Introduction
Central nervous system (CNS) metastasis of
urothelial carcinoma (UC) is rare. In patients with metastatic UC
receiving systemic chemotherapy, a cerebral metastasis sometimes
occurs during a long follow-up period after chemotherapy, and the
prognosis is usually very poor (1).
Recent development of new types of systemic chemotherapy against
UC, as well as improvements in immune checkpoint inhibitors for
patients with advanced, metastatic forms of the disease, have
contributed to improving the prognosis of patients. As
immunotherapy can produce long-term remission in some patients,
screening for CNS metastasis is important. However, to the best of
our knowledge, the present report is the first to describe a CNS
metastasis during remission induced by immunotherapy. We herein
presented two cases of cerebral metastasis occurring in the context
of systemic disease control by immunotherapy. To the best of our
knowledge, the present report is the first to describe a CNS
metastasis during remission induced by immunotherapy.
Case report
Case 1
A 54-year-old, male patient with asymptomatic gross
hematuria was referred to us by the previous hospital. He had no
past history of any serious illness or smoking. Cystoscopy revealed
a non-papillary, sessile bladder tumor, and computed tomography
(CT) revealed invasive bladder cancer but no metastasis
(cT2N0M0).
Transurethral resection of the bladder tumor (TURBT)
was performed. Pathological examination showed Grade 3, pT2, UC.
The patient was referred to our hospital for radical treatment.
Because the patient declined a radical cystourethrectomy,
intra-arterial chemotherapy with cisplatin and therarubicin for the
bladder was performed in two cycles. After the intra-arterial
chemotherapy, cystoscopy detected two recurrences of the bladder
cancer over four months, prompting TURBT to be performed each time.
One year after the intra-arterial chemotherapy, CT revealed a lung
metastasis, and systemic chemotherapy with gemcitabine and
cisplatin (GC) was begun. After four cycles, partial resection of
the right middle lung lobe was performed. Pathological analysis
revealed UC. By postoperative four months, the primary bladder
cancer had grown, and new, multiple, lung metastases had appeared.
Although an additional two cycles of GC chemotherapy were
administered, the lung metastases and primary bladder cancer
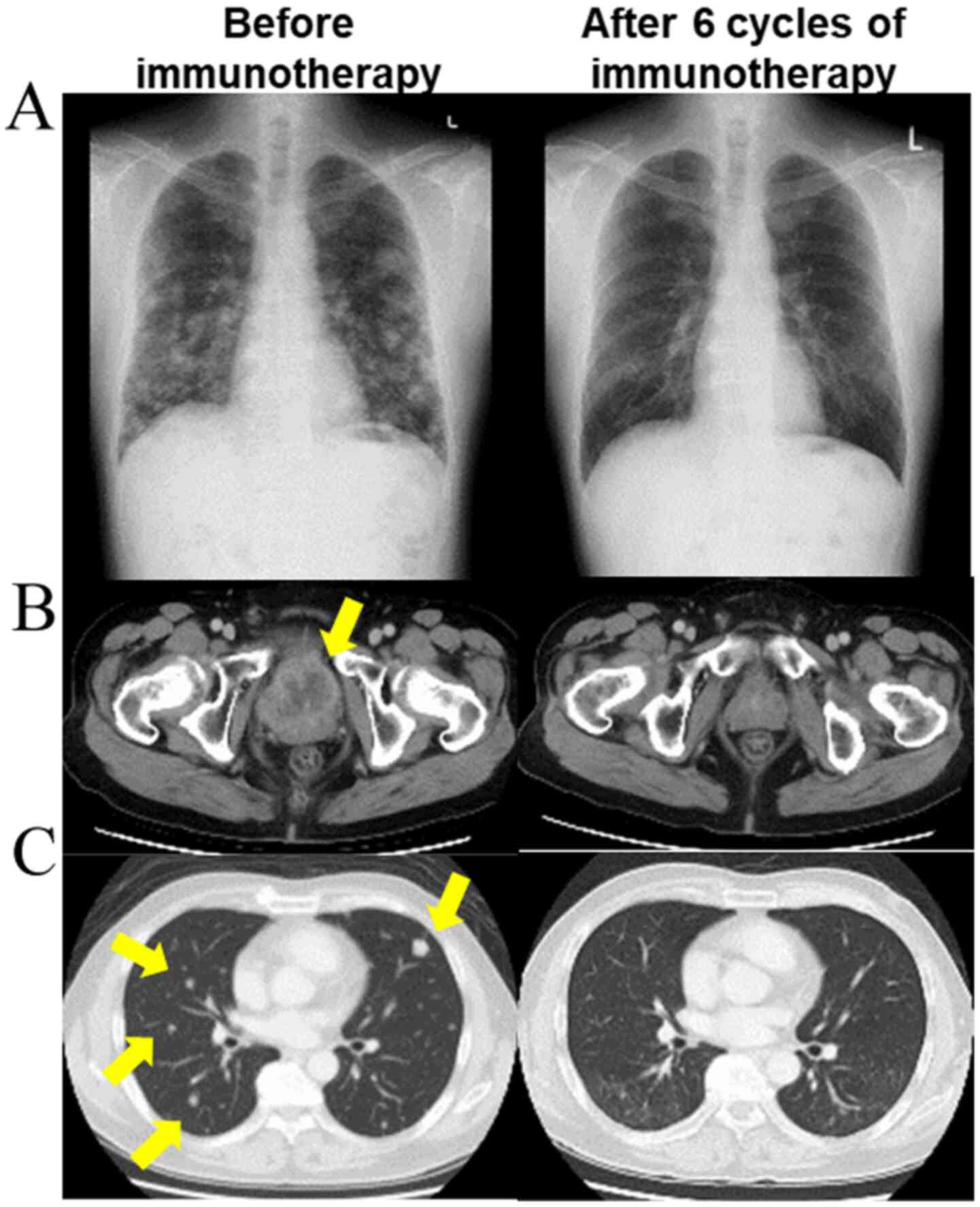
continued to grow (Fig. 1).
Immunotherapy with pembrolizumab was administered as
the second line treatment. After six cycles, the primary bladder
cancer and lung metastases resolved completely (Fig. 1), and immunotherapy was temporarily
halted. Brain magnetic resonance (MRI) imaging at this time
revealed no brain metastasis.
Two years after the immunotherapy with pembrolizmab,
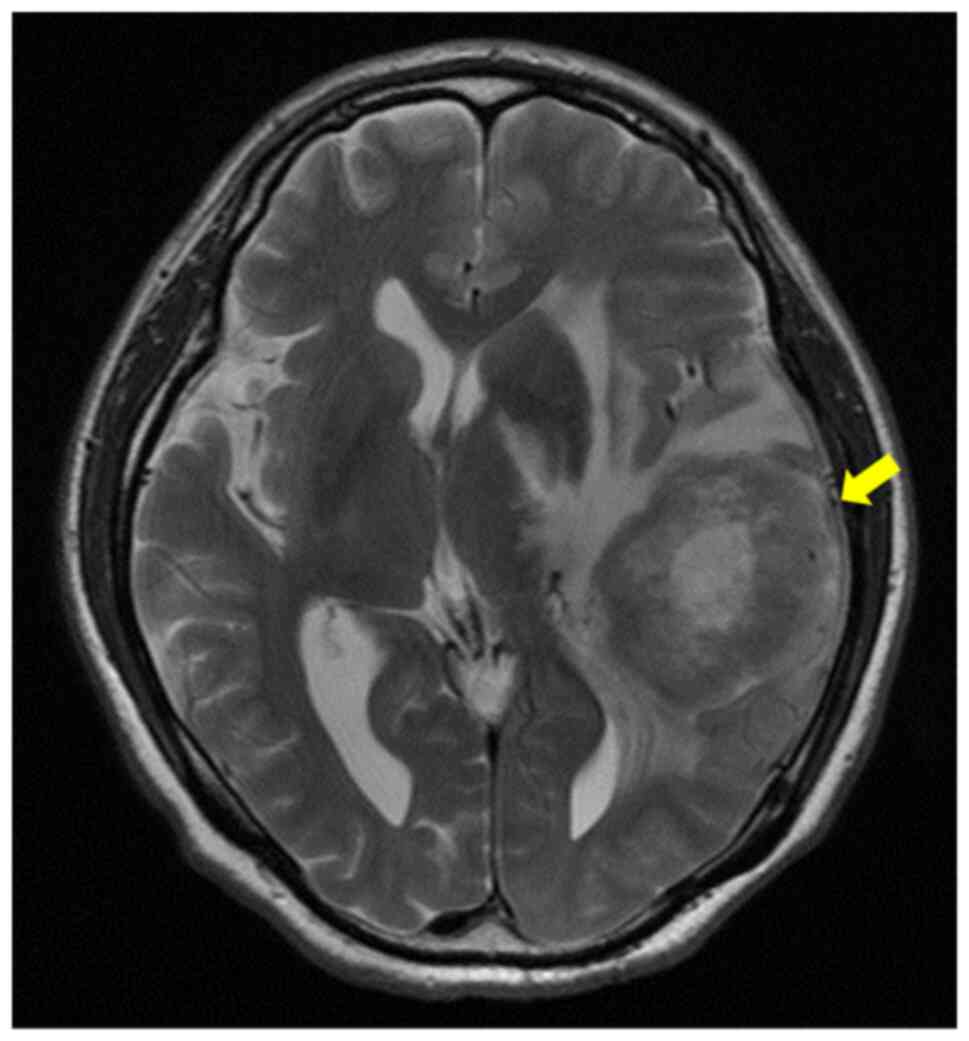
the patient was referred to our hospital due to aphasia. Brain MRI
revealed a tumor in the left parietal lobe with edema (Fig. 2). Based on these findings, a
cerebral metastasis was diagnosed, and irradiation therapy with
Gamma Knife (27 Gy) was performed. The patient was recurrence-free
18 months after the radiation therapy.
Case 2
A 77-year-old, male patient with asymptomatic gross
hematuria was referred to our hospital. He had a past history of
diabetes mellitus and no history of smoking. Cystoscopy revealed a
non-papillary, sessile bladder tumor on the left ureteral orifice.
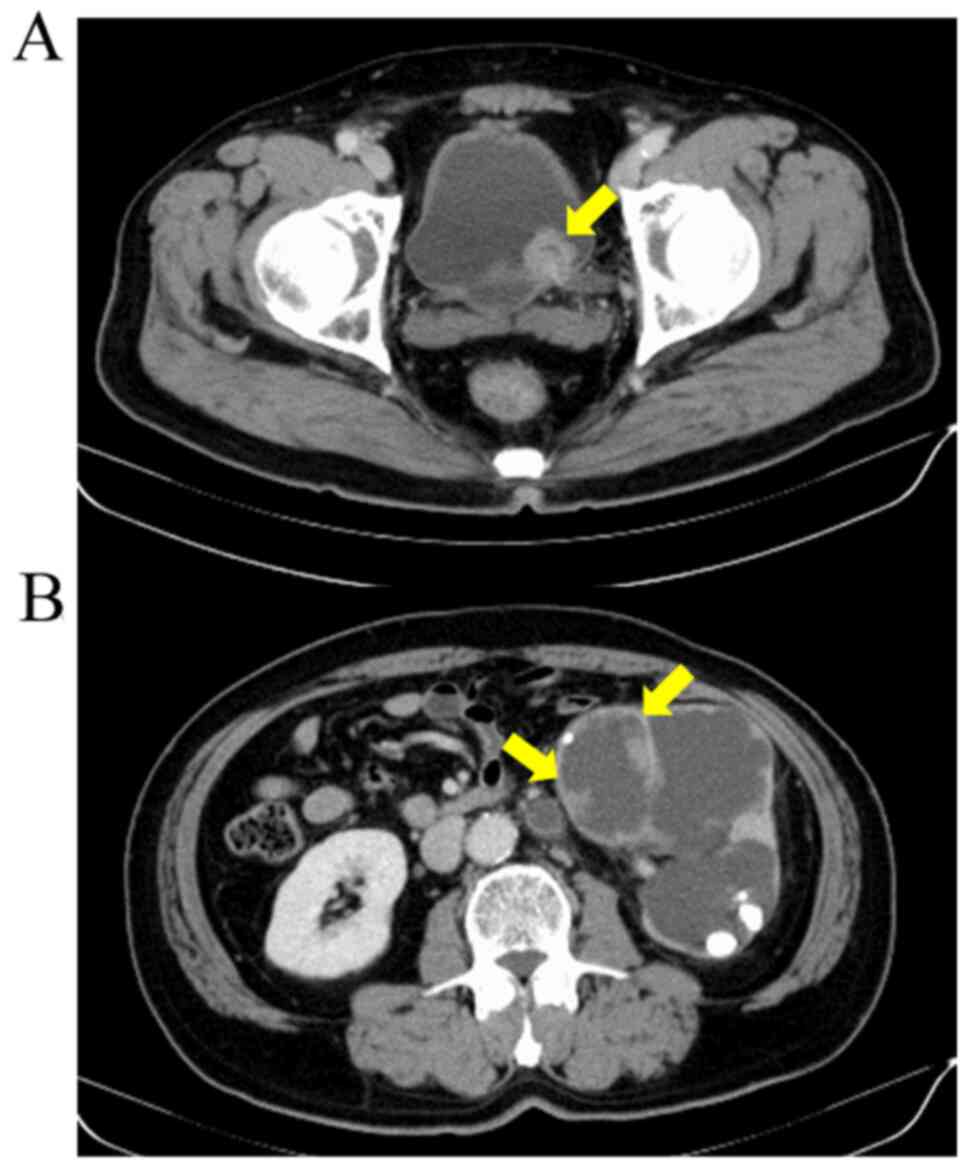
CT and MRI also revealed a left renal pelvic tumor with
hydronephrosis and renal stone but no metastasis (Fig. 3). Based on these findings, bladder
cancer and left renal pelvic cancer (cTaN0M0) were diagnosed
preoperatively.
TURBT was performed, and a subsequent pathological
examination revealed Grade 2, pTa, UC. Three months later, a left
radical nephroureterectomy was performed, and pathological analysis
revealed Grade 3, pT1, UC. Thereafter, TURBT was repeated three
times over a year, and the pathological findings were Grade 3, pT1,
each time. Because the patient declined a radical
cystourethrectomy, three cycles of intra-arterial chemotherapy with
cisplatin and therarubicin for the bladder cancer were performed.
Subsequently, two more recurrences were detected in four months,
and TURBT was performed on each occasion. The pathological analysis
findings demonstrated Grade 3, pT1. One month later after the last
TURBT, a radical cystourethrectomy with ileal conduit urinary
diversion was performed.
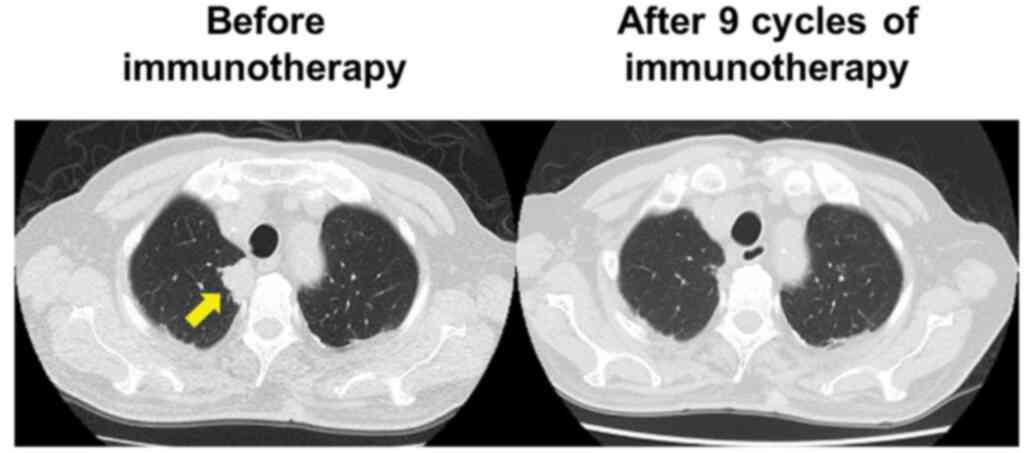
Six years after the cystectomy, CT demonstrated a
right lung tumor (Fig. 4). A
bronchoscopic biopsy was performed, with the pathological findings
revealing metastatic urothelial carcinoma. Positron emission
tomography (PET) also showed abnormal uptake in the right lung
tumor and mediastinal lymph nodes. Chemotherapy with gemcitabine
and carboplatin was begun. However, the patient was unable to
continue the treatment due to fatigue. As the second line
treatment, immunotherapy with pembrolizumab was administered. At
this time, brain MRI revealed no brain metastasis. After nine
courses of immunotherapy, all the metastases had resolved, and
immunotherapy was temporarily halted (Fig. 4).
Fifteen months after the termination of
pembrolizumab treatment, the patient was referred to our hospital
due to motor difficulties and disorientation. Neurological
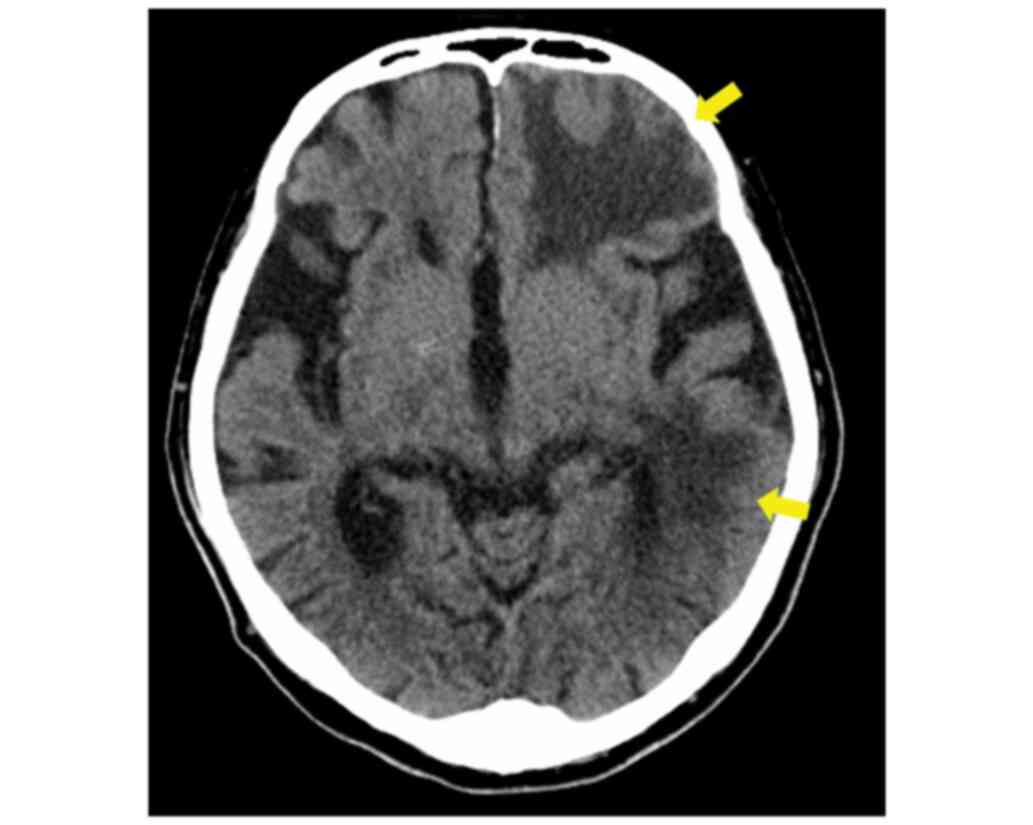
examination revealed no apparent focal deficits. Brain CT revealed
multiple, low-density areas in the left frontal, parietal, and
temporal lobes with edema (Fig. 5).
Based on these findings, multiple, cerebral metastases were
diagnosed, and whole brain irradiation therapy (30 Gy) was
performed. Thereafter, the patient's disorientation improved.
Discussion
UC metastases commonly occur in the lungs, liver,
lymph nodes, bone, adrenal glands, spleen, and intestine. CNS
metastases are rare, with an incidence of less than 1% (1). However, the rate of CNS metastasis has
meanwhile apparently increased to 16% (1). Platinum-based regimens have recently
become the standard treatment for metastatic UC and have improved
the prognosis of patients with this disease, but while chemotherapy
can help achieve systemic remission, chemotherapeutic agents cannot
cross the blood-brain barrier (BBB) to treat metastases in the
brain (2), thus leading to the
apparent increase in the incidence of CNS metastases of UC. In the
present cases, CNS metastases were discovered 15-20 months after CR
induced by immunotherapy, lending support to the above
hypothesis.
Immune checkpoint inhibitors were recently developed
for the treatment of patients with advanced cancer. With this new
therapeutic modality, patients can achieve CR, after which immune
surveillance can be deployed to insure long-term remission
(3). However, immune checkpoint
inhibitors have limited efficacy against CNS metastases thanks to
the unique immune microenvironment of the brain (4). The blood-brain barrier prevents
cytotoxic T lymphocytes (CTLs) from infiltrating brain metastases
across the BBB. Even if long-term remission is achieved via immune
checkpoint inhibitors, CNS metastases can elude treatment and
continue growing owing to the brain's unique immune
microenvironment. The following two facts in both of the present
cases corroborate this hypothesis. First, the cerebral metastases
developed more than one year after CR by immunotherapy. Second, no
metastases were present in any of the other organs.
CNS metastases are detected with high regularity
detected at our hospital. In two of three patients (66%) in whom a
cerebral metastasis of UC developed, complete, as well as
long-term, remission was achieved via immunotherapy. In both
patients, neurological symptoms prompted a CNS evaluation,
suggesting that periodic evaluations of CNS metastasis status even
in the absence of symptoms is more important for long-term disease
control.
Radiotherapy was performed for the CNS metastasis in
both of the present cases. It is likely that the radiotherapy was
not only cytotoxic to the tumor cells, but also compromised the
integrity of the BBB, possibly enabling CTLs to infiltrate the
tumors and attack the cancer cells. Moreover, the subsequent immune
surveillance reduced the risk of recurrence of CNS metastases. A
previous study demonstrated that the prognosis of patients with
brain recurrence was very poor, and that the median survival time
usually did not exceed three months (1). In contrast, one of our patients was
able to maintain remission for more than 12 months.
The present report has some limitations. First, the
timing of the occurrence and the time course of the CNS metastases
were unclear in both cases. Second, an appropriate screening
schedule for CNS metastases was lacking. Third, the follow-up
period was not long enough to determine whether there were any
secondary effects due to the radiotherapy.
Although CNS metastases are very rare in patients
with metastatic UC, evaluating for brain metastases is necessary,
given its important role in disease control, especially in patients
who have achieved CR and long-term remission by immunotherapy.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
TA, SI and SH made substantial contributions in
conception, design and interpretation of data. TA and SI wrote the
manuscript. TD, FN and SH made substantial contributions in
interpretation and acquisition of data. TA and SI were responsible
for confirming the authenticity. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Both patients provided written informed consent for
the publication of their data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Dhote R, Beuzeboc P, Thiounn N, Flam T,
Zerbib M, Christoforov B and Debré B: High incidence of brain
metastases in patients treated with an M-VAC regimen for advanced
bladder cancer. Eur Urol. 33:392–395. 1998.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Anderson RS, el-Mahdi AM, Kuban DA and
Higgins EM: Brain metastases from transitional cell carcinoma of
urinary bladder. Urology. 39:17–20. 1992.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Swann JB and Smyth MJ: Immune surveillance
of tumors. J Clin Invest. 117:1137–1146. 2007.PubMed/NCBI View
Article : Google Scholar
|
|
4
|
Ratnam NM, Gilbert MR and Giles AJ:
Immunotherapy in CNS cancers: The role of immune cell trafficking.
Neuro Oncol. 21:37–46. 2019.PubMed/NCBI View Article : Google Scholar
|