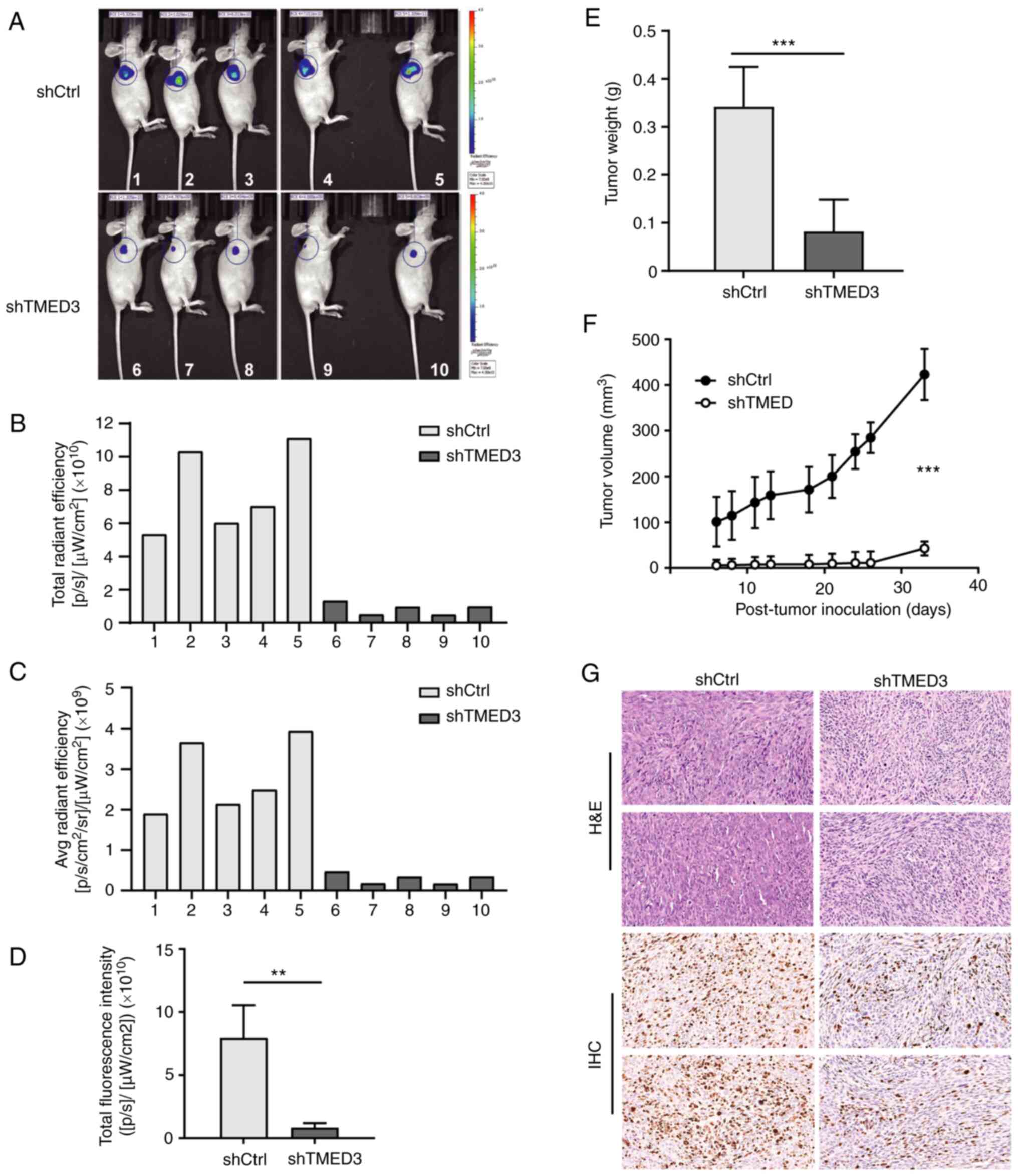
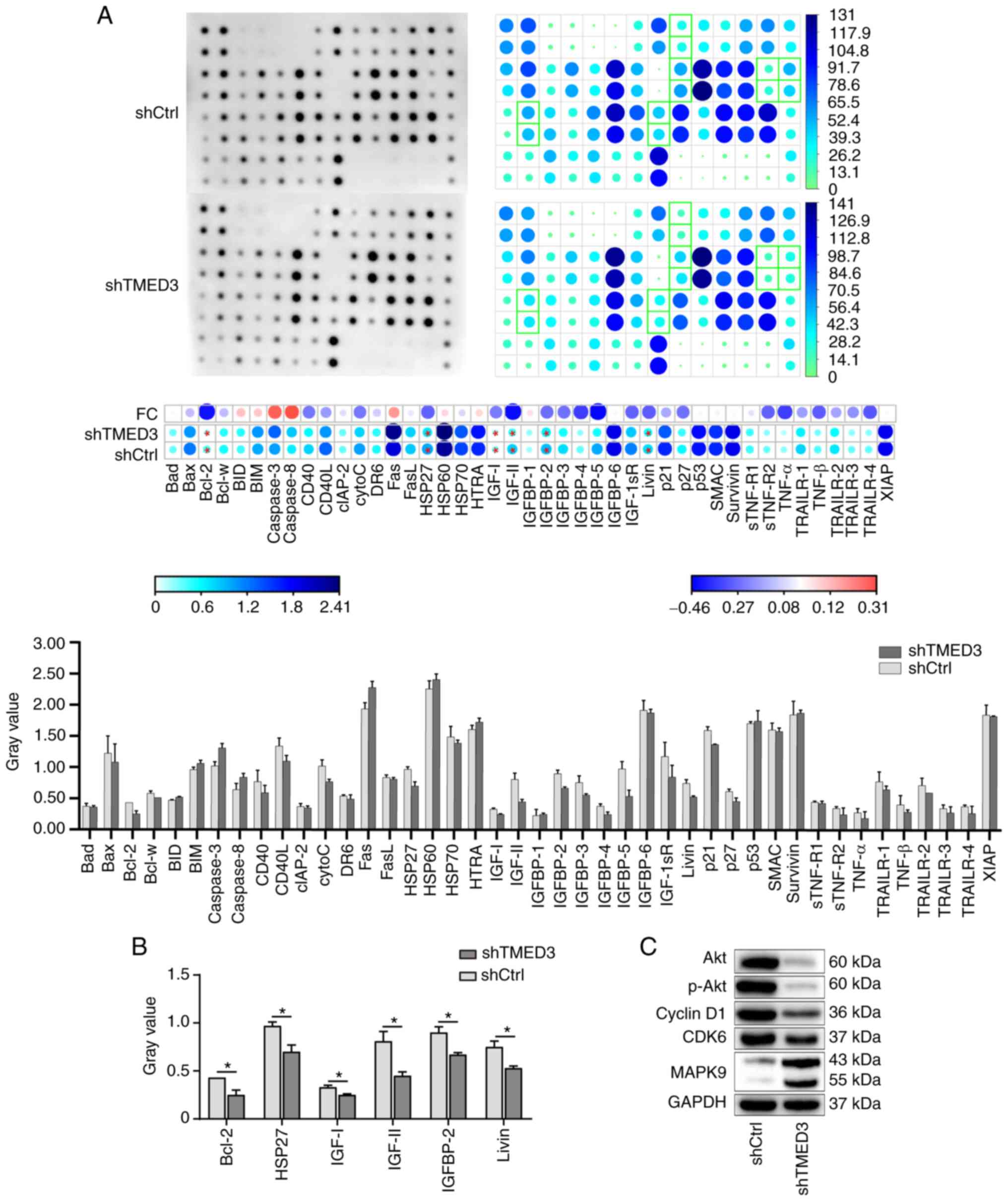
|
1
|
Sahyouni R, Goshtasbi K, Mahmoodi A and
Chen JW: A historical recount of chordoma. J Neurosurg Spine.
28:422–428. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Thanindratarn P, Dean DC, Nelson SD,
Hornicek FJ and Duan Z: Advances in immune checkpoint inhibitors
for bone sarcoma therapy. J Bone Oncol. 15:1002212019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Stacchiotti S and Sommer J; Chordoma
Global Consensus Group: Building a global consensus approach to
chordoma: A position paper from the medical and patient community.
Lancet Oncol. 16:e71–e83. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Guan JY, He XF, Chen Y, Zeng QL, Mei QL
and Li YH: Percutaneous intratumoral injection with pingyangmycin
lipiodol emulsion for the treatment of recurrent sacrococcygeal
chordomas. J Vasc Interv Radiol. 22:1216–1220. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kayani B, Hanna SA, Sewell MD, Saifuddin
A, Molloy S and Briggs TW: A review of the surgical management of
sacral chordoma. Eur J Surg Oncol. 40:1412–1420. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yang XR, Ng D, Alcorta DA, Liebsch NJ,
Sheridan E, Li S, Goldstein AM, Parry DM and Kelley MJ: T
(brachyury) gene duplication confers major susceptibility to
familial chordoma. Nat Genet. 41:1176–1178. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Stacchiotti S, Tamborini E, Lo Vullo S,
Bozzi F, Messina A, Morosi C, Casale A, Crippa F, Conca E, Negri T,
et al: Phase II study on lapatinib in advanced EGFR-positive
chordoma. Ann Oncol. 24:1931–1936. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Magnaghi P, Salom B, Cozzi L, Amboldi N,
Ballinari D, Tamborini E, Gasparri F, Montagnoli A, Raddrizzani L,
Somaschini A, et al: Afatinib is a new therapeutic approach in
chordoma with a unique ability to target EGFR and brachyury. Mol
Cancer Ther. 17:603–613. 2018. View Article : Google Scholar
|
|
9
|
Strating JR and Martens GJ: The p24 family
and selective transport processes at the ER-Golgi interface. Biol
Cell. 101:495–509. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Barr FA, Preisinger C, Kopajtich R and
Korner R: Golgi matrix proteins interact with p24 cargo receptors
and aid their efficient retention in the Golgi apparatus. J Cell
Biol. 155:885–891. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Connolly DJ, O'Neill LA and McGettrick AF:
The GOLD domain-containing protein TMED1 is involved in
interleukin-33 signaling. J Biol Chem. 288:5616–5623. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Peng C, Huang K, Liu G, Li Y and Yu C:
MiR-876-3p regulates cisplatin resistance and stem cell-like
properties of gastric cancer cells by targeting TMED3. J
Gastroenterol Hepatol. 34:1711–1719. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pei J, Zhang J, Yang X, Wu Z, Sun C, Wang
Z and Wang B: TMED3 promotes cell proliferation and motility in
breast cancer and is negatively modulated by miR-188-3p. Cancer
Cell Int. 19:752019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ha M, Moon H, Choi D, Kang W, Kim JH, Lee
KJ, Park D, Kang CD, Oh SO, Han ME, et al: Prognostic role of TMED3
in clear cell renal cell carcinoma: A retrospective Multi-cohort
analysis. Front Genet. 10:3552019. View Article : Google Scholar :
|
|
15
|
Zheng H, Yang Y, Han J, Jiang WH, Chen C,
Wang MC, Gao R, Li S, Tian T, Wang J, et al: TMED3 promotes
hepatocellular carcinoma progression via IL-11/STAT3 signaling. Sci
Rep. 6:370702016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Vainio P, Mpindi JP, Kohonen P, Fey V,
Mirtti T, Alanen KA, Perälä M, Kallioniemi O and Iljin K:
High-throughput transcriptomic and RNAi analysis identifies AIM1,
ERGIC1, TMED3 and TPX2 as potential drug targets in prostate
cancer. PLoS One. 7:e398012012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mishra S, Bernal C, Silvano M, Anand S and
Ruiz IAA: The protein secretion modulator TMED9 drives
CNIH4/TGFalpha/GLI signaling opposing TMED3-WNT-TCF to promote
colon cancer metastases. Oncogene. 38:5817–5837. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Duquet A, Melotti A, Mishra S, Malerba M,
Seth C, Conod A and Altaba AR: A novel genome-wide in vivo screen
for metastatic suppressors in human colon cancer identifies the
positive WNT-TCF pathway modulators TMED3 and SOX12. EMBO Mol Med.
6:882–901. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pfaffl MW: A new mathematical model for
relative quantification in real-time RT-PCR. Nucleic Acids Res.
29:e452001. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
21
|
Laferriere CA and Pang DS: Review of
intraperitoneal injection of sodium pentobarbital as a method of
euthanasia in laboratory rodents. J Am Assoc Lab Anim Sci.
59:254–263. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
National Research Council (US) Committee
for the Update of the Guide for the Care and se of Laboratory
Animals: Guide for the Care and Use of Laboratory Animals. 8th
edition. National Academies Press (US); Washington, DC: 2011
|
|
23
|
Thul PJ, Akesson L, Wiking M, Mahdessian
D, Geladaki A, Blal HA, Alm T, Asplund A, Björk L, Breckels LM, et
al: A subcellular map of the human proteome. Science.
356:eaal33212017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Aber R, Chan W, Mugisha S and
Jerome-Majewska LA: Transmembrane emp24 domain proteins in
development and disease. Genet Res (Camb). 101:e142019. View Article : Google Scholar
|
|
25
|
Wong RS: Apoptosis in cancer: From
pathogenesis to treatment. J Exp Clin Cancer Res. 30:872011.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Brahmkhatri VP, Prasanna C and Atreya HS:
Insulin-like growth factor system in cancer: Novel targeted
therapies. Biomed Res Int. 2015:5380192015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Simpson A, Petnga W, Macaulay VM,
Weyer-Czernilofsky U and Bogenrieder T: Insulin-like growth factor
(IGF) pathway targeting in cancer: Role of the IGF axis and
opportunities for future combination studies. Target Oncol.
12:571–597. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bach LA: IGF-binding proteins. J Mol
Endocrinol. 61:T11–T28. 2018. View Article : Google Scholar
|
|
29
|
Baxter RC: IGF binding proteins in cancer:
Mechanistic and clinical insights. Nat Rev Cancer. 14:329–341.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kasof GM and Gomes BC: Livin, a novel
inhibitor of apoptosis protein family member. J Biol Chem.
276:3238–3246. 2001. View Article : Google Scholar
|
|
31
|
Garrido C, Schmitt E, Cande C, Vahsen N,
Parcellier A and Kroemer G: HSP27 and HSP70: Potentially oncogenic
apoptosis inhibitors. Cell Cycle. 2:579–584. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Deveraux QL, Schendel SL and Reed JC:
Antiapoptotic proteins. The bcl-2 and inhibitor of apoptosis
protein families. Cardiol Clin. 19:57–74. 2001. View Article : Google Scholar
|
|
33
|
Kandel ES and Hay N: The regulation and
activities of the multifunctional serine/threonine kinase Akt/PKB.
Exp Cell Res. 253:210–229. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Fresno Vara JA, Casado E, de Castro J,
Cejas P, Belda-Iniesta C and Gonzalez-Baron M: PI3K/Akt signalling
pathway and cancer. Cancer Treat Rev. 30:193–204. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hoxhaj G and Manning BD: The PI3K-AKT
network at the interface of oncogenic signalling and cancer
metabolism. Nat Rev Cancer. 20:74–88. 2020. View Article : Google Scholar :
|
|
36
|
Tamborini E, Virdis E, Negri T, Orsenigo
M, Brich S, Conca E, Gronchi A, Stacchiotti S, Manenti G, Casali
PG, et al: Analysis of receptor tyrosine kinases (RTKs) and
downstream pathways in chordomas. Neuro Oncol. 12:776–789. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fry MJ: Phosphoinositide 3-kinase
signalling in breast cancer: How big a role might it play? Breast
Cancer Res. 3:304–312. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ippen FM, Grosch JK, Subramanian M, Kuter
BM, Liederer BM, Plise EG, Mora JL, Nayyar N, Schmidt SP,
Giobbie-Hurder A, et al: Targeting the PI3K/Akt/mTOR pathway with
the pan-Akt inhibitor GDC-0068 in PIK3CA-mutant breast cancer brain
metastases. Neuro Oncol. 21:1401–1411. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sherr CJ, Beach D and Shapiro GI:
Targeting CDK4 and CDK6: From discovery to therapy. Cancer Discov.
6:353–367. 2016. View Article : Google Scholar :
|
|
40
|
Diehl JA, Zindy F and Sherr CJ: Inhibition
of cyclin D1 phosphorylation on threonine-286 prevents its rapid
degradation via the ubiquitin-proteasome pathway. Genes Dev.
11:957–972. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Yoon CH, Kim MJ, Kim RK, Lim EJ, Choi KS,
An S, Hwang SG, Kang SG, Suh Y, Park MJ and Lee SJ: c-Jun
N-terminal kinase has a pivotal role in the maintenance of
self-renewal and tumorigenicity in glioma stem-like cells.
Oncogene. 31:4655–4666. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Bubici C and Papa S: JNK signalling in
cancer: In need of new, smarter therapeutic targets. Br J
Pharmacol. 171:24–37. 2014. View Article : Google Scholar :
|
|
43
|
Wu Q, Wu W, Fu B, Shi L, Wang X and Kuca
K: JNK signaling in cancer cell survival. Med Res Rev.
39:2082–2104. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Sabapathy K and Wagner EF: JNK2: A
negative regulator of cellular proliferation. Cell Cycle.
3:1520–1523. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Fan M, Goodwin M, Vu T, Brantley-Finley C,
Gaarde WA and Chambers TC: Vinblastine-induced phosphorylation of
Bcl-2 and Bcl-XL is mediated by JNK and occurs in parallel with
inactivation of the Raf-1/MEK/ERK cascade. J Biol Chem.
275:29980–29985. 2000. View Article : Google Scholar : PubMed/NCBI
|