Introduction
Myxofibrosarcoma (MFS) is a common type of soft
tissue sarcoma that arises in the extremities of older patients.
The principal therapeutic strategy for the tumor involves surgery
with or without radiotherapy for localized disease, and palliative
chemotherapy for metastatic disease (1,2).
It is currently estimated that ~60-70% of patients
with MFS survive >5 years, while 35% of patients with high-grade
tumors are expected to develop distant metastasis. Numerous studies
have demonstrated that both anti-angiogenic therapies and
immunotherapies improved the survival rate of patients with
metastatic sarcoma (3-5).
The present report presented a case of a 67-year-old
man with recurrent metastatic MFS, who was prescribed a combination
of the anti-angiogenic drug bevacizumab and the immunotherapy drug
nivolumab. The patient achieved a partial response for >29
months and sustained benefit. This retrospective case study was
approved by the institution review board of The People's Liberation
Army No. 904 Hospital and the requirement for informed consent was
waived.
Case report
The current case report presents a 67-year-old man
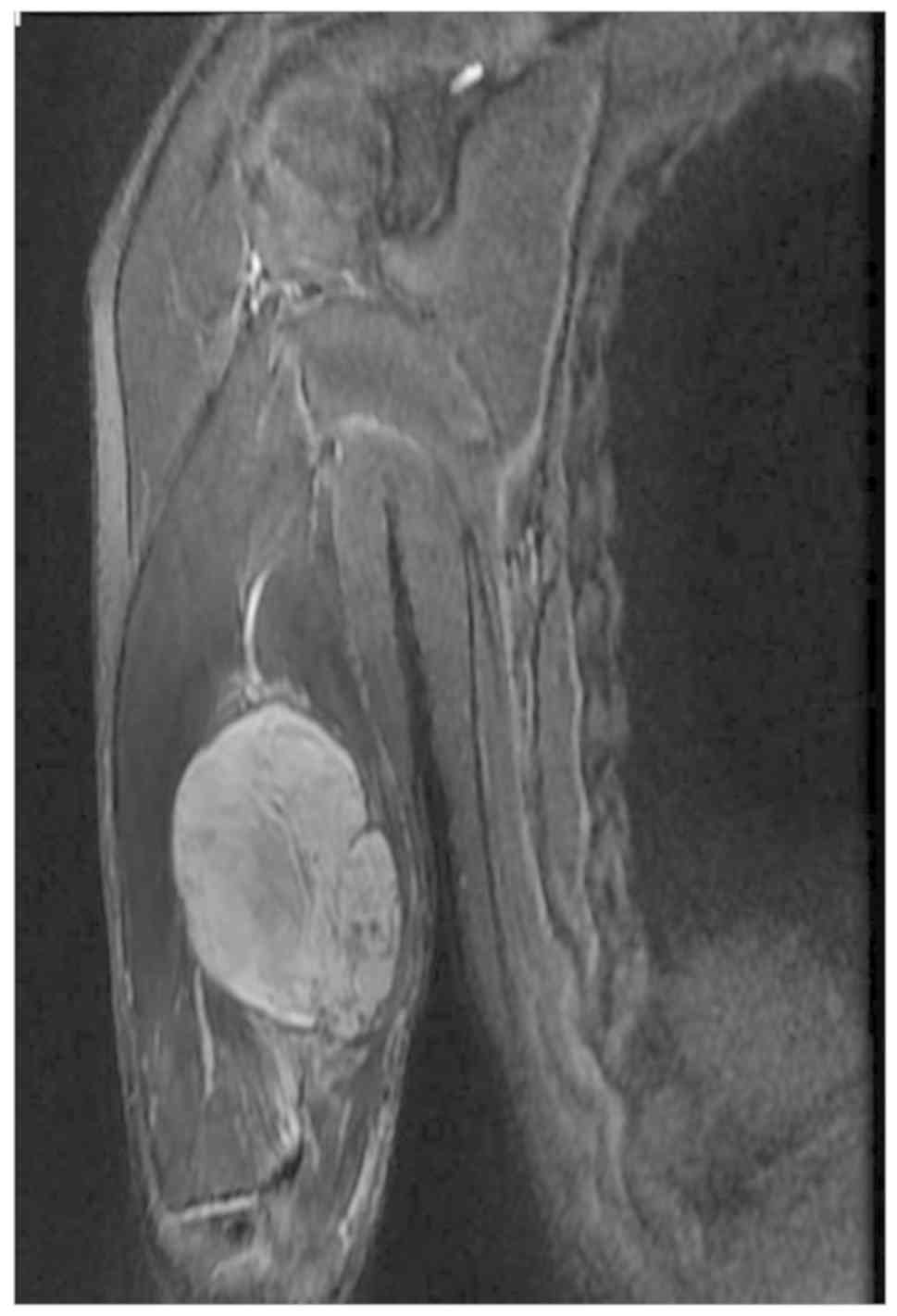
diagnosed with localized MFS in the triceps brachii in the right
upper arm (Fig. 1), who underwent
surgical resection at the resection site in June 2015. The
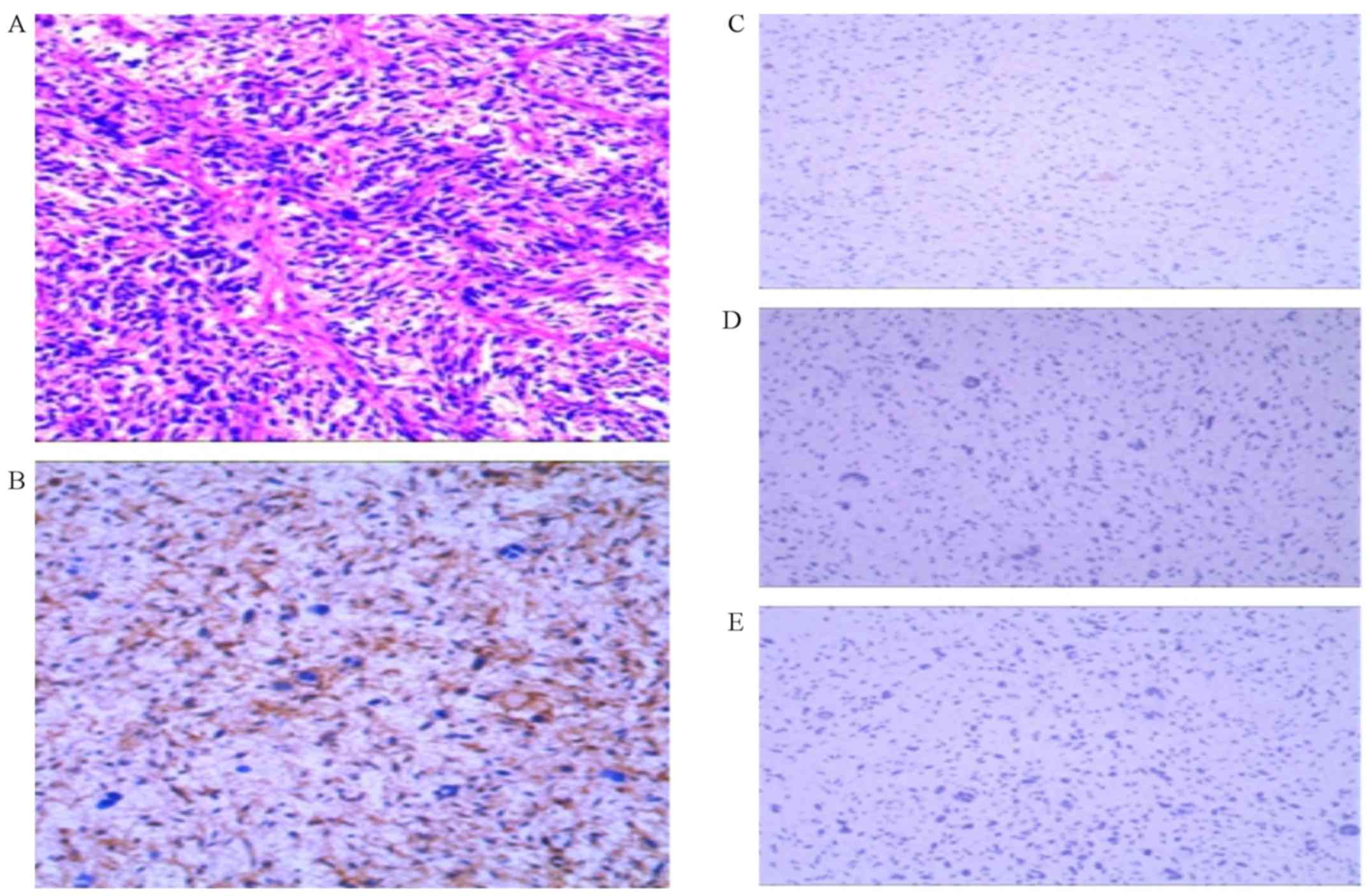
postoperative pathology revealed MFS of the right upper arm and the
following major immunohistochemical markers: CD34(+),
CD99(-), DES(-), NSE(-), SMA
(-), S100 (-) and Ki-67 (10%+)
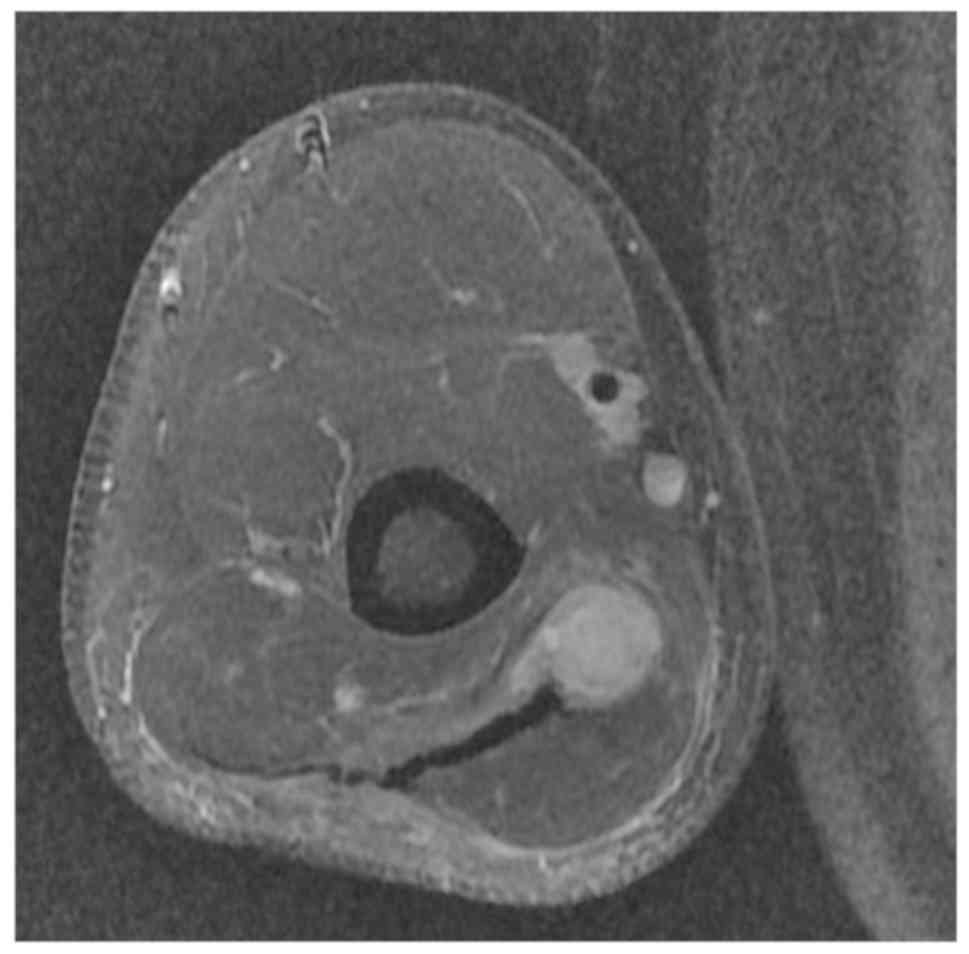
(Fig. 2). However, a residual mass
remained following the operation (Fig.
3). The first-line therapeutic strategy included surgical
resection and radiotherapy, as chemotherapy was refused due to the
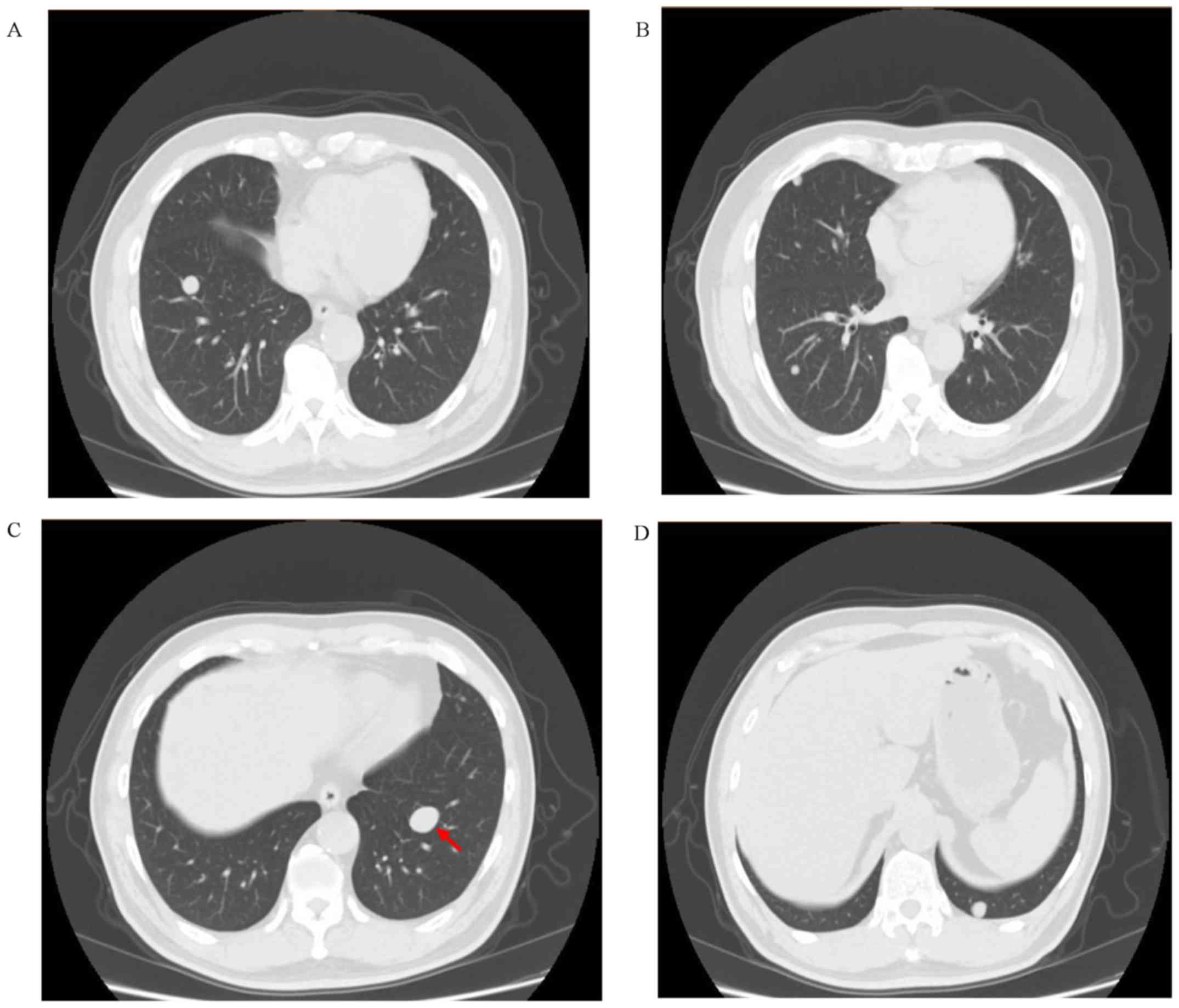
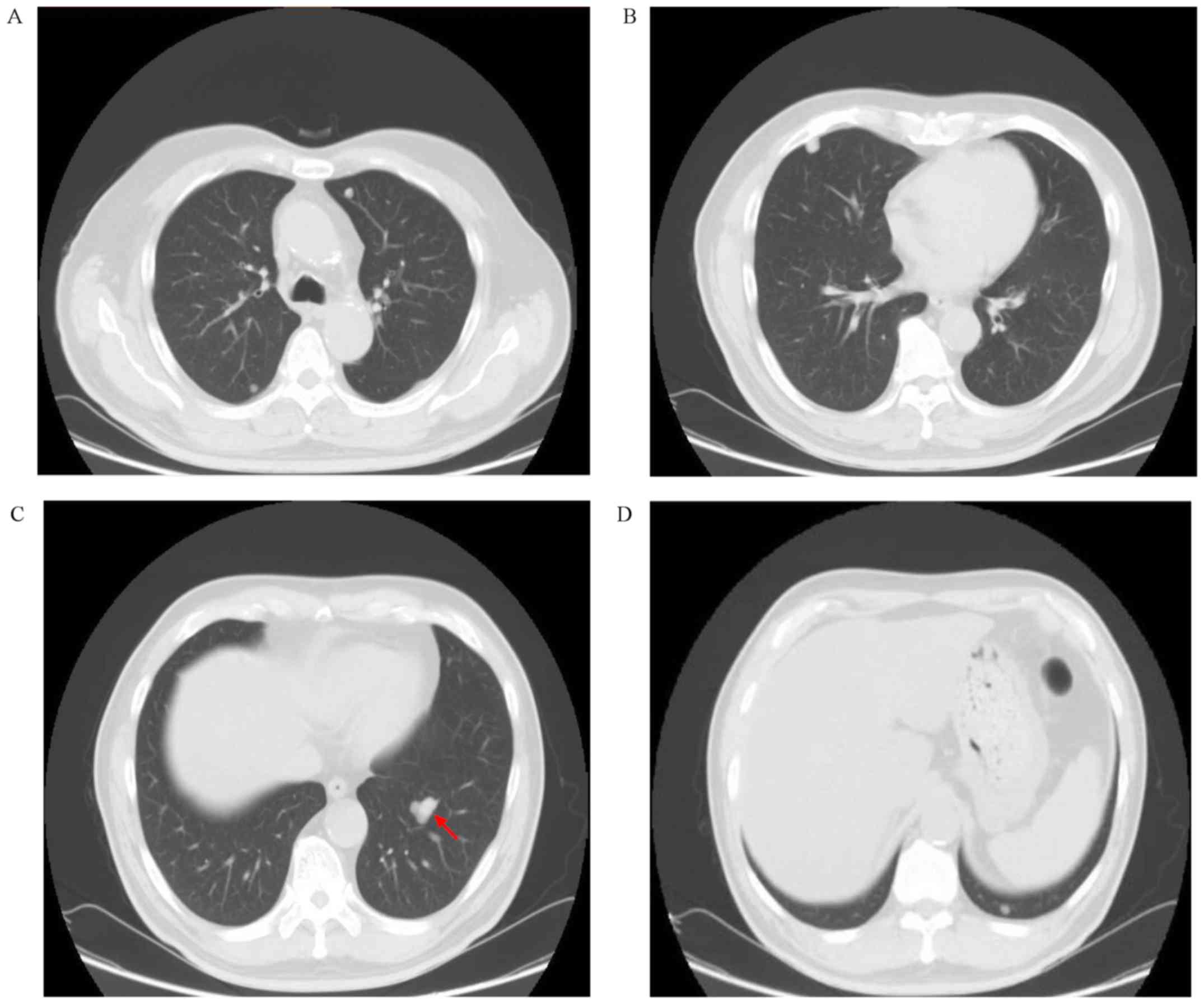
frequently occurring side effects. On February 10 2017, the CT scan
results revealed multiple lung metastases; the main lesion of the
left lung was ~21.10x14.65 mm in size (Fig. 4). The gene status of the patient was
analyzed using next-generation sequencing (NGS, whole genome
squeencing, Illumina Hiseq) and the following results were
obtained: i) A tumor mutational burden (TMB) of 6.7 mutations/MB;
ii) the absence of microsatellite instability (MSI); and iii)
negative PD-L1 expression (Panoramic gene test of Shihe 1; February
25 2017; internal sample number: b17021847377 Ge, p17021847378 Ge,
f17021847379 Ge). As chemotherapy was refused, although the TMB was
not high, nivolumab, a PD-1 inhibitor, was prescribed (3 mg/kg;
every 14 days; Hong Kong Yanghe Hospital). The treatment lasted for
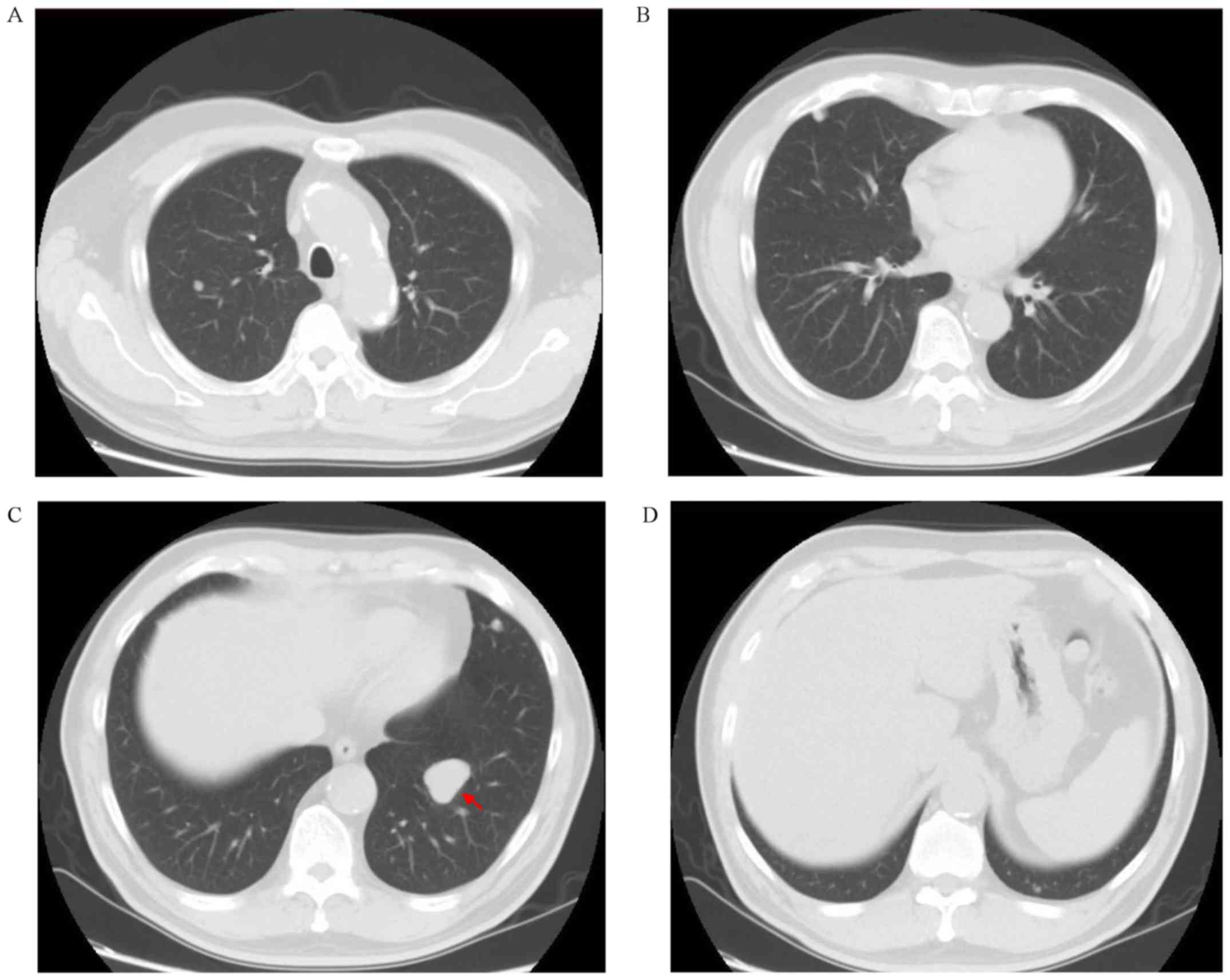
18 cycles between March and November 2017. On September 6 2017, the
CT scan results revealed that the main lesion in the left lung was
~18.96x16.64 mm in size (Fig. 5),
which suggested that the disease was in partial remission and the
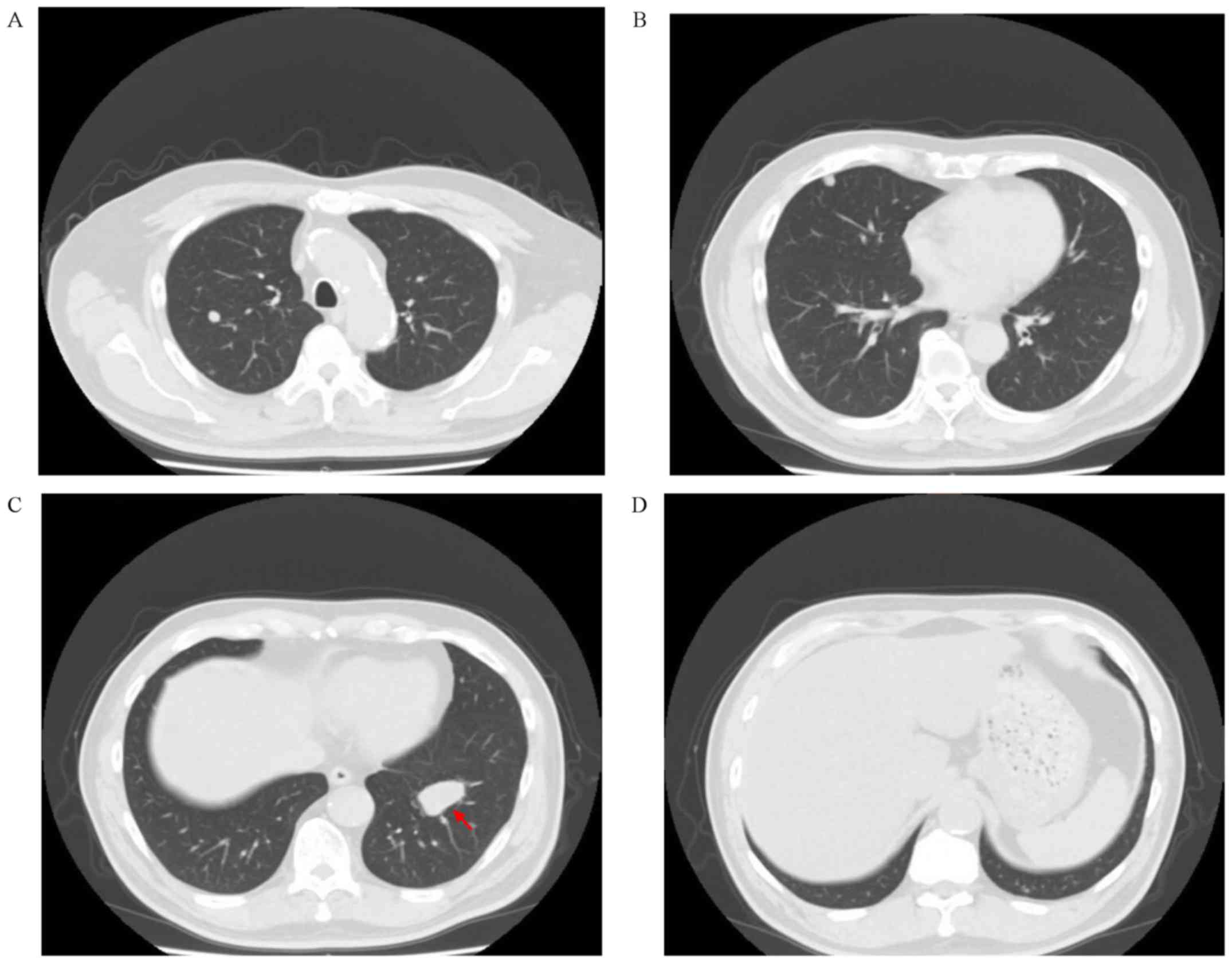
treatment was effective. On November 20 2017, the CT scan results
indicated that the right lung metastasis site was locally enlarged
(Fig. 6), and the main focus
was~29.14x26.01 mm in size, which according to the iRECICT
standards indicated that the disease had progressed. Local
radiotherapy (500 cGy; x7 times; Cancer Prevention and Treatment
Center of Sun Yat Sen University) was subsequently used to remove
the enlarged primary lesion. From December 28 2017, nivolumab (3
mg/kg; every 14 days; Hong Kong Yanghe Hospital; domestic purchase
from August 28 2018) combined with bevacizumab (7.5 mg/kg; every 14
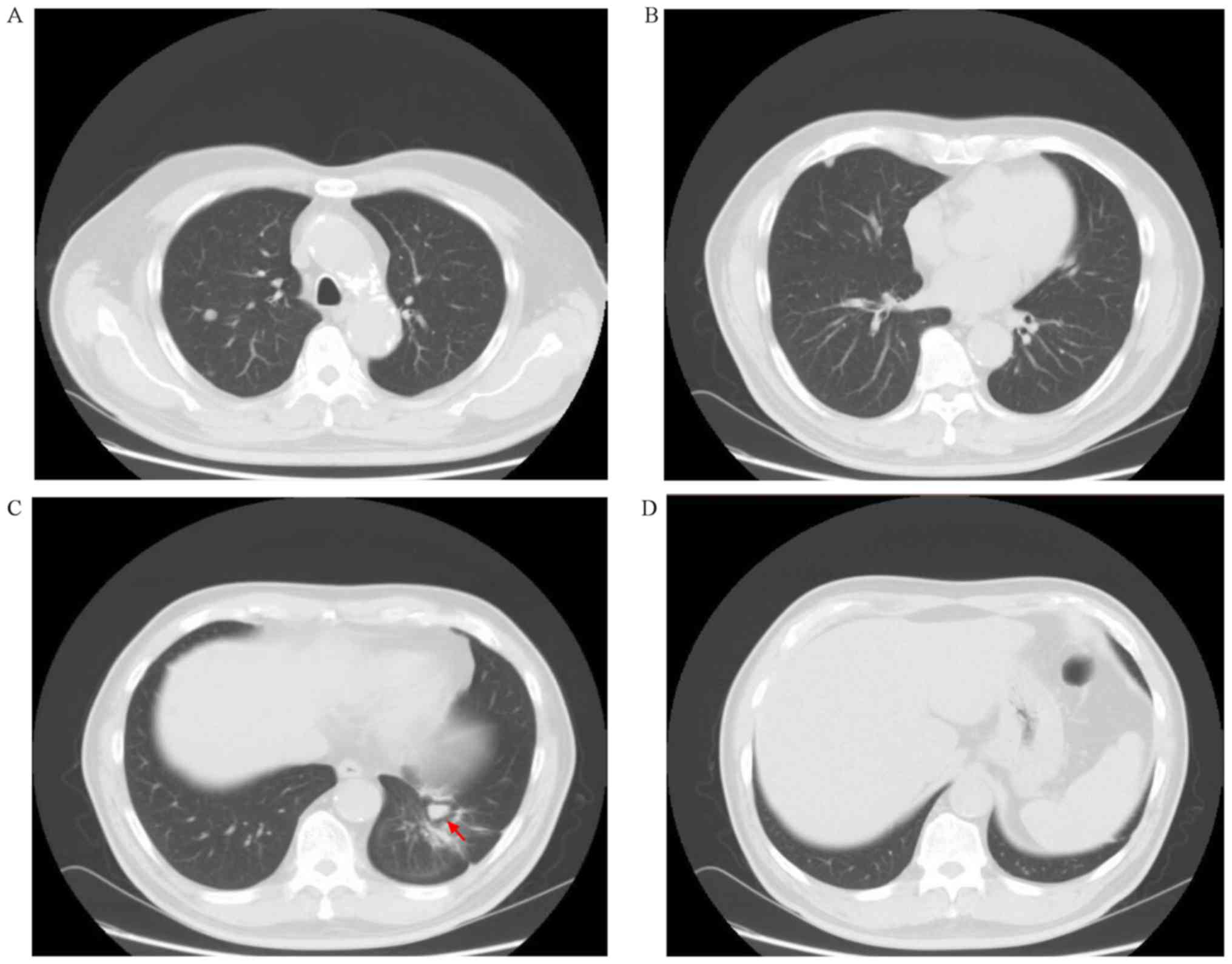
days) was prescribed. At the end of the follow-up period, the
remission time of the combined treatment was 16 months, and the
primary lesion was 14.86x11.1 mm in size, which was ~50% smaller
compared with that achieved with either agent alone before the
combination treatment. Notably, several of the foci had completely
disappeared (Figs. 7 and 8). The total survival time was >29
months and a continuous benefit was obtained from the treatment.
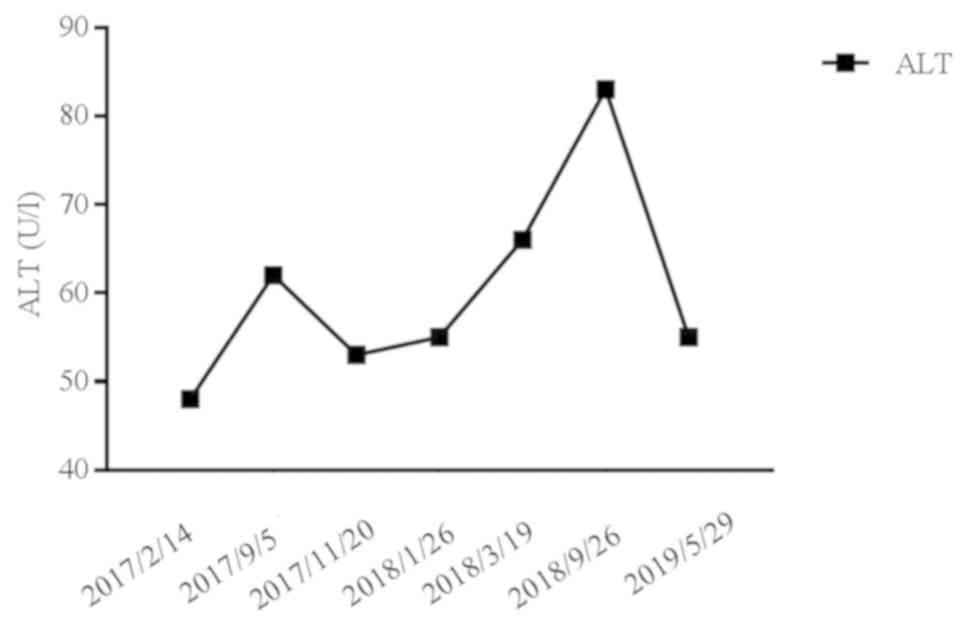
The predominant side effects of the treatment included fatigue,
elevated ALT levels, hypertension and proteinuria (Fig. 9; Table
I).
 | Table IManagement of adverse events during
treatment. |
Table I
Management of adverse events during
treatment.
| Events | Grade | Measures |
|---|
| Fatigue | 1 | Observation |
| ALT | 1 | Diammonium
glycyrrhizinate |
| Hypertension | 2 | Antihypertensive
drugs |
| Proteinuria | 1 | Observation |
Discussion
MFS is one of the common types of soft tissue
sarcoma, which occurs frequently in the extremities and
subcutaneous tissues of the middle-aged and elderly population. The
primary therapeutic strategies used to treat the disease include
surgery, radiotherapy, chemotherapy and targeted therapy (1,2). For
recurrent and metastatic MFS, the first-line chemotherapeutic
strategy involves anthracycline and ifosfamide, and the second-line
treatment includes gemcitabine and paclitaxel, of which the
curative effect was discovered to be significantly reduced compared
with the first-line treatment options (6). Trabectedin, an antineoplastic drug
isolated from Ecteinascidia turbinata, a sea squirt of the
Caribbean Sea, is currently the only second-line drug approved by
the European Union for advanced soft tissue sarcoma following the
failure of chemotherapy regimens based on anthracycline and
ifosfamide (6). The median survival
of the majority of the patients with advanced soft tissue sarcoma
is usually <1 year, with only 10% of patients surviving >5
years (7).
Pazopanib is a second generation oral multitarget
tyrosine kinase (TKI) inhibitor that targets VEGFR, FDGFR and
C-KIT, which is associated with the occurrence and development of
multiple types of tumor. A phase II clinical study has previously
demonstrated its efficacy in soft tissue sarcoma (8), while the subsequent phase III clinical
study (PALETTE) indicated that pazopanib improved the disease
progression free survival (PFS), but it was unable to improve the
overall survival (OS) (9). In 2012,
the US FDA approved the drug for the treatment of
anthracycline-based non-fat soft tissue sarcoma, which represents
the only oral drug approved by the US FDA for the treatment of
advanced soft tissue sarcoma, except for liposarcoma (10,11).
Anlotinib is a small molecular TKI compound with independent
intellectual property rights manufactured in China, of which the
targets are the same as Pazopanib. In 2016, ASCO reported the
efficacy of the single agent anlotinib in advanced soft tissue
sarcoma. In 2018, ASCO produced the results of its phase III study
(ALTER0203); compared with the placebo group, the PFS of the
anlotinib group was 4.8 months and the HR was 0.33. In addition, in
the fibrosarcoma subgroup, the PFS of anlotinib group was 4.27
months, while the placebo group was only 1.43 months (12).
VEGF is an important inducer of angiogenesis, which
has been revealed to promote the angiogenesis and proliferation of
numerous types of tumor. Bevacizumab is a completely humanized
anti-VEGF monoclonal antibody, which can specifically bind to VEGF,
prevent the binding of VEGF and inactivate VEGFR-2, thus inhibiting
the mitosis of endothelial cells and blocking the biological
effects of VEGF (7). A
retrospective study demonstrated that the sole treatment with
bevacizumab inhibited metastatic or unresectable angiosarcoma and
epithelioid hemangioendothelioma, of which 48% of the patients were
stable and 15% were in partial remission (6). Another previous study investigating
the combined treatment of bevacizumab and doxorubicin revealed that
its effective rate was 12%, demonstrating a median OS of 16 months
(5). In addition, in other soft
tissue sarcomas, the combination of bevacizumab and
chemotherapeutics, such as docetaxel, gemcitabine and temozolomide,
also promoted several curative effects, which subsequently
suggested a role for bevacizumab in the treatment of advanced soft
tissue tumors (6).
Recently, accumulating research on PD-1 and PD-L1
inhibitors has demonstrated their efficacy in various types of
malignant tumor, including soft tissue tumors (3,4). A
phase II study (SARC028) also found that of those patients with
recurrent and metastatic soft tissue sarcoma that received
pembrolizumab, a PD-1 blocker, 1/40 patients achieved complete
remission, 6 cases were in partial remission and 15 cases were in
stable condition after 17.8 months of follow-up; these results
suggested that PD-1 blockade immunotherapy for recurrent and
metastatic soft tissue sarcomas may have an improved efficacy
(4). Nivolumab is another PD-1
blocker, which has been the primary focus of several case reports
and phase II clinical studies for the treatment of soft tissue
sarcoma. It was previously hypothesized that nivolumab may serve a
definite role in the treatment of soft tissue sarcoma (13-16).
The present case report presented a case of MFS.
Following the diagnosis of lung metastasis, nivolumab was
prescribed due to the patient refusing palliative chemotherapy, and
the disease remained stable during the 18 cycles of treatment.
After 8 months, several of the lung metastases sites were enlarged,
indicating that the disease had advanced according to the iRECIST
standards. Following local radiotherapy to remove the enlarged
lesions, the combined treatment of nivolumab and bevacizumab was
administered. The patient obtained continuous disease control and
long-term survival benefits. Although several studies have
concluded that radiotherapy combined with immunotherapy exerted an
abscopal effect, the predominant principle of radiotherapy was
considered to be its ability to promote an in situ tumor
vaccine effect and reconstruct the immune microenvironment, amongst
other functions. Generally, single high-dose radiotherapy and
high-dose fractionated radiotherapy have been discovered to induce
the abscopal effect; however, whether conventional radiotherapy may
also induce the abscopal effect has not been studied in detail,
with the majority of the data stating that high-dose radiotherapy
is the predominant means to induce the abscopal effect (17). It is suggested that radiotherapy
itself may also promote immunosuppressive effects and affect the
therapeutic effects (18). In
addition, in a previous study, due to the exposure to external
antigen environments, the immunogenicity of lung tissue was
reported to be unique, and the number of infiltrating lymphocytes
in the tumors was decreased compared with in the gastrointestinal
and breast tumors, which was suggested to weaken the immune
response induced by radiotherapy (18). More importantly, numerous studies
have confirmed that the simultaneous combination of immunotherapy
with radiotherapy for PD-1/PD-L1/CTLA-4 inhibitors was recommended
to achieve the maximum effect, while immunotherapy followed by
radiotherapy or radiotherapy followed by immunotherapy reduced the
effect (19). However, in light of
the results of the present case study, further investigations
should focus on determining the efficacy of the combination with
bevacizumab. For example, the combination of bevacizumab with
PD-1/PD-L1/CTLA-4 inhibitors has previously demonstrated efficacy
in clinical studies of other types of tumor, such as lung cancer.
The principal mechanism of action of bevacizumab was discovered to
be its ability to promote antitumor neovascularization, tumor
vascularization and the improvement of the tumor microenvironment
to render a ‘hot tumor’, which increased the local tumor lymphocyte
infiltration and subsequently increased the efficacy of the
PD-1/PD-L1/CTLA-4 inhibitors (20-23).
In addition, a phase II study (APFAO; NCT03359018) revealed that
the PD-1 inhibitor camrelizumab combined with the anti-angiogenic
drug apatinib improved the PFS and OS of advanced osteosarcoma
progressing following chemotherapy [https://clinicaltrials.gov/ct2/show/NCT03359018].
In the present case report, the NGS results revealed
a TMB of 6.7 mutations/MB, the absence of MSI and negative PD-L1
expression, which also verified that the above indicators may not
be used as predictors of the efficacy of immunotherapy. Thus, based
on the improved efficacy of anlotinib following the failure of
treatments such as nivolumab combined with bevacizumab, anlotinib
may be a good choice; however, further clinical research is
required.
In conclusion, the treatment of the patient
discussed in the current case study revealed the therapeutic
effects of nivolumab, a PD-1 blocker, in combination with
bevacizumab; the patient obtained continuous remission and
long-term survival benefits. The primary side effects included
fatigue, abnormal ALT levels, hypertension and proteinuria, which
were all safe and controllable. Thus, further phase III clinical
studies are required to provide additional data to support the
treatment of advanced MFS with a combination of nivolumab and
bevacizumab.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during the current
study are included in this published article or are available from
the corresponding author upon reasonable request.
Authors' contributions
LS, DP and RZ were involved in conceiving and
designing the study. LS drafted and wrote the manuscript. DP and RZ
provided advice on the experimental design, interpreted the results
and critically revised the manuscript. All authors have read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
The access of the database for the purpose of this
study was approved by the Ethics Committee of The People's
Liberation Army No. 904 Hospital (Jiangsu, China). As the hospital
to which the patient was admitted is a teaching hospital, all
patients admitted to our hospital signed a written consent in
Chinese by which they agree that their medical data can be used in
scientific studies.
Patient consent for publication
The participation in the study was approved by the
patient and he gave his approval for the patient information
presented herein to be published.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Angervall L, Kindblom LG and Merk C:
Ayxofibrosarcoma: A study of 30 cases. Acta Pathol Microbiol Scand
A. 85A:127–140. 1977.PubMed/NCBI
|
|
2
|
Fletcher CD, Bridge JA, Hogendoorn PC, et
al: WHO classifitation of tumours of soft tissue and bone. Lyon,
IARC Press, 93-94, 2013.
|
|
3
|
Uehara T, Fujiwara T, Takeda K, Kunisada
T, Ozaki T and Udono H: Immunotherapy for bone and soft tissue
sarcomas. Biomed Res Int. 2015(820813)2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Tawbi HA, Burgess M, Bolejack V, Van Tine
BA, Schuetze SM, Hu J, D'Angelo S, Attia S, Riedel RF, Priebat DA,
et al: Pembrolizumab in advanced soft-tissue sarcoma and bone
sarcoma (SARC028): A multicentre, two-cohort,
single-arm,open-label, phase 2 trial. Lancet Oncol. 18:1493–1501.
2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
D'Adamo DR, Anderson SE, Albritton K,
Yamada J, Riedel E, Scheu K, Schwartz GK, Chen H and Maki RG: Phase
II study of doxorubicin and bevacizumab for patients with
metastatic soft-tissue sarcomas. J Clin Oncol. 23:7135–7142.
2005.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ruihua Xu: Medical Oncology. Bei Jing:
People's Medical publishing house, 549-565, 2014 (In Chinese).
|
|
7
|
Yin QL, Wu D and Yu YY: Progression in
anti-angiogenesis agents for soft tissue sarcoma. Chinese Clinical
Oncol. 22:839–844. 2017.(In Chinese).
|
|
8
|
Sleijfer S, Ray-Coquard I, Papai Z, Le
Cesne A, Scurr M, Schöffski P, Collin F, Pandite L, Marreaud S, De
Brauwer A, et al: Pazopanib, a muhikinase angiogenesis inhibitor,
in patients with relapsed or refractory advanced soft tissue
sarcoma: A phaseIIstudy from the European organisatiou for research
and treatment of eaneer-sofc tissue and bone sarcoma group (EORTC
study 62043). J Clin Oncol. 27:3126–3132. 2009.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Van der Graaf WT, Blay JY, Chawla SP, Kim
DW, Bui-Nguyen B, Casali PG, Schöffski P, Aglietta M, Staddon AP,
Beppu Y, et al: Pazopanib for metastatic soft-tissue sarcoma
(PALETTE): A randomised, double-blind, placebo-controlled phase 3
trial. Lancet. 379:1879–1886. 2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
In GK, Hu JS and Tseng WW: Treatment of
abvanced, metastatic soft tissue sarcoma: Latest evidence and
clinical considerations. Ther Adv Med Oncol. 9:533–550.
2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Sarcoma Committee of Chinese Anti-Cancer
Association; Chinese Society of Clinical Oncology. Chinese expert
consensus on diagnosis and treatment of soft tissue sarcomas
(Version 2015). Zhonghua Zhong Liu Za Zhi. 38:310–320.
2016.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
12
|
Chi Y, Fang Z, Hong X, Yao Y, Sun P, Wang
G, Du F, Sun Y, Wu Q, Qu G, et al: Safety and efficacy of
anlotinib, a multikinase angiogenesis inhibitor, in patients with
refractory metastatic soft-tissue sarcoma. Clin Cancer Res 24:
5233-5238.
|
|
13
|
Paoluzzi L, Cacario A, Ghesani M,
Karambelkar A, Rapkiewicz A, Weber J and Rosen G: Response to
anti-PD1 therapy with nivolumab in metastatic sarcomas. Clin
Sarcoma Res. 6(24)2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ben-Ami E, Barysauskas CM, Solomon S,
Tahlil K, Malley R, Hohos M, Polson K, Loucks M, Severgnini M,
Patel T, et al: Immunotherapy with single agent nivolumab foe
advanced leiomyosarcoma of the uterus: Results of a phase 2 study.
Cancer. 123:3285–3290. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
D'Angelo SP, Mahoaey MR, VanTine BA,
Atkins J, Milhem MM, Jahagirdar BN, Antonescu CR, Horvath E, Tap
WD, Schwartz GK and Streicher H: Nivolumab with or without
ipilimumab treatment for metastatic sarcoma (Alliance A091401): Two
open-label non-comparative, randomized,phase 2 trials. Lancet
oncol. 19:416–426. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wagner MJ, Ricciotti RW, Mantilla J,
Loggers ET, Pollack SM and Cranmer LD: Response to PD1 inhibition
in conventional chondrosarcoma. J Immunother Cance.
6(94)2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Linpeng Zhen, Luping Zhang, Zihan Xu, et
al: Radiation therapy combined with immunological checkpoint
inhibitors and distant effects. Chin J Lung Dis (Electronic
Edition). 11:227–230. 2018.(In Chinese).
|
|
18
|
Zhouguang Hui, Meng Yuan and Yu Men:
Application mechanism of radiotherapy combined with immune
checkpiont inhibitors. Chin J Radiat Oncol. 27:875–879. 2018.(In
Chinese).
|
|
19
|
Wang Q and Li B: Research progress in
PD-1/PD-L1 monoclonal antibody combined with radiotherapy in
treatment of solid tumors. Chin J Radiat Oncol. 25:1375–1378.
2016.(In Chinese).
|
|
20
|
Peng X and Zhou Q: First-line combination
immunotherapy in advanced non-small cell lung cancer. Zhongguo Fei
Ai Za Zhi. 21:924–930. 2018.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
21
|
Zhang J, Zhang W, Qian Z, Gao B and Xiang
Y: Tumor vascular normalization improves cancer treatment efficacy.
China Oncology. 26:188–192. 2016.(In Chinese).
|
|
22
|
Xu Y, Zhang L and Jin L: Mechanism of
tumor tolerance mediated hy immune microenvironment and its
targeted therapy. J Int Oncol. 44:594–596. 2017.(In Chinese).
|
|
23
|
Zhang B and Yue H: The mechanisms of
immune microenvironment in cancer development and progression.
Modern Oncol. 23:862–864. 2015.(In Chinese).
|