The molecular landscape of non-small cell lung
cancer (NSCLC) has been revolutionized by the discovery of
epidermal growth factor receptor (EGFR) mutations, which occur in
10-35% of Western patients and in ≤50% of Asian patients (1-3).
While EGFR tyrosine kinase inhibitors (TKIs) achieve unprecedented
initial response rates of 60-80%, the median progression-free
survival remains limited to 9-19 months due to the universal
development of acquired resistance (4-6).
Historically, resistance research has focused on tumor-autonomous
mechanisms, including secondary EGFR mutations (for example, T790M
and C797S), MET amplification and phenotypic transformation through
epithelial-mesenchymal transition (EMT) (5,7-9).
However, accumulating evidence reveals that dynamic
crosstalk between neoplastic cells and their microenvironment
contributes considerably to therapeutic evasion (10,11). The tumor microenvironment (TME),
comprising immune cells, cancer-associated fibroblasts (CAFs),
endothelial cells and extracellular matrix components, establishes
biochemical and physical barriers that compromise drug efficacy
through multiple mechanisms (12,13).
The present comprehensive review systematically
examines the molecular biology of IL-6 signaling networks in
EGFR-driven tumorigenesis, the multidimensional role of IL-6 in
sculpting therapy-resistant TME architectures, preclinical evidence
for IL-6 pathway inhibition in resensitizing refractory tumors and
current clinical challenges and future directions for
biomarker-driven combination therapies. We hypothesize that
targeting IL-6-mediated crosstalk between tumor cells and their
ecological niche represents a promising strategy to overcome
microenvironment-mediated resistance.
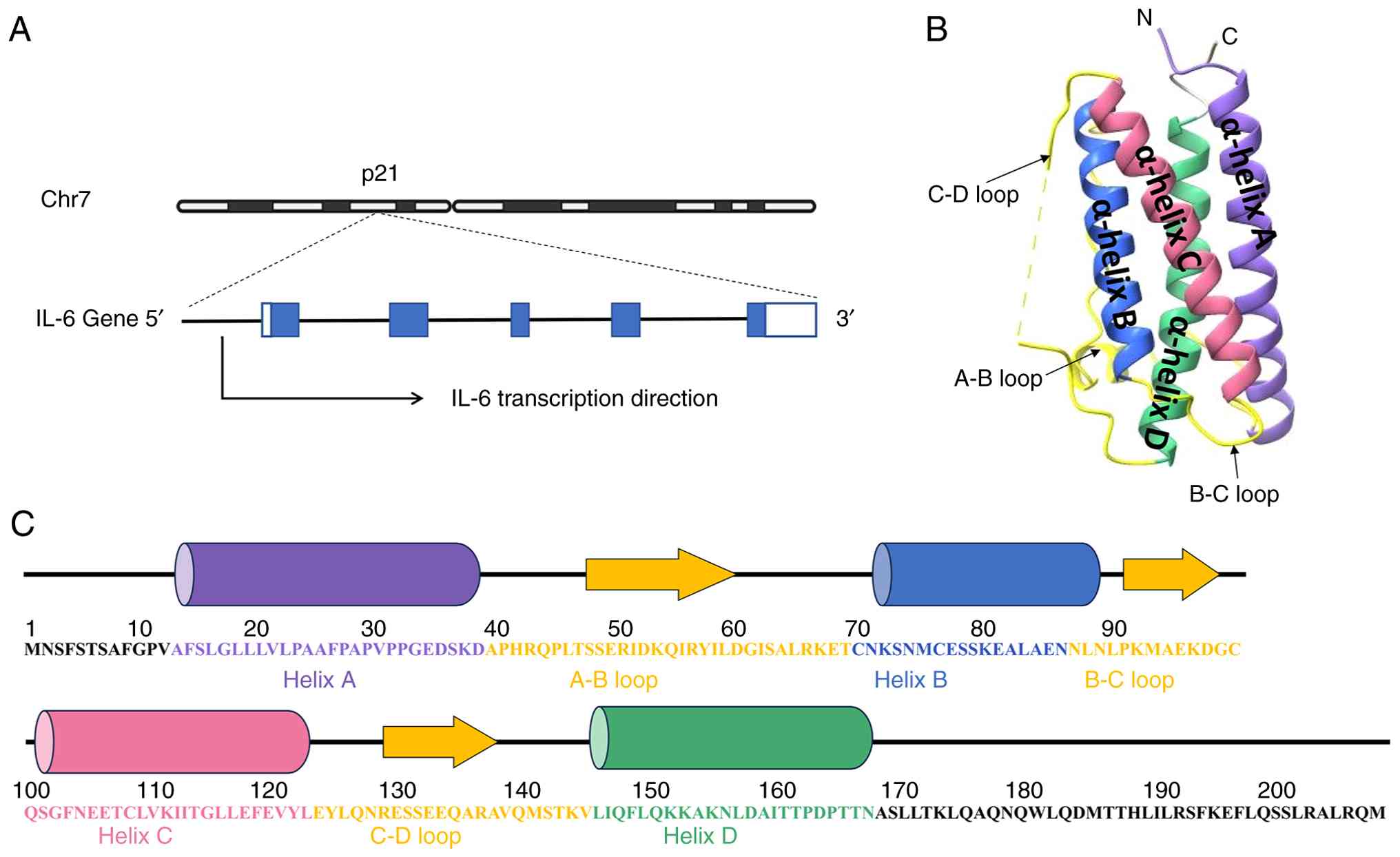
The IL-6 gene, located on chromosome 7p21, encodes a
184-amino-acid protein. Structurally, IL-6 consists of four long
α-helices and three loops at their junctions, which maturely form
following proteolytic cleavage of its signal peptide (22). IL-6 signaling is mediated by a
receptor complex consisting of IL-6, the IL-6 receptor α subunit
(IL-6Rα) and glycoprotein 130 (gp130) (23). Specifically, IL-6 first binds to
membrane-bound IL-6Rα, an 80 kDa protein featuring an extracellular
IL-6 binding domain and a minimal cytoplasmic domain. This
IL-6/IL-6Rα complex subsequently recruits gp130 (an IL-6 signal
transducer), initiating intracellular signal transduction (24). The structural organization of IL-6
is shown in Fig. 1.
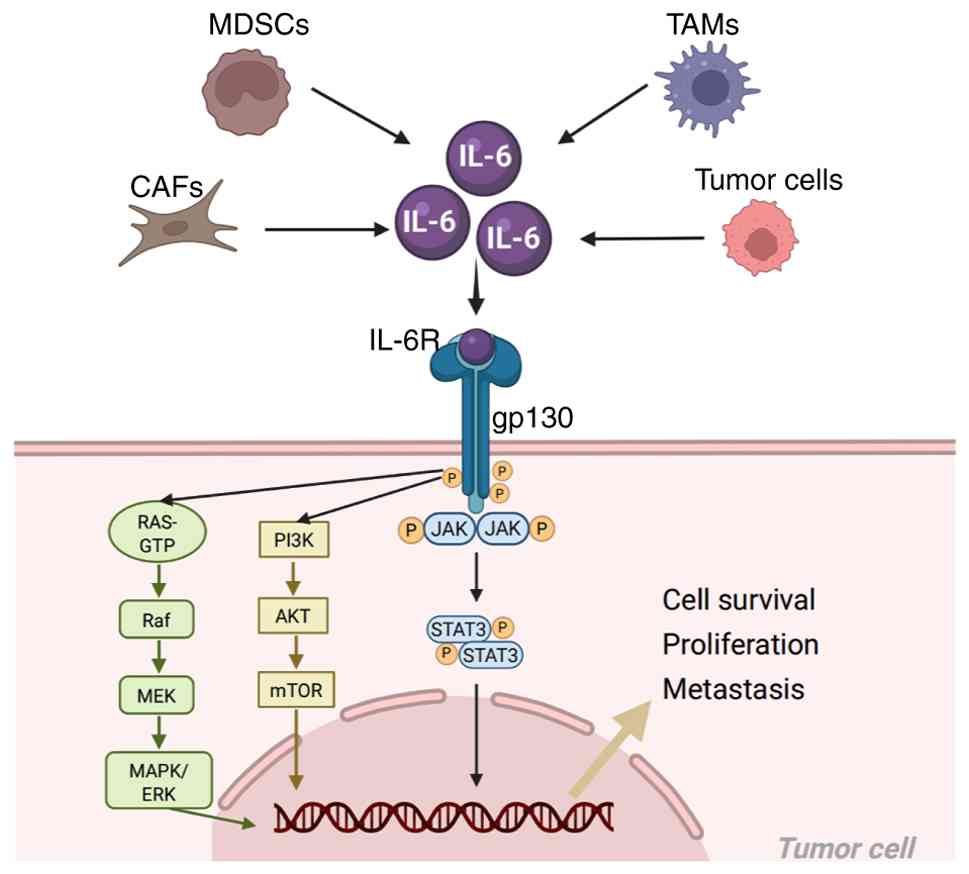
IL-6, a pleiotropic inflammatory cytokine
predominantly secreted by fibroblasts, myeloid-derived suppressor
cells (MDSCs), tumor-associated macrophages (TAMs) and tumor cells
(25-28), signals through four distinct
molecular pathways. The classical and trans-signaling pathways
constitute the two primary modes of IL-6 signal transduction
(29-31), with two additional modalities more
recently characterized (32,33). All IL-6-related cytokine receptor
complexes (IL-6/IL-6R/gp130) activate an intracellular signal: The
JAK/STAT pathway. Furthermore, gp130 phosphorylation triggers
secondary downstream pathways, including MAPK-ERK and PI3K-Akt,
which synergistically amplify oncogenic signaling (34). The nuclear translocation of
activated STAT proteins induces the transcription of
tumor-promoting genes associated with proliferation, metastasis and
immune evasion (35). Apart from
its canonical transcriptional roles in maintaining stemness,
survival, metastatic potential and immune evasion across
malignancies (36-40), cytoplasmic STAT3 drives tumor
progression through non-transcriptional mechanisms. These include
metabolic reprogramming and direct interactions with cytosolic
signaling effectors, which collectively sustain tumor expansion
(41,42). Mechanistically, through classical
membrane-bound receptor signaling and trans-signaling via soluble
IL-6R, IL-6 activates downstream JAK/STAT3, MAPK and PI3K/AKT
pathways, driving the pro-survival signaling, stemness maintenance
and immune evasion of tumor cells (43-46) (Fig.
2).
IL-6 signaling has a key influence on the prognosis
of patients with NSCLC. Accumulating evidence has demonstrated that
IL-6 is overexpressed across multiple malignancies and is
associated with tumor progression (47,48). Mechanistically, IL-6 enhances
cancer stemness properties (45,46), whereas pharmacological inhibition
of the IL-6/JAK/STAT3 axis suppresses tumorigenic potential
(45,49). Notably, IL-6 synergistically
interacts with EGFR signaling to drive NSCLC progression
(50). In EGFR-mutant
NSCLC cells, treatment with EGFR-TKI paradoxically activates
compensatory Src/IL-6/STAT3 signaling, thereby sustaining tumor
cell survival (51). Preclinical
studies have shown that IL-6 blockade markedly reduces tumor burden
(52), while JAK1/2 inhibitors
(for example ruxolitinib) effectively suppress the growth of
EGFR-mutant tumors (53).
Collectively, these findings position IL-6 as a key
molecular nexus bridging tumorigenesis and clinical prognosis in
NSCLC. The IL-6/JAK/STAT3 signaling cascade operates independently
of the mutational status of EGFR, exerting pleiotropic effects on
tumor cell behavior through both canonical and non-canonical
mechanisms.
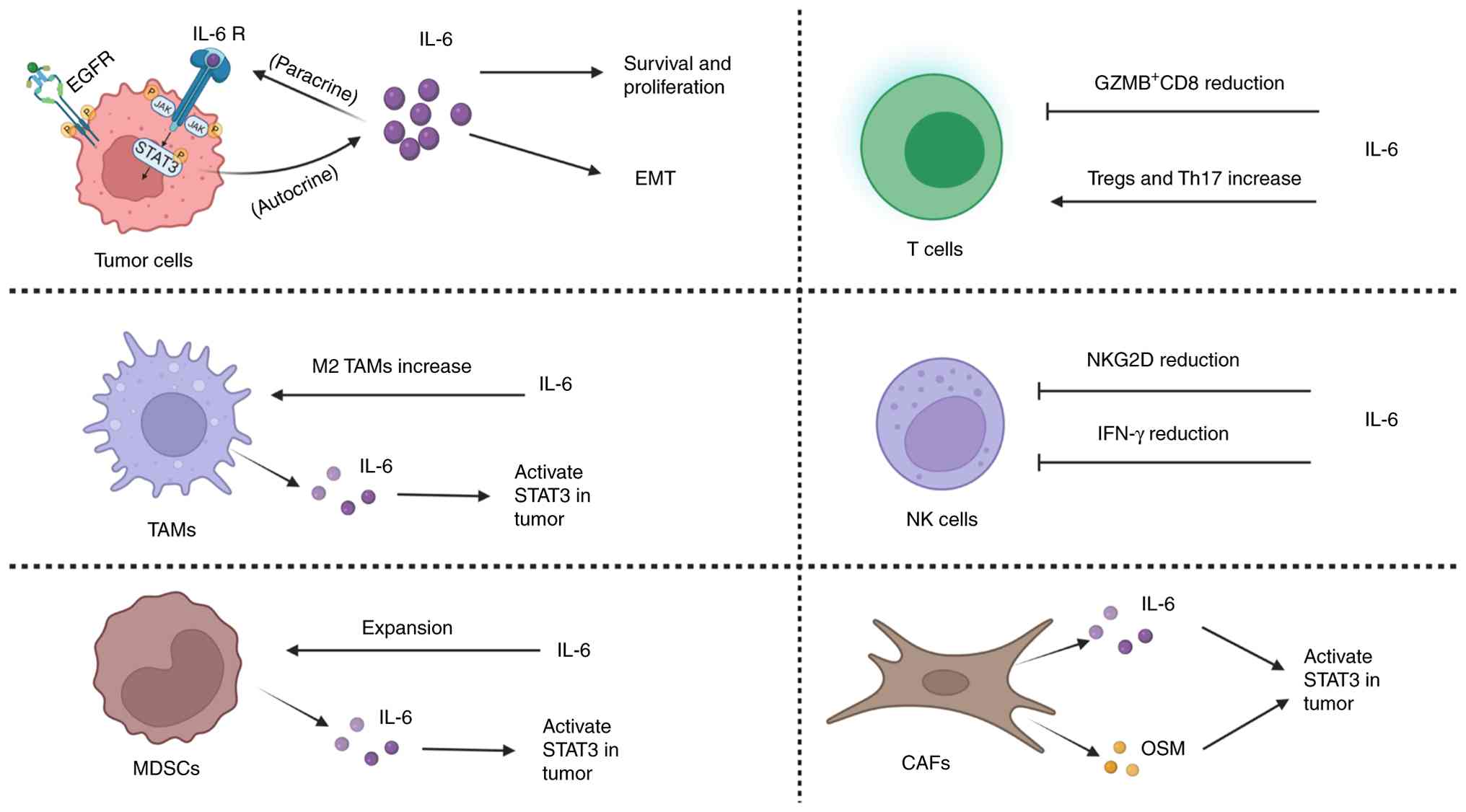
The majority of patients treated with EGFR-TKI
develop acquired resistance within 9 to 14 months of therapy
(5). Accumulating evidence
highlights the key role of IL-6 in shaping immunosuppressive
processes within the TME (63).
Furthermore, increasing data indicate a clear association between
resistance to EGFR-TKI and IL-6 signaling. IL-6 orchestrates TME
immunosuppression, leading to resistance to EGFR-TKI through
multiple mechanisms (Fig. 3).
Elevated IL-6 is consistently associated with
adverse clinical outcomes in EGFR-mutant patients with NSCLC
treated with EGFR-TKI (64).
Specifically, IL-6 positivity by immunostaining (found in 46% of
patients in one cohort) associates with notably worse PFS (43). Furthermore, higher pretreatment
serum IL-6 levels predict reduced PFS and overall survival (OS)
(65). This prognostic link
extends to the point of acquired resistance, where IL-6 levels are
substantially increased upon resistance development in both
gefitinib- and osimertinib-treated patients. Notably, after
gefitinib resistance emerges, patients with markedly elevated IL-6
have markedly shorter OS compared with those with lower levels
(44). Collectively, these
clinical observations substantiate that IL-6 plays a pivotal
regulatory role in both outcomes of and therapeutic resistance
mechanisms in EGFR-mutant patients with NSCLC.
Investigative studies reveal that IL-6 plays a
pivotal role in conferring EGFR-TKI resistance through direct
effects on tumor cells (66-68). Constitutive activation of the
IL-6/JAK2/STAT3 signaling axis is observed in resistant cellular
models (69). Mechanistically,
this cascade mediates resistance via autocrine IL-6 production and
STAT3 positive feedback activation, facilitating tumor cell
survival and proliferation both in vitro and in vivo
(67,70). Complementary preclinical
investigations reveal that pharmacological inhibition of this
signaling axis restored EGFR-TKI sensitivity in murine models,
providing therapeutic proof-of-concept (68).
Furthermore, IL-6 contributes to acquired resistance
by driving tumor cell-intrinsic EMT progression. Molecular analyses
show that IL-6 suppresses E-cadherin and transcriptionally
upregulates mesenchymal markers such as Snail and vimentin
(71). The self-sustaining
IL-6/IGF-1R/STAT3 autocrine loop has been identified as a key EMT
driver (72), with TGFβ cytokine
coactivation shown to potentiate IL-6 pathway signaling and
subsequent EMT progression. Notably, metformin administration has
been shown to suppress EGFR-TKI-resistant xenograft tumorigenesis
through dual mechanisms involving a reduction in IL-6 secretion and
a reversal of the EMT phenotype (71). These studies illustrate the key
role that IL-6 carries out in resistance to EGFR-TKI mediated by
EMT, the IL-6/JAK/STAT3 signaling pathway and other mechanisms,
thereby providing potential treatment options against resistance.
In addition, IL-6 can regulate transcription factors governing DNA
repair fidelity and cell cycle checkpoint control, potentially
impairing genomic replication accuracy (73,74). These direct actions of IL-6 on
tumor cells underscore its importance in survival, proliferation
and resistance development.
Lymphocytes are fundamental to antitumor immunity,
yet EGFR-TKI resistance is frequently characterized by the
depletion of CD8+ T cells and the expansion of
immunosuppressive subsets (75,76). IL-6 acts as a central orchestrator
of this T-cell dysfunction through distinct molecular mechanisms
(52,77-79). Mechanistically, the IL-6 signaling
exerts dual immunomodulatory effects by suppressing key cytotoxic
mediators (IFN-γ, GM-CSF and CXCL9/10) while concurrently inducing
IL-10 secretion and Th2/Th17-polarizing factors, thereby
reprogramming CD8+ T-cell differentiation toward
dysfunctional states (80). In
EGFR-TKI-resistant NSCLC microenvironments, IL-6-mediated
downregulation of granzyme B expression considerably impairs
CD8+ T-cell cytotoxic capacity, establishing a direct
association between cytokine signaling and immune effector
dysfunction (81). Importantly,
IL-6 drives T-cell exhaustion by engaging the programmed cell
death-1 (PD-1)/PD-ligand (PD-L1) checkpoint axis. In the TME,
macrophage-derived IL-6 promotes the membrane presentation of PD-1
on T cells via the Rab37/IL-6/STAT3 axis (82). Simultaneously, oncogenic
IL-6/JAK/STAT3 signaling in EGFR-mutant tumor cells
transcriptionally activates PD-L1 expression, thereby conferring a
survival advantage through immune escape (83,84).
NK cells serve as a primary innate defense, but
their surveillance capability is severely compromised in the
EGFR-TKI-resistant microenvironment. Distinct from its effects on T
cells, IL-6 blunts NK cell effector function by targeting
activating receptors. Tumor-derived IL-6 triggers STAT3
phosphorylation in NK cells, which transcriptionally downregulates
surface expression of NKp30 and NKG2D, thereby desensitizing
resistant cells to NK-mediated lysis (81,92). Additionally, STAT3 binding to the
IFN-γ promoter region directly antagonizes IFN-γ production
(93). This IL-6-dependent
downregulation of recognition receptors and cytokines establishes a
specific mechanism of innate immune evasion during therapy.
CAFs are key stromal components that drive tumor
progression through the secretion of soluble factors, notably IL-6
and TGF-β, orchestrating pro-tumorigenic processes such as
angiogenesis, invasive growth and metastatic dissemination
(94-98). CAFs themselves represent a notable
source of IL-6 within the TME. IL-6 directly impacts CAF behavior,
stimulating the proliferation of normal fibroblasts and inducing a
CAF-like phenotype (99,100). Pathophysiological investigations
have revealed that bidirectional communication between EGFR-mutant
NSCLC cells and CAFs sustains constitutive activation of the
IL-6/JAK/STAT3 cascade in malignant cells, thereby conferring
therapeutic resistance to EGFR-TKI across preclinical in
vitro and in vivo models (67,101). Targeting the paracrine
IL-6/JAK/STAT3 loop between fibroblasts and tumor cells (67) or employing agents such as
tranilast that decrease CAFs-derived IL-6, effectively abrogates
CAFs-mediated resistance (101).
Beyond IL-6, CAFs also express other cytokines such as oncostatin-M
(OSM). A preclinical study showed that combination therapy with
EGFR-TKI and JAK1 inhibitors (for example, filgotinib) potently
inhibits the fibroblast-activated OSMRs/JAK1/STAT3 axis, disrupting
stroma-driven pathway crosstalk to prevent adaptive drug resistance
(102).
TAMs, which predominantly exhibit M2 polarization
with a few M1 subpopulations, serve as pivotal mediators of tumor
progression, metastatic dissemination and formation of an
immunosuppressive microenvironment (103,104). In EGFR-TKI-resistant tissues,
IL-6/JAK/STAT3 signaling drives the polarization of TAMs toward a
protumor M2 phenotype (105).
IL-6/JAK/STAT3 signaling orchestrates three cardinal TAM features,
chemotactic migration, survival maintenance and M2 polarization,
which are characterized by arginase-1 (Arg1) overexpression coupled
with inducible nitric oxide synthase downregulation (106). Concurrently, TAMs can also
secrete IL-6 to promote lung cancer progression and metastasis
(107,108). Previous mechanistic insights
reveal that phase separation of YY1 transcriptional complexes in M2
TAMs enhances chromatin accessibility at IL-6 regulatory regions,
amplifying IL-6 transcription and creating a self-reinforcing loop
(109). Functional crosstalk
analyses also demonstrate that TAM-derived IL-6 activates
COX-2/PGE2 cascades in adjacent tumor cells, inducing EMT (110). Thus, IL-6 coordinates a dual
mechanism in the myeloid compartment: Promoting M2-mediated tissue
remodeling and enforcing intercellular communication.
MDSCs are heterogeneous progenitors that markedly
contribute to therapeutic refractoriness (111). Elevated cytokine levels in the
serum of patients with EGFR-TKI-resistant NSCLC associate with MDSC
expansion and poor prognosis (112,113). IL-6 serves as a master regulator
of MDSC biology through specific epigenetic and metabolic
reprogramming. Mechanistically, STAT3-mediated chromatin remodeling
at the Arg1 promoter drives the immunosuppressive polarization of
MDSCs (114-120). Concurrently, IL-6 primes MDSCs
to undergo metabolic rewiring (enhanced glycolysis/oxidative
phosphorylation) and suppresses their antigen presentation
machinery (121). These
functional alterations collectively potentiate MDSC-mediated T-cell
suppression, positioning the IL-6 axis as a strategic target to
dismantle the myeloid barrier in resistant tumors.
Taken together, IL-6 serves as a pivotal nexus
connecting EGFR-TKI resistance with the immunosuppressive TME.
Under persistent selective pressure from EGFR-TKI, surviving tumor
cells exhibit markedly upregulated IL-6 secretion. Once released
into the microenvironment, IL-6 triggers sustained activation of
the intrinsic JAK/STAT3 pathway via either classical or
trans-signaling modes. This establishes a self-reinforcing
autocrine loop that directly orchestrates resistance-associated
phenotypes, including EMT and the expression of pro-survival genes.
Importantly, IL-6 also actively induces and sustains an
immunosuppressive TME. This landscape is characterized by the
functional impairment of effector T cells and NK cells, coupled
with the recruitment and polarization of suppressive subsets such
as Tregs, TAMs and MDSCs. Notably, this remodeling is not a
unidirectional process; immunosuppressive cells and stromal
components (for example, TAMs and CAFs) also serve as prolific
sources of IL-6, thereby amplifying the signaling cascade.
Paracrine IL-6 from these accessory cells feeds back to the tumor
cells, further fueling downstream pathways to sustain tumor growth
and the resistant phenotype. Within the context of
EGFR-mutant NSCLC, this reciprocal 'crosstalk' and mutual
reinforcement among tumor, immune and stromal cells constitute a
vicious cycle that drives EGFR-TKI resistance. Consequently, IL-6
functions as a key bridge, inextricably linking intrinsic TKI
tolerance mechanisms with complex immune evasion strategies to
establish a synergistic resistance axis. Targeting this IL-6-driven
axis thus provides a compelling theoretical rationale for
developing novel combination therapeutic strategies to overcome
EGFR-TKI resistance.
Multiple preclinical studies and clinical trials
examining IL-6 pathway blockade in EGFR-TKI-resistant NSCLC have
been summarized in Table I
(44,53,68,69,101,102,122-128), some of which are aforementioned.
The majority of these studies have focused only on the production
of IL-6 and its signaling pathway.
Siltuximab (CNTO328), an IL-6 neutralizing antibody,
inhibited the proliferation of H1650 cells, whereas the combination
of siltuximab and erlotinib resulted in more pronounced inhibition
of tumor growth in a mouse model (122). In EGFR-mutant tumor cells
that are resistant to gefitinib due to IL-6 induction, miR-206
directly targets the 3'-UTR of intracellular IL-6 messenger RNA to
block IL-6/JAK/STAT3 signaling, thereby restoring gefitinib
sensitivity (123). Compared
with no treatment, the addition of IL-6 to erlotinib-sensitive
cells increased drug resistance. Additionally, the presence of IL-6
did not prevent the restoration of cell sensitivity to erlotinib by
treatment with P6 (a JAK1/2 inhibitor) (68). The JAK inhibitor AZD1480 showed
anticancer and antiangiogenic effects (129,130). AZD1480 alleviated
sevoflurane-induced lung metastasis by disrupting the
IL-6/JAK/STAT3 pathway (131).
Furthermore, in mice bearing EGFR-driven lung cancer,
AZD1480 showed marked antitumor activity and extended survival time
(124). However, erlotinib and
momelotinib (JAK1/2 and TBK1 inhibitors) did not appear to provide
a greater benefit compared with erlotinib monotherapy in
EGFR-mutated patients with NSCLC (125).
Ibrutinib consistently and effectively suppressed
the levels of phosphorylated STAT3, which is a powerful inhibitor
of IL-6 and laminin α5/FAK signaling. The combination of ibrutinib
and osimertinib can reverse resistance to osimertinib and inhibit
tumor growth in xenografts (44).
Similar findings have been reported for AZD9150 (an inhibitor of
STAT3), in which systemic treatment of mice bearing PC-9 tumors
with AZD9150 led to the almost complete suppression of tumor growth
(126). HKB99 (a PGAM1
allosteric inhibitor) disrupted IL-6/JAK/STAT3 signaling by
decreasing the level of phosphorylated (p)-STAT3. Additionally,
when combined with osimertinib, HKB99 exerted a synergistic
tumoricidal effect and markedly restored the sensitivity to
EGFR-TKI (69). A cell experiment
revealed that WP1066, a known STAT3 inhibitor, could cause H1650
cells to undergo apoptosis, with an inhibitory effect on tumor
growth (128). When EGFR-TKI and
TPCA-1, a dual inhibitor of both IKKs and STAT3, are coupled
together, EGFR-mutated NSCLC is more sensitive to gefitinib
(127).
Homoharringtonine possesses anticancer properties,
as demonstrated by its ability to reversibly inhibit the
IL-6-induced phosphorylation of STAT3 at the Tyr705 site in a mouse
model of EGFR-TKI resistance (132). A naturally occurring chemical
substance called polyphyllin I (PPI) has anticancer properties and
reduces the activation of the IL-6/STAT3 pathway in
erlotinib-resistant cells. The combined use of PPI and EGFR-TKI
reduces tumor growth and reverses acquired resistance in xenografts
(133).
However, following the onset of EGFR-TKI resistance,
therapeutic strategies targeting the IL-6/JAK/STAT3 pathway alone
often yield suboptimal results. One of the primary hurdles in
achieving robust clinical efficacy is the inherent cytokine
redundancy within the TME. IL-6 belongs to a larger family of
cytokines, including leukemia inhibitory factor, OSM and IL-11, all
of which converge on the common signal-transducing receptor
subunit, gp130 (48,134-136). Furthermore, the IL-6/JAK/STAT3
axis operates as an integral part of a complex, interconnected
network. Tumor cells frequently develop compensatory mechanisms to
bypass specific pathway blockade. For instance, the inhibition of
JAK/STAT3 signaling may trigger compensatory activation of the
PI3K/AKT or MEK pathways, enabling cancer cells to sustain survival
and proliferation, thereby limiting therapeutic efficacy (137,138). Clinical data (NCT00841191) from
trials of Siltuximab (a chimeric anti-IL-6 monoclonal antibody)
have shown that while systemic CRP levels (a surrogate for IL-6
activity) are successfully suppressed, intratumoral p-STAT3 levels
often persist, suggesting that the 'gp130-JAK-STAT3' hub remains
fueled by alternative ligands. This signaling bypass renders the
selective blockade of a single cytokine insufficient to dismantle
the pro-tumorigenic niche, necessitating a shift toward targeting
the shared gp130 receptor or the downstream STAT3 transcription
factor.
Immunotherapy has been among the greatest advances
in previous years for the treatment of solid tumors, including
NSCLC (139,140). EGFR-TKI resistance upregulates
PD-L1 expression in NSCLC, providing a theoretical basis for
immunotherapy (Table II).
However, negative results from large clinical studies suggest that
patients who develop resistance to EGFR-TKI have difficulty
benefiting from treatment with immunotherapy alone or immunotherapy
combined with chemotherapy (141-143). This poor response to
immunotherapy is largely attributed to an immunosuppressive TME.
Here, IL-6 carries out a pivotal role. IL-6 levels are
substantially increased upon resistance development in
EGFR-TKI-treated patients (44,65). IL-6 may orchestrate multifaceted
immunomodulatory effects within the TME of EGFR-mutant NSCLC
through the following mechanisms: First, suppression of antitumor
immunity: IL-6 exerts inhibitory effects on effector T cells, NK
cells and DCs, with experimental evidence suggesting that
IL-6/JAK/STAT3 pathway activation in these immune subsets likely
drives downregulation of the antitumor response (144-146). Second, promotion of
immunosuppressive networks: Concurrently, IL-6 enhances the
expansion and function of immunosuppressive cell populations,
including MDSCs and Tregs, while polarizing macrophages toward the
M2 phenotype (147,148). Third, immune checkpoint
modulation: IL-6 further disrupts immune-tumor crosstalk by
upregulating PD-1/PD-L1 expression, thereby fostering an
immune-evasive niche (149,150). These effects contribute to a
highly immunosuppressive TME, which in turn may mediate resistance
to EGFR-TKI. The inhibition of IL-6/JAK/STAT3 signaling can also
affect the TME and has implications for antitumor immunity.
Consequently, dual targeting of IL-6 signaling and the PD-1/PD-L1
axis represents a promising therapeutic approach to overcome
resistance to EGFR-TKI in NSCLC.
Currently, there are Phase I and II clinical trials
evaluating the efficacy and safety of the combination of anti-IL-6R
and anti-IL-6 with immunotherapy in patients with NSCLC (Table SI). The CANOPY-1 trial
demonstrated that elevated baseline plasma IL-6 levels associate
with shorter OS in immunotherapy-treated patients with NSCLC
(149). Similarly, longitudinal
increases in IL-6 levels during PD-1/PD-L1 blockade were associated
with diminished therapeutic responses in NSCLC cohorts (82). Furthermore, elevated plasma
cytokine profiles, including those of IL-6, TNF and IL-8, have been
implicated in immunotherapy resistance (151). Preclinical studies substantiate
these findings, showing that inhibition of the IL-6 pathway
augments immunotherapy efficacy through immune cell modulation
within the TME (152,153). For instance, dual administration
of anti-IL-6 and anti-PD-1 antibodies in pancreatic cancer murine
models enhanced antitumor activity and promoted T lymphocyte
infiltration (153).
Analogously, coordinated blockade of IL-6 and PD-1/PD-L1 signaling
in melanoma models upregulates the expression of T-cell-recruiting
chemokines and increases the infiltration of IFN-γ-producing
CD4+ T cells, yielding synergistic antitumor effects
(152). Notably, retrospective
analyses revealed that patients with NSCLC with low baseline IL-6
levels in plasma or tumor tissues derived greater clinical benefit
from immunotherapy. Preclinically, dual targeting of IL-6 and
immune checkpoints attenuated tumor growth and improved survival in
NSCLC-bearing mice. Mechanistically, inhibition of IL-6 expression
increases CD8+ T-cell infiltration while reducing the
numbers of PD1+CD8+-exhausted T cells and M2
macrophages within the TME (52,82). Moreover, IL-6 blockade sensitized
tumors to immunotherapy through the activation of T and NK cells in
EGFR-mutant genetically engineered mouse model (81). Depletion of IL-6 restored the
cytotoxic potential of NK cells in EGFR-TKI-resistant tumors
(81). Collectively, these
findings suggest that IL-6 is a rational immunomodulatory target
for increasing immunotherapy efficacy in EGFR-TKI-resistant NSCLC.
However, definitive clinical validation through dose-optimized
trials remains imperative.
The hyperactivation of STAT3, a downstream effector
of IL-6, a key transcriptional regulator for angiogenic factors,
most notably vascular endothelial growth factor, thereby
facilitating the neovascularization required for tumor maintenance
and dissemination (154,155). Consequently, the IL-6/JAK/STAT3
axis acts as a pro-angiogenic signaling node; its activation not
only promotes tumor cell survival but also remodels the vascular
microenvironment. Preclinical evidence supports this strategy: the
JAK inhibitor AZD1480 has demonstrated dual anticancer and
anti-angiogenic properties (129,130). Furthermore, in murine models of
EGFR-driven lung cancer, AZD1480 treatment elicits marked antitumor
activity and notably extended survival (124), underscoring the potential of
targeting this axis to suppress both tumor growth and pathological
angiogenesis.
In conclusion, the IL-6/JAK/STAT3 signaling axis
represents a pivotal mechanism of adaptive resistance in
EGFR-mutant NSCLC, orchestrated through intricate crosstalk
between tumor cells, stromal components and infiltrating immune
subsets within the TME, leading to improved immunotherapy efficacy.
While preclinical data have demonstrated that IL-6 blockade can
restore sensitivity to EGFR-TKI and potentially sensitize tumors to
immunotherapy, the translation of these findings into clinical
practice faces hurdles. Substantial preclinical and clinical
research will be needed to determine the exact efficacy of this
strategy.
Currently, large-scale Phase III clinical trials
specifically evaluating IL-6/JAK/STAT3 inhibitors in EGFR-mutant
NSCLC populations are lacking. The majority of existing evidence is
derived from broader NSCLC cohorts or early-phase studies. A
considerable challenge observed in immunotherapy trials, such as
CANOPY-1, is the variable efficacy of cytokine blockade,
underscoring the necessity of identifying specific responder
populations. Furthermore, pharmacological interactions pose a
translational barrier; for instance, elevated plasma IL-6
concentrations have been associated with reduced metabolic activity
of osimertinib, potentially altering drug exposure and efficacy
(156). This highlights the need
for rigorous pharmacokinetic evaluations when combining IL-6
inhibitors with third-generation EGFR-TKI.
The lack of robust biomarkers for patient
stratification remains a major limiting factor. Plasma IL-6 and
soluble IL-6R levels have shown prognostic value, where elevated
concentrations associate with shorter OS in patients treated with
EGFR-TKI or immunotherapy (44,52,149). Tissue p-STAT3 levels serve as a
direct indicator of downstream signaling activation. validating
these biomarkers in prospective trials is essential to transition
from general cytokine inhibition to precision medicine
strategies.
Despite the compelling preclinical rationale
connecting IL-6 signaling to EGFR-TKI resistance, several key
knowledge gaps must be bridged to facilitate successful clinical
translation. First, the spatiotemporal heterogeneity of the IL-6
pathway remains elusive. It is imperative to determine whether the
dominant cellular sources of IL-6, and the intensity of signaling,
vary between primary tumors and metastatic sites or evolve
dynamically from the initial TKI-sensitive phase to the onset of
acquired resistance (67,81). Second, the optimal timing of
intervention is currently undefined. Future studies must
distinguish whether IL-6 blockade yields superior outcomes as an
upfront prophylactic strategy to delay resistance or as a salvage
regimen upon disease progression. Third, the choice of optimal
therapeutic drugs warrants comparative investigation. The
efficacy-toxicity profiles of directly neutralizing IL-6, blocking
IL-6R, inhibiting JAK, vs. targeting STAT3 downstream, remain to be
systematically evaluated in the context of EGFR-mutant NSCLC (see
Table I for preclinical agents).
Finally, translational success will depend on a holistic
understanding of the dynamic crosstalk between IL-6 and other
oncogenic pathways, as well as the optimization of dosing schedules
and patient selection to manage potential side effects. Addressing
these complexities is essential to transform IL-6 inhibition from a
theoretical concept into a precise, effective combination strategy
for EGFR-mutant NSCLC.
Not applicable.
QW, CH and YZ wrote the manuscript and designed all
the figures and tables in consultation with the other authors. HZ,
CQ and ST contributed to the writing and editing of the manuscript.
WL, YL and PT developed the concept and reviewed and edited the
manuscript. All the authors read and approved the final version of
the manuscript. Data authentication is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
This work was supported by the National Natural Science
Foundation of China (grant nos. 82473213, 82470099 and
92159302).
|
1
|
Jänne PA, Baik C, Su WC, Johnson ML,
Hayashi H, Nishio M, Kim DW, Koczywas M, Gold KA, Steuer CE, et al:
Efficacy and safety of patritumab deruxtecan (HER3-DXd) in EGFR
inhibitor-resistant, EGFR-Mutated Non-small cell lung cancer.
Cancer Discov. 12:74–89. 2022. View Article : Google Scholar
|
|
2
|
Jordan EJ, Kim HR, Arcila ME, Barron D,
Chakravarty D, Gao J, Chang MT, Ni A, Kundra R, Jonsson P, et al:
Prospective comprehensive molecular characterization of lung
adenocarcinomas for efficient patient matching to approved and
emerging therapies. Cancer Discov. 7:596–609. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Xu K, Wang H, Li S, Zhao L, Liu X, Liu Y,
Ye L, Liu X, Li L and He Y: Changing profile of lung cancer
clinical characteristics in China: Over 8-year population-based
study. Chin Med J Pulm Crit Care Med. 1:188–194. 2023. View Article : Google Scholar
|
|
4
|
Passaro A, Jänne PA, Mok T and Peters S:
Overcoming therapy resistance in EGFR-mutant lung cancer. Nat
Cancer. 2:377–391. 2021. View Article : Google Scholar
|
|
5
|
Westover D, Zugazagoitia J, Cho BC, Lovly
CM and Paz-Ares L: Mechanisms of acquired resistance to first- and
second-generation EGFR tyrosine kinase inhibitors. Ann Oncol.
29(suppl 1): i10–i19. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tian X, Gu T, Lee MH and Dong Z: Challenge
and countermeasures for EGFR targeted therapy in non-small cell
lung cancer. Biochim Biophys Acta Rev Cancer. 1877:1886452022.
View Article : Google Scholar
|
|
7
|
Rotow J and Bivona TG: Understanding and
targeting resistance mechanisms in NSCLC. Nat Rev Cancer.
17:637–658. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Han J, Wang Y, Zhong L, Zhou H, Yu M, Li
Y, Lu Y, Wang Y and Zhu J: T790M mutation in stage Iv EGFR-mutated
NSCLC patient with acquired resistance reverted to original 19Del
mutation after administration of a series of precision treatments:
a case report. Precis Clin Med. 1:129–133. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sun SY: Taking early preventive
interventions to manage the challenging issue of acquired
resistance to third-generation EGFR inhibitors. Chin Med J Pulm
Crit Care Med. 1:3–10. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu J, Zhang Y, Liu K, Shi J, Yan M, Xu J,
Liu Y and Zhang S: Cross-talk between cancer-associated fibroblasts
and dormant cancer cells: Current status and promising therapeutic
potential. Cancer Cell Int. 26:242025. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rahal Z, El Darzi R, Moghaddam SJ, Cascone
T and Kadara H: Tumour and microenvironment crosstalk in NSCLC
progression and response to therapy. Nat Rev Clin Oncol.
22:463–482. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Spranger S and Gajewski TF: Impact of
oncogenic pathways on evasion of antitumour immune responses. Nat
Rev Cancer. 18:139–147. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
de visser KE and Joyce JA: The evolving
tumor microenvironment: From cancer initiation to metastatic
outgrowth. Cancer Cell. 41:374–403. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hou J, Karin M and Sun B: Targeting
cancer-promoting inflammation-have anti-inflammatory therapies come
of age? Nat Rev Clin Oncol. 18:261–279. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fisher DT, Appenheimer MM and Evans SS:
The two faces of IL-6 in the tumor microenvironment. Semin Immunol.
26:38–47. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Abulaiti A, Shintani Y, Funaki S, Nakagiri
T, Inoue M, Sawabata N, Minami M and Okumura M: Interaction between
non-small-cell lung cancer cells and fibroblasts via enhancement of
TGF-β signaling by IL-6. Lung Cancer. 82:204–213. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Song Z, Lin Y, Ye X, Feng C, Lu Y, Yang G
and Dong C: Expression of IL-1α and IL-6 is associated with
progression and prognosis of human cervical cancer. Med Sci Monit.
22:4475–4481. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ma Y, Ren Y, Dai ZJ, Wu CJ, Ji YH and Xu
J: IL-6, IL-8 and TNF-alpha levels correlate with disease stage in
breast cancer patients. Adv Clin Exp Med. 26:421–426. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Feng L, Qi Q, Wang P, Chen H, Chen Z, Meng
Z and Liu L: Serum levels of IL-6, IL-8, and IL-10 are indicators
of prognosis in pancreatic cancer. J Int Med Res. 46:5228–5236.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liang B, Li L, Miao R, Wang J, Chen Y, Li
Z, Zou X and Zhou M: Expression of interleukin-6 and integrin ανβ6
in colon cancer: Association with clinical outcomes and prognostic
implications. Cancer Invest. 37:174–184. 2019. View Article : Google Scholar
|
|
21
|
Tsai MS, Chen WC, Lu CH and Chen MF: The
prognosis of head and neck squamous cell carcinoma related to
immunosuppressive tumor microenvironment regulated by IL-6
signaling. Oral Oncol. 91:47–55. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kaur S, Bansal Y, Kumar R and Bansal G: A
panoramic review of IL-6: Structure, pathophysiological roles and
inhibitors. Bioorg Med Chem. 28:1153272020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rose-John S, Jenkins BJ, Garbers C, Moll
JM and Scheller J: Targeting IL-6 trans-signalling: Past, present
and future prospects. Nat Rev Immunol. 23:666–681. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yao X, Huang J, Zhong H, Shen N, Faggioni
R, Fung M and Yao Y: Targeting interleukin-6 in inflammatory
autoimmune diseases and cancers. Pharmacol Ther. 141:125–139. 2014.
View Article : Google Scholar
|
|
25
|
Nozawa H, Chiu C and Hanahan D:
Infiltrating neutrophils mediate the initial angiogenic switch in a
mouse model of multi-stage carcinogenesis. Proc Natl Acad Sci USA.
1032006.
|
|
26
|
Walter M, Liang S, Ghosh S, Hornsby PJ and
Li R: Interleukin 6 secreted from adipose stromal cells promotes
migration and invasion of breast cancer cells. Oncogene.
28:2745–2755. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nagasaki T, Hara M, Nakanishi H, Takahashi
H, Sato M and Takeyama H: Interleukin-6 released by colon
cancer-associated fibroblasts is critical for tumour angiogenesis:
Anti-interleukin-6 receptor antibody suppressed angiogenesis and
inhibited tumour-stroma interaction. Br J Cancer. 110:469–478.
2014. View Article : Google Scholar
|
|
28
|
Kumari N, Dwarakanath BS, Das A and Bhatt
AN: Role of interleukin-6 in cancer progression and therapeutic
resistance. Tumour Biol. 37:11553–11572. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wolf J, Waetzig GH, Chalaris A, Reinheimer
TM, Wege H, Rose-John S and Garbers C: Different soluble forms of
the interleukin-6 family signal transducer gp130 fine-tune the
blockade of interleukin-6 trans-signaling. J Biol Chem.
291:16186–16196. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Scheller J, Chalaris A, Schmidt-Arras D
and Rose-John S: The pro- and anti-inflammatory properties of the
cytokine interleukin-6. Biochim Biophys Acta. 1813:878–888. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Orange ST, Leslie J, Ross M, Mann DA and
Wackerhage H: The exercise IL-6 enigma in cancer. Trends Endocrinol
Metab. 34:749–763. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Arnold P, Lückstädt W, Li W, Boll I, Lokau
J, Garbers C, Lucius R, Rose-John S and Becker-Pauly C: Joint
reconstituted signaling of the IL-6 receptor via extracellular
vesicles. Cells. 9:13072020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Heink S, Yogev N, Garbers C, Herwerth M,
Aly L, Gasperi C, Husterer V, Croxford AL, Möller-Hackbarth K,
Bartsch HS, et al: Trans-presentation of IL-6 by dendritic cells is
required for the priming of pathogenic TH17 cells. Nat Immunol.
18:74–85. 2017. View Article : Google Scholar :
|
|
34
|
Schaper F and Rose-John S: Interleukin-6:
Biology, signaling and strategies of blockade. Cytokine Growth
Factor Rev. 26:475–487. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hu X, Li J, Fu M, Zhao X and Wang W: The
JAK/STAT signaling pathway: From bench to clinic. Signal Transduct
Target Ther. 6:4022021. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chen RY, Yen CJ, Liu YW, Guo CG, Weng CY,
Lai CH, Wang JM, Lin YJ and Hung LY: CPAP promotes angiogenesis and
metastasis by enhancing STAT3 activity. Cell Death Differ.
27:1259–1273. 2020. View Article : Google Scholar :
|
|
37
|
Chen H, Zhou W, Bian A, Zhang Q, Miao Y,
Yin X, Ye J, Xu S, Ti C, Sun Z, et al: Selectively targeting STAT3
using a small molecule inhibitor is a potential therapeutic
strategy for pancreatic cancer. Clin Cancer Res. 29:815–830. 2023.
View Article : Google Scholar
|
|
38
|
McGuire JJ, Frieling JS, Lo CH, Li T,
Muhammad A, Lawrence HR, Lawrence NJ, Cook LM and Lynch CC:
Mesenchymal stem cell-derived interleukin-28 drives the selection
of apoptosis resistant bone metastatic prostate cancer. Nat Commun.
12:7232021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hong C, Schubert M, Tijhuis AE, Requesens
M, Roorda M, van den Brink A, Ruiz LA, Bakker PL, van der Sluis T,
Pieters W, et al: cGAS-STING drives the IL-6-dependent survival of
chromosomally instable cancers. Nature. 607:366–373. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wong GL, Manore SG, Doheny DL and Lo HW:
STAT family of transcription factors in breast cancer: Pathogenesis
and therapeutic opportunities and challenges. Semin Cancer Biol.
86(Pt 3): 84–106. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Huynh J, Chand A, Gough D and Ernst M:
Therapeutically exploiting STAT3 activity in cancer-using tissue
repair as a road map. Nat Rev Cancer. 19:82–96. 2019. View Article : Google Scholar
|
|
42
|
Wang R, Du TT, Liu WQ, Liu YC, Yang YD, Hu
JP, Ji M, Yang BB, Li L and Chen XG: Discovery, optimization, and
evaluation of novel N-(Benzimidazol-5-yl)-1,3,4-thiadiazol-2-amine
analogues as potent STAT3 inhibitors for cancer treatment. J Med
Chem. 66:12373–12395. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Tamura T, Kato Y, Ohashi K, Ninomiya K,
Makimoto G, Gotoda H, Kubo T, Ichihara E, Tanaka T, Ichimura K, et
al: Potential influence of interleukin-6 on the therapeutic effect
of gefitinib in patients with advanced non-small cell lung cancer
harbouring EGFR mutations. Biochem Biophys Res Commun. 495:360–367.
2018. View Article : Google Scholar
|
|
44
|
Li L, Li Z, Lu C, Li J, Zhang K, Lin C,
Tang X, Liu Z, Zhang Y, Han R, et al: Ibrutinib reverses
IL-6-induced osimertinib resistance through inhibition of Laminin
α5/FAK signaling. Commun Biol. 5:1552022. View Article : Google Scholar
|
|
45
|
Hsu HS, Lin JH, Hsu TW, Su K, Wang CW,
Yang KY, Chiou SH and Hung SC: Mesenchymal stem cells enhance lung
cancer initiation through activation of IL-6/JAK2/STAT3 pathway.
Lung Cancer. 75:167–177. 2012. View Article : Google Scholar
|
|
46
|
Xie C, Liang C, Wang R, Yi K, Zhou X, Li
X, Chen Y, Miao D, Zhong C and Zhu J: Resveratrol suppresses lung
cancer by targeting cancer stem-like cells and regulating tumor
microenvironment. J Nutr Biochem. 112:1092112023. View Article : Google Scholar
|
|
47
|
Thuya WL, Cao Y, Ho PC, Wong AL, Wang L,
Zhou J, Nicot C and Goh BC: Insights into IL-6/JAK/STAT3 signaling
in the tumor microenvironment: Implications for cancer therapy.
Cytokine Growth Factor Rev. 85:26–42. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Soler MF, Abaurrea A, Azcoaga P, Araujo AM
and Caffarel MM: New perspectives in cancer immunotherapy:
Targeting IL-6 cytokine family. J Immunother Cancer.
11:e0075302023. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chen SF, Zhang ZY and Zhang JL: Matrine
increases the inhibitory effects of afatinib on H1975 cells via the
IL-6/JAK1/STAT3 signaling pathway. Mol Med Rep. 16:2733–2739. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Yuquan B, Hexiao T, Laiyi W, Gaofeng P,
Xuefeng Z, Ming X, Yanhong W, Li Z and Jinping Z: Interaction
between epidermal growth factor receptor and interleukin-6 receptor
in NSCLC progression. J Cell Biochem. 120:872–881. 2019. View Article : Google Scholar
|
|
51
|
Wang J, Wang Y, Zheng C, Hou K, Zhang T,
Qu X, Liu Y, Kang J, Hu X and Che X: Tyrosine kinase
inhibitor-induced IL-6/STAT3 activation decreases sensitivity of
EGFR-mutant non-small cell lung cancer to icotinib. Cell Biol Int.
42:1292–1299. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Liu C, Yang L, Xu H, Zheng S, Wang Z, Wang
S, Yang Y, Zhang S, Feng X, Sun N, et al: Systematic analysis of
IL-6 as a predictive biomarker and desensitizer of immunotherapy
responses in patients with non-small cell lung cancer. BMC Med.
20:1872022. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Looyenga BD, Hutchings D, Cherni I,
Kingsley C, Weiss GJ and Mackeigan JP: STAT3 is activated by JAK2
independent of key oncogenic driver mutations in non-small cell
lung carcinoma. PLoS One. 7:e308202012. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Pine SR, Mechanic LE, Enewold L,
Chaturvedi AK, Katki HA, Zheng YL, Bowman ED, Engels EA, Caporaso
NE and Harris CC: Increased levels of circulating interleukin 6,
interleukin 8, C-reactive protein, and risk of lung cancer. J Natl
Cancer Inst. 103:1112–1122. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Silva EM, Mariano VS, Pastrez PRA, Pinto
MC, Castro AG, Syrjanen KJ and Longatto-Filho A: High systemic IL-6
is associated with worse prognosis in patients with non-small cell
lung cancer. PLoS One. 12:e01811252017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Jones SA and Jenkins BJ: Recent insights
into targeting the IL-6 cytokine family in inflammatory diseases
and cancer. Nat Rev Immunol. 18:773–789. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Islas-vazquez L, Prado-Garcia H,
Aguilar-Cazares D, Meneses-Flores M, Galicia-velasco M,
Romero-Garcia S, Camacho-Mendoza C and Lopez-Gonzalez JS: LAP
TGF-Beta Subset of CD4(+)CD25(+)CD127(-) Treg cells is increased
and overexpresses LAP TGF-Beta in lung adenocarcinoma patients.
Biomed Res Int. 2015:4309432015. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Brooks GD, McLeod L, Alhayyani S, Miller
A, Russell PA, Ferlin W, Rose-John S, Ruwanpura S and Jenkins BJ:
IL6 Trans-signaling promotes KRAS-driven lung carcinogenesis.
Cancer Res. 76:866–876. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Ujiie H, Tomida M, Akiyama H, Nakajima Y,
Okada D, Yoshino N, Takiguchi Y and Tanzawa H: Serum hepatocyte
growth factor and interleukin-6 are effective prognostic markers
for non-small cell lung cancer. Anticancer Res. 32:3251–3258.
2012.PubMed/NCBI
|
|
60
|
Liao C, Yu Z, Guo W, Liu Q, Wu Y, Li Y and
Bai L: Prognostic value of circulating inflammatory factors in
non-small cell lung cancer: A systematic review and meta-analysis.
Cancer Biomark. 14:469–481. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Sheng J, Fang W, Liu X, Xing S, Zhan J, Ma
Y, Huang Y, Zhou N, Zhao H and Zhang L: Impact of gefitinib in
early stage treatment on circulating cytokines and lymphocytes for
patients with advanced non-small cell lung cancer. Onco Targets
Ther. 10:1101–1110. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Jia Y, Li X, Zhao C, Jiang T, Zhao S,
Zhang L, Liu X, Shi J, Qiao M, Luo J, et al: Impact of serum
vascular endothelial growth factor and interleukin-6 on treatment
response to epidermal growth factor receptor tyrosine kinase
inhibitors in patients with non-small-cell lung cancer. Lung
Cancer. 125:22–28. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Komi DEA and Redegeld FA: Role of mast
cells in shaping the tumor microenvironment. Clin Rev Allergy
Immunol. 58:313–325. 2020. View Article : Google Scholar :
|
|
64
|
Nilsson MB, Sun H, Diao L, Tong P, Liu D,
Li L, Fan Y, Poteete A, Lim SO, Howells K, et al: Stress hormones
promote EGFR inhibitor resistance in NSCLC: Implications for
combinations with β-blockers. Sci Transl Med. 9:eaao43072017.
View Article : Google Scholar
|
|
65
|
Jia Y, Li X, Zhao C, Jiang T, Zhao S,
Zhang L, Liu X, Shi J, Qiao M, Luo J, et al: Impact of serum
vascular endothelial growth factor and interleukin-6 on treatment
response to epidermal growth factor receptor tyrosine kinase
inhibitors in patients with non-small-cell lung cancer. Lung
Cancer. 125:22–28. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Oh YT, Chen Z, Wang D, Ramalingam SS and
Sun SY: Induction of IL6/STAT3-dependent TRAIL expression that
contributes to the therapeutic efficacy of osimertinib in EGFR
mutant NSCLC cells. Oncogene. 44:2315–2327. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Kim SM, Kwon OJ, Hong YK, Kim JH, Solca F,
Ha SJ, Soo RA, Christensen JG, Lee JH and Cho BC: Activation of
IL-6R/JAK1/STAT3 signaling induces de novo resistance to
irreversible EGFR inhibitors in non-small cell lung cancer with
T790M resistance mutation. Mol Cancer Ther. 11:2254–2264. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Yao Z, Fenoglio S, Gao DC, Camiolo M,
Stiles B, Lindsted T, Schlederer M, Johns C, Altorki N, Mittal V,
et al: TGF-beta IL-6 axis mediates selective and adaptive
mechanisms of resistance to molecular targeted therapy in lung
cancer. Proc Natl Acad Sci USA. 107:15535–15540. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Liang Q, Gong M, Zou JH, Luo MY, Jiang LL,
Wang C, Shen NX, Zhang MC, Xu L, Lei HM, et al: A phosphoglycerate
mutase 1 allosteric inhibitor overcomes drug resistance to
EGFR-targeted therapy via disrupting IL-6/JAK2/STAT3 signaling
pathway in lung adenocarcinoma. Drug Resist Updat. 68:1009572023.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Lee HJ, Zhuang G, Cao Y, Du P, Kim HJ and
Settleman J: Drug resistance via feedback activation of Stat3 in
oncogene-addicted cancer cells. Cancer Cell. 26:207–221. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Li L, Han R, Xiao H, Lin C, Wang Y, Liu H,
Li K, Chen H, Sun F, Yang Z, et al: Metformin sensitizes
EGFR-TKI-resistant human lung cancer cells in vitro and in vivo
through inhibition of IL-6 signaling and EMT reversal. Clin Cancer
Res. 20:2714–2726. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Zheng X, Lu G, Yao Y and Gu W: An
Autocrine IL-6/IGF-1R loop mediates EMT and promotes tumor growth
in non-small cell lung cancer. Int J Biol Sci. 15:1882–1891. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Shi Q, Godschalk RWL and van Schooten FJ:
Inflammation and the chemical carcinogen benzo[a]pyrene: Partners
in crime. Mutat Res Rev Mutat Res. 774:12–24. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Klammer H, Mladenov E, Li F and Iliakis G:
Bystander effects as manifestation of intercellular communication
of DNA damage and of the cellular oxidative status. Cancer Lett.
356:58–71. 2015. View Article : Google Scholar
|
|
75
|
Maynard A, McCoach CE, Rotow JK, Harris L,
Haderk F, Kerr DL, Yu EA, Schenk EL, Tan W, Zee A, et al:
Therapy-induced evolution of human lung cancer revealed by
single-cell RNA sequencing. Cell. 182:1232–1251.e22. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Isomoto K, Haratani K, Hayashi H, Shimizu
S, Tomida S, Niwa T, Yokoyama T, Fukuda Y, Chiba Y, Kato R, et al:
Impact of EGFR-TKI treatment on the tumor immune microenvironment
in EGFR mutation-positive non-small cell lung cancer. Clin Cancer
Res. 26:2037–2046. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Tsukamoto H, Nishikata R, Senju S and
Nishimura Y: Myeloid-derived suppressor cells attenuate TH1
development through IL-6 production to promote tumor progression.
Cancer Immunol Res. 1:64–76. 2013. View Article : Google Scholar
|
|
78
|
Tsukamoto H, Fujieda K, Senju S, Ikeda T,
Oshiumi H and Nishimura Y: Immune-suppressive effects of
interleukin-6 on T-cell-mediated anti-tumor immunity. Cancer Sci.
109:523–530. 2018. View Article : Google Scholar :
|
|
79
|
Ohno Y, Kitamura H, Takahashi N, Ohtake J,
Kaneumi S, Sumida K, Homma S, Kawamura H, Minagawa N, Shibasaki S
and Taketomi A: IL-6 down-regulates HLA class II expression and
IL-12 production of human dendritic cells to impair activation of
antigen-specific CD4(+) T cells. Cancer Immunol Immunother.
65:193–204. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Huseni MA, Wang L, Klementowicz JE, Yuen
K, Breart B, Orr C, Liu LF, Li Y, Gupta V, Li C, et al: CD8+ T
cell-intrinsic IL-6 signaling promotes resistance to anti-PD-L1
immunotherapy. Cell Rep Med. 4:1008782023. View Article : Google Scholar
|
|
81
|
Patel SA, Nilsson MB, Yang Y, Le X, Tran
HT, Elamin YY, Yu X, Zhang F, Poteete A, Ren X, et al: IL6 mediates
suppression of T- and NK-cell function in EMT-associated
TKI-resistant EGFR-mutant NSCLC. Clin Cancer Res. 29:1292–1304.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Kuo IY, Yang YE, Yang PS, Tsai YJ, Tzeng
HT, Cheng HC, Kuo WT, Su WC, Chang CP and Wang YC: Converged
Rab37/IL-6 trafficking and STAT3/PD-1 transcription axes elicit an
immunosuppressive lung tumor microenvironment. Theranostics.
11:7029–7044. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Zhang N, Zeng Y, Du W, Zhu J, Shen D, Liu
Z and Huang JA: The EGFR pathway is involved in the regulation of
PD-L1 expression via the IL-6/JAK/STAT3 signaling pathway in
EGFR-mutated non-small cell lung cancer. Int J Oncol. 49:1360–1368.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Kang M, Park C, Kim SH, Yoon SW, Suh KJ,
Kim YJ, Ock CY, Kim M, Keam B, Kim TM, et al: Programmed
death-ligand 1 expression level as a predictor of EGFR tyrosine
kinase inhibitor efficacy in lung adenocarcinoma. Transl Lung
Cancer Res. 10:699–711. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Li J, Xu J, Yan X, Jin K, Li W and Zhang
R: Targeting interleukin-6 (IL-6) sensitizes Anti-PD-L1 treatment
in a colorectal cancer preclinical model. Med Sci Monit.
24:5501–5508. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Fan K, Yang C, Fan Z, Huang Q, Zhang Y,
Cheng H, Jin K, Lu Y, Wang Z, Luo G, et al: MUC16 C
terminal-induced secretion of tumor-derived IL-6 contributes to
tumor-associated treg enrichment in pancreatic cancer. Cancer Lett.
418:167–175. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Hu G, Cheng P, Pan J, Wang S, Ding Q,
Jiang Z, Cheng L, Shao X, Huang L and Huang J: An IL6-adenosine
positive feedback loop between CD73+ γδTregs and CAFs promotes
tumor progression in human breast cancer. Cancer Immunol Res.
8:1273–1286. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Salazar Y, Zheng X, Brunn D, Raifer H,
Picard F, Zhang Y, Winter H, Guenther S, Weigert A, Weigmann B, et
al: Microenvironmental Th9 and Th17 lymphocytes induce metastatic
spreading in lung cancer. J Clin Invest. 130:3560–3575. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Kim HY, Jhun JY, Cho ML, Choi JY, Byun JK,
Kim EK, Yoon SK, Bae SH, Chung BH and Yang CW: Interleukin-6
upregulates Th17 response via mTOR/STAT3 pathway in
acute-on-chronic hepatitis B liver failure. J Gastroenterol.
49:1264–1273. 2014. View Article : Google Scholar
|
|
90
|
Zheng Y, Sun L, Jiang T, Zhang D, He D and
Nie H: TNFα promotes Th17 cell differentiation through IL-6 and
IL-1β produced by monocytes in rheumatoid arthritis. J Immunol Res.
2014:3853522014. View Article : Google Scholar
|
|
91
|
Tsukamoto H, Fujieda K, Hirayama M, Ikeda
T, Yuno A, Matsumura K, Fukuma D, Araki K, Mizuta H, Nakayama H, et
al: Soluble IL6R expressed by myeloid cells reduces tumor-specific
Th1 differentiation and drives tumor progression. Cancer Res.
77:2279–2291. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Wu J, Gao FX, Wang C, Qin M, Han F, Xu T,
Hu Z, Long Y, He XM, Deng X, et al: IL-6 and IL-8 secreted by
tumour cells impair the function of NK cells via the STAT3 pathway
in oesophageal squamous cell carcinoma. J Exp Clin Cancer Res.
38:3212019. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Gotthardt D, Putz EM, Straka E, Kudweis P,
Biaggio M, Poli V, Strobl B, Müller M and Sexl V: Loss of STAT3 in
murine NK cells enhances NK cell-dependent tumor surveillance.
Blood. 124:2370–2379. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Kalluri R and Zeisberg M: Fibroblasts in
cancer. Nat Rev Cancer. 6:392–401. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Karagiannis GS, Poutahidis T, Erdman SE,
Kirsch R, Riddell RH and Diamandis EP: Cancer-associated
fibroblasts drive the progression of metastasis through both
paracrine and mechanical pressure on cancer tissue. Mol Cancer Res.
10:1403–1418. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Meador CB and Hata AN: Acquired resistance
to targeted therapies in NSCLC: Updates and evolving insights.
Pharmacol Ther. 210:1075222020. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Mikubo M, Inoue Y, Liu G and Tsao MS:
Mechanism of drug tolerant persister cancer cells: The landscape
and clinical implication for therapy. J Thorac Oncol. 16:1798–1809.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Bullock E, Rozyczko A, Shabbir S, Tsoupi
I, Young AIJ, Travnickova J, Gómez-Cuadrado L, Mabruk Z, Carrasco
G, Morrow E, et al: Cancer-associated fibroblast driven paracrine
IL-6/STAT3 signaling promotes migration and dissemination in
invasive lobular carcinoma. Breast Cancer Res. 27:1212025.
View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Tan J, Zhu L, Shi J, Zhang J, Kuang J, Guo
Q, Zhu X, Chen Y, Zhou C and Gao X: Evaluation of drug resistance
for EGFR-TKIs in lung cancer via multicellular lung-on-a-chip. Eur
J Pharm Sci. 199:1068052024. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Ishiguro Y, Ishiguro H and Miyamoto H:
Epidermal growth factor receptor tyrosine kinase inhibition
up-regulates interleukin-6 in cancer cells and induces subsequent
development of interstitial pneumonia. Oncotarget. 4:550–559. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Ochi K, Suzawa K, Thu YM, Takatsu F,
Tsudaka S, Zhu Y, Nakata K, Takeda T, Shien K, Yamamoto H, et al:
Drug repositioning of tranilast to sensitize a cancer therapy by
targeting cancer-associated fibroblast. Cancer Sci. 113:3428–3436.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Shien K, Papadimitrakopoulou vA, Ruder D,
Behrens C, Shen L, Kalhor N, Song J, Lee JJ, Wang J, Tang X, et al:
JAK1/STAT3 activation through a proinflammatory cytokine pathway
leads to resistance to molecularly targeted therapy in non-small
cell lung cancer. Mol Cancer Ther. 16:2234–2245. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Mantovani A, Marchesi F, Malesci A, Laghi
L and Allavena P: Tumour-associated macrophages as treatment
targets in oncology. Nat Rev Clin Oncol. 14:399–416. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Xiang X, Wang J, Lu D and Xu X: Targeting
tumor-associated macrophages to synergize tumor immunotherapy.
Signal Transduct Target Ther. 6:752021. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Zhang B, Zhang Y, Zhao J, Wang Z, Wu T, Ou
W, Wang J, Yang B, Zhao Y, Rao Z and Gao J: M2-polarized
macrophages contribute to the decreased sensitivity of EGFR-TKIs
treatment in patients with advanced lung adenocarcinoma. Med Oncol.
31:1272014. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Zhou J, Qu Z, Sun F, Han L, Li L, Yan S,
Stabile LP, Chen LF, Siegfried JM and Xiao G: Myeloid STAT3
promotes lung tumorigenesis by transforming tumor
immunosurveillance into tumor-promoting inflammation. Cancer
Immunol Res. 5:257–268. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Hu Z, Sui Q, Jin X, Shan G, Huang Y, Yi Y,
Zeng D, Zhao M, Zhan C, Wang Q, et al: IL6-STAT3-C/EBPβ-IL6
positive feedback loop in tumor-associated macrophages promotes the
EMT and metastasis of lung adenocarcinoma. J Exp Clin Cancer Res.
43:632024. View Article : Google Scholar
|
|
108
|
Lin G, Lin L, Chen X, Chen L, Yang J, Chen
Y, Qian D, Zeng Y and Xu Y: PPAR-γ/NF-kB/AQP3 axis in M2 macrophage
orchestrates lung adenocarcinoma progression by upregulating IL-6.
Cell Death Dis. 15:5322024. View Article : Google Scholar
|
|
109
|
Chen S, Lu K, Hou Y, You Z, Shu C, Wei X,
Wu T, Shi N, Zhang G, Wu J, et al: YY1 complex in M2 macrophage
promotes prostate cancer progression by upregulating IL-6. J
Immunother Cancer. 11:e0060202023. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Che D, Zhang S, Jing Z, Shang L, Jin S,
Liu F, Shen J, Li Y, Hu J, Meng Q and Yu Y: Macrophages induce EMT
to promote invasion of lung cancer cells through the IL-6-mediated
COX-2/PGE2/β-catenin signalling pathway. Mol Immunol. 90:197–210.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Wu Y, Yi M, Niu M, Mei Q and Wu K:
Myeloid-derived suppressor cells: An emerging target for anticancer
immunotherapy. Mol Cancer. 21:1842022. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Tang J, Liu X, Gong Y, Zhu J, Huang M,
Ding Z, Yu M, Tie Y, Li Q and Wang Y: Epidermal growth factor
receptor tyrosine kinase inhibitors (EGFR-TKIs) impact on immune
microenvironment in non-small cell lung cancer (NSCLC). J Clin
Oncol. 36(15_Suppl): e211542018. View Article : Google Scholar
|
|
113
|
Yang Z, Guo J, Weng L, Tang W, Jin S and
Ma W: Myeloid-derived suppressor cells-new and exciting players in
lung cancer. J Hematol Oncol. 13:102020. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
vasquez-Dunddel D, Pan F, Zeng Q,
Gorbounov M, Albesiano E, Fu J, Blosser RL, Tam AJ, Bruno T, Zhang
H, et al: STAT3 regulates arginase-I in myeloid-derived suppressor
cells from cancer patients. J Clin Invest. 123:1580–1589. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Altorki NK, Markowitz GJ, Gao D, Port JL,
Saxena A, Stiles B, McGraw T and Mittal V: The lung
microenvironment: An important regulator of tumour growth and
metastasis. Nat Rev Cancer. 19:9–31. 2019. View Article : Google Scholar :
|
|
116
|
Neo SY, Tong L, Chong J, Liu Y, Jing X,
Oliveira MMS, Chen Y, Chen Z, Lee K, Burduli N, et al:
Tumor-associated NK cells drive MDSC-mediated tumor immune
tolerance through the IL-6/STAT3 axis. Sci Transl Med.
16:eadi29522024. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Gabrilovich DI and Nagaraj S:
Myeloid-derived suppressor cells as regulators of the immune
system. Nat Rev Immunol. 9:162–174. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Condamine T and Gabrilovich DI: Molecular
mechanisms regulating myeloid-derived suppressor cell
differentiation and function. Trends Immunol. 32:19–25. 2011.
View Article : Google Scholar
|
|
119
|
Weber R, Groth C, Lasser S, Arkhypov I,
Petrova V, Altevogt P, Utikal J and Umansky V: IL-6 as a major
regulator of MDSC activity and possible target for cancer
immunotherapy. Cell Immunol. 359:1042542021. View Article : Google Scholar
|
|
120
|
Jeong H, Koh J, Kim S, Yim J, Song SG, Kim
H, Li Y, Lee SH, Chung YK, Kim H, et al: Cell-intrinsic PD-L1
signaling drives immunosuppression by myeloid-derived suppressor
cells through IL-6/jak/Stat3 in PD-L1-high lung cancer. J
Immunother Cancer. 13:e0106122025. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Weber R, Riester Z, Hüser L, Sticht C,
Siebenmorgen A, Groth C, Hu X, Altevogt P, Utikal JS and Umansky V:
IL-6 regulates CCR5 expression and immunosuppressive capacity of
MDSC in murine melanoma. J Immunother Cancer. 8:e0009492020.
View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Song L, Rawal B, Nemeth JA and Haura EB:
JAK1 activates STAT3 activity in non-small-cell lung cancer cells
and IL-6 neutralizing antibodies can suppress JAK1-STAT3 signaling.
Mol Cancer Ther. 10:481–494. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Yang Y, Wang W, Chang H, Han Z, Yu X and
Zhang T: Reciprocal regulation of miR-206 and IL-6/STAT3 pathway
mediates IL6-induced gefitinib resistance in EGFR-mutant lung
cancer cells. J Cell Mol Med. 23:7331–7341. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Murakami T, Takigawa N, Ninomiya T, Ochi
N, Yasugi M, Honda Y, Kubo T, Ichihara E, Hotta K, Tanimoto M and
Kiura K: Effect of AZD1480 in an epidermal growth factor
receptor-driven lung cancer model. Lung Cancer. 83:30–36. 2014.
View Article : Google Scholar
|
|
125
|
Padda SK, Reckamp KL, Koczywas M, Neal JW,
Kawashima J, Kong S, Huang DB, Kowalski M and Wakelee HA: A phase
1b study of erlotinib and momelotinib for the treatment of
EGFR-mutated, tyrosine kinase inhibitor-naive metastatic non-small
cell lung cancer. Cancer Chemother Pharmacol. 89:105–115. 2022.
View Article : Google Scholar :
|
|
126
|
Hong D, Kurzrock R, Kim Y, Woessner R,
Younes A, Nemunaitis J, Fowler N, Zhou T, Schmidt J, Jo M, et al:
AZD9150, a next-generation antisense oligonucleotide inhibitor of
STAT3 with early evidence of clinical activity in lymphoma and lung
cancer. Sci Transl Med. 7:314ra1852015. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Nan J, Du Y, Chen X, Bai Q, Wang Y, Zhang
X, Zhu N, Zhang J, Hou J, Wang Q and Yang J: TPCA-1 is a direct
dual inhibitor of STAT3 and NF-κB and regresses mutant
EGFR-Associated human non-small cell lung cancers. Mol Cancer Ther.
13:617–629. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Chiu HC, Chou DL, Huang CT, Lin WH, Lien
TW, Yen KJ and Hsu JT: Suppression of Stat3 activity sensitizes
gefitinib-resistant non small cell lung cancer cells. Biochem
Pharmacol. 81:1263–1270. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Hedvat M, Huszar D, Herrmann A, Gozgit JM,
Schroeder A, Sheehy A, Buettner R, Proia D, Kowolik CM, Xin H, et
al: The JAK2 inhibitor AZD1480 potently blocks Stat3 signaling and
oncogenesis in solid tumors. Cancer Cell. 16:487–497. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Xin H, Herrmann A, Reckamp K, Zhang W, Pal
S, Hedvat M, Zhang C, Liang W, Scuto A, Weng S, et al:
Antiangiogenic and antimetastatic activity of JAK inhibitor
AZD1480. Cancer Res. 71:6601–6610. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Li R, Huang Y and Lin J: Distinct effects
of general anesthetics on lung metastasis mediated by
IL-6/JAK/STAT3 pathway in mouse models. Nat Commun. 11:6422020.
View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Cao W, Liu Y, Zhang R, Zhang B, Wang T,
Zhu X, Mei L, Chen H, Zhang H, Ming P and Huang L:
Homoharringtonine induces apoptosis and inhibits STAT3 via
IL-6/JAK1/STAT3 signal pathway in Gefitinib-resistant lung cancer
cells. Sci Rep. 5:84772015. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Lou W, Chen Y, Zhu KY, Deng H, Wu T and
Wang J: Polyphyllin I overcomes EMT-associated resistance to
erlotinib in lung cancer cells via IL-6/STAT3 pathway inhibition.
Biol Pharm Bull. 40:1306–1313. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Shi Y, Gao W, Lytle NK, Huang P, Yuan X,
Dann AM, Ridinger-Saison M, DelGiorno KE, Antal CE, Liang G, et al:
Targeting LIF-mediated paracrine interaction for pancreatic cancer
therapy and monitoring. Nature. 569:131–135. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Ernst M and Putoczki TL: Molecular
pathways: IL11 as a tumor-promoting cytokine-translational
implications for cancers. Clin Cancer Res. 20:5579–5588. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Kang S, Narazaki M, Metwally H and
Kishimoto T: Historical overview of the interleukin-6 family
cytokine. J Exp Med. 217:e201903472020. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Zhao K, Lu Y, Chen Y, Cheng J and Zhang W:
Dual inhibition of MAPK and JAK2/STAT3 pathways is critical for the
treatment of BRAF mutant melanoma. Mol Ther Oncolytics. 18:100–108.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Huang B, Lang X and Li X: The role of
IL-6/JAK2/STAT3 signaling pathway in cancers. Front Oncol.
12:10231772022. View Article : Google Scholar :
|
|
139
|
Reck M, Rodríguez-Abreu D, Robinson AG,
Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe
S, et al: Updated analysis of KEYNOTE-024: Pembrolizumab versus
platinum-based chemotherapy for advanced non-small-cell lung cancer
with PD-L1 tumor proportion score of 50% or greater. J Clin Oncol.
37:537–546. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Luo W, Wang Z, Zhang T, Yang L, Xian J, Li
Y and Li W: Immunotherapy in non-small cell lung cancer: Rationale,
recent advances and future perspectives. Precis Clin Med.
4:258–270. 2021. View Article : Google Scholar
|
|
141
|
Hayashi H, Sugawara S, Fukuda Y, Fujimoto
D, Miura S, Ota K, Ozawa Y, Hara S, Tanizaki J, Azuma K, et al: A
randomized phase II study comparing nivolumab with
carboplatin-pemetrexed for EGFR-mutated NSCLC with resistance to
EGFR tyrosine kinase inhibitors (WJOG8515L). Clin Cancer Res.
28:893–902. 2022. View Article : Google Scholar
|
|
142
|
Mok T, Nakagawa K, Park K, Ohe Y, Girard
N, Kim HR, Wu YL, Gainor J, Lee SH, Chiu CH, et al: Nivolumab plus
chemotherapy in epidermal growth factor receptor-mutated metastatic
non-small-cell lung cancer after disease progression on epidermal
growth factor receptor tyrosine kinase inhibitors: Final results of
CheckMate 722. J Clin Oncol. 42:1252–1264. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Yang JC, Lee DH, Lee JS, Fan Y, de Marinis
F, Iwama E, Inoue T, Rodríguez-Cid J, Zhang L, Yang CT, et al:
Phase III KEYNOTE-789 study of pemetrexed and platinum with or
without pembrolizumab for tyrosine kinase inhibitor-resistant,
EGFR-mutant, metastatic nonsquamous non-small cell lung cancer. J
Clin Oncol. 42:4029–4039. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Yu H, Kortylewski M and Pardoll D:
Crosstalk between cancer and immune cells: Role of STAT3 in the
tumour microenvironment. Nat Rev Immunol. 7:41–51. 2007. View Article : Google Scholar
|
|
145
|
Iwata-Kajihara T, Sumimoto H, Kawamura N,
Ueda R, Takahashi T, Mizuguchi H, Miyagishi M, Takeda K and
Kawakami Y: Enhanced cancer immunotherapy using STAT3-depleted
dendritic cells with high Th1-inducing ability and resistance to
cancer cell-derived inhibitory factors. J Immunol. 187:27–36. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Hossain DM, Dos Santos C, Zhang Q,
Kozlowska A, Liu H, Gao C, Moreira D, Swiderski P, Jozwiak A, Kline
J, et al: Leukemia cell-targeted STAT3 silencing and TLR9
triggering generate systemic antitumor immunity. Blood. 123:15–25.
2014. View Article : Google Scholar
|
|
147
|
Yu H, Pardoll D and Jove R: STATs in
cancer inflammation and immunity: A leading role for STAT3. Nat Rev
Cancer. 9:798–809. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Kortylewski M, Kujawski M, Wang T, Wei S,
Zhang S, Pilon-Thomas S, Niu G, Kay H, Mulé J, Kerr WG, et al:
Inhibiting Stat3 signaling in the hematopoietic system elicits
multicomponent antitumor immunity. Nat Med. 11:1314–1321. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Tan DSW, Felip E, de Castro G, Solomon BJ,
Greystoke A, Cho BC, Cobo M, Kim TM, Ganguly S, Carcereny E, et al:
Canakinumab versus placebo in combination with first-line
pembrolizumab plus chemotherapy for advanced non-small-cell lung
cancer: Results from the CANOPY-1 trial. J Clin Oncol. 42:192–204.
2024. View Article : Google Scholar
|
|
150
|
Zhang W, Liu Y, Yan Z, Yang H, Sun W, Yao
Y, Chen Y and Jiang R: IL-6 promotes PD-L1 expression in monocytes
and macrophages by decreasing protein tyrosine phosphatase receptor
type O expression in human hepatocellular carcinoma. J Immunother
Cancer. 8:e0002852020. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Zhou J, Lu X, Zhu H, Ding N, Zhang Y, Xu
X, Gao L, Zhou J, Song Y and Hu J: Resistance to immune checkpoint
inhibitors in advanced lung cancer: Clinical characteristics,
potential prognostic factors and next strategy. Front Immunol.
14:10890262023. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Tsukamoto H, Fujieda K, Miyashita A,
Fukushima S, Ikeda T, Kubo Y, Senju S, Ihn H, Nishimura Y and
Oshiumi H: Combined blockade of IL6 and PD-1/PD-L1 signaling
abrogates mutual regulation of their immunosuppressive effects in
the tumor microenvironment. Cancer Res. 78:5011–5022. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Mace TA, Shakya R, Pitarresi JR, Swanson
B, McQuinn CW, Loftus S, Nordquist E, Cruz-Monserrate Z, Yu L,
Young G, et al: IL-6 and PD-L1 antibody blockade combination
therapy reduces tumour progression in murine models of pancreatic
cancer. Gut. 67:320–332. 2018. View Article : Google Scholar
|
|
154
|
Jin W: Role of JAK/STAT3 signaling in the
regulation of metastasis, the transition of cancer stem cells, and
chemoresistance of cancer by epithelial-mesenchymal transition.
Cells. 9:2172020. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Danlos FX, Texier M, Job B, Mouraud S,
Cassard L, Baldini C, varga A, Yurchenko AA, Rabeau A, Champiat S,
et al: Genomic instability and protumoral inflammation are
associated with primary resistance to anti-PD-1 + antiangiogenesis
in malignant pleural mesothelioma. Cancer Discov. 13:858–879. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Yokota H, Sato K, Sakamoto S, Okuda Y,
Takeda M, Akamine Y, Nakayama K and Miura M: Influence of
interleukin-6 on the pharmacokinetics and pharmacodynamics of
osimertinib in patients with non-small cell lung cancer. Cancer
Chemother Pharmacol. 95:492025. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Martin FJ, Amode MR, Aneja A,
Austine-Orimoloye O, Azov AG, Barnes I, Becker A, Bennett R, Berry
A, Bhai J, et al: Ensembl 2023. Nucleic Acids Res. 51:D933–D941.
2023. View Article : Google Scholar :
|
|
158
|
Xie Y, Li H, Luo X, Li H, Gao Q, Zhang L,
Teng Y, Zhao Q, Zuo Z and Ren J: IBS 2.0: An upgraded illustrator
for the visualization of biological sequences. Nucleic Acids Res.
50(W1): W420–W426. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Fang Z, Ran H, Zhang Y, Chen C, Lin P,
Zhang X and Wu M: AlphaFold 3: An unprecedent opportunity for
fundamental research and drug development. Precis Clin Med.
8:pbaf0152025. View Article : Google Scholar : PubMed/NCBI
|