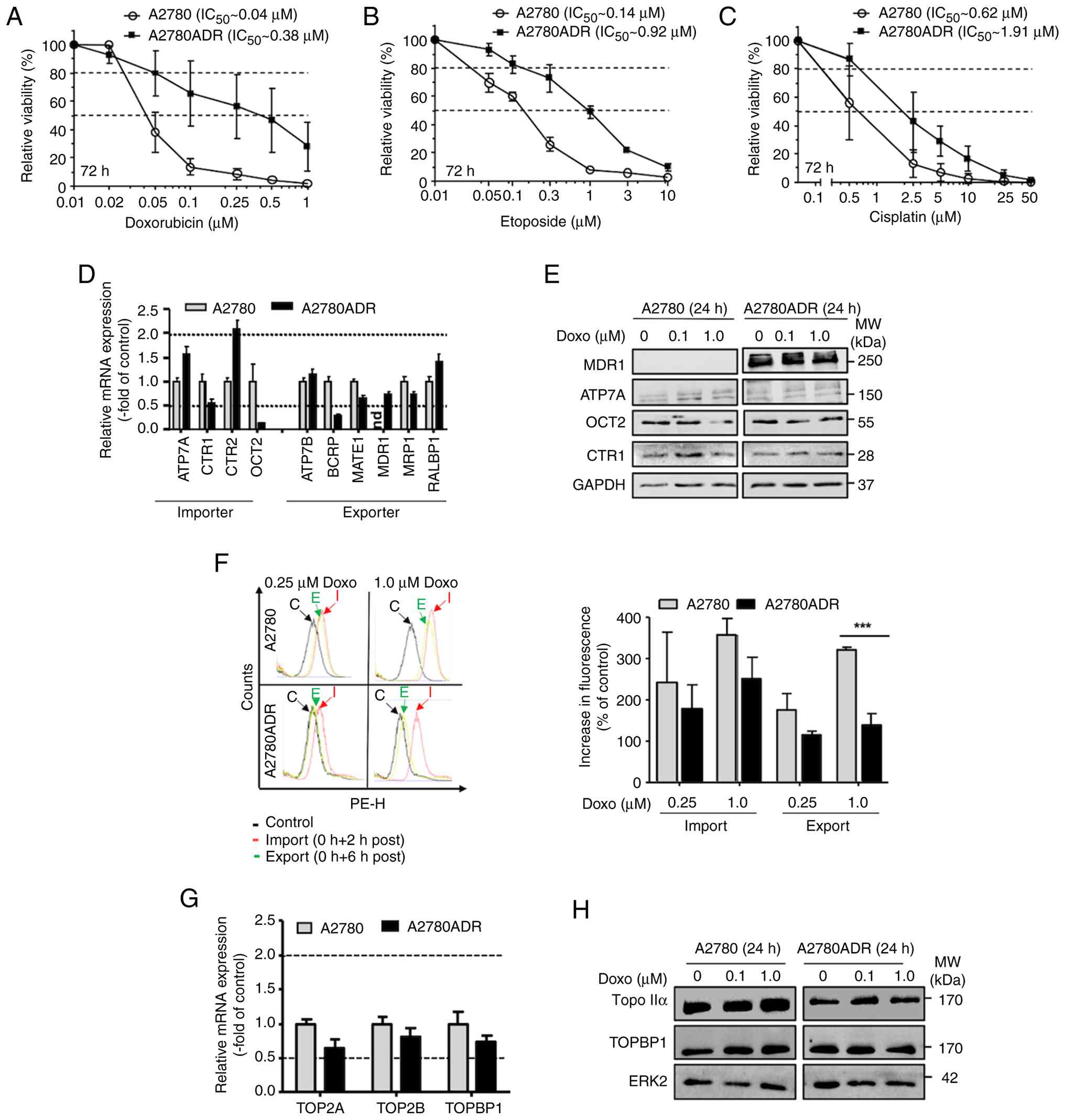
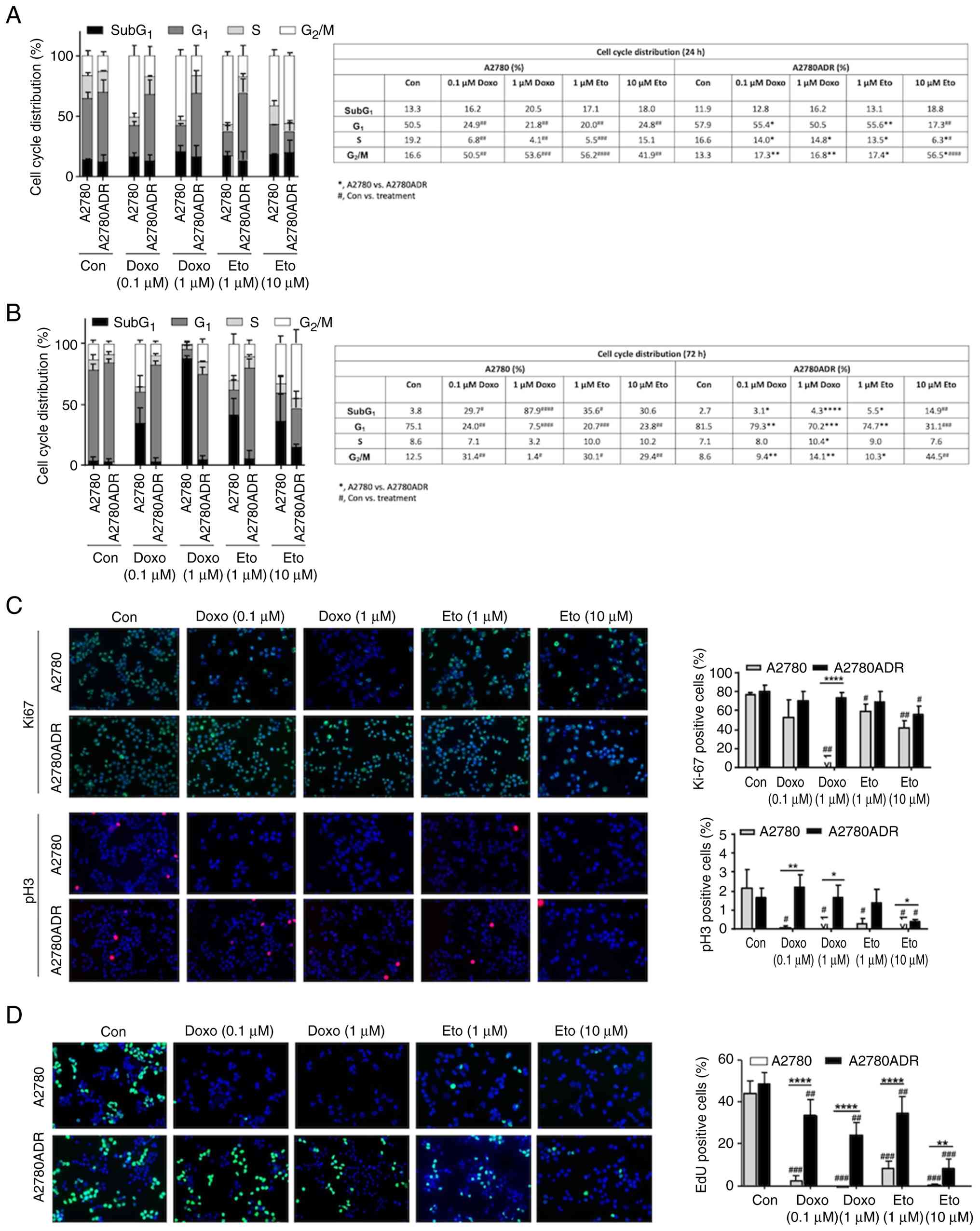
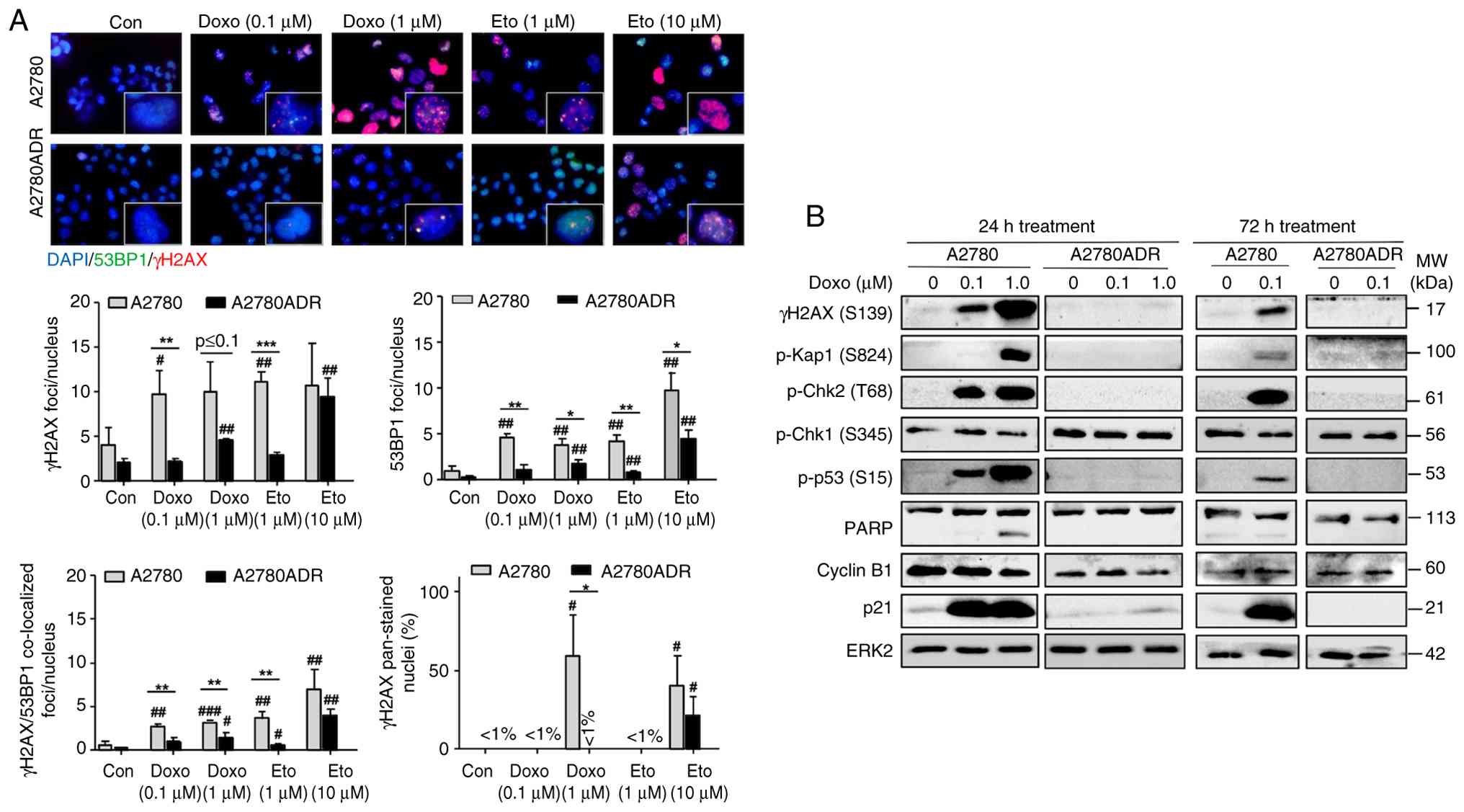
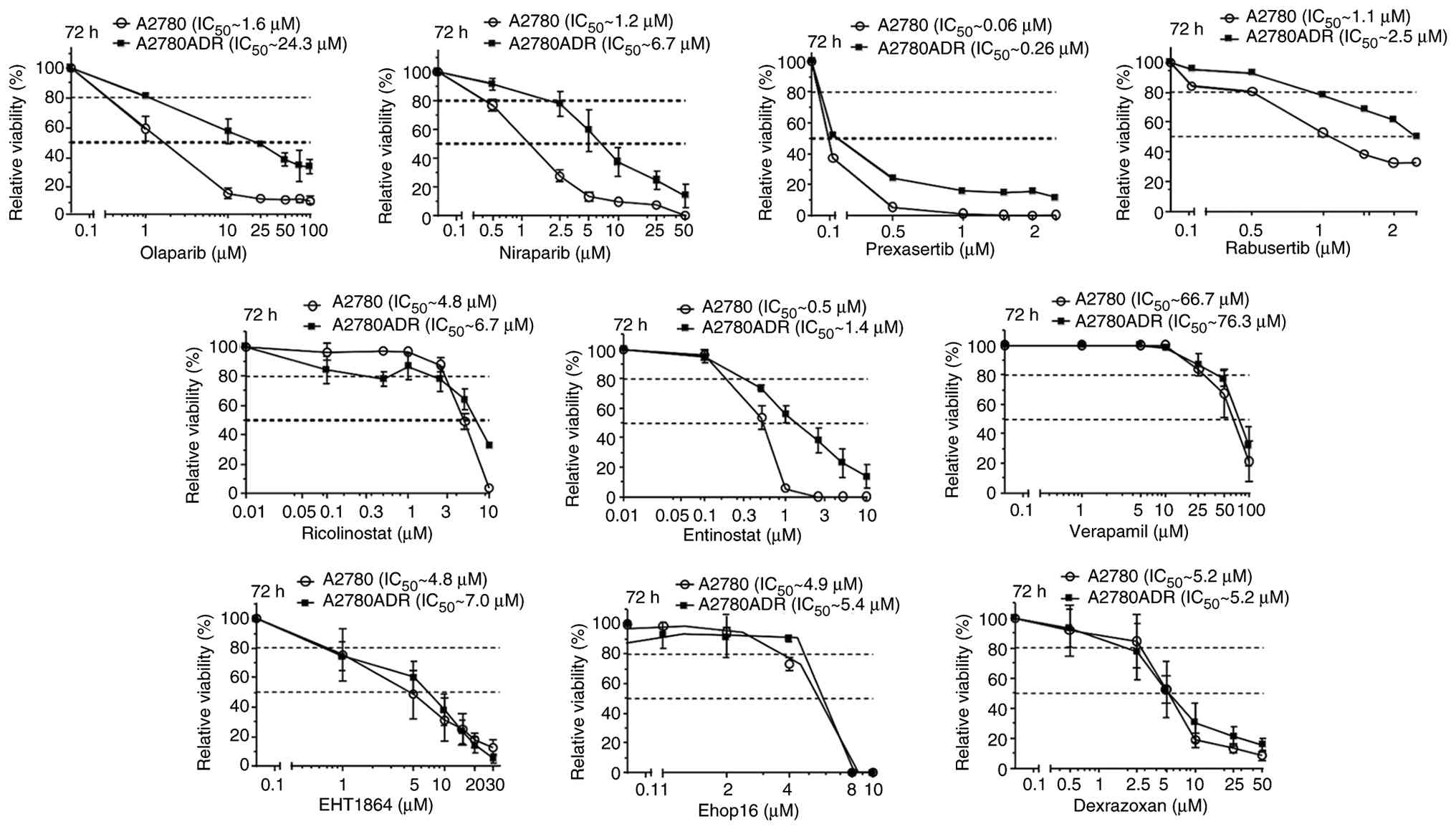
|
1
|
Minotti G, Menna P, Salvatorelli E, Cairo
G and Gianni L: Anthracyclines: Molecular advances and
pharmacologic developments in antitumor activity and
cardiotoxicity. Pharmacol Rev. 56:185–229. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Roos WP and Kaina B: DNA damage-induced
cell death: From specific DNA lesions to the DNA damage response
and apoptosis. Cancer Lett. 332:237–248. 2013. View Article : Google Scholar
|
|
3
|
Roos WP, Thomas AD and Kaina B: DNA damage
and the balance between survival and death in cancer biology. Nat
Rev Cancer. 16:20–33. 2016. View Article : Google Scholar
|
|
4
|
Rivankar S: An overview of doxorubicin
formulations in cancer therapy. J Cancer Res Ther. 10:853–858.
2014. View Article : Google Scholar
|
|
5
|
Galluzzi L, Senovilla L, Vitale I, Michels
J, Martins I, Kepp O, Castedo M and Kroemer G: Molecular mechanisms
of cisplatin resistance. Oncogene. 31:1869–1883. 2012. View Article : Google Scholar
|
|
6
|
Nielsen D, Maare C and Skovsgaard T:
Cellular resistance to anthracyclines. Gen Pharmacol. 27:251–255.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gillet JP, Efferth T and Remacle J:
Chemotherapy-induced resistance by ATP-binding cassette transporter
genes. Biochim Biophys Acta. 1775:237–262. 2007.PubMed/NCBI
|
|
8
|
Konkimalla VB, Kaina B and Efferth T: Role
of transporter genes in cisplatin resistance. In Vivo. 22:279–283.
2008.PubMed/NCBI
|
|
9
|
Woods D and Turchi JJ: Chemotherapy
induced DNA damage response: Convergence of drugs and pathways.
Cancer Biol Ther. 14:379–389. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lord CJ and Ashworth A: The DNA damage
response and cancer therapy. Nature. 481:287–294. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
O'Connor MJ: Targeting the DNA damage
response in cancer. Mol Cell. 60:547–560. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ferri A, Stagni V and Barila D: Targeting
the DNA damage response to overcome cancer drug resistance in
glioblastoma. Int J Mol Sci. 21:49102020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Harper JW and Elledge SJ: The DNA damage
response: Ten years after. Mol Cell. 28:739–745. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shiloh Y: ATM and ATR: Networking cellular
responses to DNA damage. Curr Opin Genet Dev. 11:71–77. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Matsuoka S, Ballif BA, Smogorzewska A,
McDonald ER III, Hurov KE, Luo J, Bakalarski CE, Zhao Z, Solimini
N, Lerenthal Y, et al: ATM and ATR substrate analysis reveals
extensive protein networks responsive to DNA damage. Science.
316:1160–1166. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Durocher D and Jackson SP: DNA-PK, ATM and
ATR as sensors of DNA damage: Variations on a theme? Curr Opin Cell
Biol. 13:225–231. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kumagai A and Dunphy WG: How cells
activate ATR. Cell Cycle. 5:1265–1268. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Petermann E and Caldecott KW: Evidence
that the ATR/Chk1 pathway maintains normal replication fork
progression during unperturbed S phase. Cell Cycle. 5:2203–2209.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Eich M, Roos WP, Nikolova T and Kaina B:
Contribution of ATM and ATR to the resistance of glioblastoma and
malignant melanoma cells to the methylating anticancer drug
temozolomide. Mol Cancer Ther. 12:2529–2540. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Weber AM and Ryan AJ: ATM and ATR as
therapeutic targets in cancer. Pharmacol Ther. 149:124–138. 2015.
View Article : Google Scholar
|
|
21
|
Carrassa L and Damia G: DNA damage
response inhibitors: Mechanisms and potential applications in
cancer therapy. Cancer Treat Rev. 60:139–151. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dobbelstein M and Sorensen CS: Exploiting
replicative stress to treat cancer. Nat Rev Drug Discov.
14:405–423. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Morgan MA and Lawrence TS: Molecular
pathways: Overcoming radiation resistance by targeting DNA damage
response pathways. Clin Cancer Res. 21:2898–2904. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Saini P, Li Y and Dobbelstein M: Wee1 is
required to sustain ATR/Chk1 signaling upon replicative stress.
Oncotarget. 6:13072–13087. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang Y, Lai J, Du Z, Gao J, Yang S,
Gorityala S, Xiong X, Deng O, Ma Z, Yan C, et al: Targeting
radioresistant breast cancer cells by single agent CHK1 inhibitor
via enhancing replication stress. Oncotarget. 7:34688–34702. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Toledo LI, Murga M and Fernandez-Capetillo
O: Targeting ATR and Chk1 kinases for cancer treatment: A new model
for new (and old) drugs. Mol Oncol. 5:368–373. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Murga M, Campaner S, Lopez-Contreras AJ,
Toledo LI, Soria R, Montaña MF, Artista L, Schleker T, Guerra C,
Garcia E, et al: Exploiting oncogene-induced replicative stress for
the selective killing of Myc-driven tumors. Nat Struct Mol Biol.
18:1331–1335. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gilad O, Nabet BY, Ragland RL, Schoppy DW,
Smith KD, Durham AC and Brown EJ: Combining ATR suppression with
oncogenic Ras synergistically increases genomic instability,
causing synthetic lethality or tumorigenesis in a dosage-dependent
manner. Cancer Res. 70:9693–9702. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Helleday T: The underlying mechanism for
the PARP and BRCA synthetic lethality: Clearing up the
misunderstandings. Mol Oncol. 5:387–393. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lord CJ and Ashworth A: PARP inhibitors:
Synthetic lethality in the clinic. Science. 355:1152–1158. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lord CJ, Tutt ANJ and Ashworth A:
Synthetic lethality and cancer therapy: Lessons learned from the
development of PARP inhibitors. Ann Rev Med. 66:455–470. 2015.
View Article : Google Scholar
|
|
32
|
Farmer H, McCabe N, Lord CJ, Tutt AN,
Johnson DA, Richardson TB, Santarosa M, Dillon KJ, Hickson I,
Knights C, et al: Targeting the DNA repair defect in BRCA mutant
cells as a therapeutic strategy. Nature. 434:917–921. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sriraman SK, Pan J, Sarisozen C, Luther E
and Torchilin V: Enhanced cytotoxicity of folic Acid-targeted
liposomes Co-loaded with C6 ceramide and doxorubicin: In vitro
evaluation on HeLa, A2780-ADR, and H69-AR cells. Mol Pharm.
13:428–437. 2016. View Article : Google Scholar
|
|
34
|
Claycomb WC, Lanson NA Jr, Stallworth BS,
Egeland DB, Delcarpio JB, Bahinski A and Izzo NJ Jr: HL-1 cells: A
cardiac muscle cell line that contracts and retains phenotypic
characteristics of the adult cardiomyocyte. Proc Natl Acad Sci USA.
95:2979–2984. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Nichols J, Evans EP and Smith AG:
Establishment of germ-line-competent embryonic stem (ES) cells
using differentiation inhibiting activity. Development.
110:1341–1348. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang Y and Adjaye J: A cyclic AMP analog,
8-Br-cAMP, enhances the induction of pluripotency in human
fibroblast cells. Stem Cell Rev Rep. 7:331–341. 2011. View Article : Google Scholar
|
|
37
|
O'Brien J, Wilson I, Orton T and Pognan F:
Investigation of the Alamar Blue (resazurin) fluorescent dye for
the assessment of mammalian cell cytotoxicity. Eur J Biochem.
267:5421–5426. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chou TC: Comparison of drug combinations
in vitro, in animals, and in clinics by using the combination index
method via computer simulation. Cancer Res. 692009.
|
|
39
|
Luk CK and Tannock IF: Flow cytometric
analysis of doxorubicin accumulation in cells from human and rodent
cell lines. J Natl Cancer Inst. 81:55–59. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Olive PL: Detection of DNA damage in
individual cells by analysis of histone H2AX phosphorylation.
Methods Cell Biol. 75:355–373. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Rogakou EP, Pilch DR, Orr AH, Ivanova VS
and Bonner WM: DNA double-stranded breaks induce histone H2AX
phosphorylation on serine 139. J Biol Chem. 273:5858–5868. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zimmermann M and de Lange T: 53BP1: Pro
choice in DNA repair. Trends Cell Biol. 24:108–117. 2014.
View Article : Google Scholar
|
|
43
|
Panier S and Boulton SJ: Double-strand
break repair: 53BP1 comes into focus. Nat Rev Mol Cell Biol.
15:7–18. 2014. View Article : Google Scholar
|
|
44
|
Vandesompele J, De Preter K, Pattyn F,
Poppe B, Van Roy N, De Paepe A and Speleman F: Accurate
normalization of real-time quantitative RT-PCR data by geometric
averaging of multiple internal control genes. Genome Biol.
3:RESEARCH00342002. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Xia CQ and Smith PG: Drug efflux
transporters and multidrug resistance in acute leukemia:
Therapeutic impact and novel approaches to mediation. Mol
Pharmacol. 82:1008–1021. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Mirzaei S, Gholami MH, Hashemi F, Zabolian
A, Farahani MV, Hushmandi K, Zarrabi A, Goldman A, Ashrafizadeh M
and Orive G: Advances in understanding the role of P-gp in
doxorubicin resistance: Molecular pathways, therapeutic strategies,
and prospects. Drug Discov Today. 27:436–455. 2022. View Article : Google Scholar
|
|
47
|
Tewey KM, Rowe TC, Yang L, Halligan BD and
Liu LF: Adriamycin-induced DNA damage mediated by mammalian DNA
topoisomerase II. Science. 226:466–468. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Johnson M and Keyes D: Anthracycline
Toxicity. Disclosure: Daniel Keyes declares no relevant financial
relationships with ineligible companies. StatPearls; Treasure
Island, FL: 2024
|
|
49
|
Jin MH and Oh DY: ATM in DNA repair in
cancer. Pharmacol Ther. 203:1073912019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Garzon-Hernandez C, Ramirez-Merino N and
Soberon MCM: Molecular targeted therapy in oncology focusing on DNA
repair mechanisms. Arch Med Res. 53:807–817. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wang R, Sun Y, Li C, Xue Y and Ba X:
Targeting the DNA damage response for cancer therapy. Int J Mol
Sci. 24:159072023. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Basu B, Yap TA, Molife LR and de Bono JS:
Targeting the DNA damage response in oncology: Past, present and
future perspectives. Curr Opin Oncol. 24:316–324. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Yusa K and Tsuruo T: Reversal mechanism of
multidrug resistance by verapamil: Direct binding of verapamil to
P-glycoprotein on specific sites and transport of verapamil outward
across the plasma membrane of K562/ADM cells. Cancer Res.
49:5002–5006. 1989.PubMed/NCBI
|
|
54
|
Yan T, Deng S, Metzger A, Godtel-Armbrust
U, Porter AC and Wojnowski L: Topoisomerase II{alpha}-dependent and
-independent apoptotic effects of dexrazoxane and doxorubicin. Mol
Cancer Ther. 8:1075–1085. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Jirkovský E, Jirkovská A,
Bavlovič-Piskáčková H, Skalická V, Pokorná Z, Karabanovich G,
Kollárová-Brázdová P, Kubeš J, Lenčová-Popelová O, Mazurová Y, et
al: Clinically translatable prevention of anthracycline
cardiotoxicity by dexrazoxane is mediated by topoisomerase II beta
and not metal chelation. Circ Heart Fail. 14:e0082092021.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Jirkovska A, Karabanovich G, Kubes J,
Skalická V, Melnikova I, Korábečný J, Kučera T, Jirkovský E,
Nováková L, Bavlovič Piskáčková H, et al: Structure-activity
relationship study of dexrazoxane analogues reveals ICRF-193 as the
most potent bisdioxopiperazine against anthracycline toxicity to
cardiomyocytes due to its strong topoisomerase IIβ interactions. J
Med Chem. 64:3997–4019. 2021. View Article : Google Scholar
|
|
57
|
Huelsenbeck SC, Schorr A, Roos WP,
Huelsenbeck J, Henninger C, Kaina B and Fritz G: Rac1 protein
signaling is required for DNA damage response stimulated by
topoisomerase II poisons. J Biol Chem. 287:38590–38599. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Wartlick F, Bopp A, Henninger C and Fritz
G: DNA damage response (DDR) induced by topoisomerase II poisons
requires nuclear function of the small GTPase Rac. Biochim Biophys
Acta. 1833:3093–3103. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Skvortsov S, Dudas J, Eichberger P,
Witsch-Baumgartner M, Loeffler-Ragg J, Pritz C, Schartinger VH,
Maier H, Hall J, Debbage P, et al: Rac1 as a potential therapeutic
target for chemo-radioresistant head and neck squamous cell
carcinomas (HNSCC). Br J Cancer. 110:2677–2687. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Li Q, Qin T, Bi Z, Hong H, Ding L, Chen J,
Wu W, Lin X, Fu W, Zheng F, et al: Rac1 activates non-oxidative
pentose phosphate pathway to induce chemoresistance of breast
cancer. Nat Commun. 11:14562020. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Kraft FB, Biermann L, Schäker-Hübner L,
Hanl M, Hamacher A, Kassack MU and Hansen FK: Hydrazide-based class
I selective HDAC inhibitors completely reverse chemoresistance
synergistically in platinum-resistant solid cancer cells. J Med
Chem. 67:17796–17819. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Tang SW, Thomas A, Murai J, Trepel JB,
Bates SE, Rajapakse VN and Pommier Y: Overcoming resistance to
DNA-targeted agents by epigenetic activation of schlafen 11
(SLFN11) expression with class I histone deacetylase inhibitors.
Clin Cancer Res. 24:1944–1953. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Vollmer J, Ecker J, Hielscher T,
Valinciute G, Ridinger J, Jamaladdin N, Peterziel H, van Tilburg
CM, Oehme I, Witt O, et al: Class I HDAC inhibition reduces DNA
damage repair capacity of MYC-amplified medulloblastoma cells. J
Neurooncol. 164:617–632. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Bangert A, Hacker S, Cristofanon S,
Debatin KM and Fulda S: Chemosensitization of glioblastoma cells by
the histone deacetylase inhibitor MS275. Anticancer Drugs.
22:494–499. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Gianni L, Herman EH, Lipshultz SE, Minotti
G, Sarvazyan N and Sawyer DB: Anthracycline cardiotoxicity: From
bench to bedside. J Clin Oncol. 26:3777–3784. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Ferreira AL, Matsubara LS and Matsubara
BB: Anthracycline-induced cardiotoxicity. Cardiovasc Hematol Agents
Med Chem. 6:278–281. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Henninger C and Fritz G: Statins in
anthracycline-induced cardiotoxicity: Rac and Rho, and the
heartbreakers. Cell Death Dis. 8:e25642017. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Kucuk P, Abbey L, Schmitt J, Henninger C
and Fritz G: Cardiomyocytes, cardiac endothelial cells and
fibroblasts contribute to anthracycline-induced cardiac injury
through RAS-homologous small GTPases RAC1 and CDC42. Pharmacol Res.
203:1071652024. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Yoshida M, Shiojima I, Ikeda H and Komuro
I: Chronic doxorubicin cardiotoxicity is mediated by oxidative DNA
damage-ATM-p53-apoptosis pathway and attenuated by pitavastatin
through the inhibition of Rac1 activity. J Mol Cell Cardiol.
47:698–705. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Ma J, Wang Y, Zheng D, Wei M, Xu H and
Peng T: Rac1 signalling mediates doxorubicin-induced cardiotoxicity
through both reactive oxygen species-dependent and -independent
pathways. Cardiovasc Res. 97:77–87. 2013. View Article : Google Scholar
|
|
71
|
McGowan JV, Chung R, Maulik A, Piotrowska
I, Walker JM and Yellon DM: Anthracycline chemotherapy and
cardiotoxicity. Cardiovasc Drugs Ther. 31:63–75. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Gewirtz DA: A critical evaluation of the
mechanisms of action proposed for the antitumor effects of the
anthracycline antibiotics adriamycin and daunorubicin. Biochem
Pharmacol. 57:727–741. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Fry AM, Chresta CM, Davies SM, Walker MC,
Harris AL, Hartley JA, Masters JR and Hickson ID: Relationship
between topoisomerase II level and chemosensitivity in human tumor
cell lines. Cancer Res. 51:6592–6595. 1991.PubMed/NCBI
|
|
74
|
Sun X and Kaufman PD: Ki-67: More than a
proliferation marker. Chromosoma. 127:175–186. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Ledvin L, Gassaway BM, Tawil J, Urso O,
Pizzo D, Welsh KA, Bolhuis DL, Fisher D, Bonni A, Gygi SP, et al:
The anaphase-promoting complex controls a
ubiquitination-phosphoprotein axis in chromatin during
neurodevelopment. Dev Cell. 58:2666–2683.e9. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Uxa S, Castillo-Binder P, Kohler R,
Stangner K, Muller GA and Engeland K: Ki-67 gene expression. Cell
Death Differ. 28:3357–3370. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Lamberti G, Andrini E, Sisi M, Federico AD
and Ricciuti B: Targeting DNA damage response and repair genes to
enhance anticancer immunotherapy: Rationale and clinical
implication. Future Oncol. 16:1751–1766. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Friedrich A, Assmann AS, Schumacher L,
Stuijvenberg JV, Kassack MU, Schulz WA, Roos WP, Hansen FK,
Pflieger M, Kurz T and Fritz G: In vitro assessment of the
genotoxic hazard of novel hydroxamic acid- and benzamide-type
histone deacetylase inhibitors (HDACi). Int J Mol Sci. 21:47472020.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Sandrock K, Bielek H, Schradi K, Schmidt G
and Klugbauer N: The nuclear import of the small GTPase Rac1 is
mediated by the direct interaction with karyopherin alpha2.
Traffic. 11:198–209. 2010. View Article : Google Scholar
|
|
80
|
Kitzinger R, Fritz G and Henninger C:
Nuclear RAC1 is a modulator of the doxorubicin-induced DNA damage
response. Biochim Biophys Acta Mol Cell Res. 1869:1193202022.
View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Fritz G and Henninger C: Rho GTPases:
Novel players in the regulation of the DNA damage response?
Biomolecules. 5:2417–2434. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Thurn KT, Thomas S, Raha P, Qureshi I and
Munster PN: Histone deacetylase regulation of ATM-mediated DNA
damage signaling. Mol Cancer Ther. 12:2078–2087. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Roos WP and Krumm A: The multifaceted
influence of histone deacetylases on DNA damage signalling and DNA
repair. Nucleic Acids Res. 44:10017–10030. 2016.PubMed/NCBI
|
|
84
|
Kachhap SK, Rosmus N, Collis SJ,
Kortenhorst MS, Wissing MD, Hedayati M, Shabbeer S, Mendonca J,
Deangelis J, Marchionni L, et al: Downregulation of homologous
recombination DNA repair genes by HDAC inhibition in prostate
cancer is mediated through the E2F1 transcription factor. PLoS One.
5:e112082010. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Karwatsky J, Lincoln MC and Georges E: A
mechanism for P-glycoprotein-mediated apoptosis as revealed by
verapamil hypersensitivity. Biochemistry. 42:12163–12173. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Bellamy WT, Dorr RT, Dalton WS and Alberts
DS: Direct relation of DNA lesions in multidrug-resistant human
myeloma cells to intracellular doxorubicin concentration. Cancer
Res. 48:6360–6364. 1988.PubMed/NCBI
|
|
87
|
Bellamy WT, Dalton WS, Kailey JM, Gleason
MC, McCloskey TM, Dorr RT and Alberts DS: Verapamil reversal of
doxorubicin resistance in multidrug-resistant human myeloma cells
and association with drug accumulation and DNA damage. Cancer Res.
48:6365–6370. 1988.PubMed/NCBI
|
|
88
|
Huelsenbeck J, Henninger C, Schad A,
Lackner KJ, Kaina B and Fritz G: Inhibition of Rac1 signaling by
lovastatin protects against anthracycline-induced cardiac toxicity.
Cell Death Dis. 2:e1902011. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Ohlig J, Henninger C, Zander S, Merx M,
Kelm M and Fritz G: Rac1-mediated cardiac damage causes diastolic
dysfunction in a mouse model of subacute doxorubicin-induced
cardiotoxicity. Arch Toxicol. 92:441–453. 2018. View Article : Google Scholar
|