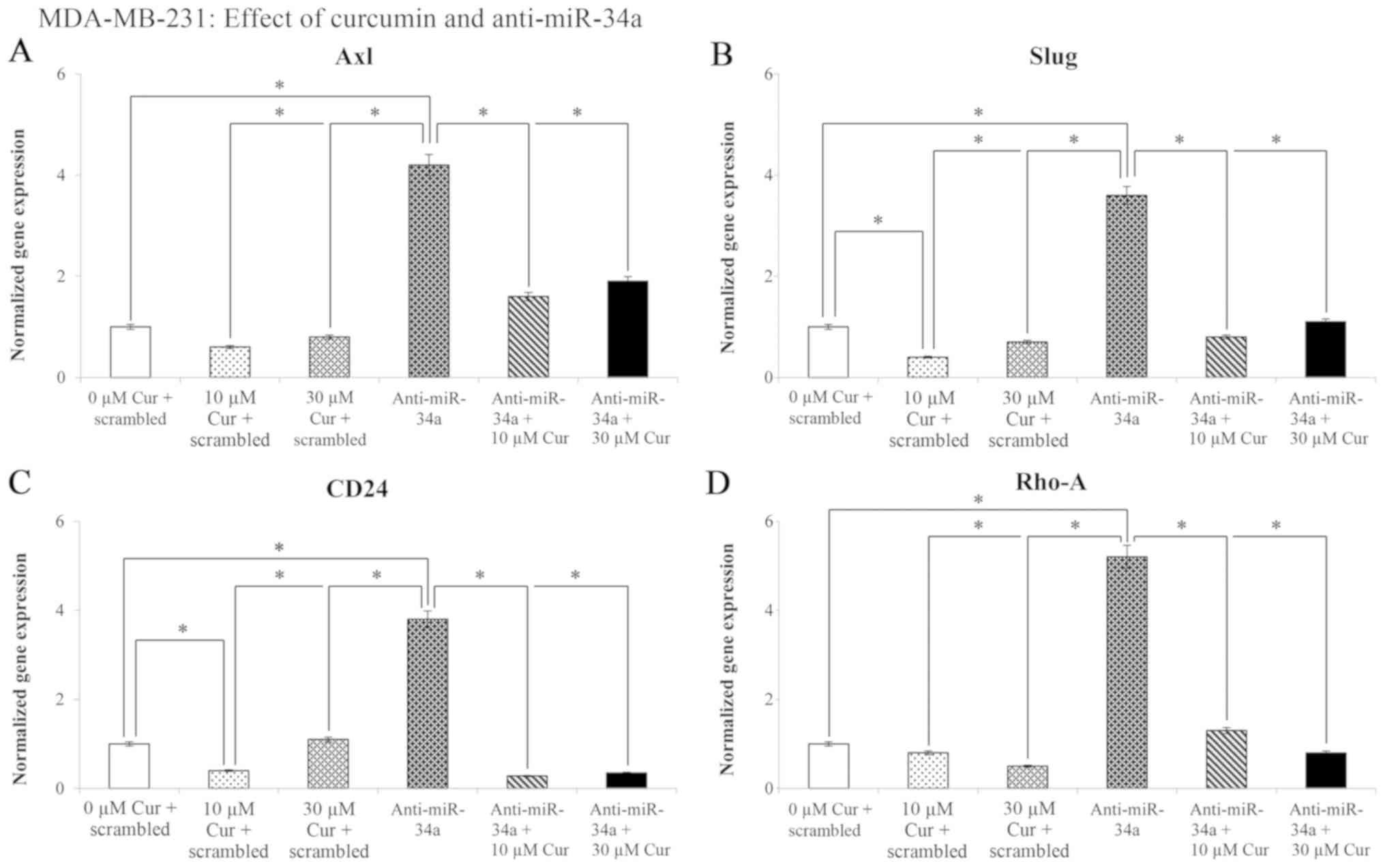
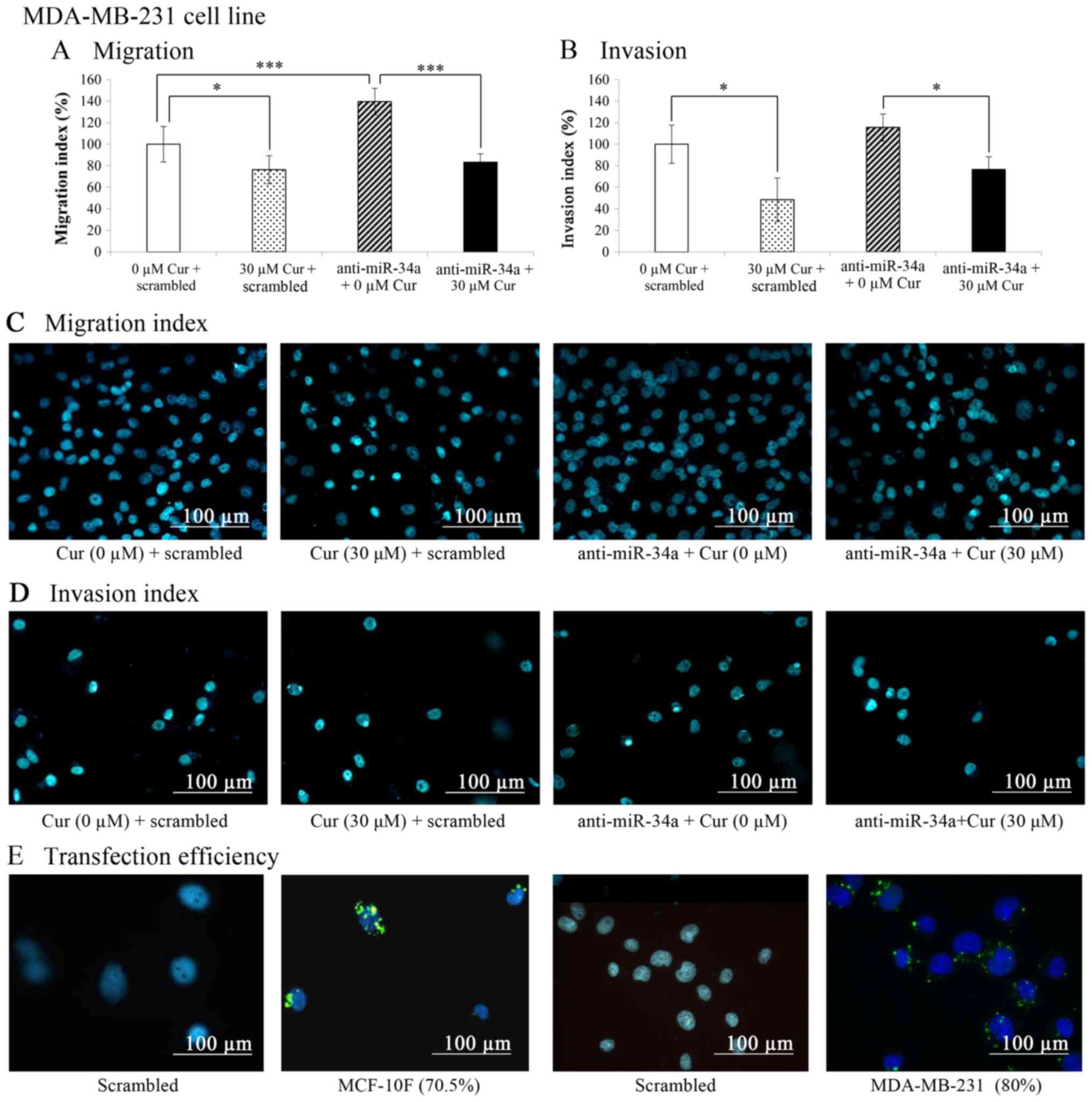
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar
|
|
2
|
Sethi N and Kang Y: Unravelling the
complexity of metastasis - molecular understanding and targeted
therapies. Nat Rev Cancer. 11:735–748. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chaffer CL and Weinberg RA: A perspective
on cancer cell metastasis. Science. 331:1559–1564. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Howlader N, Noone AM, Krapcho M, Miller D,
Bishop K, Kosary CL, Yu M, Ruhl J, Tatalovich Z, Mariotto A, et al:
SEER Cancer Statistics Review, 1975-2014. National Cancer
Institute; Bethesda, MD: 2016, https://seer.cancer.gov/csr/1975_2014/.
Accessed April 2017.
|
|
5
|
Kalluri R and Neilson EG:
Epithelial-mesenchymal transition and its implications for
fibrosis. J Clin Invest. 112:1776–1784. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Graham DK, DeRyckere D, Davies KD and Earp
HS: The TAM family: Phosphatidylserine sensing receptor tyrosine
kinases gone awry in cancer. Nat Rev Cancer. 14:769–785. 2014.
View Article : Google Scholar
|
|
7
|
Kirane A, Ludwig KF, Sorrelle N, Haaland
G, Sandal T, Ranaweera R, Toombs JE, Wang M, Dineen SP, Micklem D,
et al: Warfarin Blocks Gas6-Mediated Axl Activation Required for
Pancreatic Cancer Epithelial Plasticity and Metastasis. Cancer Res.
75:3699–3705. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Linger RM, Keating AK, Earp HS and Graham
DK: TAM receptor tyrosine kinases: Biologic functions, signaling,
and potential therapeutic targeting in human cancer. Adv Cancer
Res. 100:35–83. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Dunne PD, McArt DG, Blayney JK, Kalimutho
M, Greer S, Wang T, Srivastava S, Ong CW, Arthur K, Loughrey M, et
al: AXL is a key regulator of inherent and chemotherapy-induced
invasion and predicts a poor clinical outcome in early-stage colon
cancer. Clin Cancer Res. 20:164–175. 2014. View Article : Google Scholar
|
|
10
|
Hajra KM, Chen DY and Fearon ER: The SLUG
zinc-finger protein represses E-cadherin in breast cancer. Cancer
Res. 62:1613–1618. 2002.PubMed/NCBI
|
|
11
|
Castro Alves C, Rosivatz E, Schott C,
Hollweck R, Becker I, Sarbia M, Carneiro F and Becker KF: Slug is
overexpressed in gastric carcinomas and may act synergistically
with SIP1 and Snail in the down-regulation of E-cadherin. J Pathol.
211:507–515. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ye Y, Xiao Y, Wang W, Yearsley K, Gao JX,
Shetuni B and Barsky SH: ERalpha signaling through slug regulates
E-cadherin and EMT. Oncogene. 29:1451–1462. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Atmaca A, Wirtz RW, Werner D, Steinmetz K,
Claas S, Brueckl WM, Jäger E and Al-Batran SE: SNAI2/SLUG and
estrogen receptor mRNA expression are inversely correlated and
prognostic of patient outcome in metastatic non-small cell lung
cancer. BMC Cancer. 15:3002015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Pirruccello SJ and LeBien TW: The human B
cell-associated antigen CD24 is a single chain sialoglycoprotein. J
Immunol. 136:3779–3784. 1986.PubMed/NCBI
|
|
15
|
Daniel L, Lechevallier E, Bouvier C,
Coulange C and Pellissier JF: Adult mesoblastic nephroma. Pathol
Res Pract. 196:135–139. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kristiansen G, Denkert C, Schlüns K, Dahl
E, Pilarsky C and Hauptmann S: CD24 is expressed in ovarian cancer
and is a new independent prognostic marker of patient survival. Am
J Pathol. 161:1215–1221. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jacob J, Bellach J, Grützmann R, Alldinger
I, Pilarsky C, Dietel M and Kristiansen G: Expression of CD24 in
adenocar-cinomas of the pancreas correlates with higher tumor
grades. Pancreatology. 4:454–460. 2004. View Article : Google Scholar
|
|
18
|
Kristiansen G, Winzer KJ, Mayordomo E,
Bellach J, Schlüns K, Denkert C, Dahl E, Pilarsky C, Altevogt P,
Guski H, et al: CD24 expression is a new prognostic marker in
breast cancer. Clin Cancer Res. 9:4906–4913. 2003.PubMed/NCBI
|
|
19
|
Yu LLG and Gu JY: Advances in the role of
Rho sub-family in tumor invasion. Fudan Univ J Med Sci. 37:617–619.
2010.
|
|
20
|
Hall A: Rho GTPases and the actin
cytoskeleton. Science. 279:509–514. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
DerMardirossian C and Bokoch GM: GDIs:
Central regulatory molecules in Rho GTPase activation. Trends Cell
Biol. 15:356–363. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Olofsson B: Rho guanine dissociation
inhibitors: Pivotal molecules in cellular signalling. Cell Signal.
11:545–554. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Fritz G, Brachetti C, Bahlmann F, Schmidt
M and Kaina B: Rho GTPases in human breast tumours: Expression and
mutation analyses and correlation with clinical parameters. Br J
Cancer. 87:635–644. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Burbelo P, Wellstein A and Pestell RG:
Altered Rho GTPase signaling pathways in breast cancer cells.
Breast Cancer Res Treat. 84:43–48. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zaravinos A: The Regulatory Role of
MicroRNAs in EMT and Cancer. J Oncol. 2015:8658162015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ng EK, Wong CL, Ma ES and Kwong A:
MicroRNAs as New Players for Diagnosis, Prognosis, and Therapeutic
Targets in Breast Cancer. J Oncol. 2009:3054202009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Pichler M and Calin GA: MicroRNAs in
cancer: From developmental genes in worms to their clinical
application in patients. Br J Cancer. 113:569–573. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Croce CM: Causes and consequences of
microRNA dysregulation in cancer. Nat Rev Genet. 10:704–714. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hermeking H: The miR-34 family in cancer
and apoptosis. Cell Death Differ. 17:193–199. 2010. View Article : Google Scholar
|
|
30
|
Li Y, Kong D, Wang Z and Sarkar FH:
Regulation of microRNAs by natural agents: An emerging field in
chemoprevention and chemotherapy research. Pharm Res. 27:1027–1041.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sethi S, Li Y and Sarkar FH: Regulating
miRNA by natural agents as a new strategy for cancer treatment.
Curr Drug Targets. 14:1167–1174. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mirzaei H, Shakeri A, Rashidi B, Jalili A,
Banikazemi Z and Sahebkar A: Phytosomal curcumin: A review of
pharmacokinetic, experimental and clinical studies. Biomed
Pharmacother. 85:102–112. 2017. View Article : Google Scholar
|
|
33
|
Kuttan R, Bhanumathy P, Nirmala K and
George MC: Potential anticancer activity of turmeric (Curcuma
longa). Cancer Lett. 29:197–202. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Calaf GM, Echiburú-Chau C, Roy D, Chai Y,
Wen G and Balajee AS: Protective role of curcumin in oxidative
stress of breast cells. Oncol Rep. 26:1029–1035. 2011.PubMed/NCBI
|
|
35
|
Calaf GM: Curcumin, oxidative stress and
breast cancer. Oxidative stress and dietary antioxidants. Preedy
VR: Elsevier Inc; London: pp. 159–169. 2014
|
|
36
|
Gallardo M and Calaf GM: Curcumin and
epithelial-mesenchymal transition in breast cancer cells
transformed by low doses of radiation and estrogen. Int J Oncol.
48:2534–2542. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Echiburú-Chau C, Roy D and Calaf GM:
Metastatic suppressor CD44 is related with oxidative stress in
breast cancer cell lines. Int J Oncol. 39:1481–1489.
2011.PubMed/NCBI
|
|
38
|
Gallardo M and Calaf GM: Curcumin inhibits
invasive capabilities through epithelial mesenchymal transition in
breast cancer cell lines. Int J Oncol. 49:1019–1027. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Calaf GM and Abarca-Quinones J: Ras
protein expression as a marker for breast cancer. Oncol Lett.
11:3637–3642. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
41
|
Kunnumakkara AB, Anand P and Aggarwal BB:
Curcumin inhibits proliferation, invasion, angiogenesis and
metastasis of different cancers through interaction with multiple
cell signaling proteins. Cancer Lett. 269:199–225. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Alves CC, Carneiro F, Hoefler H and Becker
KF: Role of the epithelial-mesenchymal transition regulator Slug in
primary human cancers. Front Biosci. 14:3035–3050. 2009. View Article : Google Scholar
|
|
43
|
Lewis BP, Burge CB and Bartel DP:
Conserved seed pairing, often flanked by adenosines, indicates that
thousands of human genes are microRNA targets. Cell. 120:15–20.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zhao Y, Sun X, Jiang L, Yang F, Zhang Z
and Jia L: Differential expression of Axl and correlation with
invasion and multidrug resistance in cancer cells. Cancer Invest.
30:287–294. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Vuoriluoto K, Haugen H, Kiviluoto S,
Mpindi JP, Nevo J, Gjerdrum C, Tiron C, Lorens JB and Ivaska J:
Vimentin regulates EMT induction by Slug and oncogenic H-Ras and
migration by governing Axl expression in breast cancer. Oncogene.
30:1436–1448. 2011. View Article : Google Scholar
|
|
46
|
Calaf GM and Hei TK: Establishment of a
radiation- and estrogen-induced breast cancer model.
Carcinogenesis. 21:769–776. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Raver-Shapira N, Marciano E, Meiri E,
Spector Y, Rosenfeld N, Moskovits N, Bentwich Z and Oren M:
Transcriptional activation of miR-34a contributes to p53-mediated
apoptosis. Mol Cell. 26:731–743. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Chang TC, Wentzel EA, Kent OA,
Ramachandran K, Mullendore M, Lee KH, Feldmann G, Yamakuchi M,
Ferlito M, Lowenstein CJ, et al: Transactivation of miR-34a by p53
broadly influences gene expression and promotes apoptosis. Mol
Cell. 26:745–752. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Agostini M and Knight RA: miR-34: From
bench to bedside. Oncotarget. 5:872–881. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Beg MS, Brenner AJ, Sachdev J, Borad M,
Kang YK, Stoudemire J, Smith S, Bader AG, Kim S and Hong DS: Phase
I study of MRX34, a liposomal miR-34a mimic, administered twice
weekly in patients with advanced solid tumors. Invest New Drugs.
35:180–188. 2017. View Article : Google Scholar
|
|
51
|
Tarasov V, Jung P, Verdoodt B, Lodygin D,
Epanchintsev A, Menssen A, Meister G and Hermeking H: Differential
regulation of microRNAs by p53 revealed by massively parallel
sequencing: miR-34a is a p53 target that induces apoptosis and
G1-arrest. Cell Cycle. 6:1586–1593. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Siemens H, Jackstadt R, Hünten S, Kaller
M, Menssen A, Götz U and Hermeking H: miR-34 and SNAIL form a
double-negative feedback loop to regulate epithelial-mesenchymal
transitions. Cell Cycle. 10:4256–4271. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Mudduluru G, Ceppi P, Kumarswamy R,
Scagliotti GV, Papotti M and Allgayer H: Regulation of Axl receptor
tyrosine kinase expression by miR-34a and miR-199a/b in solid
cancer. Oncogene. 30:2888–2899. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Komina A, Palkina N, Aksenenko M,
Tsyrenzhapova S and Ruksha T: Antiproliferative and Pro-Apoptotic
Effects of MiR-4286 Inhibition in Melanoma Cells. PLoS One.
11:e01682292016. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wang X, Zhang Y, Fu Y, Zhang J, Yin L, Pu
Y and Liang G: MicroRNA-125b may function as an oncogene in lung
cancer cells. Mol Med Rep. 11:3880–3887. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Mizuno R, Chatterji P, Andres S, Hamilton
K, Simon L, Foley SW, Jeganathan A, Gregory BD, Madison B and
Rustgi AK: Differential Regulation of LET-7 by LIN28B
Isoform-Specific Functions. Mol Cancer Res. 16:403–416. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Lemecha M, Morino K, Imamura T, Iwasaki H,
Ohashi N, Ida S, Sato D, Sekine O, Ugi S and Maegawa H: MiR-494-3p
regulates mitochondrial biogenesis and thermogenesis through PGC1-α
signalling in beige adipocytes. Sci Rep. 8:150962018. View Article : Google Scholar
|
|
58
|
Inostroza-Riquelme M, Vivanco A, Lara P,
Guerrero S, Salas-Huenuleo E, Chamorro A, Leyton L, Bolaños K,
Araya E, Quest AFG, et al: Encapsulation of Gold Nanostructures and
Oil-in-Water Nanocarriers in Microgels with Biomedical Potential.
Molecules. 23:E12082018. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Bessone F, Argenziano M, Grillo G, Ferrara
B, Pizzimenti S, Barrera G, Cravotto G, Guiot C, Stura I, Cavalli
R, et al: Low-dose curcuminoid-loaded in dextran nanobubbles can
prevent metastatic spreading in prostate cancer cells.
Nanotechnology. 30:2140042019. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Guerrero S, Inostroza-Riquelme M,
Contreras-Orellana P, Diaz-Garcia V, Lara P, Vivanco-Palma A,
Cárdenas A, Miranda V, Robert P, Leyton L, et al: Curcumin-loaded
nano-emulsion: A new safe and effective formulation to prevent
tumor reincidence and metastasis. Nanoscale. 10:22612–22622. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Gera M, Sharma N, Ghosh M, Huynh DL, Lee
SJ, Min T, Kwon T and Jeong DK: Nanoformulations of curcumin: An
emerging paradigm for improved remedial application. Oncotarget.
8:66680–66698. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Yin H, Xiong G, Guo S, Xu C, Xu R, Guo P
and Shu D: Delivery of Anti-miRNA for Triple-Negative Breast Cancer
Therapy Using RNA Nanoparticles Targeting Stem Cell Marker CD133.
Mol Ther. 27:1252–1261. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Momtazi AA, Shahabipour F, Khatibi S,
Johnston TP, Pirro M and Sahebkar A: Curcumin as a MicroRNA
Regulator in Cancer: A Review. Rev Physiol Biochem Pharmacol.
171:1–38. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Rabieian R, Boshtam M, Zareei M, Kouhpayeh
S, Masoudifar A and Mirzaei H: Plasminogen Activator Inhibitor
Type-1 as a Regulator of Fibrosis. J Cell Biochem. 119:17–27. 2018.
View Article : Google Scholar
|
|
65
|
Banikazemi Z, Haji HA, Mohammadi M,
Taheripak G, Iranifar E, Poursadeghiyan M, Moridikia A, Rashidi B,
Taghizadeh M and Mirzaei H: Diet and cancer prevention: Dietary
compounds, dietary MicroRNAs, and dietary exosomes. J Cell Biochem.
119:185–196. 2018. View Article : Google Scholar
|
|
66
|
Saeedi Borujeni MJ, Esfandiary E,
Taheripak G, Codoñer-Franch P, Alonso-Iglesias E and Mirzaei H:
Molecular aspects of diabetes mellitus: Resistin, microRNA, and
exosome. J Cell Biochem. 119:1257–1272. 2018. View Article : Google Scholar
|
|
67
|
Golabchi K, Soleimani-Jelodar R, Aghadoost
N, Momeni F, Moridikia A, Nahand JS, Masoudifar A, Razmjoo H and
Mirzaei H: MicroRNAs in retinoblastoma: Potential diagnostic and
therapeutic biomarkers. J Cell Physiol. 233:3016–3023. 2018.
View Article : Google Scholar
|
|
68
|
Mirzaei H, Khataminfar S, Mohammadparast
S, Sales SS, Maftouh M, Mohammadi M, Simonian M, Parizadeh SM,
Hassanian SM and Avan A: Circulating microRNAs as Potential
Diagnostic Biomarkers and Therapeutic Targets in Gastric Cancer:
Current Status and Future Perspectives. Curr Med Chem.
23:4135–4150. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Rashidi B, Hoseini Z, Sahebkar A and
Mirzaei H: Anti-Atherosclerotic Effects of Vitamins D and E in
Suppression of Atherogenesis. J Cell Physiol. 232:2968–2976. 2017.
View Article : Google Scholar
|
|
70
|
Mirzaei H, Masoudifar A, Sahebkar A, Zare
N, Sadri Nahand J, Rashidi B, Mehrabian E, Mohammadi M, Mirzaei HR
and Jaafari MR: MicroRNA: A novel target of curcumin in cancer
therapy. J Cell Physiol. 233:3004–3015. 2018. View Article : Google Scholar
|
|
71
|
Calin GA and Croce CM: MicroRNA signatures
in human cancers. Nat Rev Cancer. 6:857–866. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Kanai M, Imaizumi A, Otsuka Y, Sasaki H,
Hashiguchi M, Tsujiko K, Matsumoto S, Ishiguro H and Chiba T:
Dose-escalation and pharmacokinetic study of nanoparticle curcumin,
a potential anticancer agent with improved bioavailability, in
healthy human volunteers. Cancer Chemother Pharmacol. 69:65–70.
2012. View Article : Google Scholar
|
|
73
|
Sinha D, Biswas J, Sung B, Aggarwal BB and
Bishayee A: Chemopreventive and chemotherapeutic potential of
curcumin in breast cancer. Curr Drug Targets. 13:1799–1819. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zhou S, Zhang S, Shen H, Chen W, Xu H,
Chen X, Sun D, Zhong S, Zhao J and Tang J: Curcumin inhibits cancer
progression through regulating expression of microRNAs. Tumour
Biol. 39:10104283176916802017. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Fadus MC, Lau C, Bikhchandani J and Lynch
HT: Curcumin: An age-old anti-inflammatory and anti-neoplastic
agent. J Tradit Complement Med. 7:339–346. 2016. View Article : Google Scholar
|