Glutathione peroxidase 3 (GPX3) is a highly
conserved selenoprotein. As a member of the GPX family, it
catalyzes the reduction of glutathione (GSH), detoxifies
water-soluble lipid hydroperoxide and protects cells. GPX3 also
provides a greater survival advantage in response to exogenous
oxidative stress, including chemotherapy (1). Furthermore, GPX3 plays a crucial
role in the growth regulation and differentiation of various
malignant tumor cells, exhibiting dual roles in different tumors.
In certain cancer types, including gastric, breast, renal clear
cell, colorectal and endometrial cancer, and myeloid leukemia, the
absence of GPX3 expression often indicates a poor prognosis and the
development of resistance to chemotherapy in patients (2). However, GPX3 expression is elevated
in epithelial ovarian cancer (OC) and clear cell carcinoma of the
ovary (3). This shows that GPX3
serves as a tumor suppressor and pro-survival protein (4). Moreover, GPX3 is the only
extracellular GPX in the oxidoreductase family. In OC, it is the
sole antioxidant enzyme with high expression that is negatively
associated with the overall survival of patients (4-6).
OC is the 5th most common malignancy in women and
the leading cause of death from gynecologic malignancies (6). Usually, patients with OC exhibit
non-specific symptoms such as abdominal distension, pain, appetite
loss, or increased frequency of urination during early development.
The most common sign in patients with advanced disease is abdominal
swelling due to the accumulation of ascites (7). Among the five major epithelioid OC
tissue types recognized by the World Health Organization criteria
in 2014, high-grade serous OC (HGSOC) is the most common
histological subtype in the clinic, accounting for 70-80% of OC
deaths (8). As there is no
reliable early screening method, most patients are diagnosed only
at stages III and IV (International Federation of Gynecology and
Obstetrics staging) (7).
Substantial advances have been made in OC detection and therapeutic
approaches in recent years; however, the 5-year survival rate for
patients with advanced OC remains low (49%, 2022) due to tumor
heterogeneity, lack of reliable early diagnostic methods and the
high incidence of chemotherapy resistance. Therefore, gaining a
deeper understanding of GPX3 and identifying new therapeutic
targets to improve the prognosis of patients with OC are crucial
(9).
The aim of the current study was to review the role
of GPX3 in OC and investigate the potential factors and effects of
GPX3 on OC stem cells (OCSCs). The current study focused on
summarizing the mechanism of action of GPX3 in OC and OCSCs, as
well as identifying potential targets for clinical
intervention.
Serum GPX3 is a highly conserved selenoprotein. The
human GPX3 gene consists of five exons in the 5q32 region of
chromosome 5, it is 10 kb in length and it encodes a 23-kDa protein
that forms a homotetramer (14).
Lee et al (12) showed
that the secreted isoform of the GPX3 protein is a homotetramer
consisting of 226 amino acids with two arginine sites that bind to
GSH. Selenocysteine is the substance at the center of the catalytic
activity of GPX (GSH-Px), and its activity is closely related to
the selenium content in the body. Each GPX3 monomer has
selenocysteine as the active center and forms a tetramer with
glutamine, tryptophan and asparagine (9).
GSH-Px, thioredoxin reductase and the thyroid
hormone deiodinase are involved in intracellular signaling, redox
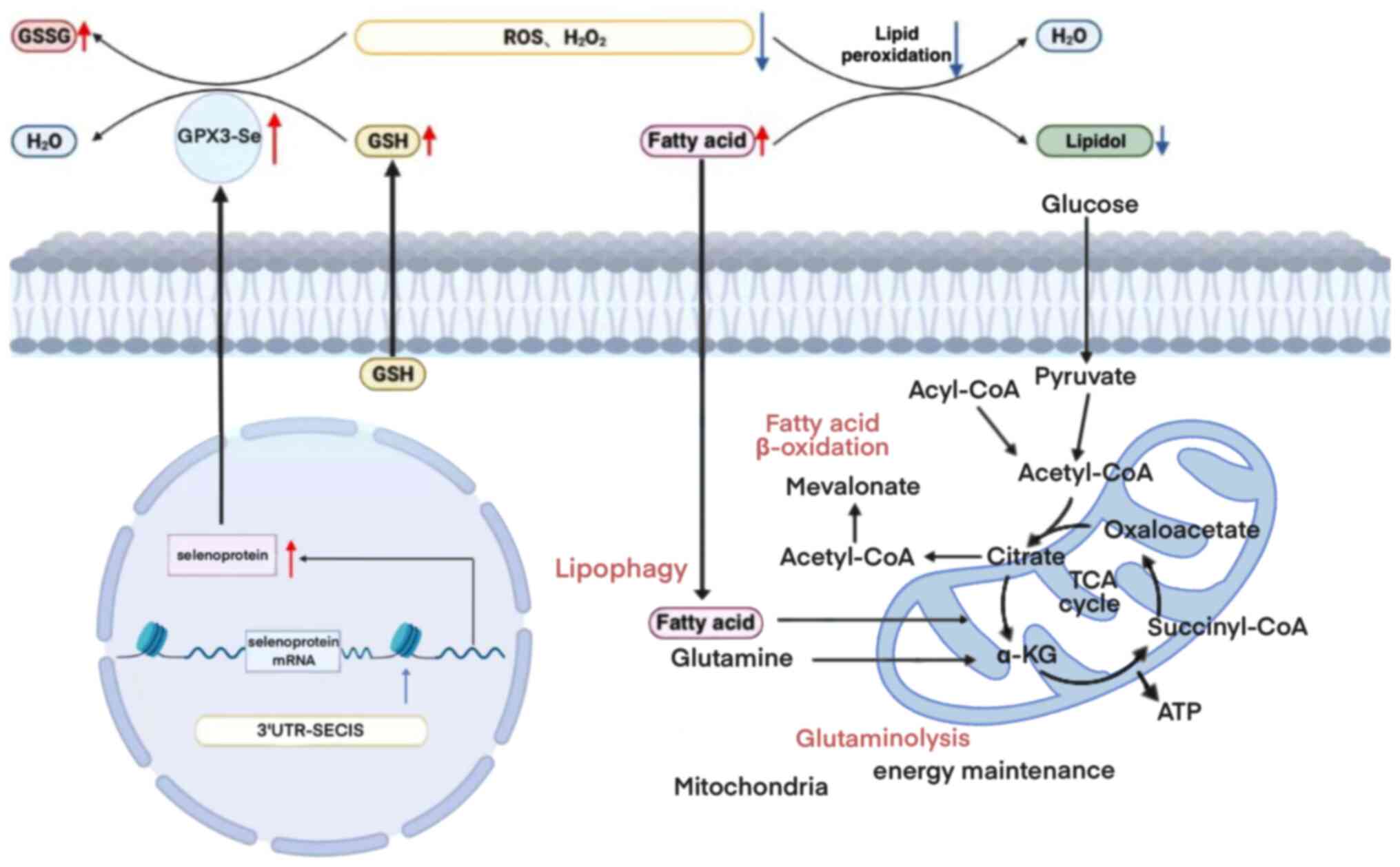
homeostasis and thyroid hormone metabolism regulation (15). GSH scavenges excess free radicals
in the body by oxidizing the sulfhydryl group (-SH) of GSH to
produce oxidized GSH (GSSG) through the catalysis of GSH-Px, which
consumes H2O2 to produce water (13). By contrast, GSH reductase uses
nicotinamide adenine dinucleotide phosphate to catalyze the
reduction of GSSG to generate GSH, thereby reducing reactive oxygen
species (ROS) in the intracellular cyclic environment and
maintaining redox homeostasis (16). GPX3 catalyzes the reduction of
hydroperoxides, including H2O2 and soluble
lipid hydroperoxides, using GSH (17). Additionally, GPX3 can interact
with GSH and thioredoxin reductase, or with thioredoxin alone, to
produce electrons for GSH in the presence of GSH depletion
(18), which in turn protects the
cells from ROS-induced deoxyribonucleic acid (DNA) and cellular
damage (15).
Extracellular GPX3 relies on the presence of a
cysteine encoded by the UGA opal codon in its active catalytic site
to undergo conversion into a functional protein (19). Typically, UGA codons serve as
signals for translation termination (20). However, in the case of
selenoprotein messenger ribonucleic acid (mRNA), the 3′untranslated
region contains selenocysteine insertion sequence (SECIS) elements,
which allow for the recognition of UGA as a selenium cysteine codon
rather than a stop signal (21).
Consequently, a deficiency in selenoproteins would result in loss
of GPX3 expression (22).
Deficiency of selenium can result in insufficient GPX3
biosynthesis, potentially elevating the susceptibility to
neurological symptoms, thyroid disorders, reduced fertility,
complications during pregnancy and other diseases that are
dependent on selenium (Fig. 1)
(12). Moreover, previous studies
have demonstrated that the expression of GPX3 is regulated by
peroxisome proliferator-activated receptor γ (PPARγ) (11,23,24). Zhou et al (11) provided evidence that alterations
in the mRNA levels of the antioxidant factor GPX3 in ovarian
tissues of rats with polycystic ovary syndrome are associated with
PPAR-γ activity. Similarly, elevated levels of GPX3 and PPARγ
expression have been observed during episodes of obesity (23,24).
GPX3 exhibits its activity within the cytoplasmic
lysosomes and plasma membranes of mammalian cells located in
various organs such as the kidney, heart, lungs, liver, brain,
adipose tissue, mammary glands and gastrointestinal tract (25). The mechanism of GPX3 function in
different types of cancers is shown in Fig. 2.
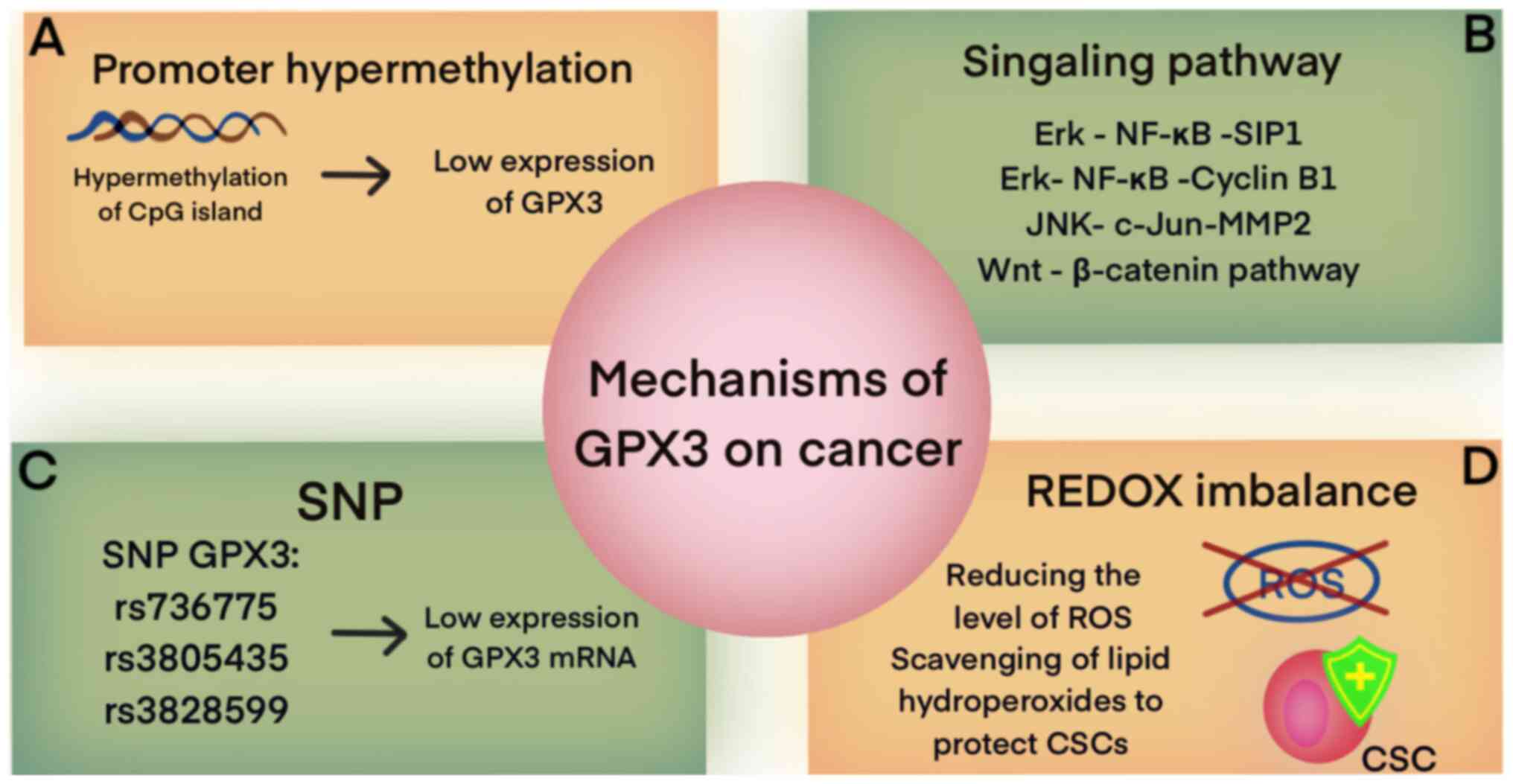
The expression level of GPX3 is strongly associated
with the density of DNA methylation (26). In various types of cancers,
including gastric (5), lung
(27), breast (17,28), endometrial (29), cervical (30), prostate (31) and head and neck tumors (21), the downregulation or complete
silencing of GPX3 gene expression, along with hypermethylation of
CpG in the CpG island of the GPX3 promoter, have been observed
(32). These molecular
alterations are indicative of a negative prognosis for patients
(Table I) (32,33,34). In the context of prostate cancer,
the interaction between GPX3 and p53-induced gene 3 has been
observed (35). However, the
expression of GPX3 is inhibited by promoter hypermethylation,
resulting in the loss of cellular antioxidant capacity. This loss
may contribute to the development of breast tumorigenesis (36). Similarly, in patients with breast
cancer, the inhibition of GPX3 expression through promoter
hypermethylation has been associated with the proliferation,
migration and invasion of tumor cells. Additionally, GPX3
hypermethylation has been linked to platinum-based chemoresistance
in head and neck cancer (32).
Platinum-based drugs can induce oxidative stress and elevate
intracellular ROS, leading to apoptosis and elimination of
proliferative signals. Therefore, alterations in the intracellular
redox state can affect the cellular response to chemotherapeutic
agents. Similarly, in cisplatin-induced renal injury, microRNA
(miR)-4835p (37) promotes the
expression of GPX3, thereby mitigating oxidative stress and
exerting a protective effect on renal injury (38).
Modifications in GPX3 play a role in the regulation
of various signaling pathways in cancer, including nuclear factor
κ-B (NF-κB), Wnt/β-catenin and JNK signaling. In lung cancer cells,
GPX3 acts as an inhibitor of proliferation, migration and invasion
of tumor cells by suppressing ROS-mediated NF-κB signaling
(27,39). The expression of GPX3 can inhibit
the activation of NF-κB through the Erk pathway, leading to the
suppression of the cell cycle proteins B1 and G2/M and the
inhibition of epithelial-mesenchymal transition (EMT) by
downregulating the Erk-NF-κ B-SIP 1 signaling axis (40). Additionally, a study by Liu et
al (41) demonstrated that
miR-196a promotes the development of non-small cell lung cancer by
downregulating the expression of GPX3 and activating the JNK
pathway, thereby enhancing the proliferation, differentiation,
self-renewal ability and invasiveness of cancer stem cells (CSCs)
(41). These findings suggest
that GPX3 and the JNK pathway could serve as potential therapeutic
targets for non-small cell lung cancer (5). GPX3 can inhibit the migration and
invasion of gastric cancer cells and selectively inhibit the
NF-κB/Wnt5a/JNK signaling pathway (42).
SNPs are primarily located in the non-coding region
of the GPX3 gene and exhibit a positive association with the
susceptibility to cancer development (43). Research has demonstrated that the
presence of the GPX3 rs736775 C allele is linked to the survival
outcomes of patients with colorectal cancer (44). In gastric cancer, the effect of
two introns of the GPX3 gene, namely rs3805435 and rs3828599, on
gene expression and disease susceptibility has been demonstrated
(26). Furthermore, the
expression of GPX3 rs736775 in patients undergoing adjuvant
chemotherapy with platinum and fluorouracil has been associated
with enhanced overall survival (45). Consequently, GPX3 rs736775 can be
regarded as a potential prognostic marker. The presence of SNPs
influences the downregulation of GPX3 mRNA, resulting in a
reduction of GPX3 expression. This reduction in GPX3 expression
leads to an imbalance in extracellular redox homeostasis,
ultimately promoting cancer development (46).
ROS can alter the energy metabolic pathways in tumor
cells, promoting glycolysis and facilitating glucose transport
through direct regulation of glucose transporter proteins (47). Additionally, ROS can react with
unsaturated fatty acids on lipid membranes, leading to the
production of free radicals and inducing fatty acid peroxidation,
resulting in the generation of fatty acid-ROS (48). This process disrupts biological
membranes, depletes antioxidants and ultimately increases oxidative
stress (40). The function of
GPX3 is to reduce ROS levels (49). GPX3 exhibits a dual role in
cancer, and these seemingly contradictory results may be closely
related to ROS. In early-stage cancer and precancerous lesions,
decreased expression of GPX3 and increased ROS production promote
cancer development. In melanoma, the upregulation of GPX3 plays a
role in regulating ROS levels by inhibiting the expression of
HIF-1α and -2α (50). HIF-1 is
upregulated in various human cancers and plays a key role in
driving tumor growth, invasion and metastasis. It induces changes
in lipid metabolism through both HIF-dependent and -independent
mechanisms. Under hypoxic conditions, mitochondria produce more
ROS. GPX3 can suppress the expression of HIF-1α by regulating ROS,
affecting tumor cell energy metabolism, and inhibiting the growth
of melanoma cells (51). However,
GPX3 also serves to protect tumor cells from exogenous oxidant
damage by enhancing the removal of hydrogen and soluble lipid
hydroperoxides from the extracellular tumor environment (52). Consequently, GPX3 promotes tumor
invasiveness and chemoresistance. Increased expression of GPX3 has
been associated with unfavorable patient prognosis in OC, stem
cells and certain colorectal cancers (44,52).
According to the 2014 World Health Organization
criteria, there are five main types of epithelioid OC tissue, among
which, epithelial OC is the most prevalent, constituting ~90% of
all cases. OC is a complex disease that can be classified into five
main subtypes: HGSOC, low-grade serous OC, serous, clear cell and
mucinous (52). Among these
subtypes, HGSOC is the most prevalent in clinical settings,
accounting for 70-80% of OC-related deaths (53). Recent studies have revealed that
precancerous lesions of HGSOC are primarily located in the
fallopian tube epithelium and are driven by TP53 mutations
(7,26,54). However, in certain cases,
plasmacytoid tubal intraepithelial carcinoma can also serve as a
metastatic counterpart to HGSOC (55).
The primary characteristics of HGSOC include TP53
mutations and frequent mutations in BRCA1 and 2 (56). Additionally, specific cases of
HGSOC exhibit overexpression of proto-oncogenes such as AKT and
ERRB2; this leads to increased genetic instability and heightened
activity of DNA repair mechanisms, such as overexpression of poly
(ADP-ribose) polymerase. Additionally, epigenetic traits, including
DNA hypomethylation and gene-specific hypermethylation, are
observed (57). Specifically,
hypermethylation of CpG sites at gene promoters affects specific
tumor suppressor genes such as SLIT2, PTEN, OPCML, RASSF1A, p16,
MLH1, E-calmodulin and APC (58).
The poor prognosis and high susceptibility to recurrence in
patients with tumors such as HGSOC can be attributed to the genetic
instability of these tumors.
CSCs are a subset of aberrant cells that possess the
unique capacity for self-renewal and differentiation, playing a
crucial role in tumor initiation, progression and metastasis
throughout the tumorigenesis process (59). Makino initially proposed the
concept of CSCs in 1959 (60). In
2005, Bapat et al (61)
made a substantial breakthrough by isolating and culturing
suspended cell spheres with CSC characteristics from ascites
obtained from patients with advanced OC. This discovery confirmed
the existence of OCSCs. Further research revealed that during
regular ovulation in women, ovarian epithelial cells undergo
continuous repair damage, leading to the continuous proliferation
and differentiation of OCSCs (62). Additionally, if OCSCs are exposed
to inflammatory mediators, undergo mesenchymal endothelial
transformation, or experience dysregulation of the redox balance in
the tumor microenvironment (TME), they may stimulate the continuous
proliferation of OCSCs, potentially leading to OC development
(63). Moreover, the unlimited
proliferation and immune evasion properties of CSCs contribute to
drug resistance and tumor recurrence in patients with OC, making
OCSCs a major factor in these clinical challenges (64). Table II presents the various markers of
OCSCs, and the names and mechanisms of drugs that have been or will
be discovered for their treatment.
The phenotype of tumor stem cells exhibits dynamic
characteristics rather than being a static attribute of tumor cells
(73). Signal transduction, redox
homeostasis, cell-to-cell contact and secreted factors present in
the TME can prompt differentiated tumor cells to reacquire stem
cell-like characteristics. Mounting evidence indicates that
conventional therapies alone are inadequate in eradicating tumor
stem cells. Furthermore, residual individual CSCs can trigger tumor
recurrence (74).
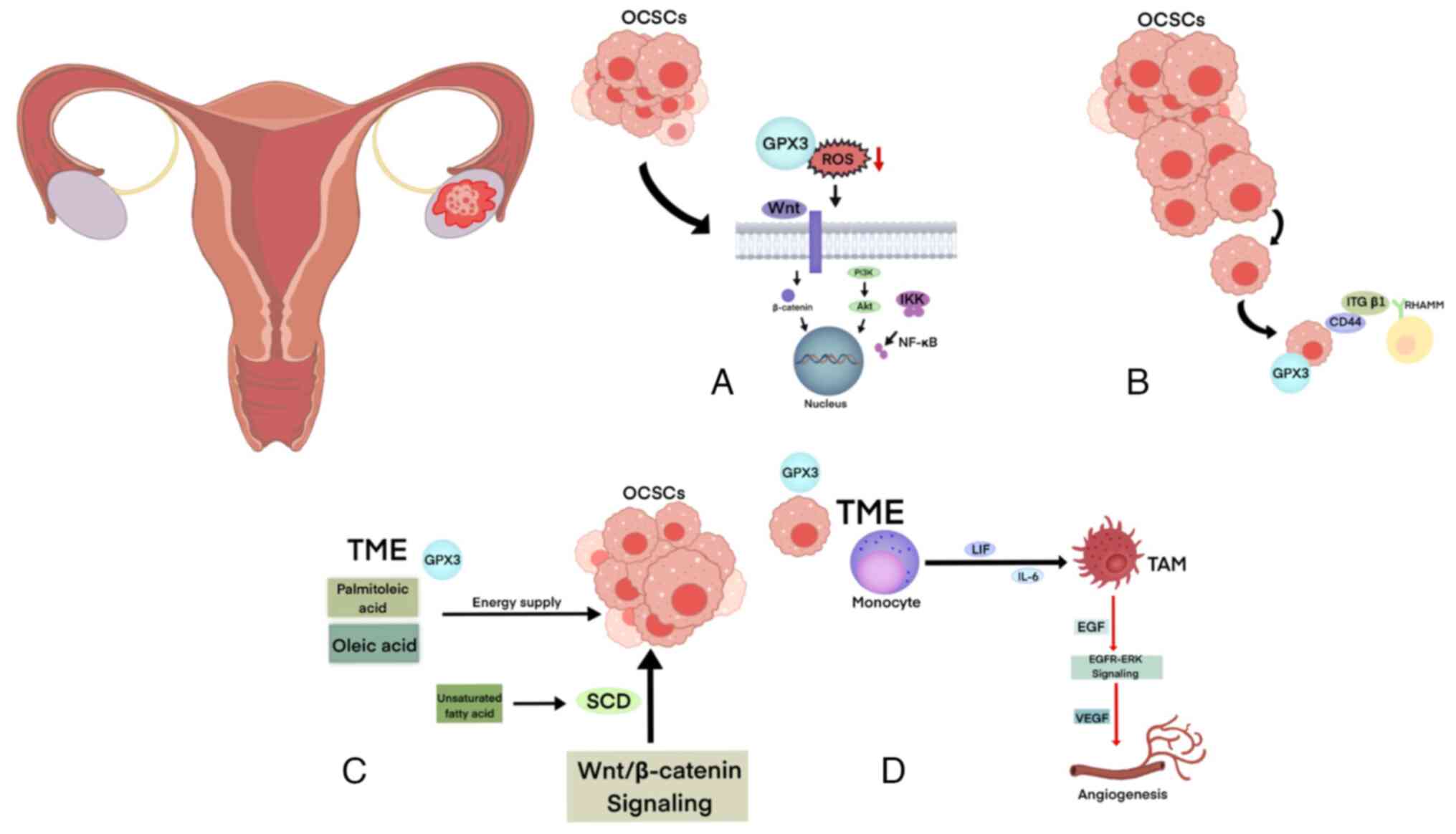
In OC development, the involvement of β-catenin in
the Wnt/β-catenin signaling pathway has been observed in stem cell
proliferation and differentiation, as well as in drug resistance in
OCSCs. Chen et al (75)
reported that the Notch signaling pathway contributes to the
survival of ovarian stem cells and their resistance to
platinum-based chemotherapeutic drugs. Additionally, the expression
of Notch3 is associated with a poor prognosis in patients with OC.
Furthermore, endothelial cells in the TME activate the expression
of the Notch1 receptor (N1ICD) and facilitate peritoneal
metastasis. Studies have demonstrated that the lifespan of mice
with OC is considerably reduced owing to the continuous activity of
N1ICD, which leads to the aging of endothelial cells, increased
expression of chemokines and the adhesion molecule VCAM1, and
facilitates recruitment of neutrophils and invasion of tumors
(70,74). Additionally, CD117 is expressed at
high levels in OCSCs and plays a role in tumor initiation and
resistance to cisplatin/paclitaxel by activating the
Wnt/β-catenin-ABCG 2 pathway (76).
Recently, the interest among researchers in the role
of non-coding RNAs in the regulation of tumorigenesis and
development is growing. Wang et al (77) conducted a study that focused on
the small nuclear kernel RNA host gene 16 (SNHG16)/Enhancer binding
protein β(CEBPB)/GATA3 axis and its influence on precursor mRNA
processing factor 6 (PRPF6) and GATA3 expression. The findings of
their study revealed that PRPF6 promotes the expression of GATA3 by
inducing SNHG16 (77). SNHG16
specifically interacts with CEBPB to upregulate the transcriptional
activity of GATA3. This upregulation of GATA3 promotes OC cell
migration and invasion, enhances resistance to paclitaxel drugs and
ultimately affects the prognosis of patients with advanced OC
(77). Liu et al (78) conducted a study that revealed the
regulatory role of the non-coding cyclic RNA circ-0000231 in the
proliferation, differentiation and invasion of OC cells. They
discovered that circ-0000231 acts through the
circ-0000231/miR-140/RAP1B axis, leading to increased expression of
E-cadherin in paclitaxel-resistant tumor cells. This discovery
highlighted circ-0000231 as a potential target for the precision
treatment of OC.
Several studies have demonstrated an aberrant
expression of GPX3 in various malignant cells, and this aberration
plays a dual role in different tumors (18). In OC, advanced papillary
plasmacytoid OC is associated with low GPX3 levels in the serum
(28). However, in plasmacytoid
OC cells, patients with increased expression of GPX3 had a reduced
median survival of 9.3 months (79). High expression of GPX3 in OC has
been potentially linked to the more prevalent subtype HGSOC.
Additionally, advanced tumors tend to exhibit higher levels of GPX3
expression. This is particularly important as patients with high
GPX3 expression and advanced-stage tumors often experience poor
survival rates. This finding may be attributed to the presence of
considerable amounts of ascites, which creates a favorable
environment for the survival of tumor cells (80). Furthermore, several findings have
indicated an upregulation of GPX3 expression in less common
subtypes of ovarian clear cell carcinoma, which has been linked to
chemoresistance development (73).
The TME refers to the specific location where tumor
cells develop, proliferate and spread. It encompasses the
structural and functional aspects of the cellular surroundings, but
also the presence of metabolites such as glucose, amino acids and
lipids within the TME, as well as environmental factors such as
hypoxia and acidity (81). CSCs
can consume metabolites within the TME. Additionally, CSCs actively
contribute to the remodeling of the TME by secreting various
metabolites, thereby creating a favorable ecological environment
for tumor initiation and progression (74). GPX3 can clear ROS through various
signaling pathways, protecting cancer cells from damage caused by
exogenous oxidants while also utilizing exogenous fatty acids
provided in the TME to help OCSCs to survive in the complex TME.
Additionally, the high expression of GPX3 may be related to the
immunosuppressive state of the TME, with its expression positively
associated with plasma cells and M0 macrophages and negatively
associated with monocytes and M1 and M2 macrophages (Fig. 3) (63).
ROS is a broad term that includes oxygen radical and
non-radical compounds, such as H2O2, hydroxyl
radicals, lipid radicals and (phospho)lipid hydroperoxides
(82). Elevated ROS levels have
been observed in various cancers and have been linked to the
activation of signaling pathways involved in cancer initiation and
progression, including the mitogen-activated protein kinase/Erk,
phosphatidylinositol-3-kinase (PI3K)/Akt, and IκB kinase/NF-κB
pathways (83). Furthermore, ROS
accumulation over time leads to irreversible damage, and Harman's
(84) theory provides further
insight into the role of extracellular ROS in triggering
age-related cancer progression (85). Transformation, alterations in
metabolism and heightened ROS production contribute to elevated
levels of oxidative damage in tumor cells compared with normal
counterparts (80). The elevation
of ROS within tumor cells has been associated with several
detrimental effects, including enhanced cell proliferation,
facilitation of mutations and heightened genetic instability
(86).
The inhibition of the GPX3 gene leads to a decrease
in the ability of tumor cells to survive without anchorage and a
reduced capacity to respond to external oxidative stress. These
findings suggest that GPX3 may play a crucial role in the reduction
of extracellular ROS. Additionally, Worley et al (4) determined whether GPX3 is essential
for OC cells to respond to extracellular oxidants. The results
showed that cells with suppressed GPX3 expression were more
susceptible to cell death induced by ascorbate, indicating that
GPX3 expression is crucial for scavenging excessive extracellular
H2O2. Another study demonstrated that GPX3
protects OC cells from damage caused by external oxidants by
scavenging H2O2 (74).
In OC, the process of EMT is necessary for tumor
metastasis. EMT is a critical biological process through which
malignant tumor cells derived from epithelial cells acquire
migratory and invasive capabilities. Through EMT, epithelial cells
lose cell polarity, connections to the basement membrane and other
epithelial phenotypes, and gain mesenchymal phenotypes such as
increased migratory and invasive abilities, anti-apoptotic features
and the capacity to degrade the extracellular matrix (79). This process confers the tumor
cells the ability to invade and metastasize. The tissue factors
Snail and Slug, involved in EMT, promote the inactivation of
cancer-generated and p53-mediated apoptotic programs (35). Additionally, Shishido et al
(80) demonstrated that the
adhesion of OC cells to the peritoneal mesothelium is facilitated
by the interaction between the OCSC markers CD44 and integrin-β1,
which are recognized by hyaluronic acid receptors on the
mesothelial cell membrane (80).
These findings indicate that mesothelial cells can enhance the stem
cell-like characteristics of OC spheres. This implies that the
adhesion and stemness of OCSCs establish a mutually reinforcing
positive feedback loop (87).
Furthermore, Hu et al (42) discovered that the downregulation
of the GPX3 gene resulted in a decrease in the wound healing
capacity and transmembrane migration rate of OC and colorectal
cancer cells. Moreover, the expression level of GPX3 was found to
be closely associated with tumor metastasis. In early-stage cancer
and precancerous lesions, decreased expression of GPX3 and
increased ROS production stimulate oxidative stress in TME,
promoting the progression of EMT and cancer development. In
advanced cancer, the high expression of GPX3 clears excess ROS in
the extracellular environment, maintaining the redox homeostasis in
the TME, protecting tumor cells and enhancing drug resistance
(88).
Lipids, such as triglycerides, play a crucial role
in supporting oncogenic signals and meeting the energy demands of
rapidly dividing cancer cells. Compared with differentiated tumor
cells, CSCs rely heavily on lipid metabolism to maintain their
stemness and fulfill intracellular biosynthesis and energy
metabolism requirements (89). In
patients with OC, the omentum and peritoneum contain a substantial
number of adipocytes. These adipocytes can transform into
cancer-associated adipocytes through interaction with OC cells.
These disease-associated adipocytes release lipids, hormones,
adipokines and tumor-promoting factors that facilitate tumor growth
and metastatic progression (90).
Additionally, the high expression of the fatty acid chaperone FABP4
by adipocytes in OC during proliferation and differentiation is
positively associated with tumor recurrence following surgery
(91). This phenomenon can be
attributed to the upregulation of FABP4 by Notch1, which
subsequently enhances tumor growth and angiogenesis in ovarian
tumor xenografts (91).
Immune cells have been observed to infiltrate the
TME in OC. Among these cells, some function as tumor-associated
immune cells, including immature/tolerogenic dendritic cells (DCs),
M2 macrophages, regulatory T cells and myeloid-derived suppressor
cells (98). These cells play a
role in maintaining immune tolerance and suppressing anti-tumor
immunity, ultimately leading to drug resistance in the ovary
(12). By contrast, mature DCs,
M1 macrophages and natural killer cells can directly inhibit tumor
growth or enhance susceptibility to OC-targeted therapies (99). Within the TME, immunosuppressive
cells, such as myeloid DCs and CD4+ Th1- and Th2-type T
cells, have been positively associated with the expression of GPX3
(100). This suggests that the
high expression of GPX3 may be linked to the immunosuppressive
state of the TME. Pei et al (64) revealed a close relationship
between pathological injury of renal tissues following renal
ischemia-reperfusion, increased GPX3 expression (a marker of
oxidative stress) and various processes (including GSH metabolism,
oxidative stress pathways and regulation of T-cell activation). The
expression of GPX3 was positively associated with plasma cells and
M0 macrophages, whereas it exhibited a negative association with
monocytes and M1 and M2 macrophages.
Tumor-associated macrophages (TAMs) are found at
high concentrations in the ascites of patients with OC due to the
induction of monocyte differentiation into TAM by factors such as
leukemia inhibitory factor and IL-6 (98). TAMs are responsible for the
release of epidermal growth factor (EGF) in OC, which directly
activates the EGF receptor (R)-ERK pathway (101). This activation, in turn,
upregulates the vascular endothelial growth factor and promotes
angiogenesis (102).
Additionally, glutamine is recognized as a crucial metabolite in
cancer cells and is emerging as having a major role in TAM
metabolism (84). Data indicate
that OC cells release N-acetyl aspartate, which in conjunction with
IL-10, synergistically induces the transformation of macrophages
into M2-type TAMs that overexpress glutamine synthetase (103). Hartwell et al (104) observed that CD8+
tumor-infiltrating T cells, under hypoglycemic and hypoxic
conditions, alter the metabolic profile of tumor cells from
glycolysis to fatty acid catabolism, thereby sustaining the energy
supply and stemness characteristics of tumor cells.
The current primary treatment approach for patients
diagnosed with OC involves performing a surgical procedure to
achieve complete tumor reduction. Tumor reduction surgery
encompasses procedures such as hysterectomy, omentectomy, and the
potential excision of other affected tissue. Despite achieving
complete remission following a combination of subtractive surgery
and first-line chemotherapy, a substantial proportion of patients
(range, 70-80%) experience recurrence within 2-5 years. This
recurrence is attributed to the presence of residual tumor tissue
and tumor stem cells which serve as the origin for future
recurrences (105). At present,
the primary approach to inhibit DNA synthesis in actively
proliferating tumor cells in patients with OC is achieved by using
platinum- and paclitaxel-dependent chemotherapy (106). However, the development of drug
resistance to these treatments affects the prognosis of patients
with OC.
Several recent studies have shown that targeted
therapy against OC cells and OCSCs will provide a new strategy for
the treatment or even cure of OC (106,107).
Levamisole, a therapeutic agent that specifically
targets CD133, a recognized marker of OCSCs, has been developed
(67). CM37, an inhibitor of
ALDH1A1, has been shown to induce the intracellular accumulation of
ROS in OCSCs, leading to DNA damage (72). Additionally, imatinib, a targeted
agent for CD117, inhibits the expression of genes associated with
OCSC. Furthermore, novel therapeutic approaches have been developed
to target signaling pathways in OCSCs (108). For instance, the combination of
inhibitors targeting the PI3K/Akt pathway with paclitaxel drugs has
been shown to enhance the sensitivity of paclitaxel (109). Alwosaibai et al (110) investigated the relationship
between programmed death-ligand 1 (PD-L1) expression, tumor
infiltrating lymphocyte expression and tumor stem cell markers in
OC (110). The researchers
discovered that inhibiting PD-L1 using immune checkpoint inhibitors
resulted in a reduction in the number of stem cells associated with
cancer recurrence (111).
Additionally, Zhou et al (112) examined the effect of miR-1307 on
the cell cycle and chemosensitivity of OCSCs. They discovered that
miR-1307 influenced the transcriptional effectors of the capicua
transcriptional repressor gene and receptor tyrosine kinase
signaling in OC cells. Furthermore, miR-1307 was found to be a
reliable predictor of chemotherapy effectiveness in patients,
providing valuable guidance for individualized treatment plans in
clinical settings. Moreover, the expression level of miR-1307 can
be utilized for more accurate and sensitive detection of OC.
With the progressive advancement of therapeutic
investigations on drug-resistant OC, clinical interventions have
increasingly emphasized targeting molecular markers associated with
OC in conjunction with conventional chemotherapy. For instance, a
recent study demonstrated that 3-hydroxy-3-methyl glutaryl coenzyme
A reductase can act as a prognostic indicator for statin treatment
(96). The concurrent
administration of statins and adriamycin enhances the DNA-damaging
effect and effectively suppresses P-glycoprotein, thereby reducing
drug resistance in patients with OC. Statins are cost-effective,
have minimal adverse effects and require comprehensive
investigation as potential alternative treatments for OC (113). Recent developments in targeted
therapeutic agents for lipid metabolism in various CSCs have been
documented and summarized in Table
III (107). Based on these
findings, it was hypothesized that GPX3 may play a role in the
regulation of redox homeostasis and lipid metabolism molecules
within the TME. This hypothesis aims to identify potential
therapeutic targets for OCSCs. Multidrug-resistant proteins, which
are commonly upregulated in cancers, can also transport GSH
(114). This finding implies
that the enhanced release of GSH into the extracellular space may
play a role in the upregulation of GPX3 activity within the TME.
Consequently, amino acid precursors involved in GSH synthesis and
substances like N-acetylcysteine could enhance the effectiveness of
GPX3 (44). In OC, the
dysregulation of signaling pathways that control lipid metabolism
disrupts the normal processing of fatty acids and cholesterol in
OCSCs. Further investigation is required to elucidate the specific
role of GPX3 in lipid metabolism within OCSCs (115).
With the advancement of research in the field of OC,
the interest in using immunotherapy and bioengineering techniques
in clinical settings is on the increase. Carboplatin and paclitaxel
have been found to induce an inflammatory state through the
upregulation of IFN-γ (116). By
contrast, the administration of carboplatin and gemcitabine creates
an immunosuppressive milieu. Platinum agents have been demonstrated
to enhance IL-6 and PGE2 production in OC cell lines. This is
followed by the activation of the STAT3 pathway and the induction
of M2 polarization, accompanied by upregulation of IL-10 (117). Platinum agents may have an
immunosuppressive effect on the TME by promoting M2 TAM
polarization (118). Chen et
al (119) used high levels
of GSH reductase in the TME and showed that the disulfide bond
(-SS-) in SSBPEI-DOX could be specifically reduced, allowing it to
bind to the conventional chemotherapeutic drug DOX and induce
apoptosis or necrosis of CSCs. The researchers developed a novel
nanoparticle system (SSBPEI-DOX@siRNAs/iRGD-PEG-HA) that was
dual-targeted and GSH-responsive. This system efficiently and
specifically delivered a combination of adriamycin and small
interfering RNAs to OCSCs (119).
The function of GPX3 as an extracellular antioxidant
enzyme in the cytosol and mitochondria can affect the fatty acid
metabolism of tumor stem cells, and the levels of GSH and
unsaturated fatty acids in the TME (120). These factors can influence the
maintenance of stemness of tumor stem cells. Additionally, the
measurement of GPX3 levels in plasma holds potential as a valuable
prognostic and diagnostic biomarker (121).
OC is a prevalent malignancy affecting the female
reproductive system, and its etiology is complex, involving a
combination of genetic, environmental and hormonal factors
(122). Due to the limited
availability of early diagnostic techniques and the prevalence of
chemotherapy resistance, the overall survival rate for individuals
with advanced OC remains considerably low. Extensive research has
indicated that GPX3 is significantly downregulated in OC, and
patients with elevated expression levels of this gene in their
tumors are more likely to experience a poor prognosis. Moreover,
the upregulation of GPX3 expression may confer protection to tumor
stem cells against exogenous oxidative stress injury, thereby
facilitating the maintenance of stemness characteristics in OCSCs.
This phenomenon, in turn, can promote OC recurrence and
chemoresistance development. However, this association is purely
observational, and further research is required to establish a
causal relationship (122).
Not applicable.
MW and DG conceived and designed the study. DG
wrote the manuscript. YZ and MW revised the manuscript. All authors
have read and approved the final manuscript. Data authentication is
not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
The present review was supported by The Outstanding Scientific
Fund of Shengjing Hospital (grant no. 201705).
|
1
|
Sun Q, Mehl S, Renko K, Seemann P, Görlich
CL, Hackler J, Minich WB, Kahaly GJ and Schomburg L: Natural
Autoimmunity to Selenoprotein P impairs selenium transport in
Hashimoto's thyroiditis. Int J Mol Sci. 22:130882021.
|
|
2
|
Chang C, Worley BL, Phaëton R and Hempel
N: Extracellular glutathione peroxidase GPx3 and its role in
cancer. Cancers. 12:21972020.
|
|
3
|
Agnani D, Camacho-Vanegas O, Camacho C,
Lele S, Odunsi K, Cohen S, Dottino P and Martignetti JA: Decreased
levels of serum glutathione peroxidase 3 are associated with
papillary serous ovarian cancer and disease progression. J Ovarian
Res. 4:182011.
|
|
4
|
Worley BL, Kim YS, Mardini J, Zaman R,
Leon KE, Vallur PG, Nduwumwami A, Warrick JI, Timmins PF, Kesterson
JP, et al: GPx3 supports ovarian cancer progression by manipulating
the extracellular redox environment. Redox Biol. 25:1010512019.
|
|
5
|
Cai M, Sikong Y, Wang Q, Zhu S, Pang F and
Cui X: Gpx3 prevents migration and invasion in gastric cancer by
targeting NFкB/Wnt5a/JNK signaling. Int J Clin Exp Pathol.
12:1194–1203. 2019.
|
|
6
|
He Q, Chen N, Wang X, Li P, Liu L, Rong Z,
Liu W, Jiang K and Zhao J: Prognostic value and immunological roles
of GPX3 in gastric cancer. Int J Med Sci. 20:1399–1416. 2023.
|
|
7
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2020. CA Cancer J Clin. 70:7–30. 2020.
|
|
8
|
Armstrong DK, Alvarez RD, Backes FJ,
Bakkum-Gamez JN, Barroilhet L, Behbakht K, Berchuck A, Chen LM,
Chitiyo VC, Cristea M, et al: NCCN Guidelines® Insights:
Ovarian cancer, version 3.2022. J Natl Compr Cancer Netw.
20:972–980. 2022.
|
|
9
|
Demircan K, Sun Q, Bengtsson Y, Seemann P,
Vallon-Christersson J, Malmberg M, Saal LH, Rydén L, Minich WB,
Borg Å, et al: Autoimmunity to selenoprotein P predicts breast
cancer recurrence. Redox Biol. 53:1023462022.
|
|
10
|
Cueto-Ureña C, Ramírez-Expósito MJ, Mayas
MD, Carrera-González MP, Godoy-Hurtado A and Martínez-Martos JM:
Glutathione Peroxidase gpx1 to gpx8 Genes expression in
experimental brain tumors reveals gender-dependent patterns. Genes
(Basel). 14:16742023.
|
|
11
|
Zhou Y, Lan H, Dong Z, Li W, Qian B, Zeng
Z, He W and Song JL: Rhamnocitrin attenuates ovarian fibrosis in
rats with letrozole-induced experimental polycystic ovary syndrome.
Oxid Med Cell Longev. 2022:1–18. 2022.
|
|
12
|
Lee HJ, Do JH, Bae S, Yang S, Zhang X, Lee
A, Choi YJ, Park DC and Ahn WS: Immunohistochemical evidence for
the over-expression of Glutathione peroxidase 3 in clear cell type
ovarian adenocarcinoma. Med Oncol. 28:522–527. 2011.
|
|
13
|
Pei J, Pan X, Wei G and Hua Y: Research
progress of glutathione peroxidase family (GPX) in redoxidation.
Front Pharmacol. 14:11474142023.
|
|
14
|
Xu S, Li X, Zhang S, Qi C, Zhang Z, Ma R,
Xiang L, Chen L, Zhu Y, Tang C, et al: Oxidative stress gene
expression, DNA methylation, and gut microbiota interaction trigger
Crohn's disease: A multi-omics Mendelian randomization study. BMC
Med. 21:1792023.
|
|
15
|
Jia Y, Dai J and Zeng Z: Potential
relationship between the selenoproteome and cancer. Mol Clin Oncol.
13:132020.
|
|
16
|
Cohen S, Mehrabi S, Yao X, Milingen S and
Aikhionbare FO: Reactive oxygen species and serous epithelial
ovarian adenocarcinoma. Cancer Res J (N Y N Y). 4:1062016.
|
|
17
|
Lou W, Ding B, Wang S and Fu P:
Overexpression of GPX3, a potential biomarker for diagnosis and
prognosis of breast cancer, inhibits progression of breast cancer
cells in vitro. Cancer Cell Int. 20:3782020.
|
|
18
|
Nirgude S and Choudhary B: Insights into
the role of GPX3, a highly efficient plasma antioxidant, in cancer.
Biochem Pharmacol. 184:1143652021.
|
|
19
|
Copeland PR and Howard MT: Ribosome fate
during decoding of UGA-sec codons. Int J Mol Sci. 22:132042021.
|
|
20
|
Hauffe R, Stein V, Chudoba C, Flore T,
Rath M, Ritter K, Schell M, Wardelmann K, Deubel S, Kopp JF, et al:
GPx3 dysregulation impacts adipose tissue insulin receptor
expression and sensitivity. JCI Insight. 5:e1362832020.
|
|
21
|
Chen B, Rao X, House MG, Nephew KP, Cullen
KJ and Guo Z: GPx3 promoter hypermethylation is a frequent event in
human cancer and is associated with tumorigenesis and chemotherapy
response. Cancer Lett. 309:37–45. 2011.
|
|
22
|
Fan Z, Yan Q, Song J and Wei J: Reactive
human plasma glutathione peroxidase mutant with diselenide bond
succeeds in tetramer formation. Antioxidants. 11:10832022.
|
|
23
|
Gusti AMT, Qusti SY, Alshammari EM, Toraih
EA and Fawzy MS: Antioxidants-related superoxide dismutase (SOD),
catalase (CAT), glutathione peroxidase (GPX),
Glutathione-S-Transferase (GST), and nitric oxide synthase (NOS)
gene variants analysis in an obese population: A preliminary
Case-control study. Antioxidants (Basel). 10:5952021.
|
|
24
|
Reddy AT, Lakshmi SP, Banno A and Reddy
RC: Role of GPx3 in PPARγ-induced protection against
COPD-associated oxidative stress. Free Radic Biol Med. 126:350–357.
2018.
|
|
25
|
Chen Y, Zhou Z and Min W: Mitochondria,
oxidative stress and innate immunity. Front Physiol.
9:14872018.
|
|
26
|
Xie J, Fu L and Zhang J: Analysis of
influencing factors on the occurrence and development of gastric
cancer in high-incidence areas of digestive tract tumors based on
high methylation of GPX3 gene. J Oncol. 2022:30948812022.
|
|
27
|
Wang Z, Zhu J, Liu Y, Wang Z, Cao X and Gu
Y: Tumor-polarized GPX3+ AT2 lung epithelial cells promote
premetastatic niche formation. Proc Natl Acad Sci.
119:e22018991192022.
|
|
28
|
Saelee P, Pongtheerat T and
Sophonnithiprasert T: Reduced expression of GPX3 in breast cancer
patients in correlation with clinical significance. Glob Med Genet.
7:87–91. 2020.
|
|
29
|
Falck E, Karlsson S, Carlsson J, Helenius
G, Karlsson M and Klinga-Levan K: Loss of glutathione peroxidase 3
expression is correlated with epigenetic mechanisms in endometrial
adenocarcinoma. Cancer Cell Int. 10:462010.
|
|
30
|
Zhang X, Zheng Z, Shen Y, Kim H, Jin R, Li
R, Lee DY, Roh MR and Yang S: Downregulation of glutathione
peroxidase 3 is associated with lymph node metastasis and prognosis
in cervical cancer. Oncol Rep. 31:2587–2592. 2014.
|
|
31
|
Rizzo A, Santoni M, Mollica V, Fiorentino
M, Brandi G and Massari F: Microbiota and prostate cancer. Semin
Cancer Biol. 86:1058–1065. 2022.
|
|
32
|
Szyfter K: Genetics and molecular biology
of head and neck cancer. Biomolecules. 11:12932021.
|
|
33
|
Uroshlev LA, Abdullaev ET, Umarova IR,
Il'icheva IA, Panchenko LA, Polozov RV, Kondrashov FA, Nechipurenko
YD and Grokhovsky SL: A method for identification of the
methylation level of CpG islands from NGS Data. Sci Rep.
10:86352020.
|
|
34
|
Dai X, Ren T, Zhang Y and Nan N:
Methylation multiplicity and its clinical values in cancer. Expert
Rev Mol Med. 23:e22021.
|
|
35
|
Wang H, Luo K, Tan LZ, Ren BG, Gu LQ,
Michalopoulos G, Luo JH and Yu YP: P53-induced gene 3 mediates cell
death induced by glutathione peroxidase 3. J Biol Chem.
287:16890–16902. 2012.
|
|
36
|
Zhao H, Li J, Li X, Han C, Zhang Y, Zheng
L and Guo M: Silencing GPX3 expression promotes tumor metastasis in
human thyroid cancer. Curr Protein Pept Sci. 16:316–321. 2015.
|
|
37
|
Zhang D, Deng JJ, Xu Q, Zeng Y and Jiang
J: MiR-146b-5p regulates the scavenging effect of GPx-3 on peroxide
in papillary thyroid cancer cells. Heliyon. 9:e184892023.
|
|
38
|
Xia Y, Pan W, Xiao X, Zhou X, Gu W, Liu Y,
Zhao Y, Li L, Zheng C, Liu J and Li M: MicroRNA-483-5p accentuates
cisplatin-induced acute kidney injury by targeting GPX3. Lab
Invest. 102:589–601. 2022.
|
|
39
|
Oh IJ, Kim HE, Song SY, Na KJ, Kim KS, Kim
YC and Lee SW: Diagnostic value of serum glutathione peroxidase 3
levels in patients with lung cancer. Thorac Cancer. 5:425–430.
2014.
|
|
40
|
Mosca L, Ilari A, Fazi F, Assaraf YG and
Colotti G: Taxanes in cancer treatment: Activity, chemoresistance
and its overcoming. Drug Resist Updat. 54:1007422021.
|
|
41
|
Liu Q, Bai W, Huang F, Tang J and Lin X:
Downregulation of microRNA-196a inhibits stem cell self-renewal
ability and stemness in non-small-cell lung cancer through
upregulating GPX3 expression. Int J Biochem Cell Biol.
115:1055712019.
|
|
42
|
Hu Q, Chen J, Yang W, Xu M, Zhou J, Tan J
and Huang T: GPX3 expression was down-regulated but positively
correlated with poor outcome in human cancers. Front Oncol.
13:9905512023.
|
|
43
|
Zhang H, Zhao W, Gu D, Du M, Gong W, Tan
Y, Wang M, Wen J, Zhai Y and Xu Z: Association of antioxidative
enzymes polymorphisms with efficacy of platin and
fluorouracil-based adjuvant therapy in gastric cancer. Cell Physiol
Biochem. 48:2247–2257. 2018.
|
|
44
|
Noci S, Dugo M, Bertola F, Melotti F,
Vannelli A, Dragani TA and Galvan A: A subset of genetic
susceptibility variants for colorectal cancer also has prognostic
value. Pharmacogenomics J. 16:173–179. 2016.
|
|
45
|
Wu S, Cheng Z, Peng Y, Cao Y and He Z:
GPx3 knockdown inhibits the proliferation and DNA synthesis and
enhances the early apoptosis of human spermatogonial stem cells via
mediating CXCL10 and cyclin B1. Front Cell Dev Biol.
11:12136842023.
|
|
46
|
Dongre A and Weinberg RA: New insights
into the mechanisms of epithelial-mesenchymal transition and
implications for cancer. Nat Rev Mol Cell Biol. 20:69–84. 2019.
|
|
47
|
Yao S, Wei W, Cao R, Lu L, Liang S, Xiong
M, Zhang C, Liang X and Ma Y: Resveratrol alleviates zea-induced
decidualization disturbance in human endometrial stromal cells.
Ecotoxicol Environ Saf. 207:1115112021.
|
|
48
|
Li Y, Zhou Y, Liu D, Wang Z, Qiu J, Zhang
J, Chen P, Zeng G, Guo Y, Wang X, et al: Glutathione Peroxidase 3
induced mitochondria-mediated apoptosis via AMPK/ERK1/2 pathway and
resisted autophagy-related ferroptosis via AMPK/mTOR pathway in
hyperplastic prostate. J Transl Med. 21:5752023.
|
|
49
|
Mao X, Xu J, Wang W, Liang C, Hua J, Liu
J, Zhang B, Meng Q, Yu X and Shi S: Crosstalk between
cancer-associated fibroblasts and immune cells in the tumor
microenvironment: New findings and future perspectives. Mol Cancer.
20:1312021.
|
|
50
|
Yi Z, Jiang L, Zhao L, Zhou M, Ni Y, Yang
Y, Yang H, Yang L, Zhang Q, Kuang Y, et al: Glutathione peroxidase
3 (GPX3) suppresses the growth of melanoma cells through reactive
oxygen species (ROS)-dependent stabilization of hypoxia-inducible
factor 1-α and 2-α. J Cell Biochem. 120:19124–19136. 2019.
|
|
51
|
Lee SH, Golinska M and Griffiths JR:
HIF-1-independent mechanisms regulating metabolic adaptation in
hypoxic cancer cells. Cells. 10:23712021.
|
|
52
|
Perets R, Wyant GA, Muto KW, Bijron JG,
Poole BB, Chin KT, Chen JYH, Ohman AWO, Stepule CD, Kwak S, et al:
Transformation of the fallopian tube secretory epithelium leads to
high-grade serous ovarian cancer in Brca;Tp53;Pten models. Cancer
Cell. 24:751–765. 2013.
|
|
53
|
Eckert MA, Pan S, Hernandez KM, Loth RM,
Andrade J, Volchenboum SL, Faber P, Montag A, Lastra R, Peter ME,
et al: Genomics of ovarian cancer progression reveals diverse
metastatic trajectories including intraepithelial metastasis to the
fallopian tube. Cancer Discov. 6:1342–1351. 2016.
|
|
54
|
Wang Y, Huang P, Wang BG, Murdock T, Cope
L, Hsu FC, Wang TL and Shih IM: Spatial Transcriptomic analysis of
ovarian cancer precursors reveals reactivation of IGFBP2 during
pathogenesis. Cancer Res. 82:4528–4541. 2022.
|
|
55
|
Yousefi B, Sadoughi F, Asemi Z, Mansournia
MA and Hallajzadeh J: Novel perspectives for the diagnosis and
treatment of gynecological cancers using Dysregulation of PIWI
Protein and PiRNAs as biomarkers. Curr Med Chem. 31:453–463.
2024.
|
|
56
|
Launonen IM, Lyytikäinen N, Casado J,
Anttila EA, Szabó A, Haltia UM, Jacobson CA, Lin JR, Maliga Z,
Howitt BE, et al: Single-cell tumor-immune microenvironment of
BRCA1/2 mutated high-grade serous ovarian cancer. Nat Commun.
13:8352022.
|
|
57
|
Eulenburg V and Hülsmann S: Synergistic
control of transmitter turnover at glycinergic synapses by GlyT1,
GlyT2, and ASC-1. Int J Mol Sci. 23:25612022.
|
|
58
|
Teodoridis JM, Hall J, Marsh S, Kannall
HD, Smyth C, Curto J, Siddiqui N, Gabra H, McLeod HL, Strathdee G
and Brown R: CpG island methylation of DNA damage response genes in
advanced ovarian cancer. Cancer Res. 65:8961–8967. 2005.
|
|
59
|
Xie Y, Ma S and Tong M: Metabolic
plasticity of cancer stem cells in response to Microenvironmental
cues. Cancers. 14:53452022.
|
|
60
|
Makino S: The role of tumor stem-cells in
regrowth of the tumor following drastic applications. Acta Unio Int
Contra Cancrum. 15:196–198. 1959.
|
|
61
|
Bapat SA, Mali AM, Koppikar CB and Kurrey
NK: Stem and progenitor-like cells contribute to the aggressive
behavior of human epithelial ovarian cancer. Cancer Res.
65:3025–3029. 2005.
|
|
62
|
Prager BC, Xie Q, Bao S and Rich JN:
Cancer Stem cells: The architects of the tumor ecosystem. Cell Stem
Cell. 24:41–53. 2019.
|
|
63
|
Li Y, Hu X, Lin R, Zhou G, Zhao L, Zhao D,
Zhang Y, Li W, Zhang Y, Ma P, et al: Single-cell landscape reveals
active cell subtypes and their interaction in the tumor
microenvironment of gastric cancer. Theranostics. 12:3818–3833.
2022.
|
|
64
|
Pei J, Tian X, Yu C, Luo J, Zhang J, Hua Y
and Wei G: GPX3 and GSTT1 as biomarkers related to oxidative stress
during renal ischemia reperfusion injuries and their relationship
with immune infiltration. Front Immunol. 14:11361462023.
|
|
65
|
Liou GY: CD133 as a regulator of cancer
metastasis through the cancer stem cells. Int J Biochem Cell Biol.
106:1–7. 2019.
|
|
66
|
Liu Q, Jin J, Ying J, Sun M, Cui Y, Zhang
L, Xu B, Fan Y and Zhang Q: Frequent epigenetic suppression of
tumor suppressor gene glutathione peroxidase 3 by promoter
hypermethylation and its clinical implication in clear cell renal
cell carcinoma. Int J Mol Sci. 16:10636–10649. 2015.
|
|
67
|
Wang YC, Bai MY, Yeh YT, Tang SL and Yu
MH: CD133 targeted PVP/PMMA Microparticle incorporating levamisole
for the treatment of ovarian cancer. Polymers (Basel).
12:4792020.
|
|
68
|
Abdellateif MS, Bayoumi AK and Mohammed
MA: c-Kit receptors as a therapeutic target in cancer: Current
insights. Oncot Targets Ther. 16:785–799. 2023.
|
|
69
|
Jiang YX, Siu MK, Wang JJ, Mo XT, Leung
TH, Chan DW, Cheung AN, Ngan HY and Chan KK: Ascites-derived
ALDH+CD44+ tumour cell subsets endow stemness, metastasis and
metabolic switch via PDK4-mediated STAT3/AKT/NF-κB/IL-8 signalling
in ovarian cancer. Br J Cancer. 123:275–287. 2020.
|
|
70
|
Robinson M, Gilbert SF, Waters JA,
Lujano-Olazaba O, Lara J, Alexander LJ, Green SE, Burkeen GA,
Patrus O, Sarwar Z, et al: Characterization of SOX2, OCT4 and NANOG
in ovarian cancer tumor-initiating cells. Cancers (Basel).
13:2622021.
|
|
71
|
Fan J, To KKW, Chen ZS and Fu L: ABC
transporters affects tumor immune microenvironment to regulate
cancer immunotherapy and multidrug resistance. Drug Resist Updat.
66:1009052023.
|
|
72
|
Kaipio K, Chen P, Roering P, Huhtinen K,
Mikkonen P, Östling P, Lehtinen L, Mansuri N, Korpela T, Potdar S,
et al: ALDH1A1-related stemness in high-grade serous ovarian cancer
is a negative prognostic indicator but potentially targetable by
EGFR/mTOR-PI3K/aurora kinase inhibitors. J Pathol. 250:159–169.
2020.
|
|
73
|
Saga Y, Ohwada M, Suzuki M, Konno R,
Kigawa J, Ueno S and Mano H: Glutathione peroxidase 3 is a
candidate mechanism of anticancer drug resistance of ovarian clear
cell adenocarcinoma. Oncol Rep. 20:1299–1303. 2008.
|
|
74
|
Xia J, Zhang J, Wu X, Du W, Zhu Y, Liu X,
Liu Z, Meng B, Guo J, Yang Q, et al: Blocking glycine utilization
inhibits multiple myeloma progression by disrupting glutathione
balance. Nat Commun. 13:40072022.
|
|
75
|
Chen B, Jiang K, Wang H, Miao L, Lin X,
Chen Q, Jing L and Lu X: NOTCH pathway genes in ovarian cancer:
Clinical significance and associations with immune cell
infiltration. Front Biosci. 28:2202023.
|
|
76
|
Rezaei F, Farhat D, Gursu G, Samnani S and
Lee JY: Snapshots of ABCG1 and ABCG5/G8: A Sterol's Journey to
cross the cellular membranes. Int J Mol Sci. 24:4842022.
|
|
77
|
Wang H, Zhou Y, Zhang S, Qi Y and Wang M:
PRPF6 promotes metastasis and paclitaxel resistance of ovarian
cancer via SNHG16/CEBPB/GATA3 axis. Oncol Res. 29:275–289.
2021.
|
|
78
|
Liu J, Wang H, Xiao S, Zhang S, Qi Y and
Wang M: Circ-0000231 promotes paclitaxel resistance in ovarian
cancer by regulating miR-140/RAP1B. Am J Cancer Res. 13:872–885.
2023.
|
|
79
|
Motohara T, Masuda K, Morotti M, Zheng Y,
El-Sahhar S, Chong KY, Wietek N, Alsaadi A, Carrami EM, Hu Z, et
al: Correction to: An evolving story of the metastatic voyage of
ovarian cancer cells: cellular and molecular orchestration of the
adipose-rich metastatic microenvironment. Oncogene. 41:3584.
2022.
|
|
80
|
Shishido A, Mori S, Yokoyama Y, Hamada Y,
Minami K, Qian Y, Wang J, Hirose H, Wu X, Kawaguchi N, et al:
Mesothelial cells facilitate cancer stem-like properties in
spheroids of ovarian cancer cells. Oncol Rep. 40:2105–2114.
2018.
|
|
81
|
Schoutrop E, Moyano-Galceran L, Lheureux
S, Mattsson J, Lehti K, Dahlstrand H and Magalhaes I: Molecular,
cellular and systemic aspects of epithelial ovarian cancer and its
tumor microenvironment. Semin Cancer Biol. 86:207–223. 2022.
|
|
82
|
Liu T, Sun L, Zhang Y, Wang Y and Zheng J:
Imbalanced GSH/ROS and sequential cell death. J Biochem Mol
Toxicol. 36:e229422022.
|
|
83
|
Zhou J, Jiang YY, Chen H, Wu YC and Zhang
L: Tanshinone I attenuates the malignant biological properties of
ovarian cancer by inducing apoptosis and autophagy via the
inactivation of PI3K/AKT/mTOR pathway. Cell Prolif.
53:e127392020.
|
|
84
|
Moulder R, Välikangas T, Hirvonen MK,
Suomi T, Brorsson CA, Lietzén N, Bruggraber SFA, Overbergh L,
Dunger DB, Peakman M, et al: Targeted serum proteomics of
longitudinal samples from newly diagnosed youth with type 1
diabetes distinguishes markers of disease and C-peptide trajectory.
Diabetologia. 66:1983–1996. 2023.
|
|
85
|
Schreckenberger ZJ, Wenceslau CF, Joe B
and McCarthy CG: Mitophagy in Hypertension-associated premature
vascular aging. Am J Hypertens. 33:804–812. 2022.
|
|
86
|
Xu H, Zhao F, Wu D, Zhang Y, Bao X, Shi F,
Cai Y and Dou J: Eliciting effective tumor immunity against ovarian
cancer by cancer stem cell vaccination. Biomed Pharmacother.
161:1145472023.
|
|
87
|
Qian J, LeSavage BL, Hubka KM, Ma C,
Natarajan S, Eggold JT, Xiao Y, Katherine CF, Krishnan V, Enejder
A, et al: Cancer-associated mesothelial cells promote ovarian
cancer chemoresistance through paracrine osteopontin signaling. J
Clin Invest. 131:e1461862021.
|
|
88
|
Byrne P, Demasi M, Jones M, Smith SM,
O'Brien KK and DuBroff R: Evaluating the association between
low-density lipoprotein cholesterol reduction and relative and
absolute effects of statin treatment: A systematic review and
Meta-analysis. JAMA Intern Med. 182:4742022.
|
|
89
|
Tesfay L, Paul BT, Konstorum A, Deng Z,
Cox AO, Lee J, Furdui CM, Hegde P, Torti FM and Torti SV:
Stearoyl-CoA Desaturase 1 protects ovarian cancer cells from
ferroptotic cell death. Cancer Res. 79:5355–5366. 2019.
|
|
90
|
Mukherjee A, Chiang CY, Daifotis HA,
Nieman KM, Fahrmann JF, Lastra RR, Romero IL, Fiehn O and Lengyel
E: Adipocyte-induced FABP4 expression in ovarian cancer cells
promotes metastasis and mediates carboplatin resistance. Cancer
Res. 80:1748–1761. 2020.
|
|
91
|
Luis G, Godfroid A, Nishiumi S, Cimino J,
Blacher S, Maquoi E, Wery C, Collignon A, Longuespée R,
Montero-Ruiz L, et al: Tumor resistance to ferroptosis driven by
Stearoyl-CoA Desaturase-1 (SCD1) in cancer cells and fatty acid
biding protein-4 (FABP4) in tumor microenvironment promote tumor
recurrence. Redox Biol. 43:1020062021.
|
|
92
|
Bhardwaj M, Lee JJ, Versace AM, Harper SL,
Goldman AR, Crissey MAS, Jain V, Singh MP, Vernon M, Aplin AE, et
al: Lysosomal lipid peroxidation regulates tumor immunity. J Clin
Invest. 133:e1645962023.
|
|
93
|
Szczuko M, Zapalowska-Chwyć M and Drozd R:
A low glycemic index decreases inflammation by increasing the
concentration of uric acid and the activity of glutathione
peroxidase (GPx3) in patients with polycystic ovary syndrome
(PCOS). Molecules. 24:15082019.
|
|
94
|
Kim WY: Therapeutic targeting of lipid
synthesis metabolism for selective elimination of cancer stem
cells. Arch Pharmacal Res. 42:25–39. 2019.
|
|
95
|
Bhat AA, Nisar S, Singh M, Ashraf B,
Masoodi T, Prasad CP, Sharma A, Maacha S, Karedath T, Hashem S, et
al: Cytokine- and chemokine-induced inflammatory colorectal tumor
microenvironment: Emerging avenue for targeted therapy. Cancer
Commun. 42:689–715. 2022.
|
|
96
|
Cervellati C, Trentini A, Rosta V, Zuliani
G, Vieceli Dalla Sega F, Fortini F, Rizzo P, Cimaglia P and Campo
G: A Nutraceutical compound containing a low dose of Monacolin K,
Polymethoxyflavones, phenolic acids, flavonoids, and hydroxytyrosol
improves HDL functionality. Curr Vasc Pharmacol. 21:433–442.
2023.
|
|
97
|
Disis ML, Taylor MH, Kelly K, Beck JT,
Gordon M, Moore KM, Patel MR, Chaves J, Park H, Mita AC, et al:
Efficacy and Safety of Avelumab for patients with recurrent or
refractory ovarian cancer: Phase 1b results from the JAVELIN Solid
tumor trial. JAMA Oncol. 5:393–401. 2019.
|
|
98
|
Binnewies M, Pollack JL, Rudolph J, Dash
S, Abushawish M, Lee T, Jahchan NS, Canaday P, Lu E, Norng M, et
al: Targeting TREM2 on tumor-associated macrophages enhances
immunotherapy. Cell Rep. 37:1098442021.
|
|
99
|
El-Arabey AA, Alkhalil SS, Al-Shouli ST,
Awadalla ME, Alhamdi HW, Almanaa TN, Mohamed SSEM and Abdalla M:
Revisiting macrophages in ovarian cancer microenvironment:
Development, function and interaction. Med Oncol. 40:1422023.
|
|
100
|
Soerens AG, Künzli M, Quarnstrom CF, Scott
MC, Swanson L, Locquiao JJ, Ghoneim HE, Zehn D, Youngblood B, Vezys
V and Masopust D: Functional T cells are capable of supernumerary
cell division and longevity. Nature. 614:762–766. 2023.
|
|
101
|
Zeng XY, Xie H, Yuan J, Jiang XY, Yong JH,
Zeng D, Dou YY and Xiao SS: M2-like tumor-associated
macrophages-secreted EGF promotes epithelial ovarian cancer
metastasis via activating EGFR-ERK signaling and suppressing lncRNA
LIMT expression. Cancer Biol Ther. 20:956–966. 2019.
|
|
102
|
Efimova I, Catanzaro E, Van Der Meeren L,
Turubanova VD, Hammad H, Mishchenko TA, Vedunova MV, Fimognari C,
Bachert C, Coppieters F, et al: Vaccination with early ferroptotic
cancer cells induces efficient antitumor immunity. J Immuno Ther
Cancer. 8:e0013692020.
|
|
103
|
Cannarile MA, Weisser M, Jacob W, Jegg AM,
Ries CH and Rüttinger D: Colony-stimulating factor 1 receptor
(CSF1R) inhibitors in cancer therapy. J Immunother Cancer.
5:532017.
|
|
104
|
Hartwell BL, Melo MB, Xiao P, Lemnios AA,
Li N, Chang JYH, Yu J, Gebre MS, Chang A, Maiorino L, et al:
Intranasal vaccination with lipid-conjugated immunogens promotes
antigen transmucosal uptake to drive mucosal and systemic immunity.
Sci Transl Med. 14:eabn14132022.
|
|
105
|
Subhankar B, Priyanka S, Bilash C and Amit
KS: Chemokines driven ovarian cancer progression, metastasis and
chemoresistance: Potential pharmacological targets for cancer
therapy. Semin Cancer Biol. 86:568–579. 2022.
|
|
106
|
Muñoz-Galván S and Carnero A: Targeting
cancer stem cells to overcome therapy resistance in ovarian cancer.
Cells. 9:14022020.
|
|
107
|
Schweer D, McAtee A, Neupane K, Richards
C, Ueland F and Kolesar J: Tumor-associated macrophages and ovarian
cancer: Implications for therapy. Cancers. 14:22202022.
|
|
108
|
Kim D, Choi B, Ryoo I and Kwak MK: High
NRF2 level mediates cancer stem cell-like properties of
aldehydemdehydrogenase (ALDH)-high ovarian cancer cells: Inhibitory
role of all-trans retinoic acid in ALDH/NRF2 signaling. Cell Death
Dis. 9:8962018.
|
|
109
|
Wang J, Hu K, Cai X, Yang B, He Q, Wang J
and Weng Q: Targeting PI3K/AKT signaling for treatment of
idiopathic pulmonary fibrosis. Acta Pharm Sin B. 12:18–32.
2022.
|
|
110
|
Alwosaibai K, Aalmri S, Mashhour M,
Ghandorah S, Alshangiti A, Azam F, Selwi W, Gharaibeh L, Alatawi Y,
Alruwaii Z and Alsaab HO: PD-L1 is highly expressed in ovarian
cancer and associated with cancer stem cells populations expressing
CD44 and other stem cell markers. BMC Cancer. 23:132023.
|
|
111
|
Laumont CM, Wouters MCA, Smazynski J,
Gierc NS, Chavez EA, Chong LC, Thornton S, Milne K, Webb JR, Steidl
C and Nelson BH: Single-cell profiles and prognostic impact of
tumor-infiltrating lymphocytes Coexpressing CD39, CD103, and PD-1
in ovarian cancer. Clin Cancer Res. 27:4089–4100. 2021.
|
|
112
|
Zhou Y, Wang M, Shuang T, Liu Y, Zhang Y
and Shi C: MiR-1307 influences the chemotherapeutic sensitivity in
ovarian cancer cells through the regulation of the CIC
transcriptional repressor. Pathol Res Pract. 215:1526062019.
|
|
113
|
Zhao G, Tan Y, Cardenas H, Vayngart D,
Wang Y, Huang H, Keathley R, Wei JJ, Ferreira CR, Orsulic S, et al:
Ovarian cancer cell fate regulation by the dynamics between
saturated and unsaturated fatty acids. Proc Natl Acad Sci.
119:e22034801192022.
|
|
114
|
Nunes SC and Serpa J: Glutathione in
ovarian cancer: A double-edged Sword. Int J Mol Sci.
19:18822018.
|
|
115
|
O'Shea AS: Clinical staging of ovarian
cancer. Methods Mol Bio. 2424:3–10. 2022.
|
|
116
|
Ozols RF, Bundy BN, Greer BE, Fowler JM,
Clarke-Pearson D, Burger RA, Mannel RS, DeGeest K, Hartenbach EM
and Baergen R; Gynecologic Oncology Group: Phase III trial of
carboplatin and paclitaxel compared with cisplatin and paclitaxel
in patients with optimally resected stage III ovarian cancer: A
Gynecologic Oncology Group study. J Clin Oncol. 21:3194–3200.
2003.
|
|
117
|
Wang K, Guan C, Shang X, Ying X, Mei S,
Zhu H, Xia L and Chai Z: A bioinformatic analysis: the
overexpression and clinical significance of FCGBP in ovarian
cancer. Aging (Albany NY). 13:7416–7429. 2021.
|
|
118
|
Shan T, Chen S, Chen X, Wu T, Yang Y, Li
S, Ma J, Zhao J, Lin W, Li W, et al: M2-TAM subsets altered by
lactic acid promote T-cell apoptosis through the PD-L1/PD-1
pathway. Oncol Rep. 44:1885–1894. 2020.
|
|
119
|
Chen L, Luo J, Zhang J, Wang S, Sun Y, Liu
Q and Cheng C: Dual targeted nanoparticles for the codelivery of
doxorubicin and siRNA cocktails to overcome ovarian cancer stem
cells. Int J Mol Sci. 24:115752023.
|
|
120
|
Koeberle SC, Kipp AP, Stuppner H and
Koeberle A: Ferroptosis-modulating small molecules for targeting
drug-resistant cancer: Challenges and opportunities in manipulating
redox signaling. Med Res Rev. 43:614–682. 2023.
|
|
121
|
Kho BG, Park HY, Cho HJ, Park CK, Kim YC,
Yun JS, Song SY, Na KJ, Choi YD, Lee SW and Oh IJ: Glutathione
peroxidase 3 as a biomarker of recurrence after lung cancer
surgery. J Clin Med. 9:38012020.
|
|
122
|
Ngoi NY, Syn NL, Goh RM, Goh BC, Huang
RYJ, Soon YY, James E, Cook A, Clamp A and Tan DS: Weekly versus
tri-weekly paclitaxel with carboplatin for first-line treatment in
women with epithelial ovarian cancer. Cochrane Db Syst Rev.
2022:CD0120072022.
|