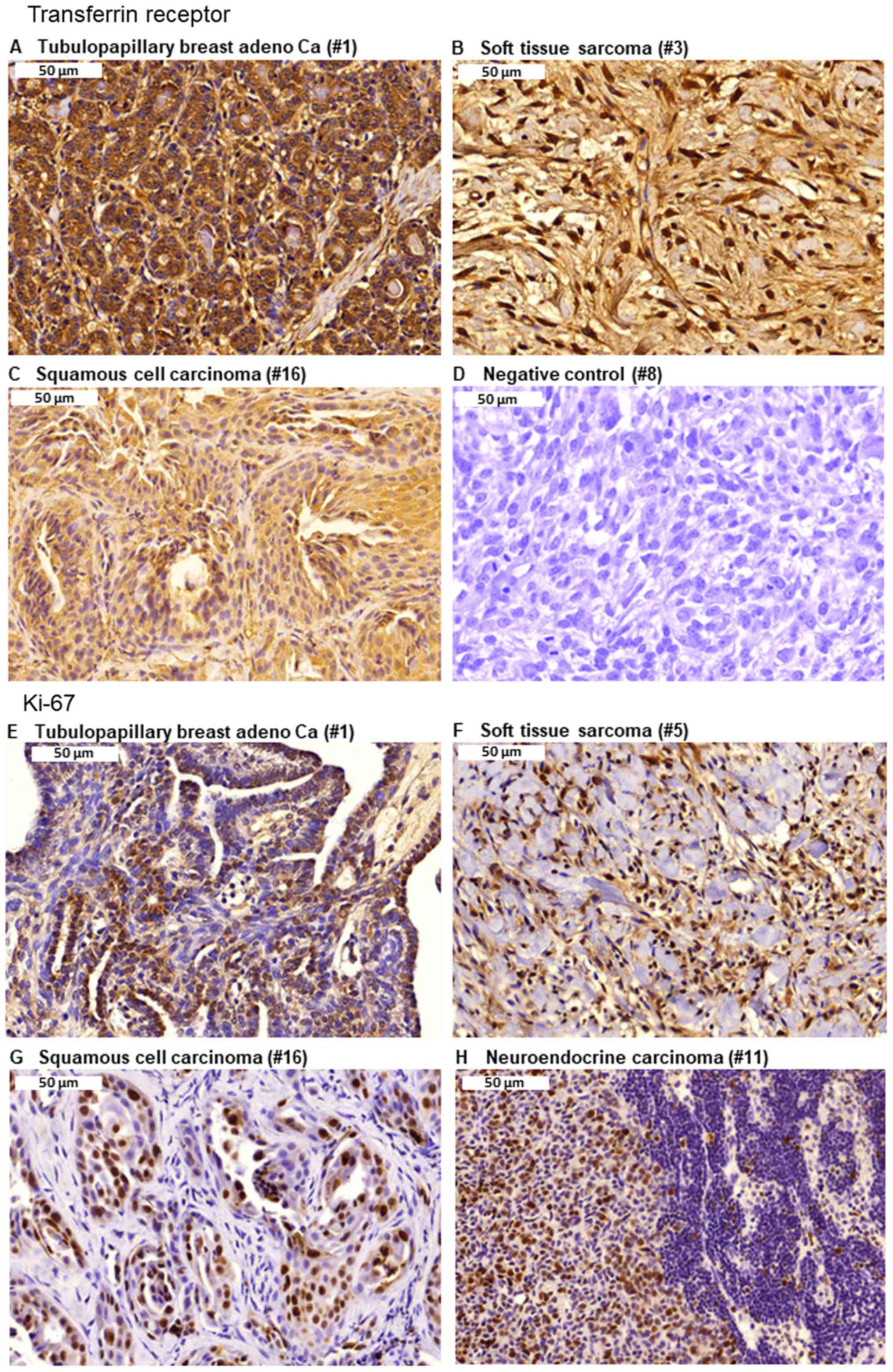
Representative images of immunostaining for TfR
expression are presented in Fig.
4A-D. Strong TfR expression was identified in biopsies of
tubulopapillary breast adenocarci-noma, soft tissue sarcoma and
squamous cell carcinoma. By contrast, the negative control did not
demonstrate any reactivity, indicating the specificity of the
immunostaining procedure. Furthermore, examples of Ki-67 expression
in tubulopapillary breast adenocarcinoma, soft tissue sarcoma,
squamous cell carcinoma and neuroendocrine carcinoma are presented
in Fig. 4E-H. Furthermore,
assessment of the presence of TILs in hematoxylin and eosin stained
tumor tissues revealed an absence of TILs in all tumor slides.
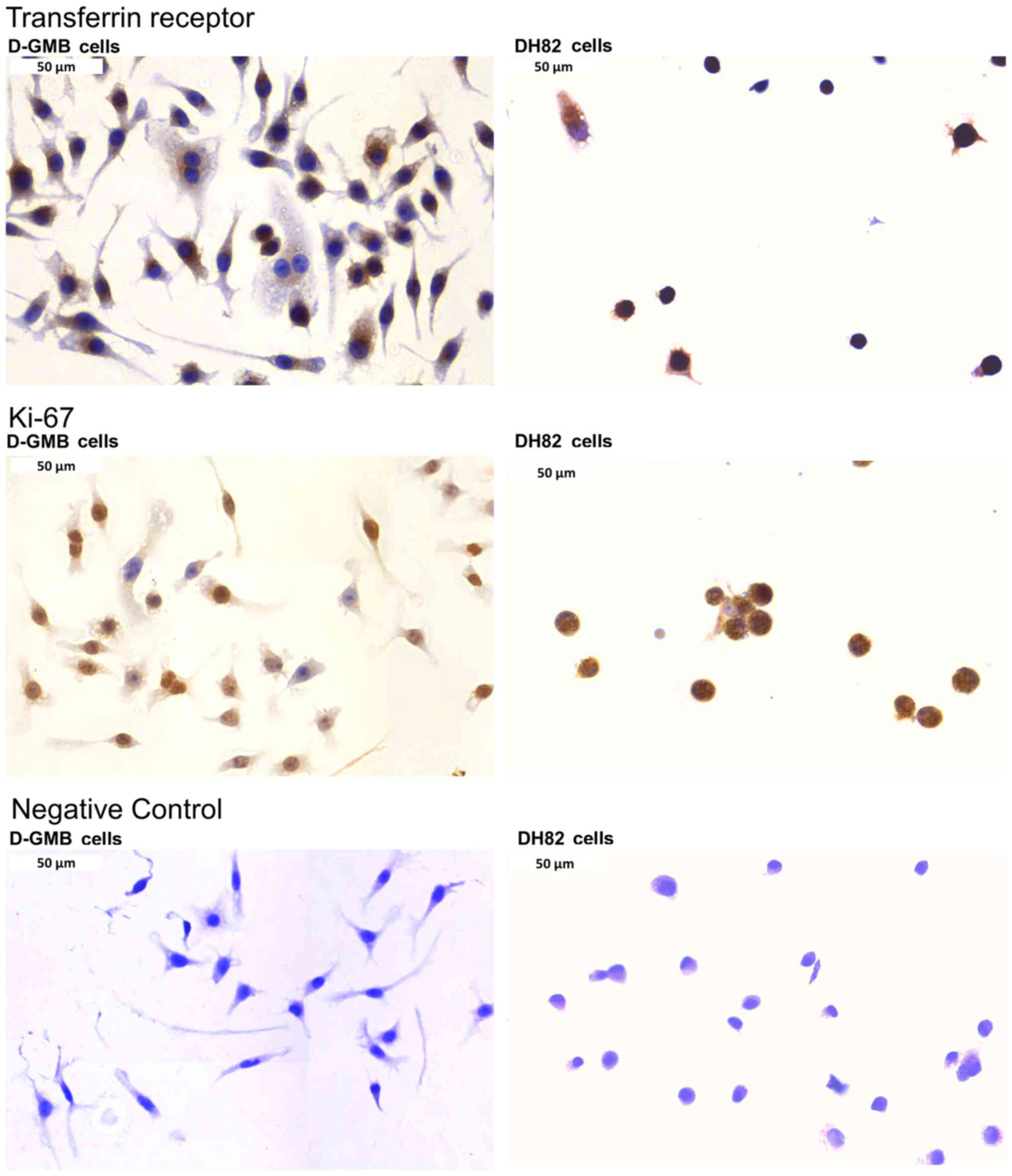
For comparison, the Ki-67 and TfR expression was
analyzed in two dog cell lines. DH82 histiocytic sarcoma and DGMB
glioblastoma cells were immunostained for both markers. Indeed,
Ki-67 and TfR were overexpressed in both cell lines as presented in
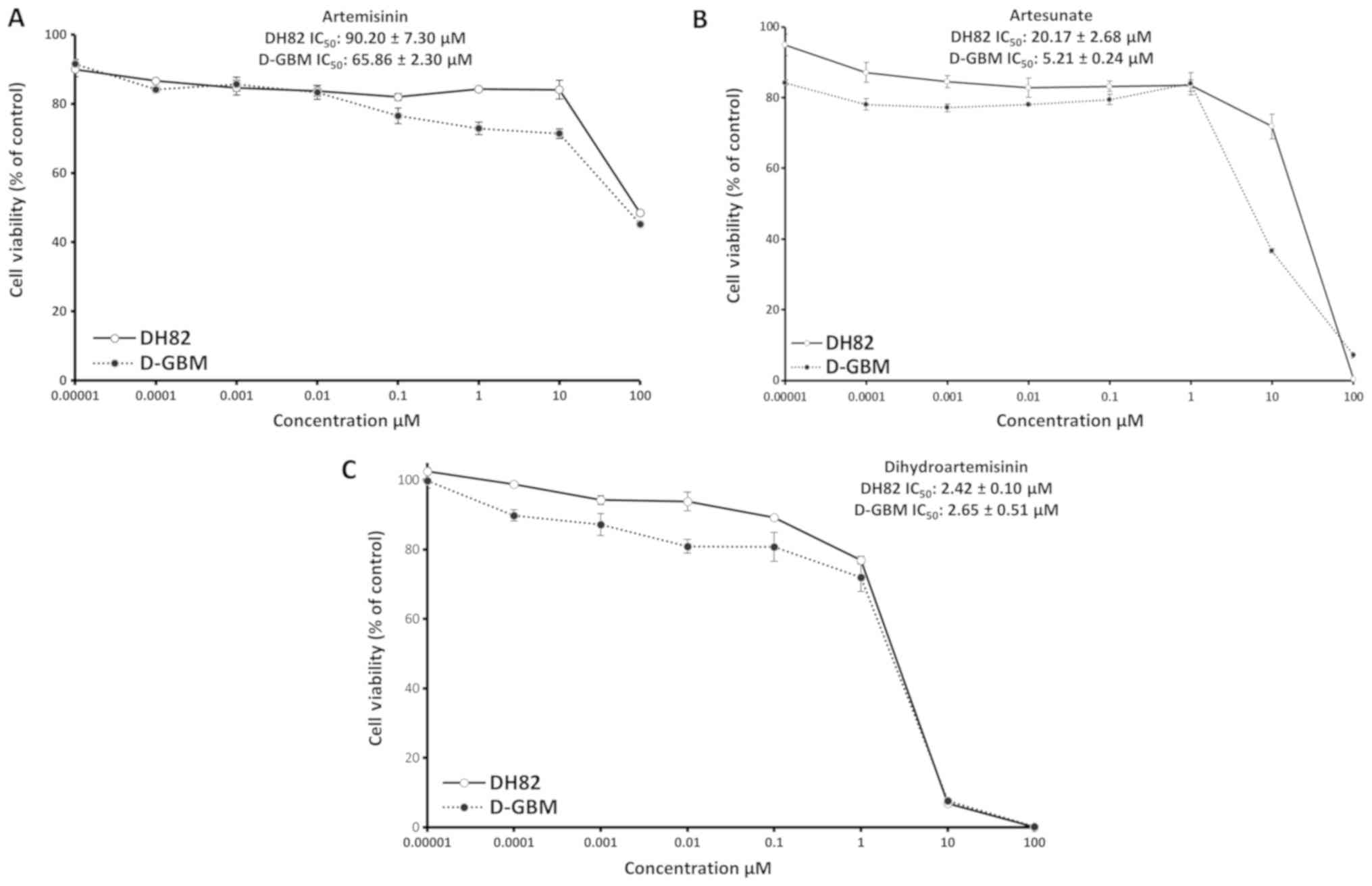
Fig. 5. Furthermore, the present
study investigated the cytotoxicity of artemisinin, artesunate and
dihydroartemis-inin towards the dog cell lines. As expected,
artemisinin and its derivatives were also active in these tumor
cell lines. The IC50 values for artemisinin, artesunate
and dihydroartemisinin towards DH82 cells and DGBM cells are
presented in Fig. 6.
Discussion
It has been a matter of discussion among
veterinarian physicians and alternative practitioners, whether or
not supplementation of iron is beneficial for the activity of
artemisinin. The role of heme has been discussed for the
antimalarial activity of artemisinin and its derivatives (57,58).
In the context of cancer, we previously reported that
Ferrosanol® and holotransferrin increase
artesunate-induced cytotoxicity and apoptosis in leukemia and
astrocytoma cells up to 10-fold. These effects were reversed by
anti-TfR monoclonal antibody RVS10, which competes with transferrin
for binding to TfR. While the TfR expression ranged between 48 and
95% in tumor cell lines, normal peripheral mononuclear blood
leukocytes revealed ≤1.3% TfR positivity, indicating that
artesunate may exert tumor specific effects at least to some
extent, because of the preferential TfR expression in tumor cells
(59).
In a subsequent study, we investigated a total of 36
cell lines of different tumor types for their response to treatment
with artesunate alone or in combination with Ferrosanol®
(60). This revealed that
artesunate plus Ferrosanol® enhances cytotoxicity
compared with artesunate alone in the majority of cell lines;
however, 11 cell lines did not demonstrate increased apoptosis and
nine lines exhibited a decrease in apoptosis following the combined
drug treatment compared with artesunate-treatment alone. It is
understood that iron acts as co-factor for proliferation-related
enzymes (61). Therefore,
Ferrosanol® may induce rather than suppress the
proliferation of these nine cell lines. Based on these in
vitro data, it cannot be reliably recommended that iron should
be added as a supplement to artemisinin-type treatment in
veterinary or human cancer therapy. The results of the present
study revealed that the blood iron content was not associated with
survival times after A. annua therapy, while the TfR content
inside the tumor was. For practical reasons, it can be assumed that
the normal iron content of the body should be sufficient for the
activity of artesunate. Only in patients with iron-deficient
conditions may iron supplementation be considered. However, in this
case it may be important that iron and artesunate reach the tumor
at the same time so that a possible enhancing effect of the drug
combination can develop. Otherwise, Ferrosanol® may
exhibit worse effects and lead to an enhancement rather than a
suppression of proliferative tumor activity.
The effect of iron has also been demonstrated in
other previous studies. It has been reported that the addition of
transferrin can inhibit cross-resistance of multidrug-resistant
H69VP small cell lung cancer cells to artemisinin (62). In addition, retinoblastoma cells
with high TfR expression are more susceptible to artesunate
compared with normal retina cells. This activity is specific, as
RNA interference-mediated TfR-knockdown increased the sensitivity
of retinoblastoma cells to artesunate (63). In accordance with these findings,
supplementation with holotransferrin increases the cytotoxicity of
dihydroartemisinin in T-cell lymphoma cells (64).
It is well documented that TfR is more highly
expressed in tumor cells compared with normal cells (61,65-68).
Therefore, experimental therapy strategies have been proposed to
address TfR as a treatment target to improve tumor-specific killing
and spare toxic side effects to normal tissues at the same time,
for example by antibody-mediated targeting of TfR or the generation
of TfR-directed immunotoxins (69-71).
The generation of transferrin-artemisinin conjugates revealed
enhanced cytotoxicity towards tumor cells compared with uncoupled
artemisinin (72-74).
Previously, we performed a clinical phase I/II trial
with artesunate in 23 dogs with cancer, which revealed 1 case of
complete remission and 7 cases of stable disease following
artesunate-treatment (39). The
results of the present retrospective study with A. annua
appear to be improved. The present study identified an increase in
survival time above the threshold of 18 months in 9 of 25 dogs
(36%). Although this trend needs to be confirmed in future studies,
it could be speculated that the anticancer effect of the whole
plant extract is better than that of isolated artemisinin or the
semisynthetic derivative, artesunate. Indeed, the plant extract
contains many more cytotoxic compounds in addition to artemisinin
(75-78), including arteanuin B, artemisitene,
scopoletin and 1,8-cineole. Therefore, A. annua preparations
alone may be used as a combination therapy. Several cytotoxic
compounds may act at the same time against the tumor, which leads
to an improved tumor inhibition.
It is well understood that rapidly proliferating
tumors respond better to standard chemotherapy compared with slowly
growing tumors (79,80). Therefore, the present study also
included Ki-67 as proliferation marker in the immunohistochemical
analysis. Ki-67 is closely associated with the cell cycle. Its role
as a predictive factor for the success of chemotherapy and as a
prognostic factor for the survival of cancer patients has been
discussed (81,82). Therefore, the significant
association between Ki-67 expression and cytotoxicity to
artemisinin in the panel of 54 human cell lines from eight
different tumor types indicated that Ki-67 expression may also be a
prognostic marker for tumor response to artemisinin.
The present study revealed a significant correlation
between TfR and Ki-67 expression in veterinary tumors and human
tumor cell lines, indicating that TfR expression is associated with
high proliferative rates and that artemisinin is more active in
fast growing tumors than in slowly growing ones. This result
supports previous data from human tumors and also speaks for the
comparability of veterinary and human tumors in this respect. The
significant correlation between TfR and Ki-67 expression has been
reported in biopsies of diverse tumor types, including leukemia,
melanoma, breast carcinoma, brain tumors, head and neck cancer and
esophageal cancer (83-88). Although a significant association
was identified between TfR and Ki-67 in the entire panel of 54
tumor cell lines, this association was not seen in the majority of
the subsets with different tumor types. This may simply be
explained by the limited number of cell lines per tumor entity. The
previously published data on the significant correlation between
TfR and Ki-67 expression were obtained in larger collectives of
tumor biopsies (83,89).
In human tumor pathology, hematoxylin and eosin
staining is also useful for the detection of tumor-infiltrating
lymphocytes (TILs) (90-92). Therefore, in the present study we
also screened the hematoxylin and eosin-stained slides of the pet
tumors for the presence of TILs, but TILs could not be detected in
the tumor tissues. Since the tumors were generally large and
already progressed at diagnosis, it was assumed that the immune
system of the animals was weakened and TIL-mediated immune defenses
against the tumor were largely destroyed.
Furthermore, it is important to mention that the
present study did not observe considerable adverse side effects
among the 25 A. annua-treated dogs and cats. In comparison
to tumor treatment in human cancer patients with the semi-synthetic
drug artesunate, A. annua appeared to be even safer. Among
the 23 dogs with cancer treated with artesunate, fever and
transient hematological and gastrointestinal toxicity were observed
in 16 dogs and 1 dog died from pneumonia (39). In human cancer patients, the
compassionate use of A. annua in 1 patient with prostate
carcinoma was well tolerated in a previous study (41). Treatment with artesunate has been
reported to lead to occasional and transient side effects,
including hematological toxicity, gastrointestinal toxicity,
asthenia and thrombosis, in colon, cervix and breast carcinoma
(42-44). Rare cases of hepatotoxicity have
been reported with artesunate (93,94).
Whether phytotherapeutic approaches with A. annua are safer
than treatment with artesunate requires further investigation.
Notably, the majority of pet owners reported that
the animals appeared to feel better after A. annua
treatment; some were more active, while others were more relaxed.
Of course, these observations are subjective and non-quantifiable,
and it is unclear whether these reports may reflect the pet owners
psychological condition after successful treatment of their pets.
Nevertheless, this observation should not be neglected, as it
cannot be excluded that there may be an unexpected and
unintentional positive side effect of A. annua-treatment.
Indeed, there are some previous studies that support these
observations. The serotonin serum levels in the brains of
artesunate-treated rabbits has been reported to be significantly
higher compared with in untreated control animals (95). Zhu et al (96) reported significant remissions in
nociceptive, anxiety and depressive behaviors by
dihydroartemisinin, artesunate or artemether. Amos et al
(97) suggested sedative
properties of artemisinin mediated by artemisinin's effect on
postsynaptic dopamine D2 receptors in the brain. This novel aspect
of artemisinin's possible activity deserves further detailed
investigation in the future.
The present investigation also raises the more
general question about the comparability of data raised in
veterinary tumors to the clinical situation in human tumors and
thereby the suitability of veterinary tumors as models of human
cancer biology and treatment. The present study determined that the
role of TfR and Ki-67 was comparable between veterinary tumors and
human tumor cell lines. TfR and Ki-67 have also been associated
with response to artemisinin and artesunate in tumors of human
patients (41-43). Although these data in human tumors
are preliminary, they indicate that veterinary tumors may represent
a suitable model for clinical human tumors in the context of
artemisinin therapy.
Independent of this situation, there are more
arguments that support the suitability of veterinary tumors for the
investigation of human cancer biology. Veterinary tumors are
spontaneously developing, which may qualify them as better models
compared with other tumors in mice and rats. Frequently tumors are
maintained by transplanting syngeneic tumors to rodents or human
xenograft tumors to immunosup-pressed nude mice. Another
possibility is to induce tumor development in mice or rats by
chemical carcinogens. Although these tumor models are indispensable
and of high value in preclinical oncology, they are to some degree
artificial. Here, tumors in dogs and cats may be advantageous,
because their spontaneous development is closer to the situation of
human tumors. Veterinary tumors have not received as much attention
as the aforementioned rodent tumor models as of yet. Further
investigations are required to investigate the full potential of
veterinary tumors as attractive in vivo models to develop
strategies for human tumor treatment.
In conclusion, the current retrospective study
involving 20 dogs and cats treated with standard therapy plus A.
annua and 11 dogs treated with standard therapy alone clearly
demonstrated that additional food supplementation of A.
annua to veterinary cancer patients resulted in an improved
survival prognosis. The activity of A. annua may be
dependent on the iron content in the tumor, but not in the blood,
since TfR expression in the tumors was significantly correlated
with survival time and artemisinin cytotoxicity in a control panel
of human tumor cell lines. The same was true for Ki-67 expression.
Tumors with high Ki-67 expression, indicating high proliferative
activity, were more susceptible to artemisinin in the human cell
line panel. The data presented in the present study should provide
guidance for the activity of A. annua against veterinary
tumors. Prospective trials are required to deliver convincing
evidence for this hypothesis.
Funding
This study was supported by intramural funding from
Johannes Gutenberg University (Mainz, Germany).
Availability of data and materials
The datasets generated and/or analyzed during the
current study are not publicly available due to restrictions of the
availability of these data but are available from the corresponding
author on reasonable request.
Authors' contributions
TE and EB designed the study. EB treated the
animals, provided the material and collected the clinical data.
MEMS performed the immunostaining. MEFH performed NMR and HPLC-MS.
TE performed the statistical analysis, supervised the work and
provided the facilities for the study. TE and MEMS wrote the
manuscript. All authors read the manuscript and approved the final
version.
Ethics approval and consent to
participate
Written permission for this retrospective study was
obtained from the Regierungspräsidium (Government Presidium)
Freiburg, Germany (Az. 35-9185.81/1, dated from February 4th,
2019). Written informed consent for experimental work was obtained
from all pet owners.
Patient consent for publication
No applicable.
Competing interests
EB commercially trades Luparte®. No part
of the experimental work at the Department of Pharmacological
Biology (Johannes Gutenberg University, Mainz, Germany) was funded
by EB. All other authors declare that they have no competing
interests.
Acknowledgments
The authors wish to thank Mrs. Doris Rohr
(Department of Pharmaceutical Biology, Institute of Pharmacy and
Biochemistry, Johannes Gutenberg University, Mainz, Germany) for
technical assistance with immunohistochem-istry staining.
Abbreviations:
|
NMR
|
nuclear magnetic resonance
|
|
TfR
|
transferrin receptor
|
|
TIL
|
tumor-infiltrating lymphocyte
|
References
|
1
|
Talmadge JE, Singh RK, Fidler IJ and Raz
A: Murine models to evaluate novel and conventional therapeutic
strategies for cancer. Am J Pathol. 170:793–804. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jantscheff P, Beshay J, Lemarchand T,
Obodozie C, Schächtele C and Weber H: Mouse-derived isograft (MDI)
in vivo tumor models I Spontaneous sMDI models: Characterization
and cancer therapeutic approaches. Cancers (Basel). 11:112019.
View Article : Google Scholar
|
|
3
|
Choi JW, Yoon HY and Jeong SW: Clinical
outcomes of surgically managed spontaneous tumors in 114
client-owned dogs. Immune Netw. 16:116–125. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hellmén E, Bergström R, Holmberg L,
Spångberg IB, Hansson K and Lindgren A: Prognostic factors in
canine mammary tumors: A multivariate study of 202 consecutive
cases. Vet Pathol. 30:20–27. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Marconato L: The staging and treatment of
multicentric high-grade lymphoma in dogs: A review of recent
developments and future prospects. Vet J. 188:34–38. 2011.
View Article : Google Scholar
|
|
6
|
Tuohy JL, Selmic LE, Worley DR, Ehrhart NP
and Withrow SJ: Outcome following curative-intent surgery for oral
melanoma in dogs: 70 cases (1998-2011). J Am Vet Med Assoc.
245:1266–1273. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Miller RL, Van Lelyveld S, Warland J,
Dobson JM and Foale RD: A retrospective review of treatment and
response of high-risk mast cell tumours in dogs. Vet Comp Oncol.
14:361–370. 2016. View Article : Google Scholar
|
|
8
|
Romano FR, Heinze CR, Barber LG, Mason JB
and Freeman LM: Association between body condition score and cancer
prognosis in dogs with lymphoma and osteosarcoma. J Vet Intern Med.
30:1179–1186. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sarowitz BN, Davis GJ and Kim S: Outcome
and prognostic factors following curative-intent surgery for oral
tumours in dogs: 234 cases (2004 to 2014). J Small Anim Pract.
58:146–153. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ettinger SN: Principles of treatment for
feline lymphoma. Clin Tech Small Anim Pract. 18:98–102. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Morris J: Mammary tumours in the cat: Size
matters, so early intervention saves lives. J Feline Med Surg.
15:391–400. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zabielska-Koczywąs K, Wojtalewicz A and
Lechowski R: Current knowledge on feline injection-site sarcoma
treatment. Acta Vet Scand. 59:472017. View Article : Google Scholar :
|
|
13
|
Martano M, Iussich S, Morello E and
Buracco P: Canine oral fibrosarcoma: Changes in prognosis over the
last 30 years? Vet J. 241:1–7. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang D, Hedlund EM, Lim S, Chen F, Zhang
Y, Sun B and Cao Y: Antiangiogenic agents significantly improve
survival in tumor-bearing mice by increasing tolerance to
chemotherapy-induced toxicity. Proc Natl Acad Sci USA.
108:4117–4122. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tiwari A, Hadley JA, Hendricks GL III,
Elkin RG, Cooper T and Ramachandran R: Characterization of
ascites-derived ovarian tumor cells from spontaneously occurring
ovarian tumors of the chicken: Evidence for E-cadherin
upregulation. PLoS One. 8:e575822013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Newman DJ and Cragg GM: Natural products
as sources of new drugs from 1981 to 2014. J Nat Prod. 79:629–661.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tu Y: The discovery of artemisinin
(qinghaosu) and gifts from Chinese medicine. Nat Med. 17:1217–1220.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bridgford JL, Xie SC, Cobbold SA, Pasaje
CFA, Herrmann S, Yang T, Gillett DL, Dick LR, Ralph SA, Dogovski C,
et al: Artemisinin kills malaria parasites by damaging proteins and
inhibiting the proteasome. Nat Commun. 9:38012018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Su XZ and Miller LH: The discovery of
artemisinin and the Nobel Prize in Physiology or Medicine. Sci
China Life Sci. 58:1175–1179. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tu Y: Artemisinin-a gift from traditional
chinese medicine to the world (Nobel Lecture). Angew Chem Int Ed
Engl. 55:10210–10226. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Daddy NB, Kalisya LM, Bagire PG, Watt RL,
Towler MJ and Weathers PJ: Artemisia annua dried leaf tablets
treated malaria resistant to ACT and i.v. artesunate: Case reports.
Phytomedicine. 32:37–40. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Saeed ME, Krishna S, Greten HJ, Kremsner
PG and Efferth T: Antischistosomal activity of artemisinin
derivatives in vivo and in patients. Pharmacol Res. 110:216–226.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Pérez del Villar L, Burguillo FJ,
López-Abán J and Muro A: Systematic review and meta-analysis of
artemisinin based therapies for the treatment and prevention of
schistosomiasis. PLoS One. 7:e458672012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Naß J and Efferth T: The activity of
Artemisia spp. and their constituents against Trypanosomiasis.
Phytomedicine. 47:184–191. 2018. View Article : Google Scholar
|
|
25
|
Efferth T: Beyond malaria: The inhibition
of viruses by arte-misinin-type compounds. Biotechnol Adv.
36:1730–1737. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Jiang W, Cen Y, Song Y, Li P, Qin R, Liu
C, Zhao Y, Zheng J and Zhou H: Artesunate attenuated progression of
atherosclerosis lesion formation alone or combined with
rosuvastatin through inhibition of pro-inflammatory cytokines and
pro-inflammatory chemokines. Phytomedicine. 23:1259–1266. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li J, Casteels T, Frogne T, Ingvorsen C,
Honore C, Courtney M, Huber KV, Schmitner N, Kimmel RA, Romanov RA,
et al: Artemisinins target GABAA receptor signaling and impair
alpha cell Identity. Cell. 168:86–100.e15. 2017. View Article : Google Scholar
|
|
28
|
Guo Y, Fu W, Xin Y, Bai J, Peng H, Fu L,
Liu J, Li L, Ma Y and Jiang H: Antidiabetic and antiobesity effects
of artemether in db/db mice. BioMed Res Int. 2018:86395232018.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Efferth T, Dunstan H, Sauerbrey A, Miyachi
H and Chitambar CR: The anti-malarial artesunate is also active
against cancer. Int J Oncol. 18:767–773. 2001.PubMed/NCBI
|
|
30
|
Efferth T, Sauerbrey A, Olbrich A, Gebhart
E, Rauch P, Weber HO, Hengstler JG, Halatsch ME, Volm M, Tew KD, et
al: Molecular modes of action of artesunate in tumor cell lines.
Mol Pharmacol. 64:382–394. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Dell'Eva R, Pfeffer U, Vené R, Anfosso L,
Forlani A, Albini A and Efferth T: Inhibition of angiogenesis in
vivo and growth of Kaposi's sarcoma xenograft tumors by the
anti-malarial arte-sunate. Biochem Pharmacol. 68:2359–2366. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Efferth T: From ancient herb to modern
drug: Artemisia annua and artemisinin for cancer therapy. Semin
Cancer Biol. 46:65–83. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Abba ML, Patil N, Leupold JH, Saeed ME,
Efferth T and Allgayer H: Prevention of carcinogenesis and
metastasis by Artemisinin-type drugs. Cancer Lett. 429:11–18. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Krusche B, Arend J and Efferth T:
Synergistic inhibition of angiogenesis by artesunate and captopril
in vitro and in vivo. Evid Based Complement Alternat Med.
2013:4547832013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Efferth T: Cancer combination therapy of
the sesquiterpenoid artesunate and the selective EGFR-tyrosine
kinase inhibitor erlotinib. Phytomedicine. 37:58–61. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Efferth T: Cancer combination therapies
with artemisinin-type drugs. Biochem Pharmacol. 139:56–70. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hosoya K, Murahari S, Laio A, London CA,
Couto CG and Kisseberth WC: Biological activity of
dihydroartemisinin in canine osteosarcoma cell lines. Am J Vet Res.
69:519–526. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hosoya K, Couto CG, London CA, Kisseberth
WC, Phelps MA and Dalton JT: Comparison of high-dose intermittent
and low-dose continuous oral artemisinin in dogs with naturally
occurring tumors. J Am Anim Hosp Assoc. 50:390–395. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Rutteman GR, Erich SA, Mol JA, Spee B,
Grinwis GC, Fleckenstein L, London CA and Efferth T: Safety and
efficacy field study of artesunate for dogs with non-resectable
tumours. Anticancer Res. 33:1819–1827. 2013.PubMed/NCBI
|
|
40
|
Berger TG, Dieckmann D, Efferth T, Schultz
ES, Funk JO, Baur A and Schuler G: Artesunate in the treatment of
metastatic uveal melanoma - first experiences. Oncol Rep.
14:1599–1603. 2005.PubMed/NCBI
|
|
41
|
Michaelsen FW, Saeed ME, Schwarzkopf J and
Efferth T: Activity of Artemisia annua and artemisinin derivatives,
in prostate carcinoma. Phytomedicine. 22:1223–1231. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Jansen FH, Adoubi I, JC KC, DE Cnodder T,
Jansen N, Tschulakow A and Efferth T: First study of oral
Artenimol-R in advanced cervical cancer: Clinical benefit,
tolerability and tumor markers. Anticancer Res. 31:4417–4422.
2011.PubMed/NCBI
|
|
43
|
Krishna S, Ganapathi S, Ster IC, Saeed ME,
Cowan M, Finlayson C, Kovacsevics H, Jansen H, Kremsner PG, Efferth
T, et al: A randomised, double blind, placebo-controlled pilot
study of oral artesunate therapy for colorectal cancer.
EBioMedicine. 2:82–90. 2014. View Article : Google Scholar
|
|
44
|
von Hagens C, Walter-Sack I, Goeckenjan M,
Osburg J, Storch-Hagenlocher B, Sertel S, Elsässer M, Remppis BA,
Edler L, Munzinger J, et al: Prospective open uncontrolled phase I
study to define a well-tolerated dose of oral artesunate as add-on
therapy in patients with metastatic breast cancer (ARTIC M33/2).
Breast Cancer Res Treat. 164:359–369. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Breuer E and Efferth T: Treatment of
iron-loaded veterinary sarcoma by Artemisia annua. Nat Prod
Bioprospect. 4:113–118. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Hegazy MF, Abdelfatah S, Hamed AR, Mohamed
TA, Elshamy AA, Saleh IA, Reda EH, Abdel-Azim NS, Shams KA, Sakr M,
et al: Cytotoxicity of 40 Egyptian plant extracts targeting
mechanisms of drug-resistant cancer cells. Phytomedicine.
59:1527712019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Chikazawa S, Hori Y, Kanai K, Ito N, Hoshi
F, Orino K, Watanabe K and Higuchi S: Factors influencing
measurement of serum iron concentration in dogs: Diurnal variation
and hyper-ferritinemia. J Vet Med Sci. 75:1615–1618. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Carpenter CE and Ward RE: Iron
determination by Ferrozine method. Food Analysis Laboratory Manual
Springer: Luxembourg: pp. 157–159. 2017, View Article : Google Scholar
|
|
49
|
Stoica G, Lungu G, Martini-Stoica H,
Waghela S, Levine J and Smith R III: Identification of cancer stem
cells in dog glioblastoma. Vet Pathol. 46:391–406. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Wellman ML, Krakowka S, Jacobs RM and
Kociba GJ: A macrophage-monocyte cell line from a dog with
malignant histiocytosis. In Vitro Cell Dev Biol. 24:223–229. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Alley MC, Scudiero DA, Monks A, Hursey ML,
Czerwinski MJ, Fine DL, Abbott BJ, Mayo JG, Shoemaker RH and Boyd
MR: Feasibility of drug screening with panels of human tumor cell
lines using a microculture tetrazolium assay. Cancer Res.
48:589–601. 1988.PubMed/NCBI
|
|
52
|
Rubinstein LV, Shoemaker RH, Paull KD,
Simon RM, Tosini S, Skehan P, Scudiero DA, Monks A and Boyd MR:
Comparison of in vitro anticancer-drug-screening data generated
with a tetra-zolium assay versus a protein assay against a diverse
panel of human tumor cell lines. J Natl Cancer Inst. 82:1113–1118.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Kuete V, Mbaveng AT, Sandjo LP, Zeino M
and Efferth T: Cytotoxicity and mode of action of a naturally
occurring naphthoquinone,
2-acetyl-7-methoxynaphtho[2,3-b]furan-4,9-quinone towards
multi-factorial drug-resistant cancer cells. Phytomedicine.
33:62–68. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
O'Brien J, Wilson I, Orton T and Pognan F:
Investigation of the Alamar Blue (resazurin) fluorescent dye for
the assessment of mammalian cell cytotoxicity. Eur J Biochem.
267:5421–5426. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Saeed ME, Mertens R, Handgretinger R and
Efferth T: Identification of fatal outcome in a childhood
nasopharyngeal carcinoma patient by protein expression profiling.
Int J Oncol. 53:1721–1731. 2018.PubMed/NCBI
|
|
56
|
Blaskó G, Cordell GA and Lankin DC:
Definitive 1H-and 13C-NMR assignments of
artemisinin (Qinghaosu). J Nat Prod. 51:1273–1276. 1988. View Article : Google Scholar
|
|
57
|
Haynes RK, Cheu KW, N'Da D, Coghi P and
Monti D: Considerations on the mechanism of action of artemisinin
anti-malarials: Part 1 - the 'carbon radical' and 'heme'
hypotheses. Infect Disord Drug Targets. 13:217–277. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Klonis N, Creek DJ and Tilley L: Iron and
heme metabolism in Plasmodium falciparum and the mechanism of
action of arte-misinins. Curr Opin Microbiol. 16:722–727. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Efferth T, Benakis A, Romero MR, Tomicic
M, Rauh R, Steinbach D, Häfer R, Stamminger T, Oesch F, Kaina B, et
al: Enhancement of cytotoxicity of artemisinins toward cancer cells
by ferrous iron. Free Radic Biol Med. 37:998–1009. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Kelter G, Steinbach D, Konkimalla VB,
Tahara T, Taketani S, Fiebig HH and Efferth T: Role of transferrin
receptor and the ABC transporters ABCB6 and ABCB7 for resistance
and differentiation of tumor cells towards artesunate. PLoS One.
2:e7982007. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Aulbert E, Disselhoff W, Sörje H, Schulz E
and Gericke D: Lysosomal accumulation of 67Ga - transferrin in
malignant tumors in relation to their growth rate. Eur J Cancer.
16:1217–1232. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Sadava D, Phillips T, Lin C and Kane SE:
Transferrin overcomes drug resistance to artemisinin in human
small-cell lung carcinoma cells. Cancer Lett. 179:151–156. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Zhao F, Wang H, Kunda P, Chen X, Liu QL
and Liu T: Artesunate exerts specific cytotoxicity in
retinoblastoma cells via CD71. Oncol Rep. 30:1473–1482. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Wang Q, Wu S, Zhao X, Zhao C, Zhao H and
Huo L: Mechanisms of dihydroartemisinin and
dihydroartemisinin/holotransferrin cytotoxicity in t-cell lymphoma
cells. PLoS One. 10:e01373312015. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Judd W, Poodry CA and Strominger JL: Novel
surface antigen expressed on dividing cells but absent from
nondividing cells. J Exp Med. 152:1430–1435. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Sutherland R, Delia D, Schneider C, Newman
R, Kemshead J and Greaves M: Ubiquitous cell-surface glycoprotein
on tumor cells is proliferation-associated receptor for
transferrin. Proc Natl Acad Sci USA. 78:4515–4519. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Trowbridge IS and Omary MB: Human cell
surface glycoprotein related to cell proliferation is the receptor
for transferrin. Proc Natl Acad Sci USA. 78:3039–3043. 1981.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Gatter KC, Brown G, Trowbridge IS,
Woolston RE and Mason DY: Transferrin receptors in human tissues:
Their distribution and possible clinical relevance. J Clin Pathol.
36:539–545. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Tortorella S and Karagiannis TC:
Transferrin receptor-mediated endocytosis: A useful target for
cancer therapy. J Membr Biol. 247:291–307. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Luria-Pérez R, Helguera G and Rodríguez
JA: Antibody-mediated targeting of the transferrin receptor in
cancer cells. Bol Méd Hosp Infant México. 73:372–379. 2016.
|
|
71
|
Akbari B, Farajnia S, Ahdi Khosroshahi S,
Safari F, Yousefi M, Dariushnejad H and Rahbarnia L: Immunotoxins
in cancer therapy: Review and update. Int Rev Immunol. 36:207–219.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Lai H, Sasaki T, Singh NP and Messay A:
Effects of arte-misinin-tagged holotransferrin on cancer cells.
Life Sci. 76:1267–1279. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Nakase I, Gallis B, Takatani-Nakase T, Oh
S, Lacoste E, Singh NP, Goodlett DR, Tanaka S, Futaki S, Lai H, et
al: Transferrin receptor-dependent cytotoxicity of
artemisinin-transferrin conjugates on prostate cancer cells and
induction of apoptosis. Cancer Lett. 274:290–298. 2009. View Article : Google Scholar
|
|
74
|
Gong Y, Gallis BM, Goodlett DR, Yang Y, Lu
H, Lacoste E, Lai H and Sasaki T: Effects of transferrin conjugates
of artemisinin and artemisinin dimer on breast cancer cell lines.
Anticancer Res. 33:123–132. 2013.
|
|
75
|
Zhong YR: Chemical constituents of
volatile oils of Artemisia annua. Zhong Yao Tong Bao. 8:31–32.
1983.In Chinese. PubMed/NCBI
|
|
76
|
Liao HW, Wang DY and Li XM: Studies on the
chemical constituents of essential oil of Hunan Artemisia annua.
Zhong Yao Cai. 29:562–564. 2006.In Chinese. PubMed/NCBI
|
|
77
|
Efferth T, Herrmann F, Tahrani A and Wink
M: Cytotoxic activity of secondary metabolites derived from
Artemisia annua L. towards cancer cells in comparison to its
designated active constituent artemisinin. Phytomedicine.
18:959–969. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Zhang X, Zhao Y, Guo L, Qiu Z, Huang L and
Qu X: Differences in chemical constituents of Artemisia annua L.
from different geographical regions in China. PLoS One.
12:e01830472017. View Article : Google Scholar
|
|
79
|
Efferth T, Konkimalla VB, Wang YF,
Sauerbrey A, Meinhardt S, Zintl F, Mattern J and Volm M: Prediction
of broad spectrum resistance of tumors towards anticancer drugs.
Clin Cancer Res. 14:2405–2412. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Volm M and Efferth T: Prediction of cancer
drug resistance and implications for personalized medicine. Front
Oncol. 5:2822015. View Article : Google Scholar
|
|
81
|
Duffy MJ, Harbeck N, Nap M, Molina R,
Nicolini A, Senkus E and Cardoso F: Clinical use of biomarkers in
breast cancer: Updated guidelines from the European Group on Tumor
Markers (EGTM). Eur J Cancer. 75:284–298. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Sun X and Kaufman PD: Ki-67: More than a
proliferation marker. Chromosoma. 127:175–186. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Scott CS, Ramsden W, Limbert HJ, Master PS
and Roberts BE: Membrane transferrin receptor (TfR) and nuclear
proliferation-associated Ki-67 expression in hemopoietic
malignancies. Leukemia. 2:438–442. 1988.PubMed/NCBI
|
|
84
|
Soyer HP, Smolle J, Smolle-Juettner FM and
Kerl H: Proliferation antigens in cutaneous melanocytic tumors - an
immunohisto-chemical study comparing the transferrin receptor and
the Ki-67 antigen. Dermatologica. 179:3–9. 1989. View Article : Google Scholar
|
|
85
|
Wrba F, Chott A, Reiner A, Reiner G,
Markis-Ritzinger E and Holzner JH: Ki-67 immunoreactivity in breast
carcinomas in relation to transferrin receptor expression, estrogen
receptor status and morphological criteria. An immunohistochemical
study. Oncology. 46:255–259. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Prior R, Reifenberger G and Wechsler W:
Transferrin receptor expression in tumours of the human nervous
system: Relation to tumour type, grading and tumour growth
fraction. Virchows Arch A Pathol Anat Histopathol. 416:491–496.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Kearsley JH, Furlong KL, Cooke RA and
Waters MJ: An immuno-histochemical assessment of cellular
proliferation markers in head and neck squamous cell cancers. Br J
Cancer. 61:821–827. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Chan KT, Choi MY, Lai KK, Tan W, Tung LN,
Lam HY, Tong DK, Lee NP and Law S: Overexpression of transferrin
receptor CD71 and its tumorigenic properties in esophageal squamous
cell carcinoma. Oncol Rep. 31:1296–1304. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Motamedi M, Xu L and Elahi S: Correlation
of transferrin receptor (CD71) with Ki67 expression on stimulated
human and mouse T cells: The kinetics of expression of T cell
activation markers. J Immunol Methods. 437:43–52. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Darb-Esfahani S, Kolaschinski I, Trillsch
F, Mahner S, Concin N, Vergote I, Van Nieuwenhuysen E,
Achimas-Cadariu P, Glajzer J, Woopen H, et al: Morphology and
tumour-infiltrating lymphocytes in high-stage, high-grade serous
ovarian carcinoma correlated with long-term survival.
Histopathology. 73:1002–1012. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Lee JS, Won HS, Sun S, Hong JH and Ko YH:
Prognostic role of tumor-infiltrating lymphocytes in gastric
cancer: A systematic review and meta-analysis. Medicine
(Baltimore). 97:e117692018. View Article : Google Scholar
|
|
92
|
Shen M, Wang J and Ren X: New insights
into tumor-infiltrating B lymphocytes in breast cancer: Clinical
impacts and regulatory mechanisms. Front Immunol. 9:4702018.
View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Uhl M, Schwab S and Efferth T: Fatal liver
and bone marrow toxicity by combination treatment of
dichloroacetate and arte-sunate in a glioblastoma multiforme
patient: Case report and review of the literature. Front Oncol.
6:2042016. View Article : Google Scholar
|
|
94
|
Efferth T, Schöttler U, Krishna S,
Schmiedek P, Wenz F and Giordano FA: Answer to the comment of Hai
Lu et al. regarding 'Hepatotoxicity by combination treatment of
temozolomide, artesunate and Chinese herbs in a glioblastoma
multiforme patient: Case report and review of the literature. Arch
Toxicol (2016). Arch Toxicol. 91:2491–2492. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Eigbibhalu UG, Albert Taiwo EO, Douglass
IA and Abimbola EA: Effect of selected anti-malarial drugs on the
blood chemistry and brain serotonin levels in male rabbits. Pak J
Pharm Sci. 26:125–129. 2013.
|
|
96
|
Zhu CY, Xu QH, Mao ZY and Lin N:
Application of three arte-misinin derivatives in neuropathic pain:
Evaluating co-curation of nociceptive and emotional syndromes in
spinal cord ligation mice. Zhongguo Zhong Yao Za Zhi. 43:3058–3063.
2018.In Chinese. PubMed/NCBI
|
|
97
|
Amos S, Chindo BA, Abbah J, Vongtau HO,
Edmond I, Binda L, Akah PA, Wambebe C and Gamaniel KS: Postsynaptic
dopamine (D(2))-mediated behavioural effects of high acute doses of
arte-misinin in rodents. Brain Res Bull. 62:255–260. 2003.
View Article : Google Scholar : PubMed/NCBI
|