Introduction
Hepatocellular carcinoma (HCC) is one of the most
common causes of cancer-related mortality worldwide and accounts
for 5.7% of new cancer cases. This malignancy tends to occur in
livers damaged by chronic infection with hepatitis B or hepatitis C
viruses, alcohol abuse and excessive fat deposits in the context of
liver cirrhosis (1-3). In
patients with early-stage HCC, while surgical resection is the best
definitive treatment for patients with adaptation, percutaneous
local treatment (PLT) is the best treatment option for patients
with early-stage HCC who are not surgical candidates (4-6).
The two most commonly used forms of PLT are radiofrequency ablation
(RFA) and microwave ablation (MWA), which are heat-based thermal
ablation methods. Because of small size of the necrotic area
achieved with one microwave ablation treatment, RFA has been the
most frequently used method worldwide and has been shown to be a
safe and effective therapeutic option (7-9).
However, nodules adjacent to large vessels may often be
incompletely ablated due to the heat sink effect. Overall, 10-25%
of HCC patients may not be eligible for RFA (10). The major limitation of conventional
MWA systems is the lack of predictability of the ablation zone size
and shape. Therefore, a specific new-generation microwave ablation
(MWA) system, the Emprint Ablation System (Covidien), which uses an
internally water-cooled microwave antenna, was designed to create
large predictable spherical zones of ablation that are not impacted
by varying tissue environments (11,12). The
purpose of this study was to verify the treatment effectiveness and
safety of this new MWA system compared with the treatment results
obtained with the RFA system and to verify whether this device is
effective for the treatment of HCC.
Patients and methods
Patients
This retrospective study population consisted of 44
patients with 52 nodules treated with MWA between July 2017 and
February 2019 and 55 patients with 70 nodules treated with RFA
between January 2016 and October 2017. To evaluate the efficacy of
PLT with regard to local control of the lesions, we excluded
patients with extrahepatic metastases or portal tumor thrombosis
detected by various imaging modalities, those with a tendency
toward severe bleeding, and those whose liver function was
Child-Pugh Class C with refractory ascites. The treatment was
limited to a solitary nodular tumor located on one subsegment. The
size of each lesion was <3.0 cm in diameter. Tumors showing
extrahepatic extension and those located near the hilar region were
excluded. Informed consent for this procedure was obtained from the
patients and his or her family members. Among the 52 nodules
treated with MWA, 40 were treated with conventional transcatheter
arterial chemoembolization (c-TACE) using iodized oil emulsion (4-6
ml, Lipiodol, Andre Guerbet) before PLT, and of the 70 nodules
treated with RFA, 58 were treated with c-TACE before PLT to
evaluate the grade of cancer in detail and to visualize the tumor
area for easy assessment of the treated margin after treatment. If
the clinical diagnosis was difficult, the histological diagnosis of
tumors was confirmed by US-guided fine needle biopsy.
The follow-up period ended in February 2019.
Table I details the baseline
clinical characteristics of the MWA and RFA groups. In both
treatment groups, the proportions of men were higher, and the
underlying liver disease was most commonly HCV infection, followed
by non-B, non-C patients. Only prothrombin time was significantly
lower in the RFA group than in the MWA group, but there were very
few differences in the clinical data between the two groups.
Additionally, mean tumor diameters and dispersion of locations were
not different between the two groups.
 | Table IBaseline clinical data and patient
characteristics. |
Table I
Baseline clinical data and patient
characteristics.
| Variables | MWA | RFA | P-value |
|---|
| Patient (n) | 44 | 55 | |
| Nodule (n) | 52 | 70 | |
| Observation period
(days, mean) | 236±137 | 602±252 | |
| Sex
(male/female) | 30/14 | 44/11 | ns |
| Age | 73.4±7.7 | 73.2±8.8 | ns |
| Background
(B/C/NBNC) | 3/29/12 | 8/31/16 | ns |
| TACE (yes/no) | 40/12 | 58/12 | ns |
| Child-Pugh class
(A/B) | 37/7 | 48/10 | ns |
| Prothrombin time
(%) | 84.5±15.9 | 78.0±13.1 | 0.0238 |
| Albumin (g/dl) | 3.9±0.5 | 3.8±0.4 | ns |
| Total bilirubin
(mg/dl) | 1.0±0.5 | 0.8±0.3 | ns |
| Platelet
(104/µl) | 13.8±5.8 | 13.1±6.8 | ns |
| ALBI | -2.59±0.56 | -2.52±0.42 | ns |
| AFP (ng/ml) | 67.2±233.8 | 32.8±111.0 | ns |
| DCP (mAU/ml) | 86.6±124.1 | 121.0±276.6 | ns |
| Segment
(left/medial/anterior/posterior) | 9/8/19/16 | 7/4/34/25 | ns |
| Tumor long diameter
(mm) | 17.2±4.9 | 17.7±6.4 | ns |
| Tumor short diameter
(mm) | 14.0±4.9 | 13.7±5.4 | ns |
The study protocol was conducted with the approval
(approval no. 2018143) of the Ethics Committee of Kansai Medical
University Medical Center (Moriguchi, Japan). All procedures
performed in studies involving human participants were in
accordance with the ethical standards of Clinical Research Board of
Kansai Medical University Medical Center and with the 1964 Helsinki
Declaration and its later amendments or comparable ethical
standards. Informed consent was obtained from all individual
participants included in the present study.
Equipment
MWA: Emprint™ Ablation Generator
with Thermosphere Technology with Emprint™ Long
Percutaneous Antenna (30 cm; Covidien).
RFA: Cool-tip RF Generator with Cool-tip RF
needle (25x3 cm; Covidien).
PLT procedure
After pretreatment with 0.1 mg of fentanyl (Janssen
Pharma) and 1.25 mg of droperidol (Daiichi Sankyo), the tumor was
detected using an ultrasound diagnostic system (TUS-A300 Aplio300;
Canon) with an ultrasound transducer (PVT-382BT; Canon).
Subsequently, local anesthesia was administered as 0.5% lidocaine
hydrochloride (Aspen Japan), and a guide needle (MWA, 12Gx140 mm;
RFA, 14Gx145 mm; Hakko Medical) was inserted into the vicinity of
the tumor under the guidance of ultrasound with a puncture adapter
(UAGV-027A; Canon) connected to the transducer. After the inner
needle of the guide was removed, the antenna (electrode) was
inserted through the outer needle of the guide to place the antenna
(electrode) in the tumor area. In the case of MWA, the output
energy was gradually increased to 45 W for 30 sec, to 60 W for 30
sec, and then to 75 W until the end of treatment, whereas in the
case of RFA, the output energy was gradually increased to 80 W for
60 sec and to 100 W for 60 sec, followed by cauterization until the
end of treatment at 120 W. Treatments were terminated until the
ablation margin was included in the high-echoic area on the
ultrasound screen.
Evaluation of treatment effect, shape
of necrotic area and follow-up
After 4 days of PLT, the direct effect of ablation
was determined by dynamic CT, and the axial and coronal necrotic
areas were measured with the long and short diameters perpendicular
to it, respectively. Furthermore, we used the equation for the
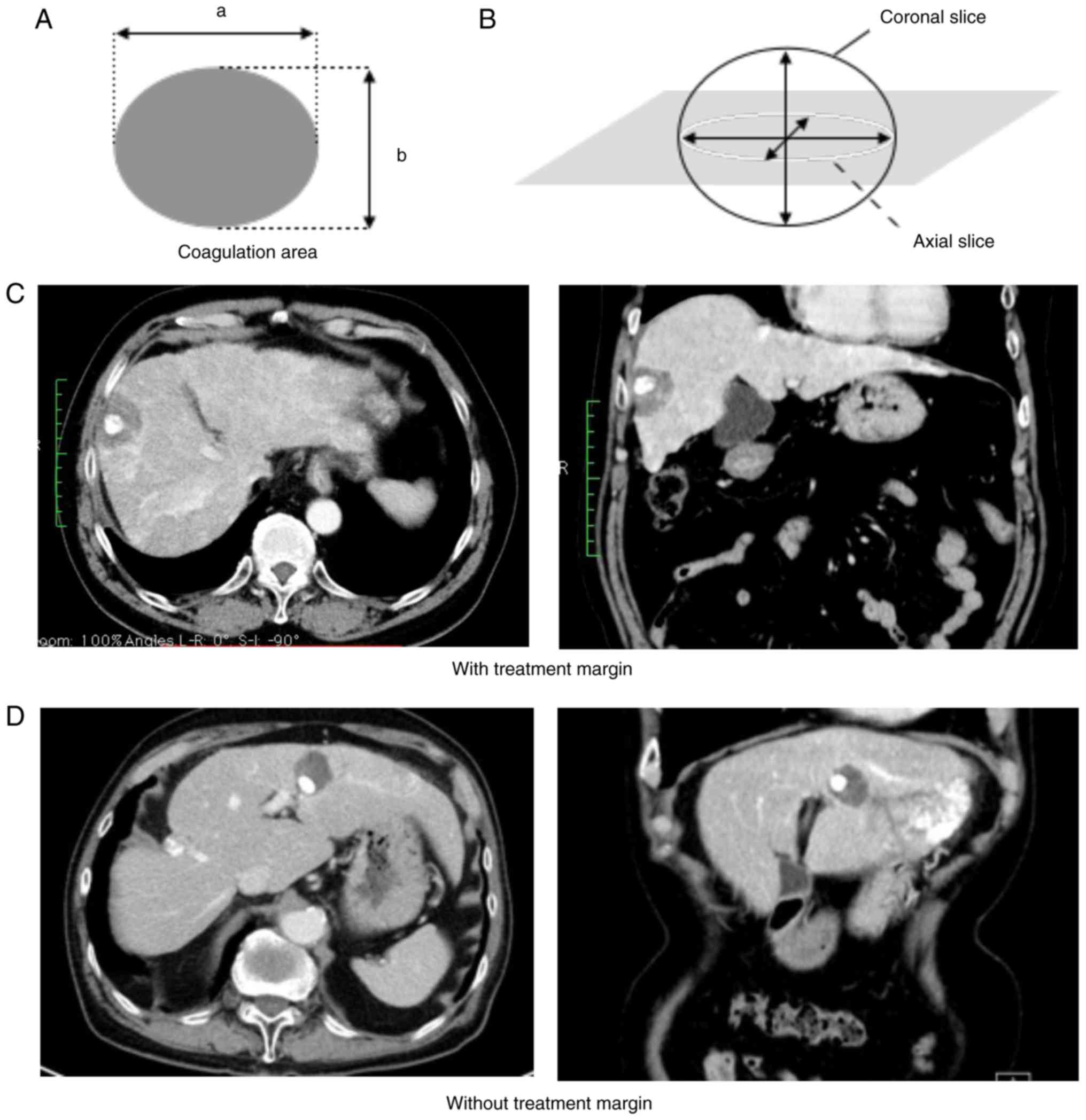
flatness ratio to find out how spherical the necrotic areas were,
which is important for predicting treatment range. The flatness
ratio of the necrotic area in all patients was calculated as
follows: f=1-b/a, where f is the flatness ratio, a is the long
diameter of the ellipse, and b is the short diameter of the
ellipse. The flatness ratio indicates how flat an ellipse is
compared with a sphere, and the more spherical the ellipse is, the
closer the flatness ratio comes to 0 (Fig. 1A and B). Complete tumor necrosis was defined as
100% tumor-necrotizing effect, and we defined the treatment margin
(TM) as the ablation region being wider across the entire
circumference than the low density area in the late phase of
pretreatment dynamic CT. In case treated with c-TACE before PLT, TM
was defined as wider than the accumulation area of the iodinated
oil emulsion over the entire circumference. We subclassified the
areas with and without TM (Fig. 1C
and D).
After their discharge from the hospital, we closely
followed all patients. Dynamic CT scans were performed every 3-4
months. When imaging studies revealed intrahepatic recurrence, the
diagnosis was confirmed by CT angiography and/or US-guided tumor
biopsy. Local recurrence was defined as recurrence of nodule in the
treatment area or the margin of the treatment area in patients.
Statistical analysis
The local recurrence rate of HCC after PLT was
determined using the Kaplan-Meier method. Local recurrence curves
were compared between MWA and RFA by means of the log-rank test.
The clinical data of the patients undergoing each procedure were
compared with the Mann-Whitney U test, while underlying liver
diseases and tumor segments were compared with Pearson's Chi-square
test. P<0.05 was considered to indicate a statistically
significant difference.
Results
Treatment efficacy
All patients completed PLT in one day. Treatment was
terminated once the ablation margin was included in the high-echoic
area on the ultrasound screen. With regard to MWA, most of the
nodules were treated with only one session (94.2%), but three
nodules required two sessions (mean: 1.05±0.23 sessions). However,
with regard to RFA, one session was used to treat 53 nodules
(75.7%), two sessions were used to treat 14 nodules (20%), and
three sessions were used to treat 3 nodules (4.2%) (mean: 1.28±0.54
sessions). The mean ablation times in MWA and RFA were 5.0±2.0 and
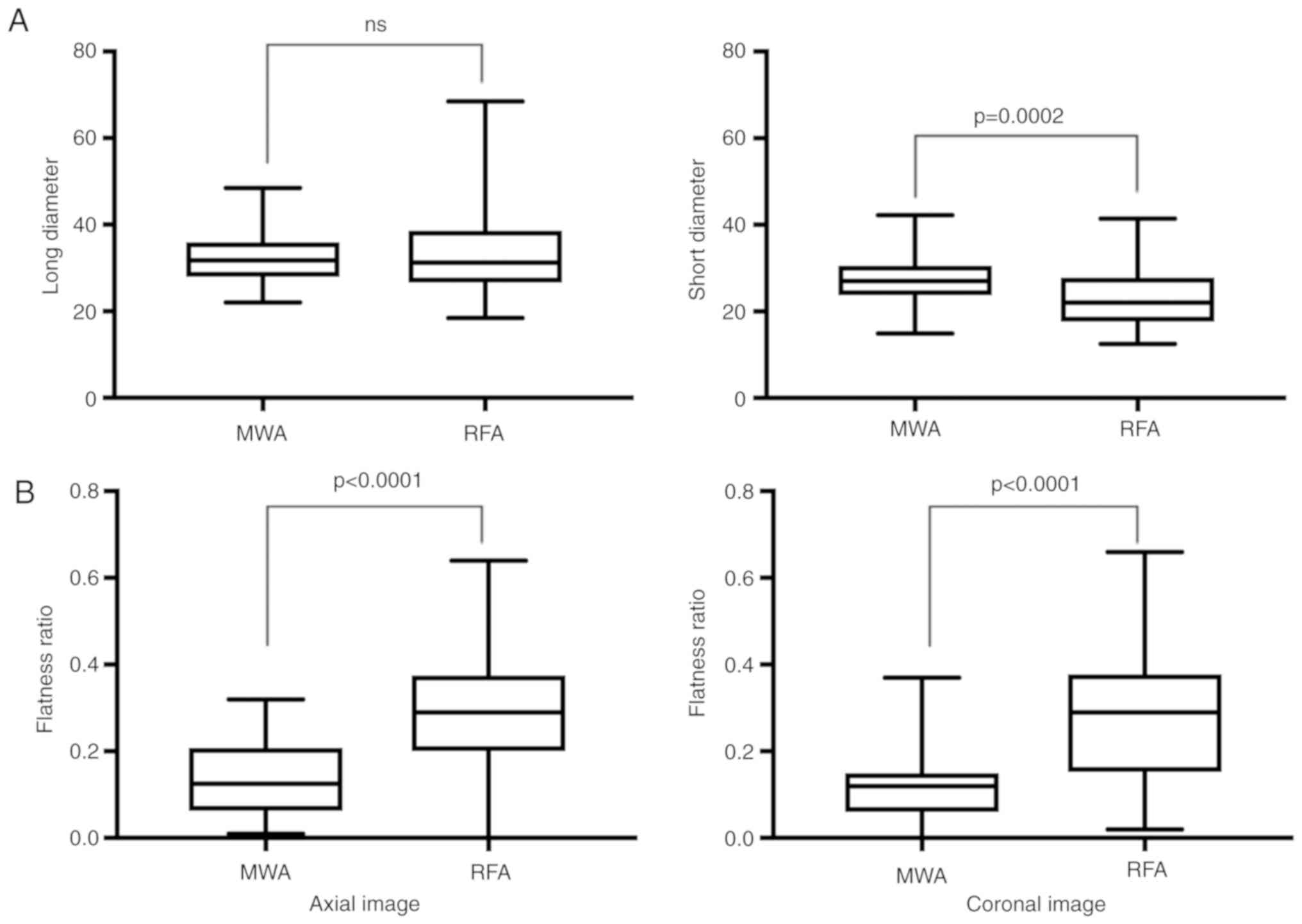
8.1±4.8 min (P=0.0066), respectively. The ablation ranges evaluated
with the axial images for MWA and RFA were 31.9±5.5 and 33.3±9.0
mm, respectively, in the long-axis diameter and 27.6±5.3 and
23.4±6.8 mm, respectively, in the short-axis diameter, and there
was a significant difference only in the short-axis diameter
(P=0.0002). The flatness ratios of the ablation region were
0.13±0.09 for MWA and 0.29±0.14 for RFA on the axial image
(P<0.0001) and 0.11±0.07 for MWA and 0.28±0.14 for RFA on the
coronal image (P<0.0001; Fig. 2).
Compared with RFA, MWA caused spherical ablation in a shorter time
with fewer punctures. Forty-eight of the 52 nodules treated with
MWA showed complete tumor necrosis (with TM 35 nodules: without TM
13 nodules), while sixty-four of the 70 nodules treated with RFA
showed complete tumor necrosis (with TM 37 nodules: Without TM 27
nodules). Patients who had insufficient ablation underwent
additional PLT or surgery at a later date. Although there was no
significant difference in the rate of complete tumor necrosis
between the two groups, the treatment margin acquisition rate was
somewhat higher for MWA than RFA (MWA/RFA: 72.9/57.8%) (Table II).
 | Table IILocal recurrence with and without a TM
and time to recurrence. |
Table II
Local recurrence with and without a TM
and time to recurrence.
| | | | Days until
recurrence |
|---|
| Variables | Nodules | Local recurrence | Shortest | Longest | Mean |
|---|
| MWA |
|
With TM | 35 | 0 | - | - | - |
|
Without
TM | 13 | 2 | 223 | 238 | 230 |
| RFA |
|
With TM | 37 | 2 | 411 | 453 | 432 |
|
Without
TM | 27 | 8 | 229 | 538 | 359 |
Side effects and complications
With regard to the clinically relevant complications
that developed during hospitalization, liver infarction occurred in
one patient undergoing MWA and in two patients undergoing RFA.
After MWA, three cases of bleeding and subcapsular hematoma
occurred; two of the cases spontaneously regained hemostasis due to
bed rest, and one case required intravascular embolization. After
RFA, three cases of subcapsular hematoma occurred, but all cases
achieved spontaneous hemostasis due to bed rest. With regard to
other complications, one patient experienced a skin burn during
RFA, and one patient experienced a marked decrease in blood
pressure due to activation of the vaso-vagal reflex during
cauterization by RFA, which necessitated the cessation of ablation.
During the observation period, we detected biloma of the treatment
area in two patients who had undergone MWA and in one patient who
had undergone RFA. There were no statistically significant
differences in the incidence rates of complications between the MWA
and RFA groups (MWA/RFA: 13.6/14.5%). None of the patients
developed local dissemination of the cancer cells along the
puncture line in this study.
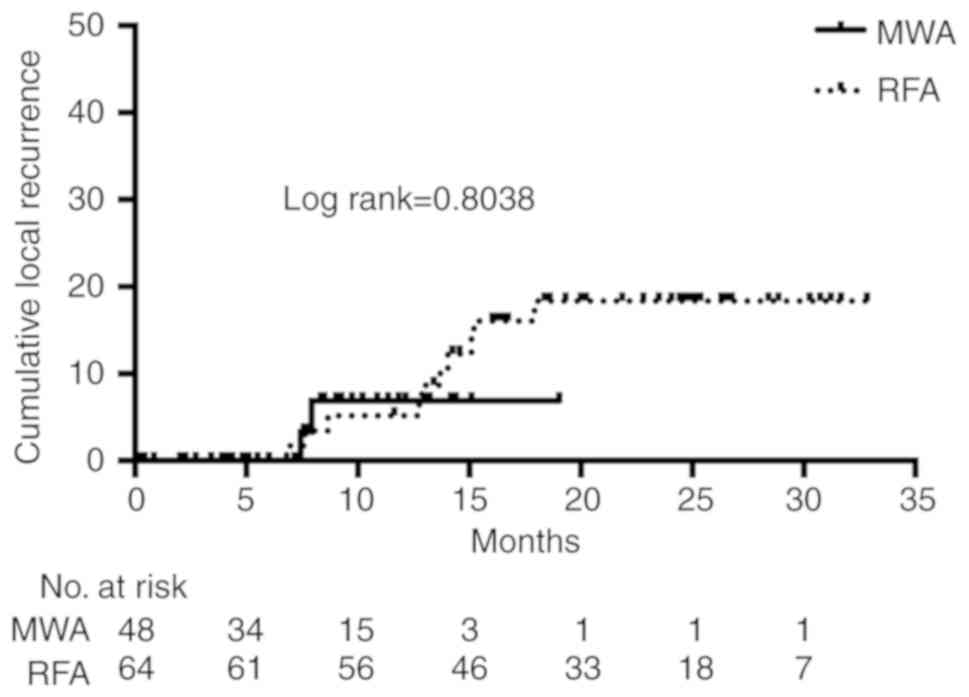
Local recurrence
Among the patients who achieved complete tumor
necrosis, local recurrence was observed in three patients treated
with MWA and ten patients treated with RFA. The cumulative local
recurrence rates were not significantly different between the MWA
and RFA groups (HR=0.82 (0.18-3.73) log-rank=0.8036, one-year
cumulative recurrence rate; MWA/RFA: 6.91/5.17%) (Fig. 3). Table
II shows the results of the subgroup analysis according to the
presence of a TM. Although local recurrence did not occur in MWA
patients who obtained complete necrosis with TM, a comparison could
not be made because the number of cases and observation period were
insufficient.
Discussion
With the positive influence of the surveillance
program in high-risk patients, HCC is being detected at
increasingly smaller sizes. MWA and RFA have high local tumor
control abilities and result in low levels of invasion and are
therefore recommended as the most appropriate treatment options of
early-HCC (13,14). Previous studies had reported that
first-generation MWA is a useful treatment for small HCC tumors
(15,16), and in several reports, MWA and RFA
have been reported to yield equivalent results in terms of
therapeutic effects and complications (17,18).
However, compared with RFA, first-generation MWA has a narrow range
of ablation in one session; therefore, more treatment sessions are
needed to obtain complete tumor necrosis with the appropriate
margin. Under these circumstances, RFA has been reported to be
equivalent to surgery in terms of the survival and tumor control
rates (19,20), and it has been adopted as the most
popular thermal ablation method.
However, RFA has a disadvantage, namely, if the
target lesion is in the vicinity of a large vessel, RFA cannot
sufficiently cauterize the tissue due to the heat sink effect; this
is a phenomenon that occurs when thermal energy diffuses away from
the target lesion due to blood flow in adjacent vessels (10). MWA is less susceptible to the heat
sink effect because of its higher temperatures and shorter ablation
times (21,22). This reduced susceptibility is
ultimately due to differences in the mechanisms of action between
MWA and RFA. RFA uses current, whereas MWA uses electromagnetic
energy (23). The limitations of
first-generation MWA were the unpredictable size and shape of the
ablation zone, but the new-generation MWA is designed to create a
large predictable spherical ablation zone that is unaffected by
changes in the tissue environment (12). The benefits of spherical ablation can
also be superior to RFA. The elliptical spherical ablation of RFA
results from the relatively narrow ablation range in the direction
vertical to the electrode, such that securing of the TM may be
insufficient. Spherical ablation of the new-generation MWA is
considered to be likely to achieve sufficient and even TM, and the
decreased number of sessions and shorter treatment times should be
very therapeutically beneficial for the patient.
We developed a protocol to gradually increase the
output to prevent popping based on Covidien's basic experiments,
but this protocol will be improved based on future clinical
experience; nevertheless, satisfactory results have been obtained
that are comparable to those of RFA in this study. In the
comparison between the two thermal ablation methods used in this
study, there was no difference in the local recurrence rates. In
patients who obtained complete necrosis with TM, there was no local
recurrence in MWA, which may be because tissue ablation was
obtained more reliably than in RFA due to the lower susceptibility
to the heat sink effect.
Unfortunately, in this study, six patients
experienced complications due to MWA. There was concern about the
risk of bile duct injury due to the higher temperature and bleeding
caused by the thicker antennas, but there were no significant
differences in complications compared with those resulting from
MWA.
There are some limitations to this study. This
retrospective cohort study has not been randomized. Although there
were very few differences in the clinical data between the two
groups, the RFA procedure was performed early and the MWA procedure
was performed later in the study, which may have resulted in bias.
For HCC<3.0 cm in diameter located outside the hilar region, the
new-generation MWA may be recommended over RFA due to its short
treatment time. However, in our daily treatment protocol, HCC
located near the hilar region is treated with c-TACE and
percutaneous ethanol injection (PEI), so this study does not
provide treatment efficacy and safety comparison between MWA and
RFA. The risk of PLT complications for HCC increases by their
perivascular presence. The possibility of bile duct injury
increases especially when the mass was adjacent to the hilar
region. Heat-based thermal ablation methods can also cause vascular
thrombosis. Extensive thrombosis can cause liver failure in
patients with poor hepatic reserves, so it must be paid a great
careful to heat-based thermal ablation of the hilar region
(24). The sample size may be small
to detect statistically significant differences in some treatment
outcomes. However, this preliminary results of early reporting on
the performance of the new-generation MWA device create the basis
for prospective study on this topic with less literature.
There is less experience with the new-generation
MWA, and the treatment strategy is not sufficiently developed.
Further accumulation of cases is necessary for a detailed
comparison with RFA.
Acknowledgements
Professor Tomoki Kitawaki; Department of
Mathematics, Kansai Medical University, for supporting statistical
analysis.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
TS and RT designed the study and wrote the initial
draft of the manuscript. TY contributed to analysis and
interpretation of data and assisted in the preparation of the
manuscript. KS, MY, MM, AN and YO contributed to data collection
and interpretation, and critically reviewed the manuscript. All
authors approved the final version of the manuscript and agree to
be accountable for all aspects of the work in ensuring that
questions related to the accuracy or integrity of any part of the
work are appropriately investigated and resolved.
Ethics approval and consent to
participate
The study protocol was conducted with the approval
(approval no. 2018143) of the Ethics Committee of Kansai Medical
University Medical Center (Moriguchi, Japan). All procedures
performed in studies involving human participants were in
accordance with the ethical standards of Clinical Research Board of
Kansai Medical University Medical Center and with the 1964 Helsinki
Declaration and its later amendments or comparable ethical
standards. Informed consent was obtained from all individual
participants included in the present study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
El-Serag HB: Epidemiology of viral
hepatitis and hepatocellular carcinoma. Gastroenterology.
142:1264–1273. 2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Lencioni R: Loco-Regional treatment of
hepatocellular carcinoma in the era of molecular targeted
therapies. Oncology. 78:107–112. 2010.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Llovet JM: Updated treatment approach to
hepatocellular carcinoma. J Gastroenterol. 40:225–235.
2005.PubMed/NCBI View Article : Google Scholar
|
|
4
|
European Association for the Study of the
Liver; European Organisation for Research and Treatment of Cancer:
EASL-EORTC clinical practice guidelines: Management of
hepatocellular carcinoma. J Hepatol 56: 908-943, 2012.
|
|
5
|
Bruix J and Sherman M: Practice Guidelines
Committee American Association for the Study of Liver Diseases:
Management of hepatocellular carcinoma. Hepatology. 42:1208–1236.
2005.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Bruix J and Sherman M: American
Association for the Study of Liver Diseases: Management of
hepatocellular carcinoma: An update. Hepatology. 53:1020–1022.
2011.PubMed/NCBI View Article : Google Scholar
|
|
7
|
McCarley JR and Soulen MC: Percutaneous
ablation of hepatic tumors. Semin Intervent Radiol. 27:255–260.
2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Shiina S, Teratani T, Obi S, Hamamura K,
Koike Y and Omata M: Nonsurgical treatment of hepatocellular
carcinoma: From percutaneous ethanol injection therapy and
percutaneous microwave coagulation therapy to radiofrequency
ablation. Oncology. 62:64–68. 2002.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Yamasaki T, Kurokawa F, Shirahashi H,
Kusano N, Hironaka K and Okita K: Percutaneous radiofrequency
ablation therapy with combined angiography and computed tomography
assistance for patients with hepatocellular carcinoma. Cancer.
91:1342–1348. 2001.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Lu DS, Raman SS, Limanond P, Aziz D,
Economou J, Busuttil R and Sayre J: Influence of large peritumoral
vessels on outcome of radiofrequency ablation of liver tumors. J
Vasc Inter Radiol. 14:1267–1274. 2003.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Ierardi AM, Mangano A, Floridi C, Dionigi
G, Biondi A, Duka E, Lucchina N, Lianos GD and Carrafiello G: A new
system of microwave ablation at 2450 MHz: Preliminary experience.
Updates Surg. 67:39–45. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Alonzo M, Bos A, Bennett S and Ferral H:
The emprint ablation system with thermosphere technology: One of
the newer next-generation microwave ablation technologies. Semin
Intervent Radiol. 32:335–338. 2015.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Thandassery RB, Goenka U and Goenka MK:
Role of local ablative therapy for hepatocellular carcinoma. J Clin
Exp Hepatol. 4 (Suppl 3):S104–S111. 2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Shiina S, Sato K, Tateishi R, Shimizu M,
Ohama H, Hatanaka T, Takawa M, Nagamatsu H and Imai Y: Percutaneous
ablation for hepatocellular carcinoma: Comparison of various
ablation techniques and surgery. Can J Gastroenterol Hepatol.
2018(4756147)2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Seki T, Wakabayashi M, Nakagawa T, Itho T,
Shiro T, Kunieda K, Sato M, Uchiyama S and Inoue K: Ultrasonically
guided percutaneous microwave coagulation therapy for small
hepatocellular carcinoma. Cancer. 74:817–825. 1994.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Seki T, Tamai T, Nakagawa T, Imamura M,
Nishimura A, Yamashiki N, Ikeda K and Inoue K: Combination therapy
with transcatheter arterial chemoembolization and percutaneous
microwave coagulation therapy for hepatocellular carcinoma. Cancer.
89:1245–1251. 2000.PubMed/NCBI
|
|
17
|
Vogl TJ, Farshid P, Naguib NN, Zangos S,
Bodelle B, Paul J, Mbalisike EC, Beeres M and Nour-Eldin NE:
Ablation therapy of hepatocellular carcinoma: A comparative study
between radiofrequency and microwave ablation. Abdom Imaging.
40:1829–1837. 2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Facciorusso A, Di Maso M and Muscatiello
N: Microwave ablation versus radiofrequency ablation for the
treatment of hepatocellular carcinoma: A systematic review and
meta-analysis. Int J Hyperthermia. 32:339–344. 2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhou Y, Zhao Y, Li B, Xu D, Yin Z, Xie F
and Yang J: Meta-analysis of radiofrequency ablation versus hepatic
resection for small hepatocellular carcinoma. BMC Gastroenterol.
10(78)2010.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Feng K, Yan J, Li X, Xia F, Ma K, Wang S,
Bie P and Dong J: A randomized controlled trial of radiofrequency
ablaion and surgical resection in the treatment of small
hepatocellular carcinoma. J Hepatol. 57:794–802. 2012.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Lucchina N, Tsetis D, Ierardi AM,
Giorlando F, Macchi E, Kehagias E, Duka E, Fontana F, Livraghi L
and Carrafiello G: Current role of microwave ablation in the
treatment of small hepatocellular carcinomas. Ann Gastroenterol.
29:460–465. 2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Dou JP, Yu J, Yang XH, Cheng ZG, Han ZY,
Liu FY, Yu XL and Liang P: Outcomes of microwave ablation for
hepatocellular carcinoma adjacent to large vessels: A propensity
score analysis. Oncotarget. 8:28758–28768. 2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Lubner MG, Brace CL, Hinshaw JL and Lee FT
Jr: Microwave tumor ablation: Mechanism of action, clinical
results, and devices. J Vasc Inter Radiol. 21 (8 Suppl):S192–S203.
2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Rhim H: Complications of radiofrequency
ablation in hepatocellular carcinoma. Abdom Imaging. 30:409–418.
2005.PubMed/NCBI View Article : Google Scholar
|