Introduction
Pulmonary metastasis (PM) is the leading cause of
mortality among patients with bone and soft tissue sarcoma (BSTS).
Furthermore, unresectable PM is associated with an extremely poor
prognosis, with a median survival of 11 months (1). Surgical resection is a generally
accepted treatment for PM, with complete resection of PM achieving
a survival of up to 33 months and 3 year survival rates of 30-42%
(1,2). However, surgical management of PM has
achieved important results in soft tissue sarcoma treatment, but
without level I or II evidence (1).
Approximately 25% of the patients are alive 5 years after surgery,
but most studies do not reveal whether these patients survive with
or without relapse (3). To the best
of our knowledge, no randomized trials comparing survival after
surgery and non-surgical treatment have been published to date
(3). Furthermore, the populations
from which patients were selected for pulmonary metastasectomy and
the prognosis of unresected patients are seldom reported.
Therefore, the aim of the present study was to retrospectively
compare the prognosis of patients with PM from BSTS receiving
surgical and non-surgical treatment.
Our preliminary study demonstrated that lung
metastasectomy had the potential to improve patient prognosis
(4). Thus, it was herein
hypothesized that pulmonary metastasectomy may be associated with
an improved prognosis in this setting, compared with non-surgical
treatment. The present study compared patients with PM from BSTS
according to whether they underwent surgical or non-surgical
treatment, with the aim to identify factors that are predictive of
a good outcome among the surgically treated patients.
Patients and methods
Patients
The present study retrospectively reviewed
prospectively collected oncology/reconstruction surgery data and
identified 56 consecutive patients (33 men and 23 women) with BSTS
who developed PM between January 2005 and March 2017. The
retrospective protocol of the present study was approved by the
Institutional Review Board of Sapporo Medical University (Sapporo,
Japan). Patients who participated in this research had complete
clinical data. The signed informed consents were obtained from the
patients or the guardians. All patients had complete local control
of the primary tumor and underwent chest computed tomography (CT)
as part of metastatic surveillance at 3-6 month intervals. PM was
defined as a growing nodule that was detected during CT
examination. Surgical staging was performed according to the
seventh edition of the TNM system of the International Union
Against Cancer (5). Histological
grading was performed based on the system for soft tissue sarcoma
from the French Federation of Cancer Centers (6), which is based on the tumor's mitotic
index, extent of necrosis, and degree of histological
differentiation. Data were collected for each patient regarding
demographic characteristics (age and sex), primary tumor
histopathology, metastasis characteristics (number and location),
surgical approach, outcome, and interval between the primary
treatment and PM diagnosis.
Treatment
The decision to perform pulmonary metastasectomy was
based on the discretion of the attending thoracic surgeon after
discussion. The eligibility criteria for pulmonary metastasectomy
were as follows: i) The primary sarcoma had been completely
resected, ii) there were ≤5 PM nodules, iii) the pulmonary lesions
were considered completely resectable, iv) the patient's general
condition was sufficient to withstand surgery and v) the patient
provided informed consent. The patients underwent video-assisted
thoracic surgery (VATS) that was performed by several thoracic
surgeons at our hospital, and the specific operative approach was
selected based on the surgeon's judgement and the feasibility of R0
resection. All patients were carefully followed up using CT every
3-6 months after surgery. Whether to administer chemotherapy was
decided through discussion with the oncologists.
Statistical analysis
Demographic characteristics and overall survival
(OS) were compared between patients who underwent pulmonary
metastasectomy or non-surgical treatment. The demographic and
clinical characteristics were compared using Fisher's exact test or
the Mann-Whitney U test, as appropriate. The OS outcomes were
calculated from the metastasectomy or initiation of non-surgical
treatment to the date of death or the date of the last follow-up.
Kaplan-Meier curves were used to describe OS, and the results were
compared using the log-rank test. All statistical data were
analyzed using GraphPad Prism, version 5.0 f (GraphPad Software
Inc.) and P<0.05 was considered to indicate statistically
significant differences.
Results
Patients and characteristics
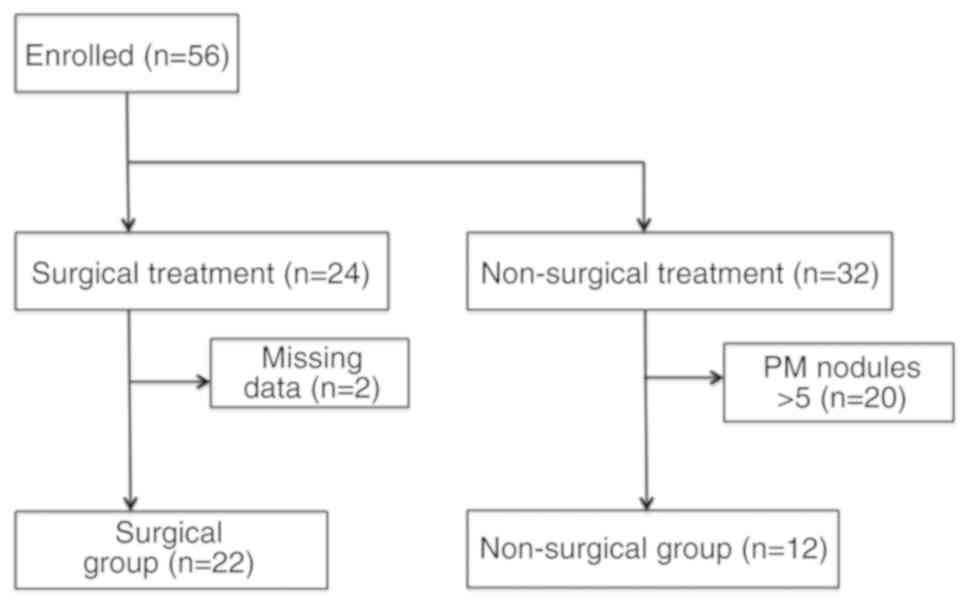
Between January 2005 and March 2017, 56 patients who
developed PM from BSTS were identified. A total of 24 patients
underwent pulmonary metastasectomy and 32 patients received
non-surgical therapy. Patients with >5 PM nodules or missing
data were excluded from the study (Fig.
1). The final analysis included 34 consecutive patients with a
median age of 64 years (range, 14-89 years) and a median follow-up
of 14.5 months (range, 1-136 months). The most common histological
type was osteosarcoma (n=10), followed by leiomyosarcoma (n=5),
undifferentiated pleomorphic sarcoma (n=5) and myxofibrosarcoma
(n=3). The patients' demographic and tumor characteristics are
summarized in Table I. Of the 34
patients, 22 underwent surgery and 15 of those patients received
chemotherapy. The remaining 12/34 patients did not undergo surgery.
However, 10 of those patients received chemotherapy. Two patients
did not receive any treatment (Table
II). Patients who received non-surgical treatment were
significantly older compared with patients who underwent pulmonary
metastasectomy (P=0.0162).
 | Table ICharacteristics of all patients. |
Table I
Characteristics of all patients.
| Characteristics | No |
|---|
| Sex
(male/female) | 19/15 |
| Age, years
(median/range) | 64.0/14-89 |
| Location
(extremity/trunk) | 25/9 |
| Adjuvant
chemotherapy | |
|
Yes | 25 |
|
No | 9 |
| Histology | |
|
Osteosarcoma | 10 |
|
Leiomyosarcoma | 5 |
|
UPS/MFS | 5/3 |
|
Dedifferentiated
liposarcoma | 3 |
|
MPNST | 2 |
|
Chondrosarcoma | 2 |
|
Chordoma | 1 |
|
Myxoid
liposarcoma | 1 |
|
Epithelioid
sarcoma | 1 |
|
Synovial
sarcoma | 1 |
| Bone sarcoma/soft
tissue sarcoma | 13/21 |
| Grade of soft tissue
sarcoma (I/II/III) | 4/3/14 |
 | Table IICharacteristics of patients according
to whether they did (surgical group) or did not undergo pulmonary
metastasectomy (non-surgical group). |
Table II
Characteristics of patients according
to whether they did (surgical group) or did not undergo pulmonary
metastasectomy (non-surgical group).
| Patient
characteristics | Non-surgical
group | Surgical group | P-value |
|---|
| No | 12 | 22 | |
| Sex
(male/female) | 7/5 | 12/10 | NS |
| Age (years
median/range) | 68.5/17-89 | 49.5/14-81 | 0.0162 |
| Location
(extremity/trunk) | 7/5 | 18/4 | NS |
| Number of metastasis
(median/range) | 2.0/1-5 | 2.0/1-5 | NS |
| Adjuvant
chemotherapy | | | NS |
|
Yes | 10 | 15 | |
|
No | 2 | 7 | |
| Histology | | | NS |
|
Osteosarcoma | 1 | 9 | |
|
Leiomyosarcoma | 2 | 3 | |
|
UPS/MFS | 3/2 | 2/1 | |
|
Dedifferentiated
liposarcoma | 0 | 3 | |
|
MPNST | 1 | 1 | |
|
Chondrosarcoma | 2 | 0 | |
|
Chordoma | 0 | 1 | |
|
Mixoid
liposarcoma | 0 | 1 | |
|
Eplithelioid
sarcoma | 1 | 0 | |
|
Synovial
sarcoma | 0 | 1 | |
| Bone sarcoma/soft
tissue sarcoma | 3/9 | 10/12 | NS |
| Tumor grade of soft
tissue sarcoma (Grade I/II/III) | 2/2/5 | 2/1/9 | NS |
Survival
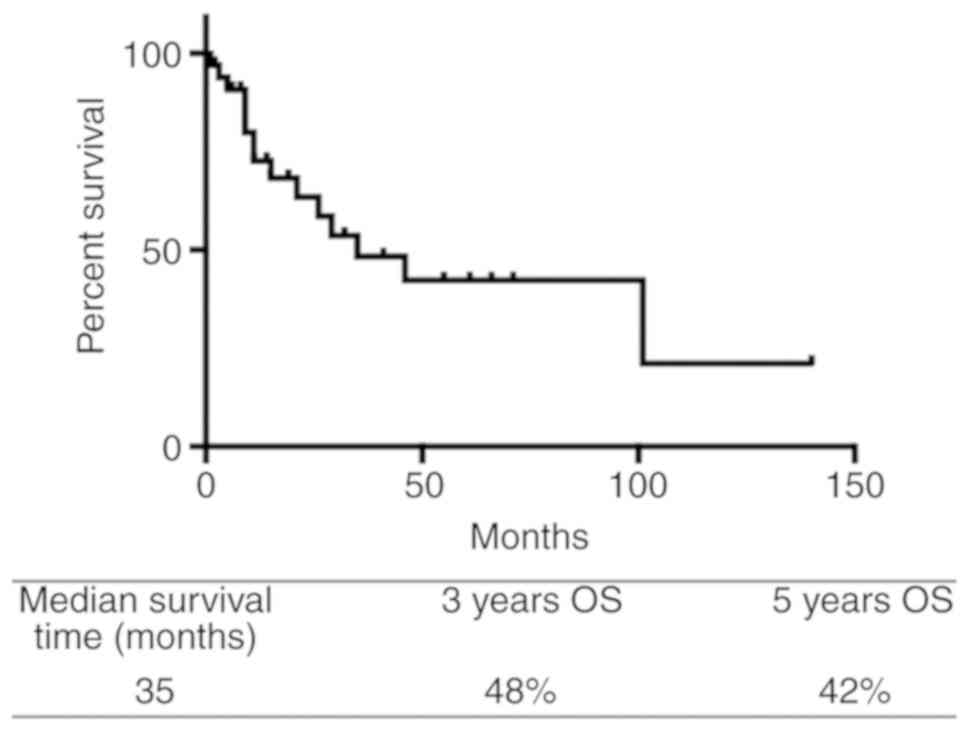
The OS curve for all patients (3 year OS, 48%) is
shown in Fig. 2. The univariate
analyses failed to identify any demographic or clinical factors
that significantly affected the prognosis among all 34 patients
(Table I). Patients who underwent
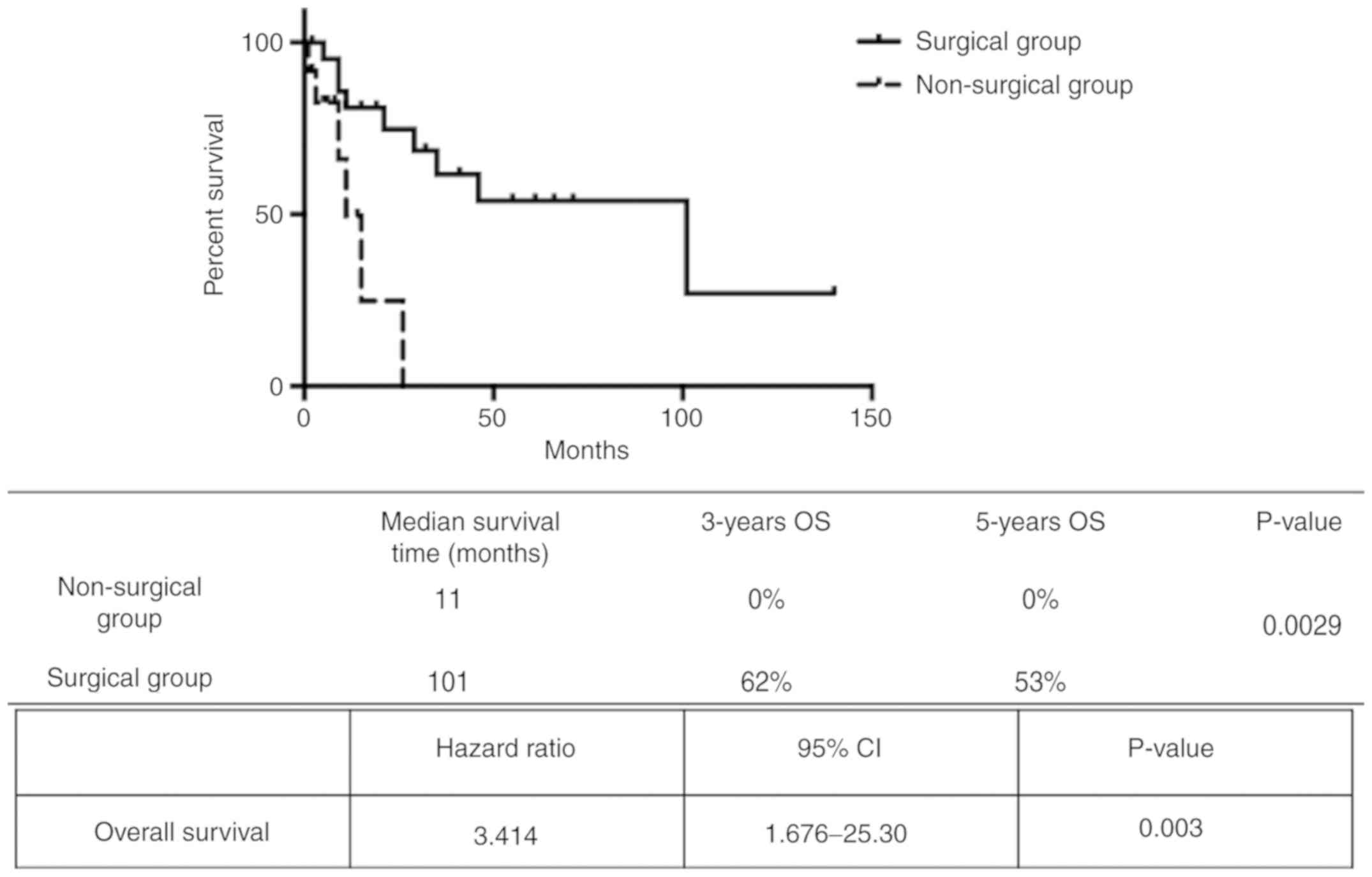
pulmonary metastasectomy had a median survival of 101 months, while
the non-surgical group had a median survival of 11 months (Fig. 3). Compared with the non-surgical
group, the pulmonary metastasectomy group had higher 3 and 5 year
OS rates (0 vs. 62% and 0 vs. 53%, respectively). The log-rank test
confirmed that these differences were statistically significant
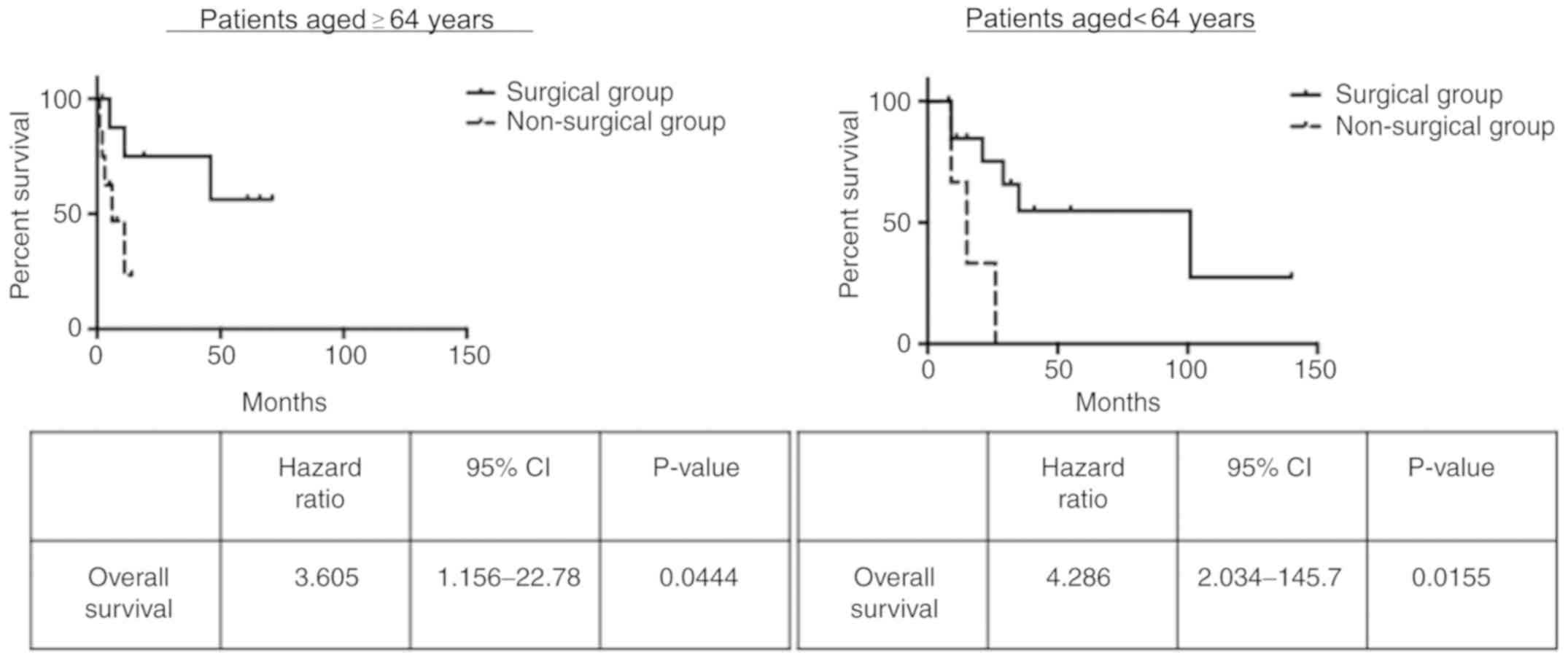
(P=0.0029). Furthermore, pulmonary metastasectomy was associated
with significantly improved survival among patients who were aged
<64 years (P=0.0155) as well as those who were aged ≥64 years
(P=0.0444; Fig. 4). This finding
demonstrated that age did not affect the survival of patients who
underwent surgical treatment.
Characteristics of the pulmonary
metastasectomy group
A total of 22 patients underwent pulmonary
metastasectomy via VATS, and their characteristics are summarized
in Table III. A total of 17
patients (77%) underwent wedge resection, 3 patients (14%)
underwent lobectomy, and 2 patients (9%) underwent segmental
resection, with a median number of resected metastases of 2 (range,
1-5). R0 resection was achieved in all patients, and the median
disease-free interval was 15 months. The mean hospital stay was 8
days (range, 5-15 days), and only 1 patient experienced
pneumothorax as a postoperative complication. There were no
postoperative fatalities. A total of 13 patients remained alive
without disease after their first thoracic surgery, and 9 patients
developed recurrence at a median interval of 9 months (range, 3-46
months).
 | Table IIICharacteristics of patients who
underwent pulmonary metastasectomy. |
Table III
Characteristics of patients who
underwent pulmonary metastasectomy.
| Variables | Overall(Range) |
|---|
| Patient number | 22 |
| Operative approach
(VATS/open) | 22/0 |
| Laterally
(unilateral/bilateral) | 22/0 |
| Type of resection
(wedge/lobectomy/segmetal) | 17/3/2 |
| Completeness of
resection (R0/R1/R2) | 23/0/0 |
| Hospitalization
(days) | 8 (5-15) |
| Complications | 1 |
| Pneumothorax | 1 |
Analysis according to outcomes in the
pulmonary metastasectomy group
The pulmonary metastasectomy group included 9
patients who succumbed to their disease during follow-up (poor
outcome) and 13 patients who survived throughout the follow-up
period (good outcome). Comparison of these subgroups failed to
detect any significant predictors of poor or good outcomes
(Table IV).
 | Table IVCharacteristics of patients who
experienced poor and good outcomes. |
Table IV
Characteristics of patients who
experienced poor and good outcomes.
| Variables | Poor outcome | Good outcome | P-value |
|---|
| Patient no | 9 | 13 | |
| Sex
(male/female) | 4/5 | 8/5 | NS |
| Age
(median/range) | 48.0/15-82 | 54.0/14-81 | NS |
| Number of
metastasis (median/range) | 1.0/1-5 | 2.0/1-5 | NS |
| Adjuvant
chemotherapy | | | NS |
|
Yes | 6 | 9 | |
|
No | 3 | 4 | |
| Location | | | NS |
|
Spine/Pelvic | 3 | 1 | |
|
Extremity | 6 | 12 | |
| Pathology | | | NS |
|
Bone | 6 | 4 | |
|
Soft
tissue | 3 | 9 | |
| DFI
(median/months) | 9/3-46 | 19/2-66 | NS |
Discussion
Previous reports have suggested that pulmonary
metastasectomy confers a survival benefit. Rusch reported that the
5 year OS rate was 17% among patients who underwent non-surgical
treatment for PM from BSTS, whereas several other reports revealed
3 year OS rates of 28-35% with resection of lung metastases and 5
year OS rates of 21-38% (7-10).
Complete resections were performed in the majority of these cases,
although certain reports included partial resections (7-11).
However, this benefit has not been fully validated, as these
reports did not analyze the oncological outcome between patients
who underwent pulmonary metastasectomy or non-surgical treatment
(7,8). Therefore, we herein compared the
prognoses between surgical and non-surgical patients with PM from
BSTS to evaluate the survival impact of surgical resection.
Patients with >5 PM nodules were excluded, as it has been
reported that patients with ≤5 nodules have a longer estimated
median survival compared with those with >5 nodules (12). The present study revealed that
pulmonary metastasectomy was associated with improved OS among
patients with PM from BSTS. These outcomes are further supported by
the fact that we attempted to identify PM as soon as possible via
frequent follow-up pulmonary CT examinations, and that all our
patients were treated using VATS, which allows for faster recovery
and less pain.
The group of patients who underwent pulmonary
metastasectomy was significantly younger compared with the
non-surgical group, which may have been associated with their
improved outcomes. However, we compared patients with a median age
of ≥64 and <64 years and found that the survival benefit of
pulmonary metastasectomy persisted in both analyses. Thus, it
appears that patient age did not significantly affect the
association between pulmonary metastasectomy and outcome.
Several studies have examined prognostic factors
among patients undergoing pulmonary metastasectomy for BSTS, which
were reported to be complete resection, disease-free interval, PM
size, PM number, and laterality (13-17).
However, the present study failed to detect significant prognostic
factors in the pulmonary metastasectomy group. This may be
attributed to the fact that we evaluated prognosis based on whether
the patients were alive with disease or had succumbed to their
disease, without considering the follow-up period. It is also
possible that the relatively small sample size affected the ability
to detect significant prognostic factors.
There were certain limitations to the present study:
i) The retrospective design and small sample size, which are
associated with risks of bias, ii) selection bias was present, as
patients who were unable to tolerate surgery did not undergo
pulmonary metastasectomy, iii) the follow-up period was shorter
compared with those in previous studies and iv) treatment was not
performed in a randomized, controlled manner.
In conclusion, the findings of the present study
revealed that, compared with non-surgical treatment, pulmonary
metastasectomy was associated with improved prognosis in patients
with PM from BSTS.
Acknowledgements
The authors would like to thank Dr Tamotsu Soma, Dr
Mikito Sasaki, Dr Mitsunori Kaya and Dr Takuro Wada for orthopedic
assistance, as well as Dr Tsukasa Hori, Dr Masaki Yamamoto and Dr
Keita Igarashi for pediatric oncological assistance.
Funding
This study was partly supported by grants from JSPS
KAKENHI (18K09112 to ME).
Availability of data and materials
The datasets generated and/or analyzed during the
present study are available from the corresponding author on
reasonable request.
Authors' contributions
JS wrote the manuscript, analyzed the data and
performed the statistical analysis. ME interpreted patient data and
wrote the manuscript. YM collected and interpreted patient data. TS
performed statistical analysis. TM, MM and AW performed pulmonary
metastasectomy. SS and TH made the pathological diagnoses. KT and
KM were involved in administering the adjuvant chemotherapy. TY
designed the present study and wrote the manuscript. All authors
read and approved the final manuscript.
Ethics approval and consent to
participate
The retrospective protocol of the present study was
approved by the Institutional Review Board of Sapporo Medical
University (Sapporo, Japan). Patients who participated in this
research had complete clinical data. The signed informed consents
were obtained from the patients or the guardians.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Billingsley KG, Burt ME, Jara E, Ginsberg
RJ, Woodruff JM, Leung DH and Brennan MF: Pulmonary metastases from
soft tissue sarcoma: Analysis of patterns of diseases and
postmetastasis survival. Ann Surg. 229:602–610. 1999.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Hamaji M, Chen F, Miyamoto E, Kondo T,
Ohata K, Motoyama H, Hijiya K, Menju T, Aoyama A, Sato T, et al:
Surgical and non-surgical management of repeat pulmonary metastasis
from sarcoma following first pulmonary metastasectomy. Surg Today.
46:1296–1300. 2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Treasure T, Fiorentino F, Scarci M, Møller
H and Utley M: Pulmonary metastasectomy for sarcoma: A systematic
review of reported outcomes in the context of thames cancer
registry data. BMJ Open. 8(e001736)2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Shimizu J, Emori M, Yasutaka M and
Yamashita T: Pulmonary metastasectomy is associated with a survival
benefit among patients with bone and soft tissue sarcoma. J East
Jpn Orthop Traumatol. 31:16–21. 2019.(In Japanese).
|
|
5
|
Sobin LH, Gospodarowicz MK and Wittekind C
(eds): TNM classification of malignant tumors 7th ed:
International Union against Cancer (UICC). Vol 78. Elsevier BV,
Netherlands, pp1278, 2010.
|
|
6
|
Coindre JM: Grading of soft tissue
sarcomas: Review and update. Arch Pathol Lab Med. 130:1448–1453.
2006.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Schur S, Hoetzenecker K, Lamm W, Koestler
WJ, Lang G, Amann G, Funovics P, Nemecek E, Noebauer I, Windhager
R, et al: Pulmonary metastasectomy for soft tissue sarcoma-report
from a dual institution experience at the medical university of
vienna. Eur J Cancer. 50:2289–2297. 2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Rehders A, Hosch SB, Scheunemann P,
Stoecklein NH, Knoefel WT and Peiper M: Benefit of surgical
treatment of lung metastasis in soft tissue sarcoma. Arch Surg.
142:70–75. 2007.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Rusch VW: Pulmonary metastasectomy.
Current indications. Chest. 107 (6 Suppl):322S–331S.
1995.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Lin AY, Kotova S, Yanagawa J, Elbuluk O,
Wang G, Kar N, Elashoff D, Grogan T, Cameron RB, Singh A, et al:
Risk stratification of patients undergoing pulmonary metastasectomy
for soft tissue and bone sarcomas. J Thorac Cardiovasc Surg.
149:85–92. 2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Casson AG, Putnam JB, Natarajan G,
Johnston DA, Mountain C, McMurtrey M and Roth JA: Five-year
survival after pulmonary metastasectomy for adult soft tissue
sarcoma. Cancer. 69:662–668. 1992.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Meyer WH, Schell MJ, Kumar AP, Rao BN,
Green AA, Champion J and Pratt CB: Thoracotomy for pulmonary
metastatic osteosarcoma. An analysis of prognostic indicators of
survival. Cancer. 59:374–379. 1987.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Thomford NR, Woolner LB and Clagett OT:
The surgical treatment of metastatic tumors in the lungs. J Thorac
Cardiovasc Surg. 49:357–363. 1965.PubMed/NCBI
|
|
14
|
Smith R, Pak Y, Kraybill W and Kane JM
III: Factors associated with actual long-term survival following
soft tissue sarcoma pulmonary metastasectomy. Eur J Surg Oncol.
35:356–361. 2009.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Garcia Franco CE, Torre W, Tamura A,
Guillen-Grima F, San-Julian M, Martin-Algarra S and Pardo FJ:
Long-Term results after resection for bone sarcoma pulmonary
metastases. Eur J Cardiothorac Surg. 37:1205–1208. 2010.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kim S, Ott HC, Wright CD, Wain JC, Morse
C, Gaissert HA, Donahue DM, Mathisen DJ and Lanuti M: Pulmonary
resection of metastatic sarcoma: Prognostic factors associated with
improved outcomes. Ann Thorac Surg. 92:1780–1786. 2011.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Mizuno T, Taniguchi T, Ishikawa Y,
Kawaguchi K, Fukui T, Ishiguro F, Nakamura S and Yokoi K: Pulmonary
metastasectomy for osteogenic and soft tissue sarcoma: Who really
benefits from surgical treatment? Eur J Cardiothorac Surg.
43:795–799. 2013.PubMed/NCBI View Article : Google Scholar
|