Introduction
Renal angiomyolipoma (RAML) is among the most common
kidney benign tumors. It consists of different proportions of
vascular, smooth muscle, and fat and has two types, namely,
tuberous sclerosis complex (TSC)-associated and sporadic types
(1-3).
TSC is an autosomal dominant disease that affects multiple organs
of the body. It is usually accompanied by the growth of hamartomas
in the skin, brain, kidneys, lung, and heart. The various
manifestations of TSC usually appear at different stages of life.
In infancy, the most common manifestations are hamartomas,
seizures, skin lesions, and rhabdomyosarcoma in the brain.
TSC-associated RAML often develops at a younger age and grows much
faster over time than sporadic RAML. Large RAML (>3-4 cm in
diameter) may develop into an aneurysm, which may rupture, bleed,
and become life-threatening, or it may compress the normal kidney
tissue, leading to renal failure. Therefore, early detection,
correct diagnosis, and reasonable treatment are very important for
patients with RAML. We present a case of giant RAML and discussed
the diagnosis and treatment of TSC-associated RAML.
Case report
The 20-year-old young female patient has been
diagnosed with epilepsy since childhood and has been treated with
valproate sustained release tablets. She was admitted to the
department of neurology of our hospital due to repeated daze,
occasionally accompanied by nausea, vomiting, and hallucinations.
Physical examination showed that the patient was slightly slow in
response and expression, and her calculation was poor. Multiple
small papules and nodules can be seen on her cheeks. Palpation of
the right upper abdomen revealed a tough mass with smooth surface,
fixed position, and no tenderness. Valproic acid test in the
laboratory showed that the patient's CYP2C9 genotype was CYP2C811,
and the enzyme activity was fast metabolism (EM) type. POLG
(A467TG>A) was of wild homozygous type, POLG (W748SC>G) wild
homozygous type SLCO1B1 genotype was 1b1b, and ApoE genotype was
E2E4. The electrolyte, liver enzymes, renal functions, cardiac
functions, routine examination of blood and urine, and coagulation
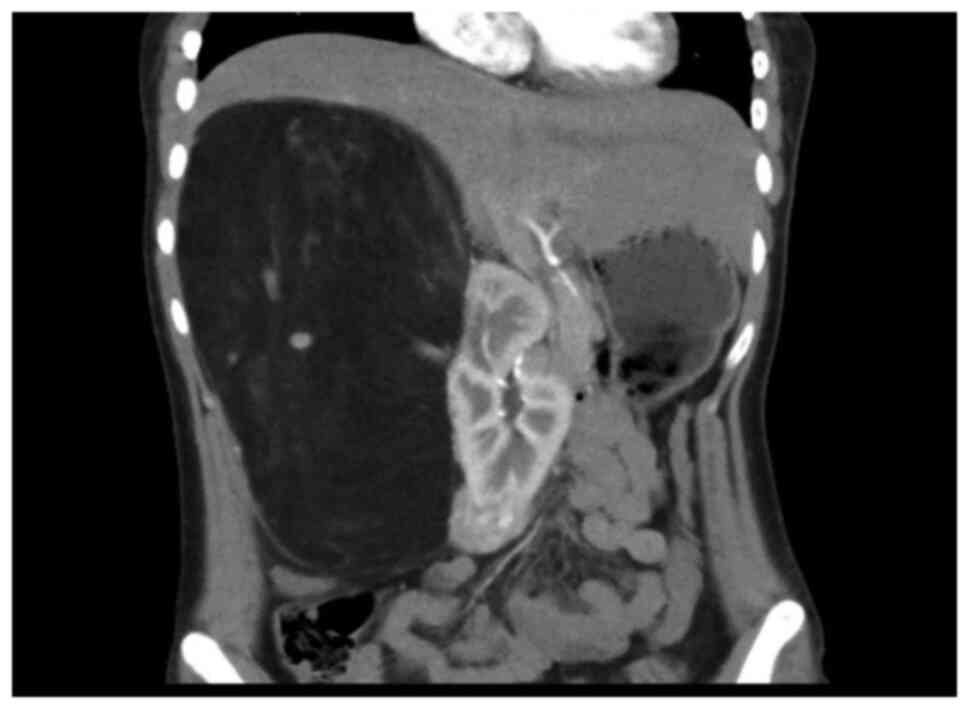
functions were normal. Abdominal enhanced CT scan showed that the
liver was obviously pushed, and the spine was scoliotic. A huge
mass occupied the right upper quadrant, with a size of ~236x125x149
mm. The CT value was about-78HU. Blood vessels could be seen
inside, and the boundary between the blood vessel and the kidney
was unclear. The surrounding tissue was pushed, and the right
kidney was displaced to the middle abdomen (Fig. 1). Moreover, two inhomogeneous
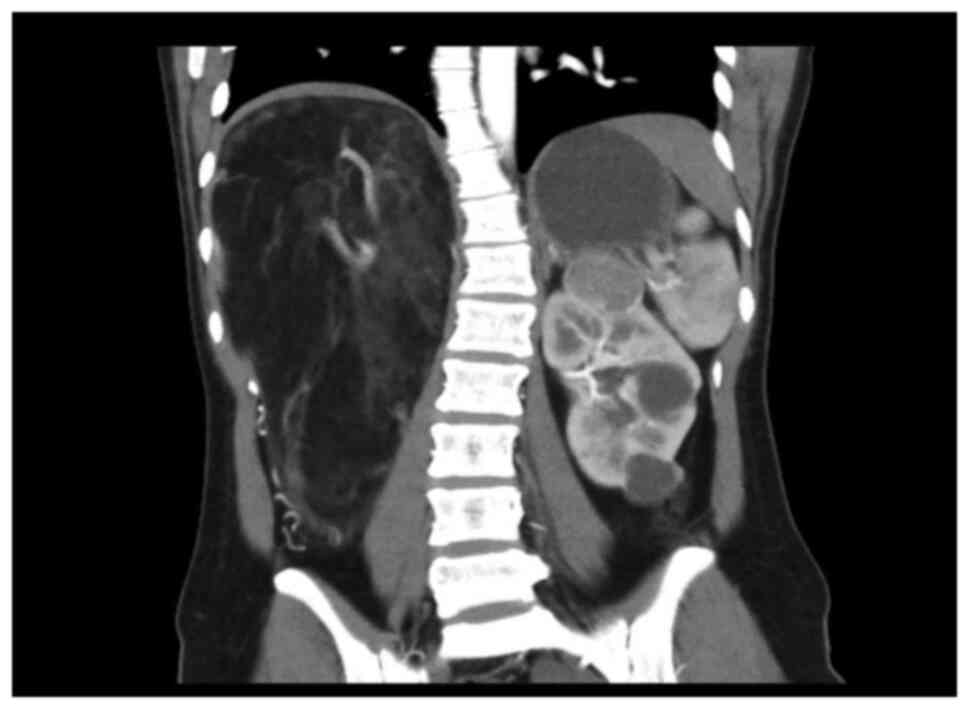
enhancement nodules were present in the middle and lower pole of
the right kidney. The larger nodule (26x33 mm in size) was in the
right kidney. Multiple low-density shadows with no enhancement were
also evident in the right kidney. In the left kidney, an
inhomogeneous enhancement nodule was present on the upper pole, and
its size was 28x29 mm. Multiple no enhancement low-density shadows
(Fig. 2) were distributed in the
left kidney. The right adrenal gland was unclear. The shape, size,
and density of the left adrenal gland were normal. No obvious
dilation and hydrops were observed in the bilateral renal pelvis
and calyces, and the intestinal curve was pushed. No obvious
enlarged lymph nodes and no ascites signs were seen behind the
peritoneum. The result of the Chest CT scan showed multiple nodular
shadows with sizes of ~2-3 mm in the right upper lobe and dorsal
right lower lobe. A nodular shadow, ~3 mm in size, was seen under
the pleura of the tongue of the left upper lobe. Cranial MRI showed
the following results. Abnormal signals were detected from the
bilateral cerebral hemisphere cortex, right putamen, and right
caudate nucleus. A slightly low signal was observed in T1WI. A
slightly high signal was observed in T2WI and FRAIR. The DWI showed
no diffusion with limited high signal. Under the bilateral
maxillary sinus mucosa, round-like long T1 long T2 signal shadows
were seen, and the larger one was ~15x17 mm in size. Considering
the patient's medical history and the results of laboratory
examination, the diagnosis included epilepsy, giant hamartoma of
the right kidney, hamartoma of the left kidney, multiple pulmonary
nodules, and abnormal head signals, which were highly consistent
with the manifestations of TSC. Although the patient had no obvious
symptoms of abdominal visceral compression, and laboratory
examination showed no obvious abnormalities in the kidney function.
The compression of a huge abdominal mass caused the liver and
kidney to deviate significantly and led to spine scoliosis. The
continued growth of the mass may affect the function of the kidney
and the digestive system. Moreover, the overgrown mass puts the
patient at risk of spontaneous rupture and bleeding, which are
life-threatening. So, the mass was indicated for surgical
resection. After comprehensive preparation before the operation,
the patient underwent open mass resection and right kidney partial
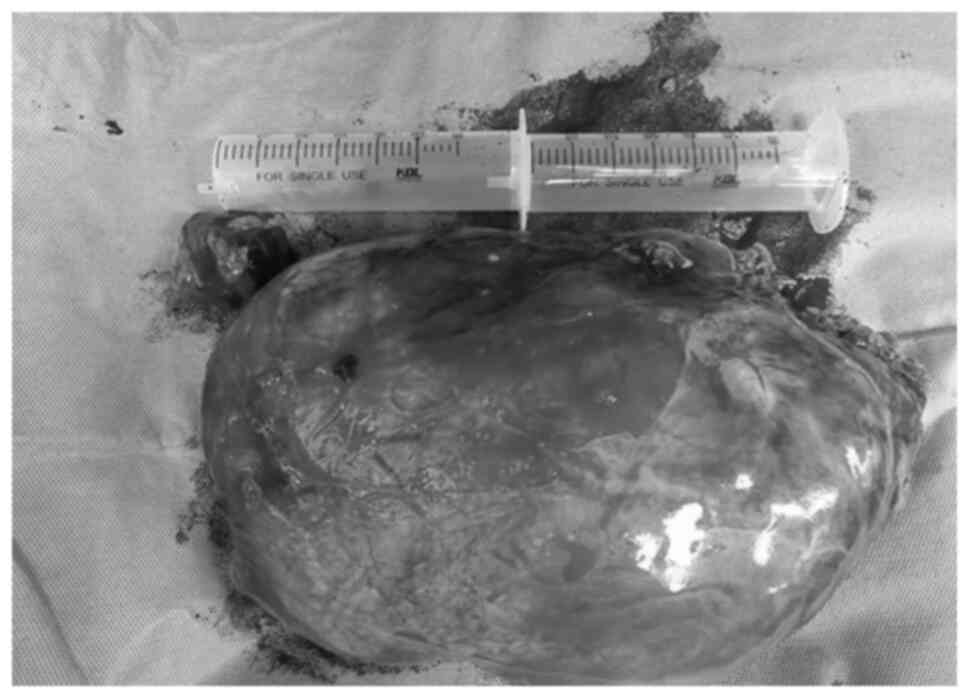
resection. The appearance of the tumor is shown below (Fig. 3). Postoperative pathological results
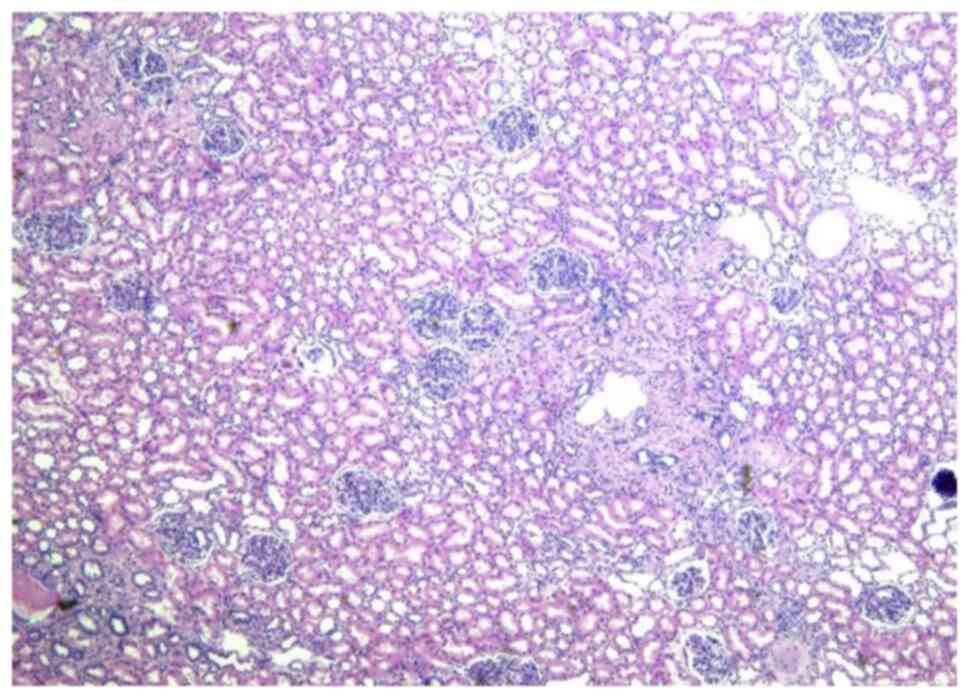
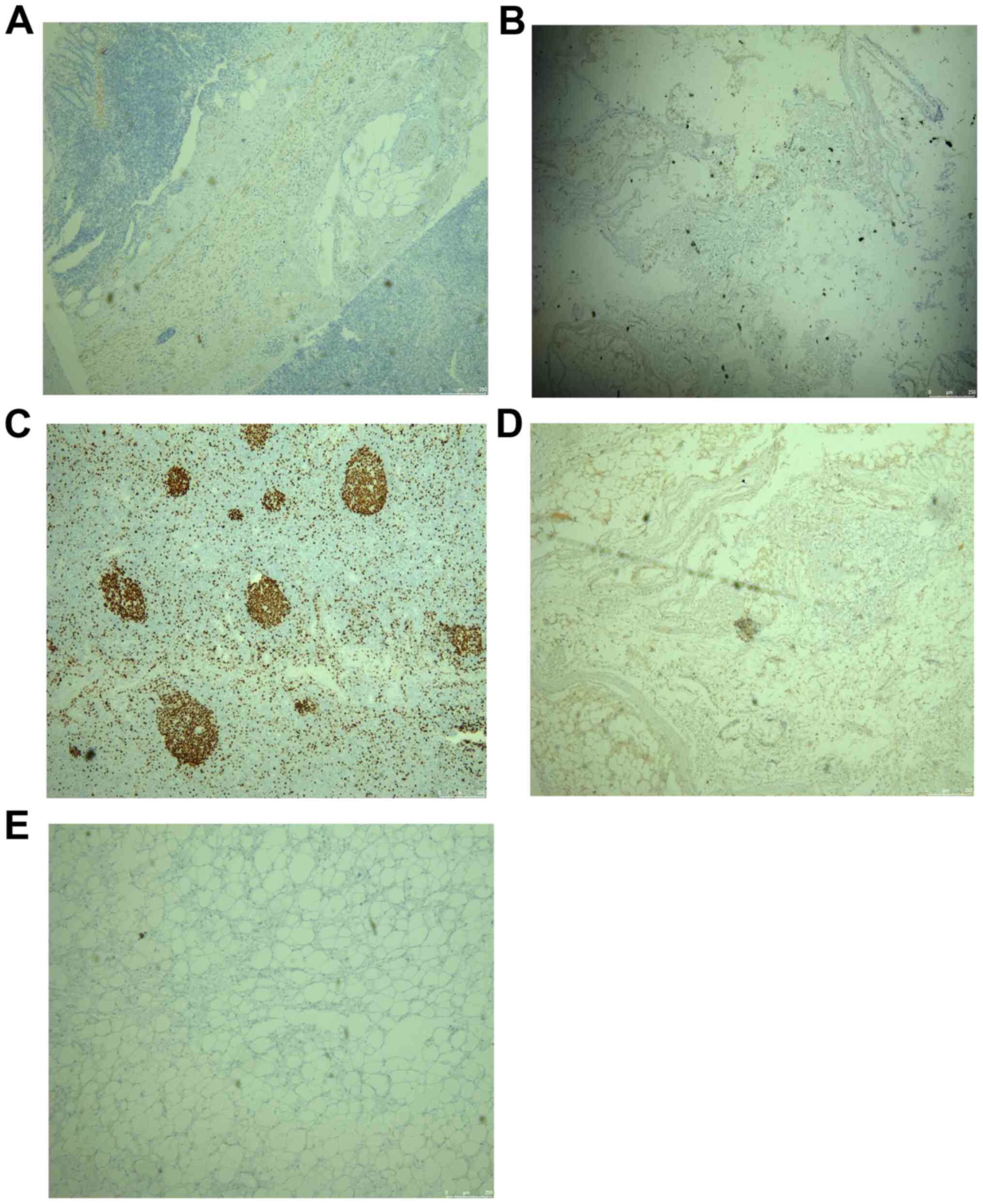
(Fig. 4) confirmed RAML, and the
immunohistochemical results were as follows: HMB45 +, SMA +, Des +,
S100 +, and Ki67 (1% +) (Fig. 5).
The patient recovered smoothly after the operation. No recurrence
of kidney tumors was found. Nodules in the lungs and abnormal head
signals remained the same after the half-year follow-up period.
Discussion
The diagnosis of RAML
RAML is the most common benign renal tumor. RAML can
be divided into sporadic and tuberous sclerosis (TSC) related
types. At present, RAML is mostly sporadic and occurs more commonly
in women are than in men. It is usually first found by B-ultrasound
and then diagnosed by CT or MRI. TSC is an autosomal dominant
inherited disease caused by mutations in the TSC 1 gene on
chromosome 9 or the TSC 2 gene on chromosome 16(4). Mutation of TSC1 or TSC2 gene causes
the abnormal function of the TSC1/TSC2 complex, releases inhibitory
effect on mechanistic target of rapamycin complex 1 (mTORC1),
promotes cell growth, and inhibits autophagy, leading to TSC. TSC
affects multiple organs of the body and is usually accompanied by
the growth of hamartomas in the skin, brain, kidney, lung, and
heart. Fat-containing angiomyolipomas have been observed in 80% of
TSC patients. Fat-poor lesions are also common in patients with
TSC, but occur in <0.1% of the general population. Renal
manifestations in TSC include RAML and multiple renal cysts
(5). Compared with sporadic RAML,
the tumor is multiple and tends to grow to a larger volume; it is
more likely to rupture, which results in hemorrhagic shock
secondary to hematuria or retroperitoneal bleeding. Angiomyolipomas
in the kidney can cause serious bleeding issues because of its
vascular nature and can necessitate dialysis and even renal
transplantation. In sporadic RAML cases, the tumor is generally
single and asymptomatic. The typical symptoms of TSC-related RAML
include abdominal pain (53%), palpable masses (47%), and gross
hematuria (23%). These symptoms are typical in patients with
multiple sclerosis whose tumor is larger than 4 cm; this is called
Lenk's triad. Other possible symptoms and signs include bleeding,
nausea, vomiting, high blood pressure, anemia, fever, shock, and
urinary tract infection. Children with TSC mutant genes have normal
kidneys at birth, but renal cysts and RAML appear with age. TSC in
the nervous system manifests as clinical symptoms related to the
growth of hamartomas, such as cortical dysplasia, subependymal
nodules, and subependymal giant cell astrocytoma (SEGA). Huge SEGA
may block the flow of cerebrospinal fluid through the orifice,
resulting in increased intracranial pressure, obstructive
hydrocephalus, focal neurological deficit, and death. The growth of
hamartomas and other disturbances to normal cell growth can cause
epilepsy and neurological cognition, as well as behavioral and
mental defects. Epilepsy occurs in 60-90% of TSC patients, usually
starting within the first year of birth. Early-onset seizures or
intractable seizures are associated with cognitive and learning
disabilities. The remaining TSC neurological symptoms also include
empirical cognitive impairment, autism, and other behavioral
disorders (50%) (6). Other
manifestations of TSC include the following: Rhabdomyosarcoma of
the heart; lymphangioleiomyomatosis, which is a common pulmonary
manifestation of TSC with a particularly high incidence in women;
retinopathy (multiple retinal hamartomas and retinal pigment
spots); and skin lesions (e.g., angiofibroma, which eventually
appears in ~90% of patients; depigmentation spots; nail fibroids;
shark plaques; oral fibroids; and others) (7). In 2012, the International TSC
Consensus Committee formulated two independent TSC diagnostic
criteria, namely, clinical and genetic diagnoses. Clinical
diagnosis depends on the 11 main features and six secondary
features of the patient (Table I)
(8), among which 2 major features
or 1 major feature + 2 or more minor features can be diagnosed as
TSC. If only 1 major feature or only 2 minor features, then
diagnosis is possible. The detection of TSC1 or TSC2 gene
pathogenic mutations in non-lesioned tissues can be diagnosed as
TSC. The use of peripheral blood samples for genetic testing is
recommended, but negative genetic testing cannot exclude the
diagnosis of TSC. In the present case, the patient presented with
RAML, multiple renal cysts, epilepsy, mental retardation, facial
hemangioma, multiple pulmonary nodules, and abnormal head signals.
The abovementioned findings support clinical diagnosis.
 | Table IMajor and minor features of the
tuberous sclerosis complex (7). |
Table I
Major and minor features of the
tuberous sclerosis complex (7).
| Major features | Minor features |
|---|
| Hypomelanotic macules
(≥3, at least 5-mm diameter) | ‘Confetti’ skin
lesions |
| Angiofibromas (≥3) or
fibrous cephalic plaque | Dental enamel pits
(>3) |
| Ungual fibromas
(≥2) | Intraoral fibromas
(≥2) |
| Shagreen patch | Retinal achromic
patch |
| Multiple retinal
hamartomas | Multiple renal
cysts |
| Cortical
dysplasiasa | Non-renal
hamartomas |
| Subependymal
nodules | - |
| Subependymal giant
cell astrocytoma | - |
| Cardiac
rhabdomyoma | - |
| LAMb | - |
| Angiomyolipomas
(≥2)b | - |
The treatment of TSC-associated
RAML
Early detection and diagnosis of RAML can reduce the
occurrence of serious complications (9). mTOR inhibitors is a new type of
treatment. Prior to the approval of everolimus in 2012, surgical
resection or transarterial embolization was the standard treatment
for most hamartomas requiring medical intervention. Excessive
surgery resection may result in the loss of healthy kidney tissue
and may damage the kidney, leading to the loss of its function.
However, if the tumor tissue is not completely removed, the
remaining tumor tissue may continue to grow, leading to recurrence
(10,11). Embolizing tumor blood vessels does
not eliminate the root cause of TSC. The 2012 consensus guidelines
recommend mTOR inhibitors as the first-line treatment for
asymptomatic RAML under 4 cm in diameter, whereas embolization and
partial resection are used as second-line treatments. Everolimus is
among the most commonly used mTOR inhibitors. Administration at 10
mg/qd oral treatment is recommended. This dosage can effectively
reduce the volume of systemic AML and allow the non-invasive
treatment of TSC patients, especially those with multiple disease
manifestations. However, long-term treatment is usually required.
The side effects associated with mTOR inhibitors and their
management need to be known. These side effects include
stomatitis/mucositis, respiratory infections, diarrhea,
hypertriglyceridemia, hypercholesterolemia, bone marrow
suppression, proteinuria, and arthralgia (12,13).
These adverse reactions are often related to drug concentration
level, and dose adjustment may reduce the occurrence of adverse
drug reactions. Interruption of the use of the drug and subsequent
adjustment of the use of mTOR inhibitors may also help reduce
systemic side effects (14). In the
present case, the patient had no typical symptoms, such as typical
hematuria and abdominal pain. Although the patient has not yet
developed symptoms, such as hematuria, bleeding, nausea, vomiting,
high blood pressure, anemia, fever, shock, and urinary tract
infection, the large size of the tumor has caused scoliosis and
pushing of the surrounding organs. With the passage of time and the
continued growth of the tumor, the patient will likely show the
abovementioned manifestations. Therefore, tumor resection is a
reasonable treatment. Unfortunately, for some reason, this patient
did not receive everolimus treatment. However, it is gratifying
that after half a year of follow-up, the patient showed no obvious
recurrence of RAML. No progress in the lesions in pulmonary and
intracranial areas was observed.
In a conclusion, clinical monitoring is essential
for the treatment of TSC patients. The current guidelines recommend
that for low-risk patients, especially asymptomatic minor patients
with tumor diameters of less than 4 cm, observation and waiting can
be a suitable treatment method. Abdominal MRI or enhanced CT need
to be performed every 1-3 years to evaluate the progress of RAML.
Renal function and blood pressure need to be monitored at least
once a year. Patients who do not take everolimus after selective
arterial embolization or surgical intervention need to to undergo
MRI or enhanced CT every 3-6 months, and their renal function and
blood pressure need to be monitored. For patients with kidney
disease, close follow-up and regular renal function monitoring are
particularly important, because unlike other TSC manifestations,
RAML and other kidney lesions may appear later in life and
accumulate over time, thereby necessitating lifelong follow-up.
Acknowledgements
Not applicable.
Funding
This work was supported by funding from the Medical
Research Foundation of Guangdong Province of China (grant no.
A2018093) and the Guangzhou Education Bureau Innovation Team
Project (grant no. 201400902).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
PS, YH and ZS acquired the data, performed the
literature review and performed surgery and pre-operative
administration. These authors also drafted, reviewed and edited the
manuscript for intellectual content. ZS conceived the current
study, analyzed the data and revised the manuscript for important
intellectual content. PS performed surgery, analyzed data and
images and edited the manuscript. SC performed pathological
staining and pathological diagnosis. SC obtained radiological
images and performed radiological diagnosis. All authors have read
and approved the manuscript.
Ethics approval and consent to
participate
The present case study was approved by the Ethics
Committee of the Fifth Affiliated Hospital of Guangzhou Medical
University of China. The patient provided consent for inclusion in
this study.
Patient consent for publication
Written informed consent was obtained from the
patient for the publication the case details and any associated
images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Franz DN, Bissler JJ and McCormack FX:
Tuberous sclerosis complex: Neurological, renal and pulmonary
manifestations. Neuropediatrics. 41:199–208. 2010.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Curatolo P, Bombardieri R and Jozwiak S:
Tuberous sclerosis. Lancet. 372:657–668. 2008.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Crino PB, Nathanson KL and Henske EP: The
tuberous sclerosis complex. N Engl J Med. 355:1345–1356.
2006.PubMed/NCBI View Article : Google Scholar
|
|
4
|
van Slegtenhorst M, de Hoogt R, Hermans C,
Nellist M, Janssen B, Verhoef S, Lindhout D, van den Ouweland A,
Halley D, Young J, et al: Identification of the tuberous sclerosis
gene TSC1 on chromosome 9q34. Science. 277:805–808. 1997.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kozłowska J and Okoń K: Renal tumors in
postmortem material. Pol J Pathol. 59:21–25. 2008.PubMed/NCBI
|
|
6
|
Krueger DA: Management of CNS-related
disease manifestations in patients with tuberous sclerosis complex.
Curr Treat Options Neurol. 15:618–633. 2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Samuels JA: Treatment of renal
angiomyolipoma and other hamartomas in patients with tuberous
sclerosis complex. Clin J Am Soc Nephrol. 12:1196–1202.
2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Northrup H and Krueger DA: International
Tuberous Sclerosis Complex Consensus Group. Tuberous sclerosis
complex diagnostic criteria update: Recommendations of the 2012
international tuberous sclerosis complex consensus conference.
Pediatr Neurol. 49:243–254. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Wheless JW and Klimo P Jr: Subependymal
giant cell astrocytomas in patients with tuberous sclerosis
complex: Considerations for surgical or pharmacotherapeutic
intervention. J Child Neurol. 29:1562–1571. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Krueger DA and Northrup H: International
Tuberous Sclerosis Complex Consensus Group. Tuberous sclerosis
complex surveillance and management: Recommendations of the 2012
international tuberous sclerosis complex consensus conference.
Pediatr Neurol. 49:255–265. 2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Boorjian SA, Frank I, Inman B, Lohse CM,
Cheville JC, Leibovich BC and Blute ML: The role of partial
nephrectomy for the management of sporadic renal angiomyolipoma.
Urology. 70:1064–1068. 2007.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Bissler JJ, McCormack FX, Young LR, Elwing
JM, Chuck G, Leonard JM, Schmithorst VJ, Laor T, Brody AS, Bean J,
et al: Sirolimus for angiomyolipoma in tuberous sclerosis complex
or lymphangioleiomyomatosis. N Engl J Med. 358:140–151.
2008.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Lebwohl D, Thomas G, Lane HA, O'Reilly T,
Escudier B, Yao JC, Pavel M, Franz D, Berg W, Baladi JF, et al:
Research and innovation in the development of everolimus for
oncology. Expert Opin Drug Discov. 6:323–338. 2011.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sheth RA, Feldman AS, Paul E, Thiele EA
and Walker TG: Angiographic and volumetric effects of mammalian
target of rapamycin inhibitors on angiomyolipomas in tuberous
sclerosis. World J Radiol. 8:308–315. 2016.PubMed/NCBI View Article : Google Scholar
|