Glycolysis is a respiratory metabolic pathway in
cells that generates two molecules of ATP and pyruvate (1). When oxygen is available, pyruvate is
oxidized by pyruvate dehydrogenase to produce acetyl coenzyme A
(CoA), which enters the tricarboxylic acid (TCA) cycle and
oxidative phosphorylation (OXPHOS) (2). When oxygen is absent, pyruvate is
catalyzed by lactate dehydrogenase to lactic acid, which fuels the
TCA cycle (3) and may also become
a gluconeogenic precursor through gluconeogenesis (4). Tumor cells have been considered major
consumers of glucose, producing lactic acid to fuel their growth
through glycolysis, while generating nicotinamide-adenine
dinucleotide phosphate through the parallel pentose phosphate
pathway, despite the presence of oxygen (5,6). By
contrast, the tumor microenvironment (TME) is the microecosystem in
which the tumor survives and thrives, comprising tumor cells,
stromal cells and associated immune cells, such as tumor-associated
macrophages (TAMs), T cells and dendritic cells, as well as their
products (e.g., cytokines and chemokines) (7). Macrophages are usually classified
into classically activated macrophages (M1 type) and alternatively
activated macrophages (M2 type) according to their activation and
function (8). M1 macrophages are
mainly energized by glycolysis, while the main source of energy for
M2 macrophages is fatty acid oxidation and OXPHOS. Furthermore, in
the presence of active OXPHOS, M2 type macrophage differentiation
does not require glycolytic stimulation (9). Of note, recent studies have indicated
that in the TME, TAMs consume the most glucose (10), while interleukin (IL)-4-induced
M2-like TAMs have significantly increased glycolytic reserves and
the highest glycolytic capacity compared with those of resting
macrophages (M0). M1-like TAMs further promote cancer metastasis
and chemoresistance (11,12). This suggests that although M2-like
TAMs are similar to M2 macrophages in terms of their
differentiation characteristics and secretory factors, they exhibit
a high dependence on glycolysis at the metabolic level (13).
During tumor development, TAMs infiltrating tumor
tissues tend to exhibit high plasticity and undergo corresponding
metabolic changes depending on oxygen and nutrient conditions,
ultimately affecting their phenotype and function (14). Cytokines in the TME, including
IL-12, tumor necrosis factor α (TNF-α) and interferon (IFN)-γ,
promote macrophage polarization to the M1 state. When stimulated by
IL-4 and colony-stimulating factor (CSF)-1 produced by cancer cells
and CD4+ T cells, as well as granulocyte macrophage
(GM)-CSF produced by cancer cells, TAMs finally polarize to the M2
state (15-17). In the early stages of tumor
development, TAMs tend to be the M1 type, while as the tumor
progresses, the M2 type gradually predominates (18). The relationship between glycolysis
and TAMs has received increasing attention and the purpose of the
present review is to summarize the effects of alterations in the
glycolytic process of TAMs on their polarization and function based
on the progress of existing studies, as well as to summarize the
role of tumor cells and immune cells in the body microenvironment
in regulating the polarization and function of TAMs through
glycolysis, to provide a comprehensive understanding of the
relationship between glycolysis and TAMs.
When macrophages change from a quiescent to an
activated state, the activities of key enzymes related to
glycolysis are frequently altered. The regulation of kinases
involved in glucose metabolism may alter the macrophage phenotype
and affect cytokine production and the expression of key surface
receptors (19). Among them, key
enzymes of glycolysis, such as hexokinase (HK), phosphofructokinase
(PFK) and pyruvate kinase M2 (PKM2), have important roles in the
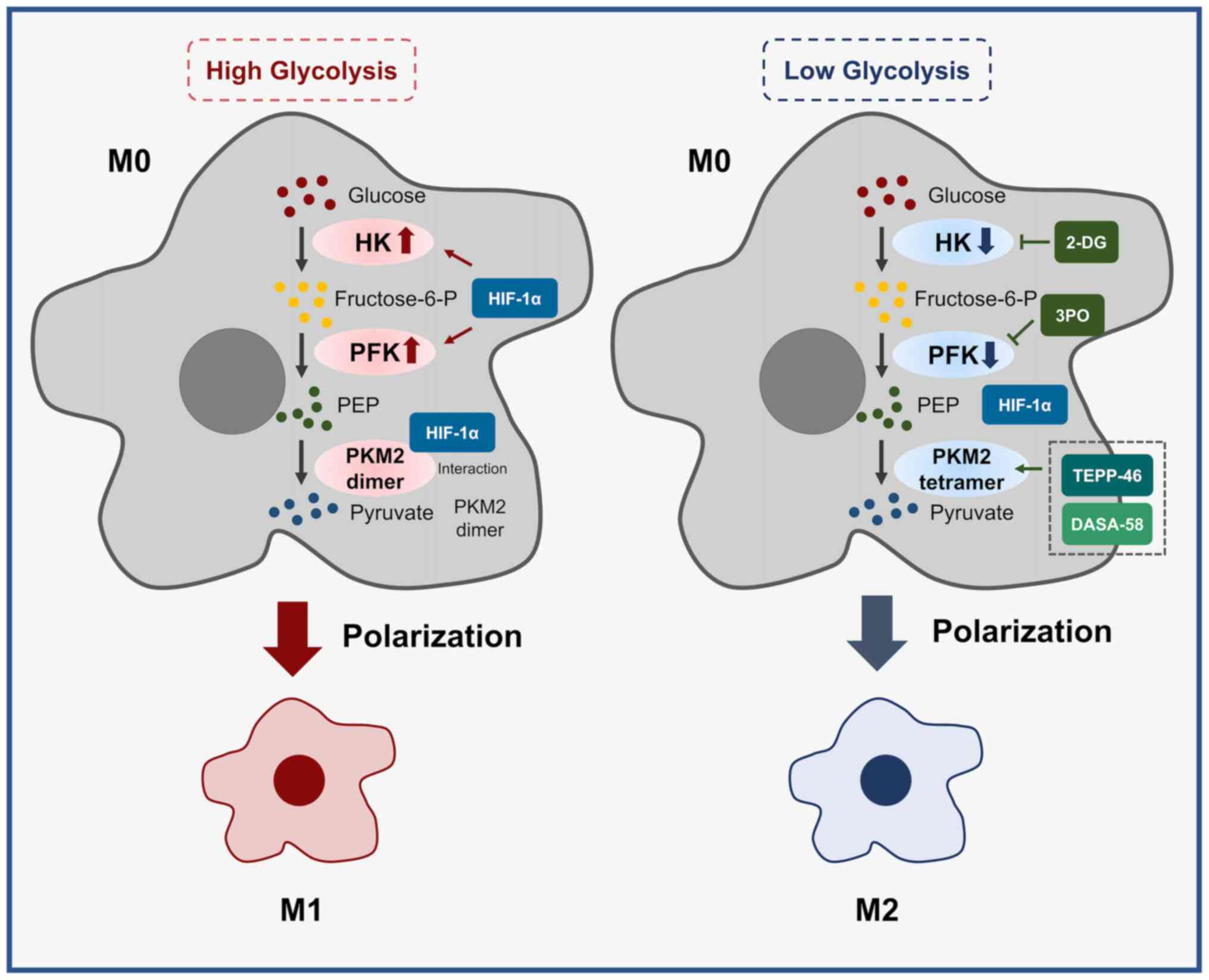
polarization and functional changes of TAMs (20-24).
HK is the first rate-limiting enzyme of glycolysis,
catalyzing the phosphorylation of glucose to glucose 6-phosphate
and entering various downstream metabolic pathways (24). In mammals, five HK isozymes (HK1,
HK2, HK3, glucokinase and HK domain containing 1) have been
identified (25). Among them, HK1-
and HK2-mediated glycolysis has a regulatory role in macrophage
polarization. IFN regulatory factor 5 increases the expression of
hypoxia-inducible factor-1α (HIF-1α) and actives HK1 through the
activation of protein kinase B β, which in turn triggers M1
polarization of macrophages (26).
However, in mouse J774A.1 macrophages, inhibition of mechanistic
target of rapamycin (mTOR) complex 1 (mTORC1), which affects
HK1-dependent glycolysis, inhibited macrophage M1 polarization
(27). In addition, in a study of
HK2, it was found that the basic helix-loop-helix family member e40
promotes the activities of HK2 and PFK by increasing macrophage
HIF1-α expression, further promoting macrophage polarization toward
the M1 type (28). Studies have
indicated that targeting HK activity using drugs effectively
controls macrophage polarization and function. Increased levels of
glucose transporter protein 1 (GLUT1), GLUT3 and HK2 in macrophages
were revealed by western blot analysis after lipopolysaccharide
(LPS) activation of RAW 264.7 macrophages (29), and targeted inhibition of HK2 by
using the glycolytic inhibitor 2-deoxy-D-glucose (2-DG) hindered
macrophage polarization to M1 (30). In response to HK2, all-trans
retinoic acid, a derivative of vitamin A, was found to promote
IL-1β maturation and secretion by enhancing HK2 gene expression and
the activation of NOD-like receptor thermal protein domain
associated protein 3 inflammatory vesicles (31). Furthermore, in the gastric cancer
TME, hypoxic conditions inhibited M1 by suppressing microRNA
(miR)-30c expression and decreasing mTOR activity and glycolysis
during TAM differentiation and function (32). Conversely, changes in M2 TAM
glycolysis are closely related to their tumor-promoting function.
In pancreatic ductal adenocarcinoma (PDAC), increased aerobic
glycolysis promotes angiogenesis, extravasation and
epithelial-mesenchymal transition, which was further supported by
TAM polarization toward M2, whereas the use of the HK2 inhibitor
2-DG altered TAM glycolytic activity to reverse this function
(33). Thus, HK2 has an important
function in macrophage glycolysis, which also provides a new
experimental basis to target and modulate HK2 to regulate the
polarization state of TAMs and thus treat tumors.
PFK catalyzes the generation of fructose 6-phosphate
to fructose 1,6-bisphosphate during glycolysis.
Fructose-2,6-bisphosphate synthesized by
PFK-2/fructose-2,6-bisphosphatase 3 (PFKFB3) is an allosteric
activator of PFK, and it was found that GM-CSF upregulates
macrophage glycolysis by enhancing PFKFB3 activity and
18F-fluorodeoxyglucose uptake, promoting macrophage M1
polarization (34). After high
glucose stimulation, bone marrow-derived macrophages polarize
toward M1 and secrete inducible nitric oxide synthase (iNOS) and
pro-inflammatory cytokines; however, silencing TGF-β activated
kinase 1 binding protein 1 inhibited macrophage M1 polarization by
affecting HIF-1α-mediated PFKFB3 activity to limit glycolysis in
mice (35). Targeted knockdown of
PFKFB3 to limit glycolysis flux also caused a decrease in NOS2
expression (36). Furthermore,
after macrophage stimulation with TNF-α, two separate inhibitors
were used to affect PFKFB3 activity, resulting in a significant
reduction in glycolysis, along with significant inhibition of
macrophage M1 polarization (22).
By contrast, the anti-inflammatory drug dexmedetomidine affected
HIF-1α binding to GLUT1, HK2 and PFKFB3 by downregulating HIF-1α
expression to inhibit glycolysis and attenuate the LPS-induced
pro-inflammatory response (37).
Taken together, these results suggested that PFKFB3-mediated
glycolysis has a key role in driving the activation of M1
macrophages (38), which may have
implications for cancer therapy by modulating
HIF-1α/PFKFB3-activated M1-type TAMs to influence tumor
progression.
PKM2 is an important pyruvate kinase that comes in
two forms, with the pyruvate lyase activity of the tetrameric form
of PKM2 being higher than that of the dimeric form (39). The dimeric form of PKM2 may
interact with HIF-1α in the nucleus and recruit hypoxia response
elements by enhancing the binding of HIF-1α and p300, thereby
promoting HIF-1α target gene activation, as well as macrophage M1
polarization (40). The use of
small molecular activators DASA-58 and TEPP-46 to promote PKM2
tetramerization during LPS activation in mouse bone marrow-derived
macrophages impaired the binding of PKM2 to HIF-1α and adversely
affected macrophage M1 polarization (21). Furthermore, PKM2 serves as a
physiological substrate for recombinant Sirtuin 5 (SIRT5) and the
hyper-glycosylation mediated by SIRT5 deficiency may promote M1
polarization by promoting the conversion of PKM2 tetramers to
dimers (41). Thus, PKM2-mediated
glycolysis influences macrophage polarization toward the M1 type,
and also has implications for targeting PKM2 structures in cancer
therapy to regulate M1 TAMs. Furthermore, in the TME, TAMs are
enriched in hypoxic regions and exhibit higher rates of glycolysis,
and secrete immunosuppressive cytokines, while also upregulating
growth factors, including vascular endothelial growth factor (VEGF)
and platelet-derived growth factor (PDGF), to induce angiogenesis
to remodel and maintain tumor growth (42,43);
furthermore, the key macrophage glycolytic enzyme PKM2 co-localized
with F-actin in filopodia (44).
Therefore, it may be speculated that glycolysis of TAMs has an
important role in their migration to hypoxic regions.
In summary, altering the activity of key glycolysis
enzymes has an impact on TAM polarization and function by mediating
inflammatory vesicle formation, functional inflammatory factor
secretion and immunosuppressive cytokine secretion (Fig. 1).
During metabolic reprogramming, GLUT1 and pyruvate
dehydrogenase kinase 1 (PDK1) in macrophages differentially affect
glucose metabolism levels through the control of glucose flux and
pyruvate, respectively (49,50).
Expression changes of GLUT1, which controls glucose
transport and related enzymes in glycolysis, mediates the metabolic
pathway shift to glycolysis (51).
GLUT1 [also known as solute carrier family 2 member 1 (SLC2A1)] has
a regulatory role in glucose flux and affects the process of
macrophage glycolysis. After stable overexpression of GLUT1 in
RAW264.7 macrophages, cellular bioenergetics analysis, metabolomics
and radioactive tracer results indicated that overexpression of
GLUT1 resulted in elevated glucose uptake and metabolism and
increased levels of intermediates of the pentose phosphate pathway.
Further detection of gene expression revealed elevated secretion of
inflammatory mediators and polarization of macrophages toward the
M1 type (52). Correspondingly,
deletion of SLC2A1 in bone marrow-derived macrophages restricted
glucose uptake, decreased macrophage glycolysis and the pentose
phosphate pathway, and caused macrophage polarization toward the M2
type (53). HIF-1 is a
heterodimeric transcription factor complex that includes two
subunits: HIF-1α, which is responsive to O2, and the
constitutively expressed HIF-1β (26). HIF-1α induces the binding of GLUT1,
which controls glucose transport, and related genes during
glycolysis by forming a dimer with HIF-1β and the intranuclear
hypoxia response element on the target gene (54). In aging skeletal muscle, HIF-1α
downregulation inhibits its downstream GLUT1, affecting macrophage
glycolysis, thereby inhibiting its M1 polarization and phagocytosis
(55). In addition, the drug
silver forgesine degrades the proteasome of HIF-1α by affecting the
expression of GLUT1, PKM, PDK1, lactate dehydrogenase (LDH)A and
PFK, which further inhibits macrophage glycolysis, thereby limiting
their polarization to the M1 type (56). By contrast, monosodium urate and
calcium pyrophosphate crystals mediate plasma membrane GLUT1
expression to promote macrophage glucose uptake and mediate
metabolic reprogramming of aerobic glycolytic pathways to promote
macrophage polarization toward the M1 type (57). However, it has also been suggested
that, although the lack of GLUT1 attenuates glycolysis and the
pentose phosphate pathway, macrophages are metabolically flexible
enough, such that a lack of GLUT1 does not severely affect their
activation status and function (54).
PDK1, a key regulatory enzyme in glucose metabolism,
regulates the conversion of pyruvate to acetyl CoA during its entry
into the TCA cycle. It was reported that the glycolysis activity
induced by LPS activation of bone marrow-derived macrophages in
mice was diminished due to PDK1 deficiency, with a corresponding
increase in mitochondrial oxidative respiratory activity, which
caused a conversion of macrophages from the M1 to the M2 type
(20). Under hypoxia, HIF-1α
enhances RAW 264.7 cell glycolysis and inhibits mitochondrial
respiration by inducing PDK1-mediated metabolic reprogramming,
preventing pyruvate from entering the TCA and converting it to
lactate, which ultimately promotes M1 polarization (44). Of note, the detection of
macrophages in mouse mammary tumor tissues by flow cytometry
revealed that PDK1 deletion significantly inhibited the
phosphorylation of protein kinase B (AKT) T308 and S6 in
macrophages and suppressed the activation of AKT/mTOR signaling in
TAMs. Further detection of related gene transcript levels revealed
that PDK1 promoted the differentiation of M2-type macrophages
(58).
Taken together, it is clear that metabolic
reprogramming mediated by alterations in glycolysis-related enzymes
GLUT1 and PDK1 has important implications for the polarization and
function of TAMs.
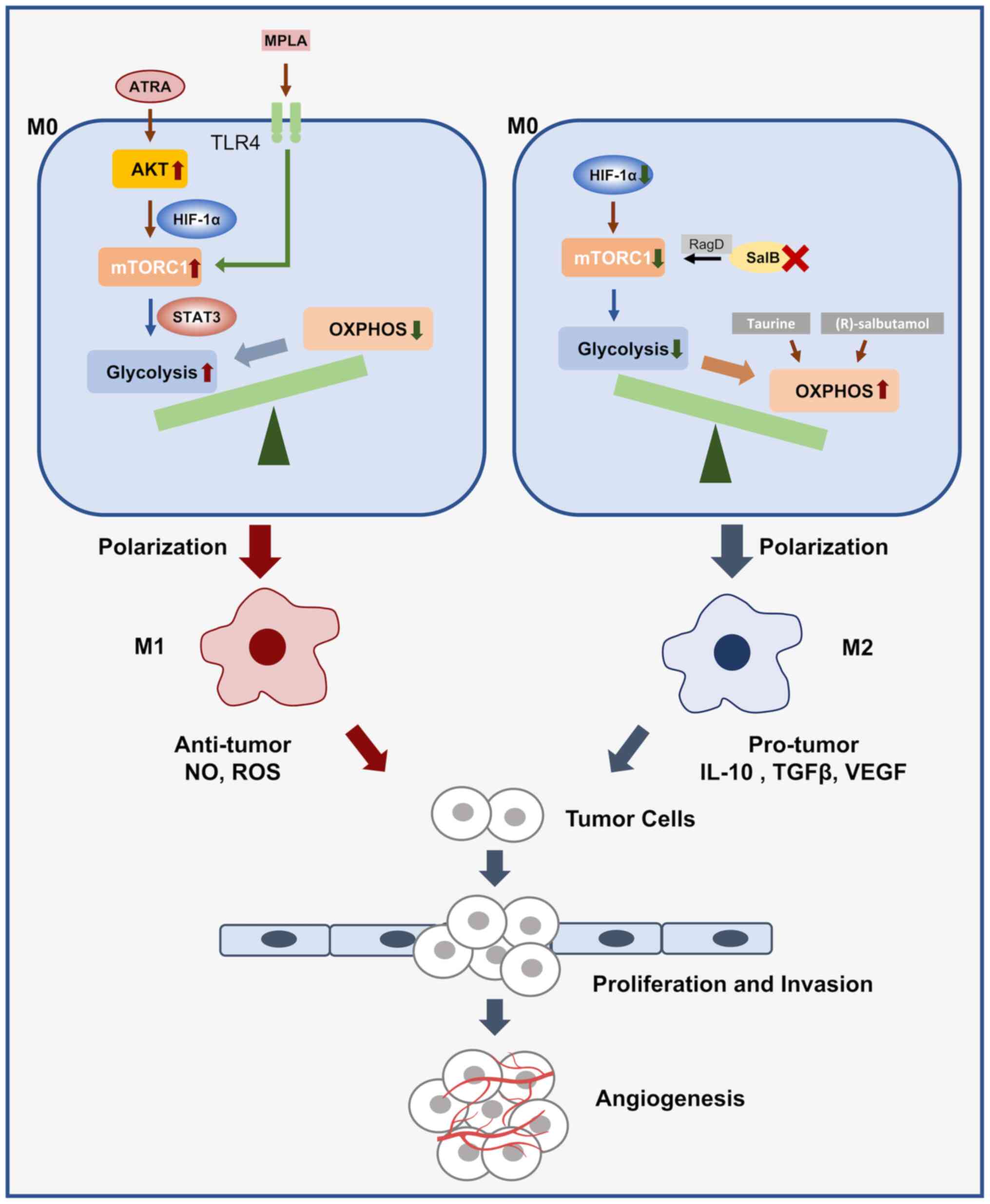
During macrophage activation, LPS signaling mainly
mediates protein Akt/mTOR enhancement of glucose uptake and
promotes IL-10 and NO production, while NO inversely mediates
OXPHOS and promotes a shift in metabolic direction toward
glycolysis (59). In bone
marrow-derived macrophages, SIRT3 deficiency promotes the
macrophage M1 phenotype by shifting metabolism from OXPHOS to
glycolysis (60). Fatty acid
transport protein 1 affects macrophage metabolic reprogramming by
controlling the activation of substrate metabolism, and its
deletion enhances glycolysis (61). Recently, it was demonstrated that
an iron-based metal-organic framework nanoparticle and an iron
induce stimulated phagocytosis of tumor cells by macrophages
through synergistic induction of mitochondrial alterations in TAMs,
leading to a switch in their metabolic mode from mitochondrial
OXPHOS to glycolysis, inducing TAMs to undergo M1 polarization
(62). On the one hand, a
clinically used Toll-like receptor 4 agonist, monophosphatidyl
lipid A, facilitated the transition from OXPHOS to glycolysis by
activating mTOR signaling (63);
on the other hand, Metformin (Met) shifted the state of TAMs to the
M1 type by targeting and inducing a decrease in OXPHOS while
increasing glycolysis (64). A
Pseudomonas aeruginosa protein, PcrV, increased glycolytic
activity and promoted the conversion of TAMs to the M1 type by
activating the PI3K/AKT/mTOR signaling pathway, and the resulting
increase in nitric oxide-related cytotoxicity induced Lewis lung
carcinoma cell apoptosis (65).
Furthermore, the anti-malarial drug chloroquine promotes the
reprogramming of TAM metabolism from OXPHOS to glycolysis by
increasing the lysosomal pH of macrophages, releasing
Ca2+ through the lysosomal Ca2+ channel
mucus-1, inducing the activation of p38 and NF-κB, and activating
transcription factor EB (TFEB), which in turn polarizes TAMs from
the M2 to the anti-tumor M1 phenotype (66). In addition, K+ in tumors
inhibits the anti-tumor ability of TAMs. Among them, Kir2.1 has an
important molecule in ion balance and its deletion causes the
metabolism of TAMs to shift from oxidative phosphorylation to
glycolysis, leading to the reactivation of TAMs' immune function,
which is not conducive to tumor growth. Kir2.1 is an important and
potential therapeutic target for restoring the anti-tumor ability
of TAMs (67). Research has
indicated that a SnSe nanosystem modeled using LDH may achieve M1
macrophage activation and restore its anti-tumor function by
altering the tumor microenvironment and reprogramming the metabolic
mode of TAMs from OXPHOS to glycolysis (68). By contrast, a novel supramolecular
nanotherapeutic reprograms TAMs from the M2 type to the M1 type by
inhibiting the TCA cycle and upregulating glycolytic metabolism,
while significantly affecting phagocytic function (69).
In contrast, the glycolysis inhibitor 2-DG induced a
shift in macrophage energy metabolism from glycolysis to OXPHOS by
upregulating p-AMPKα levels and inhibiting NF-κB activation
(70,71). Upregulation of arginase 1 (ARG1)
expression levels with a corresponding downregulation of iNOS
expression was reported, which promoted the bioenergy of
macrophages and suggested conversion of macrophages to the M2 type.
Metabolomic assays suggested an increase in glycolysis and pentose
phosphate pathway metabolites, such as those of lactate,
glyceraldehyde 3-phosphate, glycerol-3-phosphate biosynthesis,
3-phosphoglycerate, 2,3-diphosphoglycerate, fructose
1,6-bisphosphate, glucose-6-phosphate, fructose-6-phosphate and
phosphoenolpyruvate during macrophage activation; and a
corresponding decrease in mitochondrial oxidation products, such as
fumarate, succinate, citrate and isocitrate (72). Furthermore, downregulation of
HIF-1α inhibited glycolysis after overexpression of miR-223 in
RAW264.7 cells, enhanced mitochondrial respiration and promoted M2
polarization (73). In addition,
downregulation of glycolysis mediated by both mTOR and HIF-1α
attenuated IgG immune complex-induced M1 macrophage activation
in vitro (74). This
suggested that HIF-1α may affect its own polarization and function
by regulating macrophage metabolic reprogramming and stimulating
the shift from OXPHOS to glycolysis. Of note, the β2 receptor
agonist (R)-salbutamol inhibited macrophage M1 polarization by
reducing aerobic glycolysis and enhancing mitochondrial respiration
(75). By contrast, GM-CSF, an
upstream activator of mTORC2 in the pathway involving PI3K and AKT,
promotes M2 polarization by reducing glycolysis and increasing
fatty acid oxidation and OXPHOS (11). Furthermore, under IL-4 activation,
the IL-33/ST2 axis regulates mitochondrial phagocytosis levels by
affecting mTOR activity, causing a metabolic switch from OXPHOS to
glycolysis in TAMs, further increasing the expression of M2
polarization-related genes and ultimately promoting tumor growth
(76). Changes in the external
environment also alter the polarization state and functions of TAMs
by affecting their metabolic balance; it has been indicated that
gut microbiota metabolites short chain fatty acids induce a shift
in intestinal macrophage metabolism from glycolysis to OXPHOS, and
further use of antibiotics upregulates the expression of genes
involved in glycolysis, but not by inducing phosphorylation of the
mTOR signaling pathway. In turn, enhanced metabolic functions of
colonic macrophages include increased extracellular acidification
rate and oxygen consumption rate (77). In addition, studies have indicated
that after near-infra red irradiation of THP-1 cells, the activity
of citrate synthase, a key enzyme in the tricarboxylic acid cycle,
was obviously upregulated due to H3K4 trimethylation at the
promoter region (78), which would
cause metabolic reprogramming of macrophages, metabolic shift from
glycolysis to TCA and OXPHOS, and then cause macrophage to M2 type
polarization (79). Furthermore,
deletion of the glucose-6-phosphate transporter gene inhibited
glycolysis and increased mitochondrial OXPHOS, and is a cause of M1
macrophage suppression (80).
However, data from a proteomic analysis indicated
that the expression of TAM glycolysis-related genes, such as that
encoding HK2, and those encoding the downstream proteins
phosphofructokinase-1 liver type and α-enolase, were significantly
upregulated during breast cancer induction (81). Furthermore, the use of 2-DG
significantly inhibited the expression of the M2 marker IL-10 in
TAMs induced by hepatocellular carcinoma (HCC) regulatory mediators
under normoxic conditions (82).
This suggested that glycolysis in TAMs is not exclusively triggered
by hypoxic stress stimuli and that this reprogramming of glycolytic
metabolism may have an important role in the differentiation of
TAMs, while signaling molecules from tumor cells may promote TAM
glycolysis and maintain their M2 phenotype under normoxic
conditions. Transcriptomic and metabolic analyses revealed that in
mouse and human lung tumors, TAMs gradually exhibit higher
oxidative metabolism and glycolysis, while lactate generated by
glycolysis serves as an additional carbon source to support their
oxidative metabolism, causing an upregulation of ARG1 expression,
and suggesting a gradual polarization of M1 macrophages to the M2
type (83). Even MV3 human
melanoma cell-stimulated generation of M2 TAMs requires only
glycolysis, without the participation of the pentose phosphate
pathway or fatty acid oxidation (13), whereas activation of the
Wnt2b/β-catenin/c-Myc signaling pathway enhances the expression of
key glycolytic enzymes, including HK2, PKM2, LDHA and LDHB in
HCC-derived TAMs and promotes their M2 polarization (82). IL-13 extracted from the gastric
cancer cell line MKN45 not only induced elevated M2-type markers
CD163, IL-4 and IL-13 in macrophages, but also activated the
expression of glycolysis-related enzymes, including GLUT3,
glycosylphosphatidylinositol, phosphoglycerate kinase 1, LDHA,
PFKFB3 and HK2, promoting upregulation of glycolytic activity,
while assays indicated that this change was associated with TAM
amino acid metabolism and fatty acid metabolism independently
(84).
In summary, the altered metabolic balance between
glycolysis and OXPHOS affects the polarization and function of TAMs
(Fig. 2).
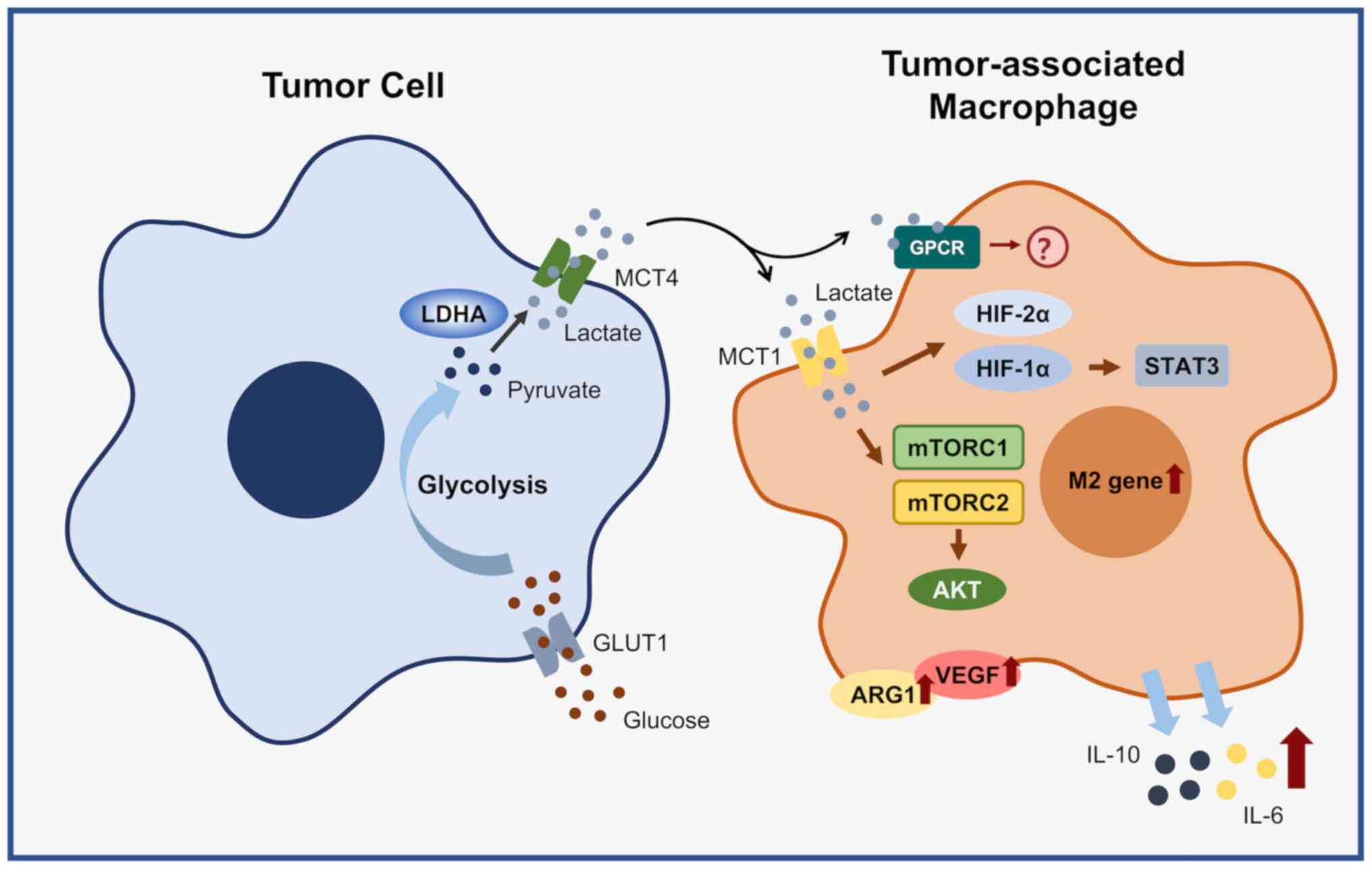
Unlike normal cells, tumor cells convert glucose
into lactic acid through aerobic glycolysis to maintain growth and
biosynthesis; this process is known as the Warburg effect. First,
with the participation of GLUT1, glucose is transported into the
cell, where pyruvate is generated under the action of key
glycolytic enzymes, including HK, PKF and PKM. Further, lactate is
generated under the catalysis of LDHA and transported out of the
cell by monocarboxylic acid transporter 4 (MCT4) and then enters
the tumor microenvironment (77).
In the Warburg effect, tumor cells release large amounts of lactate
via glycolysis, causing metabolic reprogramming of stromal cells,
including cancer-associated fibroblasts (CAFs) and TAMs (85), which undergo aerobic glycolysis and
produce metabolites to promote tumor progression, while impeding
the function of immune cells in the TME and promoting immune escape
of tumors (86). Tumor-derived
lactate induces glycolysis signaling of TAMs to further polarize
them toward the M2 type to promote tumor immune escape; however,
TAM-derived lactate promotes tumor progression while supporting
tumor energy metabolism as a signaling molecule and fuel for the
TCA cycle (87-90). Thus, the lactate-mediated
interaction between tumor cells and TAMs is a reciprocal process.
In this process, tumor-derived lactate mediates polarization and
functional changes in TAMs through two signaling pathways, the
MCT-mediated signaling pathway and the G protein-coupled
receptor-mediated signaling pathway. Among them, MCT1 deletion
blocks M2 polarization (91).
Numerous studies have indicated that in the TME, glycolysis of TAMs
and activation of HIF-1α promote each other, and together they
induce the M2 phenotype of TAMs. Lactate, produced by tumor cell
glycolysis, has a key signaling function by mediating the induction
of ARG1 and VEGF expression in TAMs by HIF-1α and promoting M2
polarization of TAMs, which is independent of the acidic
microenvironment (92,93). Furthermore, in gastric cancer,
tumor-derived lactate is able to promote TAM polarization toward
the M2-type by activating the MCT-HIF-1α pathway (94) and increasing their infiltration
(95). miR-3679-5p, delivered to
lung cancer cells via M2 macrophage exosomes, downregulates E3
ligase neural precursor cell-expressed developmentally
down-regulated 4-like expression, thereby stabilizing tumor cell
c-Myc and leading to elevated glycolysis activity, and the
resulting high lactate production induces M2 to M1 conversion of
macrophages via HIF-1α (96).
Furthermore, lactate activates MCT-mediated mTORC1, leading to
phosphorylation and inactivation of the downstream TFEB, which
further inhibits the expression of ATP6V0d2; however, blockade of
ATP6V0d2 in macrophages leads to activation of HIF-2α, which in
turn promotes M2 polarization and VEGF expression, further
contributing to their angiogenic function (97). In contrast, in breast cancer, zinc
finger E-box binding homeobox 1 induces glycolytic activity in
tumor cells through the PI3K/Akt/HIF-1α signaling axis and further
lactate production stimulates the protein kinase A/cAMP-responsive
element binding protein signaling pathway in TAMs to induce their
differentiation toward the M2 phenotype (98). Furthermore, in pituitary adenomas,
lactate further promotes tumor invasion by activating mTORC2/Akt
signaling and promoting the M2 polarization of TAMs (99). In head and neck squamous cell
carcinoma, high levels of lactate stimulate the expression of
CD163, a marker of M2 macrophages. Of note, the lactate
concentration in the TME was negatively correlated with CD68
expression, but positively with CD163 expression, implying that
lactate decreases the migratory capacity of macrophages (99). A study on pancreatic ductal
adenocarcinoma found that vascular cellular adhesion molecule-1
(VCAM-1) mediates a shift in tumor cell metabolism to aerobic
glycolysis, which increases lactate production and thus promotes M2
polarization of macrophages, while TAM C-C motif secretion of
chemokine ligand 18 (CCL18) mediates the upregulation of VCAM-1 in
PDAC and promotes the malignant progression of tumor cell migration
and invasion in vitro (100).
In addition, lactate may affect TAM-related
functions by mediating HIF-1α, including involvement in tumor
immunosuppression, drug resistance and angiogenesis. A study of
cervical cancer indicated that lactate secreted via aerobic
glycolysis of tumor cells upregulated HIF-1α expression, inhibited
NF-κB activation and further promoted IL-1β, IL-10 and IL-6
secretion (101). In addition,
glioma-derived exosome miR-1246 then activates the STAT3 signaling
pathway and inhibits NF-κB signaling pathway-induced M2 macrophage
polarization by targeting telomeric repeat-binding factor 2
interacting protein, thus promoting the formation of an
immunosuppressive microenvironment (102). Meanwhile, in tamoxifen-resistant
estrogen receptor-positive breast cancer, tumor cells highly
express a sodium/glucose cotransporter (SGLT1) to promote
glycolysis, releasing large amounts of lactic acid into the TME. In
turn, these lactates increase epidermal growth factor secretion by
activating HIF-1α/STAT3 signaling in TAMs, which in turn promotes
SGLT1 expression in tumor cells. This process creates a positive
feedback loop between tumor cells and TAMs, a cycle that promotes
tamoxifen resistance (103).
Furthermore, lactate activation of human macrophages to the TAM
phenotype simultaneously affects Notch signaling in macrophages to
stimulate CCL5 secretion. In turn, CCL5 promotes aerobic glycolysis
in breast cancer cells via AMPK signaling, thus forming a metabolic
feedback loop (104). The above
studies suggested that lactate induces M2 polarization and enhances
the tumor-promoting function of TAMs through the MCT1/HIF-1α
signaling pathway.
Therefore, certain studies have also focused on
immunotherapy induced by amplified immunogenic cell death (ICD). It
was demonstrated that the use of a regulator, panobinostat, which
induces histone acetylation in tumor cells, effectively inhibited
the glycolysis of tumor cells, resulting in a decrease in lactate,
ultimately causing the transformation of TAMs into anti-tumor M1
types (105). In addition, the
biomimetic nanosystem designed by Wang et al (106) effectively inhibits tumor growth
by consuming lactate in the TME, finally causing TAMs to polarize
from M2 to M1. Furthermore, a nanoscale mutagen sensitive to
reactive oxygen species (ROS) initiates tumor-specific effector
T-cell infiltration by inducing ICD depletion of TAMs, thereby
activating an anti-tumor immune response, which suggests the
feasibility of immunotherapy methods that mediate tumor cell
glucose metabolism and lactic acid-induced ICD (107). In addition, the use of mannose
may have a synergistic inhibitory effect on glycolysis in TAMs and
cancer cells, leading to cell apoptosis and inducing systemic
anti-tumor immune responses (108). The synergistic action with
radiotherapy may effectively inhibit tumors and their
metastasis.
In summary, altered levels of tumor cell glycolysis
affect the polarization and function of TAMs (Fig. 3); it also provides a new
perspective for indirect immunotherapy targeting glycolysis and
lactate production in tumor cells.
CAFs are the most abundant stromal cells in the TME,
and in addition to their important role in tumorigenesis and
development, CAFs regulate the TME (109). CAFs secrete IL-6, M-CSF, monocyte
chemoattractant protein-1 and stromal cell-derived factor-1, which
promote macrophage infiltration and differentiation (110). Furthermore, unlike normal
breast-derived fibroblasts, CAFs isolated from human breast cancer
may promote the differentiation of monocytes into M2 macrophages
(111). This differentiation is
evident in terms of functional and phenotypic characteristics of
pulmonary myofibroblasts after metabolic reprogramming-mediated
upregulation of glycolysis to produce lactate, which indirectly
regulates macrophage polarization (112). In addition, pulmonary
myofibroblasts were induced by TGF-β1, glycolysis was upregulated,
lactate was significantly increased and the expression of
fibrogenic mediators in macrophages was promoted. Lactate also
induced histone emulsification of the profibrotic gene (ARG1 and
PDGFA) promoter in macrophages, triggering M1 polarization
(112). In addition, CAFs
exhibited greater glucose uptake, lactate production and elevated
expression of LDHA, PKM2 and miR-21 compared with normal
fibroblast. miR-21 inhibition decreased the degree of glycolysis in
CAFs. The OXPHOS and invasive capacity of pancreatic cancer cells
were decreased after co-culture with miR-21 inhibitor-CAFs
(113), which provided new
evidence for possible crosstalk between CAFs and TAMs.
Lymphocytes generally include T lymphocytes, B
lymphocytes and natural killer cells. Based on their surface CD
molecules, T lymphocytes may be divided into various
subpopulations. It has been indicated that Met promotes ROS
production and increases glycolysis in the mitochondria of
tumor-infiltrating CD8+T lymphocytes (CD8TIL) by
activating ROS, which in turn leads to IFN-γ secretion by promoting
CD8TIL proliferation (114),
while CD36 deficiency impairs oxidized low-density
lipoprotein-stimulated monocyte-derived macrophage NF-κB
production, thereby downregulating macrophage expression of
pro-inflammatory factors IL-1 receptor antagonist, IL-1β, IL-6,
TNF-α and IFN-β (115). When
propranolol, a pan-β receptor blocker, or mice deficient in
β2-adrenoceptors (β2-AR) were used, blockade of β-AR signaling
increased glycolysis and OXPHOS in tumor-infiltrating lymphocytes,
resulting in increased expression of the effector molecules IFN-γ,
granzyme B and IL-12A and associated pro-inflammatory cytokines
(IL-1B, IL-4, IL-6 and IL-10) expression was reduced (116). Although there are relatively few
studies on the regulation of macrophage polarization and function
via lymphocyte glycolysis, they offer certain possibilities to
influence macrophage-related functions by altering the lymphocyte
glycolytic regulation of the TME.
In adipose tissue, macrophage exposure to the
saturated fatty acid palmitate triggers an upregulation of HIF-1α
that increases glycolysis and ultimately leads to IL-1β production
(117). Death of pseudohypoxic
adipocytes initiates macrophage pro-inflammatory translation, and
atherosclerotic injury associated with abnormal lipid metabolism
also shifts macrophages from the M2 to the M1 type (118). These studies suggested that when
macrophages are in adipose tissue, the hypoxic environment induces
macrophage glycolysis and polarization toward the M1 type, which
also provides ideas for inflammation therapy (117,118).
Bone MDSCs accumulate in tumors and peripheral
lymphoid organs and are divided into neutrophils polymorphonuclear
MDSCs and monocyte MDSCs (M-MDSCs), which have a role in the TME
and TAMs. The difference is caused by the CD36-mediated dependence
of tumor-associated MDSCs on fatty acid oxidation as their main
energy source. Under hypoxic conditions, activation of HIF-1α
induced a shift in MDSC metabolic mode from OXPHOS to glycolysis,
while HIF-1α promoted the differentiation of M-MDSCs toward TAMs by
downregulating CD45 and STAT3 activity (119). In addition, bone marrow
MSC-derived exosomes mediate glycolysis by inhibiting HIF-1α,
downregulating the expression of glycolytic essential proteins and
preventing LPS stimulation of M1 polarization-induced inflammation
(120).
Altered key glycolysis enzyme activity in TAMs
affects their own polarization and function. Furthermore, the
effects of metabolic reprogramming on the polarization and function
of TAMs have been intensively studied. In addition, altered levels
of glycolysis in tumor cells and other immune cells in the TME have
an important role in TAM polarization and function. Although the
effect of changes in glycolysis activity mediated by altered
activities of key enzymes in TAMs on their polarization status and
function has been investigated, whether kinase changes directly
affect the polarization and function of TAMs, as well as the
underlying mechanisms, remain to be determined. Furthermore,
studies have indicated that the metabolic homeostasis between
glycolysis and OXPHOS has an important role in the polarization and
function of TAMs, providing theoretical support for the search for
targeted therapeutic pathways. In the TME, lactate, a glycolysis
metabolite of tumor cells, induces the development of a pro-tumor
phenotype in TAMs, which also suggests that other metabolites of
tumor cells, including succinate, may have a regulatory role in TAM
polarization and function. These observations prompted us to
hypothesize that there is a competitive relationship between tumor
cells and TAMs for the uptake of glucose in the microenvironment.
Therefore, the study of the mechanism of the interaction between
glycolysis and tumor-associated macrophage polarization and
function may further clarify their intrinsic connection and provide
new perspectives for targeted therapy.
Not applicable.
JC wrote the manuscript and drew the figures. FZ and
SL performed the literature search and contributed to manuscript
revision. LC and YZ designed the study and revised the
manuscript.
Not applicable.
Not applicable.
The authors have no competing interests to
declare.
Not applicable.
The present study was supported by the National Natural Sciences
Foundation of China (grant no. 82273219) and the Hunan Provincial
Natural Science Foundation (grant nos. 2021JJ30915, 2022JJ30328 and
2021JJ70099).
|
1
|
Tang BL: Glucose, glycolysis, and
neurodegenerative diseases. J Cell Physiol. 235:7653–7662.
2020.
|
|
2
|
Fernie AR, Carrari F and Sweetlove LJ:
Respiratory metabolism: Glycolysis, the TCA cycle and mitochondrial
electron transport. Curr Opin Plant Biol. 7:254–261. 2004.
|
|
3
|
Hui S, Ghergurovich JM, Morscher RJ, Jang
C, Teng X, Lu W, Esparza LA, Reya T, Le Z, Yanxiang Guo J, et al:
Glucose feeds the TCA cycle via circulating lactate. Nature.
551:115–118. 2017.
|
|
4
|
Gerich JE, Meyer C, Woerle HJ and Stumvoll
M: Renal gluconeogenesis: Its importance in human glucose
homeostasis. Diabetes Care. 24:382–391. 2001.
|
|
5
|
Weinhouse S: Oxidative metabolism of
neoplastic tissues. Adv Cancer Res. 3:269–325. 1955.
|
|
6
|
Jin L and Zhou Y: Crucial role of the
pentose phosphate pathway in malignant tumors. Oncol Lett.
17:4213–4221. 2019.
|
|
7
|
Chen F, Zhuang X, Lin L, Yu P, Wang Y, Shi
Y, Hu G and Sun Y: New horizons in tumor microenvironment biology:
Challenges and opportunities. BMC Med. 13:452015.
|
|
8
|
Pereira M, Chen TD, Buang N, Olona A, Ko
JH, Prendecki M, Costa ASH, Nikitopoulou E, Tronci L, Pusey CD, et
al: Acute iron deprivation reprograms human macrophage metabolism
and reduces inflammation in vivo. Cell Rep. 28:498–511.e5.
2019.
|
|
9
|
Wang F, Zhang S, Vuckovic I, Jeon R,
Lerman A, Folmes CD, Dzeja PP and Herrmann J: Glycolytic
stimulation is not a requirement for M2 macrophage differentiation.
Cell Metab. 28:463–475.e4. 2018.
|
|
10
|
Reinfeld BI, Madden MZ, Wolf MM, Chytil A,
Bader JE, Patterson AR, Sugiura A, Cohen AS, Ali A, Do BT, et al:
Cell-programmed nutrient partitioning in the tumour
microenvironment. Nature. 593:282–288. 2021.
|
|
11
|
Huang SC, Smith AM, Everts B, Colonna M,
Pearce EL, Schilling JD and Pearce EJ: Metabolic reprogramming
mediated by the mTORC2-IRF4 signaling axis is essential for
macrophage alternative activation. Immunity. 45:817–830. 2016.
|
|
12
|
Shi Q, Shen Q, Liu Y, Shi Y, Huang W, Wang
X, Li Z, Chai Y, Wang H, Hu X, et al: Increased glucose metabolism
in TAMs fuels O-GlcNAcylation of lysosomal Cathepsin B to promote
cancer metastasis and chemoresistance. Cancer Cell.
40:1207–1222.e10. 2022.
|
|
13
|
M de-Brito N, Duncan-Moretti J, C da-Costa
H, Saldanha-Gama R, Paula-Neto HA, G Dorighello G, L Simões R and
Barja-Fidalgo C: Aerobic glycolysis is a metabolic requirement to
maintain the M2-like polarization of tumor-associated macrophages.
Biochim Biophys Acta Mol Cell Res. 1867:1186042020.
|
|
14
|
Saha S, Shalova IN and Biswas SK:
Metabolic regulation of macrophage phenotype and function. Immunol
Rev. 280:102–111. 2017.
|
|
15
|
Lin EY, Gouon-Evans V, Nguyen AV and
Pollard JW: The macrophage growth factor CSF-1 in mammary gland
development and tumor progression. J Mammary Gland Biol Neoplasia.
7:147–162. 2002.
|
|
16
|
Gocheva V, Wang HW, Gadea BB, Shree T,
Hunter KE, Garfall AL, Berman T and Joyce JA: IL-4 induces
cathepsin protease activity in tumor-associated macrophages to
promote cancer growth and invasion. Genes Dev. 24:241–255.
2010.
|
|
17
|
Li W, Li Y, Jin X, Liao Q, Chen Z, Peng H
and Zhou Y: CD38: A Significant Regulator of macrophage Function.
Front Oncol. 12:7756492022.
|
|
18
|
Wynn TA, Chawla A and Pollard JW:
Macrophage biology in development, homeostasis and disease. Nature.
496:445–455. 2013.
|
|
19
|
Haschemi A, Kosma P, Gille L, Evans CR,
Burant CF, Starkl P, Knapp B, Haas R, Schmid JA, Jandl C, et al:
The sedoheptulose kinase CARKL directs macrophage polarization
through control of glucose metabolism. Cell Metab. 15:813–826.
2012.
|
|
20
|
Tan Z, Xie N, Cui H, Moellering DR,
Abraham E, Thannickal VJ and Liu G: Pyruvate dehydrogenase kinase 1
participates in macrophage polarization via regulating glucose
metabolism. J Immunol. 194:6082–6089. 2015.
|
|
21
|
Palsson-McDermott EM, Curtis AM, Goel G,
Lauterbach MA, Sheedy FJ, Gleeson LE, van den Bosch MW, Quinn SR,
Domingo-Fernandez R, Johnston DG, et al: Pyruvate kinase M2
regulates Hif-1α activity and IL-1β induction and is a critical
determinant of the Warburg effect in LPS-activated macrophages.
Cell Metab. 21:65–80. 2015.
|
|
22
|
Tawakol A, Singh P, Mojena M,
Pimentel-Santillana M, Emami H, MacNabb M, Rudd JH, Narula J,
Enriquez JA, Través PG, et al: HIF-1α and PFKFB3 mediate a tight
relationship between proinflammatory activation and anerobic
metabolism in atherosclerotic macrophages. Arterioscler Thromb Vasc
Biol. 35:1463–1471. 2015.
|
|
23
|
Ruiz-Ga rcía A, Monsalve E, Novellasdemunt
L, Navarro-Sabaté A, Manzano A, Rivero S, Castrillo A, Casado M,
Laborda J, Bartrons R and Díaz-Guerra MJ: Cooperation of adenosine
with macrophage Toll-4 receptor agonists leads to increased
glycolytic flux through the enhanced expression of PFKFB3 gene. J
Biol Chem. 286:19247–19258. 2011.
|
|
24
|
Bell GI, Burant CF, Takeda J and Gould GW:
Structure and function of mammalian facilitative sugar
transporters. J Biol Chem. 268:19161–19164. 1993.
|
|
25
|
Middleton RJ: Hexokinases and
glucokinases. Biochem Soc Trans. 18:180–183. 1990.
|
|
26
|
Wang GL, Jiang BH, Rue EA and Semenza GL:
Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS
heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci
USA. 92:5510–5514. 1995.
|
|
27
|
Moon JS, Hisata S, Park MA, DeNicola GM,
Ryter SW, Nakahira K and Choi AMK: mTORC1-Induced HK1-dependent
glycolysis regulates NLRP3 inflammasome activation. Cell Rep.
12:102–115. 2015.
|
|
28
|
Zafar A, Ng HP, Kim GD, Chan ER and
Mahabeleshwar GH: BHLHE40 promotes macrophage pro-inflammatory gene
expression and functions. FASEB J. 35:e219402021.
|
|
29
|
Kim MJ, Lee CH, Lee Y, Youn H, Kang KW,
Kwon J, Alavi A, Carlin S, Cheon GJ and Chung JK:
Glucose-6-phosphatase expression-mediated [18F]FDG
efflux in murine inflammation and cancer models. Mol Imaging Biol.
21:917–925. 2019.
|
|
30
|
Yu Q, Wang Y, Dong L, He Y, Liu R, Yang Q,
Cao Y, Wang Y, Jia A, Bi Y and Liu G: Regulations of glycolytic
activities on macrophages functions in tumor and infectious
inflammation. Front Cell Infect Microbiol. 10:2872020.
|
|
31
|
Alatshan A, Kovács GE, Aladdin A,
Czimmerer Z, Tar K and Benkő S: All-trans retinoic acid enhances
both the signaling for priming and the glycolysis for activation of
NLRP3 inflammasome in human macrophage. Cells. 9:15912020.
|
|
32
|
Wenes M, Shang M, Di Matteo M, Goveia J,
Martín-Pérez R, Serneels J, Prenen H, Ghesquière B, Carmeliet P and
Mazzone M: Macrophage metabolism controls tumor blood vessel
morphogenesis and metastasis. Cell Metab. 24:701–715. 2016.
|
|
33
|
Penny HL, Sieow JL, Adriani G, Yeap WH,
See Chi Ee P, San Luis B, Lee B, Lee T, Mak SY, Ho YS, et al:
Warburg metabolism in tumor-conditioned macrophages promotes
metastasis in human pancreatic ductal adenocarcinoma.
Oncoimmunology. 5:e11917312016.
|
|
34
|
Singh P, González-Ramos S, Mojena M,
Rosales-Mendoza CE, Emami H, Swanson J, Morss A, Fayad ZA, Rudd JH,
Gelfand J, et al: GM-CSF enhances macrophage glycolytic activity in
vitro and improves detection of inflammation in vivo. J Nucl Med.
57:1428–1435. 2016.
|
|
35
|
Zeng H, Qi X, Xu X and Wu Y: TAB1
regulates glycolysis and activation of macrophages in diabetic
nephropathy. Inflamm Res. 69:1215–1234. 2020.
|
|
36
|
Xu J, Wang L, Yang Q, Ma Q, Zhou Y, Cai Y,
Mao X, Da Q, Lu T, Su Y, et al: Deficiency of myeloid Pfkfb3
protects mice from lung edema and cardiac dysfunction in
LPS-induced endotoxemia. Front Cardiovasc Med. 8:7458102021.
|
|
37
|
Meng Q, Guo P, Jiang Z, Bo L and Bian J:
Dexmedetomidine inhibits LPS-induced proinflammatory responses via
suppressing HIF1α-dependent glycolysis in macrophages. Aging
(Albany NY). 12:9534–9548. 2020.
|
|
38
|
Poels K, Schnitzler JG, Waissi F, Levels
JHM, Stroes ESG, Daemen M, Lutgens E, Pennekamp AM, De Kleijn DPV,
Seijkens TTP and Kroon J: Inhibition of PFKFB3 hampers the
progression of atherosclerosis and promotes plaque stability. Front
Cell Dev Biol. 8:5816412020.
|
|
39
|
Anastasiou D, Yu Y, Israelsen WJ, Jiang
JK, Boxer MB, Hong BS, Tempel W, Dimov S, Shen M, Jha A, et al:
Pyruvate kinase M2 activators promote tetramer formation and
suppress tumorigenesis. Nat Chem Biol. 8:839–847. 2012.
|
|
40
|
Luo W, Hu H, Chang R, Zhong J, Knabel M,
O'Meally R, Cole RN, Pandey A and Semenza GL: Pyruvate kinase M2 is
a PHD3-stimulated coactivator for hypoxia-inducible factor 1. Cell.
145:732–744. 2011.
|
|
41
|
Wang F, Wang K, Xu W, Zhao S, Ye D, Wang
Y, Xu Y, Zhou L, Chu Y, Zhang C, et al: SIRT5 Desuccinylates and
Activates Pyruvate Kinase M2 to Block Macrophage IL-1β Production
And To Prevent DSS-Induced Colitis In Mice. Cell Rep. 19:2331–2344.
2017.
|
|
42
|
Henze AT and Mazzone M: The impact of
hypoxia on tumor-associated macrophages. J Clin Invest.
126:3672–3679. 2016.
|
|
43
|
Colangelo T, Polcaro G, Muccillo L,
D'Agostino G, Rosato V, Ziccardi P, Lupo A, Mazzoccoli G, Sabatino
L and AColantuoni V: Friend or foe? The tumour microenvironment
dilemma in colorectal cancer. Biochim Biophys Acta Rev Cancer.
1867:1–18. 2017.
|
|
44
|
Semba H, Takeda N, Isagawa T, Sugiura Y,
Honda K, Wake M, Miyazawa H, Yamaguchi Y, Miura M, Jenkins DM, et
al: HIF-1α-PDK1 axis-induced active glycolysis plays an essential
role in macrophage migratory capacity. Nat Commun. 7:116352016.
|
|
45
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011.
|
|
46
|
Levine AJ and Puzio-Kuter AM: The control
of the metabolic switch in cancers by oncogenes and tumor
suppressor genes. Science. 330:1340–1344. 2010.
|
|
47
|
Vander Heiden MG, Cantley LC and Thompson
CB: Understanding the Warburg effect: The metabolic requirements of
cell proliferation. Science. 324:1029–1033. 2009.
|
|
48
|
Li C, Wang Y, Li Y, Yu Q, Jin X, Wang X,
Jia A, Hu Y, Han L, Wang J, et al: HIF1α-dependent glycolysis
promotes macrophage functional activities in protecting against
bacterial and fungal infection. Sci Rep. 8:36032018.
|
|
49
|
Wang L, Pavlou S, Du X, Bhuckory M, Xu H
and Chen M: Glucose transporter 1 critically controls microglial
activation through facilitating glycolysis. Mol Neurodegener.
14:22019.
|
|
50
|
Peng F, Wang JH, Fan WJ, Meng YT, Li MM,
Li TT, Cui B, Wang HF, Zhao Y, An F, et al: Glycolysis gatekeeper
PDK1 reprograms breast cancer stem cells under hypoxia. Oncogene.
37:1062–74. 2018.
|
|
51
|
Talreja J, Talwar H, Bauerfeld C, Grossman
LI, Zhang K, Tranchida P and Samavati L: HIF-1α regulates IL-1β and
IL-17 in sarcoidosis. Elife. 8. pp. e445192019
|
|
52
|
Freemerman AJ, Johnson AR, Sacks GN,
Milner JJ, Kirk EL, Troester MA, Macintyre AN, Goraksha-Hicks P,
Rathmell JC and Makowski L: Metabolic reprogramming of macrophages:
Glucose transporter 1 (GLUT1)-mediated glucose metabolism drives a
proinflammatory phenotype. J Biol Chem. 289:7884–7896. 2014.
|
|
53
|
Freemerman AJ, Zhao L, Pingili AK, Teng B,
Cozzo AJ, Fuller AM, Johnson AR, Milner JJ, Lim MF, Galanko JA, et
al: Myeloid Slc2a1-Deficient murine model revealed macrophage
activation and metabolic phenotype are fueled by GLUT1. J Immunol.
202:1265–1286. 2019.
|
|
54
|
Masoud GN and Li W: HIF-1α pathway: Role,
regulation and intervention for cancer therapy. Acta Pharm Sin B.
5:378–389. 2015.
|
|
55
|
Fix DK, Ekiz HA, Petrocelli JJ, McKenzie
AM, Mahmassani ZS, O'Connell RM and Drummond MJ: Disrupted
macrophage metabolic reprogramming in aged soleus muscle during
early recovery following disuse atrophy. Aging Cell.
20:e134482021.
|
|
56
|
Zhuang H, Lv Q, Zhong C, Cui Y, He L,
Zhang C and Yu J: Tiliroside ameliorates ulcerative colitis by
restoring the M1/M2 macrophage balance via the HIF-1α/glycolysis
pathway. Front Immunol. 12:6494632021.
|
|
57
|
Renaudin F, Orliaguet L, Castelli F,
Fenaille F, Prignon A, Alzaid F, Combes C, Delvaux A, Adimy Y,
Cohen-Solal M, et al: Gout and pseudo-gout-related crystals promote
GLUT1-mediated glycolysis that governs NLRP3 and interleukin-1β
activation on macrophages. Ann Rheum Dis. 79:1506–1514. 2020.
|
|
58
|
He Y, Du J and Dong Z: Myeloid deletion of
phosphoinositide-dependent kinase-1 enhances NK cell-mediated
antitumor immunity by mediating macrophage polarization.
Oncoimmunology. 9:17742812020.
|
|
59
|
Baseler WA, Davies LC, Quigley L, Ridnour
LA, Weiss JM, Hussain SP, Wink DA and McVicar DW: Autocrine IL-10
functions as a rheostat for M1 macrophage glycolytic commitment by
tuning nitric oxide production. Redox Biol. 10:12–23. 2016.
|
|
60
|
Wei T, Gao J, Huang C, Song B, Sun M and
Shen W: SIRT3 (Sirtuin-3) prevents Ang II (Angiotensin II)-Induced
macrophage metabolic switch improving perivascular adipose tissue
function. Arterioscler Thromb Vasc Biol. 41:714–7130. 2021.
|
|
61
|
Johnson AR, Qin Y, Cozzo AJ, Freemerman
AJ, Huang MJ, Zhao L, Sampey BP, Milner JJ, Beck MA, Damania B, et
al: Metabolic reprogramming through fatty acid transport protein 1
(FATP1) regulates macrophage inflammatory potential and adipose
inflammation. Mol Metab. 5:506–526. 2016.
|
|
62
|
Gu Z, Liu T, Liu C, Yang Y, Tang J, Song
H, Wang Y, Yang Y and Yu C: Ferroptosis-Strengthened metabolic and
inflammatory regulation of tumor-associated macrophages provokes
potent tumoricidal activities. Nano Lett. 21:6471–6479. 2021.
|
|
63
|
Blanco-Pérez F, Goretzki A, Wolfheimer S
and Schülke S: The vaccine adjuvant MPLA activates glycolytic
metabolism in mouse mDC by a JNK-dependent activation of
mTOR-signaling. Mol Immunol. 106:159–169. 2019.
|
|
64
|
Uehara T, Eikawa S, Nishida M, Kunisada Y,
Yoshida A, Fujiwara T, Kunisada T, Ozaki T and Udono H: Metformin
induces CD11b+-cell-mediated growth inhibition of an osteosarcoma:
Implications for metabolic reprogramming of myeloid cells and
anti-tumor effects. Int Immunol. 31:187–198. 2019.
|
|
65
|
Yu H, Bai Y, Qiu J, He X, Xiong J, Dai Q,
Wang X, Li Y, Sheng H, Xin R, et al: Pseudomonas aeruginosa PcrV
Enhances the nitric Oxide-Mediated tumoricidal activity of
Tumor-Associated macrophages via a
TLR4/PI3K/AKT/mTOR-Glycolysis-nitric oxide circuit. Front Oncol.
11:7368822021.
|
|
66
|
Chen D, Xie J, Fiskesund R, Dong W, Liang
X, Lv J, Jin X, Liu J, Mo S, Zhang T, et al: Chloroquine modulates
antitumor immune response by resetting tumor-associated macrophages
toward M1 phenotype. Nat Commun. 9:8732018.
|
|
67
|
Chen S, Cui W, Chi Z, Xiao Q, Hu T, Ye Q,
Zhu K, Yu W, Wang Z, Yu C, et al: Tumor-associated macrophages are
shaped by intratumoral high potassium via Kir2.1. Cell Metab.
34:1843–1859.e11. 2022.
|
|
68
|
Ling J, Chang Y, Yuan Z, Chen Q, He L and
Chen T: Designing lactate Dehydrogenase-Mimicking SnSe nanosheets
to reprogram tumor-associated macrophages for potentiation of
photothermal immunotherapy. ACS Appl Mater Interfaces.
14:27651–27665. 2022.
|
|
69
|
Ramesh A, Malik V, Brouillard A and
Kulkarni A: Supramolecular nanotherapeutics enable metabolic
reprogramming of tumor-associated macrophages to inhibit tumor
growth. J Biomed Mater Res A. 110:1448–1459. 2022.
|
|
70
|
Cai W, Cheng J, Zong S, Yu Y, Wang Y, Song
Y, He R, Yuan S, Chen T, Hu M, et al: The glycolysis inhibitor
2-deoxyglucose ameliorates adjuvant-induced arthritis by regulating
macrophage polarization in an AMPK-dependent manner. Mol Immunol.
140:186–195. 2021.
|
|
71
|
Qin Y, Jiang X, Yang Q, Zhao J, Zhou Q and
Zhou Y: The Functions, methods, and mobility of mitochondrial
transfer between cells. Front Oncol. 11:6727812021.
|
|
72
|
Wang T, Liu H, Lian G, Zhang SY, Wang X
and Jiang C: HIF1α-Induced glycolysis metabolism Is essential to
the activation of inflammatory macrophages. Mediators Inflamm.
2017:90293272017.
|
|
73
|
Dang CP and Leelahavanichkul A:
Over-expression of miR-223 induces M2 macrophage through glycolysis
alteration and attenuates LPS-induced sepsis mouse model, the
cell-based therapy in sepsis. PLoS One. 15:e02360382020.
|
|
74
|
Jing C, Castro-Dopico T, Richoz N, Tuong
ZK, Ferdinand JR, Lok LSC, Loudon KW, Banham GD, Mathews RJ, Cader
Z, et al: Macrophage metabolic reprogramming presents a therapeutic
target in lupus nephritis. Proc Natl Acad Sci USA. 117:15160–71.
2020.
|
|
75
|
Wang S, Liu F, Tan KS, Ser HL, Tan LT, Lee
LH and Tan W: Effect of (R)-salbutamol on the switch of phenotype
and metabolic pattern in LPS-induced macrophage cells. J Cell Mol
Med. 24:722–736. 2020.
|
|
76
|
Xu H, Li D, Ma J, Zhao Y, Xu L, Tian R,
Liu Y, Sun L and Su J: The IL-33/ST2 axis affects tumor growth by
regulating mitophagy in macrophages and reprogramming their
polarization. Cancer Biol Med. 18:172–183. 2021.
|
|
77
|
Paul S, Ghosh S and Kumar S: Tumor
glycolysis, an essential sweet tooth of tumor cells. Semin Cancer
Biol. 86:1216–1230. 2022.
|
|
78
|
Liao WT, Hung CH, Liang SS, Yu S, Lu JH,
Lee CH, Chai CY and Yu HS: Anti-Inflammatory effects induced by
near-infrared light irradiation through M2 macrophage polarization.
J Invest Dermatol. 141:2056–2066.e10. 2021.
|
|
79
|
Kelly B and O'Neill LA: Metabolic
reprogramming in macrophages and dendritic cells in innate
immunity. Cell Res. 25:771–784. 2015.
|
|
80
|
Jeon EH, Park TS, Jang Y, Hwang E, Kim SJ,
Song KD, Weinstein DA, Lee YM, Park BC and Jun HS:
Glucose-6-phosphate transporter mediates macrophage proliferation
and functions by regulating glycolysis and mitochondrial
respiration. Biochem Biophys Res Commun. 524:89–95. 2020.
|
|
81
|
Liu D, Chang C, Lu N, Wang X, Lu Q, Ren X,
Ren P, Zhao D, Wang L, Zhu Y, et al: Comprehensive proteomics
analysis reveals metabolic reprogramming of tumor-associated
macrophages stimulated by the tumor microenvironment. J Proteome
Res. 16:288–2897. 2017.
|
|
82
|
Jiang Y, Han Q, Zhao H and Zhang J:
Promotion of epithelial-mesenchymal transformation by
hepatocellular carcinoma-educated macrophages through
Wnt2b/β-catenin/c-Myc signaling and reprogramming glycolysis. J Exp
Clin Cancer Res. 40(13)2021.
|
|
83
|
Geeraerts X, Fernández-Garcia J, Hartmann
FJ, de Goede KE, Martens L, Elkrim Y, Debraekeleer A, Stijlemans B,
Vandekeere A, Rinaldi G, et al: Macrophages are metabolically
heterogeneous within the tumor microenvironment. Cell Rep.
37:1101712021.
|
|
84
|
He Z, Chen D, Wu J, Sui C, Deng X, Zhang
P, Chen Z, Liu D, Yu J, Shi J, et al: Yes associated protein 1
promotes resistance to 5-fluorouracil in gastric cancer by
regulating GLUT3-dependent glycometabolism reprogramming of
tumor-associated macrophages. Arch Biochem Biophys.
702:1088382021.
|
|
85
|
Yan W, Wu X, Zhou W, Fong MY, Cao M, Liu
J, Liu X, Chen CH, Fadare O, Pizzo DP, et al: Cancer-cell-secreted
exosomal miR-105 promotes tumour growth through the MYC-dependent
metabolic reprogramming of stromal cells. Nat Cell Biol.
20:597–609. 2018.
|
|
86
|
Wang JX, Choi SYC, Niu X, Kang N, Xue H,
Killam J and Wang Y: Lactic Acid and an Acidic Tumor
Microenvironment suppress Anticancer Immunity. Int J Mol Sci.
21:83632020.
|
|
87
|
Consiglio CR, Udartseva O, Ramsey KD, Bush
C and Gollnick SO: Enzalutamide, an androgen receptor antagonist,
enhances myeloid Cell-Mediated immune suppression and tumor
progression. Cancer Immunol Res. 8:1215–1227. 2020.
|
|
88
|
Arts RJ, Plantinga TS, Tuit S, Ulas T,
Heinhuis B, Tesselaar M, Sloot Y, Adema GJ, Joosten LA, Smit JW, et
al: Transcriptional and metabolic reprogramming induce an
inflammatory phenotype in non-medullary thyroid carcinoma-induced
macrophages. Oncoimmunology. 5:e12297252016.
|
|
89
|
Manoharan I, Prasad PD, Thangaraju M and
Manicassamy S: Lactate-Dependent regulation of immune responses by
dendritic cells and macrophages. Front Immunol. 12:6911342021.
|
|
90
|
Wu Q, Allouch A, Paoletti A, Leteur C,
Mirjolet C, Martins I, Voisin L, Law F, Dakhli H, Mintet E, et al:
NOX2-dependent ATM kinase activation dictates pro-inflammatory
macrophage phenotype and improves effectiveness to radiation
therapy. Cell Death Differ. 24:1632–1644. 2017.
|
|
91
|
Zhang J, Muri J, Fitzgerald G, Gorski T,
Gianni-Barrera R, Masschelein E, D'Hulst G, Gilardoni P, Turiel G,
Fan Z, et al: Endothelial lactate controls muscle regeneration from
ischemia by Inducing M2-like macrophage polarization. Cell Metab.
31:1136–1153.e7. 2020.
|
|
92
|
Colegio OR, Chu NQ, Szabo AL, Chu T,
Rhebergen AM, Jairam V, Cyrus N, Brokowski CE, Eisenbarth SC,
Phillips GM, et al: Functional polarization of tumour-associated
macrophages by tumour-derived lactic acid. Nature. 513:559–563.
2014.
|
|
93
|
Colgan SP, Furuta GT and Taylor CT:
Hypoxia and innate immunity: Keeping Up with the HIFsters. Annu Rev
Immunol. 38:341–363. 2020.
|
|
94
|
Zhang L and Li S: Lactic acid promotes
macrophage polarization through MCT-HIF1α signaling in gastric
cancer. Exp Cell Res. 388:1118462020.
|
|
95
|
Yao X, He Z, Qin C, Deng X, Bai L, Li G
and Shi J: SLC2A3 promotes macrophage infiltration by glycolysis
reprogramming in gastric cancer. Cancer Cell Int. 20:5032020.
|
|
96
|
Wang H, Wang L, Pan H, Wang Y, Shi M, Yu
H, Wang C, Pan X and Chen Z: Exosomes derived from macrophages
enhance aerobic glycolysis and chemoresistance in lung cancer by
stabilizing c-Myc via the Inhibition of NEDD4L. Front Cell Dev
Biol. 8:6206032020.
|
|
97
|
Liu N, Luo J, Kuang D, Xu S, Duan Y, Xia
Y, Wei Z, Xie X, Yin B, Chen F, et al: Lactate inhibits ATP6V0d2
expression in tumor-associated macrophages to promote
HIF-2α-mediated tumor progression. J Clin Invest. 129:631–646.
2019.
|
|
98
|
Jiang H, Wei H, Wang H, Wang Z, Li J, Ou
Y, Xiao X, Wang W, Chang A, Sun W, et al: Zeb1-induced metabolic
reprogramming of glycolysis is essential for macrophage
polarization in breast cancer. Cell Death Dis. 13:2062022.
|
|
99
|
Zhang A, Xu Y, Xu H, Ren J, Meng T, Ni Y,
Zhu Q, Zhang WB, Pan YB, Jin J, et al: Lactate-induced M2
polarization of tumor-associated macrophages promotes the invasion
of pituitary adenoma by secreting CCL17. Theranostics.
11:3839–3852. 2021.
|
|
100
|
Ye H, Zhou Q, Zheng S, Li G, Lin Q, Wei L,
Fu Z, Zhang B, Liu Y, Li Z and Chen R: Tumor-associated macrophages
promote progression and the Warburg effect via CCL18/NF-kB/VCAM-1
pathway in pancreatic ductal adenocarcinoma. Cell Death Dis.
9:4532018.
|
|
101
|
Stone SC, Rossetti RAM, Alvarez KLF,
Carvalho JP, Margarido PFR, Baracat EC, Tacla M, Boccardo E,
Yokochi K, Lorenzi NP and Lepique AP: Lactate secreted by cervical
cancer cells modulates macrophage phenotype. J Leukoc Biol.
105:1041–1054. 2019.
|
|
102
|
Qian M, Wang S, Guo X, Wang J, Zhang Z,
Qiu W, Gao X, Chen Z, Xu J, Zhao R, et al: Hypoxic glioma-derived
exosomes deliver microRNA-1246 to induce M2 macrophage polarization
by targeting TERF2IP via the STAT3 and NF-κB pathways. Oncogene.
39:428–442. 2020.
|
|
103
|
Niu X, Ma J, Li J, Gu Y, Yin L, Wang Y,
Zhou X, Wang J, Ji H and Zhang Q: Sodium/glucose cotransporter
1-dependent metabolic alterations induce tamoxifen resistance in
breast cancer by promoting macrophage M2 polarization. Cell Death
Dis. 12:5092021.
|
|
104
|
Lin S, Sun L, Lyu X, Ai X, Du D, Su N, Li
H, Zhang L, Yu J and Yuan S: Lactate-activated macrophages induced
aerobic glycolysis and epithelial-mesenchymal transition in breast
cancer by regulation of CCL5-CCR5 axis: A positive metabolic
feedback loop. Oncotarget. 8:110426–110443. 2017.
|
|
105
|
He Y, Fang Y, Zhang M, Zhao Y, Tu B, Shi
M, Muhitdinov B, Asrorov A, Xu Q and Huang Y: Remodeling 'cold'
tumor immune microenvironment via epigenetic-based therapy using
targeted liposomes with in situ formed albumin corona. Acta Pharm
Sin B. 12:2057–2073. 2022.
|
|
106
|
Wang H, Wu C, Tong X and Chen S: A
biomimetic Metal-Organic framework nanosystem modulates
immunosuppressive tumor microenvironment metabolism to amplify
immunotherapy. J Control Release. 353:727–737. 2023.
|
|
107
|
Zhang J, Sun X, Xu M and Zhao X, Yang C,
Li K, Zhao F, Hu H, Qiao M, Chen D and Zhao X: A Self-amplifying
ROS-sensitive prodrug-based nanodecoy for circumventing immune
resistance in chemotherapy-sensitized immunotherapy. Acta Biomater.
149:307–320. 2022.
|
|
108
|
Shen W, Liu T, Pei P, Li J, Yang S, Zhang
Y, Zhou H, Hu L and Yang K: Metabolic Homeostasis-Regulated
Nanoparticles for Antibody-Independent Cancer Radio-Immunotherapy.
Adv Mater. 34:e22073432022.
|
|
109
|
Harper J and Sainson RC: Regulation of the
anti-tumour immune response by cancer-associated fibroblasts. Semin
Cancer Biol. 25:69–77. 2014.
|
|
110
|
Sahai E, Astsaturov I, Cukierman E,
DeNardo DG, Egeblad M, Evans RM, Fearon D, Greten FR, Hingorani SR,
Hunter T, et al: A framework for advancing our understanding of
cancer-associated fibroblasts. Nat Rev Cancer. 20:174–186.
2020.
|
|
111
|
Gok Yavuz B, Gunaydin G, Gedik ME,
Kosemehmetoglu K, Karakoc D, Ozgur F and Guc D: Cancer associated
fibroblasts sculpt tumour microenvironment by recruiting monocytes
and inducing immunosuppressive PD-1+ TAMs. Sci Rep.
9:31722019.
|
|
112
|
Cui H, Xie N, Banerjee S, Ge J, Jiang D,
Dey T, Matthews QL, Liu RM and Liu G: Lung myofibroblasts promote
macrophage profibrotic activity through lactate-induced histone
lactylation. Am J Respir Cell Mol Biol. 64:115–125. 2021.
|
|
113
|
Chen S, Chen X, Shan T, Ma J, Lin W, Li W
and Kang Y: MiR-21-mediated Metabolic Alteration of
Cancer-associated Fibroblasts and Its Effect on Pancreatic Cancer
Cell Behavior. Int J Biol Sci. 14:100–110. 2018.
|
|
114
|
Nishida M, Yamashita N, Ogawa T, Koseki K,
Warabi E, Ohue T, Komatsu M, Matsushita H, Kakimi K, Kawakami E, et
al: Mitochondrial reactive oxygen species trigger
metformin-dependent antitumor immunity via activation of
Nrf2/mTORC1/p62 axis in tumor-infiltrating CD8T lymphocytes. J
Immunother Cancer. 9:e0029542021.
|
|
115
|
Janabi M, Yamashita S, Hirano K, Sakai N,
Hiraoka H, Matsumoto K, Zhang Z, Nozaki S and Matsuzawa Y: Oxidized
LDL-induced NF-kappa B activation and subsequent expression of
proinflammatory genes are defective in monocyte-derived macrophages
from CD36-deficient patients. Arterioscler Thromb Vasc Biol.
20:1953–1960. 2000.
|
|
116
|
Qiao G, Chen M, Mohammadpour H, MacDonald
CR, Bucsek MJ, Hylander BL, Barbi JJ and Repasky EA: Chronic
adrenergic stress contributes to metabolic dysfunction and an
exhausted phenotype in T cells in the tumor microenvironment.
Cancer Immunol Res. 9:651–664. 2021.
|
|
117
|
Sharma M, Boytard L, Hadi T, Koelwyn G,
Simon R, Ouimet M, Seifert L, Spiro W, Yan B, Hutchison S, et al:
Enhanced glycolysis and HIF-1α activation in adipose tissue
macrophages sustains local and systemic interleukin-1β production
in obesity. Sci Rep. 10:55552020.
|
|
118
|
Yang Q, Ma Q, Xu J, Liu Z, Zou J, Shen J,
Zhou Y, Da Q, Mao X, Lu S, et al: Prkaa1 metabolically regulates
monocyte/macrophage recruitment and viability in diet-induced
murine metabolic disorders. Front Cell Dev Biol. 8:6113542020.
|
|
119
|
Kumar V, Cheng P, Condamine T, Mony S,
Languino LR, McCaffrey JC, Hockstein N, Guarino M, Masters G,
Penman E, et al: CD45 phosphatase inhibits STAT3 transcription
factor activity in myeloid cells and promotes Tumor-Associated
macrophage differentiation. Immunity. 44:303–315. 2016.
|
|
120
|
Deng H, Wu L, Liu M, Zhu L, Chen Y, Zhou
H, Shi X, Wei J, Zheng L, Hu X, et al: Bone marrow mesenchymal stem
Cell-derived exosomes attenuate LPS-Induced ARDS by modulating
macrophage polarization through inhibiting glycolysis in
macrophages. Shock. 54:828–843. 2020.
|