Introduction
Sperm-associated antigen 6 (SPAG6), also known as
Repro-SA-1, is a homolog of Paramecium falciforme paralytic
flagellum 16 (PF16). It is located in the axonemal center and is a
microtubule-associated protein (1). SPAG6 has several functions,
including sperm acrosome formation; ciliary/flagellar movement;
immune synapse formation and function; neuronal proliferation and
differentiation; fibroblast morphology, growth and migration; and
middle ear and Eustachian tube epithelial cell function. It belongs
to the cancer/testis antigen (CTA) family and its expression is
associated with various cancers. It may also represent a tumor
prognostic marker and therapeutic target (2).
This review involved a systematic literature search
to comprehensively gather publications focused on the physiological
functions of SPAG6 and its role in oncology. The databases searched
included PubMed, Web of Science and other knowledge service
platforms, covering the time period from each database's inception
to June 2025. The retrieved literature was screened and data were
extracted based on clearly defined inclusion and exclusion
criteria. This systematic approach aimed to elucidate the molecular
functions of SPAG6 and its mechanisms in tumor initiation and
progression and provide a foundation for subsequent comprehensive
analyses.
Molecular structure and function of
SPAG6
SPAG6 was first identified by Neilson et
al (3) in 1999. They screened
a cDNA library from the testes of infertile men exhibiting
high-titer, anti-sperm autoantibodies in the serum and discovered
that SPAG6 encodes a new antigen. This gene was previously
known by several names, including Repro-SA-1, CT141
and RP11301N24.4; however, the HUGO Gene Nomenclature
Committee (https://www.genenames.org/data/gene-symbol-report/#!/hgnc_id/HGNC:11215)
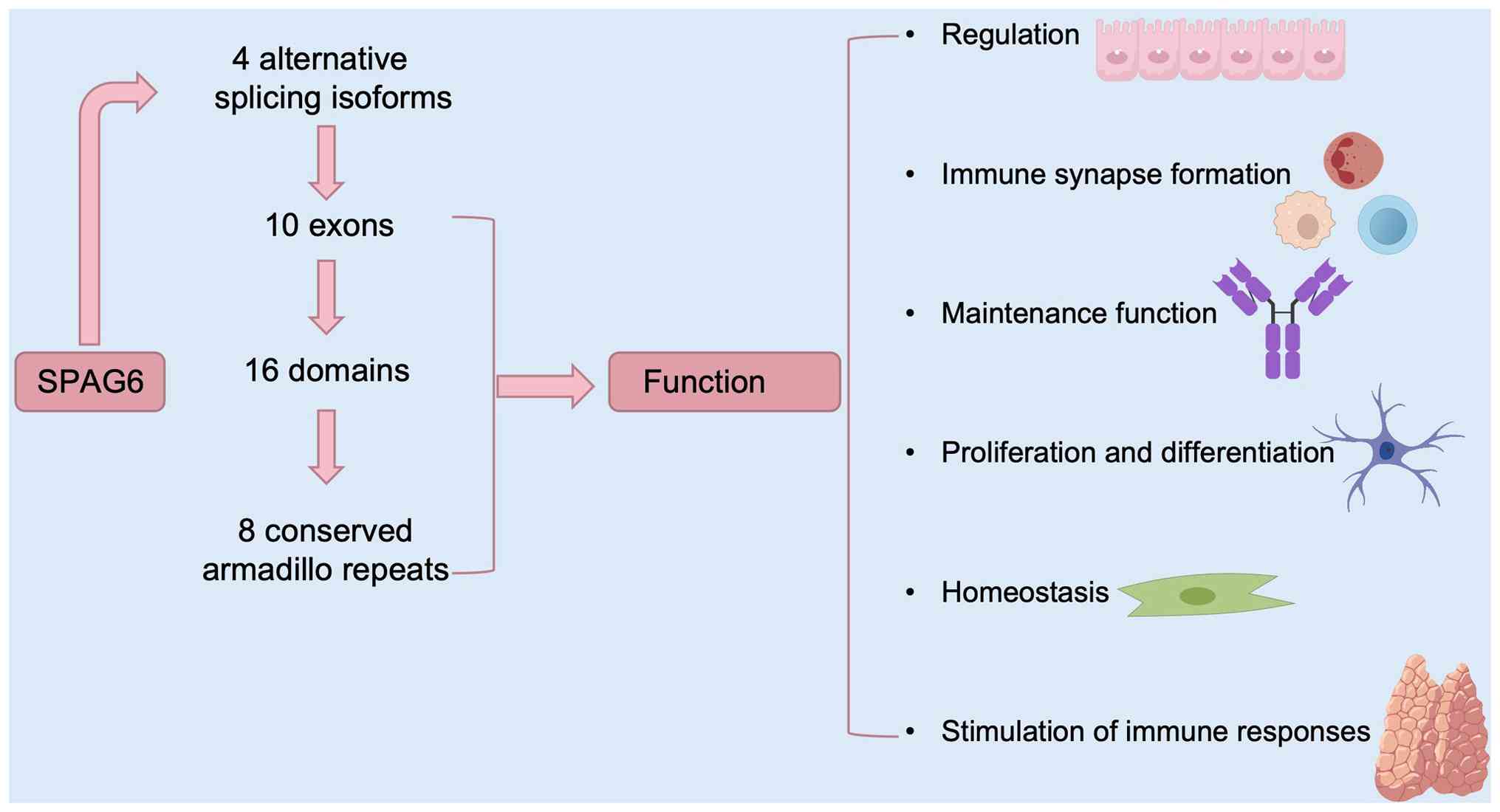
approved the official symbol SPAG6. It is located on the
10p12.2 region of human chromosome 10 and encodes four alternative
splicing isoforms. The full-length transcript consists of 10 exons
and the translated protein has 16 domains, including 8 conserved
armadillo repeats, which play a role in mediating protein-protein
interactions (4,5). SPAG6 is primarily expressed in
tissues containing ciliated cells, such as testicular germ cells,
lung tissue, nervous system and inner ear; however, it is not
present in the prostate, spleen, thymus, small intestine, colon,
peripheral blood leukocytes, heart, placenta, liver, muscles,
kidneys and pancreas. Thus, it is considered a member of the CTA
family and may serve as a tumor prognostic biomarker and
therapeutic target (2,6). SPAG6 encodes a
microtubule-associated protein that attenuates cell growth,
differentiation, migration and cell polarity regulation (7,8).
It may also represent a novel tumor serum biomarker, which supports
its inclusion as a member of the CTA family and suggests that its
transcripts are targets for immunotherapy. Abnormal SPAG6
expression or function is associated with the development of
various solid tumors, including breast and lung cancers (2,9).
The role of SPAG6 has also been reported in hematological
malignancies. Patients with acute myeloid leukemia (AML), lymphoma,
myeloproliferative neoplasms (MPNs) or myelodysplastic syndromes
(MDSs) often exhibit high and sustained SPAG6 expression,
which is significantly associated with poor outcomes (10-13).
SPAG6 regulates various physiological
functions
SPAG6 plays a role in constructing and maintaining
the cytoskeleton (2,14,15). The amino acid sequence derived
from the full-length human cDNA is highly homologous to the product
of the PF16 site in Chlamydomonas reinhardtii. The PF16
protein is localized to the central pair structure of the flagellar
axoneme, which consists of a pair of central microtubules, nine
sets of peripheral dyads and kinesin arms attached (16-18). As a CTA, PF16 exhibits
tissue-specific expression. It is frequently expressed in
immune-privileged tissues, such as the testis; however, it is
abnormally activated in tumor tissues. SPAG6 can induce spontaneous
humoral and cellular immune responses and is relatively safe in
normal tissues, indicating that it is a suitable candidate for
tumor immunotherapy (19,20). Under physiological conditions,
SPAG6 is frequently expressed in cell types with ciliary
structures, such as sperm cells, neural tissue, the inner ear and
respiratory epithelial cells. It regulates microtubule/cytoskeletal
dynamics and their resulting cell function (e.g., sperm maturation
and neural system development) through binding to microtubule
proteins (21-23). Notably, SPAG6 function is not
limited to ciliated cells, as it also plays an important role in
nonciliated cells. A study revealed its novel role in the mouse
vestibular system. SPAG6 deficiency leads to vestibular
dysfunction, abnormal ultrastructure of vestibular hair cells and
accelerated apoptosis (24).
Furthermore, it is involved in regulating neuronal migration,
developmental differentiation and neurogenesis, which emphasizes
its role in the nervous system. Because of their functional
similarities, including signal transduction and polarity
maintenance, immune synapses and cilia are closely related. Because
of the central role of SPAG6 in ciliary movement, it may also be
involved in immune regulatory processes (25,26).
SPAG6 regulates ciliary/flagellar
movement, ciliary formation and axoneme orientation and establishes
polarity in tracheal epithelial cells
SPAG6 exhibits functions similar to those of
homologous proteins in Chlamydomonas, such as regulating
ciliary/flagellar movement (27-29). Hu et al (30) reported that SPAG6 ameliorates
damage caused by brain edema following cerebral ischemic stroke
reperfusion by maintaining the structure and function of motile
cilia, attenuating the phosphoinositide 3-kinase (PI3K)/protein
kinase B (AKT)-mechanistic target of rapamycin (mTOR) signaling
pathway, and inhibiting inflammatory and autophagic responses. Mice
lacking SPAG6 do not survive to adulthood because of
hydrocephalus. Male survivors lose their reproductive capacity
because of impaired ciliary/flagellar motility and ultrastructural
abnormalities in the sperm axonemes. Compared with wild-type mice,
SPAG6 knockout mice show bronchial epithelial ciliary
dysfunction. In addition, ciliary formation defects occur in the
ventricular ependymal, middle ear and tracheal epithelial tissues
(17). Teves et al
(7) demonstrated that
SPAG6 knockout mice display a disorganized ciliary
arrangement and reduced density in tracheal epithelial cells, along
with decreased ciliary beating frequency and irregular rhythms.
Furthermore, the number of axonemes in epithelial cells is
significantly reduced, whereas the orientation of the central
microtubule pairs is random. These abnormal changes in ciliary
structure and function may be associated with disrupted microtubule
distribution, which results in misdirected axoneme/basal body
orientation and disrupted epithelial cell polarity.
SPAG6 regulates the formation and
function of immune synapses
In immune cells without cilia, SPAG6 acts through an
alternative mechanism because the formation of immune synapses and
cilia involves the same processes. The microtubule-organizing
center is a subcellular organelle responsible for forming and
organizing microtubules. In eukaryotic cells, it usually refers to
the centrosome, which is comprised of a pair of orthogonally
arranged centrioles (i.e., parent and daughter centrioles)
(31). When antigen-presenting
and effector cells undergo homologous recognition to initiate an
immune response, the centrosomes, actin cytoskeleton, Golgi
apparatus and secretory vesicles within the effector cells relocate
and aggregate at the immune synapse site. This relocation of
subcellular organelles promotes receptor-ligand interactions and
results in the release of cytokines to their target sites (26). Similarly, during targeted killing
by effector cells, the centrosome becomes re-oriented and docks
with the synapse membrane to form a synapse gap and releases lytic
enzymes that destroy the target cells (21,24). Cooley et al (25) found that SPAG6 regulates the
function of lymphocyte centrosomes and is expressed in primary and
secondary lymphoid tissues. SPAG6 defects in mice result in
a dysregulated synapse cleft, centrosome polarization and actin
clearance. The abnormal synapse formation observed in
SPAG6-deficient mice may be associated with impaired
cytotoxic T-cell function and humoral immune responses. This
manifests as weakened germinal center responses, fewer follicular
CD4+ T cells, defects in antibody class switching and abnormal B1B
cell proliferation.
SPAG6 plays a role in the functional
regulation of epithelial cells in the middle ear and Eustachian
tube
The epithelial tissue associated with the middle ear
and Eustachian tube consists of multiciliated cells (MCCs) and
non-MCCs. It is associated with otitis media with effusion
(32). MCCs contain hundreds of
cilia on their surface, and their coordinated beating facilitates
the transport of secretions from the middle ear cavity to the
nasopharynx through the Eustachian tube. Effective mucociliary
clearance requires consistent ciliary orientation within and
between cells and along the tissue axis (32). Abnormal ciliary function occurs in
primary ciliary dyskinesia or Kartagener syndrome and results in
middle ear effusion and inflammatory responses (33,34). Studies have demonstrated that
SPAG6 deletion causes hearing loss in mice. potentially by
regulating prestin expression (35). SPAG6 may affect hearing by
regulating prestin expression, whereas its deletion causes otitis
media in mice. Additionally, SPAG6 is expressed in the cilia
of the middle ear epithelium in mice, and its targeted mutation can
lead to pathological changes in the middle ear, which are
attributed to ciliary dysfunction (36,37). SPAG6 mutations disrupt
polarity maintenance in the middle ear epithelial cells, which
results in abnormal ciliary movement and reduced fluid and mucus
transport efficiency. This disrupts the balance between mucus
secretion and clearance, which ultimately causes middle ear
effusion and otitis media (38).
With respect to auditory function, cylindrical outer hair cells
(OHCs) in the organ of Corti of the mammalian cochlear detect
receptors (39). Wang et
al (39) observed SPAG6
expression in OHCs and found that it was bound to
microtubule-associated protein 1, which jointly stabilizes the
dynein structure. This suggests that SPAG6 is indispensable for
maintaining the normal physiological function of OHCs. Furthermore,
vestibular and auditory functions collaborate through shared
ciliary structures and neural pathways (the vestibulocochlear
nerve) within the hair cells of the inner ear to enable spatial
localization and perception. Li et al (24) generated SPAG6-deficient mice and
showed that its mutants exhibit vestibular disorders associated
with abnormal ultrastructural changes in the vestibular hair and
Scarpa ganglion cells of the inner ear. The changes included
swollen microvilli and reduced mitochondrial cristae. This suggests
that microtubule stability is regulated by SPAG6 and is
essential for vestibular function.
SPAG6 regulates neuronal proliferation
and differentiation processes
Normal development of the mammalian brain relies on
the coordination of the proliferative and differentiation
activities of neural progenitor cells (NPCs) (40,41). Disruption of this process results
in an abnormal number of neurons, which can lead to neurological
disorders, such as epilepsy, autism spectrum disorders and
intellectual developmental delays (42-44). Studies on chicken embryo
development indicate that SPAG6 is primarily expressed in the
ventral ventricular zone of the spinal cord (adjacent to the basal
plate region) (45). SPAG6
knockout mice have enlarged brains and reduced body size and
experience premature death because of severe hydrocephalus
(17). Thus, SPAG6 may be
involved in regulating cell proliferation and division.
Furthermore, ventricular enlargement accompanied by cortical plate
thinning (46), along with the
aforementioned cranial volume abnormalities and hydrocephalus
phenotype, suggests that SPAG6 plays an important regulatory role
in the ciliary movement function of the ependymal layer. Armadillo
repeat domain-containing proteins contribute to neural cell
division processes and related pathological mechanisms by
regulating microtubule assembly and spindle formation (38,47,48). SPAG6 is expressed in the
microtubules of COS-1 cells and plays a role in neural development
and differentiation (16). Hu
et al (49) showed that
SPAG6-overexpressing cells preferentially differentiate into
neurons. SPAG6 overexpression inhibits the proliferative activity
of NPCs, promoting their differentiation toward a neuronal lineage
while suppressing astrocyte generation. Yan et al (22) demonstrated that SPAG6
overexpression reduces neuronal migration rates and inhibits axonal
branching and extension, suggesting that it regulates neurogenesis
by stabilizing microtubule structures and inhibiting excessive
remodeling. The expansion capacity and differentiation orientation
of NPCs together determine the number of neurons produced during
brain development, which ultimately influences brain volume and
cortical thickness (50).
Cortical plate thinning observed in SPAG6-deficient mice may result
from the disruption of the balance between NPC proliferation and
differentiation. Mitchell et al (51) proposed that SPAG6 regulates
neuronal migration by targeting microtubule regulation and
primarily controls centrosome localization and somatic movement,
which further indicates the role of SPAG6 in neural development
(52). Using SPAG6-deficient mice
and mammalian spiral ganglion neuron (SGN) explants, studies have
demonstrated that the absence of SPAG6 affects neurite and growth
cone growth (1). Furthermore,
SPAG6 deficiency decreased synaptic density in SGN explants and
increased the sensitivity of SPAG6-mutant SGNs to the microtubule
stabilizer paclitaxel. These results suggest that SPAG6 contributes
to the development and function of SGNs. SPAG6 promoter methylation
is increased during the in vitro differentiation of human
embryonic stem cells into NPCs or stem cells, suggesting that its
expression is subject to stage-specific epigenetic regulation
during neurogenesis (53).
SPAG6 regulates the morphology, growth,
migration and cilia formation of fibroblasts
Primary mouse embryonic fibroblasts (MEFs) were
isolated and cultured from SPAG6 knockout and wild-type
mouse embryos (8). Compared with
wild-type MEFs, SPAG6-deficient MEFs showed various
morphological abnormalities, including generalized enlargement of
cell volume, nuclear enlargement and aggregation of vesicles in the
cytoplasm. Re-introducing SPAG6 reversed these
abnormalities. In addition, the deficient cells had slower growth
rates and reduced motility. Microtubule acetylation, which is an
important post-translational modification of microtubules (54), is significantly reduced in
SPAG6-deficient MEFs. The reduction in acetylation disrupts
the functional integrity of microtubules, yielding phenotypic
changes, including the inhibition of cell proliferation, defects in
migration, adhesion abnormalities, mitotic defects and impairment
of cilia formation. This mechanism may explain the increase in
cytoplasmic vesicles and reduced transfection efficiency observed
in SPAG6-deficient MEFs (8). A previous study confirmed that the
degree of microtubule acetylation is positively associated with
transfection efficiency (55).
The physiological function of SPAG6 is illustrated in Fig. 1 and the molecular mechanisms are
presented in Table I.
 | Table IPhysiological functions and molecular
mechanisms of sperm-associated antigen 6. |
Table I
Physiological functions and molecular
mechanisms of sperm-associated antigen 6.
| Physiological
function | Molecular
mechanisms | (Refs.) |
|---|
| Tumor
immunogenicity | Cancer/testis
antigen that stimulates immune responses
Abnormally high expression in tumor tissues, silent in normal
tissues | (10-13,19,20) |
| Regulation of
ciliary/flagellar movement | Maintains the
integrity of the axoneme microtubule structure
Mediates ciliary-directed beating and coordination
Regulates ciliogenesis | (7,17,27-29) |
| Immune synapse
formation | Regulates
microtubule-organizing center (centriole) relocation to
synapses
Maintains actin clearance
Mediates the targeted release of cytotoxic granules | (21,24,26) |
| Middle ear mucus
clearance | Establishes
polarity of multi-ciliated cells
Ensures ciliary-directed transport function | (32,36,38) |
| Auditory and
vestibular function | Stabilizes dynein
by binding to MAP1
Maintains the ultrastructure of outer hair cells
Inhibition of ciliated vestibular cells
Ciliated cell apoptosis | (24,39) |
| Neurodevelopmental
regulation | Stabilization of
microtubule structure (inhibition of excessive
remodeling)
Regulation of neural progenitor cell, proliferation/differentiation
balance
Coordination of neuronal migration (centriole-guided) | (22,46,49,50) |
| Fibroblast
homeostasis | Promotion of
microtubule protein acetylation
Maintenance of cell morphology/motility
Regulation of ciliogenesis and vesicle transport | (8,54,55) |
Role of SPAG6 in tumors
CTAs are a family of antigens that are only
expressed in testicular and placental tissues. They are abnormally
activated in various tumor tissues while maintaining
tissue-specific expression (56).
CTAs are present in human immune-privileged tissues and specific
tumor lesions. They elicit spontaneous humoral and cellular immune
responses without harming normal tissues; thus, they are ideal
candidates for tumor immunotherapy. CTAs are closely associated
with tumor cell proliferation, metastasis, invasion, disease
recurrence and poor prognosis (57-59). SPAG6 encodes a
microtubule-associated protein involved in cell growth,
differentiation, migration and polarity regulation. It is a novel
CTA with potential as a tumor serum biomarker, which confirms its
inclusion in the CTA family, and it is a promising candidate for
tumor immunotherapy. SPAG6 abnormalities are closely associated
with hematological malignancies and various solid tumors, including
breast and lung cancer (2,9).
Role of SPAG6 in hematological
malignancies
Studies have confirmed that SPAG6 has the potential
to serve as a prognostic marker and therapeutic target for
hematological malignancies.
MDS
MDS is a clonal hematopoietic stem cell disorder
characterized by significant heterogeneity (60). This disease involves ineffective
hematopoiesis, cytopenias, morphological developmental
abnormalities and transformation to AML (61). Li et al (62) used SPAG6-short hairpin RNA
lentiviral vectors to knock down SPAG6 in SKM-1 cells, which
resulted in the activation of the tumor necrosis factor-related
apoptosis-inducing ligand (TRAIL) signaling pathway, indicating
that SPAG6 attenuates apoptosis by modulating the TRAIL
pathway. Yin et al (63)
found that SPAG6 silencing in SKM-1 cells increases
phosphatase and tensin homolog deleted on chromosome 10
(PTEN) expression, which induces apoptosis through the
PI3K/AKT pathway. Jiang et al (12) showed that SPAG6 mRNA levels
were significantly higher in bone marrow cells from patients with
MDS and MDS-AML than in those from healthy controls using reverse
transcription-quantitative PCR (RT-qPCR). In vitro
experiments revealed that SPAG6 knockdown inhibits SKM-1
cell proliferation, causes cell cycle arrests at the G1/S phase and
disrupts cell differentiation. Zhang et al (64) showed that SPAG6 silencing
induces autophagy through the AMP-activated protein kinase
(AMPK)/mTOR/unc-51 like autophagy activating kinase 1 (ULK1)
signaling pathway, thereby enhancing SKM-1 cell apoptosis.
Collectively, these studies suggest that SPAG6 contributes to MDS
pathogenesis and development, indicating its potential as a novel
therapeutic target. Luo et al (65) expanded on this understanding from
an epigenetic regulation perspective by showing that SPAG6
knockout, combined with the demethylating agent decitabine (DAC),
reduces the expression of DNA methyltransferases and
methyl-CpG-binding domain proteins. This combination also enhances
apoptosis induced by DAC and the histone deacetylase inhibitor
LBH589. A study from the same group, by Luo et al (66), demonstrated that suppressing
SPAG6 expression in SKM-1 cells enhances DAC-induced
apoptosis and promotes PTEN demethylation. Overall, these
results support SPAG6 as a target for demethylation therapy in
MDS.
AML
AML is an aggressive malignancy involving white
blood cells (67,68). It primarily manifests as symptoms
associated with bone marrow failure and organ infiltration
(69). Luo et al (70) reported that SPAG6
expression in patients with AML positively correlates with risk
stratification. Patients with high SPAG6 expression had shorter
overall survival than those with low SPAG6 expression.
Furthermore, SPAG6 knockdown in the HL60 AML cell line
promotes apoptosis and arrests the cell cycle at the G1 phase, thus
confirming it as a protumor factor in AML. Steinbach et al
(10) found that SPAG6 and
six other genes are highly overexpressed in pediatric patients with
AML but were normal in patients with sustained complete remission.
This suggests that SPAG6 promotes disease progression.
SPAG6 was considered a potential indicator for evaluating
treatment efficacy and predicting prognosis in pediatric AML
(71). Skou et al
(72) found that peripheral blood
levels of SPAG6 and two other genes predict relapse in
pediatric patients with AML and are useful for minimal residual
disease (MRD) monitoring in patients lacking leukemia-specific
targets. Mu et al (73)
identified SPAG6 as a significantly upregulated gene in AML.
Its overexpression was negatively correlated with disease
prognosis. SPAG6 interacts with and relocates myosin ID
(MYO1D) from the cytoplasm to the cell membrane. This
activates the PI3K/AKT signaling pathway and extracellular
signal-regulated kinase (ERK) pathway, thereby regulating AML
growth and prognosis. Thus, SPAG6 may represent a novel
therapeutic target for AML.
Adult B-cell acute lymphoblastic
leukemia (B-ALL)
B-ALL is a genetically heterogeneous malignancy
(74) that may be classified into
distinct molecular subtypes based on recurrent gene rearrangements,
chromosomal abnormalities or specific gene mutations (75). Zhao et al (76) initially reported SPAG6
overexpression in the bone marrow of adult patients with B-ALL,
which markedly decreased after treatment and complete remission.
Studies using lentiviral transfection to knock down SPAG6 in
the human B-ALL cell lines B-ALL-1 and NALM-6 showed significant
inhibition of cell proliferation and apoptosis. These results
indicate that SPAG6 downregulation attenuates cell
proliferation and apoptosis by modulating the transforming growth
factor-β (TGF-β)/Smad signaling pathway.
Multiple myeloma (MM)
MM is a hematologic malignancy characterized by the
malignant clonal proliferation of plasma cells (77). It accounts for >10% of all
hematopoietic malignancies (78).
Li et al (79) conducted a
bioinformatics analysis for plasma cell tumor tissues and bone
marrow samples from patients with MM. Significant SPAG6
expression was observed in MM cell lines, plasma cell tumor tissues
and patient bone marrow. Increased SPAG6 mRNA levels were
positively correlated with elevated hypercalcemia, increased plasma
cell proportion and the severity of skeletal infiltration.
Functional experiments further revealed that SPAG6
overexpression enhances MM cell proliferation, migration and
antiapoptotic capacity in vitro, whereas its downregulation
showed inhibitory effects. A direct interaction was confirmed
between SPAG6 and dual-specificity phosphatase 1 (DUSP1),
which attenuates the expression of downstream molecules in the
mitogen-activated protein kinase (MAPK)/ERK signaling pathway.
These results suggest that SPAG6 plays an important role in
MM development by attenuating the DUSP1-MAPK/ERK axis and may be an
effective therapeutic target.
BCR activator of RhoGEF and GTPase
(BCR)::ABL proto-oncogene 1, non-receptor tyrosine kinase
(ABL1)-negative MPNs
MPN is a malignant clonal disorder caused by somatic
mutations in hematopoietic stem/progenitor cells (80). It is characterized by the abnormal
increase in peripheral blood cell counts and bone marrow fibrosis
(81). Xia et al (82) showed that SPAG6 is
expressed by MPN cells at the mRNA and protein level, with the
highest expression observed in nucleated erythroid precursor cells
and megakaryocytes. They hypothesized that abnormal SPAG6
expression contributes to the development of MPN and suggested that
it may serve as a novel tumor marker for BCR::ABL1-negative MPN.
Conversely, Ding et al (83) observed the significant
upregulation of SPAG6 mRNA in primary MPN cells and
MPN-derived leukemia cell lines. In vitro studies revealed
that forced SPAG6 expression enhances clonogenic potential
and accelerates the G1-to-S phase transition. Conversely,
downregulating SPAG6 enhances interferon-α (IFN-α)-mediated
apoptosis promotion and cycle arrest through the signal transducer
and transcription activator 1 (STAT1) pathway. Furthermore, the
expression of SPAG6 protein decreased concomitantly following the
inhibition of STAT1 signaling.
Burkitt lymphoma (BL)
BL is a highly aggressive malignancy originating
from mature B cells (84). It is
characterized by distinct clinical and morphological features, a
germinal center B-cell immunophenotype, high proliferative activity
and MYC rearrangements involving the immunoglobulin gene
loci (85). Zhang et al
(11) demonstrated that
suppressing SPAG6 expression reduced the viability of Daudi
and Raji cells. Conversely, PTEN inhibition using small inhibitory
RNA or the specific PTEN inhibitor SF1670 restored proliferation
and promoted apoptosis induced by SPAG6 deficiency in vitro
and in vivo. These results suggest that SPAG6 promotes
proliferation and suppresses apoptosis in BL cells through the
PTEN/PI3K/AKT signaling pathway and further indicate that SPAG6
contributes to BL progression and may serve as a prognostic
biomarker for these patients. The mechanisms of action and clinical
significance of SPAG6 in various hematological tumor types are
summarized in Table II.
 | Table IIMechanism of action and clinical
significance of SPAG6 in various hematological tumor types. |
Table II
Mechanism of action and clinical
significance of SPAG6 in various hematological tumor types.
| Tumor type | Core functions | Signaling
pathway/molecular mechanism | Clinical
significance | (Refs.) |
|---|
| Myelodysplastic
syndrome | Inhibits
apoptosis | Negative regulation
of the TRAIL apoptosis pathway Silencing activates the
PTEN/PI3K/AKT pro-apoptotic pathway Silencing activates the
AMPK/mTOR/ULK1 autophagy pathway | Enhances the
proapoptotic effect of decitabine; potential target for
demethylation therapy | (12,62-65) |
| Acute myeloid
leukemia | Promotes
proliferation/inhibits apoptosis | Combines with
MYO1D to activate PI3K/AKT and ERK pathways Knockdown blocks
the G1 phase and promotes apoptosis | Independent
prognostic marker; predictive factor for childhood AML
recurrence | (10,71-73) |
| Adult B-cell acute
lymphoblastic leukemia | Drives
proliferation/inhibits apoptosis | Regulation of
proliferation and apoptosis through the TGF-β/Smad pathway | Treatment response
monitoring biomarker; gene silencing inhibits tumor growth | (76) |
| Multiple
myeloma | Promotes
proliferation and migration and inhibits apoptosis | Combines with
DUSP1 to activate the MAPK/ERK pathway | Associated with
hypercalcemia and plasma cell proportion; potential therapeutic
target | (79) |
| BCR::ABL1-negative
myeloproliferative neoplasms | Promotes clone
formation, cell cycle progression | Inhibition of
STAT1-mediated IFN-α-induced apoptosis SPAG6 downregulation when
STAT1 signaling is blocked | Disease phenotype
biomarker associated with sensitivity to interferon therapy | (82,83) |
| Burkitt
lymphoma | Promotes
proliferation/inhibits apoptosis | Regulated by the
PTEN/PI3K/AKT pathway (PTEN inhibition reverses the silencing
effect of SPAG6) | Potential
prognostic biomarker; therapeutic target | (11) |
Role of SPAG6 in solid tumors
SPAG6 is differentially expressed at various tumor
stages and grades (86). In
osteosarcoma and lung squamous cell carcinoma (LUSC), its
expression correlates with prognosis. SPAG6 also plays a
significant role as an oncogene and serves as a prognostic
biomarker. In addition, SPAG6 influences tumor immune infiltration
and the tumor microenvironment, which indicates that it is a
promising immunotherapy target for treatment.
Breast cancer
Breast cancer is a highly heterogeneous malignant
tumor (87). Its occurrence and
development are driven by genetic and environmental factors, making
it one of the leading causes of cancer-related death in women
(88). Circular RNAs (circRNAs)
play an important regulatory role in tumor progression (89). Fan et al (90) showed that circMYH9 enhances
SPAG6 mRNA stability by recruiting the EIF4A3 protein, thus
promoting its expression. SPAG6 overexpression reverses the
inhibitory effect of circMYH9 knockdown on the malignant phenotype
of breast cancer cells. Furthermore, circMYH9 knockout inhibits
PI3K/AKT signaling by upregulating PTEN expression, which is
similarly antagonized by SPAG6 overexpression. In addition,
circMYH9 modulates the PTEN/PI3K/AKT signaling pathway through the
EIF4A3-SPAG6 axis, thereby promoting the malignant progression of
breast cancer cells.
Although mammography remains the standard imaging
modality for early breast cancer screening, it has certain
limitations. Mijnes et al (91) developed an epigenetic analysis
method based on cell-free DNA in blood. This minimally invasive
technique detects the methylation status of tumor suppressor genes
and serves as a liquid biopsy to complement traditional imaging
techniques. The combined detection of SPAG6, period circadian
regulator 1 (PER1) and inter-alpha-trypsin inhibitor heavy chain 5
(ITIH5) achieved 64% sensitivity for breast cancer detection.
Although liquid biopsy has technical challenges, the 'SNiPER'
panel, which includes SPAG6, NK2 homeobox 6, ITIH5 and PER1, holds
promise (91). Manoochehri et
al (92) demonstrated that a
methylation scoring model established from the first three
differentially methylated regions in the SPAG6,
LINC10606 and TBCD/ZNF750 regions yields high
sensitivity and specificity for detecting triple-negative breast
cancer (TNBC). LINC10606 and TBCD/ZNF750 showed
strong discriminatory power in patients with TNBC compared with
healthy controls [area under curve (AUC)=0.78 in the test set,
AUC=0.74 in the validation set]. Therefore, noninvasive DNA
methylation detection may provide novel biomarkers for the early
diagnosis of TNBC.
Nasopharyngeal carcinoma
Nasopharyngeal carcinoma is a malignant tumor
originating from the mucosal epithelium of the nasopharynx
(93), with a predilection for
the pharyngeal recess (Rosenmüller's fossa) (94). Zhang et al (95) used machine learning to identify
genes associated with nasopharyngeal carcinoma and determined their
correlation with the immune microenvironment. Of note, four genes,
including SPAG6, exhibited high predictive efficacy (AUC
>0.9) in the training and validation sets. The expression of
these genes was significantly correlated with the degree of immune
cell infiltration, with SPAG6 showing a particularly strong
association with the immune infiltration phenotype.
Thyroid cancer
Thyroid cancer is a malignant tumor originating in
the thyroid gland (96), and it
is the most commonly diagnosed endocrine malignancy worldwide
(97). Located in the anterior
neck region, the thyroid gland secretes hormones that regulate
metabolism (98). Wang (99) proposed that SPAG6 exerts
tumor-suppressing effects in thyroid cancer. SPAG6 overexpression
inhibited tumor cell invasion and proliferation. Li et al
(86) used immunofluorescence
techniques to show that SPAG6 expression positively correlates with
immune checkpoint molecules in thyroid carcinoma in vitro.
SPAG6 overexpression suppresses malignant cell behavior, including
reduced proliferation and migration, and affects the functional
phenotypes associated with DNA repair, MYC signaling, peroxidase
activity and the G2/M checkpoint.
Squamous cell carcinoma of the
skin
Squamous cell carcinoma is the second most common
nonmelanoma skin tumor, which accounts for ~20% of all skin cancers
(100). It has strong metastatic
potential and is capable of metastasizing to multiple organs in the
body, thereby posing a high risk of mortality. An in-depth study of
its molecular mechanisms is necessary to establish prevention and
treatment strategies (101). Gim
et al (102) selected
early-stage squamous cell carcinoma tissue samples representing
invasive and precancerous regions. Using the NanoString GeoMx
Digital Spatial Profiler for spatial transcriptomics analysis, they
identified SPAG6 as having the highest absolute log2-fold
change in expression among other cancer-associated genes in
fibroblasts. SPAG6 was associated with fibroblast
development and function. In addition, significant alterations in
its expression were evident during the progression of actinic
keratosis to squamous cell carcinoma.
Osteosarcoma
Osteosarcoma is the most common primary malignant
bone tumor (103). It primarily
affects adolescents and young adults and is highly invasive and
prone to metastasis. Although surgery combined with chemotherapy
significantly improves patient survival, the prognosis for
metastatic or recurrent osteosarcoma is poor (104). Bao et al (105) examined SPAG6 expression in tumor
tissues from 42 patients with osteosarcoma and 12 osteochondroma
control tissues using immunohistochemistry, RT-qPCR and western
blot analysis. The SPAG6 protein positivity rate in the
osteosarcoma tissues (71.43%) was significantly higher than that in
the control tissues (33.33%; P<0.05). Both mRNA and protein
levels were markedly increased compared with the levels in adjacent
normal tissue. High SPAG6 expression positively correlated with
higher pathological grade, metastasis and advanced Enneking stage
(P<0.05). SPAG6-positive patients experienced significantly
shorter overall survival. These results indicate that SPAG6
overexpression is associated with malignant progression and poor
prognosis in osteosarcoma, thus suggesting its potential as a
prognostic biomarker.
Lung cancer
Lung cancer is one of the most common and deadliest
malignant tumors (106). Based
on its histology, it may be broadly classified into non-small cell
lung cancer (NSCLC; accounting for ~85% of all cases) and SCLC
(accounting for ~15% of cases) (107,108). Early detection is important for
improving survival outcomes. DNA methylation is an important
epigenetic regulatory mechanism that contributes to the development
of various malignancies, including lung cancer, by modulating
transcriptional activity. It demonstrates significant potential for
predicting the early diagnosis, prognosis and treatment response of
lung cancer (109).
NSCLC originates from lung tissues, such as the
bronchial mucosa, glandular epithelium or pulmonary alveoli. It is
the most prevalent type of lung cancer (110) and has a poor prognosis (111). Altenberger et al
(112) reported that
SPAG6 and LINE-1 type transposase domain containing 1
(L1TD1) mRNA expression is significantly lower in tumor
tissues from patients with NSCLC than in normal lung tissues from
the same patients. In NSCLC cell lines exhibiting downregulated
mRNA expression, treatment with epigenetic modifiers reactivated
the expression of these genes. Tumor-specific hypermethylation of
SPAG6 and L1TD1 in NSCLC tissues with this
methylation pattern effectively distinguished tumors from normal
tissues. These results indicate that SPAG6 and L1TD1 undergo
tumor-specific methylation in NSCLC, which regulates SPAG6
expression at the transcriptional level through DNA methylation
(112).
LUSC is a type of NSCLC characterized by tumor
heterogeneity, genetic mutations, cancer stem cells, immune
resistance and chemotherapy resistance. Because it is usually
diagnosed at an advanced stage, it has a poor prognosis (113). Epigenetic modifications,
primarily DNA methylation (112), are associated with genomic
instability in LUSC. Wu et al (114) found an effect of SPAG6 DNA
methylation on its expression in LUSC. They identified contributors
to SPAG6 DNA hypermethylation. For example, DNA methyltransferase 3
b (DNMT3b)-mediated hypermethylation of the SPAG6 promoter
in LUSC resulted in SPAG6 downregulation, whereas
SPAG6 reversed the malignant phenotype of LUSC cells.
Mechanistically, SPAG6 negatively attenuates the JAK/STAT signaling
pathway by suppressing the transcriptional activity of STAT1 and
STAT3. In addition, SPAG6 expression was found to be positively
correlated with immune cell infiltration in LUSC tissues, whereas
it was negatively correlated with the expression of
immunosuppressive genes, such as cytotoxic T-lymphocyte associated
protein 4 and programmed cell death 1. Furthermore, SPAG6
suppressed tumor stem cell properties by downregulating the
stemness Nanog homeobox (Nanog), aldehyde dehydrogenase 1 family,
member A1 (ALDH1) and Sox2.
High-grade serous epithelial ovarian
cancer (HGSOC)
HGSOC is the most common and aggressive epithelial
ovarian cancer subtype (115).
It displays high invasiveness and a poor patient prognosis. A
deeper understanding of HGSOC tumorigenesis may provide insights
for the development of new therapeutics (116). Coan et al (117) observed SPAG6 expression in
ciliated cells of the fallopian tube. Impaired ciliary motility
disrupts laminar fluid flow over the tubal epithelium, which may
reduce the management of oxidative stress induced by follicular
fluid and contribute to tumorigenesis.
Bladder cancer
Bladder cancer originating from the bladder mucosa
(118) is a common malignant
tumor of the urinary system (119). Kitchen et al (120) observed frequent methylation in
the SPAG6 promoter in bladder cancer tissues. They also
observed significantly increased SPAG6 methylation levels in
recurrent and advanced bladder cancers, suggesting that
SPAG6 functions as a tumor suppressor gene in these tissues.
SPAG6 methylation may represent an independent predictor of
bladder cancer recurrence and progression (120).
All of these findings indicate that SPAG6 shows
diverse regulatory functions across different tumor
microenvironments; however, its pathogenic mechanism is primarily
attributed to dysregulated expression caused by epigenetic and
post-transcriptional regulation, rather than frequent mutations in
the gene itself. This characteristic is similar to that of other
cancer-testis antigens. Its biological functions within tumor cells
and the underlying molecular mechanisms remain to be fully
elucidated. Further in-depth studies are needed to elucidate these
mechanisms. The mechanisms of action and clinical significance of
SPAG6 in different types of solid tumors are summarized in Table III and Fig. 2. Furthermore, Fig. 3 provides a schematic of SPAG6's
role and regulation across various cancers.
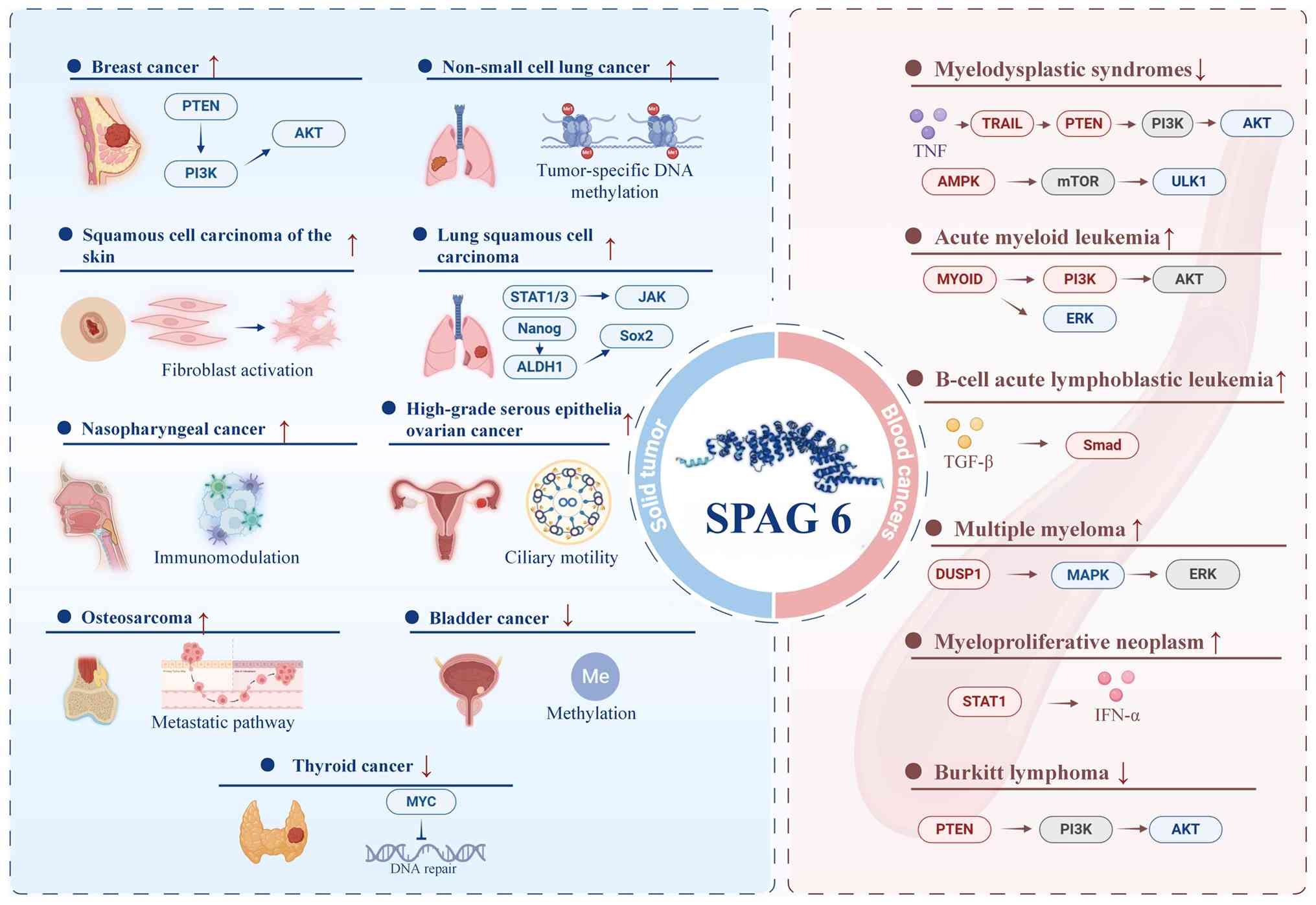 | Figure 2Alterations in SPAG6 among various
cancers as well as the associated signaling pathways and molecular
mechanisms. Arrows (↑/↓) indicate increased or decreased SPAG6
expression/activity in various cancers. The right panel depicts the
role of SPAG6 in hematological malignancies, whereas the left panel
illustrates its function in solid tumors. The regulatory pathways
annotated for each tumor type include PTEN-PI3K-AKT, JAK-STAT,
TGF-β-Smad, MAPK/ERK and AMPK-mTOR. These pathways are involved in
various biological processes, including cell proliferation, immune
regulation, metabolism and epigenetic modifications (e.g.,
methylation), as well as stem cell characteristics (e.g., Nanog,
Sox2 and ALDH1). SPAG6, sperm-associated antigen 6; AKT, AKT
serine/threonine kinase; ALDH1, aldehyde dehydrogenase 1 family
member A1; AMPK, AMP-activated protein kinase; DUSP1, dual
specificity phosphatase 1; ERK, extracellular signal-regulated
kinase; IFN-α, interferon alpha; JAK, Janus kinase; MAPK,
mitogen-activated protein kinase; mTOR, mechanistic target of
rapamycin; MYC, MYC proto-oncogene; Nanog, Nanog homeobox; PI3K,
phosphatidylinositol 3-kinase; PTEN, phosphatase and tensin
homolog; Smad, SMA- and MAD-related protein; Sox2, SRY-box
transcription factor 2; SPAG6, sperm associated antigen 6; STAT1/3,
signal transducer and activator of transcription 1/3; TGF-β,
transforming growth factor beta; TNF, tumor necrosis factor; TRAIL,
TNF-related apoptosis-inducing ligand; ULK1, Unc-51 like autophagy
activating kinase 1. |
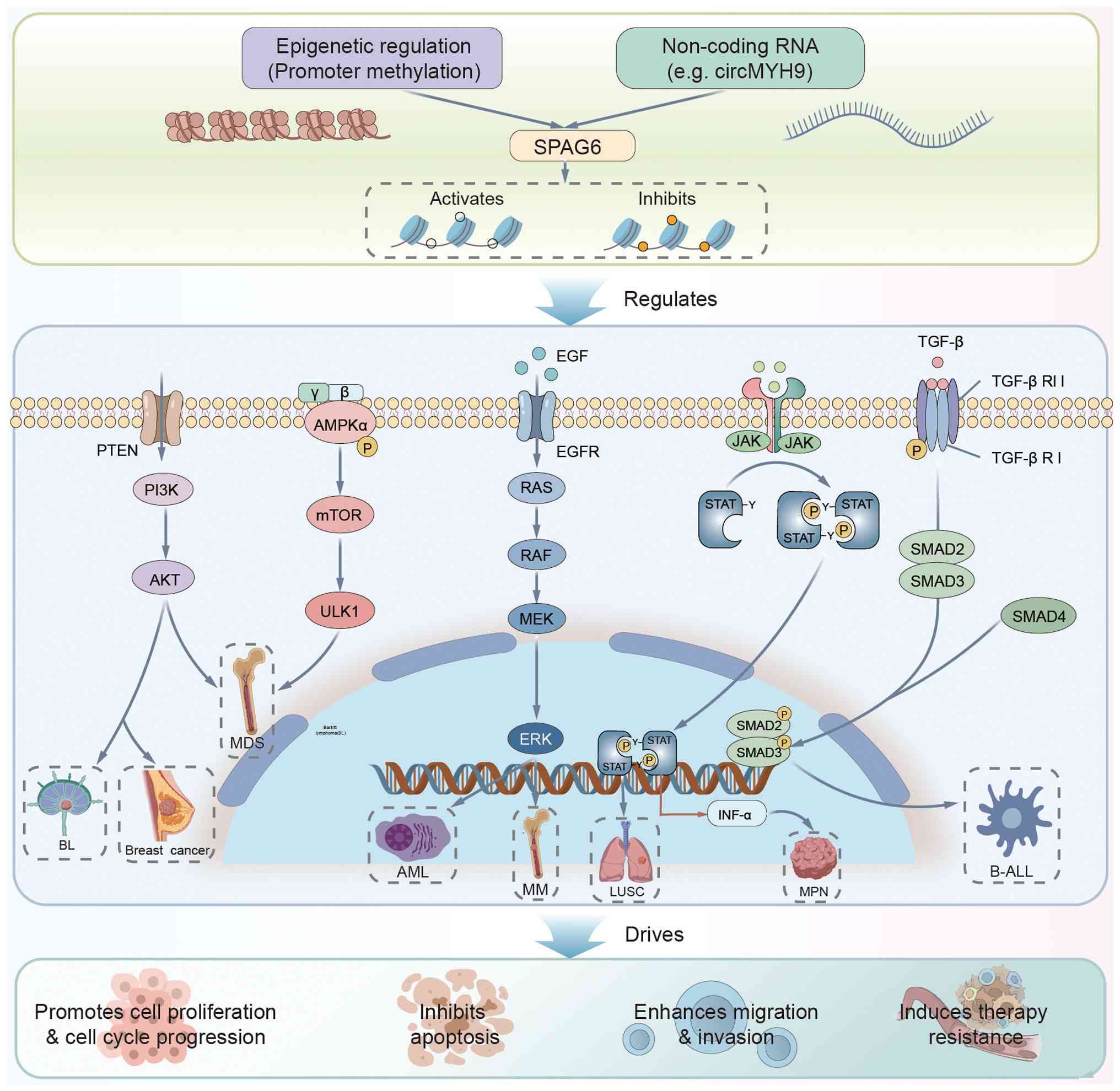 | Figure 3Schematic diagram illustrating the
central role of the SPAG6 gene in oncogenesis: Its expression is
regulated by upstream mechanisms such as promoter methylation and
non-coding RNAs (e.g., circMYH9); subsequently, SPAG6 activates or
participates in key signaling pathways, including
PTEN/PI3K/AKT/mTOR, ERK, and JAK/STAT, playing a core functional
role in various hematological malignancies (such as multiple
myeloma and acute myeloid leukemia) and solid tumors (such as
breast cancer and lung squamous cell carcinoma); ultimately, it
drives malignant progression by promoting cell proliferation,
inhibiting apoptosis, enhancing migration and invasion and inducing
therapy resistance. SPAG6, sperm-associated antigen 6. AKT, AKT
serine/threonine kinase; ALDH1, aldehyde dehydrogenase 1 family
member A1; AML, acute myeloid leukemia; AMPK, AMP-activated protein
kinase; BL, Burkitt lymphoma; B-ALL, B-cell acute lymphoblastic
leukemia; circMYH9, circular RNA myosin heavy chain 9; ERK,
extracellular signal-regulated kinase; LUSC, lung squamous cell
carcinoma; MDS, myelodysplastic syndromes; MM, multiple myeloma;
MPN, myeloproliferative neoplasm; mTOR, mechanistic target of
rapamycin; PI3K, phosphatidylinositol 3-kinase; PTEN, phosphatase
and tensin homolog; Smad, SMA- and MAD-related protein; Sox2,
SRY-box transcription factor 2; SPAG6, sperm associated antigen 6;
STAT1/3, signal transducer and activator of transcription 1/3;
TGF-β, transforming growth factor beta; ULK1, Unc-51 like autophagy
activating kinase 1. |
 | Table IIIMechanism of action and clinical
significance of SPAG6 in different types of solid tumor. |
Table III
Mechanism of action and clinical
significance of SPAG6 in different types of solid tumor.
| Tumor type | Increase or
decrease in molecular expression/activity and Core functions | Signaling
pathway/molecular mechanism | Clinical
significance | (Refs.) |
|---|
| Breast cancer | ↑Promotes
cancer |
circMYH9/EIF4A3→↑SPAG6→↓PTEN→activation of
PI3K/AKT
Drives cell proliferation and metastasis | Liquid biopsy
markers (SNiPER combination sensitivity 64%) | (90-92) |
| Nasopharyngeal
carcinoma | ↑Promotes
cancer | Significantly
positively correlated with immune cell infiltration
Machine learning screening of key genes | Potential targets
for immunotherapy | (95) |
| Thyroid cancer | ↑Anti-cancer | Inhibits cell
proliferation/migration
Positively correlated with immune checkpoint genes
Regulates DNA repair/MYC target pathways | Protective factor
for differentiated thyroid cancer | (86,99) |
| Squamous cell
carcinoma of the skin | ↑Promotes
cancer | Regulation of
fibroblast development and function
Significant changes in AK expression during progression | Potential markers
for early diagnosis | (102) |
| Osteosarcoma | ↑Promotes
cancer | Expression is
positively correlated with pathological
grading/metastasis/staging
Significant increase in mRNA and protein levels | Independent
prognostic marker (high expression=low survival rate) | (105) |
| Lung squamous cell
carcinoma | ↓Anti-cancer | DNA
hypermethylation silences expression
Inhibits STAT1/STAT3 → blocks JAK/STAT
Negatively regulates stemness markers (Nanog/ALDH1/Sox2)
Negatively correlated with immunosuppressive genes | Positively
correlates with immune infiltration markers
Reverses malignant phenotype | (114) |
| Non-small cell lung
cancer | ↓Anti-cancer | Tumor-specific DNA
methylation
Epigenetic drugs can re-express | Methylation
detection distinguishes between tumor and normal tissue | (112) |
| High-grade serous
epithelial ovarian cancer | No conclusion | Ciliary dysfunction
→ reduced oxidative stress clearance
May be involved in tumor initiation | Early onset
mechanism hypothesis | (117) |
| Bladder cancer | ↓Anti-cancer | Tumor-specific DNA
methylation | Independent
predictors of tumor relapse and progression | (119,120) |
Other syndromes
As a gene initially identified from a human
testicular cDNA expression library (3), the encoded product of SPAG6
is a component of the '9+2' microtubule-based centriole complex,
which plays an important role in maintaining the structural
integrity of sperm tail microtubules and ensuring proper flagellar
motility (3,16,17). Abnormal SPAG6 expression is
associated with male infertility, particularly with phenotypes,
such as asthenozoospermia, teratozoospermia and azoospermia, which
are linked to multiple morphological abnormalities of the sperm
flagella (MMAF). Significantly reduced SPAG6 mRNA and protein
expression was observed in patients with idiopathic asthenospermia
(121). Whole-exome sequencing
and ultrastructural analysis of patients with teratospermia
consistently reveal downregulated SPAG6 expression (122). SPAG6 protein levels are also
markedly decreased in patients with MMAF (123). Proteomic analysis of
spermatogenic efferent duct mutation carriers revealed differential
SPAG6 expression associated with flagellar assembly processes
(124). In addition, compound
heterozygous SPAG6 mutations were identified in patients
with primary ciliary dyskinesia (125), and biallelic dynein heavy chain
domain 1 variants were observed in patients with azoospermia, which
was associated with reduced sperm SPAG6 levels (126). SPAG6 mutations may influence
pregnancy outcomes following intracytoplasmic sperm injection
(127). Functional studies
indicate that targeted disruption of SPAG6 expression
results in decreased sperm motility, increased apoptosis (128) and markedly reduced levels of
axonemal proteins (15,129,130). Studies using animal models
confirm that SPAG6 deficiency causes sperm motility defects
and abnormal microtubule architecture, which results in infertility
(15,17). It also disrupts centrosome
polarization and immunological synapse formation, thereby
compromising lymphocyte function (25,131). Furthermore, significant
downregulation of SPAG6 was observed in cryptic testicular
tissue (131), further
indicating a role in normal testicular physiology. Overall,
SPAG6 is an important factor in sperm flagellar development
and motility. Its abnormal expression or function constitutes a
genetic basis for various male reproductive disorders and provides
a foundation for the development of related gene therapies.
Clinical significance of SPAG6
In hematologic malignancies, high SPAG6 expression
in MDS promotes disease progression by inhibiting the TRAIL
apoptosis pathway (reducing FAS-Associated Via Death Domain binding
to death receptors), activating the PI3K/AKT signaling pathway
(downregulating PTEN) (63), and
attenuating the G1/S transition of the cell cycle (12). SPAG6 silencing induces
AMPK/mTOR/ULK1-mediated autophagic apoptosis and enhances the DNA
demethylation efficacy of decitabine (64). In AML, SPAG6 serves as an
independent prognostic marker (71), with high expression activating the
PI3K/AKT and ERK pathways through the formation of a
SPAG6-MYO1D complex that drives leukemia growth and
is associated with MRD monitoring (73). SPAG6 expression is significantly
reduced in adult patients with B-ALL during remission and it
maintains tumor cell proliferation through the TGF-β/Smad signaling
pathway (76). In MM,
SPAG6 binds to DUSP1 and activates the MAPK/ERK
pathway, thereby promoting the malignant phenotype and exhibiting
an association with bone infiltration (79). In BCR-ABL1-negative MPN, SPAG6
forms a positive feedback loop with STAT1 (83). Its silencing enhances the
proapoptotic effect of IFN-α. In BL, SPAG6 activates the
PI3K/AKT pathway by inhibiting PTEN, and its downregulation
inhibits tumor growth in vivo and in vitro (11).
In solid tumors, circMYH9 activates the
PTEN/PI3K/AKT pathway in breast cancer by stabilizing SPAG6
mRNA (90). SPAG6 methylation
patterns serve as liquid biopsy markers. In nasopharyngeal
carcinoma, SPAG6 is downregulated because of high promoter
methylation (95). It is
associated with immune infiltration and considered an important
diagnostic gene. In thyroid cancer, SPAG6 overexpression
inhibits proliferation and migration and regulates the DNA
repair/MYC target pathway (86).
In osteosarcoma, the SPAG6 positivity rate (71.43%) is
significantly increased and associated with disease grade,
metastasis and poor prognosis (105). In LUSC, DNMT3b-mediated
hypermethylation of SPAG6 results in its silencing, which
activates the JAK/STAT pathway and enhances cancer stemness
(114). In NSCLC, SPAG6
expression is downregulated by tumor-specific methylation (112). Ovarian cancer studies have
reported that SPAG6-related ciliary dysfunction contributes to a
tumor initiation microenvironment (117). In bladder cancer, frequent
methylation occurs in the SPAG6 promoter region, and
SPAG6 methylation levels are significantly increased in
recurrent and advanced bladder cancer (120).
Although the protumor/antitumor mechanisms of SPAG6
in various tumor types have become gradually clearer, several
unresolved issues persist in this field. For instance, although
SPAG6 acts as an oncogene in most tumors, such as hematological
malignancies and osteosarcoma, it exhibits tumor-suppressing
effects in a minority of cancers, including nasopharyngeal
carcinoma and LUSC. The reasons for this contrasting role warrant
further investigation. Second, studies have predominantly focused
on SPAG6's regulation of a few classical pathways, such as
PTEN/PI3K/AKT and MAPK/ERK; however, the precise regulatory factors
upstream (e.g., transcription factors or noncoding RNAs governing
its expression) and its broader downstream effector networks remain
elusive. In particular, its role in modulating the tumor immune
microenvironment and DNA damage response is unknown. This lack of
mechanistic insight hinders its clinical translation, while the
development of specific inhibitors targeting SPAG6 remains in its
infancy. To date, there have been no reports of small-molecule
inhibitors or traditional Chinese medicine-derived inhibitors of
SPAG6, as research is primarily focused on identifying the
underlying mechanism. Based on the latest findings from these
mechanistic studies, the development of targeted strategies for
attenuating SPAG6 has significant clinical implications. Because of
the differential expression of SPAG6 in various tumors, the
following targeted treatment strategies should be considered:
First, for tumors with high SPAG6 expression, such as
hematological tumors and osteosarcoma, developing specific
inhibitors to block key oncogenic pathways mediated by
SPAG6, such as the PTEN/PI3K/AKT signaling pathway, may be
fruitful. Second, for tumor types with silenced SPAG6
expression, such as nasopharyngeal carcinoma and LUSC, identifying
epigenetic regulatory approaches, such as demethylation drugs, to
restore its expression is necessary. Finally, because SPAG6
silencing enhances the efficacy of drugs, such as decitabine and
IFN-α, in MDS and MPN models, SPAG6 inhibitors combined with
traditional chemotherapy/targeted drugs offer synergistic
therapeutic potential. Such multifaceted treatment strategies will
provide personalized intervention for patients with cancer
harboring various SPAG6 expression profiles.
Summary and outlook
In summary, SPAG6 is a tubulin protein with
multiple physiological functions, including regulation of
ciliary/flagellar movement, mediation of the formation and function
of immune synapses, neuronal proliferation and differentiation. It
regulates the morphology, growth and migration of fibroblasts and
attenuates the function of middle ear and Eustachian tube
epithelial cells. In addition, SPAG6 acts as an oncogene in most
tumors, promoting tumorigenesis through its high expression and
regulating signaling pathways, such as PTEN/PI3K/AKT and MAPK/ERK,
to promote tumor proliferation, migration and drug resistance
(e.g., hematological tumors, osteosarcoma and breast cancer). It
also exhibits tumor-suppressing effects in bladder cancer
(methylation silencing) and thyroid cancer (overexpression inhibits
the malignant phenotype). It exhibits significant tissue
specificity in hematological tumors (AML, MDS and lymphoma) and
serves as a prognostic marker and therapeutic target. SPAG6
silencing enhances tumor cell sensitivity to chemotherapy. In solid
tumors, SPAG6 is closely associated with immune infiltration,
cancer stemness and epigenetic regulation (e.g., breast cancer
circRNA stabilizes SPAG6 mRNA), and pancancer analyses
suggest that its expression is associated with immune
microenvironment remodeling. Overall, SPAG6 is a potential
biomarker for tumor classification, prognosis assessment and
targeted intervention. It may be used to guide treatment selection
and assess disease prognosis. Nevertheless, SPAG6-related studies
have several limitations. In vitro and in vivo
studies are needed to evaluate its potential as a therapeutic
target and prognostic marker in tumors. In addition, it is
necessary to examine its upstream and downstream regulatory
pathways and identify proteins that interact with SPAG6 through
protein-protein interactions to elucidate the mechanisms underlying
its role in tumorigenesis and tumor progression.
Availability of data and materials
Not applicable.
Authors' contributions
YL and YW conceptualized the structure and main
ideas of this paper and provided guidance for the writing process.
YL and PZ performed the literature search. QY, PZ and HX drafted
the main body of the paper. RZ and RW revised and polished the
paper. All authors have read and approved the final version of the
manuscript. Data authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, the authors
used Grammarly (https://www.grammarly.com/) for grammar checking and
language enhancement. After using this tool, the authors reviewed
and edited the content as needed and take full responsibility for
the content of the publication.
Abbreviations:
|
AK
|
adenylate kinase
|
|
AKT
|
AKT serine/threonine kinase
|
|
ALDH1
|
aldehyde dehydrogenase 1 family
member A1
|
|
ALL
|
acute lymphoblastic leukemia
|
|
AML
|
acute myeloid leukemia
|
|
AMPK
|
AMP-activated protein kinase
|
|
B-ALL
|
B-cell acute lymphoblastic
leukemia
|
|
BCR::ABL1
|
BCR (BCR activator of RhoGEF and
GTPase)::ABL1 (ABL proto-oncogene 1 non-receptor tyrosine
kinase)
|
|
BL
|
burkitt lymphoma
|
|
circMYH9
|
circular RNA myosin heavy chain 9
|
|
DNA
|
deoxyribonucleic acid
|
|
DUSP1
|
dual specificity phosphatase 1
|
|
EIF4A3
|
eukaryotic translation initiation
factor 4A3
|
|
ERK
|
extracellular signal-regulated
kinase
|
|
G1 phase
|
Gap 1 phase of the cell cycle
|
|
IFN-α
|
interferon alpha
|
|
JAK
|
Janus kinase
|
|
LUSC
|
lung squamous cell carcinoma
|
|
MAP1
|
microtubule-associated protein 1
|
|
MAPK
|
mitogen-activated protein kinase
|
|
MDS
|
myelodysplastic syndromes
|
|
MM
|
multiple myeloma
|
|
MPN
|
myeloproliferative neoplasm
|
|
mRNA
|
messenger RNA
|
|
mTOR
|
mechanistic target of rapamycin
|
|
MYC
|
MYC proto-oncogene
|
|
MYO1D
|
myosin ID
|
|
Nanog
|
Nanog homeobox
|
|
PI3K
|
phosphatidylinositol 3-kinase
|
|
PTEN
|
phosphatase and tensin homolog
|
|
Smad
|
SMA- and MAD-related protein
|
|
Sox2
|
SRY-box transcription factor 2
|
|
SPAG6
|
sperm associated antigen 6
|
|
STAT1
|
signal transducer and activator of
transcription 1
|
|
TGF-β
|
transforming growth factor beta
|
|
TNF
|
tumor necrosis factor
|
|
TRAIL
|
TNF-related apoptosis-inducing
ligand
|
|
ULK1
|
Unc-51 like autophagy activating
kinase 1
|
Acknowledgements
The authors would like to express their heartfelt
gratitude to Professor Zhaoyun Liu from the Hematology Department,
General Hospital of Tianjin Medical University (Tianjin, China),
for his invaluable help, guidance and patience throughout this
project. The authors also thank Professor Jin Huang from West China
Hospital Sichuan University (Chengdu, China) for his invaluable and
thoughtful advice during the interpretation of relevant literature.
Lastly, the authors thank Mr. Kun Sun, another master's student at
Sichuan University School of Business (Chengdu, China), for his
technical guidance.
Funding
The authors are grateful to Mr. Dongsheng Dai, a master's
student at Sichuan University School of Business (Chengdu, China),
for financial support.
References
|
1
|
Li X, Xu L, Sun G, Wu X, Bai X, Li J,
Strauss JF, Zhang Z and Wang H: Spag6 mutant mice have defects in
development and function of spiral ganglion neurons, apoptosis, and
higher sensitivity to paclitaxel. Sci Rep. 7:86382017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Siliņa K, Zayakin P, Kalniņa Z, Ivanova L,
Meistere I, Endzeliņš E, Abols A, Stengrēvics A, Leja M, Ducena K,
et al: Sperm-associated antigens as targets for cancer
immunotherapy: Expression pattern and humoral immune response in
cancer patients. J Immunother. 34:28–44. 2011. View Article : Google Scholar
|
|
3
|
Neilson LI, Schneider PA, Van Deerlin PG,
Kiriakidou M, Driscoll DA, Pellegrini MC, Millinder S, Yamamoto KK,
French CK and Strauss JF III: cDNA cloning and characterization of
a human sperm antigen (SPAG6) with homology to the product of the
Chlamydomonas PF16 locus. Genomics. 60:272–280. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Qiu H, Gołas A, Grzmil P and Wojnowski L:
Lineage-specific duplications of Muroidea Faim and Spag6 genes and
atypical accelerated evolution of the parental Spag6 gene. J Mol
Evol. 77:119–129. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tewari R, Bailes E, Bunting KA and Coates
JC: Armadillo-repeat protein functions: Questions for little
creatures. Trends Cell Biol. 20:470–481. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Scanlan MJ, Simpson AJ and Old LJ: The
cancer/testis genes: Review, standardization, and commentary.
Cancer Immun. 4:12004.PubMed/NCBI
|
|
7
|
Teves ME, Sears PR, Li W and Zhang Z, Tang
W, van Reesema L, Costanzo RM, Davis CW, Knowles MR, Strauss JF III
and Zhang Z: Sperm-associated antigen 6 (SPAG6) deficiency and
defects in ciliogenesis and cilia function: Polarity, density, and
beat. PLoS One. 9:e1072712014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li W, Mukherjee A, Wu J, Zhang L, Teves
ME, Li H, Nambiar S, Henderson SC, Horwitz AR, Strauss JF III, et
al: Sperm associated antigen 6 (SPAG6) regulates fibroblast cell
growth, morphology, migration and ciliogenesis. Sci Rep.
5:165062015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lonergan KM, Chari R, Deleeuw RJ, Shadeo
A, Chi B, Tsao MS, Jones S, Marra M, Ling V, Ng R, et al:
Identification of novel lung genes in bronchial epithelium by
serial analysis of gene expression. Am J Respir Cell Mol Biol.
35:651–661. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Steinbach D, Schramm A, Eggert A, Onda M,
Dawczynski K, Rump A, Pastan I, Wittig S, Pfaffendorf N, Voigt A,
et al: Identification of a set of seven genes for the monitoring of
minimal residual disease in pediatric acute myeloid leukemia. Clin
Cancer Res. 12:2434–2441. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang R, Zhu H, Yuan Y, Wang Y and Tian Z:
SPAG6 promotes cell proliferation and inhibits apoptosis through
the PTEN/PI3K/AKT pathway in Burkitt lymphoma. Oncol Rep.
44:2021–2030. 2020.PubMed/NCBI
|
|
12
|
Jiang M, Chen Y, Deng L, Luo X, Wang L and
Liu L: Upregulation of SPAG6 in myelodysplastic syndrome: Knockdown
inhibits cell proliferation via AKT/FOXO signaling pathway. DNA
Cell Biol. 38:476–484. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ding L, Luo J, Zhang JP, Wang J, Li ZQ,
Huang J, Chai L, Mu J, Zhao B, Zhong YR, et al: Aberrant expression
of SPAG6 may affect the disease phenotype and serve as a tumor
biomarker in BCR/ABL1-negative myeloproliferative neoplasms. Oncol
Lett. 23:102022. View Article : Google Scholar
|
|
14
|
Zheng DF, Wang Q, Wang JP, Bao ZQ, Wu SW,
Ma L, Chai DM, Wang ZP and Tao YS: The emerging role of
sperm-associated antigen 6 gene in the microtubule function of
cells and cancer. Mol Ther Oncolytics. 15:101–107. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang Z, Sapiro R, Kapfhamer D, Bucan M,
Bray J, Chennathukuzhi V, McNamara P, Curtis A, Zhang M,
Blanchette-Mackie EJ and Strauss JF III: A sperm-associated WD
repeat protein orthologous to Chlamydomonas PF20 associates with
Spag6, the mammalian orthologue of Chlamydomonas PF16. Mol Cell
Biol. 22:7993–8004. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sapiro R, Tarantino LM, Velazquez F,
Kiriakidou M, Hecht NB, Bucan M and Strauss JF III: Sperm antigen 6
is the murine homologue of the Chlamydomonas reinhardtii central
apparatus protein encoded by the PF16 locus. Biol Reprod.
62:511–518. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sapiro R, Kostetskii I, Olds-Clarke P,
Gerton GL, Radice GL and Strauss JF III: Male infertility, impaired
sperm motility, and hydrocephalus in mice deficient in
sperm-associated antigen 6. Mol Cell Biol. 22:6298–6305. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Smith EF and Lefebvre PA: PF16 encodes a
protein with armadillo repeats and localizes to a single
microtubule of the central apparatus in Chlamydomonas flagella. J
Cell Biol. 132:359–370. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Meng X, Sun X, Liu Z and He Y: A novel era
of cancer/testis antigen in cancer immunotherapy. Int
Immunopharmacol. 98:1078892021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yang P, Meng M and Zhou Q: Oncogenic
cancer/testis antigens are a hallmarker of cancer and a sensible
target for cancer immunotherapy. Biochim Biophys Acta Rev Cancer.
1876:1885582021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wu Q, Liu J, Fang A, Li R, Bai Y,
Kriegstein AR and Wang X: The dynamics of neuronal migration. Adv
Exp Med Biol. 800:25–36. 2014. View Article : Google Scholar
|
|
22
|
Yan R, Hu X, Zhang Q, Song L, Zhang M,
Zhang Y and Zhao S: Spag6 negatively regulates neuronal migration
during mouse brain development. J Mol Neurosci. 57:463–469. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bogoyevitch MA, Yeap YY, Qu Z, Ngoei KR,
Yip YY, Zhao TT, Heng JI and Ng DC: WD40-repeat protein 62 is a
JNK-phosphorylated spindle pole protein required for spindle
maintenance and timely mitotic progression. J Cell Sci.
125:5096–5109. 2012.PubMed/NCBI
|
|
24
|
Li X, Zhang D, Xu L, Liu W, Zhang N,
Strauss JF III, Zhang Z and Wang H: Sperm-associated antigen 6
(Spag6) mutation leads to vestibular dysfunction in mice. J
Pharmacol Sci. 147:325–330. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cooley LF, El Shikh ME, Li W, Keim RC,
Zhang Z, Strauss JF, Zhang Z and Conrad DH: Impaired immunological
synapse in sperm associated antigen 6 (SPAG6) deficient mice. Sci
Rep. 6:258402016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
de la Roche M, Ritter AT, Angus KL,
Dinsmore C, Earnshaw CH, Reiter JF and Griffiths GM: Hedgehog
signaling controls T cell killing at the immunological synapse.
Science. 342:1247–1250. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ralston KS, Lerner AG, Diener DR and Hill
KL: Flagellar motility contributes to cytokinesis in Trypanosoma
brucei and is modulated by an evolutionarily conserved dynein
regulatory system. Eukaryot Cell. 5:696–711. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Branche C, Kohl L, Toutirais G, Buisson J,
Cosson J and Bastin P: Conserved and specific functions of axoneme
components in trypanosome motility. J Cell Sci. 119:3443–3455.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Straschil U, Talman AM, Ferguson DJP,
Bunting KA, Xu Z, Bailes E, Sinden RE, Holder AA, Smith EF, Coates
JC and Tewari R: The Armadillo repeat protein PF16 is essential for
flagellar structure and function in Plasmodium male gametes. PLoS
One. 5:e129012010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hu M, Ayub Q, Guerra-Assunção JA, Long Q,
Ning Z, Huang N, Romero IG, Mamanova L, Akan P, Liu X, et al:
Exploration of signals of positive selection derived from
genotype-based human genome scans using re-sequencing data. Hum
Genet. 131:665–674. 2012. View Article : Google Scholar :
|
|
31
|
Doxsey S: Re-evaluating centrosome
function. Nat Rev Mol Cell Biol. 2:688–698. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Vladar EK, Bayly RD, Sangoram AM, Scott MP
and Axelrod JD: Microtubules enable the planar cell polarity of
airway cilia. Curr Biol. 22:2203–2212. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Majithia A, Fong J, Hariri M and Harcourt
J: Hearing outcomes in children with primary ciliary dyskinesia-a
longitudinal study. Int J Pediatr Otorhinolaryngol. 69:1061–1064.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Leigh MW, Pittman JE, Carson JL, Ferkol
TW, Dell SD, Davis SD, Knowles MR and Zariwala MA: Clinical and
genetic aspects of primary ciliary dyskinesia/Kartagener syndrome.
Genet Med. 11:473–487. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Li H, Lv J, Zhou Q, Jin L, Kang Z and
Huang Y: Establishment of sperm associated antigen 6 gene knockout
mouse model and its mechanism of deafness. Saudi J Biol Sci.
27:1289–1295. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Li X, Xu L, Li J, Li B, Bai X, Strauss JF
III, Zhang Z and Wang H: Otitis media in sperm-associated antigen 6
(Spag6)-deficient mice. PLoS One. 9:e1128792014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Li X, Zhang D, Xu L, Han Y, Liu W, Li W,
Fan Z, Costanzo RM, Strauss JF III, Zhang Z and Wang H: Planar cell
polarity defects and hearing loss in sperm-associated antigen 6
(Spag6)-deficient mice. Am J Physiol Cell Physiol. 320:C132–C141.
2021.
|
|
38
|
Vallee RB and Tsai JW: The cellular roles
of the lissencephaly gene LIS1, and what they tell us about brain
development. Genes Dev. 20:1384–1393. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang J, Li X, Zhang Z, Wang H and Li J:
Expression of prestin in OHCs is reduced in Spag6 gene knockout
mice. Neurosci Lett. 592:42–47. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Farkas LM and Huttner WB: The cell biology
of neural stem and progenitor cells and its significance for their
proliferation versus differentiation during mammalian brain
development. Curr Opin Cell Biol. 20:707–715. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Florio M and Huttner WB: Neural
progenitors, neurogenesis and the evolution of the neocortex.
Development. 141:2182–2194. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gilmore EC and Walsh CA: Genetic causes of
microcephaly and lessons for neuronal development. Wiley
Interdiscip Rev Dev Biol. 2:461–478. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Guerrini R, Dobyns WB and Barkovich AJ:
Abnormal development of the human cerebral cortex: Genetics,
functional consequences and treatment options. Trends Neurosci.
31:154–162. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Martin CA, Ahmad I, Klingseisen A, Hussain
MS, Bicknell LS, Leitch A, Nürnberg G, Toliat MR, Murray JE, Hunt
D, et al: Mutations in PLK4, encoding a master regulator of
centriole biogenesis, cause microcephaly, growth failure and
retinopathy. Nat Genet. 46:1283–1292. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Hamada T, Teraoka M, Imaki J, Ui-Tei K,
Ladher RK and Asahara T: Gene expression of Spag6 in chick central
nervous system. Anat Histol Embryol. 39:227–232. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhang Z, Tang W, Zhou R, Shen X, Wei Z,
Patel AM, Povlishock JT, Bennett J and Strauss JF III: Accelerated
mortality from hydrocephalus and pneumonia in mice with a combined
deficiency of SPAG6 and SPAG16L reveals a functional
interrelationship between the two central apparatus proteins. Cell
Motil Cytoskeleton. 64:360–376. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Chen JF, Zhang Y, Wilde J, Hansen KC, Lai
F and Niswander L: Microcephaly disease gene Wdr62 regulates
mitotic progression of embryonic neural stem cells and brain size.
Nat Commun. 5:38852014. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Moon HM, Youn YH, Pemble H, Yingling J,
Wittmann T and Wynshaw-Boris A: LIS1 controls mitosis and mitotic
spindle organization via the LIS1-NDEL1-dynein complex. Hum Mol
Genet. 23:449–466. 2014. View Article : Google Scholar
|
|
49
|
Hu X, Yan R, Cheng X, Song L, Zhang W, Li
K and Zhao S: The function of sperm-associated antigen 6 in
neuronal proliferation and differentiation. J Mol Histol.
47:531–540. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Fang WQ, Chen WW, Fu AKY and Ip NY: Axin
directs the amplification and differentiation of intermediate
progenitors in the developing cerebral cortex. Neuron. 79:665–679.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Mitchell B, Stubbs JL, Huisman F, Taborek
P, Yu C and Kintner C: The PCP pathway instructs the planar
orientation of ciliated cells in the Xenopus larval skin. Curr
Biol. 19:924–929. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Fulda S and Debatin KM: Extrinsic versus
intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene.
25:4798–4811. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Shen Y, Chow J, Wang Z and Fan G: Abnormal
CpG island methylation occurs during in vitro differentiation of
human embryonic stem cells. Hum Mol Genet. 15:2623–3635. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Janke C and Bulinski JC:
Post-translational regulation of the microtubule cytoskeleton:
Mechanisms and functions. Nat Rev Mol Cell Biol. 12:773–786. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Vaughan EE, Geiger RC, Miller AM,
Loh-Marley PL, Suzuki T, Miyata N and Dean DA: Microtubule
acetylation through HDAC6 inhibition results in increased
transfection efficiency. Mol Ther. 16:1841–1847. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Bullinger L, Döhner K and Döhner H:
Genomics of acute myeloid leukemia diagnosis and pathways. J Clin
Oncol. 35:934–946. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Kim Y, Yeon M and Jeoung D: DDX53
regulates cancer stem cell-like properties by binding to SOX-2. Mol
Cells. 40:322–330. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Li Y, Li J, Wang Y, Zhang Y, Chu J, Sun C,
Fu Z, Huang Y, Zhang H, Yuan H and Yin Y: Roles of cancer/testis
antigens CTAs) in breast cancer. Cancer Lett. 399:64–73. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
van Duin M, Broyl A, de Knegt Y,
Goldschmidt H, Richardson PG, Hop WC, van der Holt B,
Joseph-Pietras D, Mulligan G, Neuwirth R, et al: Cancer testis
antigens in newly diagnosed and relapse multiple myeloma:
Prognostic markers and potential targets for immunotherapy.
Haematologica. 96:1662–1669. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Gera K, Chauhan A, Castillo P, Rahman M,
Mathavan A, Mathavan A, Oganda-Rivas E, Elliott L, Wingard JR and
Sayour EJ: Vaccines: A promising therapy for myelodysplastic
syndrome. J Hematol Oncol. 17:42024. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Lee P, Yim R, Yung Y, Chu HT, Yip PK and
Gill H: Molecular targeted therapy and immunotherapy for
myelodysplastic syndrome. Int J Mol Sci. 22:102322021. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Li X, Yang B, Wang L, Chen L, Luo X and
Liu L: SPAG6 regulates cell apoptosis through the TRAIL signal
pathway in myelodysplastic syndromes. Oncol Rep. 37:2839–2846.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Yin J, Li X, Zhang Z, Luo X, Wang L and
Liu L: SPAG6 silencing induces apoptosis in the myelodysplastic
syndrome cell line SKM-1 via the PTEN/PI3K/AKT signaling pathway in
vitro and in vivo. Int J Oncol. 53:297–306. 2018.PubMed/NCBI
|
|
64
|
Zhang M, Luo J, Luo X and Liu L: SPAG6
silencing induces autophagic cell death in SKM-1 cells via the
AMPK/mTOR/ULK1 signaling pathway. Oncol Lett. 20:551–560. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Luo J, Mu J, Zhang M, Zhao B and Liu L:
SPAG6-silencing enhances decitabine-induced apoptosis and
demethylation of PTEN in SKM-1 cells and in a xenograft mouse
model. Leuk Lymphoma. 62:2242–2252. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Luo J, Mu J and Liu L: Effects of SPAG6
silencing and decitabine treatment on apoptosis and phosphatase and
tensin homolog methylation in SKM-1 cells. Zhonghua Xue Ye Xue Za
Zhi. 42:1005–1010. 2021.In Chinese.
|
|
67
|
Papaemmanuil E, Gerstung M, Bullinger L,
Gaidzik VI, Paschka P, Roberts ND, Potter NE, Heuser M, Thol F,
Bolli N, et al: Genomic classification and prognosis in acute
myeloid leukemia. N Engl J Med. 374:2209–2221. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Patel JP, Gönen M, Figueroa ME, Fernandez
H, Sun Z, Racevskis J, Van Vlierberghe P, Dolgalev I, Thomas S,
Aminova O, et al: Prognostic relevance of integrated genetic
profiling in acute myeloid leukemia. N Engl J Med. 366:1079–1089.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Stubbins RJ, Francis A, Kuchenbauer F and
Sanford D: Management of acute myeloid leukemia: A review for
general practitioners in oncology. Curr Oncol. 29:6245–6259. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Luo J, Zhao H, Zang X and Liu L: High
expression of SPAG6 acts as a pro-tumor factor and associated with
poor prognosis in acute myeloid leukemia. Int J Lab Hematol.
47:680–689. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Steinbach D, Bader P, Willasch A,
Bartholomae S, Debatin KM, Zimmermann M, Creutzig U, Reinhardt D
and Gruhn B: Prospective validation of a new method of monitoring
minimal residual disease in childhood acute myelogenous leukemia.
Clin Cancer Res. 21:1353–1359. 2015. View Article : Google Scholar
|
|
72
|
Skou AS, Juul-Dam KL, Hansen M, Lausen B,
Stratmann S, Holmfeldt L, Aggerholm A, Nyvold CG, Ommen HB and
Hasle H: Measurable residual disease monitoring of SPAG6, ST18,
PRAME, and XAGE1A expression in peripheral blood may detect
imminent relapse in childhood acute myeloid leukemia. J Mol Diagn.
23:1787–1799. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Mu J, Yuan P, Luo J, Chen Y, Tian Y, Ding
L, Zhao B, Wang X, Wang B and Liu L: Upregulated SPAG6 promotes
acute myeloid leukemia progression through MYO1D that regulates the
EGFR family expression. Blood Adv. 6:5379–5394. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Passet M, Kim R and Clappier E: Genetic
subtypes of B-cell acute lymphoblastic leukemia in adults. Blood.
145:1451–1463. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Yasuda T, Sanada M, Tsuzuki S and Hayakawa
F: Oncogenic lesions and molecular subtypes in adults with B-cell
acute lymphoblastic leukemia. Cancer Sci. 114:8–15. 2023.
View Article : Google Scholar :
|
|
76
|
Zhao B, Yin J, Ding L, Luo J, Luo J, Mu J,
Pan S, Du J, Zhong Y, Zhang L and Liu L: SPAG6 regulates cell
proliferation and apoptosis via TGF-β/Smad signal pathway in adult
B-cell acute lymphoblastic leukemia. Int J Hematol. 119:119–129.
2024. View Article : Google Scholar
|
|
77
|
Cowan AJ, Green DJ, Kwok M, Lee S, Coffey
DG, Holmberg LA, Tuazon S, Gopal AK and Libby EN: Diagnosis and
management of multiple myeloma: A review. JAMA. 327:464–477. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Kyle RA and Rajkumar SV: Multiple myeloma.
Blood. 111:2962–2972. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Li J, Yan X, Ding L, Yin J, Li P and Liu
L: SPAG6 promotes multiple myeloma through activation of the
MAPK/ERK signaling pathway. Front Pharmacol. 16:15726212025.
View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Luque Paz D, Kralovics R and Skoda RC:
Genetic basis and molecular profiling in myeloproliferative
neoplasms. Blood. 141:1909–1921. 2023. View Article : Google Scholar
|
|
81
|
Morishita S and Komatsu N: Diagnosis- and
prognosis-related gene alterations in BCR::ABL1-negative
myeloproliferative neoplasms. Int J Mol Sci. 24:130082023.
View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Xia Y, Li X, Tian X and Zhao Q:
Identification of a five-gene signature derived from MYCN
amplification and establishment of a nomogram for predicting the
prognosis of neuroblastoma. Front Mol Biosci. 8:7696612021.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Ding L, Luo J, Du J, Zhao B, Luo J, Pan S,
Zhang L, Yan X, Li J and Liu L: Upregulated SPAG6 correlates with
increased STAT1 and is associated with reduced sensitivity of
interferon-α response in BCR::ABL1 negative myeloproliferative
neoplasms. Cancer Sci. 114:4445–4458. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
López C, Burkhardt B, Chan JKC, Leoncini
L, Mbulaiteye SM, Ogwang MD, Orem J, Rochford R, Roschewski M and
Siebert R: Burkitt lymphoma. Nat Rev Dis Primers. 8:782022.
View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Fang H, Wang W and Medeiros LJ: Burkitt
lymphoma. Hum Pathol. 156:1057032025. View Article : Google Scholar
|
|
86
|
Li X, Wang Y, Li X, Kong L, Díez JJ, Wang
H and Zhang D: A comprehensive pan-cancer analysis revealing SPAG6
as a novel diagnostic, prognostic and immunological biomarker in
tumor. Gland Surg. 13:999–1015. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Hutchinson L: Breast cancer: Challenges,
controversies, breakthroughs. Nat Rev Clin Oncol. 7:669–670. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Xiong X, Zheng LW, Ding Y, Chen YF, Cai
YW, Wang LP, Huang L, Liu CC, Shao ZM and Yu KD: Breast cancer:
Pathogenesis and treatments. Signal Transduct Target Ther.
10:492025. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Conn VM, Chinnaiyan AM and Conn SJ:
Circular RNA in cancer. Nat Rev Cancer. 24:597–613. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Fan S, Cui Y, Liu Y, Li Y, Huang H and Hu
Z: CircMYH9 promotes the mRNA stability of SPAG6 by recruiting
EIF4A3 to facilitate the progression of breast cancer. Epigenetics.
20:24823822025. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Mijnes J, Tiedemann J, Eschenbruch J,
Gasthaus J, Bringezu S, Bauerschlag D, Maass N, Arnold N, Weimer J,
Anzeneder T, et al: SNiPER: A novel hypermethylation biomarker
panel for liquid biopsy based early breast cancer detection.
Oncotarget. 10:6494–6508. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Manoochehri M, Borhani N, Gerhäuser C,
Assenov Y, Schönung M, Hielscher T, Christensen BC, Lee MK, Gröne
HJ, Lipka DB, et al: DNA methylation biomarkers for noninvasive
detection of triple-negative breast cancer using liquid biopsy. Int
J Cancer. 152:1025–1035. 2023. View Article : Google Scholar
|
|
93
|
Tang LL, Chen YP, Chen CB, Chen MY, Chen
NY, Chen XZ, Du XJ, Fang WF, Feng M, Gao J, et al: The Chinese
society of clinical oncology CSCO) clinical guidelines for the
diagnosis and treatment of nasopharyngeal carcinoma. Cancer Commun
(Lond). 41:1195–1227. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Chen YP, Chan ATC, Le QT, Blanchard P, Sun
Y and Ma J: Nasopharyngeal carcinoma. Lancet. 394:64–80. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Zhang H, Ma J, An S, Xu L, Lu J and Jiang
C: Screen of key characteristic genes of nasopharyngeal carcinoma
(NPC) base on machine learning and analysis of their correlation
with immune cells. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 39:988–995.
2023.In Chinese. PubMed/NCBI
|
|
96
|
Cabanillas ME, McFadden DG and Durante C:
Thyroid cancer. Lancet. 388:2783–2795. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Pacifico F and Leonardi A: Role of
NF-kappaB in thyroid cancer. Mol Cell Endocrinol. 321:29–35. 2010.
View Article : Google Scholar
|
|
98
|
Abaandou L, Ghosh R and Klubo-Gwiezdzinska
J: The role of the hypothalamic-pituitary-thyroid axis in thyroid
cancer. Lancet Diabetes Endocrinol. 13:333–346. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Wang H: LINC00092 enhances LPP expression
to repress thyroid cancer development via sponging miR-542-3p. Horm
Metab Res. 56:150–158. 2024. View Article : Google Scholar
|
|
100
|
Waldman A and Schmults C: Cutaneous
squamous cell carcinoma. Hematol Oncol Clin North Am. 33:1–12.
2019. View Article : Google Scholar
|
|
101
|
Burton KA, Ashack KA and Khachemoune A:
Cutaneous squamous cell carcinoma: A review of high-risk and
metastatic disease. Am J Clin Dermatol. 17:491–508. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Gim JA, Kim C, Oh HJ, Kim KE, Jeon J, Kim
A and Baek YS: Spatial transcriptomics shows a distinctive tumour
microenvironment in the invasive versus premalignant portion of
early cutaneous squamous cell carcinoma. Exp Dermatol.
34:e701252025. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Kansara M, Teng MW, Smyth MJ and Thomas
DM: Translational biology of osteosarcoma. Nat Rev Cancer.
14:722–735. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Chen C, Xie L, Ren T, Huang Y, Xu J and
Guo W: Immunotherapy for osteosarcoma: Fundamental mechanism,
rationale, and recent breakthroughs. Cancer Lett. 500:1–10. 2021.
View Article : Google Scholar
|
|
105
|
Bao Z, Zhu R, Fan H, Ye Y, Li T and Chai
D: Aberrant expression of SPAG6 and NM23 predicts poor prognosis of
human osteosarcoma. Front Genet. 13:10125482022. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Coscio AM and Garst J: Lung cancer in
women. Curr Oncol Rep. 8:248–251. 2006. View Article : Google Scholar
|
|
107
|
Thai AA, Solomon BJ, Sequist LV, Gainor JF
and Heist RS: Lung cancer. Lancet. 398:535–554. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Zheng Y, Sadée C, Ozawa M, Howitt BE and
Gevaert O: Single-cell multimodal analysis reveals tumor
microenvironment predictive of treatment response in non-small cell
lung cancer. Sci Adv. 11:eadu21512025. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Kontic M and Markovic F: Use of DNA
methylation patterns for early detection and management of lung
cancer: Are we there yet? Oncol Res. 33:781–793. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Bonanno L, Favaretto A, Rugge M, Taron M
and Rosell R: Role of genotyping in non-small cell lung cancer
treatment: Current status. Drugs. 71:2231–2246. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Rigas JR and Kelly K: Current treatment
paradigms for locally advanced non-small cell lung cancer. J Thorac
Oncol. 2(Suppl 2): S77–S85. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Altenberger C, Heller G, Ziegler B,
Tomasich E, Marhold M, Topakian T, Müllauer L, Heffeter P, Lang G,
End-Pfützenreuter A, et al: SPAG6 and L1TD1 are transcriptionally
regulated by DNA methylation in non-small cell lung cancers. Mol
Cancer. 16:12017. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Wang Y, Liu Y, Wang R, Cao F, Guan Y, Chen
Y, An B, Qin S and Yao S: Establishment of a prognostic model
toward lung squamous cell carcinoma based on m7G-related
genes in the cancer genome atlas. Physiol Genomics. 55:427–439.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Wu Q, Yan Y, Shi S, Qi Q and Han J:
DNMT3b-mediated SPAG6 promoter hypermethylation affects lung
squamous cell carcinoma development through the JAK/STAT pathway.
Am J Transl Res. 14:6964–6977. 2022.PubMed/NCBI
|
|
115
|
Hao Q, Li J, Zhang Q, Xu F, Xie B, Lu H,
Wu X and Zhou X: Single-cell transcriptomes reveal heterogeneity of
high-grade serous ovarian carcinoma. Clin Transl Med. 11:e5002021.
View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Wang Y, Xie H, Chang X, Hu W, Li M, Li Y,
Liu H, Cheng H, Wang S, Zhou L, et al: Single-cell dissection of
the multiomic landscape of high-grade serous ovarian cancer. Cancer
Res. 82:3903–3916. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Coan M, Rampioni Vinciguerra GL, Cesaratto
L, Gardenal E, Bianchet R, Dassi E, Vecchione A, Baldassarre G,
Spizzo R and Nicoloso MS: Exploring the role of fallopian ciliated
cells in the pathogenesis of high-grade serous ovarian cancer. Int
J Mol Sci. 19:25122018. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Berdik C: Unlocking bladder cancer.
Nature. 551:S34–S35. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Grayson M: Bladder cancer. Nature.
551:S332017. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Kitchen MO, Bryan RT, Haworth KE, Emes RD,
Luscombe C, Gommersall L, Cheng KK, Zeegers MP, James ND, Devall
AJ, et al: Methylation of HOXA9 and ISL1 predicts patient outcome
in high-grade non-invasive bladder cancer. PLoS One.
10:e01370032015. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Huo FY, Li YS, Yang XY, Wang QX, Liu JJ,
Wang LK, Su YH and Sun L: Expressions of SLC22A14 and SPAG6
proteins in the ejaculated sperm of idiopathic asthenozoospermia
patients. Zhonghua Nan Ke Xue. 23:703–707. 2017.In Chinese.
|
|
122
|
Yu H, Shi X, Shao Z, Geng H, Guo S, Li K,
Gu M, Xu C, Gao Y, Tan Q, et al: Novel HYDIN variants associated
with male infertility in two Chinese families. Front Endocrinol
(Lausanne). 14:11188412023. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Xu C, Tang D, Shao Z, Geng H, Gao Y, Li K,
Tan Q, Wang G, Wang C, Wu H, et al: Homozygous SPAG6 variants can
induce nonsyndromic asthenoteratozoospermia with severe MMAF.
Reprod Biol Endocrinol. 20:412022. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Li DY, Yang XX, Tu CF, Wang WL, Meng LL,
Lu GX, Tan YQ, Zhang QJ and Du J: Sperm flagellar 2 (SPEF2) is
essential for sperm flagellar assembly in humans. Asian J Androl.
24:359–366. 2022. View Article : Google Scholar :
|
|
125
|
Wu H, Wang J, Cheng H, Gao Y, Liu W, Zhang
Z, Jiang H, Li W, Zhu F, Lv M, et al: Patients with severe
asthenoteratospermia carrying SPAG6 or RSPH3 mutations have a
positive pregnancy outcome following intracytoplasmic sperm
injection. J Assist Reprod Genet. 37:829–840. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Tan C, Meng L, Lv M, He X, Sha Y, Tang D,
Tan Y, Hu T, He W, Tu C, et al: Bi-allelic variants in DNHD1 cause
flagellar axoneme defects and asthenoteratozoospermia in humans and
mice. Am J Hum Genet. 109:157–171. 2022. View Article : Google Scholar :
|
|
127
|
Jiang T, Wang Y, Wu W, Yang Q, Wu S, Zhang
X and Xu W: Distinct germ-line genetic mutation patterns correlate
with reproductive outcomes in ICSI patients: A pilot study. Front
Genet. 16:16109432025. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Ren H, Zhang Y, Bi Y, Wang H, Fang G and
Zhao P: Target silencing of porcine SPAG6 and PPP1CC by shRNA
attenuated sperm motility. Theriogenology. 219:138–146. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Ye JW, Abbas T, Zhou JT, Chen J, Yang ML,
Huang XH, Zhang H, Ma H, Ma A, Xu B, et al: Homozygous CCDC146
mutation causes oligoasthenoteratozoospermia in humans and mice.
Zool Res. 45:1073–1087. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
He X, Liu C, Yang X, Lv M, Ni X, Li Q,
Cheng H, Liu W, Tian S, Wu H, et al: Bi-allelic loss-of-function
variants in CFAP58 cause flagellar axoneme and mitochondrial sheath
defects and asthenoteratozoospermia in humans and mice. Am J Hum
Genet. 107:514–526. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Sun W, Zhang X, Wang L, Ren G, Piao S,
Yang C and Liu Z: RNA sequencing profiles reveals progressively
reduced spermatogenesis with progression in adult cryptorchidism.
Front Endocrinol (Lausanne). 14:12717242023. View Article : Google Scholar : PubMed/NCBI
|