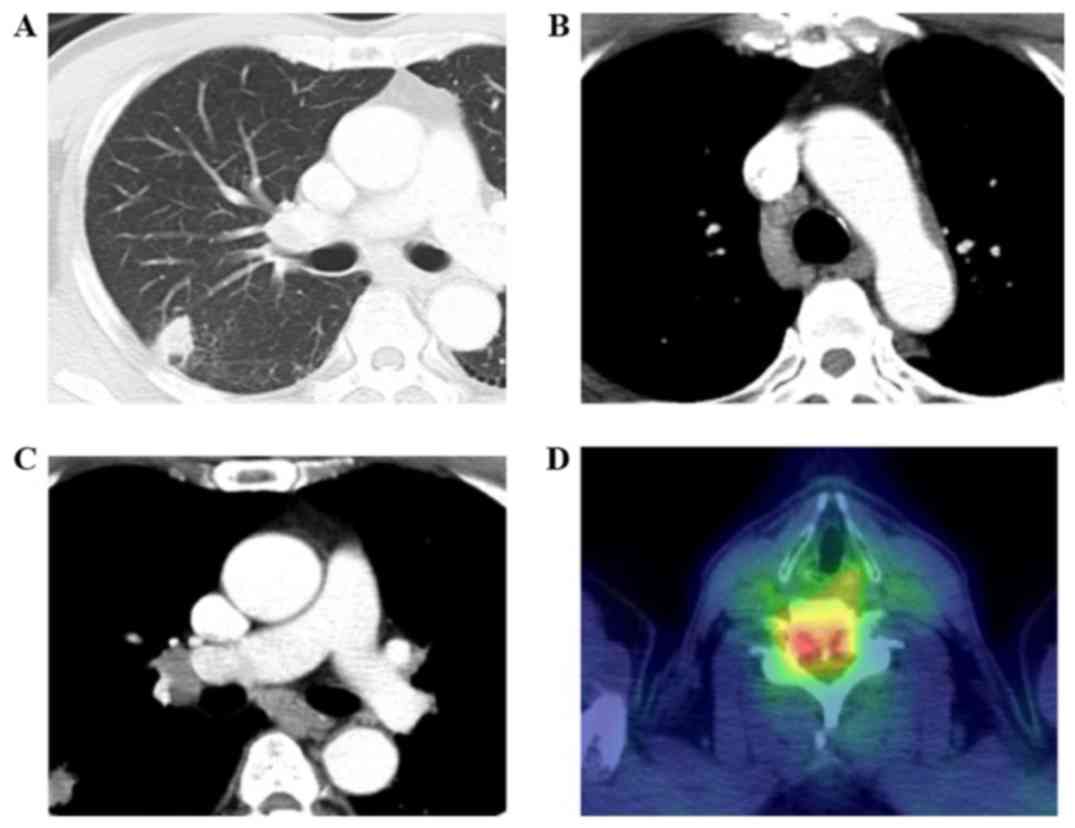
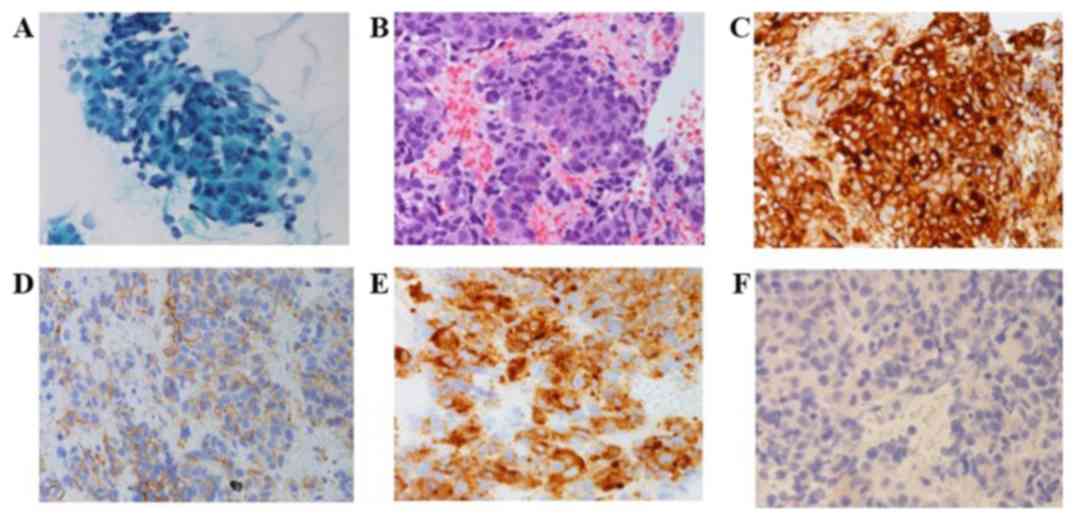
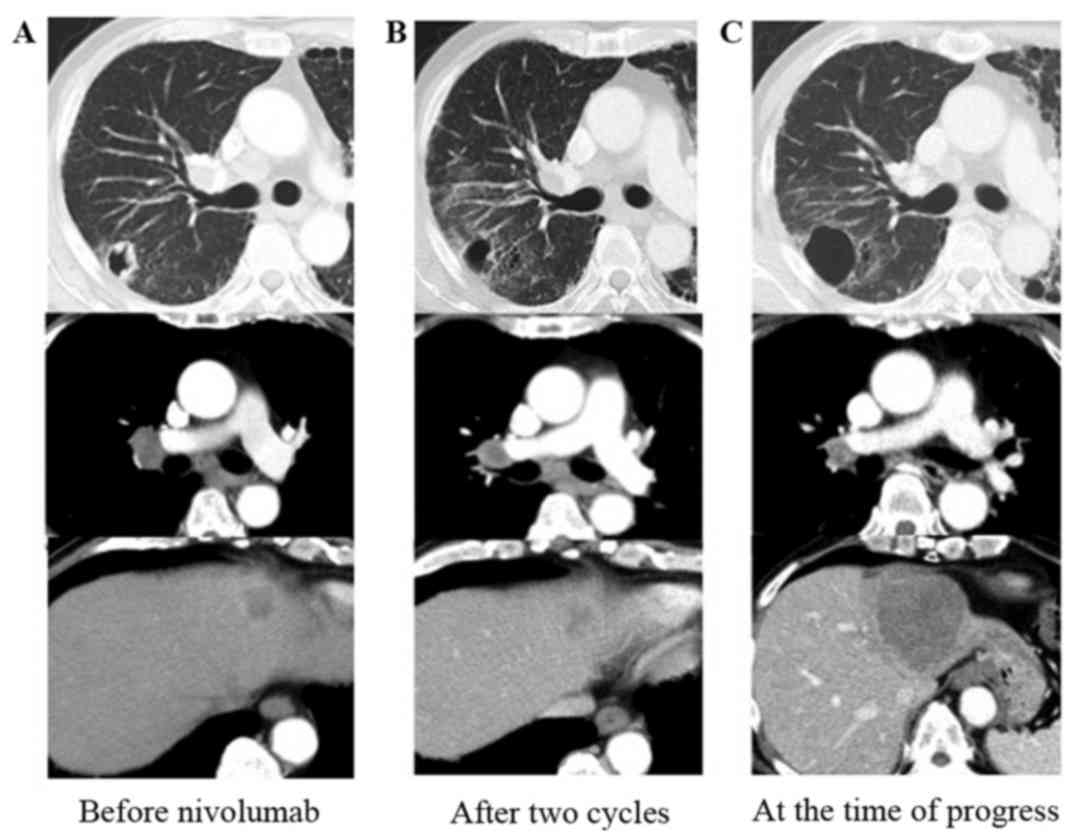
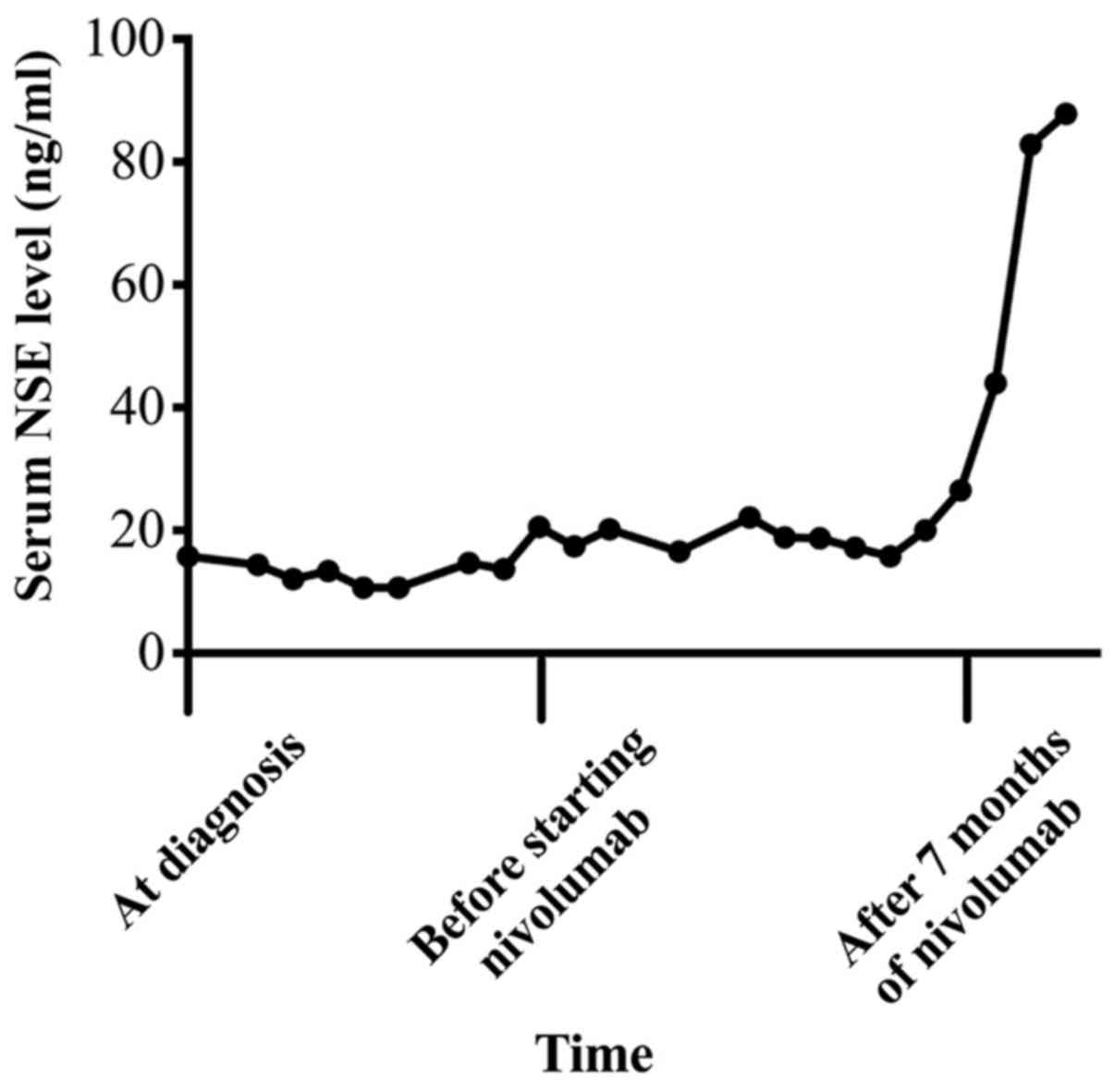
|
1
|
Pardoll DM: The blockade of immune
checkpoints in cancer immunotherapy. Nat Rev Cancer. 12:252–264.
2012.PubMed/NCBI View
Article : Google Scholar
|
|
2
|
Brahmer J, Reckamp KL, Baas P, Crino L,
Eberhardt WE, Poddubskaya E, Antonia S, Pluzanski A, Vokes EE,
Holgado E, et al: Nivolumab versus docetaxel in advanced
squamous-cell non-small-cell lung cancer. N Engl J Med.
373:123–135. 2015.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Borghaei H, Paz-Ares L, Horn L, Spigel DR,
Steins M, Ready NE, Chow LQ, Vokes EE, Felip E, Holgado E, et al:
Nivolumab versus docetaxel in advanced nonsquamous non-small-cell
lung cancer. N Engl J Med. 373:1627–1639. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Gettinger S, Rizvi NA, Chow LQ, Borghaei
H, Brahmer J, Ready N, Gerber DE, Shepherd FA, Antonia S, Goldman
JW, et al: Nivolumab monotherapy for first-line treatment of
advanced non-small-cell lung cancer. J Clin Oncol. 34:2980–2987.
2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Derks JL, Hendriks LE, Buikhuisen WA,
Groen HJ, Thunnissen E, van Suylen RJ, Houben R, Damhuis RA, Speel
EJ and Dingemans AM: Clinical features of large cell neuroendocrine
carcinoma: A population-based overview. Eur Respir J. 47:615–624.
2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Travis WD, Brambilla E, Burke AP, Marx A
and Nicholson AG (eds): WHO classification of tumours of the lung,
pleura, thymus and heart. 4th edition. Lyon, Int Agency Res Cancer,
2015.
|
|
7
|
Niho S, Kenmotsu H, Sekine I, Ishii G,
Ishikawa Y, Noguchi M, Oshita F, Watanabe S, Nakajima R, Tada H and
Nagai K: Combination chemotherapy with irinotecan and cisplatin for
large-cell neuroendocrine carcinoma of the lung: A multicenter
phase II study. J Thorac Oncol. 8:980–984. 2013.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Le Treut J, Sault MC, Lena H, Souquet PJ,
Vergnenegre A, Le Caer H, Berard H, Boffa S, Monnet I, Damotte D
and Chouaid C: Multicentre phase II study of cisplatin-etoposide
chemotherapy for advanced large-cell neuroendocrine lung carcinoma:
The GFPC 0302 study. Ann Oncol. 24:1548–1552. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Kasahara N, Wakuda K, Omori S, Nakashima
K, Ono A, Taira T, Kenmotsu H, Naito T, Murakami H, Mori K, et al:
Amrubicin monotherapy may be an effective second-line treatment for
patients with large-cell neuroendocrine carcinoma of high-grade
non-small-cell neuroendocrine carcinoma. Mol Clin Oncol. 6:718–722.
2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Derks JL, van Suylen RJ, Thunnissen E, den
Bakker MA, Groen HJ, Smit EF, Damhuis RA, van den Broek EC, Speel
EM and Dingemans AC: PALGA group: Chemotherapy for pulmonary large
cell neuroendocrine carcinomas: Does the regimen matter? Eur Respir
J. 49(pii: 1601838)2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Chen YB, Mu CY and Huang JA: Clinical
significance of programmed death-1 ligand-1 expression in patients
with non-small cell lung cancer: A 5-year-follow-up study. Tumori.
98:751–755. 2012.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Garon EB, Rizvi NA, Hui R, Leighl N,
Balmanoukian AS, Eder JP, Patnaik A, Aggarwal C, Gubens M, Horn L,
et al: Pembrolizumab for the treatment of non-small-cell lung
cancer. N Engl J Med. 372:2018–2028. 2015.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Tsuruoka K, Horinouchi H, Goto Y, Kanda S,
Fujiwara Y, Nokihara H, Yamamoto N, Asakura K, Nakagawa K, Sakurai
H, et al: PD-L1 expression in neuroendocrine tumors of the lung.
Lung Cancer. 108:115–120. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Wang VE, Urisman A, Albacker L, Ali S,
Miller V, Aggarwal R and Jablons D: Checkpoint inhibitor is active
against large cell neuroendocrine carcinoma with high tumor
mutation burden. J Immunother Cancer. 5(75)2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Mauclet C, Duplaquet F, Pirard L, Rondelet
B, Dupont M, Pop-Stanciu C, Vander Borght T, Remmelink M, D'Haene
N, Lambin S, et al: Complete tumor response of a locally advanced
lung large-cell neuroendocrine carcinoma after palliative thoracic
radiotherapy and immunotherapy with nivolumab. Lung Cancer.
128:53–56. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
McLaughlin J, Han G, Schalper KA,
Carvajal-Hausdorf D, Pelekanou V, Rehman J, Velcheti V, Herbst R,
LoRusso P and Rimm DL: Quantitative assessment of the heterogeneity
of PD-L1 expression in non-small-cell lung cancer. JAMA Oncol.
2:46–54. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Munari E, Zamboni G, Marconi M, Sommaggio
M, Brunelli M, Martignoni G, Netto GJ, Moretta F, Mingari MC,
Salgarello M, et al: PD-L1 expression heterogeneity in non-small
cell lung cancer: Evaluation of small biopsies reliability.
Oncotarget. 8:90123–90131. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Sabari JK, Julian RA, Ni A, Halpenny D,
Hellman MD, Drilon AE, Li BT, Poirier JT, Rudin CM and Rekhtman N:
Outcomes of advanced pulmonary large cell neuroendocrine carcinoma
stratified by RB1 loss, SLFN11 expression, and tumor mutational
burden. J Clin Oncol. 36 (15 Suppl)(e20568)2018.
|
|
19
|
Grangeon M, Tomasini P, Chaleat S, Jeanson
A, Souquet-Bressand M, Khobta N, Bermudez J, Trigui Y, Greillier L,
Blanchon M, et al: Association between immune-related adverse
events and efficacy of immune checkpoint inhibitors in
non-small-cell lung cancer. Clin Lung Cancer. 20:201–207.
2019.PubMed/NCBI View Article : Google Scholar
|