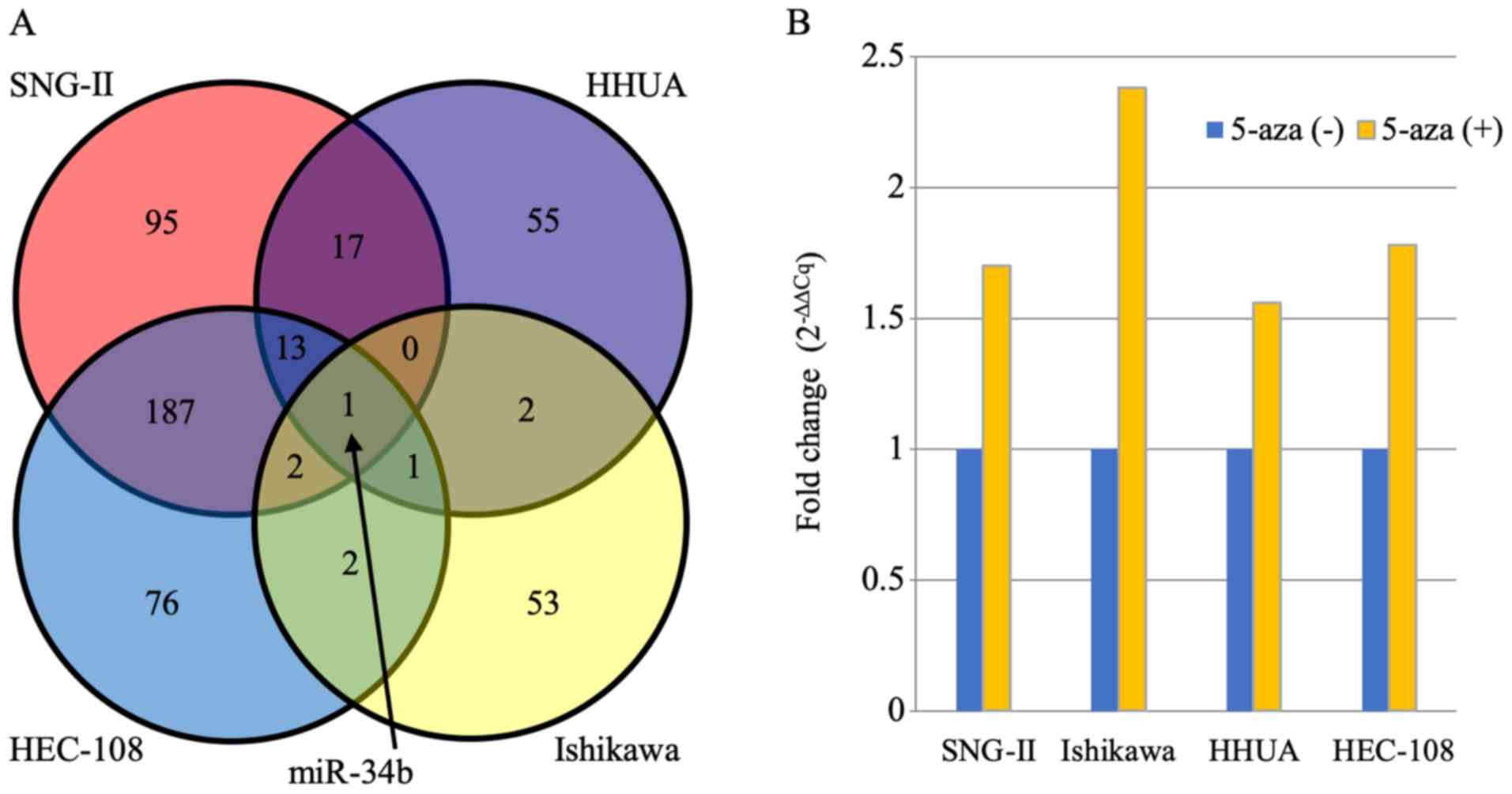
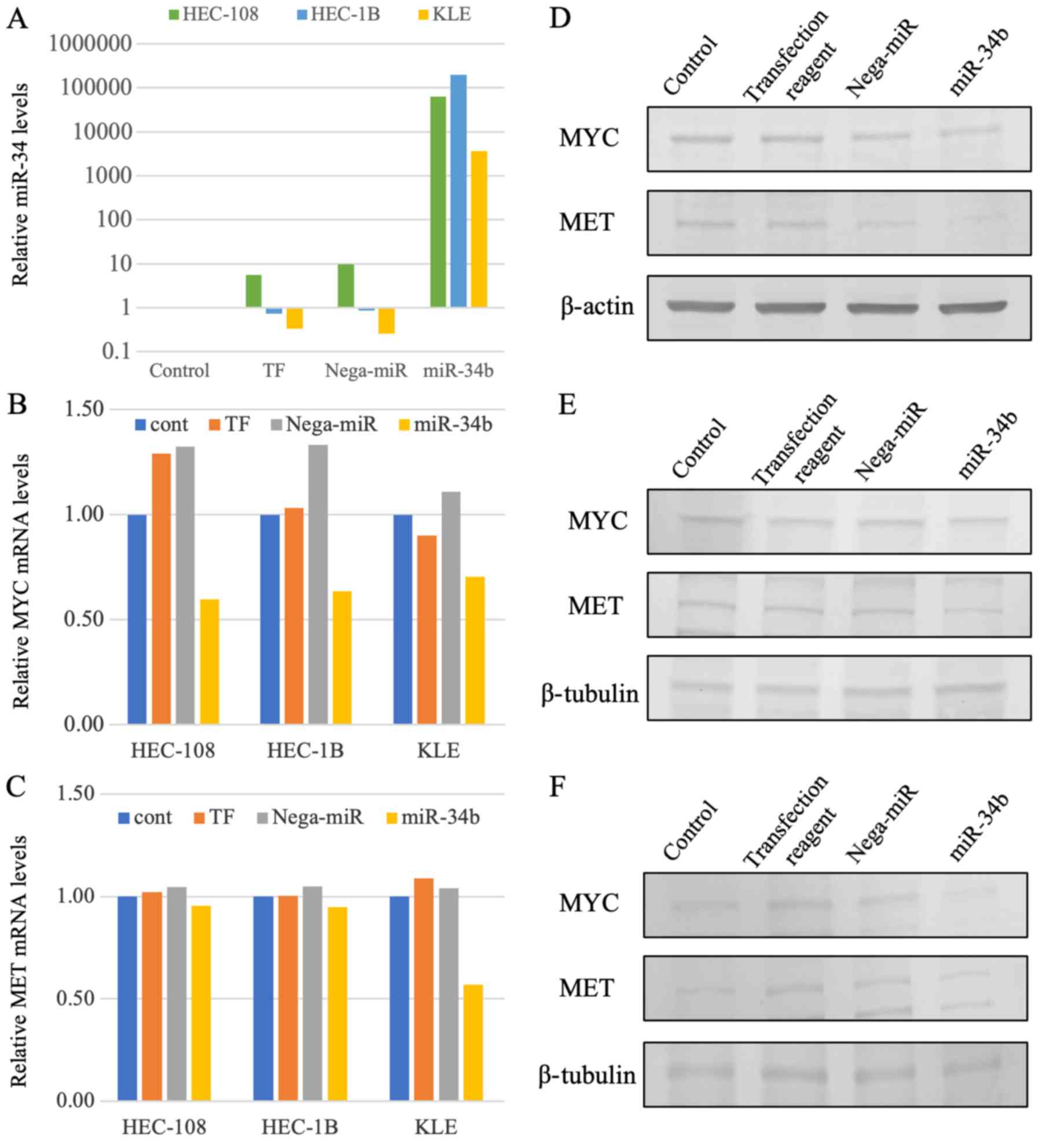
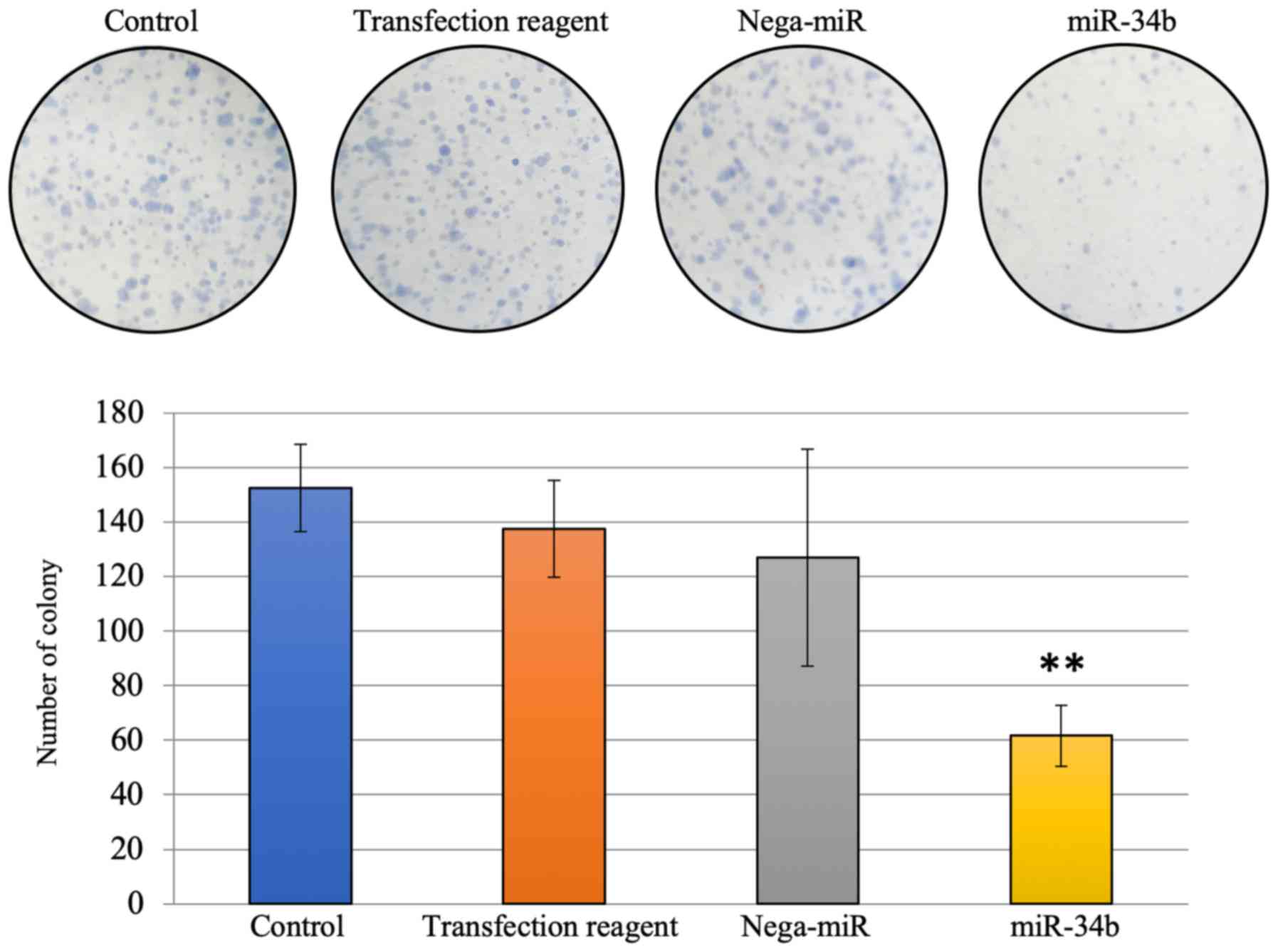
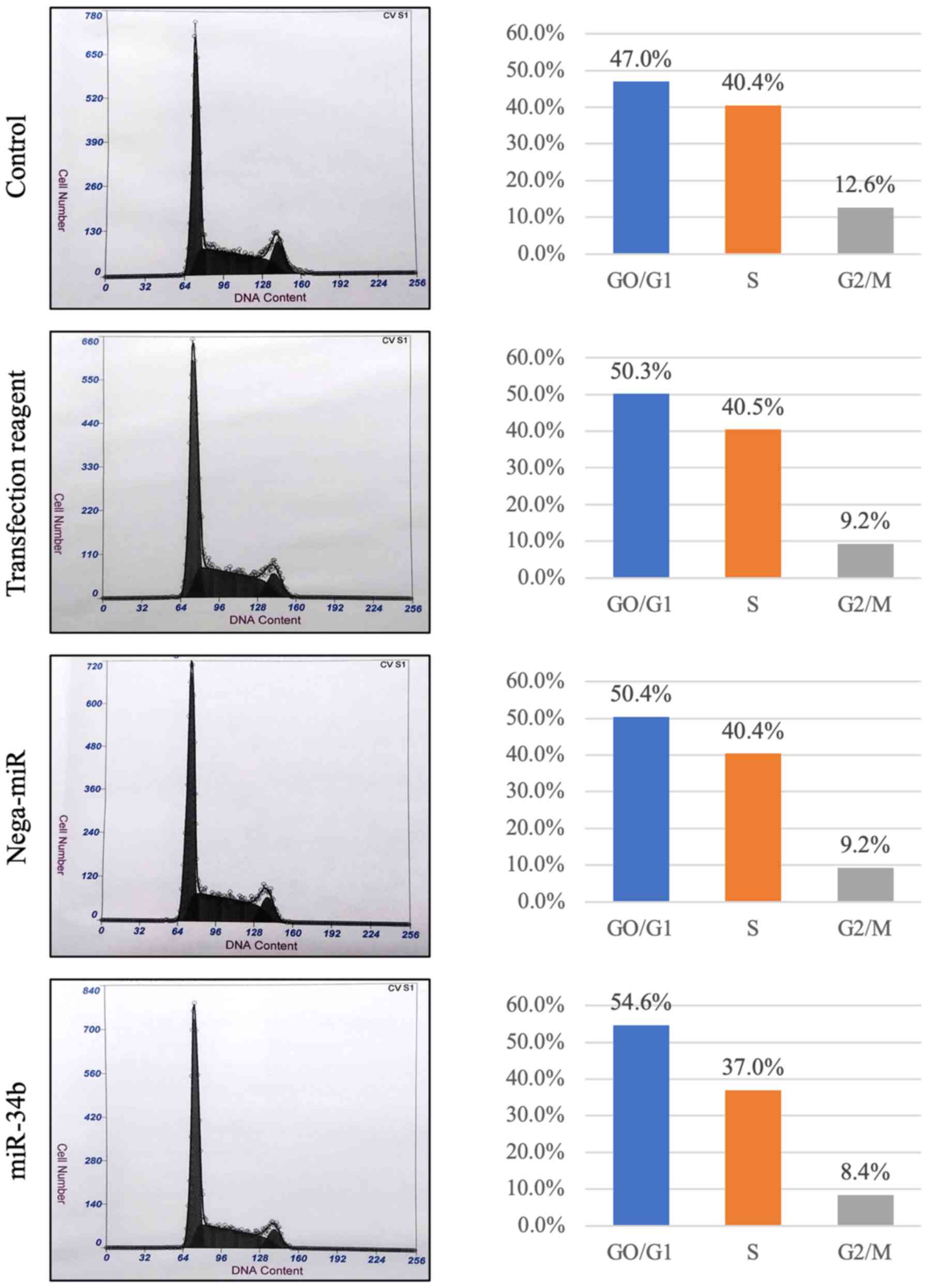
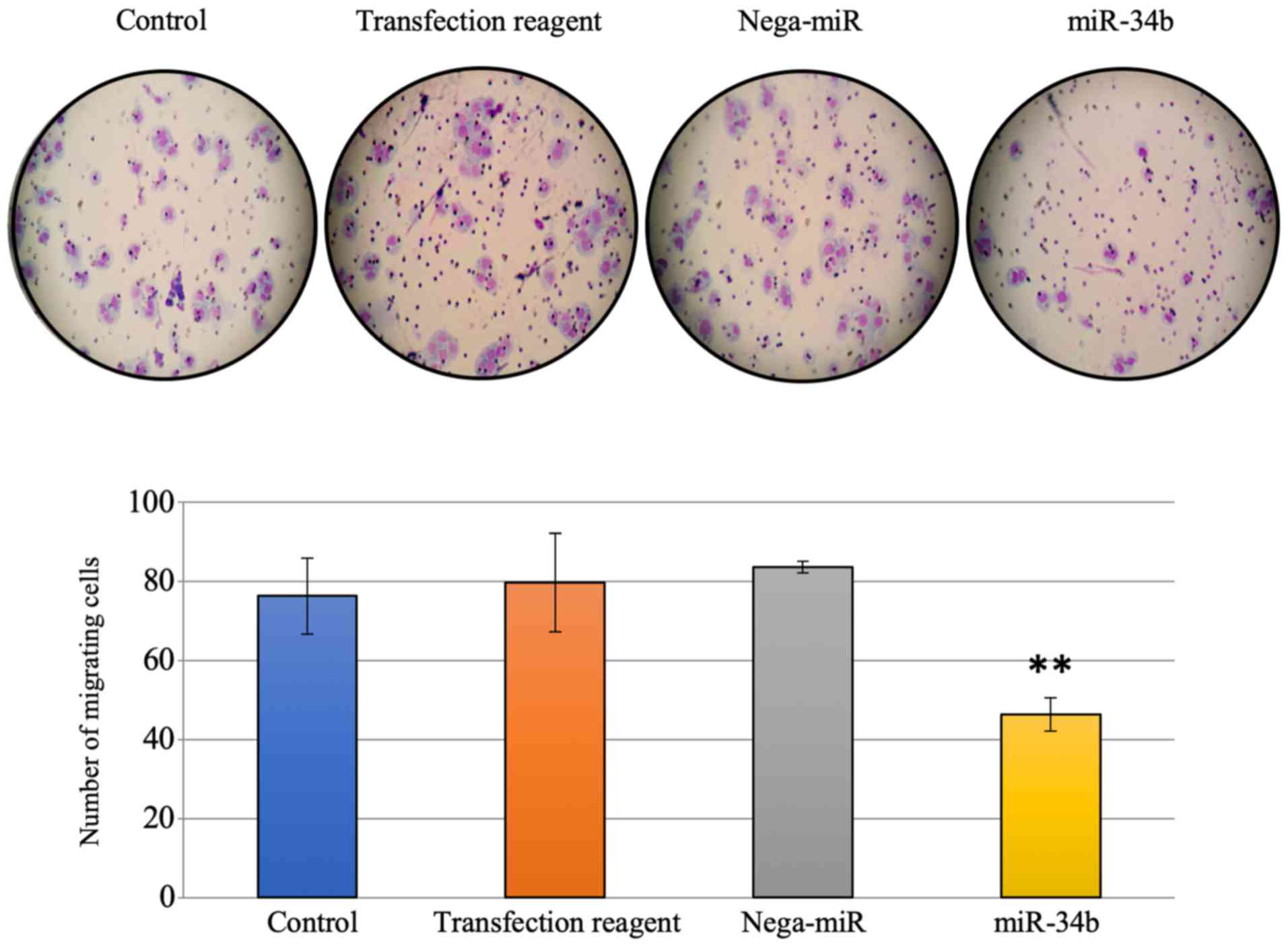
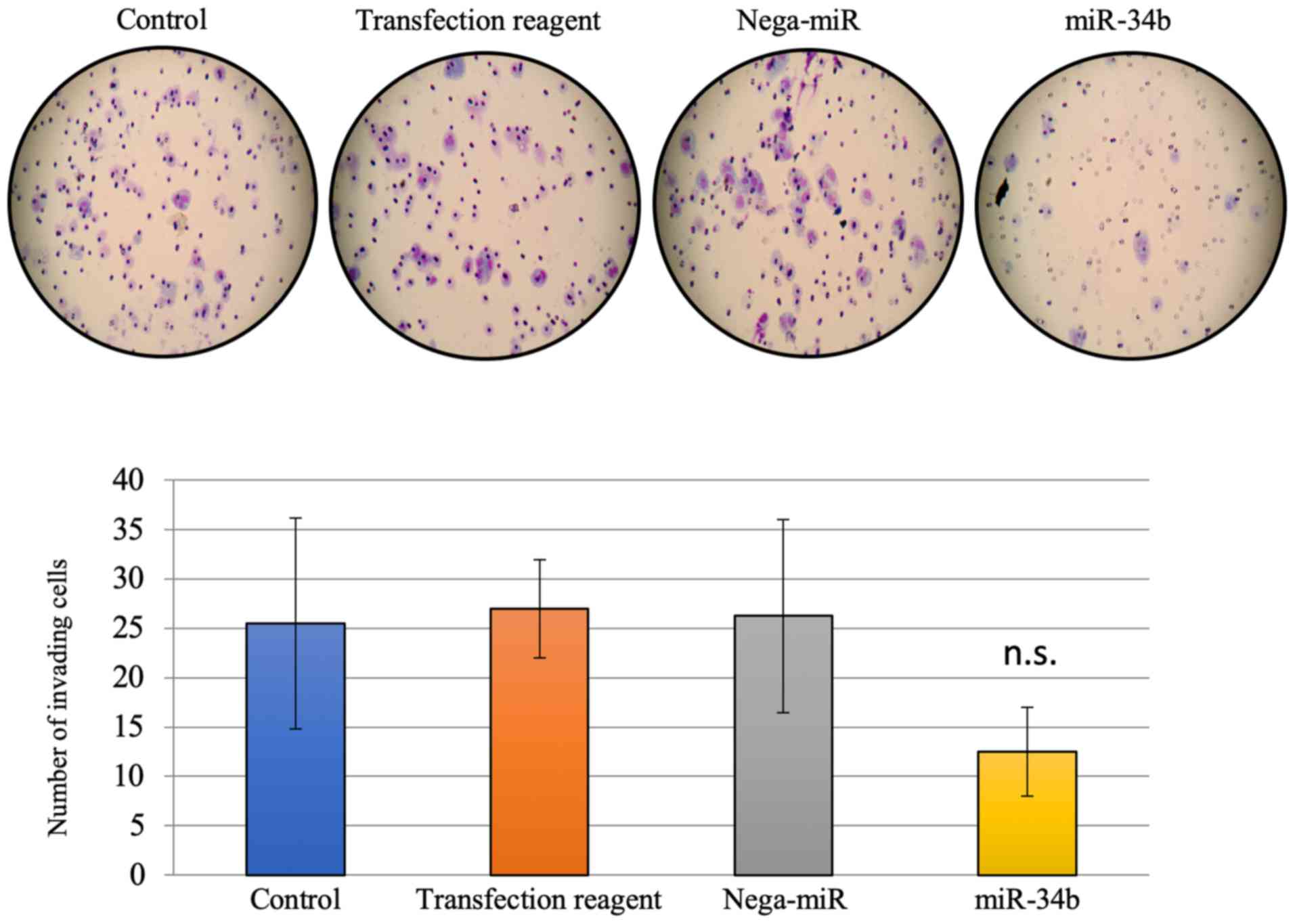
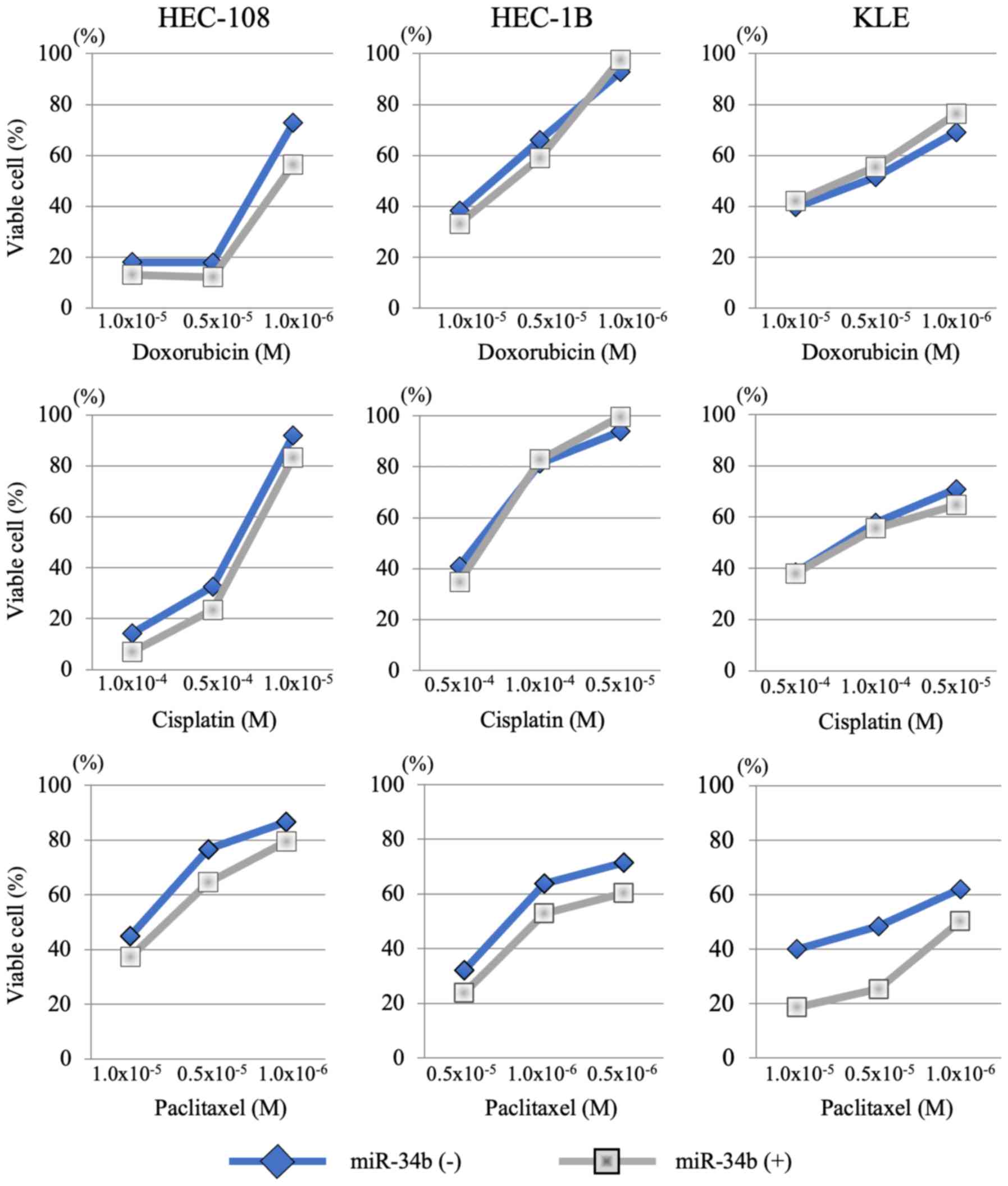
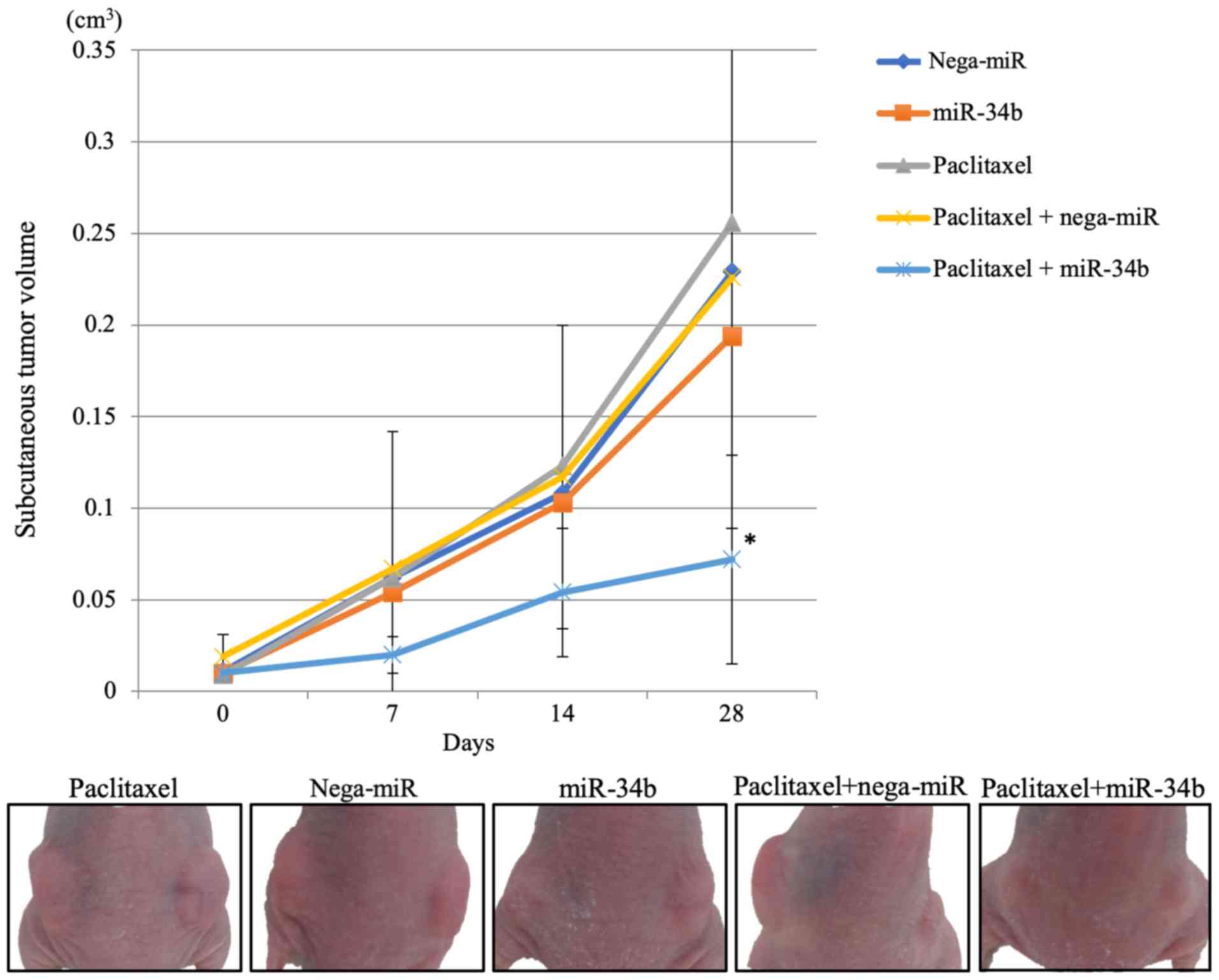
|
1
|
Lee JM, Yoo JK, Yoo H, Jung HY, Lee DR,
Jeong HC, Oh SH, Chung HM and Kim JK: The novel miR-7515 decreases
the proliferation and migration of human lung cancer cells by
targeting c-Met. Mol Cancer Res. 11:43–53. 2013. View Article : Google Scholar
|
|
2
|
Kozloski GA, Jiang X, Bhatt S, Ruiz J,
Vega F, Shaknovich R, Melnick A and Lossos IS: miR-181a negatively
regulates NF-κB signaling and affects activated B-cell-like diffuse
large B-cell lymphoma pathogenesis. Blood. 127:2856–2866. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ivey KN and Srivastava D: MicroRNAs as
regulators of differ-entiation and cell fate decisions. Cell Stem
Cell. 7:36–41. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bueno MJ and Malumbres M: MicroRNAs and
the cell cycle. Biochim Biophys Acta. 1812:592–601. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Su Z, Yang Z, Xu Y, Chen Y and Yu Q:
MicroRNAs in apoptosis, autophagy and necroptosis. Oncotarget.
6:8474–8490. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rachagani S, Macha MA, Menning MS, Dey P,
Pai P, Smith LM, Mo YY and Batra SK: Changes in microRNA (miRNA)
expression during pancreatic cancer development and progression in
a genetically engineered KrasG12D;Pdx1-Cre mouse (KC) model.
Oncotarget. 6:40295–40309. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Balacescu O, Sur D, Cainap C, Visan S,
Cruceriu D, Manzat-Saplacan R, Muresan MS, Balacescu L, Lisencu C
and Irimie A: The Impact of miRNA in colorectal cancer progression
and its liver metastases. Int J Mol Sci. 19:37112018. View Article : Google Scholar
|
|
8
|
Mavrogiannis AV, Kokkinopoulou I, Kontos
CK and Sideris DC: Effect of vinca alkaloids on the expression
levels of microRNAs targeting apoptosis-related genes in breast
cancer cell lines. Curr Pharm Biotechnol. 19:1076–1086. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bao W, Zhang Y, Li S, Fan Q, Qiu M, Wang
Y, Li Y, Ji X, Yang Y, Sang Z, et al: miR1075p promotes tumor
proliferation and invasion by targeting estrogen receptoralpha in
endometrial carcinoma. Oncol Rep. 41:1575–1585. 2019.
|
|
10
|
Li L, Shou H, Wang Q and Liu S:
Investigation of the potential theranostic role of KDM5B/miR-29c
signaling axis in paclitaxel resistant endometrial carcinoma. Gene.
694:76–82. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shu S, Liu X, Xu M, Gao X, Chen S, Zhang L
and Li R: MicroRNA-320a acts as a tumor suppressor in endometrial
carcinoma by targeting IGF-1R. Int J Mol Med. 43:1505–1512.
2019.PubMed/NCBI
|
|
12
|
Tan A, Luo R and Ruan P: miR-495 promotes
apoptosis and inhibits proliferation in endometrial cells via
targeting PIK3R1. Pathol Res Pract. 215:594–599. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Engkvist ME, Stratford EW, Lorenz S,
Meza-Zepeda LA, Myklebost O and Munthe E: Analysis of the miR-34
family functions in breast cancer reveals annotation error of
miR-34b. Sci Rep. 7:96552017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fang LL, Sun BF, Huang LR, Yuan HB, Zhang
S, Chen J, Yu ZJ and Luo H: Potent inhibition of miR-34b on
migration and inva-sion in metastatic prostate cancer cells by
regulating the TGF-β pathway. Int J Mol Sci. 18:27622017.
View Article : Google Scholar
|
|
15
|
Wang Y, Wu Z and Hu L: The regulatory
effects of metformin on the [SNAIL/miR-34]:[ZEB/miR-200] system in
the epithelial-mesenchymal transition (EMT) for colorectal cancer
(CRC). Eur J Pharmacol. 834:45–53. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Metheetrairut C, Chotigavanich C,
Amornpichetkul K, Keskool P, Ongard S and Metheetrairut C:
Expression levels of miR-34-family microRNAs are associated with
TP53 mutation status in head and neck squamous cell carcinoma. Eur
Arch Otorhinolaryngol. 276:521–533. 2019. View Article : Google Scholar
|
|
17
|
Zhang L, Wang L, Dong D, Wang Z, Ji W, Yu
M, Zhang F, Niu R and Zhou Y: MiR-34b/c-5p and the neurokinin-1
receptor regulate breast cancer cell proliferation and apoptosis.
Cell Prolif. 52:e125272019. View Article : Google Scholar
|
|
18
|
Yang L, Song X, Zhu J, Li M, Ji Y, Wu F,
Chen Y, Cui X, Hu J, Wang L, et al: Tumor suppressor microRNA-34a
inhibits cell migration and invasion by targeting
MMP-2/MMP-9/FNDC3B in esophageal squamous cell carcinoma. Int J
Oncol. 51:378–388. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Corney DC, Hwang CI, Matoso A, Vogt M,
Flesken-Nikitin A, Godwin AK, Kamat AA, Sood AK, Ellenson LH,
Hermeking H and Nikitin AY: Frequent downregulation of miR-34
family in human ovarian cancers. Clin Cancer Res. 16:1119–1128.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Javeri A, Ghaffarpour M, Taha MF and
Houshmand M: Downregulation of miR-34a in breast tumors is not
associated with either p53 mutations or promoter hypermethylation
while it correlates with metastasis. Med Oncol. 30:4132013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Okada N, Lin CP, Ribeiro MC, Biton A, Lai
G, He X, Bu P, Vogel H, Jablons DM, Keller AC, et al: A positive
feedback between p53 and miR-34 miRNAs mediates tumor suppression.
Genes Deve. 28:438–450. 2014. View Article : Google Scholar
|
|
22
|
Xie K, Liu J, Chen J, Dong J, Ma H, Liu Y
and Hu Z: Methylation-associated silencing of microRNA-34b in
hepatocellular carcinoma cancer. Gene. 543:101–107. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kawaguchi M, Yanokura M, Banno K,
Kobayashi Y, Kuwabara Y, Kobayashi M, Nomura H, Hirasawa A, Susumu
N and Aoki D: Analysis of a correlation between the BRAF V600E
mutation and abnormal DNA mismatch repair in patients with sporadic
endometrial cancer. Int J Oncol. 34:1541–1547. 2009.PubMed/NCBI
|
|
24
|
Yanokura M, Banno K, Kawaguchi M, Hirao N,
Hirasawa A, Susumu N, Tsukazaki K and Aoki D: Relationship of
aberrant DNA hypermethylation of CHFR with sensitivity to taxanes
in endometrial cancer. Oncol Rep. 17:41–48. 2007.
|
|
25
|
Yanokura M, Banno K, Susumu N, Kawaguchi
M, Kuwabara Y, Tsukazaki K and Aoki D: Hypermethylation in the p16
promoter region in the carcinogenesis of endometrial cancer in
Japanese patients. Anticancer Res. 26:851–856. 2006.PubMed/NCBI
|
|
26
|
Banno K, Yanokura M, Susumu N, Kawaguchi
M, Hirao N, Hirasawa A, Tsukazaki K and Aoki D: Relationship of the
aberrant DNA hypermethylation of cancer-related genes with
carcinogenesis of endometrial cancer. Oncol Reps. 16:1189–1196.
2006.
|
|
27
|
Agostini M and Knight RA: miR-34: From
bench to bedside. Oncotarget. 5:872–881. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
29
|
Mondal A and Bennett LL: Resveratrol
enhances the efficacy of sorafenib mediated apoptosis in human
breast cancer MCF7 cells through ROS, cell cycle inhibition,
caspase 3 and PARP cleavage. Biomed Pharmacother. 84:1906–1914.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Migliore C, Petrelli A, Ghiso E, Corso S,
Capparuccia L, Eramo A, Comoglio PM and Giordano S: MicroRNAs
impair MET-mediated invasive growth. Cancer Res. 68:10128–10136.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Seviour EG, Sehgal V, Lu Y, Luo Z, Moss T,
Zhang F, Hill SM, Liu W, Maiti SN, Cooper L, et al: Functional
proteomics identifies miRNAs to target a p27/Myc/phospho-Rb
signature in breast and ovarian cancer. Oncogene. 35:691–701. 2016.
View Article : Google Scholar
|
|
32
|
Siemens H, Jackstadt R, Hunten S, Kaller
M, Menssen A, Gotz U and Hermeking H: miR-34 and SNAIL form a
double-negative feedback loop to regulate epithelial-mesenchymal
transitions. Cell Cycle. 10:4256–4271. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wang J and Cheng JX: c-Met inhibition
enhances chemosensitivity of human ovarian cancer cells. Clin Exp
Pharmacol Physiol. 44:79–87. 2017. View Article : Google Scholar
|
|
34
|
Yang L, Song Z, Wang X, Yang W, Wang M and
Liu H: Huaier extract enhances the treatment efficacy of paclitaxel
in breast cancer cells via the NF-κB/IκBα pathway. Oncol Rep.
38:3455–3464. 2017.PubMed/NCBI
|
|
35
|
Kress TR, Cannell IG, Brenkman AB, Samans
B, Gaestel M, Roepman P, Burgering BM, Bushell M, Rosenwald A and
Eilers M: The MK5/PRAK kinase and Myc form a negative feedback loop
that is disrupted during colorectal tumorigenesis. Mol Cell.
41:445–457. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Cannell IG, Kong YW, Johnston SJ, Chen ML,
Collins HM, Dobbyn HC, Elia A, Kress TR, Dickens M, Clemens MJ, et
al: p38 MAPK/MK2-mediated induction of miR-34c following DNA damage
prevents Myc-dependent DNA replication. Proc Natl Acad Sci USA.
107:5375–5380. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hydbring P, Bahram F, Su Y, Tronnersjo S,
Hogstrand K, von der Lehr N, Sharifi HR, Lilischkis R, Hein N, Wu
S, et al: Phosphorylation by Cdk2 is required for Myc to repress
Ras-induced senescence in cotransformation. Proc Natl Acad Sci USA.
107:58–63. 2010. View Article : Google Scholar
|
|
38
|
Watanabe K, Emoto N, Hamano E, Sunohara M,
Kawakami M, Kage H, Kitano K, Nakajima J, Goto A, Fukayama M, et
al: Genome structure-based screening identified epigenetically
silenced microRNA associated with invasiveness in non-small-cell
lung cancer. Int J Cancer. 130:2580–2590. 2012. View Article : Google Scholar
|
|
39
|
Chu SH, Ma YB, Feng DF, Zhang H, Qiu JH
and Zhu ZA: c-Met antisense oligodeoxynucleotides increase
sensitivity of human glioma cells to paclitaxel. Oncol Rep.
24:189–194. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Begg AC, Stewart FA and Vens C: Strategies
to improve radiotherapy with targeted drugs. Nat Rev Cancer.
11:239–253. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Shah MA and Schwartz GK: Cell
cycle-mediated drug resistance: An emerging concept in cancer
therapy. Clin Cancer Res. 7:2168–2181. 2001.PubMed/NCBI
|
|
42
|
Choi J, Lee HE, Kim MA, Jang BG, Lee HS
and Kim WH: Analysis of MET mRNA expression in gastric cancers
using RNA in situ hybridization assay: Its clinical implication and
comparison with immunohistochemistry and silver in situ
hybridization. PLoS One. 9:e1116582014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Fu P, Du F, Yao M, Lv K and Liu Y:
MicroRNA-185 inhibits proliferation by targeting c-Met in human
breast cancer cells. Exp Ther Med. 8:1879–1883. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Rubin MA: Insights into the mechanism of
organ-specific cancer metastasis. Cancer Discov. 4:1262–1264. 2014.
View Article : Google Scholar : PubMed/NCBI
|