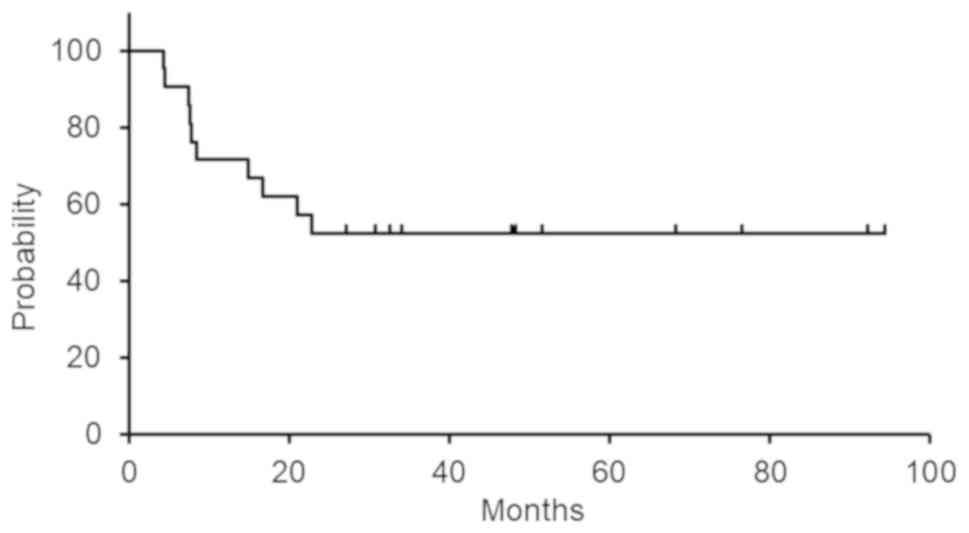
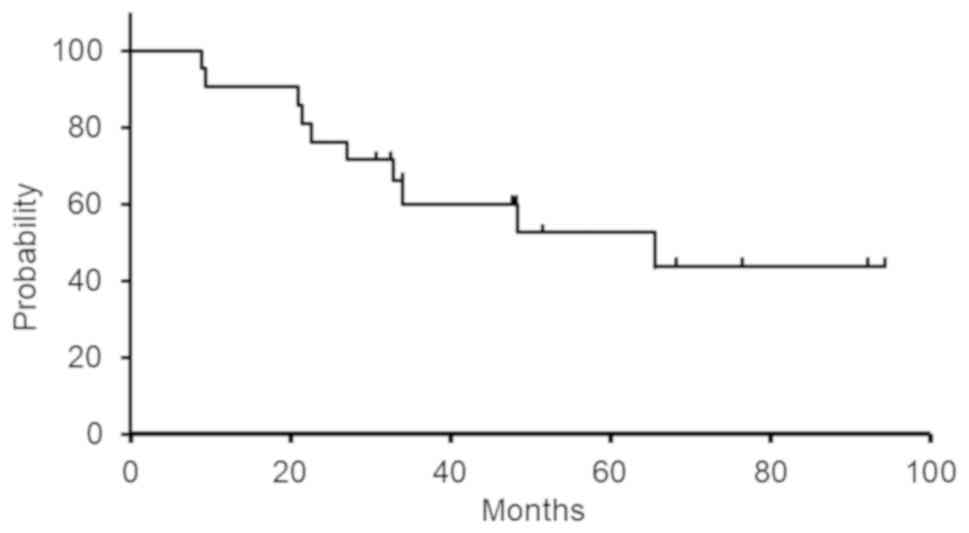
|
1
|
Forastiere AA, Goepfert H, Maor M, Pajak
TF, Weber R, Morrison W, Glisson B, Trotti A, Ridge JA, Chao C, et
al: Concurrent chemotherapy and radiotherapy for organ preservation
in advanced laryngeal cancer. N Engl J Med. 349:2091–2098.
2003.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Adelstein DJ, Li Y, Adams GL, Wagner H Jr,
Kish JA, Ensley JF, Schuller DE and Forastiere AA: An intergroup
phase III comparison of standard radiation therapy and two
schedules of concurrent chemoradiotherapy in patients with
unresectable squamous cell head and neck cancer. J Clin Oncol.
21:92–98. 2003.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Pignon JP, Bourhis J, Domenge C and
Designé L: Chemotherapy added to locoregional treatment for head
and neck squamous-cell carcinoma: Three meta-analyses of updated
individual data. MACH-NC collaborative group. Meta-analysis of
chemotherapy on head and neck cancer. Lancet. 355:949–955.
2000.PubMed/NCBI
|
|
4
|
Pignon JP, le Maître A, Maillard E and
Bourhis J: MACH-NC Collaborative Group. Meta-analysis of
chemotherapy in head and neck cancer (MACH-NC): An update on 93
randomised trials and 17,346 patients. Radiother Oncol. 92:4–14.
2009.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zhang L, Jiang N, Shi Y, Li S, Wang P and
Zhao Y: Induction chemotherapy with concurrent chemoradiotherapy
versus concurrent chemoradiotherapy for locally advanced squamous
cell carcinoma of head and neck: A meta-analysis. Sci Rep.
5(10798)2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Haddad RI, Posner M, Hitt R, Cohen EEW,
Schulten J, Lefebvre JL and Vermorken JB: Induction chemotherapy in
locally advanced squamous cell carcinoma of the head and neck:
Role, controversy, and future directions. Ann Oncol. 29:1130–1140.
2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Budach W, Bölke E, Kammers K, Gerber PA,
Orth K, Gripp S and Matuschek C: Induction chemotherapy followed by
concurrent radio-chemotherapy versus concurrent radio-chemotherapy
alone as treatment of locally advanced squamous cell carcinoma of
the head and neck (HNSCC): A meta-analysis of randomized trials.
Radiother Oncol. 118:238–243. 2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Haddad R, O'Neill A, Rabinowits G, Tishler
R, Khuri F, Adkins D, Clark J, Sarlis N, Lorch J, Beitler JJ, et
al: Induction chemotherapy followed by concurrent chemoradiotherapy
(sequential chemoradiotherapy) versus concurrent chemoradiotherapy
alone in locally advanced head and neck cancer (PARADIGM): A
randomised phase 3 trial. Lancet Oncol. 14:257–264. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Vermorken JB, Remenar E, van Herpen C,
Gorlia T, Mesia R, Degardin M, Stewart JS, Jelic S, Betka J, Preiss
JH, et al: Cisplatin, fluorouracil, and docetaxel in unresectable
head and neck cancer. N Engl J Med. 357:1695–1704. 2007.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Posner MR, Hershock DM, Blajman CR,
Mickiewicz E, Winquist E, Gorbounova V, Tjulandin S, Shin DM,
Cullen K, Ervin TJ, et al: Cisplatin and fluorouracil alone or with
docetaxel in head and neck cancer. N Engl J Med. 357:1705–1715.
2007.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Blanchard P, Bourhis J, Lacas B, Posner
MR, Vermorken JB, Cruz Hernandez JJ, Bourredjem A, Calais G,
Paccagnella A, Hitt R, et al: Taxane-cisplatin-fluorouracil as
induction chemotherapy in locally advanced head and neck cancers:
An individual patient data meta-analysis of the meta-analysis of
chemotherapy in head and neck cancer group. J Clin Oncol.
31:2854–2860. 2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Caballero M, Mackers P, Reig O, Buxo E,
Navarrete P, Blanch JL and Grau JJ: The role of audiometry prior to
high-dose cisplatin in patients with head and neck cancer.
Oncology. 93:75–82. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Mizuno T, Ishikawa K, Sato W, Koike T,
Kushida M, Miyagawa Y, Yamada K, Hirata S, Imai E and Noda Y: The
risk factors of severe acute kidney injury induced by cisplatin.
Oncology. 85:364–369. 2013.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Nguyen-Tan PF, Zhang Q, Ang KK, Weber RS,
Rosenthal DI, Soulieres D, Kim H, Silverman C, Raben A, Galloway
TJ, et al: Randomized phase III trial to test accelerated versus
standard fractionation in combination with concurrent cisplatin for
head and neck carcinomas in the radiation therapy oncology group
0129 trial: Long-term report of efficacy and toxicity. J Clin
Oncol. 32:3858–3866. 2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Loong HH, Ma BB, Leung SF, Mo F, Hui EP,
Kam MK, Chan SL, Yu BK and Chan AT: Prognostic significance of the
total dose of cisplatin administered during concurrent
chemoradiotherapy in patients with locoregionally advanced
nasopharyngeal carcinoma. Radiother Oncol. 104:300–304.
2012.PubMed/NCBI View Article : Google Scholar
|
|
16
|
NCCN guidelines with NCCN evidence
blocks™-head and neck cancers version 1, 2020.
|
|
17
|
Lefebvre JL, Pointreau Y, Rolland F,
Alfonsi M, Baudoux A, Sire C, de Raucourt D, Malard O, Degardin M,
Tuchais C, et al: Induction chemotherapy followed by either
chemoradiotherapy or bioradiotherapy for larynx preservation: The
TREMPLIN randomized phase II study. J Clin Oncol. 31:853–859.
2013.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Hitt R, Grau JJ, López-Pousa A, Berrocal
A, García-Girón C, Irigoyen A, Sastre J, Martínez-Trufero J,
Brandariz Castelo JA, Verger E, et al: A randomized phase III trial
comparing induction chemotherapy followed by chemoradiotherapy
versus chemoradiotherapy alone as treatment of unresectable head
and neck cancer. Ann Oncol. 25:216–225. 2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Adelstein DJ, Moon J, Hanna E, Giri PG,
Mills GM, Wolf GT and Urba SG: Docetaxel, cisplatin, and
fluorouracil induction chemotherapy followed by accelerated
fractionation/concomitant boost radiation and concurrent cisplatin
in patients with advanced squamous cell head and neck cancer. A
Southwest oncology group phase II trial (S0216). Head Neck.
32:221–228. 2010.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Driessen CML, Leijendeckers J, Snik A, van
der Graaf WTA, de Boer JP, Gelderblom H, Kaanders JHAM, Takes R and
van Herpen CML: Ototoxicity in locally advanced head and neck
cancer patients treated with induction chemotherapy followed by
intermediate or high-dose cisplatin-based chemoradiotherapy. Head
Neck. 41:488–494. 2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Lieberthal W, Triaca V and Levine J:
Mechanisms of death induced by cisplatin in proximal tubular
epithelial cells: Apoptosis vs. necrosis. Am J Phys. 270:F700–F708.
1996.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Lee RH, Song JM, Park MY, Kang SK, Kim YK
and Jung JS: Cisplatin-induced apoptosis by translocation of
endogenous Bax in mouse collecting duct cells. Biochem Pharmacol.
62:1013–1023. 2001.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Ozkok A and Edelstein CL: Pathophysiology
of cisplatin-induced acute kidney injury. Biomed Res Int.
2014(967826)2014.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Volarevic V, Djokovic B, Jankovic MG,
Harrell CR, Fellabaum C, Djonov V and Arsenijevic N: Molecular
mechanisms of cisplatin-induced nephrotoxicity: A balance on the
knife edge between renoprotection and tumor toxicity. J Biomed Sci.
26(25)2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Park SB, Goldstein D, Krishnan AV, Lin CS,
Friedlander ML, Cassidy J, Koltzenburg M and Kiernan MC:
Chemotherapy-induced peripheral neurotoxicity: a critical analysis.
CA Cancer J Clin. 63:419–437. 2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Oldenburg J, Fosså S and Dahl A: Scale for
chemotherapy-induced long-term neurotoxicity (SCIN): Psychometrics,
validation, and findings in a large sample of testicular cancer
survivors. Qual Life Res. 15:791–800. 2006.PubMed/NCBI View Article : Google Scholar
|
|
27
|
LoMonaco M, Milone M, Batocchi AP, Padua
L, Restuccia D and Tonali P: Cisplatin neuropathy: Clinical course
and neurophysiological findings. J Neurol. 239:199–204.
1992.PubMed/NCBI View Article : Google Scholar
|
|
28
|
van den Bent MJ, van Putten WL, Hilkens
PH, de Wit R and van der Burg ME: Retreatment with dose-dense
weekly cisplatin after previous cisplatin chemotherapy is not
complicated by significant neuro-toxicity. Eur J Cancer.
38:387–391. 2002.PubMed/NCBI View Article : Google Scholar
|
|
29
|
von Schlippe M, Fowler CJ and Harland SJ:
Cisplatin neurotoxicity in the treatment of metastatic germ cell
tumour: Time course and prognosis. Br J Cancer. 85:823–826.
2001.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Cavaletti G, Marzorati L, Bogliun G,
Colombo N, Marzola M, Pittelli MR and Tredici G: Cisplatin-induced
peripheral neurotoxicity is dependent on total-dose intensity and
single-dose intensity. Cancer. 69:203–207. 1992.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Brydøy M, Oldenburg J, Klepp O, Bremnes
RM, Wist EA, Wentzel-Larsen T, Hauge ER, Dahl O and Fosså SD:
Observational study of prevalence of long-term Raynaud-like
phenomena and neurological side effects in testicular cancer
survivors. J Natl Cancer Inst. 101:1682–1695. 2009.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Okano S, Enokida T, Onoe T, Ota Y, Motegi
A, Zenda S, Akimoto T and Tahara M: Induction TPF chemotherapy
followed by CRT with fractionated administration of cisplatin in
patients with unresectable locally advanced head and neck cancer.
Int J Clin Oncol. 24:789–797. 2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Hitt R, Mesia R, Grau JJ, Iglesias L, Del
Barco E, Lozano A, Trufero JM, Giron CG, Martin AL and Hernandez
JJC: Randomized phase III trial of induction chemotherapy (ICT)
with docetaxel-cisplatin-5fluorouracil (DCF) followed by
cisplatin-radiotherapy (CRT) or cetuximab-radiotherapy (CetRT) in
patients (pts) with locally advanced unresectable head and neck
cancer (LAUHNC). J Clin Oncol. 34: (15 Suppl)(S6001)2016.
|