Introduction
Introduction of immunotherapy with immune checkpoint
inhibitors (ICI) has changed the therapeutic landscape of advanced
head and neck squamous cell carcinoma (HNSCC), but only a small
subset of patients with HNSCC benefit from ICI, with an overall
response rate of 13-17% as a monotherapy (1-3).
Programmed death-ligand 1 (PD-L1) expression is currently used as a
predictive biomarker to select patients for anti-PD1 inhibitory
therapy. However, PD-L1 expression is a dynamic biomarker with
significant intratumoral heterogeneity and has a low predictive
value overall. Additionally, obtaining tissue for PD-L1 testing is
not always feasible. Identification of a reliable and accessible
clinical marker for optimal patient selection and early prediction
of response is an unmet need.
Lymphopenia has been suggested as a negative
predictive marker for ICI therapy response in several solid tumors
including HNSCC (4,5). Several studies have previously
assessed the predictive value of various peripheral lymphocyte
measures for ICI therapy response, including absolute or relative
lymphocyte count at the beginning of treatment, on-treatment, or
dynamic changes during treatment. High baseline ALC and increase in
ALC during the treatment were associated with better response to
anti-PD-1/PD-L1 therapy (4,6,7).
Similarly, low NLR and decrease in NLR with anti-PD-1 blockade were
associated with improved outcomes in various solid tumors (5,8,9). The
measure of peripheral lymphocyte with the best predictive value in
HNSCC is unknown. In this study, we examined comparative predictive
values of multiple lymphocyte level measures for anti-PD-1 ICI
therapy in patients with advanced HNSCC.
Materials and methods
Patient selection
We retrospectively collected clinicopathologic data
on patients diagnosed with advanced HNSCC who have received at
least two doses of anti-PD-1 ICI therapy at Massachusetts General
Hospital between 2015 and 2020. Patients who had underlying
hematologic malignancy, were taking systemic corticosteroids at the
time of therapy, were on antibiotic therapy for an active
infection, and/or whose laboratory values were not available were
excluded from the study.
Variables and follow up
Demographic data, clinicopathologic data, medical
and social history, treatment history, and treatment responses were
obtained from the patients' records. Baseline and week 6 absolute
lymphocyte count (ALC) and absolute neutrophil count (ANC) were
collected, and neutrophil-to-lymphocyte ratios (NLR) were
calculated. ALC and NLR changes (Δ) from baseline to week 6 were
measured.
Statistical analysis
Progression-free survival (PFS), defined as the time
elapsed between the initiation of ICI therapy and tumor progression
or death from any cause, was the primary clinical outcome measure.
Overall survival (OS) was defined as the time from the initiation
of ICI treatment to the date of death from any cause. The
Kaplan-Meier method was used to estimate PFS and OS, and the Cox
proportional hazards regression model was used to estimate hazard
ratios (HR) and confidence intervals (CI). Treatment response was
defined as unequivocal radiographic and/or clinical improvement on
ICI treatment, and was summarized using exact 95% CIs for each
subgroup. The multivariable models were adjusted for human
papilloma virus (HPV)-association, smoking history, Eastern
Cooperative Oncology Group (ECOG) performance status, and the line
of ICI therapy. All statistical tests were 2-sided, and a P value
of <0.05 was considered statistically significant. This study
was conducted under the IRB Protocol no. 2018P001456.
Results
Patient characteristics
Between the years 2015 and 2020, a total of 108
patients with advanced HNSCC that met the selection criteria for
this study were identified. The patients' baseline demographic and
clinicopathologic characteristics and treatment history are
summarized in Table I. The median
(range) age was 67 (26-92) years. Eighty-one point five percent of
the patients were male and 88.0% of the patients were white. The
oropharynx was the most common primary tumor site (31.5%) followed
by the oral cavity (26.9%). Anti-PD-1 therapy was given as a
first-line therapy in 47.2% of the patients. Thirty-one (28.7%)
cases were human papilloma virus (HPV) associated (Table I). Median baseline ALC, ANC, and NLR
were 790 cells/µl, 4,780 cells/µl, and 6.7, respectively. Week 6
ALC, ANC, and NLR were 770 cells/µl, 4,670 cells/µl, and 6.2,
respectively. Median ΔALC and ΔNLR were -15 and 6.1,
respectively.
 | Table IBaseline demographic and
clinicopathologic characteristics and treatment history of the
total population (n=108). |
Table I
Baseline demographic and
clinicopathologic characteristics and treatment history of the
total population (n=108).
| Clinicopathologic
characteristics | Value |
|---|
| Median age, years
(range) | 67 (29-92) |
| Sex, n (%) | |
|
Male | 88 (81.5) |
|
Female | 20 (18.5) |
| Ethnicity, n (%) | |
|
White | 95 (88.0) |
|
Asian | 11 (10.2) |
|
Black | 2 (0.2) |
| Performance status
(ECOG), n (%) | |
|
0, 1 | 77 (71.3) |
|
2, 3, 4 | 31 (28.7) |
| Smoking, n (%) | |
|
≥10 PY | 53 (49.1) |
|
Never or
<10 PY | 55 (50.9) |
| Primary site, n
(%) | |
|
Oropharynx | 34 (31.5) |
|
Oral
cavity | 29 (26.9) |
|
Nasopharynx | 17 (15.7) |
|
Larynx/hypopharynx | 15 (13.9) |
|
Sinonasal | 9 (8.3) |
|
Unknown
primary | 4 (3.7) |
| HPV status, n
(%) | |
|
Positive | 31 (28.7) |
|
Unknown/Negative | 71 (72.3) |
| Treatment
history | |
|
Anti-PD-1
agent, n (%) | |
|
Pembrolizumab | 76 (63.7) |
|
Nivolumab | 32 (35.4) |
|
Line of ICI
therapy, n (%) | |
|
First
line | 51 (47.2) |
|
≥ Second
line | 57 (52.8) |
Survival analysis
With a median follow-up of 11.7 months (range,
1.4-51.1), the median OS (mOS) for the total population was 18.4
months (95% CI, 14.5-22.3) and the median PFS (mPFS) was 4.1 months
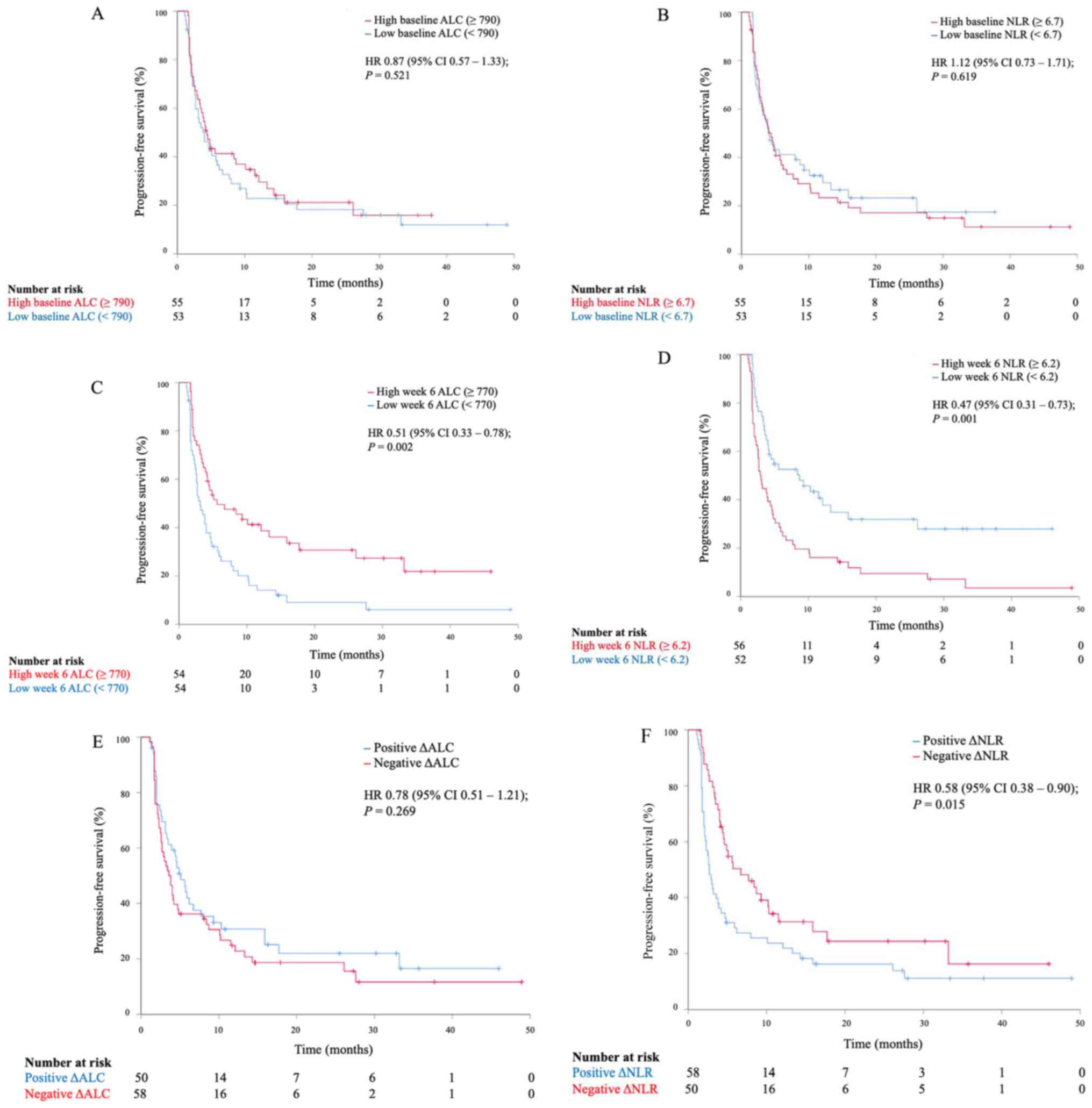
(95% CI, 3.1-5.1). Neither baseline ALC nor NLR provided predictive
value for PFS. The mPFS for patients with high (≥790) vs. low
(<790) baseline ALC were 4.0 months (95% CI, 3.3-5.5) and 4.0
months (95% CI, 2.2-5.8), respectively (Fig. 1A). The mPFS for patients with high
(≥6.7) vs. low (<6.7) baseline NLR were 4.4 months (95% CI,
3.0-5.8) and 4.1 months (95% CI, 2.6-5.6), respectively (Fig. 1B). On the other hand, both week 6
ALC and week 6 NLR were predictive for PFS. High week 6 ALC levels
were associated with a longer PFS. The mPFS for patients with high
(≥770) vs. low (<770) levels at week 6 ALC were 5.6 months (95%
CI, 0.2-11.0) and 3.1 months (95% CI, 2.2-4.1), respectively (HR,
0.51; 95% CI, 0.33-0.78; P=0.002, Fig.
1C). Low week 6 NLR was also predictive for improved PFS. The
mPFS for patients with high (≥6.2) vs. low (<6.2) levels at week
6 NLR were 2.9 months (95% CI, 2.3-3.5) and 8.7 months (95% CI,
2.7-14.7), respectively (HR, 0.47; 95% CI; 0.31-0.73, P=0.001,
Fig. 1D). Increase in ALC
on-treatment at week 6 (positive ΔALC) was associated with a trend
for longer PFS but was not statistically significant (Fig. 1E). Decrease in NLR on-treatment at
week 6 (negative ΔNLR) was associated with a superior PFS. The mPFS
for patients with negative vs. positive ΔNLR were 6.7 months (95%
CI, 2.9-10.5) and 2.7 months (95% CI, 2.1-3.3), respectively (HR,
0.58; 95% CI, 0.38-0.90; P=0.015, Fig.
1F). Other clinicopathologic characteristics, including HPV
status, line of ICI therapy, ECOG status, and smoking history, were
examined for correlation with disease progression. None of the
subgroups showed a differential impact on PFS (Table II). The differential predictive
value of week 6 ALC, week 6 NLR, and ΔNLR for PFS all remained
statistically significant after a multivariable analysis (P=0.009,
0.005, and 0.037, respectively).
 | Table IIPFS of subgroups based on various
peripheral lymphocyte values and other clinical variables. |
Table II
PFS of subgroups based on various
peripheral lymphocyte values and other clinical variables.
| Variable | Median PFS, months
(95% CI) | HR | P-value |
|---|
| Total population | 4.1 (3.1-5.1) | N/A | N/A |
| ALC values | | | |
|
Baseline
ALC | | | |
|
High
(≥790) | 4.0 (3.3-5.5) | 0.87 | 0.521 |
|
Low
(<790) | 4.0 (2.2-5.8) | | |
|
Baseline
NLR | | | |
|
High
(≥6.7) | 4.4 (3.0-5.8) | 1.12 | 0.619 |
|
Low
(<6.7) | 4.1 (2.6-5.6) | | |
|
Week 6
ALC | | | |
|
High
(≥770) | 5.6 (0.2-11.0) | 0.51 | 0.002 |
|
Low
(<770) | 3.1 (2.2-4.1) | | |
|
Week 6
NLR | | | |
|
High
(≥6.2) | 2.9 (2.3-3.50) | 0.47 | 0.001 |
|
Low
(<6.2) | 8.7 (2.7-14.7) | | |
|
ΔALC | | | |
|
Positive | 5.1 (3.5-6.7) | 0.78 | 0.269 |
|
Negative | 3.6 (2.5-4.7) | | |
|
ΔNLR | | | |
|
Positive | 2.7 (2.1-3.3) | 0.58 | 0.015 |
|
Negative | 6.7 (2.9-10.5) | | |
| Other
characteristics | | | |
|
HPV | | | |
|
Positive | 4.2 (0.7-7.7) | 0.90 | 0.660 |
|
Unknown/negative | 4.1 (3.1-5.1) | | |
|
Line of ICI
therapy | | | |
|
First
line | 5.1 (2.7-7.5) | 0.76 | 0.216 |
|
≥
Second line | 3.8 (3.0-4.6) | | |
|
ECOG | | | |
|
0,
1 | 4.4 (2.6-6.2) | 0.69 | 0.117 |
|
2,
3, 4 | 3.1 (1.4-4.8) | | |
|
Smoking | | | |
|
≥10
PY | 3.8 (2.9-4.7) | 1.17 | 0.461 |
|
Never
or <10 PY | 4.7 (2.3-7.1) | | |
Response rate analysis
The response rate to anti-PD-1 therapy for the total
group of patients with advanced HNSCC was 22.2% (95% CI,
14.8-31.2), including three complete responses and 21 partial
responses (Table III). Response
rates were not significantly different between high vs. low
baseline ALC and NLR cohorts, although patients with high ALC and
low NLR values at baseline and week 6 had numerically higher
response rates. The response rate for patients with high (≥790) or
low (<790) baseline ALC were 24.1% (95% CI, 13.5-37.6) and 20.4%
(95% CI, 10.6-33.5), and the response rate for patients with high
(≥6.7) or low (<6.7) baseline NLR were 25.5% (95% CI, 14.7-39.0)
and 18.9% (95% CI, 9.4-32.0), respectively. Similarly, the response
rates for patients with high (≥770) or low (<770) week 6 ALC
were 24.1% (95% CI, 21.1-47.5) and 20.4% (95% CI, 10.6-33.5), and
the response rate for patients with high (≥6.2) or low (<6.2)
week 6 NLR were 17.9% (95% CI, 8.9-7-30.4) and 26.9% (95% CI,
15.6-41.0), respectively. On-treatment dynamic changes of ALC and
NLR and other baseline clinicopathologic parameters showed no
differential effect on response rate (Table III).
 | Table IIIResponse rate of subgroups based on
various peripheral lymphocyte values and other clinical
variables. |
Table III
Response rate of subgroups based on
various peripheral lymphocyte values and other clinical
variables.
| Variable | Response rate (95%
CI) | HR |
|---|
| Total population | 22.2 (14.8-31.2) | N/A |
| ALC values | | |
|
Baseline
ALC | | |
|
High
(≥790) | 24.1 (13.5-37.6) | 0.645 |
|
Low
(<790) | 20.4
(10.6-33.5) | |
|
Baseline
NLR | | |
|
High
(≥6.7) | 25.5
(14.7-39.0) | 0.412 |
|
Low
(<6.7) | 18.9
(9.4-32.0) | |
|
Week 6
ALC | | |
|
High
(≥770) | 24.1
(21.1-47.5) | 0.645 |
|
Low
(<770) | 20.4
(10.6-33.5) | |
|
Week 6
NLR | | |
|
High
(≥6.2) | 17.9
(8.9-30.4) | 0.263 |
|
Low
(<6.2) | 26.9
(15.6-41.0) | |
|
ΔALC | | |
|
Positive | 20.0
(10.0-33.7) | 0.611 |
|
Negative | 24.1
(13.9-37.2) | |
|
ΔNLR | | |
|
Positive | 20.7
(11.2-33.4) | 0.682 |
|
Negative | 24.0
(13.1-38.2) | |
| Other
characteristics | | |
|
HPV | | |
|
Positive | 22.6
(9.6-41.1) | 0.955 |
|
Unknown/negative | 22.1
(13.4-33.0) | |
|
Line of ICI
therapy | | |
|
First
line | 27.5
(15.9-41.7) | 0.214 |
|
≥
Second line | 17.5
(8.7-29.9) | |
|
ECOG | | |
|
0,
1 | 22.1
(13.4-33.0) | 0.955 |
|
2,
3, 4 | 22.6
(9.6-41.1) | |
|
Smoking | | |
|
≥10
PY | 22.6
(12.3-36.2) | 0.921 |
|
Never
or <10 PY | 21.8
(11.8-35.0) | |
Discussion
In this single institution retrospective study, we
examined various measures of peripheral lymphocyte levels in
association with response to anti-PD-1 ICI therapy in patients with
advanced HNSCC. To our knowledge, this is the first study to
explore the correlation of anti-PD-1 immune checkpoint inhibitor
therapy with multiple lymphocyte variables in HNSCC. Baseline
lymphocyte measures either in absolute level or in proportion with
neutrophil count did not predict anti-PD-1 therapy response.
However, early on-treatment lymphocyte values for both ALC and NLR
were significantly associated with ICI treatment outcome. A high
week 6 ALC and higher week 6 NLR were associated with longer
disease control. A low week 6 NLR was also associated with a trend
for higher response rate but was not statistically significant. As
per dynamic changes of lymphocyte, decreasing NLR (increasing
lymphocyte proportion) change during treatment was associated with
a longer PFS but ALC change was not correlated with the
outcome.
Several studies previously explored the correlation
between peripheral lymphocyte count and ICI therapy outcomes in
variable solid tumors. Ho et al (4) reported a correlation between
pretreatment ALC ≥ 600 cells/µl and NLR <7 with disease control
with anti-PD-1 therapy in recurrent or metastatic HNSCC. Similar
association between baseline ALC and NLR with ICI therapy outcomes
was demonstrated in lung cancer, melanoma, and other solid tumors
(5,9-11).
In our patient cohort, baseline lymphocyte values had no
correlation with ICI outcome. Lalani et al (9) demonstrated that week 6 NLR had a
stronger correlation with response rate, PFS, and OS of advanced
renal cell carcinoma patients who were treated with ICI therapy,
which was more consistent with our findings. In addition, as shown
in our HNSCC cohort, the association between NLR changes with ICI
outcomes has been observed in various solid tumors (5,9,12).
The major limitation of our study was the inherent
bias of retrospective analyses in uncontrolled groups. Despite
multivariable analyses, the potential selection bias and the
imbalance of the baseline characteristics and treatment history may
have contributed to the treatment outcomes. The relatively small
sample size may have limited statistical power for subgroup
analyses.
In conclusion, our single institution retrospective
analysis suggests that week 6 ALC, week 6NLR, and on-treatment NLR
dynamic have a predictive value for anti-PD-1 therapy responses.
Our findings warrant further investigation of peripheral lymphocyte
as a potential clinical marker for ICI therapy in advanced
HNSCC.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
JCP designed the study and wrote the manuscript. JD
analyzed the data, created figures and reviewed the manuscript. JRC
participated in the study design and reviewed the manuscript. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was conducted under the IRB
Protocol Partners #2018P001456.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ferris RL, Blumenschein G Jr, Fayette J,
Guigay J, Colevas AD, Licitra L, Harrington K, Kasper S, Vokes EE,
Even C, et al: Nivolumab for recurrent squamous-cell carcinoma of
the head and neck. N Engl J Med. 375:1856–1867. 2016.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Burtness B, Harrington KJ, Greil R,
Soulières D, Tahara M, de Castro G Jr, Psyrri A, Basté N, Neupane
P, Bratland Å, et al: Pembrolizumab alone or with chemotherapy
versus cetuximab with chemotherapy for recurrent or metastatic
squamous cell carcinoma of the head and neck (KEYNOTE-048): A
randomised, open-label, phase 3 study. Lancet. 394:1915–1928.
2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Cohen EEW, Soulieres D, Le Tourneau C,
Dinis J, Licitra L, Ahn MJ, Soria A, Machiels JP, Mach N, Mehra R,
et al: Pembrolizumab versus methotrexate, docetaxel, or cetuximab
for recurrent or metastatic head-and-neck squamous cell carcinoma
(KEYNOTE-040): A randomised, open-label, phase 3 study. Lancet.
393:156–167. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Ho WJ, Yarchoan M, Hopkins A, Mehra R,
Grossman S and Kang H: Association between pretreatment lymphocyte
count and response to PD1 inhibitors in head and neck squamous cell
carcinomas. J Immunother Cancer. 6(84)2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ameratunga M, Chénard-Poirier M, Candilejo
IM, Pedregal M, Lui A, Dolling D, Aversa C, Garces AI, Ang EJ,
Banerji U, et al: Neutrophil-lymphocyte ratio kinetics in patients
with advanced solid tumours on phase I trials of PD-1/PD-L1
inhibitors. Eur J Cancer. 89:56–63. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Huemer F, Lang D, Westphal T, Gampenrieder
SP, Hutarew G, Weiss L, Hackl H, Lamprecht B, Rinnerthaler G and
Greil R: Baseline absolute lymphocyte count and ECOG performance
score are associated with survival in advanced non-small cell lung
cancer undergoing PD-1/PD-L1 blockade. J Clin Med.
8(1014)2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Maltese M, Panni S, Lazzarelli S,
Brighenti M, Negri F and Ratti M: High baseline lymphocyte count is
a predictive biomarker of prolonged time to progression in patients
with advanced solid tumors receiving checkpoint inhibitors. J Clin
Oncol. 35 (Suppl 15):e14532. 2017.
|
|
8
|
Zer A, Sung MR, Walia P, Khoja L, Maganti
M, Labbe C, Shepherd FA, Bradbury PA, Feld R, Liu G, et al:
Correlation of neutrophil to lymphocyte ratio and absolute
neutrophil count with outcomes with PD-1 axis inhibitors in
patients with advanced non-small-cell lung cancer. Clin Lung
Cancer. 19:426–434. e421. 2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Lalani AKA, Xie W, Martini DJ, Steinharter
JA, Norton CK, Krajewski KM, Duquette A, Bossé D, Bellmunt J, Van
Allen EM, et al: Change in neutrophil-to-lymphocyte ratio (NLR) in
response to immune checkpoint blockade for metastatic renal cell
carcinoma. J Immunother Cancer. 6(5)2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Bennati C, Mazza V, D'Arcangelo M, Minuti
G, Vecchiarelli S, Attilia L, Gili A, Montanari M, Landi L and
Cappuzzo F: E5Integrating programmed cell death ligand 1 (PD-L1)
and neutrophil to lymphocyte ratio (NLR) as predictive panel of
response to nivolumab in non-small cell lung cancer (NSCLC). Ann
Oncol 28 (Suppl_6): doi: 10.1093/annonc/mdx426.004.
|
|
11
|
Capone M, Giannarelli D, Mallardo D,
Madonna G, Festino L, Grimaldi AM, Vanella V, Simeone E, Paone M,
Palmieri G, et al: Baseline neutrophil-to-lymphocyte ratio (NLR)
and derived NLR could predict overall survival in patients with
advanced melanoma treated with nivolumab. J Immunother Cancer.
6(74)2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Moschetta M, Uccello M, Kasenda B, Mak G,
McClelland A, Boussios S, Forster M and Arkenau HT: Dynamics of
neutrophils-to-lymphocyte ratio predict outcomes of PD-1/PD-L1
blockade. Biomed Res Int. 2017(1506824)2017.PubMed/NCBI View Article : Google Scholar
|